Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila
|
|
|
- Kevin Ross
- 5 years ago
- Views:
Transcription
1 Hereditas 147: (2010) Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila ANJA C. NAGEL, JENS SCHMID, JASMIN S. AUER, ANETTE PREISS and DIETER MAIER Institut für Genetik (240), Universität Hohenheim, Stuttgart, Germany Nagel, A. C., Schmid, J., Auer, J. S., Preiss, A. and Maier, D Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila. Hereditas 147 : Lund, Sweden. eissn Received 16 July Accepted 27 August The mammalian protein kinase D family is involved in manifold cellular processes including cell migration and motility. Recently it was shown that human PKD1 and PKD2 phosphorylate and thereby inhibit Slingshot 1 Like (SSH1L), a phosphatase which is central to the regulation of actin cytoskeletal dynamics. We noted before that the overexpression of a constitutively active form of Drosophila PKD (PKD-SE) affects the fly retina and the resultant phenotypes suggest underlying defects in the actin cytoskeleton. Drosophila Slingshot, however, does not possess the phosphorylation site known to be targeted in SSH1L by human PKD1. Here we show that Drosophila PKD, despite this lack of conservation, nevertheless negatively regulates Slingshot. Overexpression of the active PKD-SE protein causes cellular defects that are similar to those of slingshot mutants. These include aberrant bristle morphology and positioning of photoreceptor nuclei. Interestingly, the observed nuclear mispositioning is due to a disturbance of the cytoskeleton rather than the epithelial organization. In accordance, overexpression of PKD-SE results in an accumulation of filamentous actin. This enrichment is modified by changes in slingshot gene doses, in line with an antagonistic relationship between PKD and slingshot. We conclude that similar to mammals, Drosophila PKD is a negative regulator of Ssh, with the premise of a different target phosphorylation site in Ssh. Dieter Maier, Institut f ü r Genetik (240), Universit ä t Hohenheim, Garbenstr. 30, DE Stuttgart, Germany. dieter. maier@uni-hohenheim.de The actin cytoskeleton is a highly dynamic structure that lies at the basis of cell shape and motility. There are several well documented examples that changes in cell shape provide the driving force for morphogenesis. These include the morphogenetic movements during gastrulation and the dorsal closure of the Drosophila embryo. Regular structures like the microvilli stacks within rhabdomeres of the fly retina are built up with the help of actin filaments. Moreover, actin filament based structures like bristles or fine hairs of the fly, which serve as mechano-sensors, can be extremely elaborate and stable structures (reviewed by JACINTO and B AUM 2003). The principle players that regulate actin dynamics have been identified in the past (reviewed by POLLARD and BORISY 2003). In particular, the actin depolymerising factor Cofilin promotes filament disassembly and creates new ends for elongation, thus setting the direction of cell movement (overview by DESMARAIS et al. 2005). Cofilin is inhibited by two families of kinases (LIMK1, 2 and TESK1, 2), which phosphorylate Cofilin on serine 3, thereby abolishing its actin-binding property. Notably the family of Slingshot (Ssh) phosphatases reactivates phospho-cofilin by dephosphorylation ( NIWA et al. 2002; overview by DESMARAIS et al. 2005). In human cells Slingshot 1 Like (SSH1L) phosphatase activity is dependent on actin binding ( NAGATA-OHASHI et al. 2004). However, SSH1L phosphorylated at Ser-937 and Ser-978 is bound by scaffolding proteins at these residues, thereby sequestering it from the actin/cofilin complex in the cytoplasm ( NAGATA- OHASHI et al. 2004). Recently it was shown that this C- terminal phosphorylation of SSH1L is dependent on protein kinase D 1 and 2 (PKD1, PKD2) ( EISELER et al. 2009; P ETERBURS et al. 2009). Hence, human PKD1 and PKD2 act as a negative regulator on the reactivation of the Slingshot- Cofilin signaling network which regulates actin dynamics in the cell. Albeit very similar in structure and function, mammalian SSHL phosphatases differ from Drosophila Slingshot (Ssh) in their C-terminal end ( NIWA et al. 2002; OHTA et al. 2003). Hence, Drosophila Ssh does not contain the sequence recognition site that was identified in SSH1L as target of human PKD. However, we have noted before that manipulation of Drosophila PKD activity affected the development of different organs in a way that conforms to a role of PKD in the regulation of actin dynamics in Drosophila as well ( MAIER et al. 2006, 2007). In this work we set out to determine the mode of action of Drosophila PKD on actin dynamics. We find that overexpression of a constitutively active PKD isoform, PKD-SE causes phenotypes very similar to those seen in ssh mutants. These include aberrant bristle morphology and positioning of photoreceptor nuclei. The latter phenotype is due to a disturbance of the actin cytoskeleton rather than a general defect in epithelial architecture. At the subcellular level, we note a strong enrichment of 2010 The Authors. This is an Open Access article. DOI: /j x
2 238 A. C. Nagel et al. Hereditas 147 (2010) filamentous actin as a consequence of either loss of ssh or gain of PKD-SE. Moreover we show, that there is an antagonistic genetic relationship between ssh and PKD. In summary, Drosophila PKD may act as a negative regulator of Ssh in the fly as well, suggesting that it targets a different phosphorylation site in Ssh than the ones identified earlier in human SSH1L. MATERIAL AND METHODS Fly stocks and clonal analysis The following fly stocks were used: UAS-PKD-SE GFP ( MAIER et al. 2007); FRT82B ssh 1-63 / TM3 Ser; UASssh.N 30, UAS-ssh.CS 11 (NIWA et al. 2002); y w, FRT82B ubi-gfp /TM6B; y w hs-flp; act CD2 Gal4, UAS- GFPnls / TM6B ( STRUHL and BASLER 1993; kindly obtained from K. Basler); UAS-lacZ, gmr-gal4; y 1 w 67c23. Flies were obtained from the Bloomington fly collection if not noted otherwise. Clones lacking ssh function were generated with the FLP/FRT system ( XU and RUBIN 1993) using flies of the genotype y w hs-flp; FRT82B ssh 1-63 / FRT82B Ubi-GFP. Homozygous mutant cell clones were recognized by the lack of GFP expression. Clones overexpressing PKD-SE- GFP were generated as described before ( STRUHL and B ASLER 1993; PROTZER et al. 2008) in animals of the genotype y w hs-flp, UAS-PKD-SE GFP / act CD2 Gal4, UAS-GFPnls. Overexpression clones were recognized by the expression of GFP. A given UAS-line was overexpressed in the developing eye by combining it with the gmr-gal4 driver line ( BRAND and PERRIMON 1993); UASlacZ served as control. For clonal analysis in larval or early pupal tissues, a single heat shock of 30 min was applied during second larval instar. To observe clones in adult animals, three heat shocks were given in 48 h intervals starting from late first instar larval stage. Animals were allowed to develop further into late larval or early pupal stage or to adulthood before tissues were dissected, stained by help of appropriate antibodies and mounted for microscopy. Immunohistochemistry, phenotypic analyses and imaging Larval and pupal tissues were dissected and stained with antibodies as described before ( MAIER et al. 1999, 2007). Monoclonal antibodies directed against Elav, Dlg and Arm, developed by G. M. Rubin, C. Goodman and E. Wieschaus, respectively, were obtained from Developmental Studies Hybridoma Bank developed under the auspices of the NICHD and maintained by the Univ. of Iowa (Dept of Biological Sciences, Iowa City, IA 52242). Rhodamin coupled phalloidin was purchased from Invitrogen and secondary Cy3-coupled antibodies from Jackson Laboratory (Dianova, Hamburg). Fluorescently labelled tissues were mounted in Vectashield (Vector Laboratories) and analyzed on a Zeiss Axiophot linked to a Bio-Rad MRC1024 confocal microscope. Pictures were captured using BioRad Laser Sharp 3.1 software. Adult specimens were mounted in Hoyers medium (VAN DER MEER 1977) and analyzed as using a Zeiss Axioskop light microscope. Pictures were taken with a Pixera Camera (Optronics) and captured using the Viewfinder 2.0 software. Pictures were compiled using Corel Photo Paint and Corel Draw software. RESULTS Drosophila PKD-SE induces phenotypes comparable to the loss of Slingshot phosphatase We had noted before that overexpression of a constitutively active PKD isoform PKD-SE caused a variety of defects in the fly s retina that may have their origin in a disturbance of the actin cytoskeleton ( MAIER et al. 2007). In the course of our further examinations we observed a peculiar feature within the developing retina of larvae overexpressing PKD- SE: the nuclei of the photoreceptor cells that are normally localized at the apical side of the cell were mislocalized to a more basal position. We knew from the literature that this phenotype also occurs in photoreceptor cells lacking the phosphatase Slingshot (Ssh) ( ROGERS et al. 2005). Indeed, clones of photoreceptor cells either lacking Ssh or overexpressing the active PKD-SE isoform are characterized by a more basal position of their nuclei in comparison to their wild type sister cells (Fig. 1a d). To further compare the effects of PKD-SE overexpression and loss of ssh, we induced clones throughout development and noted a number of phenotypes affecting primarily bristle formation: mutation of ssh caused a characteristic bending, thickening and splitting of the bristles (Fig. 1g h) compared to the wild type (Fig. 1e). The latter phenotype is name giving to the mutant and arises from the failure to form correct actin bundles ( NIWA et al. 2002). Overexpression of PKD-SE resulted in similar bristle forms, albeit never as extreme (Fig. 1f). Although such bristle defects may be caused in several ways, the similarity of phenotypes is in agreement with a negative regulation of Ssh by PKD. In addition, PKD-SE clones showed conspicuous dark patches that were not seen in ssh mutant clones. The dark patches may result from secretion defects as a consequence of PKD-SE overexpression (Fig. 1f; MAIER et al. 2007). Ectopic expression of PKD-SE and loss of ssh cause an enrichment of F-actin but do not affect the overall epithelial organization The similarity between overexpression of PKD-SE and ssh mutants with regard to nuclear positioning in
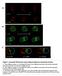
3 Hereditas 147 (2010) Drosophila PKD inhibits Ssh phosphatase 239 Fig. 1. Gain of PKD-SE and loss of slingshot cause similar phenotypes. (a d ) Apical (a, c) and basal (b, d) view of eye imaginal discs that were stained with anti-elav antibodies (red) to label the nuclei of the photoreceptor cells. Cell clones were induced that either overexpress PKD-SE (marked by GFP, green in a, a and b, b ) or that lack Ssh (marked by the absence of green GFP in c, c and d, d ). Note that in either cell clone, the nuclei of the developing photoreceptor cells have a more basal position than the surrounding wild type cells. (e) A wild type bristle is composed of a socket (so) and a long shaft (sh) that is regular and slender with a slight curvature, ending in a fine tip. (f) Overexpession of PKD-SE in small cell clones induces bristle abnormalities. Frequently, the shaft is bent or curved more strongly than normal (small arrows). Bristle shafts appear uneven because the centre is thicker (open arrow). The bristles appear brittle (arrowhead) and are frequently absent. In addition, PKD-SE cell clones show dark cuticle patches (also on the wing, not shown), that appear to result from superfluous cuticle (large arrow). (g) A strong ssh mutant bristle phenotype is characterized by failure of building normal actin bundles resulting in bents of the bristle shaft (sh). (h) Cell clones lacking ssh show various bristle phenotypes including greater curvature (small arrows), thick and stumpy shafts (open arrow) and less often split shafts (asterisk). Moreover, the trichomes that are present on each cell are also affected and curved more strongly. In all panels, the size bar is 20μm.
4 240 A. C. Nagel et al. photoreceptor cells prompted us to compare more closely the effects of either mutation of the fly. The mislocalization of the photoreceptor nuclei could be a result of a disturbance of the cellular cytoskeleton like in ssh mutants or of the epithelial organization. We investigated this possibility further by comparing clones lacking ssh with those overexpressing PKD-SE in the larval retina. The cytoskeleton was monitored directly by staining for filamentous actin (F-actin). We noted extreme changes in ssh mutant cell clones that generally showed a strong disturbance of the cell shape. As expected, the ssh mutant cells were strongly enriched in F-actin which frequently accumulated along scars rather than within the entire clone (Fig. 2b b ). The scars may arise from a contraction of the apical cell surface as a consequence of Ssh loss and also from cell death of mutant cells (NIWA et al. 2002; ROGERS et al. 2005). The epithelial organization was analyzed by monitoring several proteins that mark different apical-basal positions within the cell including Armadillo (Arm) that is present at the zonula adherens and Disc large (Dlg) that marks the septate junctions (MÜLLER and BOSSINGER 2003). Typically, the markers appeared enriched in the mutant clones, however, were all present at the expected position (Fig. 2d d, 2f f ). Overexpression of PKD-SE in cell clones had similar effects, that were however considerably Hereditas 147 (2010) weaker (Fig. 2a a, 2c c, 2e e ). Notably, F-actin was strongly enriched, whereas the epithelial organization remained intact. We conclude that activation of PKD influences the dynamics of the actin cytoskeleton in a similar way as does the loss of Ssh. Hence PKD might function as a negative regulator of Ssh. Antagonistic relationship between PKD-SE and ssh In order to address the genetic relationship between PKD and ssh, we made use of the F-actin accumulation in PKDSE overexpressing cells and asked, whether it was dependent on ssh gene dosis. To combine PKD-SE overexpression with either gain or loss of ssh gene activity, we made use of the Gal4/UAS system that allows a tissue specific overexpression of the desired genes (BRAND and PERRIMON 1993). We utilized a dominant negative form of Ssh (ssh. CS; NIWA et al. 2002) and a wild type form of Ssh (ssh.n; NIWA et al. 2002) to generate a loss and a gain of function situation, respectively. Indeed, we observed a strong accumulation of F-actin in the retina of pupae in which PKDSE was overexpressed. F-actin accumulation was extremely strong in the homozygous animals compared to the heterozygotes and the controls (Fig. 3a c). A combined overexpression of the dominant negative ssh.cs together with PKD-SE resulted in an enhanced F-actin Fig. 2. Sub-cellular effects of PKD-SE overexpression match those of Ssh loss. Cell clones were induced in eye imaginal discs. Clones overexpressing PKD-SE are marked by GFP (green; a, a, c, c, e, e ), whereas clones lacking ssh are marked by the absence of GFP (b, b, d, d, f, f ). The discs were stained with either rhodamine-coupled phalloidin to visualize F-actin (a b ), or antibodies directed against Armadillo (Arm) to mark the zonula adherence (c d ) or against Disc large (Dlg) to mark the septate junctions (e f ). The markers are shown in red. (a f) shows the double staining; a f the single markers (red) and a f the clones (green and absence of green, respectively; clones are outlined). Size bar is 20μm.
5 Hereditas 147 (2010) Drosophila PKD inhibits Ssh phosphatase 241 enrichment (Fig. 3g). Since ssh.cs overexpression alone did not elicit this effect (Fig. 3f), we conclude that PKD and Ssh act antagonistically. Accordingly, F-actin enrichment appeared reduced when the wild type ssh.n gene copy was overexpressed together with PKD-SE (Fig. 1d e). These results are indicative of a negative relationship between PKD and Ssh also in Drosophila. DISCUSSION Drosophila PKD-SE acts as negative regulator of Slingshot phosphatase In this study, we have used Drosophila as an in vivo model system to investigate the role of PKD in the regulation of actin dynamics. Our results suggest that a constitutively active Drosophila PKD acts as negative regulator Slingshot phosphatase. Expression of PKD-SE results in the accumulation of F-actin, thus affecting the cytoskeleton. The observed phenotypes include defects in cell morphology, e.g. positioning of photoreceptor nuclei or rhabdomere formation in the ommatidia ( MAIER et al. 2007). Malformation of bristles and hairs point to a major role of PKD in the formation of parallel actin-filament bundles, resembling mutations in ssh ( NIWA et al. 2002). However, the phenotypes that arise from overactivation of Drosophila PKD are always much weaker than those induced by the loss of Ssh activity. This is not unexpected since it is already known that several players regulate Ssh activity apart from PKD. One example is PAK4 (p21 activated kinase 4), which acts as a dual regulator in activating LIMK1 and inhibiting SSH1L by phosphorylation ( SOOSAIRAJAH et al. 2005). Drosophila dpak has been shown to activate LIMK-1, acting as an essential regulator of actin dynamics as well ( CONDER et al. 2004; NG and L UO 2004). Drosophila Ssh harbors several presumptive PAK-consensus sites ( RENNEFAHRT et al. 2007). Hence, it is conceivable that dpak may phosphorylate and thereby inactivate Ssh in Drosophila like in mammals. Fig. 3. PKD-SE induces F-actin accumulation that is modified by Ssh activity. Upper panels, apical view and lower panels, basal view of a pupal retina stained with rhodaminecoupled Phalloidin to visualize filamentous actin. Note strong enrichment of F-actin upon overexpression of active PKD-SE (a, a wild type vs b, b one and c, c two copies of PKD-SE overexpression construct, respectively). Concomitant overexpression of the dominant negative Ssh form (ssh.cs) slightly increases F-actin accumulation (g, g ) compared with the overexpression of ssh-cs alone (f, f ). In contrast, overexpression of the wild type Ssh (ssh.n), which on its own has no effect (d, d ), decreased the F-actin accumulation by PKD-SE to more normal levels (e, e ). Size bar is 20 μm. The following genotypes are shown: (a a ) gmr-gal4 UAS-lacZ control retina. (b b ) gmr-gal4 UAS-PKD-SE / ; UAS-lacZ /. (c c ) gmr-gal4 UAS-PKD-SE / gmr-gal4 UAS-PKD-SE. (d d ) gmr-gal4/ ; UAS-ssh.N /. (e e ) gmr-gal4 UAS-PKD-SE / ; UASssh.N /. (f f ) gmr-gal4/ ; UAS-ssh.CS/. (g g ) gmr-gal4 UAS-PKD-SE / ; UAS-ssh.CS/. Molecular differences between Drosophila Slingshot and human Slingshot like proteins SSH1L, SSH2L and SSH3L From studies in mammals it is known that Slingshot like proteins (SSH1L, SSH2L) bind to F-actin with the C-terminal domain, which is a prerequisite for the activation of the phosphatase ( OHTA et al. 2003; NAGATA-OHASHI et al. 2004; SOOSAIRAJAH et al. 2005). Hence, the C-terminal domain acts auto-inhibitory to SSH activity. In SSH1L the C-terminal domain contains two Serine residues (Ser 937 and Ser 978 ) that are phosphorylated by human PKD1 and PKD2 ( EISELER et al. 2009; PETERBURS et al. 2009). Phosphorylation of Ser 937 and Ser 978 triggers the binding of scaffolding proteins, and the sequestration of SSH1L to the cytoplasm, thereby limiting the amount of actin-bound active SSH1L ( NAGATA-OHASHI et al. 2004). Interestingly, these phosphorylation sites are neither conserved in SSH3L nor in the Drosophila Ssh protein ( OHTA et al. 2003). Instead, there is a potential PKD target sequence that is located next to the phosphatase catalytic centre and that is well conserved in human SSH1L, SSH2L and Drosophila Slingshot proteins. This raises the possibility of an alternative mechanism of Slingshot inhibition by PKD kinases: phosphorylation of Slingshot at the catalytic centre may directly interfere with its enzymatic activity. It is tempting to speculate that such an inhibitory mechanism is crucial
6 242 A. C. Nagel et al. Hereditas 147 (2010) to the regulation of Slingshot and it may be conserved from fly to human. Acknowledgements We are indebted to I. Wech for excellent technical help. We acknowledge K. Basler, the Bloomington stock centre, the DSHB and DGRC for fly stocks, monoclonal antibodies and cdna clones, respectively. This work was supported by the German Research Foundation (DFG) through a grant to DM (MA 1328/8-1). REFERENCES Brand, A. H. and Perrimon, N Targeted gene expression as a means of altering cell fates and generating dominant phenotypes. Development 118: Conder, R., Yu, H., Ricos, M. et al dpak is required for integrity of the leading edge cytoskeleton during Drosophila dorsal closure but does not signal through the JNK cascade. Dev. Biol. 276: DesMarais, V., Ghosh, M., Eddy, R. et al Cofilin takes the lead. J. Cell Sci. 118: Eiseler, T., D ö ppler, H., Yan, I. K. et al Protein kinase D1 regulates cofilin-mediated F-actin reorganization and cell motility through slingshot. Nat. Cell Biol. 11: Jacinto, A. and Baum, B Actin in development. Mech. Dev. 120: Maier, D., Nagel, A. C., Johannes, B. et al Subcellular localization of Hairless protein shows a major focus of activity within the nucleus. Mech. Dev. 89: Maier, D., Hausser, A., Nagel, A. C. et al Drosophila protein kinase D is broadly expressed and a fraction localizes to the Golgi compartment. Gene Expr. Patterns 6: Maier, D., Nagel, A. C., Gloc, H. et al Protein kinase D regulates several aspects of development in Drosophila melanogaster. BMC Dev. Biol. 25; 7:74. M ü ller, H.-A. and Bossinger, O Molecular networks controlling epithelial cell polarity in development. Mech. Dev. 120: Nagata-Ohashi, K., Ohta, Y, Goto, K. et al A pathway of neuregulin-induced activation of cofilin-phosphatase Slingshot and cofilin in lamellipodia. J. Cell. Biol. 165: Ng, J. and Luo, L Rho GTPases regulate axon growth through convergent and divergent signalling. Neuron 44: Niwa, R., Nagata-Ohashi, K., Takeichi, M. et al Control of actin reorganization by Slingshot, a family of phosphatases that dephosphorylate ADF/cofilin. Cell 108: Ohta, Y., Kousaka, K., Nagata-Ohashi, K. et al Differential activities, subcellular distribution and tissue expression patterns of three members of Slingshot family phosphatases that dephosphorylate cofilin. Genes Cells 8: Peterburs, O., Heering, J., Link, G. et al Protein kinase D (PKD) regulates cell migration by direct phosphorylation of the cofilin phosphatase Slinghot 1 like (SSH1L). Cancer Res. 14: Pollard, T. D. and Borisy, G. G Cellular motility driven by assembly and disassembly of actin filaments. Cell 112: Protzer, C. E., Wech, I. and Nagel, A. C Hairless induces cell death by downregulation of EGFR signalling activity. J. Cell Sci. 121: Rennefahrt, U. E., Deacon, S. W., Parker, S. A. et al Specificity profiling of PAK kinases allows identification of novel phosphorylation sites. J. Biol. Chem. 282: Rogers, E. M., Hsiung, F., Rodrigues, A. B. et al Slingshot cofilin phosphatase localization is regulated by Receptor Tyrosine Kinases and cytoskeletal structure in the developing Drosophila eye. Mech. Dev. 122: Soosairajah, J., Maiti, S., Wiggan, O N. et al Interplay between components of a novel LIM kinase slingshot phosphatase complex regulates cofilin. EMBO J. 24: Struhl, G. and Basler, K Organizing activity of wingless protein in Drosophila. Cell 72: van der Meer, J Optical clean and permanent whole mount preparations for phase contrast microscopy of cuticular structures of insect larvae. Drosophila Inf. Serv. 52: 160. Xu, T. and Rubin, G. M Analysis of genetic mosaics in the developing and adult Drosophila tissues. Development 117:
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila
 Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon 97403 Correspondence
Chapter 4 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays.
 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290
 Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Supplementary Information
 Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Hairless induces cell death by downregulation of EGFR signalling activity
 Research Article 3167 Hairless induces cell death by downregulation of EGFR signalling activity Cornelia E. Protzer, Irmgard Wech and Anja C. Nagel* University of Hohenheim, Institute of Genetics (240),
Research Article 3167 Hairless induces cell death by downregulation of EGFR signalling activity Cornelia E. Protzer, Irmgard Wech and Anja C. Nagel* University of Hohenheim, Institute of Genetics (240),
Sang-Chul Nam a,1, Bibhash Mukhopadhyay b, Kwang-Wook Choi a,b,c,
 Developmental Biology 306 (2007) 624 635 www.elsevier.com/locate/ydbio Antagonistic functions of Par-1 kinase and protein phosphatase 2A are required for localization of Bazooka and photoreceptor morphogenesis
Developmental Biology 306 (2007) 624 635 www.elsevier.com/locate/ydbio Antagonistic functions of Par-1 kinase and protein phosphatase 2A are required for localization of Bazooka and photoreceptor morphogenesis
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
Different cell types exhibit different degrees of complexity in
 Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Cell biology: Death drags down the neighbourhood
 Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Developmental Biology
 Developmental Biology 334 (2009) 161 173 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Wingless signaling and the control of cell
Developmental Biology 334 (2009) 161 173 Contents lists available at ScienceDirect Developmental Biology journal homepage: www.elsevier.com/developmentalbiology Wingless signaling and the control of cell
5- Semaphorin-Plexin-Neuropilin
 5- Semaphorin-Plexin-Neuropilin 1 SEMAPHORINS-PLEXINS-NEUROPILINS ligands receptors co-receptors semaphorins and their receptors are known signals for: -axon guidance -cell migration -morphogenesis -immune
5- Semaphorin-Plexin-Neuropilin 1 SEMAPHORINS-PLEXINS-NEUROPILINS ligands receptors co-receptors semaphorins and their receptors are known signals for: -axon guidance -cell migration -morphogenesis -immune
Neurite initiation. Neurite formation begins with a bud that sprouts from the cell body. One or several neurites can sprout at a time.
 Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Role of Mitochondrial Remodeling in Programmed Cell Death in
 Developmental Cell, Vol. 12 Supplementary Data Role of Mitochondrial Remodeling in Programmed Cell Death in Drosophila melanogaster Gaurav Goyal, Brennan Fell, Apurva Sarin, Richard J. Youle, V. Sriram.
Developmental Cell, Vol. 12 Supplementary Data Role of Mitochondrial Remodeling in Programmed Cell Death in Drosophila melanogaster Gaurav Goyal, Brennan Fell, Apurva Sarin, Richard J. Youle, V. Sriram.
Reading: Chapter 5, pp ; Reference chapter D, pp Problem set F
 Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Three Distinct Roles for Notch in Drosophila R7 Photoreceptor Specification
 Three Distinct Roles for Notch in Drosophila R7 Photoreceptor Specification Andrew Tomlinson 1. *, Yannis Emmanuel Mavromatakis 1., Gary Struhl 1,2 1 Department of Genetics and Development, College of
Three Distinct Roles for Notch in Drosophila R7 Photoreceptor Specification Andrew Tomlinson 1. *, Yannis Emmanuel Mavromatakis 1., Gary Struhl 1,2 1 Department of Genetics and Development, College of
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Segment boundary formation in Drosophila embryos
 Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
L œil de la Drosophile pour l étude du développement et de l apoptose
 L œil de la Drosophile pour l étude du développement et de l apoptose Pr Bertrand Mollereau Apoptosis and Neurogenetic LBMC UMR5239 ENS Lyon http://www.ens-lyon.fr/lbmc/apodroso/ Drosophila is a powerful
L œil de la Drosophile pour l étude du développement et de l apoptose Pr Bertrand Mollereau Apoptosis and Neurogenetic LBMC UMR5239 ENS Lyon http://www.ens-lyon.fr/lbmc/apodroso/ Drosophila is a powerful
Conclusions. The experimental studies presented in this thesis provide the first molecular insights
 C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
Nature Neuroscience: doi: /nn.2662
 Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
BIO 311C Spring 2010
 BIO 311C Spring 2010 Prokaryotic cells contain structures that are very similar to structures of the eukaryotic cytoskeleton. Prokaryotic cytoskeletal elements are required for cell division, maintaining
BIO 311C Spring 2010 Prokaryotic cells contain structures that are very similar to structures of the eukaryotic cytoskeleton. Prokaryotic cytoskeletal elements are required for cell division, maintaining
mirror controls planar polarity and equator formation through repression of fringe expression and through control of cell affinities
 Development 126, 5857-5866 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5359 5857 mirror controls planar polarity and equator formation through repression of fringe expression
Development 126, 5857-5866 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5359 5857 mirror controls planar polarity and equator formation through repression of fringe expression
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
targets. clustering show that different complex pathway
 Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Supplementary Figure 1. CLICR allows clustering and activation of cytoplasmic protein targets. (a, b) Upon light activation, the Cry2 (red) and LRP6c (green) components co-cluster due to the heterodimeric
Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A)
 Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A) and mutant (B) 4 dpf retinae. The central retina of the
Fig. S1. Proliferation and cell cycle exit are affected by the med mutation. (A,B) M-phase nuclei are visualized by a-ph3 labeling in wild-type (A) and mutant (B) 4 dpf retinae. The central retina of the
7.06 Problem Set #4, Spring 2005
 7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
7.06 Problem Set #4, Spring 2005 1. You re doing a mutant hunt in S. cerevisiae (budding yeast), looking for temperaturesensitive mutants that are defective in the cell cycle. You discover a mutant strain
10/2/2015. Chapter 4. Determination and Differentiation. Neuroanatomical Diversity
 Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
purpose of this Chapter is to highlight some problems that will likely provide new
 119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
Richik N. Ghosh, Linnette Grove, and Oleg Lapets ASSAY and Drug Development Technologies 2004, 2:
 1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
1 3/1/2005 A Quantitative Cell-Based High-Content Screening Assay for the Epidermal Growth Factor Receptor-Specific Activation of Mitogen-Activated Protein Kinase Richik N. Ghosh, Linnette Grove, and Oleg
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 2, 2006 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Ultrabithorax and the control of cell morphology in Drosophila halteres
 Development 127, 97-107 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV7766 97 Ultrabithorax and the control of cell morphology in Drosophila halteres Fernando Roch and Michael
Development 127, 97-107 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV7766 97 Ultrabithorax and the control of cell morphology in Drosophila halteres Fernando Roch and Michael
Drosophila wing. Temporal regulation of Apterous activity during development of the. Marco Milán and Stephen M. Cohen* SUMMARY
 Development 127, 3069-3078 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2547 3069 Temporal regulation of Apterous activity during development of the Drosophila wing Marco Milán
Development 127, 3069-3078 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV2547 3069 Temporal regulation of Apterous activity during development of the Drosophila wing Marco Milán
Suppression of the rbf null mutants by a de2f1 allele that lacks transactivation domain
 Development 127, 367-379 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV5342 367 Suppression of the rbf null mutants by a de2f1 allele that lacks transactivation domain Wei Du
Development 127, 367-379 (2000) Printed in Great Britain The Company of Biologists Limited 2000 DEV5342 367 Suppression of the rbf null mutants by a de2f1 allele that lacks transactivation domain Wei Du
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins
 13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
13-3. Synthesis-Secretory pathway: Sort lumenal proteins, Secrete proteins, Sort membrane proteins Molecular sorting: specific budding, vesicular transport, fusion 1. Why is this important? A. Form and
16 The Cell Cycle. Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization
 The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
The Cell Cycle 16 The Cell Cycle Chapter Outline The Eukaryotic Cell Cycle Regulators of Cell Cycle Progression The Events of M Phase Meiosis and Fertilization Introduction Self-reproduction is perhaps
Development of the Drosophila mushroom bodies: sequential generation of three distinct types of neurons from a neuroblast
 Development 126, 4065-4076 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8625 4065 Development of the Drosophila mushroom bodies: sequential generation of three distinct types
Development 126, 4065-4076 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8625 4065 Development of the Drosophila mushroom bodies: sequential generation of three distinct types
The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also
 c Indian Academy of Sciences RESEARCH ARTICLE The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also MUKULIKA RAY and SUBHASH
c Indian Academy of Sciences RESEARCH ARTICLE The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also MUKULIKA RAY and SUBHASH
Why Flies? stages of embryogenesis. The Fly in History
 The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
Axis Specification in Drosophila
 Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental Biology Biology 4361 Axis Specification in Drosophila November 6, 2007 Axis Specification in Drosophila Fertilization Superficial cleavage Gastrulation Drosophila body plan Oocyte formation
Developmental, Cellular, and Molecular Biology Group, Duke University, Durham, North Carolina
 Molecular Biology of the Cell Vol. 9, 3505 3519, December 1998 Drosophila coracle, a Member of the Protein 4.1 Superfamily, Has Essential Structural Functions in the Septate Junctions and Developmental
Molecular Biology of the Cell Vol. 9, 3505 3519, December 1998 Drosophila coracle, a Member of the Protein 4.1 Superfamily, Has Essential Structural Functions in the Septate Junctions and Developmental
SUPPLEMENTARY INFORMATION
 GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
GP2 Type I-piliated bacteria FAE M cell M cell pocket idc T cell mdc Generation of antigenspecific T cells Induction of antigen-specific mucosal immune response Supplementary Figure 1 Schematic diagram
with%dr.%van%buskirk%%%
 with%dr.%van%buskirk%%% How$to$do$well?$ Before$class:$read$the$corresponding$chapter$ Come$to$class$ready$to$par9cipate$in$Top$Hat$ Don t$miss$an$exam!!!!!!!!!!!!!!!!!!!!!!!!!!$ But$I m$not$good$with$science
with%dr.%van%buskirk%%% How$to$do$well?$ Before$class:$read$the$corresponding$chapter$ Come$to$class$ready$to$par9cipate$in$Top$Hat$ Don t$miss$an$exam!!!!!!!!!!!!!!!!!!!!!!!!!!$ But$I m$not$good$with$science
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
SUPPLEMENTARY INFORMATION doi:10.1038/nature12791 Supplementary Figure 1 (1/3) WWW.NATURE.COM/NATURE 1 RESEARCH SUPPLEMENTARY INFORMATION Supplementary Figure 1 (2/3) 2 WWW.NATURE.COM/NATURE SUPPLEMENTARY
Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and
 Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and Lifeact-Ruby (red) were imaged in vivo to visualize secretion
Supplementary Figure 1. Real time in vivo imaging of SG secretion. (a) SGs from Drosophila third instar larvae that express Sgs3-GFP (green) and Lifeact-Ruby (red) were imaged in vivo to visualize secretion
Signal Transduction. Dr. Chaidir, Apt
 Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Signal Transduction Dr. Chaidir, Apt Background Complex unicellular organisms existed on Earth for approximately 2.5 billion years before the first multicellular organisms appeared.this long period for
Chapter 11. Development: Differentiation and Determination
 KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY ACTIVITY STEPHANIE E. CRILLY SPRING 2015
 THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY INVESTIGATING THE ROLE OF βheavy-spectrin IN MODULATION OF RAC1 ACTIVITY STEPHANIE E. CRILLY SPRING
THE PENNSYLVANIA STATE UNIVERSITY SCHREYER HONORS COLLEGE DEPARTMENT OF BIOCHEMISTRY AND MOLECULAR BIOLOGY INVESTIGATING THE ROLE OF βheavy-spectrin IN MODULATION OF RAC1 ACTIVITY STEPHANIE E. CRILLY SPRING
Autophagy-independent function of Atg1 for apoptosis-induced compensatory proliferation
 University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 8-19-2016 Autophagy-independent function of Atg1 for apoptosis-induced
University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 8-19-2016 Autophagy-independent function of Atg1 for apoptosis-induced
Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway
 Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway Justin Hogan, Meagan Valentine, Chris Cox, Kristy Doyle, Simon Collier* Department
Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway Justin Hogan, Meagan Valentine, Chris Cox, Kristy Doyle, Simon Collier* Department
Supporting Information
 Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family
 Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Leucine-rich repeat receptor-like kinases (LRR-RLKs), HAESA, ERECTA-family GENES & DEVELOPMENT (2000) 14: 108 117 INTRODUCTION Flower Diagram INTRODUCTION Abscission In plant, the process by which a plant
Follow this and additional works at: Part of the Medical Sciences Commons
 Bucknell University Bucknell Digital Commons Master s Theses Student Theses 2010 The overexpression of homeotic complex gene Ultrabithorax in the post-embryonic neuronal lineages of the ventral nervous
Bucknell University Bucknell Digital Commons Master s Theses Student Theses 2010 The overexpression of homeotic complex gene Ultrabithorax in the post-embryonic neuronal lineages of the ventral nervous
Chapter 16. Cellular Movement: Motility and Contractility. Lectures by Kathleen Fitzpatrick Simon Fraser University Pearson Education, Inc.
 Chapter 16 Cellular Movement: Motility and Contractility Lectures by Kathleen Fitzpatrick Simon Fraser University Two eukaryotic motility systems 1. Interactions between motor proteins and microtubules
Chapter 16 Cellular Movement: Motility and Contractility Lectures by Kathleen Fitzpatrick Simon Fraser University Two eukaryotic motility systems 1. Interactions between motor proteins and microtubules
 DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016
 Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Molecular Cell Biology 5068 In Class Exam 2 November 8, 2016 Exam Number: Please print your name: Instructions: Please write only on these pages, in the spaces allotted and not on the back. Write your
Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells
 Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells Understand how a simple biochemical oscillator can drive the
Life Sciences 1a: Section 3B. The cell division cycle Objectives Understand the challenges to producing genetically identical daughter cells Understand how a simple biochemical oscillator can drive the
Wingless transduction by the Frizzled and Frizzled2 proteins of Drosophila
 Development 126, 5441-5452 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV7755 5441 Wingless transduction by the Frizzled and Frizzled2 proteins of Drosophila Chiann-mun Chen
Development 126, 5441-5452 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV7755 5441 Wingless transduction by the Frizzled and Frizzled2 proteins of Drosophila Chiann-mun Chen
SUPPLEMENTARY INFORMATION
 Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
Neurite formation & neuronal polarization. The cytoskeletal components of neurons have characteristic distributions and associations
 Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
Mechanisms of neuronal migration & Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 The cytoskeletal
ADAM FAMILY. ephrin A INTERAZIONE. Eph ADESIONE? PROTEOLISI ENDOCITOSI B A RISULTATO REPULSIONE. reverse. forward
 ADAM FAMILY - a family of membrane-anchored metalloproteases that are known as A Disintegrin And Metalloprotease proteins and are key components in protein ectodomain shedding Eph A INTERAZIONE B ephrin
ADAM FAMILY - a family of membrane-anchored metalloproteases that are known as A Disintegrin And Metalloprotease proteins and are key components in protein ectodomain shedding Eph A INTERAZIONE B ephrin
Regulation and signaling. Overview. Control of gene expression. Cells need to regulate the amounts of different proteins they express, depending on
 Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
Regulation and signaling Overview Cells need to regulate the amounts of different proteins they express, depending on cell development (skin vs liver cell) cell stage environmental conditions (food, temperature,
09/30/2017. Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center
 09/30/2017 Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs
09/30/2017 Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs
Bio 127 Section I Introduction to Developmental Biology. Cell Cell Communication in Development. Developmental Activities Coordinated in this Way
 Bio 127 Section I Introduction to Developmental Biology Cell Cell Communication in Development Gilbert 9e Chapter 3 It has to be EXTREMELY well coordinated for the single celled fertilized ovum to develop
Bio 127 Section I Introduction to Developmental Biology Cell Cell Communication in Development Gilbert 9e Chapter 3 It has to be EXTREMELY well coordinated for the single celled fertilized ovum to develop
Integrin-ECM interactions regulate the changes in cell shape driving the morphogenesis of the Drosophila wing epithelium
 Research Article 1061 Integrin-ECM interactions regulate the changes in cell shape driving the morphogenesis of the Drosophila wing epithelium Paloma Domínguez-Giménez 1, Nicholas H. Brown 2 and María
Research Article 1061 Integrin-ECM interactions regulate the changes in cell shape driving the morphogenesis of the Drosophila wing epithelium Paloma Domínguez-Giménez 1, Nicholas H. Brown 2 and María
TGF-β/BMP superfamily members, Gbb-60A and Dpp, cooperate to provide pattern information and establish cell identity in the Drosophila wing
 Development 125, 2723-2734 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV5189 2723 TGF-β/BMP superfamily members, Gbb-60A and Dpp, cooperate to provide pattern information and
Development 125, 2723-2734 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV5189 2723 TGF-β/BMP superfamily members, Gbb-60A and Dpp, cooperate to provide pattern information and
Heather Currinn, Benjamin Guscott, Zita Balklava, Alice Rothnie and Thomas Wassmer*
 Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Online Resources APP controls the formation of PI(3,5)P 2 vesicles through its binding of the PIKfyve complex. Cellular and Molecular Life Sciences Heather Currinn, Benjamin Guscott, Zita Balklava, Alice
Drosophila IAP1-mediated ubiquitylation controls activation of the initiator caspase DRONC independent of protein degradation
 University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 9-1-2011 Drosophila IAP1-mediated ubiquitylation controls
University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 9-1-2011 Drosophila IAP1-mediated ubiquitylation controls
Drosophila receptor tyrosine kinase signaling
 Development 129, 71-82 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV5966 71 MASK, a large ankyrin repeat and KH domain-containing protein involved in Drosophila receptor tyrosine
Development 129, 71-82 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV5966 71 MASK, a large ankyrin repeat and KH domain-containing protein involved in Drosophila receptor tyrosine
4) Please cite Dagda et al J Biol Chem 284: , for any publications or presentations resulting from use or modification of the macro.
 Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Supplement Figure S1. Algorithmic quantification of mitochondrial morphology in SH- SY5Y cells treated with known fission/fusion mediators. Parental SH-SY5Y cells were transiently transfected with an empty
Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1
 1362 Research Article Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1 Thomas J. Widmann and Christian Dahmann* Max Planck Institute
1362 Research Article Dpp signaling promotes the cuboidal-to-columnar shape transition of Drosophila wing disc epithelia by regulating Rho1 Thomas J. Widmann and Christian Dahmann* Max Planck Institute
Drosophila. The wingless signalling pathway and the patterning of the wing margin in. Juan Pablo Couso*, Sarah A. Bishop and Alfonso Martinez Arias
 Development 120, 621-636 (1994) Printed in Great Britain The Company of Biologists Limited 1994 621 The wingless signalling pathway and the patterning of the wing margin in Drosophila Juan Pablo Couso*,
Development 120, 621-636 (1994) Printed in Great Britain The Company of Biologists Limited 1994 621 The wingless signalling pathway and the patterning of the wing margin in Drosophila Juan Pablo Couso*,
Nature Neuroscience: doi: /nn.2717
 Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Figure 1. Automated 4D detection and tracking of endosomes and polarity markers.
 A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
Systematic in vivo RNAi analysis of putative components of the Drosophila cell death machinery Introduction UNCORRECTED PROOF
 (2006) 00, 1 12 & 2006 Nature Publishing Group All rights reserved 1350-9047/06 $30.00 www.nature.com/cdd Systematic in vivo RNAi analysis of putative components of the Drosophila cell death machinery
(2006) 00, 1 12 & 2006 Nature Publishing Group All rights reserved 1350-9047/06 $30.00 www.nature.com/cdd Systematic in vivo RNAi analysis of putative components of the Drosophila cell death machinery
RACK-1 Acts with Rac GTPase Signaling and UNC-115/ ablim in Caenorhabditis elegans Axon Pathfinding and Cell Migration
 RACK-1 Acts with Rac GTPase Signaling and UNC-115/ ablim in Caenorhabditis elegans Axon Pathfinding and Cell Migration Rafael S. Demarco, Erik A. Lundquist* Programs in Genetics and Molecular, Cellular,
RACK-1 Acts with Rac GTPase Signaling and UNC-115/ ablim in Caenorhabditis elegans Axon Pathfinding and Cell Migration Rafael S. Demarco, Erik A. Lundquist* Programs in Genetics and Molecular, Cellular,
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila
 Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
S Phase Coupled E2f1 Destruction Ensures Homeostasis in Proliferating Tissues
 S Phase Coupled E2f1 Destruction Ensures Homeostasis in Proliferating Tissues Jean M. Davidson 1, Robert J. Duronio 1,2,3 * 1 Department of Biology, The University of North Carolina at Chapel Hill, Chapel
S Phase Coupled E2f1 Destruction Ensures Homeostasis in Proliferating Tissues Jean M. Davidson 1, Robert J. Duronio 1,2,3 * 1 Department of Biology, The University of North Carolina at Chapel Hill, Chapel
Chapter 18 Lecture. Concepts of Genetics. Tenth Edition. Developmental Genetics
 Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
Drosophila through Notch
 Development 126, 2993-3003 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV1389 2993 Composite signalling from Serrate and Delta establishes leg segments in Drosophila through
Development 126, 2993-3003 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV1389 2993 Composite signalling from Serrate and Delta establishes leg segments in Drosophila through
Spatial pattern of nonmuscle myosin-ii distribution during the development of the Drosophila compound eye and implications for retinal morphogenesis
 Developmental Biology 269 (2004) 519 533 www.elsevier.com/locate/ydbio Spatial pattern of nonmuscle myosin-ii distribution during the development of the Drosophila compound eye and implications for retinal
Developmental Biology 269 (2004) 519 533 www.elsevier.com/locate/ydbio Spatial pattern of nonmuscle myosin-ii distribution during the development of the Drosophila compound eye and implications for retinal
Abl Kinase Inhibits the Engulfment of Apoptotic [corrected] Cells in Caenorhabditis elegans
![Abl Kinase Inhibits the Engulfment of Apoptotic [corrected] Cells in Caenorhabditis elegans Abl Kinase Inhibits the Engulfment of Apoptotic [corrected] Cells in Caenorhabditis elegans](/thumbs/74/71020578.jpg) Abl Kinase Inhibits the Engulfment of Apoptotic [corrected] Cells in Caenorhabditis elegans The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story
Abl Kinase Inhibits the Engulfment of Apoptotic [corrected] Cells in Caenorhabditis elegans The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story
Isoform-specific functions of Mud/NuMA mediate binucleation of Drosophila male accessory gland cells
 Taniguchi et al. BMC Developmental Biology (2014) 14:46 DOI 10.1186/s12861-014-0046-5 RESEARCH ARTICLE Open Access Isoform-specific functions of Mud/NuMA mediate binucleation of Drosophila male accessory
Taniguchi et al. BMC Developmental Biology (2014) 14:46 DOI 10.1186/s12861-014-0046-5 RESEARCH ARTICLE Open Access Isoform-specific functions of Mud/NuMA mediate binucleation of Drosophila male accessory
Functional and regulatory interactions between Hox and extradenticle genes
 Functional and regulatory interactions between Hox and extradenticle genes Natalia Azpiazu and Ginés Morata 1 Centro de Biologia Molecular Centro Superior de Investigaciones Cientificas-Universidad Autońoma
Functional and regulatory interactions between Hox and extradenticle genes Natalia Azpiazu and Ginés Morata 1 Centro de Biologia Molecular Centro Superior de Investigaciones Cientificas-Universidad Autońoma
Lecture 10: Cyclins, cyclin kinases and cell division
 Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Chem*3560 Lecture 10: Cyclins, cyclin kinases and cell division The eukaryotic cell cycle Actively growing mammalian cells divide roughly every 24 hours, and follow a precise sequence of events know as
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Overexpression of YFP::GPR-1 in the germline.
 Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Drosophila. Regulation of Wingless and Vestigial expression in wing and haltere discs of. Prasad Mohit, Ruchi Bajpai and L. S. Shashidhara* SUMMARY
 Development 130, 1537-1547 2003 The Company of Biologists Ltd doi:10.1242/dev.00393 1537 Regulation of Wingless and Vestigial expression in wing and haltere discs of Drosophila Prasad Mohit, Ruchi Bajpai
Development 130, 1537-1547 2003 The Company of Biologists Ltd doi:10.1242/dev.00393 1537 Regulation of Wingless and Vestigial expression in wing and haltere discs of Drosophila Prasad Mohit, Ruchi Bajpai
Neurite formation & neuronal polarization
 Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Neurite formation & neuronal polarization Paul Letourneau letou001@umn.edu Chapter 16; The Cytoskeleton; Molecular Biology of the Cell, Alberts et al. 1 An immature neuron in cell culture first sprouts
Biol403 - Receptor Serine/Threonine Kinases
 Biol403 - Receptor Serine/Threonine Kinases The TGFβ (transforming growth factorβ) family of growth factors TGFβ1 was first identified as a transforming factor; however, it is a member of a family of structurally
Biol403 - Receptor Serine/Threonine Kinases The TGFβ (transforming growth factorβ) family of growth factors TGFβ1 was first identified as a transforming factor; however, it is a member of a family of structurally
7.013 Spring 2005 Problem Set 4
 MIT Department of Biology 7.013: Introductory Biology - Spring 2005 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel NAME TA 7.013 Spring 2005 Problem Set 4 FRIDAY April 8th,
MIT Department of Biology 7.013: Introductory Biology - Spring 2005 Instructors: Professor Hazel Sive, Professor Tyler Jacks, Dr. Claudette Gardel NAME TA 7.013 Spring 2005 Problem Set 4 FRIDAY April 8th,
1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms.
 Practicing Biology BIG IDEA 3.A 1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms. 2. Using at least 2 well-known experiments, describe which features of DNA and RNA
Practicing Biology BIG IDEA 3.A 1. Draw, label and describe the structure of DNA and RNA including bonding mechanisms. 2. Using at least 2 well-known experiments, describe which features of DNA and RNA
Drosophila Life Cycle
 Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
Drosophila Life Cycle 1 Early Drosophila Cleavage Nuclei migrate to periphery after 10 nuclear divisions. Cellularization occurs when plasma membrane folds in to divide nuclei into cells. Drosophila Superficial
orthodenticle homeobox gene
 Development 121, 3561-3572 (1995) Printed in Great Britain The Company of Biologists Limited 1995 DEV8275 3561 Pattern formation in Drosophila head development: the role of the orthodenticle homeobox gene
Development 121, 3561-3572 (1995) Printed in Great Britain The Company of Biologists Limited 1995 DEV8275 3561 Pattern formation in Drosophila head development: the role of the orthodenticle homeobox gene
BMC Developmental Biology
 BMC Developmental Biology BioMed Central Research article Rho-kinase regulates tissue morphogenesis via non-muscle myosin and LIM-kinase during Drosophila development Valerie Verdier, Guang-Chao-Chen and
BMC Developmental Biology BioMed Central Research article Rho-kinase regulates tissue morphogenesis via non-muscle myosin and LIM-kinase during Drosophila development Valerie Verdier, Guang-Chao-Chen and
Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype by altering frizzled signaling
 Development 125, 959-968 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV7601 959 Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype
Development 125, 959-968 (1998) Printed in Great Britain The Company of Biologists Limited 1998 DEV7601 959 Mutations in the cadherin superfamily member gene dachsous cause a tissue polarity phenotype
Serrate and wingless cooperate to induce vestigial gene expression and wing formation in Drosophila
 Serrate and wingless cooperate to induce vestigial gene expression and wing formation in Drosophila J.P. Couso*t, E. Knust* and A. Martinez Arias* *Department of Zoology, University of Cambridge, Downing
Serrate and wingless cooperate to induce vestigial gene expression and wing formation in Drosophila J.P. Couso*t, E. Knust* and A. Martinez Arias* *Department of Zoology, University of Cambridge, Downing
