Electrophilic substitution Both words needed Ignore minor misspellings 1
|
|
|
- Gary Smith
- 5 years ago
- Views:
Transcription
1 M.(a) Electrophilic substitution Both words needed Ignore minor misspellings (b) (i) Sn / HCl OR H 2 / Ni OR H 2 / Pt OR Fe / HCl OR Zn / HCl OR SnCl 2 / HCl Ignore conc or dil with HCl, Allow (dil) H 2 SO 4 but not conc H 2 SO 4 Not allow HNO 3 or H + Ignore NaOH after Sn / HCl Ignore catalyst (ii) CH 3 C 6 H 4 NO 2 + 6[H] CH 3 C 6 H 4 NH 2 + 2H 2 O OR Allow molecular formulae as structures given C 7 H 7 NO 2 + 6[H] C 7 H 9 N + 2H 2 O Qu states use [H], so penalised 3H 2 (iii) making dyes OR making quaternary ammonium salts OR making (cationic) surfactants OR making hair conditioner OR making fabric softener OR making detergents (c) Page 2
2 NO Mark for name of mechanism Allow SN M for lone pair on N and arrow to C or mid point of space between N and C M2 for arrow from bond to Br M3 for structure of protonated secondary amine M4 for arrow from bond to N or + on N For M4: ignore RNH 2 or NH 3 removing H + but penalise Br 4 M3 (d) lone or electron pair on N If no mention of lone pair CE = 0 If lone pair mentioned but not on N then lose M and mark on M in J spread / delocalised into ring (or not delocalised in K) Ignore negative inductive effect of benzene Allow interacts with Π cloud for M2 M2 less available (for protonation or donation in J) M3 OR in K there is a positive inductive effect / electron releasing) M2 more available (for protonation or donation in K) Page 3
3 M3 [] M2.(a) M Ester If Ester 2, can score M3 only. M2 peak at δ = 4. due to When marking M2 and M3, check any annotation of structures in the stem at the top of the page. M3 (δ = 4. peak is) quartet as adjacent / next to / attached to CH 3 M4 Other spectrum quartet at δ = (or value in this range) (b) M Quaternary (alkyl) ammonium salt / bromide M2 CH 3Br or bromomethane Penalise contradictory formula and name. M3 Excess ( CH 3Br or bromomethane) Mention of acid eg H 2SO 4 OR alkali eg NaOH loses both M2 and M3. Page 4
4 M4 Nucleophilic substitution Can only score M3 if reagent correct. Ignore alcohol or ethanol (conditions) or Temp. (c) Bromine (penalise Br but mark on) Acidified KMnO 4 (Penalise missing acid but mark on) Wrong reagent = no marks. If bromine colour stated it must be red, yellow, orange, brown or any combination, penalise wrong starting colour. Benzene no reaction / c olour remains / no (visible) change no reaction / colour remains / no (visible) change Ignore clear, nothing. Allow colour fades slowly. Allow nvc for no visible change. cyclohexene (Bromine) decolourised (Acidified KMnO 4) decolourised [] M3. (a) (i) CH 3CH=CHCH 3 Addition or radical (QoL) Page 5
5 (ii) CH 3CH(OH)CH(OH)CH 3 or with no brackets butan(e) 2,3 diol or 2,3 butan(e)diol 2,3 dimethylbutan(e)dioic acid ignore,4 condensation (QoL) 2,3 dimethylbutan(e)dioyl chloride (iii) NaOH or HCl etc or Na 2CO 3 Allow conc sulphuric/nitric NOT water nor acidified water nor weak acids (b) Structure Allow CONH and COHN Allow zwitterions NOT polypeptides/repeating units Structure 2 either of (c) (i) CH 3CH 2CH 2Br allow Cl, I (ii) CH 3CH 2CN Page 6
6 (iii) (nucleophilic) substitution or from CH 3CH 2CH 2Br if reduction written here, no further marks further substitution/reaction occurs or other products are formed Allow reduction forms only one product one of (CH 3CH 2CH 2) 2NH (CH 3CH 2CH 2) 3N (CH 3CH 2CH 2) 4N + Br Allow salts including NH 4Br Allow HBr [5] M4.(a) (nucleophilic) addition-elimination M4 for 3 arrows and lp Allow wrong amine in M but penalise in M3 Allow C 3H 7 in M3 Minus sign on NH 3 loses M (but not M4 if NH 3 also shown here) Allow attack by: NH 2CH 2CH 2CH 3 M2 not allowed independent of M, but allow M for correct attack on C + + rather than δ+ on C=O loses M2 If Cl lost with C=O breaking, max for M Page 7
7 M3 for correct structure with charges but lone pair on O is part of M4 3 arrows in M4 can be shown in two separate steps. If M3 drawn twice, mark first answer eg ignore missing + if missed off second structure Only allow M4 after correct / very close M3 For M4, ignore RNH 2 removing H + but lose M4 for Cl removing H + in mechanism, but ignore HCl shown as a product. 4 N-propylethanamide must be this name even if wrong amine used NOT N-propylethaneamide (b) (i) Not allow ambiguous C 3H 7NH 2 BEWARE No mark for the original amine CH 3CH 2CH 2NH 2 Label and structure must both be correct for each type to score the mark. Allow C 2H 5 Penalize wrong number of carbons but otherwise correct, first time only. (ii) Absorption at (cm ) in spectrum Allow trough, peak, spike. Ignore absorption at for C C bond in secondary - this is within fingerprint region. Page 8
8 Allow any number in this range. If range missing, no further marks. If range linked to tertiary, no further marks. N H (bond) (only) present in secondary amine or not present in tertiary amine OR This peak or N H absorption (only) present in spectrum of secondary amine or not present in spectrum of tertiary amine (c) (i) M Route A: stage KCN Apply list principle for extra reagents or catalysts NOT HCN NOT KCN / acid Not KCN / HCN M2 Aqueous or ethanolic M2 only scores after correct M ignore warm; acid here loses M & M2 M3 Route A Intermediate CH 3CH 2CN or propanenitrile If M3 intermediate wrong, max 2 for M & M2 ie no mark for stage 2 Name alone must be exactly correct to gain M but mark on if name close But if M3 intermediate close, eg nitrile or wrong nitrile, can award marks in stage 2 correct formula gains M (ignore name if close) If stage correct and intermediate is missing, can award marks in stage 2 contradiction of name and formula loses mark stage wrong & intermediate missing, no marks. M4 Route A: stage 2 H 2 H loses M4 but mark on LiAlH 4 Apply list principle for extra reagents or catalysts. Page 9
9 M5 only scores after correct M4 Not NaBH 4 not Sn or Fe / HCl Allow (dil) acid after but not with LiAlH 4 Penalise conc acid. M5 Ni or Pt or Pd ether M6 Route B NH 3 With acid loses M6 & M7 Apply list principle for extra reagents or catalysts. M7 Excess NH 3 Ignore conc, ignore high P, ignore solvent. (ii) Route A disadv Toxic / poisonous KCN or cyanide or CN or HCN Expensive LiAlH 4 ignore acidified OR lower yield because 2 steps Allow H 2 flammable / explosive etc. Not just dangerous. Ignore time reasons. Route B disadv Allow impure product. Further reaction / substitution likely [20] Page 0
Electrophilic substitution Both words needed Ignore minor misspellings 1
 M.(a) Electrophilic substitution Both words needed Ignore minor misspellings (b) (i) Sn / HCl H 2 / Ni H 2 / Pt Fe / HCl Zn / HCl SnCl 2 / HCl Ignore conc or dil with HCl, Allow (dil) H 2 SO 4 but not
M.(a) Electrophilic substitution Both words needed Ignore minor misspellings (b) (i) Sn / HCl H 2 / Ni H 2 / Pt Fe / HCl Zn / HCl SnCl 2 / HCl Ignore conc or dil with HCl, Allow (dil) H 2 SO 4 but not
Final. Mark Scheme. Chemistry CHEM4. (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry
 ERRY ILL TUITI AQA EMISTRY A2 PAPER 30 MARK SEME Question : /A Question 2: /A Question 3: /A General ertificate of Education (A-level) June 203 hemistry EM4 (Specification 2420) Unit 4: Kinetics, Equilibria
ERRY ILL TUITI AQA EMISTRY A2 PAPER 30 MARK SEME Question : /A Question 2: /A Question 3: /A General ertificate of Education (A-level) June 203 hemistry EM4 (Specification 2420) Unit 4: Kinetics, Equilibria
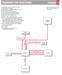
Consider the following reaction sequence starting from methylbenzene.... (1)... (1)
 Consider the following reaction sequence starting from methylbenzene. (a) Name the type of mechanism for reaction... () (b) Compound J is formed by reduction in reaction 2. (i) Give a reducing agent for
Consider the following reaction sequence starting from methylbenzene. (a) Name the type of mechanism for reaction... () (b) Compound J is formed by reduction in reaction 2. (i) Give a reducing agent for
Mechanisms. . CCl2 F + Cl.
 Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
Mechanisms 1) Free radical substitution Alkane à halogenoalkane Initiation: Propagation: Termination: Overall: 2) Ozone depletion UV light breaks the C Cl bond releasing chlorine radical CFCl 3 F à. CCl2
Allow CONH- or - COHN - 1(a)(i) Mark two halves separately
 ERRY ILL TUITIN AQA EMISTRY A2 PAPER 27 MARK SEME Question Marking Guidance (a)(i) 2 2 N N 2 Mark Allow N- or - N - N 6 Mark two halves separately 2 N 6 omments lose each for missing trailing bonds at
ERRY ILL TUITIN AQA EMISTRY A2 PAPER 27 MARK SEME Question Marking Guidance (a)(i) 2 2 N N 2 Mark Allow N- or - N - N 6 Mark two halves separately 2 N 6 omments lose each for missing trailing bonds at
Page 2. (1)-methylethyl ethanoate OR. Propan-2-yl ethanoate Ignore extra or missing spaces, commas or hyphens 1. (ii) M4 for 3 arrows and lp
 M.(a) (i) (CH 3 ) 2 CHOH + (CH 3 CO) 2 O CH 3 COOCH(CH 3 ) 2 + CH 3 COOH Allow CH 3 CO 2 CH(CH 3 ) 2 and CH 3 CO 2 H Ignore (CH 3 ) 2 C in equation ()-methylethyl ethanoate OR Propan-2-yl ethanoate Ignore
M.(a) (i) (CH 3 ) 2 CHOH + (CH 3 CO) 2 O CH 3 COOCH(CH 3 ) 2 + CH 3 COOH Allow CH 3 CO 2 CH(CH 3 ) 2 and CH 3 CO 2 H Ignore (CH 3 ) 2 C in equation ()-methylethyl ethanoate OR Propan-2-yl ethanoate Ignore
AMINES HYDROXYNITRILES
 AMINES ACYL CHLORIDES HYDROXYNITRILES ALDEHYDE/KETONE REDUCTION Amines are nitrogen- containing organic compounds derived from ammonia, where one or more of the hydrogen atoms has been replaced by an alkyl
AMINES ACYL CHLORIDES HYDROXYNITRILES ALDEHYDE/KETONE REDUCTION Amines are nitrogen- containing organic compounds derived from ammonia, where one or more of the hydrogen atoms has been replaced by an alkyl
OCR (A) Chemistry A-level. Module 6: Organic Chemistry and Analysis
 OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
Unit 4. Answers Marks Examiner s tips. 1 a) order with respect to A = 1 order with respect to B = 2. b) i) rate = k [C][D] 2 1
![Unit 4. Answers Marks Examiner s tips. 1 a) order with respect to A = 1 order with respect to B = 2. b) i) rate = k [C][D] 2 1 Unit 4. Answers Marks Examiner s tips. 1 a) order with respect to A = 1 order with respect to B = 2. b) i) rate = k [C][D] 2 1](/thumbs/73/69433234.jpg) a) order with respect to A = order with respect to B = b) i) rate = k [C][D] 4.45 0 k = (.5 0 ) (6.65 0 ) Rate =.3 mol dm 6 s 3 If you place the units in the equation and cancel them out as you would with
a) order with respect to A = order with respect to B = b) i) rate = k [C][D] 4.45 0 k = (.5 0 ) (6.65 0 ) Rate =.3 mol dm 6 s 3 If you place the units in the equation and cancel them out as you would with
COCH 3. + HCl H 5 C 6. Not molecular formulae Not allow C. phenylethanone Ignore number 1 in name but penalise other numbers
 Not molecular formulae Not allow C 6 H 5 CH CO M. (a) (i) CH COCl + C 6 H 6 C 6 H 5 COCH + HCl OR phenylethanone Ignore number in name but penalise other numbers AlCl can be scored in equation Allow RHS
Not molecular formulae Not allow C 6 H 5 CH CO M. (a) (i) CH COCl + C 6 H 6 C 6 H 5 COCH + HCl OR phenylethanone Ignore number in name but penalise other numbers AlCl can be scored in equation Allow RHS
Page 2. M1.(a) P 3,3 dimethylbut 1 ene OR accept 3,3 dimethylbutene Ignore absence of commas, hyphens and gaps Require correct spelling Q OR
 M.(a) P 3,3 dimethylbut ene OR accept 3,3 dimethylbutene Ignore absence of commas, hyphens and gaps Require correct spelling Q OR 3 chloro 2,2 dimethylbutane accept 2 chloro 3,3 dimethylbutane In Q, chloro
M.(a) P 3,3 dimethylbut ene OR accept 3,3 dimethylbutene Ignore absence of commas, hyphens and gaps Require correct spelling Q OR 3 chloro 2,2 dimethylbutane accept 2 chloro 3,3 dimethylbutane In Q, chloro
Amines. Types of Amines. chemrevise.org. N Goalby Chemrevise.org. primary amine (one C attached to N) H. Secondary amine (two C s.
 Amines Goalby hemrevise.org Types of Amines primary amine (one attached to ) Secondary amine (two s attached to ) 3 2 2 3 name : diethylamine It is also possible to have aromatic amines 2 phenylamine name:
Amines Goalby hemrevise.org Types of Amines primary amine (one attached to ) Secondary amine (two s attached to ) 3 2 2 3 name : diethylamine It is also possible to have aromatic amines 2 phenylamine name:
Summary of mechanisms. Type of reaction: Nucleophilic subsitution/hydrolysis
 S Summary of mechanisms S Summary of mechanisms electrophilic addition Electrophiles: H δ in H (Ni catalyst needed), H δ in H-X; X δ in X ; H δ in H O (g) (conc H 3 PO 4 cat needed); H δ in NH 3 ; H δ
S Summary of mechanisms S Summary of mechanisms electrophilic addition Electrophiles: H δ in H (Ni catalyst needed), H δ in H-X; X δ in X ; H δ in H O (g) (conc H 3 PO 4 cat needed); H δ in NH 3 ; H δ
2.8 EXTRA QUESTIONS MS. (iii) C 6 H 5 CHCl 2 / C 6 H 5 CCl 3 / C 6 H 5 CHCHC 6 H 5 / other correct possible answer (1) 1
 .8 EXTRA QUESTIONS MS 1. (i) l l / ½ l l 1 (ii) l 6 5 6 5 l 6 5 l l 6 5 l (iii) 6 5 l / 6 5 l / 6 5 6 5 / other correct possible answer 1 [4]. (a) (i) (free)radical substitution 1 (both words required
.8 EXTRA QUESTIONS MS 1. (i) l l / ½ l l 1 (ii) l 6 5 6 5 l 6 5 l l 6 5 l (iii) 6 5 l / 6 5 l / 6 5 6 5 / other correct possible answer 1 [4]. (a) (i) (free)radical substitution 1 (both words required
Class XII: Chemistry Chapter 13: Amines Top concepts
 Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Class XII: Chemistry Chapter 13: Amines Top concepts 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification
Page 2. M1.(a) (i) (nucleophilic) addition-elimination Not electrophilic addition-elimination Ignore esterification 1
 M.(a) (i) (nucleophilic) addition-elimination Not electrophilic addition-elimination Ignore esterification M3 for structure If wrong nucleophile used or O H broken in first step, can only score M2. M2
M.(a) (i) (nucleophilic) addition-elimination Not electrophilic addition-elimination Ignore esterification M3 for structure If wrong nucleophile used or O H broken in first step, can only score M2. M2
6.10 Amines. Naming. N Goalby chemrevise.org 1 CH2 CH2
 6.10 Amines aming Amines These end in amine. There is, however, rather confusingly two ways of using this suffix. The exam board tend to use the common version where the name stem ends in -yl propylamine.
6.10 Amines aming Amines These end in amine. There is, however, rather confusingly two ways of using this suffix. The exam board tend to use the common version where the name stem ends in -yl propylamine.
CHEM4 Kinetics, Equilibria and Organic Chemistry Mark scheme
 AQA Qualifications A-LEVEL EMISTRY EM4 Kinetics, Equilibria and rganic hemistry Mark scheme 2420 June 204 Version:. Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
AQA Qualifications A-LEVEL EMISTRY EM4 Kinetics, Equilibria and rganic hemistry Mark scheme 2420 June 204 Version:. Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
klm Mark Scheme Chemistry 6421 General Certificate of Education Further Physical and Organic Chemistry 2010 examination - January series
 2 nd February 200 klm General Certificate of Education Chemistry 642 CHM4 Further Physical and Organic Chemistry Mark Scheme 200 examination - January series Mark schemes are prepared by the Principal
2 nd February 200 klm General Certificate of Education Chemistry 642 CHM4 Further Physical and Organic Chemistry Mark Scheme 200 examination - January series Mark schemes are prepared by the Principal
GCE A level 1094/01 CHEMISTRY CH4
 Surname ther Names Centre 2 Candidate GCE A level 1094/01 CHEMISTRY CH4 P.M. MNDAY, 14 January 2013 1¾ hours ADDITINAL MATERIALS In addition to this examination paper, you will need: Data Sheet Periodic
Surname ther Names Centre 2 Candidate GCE A level 1094/01 CHEMISTRY CH4 P.M. MNDAY, 14 January 2013 1¾ hours ADDITINAL MATERIALS In addition to this examination paper, you will need: Data Sheet Periodic
1,4-diaminobenzene is an important intermediate in the production of polymers such as Kevlar and also of polyurethanes, used in making foam seating.
 ,4-diaminobenzene is an important intermediate in the production of polymers such as Kevlar and also of polyurethanes, used in making foam seating. A possible synthesis of,4-diaminobenzene from phenylamine
,4-diaminobenzene is an important intermediate in the production of polymers such as Kevlar and also of polyurethanes, used in making foam seating. A possible synthesis of,4-diaminobenzene from phenylamine
 Top concepts Chapter: Amines 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification of amines: 3. Preparation
Top concepts Chapter: Amines 1. Amines are regarded as derivatives of ammonia in which one, two or all three hydrogen atoms are replaced by alkyl or aryl group 2. Classification of amines: 3. Preparation
abc Mark Scheme Chemistry 6421 General Certificate of Education CHM examination January series Further Physical and Organic Chemistry
 Version : 3/0/07 abc General Certificate of Education Chemistry 64 CM4 Further Physical and rganic Chemistry Mark Scheme 007 examination January series Mark schemes are prepared by the Principal Examiner
Version : 3/0/07 abc General Certificate of Education Chemistry 64 CM4 Further Physical and rganic Chemistry Mark Scheme 007 examination January series Mark schemes are prepared by the Principal Examiner
Which of the following is a correct mechanism for the formation of 2-methylbut-2-ene from 2-bromo-3-methylbutane?
 Which of the following is a correct mechanism for the formation of 2-methylbut-2-ene from 2-bromo-3-methylbutane? A B C D (Total mark) Page of 75 2 There are many uses of halogenated organic compounds
Which of the following is a correct mechanism for the formation of 2-methylbut-2-ene from 2-bromo-3-methylbutane? A B C D (Total mark) Page of 75 2 There are many uses of halogenated organic compounds
AQA Qualifications. A-LEVEL Chemistry. CHEM4 Kinetic, Equilibria and Organic Chemistry Mark scheme June Version: 1.
 AQA Qualifications A-LEVEL Chemistry CEM4 Kinetic, Equilibria and rganic Chemistry Mark scheme 2420 June 206 Version:.0 Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
AQA Qualifications A-LEVEL Chemistry CEM4 Kinetic, Equilibria and rganic Chemistry Mark scheme 2420 June 206 Version:.0 Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
7 Benzene and aromatic compounds Answers
 Practice: pages 161 163 1 Answer is D. Methyl takes precedence over nitro and, therefore, automatically takes position 1, which doesn t have to be included in the name. [1] 2 Answer is C. If Kekulé was
Practice: pages 161 163 1 Answer is D. Methyl takes precedence over nitro and, therefore, automatically takes position 1, which doesn t have to be included in the name. [1] 2 Answer is C. If Kekulé was
abc Mark Scheme Chemistry 5421 General Certificate of Education 2005 examination - June series CHM4 Further Physical and Organic Chemistry
 Version.0 General Certificate of Education abc Chemistry 542 CM4 Further Physical and Organic Chemistry Mark Scheme 2005 examination - June series Mark schemes are prepared by the Principal Examiner and
Version.0 General Certificate of Education abc Chemistry 542 CM4 Further Physical and Organic Chemistry Mark Scheme 2005 examination - June series Mark schemes are prepared by the Principal Examiner and
IGNORE Just benzene has a delocalised ring Benzene does not have C=C double bonds Any references to shape/ bond angles. Acceptable Answers Reject Mark
 1(a) All carbon to carbon bonds same length/ longer C-C and shorter C=C not present 1 IGNORE Just benzene has a delocalised ring Benzene does not have C=C double bonds Any references to shape/ bond angles
1(a) All carbon to carbon bonds same length/ longer C-C and shorter C=C not present 1 IGNORE Just benzene has a delocalised ring Benzene does not have C=C double bonds Any references to shape/ bond angles
Acceptable sequence of stages are: chlorination. nitration, reduction, chlorination nitration. nitration, chlorination, reduction, reduction
 Question er Mark Guidance 1 (a) (i) Response requires three stages Acceptable sequence of stages are: chlorination nitration, reduction, chlorination nitration nitration, chlorination, reduction, reduction
Question er Mark Guidance 1 (a) (i) Response requires three stages Acceptable sequence of stages are: chlorination nitration, reduction, chlorination nitration nitration, chlorination, reduction, reduction
Topic 4.10 ORGANIC SYNTHESIS AND ANALYSIS. Organic analysis Organic synthesis
 Topic 4.10 ORGANIC SYNTHESIS AND ANALYSIS Organic analysis Organic synthesis DISTINGUISHING BETWEEN DIFFERENT ORGANIC COMPOUNDS Many of the organic compounds prepared in AS Unit 2 and in A2 Unit 4 can
Topic 4.10 ORGANIC SYNTHESIS AND ANALYSIS Organic analysis Organic synthesis DISTINGUISHING BETWEEN DIFFERENT ORGANIC COMPOUNDS Many of the organic compounds prepared in AS Unit 2 and in A2 Unit 4 can
Final. Mark Scheme. Chemistry CHEM4. (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry
 Version.0 General ertificate of Education (A-level) June 203 hemistry EM4 (Specification 2420) Unit 4: Kinetics, Equilibria and rganic hemistry Final Mark Scheme Mark schemes are prepared by the Principal
Version.0 General ertificate of Education (A-level) June 203 hemistry EM4 (Specification 2420) Unit 4: Kinetics, Equilibria and rganic hemistry Final Mark Scheme Mark schemes are prepared by the Principal
TOPIC 13 ANSWERS & MARK SCHEMES QUESTIONSHEET 1 ALKANES
 QUESTIONSHEET 1 ALKANES a) (i) UV light / temperatures > 500 ºC (ii) CH 4 (g) + Cl 2 (g) Cl(g) + HCl(g) Cl(g) + Cl 2 (g) Cl 2 (l) + HCl(g) Cl 2 (l) + Cl 2 (g) CHCl 3 (l) + HCl(g) CHCl 3 (l) + Cl 2 (g)
QUESTIONSHEET 1 ALKANES a) (i) UV light / temperatures > 500 ºC (ii) CH 4 (g) + Cl 2 (g) Cl(g) + HCl(g) Cl(g) + Cl 2 (g) Cl 2 (l) + HCl(g) Cl 2 (l) + Cl 2 (g) CHCl 3 (l) + HCl(g) CHCl 3 (l) + Cl 2 (g)
Version 1.0: 1006 abc. General Certificate of Education examination - June series
 Version.0: 006 abc General Certificate of Education Chemistry 542 CHM3/W Introduction to Organic Chemistry Mark Scheme 2006 examination - June series Mark schemes are prepared by the Principal Examiner
Version.0: 006 abc General Certificate of Education Chemistry 542 CHM3/W Introduction to Organic Chemistry Mark Scheme 2006 examination - June series Mark schemes are prepared by the Principal Examiner
Final. Mark Scheme. Chemistry CHEM4. (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry
 Version General Certificate of Education (A-level) January 202 Chemistry CHEM4 (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry Final Mark Scheme Mark schemes are prepared by the
Version General Certificate of Education (A-level) January 202 Chemistry CHEM4 (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry Final Mark Scheme Mark schemes are prepared by the
Organic Chemistry SL IB CHEMISTRY SL
 Organic Chemistry SL IB CHEMISTRY SL 10.1 Fundamentals of organic chemistry Understandings: A homologous series is a series of compounds of the same family, with the same general formula, which differ
Organic Chemistry SL IB CHEMISTRY SL 10.1 Fundamentals of organic chemistry Understandings: A homologous series is a series of compounds of the same family, with the same general formula, which differ
Química Orgânica I. Ciências Farmacêuticas Bioquímica Química AFB QO I 2007/08 1
 Química rgânica I Ciências Farmacêuticas Bioquímica Química AFB Q I 2007/08 1 alcohols Adaptado de rganic Chemistry, 6th Edition; Wade rganic Chemistry, 6 th Edition; McMurry AFB Q I 2007/08 2 Typical
Química rgânica I Ciências Farmacêuticas Bioquímica Química AFB Q I 2007/08 1 alcohols Adaptado de rganic Chemistry, 6th Edition; Wade rganic Chemistry, 6 th Edition; McMurry AFB Q I 2007/08 2 Typical
Amines. Introduction Organic derivatives of ammonia. Many are biologically active.
 Amines Introduction Organic derivatives of ammonia. Many are biologically active. 1 Biological Activity Neurotransmitters: dopamine Bioregulators: epinephrine Vitamins: niacin, B 6 Alkaloids: nicotine,
Amines Introduction Organic derivatives of ammonia. Many are biologically active. 1 Biological Activity Neurotransmitters: dopamine Bioregulators: epinephrine Vitamins: niacin, B 6 Alkaloids: nicotine,
CHEM 343 Principles of Organic Chemistry II Summer Instructor: Paul J. Bracher. Quiz # 3. Monday, July 21 st, :30 a.m.
 CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Solutions Key Page 1 of 9 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21 st,
CHEM 343 Principles of Organic Chemistry II Summer 2014 Quiz # 3 Solutions Key Page 1 of 9 CHEM 343 Principles of Organic Chemistry II Summer 2014 Instructor: Paul J. Bracher Quiz # 3 Monday, July 21 st,
18.1 Arenes benzene compounds Answers to Exam practice questions
 Pages 230 232 1 a) Benzene has a planar molecule ; with six carbon atoms in a regular hexagon. Each carbon atom forms a normal covalent ( ) bond with its two adjacent carbons atoms and a hydrogen atom.
Pages 230 232 1 a) Benzene has a planar molecule ; with six carbon atoms in a regular hexagon. Each carbon atom forms a normal covalent ( ) bond with its two adjacent carbons atoms and a hydrogen atom.
Ammonia Primary Secondary Tertiary Quarternary Ammonium Ion
 1 Chapter 19: Amines I. Introduction: Classification: NH 3 RNH 2 R 2 NH R 3 N R 4 N + Ammonia Primary Secondary Tertiary Quarternary Ammonium Ion Amines are a very common functional group in organic chemistry,
1 Chapter 19: Amines I. Introduction: Classification: NH 3 RNH 2 R 2 NH R 3 N R 4 N + Ammonia Primary Secondary Tertiary Quarternary Ammonium Ion Amines are a very common functional group in organic chemistry,
AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS
 AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS 1 1. Consider the following reaction sequence. CH 3 CH 3 CH 3 Step 1
AQA A2 CHEMISTRY TOPIC 4.10 ORGANIC SYNTHESIS AND ANALYSIS TOPIC 4.11 STRUCTURE DETERMINATION BOOKLET OF PAST EXAMINATION QUESTIONS 1 1. Consider the following reaction sequence. CH 3 CH 3 CH 3 Step 1
Cambridge Assessment International Education Cambridge International Advanced Subsidiary and Advanced Level. Published
 Cambridge Assessment International Education Cambridge International Advanced Subsidiary and Advanced Level CHEMISTRY 970/4 Paper 4 A Level Structured Questions 207 MARK SCHEME Maximum Mark: 00 Published
Cambridge Assessment International Education Cambridge International Advanced Subsidiary and Advanced Level CHEMISTRY 970/4 Paper 4 A Level Structured Questions 207 MARK SCHEME Maximum Mark: 00 Published
Haloalkanes. Isomers: Draw and name the possible isomers for C 5 H 11 Br
 Haloalkanes The basics: The functional group is a halogen atom: F, Cl, Br or I General formula C n H 2n+1 X Use the prefixes: fluoro, chloro, bromo and iodo. Isomers: Draw and name the possible isomers
Haloalkanes The basics: The functional group is a halogen atom: F, Cl, Br or I General formula C n H 2n+1 X Use the prefixes: fluoro, chloro, bromo and iodo. Isomers: Draw and name the possible isomers
3.2.9 Alkenes. Polymerisation. 152 minutes. 149 marks. Page 1 of 22
 ..9 Alkenes Polymerisation 5 minutes 49 marks Page of M. Oxygen or air () Can score from the equation Silver catalyst () Linked to process: e.g. Consequence of leaks etc. () Hazard = flammable or explosive
..9 Alkenes Polymerisation 5 minutes 49 marks Page of M. Oxygen or air () Can score from the equation Silver catalyst () Linked to process: e.g. Consequence of leaks etc. () Hazard = flammable or explosive
MARK SCHEME for the May/June 2010 question paper for the guidance of teachers 9701 CHEMISTRY
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS GCE Advanced Subsidiary Level and GCE Advanced Level MARK SCHEME for the May/June 2010 question paper for the guidance of teachers 9701 CHEMISTRY 9701/41
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS GCE Advanced Subsidiary Level and GCE Advanced Level MARK SCHEME for the May/June 2010 question paper for the guidance of teachers 9701 CHEMISTRY 9701/41
AQA Qu a lifica t ions A- LEVEL CHEMISTRY. CHEM4 Kinetics, Equilibria and Organic Chemistry Mark scheme June 2015.
 AQA Qu a lifica t ions A- LEVEL EMISTRY EM4 Kinetics, Equilibria and Organic hemistry Mark scheme 40 June 05 Version : Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
AQA Qu a lifica t ions A- LEVEL EMISTRY EM4 Kinetics, Equilibria and Organic hemistry Mark scheme 40 June 05 Version : Final Mark schemes are prepared by the Lead Assessment Writer and considered, together
Page (Extra space) (4) Benzene can be converted into amine U by the two-step synthesis shown below.
 Q1. The hydrocarbons benzene and cyclohexene are both unsaturated compounds. Benzene normally undergoes substitution reactions, but cyclohexene normally undergoes addition reactions. (a) The molecule cyclohexatriene
Q1. The hydrocarbons benzene and cyclohexene are both unsaturated compounds. Benzene normally undergoes substitution reactions, but cyclohexene normally undergoes addition reactions. (a) The molecule cyclohexatriene
M1. (a) Functional group (isomerism) 1
 M. (a) Functional group (isomerism) (b) M Tollens (reagent) (Credit ammoniacal silver nitrate a description of making Tollens ) (Ignore either AgNO or [Ag(NH ) 2 + ] or the silver mirror test on their
M. (a) Functional group (isomerism) (b) M Tollens (reagent) (Credit ammoniacal silver nitrate a description of making Tollens ) (Ignore either AgNO or [Ag(NH ) 2 + ] or the silver mirror test on their
Mechanism Summary for A-level AQA Chemistry
 Mechanism Summary for Alevel AQA hemistry Electrophilic Addition of Alkenes with omine Electrophilic Addition of Alkenes with sulphuric acid 3 S 2 3 3 S 2 S 2 Electrophilic Addition of Alkenes with hydrogen
Mechanism Summary for Alevel AQA hemistry Electrophilic Addition of Alkenes with omine Electrophilic Addition of Alkenes with sulphuric acid 3 S 2 3 3 S 2 S 2 Electrophilic Addition of Alkenes with hydrogen
CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher. Quiz # 4. Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m.
 CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
CHEM 347 Quiz # 4 Spring 2014 Page 1 of 9 CHEM 347 Organic Chemistry II (for Majors) Instructor: Paul J. Bracher Quiz # 4 Due in Monsanto Hall 103 by: Friday, April 4 th, 2014, 7:00 p.m. Student Name (Printed)
Downloaded from
 Page 1 of 6 AMINES Amines are derivatives of ammonia (NH3), obtained by replacement of 1, 2 or all the 3 hydrogen atoms by alkyl and/or aryl groups. In nature amines are present in - proteins, vitamins,
Page 1 of 6 AMINES Amines are derivatives of ammonia (NH3), obtained by replacement of 1, 2 or all the 3 hydrogen atoms by alkyl and/or aryl groups. In nature amines are present in - proteins, vitamins,
Essential Organic Chemistry. Chapter 9
 Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Essential Organic Chemistry Paula Yurkanis Bruice Chapter 9 Substitution and Elimination Reactions of Alkyl Halides 9.1 How Alkyl Halides React Substitution Reactions One group takes the place of another.
Subject: Chains, Rings and Spectroscopy Code: Session: June Year: Final Mark Scheme
 Subject: hains, Rings and Spectroscopy ode: : e : Mark Scheme MAXIMUM MARK 90 ADVIE T EXAMINERS N TE ANNTATIN F SRIPTS 1. Please ensure that you use the final version of the Mark Scheme. You are advised
Subject: hains, Rings and Spectroscopy ode: : e : Mark Scheme MAXIMUM MARK 90 ADVIE T EXAMINERS N TE ANNTATIN F SRIPTS 1. Please ensure that you use the final version of the Mark Scheme. You are advised
Name/CG: 2012 Term 2 Organic Chemistry Revision (Session II) Deductive Question
 Name/G: 2012 Term 2 rganic hemistry Revision (Session II) Deductive Question 1(a) A yellow liquid A, 7 7 N 2, reacts with alkaline potassium manganate (VII) and on acidification gives a yellow solid B,
Name/G: 2012 Term 2 rganic hemistry Revision (Session II) Deductive Question 1(a) A yellow liquid A, 7 7 N 2, reacts with alkaline potassium manganate (VII) and on acidification gives a yellow solid B,
Unit 13-NITROGEN CONTAINING ORGANIC COMPOUNDS
 Unit 13-NITROGEN CONTAINING ORGANIC COMPOUNDS Two marks: 1. Name the product obtained when a nitrile is reduced by H 2 /Ni,. Give the equation. H 2 /Ni, Primary amine: RCN RCH 2 NH 2. 2. How is nitrobenzene
Unit 13-NITROGEN CONTAINING ORGANIC COMPOUNDS Two marks: 1. Name the product obtained when a nitrile is reduced by H 2 /Ni,. Give the equation. H 2 /Ni, Primary amine: RCN RCH 2 NH 2. 2. How is nitrobenzene
Figure (1) Name a laboratory technique that could be used to separate isooctane from a mixture of octane and isooctane.
 Octane and isooctane are structural isomers with the molecular formula C 8 H 8. The displayed formulas and boiling points of octane and isooctane are shown in Figure. Figure (a) Give the IUPAC name for
Octane and isooctane are structural isomers with the molecular formula C 8 H 8. The displayed formulas and boiling points of octane and isooctane are shown in Figure. Figure (a) Give the IUPAC name for
4.4, 4.5 HW MS 1. (a) nucleophilic addition 1 M2. (b) (i) 2-hydroxybutanenitrile 1 (ii) 2 H 3C O H
 4.4, 4.5 W MS 1. (a) nucleophilic addition 1 M N N M1 M M4 4 (b) (i) -hydroxybutanenitrile 1 (ii) N (allow 1 for amide even if not 4 7 N, i.e. RN ) (if not amide, allow one for any isomer of 4 7 N which
4.4, 4.5 W MS 1. (a) nucleophilic addition 1 M N N M1 M M4 4 (b) (i) -hydroxybutanenitrile 1 (ii) N (allow 1 for amide even if not 4 7 N, i.e. RN ) (if not amide, allow one for any isomer of 4 7 N which
Chemistry of Benzene: Electrophilic Aromatic Substitution
 Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Chemistry of Benzene: Electrophilic Aromatic Substitution Why this Chapter? Continuation of coverage of aromatic compounds in preceding chapter focus shift to understanding reactions Examine relationship
Organic Chemistry Review: Topic 10 & Topic 20
 Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Mark Scheme. Final. Chemistry CHEM4. (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry
 Version General Certificate of Education (A-level) June 20 Chemistry CEM4 (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry Final Mark Scheme Mark schemes are prepared by the Principal
Version General Certificate of Education (A-level) June 20 Chemistry CEM4 (Specification 2420) Unit 4: Kinetics, Equilibria and Organic Chemistry Final Mark Scheme Mark schemes are prepared by the Principal
Acceptable Answers Reject Mark. Acceptable Answers Reject Mark. ALLOW Iron/Fe or Zn/Zinc for tin Conc for concentrated. Acceptable Answers Reject Mark
 (a)(i) (a)(ii) Concentrated nitric acid AND concentrated sulfuric acid concentrated nitric and sulfuric acids Concentrated HNO and concentrated H SO 4 Extra reagents To prevent multiple substitutions/
(a)(i) (a)(ii) Concentrated nitric acid AND concentrated sulfuric acid concentrated nitric and sulfuric acids Concentrated HNO and concentrated H SO 4 Extra reagents To prevent multiple substitutions/
TOPIC 25 ANSWERS & MARK SCHEMES QUESTIONSHEET 1 SINGLE STAGE ALIPHATIC SYNTHESES NOT INVOLVING NITROGEN COMPOUNDS SO 4 / H 3 PO 4
 QUESTIONSHEET 1 SINGLE STAGE ALIPHATIC SYNTHESES NOT INVOLVING NITROGEN COMPOUNDS a) (i) Reagent Br 2 Conditions uv light / heat > 400 0 C (ii) Reagent HBr Conditions Gas (allow Concentrated HBr(aq) (½)
QUESTIONSHEET 1 SINGLE STAGE ALIPHATIC SYNTHESES NOT INVOLVING NITROGEN COMPOUNDS a) (i) Reagent Br 2 Conditions uv light / heat > 400 0 C (ii) Reagent HBr Conditions Gas (allow Concentrated HBr(aq) (½)
Alkenes and Alcohols Answers
 Alkenes and Alcohols Answers Chemistry - AQA GCE Mark Scheme 200 June series Q Part Sub Part Marking Guidance Mark Comments 8 a Three conditions in any order for M to M3 4 Mark independently M M2 M3 yeast
Alkenes and Alcohols Answers Chemistry - AQA GCE Mark Scheme 200 June series Q Part Sub Part Marking Guidance Mark Comments 8 a Three conditions in any order for M to M3 4 Mark independently M M2 M3 yeast
Chemistry 2.5 AS WORKBOOK. Working to Excellence Working to Excellence
 Chemistry 2.5 AS 91165 Demonstrate understanding of the properties of selected organic compounds WORKBOOK Working to Excellence Working to Excellence CONTENTS 1. Writing Excellence answers to Cis-Trans
Chemistry 2.5 AS 91165 Demonstrate understanding of the properties of selected organic compounds WORKBOOK Working to Excellence Working to Excellence CONTENTS 1. Writing Excellence answers to Cis-Trans
... (1) Explain the origin of the interaction represented by the dotted lines in the figure above
 The following figure shows a simplified representation of the arrangement of some amino acids in a portion of a protein structure in the form of an α-helix. (a) Name the type of protein structure in the
The following figure shows a simplified representation of the arrangement of some amino acids in a portion of a protein structure in the form of an α-helix. (a) Name the type of protein structure in the
2. Examining the infrared spectrum of a compound allows us to:
 CHEM 204 2010 Ass. 1 Problem 1. The amount of energy in infrared light corresponds to: a. the amount of energy needed to promote one electron from a bonding to an antibonding molecular orbital b. the amount
CHEM 204 2010 Ass. 1 Problem 1. The amount of energy in infrared light corresponds to: a. the amount of energy needed to promote one electron from a bonding to an antibonding molecular orbital b. the amount
must show C=C Penalise sticks once per pair 1 H 10 Penalise sticks once per pair 1 or CH 3 C 2 Penalise sticks once per pair 1
 M. (a) A allow CH COCH B must show C=C Penalise sticks once per pair (b) C CH CH CH CH CH D NOT cyclopentane which is only C 5 H 0 Penalise sticks once per pair (c) E CH CH COOCH Allow C H 5 CO CH F CH
M. (a) A allow CH COCH B must show C=C Penalise sticks once per pair (b) C CH CH CH CH CH D NOT cyclopentane which is only C 5 H 0 Penalise sticks once per pair (c) E CH CH COOCH Allow C H 5 CO CH F CH
1 (a) (CH 3 CO) 2 O + CH 3 CH(OH)CH 3 CH 3 COOCH(CH 3 ) 2 + CH 3 COOH
 Question er Mark Guidance 1 (a) ( 3 ) 2 + 3 () 3 3 ( 3 ) 2 + 3 1st mark orrect structure of ester: 3 ( 3 ) 2 2nd mark Equation contains correct formulae for ( 3 ) 2, 3 () 3 AND 3 2 ALLW correct structural
Question er Mark Guidance 1 (a) ( 3 ) 2 + 3 () 3 3 ( 3 ) 2 + 3 1st mark orrect structure of ester: 3 ( 3 ) 2 2nd mark Equation contains correct formulae for ( 3 ) 2, 3 () 3 AND 3 2 ALLW correct structural
2.9 ALKENES EXTRA QUESTIONS MS. (b) Addition 1 (ignore self or chain as a preface to addition ) (penalise additional)
 2.9 ALKENES EXTRA QUESTIONS MS 1. (a) 2 6 2 4 2 1 (b) Addition 1 (ignore self or chain as a preface to addition ) (penalise additional) 2. Electrophilic addition 1 M1: curly arrow from = bond towards/alongside
2.9 ALKENES EXTRA QUESTIONS MS 1. (a) 2 6 2 4 2 1 (b) Addition 1 (ignore self or chain as a preface to addition ) (penalise additional) 2. Electrophilic addition 1 M1: curly arrow from = bond towards/alongside
M1.(a) (i) 2Cl Cl 2 + 2e Ignore state symbols Credit loss of electrons from LHS Credit multiples Do not penalise absence of charge on electron 1
 M.(a) (i) 2Cl Cl 2 + 2e Credit loss of electrons from LHS Credit multiples Do not penalise absence of charge on electron (ii) (iii) +7 7 VII +VII Allow Mn +7 and 7+ MnO 4 + 8H + + 5e Mn 2+ + 4H 2 O Credit
M.(a) (i) 2Cl Cl 2 + 2e Credit loss of electrons from LHS Credit multiples Do not penalise absence of charge on electron (ii) (iii) +7 7 VII +VII Allow Mn +7 and 7+ MnO 4 + 8H + + 5e Mn 2+ + 4H 2 O Credit
Organic Chemistry, 7 L. G. Wade, Jr. Chapter , Prentice Hall
 Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
Organic Chemistry, 7 th Edition L. G. Wade, Jr. Chapter 17 Reactions of Aromatic Compounds 2010, Prentice Hall Electrophilic Aromatic Substitution Although h benzene s pi electrons are in a stable aromatic
DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE
 DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 6 Dr Ali El-Agamey 1 Oxidation States Easy for inorganic salts: CrO 4 2- reduced to Cr 2 O 3. KMnO 4 reduced to MnO 2. Oxidation: Gain of O,
DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 6 Dr Ali El-Agamey 1 Oxidation States Easy for inorganic salts: CrO 4 2- reduced to Cr 2 O 3. KMnO 4 reduced to MnO 2. Oxidation: Gain of O,
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Advanced Subsidiary Level and Advanced Level
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Advanced Subsidiary Level and Advanced Level * 7524377035* CHEMISTRY 9701/23 Paper 2 Structured Questions AS Core May/June
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Advanced Subsidiary Level and Advanced Level * 7524377035* CHEMISTRY 9701/23 Paper 2 Structured Questions AS Core May/June
A mass spectrometer can be used to distinguish between samples of butane and propanal. The table shows some precise relative atomic mass values.
 Butane and propanal are compounds with M r = 58.0, calculated using data from your Periodic Table. (a) A mass spectrometer can be used to distinguish between samples of butane and propanal. The table shows
Butane and propanal are compounds with M r = 58.0, calculated using data from your Periodic Table. (a) A mass spectrometer can be used to distinguish between samples of butane and propanal. The table shows
MARK SCHEME for the October/November 2015 series 9701 CHEMISTRY
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International Advanced Level MARK SCHEME for the October/November 015 series 9701 CHEMISTRY 9701/4 Paper 4 (A Structured Questions), maximum raw mark 100
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International Advanced Level MARK SCHEME for the October/November 015 series 9701 CHEMISTRY 9701/4 Paper 4 (A Structured Questions), maximum raw mark 100
But in organic terms: Oxidation: loss of H 2 ; addition of O or O 2 ; addition of X 2 (halogens).
 Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Unit 2. Answers to examination-style questions. Answers Marks Examiner s tips. 1 (a) heat energy change at constant pressure
 (a) heat energy change at constant pressure This is in the spec but not so well known. Learn it. (b) N 2 (g) + ½O 2 (g) N 2 O(g) (c) (i) D = (bonds broken) (bonds made) = ½(945) + (3/2)(59) 3(278) = 23
(a) heat energy change at constant pressure This is in the spec but not so well known. Learn it. (b) N 2 (g) + ½O 2 (g) N 2 O(g) (c) (i) D = (bonds broken) (bonds made) = ½(945) + (3/2)(59) 3(278) = 23
(nucleophilic) addition-elimination Not electrophilic addition-elimination 1. The minimum quantity of hot water was used:
 M.(a) (nucleophilic) addition-elimination Not electrophilic addition-elimination Allow C 6H 5 or benzene ring Allow attack by :NH 2C 6H 5 M2 not allowed independent of M, but allow M for correct attack
M.(a) (nucleophilic) addition-elimination Not electrophilic addition-elimination Allow C 6H 5 or benzene ring Allow attack by :NH 2C 6H 5 M2 not allowed independent of M, but allow M for correct attack
Downloaded from
 1 Class XII Chemistry Chapter: Alcohols, Phenols And Ethers Top concepts: 1. Structure of alcohols, phenols and ethers: 2. Preparation of alcohols: 3. Preparation of phenols: 2 4. Physical properties of
1 Class XII Chemistry Chapter: Alcohols, Phenols And Ethers Top concepts: 1. Structure of alcohols, phenols and ethers: 2. Preparation of alcohols: 3. Preparation of phenols: 2 4. Physical properties of
Answer Marks Guidance
 Question number (a) molecular formula: C 4 H 8 Answer Marks Guidance empirical formula: CH 2 This is a revision of earlier chapters. (b) (i) name of mechanism: electrophilic addition Remember that reactions
Question number (a) molecular formula: C 4 H 8 Answer Marks Guidance empirical formula: CH 2 This is a revision of earlier chapters. (b) (i) name of mechanism: electrophilic addition Remember that reactions
ammonia primary amine secondary amine tertiary amine quaternary ammonium salt (NOT amines) H CH 3 CH 3 CH 2
 AMIES 1) TYPES F AMIES Amines can be classed as primary, secondary and tertiary. Amines are related to ammonia ( 3 ). Aromatic amines have the joined directly to the benzene ring. Quaternary ammonium salts
AMIES 1) TYPES F AMIES Amines can be classed as primary, secondary and tertiary. Amines are related to ammonia ( 3 ). Aromatic amines have the joined directly to the benzene ring. Quaternary ammonium salts
Q.1 Draw structures for all amines of molecular formula C 4 H 11 N. Classify them as primary, secondary or tertiary amines.
 1 AMIES Structure ontain the 2 group. lassification primary (1 ) amines secondary (2 ) amines tertiary (3 ) amines quarternary (4 ) ammonium salts + 1 2 3 4 Aliphatic Aromatic methylamine, ethylamine,
1 AMIES Structure ontain the 2 group. lassification primary (1 ) amines secondary (2 ) amines tertiary (3 ) amines quarternary (4 ) ammonium salts + 1 2 3 4 Aliphatic Aromatic methylamine, ethylamine,
Amines Reading Study Problems Key Concepts and Skills Lecture Topics: Amines: structure and nomenclature
 Amines Reading: Wade chapter 19, sections 19-1-19-19 Study Problems: 19-37, 19-39, 19-40, 19-41, 19-44, 19-46, 19-47, 19-48, 19-51, 19-54 Key Concepts and Skills: Explain how the basicity of amines varies
Amines Reading: Wade chapter 19, sections 19-1-19-19 Study Problems: 19-37, 19-39, 19-40, 19-41, 19-44, 19-46, 19-47, 19-48, 19-51, 19-54 Key Concepts and Skills: Explain how the basicity of amines varies
2A - Amines. 2 H atoms replaced: 2 attached C's to N. 3 H atom replaced: 3 attached C's to N Ammonia, NH3 Primary amine Secondary amine Tertiary amine
 2A - Amines Something fishy about amines: Have an NH 2, amine group. Amines are derivatives of ammonia: 3 H atoms 1 H atom replaced: 1 attached C to N 2 H atoms replaced: 2 attached C's to N 3 H atom replaced:
2A - Amines Something fishy about amines: Have an NH 2, amine group. Amines are derivatives of ammonia: 3 H atoms 1 H atom replaced: 1 attached C to N 2 H atoms replaced: 2 attached C's to N 3 H atom replaced:
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
334/01 CHEMISTRY CH4. A.M. MONDAY, 23 January (1 hour 40 minutes)
 Candidate Name Centre Number Candidate Number WELSH JOINT EDUCATION COMMITTEE General Certificate of Education Advanced CYD-BWYLLGOR ADDYSG CYMRU Tystysgrif Addysg Gyffredinol Uwch 334/01 CHEMISTRY CH4
Candidate Name Centre Number Candidate Number WELSH JOINT EDUCATION COMMITTEE General Certificate of Education Advanced CYD-BWYLLGOR ADDYSG CYMRU Tystysgrif Addysg Gyffredinol Uwch 334/01 CHEMISTRY CH4
4.3 ANSWERS TO EXAM QUESTIONS
 4. ANSWERS TO EXAM QUESTIONS. (a) (i) A proton donor () (ii) Fully ionised or fully dissociated () (iii) 0 0 4 () mol dm 6 () 4 (b) (i) 50 0 /5 000 () = 0 06 mol dm () () (ii) Mol OH added = 50 0 50/000
4. ANSWERS TO EXAM QUESTIONS. (a) (i) A proton donor () (ii) Fully ionised or fully dissociated () (iii) 0 0 4 () mol dm 6 () 4 (b) (i) 50 0 /5 000 () = 0 06 mol dm () () (ii) Mol OH added = 50 0 50/000
Acceptable Answers Reject Mark. The sulfur trioxide can accept a pair of electrons An electron 1
 (a)(i sulfuric acid / fuming H SO 4 / oleum / H S O 7 Conc. (for fuming) Fuming dilute sulfuric acid Just sulfuric acid Just H SO 4 (a)(ii) Sulfur is δ+ and on at least one oxygen δ- Full + or charge(s)
(a)(i sulfuric acid / fuming H SO 4 / oleum / H S O 7 Conc. (for fuming) Fuming dilute sulfuric acid Just sulfuric acid Just H SO 4 (a)(ii) Sulfur is δ+ and on at least one oxygen δ- Full + or charge(s)
AMINES. 3. Secondary When two hydrogen atoms are replaced by two alkyl or aryl groups.
 AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
8 (c) (i) H 2 /Ni or H 2 /Pt or Sn/HCl or Fe/HCl (conc or dil or neither) allow dil H 2 SO 4 ignore mention of NaOH
 Polymers Answers EM4 - AQA GE hemistry Mark Scheme 200 January series 8 (c) (i) 2 /i or 2 /Pt or Sn/l or e/l (conc or dil or neither) allow dil 2 S 4 ignore mention of a ot ab 4 ot LiAl 4 ot a/ 2 5 not
Polymers Answers EM4 - AQA GE hemistry Mark Scheme 200 January series 8 (c) (i) 2 /i or 2 /Pt or Sn/l or e/l (conc or dil or neither) allow dil 2 S 4 ignore mention of a ot ab 4 ot LiAl 4 ot a/ 2 5 not
Organic Chemistry. REACTIONS Grade 12 Physical Science Mrs KL Faling
 Organic Chemistry REACTIONS Grade 12 Physical Science Mrs KL Faling SUBSTITUTION REACTIONS This is a reaction where an atom or group of atoms is replaced by another atom or group of atoms Substitution
Organic Chemistry REACTIONS Grade 12 Physical Science Mrs KL Faling SUBSTITUTION REACTIONS This is a reaction where an atom or group of atoms is replaced by another atom or group of atoms Substitution
3.10 Benzene : Aromatic Hydrocarbons / Arenes
 3.10 Benzene : Aromatic ydrocarbons / Arenes There are two major classes of organic chemicals aliphatic : straight or branched chain organic substances aromatic or arene: includes one or more ring of six
3.10 Benzene : Aromatic ydrocarbons / Arenes There are two major classes of organic chemicals aliphatic : straight or branched chain organic substances aromatic or arene: includes one or more ring of six
MARK SCHEME for the October/November 2015 series 9701 CHEMISTRY
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International Advanced Level MARK SCHEME for the October/November 05 series 970 CHEMISTRY 970/43 Paper 4 (A Structured Questions), maximum raw mark 00 This
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International Advanced Level MARK SCHEME for the October/November 05 series 970 CHEMISTRY 970/43 Paper 4 (A Structured Questions), maximum raw mark 00 This
Chapter 10: Carboxylic Acids and Their Derivatives
 Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chapter 10: Carboxylic Acids and Their Derivatives The back of the white willow tree (Salix alba) is a source of salicylic acid which is used to make aspirin (acetylsalicylic acid) The functional group
Chemistry Questions ans Answers BASED ON HIGH ORDER THINKING SKILL (HOTS) UNIT- 13 ORGANIC COMPOUNDS CONTAINING NITROGEN
 Chemistry Questions ans Answers BASED N HIGH RDER THINKING SKILL (HTS) UNIT- 13 RGANIC CMPUNDS CNTAINING NITRGEN 1 MARK QUESTINS Q. 1. Why the presence of a base is essential in the ammonolysis of alkyl
Chemistry Questions ans Answers BASED N HIGH RDER THINKING SKILL (HTS) UNIT- 13 RGANIC CMPUNDS CNTAINING NITRGEN 1 MARK QUESTINS Q. 1. Why the presence of a base is essential in the ammonolysis of alkyl
ROADMAP FOR REACTIONS Chapter 6
 RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
Question Answer Marks Guidance 1 (a) M1 EITHER in words: (pyruvic acid forms) hydrogen bonds with water
 1 (a) M1 EITER in words: (pyruvic acid forms) hydrogen bonds with water 2 R correctly labelled diagram showing hydrogen bond between pyruvic acid and water FR M1 only: if use diagram ALLW a labelled hydrogen
1 (a) M1 EITER in words: (pyruvic acid forms) hydrogen bonds with water 2 R correctly labelled diagram showing hydrogen bond between pyruvic acid and water FR M1 only: if use diagram ALLW a labelled hydrogen
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry
 UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
Ch 16 Electrophilic Aromatic Substitution
 Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
Ch 16 Electrophilic Aromatic Substitution Mechanism - Aromatic rings typically undergo substitution, where an H is replaced with an electrophile (E+). - The rings do not typically undergo addition across
Page 2. Q1.Consider the following scheme of reactions. (a) Give the IUPAC name for compound P and that for compound Q. P... Q...
 Q1.Consider the following scheme of reactions. (a) Give the IUPAC name for compound P and that for compound Q. P... Q... (2) (b) The conversion of P into Q in Reaction 1 uses HCl Name and outline a mechanism
Q1.Consider the following scheme of reactions. (a) Give the IUPAC name for compound P and that for compound Q. P... Q... (2) (b) The conversion of P into Q in Reaction 1 uses HCl Name and outline a mechanism
