Supporting Information
|
|
|
- Hubert Reed
- 5 years ago
- Views:
Transcription
1 Supporting Information On the Minimal Size of Coffee Ring Structure Xiaoying Shen, Chih-Ming Ho and Tak-Sing Wong * Mechanical and Aerospace Engineering Department, University of California, Los Angeles, California Institute of Microelectronics Engineering, Peking University, Beijing, P. R. China *Corresponding author. tswong@ucla.edu Experimental Section Materials. Polystyrene latex beads (SIGMA, LB-1) surfactant-stabilized suspension with a particle diameter of 113 ± 11 nm (m = 20, where m is the number of the independent measurements from scanning electron microscope, SEM), was used as received, where the particle density was particles/ml. In some of the experiments, the suspension was further diluted by 10 or 100 times, with the addition of trace amount of anionic surfactant sodium dodecyl sulphate (SDS) (Fluka, Switzerland, diluted to < 0.01%) to stabilize the particle suspension. In addition, carboxyl latex beads (Molecular Probes ) charge-stabilized suspension was used as received. Three different particle sizes were used in the experiments: 28 ± 5 nm, 3.3 x particles/ml; 62 ± 6 nm, 3.1 x particles/ml; 140 ± 13 nm, 2.7 x particles/ml. Purified deionized water with a resistivity of ~18.3 MΩ/cm was used for the dilution. S1
2 Fabrication of Chemically Heterogeneous Surfaces. Following the published experimental protocol (Wong T.S. et al., Langmuir 2009, 25, 12, ), standard photolithography procedures were used to create engineered surfaces with patterned platinum (Pt) hydrophilic cylindrical structures on a relatively nonwetting surface coated with hexamthyldisilazane (HMDS). These surfaces were utilized to control the size of liquid droplets. The diameters of the hydrophilic cylindrical structures ranged from 3 µm to 1 mm, and the height of the structures measured ~20 nm, as confirmed by atomic force microscopy (AFM, Veeco Dimension 3100; AFM tip used: RTESP Veeco, NY, with specified tip radius of curvature < 10 nm) measurement. Based on the patterned heterogeneous surfaces, a colloidal solution, with known particle size and particle concentration, receded from a relatively nonwetting substrate onto the hydrophilic structures. Liquid droplets were retained on the structures as a result of pinning to the solid edge. Nanoparticle deposits were formed on the hydrophilic surfaces upon solvent evaporation under a humidity-controlled environment at room temperature. Surface Characterization. Contact angle measurements were carried out for all the particle suspensions on both Pt and HMDS surfaces by a drop shape analysis system (FTA 4000) at ambient temperature (24 26 o C) with ~50% relative humidity under an enclosed environment. The droplet volume for the measurement was ~0.5 µl (for static contact angle), and the droplet profile was captured through a camera equipped with an optical system for the amplification of the captured images. All the contact angle measurements were performed within 30 seconds; otherwise the data point will be discarded due to excessive evaporation of the droplet. The droplet profile was fitted into a spherical cap profile by an in-built computer program provided by the system, in order to determine the contact angle. In addition, dynamic receding and advancing contact angle measurements were achieved by withdrawal and addition of tested colloidal solutions through an automated syringe system, and the droplet profiles were captured subsequently for the measurements. The droplet evaporation history was also studied using the FTA 4000 system. The contact angle and the base diameter of a droplet in each frame were measured separately using the in-built program. S2
3 Formation of Particle Deposits. A chip with patterned surfaces was transferred to a humidity-controlled chamber (Electro-Tech Systems Inc, PA) for the particle deposits formation process. The relative humidity of the chamber can be controlled from 10% to 90% (with accuracy of ±2%) at room temperature of 22 o C 25 o C. The humidity chamber was adjusted to the targeted relative humidity (e.g., 50%) for ~1 hour prior to the experiments. In all of the experiments, ~80 µl of the colloidal solution was placed on the chip, where it was tilted to remove the excess solution. The chip was left in the humidity chamber for ~30 min until the solvent was fully evaporated. The pinning of the liquid to the hydrophilic surfaces and the subsequent formation of liquid droplets were further confirmed through the use of a digital video camera connected to an optical microscope. Observation and Measurement. A combination of scanning electron microscope (SEM, Hitachi S4700) and AFM were utilized to obtain the quantitative measurements of the particle deposits. SEM was utilized to obtain two-dimensional information of the deposits (i.e. particle density distribution and the distance between the edge of the ring structure and the initial contact line) and AFM was used to obtain the third dimensional information (i.e. height). Besides, SEM was also utilized to measure the diameter of the hydrophilic surfaces and particle diameters. Data Analysis. The diameters of the hydrophilic sites used for the experiments are listed as follow: 3 µm, 5 µm, 10 µm, 25 µm, 50 µm, 100 µm, 250 µm, 500 µm, and 1 mm. The captured SEM images were processed and analyzed using an image processing software ImageJ (developed at National Institute of Health, USA). The SEM images ( pixels) were processed to obtain the dimension of each pixel, which will be used for the quantitative measurements of the nanoparticle deposits. A minimum of 10 independent measurements were carried out for each hydrophilic site to obtain L p for all of the experiments. S3
4 Engineered Heterogeneous Surfaces and the Liquid Receding Experiment Figure S1. (a) SEM image showing the engineered heterogeneous surfaces. The diameter of the hydrophilic sites ranged from 1 mm down to 3 µm. (b) Time sequence optical images showing the droplet formation process on the hydrophilic sites (droplet size: 100 µm and 250 µm). A macroscopic colloidal solution was first applied onto the surface, where the solution was withdrew from the surface, leaving part of the liquid being trapped onto the hydrophilic structures due to pinning. Upon the solvent evaporation, nanoparticles deposit form on the hydrophilic sites. S4
5 Summary of macroscopic contact angle measurements Table S1. Contact angle measurements of water and colloidal solutions used in the experiments. Measurement Type Water 100 nm Surfactantstablized 100 nm Chargestablized 60 nm Chargestablized 20 nm Chargestablized Pt θ adv 47.0 ± ± ± ± ±4.1 θ static 22.8 ± ± ± ± ±0.3 θ rec 11.3 ± HMDS θ adv 78.4 ± ± ± ± ±5.8 θ rec 63.9 ± Note: The receding angles of the colloidal droplets cannot be measured accurately due to the strong CL pinning by the coffee ring structures. S5
6 Droplet Evaporation History of Various Colloidal Solutions on Pt surfaces. Figure S2. Evaporation history of different particle suspensions on Pt surfaces Table S2. Evaporation history of different particle suspensions on Pt surfaces Water 100 nm Surfactantstabilized 100 nm Chargestabilized 60 nm Chargestabilized 20 nm Chargestabilized Slope θ initial 21.9 ± ± ± ± ±0.6 The initial stage of the evaporation dynamics for both 100 nm (b) surfactant-stabilized solution and (c) charged-stabilized solution are similar to that of the (a) pure water, where they kept a constant area mode of evaporation, indicating strong pinning of the CL to the edge of the hydrophilic sites. For colloidal particles with smaller particle sizes, the droplet first followed a constant area mode of evaporation, and then transited to a constant contact angle mode of evaporation. The observation was more pronounced for the colloidal solution with 20 nm particles, indicating that the CL pinning ability is S6
7 weaker compared to that of 100 nm particles. Table S1 shows the slope (i.e., evaporation rate) and initial contact angle obtained from the evaporation history plots. S7
8 Nanoparticle Deposits at Different Length Scales Figure S3. SEM images for nanoparticle deposits on hydrophilic Pt structures with diameters ranging from 3 µm to 500 µm. Representative SEM images showing the deposit patterns at different droplet diameters: (a) 3 µm, (b) 5 µm, (c) 10 µm, (d) 25 µm, (e) 50 µm, (f) 100 µm, (g) 250 µm, (h) 500µm. Two different regimes of the deposit patterns are clearly distinguishable. SEM images show that for the droplet smaller than 10µm, the nanoparticles are dispersed homogeneously on the surface. For the droplet large enough, nanoparticles form coffee ring structure after droplet evaporation. It is important to note that the image contrast of the SEM images is dependent on the local thickness of the coffee ring structure. Therefore the perimeters of the coffee ring structures appear to be darker as the number of the nanoparticles layers increase. S8
9 Effect of Surfactants to the Coffee Ring Formation. Figure S4. Comparison of the evaporation rate and L p between the surfactant-stabilized particle suspension and surfactant-free charge-stabilized colloidal solutions. The plots indicate that the presence of surfactants does not severely impact the evaporation rate of the colloidal solution, as well as L p for the coffee ring structures. S9
10 Nanoparticle Deposits for Small Droplets Figure S5. (a) SEM image of 100 nm particles deposited in a 3 µm droplet. (particle concentration: particles/ml, relative humidity = 50% at room temperature). (b) SEM image of 100 nm particles deposit in a 5 µm droplet (particle concentration: particles/ml, relative humidity = 50% at room temperature). In these SEM images, it is clear that there were sufficient nanoparticles to form at least one concentric coffee ring structure at the edge of the droplet, yet no coffee rings were formed due to the fast evaporation of small droplets. This further highlights that the evaporation time of the droplets plays an important role in the coffee ring formation process. S10
11 Monolayer Layer Formation near the Contact Line Figure S6. (a) An AFM image showing the surface topography of a coffee ring section (A A ) formed by surfactant-stabilized 100 nm particles. The monolayer formation at the boundary of the coffee ring structure is confirmed by the particle height measurement, where the average particle height (i.e., the difference between the red dotted lines) matches to the diameter of a single particle (i.e., ~100 nm). This indicates that the formation of the coffee ring structure is first initiated from the formation of monolayer, which plays an important role for the CL pinning. (b) SEM images showing a coffee ring structure formed for a 25 µm droplet. From the magnified image at the corner of coffee ring structures, we can see that a small number of concentric rings of nanoparticles are sufficient for the CL pinning. S11
12 Nanoparticle Deposit Patterns for 20 nm and 60 nm particles Figure S7. Representative SEM images showing the nanoparticle deposit patterns for (a) 20 nm and (b) 60 nm particle suspension. For the 20 nm particles, all of the nanoparticles were distributed uniformly on the surface; whereas for the 60 nm particles, concentric circular patterns were formed on the hydrophilic sites instead of a single coffee ring structure, indicating repeated CL pinning and depinning. S12
13 Derivation of Eq. 1 in the Main Text Figure S8. (a) SEM image showing the monolayer formation of nanoparticles close to the contact line, where a finite distance is observed between the edge of the particle monolayer and the initial contact line of droplet. By simple geometry, we derived a relationship which relates L p and the particle sizes of the colloidal suspension, L p = r θ tan( local 2) (S-e1) where r is the radius of the nanoparticles, and θ local is the local contact angle between the three phase CL at the particle surface and the horizontal Pt surface. Notice that this equation is derived under the conditions where D >> D c and r << D, where D is the diameter of the hydrophilic site. S13
14 Derivation of Eqs. 2 to 7 in the Main Text Time of Evaporation ( τ evap ): We consider the evaporation process of a sessile droplet of particle suspension on a horizontal surface. In our experiments, the droplet diameter is sufficiently small (D < 1 mm) so that surface tension is dominant, and the gravitational effect can be neglected. Therefore the droplet can be modeled as a spherical-cap shape. As supported by our experimental measurements on the evaporation dynamics of colloidal liquid droplets, the initial phase of the evaporation process is predominately in constant contact base diameter mode. Therefore we employed the constant contact base diameter evaporation model for our calculation. The contact angle is small enough (θ << 57.2 ) and the maximum height of droplet is much smaller than its radius. We assume that the droplet profile is not affected by either the particle transport or the capillary flow developed within droplet. Based on our assumptions and experimental conditions, we applied the theoretical model by Popov et al. (Popov et al. Physical Review E 71, ) to deduce τ evap, which is described as follow. Figure S9. A schematic showing a droplet sitting on a solid surface. Considering in Figures S6 where a liquid droplet sits on a horizontal solid surface with base diameter, D, radius, R = 2 D and a contact angle of θ (i.e., the angle between the liquid-vapor interface and the horizontal plane of the substrate). The volume of the liquid droplet, V d (t), can be calculated as, 3 R( t) 1 R ( t) 2 Vd ( t) = h( r, t)2π rdr= π (1 cosθ ( t)) (2+ cosθ ( t)) (S-e2) sin θ ( t) where R( t) sinθ ( t) is the curvature radius of the droplet surface, and the height of droplet surface at r can be expressed as S14
15 2 R ( t) 2 h( r, t) = r R( t) cotθ ( t) 2 sin θ ( t) (S-e3) For small contact angle, θ << 57.2, Eq. S-e3 can be simplified as 2 2 R ( t) r 3 h ( r, t) = θ ( t) + O( θ ) 2R( t) (S-e4) 3 πr dv d = dθ 4 (S-e5) Based on our experimental observations, the contact base diameter of the liquid droplet stayed constant for the majority of the evaporation process, therefore we have D(t)=D i, where D i is the initial base diameter. In addition, we further assume that the evaporation process is not influenced by of the presence of solute inside the droplet (see for example, Drisdell et al., Proc. Natl. Acad. Sci. USA 2009, 106, ). The evaporation rate can be expressed as 2 Dv ( c J ( r) = π R 0 c ) 2 i r 2 (S-e6) where D v is the molecular diffusion constant of the vapor in air, c 0 is the density of the saturated vapor immediate above the liquid-vapor interface, c is the ambient vapor density. M wpvs c0 c = ( )(1 RH ) (S-e7) R T g where M w is the molar mass of water, Pvs is the saturation water vapor pressure, R g is the ideal gas constant, and T is temperature in Kelvin. From Eq. (S-e4) to (S-e7), the time derivative of droplet volume can be expressed as dv dt d = 0 R i J ( r) ρ 1+ ( h) r 2 4 v ( 0 ) 2πrdr = D c c ρ L R i (S-e8) By integrating Eq. (S-e5) and Eq. (S-e8), we can obtain the relation of θ with time. 3 i π R dθ 4D ( c c ) 4 dt v 0 = Ri (S-e9) ρl S15
16 4 θ ( t) = θ Kt (S-e10) initial πr 2 where K is a characteristic parameter represents droplet evaporation rate in the following form, i K 4Dv ( c0 c ) = (S-e11) ρ L It is important to correctly define the characteristic time scale, τ evap. Based on our experiments, we found that under the conditions where the coffee rings were well formed, there always existed a finite distance, L p, between the initial pinning point and the boundary of the coffee ring. This distance can be estimated by the following simple geometrical relationship as shown earlier, L p r = (S-e12) θ / 2) tan( local where θ local is the local contact angle at the time when the particles start to form ring-like deposit. By using experimentally measured L p and the size of the particles, r, one can determine the θ local during the coffee ring formation process. Specifically, the evaporation time from θ initial to θ local represents the time required to initiate the coffee ring formation. Based on the measured L p, we found that θ local is consistently between θ initial and θ receding. Based on our experimental observations together with (S-e10) and (S-e11), the time for a droplet contact angle changes from the initial contact angle θ initial to receding contact angle θ receding can be expressed as, τ evap θinitial θ receding 2 = πri (S-e13) 4K Time of Particle Movement ( τ particle ): To determine τ particle, we consider the diffusion time required for two adjacent nanoparticles near the CL to meet each others. The mean distance between two particles within the liquid suspension, L m, can be estimated as 3 V d / n, where V d is the volume of the droplet and n is the number of nanoparticles within the droplet. The induced capillary flow velocity is very small S16
17 when it is close to the substrate surface due to the no-slip flow boundary condition, therefore it is reasonable to neglect the impact of flow velocity on the particle movement. To estimate the time for the nanoparticles to travel through L m, we use the Einstein diffusion equation to estimate τ particle, (Einstein, 1956, ISBN ) the particle motion can be described as x 2 = 2D t (S-e14) where the diffusion coefficient of a spherical object, D p, is given by the Stokes-Einstein equation, p D p k BT =, where η is the viscosity of the suspending fluid and r is the radius of the sphere. Since the 6πηr average diffusion distance can be approximately by L m, we get 2 m L τ particle = (S-e15) 2D p By equating (S-e13) and (S-e15), we can determine a critical length scale important to the coffee ring formation, which can be expressed as, D c 8K = 2R = Lm (S-e16) = D i τ evap τ particle π ( θinitial θ receding ) p Eq. (S-e16) conveys a number of critical information for an evaporating colloidal liquid droplet. For example, the parameter K indicates the evaporation rate, which is dictated by the liquid medium and the evaporating environment. L m and D p are parameters related to the properties of the particle suspensions, such as particle concentration and sizes. The contact angle (θ initial θ receding ) characterizes the surface property. All of these parameters collectively give rise to the compact form of Eq. (S-e16) that describes the minimal size of the coffee ring structure. S17
18 Table S3. Physical Constants Used in the Calculations Symbol Physical Parameter Value Unit & Specification Ref. R g Ideal gas constant J mol -1 K -1 η Viscosity of water Pa s at 20 C [S1] *P Pressure Pa *T Temperature 25 C M w Molar mass of water g mol -1 [S1] P vs Saturation water vapor pressure Pa for T = 0 80 C [S2] k B Boltzmann constant J K -1 ρ L Density of water kg m -3 [S3] D v Diffusion coefficient of water vapor in air for T= 0 50 C error <0.1 kg m m 2 s -1 for T = 0-45 C, error < m 2 s -1 [S4] Note: * Ambient conditions in our experiments. Saturated water vapor pressure in air, empirical relation: degree Celcius ( C); range of validity, T = 0 80 C. P vs = 7.5T / T ; Temperature T in Density of water, empirical relation: ρ L = ( T 3.98) ( T 3.98) ; range of validity, T = 0 50 C, error <0.1 kg m -3 6 Diffusion coefficient of water vapor in air, empirical relation: D v = ( T ) ; T: temperature, degree Celcius( C); range of validity, T = 0 45 C, error < m 2 s -1 References (1) Aylward, G.; Findlay, T. Pyrodynamic 1967, 5. (2) Monteith, J. L.; Unsworth, M. H. Principles of environmental physics; Edward Arnold: London, (3) Weast, R. C. Handbook of Chemistry and Physics, 67th edition ed.; CRC Press Inc.: Boca Raton, Florida, USA, (4) Gates, D. M. Biophysical ecology Springer VerlagNew York Heidelberg Berlin, S18
Supplementary Information. In colloidal drop drying processes, multi-ring depositions are formed due to the stick-slip
 Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Electronic Supplementary Material (ESI for Soft Matter. This journal is The Royal Society of Chemistry 14 Supplementary Information A1. Contact line receding velocity of an evaporating drop In colloidal
Supplementary Information. for
 Supplementary Information for Discrete Element Model for Suppression of Coffee-Ring Effect Ting Xu, 1 Miu Ling Lam, 2,3,4 and Ting-Hsuan Chen 1,2,3,4 1 Department of Mechanical and Biomedical Engineering,
Supplementary Information for Discrete Element Model for Suppression of Coffee-Ring Effect Ting Xu, 1 Miu Ling Lam, 2,3,4 and Ting-Hsuan Chen 1,2,3,4 1 Department of Mechanical and Biomedical Engineering,
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning
 The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
The evaporation of sessile droplets onto solid surfaces : experiments and simulations of the contact line pinning-depinning L.Kabeya-Mukeba, N.Vandewalle and S.Dorbolo GRASP, Institut de Physique B5a,
Supplementary Information
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supplementary Information Visualization of equilibrium position of colloidal particles at fluid-water
Microfluidics 2 Surface tension, contact angle, capillary flow
 MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
MT-0.6081 Microfluidics and BioMEMS Microfluidics 2 Surface tension, contact angle, capillary flow 28.1.2017 Ville Jokinen Surface tension & Surface energy Work required to create new surface = surface
Droplet Migration during Condensation on Chemically Patterned. Micropillars
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry Please do 2016 not adjust margins RSC Advances ELECTRONIC SUPPORTING INFORMATION (ESI) Droplet Migration
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
On the Effect of an Atmosphere of Nitrogen on the Evaporation of Sessile Droplets of Water
 On the Effect of an Atmosphere of Nitrogen on the Evaporation of Sessile Droplets of Water S. K. Wilson 1, K. Sefiane 2, S. David 2, G. J. Dunn 1 and B. R. Duffy 1 1 Department of Mathematics, University
On the Effect of an Atmosphere of Nitrogen on the Evaporation of Sessile Droplets of Water S. K. Wilson 1, K. Sefiane 2, S. David 2, G. J. Dunn 1 and B. R. Duffy 1 1 Department of Mathematics, University
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/8/eaat3276/dc1 Supplementary Materials for Free-standing liquid membranes as unusual particle separators Birgitt Boschitsch Stogin, Luke Gockowski, Hannah Feldstein,
advances.sciencemag.org/cgi/content/full/4/8/eaat3276/dc1 Supplementary Materials for Free-standing liquid membranes as unusual particle separators Birgitt Boschitsch Stogin, Luke Gockowski, Hannah Feldstein,
Leandra Boucheron Shpyrko Research Group July 19, 2012
 Leandra Boucheron Shpyrko Research Group July 19, 2012 Outline Introduction How and Why do Coffee Rings Form? Manipulating Coffee Rings Reversing the Effect Particle Shape and Size Other Factors Conclusion
Leandra Boucheron Shpyrko Research Group July 19, 2012 Outline Introduction How and Why do Coffee Rings Form? Manipulating Coffee Rings Reversing the Effect Particle Shape and Size Other Factors Conclusion
A droplet of colloidal solution is left to evaporate on a superhydrophobic surface. Avijit Baidya
 A droplet of colloidal solution is left to evaporate on a superhydrophobic surface. Avijit Baidya 14.03.15 In this paper Evaporation-driven particle self-assembly can be used to generate three-dimensional
A droplet of colloidal solution is left to evaporate on a superhydrophobic surface. Avijit Baidya 14.03.15 In this paper Evaporation-driven particle self-assembly can be used to generate three-dimensional
CHAPTER TWO: EXPERIMENTAL AND INSTRUMENTATION TECHNIQUES
 CHAPTER TWO: EXPERIMENTAL AND INSTRUMENTATION TECHNIQUES 25 2.1 INSTRUMENTATION The prepared samples were characterized using various techniques. Among which are Dynamic Light Scattering, Zeta Potential
CHAPTER TWO: EXPERIMENTAL AND INSTRUMENTATION TECHNIQUES 25 2.1 INSTRUMENTATION The prepared samples were characterized using various techniques. Among which are Dynamic Light Scattering, Zeta Potential
Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena
 Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena Saurabh Nath and Jonathan B. Boreyko Department of Biomedical Engineering and Mechanics, Virginia
Supporting Information: On Localized Vapor Pressure Gradients Governing Condensation and Frost Phenomena Saurabh Nath and Jonathan B. Boreyko Department of Biomedical Engineering and Mechanics, Virginia
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
Supporting Information. Controlled mineralization by extracellular matrix: monodisperse, colloidal stable calcium phosphate-hyaluronan
 Supporting Information Controlled mineralization by extracellular matrix: monodisperse, colloidal stable calcium phosphate-hyaluronan hybrid nanospheres Zhenhua Chen, a Huihui Zhou, b Xiaoliang Wang, a
Supporting Information Controlled mineralization by extracellular matrix: monodisperse, colloidal stable calcium phosphate-hyaluronan hybrid nanospheres Zhenhua Chen, a Huihui Zhou, b Xiaoliang Wang, a
Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity
 Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity M. R. Barmi 1, B. D. Piorek 1, M. Moskovits 2, C. D. Meinhart 1* 1 Department of Mechanical Engineering, University
Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity M. R. Barmi 1, B. D. Piorek 1, M. Moskovits 2, C. D. Meinhart 1* 1 Department of Mechanical Engineering, University
Electronic supplementary information
 Electronic supplementary information Multi-Scale Structured, Superhydrophobic and Wide-Angle, Antireflective Coating in the Near-Infrared Region Kelly C. Camargo,, * Alexandre F. Michels, Fabiano S. Rodembusch,
Electronic supplementary information Multi-Scale Structured, Superhydrophobic and Wide-Angle, Antireflective Coating in the Near-Infrared Region Kelly C. Camargo,, * Alexandre F. Michels, Fabiano S. Rodembusch,
cavity (inner rectangular mark) in the frozen decane (dark background) is clearly visible.
 Supplementary Figure S1. Low magnification image showing one half of the sandwich holder from the top. The rectangular print left by the water droplet contained in the holder cavity (inner rectangular
Supplementary Figure S1. Low magnification image showing one half of the sandwich holder from the top. The rectangular print left by the water droplet contained in the holder cavity (inner rectangular
Supporting Information. Three-Dimensional Super-Resolution Imaging of Single Nanoparticle Delivered by Pipettes
 Supporting Information Three-Dimensional Super-Resolution Imaging of Single Nanoparticle Delivered by Pipettes Yun Yu,, Vignesh Sundaresan,, Sabyasachi Bandyopadhyay, Yulun Zhang, Martin A. Edwards, Kim
Supporting Information Three-Dimensional Super-Resolution Imaging of Single Nanoparticle Delivered by Pipettes Yun Yu,, Vignesh Sundaresan,, Sabyasachi Bandyopadhyay, Yulun Zhang, Martin A. Edwards, Kim
Evaporative Deposition Patterns Revisited: Spatial Dimensions of the Deposit
 arxiv:cond-mat/0408106v2 [cond-mat.soft] 7 Dec 2004 Evaporative Deposition Patterns Revisited: Spatial Dimensions of the Deposit Yuri O. Popov Department of Physics, University of Chicago, 5640 S. Ellis
arxiv:cond-mat/0408106v2 [cond-mat.soft] 7 Dec 2004 Evaporative Deposition Patterns Revisited: Spatial Dimensions of the Deposit Yuri O. Popov Department of Physics, University of Chicago, 5640 S. Ellis
Supplementary information
 1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
1 2 Supplementary information 3 4 5 6 Supplementary Figure 1 7 8 Supplementary Figure 1 ǀ Characterization of the lysozyme fibrils by atomic force microscopy 9 (AFM) and scanning electron microscopy (SEM).
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen
 Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Brownian diffusion of a partially wetted colloid
 SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
SUPPLEMENTARY INFORMATION DOI: 1.138/NMAT4348 Brownian diffusion of a partially wetted colloid Giuseppe Boniello, Christophe Blanc, Denys Fedorenko, Mayssa Medfai, Nadia Ben Mbarek, Martin In, Michel Gross,
Effect of Non-Ionic Surfactants on Dispersion and. Polar Interactions in the Adsorption of Cellulases. onto Lignin
 Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
Supporting Information Effect of Non-Ionic Surfactants on Dispersion and Polar Interactions in the Adsorption of Cellulases onto Lignin Feng Jiang, Chen Qian, Alan R. Esker and Maren Roman, * Macromolecules
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. Phagocytosis- inspired behaviour in synthetic protocell communities of compartmentalized colloidal objects Laura Rodríguez- Arco, Mei Li and Stephen
In the format provided by the authors and unedited. Phagocytosis- inspired behaviour in synthetic protocell communities of compartmentalized colloidal objects Laura Rodríguez- Arco, Mei Li and Stephen
Initial position, x p (0)/L
 .4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
.4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
Measuring particle aggregation rates by light scattering
 Measuring particle aggregation rates by light scattering Gregor Trefalt, Istvan Szilagyi, Michal Borkovec Email. gregor.trefalt@unige.ch, istvan.szilagyi@unige.ch, michal.borkovec@unige.ch Introduction
Measuring particle aggregation rates by light scattering Gregor Trefalt, Istvan Szilagyi, Michal Borkovec Email. gregor.trefalt@unige.ch, istvan.szilagyi@unige.ch, michal.borkovec@unige.ch Introduction
Investigations on the Evaporation of Charged Droplets on Hydrophobic Surface
 Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
Proceedings of the International Conference on Heat Transfer and Fluid Flow Prague, Czech Republic, August 11-12, 2014 Paper No. 15 Investigations on the Evaporation of Charged Droplets on Hydrophobic
Self-assembled nanostructures for antireflection optical coatings
 Self-assembled nanostructures for antireflection optical coatings Yang Zhao 1, Guangzhao Mao 2, and Jinsong Wang 1 1. Deaprtment of Electrical and Computer Engineering 2. Departmentof Chemical Engineering
Self-assembled nanostructures for antireflection optical coatings Yang Zhao 1, Guangzhao Mao 2, and Jinsong Wang 1 1. Deaprtment of Electrical and Computer Engineering 2. Departmentof Chemical Engineering
Formation of Two-Dimensional Colloidal Sphere Arrays on Micro-Patterns
 Formation of Two-Dimensional Colloidal Sphere Arrays on Micro-Patterns Neil A. Bernotski 1, Xiaorong Xiong 2, Kerwin Wang 3, Nels E. Jewell-Larsen 4, and Karl F. Böhringer 5 Department of Electrical Engineering,
Formation of Two-Dimensional Colloidal Sphere Arrays on Micro-Patterns Neil A. Bernotski 1, Xiaorong Xiong 2, Kerwin Wang 3, Nels E. Jewell-Larsen 4, and Karl F. Böhringer 5 Department of Electrical Engineering,
Reaction at the Interfaces
 Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Reaction at the Interfaces Lecture 1 On the course Physics and Chemistry of Interfaces by HansJürgen Butt, Karlheinz Graf, and Michael Kappl Wiley VCH; 2nd edition (2006) http://homes.nano.aau.dk/lg/surface2009.htm
Supporting Information: Mass Spectrometric Sampling of. a Liquid Surface by Nanoliter Droplet Generation from
 Supporting Information: Mass Spectrometric Sampling of a Liquid Surface by Nanoliter Droplet Generation from Bursting Bubbles and Focused Acoustic Pulses: Application to Studies of Interfacial Chemistry
Supporting Information: Mass Spectrometric Sampling of a Liquid Surface by Nanoliter Droplet Generation from Bursting Bubbles and Focused Acoustic Pulses: Application to Studies of Interfacial Chemistry
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Laure Fabié, Hugo Durou, Thierry Ondarçuhu Nanosciences Group, CEMES CNRS Toulouse (France)
 Nanodroplet deposition and manipulation with an AFM tip Laure Fabié, Hugo Durou, Thierry Ondarçuhu Nanosciences Group, CEMES CNRS Toulouse (France) Direct deposition methods Liquid lithography Pin and
Nanodroplet deposition and manipulation with an AFM tip Laure Fabié, Hugo Durou, Thierry Ondarçuhu Nanosciences Group, CEMES CNRS Toulouse (France) Direct deposition methods Liquid lithography Pin and
Supporting Information
 Supporting Information Wu et al. 1.173/pnas.15492112 SI Text S1) Derivation of the Wind-Chime Model Each pulse generates an angular momentum on each MoS 2 flake that makes them rotate unless they develop
Supporting Information Wu et al. 1.173/pnas.15492112 SI Text S1) Derivation of the Wind-Chime Model Each pulse generates an angular momentum on each MoS 2 flake that makes them rotate unless they develop
Supplementary table I. Table of contact angles of the different solutions on the surfaces used here. Supplementary Notes
 1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
Supplementary Information. Synthesis of soft colloids with well controlled softness
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information Synthesis of soft colloids with well controlled softness Fuhua Luo, Zhifeng
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Supplementary Information Synthesis of soft colloids with well controlled softness Fuhua Luo, Zhifeng
COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS
 18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
18 TH INTERNATIONAL CONFERENCE ON COMPOSITE MATERIALS COMPARISON OF WETTABILITY AND CAPILLARY EFFECT EVALUATED BY DIFFERENT CHARACTERIZING METHODS S.K. Wang*, M. Li*, Y.Z. Gu, Y.X. Li and Z.G. Zhang Key
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Interfaces and interfacial energy
 Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
Interfaces and interfacial energy 1/14 kinds: l/g }{{ l/l } mobile s/g s/l s/s Example. Estimate the percetage of water molecules on the surface of a fog droplet of diameter (i) 0.1 mm (naked eye visibility
Jahresbericht 2003 der Arbeitsgruppe Experimentalphysik Prof. Dr. Michael Farle
 Self-assembly of Fe x Pt 1-x nanoparticles. M. Ulmeanu, B. Stahlmecke, H. Zähres and M. Farle Institut für Physik, Universität Duisburg-Essen, Lotharstr. 1, 47048 Duisburg Future magnetic storage media
Self-assembly of Fe x Pt 1-x nanoparticles. M. Ulmeanu, B. Stahlmecke, H. Zähres and M. Farle Institut für Physik, Universität Duisburg-Essen, Lotharstr. 1, 47048 Duisburg Future magnetic storage media
spreading of drops on soft surfaces
 Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supporting Information
 Supporting Information Clustered Ribbed-Nanoneedle Structured Copper Surfaces with High- Efficiency Dropwise Condensation Heat Transfer Performance Jie Zhu, Yuting Luo, Jian Tian, Juan Li and Xuefeng Gao*
Supporting Information Clustered Ribbed-Nanoneedle Structured Copper Surfaces with High- Efficiency Dropwise Condensation Heat Transfer Performance Jie Zhu, Yuting Luo, Jian Tian, Juan Li and Xuefeng Gao*
Evaporative deposition patterns: Spatial dimensions of the deposit
 Evaporative deposition patterns: Spatial dimensions of the deposit Yuri O. Popov* Department of Physics, University of Chicago, 5640 S. Ellis Avenue, Chicago, Illinois 60637, USA Received 4 August 2004;
Evaporative deposition patterns: Spatial dimensions of the deposit Yuri O. Popov* Department of Physics, University of Chicago, 5640 S. Ellis Avenue, Chicago, Illinois 60637, USA Received 4 August 2004;
A Hydrophilic/Hydrophobic Janus Inverse-Opal
 Supporting information A Hydrophilic/Hydrophobic Janus Inverse-Opal Actuator via Gradient Infiltration Dajie Zhang #, Jie Liu //#, Bo Chen *, Yong Zhao, Jingxia Wang * //, Tomiki Ikeda, Lei Jiang //. CAS
Supporting information A Hydrophilic/Hydrophobic Janus Inverse-Opal Actuator via Gradient Infiltration Dajie Zhang #, Jie Liu //#, Bo Chen *, Yong Zhao, Jingxia Wang * //, Tomiki Ikeda, Lei Jiang //. CAS
Introduction to Dynamic Light Scattering for Particle Size Determination
 www.horiba.com/us/particle Jeffrey Bodycomb, Ph.D. Introduction to Dynamic Light Scattering for Particle Size Determination 2016 HORIBA, Ltd. All rights reserved. 1 Sizing Techniques 0.001 0.01 0.1 1 10
www.horiba.com/us/particle Jeffrey Bodycomb, Ph.D. Introduction to Dynamic Light Scattering for Particle Size Determination 2016 HORIBA, Ltd. All rights reserved. 1 Sizing Techniques 0.001 0.01 0.1 1 10
Bioassay on a Robust and Stretchable Extreme Wetting. Substrate through Vacuum-Based Droplet Manipulation
 Supporting Information for A Single-Droplet Multiplex Bioassay on a Robust and Stretchable Extreme Wetting Substrate through Vacuum-Based Droplet Manipulation Heetak Han, Jung Seung Lee, Hyunchul Kim,
Supporting Information for A Single-Droplet Multiplex Bioassay on a Robust and Stretchable Extreme Wetting Substrate through Vacuum-Based Droplet Manipulation Heetak Han, Jung Seung Lee, Hyunchul Kim,
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Supporting Information. Railing Cells along 3D Microelectrode Tracks for a. Continuous-Flow Dielectrophoretic Sorting
 Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 2018 Supporting Information Railing Cells along 3D Microelectrode Tracks for a Continuous-Flow
Electronic Supplementary Material (ESI) for Lab on a Chip. This journal is The Royal Society of Chemistry 2018 Supporting Information Railing Cells along 3D Microelectrode Tracks for a Continuous-Flow
Clouds associated with cold and warm fronts. Whiteman (2000)
 Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Clouds associated with cold and warm fronts Whiteman (2000) Dalton s law of partial pressures! The total pressure exerted by a mixture of gases equals the sum of the partial pressure of the gases! Partial
Basic Laboratory. Materials Science and Engineering. Atomic Force Microscopy (AFM)
 Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Basic Laboratory Materials Science and Engineering Atomic Force Microscopy (AFM) M108 Stand: 20.10.2015 Aim: Presentation of an application of the AFM for studying surface morphology. Inhalt 1.Introduction...
Collision and Oxidation of Silver Nanoparticles on a Gold Nanoband Electrode
 Supporting Information for: Collision and Oxidation of Silver Nanoparticles on a Gold Nanoband Electrode Fan Zhang, Martin A. Edwards, Rui Hao, Henry S. White, * and Bo Zhang * Department of Chemistry,
Supporting Information for: Collision and Oxidation of Silver Nanoparticles on a Gold Nanoband Electrode Fan Zhang, Martin A. Edwards, Rui Hao, Henry S. White, * and Bo Zhang * Department of Chemistry,
When a drop of a colloidal solution of nanoparticles
 Kinetically driven self assembly of highly ordered nanoparticle monolayers LETTERS TERRY P. BIGIONI, XIAO-MIN LIN 2,TOANT.NGUYEN,ERICI.CORWIN,3, THOMAS A. WITTEN,3 AND HEINRICH M. JAEGER,3 * James Franck
Kinetically driven self assembly of highly ordered nanoparticle monolayers LETTERS TERRY P. BIGIONI, XIAO-MIN LIN 2,TOANT.NGUYEN,ERICI.CORWIN,3, THOMAS A. WITTEN,3 AND HEINRICH M. JAEGER,3 * James Franck
Fabrication of ordered array at a nanoscopic level: context
 Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Fabrication of ordered array at a nanoscopic level: context Top-down method Bottom-up method Classical lithography techniques Fast processes Size limitations it ti E-beam techniques Small sizes Slow processes
Drop friction on liquid-infused materials
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 207 Drop friction on liquid-infused materials Armelle Gas,2, Ludovic Keiser,3, Christophe Clanet,2
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 207 Drop friction on liquid-infused materials Armelle Gas,2, Ludovic Keiser,3, Christophe Clanet,2
Supporting information
 Electronic Supplementary Material (ESI) for anoscale. This journal is The Royal Society of Chemistry 2014 Supporting information On-demand shape and size purification of nanoparticle based on surface area
Electronic Supplementary Material (ESI) for anoscale. This journal is The Royal Society of Chemistry 2014 Supporting information On-demand shape and size purification of nanoparticle based on surface area
Rate of change of velocity. a=dv/dt. Acceleration is a vector quantity.
 9.7 CENTRIFUGATION The centrifuge is a widely used instrument in clinical laboratories for the separation of components. Various quantities are used for the description and the calculation of the separation
9.7 CENTRIFUGATION The centrifuge is a widely used instrument in clinical laboratories for the separation of components. Various quantities are used for the description and the calculation of the separation
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/10/eaao3530/dc1 Supplementary Materials for Topological liquid diode Jiaqian Li, Xiaofeng Zhou, Jing Li, Lufeng Che, Jun Yao, Glen McHale, Manoj K. Chaudhury,
advances.sciencemag.org/cgi/content/full/3/10/eaao3530/dc1 Supplementary Materials for Topological liquid diode Jiaqian Li, Xiaofeng Zhou, Jing Li, Lufeng Che, Jun Yao, Glen McHale, Manoj K. Chaudhury,
Fast Nucleation for Silica Nanoparticle Synthesis in. Sol-Gel Method
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Fast Nucleation for lica Nanoparticle Synthesis in Sol-Gel Method Chandra K. Dixit*, Snehasis
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Fast Nucleation for lica Nanoparticle Synthesis in Sol-Gel Method Chandra K. Dixit*, Snehasis
2. Modeling of shrinkage during first drying period
 2. Modeling of shrinkage during first drying period In this chapter we propose and develop a mathematical model of to describe nonuniform shrinkage of porous medium during drying starting with several
2. Modeling of shrinkage during first drying period In this chapter we propose and develop a mathematical model of to describe nonuniform shrinkage of porous medium during drying starting with several
SUPPORTING INFORMATION
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 SUPPORTING INFORMATION Materials Graphite powder (SP-1 graphite) was obtained from Bay carbon.
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 SUPPORTING INFORMATION Materials Graphite powder (SP-1 graphite) was obtained from Bay carbon.
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Step 1. Step 2. g l = g v. dg = 0 We have shown that over a plane surface of water. g v g l = ρ v R v T ln e/e sat. this can be rewritten
 The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
The basic question is what makes the existence of a droplet thermodynamically preferable to the existence only of water vapor. We have already derived an expression for the saturation vapor pressure over
Supporting Information
 Supporting Information Wiley-VCH 2013 69451 Weinheim, Germany Colloidal Clusters by Using Emulsions and Dumbbell-Shaped Particles: Experiments and Simulations** Bo Peng,* Frank Smallenburg,* Arnout Imhof,
Supporting Information Wiley-VCH 2013 69451 Weinheim, Germany Colloidal Clusters by Using Emulsions and Dumbbell-Shaped Particles: Experiments and Simulations** Bo Peng,* Frank Smallenburg,* Arnout Imhof,
Gold-poly(N-isopropylacrylamide) core-shell colloids with homogeneous density profiles: A small angle scattering study
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information Gold-poly(N-isopropylacrylamide) core-shell colloids with
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Supporting Information Gold-poly(N-isopropylacrylamide) core-shell colloids with
Shape of the Interfaces
 NPTEL Chemical Engineering Interfacial Engineering Module : Lecture 3 Shape of the Interfaces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039 India
NPTEL Chemical Engineering Interfacial Engineering Module : Lecture 3 Shape of the Interfaces Dr. Pallab Ghosh Associate Professor Department of Chemical Engineering IIT Guwahati, Guwahati 781039 India
Magnetic nanoparticles-induced anisotropic shrinkage of polymer emulsion droplets. Table of contents
 Electronic Supplemental Information Magnetic nanoparticles-induced anisotropic shrinkage of polymer emulsion droplets Bing Liu, * a Julius W.J. de Folter b and Helmuth Möhwald a a Department of Interfaces,
Electronic Supplemental Information Magnetic nanoparticles-induced anisotropic shrinkage of polymer emulsion droplets Bing Liu, * a Julius W.J. de Folter b and Helmuth Möhwald a a Department of Interfaces,
Vibration of submillimeter-size supported droplets
 PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
3D Micropatterned Surface Inspired by Salvinia
 Supporting information 3D Micropatterned Surface Inspired by Salvinia molesta via Direct Laser Lithography. Omar Tricinci*,, Tercio Terencio,#, Barbara Mazzolai, Nicola M. Pugno,,, Francesco Greco*,, Virgilio
Supporting information 3D Micropatterned Surface Inspired by Salvinia molesta via Direct Laser Lithography. Omar Tricinci*,, Tercio Terencio,#, Barbara Mazzolai, Nicola M. Pugno,,, Francesco Greco*,, Virgilio
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
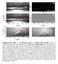 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Critical Micellization Concentration Determination using Surface Tension Phenomenon
 Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Supporting Information. by Hexagonal Boron Nitride
 Supporting Information High Velocity Saturation in Graphene Encapsulated by Hexagonal Boron Nitride Megan A. Yamoah 1,2,, Wenmin Yang 1,3, Eric Pop 4,5,6, David Goldhaber-Gordon 1 * 1 Department of Physics,
Supporting Information High Velocity Saturation in Graphene Encapsulated by Hexagonal Boron Nitride Megan A. Yamoah 1,2,, Wenmin Yang 1,3, Eric Pop 4,5,6, David Goldhaber-Gordon 1 * 1 Department of Physics,
Digital Holographic Measurement of Nanometric Optical Excitation on Soft Matter by Optical Pressure and Photothermal Interactions
 Ph.D. Dissertation Defense September 5, 2012 Digital Holographic Measurement of Nanometric Optical Excitation on Soft Matter by Optical Pressure and Photothermal Interactions David C. Clark Digital Holography
Ph.D. Dissertation Defense September 5, 2012 Digital Holographic Measurement of Nanometric Optical Excitation on Soft Matter by Optical Pressure and Photothermal Interactions David C. Clark Digital Holography
Application of Micro-Flow Imaging (MFI TM ) to The Analysis of Particles in Parenteral Fluids. October 2006 Ottawa, Canada
 Application of Micro-Flow Imaging (MFI TM ) to The Analysis of Particles in Parenteral Fluids October 26 Ottawa, Canada Summary The introduction of a growing number of targeted protein-based drug formulations
Application of Micro-Flow Imaging (MFI TM ) to The Analysis of Particles in Parenteral Fluids October 26 Ottawa, Canada Summary The introduction of a growing number of targeted protein-based drug formulations
Part I.
 Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Topography driven spreading. School of Biomedical & Natural Sciences, Nottingham Trent University. Clifton Lane, Nottingham NG11 8NS, UK.
 Postprint Version G. McHale, N. J. Shirtcliffe, S. Aqil, C. C. Perry and M. I. Newton, Topography driven spreading, Phys. Rev. Lett. 93, Art. No. 036102 (2004); DOI: 10.1103/PhysRevLett.93.036102. The
Postprint Version G. McHale, N. J. Shirtcliffe, S. Aqil, C. C. Perry and M. I. Newton, Topography driven spreading, Phys. Rev. Lett. 93, Art. No. 036102 (2004); DOI: 10.1103/PhysRevLett.93.036102. The
8.6 Drag Forces in Fluids
 86 Drag Forces in Fluids When a solid object moves through a fluid it will experience a resistive force, called the drag force, opposing its motion The fluid may be a liquid or a gas This force is a very
86 Drag Forces in Fluids When a solid object moves through a fluid it will experience a resistive force, called the drag force, opposing its motion The fluid may be a liquid or a gas This force is a very
Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets
 Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets Qiaolan Zhang, a,b Min He, a Jing Chen, a,b Jianjun Wang,* a Yanlin Song* a and Lei Jiang a a Beijing National
Anti-icing surfaces based on enhanced self-propelled jumping of condensed water microdroplets Qiaolan Zhang, a,b Min He, a Jing Chen, a,b Jianjun Wang,* a Yanlin Song* a and Lei Jiang a a Beijing National
Carbon nanotube coated snowman-like particles and their electro-responsive characteristics. Ke Zhang, Ying Dan Liu and Hyoung Jin Choi
 Supporting Information: Carbon nanotube coated snowman-like particles and their electro-responsive characteristics Ke Zhang, Ying Dan Liu and Hyoung Jin Choi Experimental Section 1.1 Materials The MWNT
Supporting Information: Carbon nanotube coated snowman-like particles and their electro-responsive characteristics Ke Zhang, Ying Dan Liu and Hyoung Jin Choi Experimental Section 1.1 Materials The MWNT
Mapping the mechanical stiffness of live cells with the scanning ion conductance microscope
 SUPPLEMENTARY INFORMATION Mapping the mechanical stiffness of live cells with the scanning ion conductance microscope Johannes Rheinlaender and Tilman E. Schäffer Supplementary Figure S1 Supplementary
SUPPLEMENTARY INFORMATION Mapping the mechanical stiffness of live cells with the scanning ion conductance microscope Johannes Rheinlaender and Tilman E. Schäffer Supplementary Figure S1 Supplementary
Surface Tension and its measurements
 Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
Surface Tension and its measurements Surface Tension Surface tension is a fundamental property by which the gas liquid interfaces are characterized. The zone between a gaseous phase and a liquid phase
New Methods for Measuring Water Desorption and Vapour Permeation Rates in Membranes
 New Methods for Measuring Water Desorption and Vapour Permeation Rates in Membranes L. I. iortea, D. O Driscoll, E. P. Berg, P. Xiao, F.. Pascut and R. E. Imhof School of Engineering, South Bank University,
New Methods for Measuring Water Desorption and Vapour Permeation Rates in Membranes L. I. iortea, D. O Driscoll, E. P. Berg, P. Xiao, F.. Pascut and R. E. Imhof School of Engineering, South Bank University,
SUPPLEMENTAL MATERIAL I: SEM IMAGE OF PHOTONIC CRYSTAL RESONATOR
 1 SUPPLEMENTAL MATERIAL I: SEM IMAGE OF PHOTONIC CRYSTAL RESONATOR Figure S1 below is a scanning electronic microscopy image of a typical evanescently coupled photonic crystal resonator used in these experiments.
1 SUPPLEMENTAL MATERIAL I: SEM IMAGE OF PHOTONIC CRYSTAL RESONATOR Figure S1 below is a scanning electronic microscopy image of a typical evanescently coupled photonic crystal resonator used in these experiments.
2. Determine the surface tension of water with the capillary-rise method.
 Fakultät für Physik und Geowissenschaften Physikalisches Grundpraktikum M19e Surface Tension Tasks 1. Determine the surface tension σ of an organic liquid using the anchor-ring method. Use three different
Fakultät für Physik und Geowissenschaften Physikalisches Grundpraktikum M19e Surface Tension Tasks 1. Determine the surface tension σ of an organic liquid using the anchor-ring method. Use three different
Particles, drops, and bubbles. Lecture 3
 Particles, drops, and bubbles Lecture 3 Brownian Motion is diffusion The Einstein relation between particle size and its diffusion coefficient is: D = kt 6πηa However gravitational sedimentation tends
Particles, drops, and bubbles Lecture 3 Brownian Motion is diffusion The Einstein relation between particle size and its diffusion coefficient is: D = kt 6πηa However gravitational sedimentation tends
SUPPORTING INFORMATION. Hierarchical Self-Assembly of Gold Nanoparticles into Patterned Plasmonic Nanostructures
 SUPPORTING INFORMATION Hierarchical Self-Assembly of Gold Nanoparticles into Patterned Plasmonic Nanostructures Cyrille Hamon 1, Sergey Novikov 1, Leonardo Scarabelli 1, Lourdes Basabe-Desmonts 2,3 Luis
SUPPORTING INFORMATION Hierarchical Self-Assembly of Gold Nanoparticles into Patterned Plasmonic Nanostructures Cyrille Hamon 1, Sergey Novikov 1, Leonardo Scarabelli 1, Lourdes Basabe-Desmonts 2,3 Luis
Courtesy: Images on the internet
 Courtesy: Images on the internet Focus: Physics of Blowing Bubbles February 19, 2016 Physics 9, 21 Using a bubble-blowing apparatus, researchers developed a model that explains the effects of several factors,
Courtesy: Images on the internet Focus: Physics of Blowing Bubbles February 19, 2016 Physics 9, 21 Using a bubble-blowing apparatus, researchers developed a model that explains the effects of several factors,
Supporting Information
 Supporting Information Visible Light-Driven BiOI-Based Janus Micromotors in Pure Water Renfeng Dong, a Yan Hu, b Yefei Wu, b Wei Gao, c Biye Ren, b* Qinglong Wang, a Yuepeng Cai a* a School of Chemistry
Supporting Information Visible Light-Driven BiOI-Based Janus Micromotors in Pure Water Renfeng Dong, a Yan Hu, b Yefei Wu, b Wei Gao, c Biye Ren, b* Qinglong Wang, a Yuepeng Cai a* a School of Chemistry
Supporting Information
 Supporting Information High-Efficiency Fog Collector: Water Unidirectional Transport on Heterogeneous Rough Conical Wires Ting Xu, Yucai Lin, Miaoxin Zhang, Weiwei Shi, Yongmei Zheng* Key Laboratory of
Supporting Information High-Efficiency Fog Collector: Water Unidirectional Transport on Heterogeneous Rough Conical Wires Ting Xu, Yucai Lin, Miaoxin Zhang, Weiwei Shi, Yongmei Zheng* Key Laboratory of
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10447 Supplementary Methods Materials. The lubricating fluids used for the experiment were perfluorinated fluids (e.g., 3M Fluorinert FC-70, Dupont Krytox 100 and 103). Unless otherwise
doi:10.1038/nature10447 Supplementary Methods Materials. The lubricating fluids used for the experiment were perfluorinated fluids (e.g., 3M Fluorinert FC-70, Dupont Krytox 100 and 103). Unless otherwise
LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE
 Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
Supplementary Figures
 Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
Supplementary Figures 1 Supplementary Figure 1 Micro and nano-textured boiling surfaces. (a) A schematic of the textured boiling surfaces. (b) An isometric view of the square array of square micropillars.
AGITATION AND AERATION
 AGITATION AND AERATION Although in many aerobic cultures, gas sparging provides the method for both mixing and aeration - it is important that these two aspects of fermenter design be considered separately.
AGITATION AND AERATION Although in many aerobic cultures, gas sparging provides the method for both mixing and aeration - it is important that these two aspects of fermenter design be considered separately.
Atomic Force Microscopy Characterization of Room- Temperature Adlayers of Small Organic Molecules through Graphene Templating
 Atomic Force icroscopy Characterization of Room- Temperature Adlayers of Small Organic olecules through Graphene Templating Peigen Cao, Ke Xu,2, Joseph O. Varghese, and James R. Heath *. Kavli Nanoscience
Atomic Force icroscopy Characterization of Room- Temperature Adlayers of Small Organic olecules through Graphene Templating Peigen Cao, Ke Xu,2, Joseph O. Varghese, and James R. Heath *. Kavli Nanoscience
3D Motion of DNA-Au Nanoconjugates in Graphene Liquid Cell EM
 Supporting Information for 3D Motion of DNA-Au Nanoconjugates in Graphene Liquid Cell EM Qian Chen,,, Jessica Smith,, Jungwon Park,,1, Kwanpyo Kim,,2, Davy Ho, Haider I. Rasool,,!Alex Zettl,, A. Paul Alivisatos,,*!
Supporting Information for 3D Motion of DNA-Au Nanoconjugates in Graphene Liquid Cell EM Qian Chen,,, Jessica Smith,, Jungwon Park,,1, Kwanpyo Kim,,2, Davy Ho, Haider I. Rasool,,!Alex Zettl,, A. Paul Alivisatos,,*!
Experimental Study on the Effective Thermal Conductivity and Thermal Diffusivity of Nanofluids
 International Journal of Thermophysics, Vol. 27, No. 2, March 2006 ( 2006) DOI: 10.1007/s10765-006-0054-1 Experimental Study on the Effective Thermal Conductivity and Thermal Diffusivity of Nanofluids
International Journal of Thermophysics, Vol. 27, No. 2, March 2006 ( 2006) DOI: 10.1007/s10765-006-0054-1 Experimental Study on the Effective Thermal Conductivity and Thermal Diffusivity of Nanofluids
BAE 820 Physical Principles of Environmental Systems
 BAE 820 Physical Principles of Environmental Systems Estimation of diffusion Coefficient Dr. Zifei Liu Diffusion mass transfer Diffusion mass transfer refers to mass in transit due to a species concentration
BAE 820 Physical Principles of Environmental Systems Estimation of diffusion Coefficient Dr. Zifei Liu Diffusion mass transfer Diffusion mass transfer refers to mass in transit due to a species concentration
Kelvin Effect. Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics
 Kelvin Effect Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Vapor Pressure (e) e < e # e = e # Vapor Pressure e > e # Relative humidity RH =
Kelvin Effect Covers Reading Material in Chapter 10.3 Atmospheric Sciences 5200 Physical Meteorology III: Cloud Physics Vapor Pressure (e) e < e # e = e # Vapor Pressure e > e # Relative humidity RH =
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading
 Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
Loughborough University Institutional Repository Surface forces action in a vicinity of three phase contact line and other current problems in kinetics of wetting and spreading This item was submitted
