1 Introduction. ISSN , England, UK World Journal of Modelling and Simulation Vol. 14 (2018) No. 1, pp. 3-11
|
|
|
- Linette Day
- 5 years ago
- Views:
Transcription
1 ISSN , England, UK World Journal of Modelling and Simulation Vol. 14 (2018) No. 1, pp Molecular Structure, Mulliken charges, HOMO-LUMO, Electrostatic Potential and Nonlinear Optical Properties of Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate molecule by HF and DFT methods Nadia Benhalima 1, Amel Djedouani 2, Rachida Rahmani 1, Abdelkader Chouaih 1 * Fodil Hamzaoui 3, El Hadj Elandaloussi 4 1 Laboratory of Technology and Solid s Properties, Faculty of Sciences and Technology, Abdelhamid Ibn Badis University, BP 227 Mostaganem 27000, Algeria 2 Ecole Normale Superieure de Constantine, Constantine 25000, Algeria 3 LPFM Academie de Montpellier, France 4 Laboratoire de Valorisation des Materiaux, Université Abdelhamid Ibn Badis, B.P. 227, Mostaganem, Algeria (Received December , Accepted May ) Abstract. In this work, we report a oretical study on molecular structure, electrostatic potential and nonlinear optical properties of Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate molecule (monomer and dimer ). The molecular geometry in ground state was investigated by ab initio and density functional method (B3LYP) with 6-31G(d) and 6-31+G(d,p) basis sets. The results show that computed geometrical parameters are in good agreement with experimental values. The molecular electrostatic potential and HOMO-LUMO of title molecule have been also calculated using oretical methods. The calculated HOMO and LUMO energies show that charge transfer occurs within molecule. In order to investigate nonlinear optical behavior, electric dipole moment µ, polarizability α and hyperpolarizability β were computed. All β values that we report here are magnitude of static hyperpolarizability. Theoretically predicted β values exhibit higher nonlinear optical activity. Keywords: oretical calculation, charge transfer, atomic charges, molecular electrostatic potential, HOMO- LUMO 1 Introduction Nonlinear optical (NLO) materials have been extensively studied for many years [2]. Materials with high NLO response are of great technological interest because of ir potential applications in data storage, signalprocessing, and telecommunication technologies [29]. In particular, organic molecules have received large attention due to combination of molecular design flexibility, chemical tenability and choice of syntic strategies [9]. In last two decades, organic molecules have been synsized, some of m showing impressively high hyperpolarizabilities [19]. Theoretical investigations have been of great help in achievement of se results; y gave first hints about possible high NLO responses of organic molecules with extended conjugated π systems [3], and provided guidelines for a rational design of such molecules [23]. The recent synses of zwitterionic molecules, some of m exhibiting extremely high NLO activity, have also been inspired by oretical investigations [8]. Organic molecules with -electron delocalization are currently of wide interest as NLO materials with potential applications in optical switches and or NLO devices [22]. Large Corresponding author. address: achouaih@gmail.com Published by World Academic Press, World Academic Union
2 4 N. Benhalima & A. Djedouani & R. Rahmani et al: Title Suppressed Due to Excessive Length optical non-linearity could be seen in organic conjugated molecules incorporating an electron acceptor group at one moiety and a donor group at opposite one [27]. For prototypical push-pull molecule, latter would exhibit an asymmetric polarization response to a symmetric electric field at intense field strengths since electron intensity is more easily displaced toward an acceptor substituent than toward a substituent donor [13]. Consequently, dipolar aromatic molecules possessing an electron donor group and an electron acceptor group contribute to large optical non-linearity arising from intramolecular charge transfer between two groups of opposite nature. It is also well known that first and second hyperpolarizabilities in π-electron systems with donor-acceptor groups increase with increasing donor and acceptor strengths. Despite relevant role played by ory in development of field of NLO organic molecules, re are so far only few papers reported comparison between experimental and computed hyperpolarizability for large size NLO chromophores [11]. Zwitterionic compounds have received interest of chemists and physicists due to ir applications as nonlinear optical materials [18]. There are some reports concerning formation of zwitterionic form in crystal structure of ortho-hydroxy Schiff bases [28]. The present work was assessed to determination of molecular structure, electrostatic potential and nonlinear optical properties of Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate compound. A combined experimental and oretical study was carried out and a detailed description of structural features of studied molecule is reported. The ground state molecular structure was optimized and compared with experimental data obtained by X-ray analysis. In addition, various nonlinear optical properties such as electric dipole moment, molecular electrostatic potential, polarizability and hyperpolarizability were also addressed oretically. The HOMO-LUMO energy gap has been computed and NLO activity of molecule was analyzed. 2 Computational details The molecular geometries of title compound were fully optimized at various ory levels using Gaussian 03 program [17] and Gauss-View molecular visualization program [1]. From X-ray crystal structure data [15], Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate compound crystallizes in monoclinic space group P21/c with four molecules per unit cell (Z = 4) and following cell dimensions: a = (4)A, b = (10)A, c = (3)A. β = (6). The data were collected at low temperature (173 K). The spatial coordinate positions of title compound, as obtained from X-ray structural analysis, were used as initial coordinates for oretical calculations. For title molecule, full geometry optimizations have been carried out at Hartree-Fock (HF) [10] level using 6 31G(d) and 6-31+G(d,p) basis sets, by Berny method [6, 16]. The effects of electron correlation on geometry optimization are taken into account by Becke s three parameter hybrid exchange functions and Lee-Young-Parr correlation functional (B3LYP) [7, 30] level in DFT method. The ab initio and DFT optimized geometries of crystal structure of investigated compound correspond to slightly non-planar conformation having 105 dihedral angle. The optimized structures were n used to calculate electric dipole moments and hyperpolarizabilities of title compound. Nonlinear optical calculations were performed at both Hartree-Fock (HF) ab initio and DFT levels with 6 31G(d) and G(d, p) basis sets. The 6 31G(d) basis set has been found to be more adequate for obtaining reliable trends in β values [14, 33]. 3 Results and discussion 3.1 Optimized geometry The optimized bond lengths, bond and dihedral angles of title compound calculated by B3LYP and HF methods using different basis sets are listed in Tab. 1, in accordance with atom numbering shown in Fig. 1. Our optimized structural parameters are now compared with exact experimental X-ray data [15]. Tab. 1 shows that most of bond lengths are found to be greater than experimental ones. For example, calculated bond lengths (C1 N1, C1 N2, C2 N3, C4 C8, C4 C5, N2 N3) at B3LYP level are slightly larger than corresponding WJMS for contribution: submit@wjms.org.uk
3 World Journal of Modelling and Simulation, Vol. 14 (2018) No. 1, pp experimental values. This overestimation can be explained by fact that oretical calculations belong to isolated molecule in gaseous phase and experimental results belong to similar molecules in solid state. The C = O bond length namely C1 O4 (1.222)A, C5 O1 (1.215A ) and C8 O3 (1.258A ) calculated by means of B3LYP/6-31+G(d,p) method are most close to measured ones. These values agree well with data reported in literature [4, 5]. On or hand, some computed bond angles deviate about 1 2 from experimental data. The bond angles of O4 C1 N2, C2 N3 N2 and N3 C2 C3 were measured to be (17), (15) and (17). The calculated values using B3LYP/6-31G(d) are of order of , and , respectively. According to above comparisons, biggest differences of bond lengths and bond angles mainly occur in groups involved in bonding [i.e., C1 O4, C8 O3 and N1 C1 N2], which can also easily be understood taking into account intermolecular bond interactions occurring in crystal. When X-ray structure of title compound is compared with its optimized counterparts (see Fig. 2), slight conformational discrepancies are observed between m. The calculated value of dihedral angle (C1 N2 N3 C2) using both B3LYP/6-31G(d) (108.4 ) and HF/6-31G(d) (105.8 ) methods is slightly different from experimental one (0.00 ). In case of calculated dihedral angle, re is a good agreement between B3LYP and HF methods. In spite of differences observed, calculated geometric parameters are in general in good agreement with X-ray structure. N1 C1 N2 O4 C3 C2 N3 C4 C8 O3 O1 C5 O2 C6 C7 C9 O1w Fig. 1: Optimized structure with B3LYP/6-31G (d) level of title molecule 3.2 Mulliken charge analysis It is clear that Mulliken [24] populations provide simplest picture of charge distribution. Knowledge of charge density function can lead to some important properties of molecule such as charges on different atoms, molecular dipole moment and electrostatic potential around molecule. The Mulliken charges for non-h atoms of title compound were calculated at HF and B3LYP level with 6-31G(d) basis set in gas-phase. The atomic charges show that N1, N2, N3 and O1, O1w, O2, O3, O4 atoms have bigger negative atomic charges in gas phase compared to carbon atoms. This behavior can be result of intramolecular N HO bond and C8 = O3 double bond features. The obtained results by both methods are summarized in Tab Molecular electrostatic potential Molecular electrostatic potential (MEP) is associated to electronic density and has been used primarily for predicting sites and relative reactivities toward electrophilic attack in studies of biological recognition and WJMS for subscription: info@wjms.org.uk
4 6 N. Benhalima & A. Djedouani & R. Rahmani et al: Title Suppressed Due to Excessive Length Table 1: Calculated geometrical parameters for title molecule (Distance, Å; Angle, ) Geometrical X ray α HF DFT parameters 6-31G(d) 6-31+G(d,p) 6-31G(d) 6-31+G(d,p) C1-O (2) C1-N (2) C1-N (2) C2-N (2) C2-C (2) C2-C (2) C4-C (2) C4-C (2) C5-O (2) C5-O (2) C6-C (2) C6-O (2) C6-C (3) C7-C (2) C8-O (2) N2-N (2) O4-C1-N (17) O4-C1-N (17) N1-C1-N (16) N3-C2-C (15) N3-C2-C (17) C4-C2-C (16) C8-C4-C (16) C8-C4-C (15) C5-C4-C (15) O1-C5-O (16) O1-C5-C (18) O2-C5-C (15) C7-C6-O (17) C7-C6-C (18) O2-C6-C (16) C6-C7-C (17) O3-C8-C (16) O3-C8-C (15) C4-C8-C (15) C1-N2-N (15) C2-N3-N (15) C6-O2-C (14) C1-N2-N3-C bonding interactions [25, 32]. Molecular electrostatic potential, V(r), at a point r(x,y,z) is described as following: V (r) = Z A ρ(r)dr R A r r r A ZA is charge on nucleus A located at RA and ρ(r) is electron density. The electrostatic potential plots are shown in Fig. 2 ((a) monomer and (b) dimer). MEP was calculated at B3LYP/6-31G(d) optimized geometry. The negative regions of MEP surface correspond to an attraction and are colored in red. While, positive regions correspond to a repulsion and are colored in blue. The negative regions are related to electrophilic sites and positive ones to nucleophilic sites. The MEP maps of title molecule show that negative regions were found around oxygen atoms and that positive potential sites are around atoms. WJMS for contribution: submit@wjms.org.uk
5 N N N World O1 Journal of Modelling and Simulation, Vol. 14 (2018) No. 1, pp O1w Table O2 2: Mulliken0.070 Charges (q) in non- atoms of title molecule calculated with 6-31G(d) basis set O O q Atoms 3.3. Molecular electrostatic potential HF DFT Molecular electrostatic potential (MEP) is C1 associated to electronic density and has been used primarily C for predicting sites and relative reactivities toward electrophilic attack in studies of biological recognition C and bonding interactions [27, C4 28]. Molecular electrostatic potential, V(r), at a point r(x,y,z) is described as following: C C C C Z C A X ( r) dr ' V (r) N1 A R A r r ' r ZA is charge on nucleus A located at N2RA and (r) is electron density. The electrostatic potential plots are shown in Figure 2 ((a) monomer N3 and (b) dimer) MEP was calculated at B3LYP/6-31G(d) O optimized geometry. The negative regions of MEP surface correspond to an attraction and are colored in O1w red. While, positive regions correspond O2 to a repulsion and are colored in blue. The negative regions are related to electrophilic sites and O3 positive 0.06 ones to nucleophilic sites. The MEP maps of title molecule show that negative regions O4were found around oxygen atoms and that positive potential sites are around atoms. Fig. 2: Molecular electrostatic potential maps (a) and (b) calculated at B3LYP/6-31G(d) level Fig. 2: Molecular electrostatic potential maps (a) and (b) calculated at B3LYP/6-31G(d) level 3.4 Homo-lumo calculation The highest occupied molecular orbitals (HOMOs) and Lowest-lying unoccupied molecular orbitals (LU- MO) are very useful for physicists and chemists because energy difference between se orbitals (energy gap) represents minimum energy required to promote an electron, and is refore often mostfrequent and important energy transfer mechanism within a system. Furr, charge transport properties of molecule [12] are determined by difference of energy between HOMO and LUMO. The orbitals also provide important electron density information which can help determine which part of molecule is most actively participating in an energy transfer event. The HOMO-LUMO energy gap of molecule was calculated using HF/(6 31G(d) and G(d, p)) and B3LY P/(6 31G(d) and G(d, p)) level as presented in Tab. 3. The energy gap between HOMO and LUMO indicates molecular chemical stability. In addition, a lower HOMO-LUMO energy gap explains fact that eventual charge transfer interaction is taking place within molecule. The HOMO-LUMO plots are given in Fig Hydrogen bonding and non linear optical properties Hydrogen bonds are very important dipole interactions in stabilizing structures. In amino acids having zwitterionic form, NH2 moiety is a good donor and carboxylate or nitrate group is an excellent acceptor. In NLO crystals, bonds linked with adjacent molecules, where O-H O bonds are relatively stronger than N-H O bonds [26]. Fig. 4 shows molecular packing diagram of molecule where bond interactions N- H O and O-H O were observed. These interactions are responsible for NLO effect in Zwitterionic WJMS for subscription: info@wjms.org.uk
6 31+G(d,p)) level as presented in Table 3. The energy gap between HOMO and LUMO indicates molecular chemical stability. In addition, a lower HOMO-LUMO energy gap explains fact that 8 eventual charge transfer N. Benhalima interaction & is A. taking Djedouani place & R. within Rahmani et molecule. al: Title Suppressed The HOMO-LUMO Due to Excessive plots Length are given in Figure 3. Fig. Fig. 3: The 3 The HOMO and and LUMO plotof of title title molecule molecule 3.5. Hydrogen bonding and non linear optical properties 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate Hydrogen bonds are very important dipole interactions in stabilizing monohydrate compound. structures. In The amino NLOacids effect having may also be zwitterionic associated form, to push-pull NH 2 moiety type is structure. a good donor As expected, and carboxylate best donors or nitrate interact group with is an excellent best acceptors acceptor. leading to In strong NLO crystals, mentioned bonds bonds. linked These with rigid adjacent molecules, bonds link where symmetry-independent O-H O molecules bonds are into relatively a dimer, stronger which can than be N-H O considered bonds as [30]. building block of structure. Figure It is evident 4 shows that molecular crystal structures packing diagram are generated of molecule and stabilized where by bonds bond but interactions more evidently N that H O y also and O H O participate were considerably observed. These to interactions increase of are hyperpolarizability responsible for of NLO effect bonded in Zwitterionic molecular systems. 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate For organic NLO materials, oretical and experimental monohydrate studies are performed compound. in The order NLO to understand effect microscopic may also be origin associated of nonlinear to "push pull" behavior [20, type 31]. structure. As expected, best donors interact with best In this acceptors context, leading this study to is strong extended mentioned to determination bonds. of These electric rigid dipole moment bonds µ, link isotropic polarizability symmetry-independent α and first molecules hyperpolarizability into a dimer, β which can be considered as building block of tot of title compound (monomer and dimer). The dipole moment structure. (µ), isotropic polarizability β tot, first-order hyperpolarizability (α) tensor, can be obtained using following It is evident equations: that crystal structures are generated and stabilized by bonds but more evidently that y also participate considerably to increase of hyperpolarizability of bonded molecular systems. For organic NLO materials, oretical and experimental studies are performed in order to understand microscopic origin µ 0 of = nonlinear (µ 2 behavior [31, 32]. x + µ 2 y + µ 2 z) 1/2 In this context, this study is extended to determination of electric dipole moment µ, isotropic polarizability α and first hyperpolarizability α = 1 3 (α xx + of α yy title + αcompound zz ) (monomer and dimer). The dipole moment, isotropic polarizability β, first-order hyperpolarizability tensor, can be tot = (βx 2 + βy 2 + βz 2 ) 1/2. The whole equation for computing magnitude of first hyeprpolarizability (β) from Gaussian 03 output is given below: β tot = [(β xxx + β yyy + β zzz ) 2 + (β yyy + β yzz + β yxx ) 2 ] + (β zzz + β zxx + β zyy ) 2. The first hyperpolarizability is a third rank tensor that can be depicted by a matrices. The 27 components of 3D matrix can be reduced to 10 components because of Kleinman symmetry [21]. The Gaussian 03 output provides 10 components of this matrix as βxxx, βxxy, βxyy, βyyy, βxxz, βxyz, βyyz, βxzz, βyzz, βzzz respectively. The first hyperpolarizability tensors provided by Gaussian 03 are given in atomic units (a.u.), computed values were converted into electrostatic units. (α : 1a.u. = esu; β : 1a.u. = esu). The dipole moment (µ0), mean polarizability (α) and first hyperpolarizability (β) for monomer and dimer are calculated at 6 31G(d) and G(d, p) basis sets. Tab. 3 gives HF and B3LYP results of electronic dipole moment µ i (i = x, y, z), polarizability αij and first hyperpolarizability βijk for Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate compound (monomer and dimer ). Theoretical calculation plays a significant role in understanding structure-property relationship WJMS for contribution: submit@wjms.org.uk
7 Table 3. The electric dipole moment µ (D), average polarizability α ( esu) and first hyperpolarizability β ( esu) of title molecule. World Journal of Modelling HF and Simulation, Vol. 14 (2018) No. 1, pp DFT 9 Parameters 6-31G* 6-31+G** 6-31G* 6-31+G** which is able to help Monomer in designing Dimer novel NLO Monomer materials. Dimer It is well established Monomer that Dimer higher Monomer values of Dimer dipole moment, µ x molecular 2.43 polarizability, and hyperpolarizability are very important 2.43 for 7.52 active NLO 2.20 properties The highest µ y value of dipole 1.01 moment obtained 1.16 with 0.85 B3LYP /Lanl2dz 1.52 is equal 1.49 to 3.68 D3.92 for monomer 1.75 and D for dimer. µ z The highest 1.46 value of dipole 2.88 moment 1.76 is observed for 1.31 component 1.12 µ x. In this 0.02 direction, 1.68 this value is 1.41 equal µ to 2.67 and D for monomer and dimer respectively. The calculated polarizability (α), is equal to α xx α xy 24 esu and esu for monomer 5.71 and22.45 dimer respectively obtained with 6-31G+(d,p) As it canαbe yy seen in Tab. 3, calculated polarizability αij have non zero values and was dominated by diagonal components. α xz The first hyperpolarizability 4.70 values 7.72 β tot of 3.38 title molecule obtained 3.19 with B3LYP/Lanl2dz are α equal yz to esu for monomer and esu for dimer. The calculated results show that α zz α title (a.u) molecule might have microscopic nonlinear (NLO) behavior with non-zero values. The calculated first order α (esu) hyperpolarizability of title molecule20.29 has a low value of esu obtained23.89 with HF method using β xxx G(d, p) basis set and a high value of esu obtained with DFT method using β same xxy basis set. Thus, calculated low β total value for title molecule ( esu) is found to be 8 β xyy times β yyy greater than β total value of urea ( esu), refore, predicting that43.70 our molecule is a powerful β xxz candidate 5.92 for NLO material β xyz From cited above results, molecule having greatest β tot value, corresponds to low HOMO LUMO β yyz energy gap These results39.50 show that HOMO-LUMO gap have a substantial influence on first hyperpolarizability. β xzz The high β value and low HOMO-LUMO energy gap show that title molecule is highly NLO β yzz active β zzz material and it might be efficient for optoelectronic applications β Consequently, (a.u) we can finally infer from above discussion of contents of Tab. 3 that introduction of electron β (esu) correlation in method applied1.612 for analysis of hyperpolarizability, such3.343 as DFT method, HOMO will probably predict more reasonable values as opposed to those converged upon use of HF method. LUMO Gap (Hartrees) Gap (ev) O4 N1 O1w Å H1w H1A Å O3 Fig. 4: Fig. N-H4: ON H O and O-H and OO H O interactions interactions calculated calculated at B3LYP/6-31G at (d) (d) level 4. Conclusion 4 Conclusion In this work we have calculated geometric parameters and nonlinear optical properties of Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate molecule by using HF and B3LYP methods with 6-31G(d) and 6-31+G(d,p) basis sets. The optimized bond lengths and bond angles obtained with B3LYP level of calculation show excellent agreement with experimental values. The energy gap between HOMO and LUMO calculated using B3LYP is lower than value calculated using HF level. The energy gap values indicate molecular chemical stability. Results show that first hyperpolarizability β of Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate molecule is directly related to HOMO-LUMO energy gap. Molecular electrostatic potential (MEP) was calculated at B3LYP/6-31G (d) optimized geometry. The MEP map shows that negative potential sites are on electronegative atoms as well as positive potential sites are around atoms. These sites give WJMS for subscription: info@wjms.org.uk
8 10 N. Benhalima & A. Djedouani & R. Rahmani et al: Title Suppressed Due to Excessive Length Table 3: The electric dipole moment µ (D), average polarizability α( 10 24esu) and first hyperpolarizability β( 10 30esu) of title molecule HF DFT Parameters 6 31G G 6 31G G Monomer Dimer Monomer Dimer Monomer Dimer Monomer Dimer µ x µ y µ z µ α xx α xy α yy α xz α yz α zz α(a.u) α(esu) β xxx β xxy β xyy β yyy β xxz β xyz β yyz β xzz β yzz β zzz β ( a.u) β ( esu) HOMO LUMO Gap (Hartrees) Gap (ev) information about region from where compound can have intramolecular interactions. This study reveals that Zwitterionic 6-methyl-2-oxo-3-[1-(ureidoiminio)ethyl]-2H-pyran-4-olate monohydrate molecule has a large first static hyperpolarizabilities and may have potential applications in development of NLO materials. References [1] A. H. A. Frisch, A.B. Neilson. Gauss view 4, gaussian, inc., wallingford ct usa, [2] P. Agarwal, N. Choudhary. Density functional ory studies on structure, spectra (ft-ir, ft-raman, and uv) and first order molecular hyperpolarizability of 2-hydroxy-3-methoxy-n-(2-chloro-benzyl)-benzaldehyde-imine: Comparison to experimental data. Vibrational Spectroscopy, 2013, 64((2013)): [3] G. P. Agrawal, C. Flytzanis. Delocalization and superalternation effects in nonlinear susceptibilities of onedimensional systems. Chemical Physics Letters, 1976, 44(2): [4] M. I. Attia, N. R. El-Brollosy. Synsis, single crystal x-ray structure, and antimicrobial activity of 6-(1,3- benzodioxol-5-ylmethyl)-5-ethyl-2-[2-(morpholin-4-yl)ethyl]sulfanylpyrimidin-4(3h)-one. Journal of Chemistry, 2015, 2014(4): 1 8. [5] M. I. Attia, H. A. Ghabbour et al Synsis and single crystal x-ray structure of new (2e)-2-[3-(1h-imidazol-1-yl)-1- phenylpropylidene]-n-phenylhydrazinecarboxamide. Journal of Chemistry, 2013, [6] R. F. W. Bader. Atoms in molecules: A quantum ory. 1994, 360(1-3). [7] A. D. Becke. Density-functional rmochemistry. v. systematic optimization of exchange-correlation functionals. The Journal of chemical physics, 1997, 107(20): WJMS for contribution: submit@wjms.org.uk
9 World Journal of Modelling and Simulation, Vol. 14 (2018) No. 1, pp [8] A. Capobianco, A. Esposito, T. Caruso. Tuning wavefunction mixing in pushcpull molecules: From neutral to zwitterionic compounds. European Journal of Organic Chemistry, 2012, 2012(15): [9] R. Centore, A. Concilio, F. Borbone. Quadratic nonlinear optical and preliminary piezoelectric investigation of crosslinked samples obtained from a liquid chromophore. Journal of Polymer Science Part B Polymer Physics, 2012, 50(9): [10] H. D. Cohen, C. C. J. Roothaan. Electric dipole polarizability of atoms by hartree-fock method. Journal of Chemical Physics, 1965, 43(10): S34 S39. [11] L. R. Dalton, S. J. Benight. Systematic nanoengineering of soft matter organic electro-optic materials. Chemistry of Materials, 2011, 23(3): [12] W. B. Davis, W. A. Svec, M. A. Ratner. Molecular-wire behaviour in p -phenylenevinylene oligomers. Nature, 1998, 396(6706): [13] B. L. Davydov, L. D. Derkacheva, V. V. Dunina. Connection between charge transfer and laser second harmonic generation. Zhetf Pisma Redaktsiiu, 1970, 12(1): 24. [14] C. Dehu, F. Meyers, et al Solvent effects on second-order nonlinear optical response of. pi.-conjugated molecules: A combined evaluation through self-consistent reaction field calculations and hyper-rayleigh scattering measurements. Journal of American Chemical Society, 1995, 117(40): [15] A. Djedouani, S. Boufas. Djedouani, a., boufas, s., allain, m., bouet, g., khan, m.: Zwitterionic 6-methyl-2-oxo-3- [1-(ureidoiminio)ethyl]-2h-pyran-4-olate monohydrate. Acta Crystallogr Sect E Struct Rep Online, 2008, 64(Pt 9): [16] R. Fletcher, M. J. D. Powell. A rapidly convergent descent method for minimization. Computer Journal, 1963, 6(6): [17] M. Frisch, G. Trucks, et al J. Cheeseman, et al. Gaussian 03, revision b. 04. gaussian, inc., pittsburgh, [18] M. P. Hill, E. C. Carroll et al Rapid photodynamics of vitamin b6 coenzyme pyridoxal 5 -phosphate and its schiff bases in solution. Journal of Physical Chemistry B, 2008, 112(18): [19] H. Kang, A. Facchetti, H. Jiang. Ultralarge hyperpolarizability twisted pi-electron system electro-optic chromophores: synsis, solid-state and solution-phase structural characteristics, electronic structures, linear and nonlinear optical properties, and computational studies. Journal of American Chemical Society, 2007, 129(11): [20] P. Kerkoc, M. Zgonik, K. Sutter. 4-(n,n-dimethylamino)-3-acetamidonitrobenzene single crystals for nonlinearoptical applications. Journal of Optical Society of America B, 1990, 7(3): [21] D. A. Kleinman. Nonlinear dielectric polarization in optical media. Physical Review, 1962, 126(6): [22] M. Lanata, C. Bertarelli et al Molecules with quinoid ground state: a new class of large molecular optical nonlinearities. Syntic Metals, 2003, 138(1-2): [23] S. R. Marder, B. Kippelen, et al Design and synsis of chromophores and polymers for electro-optic and photorefractive applications. Nature, 1997, 388(6645): 845. [24] R. S. Mulliken. Electronic population analysis on lcao mo molecular wave functions. i. The Journal of Chemical Physics, 1955, 23(10): [25] J. S. Murray, K. D. Sen. Molecular electrostatic potentials : concepts and applications. Elsevier, [26] H. S. Nalwa, M. Hanack. Third-order nonlinear optical properties of porphyrazine, phthalocyanine and naphthalocyanine germanium derivatives: Demonstrating effect of -conjugation length on third-order optical nonlinearity of two-dimensional molecules. Chemical physics, 1999, 245(1): [27] H. S. e. a. Nalwa. Nonlinear Optics of Organic Molecules and Polymers [28] A. Ozek, C. Albayrak. Three (e)-2-[(bromophenyl)iminomethyl]-4-methoxyphenols. Acta Crystallographica Section C-crystal Structure Communications, 2007, 63(3): [29] A. Petrosyan. Salts of l-histidine as nonlinear optical materials: a review. J Cryst Phys Chem, 2010, 1(1): [30] G. Rauhut, P. Pulay. Transferable scaling factors for density functional derived vibrational force fields. J.phys.chem, 1995, 99(10): [31] D. Sajan, H. J. Ravindra, N. Misra. Intramolecular charge transfer and bonding interactions of nonlinear optical material n -benzoyl glycine: Vibrational spectral study. Vibrational Spectroscopy, 2010, 54(1): [32] E. Scrocco, J. Tomasi. Electronic molecular structure, reactivity and intermolecular forces: An euristic interpretation by means of electrostatic molecular potentials. Advances in Quantum Chemistry, 1978, 11: [33] K. S. Thanthiriwatte, K. M. N. D. Silva. Non-linear optical properties of novel fluorenyl derivativesłab initio quantum chemical calculations. Journal of Molecular Structure Theochem, 2002, 617(1-3): WJMS for subscription: info@wjms.org.uk
First Order Hyperpolarizability and Homo-Lumo Analysis of L-Arginine Maleate (LArM) by Density Functional Theory Methods
 51 Sciencia Acta Xaveriana An International Science Journal ISSN. 0976-1152 Volume 4 No. 2 pp. 51-58 September 2013 First Order Hyperpolarizability and Homo-Lumo Analysis of L-Arginine Maleate (LArM) by
51 Sciencia Acta Xaveriana An International Science Journal ISSN. 0976-1152 Volume 4 No. 2 pp. 51-58 September 2013 First Order Hyperpolarizability and Homo-Lumo Analysis of L-Arginine Maleate (LArM) by
CHAPTER 8 REPORT ON HIGHER SHG EFFICIENCY IN BIS (CINNAMIC ACID): HEXAMINE COCRYSTAL
 CHAPTER 8 REPORT ON HIGHER SHG EFFICIENCY IN BIS (CINNAMIC ACID): HEXAMINE COCRYSTAL 8.1. Introduction In recent times higher Second Harmonic Generation (SHG) efficiency organic materials receive great
CHAPTER 8 REPORT ON HIGHER SHG EFFICIENCY IN BIS (CINNAMIC ACID): HEXAMINE COCRYSTAL 8.1. Introduction In recent times higher Second Harmonic Generation (SHG) efficiency organic materials receive great
THEORETICAL INVESTIGATION OF ELECTROSTATIC POTENTIAL AND NON LINEAR OPTICAL PROPERTIES OF M NITROACETANILIDE
 ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in THEORETICAL INVESTIGATION OF ELECTROSTATIC POTENTIAL AND NON LINEAR OPTICAL PROPERTIES OF N.
ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in THEORETICAL INVESTIGATION OF ELECTROSTATIC POTENTIAL AND NON LINEAR OPTICAL PROPERTIES OF N.
THE ELECTROSTATIC PROPERTIES OF 1,2-DIMETHYL-3- NITROBENZENE COMPOUND: ab initio CALCULATION AND X-RAY CHARGE DENSITY ANALYSIS
 ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in THE ELECTROSTATIC PROPERTIES OF 1,2-DIMETHYL-3- NITROBENZENE COMPOUND: ab initio CALCULATION
ISSN: 0974-1496 e-issn: 0976-0083 CODEN: RJCABP http://www.rasayanjournal.com http://www.rasayanjournal.co.in THE ELECTROSTATIC PROPERTIES OF 1,2-DIMETHYL-3- NITROBENZENE COMPOUND: ab initio CALCULATION
Theoretical and Experimental Electrostatic Potential around the m-nitrophenol Molecule
 Molecules 2015, 20, 4042-4054; doi:10.3390/molecules20034042 Article OPEN ACCESS molecules ISSN 1420-3049 www.mdpi.com/journal/molecules Theoretical and Experimental Electrostatic Potential around the
Molecules 2015, 20, 4042-4054; doi:10.3390/molecules20034042 Article OPEN ACCESS molecules ISSN 1420-3049 www.mdpi.com/journal/molecules Theoretical and Experimental Electrostatic Potential around the
AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES
 Int. J. Chem. Sci.: 9(4), 2011, 1564-1568 ISSN 0972-768X www.sadgurupublications.com AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C.
Int. J. Chem. Sci.: 9(4), 2011, 1564-1568 ISSN 0972-768X www.sadgurupublications.com AB INITIO MODELING OF THE STRUCTURAL DEFECTS IN AMIDES M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C.
Structure, Electronic and Nonlinear Optical Properties of Furyloxazoles and Thienyloxazoles
 Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
Journal of Physics: Conference Series PAPER OPEN ACCESS Structure, Electronic and Nonlinear Optical Properties of Furyls and Thienyls To cite this article: Ozlem Dagli et al 6 J. Phys.: Conf. Ser. 77 View
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes
 Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
CHEM 344 Molecular Modeling
 CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Part 1: Molecular Orbital Theory, Hybridization, & Formal Charge * all calculation data obtained
CHAPTER-IV. FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations
 4.1. Introduction CHAPTER-IV FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations m-xylol is a material for thermally stable aramid fibers or alkyd resins [1]. In recent
4.1. Introduction CHAPTER-IV FT-IR and FT-Raman investigation on m-xylol using ab-initio HF and DFT calculations m-xylol is a material for thermally stable aramid fibers or alkyd resins [1]. In recent
International Journal of Materials Science ISSN Volume 12, Number 2 (2017) Research India Publications
 HF, DFT Computations and Spectroscopic study of Vibrational frequency, HOMO-LUMO Analysis and Thermodynamic Properties of Alpha Bromo Gamma Butyrolactone K. Rajalakshmi 1 and A.Susila 2 1 Department of
HF, DFT Computations and Spectroscopic study of Vibrational frequency, HOMO-LUMO Analysis and Thermodynamic Properties of Alpha Bromo Gamma Butyrolactone K. Rajalakshmi 1 and A.Susila 2 1 Department of
with the larger dimerization energy also exhibits the larger structural changes.
 A7. Looking at the image and table provided below, it is apparent that the monomer and dimer are structurally almost identical. Although angular and dihedral data were not included, these data are also
A7. Looking at the image and table provided below, it is apparent that the monomer and dimer are structurally almost identical. Although angular and dihedral data were not included, these data are also
Practical 1: Structure and electronic properties of organic molecules. B/ Structure, electronic and vibrational properties of the water molecule
 D1CH9116 - MMCO Molecular Modelling applied to organic chemistry Practical 1: tructure and electronic properties of organic molecules B/ tructure, electronic and vibrational properties of the water molecule
D1CH9116 - MMCO Molecular Modelling applied to organic chemistry Practical 1: tructure and electronic properties of organic molecules B/ tructure, electronic and vibrational properties of the water molecule
IR, Raman, First Hyperpolarizability and Computational Study of 1-chloroethyl Benzene
 Material Science Research India Vol. 9(1), 117-121 (2012) IR, Raman, First Hyperpolarizability and Computational Study of 1-chloroethyl Benzene HEMA TRESA VARGHESE¹, C.YOHANNAN PANICKER²* and SHEENA MARY
Material Science Research India Vol. 9(1), 117-121 (2012) IR, Raman, First Hyperpolarizability and Computational Study of 1-chloroethyl Benzene HEMA TRESA VARGHESE¹, C.YOHANNAN PANICKER²* and SHEENA MARY
Competition between Alkalide Characteristics and Nonlinear Optical Properties in OLi 3 M Li 3 O (M = Li, Na, and K) Complexes
 Competition between Alkalide Characteristics and Nonlinear Optical Properties in OLi 3 M Li 3 O (M = Li, Na, and K) Complexes Ambrish Kumar Srivastava and Neeraj Misra * Department of Physics, University
Competition between Alkalide Characteristics and Nonlinear Optical Properties in OLi 3 M Li 3 O (M = Li, Na, and K) Complexes Ambrish Kumar Srivastava and Neeraj Misra * Department of Physics, University
Department of Physics, A.V.C. College, Mayiladuthurai, Tamilnadu, India.
 IOSR Journal of Applied Physics (IOSR-JAP) e-issn: 2278-4861.Volume 8, Issue 3 Ver. II (May. - Jun. 2016), PP 01-14 www.iosrjournals.org The Spectroscopic (FT-IR, FT-Raman, NMR & UV-Vis) and first order
IOSR Journal of Applied Physics (IOSR-JAP) e-issn: 2278-4861.Volume 8, Issue 3 Ver. II (May. - Jun. 2016), PP 01-14 www.iosrjournals.org The Spectroscopic (FT-IR, FT-Raman, NMR & UV-Vis) and first order
Vibrational spectra, nonlinear optical properties, NBO analysis and thermodynamic properties of N-phenylethanolamine by density functional method
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 016, 8(6):184-05 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Vibrational spectra, nonlinear optical properties,
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 016, 8(6):184-05 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Vibrational spectra, nonlinear optical properties,
FT-IR, FT-Raman And UV Spectra and Ab-Initio HF and DFT Vibrational Study of 1-Propyl 4-Piperidone
 FT-IR, FT-Raman And UV Spectra and Ab-Initio HF and DFT Vibrational Study of 1-Propyl 4-Piperidone K. Rajalakshmi 1 and E.Elumalai 2 1 Department of Physics, Sri Chandrasekharendra Saraswathi Viswa Mahavidyalaya,
FT-IR, FT-Raman And UV Spectra and Ab-Initio HF and DFT Vibrational Study of 1-Propyl 4-Piperidone K. Rajalakshmi 1 and E.Elumalai 2 1 Department of Physics, Sri Chandrasekharendra Saraswathi Viswa Mahavidyalaya,
Atomic and molecular interaction forces in biology
 Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
Growth and Characterization of an Organic Crystal and DFT Studies of 2-amino 5-methyl Pyridinium Salicylate
 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2016, Vol. 32, No. (4): Pg. 1937-1945 Growth and Characterization
ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2016, Vol. 32, No. (4): Pg. 1937-1945 Growth and Characterization
Density Functional Theory Study of Exohedral Carbon Atoms Effect on Electrophilicity of Nicotine: Comparative Analysis
 Computational Chemistry, 2016, 4, 17-31 Published Online January 2016 in SciRes. http://www.scirp.org/journal/cc http://dx.doi.org/10.4236/cc.2016.41003 Density Functional Theory Study of Exohedral Carbon
Computational Chemistry, 2016, 4, 17-31 Published Online January 2016 in SciRes. http://www.scirp.org/journal/cc http://dx.doi.org/10.4236/cc.2016.41003 Density Functional Theory Study of Exohedral Carbon
Australian Journal of Basic and Applied Sciences
 AENSI Journals Australian Journal of Basic and Applied Sciences ISSN:1991-8178 Journal home page: www.ajbasweb.com Theoretical Study for the Effect of Hydroxyl Radical on the Electronic Properties of Cyclobutadiene
AENSI Journals Australian Journal of Basic and Applied Sciences ISSN:1991-8178 Journal home page: www.ajbasweb.com Theoretical Study for the Effect of Hydroxyl Radical on the Electronic Properties of Cyclobutadiene
STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY
 Int. J. Chem. Sci.: 9(4), 2011, 1763-1767 ISSN 0972-768X www.sadgurupublications.com STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C. YOHANNAN
Int. J. Chem. Sci.: 9(4), 2011, 1763-1767 ISSN 0972-768X www.sadgurupublications.com STRUCTURAL DEFECTS IN IMIDATES : AN AB INITIO STUDY M. FATHIMA BEGUM, HEMA TRESA VARGHESE a, Y. SHEENA MARY a, C. YOHANNAN
The linear, nonlinear optical properties and quantum chemical parameters of some sudan dyes
 The linear, nonlinear optical properties and quantum chemical parameters of some sudan dyes Aslı EŞME 1,*, Seda GÜNEŞDOĞDU SAĞDINÇ 2 1 Department of Elementary Science Education, Kocaeli University, Umuttepe
The linear, nonlinear optical properties and quantum chemical parameters of some sudan dyes Aslı EŞME 1,*, Seda GÜNEŞDOĞDU SAĞDINÇ 2 1 Department of Elementary Science Education, Kocaeli University, Umuttepe
( ) R kj. = y k y j. y A ( ) z A. y a. z a. Derivatives of the second order electrostatic tensor with respect to the translation of ( ) δ yβ.
 Supporting information Derivatives of R with respect to the translation of fragment along the y and z axis: y = y k y j (S1) z ( = z z k j) (S2) Derivatives of S with respect to the translation of fragment
Supporting information Derivatives of R with respect to the translation of fragment along the y and z axis: y = y k y j (S1) z ( = z z k j) (S2) Derivatives of S with respect to the translation of fragment
CHAPTER INTRODUCTION
 CHAPTER 3 A SCALED QUANTUM MECHANICAL APPROACH OF VIBRATIONAL ANALYSIS OF O-TOLUNITRILE BASED ON FTIR AND FT RAMAN SPECTRA, AB INITIO, HARTREE FOCK AND DFT METHODS 3.1. INTRODUCTION o-tolunitrile or ortho
CHAPTER 3 A SCALED QUANTUM MECHANICAL APPROACH OF VIBRATIONAL ANALYSIS OF O-TOLUNITRILE BASED ON FTIR AND FT RAMAN SPECTRA, AB INITIO, HARTREE FOCK AND DFT METHODS 3.1. INTRODUCTION o-tolunitrile or ortho
2x (x 2 + y 2 + 1) 2 2y. (x 2 + y 2 + 1) 4. 4xy. (1, 1)(x 1) + (1, 1)(y + 1) (1, 1)(x 1)(y + 1) 81 x y y + 7.
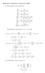 Homework 8 Solutions, November 007. (1 We calculate some derivatives: f x = f y = x (x + y + 1 y (x + y + 1 x = (x + y + 1 4x (x + y + 1 4 y = (x + y + 1 4y (x + y + 1 4 x y = 4xy (x + y + 1 4 Substituting
Homework 8 Solutions, November 007. (1 We calculate some derivatives: f x = f y = x (x + y + 1 y (x + y + 1 x = (x + y + 1 4x (x + y + 1 4 y = (x + y + 1 4y (x + y + 1 4 x y = 4xy (x + y + 1 4 Substituting
Journal of Chemical and Pharmaceutical Research, 2015, 7(2): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(2):641-645 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Synthesis, characterization and theoretical study
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2015, 7(2):641-645 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Synthesis, characterization and theoretical study
The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then
 1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
1 The symmetry properties & relative energies of atomic orbitals determine how they react to form molecular orbitals. These molecular orbitals are then filled with the available electrons according to
Research & Development Centre, Bharathiar University, Coimbatore , India. 2
 EXPERIMENTAL FT-IR, FT-RAMAN AND THEORETICAL QUANTUM CHEMICAL COMPUTATIONS ON THE MOLECULAR STRUCTURE P-BROMOBENZOTRIFLUORIDE V. Anbu 1, K.A. Vijayalakshmi *, T. Karthick 3, Poonam Tandon 4 1 Research
EXPERIMENTAL FT-IR, FT-RAMAN AND THEORETICAL QUANTUM CHEMICAL COMPUTATIONS ON THE MOLECULAR STRUCTURE P-BROMOBENZOTRIFLUORIDE V. Anbu 1, K.A. Vijayalakshmi *, T. Karthick 3, Poonam Tandon 4 1 Research
Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8. Scholars Research Library (
 Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Theoretical study
Journal of Computational Methods in Molecular Design, 2013, 3 (1):1-8 Scholars Research Library (http://scholarsresearchlibrary.com/archive.html) ISSN : 2231-3176 CODEN (USA): JCMMDA Theoretical study
Quantum Chemical Simulations and Descriptors. Dr. Antonio Chana, Dr. Mosè Casalegno
 Quantum Chemical Simulations and Descriptors Dr. Antonio Chana, Dr. Mosè Casalegno Classical Mechanics: basics It models real-world objects as point particles, objects with negligible size. The motion
Quantum Chemical Simulations and Descriptors Dr. Antonio Chana, Dr. Mosè Casalegno Classical Mechanics: basics It models real-world objects as point particles, objects with negligible size. The motion
Electronic structures of one-dimension carbon nano wires and rings
 IOP Publishing Journal of Physics: Conference Series 61 (2007) 252 256 doi:10.1088/1742-6596/61/1/051 International Conference on Nanoscience and Technology (ICN&T 2006) Electronic structures of one-dimension
IOP Publishing Journal of Physics: Conference Series 61 (2007) 252 256 doi:10.1088/1742-6596/61/1/051 International Conference on Nanoscience and Technology (ICN&T 2006) Electronic structures of one-dimension
CHEM 344 Molecular Modeling
 CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Day 1 all calculation data obtained from Gaussian09 using B3LYP/6-31G(d) unless otherwise noted.
CHEM 344 Molecular Modeling The Use of Computational Chemistry to Support Experimental Organic Chemistry Day 1 all calculation data obtained from Gaussian09 using B3LYP/6-31G(d) unless otherwise noted.
CHAPTER 5 FT INFRARED, FT RAMAN, VIBRATIONAL ASSIGNMENTS AND QUANTUM CHEMICAL STUDIES OF NICOTINIC ACID (NIASIN)
 CHAPTER 5 FT INFRARED, FT RAMAN, VIBRATIONAL ASSIGNMENTS AND QUANTUM CHEMICAL STUDIES OF NICOTINIC ACID (NIASIN) 5.1. INTRODUCTION Nicotinic acid widely known as Niacin are also known as vitamin B 3 and
CHAPTER 5 FT INFRARED, FT RAMAN, VIBRATIONAL ASSIGNMENTS AND QUANTUM CHEMICAL STUDIES OF NICOTINIC ACID (NIASIN) 5.1. INTRODUCTION Nicotinic acid widely known as Niacin are also known as vitamin B 3 and
EJL R Sβ. sum. General objective: Reduce the complexity of the analysis by exploiting symmetry. Garth J. Simpson
 e sum = EJL R Sβ General objective: Reduce the complexity of the analysis by exploiting symmetry. Specific Objectives: 1. The molecular symmetry matrix S. How to populate it.. Relationships between the
e sum = EJL R Sβ General objective: Reduce the complexity of the analysis by exploiting symmetry. Specific Objectives: 1. The molecular symmetry matrix S. How to populate it.. Relationships between the
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer. Lecture 31, April 12, 2006
 Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 31, April 12, 2006 (Some material in this lecture has been adapted from Cramer, C.
Chem 3502/4502 Physical Chemistry II (Quantum Mechanics) 3 Credits Spring Semester 2006 Christopher J. Cramer Lecture 31, April 12, 2006 (Some material in this lecture has been adapted from Cramer, C.
Why Is Molecular Interaction Important in Our Life
 Why Is Molecular Interaction Important in ur Life QuLiS and Graduate School of Science iroshima University http://www.nabit.hiroshima-u.ac.jp/iwatasue/indexe.htm Suehiro Iwata Sept. 29, 2007 Department
Why Is Molecular Interaction Important in ur Life QuLiS and Graduate School of Science iroshima University http://www.nabit.hiroshima-u.ac.jp/iwatasue/indexe.htm Suehiro Iwata Sept. 29, 2007 Department
Quantum Chemical Designing of Novel Organic Non-Linear Optical Compounds
 Quantum Chemical Designing of Novel NLO Compounds Bull. Korean Chem. Soc. 2014, Vol. 35, No. 5 1391 http://dx.doi.org/10.5012/bkcs.2014.35.5.1391 Quantum Chemical Designing of Novel Organic Non-Linear
Quantum Chemical Designing of Novel NLO Compounds Bull. Korean Chem. Soc. 2014, Vol. 35, No. 5 1391 http://dx.doi.org/10.5012/bkcs.2014.35.5.1391 Quantum Chemical Designing of Novel Organic Non-Linear
Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study
 pubs.acs.org/jpca Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study Tomoo Miyahara, Hiroshi Nakatsuji,*, and Hiroshi Sugiyama Quantum Chemistry Research Institute, JST, CREST,
pubs.acs.org/jpca Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study Tomoo Miyahara, Hiroshi Nakatsuji,*, and Hiroshi Sugiyama Quantum Chemistry Research Institute, JST, CREST,
Spectroscopic properties of dipicolinic acid and its dianion
 Chemical Physics 322 (2006) 254 268 www.elsevier.com/locate/chemphys Spectroscopic properties of dipicolinic acid and its dianion John Rui-Hua Xie a, Vedene H. Smith Jr. b, Roland E. Allen a, * a Department
Chemical Physics 322 (2006) 254 268 www.elsevier.com/locate/chemphys Spectroscopic properties of dipicolinic acid and its dianion John Rui-Hua Xie a, Vedene H. Smith Jr. b, Roland E. Allen a, * a Department
Chapter 6. Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications
 Chapter 6 Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications 6.1 Structures of Ni, Cu substituted Phthalocyanine Almost all of the metals
Chapter 6 Electronic spectra and HOMO-LUMO studies on Nickel, copper substituted Phthalocyanine for solar cell applications 6.1 Structures of Ni, Cu substituted Phthalocyanine Almost all of the metals
NITRATE. HOWRAH (West Bengal) INDIA e Thushara, Neethinagar-64, Pattathanam, KOLLAM (Kerala) INDIA ABSTRACT
 Int. J. Chem. Sci.: 8(1), 2010, 176-182 SPECTROSCOIC INVESTIGATIONS OF ANILINIUM NITRATE C. YOHANNAN PANICKER *, HEMA TRESA VARGHESE a, P. E. EAPEN b, K. RAJU c, SUBARNA GANGULI d, FATHIMA BEEGUM and Y.
Int. J. Chem. Sci.: 8(1), 2010, 176-182 SPECTROSCOIC INVESTIGATIONS OF ANILINIUM NITRATE C. YOHANNAN PANICKER *, HEMA TRESA VARGHESE a, P. E. EAPEN b, K. RAJU c, SUBARNA GANGULI d, FATHIMA BEEGUM and Y.
Computer Laboratory DFT and Frequencies
 Computer Laboratory DFT and Frequencies 1. DFT KEYWORDS FOR DFT METHODS Names for the various pure DFT models are given by combining the names for the exchange and correlation functionals. In some cases,
Computer Laboratory DFT and Frequencies 1. DFT KEYWORDS FOR DFT METHODS Names for the various pure DFT models are given by combining the names for the exchange and correlation functionals. In some cases,
Structural Properties, Natural Bond Orbital, Theory Functional Calculations (DFT), and Energies for the α Halorganic Compounds
 Current World Environment Vol. 7(2), 221-226 (2012) Structural Properties, Natural Bond Orbital, Theory Functional Calculations (DFT), and Energies for the α Halorganic Compounds NAJLA SEIDY and SHAHRIAR
Current World Environment Vol. 7(2), 221-226 (2012) Structural Properties, Natural Bond Orbital, Theory Functional Calculations (DFT), and Energies for the α Halorganic Compounds NAJLA SEIDY and SHAHRIAR
Excited States Calculations for Protonated PAHs
 52 Chapter 3 Excited States Calculations for Protonated PAHs 3.1 Introduction Protonated PAHs are closed shell ions. Their electronic structure should therefore be similar to that of neutral PAHs, but
52 Chapter 3 Excited States Calculations for Protonated PAHs 3.1 Introduction Protonated PAHs are closed shell ions. Their electronic structure should therefore be similar to that of neutral PAHs, but
Quantum Mechanical Study on the Adsorption of Drug Gentamicin onto γ-fe 2
 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-00 X CODEN: OJCHEG 015, Vol. 31, No. (3): Pg. 1509-1513 Quantum Mechanical
ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-00 X CODEN: OJCHEG 015, Vol. 31, No. (3): Pg. 1509-1513 Quantum Mechanical
IFM Chemistry Computational Chemistry 2010, 7.5 hp LAB2. Computer laboratory exercise 1 (LAB2): Quantum chemical calculations
 Computer laboratory exercise 1 (LAB2): Quantum chemical calculations Introduction: The objective of the second computer laboratory exercise is to get acquainted with a program for performing quantum chemical
Computer laboratory exercise 1 (LAB2): Quantum chemical calculations Introduction: The objective of the second computer laboratory exercise is to get acquainted with a program for performing quantum chemical
MO Calculation for a Diatomic Molecule. /4 0 ) i=1 j>i (1/r ij )
 MO Calculation for a Diatomic Molecule Introduction The properties of any molecular system can in principle be found by looking at the solutions to the corresponding time independent Schrodinger equation
MO Calculation for a Diatomic Molecule Introduction The properties of any molecular system can in principle be found by looking at the solutions to the corresponding time independent Schrodinger equation
Available online at WSN 61(2) (2017) EISSN
 Available online at www.worldscientificnews.com WSN 61(2) (2017) 150-185 EISSN 2392-2192 FT-IR, FT-Raman, NMR and U-V Spectral investigation: Computation of vibrational frequency, chemical shifts and electronic
Available online at www.worldscientificnews.com WSN 61(2) (2017) 150-185 EISSN 2392-2192 FT-IR, FT-Raman, NMR and U-V Spectral investigation: Computation of vibrational frequency, chemical shifts and electronic
Chapter 5. Molecular Orbitals
 Chapter 5. Molecular Orbitals MO from s, p, d, orbitals: - Fig.5.1, 5.2, 5.3 Homonuclear diatomic molecules: - Fig. 5.7 - Para- vs. Diamagnetic Heteronuclear diatomic molecules: - Fig. 5.14 - ex. CO Hybrid
Chapter 5. Molecular Orbitals MO from s, p, d, orbitals: - Fig.5.1, 5.2, 5.3 Homonuclear diatomic molecules: - Fig. 5.7 - Para- vs. Diamagnetic Heteronuclear diatomic molecules: - Fig. 5.14 - ex. CO Hybrid
AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS
 Journal of Undergraduate Chemistry Research, 2004, 1, 15 AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS J.R. Foley* and R.D. Parra Chemistry
Journal of Undergraduate Chemistry Research, 2004, 1, 15 AN AB INITIO STUDY OF INTERMOLECULAR INTERACTIONS OF GLYCINE, ALANINE AND VALINE DIPEPTIDE-FORMALDEHYDE DIMERS J.R. Foley* and R.D. Parra Chemistry
Ab Initio Study of the Dimers of Nodifloridin B
 Ab Initio Study of the Dimers of Nodifloridin B Liliana Mammino Abstract Nodifloridin B is an acylated phloroglucinol derivative in which both the acyl chain and a substituent in meta to the acyl chain
Ab Initio Study of the Dimers of Nodifloridin B Liliana Mammino Abstract Nodifloridin B is an acylated phloroglucinol derivative in which both the acyl chain and a substituent in meta to the acyl chain
Supporting Information
 Supporting Information Tuning of Second-Order Nonlinear Optical Response Properties of Aryl-Substituted Boron-Dipyrromethene Dyes: Unidirectional Charge Transfer Coupled with Structural Tailoring Ramprasad
Supporting Information Tuning of Second-Order Nonlinear Optical Response Properties of Aryl-Substituted Boron-Dipyrromethene Dyes: Unidirectional Charge Transfer Coupled with Structural Tailoring Ramprasad
Supporting information for: First hyperpolarizability of collagen using the. point dipole approximation
 Supporting information for: First hyperpolarizability of collagen using the point dipole approximation Ignat Harczuk, Olav Vahtras, and Hans Ågren KTH Royal Institute of Technology, School of Biotechnology,
Supporting information for: First hyperpolarizability of collagen using the point dipole approximation Ignat Harczuk, Olav Vahtras, and Hans Ågren KTH Royal Institute of Technology, School of Biotechnology,
AB INITIO CALCULATIONS AND VIBRATIONAL SPECTROSCOPIC STUDIES OF 2-CHLORO-6- METHOXYPYRIDINE
 AB INITIO CALCULATIONS AND VIBRATIONAL SPECTROSCOPIC STUDIES OF 2-CHLORO-6- METHOXYPYRIDINE L.Usha Kumari a, M.Fathima Beegum a, B.Harikumar b, Hema Tresa Varghese c, C.Yohannan Panicker a* a Department
AB INITIO CALCULATIONS AND VIBRATIONAL SPECTROSCOPIC STUDIES OF 2-CHLORO-6- METHOXYPYRIDINE L.Usha Kumari a, M.Fathima Beegum a, B.Harikumar b, Hema Tresa Varghese c, C.Yohannan Panicker a* a Department
Hong Zhou, Guang-Xiang Liu,* Xiao-Feng Wang and Yan Wang * Supporting Information
 Three cobalt(ii) coordination polymers based on V-shaped aromatic polycarboxylates and rigid bis(imidazole) ligand: Syntheses, crystal structures, physical properties and theoretical studies Hong Zhou,
Three cobalt(ii) coordination polymers based on V-shaped aromatic polycarboxylates and rigid bis(imidazole) ligand: Syntheses, crystal structures, physical properties and theoretical studies Hong Zhou,
Same idea for polyatomics, keep track of identical atom e.g. NH 3 consider only valence electrons F(2s,2p) H(1s)
 XIII 63 Polyatomic bonding -09 -mod, Notes (13) Engel 16-17 Balance: nuclear repulsion, positive e-n attraction, neg. united atom AO ε i applies to all bonding, just more nuclei repulsion biggest at low
XIII 63 Polyatomic bonding -09 -mod, Notes (13) Engel 16-17 Balance: nuclear repulsion, positive e-n attraction, neg. united atom AO ε i applies to all bonding, just more nuclei repulsion biggest at low
Hints on Using the Orca Program
 Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Hints on Using the Orca Program
Computational Chemistry Workshops West Ridge Research Building-UAF Campus 9:00am-4:00pm, Room 009 Electronic Structure - July 19-21, 2016 Molecular Dynamics - July 26-28, 2016 Hints on Using the Orca Program
Molecular Orbital Theory. Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions between two bonded atoms
 Molecular Orbital Theory Valence Bond Theory: Electrons are located in discrete pairs between specific atoms Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions
Molecular Orbital Theory Valence Bond Theory: Electrons are located in discrete pairs between specific atoms Molecular Orbital Theory: Electrons are located in the molecule, not held in discrete regions
Example questions for Molecular modelling (Level 4) Dr. Adrian Mulholland
 Example questions for Molecular modelling (Level 4) Dr. Adrian Mulholland 1) Question. Two methods which are widely used for the optimization of molecular geometies are the Steepest descents and Newton-Raphson
Example questions for Molecular modelling (Level 4) Dr. Adrian Mulholland 1) Question. Two methods which are widely used for the optimization of molecular geometies are the Steepest descents and Newton-Raphson
Rotational Raman Spectroscopy
 Rotational Raman Spectroscopy If EM radiation falls upon an atom or molecule, it may be absorbed if the energy of the radiation corresponds to the separation of two energy levels of the atoms or molecules.
Rotational Raman Spectroscopy If EM radiation falls upon an atom or molecule, it may be absorbed if the energy of the radiation corresponds to the separation of two energy levels of the atoms or molecules.
Molecular Symmetry 10/25/2018
 Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy). Predict IR spectra or Interpret UV-Vis spectra Predict optical activity
Symmetry helps us understand molecular structure, some chemical properties, and characteristics of physical properties (spectroscopy). Predict IR spectra or Interpret UV-Vis spectra Predict optical activity
Density Functional Theory Investigation of The Physical Properties of Dicyano Pyridazine Molecules
 Density Functional Theory Investigation of The Physical Properties of Dicyano Pyridazine Molecules Fouad N. Ajeel 1, Alaa M. Khudhair 2, Anees A. Mohammed 3 1,2,3 Department of Physics, College of Sciences,
Density Functional Theory Investigation of The Physical Properties of Dicyano Pyridazine Molecules Fouad N. Ajeel 1, Alaa M. Khudhair 2, Anees A. Mohammed 3 1,2,3 Department of Physics, College of Sciences,
Structural, Electronic, Topological and Vibrational Properties of Isomers of Thymidine - A Study Combining Spectroscopic Data with DFT Calculations
 Human Journals Research Article December 2017 Vol.:8, Issue:2 All rights are reserved by Silvia Antonia Brandán et al. Structural, Electronic, Topological and Vibrational Properties of Isomers of Thymidine
Human Journals Research Article December 2017 Vol.:8, Issue:2 All rights are reserved by Silvia Antonia Brandán et al. Structural, Electronic, Topological and Vibrational Properties of Isomers of Thymidine
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Tuning Color Through Substitution
 1 Tuning Color Through Substitution Introduction In this experiment, the effect of substituents on the absorbance spectra of molecules will be related to the structure of the molecular orbitals involved
1 Tuning Color Through Substitution Introduction In this experiment, the effect of substituents on the absorbance spectra of molecules will be related to the structure of the molecular orbitals involved
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Enhanced Catalytic Activity and Magnetization of Encapsulated
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Enhanced Catalytic Activity and Magnetization of Encapsulated
Density Functional Theory Analysis of Electronic Structure Variations across the Orthoquinone/Semiquinone/Catechol Redox Series
 J. Phys. Chem. A 1999, 103, 4101-4112 4101 Density Functional Theory Analysis of Electronic Structure Variations across the Orthoquinone/Semiquinone/Catechol Redox Series Daniel E. Wheeler, Jorge H. Rodriguez,
J. Phys. Chem. A 1999, 103, 4101-4112 4101 Density Functional Theory Analysis of Electronic Structure Variations across the Orthoquinone/Semiquinone/Catechol Redox Series Daniel E. Wheeler, Jorge H. Rodriguez,
One Q partial negative, the other partial negative Ø H- bonding particularly strong. Abby Carroll 2
 Chemistry Notes v Polarity Experiment Ø Things involved Polarity Solubility Dispersion Ø Polarity Shaving cream has soap steric acid Water is polar Food coloring is polar/ionic because dissolved Like dissolves
Chemistry Notes v Polarity Experiment Ø Things involved Polarity Solubility Dispersion Ø Polarity Shaving cream has soap steric acid Water is polar Food coloring is polar/ionic because dissolved Like dissolves
Molecular Symmetry. Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals
 Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Molecular Symmetry Symmetry is relevant to: spectroscopy, chirality, polarity, Group Theory, Molecular Orbitals - A molecule has a symmetry element if it is unchanged by a particular symmetry operation
Electronic structure Crystal-field theory Ligand-field theory. Electronic-spectra electronic spectra of atoms
 Chapter 19 d-metal complexes: electronic structure and spectra Electronic structure 19.1 Crystal-field theory 19.2 Ligand-field theory Electronic-spectra 19.3 electronic spectra of atoms 19.4 electronic
Chapter 19 d-metal complexes: electronic structure and spectra Electronic structure 19.1 Crystal-field theory 19.2 Ligand-field theory Electronic-spectra 19.3 electronic spectra of atoms 19.4 electronic
DENSITY FUNCTIONAL THEORY STUDIES ON IR SPECTRA OF THE TRIPHENYLENE DERIVATIVES. A SCALED QUANTUM MECHANICAL FORCE FIELD APPROACH
 Vol. 98 (2000) ACTA PHYSICA POLONICA A No. 5 Proceedings of the International Conference "Condensed Matter Physics", Jaszowiec 2000 DENSITY FUNCTIONAL THEORY STUDIES ON IR SPECTRA OF THE TRIPHENYLENE DERIVATIVES.
Vol. 98 (2000) ACTA PHYSICA POLONICA A No. 5 Proceedings of the International Conference "Condensed Matter Physics", Jaszowiec 2000 DENSITY FUNCTIONAL THEORY STUDIES ON IR SPECTRA OF THE TRIPHENYLENE DERIVATIVES.
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding.
 Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
Quantum Chemistry. NC State University. Lecture 5. The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy
 Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
Quantum Chemistry Lecture 5 The electronic structure of molecules Absorption spectroscopy Fluorescence spectroscopy NC State University 3.5 Selective absorption and emission by atmospheric gases (source:
A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility
 (P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
(P&S Ch 5; Fer Ch 2, 9; Palm Ch 10,11; Zub Ch 9) A. Reaction Mechanisms and Catalysis (1) proximity effect (2) acid-base catalysts (3) electrostatic (4) functional groups (5) structural flexibility B.
Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory
 Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory Electronic Structure of Six and Four-Coordinate Complexes Using Crystal Field Theory, we can generate energy level
Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory Electronic Structure of Six and Four-Coordinate Complexes Using Crystal Field Theory, we can generate energy level
Chem 673, Problem Set 5 Due Thursday, November 29, 2007
 Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
one ν im: transition state saddle point
 Hypothetical Potential Energy Surface Ethane conformations Hartree-Fock theory, basis set stationary points all ν s >0: minimum eclipsed one ν im: transition state saddle point multiple ν im: hilltop 1
Hypothetical Potential Energy Surface Ethane conformations Hartree-Fock theory, basis set stationary points all ν s >0: minimum eclipsed one ν im: transition state saddle point multiple ν im: hilltop 1
Journal of Chemical and Pharmaceutical Research, 2012, 4(6): Research Article
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2012, 4(6): 3287-3296 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Vibrational, Structural and Electronic properties
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research, 2012, 4(6): 3287-3296 Research Article ISSN : 0975-7384 CODEN(USA) : JCPRC5 Vibrational, Structural and Electronic properties
Visualization in Chemistry. Giovanni Morelli
 Visualization in Chemistry Giovanni Morelli What is Chemistry? If we provide a good answer to this question maybe we can understand what graphics can do for chemistry and chemists What is Chemistry? A
Visualization in Chemistry Giovanni Morelli What is Chemistry? If we provide a good answer to this question maybe we can understand what graphics can do for chemistry and chemists What is Chemistry? A
Journal of Chemical and Pharmaceutical Research
 Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4): 589-595 Altering the electronic properties of adamantane
Available on line www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(4): 589-595 Altering the electronic properties of adamantane
A Eşme* Department of Elementary Science Education, Kocaeli University, Kocaeli 41380, Turkey
 Indian Journal of Pure & Applied Physics Vol. 55, July 2017, pp. 478-489 Quantum chemical calculations on the geometrical, conformational, spectroscopic (FTIR, FT-Raman) analysis and NLO activity of milrinone
Indian Journal of Pure & Applied Physics Vol. 55, July 2017, pp. 478-489 Quantum chemical calculations on the geometrical, conformational, spectroscopic (FTIR, FT-Raman) analysis and NLO activity of milrinone
NMR and IR spectra & vibrational analysis
 Lab 5: NMR and IR spectra & vibrational analysis A brief theoretical background 1 Some of the available chemical quantum methods for calculating NMR chemical shifts are based on the Hartree-Fock self-consistent
Lab 5: NMR and IR spectra & vibrational analysis A brief theoretical background 1 Some of the available chemical quantum methods for calculating NMR chemical shifts are based on the Hartree-Fock self-consistent
J.Phys. & Theo.Chem.I.A.U. Iran M.Monajjemi et al. Vol.4, No.1, Spring 2007
 Journal of Physical & Theoretical Chemistry Islamic Azad University of Iran 4 (1) (27) Science and Research Campus ISSN: 1735-2126 AB Initio Calculations and IR Studies of Tautometric forms of Uracil and
Journal of Physical & Theoretical Chemistry Islamic Azad University of Iran 4 (1) (27) Science and Research Campus ISSN: 1735-2126 AB Initio Calculations and IR Studies of Tautometric forms of Uracil and
Ab initio and DFT study of the thermodynamic properties and nuclear magnetic resonance of (4, 4) armchair AlN nanotubes
 Ab initio and DFT study of the thermodynamic properties and nuclear magnetic resonance of (4, 4) armchair AlN nanotubes Elham Pournamdari * Department of science, Islamshahr Branch, Islamic Azad University,
Ab initio and DFT study of the thermodynamic properties and nuclear magnetic resonance of (4, 4) armchair AlN nanotubes Elham Pournamdari * Department of science, Islamshahr Branch, Islamic Azad University,
DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE
 INTERNATIONAL JOURNAL OF RESEARCH IN PHARMACY AND CHEMISTRY Available online at www.ijrpc.com Research Article DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE Raghab Parajuli* Department
INTERNATIONAL JOURNAL OF RESEARCH IN PHARMACY AND CHEMISTRY Available online at www.ijrpc.com Research Article DFT STUDY OF Co 2+ AND Fe 2+ - URACIL COMPLEXES IN THE GAS PHASE Raghab Parajuli* Department
Nonlinear Polarizabilities of Donor-Acceptor Substituted Conjugated Polyenes
 11080 J. Phys. Chem. 1996, 100, 11080-11085 Nonlinear Polarizabilities of Donor-Acceptor Substituted Conjugated Polyenes Guanhua Chen and Shaul Mukamel* Department of Chemistry, UniVersity of Rochester,
11080 J. Phys. Chem. 1996, 100, 11080-11085 Nonlinear Polarizabilities of Donor-Acceptor Substituted Conjugated Polyenes Guanhua Chen and Shaul Mukamel* Department of Chemistry, UniVersity of Rochester,
Be H. Delocalized Bonding. Localized Bonding. σ 2. σ 1. Two (sp-1s) Be-H σ bonds. The two σ bonding MO s in BeH 2. MO diagram for BeH 2
 The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
The Delocalized Approach to Bonding: The localized models for bonding we have examined (Lewis and VBT) assume that all electrons are restricted to specific bonds between atoms or in lone pairs. In contrast,
Supporting Information
 Supporting Information Remote Stereoinductive Intramolecular Nitrile Oxide Cycloaddition: Asymmetric Total Synthesis and Structure Revision of ( )-11 -Hydroxycurvularin Hyeonjeong Choe, Thuy Trang Pham,
Supporting Information Remote Stereoinductive Intramolecular Nitrile Oxide Cycloaddition: Asymmetric Total Synthesis and Structure Revision of ( )-11 -Hydroxycurvularin Hyeonjeong Choe, Thuy Trang Pham,
Hydrogen bonding in oxalic acid and its complexes: A database study of neutron structures
 PRAMANA c Indian Academy of Sciences Vol. 63, No. 2 journal of August 2004 physics pp. 263 269 Hydrogen bonding in oxalic acid and its complexes: A database study of neutron structures R CHITRA, AMIT DAS,
PRAMANA c Indian Academy of Sciences Vol. 63, No. 2 journal of August 2004 physics pp. 263 269 Hydrogen bonding in oxalic acid and its complexes: A database study of neutron structures R CHITRA, AMIT DAS,
Exchange Correlation Functional Investigation of RT-TDDFT on a Sodium Chloride. Dimer. Philip Straughn
 Exchange Correlation Functional Investigation of RT-TDDFT on a Sodium Chloride Dimer Philip Straughn Abstract Charge transfer between Na and Cl ions is an important problem in physical chemistry. However,
Exchange Correlation Functional Investigation of RT-TDDFT on a Sodium Chloride Dimer Philip Straughn Abstract Charge transfer between Na and Cl ions is an important problem in physical chemistry. However,
Close agreement between the orientation dependence of hydrogen bonds observed in protein structures and quantum mechanical calculations
 Close agreement between the orientation dependence of hydrogen bonds observed in protein structures and quantum mechanical calculations Alexandre V. Morozov, Tanja Kortemme, Kiril Tsemekhman, David Baker
Close agreement between the orientation dependence of hydrogen bonds observed in protein structures and quantum mechanical calculations Alexandre V. Morozov, Tanja Kortemme, Kiril Tsemekhman, David Baker
Analysis of Permanent Electric Dipole Moments of Aliphatic Amines.
 Analysis of Permanent Electric Dipole Moments of Aliphatic Amines. Boris Lakard* LPUB, UMR CNRS 5027, University of Bourgogne, F-21078, Dijon, France Internet Electronic Conference of Molecular Design
Analysis of Permanent Electric Dipole Moments of Aliphatic Amines. Boris Lakard* LPUB, UMR CNRS 5027, University of Bourgogne, F-21078, Dijon, France Internet Electronic Conference of Molecular Design
= (1) V = (12) Å 3 Z =4 Mo K radiation. Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections
![= (1) V = (12) Å 3 Z =4 Mo K radiation. Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections = (1) V = (12) Å 3 Z =4 Mo K radiation. Data collection. Refinement. R[F 2 >2(F 2 )] = wr(f 2 ) = S = reflections](/thumbs/81/83180848.jpg) organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 S-Benzylisothiouronium nitrate P. Hemalatha a and V. Veeravazhuthi b * a Department of Physics, PSG College of
organic compounds Acta Crystallographica Section E Structure Reports Online ISSN 1600-5368 S-Benzylisothiouronium nitrate P. Hemalatha a and V. Veeravazhuthi b * a Department of Physics, PSG College of
Trace Solvent as a Predominant Factor to Tune Dipeptide. Self-Assembly
 Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
Electronic and Optoelectronic Polymers
 Electronic and Optoelectronic Polymers Wen-Chang Chen Department of Chemical Engineering Institute of Polymer Science and Engineering National Taiwan University Outlines History of Conjugated Polymers
Electronic and Optoelectronic Polymers Wen-Chang Chen Department of Chemical Engineering Institute of Polymer Science and Engineering National Taiwan University Outlines History of Conjugated Polymers
Vibrational Spectroscopy
 Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
QUANTUM MECHANICS AND MOLECULAR STRUCTURE
 6 QUANTUM MECHANICS AND MOLECULAR STRUCTURE 6.1 Quantum Picture of the Chemical Bond 6.2 Exact Molecular Orbital for the Simplest Molecule: H + 2 6.3 Molecular Orbital Theory and the Linear Combination
6 QUANTUM MECHANICS AND MOLECULAR STRUCTURE 6.1 Quantum Picture of the Chemical Bond 6.2 Exact Molecular Orbital for the Simplest Molecule: H + 2 6.3 Molecular Orbital Theory and the Linear Combination
GROWTH, THERMAL, OPTICAL, VIBRATIONAL PROPERTIES AND HYPERPOLARIZABILITY OF PICRATES : ORTHONITROANILINE WITH PICRIC ACID AND IMIDAZOLIUM PICRATE
 39 CHAPTER 2 GROWTH, THERMAL, OPTICAL, VIBRATIONAL PROPERTIES AND HYPERPOLARIZABILITY OF PICRATES : ORTHONITROANILINE WITH PICRIC ACID AND IMIDAZOLIUM PICRATE 2.1 INTRODUCTION Nonlinear optics plays a
39 CHAPTER 2 GROWTH, THERMAL, OPTICAL, VIBRATIONAL PROPERTIES AND HYPERPOLARIZABILITY OF PICRATES : ORTHONITROANILINE WITH PICRIC ACID AND IMIDAZOLIUM PICRATE 2.1 INTRODUCTION Nonlinear optics plays a
