CHM 233 : Fall 2018 Quiz #10 - Answer Key
|
|
|
- Fay Bishop
- 5 years ago
- Views:
Transcription
1 M 233 : Fall 2018 Quiz #10 - nswer Key Question 1 M20a Which of the following is The strongest Bronsted cid? R- 2 is a carboxylic acid and R-S 3 is a solfonic acid, you will want to draw these two as Lewis structures and also draw the conjugate base anions to answer this question F 3 S S 3 3 S 3 B D The strongest acid has the most stable conjugate base anion 3 more acidic, lower pka 3 more stable less acidic, higher pka B 3 3 B less stable Bronsted acidity is controlled by the following factors: 1. Electronegativity of the atom with the negative charge. 2. The size of the atom bearing the negative charge. 3. Resonance. 4. Substituent effects. 2 3 carboxylic acid 3 S S S S sulfonic acid Sulfonic acids are generally stronger than carboxylic acids since the conjugate base is stabilized by 3 resonance structures compared to 2 for the carboxylic acids, therefore the answer is not B. Increasing Stability F 3 electronegative group S > S > 3 resonance donating group The remaining difference is the substituents. The electron withdrawing F 3 group (3 electronegative F's) stabilizes the negative charge and thus the anion. Later we will see that a resonance electron donating group such as -3 destabilizes the negative charge and thus the anion. S
2 acetone pk a = 19 Question 2 M20c Which of the following statements about the equilibrium below is true? Stronger Stronger Weaker Weaker cid Base Base cid allyl anion acetone enolate propene pk a = 43 I the equilibrium will lie on the left II the equilibrium will lie on the right III the allyl anion is the strongest base in the equilibrium IV propene is the weakest acid in the equilibrium I and IV only II, III and IV only B II and IV only D I, III and IV only acetone and propene are the acids on each side of the equilibrium, the two anions are the bases. The strongest acid is the one with the lower pka the stronger acid has the weaker conjugate base and vica versa, thus the allyl anion is the strongest base. The strongest acid and the strongest base "push" the equilibrium to the RIGT because they react faster than the weaker acid and base react in the reverse direction both of these answers are easy to obtain since the pka's are given, the smaller pka corresponds to the stronger acid you could also have worked this out based on electron energies. cetone is the stronger acid because it "makes" the more stable conjugate base, acetone enolate. omparing resonance structures of the two anions, the negative charge is "shared" by two carbons in the allyl anion, but by a carbon and a more electronegative oxygen in the enolate, the electrons are lower in energy in the enolate, the enolate is less reactive, is a weaker base. acetone enolate allyl anion
3 Question 3 M20d Which of the following is the product of the acid/base reaction between aluminum trichloride (ll 3 ) and acetone ( 3 3 )? l l l l ll 3 ll 3 ll 3 ll 3 B D although ll3 has lots of non-bonding electrons, they are on the electronegative chlorines and are thus not very high in energy, not very reactive. the nonbonding electrons on acetone are also on an electronegative atom, oxygen, and whether acetone reacts as a base or an acid depends upon what it reacts with L l l l LB l in this case the key point is that aluminum has only 3 valence electrons, thus with 3 bonds to chlorine it can easily accept a pair of electrons from oxygen, thus forming a bond without having to break a bond, this is an ideal Lewis acid/base reaction. note that if the pi-electrons in the acetone had ben used, a bond would have been broken in the reaction, thus D is not a good answer the oxygen will develop a formal positive charge since is loses a half share of the the non-bonding electrons, and the aluminum gains a formal negative charge since it gains a half share of the non-bonding pair the electrons in the new bond are considerably lowered in energy, we expect this Lewis acid/base reaction to be exothermic l l l l
4 Question 4 M20e Give the major product of the following Lewis cid/base reaction. ote that the non-bonding electrons are T shown on the fluorines in either the reactants or products (hint, you will need to draw a minor resonance contributor of the ester to determine the most reactive pair of nonbonding electrons in the ester) BF 3 LB BF 3 L BF 3 BF 3 B D BF 3 B and D are impossible, boron does not have a pair of electrons that it can use to form a new bond To decide between and, consider the following minor resonance contributor R, consider the following bond dipole moments is bigger than because of the pi electrons in the double bond Either way it is clear that there is a larger partial negative charge on the carbonyl (=) oxygen, thus the electron energy will be higher on this oxygen, hence these electrons are more reactive, these represent the Lewis base site.
5 Question 5 M20i Which of the following reactions is most likely to occur? makes a σ-bond, breaks a π-bond and puts negative charge on oxygen, this is the best. B gives electrons to hydride, nowhere for them to go, very bad reaction! makes a σ-bond, breaks a π-bond and puts negative charge on carbon, which is not as good as. D gives electrons to hydride, nowhere for them to go, very bad reaction! B D
6 Question 6 M20k Which is the most acidic proton in this molecule, a, b, c or d (not all of the - bonds are shown in this molecule)? hint, draw all of the conjugate base anions and decide which anion has the lowest energy electrons) a b c d a B b c D d a b c d b c d b c d 2 resonance contributors to the anion structure a b c d a c d X no other resonance contributors to the anion structure a b c d a b d X no other resonance contributors to the anion structure a a a a b b b b c d c c c TREE resonance contributors for the anion structure, most delocalized lowest energy nonbonding electrons, most stable conjugate base anion, most acidic proton
7 Question 7 M20l Which is the weakest base? (hint, look for minor resonance contributors) B D X no minor resonance contributors to the structure, non-bonding electrons are not particularly stabilized or low in energy B X no minor resonance contributors to the structure, non-bonding electrons are not particularly stabilized or low in energy minor resonance contributor to the structure shows how the nonbonding electrons are somewhat stabilized by delocalization, and this lower in energy than in or B D two minor resonance contributors to the structure shows how the non-bonding electrons are even more stabilized by delocalization than in, these non-bonding electrons are thus lowest in energy of all, are thus least reactive, D is thus the weakest base
8 Question 8 M20b What are the most likely products of the following Bronsted cid/base reaction? 3 2? I II 3 3 III 3 IV 2 2 V = I & IV B = I & III = II & IV D = II & V highest energy, most reactive electrons are non-bonding on nitrogen, the amine will be the Lewis base L/B LB/BB making a bond to the hydrogen requires breaking this - bond, which generates a resonance stabilized anion on oxygen, this is the bst reaction for the Lewis acid since these are the lowest electron energy products that can be obtained from the Lewis acid also, it isn't cheating just to know that a carboxylic acid will donate the proton on the oxygen when reacting with a base!!! in addition to being a Lewis acid base reaction the base provides the electrons to form a new bond to one of the atoms on the acid, LB and L), this is also a Bronsted acid base reaction, since overall a proton is transferred from teh acid to the base, thus the reaction is labelled as L/B and LB/BB.
CHEM 109A Organic Chemistry
 CHEM 109A Organic Chemistry https://labs.chem.ucsb.edu/zakarian/armen/courses.html Chapter 2 Acids and Bases Central to Understanding Organic Chemistry Draw the conjugate acid of each of the following:
CHEM 109A Organic Chemistry https://labs.chem.ucsb.edu/zakarian/armen/courses.html Chapter 2 Acids and Bases Central to Understanding Organic Chemistry Draw the conjugate acid of each of the following:
Chapter 2: Acids and Bases
 hapter 2: Acids and Bases 32 hapter 2: Acids and Bases Problems 2.1 Write each acid- reaction as a proton-transfer reaction. Label which reactant is the acid and which the, as well as which product is
hapter 2: Acids and Bases 32 hapter 2: Acids and Bases Problems 2.1 Write each acid- reaction as a proton-transfer reaction. Label which reactant is the acid and which the, as well as which product is
Ch 2 Polar Covalent Bonds
 h 2 Polar ovalent Bonds Two primary bond types: ovalent (shared e -1 s) and Ionic (transferred e -1 s) Ionic bonds can have covalent character, such as with Na:l. An e -1 pair on l -1 can fill the 3s orbital
h 2 Polar ovalent Bonds Two primary bond types: ovalent (shared e -1 s) and Ionic (transferred e -1 s) Ionic bonds can have covalent character, such as with Na:l. An e -1 pair on l -1 can fill the 3s orbital
Acid/Base stuff Beauchamp 1
 cid/base stuff Beauchamp 1 Problems You should be able to match a pk a value with its acid in each group below and explain the differences. You should be able to draw an arrow-pushing mechanism with general
cid/base stuff Beauchamp 1 Problems You should be able to match a pk a value with its acid in each group below and explain the differences. You should be able to draw an arrow-pushing mechanism with general
Chapter 1 Introduction and Review
 Chapter 1 Introduction and Review Concept to review: It is your responsibility to review the following concepts before the first class to ensure success in understanding new concepts: Atomic structure
Chapter 1 Introduction and Review Concept to review: It is your responsibility to review the following concepts before the first class to ensure success in understanding new concepts: Atomic structure
HOMEWORK PROBLEMS: POLAR BONDS, RESONANCE, ACIDS & BASES 1. Which of the following molecules is the most polar?
 CEM 31 MEWRK PRBLEMS: PLAR BDS, RESACE, ACIDS & BASES 1. Which of the following molecules is the most polar? 2. Trans-dichlorodifluoroethylene, C 2 Cl 2 2, has a number of polar bonds but no net dipole
CEM 31 MEWRK PRBLEMS: PLAR BDS, RESACE, ACIDS & BASES 1. Which of the following molecules is the most polar? 2. Trans-dichlorodifluoroethylene, C 2 Cl 2 2, has a number of polar bonds but no net dipole
Covalent bonds can have ionic character These are polar covalent bonds
 Polar Covalent Bonds: Electronegativity Covalent bonds can have ionic character These are polar covalent bonds Bonding electrons attracted more strongly by one atom than by the other Electron distribution
Polar Covalent Bonds: Electronegativity Covalent bonds can have ionic character These are polar covalent bonds Bonding electrons attracted more strongly by one atom than by the other Electron distribution
2. Polar Covalent Bonds: Acids and Bases
 2. Polar Covalent Bonds: Acids and Bases Based on McMurry s Organic Chemistry, 6 th edition, Chapter 2 2003 Ronald Kluger Department of Chemistry University of Toronto 2.1 Polar Covalent Bonds: Electronegativity!
2. Polar Covalent Bonds: Acids and Bases Based on McMurry s Organic Chemistry, 6 th edition, Chapter 2 2003 Ronald Kluger Department of Chemistry University of Toronto 2.1 Polar Covalent Bonds: Electronegativity!
6.2 Electron Movements in Brønsted Acid Base Reactions. Copyright 2018 by Nelson Education Limited 1
 6.2 Electron Movements in Brønsted Acid Base Reactions Copyright 2018 by Nelson Education Limited 1 Recall: Brønsted Acid Base Reactions often simply termed acid base reactions Recall: H + does not actually
6.2 Electron Movements in Brønsted Acid Base Reactions Copyright 2018 by Nelson Education Limited 1 Recall: Brønsted Acid Base Reactions often simply termed acid base reactions Recall: H + does not actually
Chemistry 250A -- Exam #1 Answer Key -- September 23, 2008 (There are 5 pages.)
 ame: 1 hemistry 250 -- Exam #1 nswer Key -- September 23, 2008 (There are 5 pages.) 1. (15 pts) Show the major product(s) from the following acid-base reactions. If there is no reaction then say o Reaction.
ame: 1 hemistry 250 -- Exam #1 nswer Key -- September 23, 2008 (There are 5 pages.) 1. (15 pts) Show the major product(s) from the following acid-base reactions. If there is no reaction then say o Reaction.
Chapter Bonding. Atoms trying to attain the stable configuration of a noble (inert) gas - often referred to as the octet rule
 Chapter 1 1.2-1.3 Bonding Atoms trying to attain the stable configuration of a noble (inert) gas - often referred to as the octet rule 1.2 Ionic Bonding - Electrons Transferred 1.3 Covalent Bonding - Electrons
Chapter 1 1.2-1.3 Bonding Atoms trying to attain the stable configuration of a noble (inert) gas - often referred to as the octet rule 1.2 Ionic Bonding - Electrons Transferred 1.3 Covalent Bonding - Electrons
Ch.2 Polar Bonds and Their Consequences. 2.1 Polar Covalent Bonds and Electronegativity. polar covalent bonds: electron distribution is unsymmetrical
 2.1 Polar ovalent Bonds and Electronegativity polar covalent bonds: electron distribution is unsymmetrical Ionic haracter δ+ δ- + - X Y X Y X Y symmetrical covalent bond polar covalent bond ionic bond
2.1 Polar ovalent Bonds and Electronegativity polar covalent bonds: electron distribution is unsymmetrical Ionic haracter δ+ δ- + - X Y X Y X Y symmetrical covalent bond polar covalent bond ionic bond
Chapter 2 Acids and Bases. Arrhenius Acid and Base Theory. Brønsted-Lowry Acid and Base Theory
 hapter 2 Acids and Bases A significant amount of chemistry can be described using different theories of acids and bases. We ll consider three different acid-base theories (listed in order of increasing
hapter 2 Acids and Bases A significant amount of chemistry can be described using different theories of acids and bases. We ll consider three different acid-base theories (listed in order of increasing
Chapter 2 Polar Covalent Bonds; Acids and Bases SAMPLE. Chapter Outline
 Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
2. Acids and Bases (text )
 2009, Department of hemistry, The University of Western ntario 2.1 2. Acids and Bases (text 2.1 2.6) Acid-base reactions are one of the most important reaction types in organic chemistry and biology, e.g.:
2009, Department of hemistry, The University of Western ntario 2.1 2. Acids and Bases (text 2.1 2.6) Acid-base reactions are one of the most important reaction types in organic chemistry and biology, e.g.:
Chemistry 304B, Spring 99 Lecture 2 1
 Chemistry 04B, Spring 99 Lecture 2 1 Consider Acidity and Basicity Recall carboxylic acids and amines (base). Jones: p 27-8; 149, 291, 822-826, 1086-1090, 958-96, 878-882 General terms: Lewis acid: capable
Chemistry 04B, Spring 99 Lecture 2 1 Consider Acidity and Basicity Recall carboxylic acids and amines (base). Jones: p 27-8; 149, 291, 822-826, 1086-1090, 958-96, 878-882 General terms: Lewis acid: capable
Chapter 2: Acids and Bases
 1. Which of the following statements is a correct definition for a Brønsted-Lowry acid? A) Proton acceptor C) Electron pair acceptor B) Electron pair donor D) Proton donor 2. Which of the following statements
1. Which of the following statements is a correct definition for a Brønsted-Lowry acid? A) Proton acceptor C) Electron pair acceptor B) Electron pair donor D) Proton donor 2. Which of the following statements
Chapter 2 Polar Covalent Bonds; Acids and Bases. Chapter Outline
 rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
Organic Chem Chapter 3: Acids and Bases
 Organic Chem Chapter 3: Acids and Bases Title and Highlight Right side: NOTES! Topic: EQ: Date NOTES: Write out the notes from my website. Use different types of note-taking methods to help you recall
Organic Chem Chapter 3: Acids and Bases Title and Highlight Right side: NOTES! Topic: EQ: Date NOTES: Write out the notes from my website. Use different types of note-taking methods to help you recall
Acids and Bases: Molecular Structure and Acidity
 Tutorial Contents A. Introduction B. Resonance C. Atomic Radius D. Electronegativity E. Inductive Effect F. Exercises G. Exercise Solutions Acids and Bases: Molecular Structure and Acidity Review the Acids
Tutorial Contents A. Introduction B. Resonance C. Atomic Radius D. Electronegativity E. Inductive Effect F. Exercises G. Exercise Solutions Acids and Bases: Molecular Structure and Acidity Review the Acids
Chapter 01 Structure Determines Properties part 2
 Chapter 01 Structure Determines Properties part 2 CEM 341: Spring 2012 Prof. Greg Cook Resonance Section 1.8 Resonance Some molecules may have more than one correct Lewis structure These are NT isomers.
Chapter 01 Structure Determines Properties part 2 CEM 341: Spring 2012 Prof. Greg Cook Resonance Section 1.8 Resonance Some molecules may have more than one correct Lewis structure These are NT isomers.
Chapter 3 An Introduction to Organic Reactions: Acids and Bases
 There are 4 types of Organic Reactions Chapter 3 An Introduction to Organic Reactions: SUBSTITUTION: ADDITION: X Y + A X A + Y Example Example A B + X Y A B X Y ELIMINATION There are 4 Types of Organic
There are 4 types of Organic Reactions Chapter 3 An Introduction to Organic Reactions: SUBSTITUTION: ADDITION: X Y + A X A + Y Example Example A B + X Y A B X Y ELIMINATION There are 4 Types of Organic
Acids and Bases. Acids and Bases
 BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
BrØnsted-Lowry A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton BrØnsted-Lowry Some molecules contain both hydrogen atoms and lone pairs and thus, can act
ORGANIC CHEMISTRY. Meaning of Organic?
 ORGANIC CHEMISTRY Meaning of Organic? Initially scientists believed there was a special force in living organisms -this was assumed the unique component of organic material In 1828 Wöhler synthesized urea
ORGANIC CHEMISTRY Meaning of Organic? Initially scientists believed there was a special force in living organisms -this was assumed the unique component of organic material In 1828 Wöhler synthesized urea
O Enolate. = Electrophile/ Lewis acid. = Electrophile/ Lewis acid O B. Enolate. = Electrophile/ Lewis acid. Enolate. O O Enolate
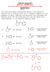 CM 234, Spring 2017 QUIZ #12 ANSWER KEY (hit the RETURN Button to return to the Main Quiz Page) QUESTIN 1 MC34r The following Aldol condensation product was formed by reaction of an enolate anion and a
CM 234, Spring 2017 QUIZ #12 ANSWER KEY (hit the RETURN Button to return to the Main Quiz Page) QUESTIN 1 MC34r The following Aldol condensation product was formed by reaction of an enolate anion and a
Carbonyl Chemistry: Fundamentals
 Carbonyl Chemistry: Fundamentals What is a carbonyl group and what are its properties? A carbonyl is any compound that has a carbon double-bonded to an oxygen Carbonyls can behave as a nucleophile OR an
Carbonyl Chemistry: Fundamentals What is a carbonyl group and what are its properties? A carbonyl is any compound that has a carbon double-bonded to an oxygen Carbonyls can behave as a nucleophile OR an
Full file at
 Essential Organic Chemistry, 2e (Bruice) Chapter 2 Acids and Bases 1) Which of the following is not a conjugate acid-base pair? A) B) C) HSO- 4, H2SO4 D) -OH, O2- E) NO3 -, NO2-2) Which is defined as a
Essential Organic Chemistry, 2e (Bruice) Chapter 2 Acids and Bases 1) Which of the following is not a conjugate acid-base pair? A) B) C) HSO- 4, H2SO4 D) -OH, O2- E) NO3 -, NO2-2) Which is defined as a
If somehow it is possible to increase the stability of a given base, then it
 Text Related to Segment.04 00 laude E. Wintner If somehow it is possible to increase the stability of a given base, then it should be true that the corresponding acid will be stronger than would be so
Text Related to Segment.04 00 laude E. Wintner If somehow it is possible to increase the stability of a given base, then it should be true that the corresponding acid will be stronger than would be so
Acid Dissociation Constant
 CE 131 Lecture 37 Lewis Acids and Bases Chapter 16: pp. 800-802. Acid Dissociation Constant C 2 3 2 + 2 3 + + C 2 3-2 [ 3 + ][C 2 3-2 ] K = [ 2 ][C 2 3 2 ] [ 3 + ][C 2 3-2 ] K a = K [ 2 ] = [C 2 3 2 ]
CE 131 Lecture 37 Lewis Acids and Bases Chapter 16: pp. 800-802. Acid Dissociation Constant C 2 3 2 + 2 3 + + C 2 3-2 [ 3 + ][C 2 3-2 ] K = [ 2 ][C 2 3 2 ] [ 3 + ][C 2 3-2 ] K a = K [ 2 ] = [C 2 3 2 ]
Learning Guide for Chapter 7 - Organic Reactions I
 Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. cleophiles and Electrophiles V. Acids and Bases What a chemical
The energy associated with electrostatic interactions is governed by Coulomb s law:
 Chapter 8 Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other
Chapter 8 Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other
Two atoms share electrons to make a covalent bond. The differenceof the electronegativites of those two atoms determines how polar that bond is.
 Chapter 2: Polar covalent bonds; Acids and bases The reactivity of organic compounds is often defined by the polarities of the covalent bonds in the molecule. Polar covalent bonds: an intermediate between
Chapter 2: Polar covalent bonds; Acids and bases The reactivity of organic compounds is often defined by the polarities of the covalent bonds in the molecule. Polar covalent bonds: an intermediate between
1. methyl 2. methylene 3. methine 4. primary 5. secondary 6. tertiary 7. quarternary 8. isopropyl
 hem 201 Sample Midterm Beauchamp Exams are designed so that no one question will make or break you. The best strategy is to work steadily, starting with those problems you understand best. Make sure you
hem 201 Sample Midterm Beauchamp Exams are designed so that no one question will make or break you. The best strategy is to work steadily, starting with those problems you understand best. Make sure you
Chapter 8 Basic concepts of bonding
 Chapter 8 Basic concepts of bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule When atoms or ions are strongly attracted to one another, we say that there is a chemical bond between them. Types
Chapter 8 Basic concepts of bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule When atoms or ions are strongly attracted to one another, we say that there is a chemical bond between them. Types
Chemistry 2000 Lecture 19: Organic acids
 Chemistry 2000 Lecture 19: Organic acids Marc R. Roussel March 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 19: Organic acids March 8, 2018 1 / 22 Organic acids The acid dissociation constant, K a The
Chemistry 2000 Lecture 19: Organic acids Marc R. Roussel March 8, 2018 Marc R. Roussel Chemistry 2000 Lecture 19: Organic acids March 8, 2018 1 / 22 Organic acids The acid dissociation constant, K a The
Practice Hour Examination # 1-1
 CHEM 346 Organic Chemistry I Fall 2013 Practice Hour Examination # 1-1 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I (for Majors) Instructor: Paul J. Bracher Practice Hour Examination # 1-1 Monday,
CHEM 346 Organic Chemistry I Fall 2013 Practice Hour Examination # 1-1 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I (for Majors) Instructor: Paul J. Bracher Practice Hour Examination # 1-1 Monday,
Chapter 9 Bonding. Dr. Sapna Gupta
 Chapter 9 Bonding Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and right)
Chapter 9 Bonding Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and right)
Learning Guide for Chapter 7 - Organic Reactions I
 Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. Nucleophiles and Electrophiles V. Acids and Bases What a
Learning Guide for Chapter 7 - rganic Reactions I I. Introduction to Reactions II. Principles of Kinetics III. Principles of Thermodynamics IV. Nucleophiles and Electrophiles V. Acids and Bases What a
Organic Chemistry Peer Tutoring Department University of California, Irvine
 Organic Chemistry Peer Tutoring Department University of California, Irvine Arash Khangholi (akhangho@uci.edu) Cassandra Amezquita (camezqu1@uci.edu) Jiana Machhor (jmachhor@uci.edu) OCHEM 51A Professor
Organic Chemistry Peer Tutoring Department University of California, Irvine Arash Khangholi (akhangho@uci.edu) Cassandra Amezquita (camezqu1@uci.edu) Jiana Machhor (jmachhor@uci.edu) OCHEM 51A Professor
Chapter 8: Concepts of Chemical Bonding
 Chapter 8: Concepts of Chemical Bonding Learning Outcomes: Write Lewis symbols for atoms and ions. Define lattice energy and be able to arrange compounds in order of increasing lattice energy based on
Chapter 8: Concepts of Chemical Bonding Learning Outcomes: Write Lewis symbols for atoms and ions. Define lattice energy and be able to arrange compounds in order of increasing lattice energy based on
Organic Chemistry 6 th Edition Paula Yurkanis Bruice. Chapter 1. Electronic Structure and Bonding. Acids and Bases Pearson Education, Inc.
 Organic Chemistry 6 th Edition Paula Yurkanis Bruice Chapter 1 Electronic Structure and Bonding Acids and Bases 2011 Pearson Education, Inc. 1 Organic Chemistry Carbon-containing compounds were once considered
Organic Chemistry 6 th Edition Paula Yurkanis Bruice Chapter 1 Electronic Structure and Bonding Acids and Bases 2011 Pearson Education, Inc. 1 Organic Chemistry Carbon-containing compounds were once considered
Name. Chapter 4 covers acid-base chemistry. That should help you get going.
 Name Chapter 4 covers acid-base chemistry. That should help you get going. 1 Use curved arrows to illustrate the transfer of a proton (i.e. an + ) from benzoic acid to phenoxide, and draw the products.
Name Chapter 4 covers acid-base chemistry. That should help you get going. 1 Use curved arrows to illustrate the transfer of a proton (i.e. an + ) from benzoic acid to phenoxide, and draw the products.
H H H H S H H H NO 2 O H H 3 C. OMe CH 3
 Fall Workshop #9 Question #1 Using the tables on pages 18-19 of SAM, the table inside the back cover of Jones, and the figure we created in class: (a) Report an estimated pka value for each hydrogen atom
Fall Workshop #9 Question #1 Using the tables on pages 18-19 of SAM, the table inside the back cover of Jones, and the figure we created in class: (a) Report an estimated pka value for each hydrogen atom
Objective 3. Draw resonance structures, use curved arrows, determine extent of delocalization. Identify major/minor contributor.
 Objective 3 Draw resonance structures, use curved arrows, determine extent of delocalization. Identify major/minor contributor. Structure Should Fit Experimental Data The chemical formula of benzene is
Objective 3 Draw resonance structures, use curved arrows, determine extent of delocalization. Identify major/minor contributor. Structure Should Fit Experimental Data The chemical formula of benzene is
Chapter 3 Acids and Bases. The Curved-Arrow Notation
 Organic Chemistry, 5th ed. Marc Loudon Chapter 3 Acids and Bases. The Curved-Arrow Notation Eric J. Kantorows ki California Polytechnic State University San Luis Obispo, CA Chapter 3 Overview 3.1 Lewis
Organic Chemistry, 5th ed. Marc Loudon Chapter 3 Acids and Bases. The Curved-Arrow Notation Eric J. Kantorows ki California Polytechnic State University San Luis Obispo, CA Chapter 3 Overview 3.1 Lewis
CHAPTER 9 THEORY OF RESONANCE BY, G.DEEPA
 CHAPTER 9 THEORY OF RESONANCE BY, G.DEEPA Conjugation in Alkadienes and Allylic Systems conjugation a series of overlapping p orbitals The Allyl Group allylic position is the next to a double bond 1 allyl
CHAPTER 9 THEORY OF RESONANCE BY, G.DEEPA Conjugation in Alkadienes and Allylic Systems conjugation a series of overlapping p orbitals The Allyl Group allylic position is the next to a double bond 1 allyl
NAME: SPRING 2015 MIDTERM
 page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
page 1 pts NAME: SPRING 2015 MIDTERM hemistry 231 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 4/6) Do your best on this take-home portion of your mid-term. I may grade
Module 2 Acids and Bases. Lecture 3 Acids and Bases
 Module 2 Acids and Bases Lecture 3 Acids and Bases 2.1 Concepts A compound is classified as an acid or a base based on certain properties. At present there are several theories which define the concepts
Module 2 Acids and Bases Lecture 3 Acids and Bases 2.1 Concepts A compound is classified as an acid or a base based on certain properties. At present there are several theories which define the concepts
Chapter 1 Atomic and Molecular Structure
 Name Date PEP Organic Chemistry Think About It: What is organic chemistry? Chapter 1 Atomic and Molecular Structure Describe some of the ways that ancient civilizations have taken advantage of organic
Name Date PEP Organic Chemistry Think About It: What is organic chemistry? Chapter 1 Atomic and Molecular Structure Describe some of the ways that ancient civilizations have taken advantage of organic
Full file at https://fratstock.eu
 1. From the list below, pick the one species that cannot act as both a Bronsted-Lowry acid and base. A) HCO3 D) OH B) H2SO4 E) HPO4 2 C) H2PO4 2. From the list below, pick the one species that is not a
1. From the list below, pick the one species that cannot act as both a Bronsted-Lowry acid and base. A) HCO3 D) OH B) H2SO4 E) HPO4 2 C) H2PO4 2. From the list below, pick the one species that is not a
Nucleophilic Addition Reactions of Carboxylic Acid Derivatives
 Lecture 5: bjectives: Nucleophilic Addition eactions of Carboxylic Acid Derivatives By the end of this lecture you will be able to: draw the mechanism of a nucleophilic addition-elimination reaction with
Lecture 5: bjectives: Nucleophilic Addition eactions of Carboxylic Acid Derivatives By the end of this lecture you will be able to: draw the mechanism of a nucleophilic addition-elimination reaction with
Chemistry 201. MW 12pm 1:15pm Examination #1 July 20 th Bronco ID. Question Score Possible Points. 1 (17pts) 2 (28pts) 3 (14pts) 4...
 Chemistry 201 MW 12pm 1:15pm Examination #1 July 20 th 2016 Name Bronco ID. Question Score Possible Points 1 (17pts) 2 (28pts) 3 (14pts) 4... (22pts) 5 (19pts). Total (100pts) 1. Read each question carefully.
Chemistry 201 MW 12pm 1:15pm Examination #1 July 20 th 2016 Name Bronco ID. Question Score Possible Points 1 (17pts) 2 (28pts) 3 (14pts) 4... (22pts) 5 (19pts). Total (100pts) 1. Read each question carefully.
Chapter 8. Acidity, Basicity and pk a
 Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Chemistry: The Central Science. Chapter 8: Basic Concepts of Chemical Bonding
 Chemistry: The Central Science Chapter 8: Basic Concepts of Chemical Bonding The properties of substances are determined in large part by the chemical bonds that hold their atoms together 8.1: Chemical
Chemistry: The Central Science Chapter 8: Basic Concepts of Chemical Bonding The properties of substances are determined in large part by the chemical bonds that hold their atoms together 8.1: Chemical
Chapter Nine. Chemical Bonding I
 Chapter Nine Chemical Bonding I 1 The Ionic Bond and Lattice Energies 2 Lewis Dot Symbols Consists of atomic symbol surrounded by 1 dot for each valence electron in the atom Only used for main group elements
Chapter Nine Chemical Bonding I 1 The Ionic Bond and Lattice Energies 2 Lewis Dot Symbols Consists of atomic symbol surrounded by 1 dot for each valence electron in the atom Only used for main group elements
Exam Analysis: Organic Chemistry, Midterm 1
 Exam Analysis: Organic Chemistry, Midterm 1 1) TEST BREAK DOWN: There are three independent topics covered in the first midterm, which are hybridization, structure and isomerism, and resonance. The test
Exam Analysis: Organic Chemistry, Midterm 1 1) TEST BREAK DOWN: There are three independent topics covered in the first midterm, which are hybridization, structure and isomerism, and resonance. The test
Chem 3719 Klein Chapter Practice Problems
 Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
Chem 379 Klein Chapter Practice Problems Dr. Peter Norris, 208 Klein Chapter Problems : Review of General Chemistry. Draw viable structures for molecules with the following molecular formulae. Remember
 Organic Chemistry I Dr Alex Roche Organic chemistry is the chemistry of Carbon and its compounds. Organic molecules constitute the essence of life (fats, sugars, proteins, DNA), and also permeate our everyday
Organic Chemistry I Dr Alex Roche Organic chemistry is the chemistry of Carbon and its compounds. Organic molecules constitute the essence of life (fats, sugars, proteins, DNA), and also permeate our everyday
Chapter 2 Lecture Outline
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 2 Lecture Outline Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 2 Lecture Outline Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
What is reactivity based on? What do all elements want to be happy? Draw the Lewis Dot Structure for Sodium and Chlorine.
 What is reactivity based on? What do all elements want to be happy? Draw the Lewis Dot Structure for Sodium and Chlorine. FORCES What is a chemical bond? The force that holds two atoms together Between
What is reactivity based on? What do all elements want to be happy? Draw the Lewis Dot Structure for Sodium and Chlorine. FORCES What is a chemical bond? The force that holds two atoms together Between
Facebook: UCI ORganic Chemistry Peer Tutoring King 51 Fall 2017
 1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
1 Organic Chemistry 51A Professor King Final Exam Review Session December 7 th, 2017 Will Cabanela (rcabanel@uci.edu) Amanda Pinski (apinski@uci.edu) Zachary Valley (zvalley@uci.edu) http://sites.uci.edu/ochemtutors
Reaction mechanisms offer us insights into how reactions work / how molecules react with one another.
 Introduction 1) Lewis Structures 2) Representing Organic Structures 3) Geometry and Hybridization 4) Electronegativities and Dipoles 5) Resonance Structures (a) Drawing Them (b) Rules for Resonance 6)
Introduction 1) Lewis Structures 2) Representing Organic Structures 3) Geometry and Hybridization 4) Electronegativities and Dipoles 5) Resonance Structures (a) Drawing Them (b) Rules for Resonance 6)
Chapter 9 Bonding - 1. Dr. Sapna Gupta
 Chapter 9 Bonding - 1 Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and
Chapter 9 Bonding - 1 Dr. Sapna Gupta Lewis Dot Symbol Lewis dot symbols is a notation where valence electrons are shown as dots. Draw the electrons symmetrically around the sides (top, bottom, left and
MULTIPLE CHOICE 2 points each
 Name: Date: Score: / 110 Chapter 1/ TEST 1 OPEN BOOK KEY Organic Chemistry MULTIPLE CHOICE 2 points each 1. An atom of which element would have an electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 1? a.
Name: Date: Score: / 110 Chapter 1/ TEST 1 OPEN BOOK KEY Organic Chemistry MULTIPLE CHOICE 2 points each 1. An atom of which element would have an electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 1? a.
Chemistry: The Central Science
 Chemistry: The Central Science Fourteenth Edition Chapter 8 Basic Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions Covalent Sharing of
Chemistry: The Central Science Fourteenth Edition Chapter 8 Basic Concepts of Chemical Bonding Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions Covalent Sharing of
Many Organic compounds are acids or bases (or both) Many Organic compounds undergo acid-base reactions
 Objective 4 Intro to Reactivity 1: identify acids and bases using Lewis definition. Use curved arrows to show how base reacts with acid. Relate strength to pk a. Determine direction of equilibrium. Use
Objective 4 Intro to Reactivity 1: identify acids and bases using Lewis definition. Use curved arrows to show how base reacts with acid. Relate strength to pk a. Determine direction of equilibrium. Use
Chapter 3. Acids and Bases
 Chapter 3 Acids and Bases 3.1 Acids and Bases Brønsted-Lowry definition Acids donate a proton Bases accept a proton Recall from General Chemistry this classic example 3-2 3.1 Conjugate Acids and Bases
Chapter 3 Acids and Bases 3.1 Acids and Bases Brønsted-Lowry definition Acids donate a proton Bases accept a proton Recall from General Chemistry this classic example 3-2 3.1 Conjugate Acids and Bases
Why do I care, Dr. P?
 Chapter 2- Polar Covalent Bonds; Acids and Bases Ashley Piekarski, Ph.D. Why do I care, Dr. P? In Chapter 1, we studied valence bond theory which uses hybrid orbitals to account for the observed shapes
Chapter 2- Polar Covalent Bonds; Acids and Bases Ashley Piekarski, Ph.D. Why do I care, Dr. P? In Chapter 1, we studied valence bond theory which uses hybrid orbitals to account for the observed shapes
Chapter 12. Reactions of Arenes: Electrophilic Aromatic Substitution. Class Notes. A. The method by which substituted benzenes are synthesized
 Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Chapter 12 Reactions of Arenes: Electrophilic Aromatic Substitution Chapter 12 suggested problems: 22, 23, 26, 27, 32, 33 Class Notes I. Electrophilic aromatic substitution reactions A. The method by which
Resonance and M.O. View of Butadiene. Super-Conjugated or Aromatic p e - Systems
 Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
Resonance and M.. View of Butadiene The different resonance forms of butadiene suggest p bonding character between the two central carbon atoms. 2 2 2 2 carbanion 2 2 carbocation The M.. view of butadiene
Acid-Base Chemistry. Introduction to Reaction Mechanism
 Acid-Base Chemistry Introduction to Reaction Mechanism What is an acid and what is a base? Bronsted-Lowry definition Acids are proton donors (A-H) e.g. HCl, H 2 SO 4, HBr, HNO 3, HI etc. Bronsted-Lowry
Acid-Base Chemistry Introduction to Reaction Mechanism What is an acid and what is a base? Bronsted-Lowry definition Acids are proton donors (A-H) e.g. HCl, H 2 SO 4, HBr, HNO 3, HI etc. Bronsted-Lowry
Chapter In each case the conjugate base is obtained by removing a proton from the acid: (a) OH (b) I (c)
 Practice Exercises 16.1 Conjugate acid base pairs (a), (c), and (f) (b) The conjugate base of I is I (d) The conjugate base of N 2 is N 2 and the conjugate base of N 4 is N 3 (e) The conjugate acid of
Practice Exercises 16.1 Conjugate acid base pairs (a), (c), and (f) (b) The conjugate base of I is I (d) The conjugate base of N 2 is N 2 and the conjugate base of N 4 is N 3 (e) The conjugate acid of
Organic Chemistry CHM 314 Dr. Laurie S. Starkey, Cal Poly Pomona Structure & Shape of Organic Molecules - Chapter 1 (Wade)
 rganic hemistry M 314 Dr. Laurie S. Starkey, al Poly Pomona Structure & Shape of rganic Molecules - hapter 1 (Wade) rganic (living things, chemistry of carbon) Examples of rganic ompounds: Inorganic (rocks,
rganic hemistry M 314 Dr. Laurie S. Starkey, al Poly Pomona Structure & Shape of rganic Molecules - hapter 1 (Wade) rganic (living things, chemistry of carbon) Examples of rganic ompounds: Inorganic (rocks,
Chapter 02 - Polar Covalent Bonds; Acids and Bases
 Exhibit 2-1 Give the corresponding letter of the term that best matches the given definition. a. Brønsted-Lowry Acid f. Ionic Bond b. Brønsted-Lowry Base g. Covalent Bond c. Lewis Acid h. Polar-Covalent
Exhibit 2-1 Give the corresponding letter of the term that best matches the given definition. a. Brønsted-Lowry Acid f. Ionic Bond b. Brønsted-Lowry Base g. Covalent Bond c. Lewis Acid h. Polar-Covalent
CHE1502. Tutorial letter 203/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry
 E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
Final Exam. Ian R. Gould PRINTED FIRST NAME
 CEM 234, Spring 2015 Final Exam Ian R. Gould PRITED FIRST AME ASWER PRITED LAST AME KEY ASU ID or Posting ID Person on your LEFT (or Aisle) Person on your RIGT (or Aisle) 1 /12 2 /20 3 /25 4 /12 5 /20
CEM 234, Spring 2015 Final Exam Ian R. Gould PRITED FIRST AME ASWER PRITED LAST AME KEY ASU ID or Posting ID Person on your LEFT (or Aisle) Person on your RIGT (or Aisle) 1 /12 2 /20 3 /25 4 /12 5 /20
Chapter 3 Acids and Bases"
 Chapter 3 Acids and Bases BrØnsted-Lowry Acids and Bases A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton Acids and Bases Reactions of BrØnsted-Lowry Acids
Chapter 3 Acids and Bases BrØnsted-Lowry Acids and Bases A BrØnsted-Lowry acid is a proton donor. A BrØnsted-Lowry base is a proton acceptor. H + = proton Acids and Bases Reactions of BrØnsted-Lowry Acids
Normal Bonds (Sections ) Summary of Normal, Ideal Bonding (No Formal Charge)
 hem 350 Jasperse h. 1 Normal Bonding; Drawing Lewis Structures 1 Normal Bonds (Sections 1.2-1.5) Summary of Normal, Ideal Bonding (No Formal harge) Valence Electrons Valence Bonds Lone Pairs 4 4 0 N 5
hem 350 Jasperse h. 1 Normal Bonding; Drawing Lewis Structures 1 Normal Bonds (Sections 1.2-1.5) Summary of Normal, Ideal Bonding (No Formal harge) Valence Electrons Valence Bonds Lone Pairs 4 4 0 N 5
OH OH OH CH 2 CH 2 C(CH 3 ) 2 (a) CH 3 CHCH 2 CHCH(CH 3 ) 2. (b)
 hem 226 Problem Set #8 Fundamentals of rganic hemistry, 4 th edition, John McMurry. hapter 8 1. Give IUPA names for the following alcohols. 2 2 ( 3 ) 2 3 2 ( 3 ) 2 Longest chain = 6 carbons:...hexanediol.
hem 226 Problem Set #8 Fundamentals of rganic hemistry, 4 th edition, John McMurry. hapter 8 1. Give IUPA names for the following alcohols. 2 2 ( 3 ) 2 3 2 ( 3 ) 2 Longest chain = 6 carbons:...hexanediol.
CHE2060 Lecture 5: Acid-base chemistry. CHE2060 Lecture 5: Acid-base chemistry
 CHE2060 Lecture 5: Acid-base chemistry 5.1 Acids & bases: overview & basics 5.2 Acid & base strength 5.3 Equilibrium acid-base reactions 5.4 The leveling effect of solvents 5.5 Estimation of acidity by
CHE2060 Lecture 5: Acid-base chemistry 5.1 Acids & bases: overview & basics 5.2 Acid & base strength 5.3 Equilibrium acid-base reactions 5.4 The leveling effect of solvents 5.5 Estimation of acidity by
Chapter 8. Basic Concepts of Chemical Bonding
 Chapter 8. Basic Concepts of Chemical Bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule 8.2 Ionic Bonding positive and negative ions form an ionic lattice, in which each cation is surrounded
Chapter 8. Basic Concepts of Chemical Bonding 8.1 Chemical Bonds, Lewis Symbols, and the Octet Rule 8.2 Ionic Bonding positive and negative ions form an ionic lattice, in which each cation is surrounded
Chapter 8. Basic Concepts of Chemical Bonding. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 8 of Chemical John D. Bookstaver St. Charles Community College Cottleville, MO Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions. Covalent
Lecture Presentation Chapter 8 of Chemical John D. Bookstaver St. Charles Community College Cottleville, MO Chemical Bonds Three basic types of bonds Ionic Electrostatic attraction between ions. Covalent
**YOU ARE NOT ALLOWED TO TAKE SPARE COPIES OF THIS EXAM FROM THE TESTING ROOM**
 EM 233, Fall 2017 Midterm #3 Ian R. Gould MPLETE TIS SETIN : Up to TW PINTS will be removed for incorrect/missing information! PRINTED FIRST NME answer key PRINTED LST NME Person on your LEFT (or Empty
EM 233, Fall 2017 Midterm #3 Ian R. Gould MPLETE TIS SETIN : Up to TW PINTS will be removed for incorrect/missing information! PRINTED FIRST NME answer key PRINTED LST NME Person on your LEFT (or Empty
Chapter 6 Molecular Structure
 hapter 6 Molecular Structure 1. Draw the Lewis structure of each of the following ions, showing all nonzero formal charges. Indicate whether each ion is linear or bent. If the ion is bent, what is the
hapter 6 Molecular Structure 1. Draw the Lewis structure of each of the following ions, showing all nonzero formal charges. Indicate whether each ion is linear or bent. If the ion is bent, what is the
NAME: SUMMER 2015 MIDTERM
 page 1 pts NAME: SUMMER 2015 MIDTERM hemistry 350 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 6/16) Do your best on this take-home portion of your midterm. I may grade
page 1 pts NAME: SUMMER 2015 MIDTERM hemistry 350 Professor: Dr. Gergens take-home portion (DUE at the beginning of the period, 6/16) Do your best on this take-home portion of your midterm. I may grade
Basic chemistry for general biology. Electrons and orbitals, and how bonds make happy atoms
 Basic chemistry for general biology Electrons and orbitals, and how bonds make happy atoms A review (I hope) Atoms are composed of three elementary particles: protons, electrons, and neutrons Protons (H+)
Basic chemistry for general biology Electrons and orbitals, and how bonds make happy atoms A review (I hope) Atoms are composed of three elementary particles: protons, electrons, and neutrons Protons (H+)
ORGANIC - BROWN 8E CH.4 - ACIDS AND BASES.
 !! www.clutchprep.com CONCEPT: FREE ENERGY DIAGRAMS Atoms save energy by forming bonds. Free energy diagrams show overall changes in potential energy during reactions. Free energy diagrams give us information
!! www.clutchprep.com CONCEPT: FREE ENERGY DIAGRAMS Atoms save energy by forming bonds. Free energy diagrams show overall changes in potential energy during reactions. Free energy diagrams give us information
Chapter 02 - Polar Covalent Bonds; Acids and Bases. Exhibit 2-1
 Exhibit 2-1 Organic Chemistry 9th Edition McMurry TEST BANK Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurry-test-bank/ Organic Chemistry 9th Edition McMurry
Exhibit 2-1 Organic Chemistry 9th Edition McMurry TEST BANK Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurry-test-bank/ Organic Chemistry 9th Edition McMurry
Valence electrons octet rule. Lewis structure Lewis structures
 Lewis Dot Diagrams Valence electrons are the electrons in the outermost energy level of an atom. An element with a full octet of valence electrons has a stable configuration. The tendency of bonded atoms
Lewis Dot Diagrams Valence electrons are the electrons in the outermost energy level of an atom. An element with a full octet of valence electrons has a stable configuration. The tendency of bonded atoms
Organic Chemistry, 7e (Bruice) Chapter 2 Acids and Bases: Central to Understanding Organic Chemistry
 Organic Chemistry, 7e (Bruice) Chapter 2 Acids and Bases: Central to Understanding Organic Chemistry 1) Which of the following is not a conjugate acid-base pair? A) H2O, HO- B) H2O, H3O+ C) HSO4 -, H2SO4
Organic Chemistry, 7e (Bruice) Chapter 2 Acids and Bases: Central to Understanding Organic Chemistry 1) Which of the following is not a conjugate acid-base pair? A) H2O, HO- B) H2O, H3O+ C) HSO4 -, H2SO4
Chapter 8 Concepts of Chemical. Bonding
 Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 8 Concepts of John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 8 Concepts of John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice
What Is Organic Chemistry?
 What Is Organic Chemistry? EQ: What is Organic Chemistry? Read: pages 1-3 Answer the questions in your packet Basics of Organic Chem 1 Chapter 1: Structure and Bonding Key terms Organic Chemistry Inorganic
What Is Organic Chemistry? EQ: What is Organic Chemistry? Read: pages 1-3 Answer the questions in your packet Basics of Organic Chem 1 Chapter 1: Structure and Bonding Key terms Organic Chemistry Inorganic
(1) Recall the different types of intermolecular interactions. (2) Look at the structure and determine the correct answer.
 MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
MCAT rganic Chemistry - Problem Drill 11: Carboxylic Acids Question No. 1 of 10 Question 1. What kinds of interactions are holding together the carboxylic acid dimer shown? Question #01 3 C C 3 (A) Van
Learning Guide for Chapter 1 - Atoms and Molecules
 Learning Guide for hapter 1 - Atoms and Molecules I. Introduction to organic chemistry - p 1 II. Review of atomic structure - p 3 Elementary particles Periodic Table of Elements Electronegativity Atomic
Learning Guide for hapter 1 - Atoms and Molecules I. Introduction to organic chemistry - p 1 II. Review of atomic structure - p 3 Elementary particles Periodic Table of Elements Electronegativity Atomic
Chapter 19: Aromatic Substitution Reactions
 Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
Chem A225 Notes Page 52 Chapter 19: Aromatic Substitution Reactions Topic One: lectrophilic Aromatic Substitution I. Introduction to lectrophilic Aromatic Substitution (AS) A. eneral Reaction Pattern B.
px3 All bonding with all hybrid orbitals will occur on the larger lobe for each hybrid.
 Reasons for ybridization 1. Allows atoms to have the correct number of unpaired electrons to make necessary number of bonds. 2. Best spatial orientation achieved least amount of electron repulsions 3.
Reasons for ybridization 1. Allows atoms to have the correct number of unpaired electrons to make necessary number of bonds. 2. Best spatial orientation achieved least amount of electron repulsions 3.
Chem 350 Jasperse Ch. 1 Notes 1
 Chem 350 Jasperse Ch. 1 Notes 1 Ch. 1 Intro and Review 1.1 Intro to rganic Chemistry rganic : rganic Chemistry : Focus on carbon, with, N,, and halogens all major contributors Biochemicals are all carbon-based
Chem 350 Jasperse Ch. 1 Notes 1 Ch. 1 Intro and Review 1.1 Intro to rganic Chemistry rganic : rganic Chemistry : Focus on carbon, with, N,, and halogens all major contributors Biochemicals are all carbon-based
Chemistry 201. MW 12pm 1:15pm Examination #1 July 22 nd Bronco ID. Question Score Possible Points. 1 (10pts) 2 (24pts) 3 (14pts) 4...
 Chemistry 201 MW 12pm 1:15pm Examination #1 July 22 nd 2015 Name Bronco ID. Question Score Possible Points 1 (10pts) 2 (24pts) 3 (14pts) 4... (22pts) 5 (30pts). Total (100pts) 1. Read each question carefully.
Chemistry 201 MW 12pm 1:15pm Examination #1 July 22 nd 2015 Name Bronco ID. Question Score Possible Points 1 (10pts) 2 (24pts) 3 (14pts) 4... (22pts) 5 (30pts). Total (100pts) 1. Read each question carefully.
CHEM 3013 ORGANIC CHEMISTRY I LECTURE NOTES CHAPTER 2
 EM 3013 RGANI EMISTRY I LETURE NTES 1 APTER 2 1. ormal harge The Lewis structures we have drawn thus far have all been neutral covalent molecules. owever, some covalently bonded molecules may contain charged
EM 3013 RGANI EMISTRY I LETURE NTES 1 APTER 2 1. ormal harge The Lewis structures we have drawn thus far have all been neutral covalent molecules. owever, some covalently bonded molecules may contain charged
Course Goals for CHEM 202
 Course Goals for CHEM 202 Students will use their understanding of chemical bonding and energetics to predict and explain changes in enthalpy, entropy, and free energy for a variety of processes and reactions.
Course Goals for CHEM 202 Students will use their understanding of chemical bonding and energetics to predict and explain changes in enthalpy, entropy, and free energy for a variety of processes and reactions.
