End-of-chapter problems from Hornback: Ch 8: 23, 25, 26, 27b,c,h,i, 28-30, 32-36, 38-40, 44, 46-48, 50, 53. Ch 9: 17d,m, 18, 19, 26, 28.
|
|
|
- Stanley Turner
- 5 years ago
- Views:
Transcription
1 Chem 21 Homework set 9 End-of-chapter problems from Hornback: Ch 8: 23, 25, 26, 27b,c,h,i, 28-30, 32-36, 38-40, 44, 46-48, 50, 53. Ch 9: 17d,m, 18, 19, 26, Draw the products of the E2 reaction of each of the following compounds with CH 3 O in (solvent). Pay attention to stereochemistry. For a and b, be sure to consider the proper conformation around the C2-C3 bond. For c and d, you'll need to draw the chair conformations. (c) (d) 2. Write a mechanism for each the following S N 1 solvolyses, and draw the products, including stereochemistry. A small amount of E1 occurs also, but let's forget about E1 for now. Your mechanism should show each step clearly and include curved arrows indicating how electron pairs move in each step. (yes, there is a reaction in b.) Cl (c) For this one you ll need to draw the structure of solvent, acetic acid to have any hope of drawing a sensible product structure (the product is an ester, not a carboxylic acid). Think also about which oxygen of acetic acid adds to the cation. (hint: which oxygen is protonated by a CH strong acid?) One adds to make a much more stable 3 CO 2 H intermediate, although both possibilities actually lead to the tbu same product. 3. Draw the products from the following S N 1 solvolyses, paying attention to stereochemistry. n each case, state whether the products are enantiomers, diastereomers, or constitutional isomers. CH 3 (c) cis-1-bromo-4-methylcyclohexane in formic acid (HCO 2 H). (Hint: structure, structure, structure; no swarms of letters.)
2 (d) note the mixed solvent only water can react with the carbocation to make a stable product. water (S)-3-iodo-3-methylhexane acetone (e) (R)-1-chloro-3-bromobutane MeOH (f) Small amounts of alkenes are also produced in each of these reactions. Draw the alkenes produced in parts a, b, and c via E1 reactions. 4. Write a mechanism for each the following S N 1 solvolyses. Let s ignore stereochemistry this time. Let's also ignore the structure of that thing over the arrow in part a... NOT. CH 3 CO 2 H OCOCH 3 H 2 O acetone OH (c) Cl Et OH (d) Sketch a reaction coordinate diagram for the rearrangement in part c. 5. Here's something a little different. When the optically active tosylate below is solvolyzed in methanol, a substitution occurs, but with a rather unusual stereochemical outcome, as shown below only the trans-dimethoxy product is formed; not the cis. Furthermore, the product is racemic. One more interesting bit of evidence is that the trans-tosylate (below) solvolyzes much faster than the cis stereoisomer. Write a mechanism that accounts for these observations. O OTs OCH 3 OCH 3 OCH 3
3 (this is a continuation of the non-graded part; ignore the boxes in the header above) 6. The reactions below may proceed by S N 2, E2, or S N 1/E1 pathways, or not at all. Draw the products, and indicate which reaction mechanism(s) is/are operative, or write NR for "no rxn". (f you're confident that you understand all the reaction mechanisms, but you're having trouble with these, it's probably because you're not taking a systematic approach use the step-by-step procedure outlined in class.) KOEt CH 3 CO 2 Na + DMF (c) Cl! (d) HC C Na + (e) TsO NaOCH 3 acetone (f) tbuo K + DMSO (g) CH 3 O Na +
4 (h) HCO 2 Na acetonitrile (i) CH 3 CO 2 H! (j) OMs KOH DMSO (k) NaOEt DMF (l) CO 2 H CH 3 OK Et 2 O (m) tbu hexane!
5 Chem 21 Fall 2009 HW set 9 30 points; due Wed, Nov 11 Name 1. Think carefully about the possible conformations of its cyclohexane ring, and then draw the alkene(s) formed via the E2 reaction of the compound below with potassium ethoxide in ethanol. KOEt Would you expect S N 2 reaction to occur as well? f so, draw the product. (c) f the bromide above is instead dissolved in ethanol, with no base present, S N 1 and E1 reactions occur. What are the products of these reaction pathways? 2. Explain why the E2 reaction of 1-bromo-1-methylcyclobutane produces methylenecyclobutane rather than 1-methylcyclobutene. Note that the two alkenes have virtually identical enthalpies of formation, so product stability isn't an issue in this case. (Yes, the second one has a trisubstituted double bond, but it also has a wee bit more ring strain.) NaOCH 3 not
6 3. Write the mechanism of the S N 1 solvolysis below. Two constitutionally isomeric cyclopentenes are formed (let's not worry about stereochemistry for this problem). Write the mechanism and then draw the products. CH 3 OH 4. Same instructions as for #6 of the non-graded part. CH 3 CO 2 H SNa OMs CH 3 O DMSO (c) CO 2 tbu KOtBu tbuoh
CHE 321 Summer 2012 Exam 2 Form Select the correct number of chirality centers in heroin, the illicit drug shown below.
 Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
B. A transition state represents a maximum on the reaction path diagram and can be isolated.
 Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
Practice Hour Exam 2, Chemistry 2210, Organic Chemistry I 1. The most stable carbocation is: 2. Which of the following statements is true of transition states? A. A transition state represents a minimum
CHE 321 Summer 2010 Exam 2 Form Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III
 CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CHEM 302 Organic Chemistry I Problem Set VII Chapter 7 Answers
 CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
CEM 302 rganic Chemistry Problem Set V Chapter 7 Answers 1) The pk a values of acetic acid and trifluroacetic acid are 4.5 and 0.9 respectively. Based on this information, which anion, C 3 C - or CF 3
1. Complete the template below to show the stereochemistry of (3R,4R) 3-chloro-4,5,5- trimethyl-hexane-1,4-diol.
 Chemistry 51 Exam 1, Fall 2004 This is a closed book exam. The exam lasts 50 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer sheet
Chemistry 51 Exam 1, Fall 2004 This is a closed book exam. The exam lasts 50 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer sheet
CH Organic Chemistry I (Katz) Practice Exam #3- Fall 2013
 C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
C2710 - rganic Chemistry I (Katz) Practice Exam #3- all 2013 Name: Score: Part I - Choose the best answer and write the letter of your choice in the space provided. (32 pts) 1. f the following, which reaction
PRACTICE PROBLEMS UNIT 8
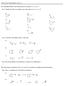 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron sheet.
 Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Chem 30A Winter Mon March 21st
 Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat #
 Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
THE UNIVERSITY OF CALGARY FACULTY OF SCIENCE FINAL EXAMINATION CHEMISTRY 351 READ ALL THE INSTRUCTIONS CAREFULLY
 TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
TE UNIVERSITY F CALGARY FACULTY F SCIENCE FINAL EXAMINATIN CEMISTRY 351 DECEMBER 22nd 1999 Time: 3 ours READ ALL TE INSTRUCTINS CAREFULLY PLEASE WRITE YUR NAME, STUDENT I.D. NUMBER N BT YUR EXAM ANSWER
OChem1 Old Exams. Chemistry 3719 Practice Exams
 OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
OChem1 Old Exams Chemistry 3719 Practice Exams Fall 2017 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete
diene. If stereoisomeric mixtures form, indicate by drawing one and writing "+ enantiomer" and/or "+ diastereomer" in the box.
 I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
UCF - ORGANIC CHEMISTRY 1 - PROF. DAOUDI UCF PROF. DAOUDI EXAM 3 REVIEW.
 UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
FOURTH EXAMINATION. (1)..20 pts... (2)..24 pts... (3)..18 pts... (4)..12 pts... (5)..12 pts... (6).. 6 pts... (7).. 8 pts... Bonus 4 pts...
 Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
Name: CHEM 331 FURTH EXAMINATIN All answers should be written on the exam in the spaces provided. Clearly indicate your answers in the spaces provided; if I have to guess as to what or where your answer
an axial "X" is necessary for a succesful E2 reaction and also works better for S N 2
 cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
cleophilic Substitution & Elimination hemistry 1 Templates for predicting S2 and E2 reactions primary - β α α β two different perspectives secondary - β β α β β α two different perspectives tertiary -
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
EM 331: hapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N 2 N 2 N 1 2 3 4 2. What hybrid orbitals are used to
CHAPTER 8 HW: ELIMINATIONS REACTIONS
 APTER 8 W: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult to
APTER 8 W: ELMNATNS REATNS S-TRANS SMERSM 1. Use a discussion and drawing of orbitals to help explain why it is generally easy to rotate around single bonds at room temperature, while it is difficult to
STEREOCHEMISTRY A STUDENT SHOULD BE ABLE TO:
 A STUDENT SHOULD BE ABLE TO: STEREOCHEMISTRY 1. Determine the relationship between two given structures (which may be any of the kinds below). Also, define the following terms, and give examples of pairs
A STUDENT SHOULD BE ABLE TO: STEREOCHEMISTRY 1. Determine the relationship between two given structures (which may be any of the kinds below). Also, define the following terms, and give examples of pairs
1. Name the following compound. Use the IUPAC system and include the stereochemical designations.
 Chemistry 51 Exam 1, Summer 2004 This is a closed book exam. The exam lasts 100 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer
Chemistry 51 Exam 1, Summer 2004 This is a closed book exam. The exam lasts 100 minutes. All answers must appear on the answer sheet. Only the answer sheet will be collected. Put your name on the answer
(b) (CH 3 ) 2 CCH 2 CH 3 D 2 O. (e) A. CH 3 CCl OSO 2 CH 3 C 6 H 5 H 3 C
 278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
278 h a p t e r 7 Further Reactions of aloalkanes 3. arbocations are stabilized by hyperconjugation: Tertiary are the most stable, followed by secondary. Primary and methyl cations are too unstable to
CH 253, Fall Question Points Possible Points Received
 CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
Chem 3719 Example Exams. Chemistry 3719 Practice Exams
 Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
If Compound 2 (below) was used in the above reaction, how might things change? Name Page 1. I. (23 points)
 I. (2 points) ame Page itrogen atoms in amines can undergo substitution reactions repeatedly under mildly basic conditions. Provide the structure of the starting amine and supply the missing reagent(s).
I. (2 points) ame Page itrogen atoms in amines can undergo substitution reactions repeatedly under mildly basic conditions. Provide the structure of the starting amine and supply the missing reagent(s).
Dr. Anand Gupta Mr Mahesh Kapil
 Dr. Anand Gupta Mr Mahesh Kapil 09356511518 09888711209 anandu71@yahoo.com mkapil_foru@yahoo.com Preparation of Haloalkanes From Alkanes Alkenes Alcohols Carboxylic Acids (Hundsdicker Reaction) Halide
Dr. Anand Gupta Mr Mahesh Kapil 09356511518 09888711209 anandu71@yahoo.com mkapil_foru@yahoo.com Preparation of Haloalkanes From Alkanes Alkenes Alcohols Carboxylic Acids (Hundsdicker Reaction) Halide
EXAMINATION 2 Chemistry 3A
 000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1. In the reaction shown above the nucleophile is. (a) Na (b) NaC CH (c) HC C (d) HC CH. 2. In the reaction shown above the nucleophile is
 Chemistry 247B Hanson Sample Exam 4B In each case, read each possible answer, use a process of elimination, and circle the BEST answer. If you are having trouble deciding between two answers, briefly explain
Chemistry 247B Hanson Sample Exam 4B In each case, read each possible answer, use a process of elimination, and circle the BEST answer. If you are having trouble deciding between two answers, briefly explain
1. (6 points) Provide IUPAC accepted names for the following compounds. 2. (6 points) Provide a structure for the following compounds.
 Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
8.8 Unimolecular Nucleophilic Substitution S N 1
 8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
8.8 Unimolecular Nucleophilic Substitution S N 1 A question. Tertiary alkyl halides are very unreactive in substitutions that proceed by the S N 2 mechanism. Do they undergo nucleophilic substitution at
EXAMINATION 2 Chemistry 3A
 1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound?
 CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
CEM 331: Chapter 1/2: Structures (Atoms, Molecules, Bonding) 1. What are the respective hybridizations of the atoms numbered 1 to 4 in this compound? N C 2 C N C 2 C N 1 2 3 4 1: three sigma bonds and
Partial Periodic Table
 CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
HO HO. 1) BH 3 HCl. + enantiomer either one may be drawn 4. + enantiomer either one may be drawn 4 1) O 3
 . (0 points) Page 1 A. Reaction analysis: provide the requested information in each of the following chemical transformations. how all missing starting materials and products unless otherwise indicated,
. (0 points) Page 1 A. Reaction analysis: provide the requested information in each of the following chemical transformations. how all missing starting materials and products unless otherwise indicated,
Chapter 9:Nucleophiles & Substitution Reactions
 1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
Copyright 2009 James K Whitesell
 Copyright 2009 James K Whitesell 5-1 These two molecules, cyclopropylcyclopentane and cyclobutycyclobutane have the same number of carbon and hydrogen atoms and thus they are constitutional isomers. 5-2
Copyright 2009 James K Whitesell 5-1 These two molecules, cyclopropylcyclopentane and cyclobutycyclobutane have the same number of carbon and hydrogen atoms and thus they are constitutional isomers. 5-2
Homework for Chapter 17 Chem 2320
 Homework for Chapter 17 Chem 2320 I. Cumulated, isolated, and conjugated dienes Name 1. Draw structures which fit the following descriptions. Use correct geometry! a conjugated diene with the formula C
Homework for Chapter 17 Chem 2320 I. Cumulated, isolated, and conjugated dienes Name 1. Draw structures which fit the following descriptions. Use correct geometry! a conjugated diene with the formula C
Homework - Chapter 9 Chem 2310
 Homework - Chapter 9 Chem 2310 me:. ntroduction to organic halides 1. Draw a line structure for a compound with the following description: an alkyl halide with the formula C 5 H 11 a vinyl halide with
Homework - Chapter 9 Chem 2310 me:. ntroduction to organic halides 1. Draw a line structure for a compound with the following description: an alkyl halide with the formula C 5 H 11 a vinyl halide with
CHEM 3311 (Richardson) Third Exam Nov. 27, 2018
 CHEM 3311 (Richardson) Third Exam Nov. 27, 2018 Your Name: Question Score Out of 1 15 Student ID: 2 20 3 20 Recitation (check one) O 10:00 Mon (Shafer Soars) 4 10 O 11:00 Mon (Matthew Farmer) O 1:00 Mon
CHEM 3311 (Richardson) Third Exam Nov. 27, 2018 Your Name: Question Score Out of 1 15 Student ID: 2 20 3 20 Recitation (check one) O 10:00 Mon (Shafer Soars) 4 10 O 11:00 Mon (Matthew Farmer) O 1:00 Mon
C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions. 5-Androstene, the parent alkene for most anabolic steroids
 C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions 5-Androstene, the parent alkene for most anabolic steroids CHM 321: Summary of Important Concepts YConcepts for Chapter
C h a p t e r E i g h t: Alkenes: Structure and Preparation via Elimination Reactions 5-Androstene, the parent alkene for most anabolic steroids CHM 321: Summary of Important Concepts YConcepts for Chapter
C sp2 trigonal planar trigonal planar 3
 I. ( points) ame Page A. Rocuronium bromide (Zemuron) is a muscle relaxant that is used in the application of anesthesia. It is used most often to facilitate endotracheal intubation because of its powerful
I. ( points) ame Page A. Rocuronium bromide (Zemuron) is a muscle relaxant that is used in the application of anesthesia. It is used most often to facilitate endotracheal intubation because of its powerful
Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm
 - 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
- 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
CHEM 2423 Unit 8 Homework Answers
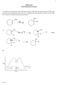 1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
1 CEM 2423 Unit 8 omework Answers 1. Show the mechanism of the following reaction. Label the rate-limiting step. Draw the potential energy diagram for this reaction. (c) Explain the regiochemistry of this
Points: 1 / 15 / Total: 100 points /
 Chemistry 6372 Fall 2002 omework #6 Answer Key Name Points: 1 / 15 / 2 / 10 / 3 / 10 / 4 / 10 / 5 / 10 / 6 / 10 / 7 / 35 / Total: 100 points / Average: 73 1) Answer the following questions from our book:
Chemistry 6372 Fall 2002 omework #6 Answer Key Name Points: 1 / 15 / 2 / 10 / 3 / 10 / 4 / 10 / 5 / 10 / 6 / 10 / 7 / 35 / Total: 100 points / Average: 73 1) Answer the following questions from our book:
= ( 3 ) + 2 ( 3 ) = ( ) = = ( ) = 20 H H
 Columbia University 92CORG12.DOC CEM S3443D Summer 1992 Professor Grace B. Borowitz Exam No. 2 June 17, 1992 Name: Grade: Please use a non-red pen. Answer questions in the provided space. If you write
Columbia University 92CORG12.DOC CEM S3443D Summer 1992 Professor Grace B. Borowitz Exam No. 2 June 17, 1992 Name: Grade: Please use a non-red pen. Answer questions in the provided space. If you write
1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below.
 1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below. E Rot = 14.2 kj/mol E Rot = 19.6 kj/mol a. Why does the potential energy of a molecule increase
1. The barrier to rotation around the C-C bonds for 2-methylpropane and 2,2-dimethylpropane are shown below. E Rot = 14.2 kj/mol E Rot = 19.6 kj/mol a. Why does the potential energy of a molecule increase
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1
 Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Chemistry 232 PRACTICE Midterm 3 October 2010 Your name: 1 You will need to be able to show picture ID to take the test. DO NOT OPEN TIS TEST UNTIL EVERYONE AS ONE I encourage following instructions: ten
Learning Guide for Chapter 17 - Dienes
 Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
Learning Guide for Chapter 17 - Dienes I. Isolated, conjugated, and cumulated dienes II. Reactions involving allylic cations or radicals III. Diels-Alder Reactions IV. Aromaticity I. Isolated, Conjugated,
Chapter 8 Alkyl Halides and Elimination Reactions
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Columbia University C99ORG12.DOC S3443D Summer 99 Professor Grace B. Borowitz Exam No. 2 June 14, 1999
 olumbia University 99RG12.D SD Summer 99 Professor Grace B. Borowitz Exam No. 2 June 1, 1999 Name: Sterie hemistry Grade: Please use a non-red pen. Answer questions in the provided space. If you write
olumbia University 99RG12.D SD Summer 99 Professor Grace B. Borowitz Exam No. 2 June 1, 1999 Name: Sterie hemistry Grade: Please use a non-red pen. Answer questions in the provided space. If you write
2/26/18. Practice Questions. Practice Questions B F. How many steps are there in this reaction?
 Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
CHEM Exam 2 ANSWER KEY
 CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
FINAL EXAMINATION Chemistry 3A
 1 FINAL EXAMINATION Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt May 11, 2016 GSI (if you are taking Chem 3AL): Please provide the following information
1 FINAL EXAMINATION Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt May 11, 2016 GSI (if you are taking Chem 3AL): Please provide the following information
Test 2A Chemistry 21 - Dr. Kline November 15, 2017
 Test 2A Chemistry 21 - Dr. Kline November 15, 2017 This test consists of a combination of multiple-choice and other questions. There should be 21 questions on eight pages. Do not use your own tables, scratch
Test 2A Chemistry 21 - Dr. Kline November 15, 2017 This test consists of a combination of multiple-choice and other questions. There should be 21 questions on eight pages. Do not use your own tables, scratch
(CH 3 ) 3 COH. CH 3 ONa
 1. Rank the following compounds in the trend requested. (15 points each) a. Rank by nucleophilicity. The strongest nucleophile is 1, while the weakest nucleophile is 5. C 3 PNa (C 3 ) 3 C C 3 Na C 3 C
1. Rank the following compounds in the trend requested. (15 points each) a. Rank by nucleophilicity. The strongest nucleophile is 1, while the weakest nucleophile is 5. C 3 PNa (C 3 ) 3 C C 3 Na C 3 C
C h a p t e r S e v e n : Substitution Reactions S N 2 O H H H O H H. Br -
 C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
C h a p t e r S e v e n : Substitution Reactions Br Br S N 2 CM 321: Summary of Important Concepts YConcepts for Chapter 7: Substitution Reactions I. Nomenclature of alkyl halides, R X A. Common name:
Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). Cl E- 6 chloro, 5 ethyl, 4 methyl 3-octene
 Problem Set 6 Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). E- 6 chlo, 5 ethyl, 4 methyl 3-octene 5 methyl, 3 vinyl cyclohexene 7Z- 7 bromo 4 ethyl, 6 methyl
Problem Set 6 Name the following compounds (include stereochemistry, cis/trans, E/Z when appropriate). E- 6 chlo, 5 ethyl, 4 methyl 3-octene 5 methyl, 3 vinyl cyclohexene 7Z- 7 bromo 4 ethyl, 6 methyl
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chemistry 123: Physical and Organic Chemistry Topic 1: Organic Chemistry
 Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
Concept Check: Topic 1: Conformation Winter 2009 Page 112 Concept Check: Topic 1: Conformation Winter 2009 Page 113 1 STEREOCHEMISTRY Winter 2009 Page 114 We have already covered two kinds of isomerism:
CHEMpossible. 261 Exam 1 Review
 CHEMpossible 261 Exam 1 Review A. Rank the following carboxylic acids from least acidic to most acidic: B. Draw the line-angle formulas for three acylic (non-cyclic) esters with the molecular formula C
CHEMpossible 261 Exam 1 Review A. Rank the following carboxylic acids from least acidic to most acidic: B. Draw the line-angle formulas for three acylic (non-cyclic) esters with the molecular formula C
(2/94)(6,7,9/95)(8,9/97)(12/99)(1/00) Neuman Chapter 4
 4: Stereochemistry Tetrahedral Carbon Configurations Stereoisomers and R,S Assignments The Number and Types of Stereoisomers Drawing Structures of Stereoisomers Cyclic Molecules Optical Activity Preview
4: Stereochemistry Tetrahedral Carbon Configurations Stereoisomers and R,S Assignments The Number and Types of Stereoisomers Drawing Structures of Stereoisomers Cyclic Molecules Optical Activity Preview
REVIEW PROBLEMS Key. 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. H H.
 rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
rganic hemistry II (E325) REVIEW PRBLEMS Key 1. Draw a complete orbital picture for the molecule shown below. Is this molecule chiral? Explain. 3 3 sp3 orbital p orbital sp2 orbital s orbital molecule
Due Date: 2) What is the relationship between the following compounds?
 Assignment #5 Name CHEM201 Student #: Due Date: MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) What type of isomers are CH3CH2OCH3 and CH3CH2CH2OH?
Assignment #5 Name CHEM201 Student #: Due Date: MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers the question. 1) What type of isomers are CH3CH2OCH3 and CH3CH2CH2OH?
Chem 3719 Klein Chapter Practice Problems
 Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail
 Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
Dehydrohalogenation of Alkyl Halides E2 and E1 Reactions in Detail b-elimination Reactions Overview dehydration of alcohols: X = H; Y = OH dehydrohalogenation of alkyl halides: X = H; Y = Br, etc. X C
!!!! Organic Chemistry CHM 224. Exam I Questions
 ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
OChem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards
 Chem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards Chem1 Practice Exams Dr. Peter Norris, 2014 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total
Chem1 Course Pack Practice Exams Practice Problems by Chapter Mechanism Flashcards Chem1 Practice Exams Dr. Peter Norris, 2014 Chemistry 3719 Practice Exam A1 This exam is worth 100 points out of a total
CHAPTER 7. Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination
 CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
CHAPTER 7 Further Reactions of Haloalkanes: Unimolecular Substitution and Pathways of Elimination 7-1 Solvolysis of Tertiary and Secondary Haloalkanes The rate of S N 2 reactions decrease dramatically
+ + CH 11: Substitution and Elimination Substitution reactions
 C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
C 11: Substitution and Elimination Substitution reactions Things to sort out: Nucleophile Electrophile -- > substrate Leaving Group S N 2 S N 1 E 1 E 2 Analysis Scheme Kinetics Reaction profile Substrates
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER
 UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER TITLE OF PAPER COURSE NUMBER Introductory Organic Chemistry C203 TIME Three Hours INSTRUCTIONS Section A is compulsory. Answer any three questions
UNIVERSITY OF SWAZILAND FINAL EXAMINATION 2013, DECEMBER TITLE OF PAPER COURSE NUMBER Introductory Organic Chemistry C203 TIME Three Hours INSTRUCTIONS Section A is compulsory. Answer any three questions
Chem 112A: Final Exam
 Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Chapter 7 Substitution Reactions
 Chapter 7 Substitution Reactions Review of Concepts Fill in the blanks below. To verify that your answers are correct, look in your textbook at the end of Chapter 7. Each of the sentences below appears
Chapter 7 Substitution Reactions Review of Concepts Fill in the blanks below. To verify that your answers are correct, look in your textbook at the end of Chapter 7. Each of the sentences below appears
CHEM 2430 Organic Chemistry I Fall Instructor: Paul Bracher. Final Examination
 CHEM 2430 Organic Chemistry I Fall 2015 Final Exam Solutions Key Page 1 of 10 CHEM 2430 Organic Chemistry I Fall 2015 Instructor: Paul Bracher Final Examination Friday, December 11 th, 2015 12:00 1:50
CHEM 2430 Organic Chemistry I Fall 2015 Final Exam Solutions Key Page 1 of 10 CHEM 2430 Organic Chemistry I Fall 2015 Instructor: Paul Bracher Final Examination Friday, December 11 th, 2015 12:00 1:50
Chapter 7 Substitution Reactions 7.1 Introduction to Substitution Reactions Substitution Reactions: two reactants exchange parts to give new products
 hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
hapter 7 Substitution eactions 7.1 Introduction to Substitution eactions Substitution eactions: two reactants exchange parts to give new products A-B + -D A-D + B- 3 2 + Br 3 2 Br + Elimination eaction:
CEM 251, Fall 2011 Sections Tuesday Thursday 6:00 7:30 pm Midterm #2 October 27, 2011
 CEM 251, Fall 2011 Sections 25 36 Tuesday Thursday 6:00 7:30 pm ctober 27, 2011 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only what is
CEM 251, Fall 2011 Sections 25 36 Tuesday Thursday 6:00 7:30 pm ctober 27, 2011 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only what is
Cl Cl CH 2 OH H 3 C CH 2 CH 3 H. cis-1-(bromomethyl)-2-(2-chloroethyl)cyclobutane. Meso-1,2,3,4,butanetetraol (show as a Fischer projection)
 Page 1 2. 12 Points. (a) Name or write a structure for the following compounds (IUPAC nomenclature). Be sure to pay attention to stereochemistry! Note that most of these questions are taken directly from
Page 1 2. 12 Points. (a) Name or write a structure for the following compounds (IUPAC nomenclature). Be sure to pay attention to stereochemistry! Note that most of these questions are taken directly from
1) (100 pts) 5) (20 pts) 3) (35 pts) 4) (25pts. Total (200 pts) More Tutorial at
 Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
2 h; 240 points please print clearly Dr. Kathleen Nolta Signature UM ID # Problem Points Score GSI I 42 II 44 III 40 IV 43 V 31 VI 40 Total 240
 hemistry 10 First ame Final Examination Last ame h; 0 points please print clearly Dr. Kathleen olta ignature UM ID # PRIT TE FIRT LETTER F YUR LAT AME ERE: Problem Points core GI I II III 0 IV V 1 VI 0
hemistry 10 First ame Final Examination Last ame h; 0 points please print clearly Dr. Kathleen olta ignature UM ID # PRIT TE FIRT LETTER F YUR LAT AME ERE: Problem Points core GI I II III 0 IV V 1 VI 0
CH Organic Chemistry I (Katz) Practice Exam #2- Fall 2015 (With Answers)
 2710 - rganic hemistry I (Katz) Practice Exam #2- all 2015 (With nswers) Name: Score: Part I - hoose the best answer and write the letter of your choice in the space provided. 1. The Newman projection
2710 - rganic hemistry I (Katz) Practice Exam #2- all 2015 (With nswers) Name: Score: Part I - hoose the best answer and write the letter of your choice in the space provided. 1. The Newman projection
KEY Page 1. Name H H H 3 C C 3. Cl CH. AlCl 3 H H. + enantiomer H 3 C CH 3. Cl CH3 HNO 3. AlCl NO 2 CH 3 1) O 3 2) H 2 O 2 OH H 3 C C.
 ame Page 1 I. (8 points) omplete the following reactions as directed. Transformations requiring sequential experimental steps should be numbered appropriately. Show the major organic product(s) unless
ame Page 1 I. (8 points) omplete the following reactions as directed. Transformations requiring sequential experimental steps should be numbered appropriately. Show the major organic product(s) unless
Partial Periodic Table
 CEM 33, Fall 00 Professor Walba Third our Exam November 8, 00 Scores: ) 0 ) 0 3) 0 4) 0 5) 0 00 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, have neither given nor received
CEM 33, Fall 00 Professor Walba Third our Exam November 8, 00 Scores: ) 0 ) 0 3) 0 4) 0 5) 0 00 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, have neither given nor received
Departmental Final Examination. Organic Chemistry I Caffein
 Departmental Final Examination rganic Chemistry I 2423 Caffein Name CEMISTRY 2423 FINAL EXAM FALL, 2005 DIRECTINS: A periodic table is attached at the end of this exam. Please answer all questions as completely
Departmental Final Examination rganic Chemistry I 2423 Caffein Name CEMISTRY 2423 FINAL EXAM FALL, 2005 DIRECTINS: A periodic table is attached at the end of this exam. Please answer all questions as completely
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Text Sections (N0 1.9, 9-11) Homework: Chapter 1:
 CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
CHEM 261 HOME WORK Lecture Topics: MODULE 1: The Basics: Bonding and Molecular Structure Atomic Structure - Valence Electrons Chemical Bonds: The Octet Rule - Ionic bond - Covalent bond How to write Lewis
CHEM 2311 HW1. COMPLETELY FILL THE BOXES IN DARK PENCIL, i.e.:, NOT or or STRUCTURE
 EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
EM 2311 W1 Provide answers to the following multiple choice questions on the Scantron form provided in lecture. Make sure that you put your name and bubble in your GTID # on the answer form. MPLETELY FILL
CHEMISTRY 341. Final Exam Tuesday, December 16, Problem 1 15 pts Problem 9 8 pts. Problem 2 5 pts Problem pts
 CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
CEMISTRY 341 Final Exam Tuesday, December 16, 1997 Name NAID Problem 1 15 pts Problem 9 8 pts Problem 2 5 pts Problem 10 21 pts Problem 3 26 pts Problem 11 15 pts Problem 4 10 pts Problem 12 6 pts Problem
Chemistry 3719, Fall 2012 Exam 1 Name: Student number:
 Chemistry 3719, Fall 2012 Exam 1 Name: Student number: This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete the exam and you may use molecular
Chemistry 3719, Fall 2012 Exam 1 Name: Student number: This exam is worth 100 points out of a total of 600 points for Chemistry 3719/3719L. You have 50 minutes to complete the exam and you may use molecular
Chm 222 Spring 2015, Exercise Set 5 The Stereochemistry Pillar
 Chm 222 Spring 2015, Exercise Set 5 The Stereochemistry Pillar Mr. Linck c Boniface Beebe Productions January 19, 2016 Version 3.0. NOTE: An asterisk in the problem title indicates that it is a continuing
Chm 222 Spring 2015, Exercise Set 5 The Stereochemistry Pillar Mr. Linck c Boniface Beebe Productions January 19, 2016 Version 3.0. NOTE: An asterisk in the problem title indicates that it is a continuing
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
1. Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C bond length bond strength 3 C C 3 3 C C 3 3 C C 3 3 C 2 C C 2 3 C a C=C double bond
3.2.8 Haloalkanes. Elimination. 148 minutes. 145 marks. Page 1 of 22
 3.2.8 Haloalkanes Elimination 148 minutes 145 marks Page 1 of 22 Q1. Reaction of 2-bromobutane with potassium hydroxide can produce two types of product depending on the solvent used. In aqueous solution,
3.2.8 Haloalkanes Elimination 148 minutes 145 marks Page 1 of 22 Q1. Reaction of 2-bromobutane with potassium hydroxide can produce two types of product depending on the solvent used. In aqueous solution,
Chemistry 201. MW 12:00pm 1:15pm Examination #2 August 15 th Bronco ID. Question Score Possible Points. 1 (12pts) 2 (24pts) 3 (25pts)
 Chemistry 201 MW 12:00pm 1:15pm Examination #2 August 15 th 2016 Name Bronco ID. Question Score Possible Points 1 (12pts) 2 (24pts) 3 (25pts) 4... (12pts) 5 (27pts). Total (100pts) 1. Read each question
Chemistry 201 MW 12:00pm 1:15pm Examination #2 August 15 th 2016 Name Bronco ID. Question Score Possible Points 1 (12pts) 2 (24pts) 3 (25pts) 4... (12pts) 5 (27pts). Total (100pts) 1. Read each question
CEM 351 2nd EXAM/Version A Friday, October 17, :50 2:40 p.m. Room 138, Chemistry
 Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
EXAMINATION 1 Chemistry 3A. Making up an I Grade If you are, please indicate the semester during which you took previous Chem 3A and the Instructor
 1 EXAMINATION 1 Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt February 28, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1 EXAMINATION 1 Chemistry 3A Key Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt February 28, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
Organic Chemistry Chapter 5 Stereoisomers H. D. Roth
 Organic Chemistry Chapter 5 Stereoisomers. D. Roth 11. Chirality of conformationally mobile systems ring compounds Monosubstituted cycloalkanes cannot have an asymmetric carbon in the ring, because there
Organic Chemistry Chapter 5 Stereoisomers. D. Roth 11. Chirality of conformationally mobile systems ring compounds Monosubstituted cycloalkanes cannot have an asymmetric carbon in the ring, because there
Organic Chemistry. Alkenes (2)
 For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
