Infra Red Spectroscopy
|
|
|
- Everett Todd
- 5 years ago
- Views:
Transcription
1 CH 2252 Instrumental Methods of Analysis Unit I Infra Red Spectroscopy M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam , Kanchipuram (Dist) Tamil Nadu, India msubbu.in@gmail.com 22-Jan-2010
2 Contents Instrumentation (Source, Optical parts and Detectors)
3 Applications: Introduction Suited for both qualitative and quantitative determination, particularly of inorganic compounds Its use in the applications to inorganic compounds is limited, because of the strong absorption of IR radiation by water. This necessitates the study of inorganic materials in the solid state (else needs D 2 O) Wavelength region: IR spectrum extends from 0.8 to 200 µm in the electromagnetic spectrum. However, most of the commercial instruments are available in the region from 0.8 to 50 µm. The region of IR spectrum which is of great interest to organic chemists is the wavelength range 2.5 to 15µm. In practice, wave number (cm -1 ) rather than wavelength is commonly used. (4000 to 600 cm -1 )
4 Introduction (contd.) IR spectroscopy deals with transitions between vibrational energy levels in molecules, and is therefore also called vibrational spectroscopy IR spectrum: It is a plot of the energy of the infrared radiation expressed in wavelength (µm) or wave number(cm -1 ) versus the percentage of light transmitted by the compound IR spectroscopy is applicable to solids, liquids, and gases. It is fast and requires small sample sizes
5 Various Regions of the Infrared Range of Spectrum Region Wavelength range (µm) Wave number range (cm -1 ) Near Middle Far Most useful IR region
6
7 Interpretation of Spectra IR rays produce vibrations and rotations in a molecule by the absorption of radiation The IR spectrum based on these absorption constitutes a powerful tool for the study of molecular structures and identification. Chemical identification is based on empirical correlations of vibrating groups, with specific absorption bands; and the quantitative determinations are dependent on the intensity measurements Interpretation of IR spectra is highly empirical and requires huge libraries of reference spectra; the interpretation of spectra is a skilled art.
8 Group IR Spectrum Bond Wave number range (cm -1 ) hydroxyl O-H amines N-H aromatic rings C-H alkenes C-H alkanes C-H nitriles C=N carbonyl C=O amines C=N Since most organic molecules have single bonds, the region below 1500 cm-1 can become quite complex and is often referred to as the fingerprint region.
9 Infrared Spectrophotometer - Instrumentation Same basic elements as in UV-vis spectrophotometer Radiation sources: blackbody radiator The optimum radiation source is an inert solid, heated electrically to temperatures between 1500 and 2000 K Practical infrared sources: Globar rod, Nernst filament, nichrome wire Globar is a silicon carbide rod Nernst filament is composed of fused rare earth oxides of zirconium and yttrium Monochromators: prism (for wavelengths less than 40µm), and gratings (for wavelengths higher than 40µm).
10 Sample Handling Solids, liquids, or gases can be handled Sample handling presents a number of problems, since no rugged window material for cell exists, which is transparent over the entire range and is also inert The most commonly used window material is NaCl, which transmits down to about 650 cm -1. KBr transmits down to about 400 cm -1, CaBr to about 250 cm -1 and CsI to about 200 cm -1 Since these materials are all water soluble, the surfaces of the windows made from them are easily fogged by exposure to atmospheric water vapor or moist samples. Therefore, they require frequent policing when used under such conditions
11 Sample Handling (contd.) Gas cells: usually have a path length of 100 mm, since this thickness gives a reasonable absorbance level for the majority of gases and vapors at the normally encountered partial pressures Liquid cells: The extinction coefficient of most liquid hydrocarbons in the infrared region is such that a pure sample of thickness between 0.01 and 0.05 mm gives an absorption spectrum that is quite suitable for analysis. The transmittance lies between 15 and 70 percent Typical path length of cells: 0.02, 0.04, 0.06, 0.1, 0.16, 0.25, 0.4, 0.6 and 1.0 mm. The external cell diameter is 30 mm. About 100 µl sample volume per 0.1 mm path length is required
12 Sampling of Solids Solids dissolved in solutions: there is no single solvent which is transparent through the entire infrared range In order to cover the main spectral range between 4000 and 650 cm -1, a combination of CCl 4 and CS 2 are employed Pressed pellet technique Sample is finely ground and mixed with an alkali halide like KBr, and then pressed into the form of a disc for examination in the instrument Mull technique Finely powdered solids are dispersed within a liquid medium to form a thick slurry. The liquid is so chosen that it has the same refractive index as the sample, so as to minimize scattering losses. The most commonly used liquid mulling agent is a mixture of liquid paraffins, known as Nujol.
13 Detectors Convert the thermal radiant energy into electrical energy Thermal detectors thermopiles, bolometers, pneumatic detectors and pyroelectric detectors. Has low response speed and detectivity, operable at room temperature Quantum type detectors photoconductive cells, and photo diodes. Has high detectivity and fast response speed, requires cooling
PAPER No. 12: ORGANIC SPECTROSCOPY MODULE No. 4: Basic principles and Instrumentation for IR spectroscopy
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Module 4: Basic principles and Instrumentation for IR spectroscopy CHE_P12_M4_e-Text TABLE OF CONTENTS
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Module 4: Basic principles and Instrumentation for IR spectroscopy CHE_P12_M4_e-Text TABLE OF CONTENTS
Advanced Pharmaceutical Analysis
 Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
Atomic Absorption Spectroscopy
 CH 2252 Instrumental Methods of Analysis Unit IV Atomic Absorption Spectroscopy Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering
CH 2252 Instrumental Methods of Analysis Unit IV Atomic Absorption Spectroscopy Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering
Infrared Spectroscopy: Identification of Unknown Substances
 Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Infrared Spectroscopy: Identification of Unknown Substances Suppose a white powder is one of the four following molecules. How can they be differentiated? H N N H H H H Na H H H H H A technique that is
Infrared spectroscopy. Siriphorn Laomanacharoen Bureau of Drug and Narcotic Department of Medical Sciences 2 March 2012
 Siriphorn Laomanacharoen Bureau of Drug and Narcotic Department of Medical Sciences 2 March 2012 1 Infrared region 2 Infrared region below red in the visible region at wavelengths between 2.5-25 µm more
Siriphorn Laomanacharoen Bureau of Drug and Narcotic Department of Medical Sciences 2 March 2012 1 Infrared region 2 Infrared region below red in the visible region at wavelengths between 2.5-25 µm more
25 Instruments for Optical Spectrometry
 25 Instruments for Optical Spectrometry 25A INSTRUMENT COMPONENTS (1) source of radiant energy (2) wavelength selector (3) sample container (4) detector (5) signal processor and readout (a) (b) (c) Fig.
25 Instruments for Optical Spectrometry 25A INSTRUMENT COMPONENTS (1) source of radiant energy (2) wavelength selector (3) sample container (4) detector (5) signal processor and readout (a) (b) (c) Fig.
Ultraviolet-Visible and Infrared Spectrophotometry
 Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Introduction. The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants
 Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Introduction The analysis of the outcome of a reaction requires that we know the full structure of the products as well as the reactants Spectroscopy and the Electromagnetic Spectrum Unlike mass spectrometry,
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Spectroscopy. Page 1 of 8 L.Pillay (2012)
 Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
Spectroscopy Electromagnetic radiation is widely used in analytical chemistry. The identification and quantification of samples using electromagnetic radiation (light) is called spectroscopy. Light has
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER
 SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
SPECTROSCOPY MEASURES THE INTERACTION BETWEEN LIGHT AND MATTER c = c: speed of light 3.00 x 10 8 m/s (lamda): wavelength (m) (nu): frequency (Hz) Increasing E (J) Increasing (Hz) E = h h - Planck s constant
Ultraviolet-Visible and Infrared Spectrophotometry
 Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
Ultraviolet-Visible and Infrared Spectrophotometry Ahmad Aqel Ifseisi Assistant Professor of Analytical Chemistry College of Science, Department of Chemistry King Saud University P.O. Box 2455 Riyadh 11451
CH 2252 Instrumental Methods of Analysis Unit V Gas Chromatography. M. Subramanian
 CH 2252 Instrumental Methods of Analysis Unit V Gas Chromatography M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603
CH 2252 Instrumental Methods of Analysis Unit V Gas Chromatography M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603
Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different types are classified by frequency or
 CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
CHEM 241 UNIT 5: PART B INFRA-RED RED SPECTROSCOPY 1 Spectroscopy of the Electromagnetic Spectrum Radiant energy is proportional to its frequency (cycles/s = Hz) as a wave (Amplitude is its height) Different
Infrared Spectroscopy
 Reminder: These notes are meant to supplement, not replace, the laboratory manual. Infrared Spectroscopy History and Application: Infrared (IR) radiation is simply one segment of the electromagnetic spectrum
Reminder: These notes are meant to supplement, not replace, the laboratory manual. Infrared Spectroscopy History and Application: Infrared (IR) radiation is simply one segment of the electromagnetic spectrum
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
12. Structure Determination: Mass Spectrometry and Infrared Spectroscopy Determining the Structure of an Organic Compound The analysis of the outcome of a reaction requires that we know the full structure
FTIR Spectrometer. Basic Theory of Infrared Spectrometer. FTIR Spectrometer. FTIR Accessories
 FTIR Spectrometer Basic Theory of Infrared Spectrometer FTIR Spectrometer FTIR Accessories What is Infrared? Infrared radiation lies between the visible and microwave portions of the electromagnetic spectrum.
FTIR Spectrometer Basic Theory of Infrared Spectrometer FTIR Spectrometer FTIR Accessories What is Infrared? Infrared radiation lies between the visible and microwave portions of the electromagnetic spectrum.
Learning Guide for Chapter 3 - Infrared Spectroscopy
 Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Instrumental Chemical Analysis
 L11 page 1 Instrumental Chemical Analysis Infrared Spectroscopy Dr. Ahmad Najjar Philadelphia University Faculty of Pharmacy Department of Pharmaceutical Sciences 2 nd semester, 2016/2017 Infrared Spectroscopy
L11 page 1 Instrumental Chemical Analysis Infrared Spectroscopy Dr. Ahmad Najjar Philadelphia University Faculty of Pharmacy Department of Pharmaceutical Sciences 2 nd semester, 2016/2017 Infrared Spectroscopy
Lecture 11. IR Theory. Next Class: Lecture Problem 4 due Thin-Layer Chromatography
 Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Lecture 11 IR Theory Next Class: Lecture Problem 4 due Thin-Layer Chromatography This Week In Lab: Ch 6: Procedures 2 & 3 Procedure 4 (outside of lab) Next Week in Lab: Ch 7: PreLab Due Quiz 4 Ch 5 Final
Welcome to Organic Chemistry II
 Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Welcome to Organic Chemistry II Erika Bryant, Ph.D. erika.bryant@hccs.edu Class Syllabus 3 CHAPTER 12: STRUCTURE DETERMINATION 4 What is this solution Soda Tea Coffee??? 5 What is this solution Soda Tea
Infrared Spectroscopy
 Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Infrared Spectroscopy IR Spectroscopy Used to identify organic compounds IR spectroscopy provides a 100% identification if the spectrum is matched. If not, IR at least provides information about the types
Infrared spectroscopy
 Infrared spectroscopy Chapter content Theory Instrumentation Measurement techniques Midinfrared (MIR) Identification of organic compounds Quantitative analysis Applications in food analysis Nearinfrared
Infrared spectroscopy Chapter content Theory Instrumentation Measurement techniques Midinfrared (MIR) Identification of organic compounds Quantitative analysis Applications in food analysis Nearinfrared
MASS and INFRA RED SPECTROSCOPY
 MASS and INFRA RED SPECTRSCPY Mass Spectroscopy The mass spectrometer was looked at in Unit 1. It was noted there that compounds produce fragmentation patterns when passes through a mass spectrometer.
MASS and INFRA RED SPECTRSCPY Mass Spectroscopy The mass spectrometer was looked at in Unit 1. It was noted there that compounds produce fragmentation patterns when passes through a mass spectrometer.
ORGANIC - BROWN 8E CH INFRARED SPECTROSCOPY.
 !! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
!! www.clutchprep.com CONCEPT: PURPOSE OF ANALYTICAL TECHNIQUES Classical Methods (Wet Chemistry): Chemists needed to run dozens of chemical reactions to determine the type of molecules in a compound.
Chapter 13 An Introduction to Ultraviolet/Visible Molecular Absorption Spectrometry
 Chapter 13 An Introduction to Ultraviolet/Visible Molecular Absorption Spectrometry 13A Measurement Of Transmittance and Absorbance Absorption measurements based upon ultraviolet and visible radiation
Chapter 13 An Introduction to Ultraviolet/Visible Molecular Absorption Spectrometry 13A Measurement Of Transmittance and Absorbance Absorption measurements based upon ultraviolet and visible radiation
EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY
 EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY Structure 7.1 Introduction Objectives 7.2 Principle 7.3 Requirements 7.4 Strategy for the Interpretation of IR Spectra 7.5 Practice Problems
EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY Structure 7.1 Introduction Objectives 7.2 Principle 7.3 Requirements 7.4 Strategy for the Interpretation of IR Spectra 7.5 Practice Problems
Infrared Spectroscopy
 x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
6. CHARACTERIZATION OF AS (III) IONS BIOSORPTION BY THE LIVE, HEAT AND ALKALINE- TREATED FUNGAL BIOMASS ON THE BASICS OF SURFACE STUDIES
 6. CHARACTERIZATION OF AS (III) IONS BIOSORPTION BY THE LIVE, HEAT AND ALKALINE- TREATED FUNGAL BIOMASS ON THE BASICS OF SURFACE STUDIES 6. Introduction Infrared spectroscopy is a technique used to identify
6. CHARACTERIZATION OF AS (III) IONS BIOSORPTION BY THE LIVE, HEAT AND ALKALINE- TREATED FUNGAL BIOMASS ON THE BASICS OF SURFACE STUDIES 6. Introduction Infrared spectroscopy is a technique used to identify
CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710)
 CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710) Identification of an Unknown by IR PRELAB (PreLab is due before entering the lab.) Every student has to prepare for each experiment by answering the Pre-Laboratory
CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710) Identification of an Unknown by IR PRELAB (PreLab is due before entering the lab.) Every student has to prepare for each experiment by answering the Pre-Laboratory
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
CHEM 51LB: EXPERIMENT 7 SPECTROSCOPIC METHODS: INFRARED SPECTROSCOPY (IDENTIFICATION OF FUNCTIONAL GROUPS)
 REACTIONS: None TECHNIQUES: IR CHEM 51LB: EXPERIMENT 7 SPECTROSCOPIC METHODS: INFRARED SPECTROSCOPY (IDENTIFICATION OF FUNCTIONAL GROUPS) After a reaction is completed, the identity of the product must
REACTIONS: None TECHNIQUES: IR CHEM 51LB: EXPERIMENT 7 SPECTROSCOPIC METHODS: INFRARED SPECTROSCOPY (IDENTIFICATION OF FUNCTIONAL GROUPS) After a reaction is completed, the identity of the product must
Infrared Spectroscopy
 Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Infrared Spectroscopy Introduction Spectroscopy is an analytical technique which helps determine structure. It destroys little or no sample. The amount of light absorbed by the sample is measured as wavelength
Determination of Chemical Composition and Molecular Microstructures: Infrared Spectroscopy
 Determination of Chemical Composition and Molecular Microstructures: Infrared Spectroscopy 1 Infrared Spectroscopy Infrared spectrometry is applied to the qualitative and quantitative determination of
Determination of Chemical Composition and Molecular Microstructures: Infrared Spectroscopy 1 Infrared Spectroscopy Infrared spectrometry is applied to the qualitative and quantitative determination of
Introduction to Electromagnetic Radiation and Radiative Transfer
 Introduction to Electromagnetic Radiation and Radiative Transfer Temperature Dice Results Visible light, infrared (IR), ultraviolet (UV), X-rays, γ-rays, microwaves, and radio are all forms of electromagnetic
Introduction to Electromagnetic Radiation and Radiative Transfer Temperature Dice Results Visible light, infrared (IR), ultraviolet (UV), X-rays, γ-rays, microwaves, and radio are all forms of electromagnetic
Increasing energy. ( 10 4 cm -1 ) ( 10 2 cm -1 )
 The branch of science which deals with the interaction of electromagnetic radiation with matter is called spectroscopy The energy absorbed or emitted in each transition corresponds to a definite frequency
The branch of science which deals with the interaction of electromagnetic radiation with matter is called spectroscopy The energy absorbed or emitted in each transition corresponds to a definite frequency
m m lighter 'atom' dominates 2 INFRARED SPECTROSCOPY All modes of vibrations are not IR active.
 INFRARED SPECTROSCOPY Infrared spectroscopy probes the interaction of infrared radiation with covalent bonds in molecules. Absorption of IR radiation results in the transitions between vibrational energy
INFRARED SPECTROSCOPY Infrared spectroscopy probes the interaction of infrared radiation with covalent bonds in molecules. Absorption of IR radiation results in the transitions between vibrational energy
CHEM 3760 Orgo I, S12, Exp 5 (Lab #6) (TECH 710: IR Unknown)
 CHEM 3760 rgo I, S12, Exp 5 (Lab #6) (TECH 710: IR Unknown) LAB REPRT ISTRUCTIS Identifying an Unknown Compound by Infrared Spectroscopy (IR) Carefully read all instructions and complete Section I on page
CHEM 3760 rgo I, S12, Exp 5 (Lab #6) (TECH 710: IR Unknown) LAB REPRT ISTRUCTIS Identifying an Unknown Compound by Infrared Spectroscopy (IR) Carefully read all instructions and complete Section I on page
Differential Scanning Calorimetry
 CH 2252 Instrumental Methods of Analysis Unit III Differential Scanning Calorimetry M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering
CH 2252 Instrumental Methods of Analysis Unit III Differential Scanning Calorimetry M. Subramanian Assistant Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering
Structure Determination. How to determine what compound that you have? One way to determine compound is to get an elemental analysis
 Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
Structure Determination How to determine what compound that you have? ne way to determine compound is to get an elemental analysis -basically burn the compound to determine %C, %H, %, etc. from these percentages
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
CH2407 Process Equipment Design II Reboiler Design. Dr. M. Subramanian
 CH2407 Process Equipment Design II Reboiler Design Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603 110, Kanchipuram
CH2407 Process Equipment Design II Reboiler Design Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam 603 110, Kanchipuram
Chapter 12 Mass Spectrometry and Infrared Spectroscopy
 Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 12 Mass Spectrometry and Infrared Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice
Instrumental Analysis: Spectrophotometric Methods
 Instrumental Analysis: Spectrophotometric Methods 2007 By the end of this part of the course, you should be able to: Understand interaction between light and matter (absorbance, excitation, emission, luminescence,fluorescence,
Instrumental Analysis: Spectrophotometric Methods 2007 By the end of this part of the course, you should be able to: Understand interaction between light and matter (absorbance, excitation, emission, luminescence,fluorescence,
1901 Application of Spectrophotometry
 1901 Application of Spectrophotometry Chemical Analysis Problem: 1 Application of Spectroscopy Organic Compounds Organic compounds with single bonds absorb in the UV region because electrons from single
1901 Application of Spectrophotometry Chemical Analysis Problem: 1 Application of Spectroscopy Organic Compounds Organic compounds with single bonds absorb in the UV region because electrons from single
Chemistry Instrumental Analysis Lecture 15. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 15 IR Instruments Types of Instrumentation Dispersive Spectrophotometers (gratings) Fourier transform spectrometers (interferometer) Single beam Double beam
Chemistry 4631 Instrumental Analysis Lecture 15 IR Instruments Types of Instrumentation Dispersive Spectrophotometers (gratings) Fourier transform spectrometers (interferometer) Single beam Double beam
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups
 Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
Chapter 15 Molecular Luminescence Spectrometry
 Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
Chapter 15 Molecular Luminescence Spectrometry Two types of Luminescence methods are: 1) Photoluminescence, Light is directed onto a sample, where it is absorbed and imparts excess energy into the material
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
2001 Spectrometers. Instrument Machinery. Movies from this presentation can be access at
 2001 Spectrometers Instrument Machinery Movies from this presentation can be access at http://www.shsu.edu/~chm_tgc/sounds/sound.html Chp20: 1 Optical Instruments Instrument Components Components of various
2001 Spectrometers Instrument Machinery Movies from this presentation can be access at http://www.shsu.edu/~chm_tgc/sounds/sound.html Chp20: 1 Optical Instruments Instrument Components Components of various
Symmetric Stretch: allows molecule to move through space
 BACKGROUND INFORMATION Infrared Spectroscopy Before introducing the subject of IR spectroscopy, we must first review some aspects of the electromagnetic spectrum. The electromagnetic spectrum is composed
BACKGROUND INFORMATION Infrared Spectroscopy Before introducing the subject of IR spectroscopy, we must first review some aspects of the electromagnetic spectrum. The electromagnetic spectrum is composed
Chapter 17: Fundamentals of Spectrophotometry
 Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
Chapter 17: Fundamentals of Spectrophotometry Spectroscopy: the science that deals with interactions of matter with electromagnetic radiation or other forms energy acoustic waves, beams of particles such
CH2351 Chemical Engineering Thermodynamics II Unit I, II Phase Equilibria. Dr. M. Subramanian
 CH2351 Chemical Engineering Thermodynamics II Unit I, II Phase Equilibria Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
CH2351 Chemical Engineering Thermodynamics II Unit I, II Phase Equilibria Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
Electro Analytical Methods
 CH 2252 Instrumental Methods of Analysis Unit II Electro Analytical Methods Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
CH 2252 Instrumental Methods of Analysis Unit II Electro Analytical Methods Dr. M. Subramanian Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College of Engineering Kalavakkam
CHAPTER 8 ISOLATION AND CHARACTERIZATION OF PHYTOCONSTITUENTS BY COLUMN CHROMATOGRAPHY
 146 CHAPTER 8 ISLATIN AND CHARACTERIZATIN F PHYTCNSTITUENTS BY CLUMN CHRMATGRAPHY 8.1 INTRDUCTIN Column chromatography is an isolation technique in which the phytoconstituents are being eluted by adsorption.
146 CHAPTER 8 ISLATIN AND CHARACTERIZATIN F PHYTCNSTITUENTS BY CLUMN CHRMATGRAPHY 8.1 INTRDUCTIN Column chromatography is an isolation technique in which the phytoconstituents are being eluted by adsorption.
Lab 6. Use of VSEPR to Predict Molecular Structure and IR Spectroscopy to Identify an Unknown
 Lab 6. Use of VSEPR to Predict Molecular Structure and IR Spectroscopy to Identify an Unknown Prelab Assignment Before coming to lab: In addition to reading introduction of this lab handout, read and understand
Lab 6. Use of VSEPR to Predict Molecular Structure and IR Spectroscopy to Identify an Unknown Prelab Assignment Before coming to lab: In addition to reading introduction of this lab handout, read and understand
Molecular Spectroscopy. H 2 O e -
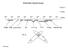 Molecular Spectroscopy ν (cm -1 ) λ (cm) 10 6 10 8 10 10 10 12 10 14 10 16 10 18 10 20 10 22 ν (Hz) NMR ESR microwave IR UV/Vis VUV X-Ray Gamma Ray H 2 e - UV/Vis Spectroscopy absorption technique X hν
Molecular Spectroscopy ν (cm -1 ) λ (cm) 10 6 10 8 10 10 10 12 10 14 10 16 10 18 10 20 10 22 ν (Hz) NMR ESR microwave IR UV/Vis VUV X-Ray Gamma Ray H 2 e - UV/Vis Spectroscopy absorption technique X hν
10/2/2008. hc λ. νλ =c. proportional to frequency. Energy is inversely proportional to wavelength And is directly proportional to wavenumber
 CH217 Fundamentals of Analytical Chemistry Module Leader: Dr. Alison Willows Electromagnetic spectrum Properties of electromagnetic radiation Many properties of electromagnetic radiation can be described
CH217 Fundamentals of Analytical Chemistry Module Leader: Dr. Alison Willows Electromagnetic spectrum Properties of electromagnetic radiation Many properties of electromagnetic radiation can be described
Reference literature. (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters )
 September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
September 17, 2018 Reference literature (See: CHEM 2470 notes, Module 8 Textbook 6th ed., Chapters 13-14 ) Reference.: https://slideplayer.com/slide/8354408/ Spectroscopy Usual Wavelength Type of Quantum
Glossary. Analyte - the molecule of interest when performing a quantitative analysis.
 Glossary This glossary contains definitions of many important FTIR terms. Many of the terms listed here appeared in italics in the body of the book. Words that appear in italics in the glossary are defined
Glossary This glossary contains definitions of many important FTIR terms. Many of the terms listed here appeared in italics in the body of the book. Words that appear in italics in the glossary are defined
VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur
 VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur 603 203 DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING QUESTION BANK V SEMESTER EI6501 Analytical Instruments Regulation 2013 Academic
VALLIAMMAI ENGINEERING COLLEGE SRM Nagar, Kattankulathur 603 203 DEPARTMENT OF ELECTRONICS AND INSTRUMENTATION ENGINEERING QUESTION BANK V SEMESTER EI6501 Analytical Instruments Regulation 2013 Academic
February 8, 2018 Chemistry 328N
 Lecture 7 UV-Vis spectroscopy February 8, 2018 First Midterm Exam When: Wednesday, 2/14 When: 7-9 PM (please do not be late) Where: WEL 2.122 This room!!! What: Covers material through today s lecture
Lecture 7 UV-Vis spectroscopy February 8, 2018 First Midterm Exam When: Wednesday, 2/14 When: 7-9 PM (please do not be late) Where: WEL 2.122 This room!!! What: Covers material through today s lecture
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Molecular Spectroscopy
 Molecular Spectroscopy Types of transitions: 1) Electronic (UV-Vis-Near IR) 2) Vibrational (IR) 3) Rotational (microwave) Electronic Absorption Spectra π π* Gary L. Miessler and Donald A. Tarr, Inorganic
Molecular Spectroscopy Types of transitions: 1) Electronic (UV-Vis-Near IR) 2) Vibrational (IR) 3) Rotational (microwave) Electronic Absorption Spectra π π* Gary L. Miessler and Donald A. Tarr, Inorganic
UNIT 3 CHEMISTRY. Fundamental Principles in Chemistry
 UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
SPECTROPHOTOMETRY AND SPECTROMETRY - CONCEPT AND APPLICATIONS
 SPECTROPHOTOMETRY AND SPECTROMETRY - CONCEPT AND APPLICATIONS Renjini A 1, Dani Dileep 2 1 Assistant Professor, Department of ECE, Rajadhani Institute of Engineering and Technology, Kerala, India 2 PG
SPECTROPHOTOMETRY AND SPECTROMETRY - CONCEPT AND APPLICATIONS Renjini A 1, Dani Dileep 2 1 Assistant Professor, Department of ECE, Rajadhani Institute of Engineering and Technology, Kerala, India 2 PG
Synthesis and Infrared Spectrum of Nitric Oxide 1
 Synthesis and Infrared Spectrum of Nitric Oxide 1 Purpose: Infrared spectroscopy is used to determine the force constant of the bond in nitric oxide. Prelab: Reading: Section 6.1 and 6.2 in Brown, LeMay,
Synthesis and Infrared Spectrum of Nitric Oxide 1 Purpose: Infrared spectroscopy is used to determine the force constant of the bond in nitric oxide. Prelab: Reading: Section 6.1 and 6.2 in Brown, LeMay,
First Law of Thermodynamics
 CH2303 Chemical Engineering Thermodynamics I Unit II First Law of Thermodynamics Dr. M. Subramanian 07-July-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College
CH2303 Chemical Engineering Thermodynamics I Unit II First Law of Thermodynamics Dr. M. Subramanian 07-July-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College
CHAPTER - 3 CHARACTERIZATION OF SINGLE CRYSTALS USING DIFFERENT TECHNIQUES. 3.1 Instrumentations used for characterization
 CHAPTER - 3 CHARACTERIZATION OF SINGLE CRYSTALS USING DIFFERENT TECHNIQUES 3.1 Instrumentations used for characterization Crystals grown using any method of crystal growth have to be characterized for
CHAPTER - 3 CHARACTERIZATION OF SINGLE CRYSTALS USING DIFFERENT TECHNIQUES 3.1 Instrumentations used for characterization Crystals grown using any method of crystal growth have to be characterized for
CHEM*3440. Photon Energy Units. Spectrum of Electromagnetic Radiation. Chemical Instrumentation. Spectroscopic Experimental Concept.
 Spectrum of Electromagnetic Radiation Electromagnetic radiation is light. Different energy light interacts with different motions in molecules. CHEM*344 Chemical Instrumentation Topic 7 Spectrometry Radiofrequency
Spectrum of Electromagnetic Radiation Electromagnetic radiation is light. Different energy light interacts with different motions in molecules. CHEM*344 Chemical Instrumentation Topic 7 Spectrometry Radiofrequency
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee
 Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Analytical Technologies in Biotechnology Prof. Dr. Ashwani K Sharma Department of Biotechnology Indian Institute of Technology, Roorkee Module - 6 Spectroscopic Techniques Lecture - 2 UV-Visible Spectroscopy
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications
 Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Design and Development of a Smartphone Based Visible Spectrophotometer for Analytical Applications Bedanta Kr. Deka, D. Thakuria, H. Bora and S. Banerjee # Department of Physicis, B. Borooah College, Ulubari,
Spectroscopy and Chromatography
 Spectroscopy and Chromatography Introduction Visible light is one very small part of the electromagnetic spectrum. The different properties of the various types of radiation depend upon their wavelength.
Spectroscopy and Chromatography Introduction Visible light is one very small part of the electromagnetic spectrum. The different properties of the various types of radiation depend upon their wavelength.
More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages in your laboratory manual.
 CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
CHEM 3780 rganic Chemistry II Infrared Spectroscopy and Mass Spectrometry Review More information can be found in Chapter 12 in your textbook for CHEM 3750/ 3770 and on pages 13-28 in your laboratory manual.
Unit 3 Organic Chemistry. 3.3 Structural Analysis Part 2:
 Unit 3 Organic Chemistry 3.3 Structural Analysis Part 2: Mass Spectroscopy Infra-red Spectroscopy NMR Proton Spectroscopy Answers to Questions in Notes Learning Outcomes Exam Questions & Answers MODIFIED
Unit 3 Organic Chemistry 3.3 Structural Analysis Part 2: Mass Spectroscopy Infra-red Spectroscopy NMR Proton Spectroscopy Answers to Questions in Notes Learning Outcomes Exam Questions & Answers MODIFIED
Ch 313 FINAL EXAM OUTLINE Spring 2010
 Ch 313 FINAL EXAM OUTLINE Spring 2010 NOTE: Use this outline at your own risk sometimes a topic is omitted that you are still responsible for. It is meant to be a study aid and is not meant to be a replacement
Ch 313 FINAL EXAM OUTLINE Spring 2010 NOTE: Use this outline at your own risk sometimes a topic is omitted that you are still responsible for. It is meant to be a study aid and is not meant to be a replacement
Determining the Structure of an Organic Compound
 Chapter 12- Structure Determination: Mass Spectrometry and Infrared Spectroscopy Ashley Piekarski, Ph.D. Determining the Structure of an Organic Compound The analysis of the outcome of a reac=on requires
Chapter 12- Structure Determination: Mass Spectrometry and Infrared Spectroscopy Ashley Piekarski, Ph.D. Determining the Structure of an Organic Compound The analysis of the outcome of a reac=on requires
Unit 11 Instrumentation. Mass, Infrared and NMR Spectroscopy
 Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place.
 RADIATION INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place. Radiation: The energy emitted by matter in the form
RADIATION INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place. Radiation: The energy emitted by matter in the form
ANALYSIS USING INFRARED SPECTROSCOPIC METHODS
 ANALYSIS USING INFRARED SPECTROSCOPIC METHODS ROBERT A. OETJEN Department of Physics, The Ohio State University, Columbus 10 REQUIREMENTS OF A PHYSICAL-ANALYTICAL METHOD The essential requirements in any
ANALYSIS USING INFRARED SPECTROSCOPIC METHODS ROBERT A. OETJEN Department of Physics, The Ohio State University, Columbus 10 REQUIREMENTS OF A PHYSICAL-ANALYTICAL METHOD The essential requirements in any
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Spectrophotometry. Dr. Shareef SHAIK ASST. PROFESSOR Pharmacology
 Spectrophotometry Dr. Shareef SHAIK ASST. PROFESSOR Pharmacology Content Introduction Beer-Lambert law Instrument Applications Introduction 3 Body fluids such as blood, csf and urine contain organic and
Spectrophotometry Dr. Shareef SHAIK ASST. PROFESSOR Pharmacology Content Introduction Beer-Lambert law Instrument Applications Introduction 3 Body fluids such as blood, csf and urine contain organic and
Solution reduction synthesis of amine terminated carbon quantum dots
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Solution reduction synthesis of amine terminated carbon quantum dots Keith Linehan and Hugh
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Solution reduction synthesis of amine terminated carbon quantum dots Keith Linehan and Hugh
Absorption photometry
 The light Absorption photometry Szilvia Barkó University of Pécs, Faculty of Medicines, Dept. Biophysics February 2011 Transversal wave E Electromagnetic wave electric gradient vector wavelength The dual
The light Absorption photometry Szilvia Barkó University of Pécs, Faculty of Medicines, Dept. Biophysics February 2011 Transversal wave E Electromagnetic wave electric gradient vector wavelength The dual
Lecture 13 Organic Chemistry 1
 EM 232 rganic hemistry I at hicago Lecture 13 rganic hemistry 1 Professor Duncan Wardrop February 23, 2010 1 EM 232 rganic hemistry I at hicago Spectroscopy & Spectrometry hapter 13 2 EM 232 rganic hemistry
EM 232 rganic hemistry I at hicago Lecture 13 rganic hemistry 1 Professor Duncan Wardrop February 23, 2010 1 EM 232 rganic hemistry I at hicago Spectroscopy & Spectrometry hapter 13 2 EM 232 rganic hemistry
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU
 The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
The Fundamentals of Spectroscopy: Theory BUILDING BETTER SCIENCE AGILENT AND YOU 1 Agilent is committed to the educational community and is willing to provide access to company-owned material. This slide
Chapter 4 Scintillation Detectors
 Med Phys 4RA3, 4RB3/6R03 Radioisotopes and Radiation Methodology 4-1 4.1. Basic principle of the scintillator Chapter 4 Scintillation Detectors Scintillator Light sensor Ionizing radiation Light (visible,
Med Phys 4RA3, 4RB3/6R03 Radioisotopes and Radiation Methodology 4-1 4.1. Basic principle of the scintillator Chapter 4 Scintillation Detectors Scintillator Light sensor Ionizing radiation Light (visible,
Preface to the Second Edition. Preface to the First Edition
 Contents Preface to the Second Edition Preface to the First Edition iii v 1 Introduction 1 1.1 Relevance for Climate and Weather........... 1 1.1.1 Solar Radiation.................. 2 1.1.2 Thermal Infrared
Contents Preface to the Second Edition Preface to the First Edition iii v 1 Introduction 1 1.1 Relevance for Climate and Weather........... 1 1.1.1 Solar Radiation.................. 2 1.1.2 Thermal Infrared
Spectroscopic techniques: why, when, where,and how Dr. Roberto GIANGIACOMO
 Spectroscopic techniques: why, when, where,and how Dr. Roberto GIANGIACOMO BASIC INFORMATION Spectroscopy uses light to analyze substances or products by describing the energy transfer between light and
Spectroscopic techniques: why, when, where,and how Dr. Roberto GIANGIACOMO BASIC INFORMATION Spectroscopy uses light to analyze substances or products by describing the energy transfer between light and
Introduction to Fourier Transform Infrared Spectroscopy
 molecular spectroscopy Introduction to Fourier Transform Infrared Spectroscopy Part of Thermo Fisher Scientific Introduction What is FT-IR? FT-IR stands for Fourier Transform InfraRed, the preferred method
molecular spectroscopy Introduction to Fourier Transform Infrared Spectroscopy Part of Thermo Fisher Scientific Introduction What is FT-IR? FT-IR stands for Fourier Transform InfraRed, the preferred method
Spectrophotometry. Introduction
 Spectrophotometry Spectrophotometry is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. The basic principle
Spectrophotometry Spectrophotometry is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. The basic principle
Learning Guide for Chapter 3 - Infrared Spectroscopy
 Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Learning Guide for hapter 3 - Infrared Spectroscopy I. Introduction to spectroscopy - p 1 II. Molecular vibrations - p 3 III. Identifying functional groups - p 6 IV. Interpreting an IR spectrum - p 12
Questions on Instrumental Methods of Analysis
 Questions on Instrumental Methods of Analysis 1. Which one of the following techniques can be used for the detection in a liquid chromatograph? a. Ultraviolet absorbance or refractive index measurement.
Questions on Instrumental Methods of Analysis 1. Which one of the following techniques can be used for the detection in a liquid chromatograph? a. Ultraviolet absorbance or refractive index measurement.
Preview from Notesale.co.uk Page 1 of 38
 F UNDAMENTALS OF PHOTONICS Module 1.1 Nature and Properties of Light Linda J. Vandergriff Director of Photonics System Engineering Science Applications International Corporation McLean, Virginia Light
F UNDAMENTALS OF PHOTONICS Module 1.1 Nature and Properties of Light Linda J. Vandergriff Director of Photonics System Engineering Science Applications International Corporation McLean, Virginia Light
Absorption spectrometry summary
 Absorption spectrometry summary Rehearsal: Properties of light (electromagnetic radiation), dual nature light matter interactions (reflection, transmission, absorption, scattering) Absorption phenomena,
Absorption spectrometry summary Rehearsal: Properties of light (electromagnetic radiation), dual nature light matter interactions (reflection, transmission, absorption, scattering) Absorption phenomena,
VIBRATION-ROTATION SPECTRUM OF CO
 Rice University Physics 332 VIBRATION-ROTATION SPECTRUM OF CO I. INTRODUCTION...2 II. THEORETICAL CONSIDERATIONS...3 III. MEASUREMENTS...8 IV. ANALYSIS...9 April 2011 I. Introduction Optical spectroscopy
Rice University Physics 332 VIBRATION-ROTATION SPECTRUM OF CO I. INTRODUCTION...2 II. THEORETICAL CONSIDERATIONS...3 III. MEASUREMENTS...8 IV. ANALYSIS...9 April 2011 I. Introduction Optical spectroscopy
Spectroscopy. Fourier Transform Infrared (FT-IR) Spectroscopy
 Fourier Transform Infrared (FT-IR) Spectroscopy Learning objectives Learning outcomes After completing this course, the student will be able to: Recognize the concept and principle of FT-IR Spectroscopy
Fourier Transform Infrared (FT-IR) Spectroscopy Learning objectives Learning outcomes After completing this course, the student will be able to: Recognize the concept and principle of FT-IR Spectroscopy
Raman and stimulated Raman spectroscopy of chlorinated hydrocarbons
 Department of Chemistry Physical Chemistry Göteborg University KEN140 Spektroskopi Raman and stimulated Raman spectroscopy of chlorinated hydrocarbons WARNING! The laser gives a pulsed very energetic and
Department of Chemistry Physical Chemistry Göteborg University KEN140 Spektroskopi Raman and stimulated Raman spectroscopy of chlorinated hydrocarbons WARNING! The laser gives a pulsed very energetic and
Topic 2.11 ANALYTICAL TECHNIQUES. High Resolution Mass Spectrometry Infra-red Spectroscopy
 Topic 2.11 ANALYTICAL TECHNIQUES High Resolution Mass Spectrometry Infra-red Spectroscopy HIGH RESOLUTION MASS SPECTROMETRY The technique of mass spectrometry was used in Unit 1 to: a) determine the relative
Topic 2.11 ANALYTICAL TECHNIQUES High Resolution Mass Spectrometry Infra-red Spectroscopy HIGH RESOLUTION MASS SPECTROMETRY The technique of mass spectrometry was used in Unit 1 to: a) determine the relative
Monday 9 September, :30-11:30 Class#03
 Monday 9 September, 2013 10:30-11:30 Class#03 Topics for the hour Solar zenith angle & relationship to albedo Blackbody spectra Stefan-Boltzman Relationship Layer model of atmosphere OLR, Outgoing longwave
Monday 9 September, 2013 10:30-11:30 Class#03 Topics for the hour Solar zenith angle & relationship to albedo Blackbody spectra Stefan-Boltzman Relationship Layer model of atmosphere OLR, Outgoing longwave
