Evidence for Nuclear Reactions in Ni-H Systems I: Heat Excess Measurements
|
|
|
- Herbert Phillips
- 5 years ago
- Views:
Transcription
1 Evidence for Nuclear Reactions in Ni-H Systems I: Heat Excess Measurements S. FOCARDI (I), V. GABBANI F. Pl.-\! "TELLI n, v. YIO!"TALBANO e), S. VERONESI e) (') Dipartimento di Fiaica, Uniuersita di Bologna e INFN ae=ione di Bologna, Bologna " Italy (') Dipm timento di Fisica, Uniuersita di Siena e centro IMO Siena" Italy SUlumary. This is the first of a series of papers in which evidence is given for the occurrence of nuclear reactions during energy production in Ni-H systems. Details of the calorimetric measurements are reported in this paper. Evidence for nuclear emission and presence of nuclear ashes will be reported in the following papers. 1 Introduction In a.previous paper [11 some of us reported on the existence of an anomalous heat production observed.in hydrogen-loaded nickel rods. The phen<jrnenon occurs when a cell containing a nickel rod is maintained at temperatures above a critical value and is filled with gaseous H 2 at subatmospheric pressures. A constant input power was used to raise and keep the cell temperature constant ac its working value (corresponding to about 700 J( for the Ni rod). It was possible to induce an increase of the sample temperature from its working value to about 820 K. This anomalous equilibrium condition will be referred in the following as excited state. The system was able to stay in the excited state for times longer than one month, so producing an excess heat greater than 90 MJ. The experimental cell described in reference [II was successively modified and also a new cell was built with an improvement which allows the measurement and the monitoring of the external surface temperature. _ \Vith this new set UP, the external temperature increase, together with the internal one, have been utilized to characterize the excited state of the Ni sample. The existence of an exothermic effect, whose heat yield is well above that of any known chemical reaction, has been unambiguously confirmed by evaluating the thermal flux coming from the cells. J\Ioreover, in order to check a supposed nuclear origin of the phenomenon we measured the radiation coming from the cells and performed measurements to detect nuclear a,hes. I
2 Evidence of heat excess have been obtained by many authors [21 but almost all of the experimental and theoretical researches in this field is concerned with palladiumdeuterium systems in electrolytic devices. Very little evidence of radiation emission in the experiments was reported. They are normally qualitati,ve and when quantitative [3] are very close to a background level. For the sake of clarity, experimental data are presented in different papers focusing on the following topics: calorimetric measurements, nuclear measurements, tests for nuclear ashes. 2 Experimental apparatus In the following we shall describe two experimental cells which will be referred to cell A and B. Cell A (fig. la) is the same as used in the experiment described in reference [II, B is a new cell. The cell Al containing only one metal rod, is now equipped with a type K chromel-alumel thermocouple having a sensitivity of about 10 p.v/k in the region of interest. One of the two thermocouple junctions is fixed to the external surface, while the reference one is immersed in a water- containing dewar. \rvater temperature and room temperature are continuously recorded. Voltage measurements are performed with multimeters METEX M4650. Cell B (see fig. Ib) is constructed in a similar way. The cell vessel is a stainless steel tube (din = 22 mm, d ext = 28 mm,length = 150 mm) ending with commercia.l vacuum fittings flanges CF35. A second stainless steel tube (din = 34 mm) is placed around this vessel to create an interspace and to allow a fast cooling of the cell, if necessary. The heater, supplied by means of a d.c. voltage stabilized power supply (Alpha Electronica mod. AL81S), is a Ni-Cr wire fastened around a ceramic cylinder (external diameter 21 mm). A «ramic holder keeps four metal rods symmetrically set around the cell axis where the thermocouple: Tc.2 is placed. Due to the high cell working temperature (about 700 K) electrical insulation and gluing inside the cell are done with ceramic bonding. The cell is equipped with three other thermocouples, Tc" Tc" (extermi) and TC3 (internall placed as shown in fig. Ib, to have a more complete temperature map. All the thermocouples are type K, with an accuracy better than 0.3 K; their reference junctions are immersed in a water containing dewar, at a controlled temperature. Electrical feedthroughs are done by.means of a BTJ8 vacuum fitting (from RIAL Parma, Italy) and the cell is connected to the gas handling system by means of a commercial DN2.5,,:acuum fitting with aluminium seal. The pressure inside the cell is measured by a piezomanometer (vvse-vvaldscc Electronic). Data acquisition (temperatures, pressure, voltage and current) is performed by means of a PC (cpu 80486) equipped with a 12 bit National Instrument interface card utilizing a Labview softwan:~. vve have utilized a rotary pump for primary vacuum and a turbomolecular pump for degassing of cells and samples. Fig. lc shows the experimental set up utilized for both the cells. The metal rods used in the experiments here described were coated with a thick {'" 2
3 0.1 mm) nickel layer in an usual nickel-plating bath [41 After the introduction in the cells, the rods were annea!en under vacuum (p < 10-4 mbar) a.t temperatures uij to about 900 K. Successive thermal cycles were also performed in hydrogen atmosphere below 1 bar. 3 Calorimetry OUf experimental apparatuses were mainly built to allow the measurement of the heat production triggered in Ni sample after several thermal cycles in H 2 atmosphere. Each calorimetric cell allows a continuous monitoring of its energy balance by measuring the equilibrium temperature at different cell positions ( see figs. 1 for temperature sensors position). A constant power is supplied to each cell and the power output is obtained from the calibration procedure described below. The presence of an excess heat source is detected by the increase of all the monitored temperatures. In particular, the external wall temperature of the cell depends on its heat exchange coefficient and on the thermal power produced or applied inside the cell. OUf calorimetric cells can be regarded from this point of view as black boxes. At constant external conditions. an equilibrium temperature increase of the wall is related to an extra heat production, if the power input remains unchanged. Let Pin be the electrical power supplied to the cell heater and To the thermal bath temperature (room temperature in our case). At the thermal equilibrium a difference between the temperature of the wall T w (T, for cell.\ and T, for cell B) and To is measured. If A(T) is the heat exchange coefficient between the wall and the bath, the following rela.tion must be verified (1) If some kind of physical-chemical process occurs into the cell and Hi are the related enthalpy variations, the following relation holds (2) where C is the cell heat capacity. Equation (2) is valid if the characteristic times of the heat exchanges inside the cell are shorter than T, = C/ A (cell time constant). II all the physical-chemical processes last for a limited time period before stopping, equation (2) reduces to equation (1) in few cell time constants. In the case for which one process (the j-th) does not stop and Pj = d;, --+ constant, on assuming quasi-stationary conditions, that is that the temperatures of the cell remain practically constant during a few cell time constant, one obtains (3) 3
4 v, here T j is the final wall equilibrium temperature. In our experiments, since the processes are exothermic, P j > 0 and T f > T w. From equation (3) and (1), for the same input power, P j becomes (4) if A can be considered as a constant in the explored temperature range. Thus, the power generated or absorbed by the j-th process occurring in the cell is proportional to the temperature variation of the cell when the new equilibrium temperature T j is reached, at a constant input power. The instrumental constant Acan be obtained from a calibration procedure during which a known power is supplied to the cell and the equilibrium temperatures T w and To are measured. We have measured the temperatures T w by supplying power up to about 150 W for the cell A and up to 70 VV for the cell B in successive steps. Results are shown in figs. 2a and 2b. From the calibrations, it is possible to evaluate the heat exchange coefficient, A...;. = 1.30 ± 0.05 W /F. and AB = 2.71 ± 0.06 W /1( for celfs A and B respectively at a working temperature of the sample (T '" 700 I(). 4 Results and discussion The sample 103tding in a natural hydrogen atmosphere was performed in successive steps. In each step, we started with an initial gas pressure in the range 400-:-800 mbar and thereafter a little amount of hydrogen was introduced into the cell through a suitable valve ("'-p'" mbar). When the pressure decrea~ed down to its starting vo-tue, new hydrogen wo-os ~dded (see fig. 3). After several loading cycles) the sample was ready and it was possible to trigger the exothermic process. Such an operation can be performed by lowering the input power, waiting for the sample temperature to decrease down to about 300 K, then suddenly restoring the previous power level. After this operation an increased equilibrium temperature, as shown in fig. 4, is obtained: the cell is producing an excess heat. Another way to trigger the process is to provoke a pressure step-like variation. as sho~vn in fig. 5. After the triggering procedure, the production ofexcess heat is maintained for moflths. It must be underlined that, once the heat producing process has been started, the cells are kept sealed for all the time, that is no H, or anything else is introduced or extracted from the cell, except heat, of course, while the input power is maintained constant. The initial temperatures are newly obtained when the heat producing process is stopped by following a suitable procedure (see fig. 5 and relative caption). Cell A [nitially on supplying W to the cell the equilibrium temperatures were I( and 1):3.6 I( above To for the sample (Tp,) and the cell (T,) respectively. After the triggering procedure the temperatures showed a sudden increase (to K and 118.0
5 K for Tp, and T, respectively) while the supplied power decreased to W (due to the heater resistance increase). Then, the temperatures slowly increased forcing us to lower the input power twice. The equilibrium was reached at K and K above To for Tp, and T, respectively with a supplied powe,r of W. Summarizing, the input power decrease (48.1 VV) would produce in OUf experimental conditions a decrease of 37 K for Tel while a decrease of only 7.1 K was observed. In spite of the input power reduction, the sample temperature T pt increased by 31.7 K. From the..\.4. value and the supplied power difference, it is possible to evaluate a power excess produced by the sample of 38.9±1.5 W. The power excess from the sample temperature variation, evaluated by the method used in reference [1], gives a value of 68 "V. Such a discrepancy can be explained on taking into account that the sample calibration curve was obtained before the process was triggered. \Vhen the cell is in the excited state, the nickel rod becomes a heat emitter. The thermometer outside the cell probes the total heat, regardless of its origin, while the thermometer close to the nickel feels differently the heat emitted from the coil and that emitted from the sample. Moreover the sample is thermally connected with the outside along the cell axis (see fig. la), and the power measured from the wall temperature does not take into account such contribution. In any case, in the follo\ving, we will refer to the lower value of the power, that is that measured externa.lly. Cell A produced an excess power continuously for 278 days and it increased slowly during this-period. Before the shut-down, this cell was working with an input po\ver of 94.3 \;V and the sample temperature Tpt of K above To. Moreover as a further proof of an extra power in the nickel rod we performed the following measurement: after the shut-down the same working temperature was obtained by supplying \V. Cell B The behaviour of this cell wa~ very similar to that of cell A. In the experimental conditions the input power "vas 61.8 \;V and the equilibrium temperatures (above the ambient temperature To) K, K, K, 22.9 K for T" T" T 3 and T. respectively. After the triggering process these temperatures increased up to K, K, K and 31.4 K respectively, at a constant input power. From the calibration data it is possible to evaluate an excess power, in the cell, of 23.0±1.3 VV (from T~). By utilizing the T 1 calibration curves it is possible to give a rough evaluation of ~ IS \V extra power. Fig. 6 shows the temperature T 4 versus the input power and the experimental points obtained when the exothermic process was active. Vie can observe that the excited state exists in a wide temperature range. The excited sta.te curve becomes nearer to the calibration one when the power is lower, as already shown in reference [1]. The mean power excess of each cell multiplied by its activity period (278 days for cell A and 319 days for cell B) gives an energy excess of about 900 MJ for cell A and 600 l\[j for cell B. Such an energy amount cannot be accounted for any chemical reaction occurring inside the cells.
6 In conclusion we can say that all data coherently support the evidence of an excess heat produced by the sample when it is in the physical conditions provoked by the annealing and storing in a H 2 atmosphere at high temperature as we have described. Aknowledgements We wish thank Prof. C. Salvetti, Dr. E. Tombari, Prof. C. Stremmenos, Dr. V. Palleschi, Dr. E. Tognoni and Dr. C. Ferrari for helpful suggestions and discussions. Special thanks are also due to Prof. R. Habel, Dr. E. Majorana Dr. I. Usai and Ing.!\I. Pianigiani for their collaboration in the initial phase of the experiment. References [11 FOCARDI S., HABEL R.. and PIANTEI.I.1 F., Nfl""" Cim. A, 107 (1994) 163. [2] see, for instance, Proceeding of the Jh international conference on cold fusion, Monte-Carlo, Monaco, 9-15 Apri/1995 and references therein [:31 SANKARANARAYANAN T. K., SRINIVASAN M., BAJPAI Ivi. B., and GUPTA D. S., Proceeding of the Sh international conference on cold fusion, ly/onte-carlo, Monaco, 9-15 Apri/1995, (199-5) 173; SHYAM A., SRINIVASAN M., KAUSHIK T. C. and KULKARNI L. V., P1'Oceeding of the st h international conference on cold jllsion, Monte-Carlo, Monaco, 9-15 April 1995, (199.1) [41 BERTORELLE E., Tratt"to di g>li""notecnica, vol 1, ed. Hoepli (19i1). FIGURE CAPTIONS Fig.-la Schematic drawing of cell A. Tee and Thpl show the temperature sensor positions. Fig.-lb Schematic drawing of cell B. TC11 Tc1., TC3 and TC4 show the thermocouples positions. Fig.-lc Experimental set up. Fig.-2a Cell A. Calibration curves; temperature (relative ';0 the room temperatlire) vs polyer ( Tp,(o) and T,(o)). - Fig.-2b Cell B. Calibration curves; temperature (relative to the room temperature) vs polyer (T,(o), T,(o), T 3 (0) and T,(*). Fig.-3 A typical hydrogen loading of a sample. The plot refers to cell B. The experimental data can be fitted with an exponential law. The characteristic time for this data set is T = 72 minutes.. Fig.-4 A sample excitation performed with a temperatllre jllmp (71(0), 7,(0), T 3 (0)). An inversion between T'l and T 3 can be observed. Such an effect is due to the extra power produced by the nickel rod. 6
7 Fig.-5 A sample excitation (three negative pressure jumps) and a successive deexcitation (vacuum followed by a pressure jump) for cell B (T,(o), To(')' T 3 (O)). The detail of the excitation is shown in the square. Fig.-6 T, (relative to the room temperature) vs power for.cell B. The upper points(o) refer to the excited state; the lower curve is the calibration one. -,
8 Cu Jjfr 1'h,., Te cell vessel external SS tube CF'35 flanges heater ceramic cylinder sample holder PllOO temperalure sensor lype J{ thermocouple 3 Te. 4 3 'l ~~""""'~''''''~ f-' 1 ('"/6'..1. A electrical ~o..noo.."". \','I 1- C\-
9 cell vp~<:pl exlernal 55 lube CF'35 nanges healer ceramic cylinder un Ga,, type K lhermocouple Te. ~ 3 1, 3 lj l -\._--,,:.,.._.. II., II ,!"":::::T"""" ';;'" ':.:.rff' I, Te, 1. "..,.. _ 1- J7lv, 1.,8 t:1~cl"c.1 Connecllons
10 ROTARY PUMP I X X I Y 1>< TURBOMOLECULAR PUMP IX I,..-- L PRESSURE 1>< I I EXP. CELL I GAUG' E il POWER SUPPLY I PC. I U.PS. POWER LINE 1-c
11 '"T [K] 500,---,---,---,----,------, , o o o P [Wj FIG'. d A '!'d- ) - CZ-
12 400!1T [K] o o P [W] FIG-. <1.13 +J- 1- b
13 ~ '- 950 (1J -0 E CL , r-.----,-,.----r-.--,,--,--r-,.----,,-, ,-,----,,-, a y/6-3 t [min] h -3
14 , 400 ~T [K] o t [min] FI6'. tf +~ 4
15 tit [K] l- I- 1 '1~ :~ 300 ~ ". '00 r )f ~ '" ~ l.,~ ~- m '" m '" '00,,,,., o t [min] FIG- '- -1'<'), 5
16 40, , '"T [K] o 30 o o P[W] 80 FIG- 6
Figure 1: Two pressure shocks causing the sample temperature increase (Termocouple Tc1, figure 1b of reference [2]). 2 Heat production Apart from the
![Figure 1: Two pressure shocks causing the sample temperature increase (Termocouple Tc1, figure 1b of reference [2]). 2 Heat production Apart from the Figure 1: Two pressure shocks causing the sample temperature increase (Termocouple Tc1, figure 1b of reference [2]). 2 Heat production Apart from the](/thumbs/91/107515788.jpg) Ni-H Systems E.G. Campari(1), S. Focardi (1), V. Gabbani (2), V. Montalbano (2), F. Piantelli (2), E. Porcu (3), E. Tosti (3) and S. Veronesi (2) ( 1 ) Dipartimento di Fisica, Universitá di Bologna, Bologna
Ni-H Systems E.G. Campari(1), S. Focardi (1), V. Gabbani (2), V. Montalbano (2), F. Piantelli (2), E. Porcu (3), E. Tosti (3) and S. Veronesi (2) ( 1 ) Dipartimento di Fisica, Universitá di Bologna, Bologna
Evidence of electromagnetic radiation from Ni-H Systems
 Focardi, S., et al. Evidence of electromagnetic radiation from Ni-H Systems. in Eleventh International Conference on Condensed Matter Nuclear Science. 2004. Marseille, France. Evidence of electromagnetic
Focardi, S., et al. Evidence of electromagnetic radiation from Ni-H Systems. in Eleventh International Conference on Condensed Matter Nuclear Science. 2004. Marseille, France. Evidence of electromagnetic
Photon and particle emission, heat production and surface transformation in Ni-H system
 Campari, E.G., et al. Photon and particle emission, heat production and surface transformation in Ni-H system. in Eleventh International Conference on Condensed Matter Nuclear Science. 2004. Marseille,
Campari, E.G., et al. Photon and particle emission, heat production and surface transformation in Ni-H system. in Eleventh International Conference on Condensed Matter Nuclear Science. 2004. Marseille,
Investigation of anomalous heat production in Ni-H systems
 Cerron-Zeballos, E., et al., Investigation of anomalous heat production in Ni-H systems. Nuovo Cimento Soc. Ital. Fis. A, 1996. 109A: p. 1645. Investigation of anomalous heat production in Ni-H systems
Cerron-Zeballos, E., et al., Investigation of anomalous heat production in Ni-H systems. Nuovo Cimento Soc. Ital. Fis. A, 1996. 109A: p. 1645. Investigation of anomalous heat production in Ni-H systems
On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness
 JET P(98)57 P A eladarakis W Obert On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness This document is intended for publication in the open literature.
JET P(98)57 P A eladarakis W Obert On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness This document is intended for publication in the open literature.
Surface Analysis of hydrogen loaded nickel alloys
 Surface Analysis of hydrogen loaded nickel alloys E. Campari *, S. Focardi *, V. Gabbani **, V. Montalbano **, F. Piantelli **, S. Veronesi +** * Dipartimento di fisica, Università di Bologna Centro I.M.O.
Surface Analysis of hydrogen loaded nickel alloys E. Campari *, S. Focardi *, V. Gabbani **, V. Montalbano **, F. Piantelli **, S. Veronesi +** * Dipartimento di fisica, Università di Bologna Centro I.M.O.
Laboratory 12: Three Thermodynamics Experiments
 Laboratory 12: Three Thermodynamics Experiments Experiment 1: Coefficient of Linear Expansion of Metals The fact that most objects expand when heated is common knowledge. The change in the linear dimensions
Laboratory 12: Three Thermodynamics Experiments Experiment 1: Coefficient of Linear Expansion of Metals The fact that most objects expand when heated is common knowledge. The change in the linear dimensions
Temperature Scales. Temperature, and Temperature Dependent on Physical Properties. Temperature. Temperature Scale
 Temperature Scales The Celsius, Fahrenheit, and Kelvin Temperature Scales: Temperature, and Temperature Dependent on Physical Properties Physics Enhancement Programme Dr. M.H. CHAN, HKBU 9 T F T 5 T T
Temperature Scales The Celsius, Fahrenheit, and Kelvin Temperature Scales: Temperature, and Temperature Dependent on Physical Properties Physics Enhancement Programme Dr. M.H. CHAN, HKBU 9 T F T 5 T T
He II Heat transfer through a Corrugated Tube - Test Report
 He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
Experiment 1. Measurement of Thermal Conductivity of a Metal (Brass) Bar
 Experiment 1 Measurement of Thermal Conductivity of a Metal (Brass) Bar Introduction: Thermal conductivity is a measure of the ability of a substance to conduct heat, determined by the rate of heat flow
Experiment 1 Measurement of Thermal Conductivity of a Metal (Brass) Bar Introduction: Thermal conductivity is a measure of the ability of a substance to conduct heat, determined by the rate of heat flow
CORRELATED MEASUREMENTS OF D 2 LOADING AND 4 He PRODUCTION IN Pd LATTICE
 Iazzi, F., et al. Correlated Measurements of D2 Loading and 4He Production in Pd Lattice. in The Seventh International Conference on Cold Fusion. 1998. Vancouver, Canada: ENECO, Inc., Salt Lake City, UT.
Iazzi, F., et al. Correlated Measurements of D2 Loading and 4He Production in Pd Lattice. in The Seventh International Conference on Cold Fusion. 1998. Vancouver, Canada: ENECO, Inc., Salt Lake City, UT.
PAPER 2 THEORY QUESTIONS
 PAPER 2 THEORY QUESTIONS 1 Fig. 1.1 shows the arrangement of atoms in a solid block. Fig. 1.1 (a) End X of the block is heated. Energy is conducted to end Y, which becomes warm. (i) Explain how heat is
PAPER 2 THEORY QUESTIONS 1 Fig. 1.1 shows the arrangement of atoms in a solid block. Fig. 1.1 (a) End X of the block is heated. Energy is conducted to end Y, which becomes warm. (i) Explain how heat is
Department of Mechanical Engineering ME 96. Free and Forced Convection Experiment. Revised: 25 April Introduction
 CALIFORNIA INSTITUTE OF TECHNOLOGY Department of Mechanical Engineering ME 96 Free and Forced Convection Experiment Revised: 25 April 1994 1. Introduction The term forced convection refers to heat transport
CALIFORNIA INSTITUTE OF TECHNOLOGY Department of Mechanical Engineering ME 96 Free and Forced Convection Experiment Revised: 25 April 1994 1. Introduction The term forced convection refers to heat transport
NUCLEAR TRANSMUTATION IN DEUTERED PD FILMS IRRADIATED BY AN UV LASER
 Castellano, et al. Nuclear Transmutation in Deutered Pd Films Irradiated by an UV Laser. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna,
Castellano, et al. Nuclear Transmutation in Deutered Pd Films Irradiated by an UV Laser. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna,
Measurements of temperature on LHC thermal models
 Measurements of temperature on LHC thermal models Christine Darve 1, Juan Casas 2, Moyses Kuchnir 1 1 : Fermi National Accelerator Laboratory, Batavia, IL, USA 2 : CERN, European Laboratory for Particle
Measurements of temperature on LHC thermal models Christine Darve 1, Juan Casas 2, Moyses Kuchnir 1 1 : Fermi National Accelerator Laboratory, Batavia, IL, USA 2 : CERN, European Laboratory for Particle
Thermochemistry/Calorimetry. Determination of the enthalpy of combustion with a calorimetric bomb LEC 02. What you need:
 LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
Earlier Lecture. In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide.
 41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
Evidence for Tritium generation in Self-heated Nickel Wires subjected To Hydrogen Gas Absroption/Desorption Cycles
 Sankaranarayanan, T.K., et al. Evidence for Tritium Generation in Self-Heated Nickel Wires Subjected to Hydrogen Gas Absorption/Desorption Cycles. in 5th International Conference on Cold Fusion. 1995.
Sankaranarayanan, T.K., et al. Evidence for Tritium Generation in Self-Heated Nickel Wires Subjected to Hydrogen Gas Absorption/Desorption Cycles. in 5th International Conference on Cold Fusion. 1995.
TRITIUM PRODUCTION IN PALLADIUM DEUTERIDE/HYDRIDE IN EVACUATED CHAMBER
 Yamada, H., et al. Tritium Production in Palladium Deuteride/Hydride in Evacuated Chamber. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna,
Yamada, H., et al. Tritium Production in Palladium Deuteride/Hydride in Evacuated Chamber. in 8th International Conference on Cold Fusion. 2000. Lerici (La Spezia), Italy: Italian Physical Society, Bologna,
Introduction to Blackbody Sources
 Introduction to s This section contains dedicated blackbody sources for low uncertainty calibration of infrared thermometers. A range of portable primary blackbody sources combine high emissivity with
Introduction to s This section contains dedicated blackbody sources for low uncertainty calibration of infrared thermometers. A range of portable primary blackbody sources combine high emissivity with
Sensors and Actuators Sensors Physics
 Sensors and ctuators Sensors Physics Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC SENSORS (Chapter 3.9, 16.4) 3 Thermoelectric effect thermoelectric
Sensors and ctuators Sensors Physics Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC SENSORS (Chapter 3.9, 16.4) 3 Thermoelectric effect thermoelectric
Veerapong Kanchanawongkul*
 Using LabVIEW to Development of Temperature Measurement System with Thermocouple and Thermistor AIS 08 Veerapong Kanchanawongkul* Department of Mechanical Engineering, Faculty of Engineering, South-East
Using LabVIEW to Development of Temperature Measurement System with Thermocouple and Thermistor AIS 08 Veerapong Kanchanawongkul* Department of Mechanical Engineering, Faculty of Engineering, South-East
Semiconductor thermogenerator
 Semiconductor thermogenerator LEP 4.1.07 Related topics Seebeck effect (thermoelectric effect), thermoelectric e.m.f., efficiency, Peltier coefficient, Thomson coefficient, Seebeck coefficient, direct
Semiconductor thermogenerator LEP 4.1.07 Related topics Seebeck effect (thermoelectric effect), thermoelectric e.m.f., efficiency, Peltier coefficient, Thomson coefficient, Seebeck coefficient, direct
Reproduction of Fleischmann and Pons experiments
 onchampt, G., L. Bonnetain, and P. Hieter. Reproduction of Fleischmann and Pons Experiments. in Sixth International Conference on Cold Fusion, Progress in New Hydrogen Energy. 1996. Lake Toya, Hokkaido,
onchampt, G., L. Bonnetain, and P. Hieter. Reproduction of Fleischmann and Pons Experiments. in Sixth International Conference on Cold Fusion, Progress in New Hydrogen Energy. 1996. Lake Toya, Hokkaido,
Thermal Interface Material Performance Measurement
 Thermal Interface Material Performance Measurement Long Win Science & Technology Co., Ltd. www.longwin.com longwin@longwin.com 886-3-4643221 886-3-4986875 2007/07/16 Contents 1. Introduction Heat Transfer
Thermal Interface Material Performance Measurement Long Win Science & Technology Co., Ltd. www.longwin.com longwin@longwin.com 886-3-4643221 886-3-4986875 2007/07/16 Contents 1. Introduction Heat Transfer
Thermal Coatings for In-vacuum Radiation Cooling LIGO-T C R. Abbott, S. Waldman, Caltech 12 March, 2007
 Thermal Coatings for In-vacuum Radiation Cooling LIGO-T070054-00-C R. Abbott, S. Waldman, Caltech 12 March, 2007 1. Overview and Background 1.1. There are instances in LIGO where the use of electronics
Thermal Coatings for In-vacuum Radiation Cooling LIGO-T070054-00-C R. Abbott, S. Waldman, Caltech 12 March, 2007 1. Overview and Background 1.1. There are instances in LIGO where the use of electronics
A simple calorimetric method to avoid artifacts in a controversial field: the ice calorimeter
 Dufour, J., et al. A simple calorimetric method to avoid artifacts in a controversial field: The ice calorimeter. in ICCF-14 International Conference on Condensed Matter Nuclear Science. 2008. Washington,
Dufour, J., et al. A simple calorimetric method to avoid artifacts in a controversial field: The ice calorimeter. in ICCF-14 International Conference on Condensed Matter Nuclear Science. 2008. Washington,
Yasuhiro Iwamura, Takehiko Itoh, Nobuaki Gotoh and Ichiro Toyoda
 Iwamura, Y., et al. Correlation between behavior of deuterium in palladium and occurance of nuclear reactions observed by simultaneous measurement of excess heat and nuclear products. in Sixth International
Iwamura, Y., et al. Correlation between behavior of deuterium in palladium and occurance of nuclear reactions observed by simultaneous measurement of excess heat and nuclear products. in Sixth International
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
ME 105 Mechanical Engineering Laboratory Spring Quarter Experiment #2: Temperature Measurements and Transient Conduction and Convection
 ME 105 Mechanical Engineering Lab Page 1 ME 105 Mechanical Engineering Laboratory Spring Quarter 2010 Experiment #2: Temperature Measurements and Transient Conduction and Convection Objectives a) To calibrate
ME 105 Mechanical Engineering Lab Page 1 ME 105 Mechanical Engineering Laboratory Spring Quarter 2010 Experiment #2: Temperature Measurements and Transient Conduction and Convection Objectives a) To calibrate
Confirmation of Heat Generation during Hydrogenation of Oil
 Session 7 O_8 Material Science Confirmation of Heat Generation during Hydrogenation of Oil Tadahiko Mizuno Graduate School of Engineering, Hokkaido University, Division of Material Chemistry Kita-ku Kita
Session 7 O_8 Material Science Confirmation of Heat Generation during Hydrogenation of Oil Tadahiko Mizuno Graduate School of Engineering, Hokkaido University, Division of Material Chemistry Kita-ku Kita
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
Physics Module Form 4 Chapter 4 - Heat GCKL 2010 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. (, Temperature ) is a form of energy that flows from a hot body to a cold body.
Measurements & Instrumentation. Module 3: Temperature Sensors
 Measurements & Instrumentation PREPARED BY Academic Services Unit August 2013 Institute of Applied Technology, 2013 Module Objectives Upon successful completion of this module, students should be able
Measurements & Instrumentation PREPARED BY Academic Services Unit August 2013 Institute of Applied Technology, 2013 Module Objectives Upon successful completion of this module, students should be able
Put sufficient ice cubes into water (1 M) and wait for equilibrium (both exist) (1 M)
 NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
NAME : F.5 ( ) Marks: /70 FORM FOUR PHYSICS REVISION TEST on HEAT Allowed: 70 minutes This paper consists of two sections. Section A (50 marks) consists of the structure-type questions, and Section B (20
Sensing, Computing, Actuating
 Sensing, Computing, ctuating Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC EFFECT (Chapter 5.11) 3 Thermocouple cylinder head temperature (thermocouple)
Sensing, Computing, ctuating Sander Stuijk (s.stuijk@tue.nl) Department of Electrical Engineering Electronic Systems 2 THERMOELECTRIC EFFECT (Chapter 5.11) 3 Thermocouple cylinder head temperature (thermocouple)
15. Compare the result with the value you have taken above Compare the calculated pressure value with the actual pressure value that you have
 105) to convert it to. 15. Compare the result with the value you have taken above. 17. 16. Compare the calculated pressure value with the actual pressure value that you have taken from the, is it the same?
105) to convert it to. 15. Compare the result with the value you have taken above. 17. 16. Compare the calculated pressure value with the actual pressure value that you have taken from the, is it the same?
Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens
 Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens T. Dasgupta and A. M. Umarji a Materials Research Centre, Indian Institute of Science, Bangalore-560012,
Apparatus to measure high-temperature thermal conductivity and thermoelectric power of small specimens T. Dasgupta and A. M. Umarji a Materials Research Centre, Indian Institute of Science, Bangalore-560012,
Critical parameters of
 Critical parameters of superconductors 2005-03-30 Why do this experiment? Superconductivity is a very interesting property from both commercial and basic scientific points of view. Superconductors are
Critical parameters of superconductors 2005-03-30 Why do this experiment? Superconductivity is a very interesting property from both commercial and basic scientific points of view. Superconductors are
1. How much heat was needed to raise the bullet to its final temperature?
 Name: Date: Use the following to answer question 1: A 0.0500-kg lead bullet of volume 5.00 10 6 m 3 at 20.0 C hits a block that is made of an ideal thermal insulator and comes to rest at its center. At
Name: Date: Use the following to answer question 1: A 0.0500-kg lead bullet of volume 5.00 10 6 m 3 at 20.0 C hits a block that is made of an ideal thermal insulator and comes to rest at its center. At
Observation of High Multiplicity Bursts of Neutrons During Electrolysis of Heavy Water with Palladium Cathode Using the Dead-Time Filtering Technique
 Shyam, A., et al. Observation of High Multiplicity Bursts of Neutrons During Electrolysis of Heavy Water with Palladium Cathode Using the Dead-Time Filtering Technique. in 5th International Conference
Shyam, A., et al. Observation of High Multiplicity Bursts of Neutrons During Electrolysis of Heavy Water with Palladium Cathode Using the Dead-Time Filtering Technique. in 5th International Conference
A New Energy caused by Spillover-Deuterium
 Arata, Y. and Y.C. Zhang, A new energy caused by "Spillover-deuterium". Proc. Jpn. Acad., Ser. B, 1994. 70 ser. B: p. 106. A New Energy caused by Spillover-Deuterium By Yoshiaki ARATA, M. J. A., and Yue-Chang
Arata, Y. and Y.C. Zhang, A new energy caused by "Spillover-deuterium". Proc. Jpn. Acad., Ser. B, 1994. 70 ser. B: p. 106. A New Energy caused by Spillover-Deuterium By Yoshiaki ARATA, M. J. A., and Yue-Chang
Chapter 11. Energy in Thermal Processes
 Chapter 11 Energy in Thermal Processes Energy Transfer When two objects of different temperatures are placed in thermal contact, the temperature of the warmer decreases and the temperature of the cooler
Chapter 11 Energy in Thermal Processes Energy Transfer When two objects of different temperatures are placed in thermal contact, the temperature of the warmer decreases and the temperature of the cooler
THIN FLEXIBLE POLYMER SUBSTRATES COATED BY THICK FILMS IN ROLL-TO-ROLL VACUUM
 ARCOTRONICS INDUSTRIES SpA Via San Lorenzo, 19 40037 Sasso Marconi (BO) Italy Tel. (+39) 051939111 Fax (+39) 051840684 http://www.arcotronics.com THIN FLEXIBLE POLYMER SUBSTRATES COATED BY THICK FILMS
ARCOTRONICS INDUSTRIES SpA Via San Lorenzo, 19 40037 Sasso Marconi (BO) Italy Tel. (+39) 051939111 Fax (+39) 051840684 http://www.arcotronics.com THIN FLEXIBLE POLYMER SUBSTRATES COATED BY THICK FILMS
Lecture 36: Temperatue Measurements
 Lecture 36: Temperatue Measurements Contents Principle of thermocouples Materials for themocouples Cold junction compensation Compensating wires Selection of thermocouples Illustration of gas temperature
Lecture 36: Temperatue Measurements Contents Principle of thermocouples Materials for themocouples Cold junction compensation Compensating wires Selection of thermocouples Illustration of gas temperature
Thermocouples calibration and analysis of the influence of the length of the sensor coating
 Journal of Physics: Conference Series OPEN ACCESS Thermocouples calibration and analysis of the influence of the length of the sensor coating To cite this article: M Noriega et al 2015 J. Phys.: Conf.
Journal of Physics: Conference Series OPEN ACCESS Thermocouples calibration and analysis of the influence of the length of the sensor coating To cite this article: M Noriega et al 2015 J. Phys.: Conf.
Solar Flat Plate Thermal Collector
 Solar Flat Plate Thermal Collector INTRODUCTION: Solar heater is one of the simplest and basic technologies in the solar energy field. Collector is the heart of any solar heating system. It absorbs and
Solar Flat Plate Thermal Collector INTRODUCTION: Solar heater is one of the simplest and basic technologies in the solar energy field. Collector is the heart of any solar heating system. It absorbs and
Electricity and Energy 1 Content Statements
 Keep this in good condition, it will help you pass your final exams. The school will only issue one paper copy per pupil. An e-copy will be placed on the school s web-site. Electricity and Energy 1 Content
Keep this in good condition, it will help you pass your final exams. The school will only issue one paper copy per pupil. An e-copy will be placed on the school s web-site. Electricity and Energy 1 Content
Thermal and electrical conductivity of metals
 Thermal and electrical conductivity of metals (Item No.: P2350200) Curricular Relevance Area of Expertise: Physics Education Level: University Topic: Thermodynamics Subtopic: Heat, Work, and the First
Thermal and electrical conductivity of metals (Item No.: P2350200) Curricular Relevance Area of Expertise: Physics Education Level: University Topic: Thermodynamics Subtopic: Heat, Work, and the First
5.5. Calibration and Test Procedures
 5.5. Calibration and Test Procedures - List of Calibration Procedures 1. C-18-010-2000, Calibration procedure of melting point measuring apparatus 2. C-18-011-2000, Calibration procedure of calorimeter
5.5. Calibration and Test Procedures - List of Calibration Procedures 1. C-18-010-2000, Calibration procedure of melting point measuring apparatus 2. C-18-011-2000, Calibration procedure of calorimeter
RECORD AND ANALYSE THE PRESSURE-ENTHALPY DIAGRAM FOR A COMPRESSION HEAT PUMP
 Thermodynamics Heat cycles Heat Pump RECORD AND ANALYSE THE PRESSURE-ENTHALPY DIAGRAM FOR A COMPRESSION HEAT PUMP Demonstrate how an electric compression heat pump works Quantitatively investigate of the
Thermodynamics Heat cycles Heat Pump RECORD AND ANALYSE THE PRESSURE-ENTHALPY DIAGRAM FOR A COMPRESSION HEAT PUMP Demonstrate how an electric compression heat pump works Quantitatively investigate of the
Thermal design of the CFRP support struts for the spatial framework of the Herschel Space Observatory
 Thermal design of the CFRP support struts for the spatial framework of the Herschel Space Observatory McDonald P.C.* Jaramillo E.** and B. Baudouy*** *Institute of Cryogenics, School of Engineering Sciences,
Thermal design of the CFRP support struts for the spatial framework of the Herschel Space Observatory McDonald P.C.* Jaramillo E.** and B. Baudouy*** *Institute of Cryogenics, School of Engineering Sciences,
Schematic for resistivity measurement
 Module 9 : Experimental probes of Superconductivity Lecture 1 : Experimental probes of Superconductivity - I Among the various experimental methods used to probe the properties of superconductors, there
Module 9 : Experimental probes of Superconductivity Lecture 1 : Experimental probes of Superconductivity - I Among the various experimental methods used to probe the properties of superconductors, there
Renewable Energy. Theory: The Ideal Gas Law The equation of state for an ideal gas is written: PV = nrt
 Lab 3 Gas Laws and Heat Engines Fall 2010 Introduction/Purpose: In this exercise you will test some of the aspects of the ideal gas law under conditions of constant pressure, constant temperature, and
Lab 3 Gas Laws and Heat Engines Fall 2010 Introduction/Purpose: In this exercise you will test some of the aspects of the ideal gas law under conditions of constant pressure, constant temperature, and
ANOMALOUS HEAT FLOW AND ITS CORRELATION WITH DEUTERIUM FLUX IN A GAS-LOADING DEUTERIUM-PALLADIUM SYSTEM
 Tian, J., et al. Anomalous heat flow and its correlation with deuterium flux in a gasloading deuteriumpalladium system. in The 9th International Conference on Cold Fusion, Condensed Matter Nuclear Science.
Tian, J., et al. Anomalous heat flow and its correlation with deuterium flux in a gasloading deuteriumpalladium system. in The 9th International Conference on Cold Fusion, Condensed Matter Nuclear Science.
EXCESS HEAT AND HEAT AFTER DEATH IN A GAS- LOADING HYDROGEN/PALLADIUM SYSTEM
 Tian, J., et al. "Excess heat" and "heat after death" in a gas loading hydrogen/palladium system. in The 9th International Conference on Cold Fusion, Condensed Matter Nuclear Science. 2002. Tsinghua Univ.,
Tian, J., et al. "Excess heat" and "heat after death" in a gas loading hydrogen/palladium system. in The 9th International Conference on Cold Fusion, Condensed Matter Nuclear Science. 2002. Tsinghua Univ.,
Chapter 1 Heating Processes
 Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
EVIDENCE FOR PRODUCTION OF TRITIUM VIA COLD FUSION REACTIONS IN DEUTERIUM GAS LOADED PALLADIUM
 Krishnan, M.S., et al., Evidence for Production of Tritium via Cold Fusion Reactions in Deuterium Gas Loaded Palladium, in BARC Studies in Cold Fusion, P.K. Iyengar and M. Srinivasan, Editors. 1989, Atomic
Krishnan, M.S., et al., Evidence for Production of Tritium via Cold Fusion Reactions in Deuterium Gas Loaded Palladium, in BARC Studies in Cold Fusion, P.K. Iyengar and M. Srinivasan, Editors. 1989, Atomic
Thermocouple Calibrations and Heat Transfer Coefficients
 Laboratory Experiment 5: Thermocouple Calibrations and Heat Transfer Coefficients Presented to the University of California, San Diego Department of Mechanical and Aerospace Engineering MAE 170 Prepared
Laboratory Experiment 5: Thermocouple Calibrations and Heat Transfer Coefficients Presented to the University of California, San Diego Department of Mechanical and Aerospace Engineering MAE 170 Prepared
Determination of freezing points of pure substances with Cobra4 TEC
 Determination of freezing points of pure substances TEC Related concept Crystallization point, Gibbs free energy, enthalpy, entropy, heat of fusion, freezing point depression. Principle When a pure substance
Determination of freezing points of pure substances TEC Related concept Crystallization point, Gibbs free energy, enthalpy, entropy, heat of fusion, freezing point depression. Principle When a pure substance
Cryogenic Instrumentation I Thermometry OUTLINE Thermometry Pt (pure metal) Temperature Ranges of Thermometer Application Typical Resistive Thermal
 Cryogenic Instrumentation I 1. Thermometry 2. anges of Application 3. Constant Volume 4. Thermocouples 5. Time esponse Data 6. 4 Terminal esistance Measurement OUTLINE 8. Pt (pure metal) 9. Typical esistive
Cryogenic Instrumentation I 1. Thermometry 2. anges of Application 3. Constant Volume 4. Thermocouples 5. Time esponse Data 6. 4 Terminal esistance Measurement OUTLINE 8. Pt (pure metal) 9. Typical esistive
THERMOCOUPLE CHARACTERISTICS TRAINER
 THERMOCOUPLE CHARACTERISTICS TRAINER (Model No : ) User Manual Version 2.0 Technical Clarification /Suggestion : / Technical Support Division, Vi Microsystems Pvt. Ltd., Plot No :75,Electronics Estate,
THERMOCOUPLE CHARACTERISTICS TRAINER (Model No : ) User Manual Version 2.0 Technical Clarification /Suggestion : / Technical Support Division, Vi Microsystems Pvt. Ltd., Plot No :75,Electronics Estate,
Thermal Capacity Measurement of Engineering Alloys in Dependence on Temperature.
 Thermal Capacity Measurement of Engineering Alloys in Dependence on Temperature. Z. Jedlicka, 1 I. Herzogova 2 ABSTRACT Thermophysical laboratory at Department of Thermal Engineering of Technical University
Thermal Capacity Measurement of Engineering Alloys in Dependence on Temperature. Z. Jedlicka, 1 I. Herzogova 2 ABSTRACT Thermophysical laboratory at Department of Thermal Engineering of Technical University
A 3 Vapor pressure of volatile liquids
 Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
Versuchsanleitungen zum Praktikum Physikalische Chemie für Anfänger 1 A 3 Vapor pressure of volatile liquids Purpose The purpose of this experiment is to determine the vaporization enthalpy, entropy and
QPR No. 68 RADIO PHYSICS I. PHYSICAL ELECTRONICS. Prof. W. B. Nottingham J. L. Coggins B. L. Blackford L. E. Sprague RESEARCH OBJECTIVES
 RADIO PHYSICS I. PHYSICAL ELECTRONICS RESEARCH OBJECTIVES Prof. W. B. Nottingham J. L. Coggins B. L. Blackford L. E. Sprague 1. Theory of Energy Conversion Electronics The effectiveness of energy conversion
RADIO PHYSICS I. PHYSICAL ELECTRONICS RESEARCH OBJECTIVES Prof. W. B. Nottingham J. L. Coggins B. L. Blackford L. E. Sprague 1. Theory of Energy Conversion Electronics The effectiveness of energy conversion
THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS
 THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS R.L. ROGERS* Dewar Calorimetry is one of the simplest and most useful techniques used in the assessment of chemical reaction
THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS R.L. ROGERS* Dewar Calorimetry is one of the simplest and most useful techniques used in the assessment of chemical reaction
Resistivity and Temperature Coefficients (at 20 C)
 Homework # 4 Resistivity and Temperature Coefficients (at 0 C) Substance Resistivity, Temperature ( m) Coefficient, (C ) - Conductors Silver.59 x 0-0.006 Copper.6 x 0-0.006 Aluminum.65 x 0-0.0049 Tungsten
Homework # 4 Resistivity and Temperature Coefficients (at 0 C) Substance Resistivity, Temperature ( m) Coefficient, (C ) - Conductors Silver.59 x 0-0.006 Copper.6 x 0-0.006 Aluminum.65 x 0-0.0049 Tungsten
DIRECT RADIOMETRIC TECHNIQUES
 EMISSIVITY AND OTHER INFRARED-OPTICAL PROPERTIES MEASUREMENT METHODS DIRECT RADIOMETRIC TECHNIQUES Measuring principle The principle of direct radiometric techniques is shown schematically in the figure
EMISSIVITY AND OTHER INFRARED-OPTICAL PROPERTIES MEASUREMENT METHODS DIRECT RADIOMETRIC TECHNIQUES Measuring principle The principle of direct radiometric techniques is shown schematically in the figure
Harnessing the Power of Arduino for the Advanced Lab
 P P Herbert Jaeger + Harnessing the Power of Arduino for the Advanced Lab (Final Version) ALPhA Immersion Workshop July 27 29, 2017 Department of Physics Indiana University Purdue University Ft. Wayne,
P P Herbert Jaeger + Harnessing the Power of Arduino for the Advanced Lab (Final Version) ALPhA Immersion Workshop July 27 29, 2017 Department of Physics Indiana University Purdue University Ft. Wayne,
4.1. Physics Module Form 4 Chapter 4 - Heat GCKL UNDERSTANDING THERMAL EQUILIBRIUM. What is thermal equilibrium?
 4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
4.1 4 UNDERSTANDING THERMAL EQUILIBRIUM What is thermal equilibrium? 1. ( Heat, Temperature ) is a form of energy that flows from a hot body to a cold body. 2. The SI unit for ( heat, temperature) is Joule,
DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD
 Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
HIMARC Simulations Divergent Thinking, Convergent Engineering
 HIMARC Simulations Divergent Thinking, Convergent Engineering 8117 W. Manchester Avenue, Suite 504 Los Angeles, CA 90293 Ph: (310) 657-7992 Horizontal Superconducting Magnet, ID 1.6m 1 1 Design definition
HIMARC Simulations Divergent Thinking, Convergent Engineering 8117 W. Manchester Avenue, Suite 504 Los Angeles, CA 90293 Ph: (310) 657-7992 Horizontal Superconducting Magnet, ID 1.6m 1 1 Design definition
High-Pressure Volumetric Analyzer
 High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics. Thermodynamics and Statistical Physics
 Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Thermodynamics and Statistical Physics Key contents: Temperature scales Thermal expansion Temperature and heat, specific heat Heat and
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics Thermodynamics and Statistical Physics Key contents: Temperature scales Thermal expansion Temperature and heat, specific heat Heat and
THERMAL PROPERTY MEASUREMENT TECHNIQUES. AND SOME RESULTS FOR CdTe. H. F. Poppendiek D. J. Connelly R. K. Livett
 THERMAL PROPERTY MEASUREMENT TECHNIQUES AND SOME RESULTS FOR CdTe H. F. Poppendiek D. J. Connelly R. K. Livett QUARTERLY REPORT NO. 1 PERIOD: MARCH 10 TO JUNE 10, 1983 "The views and conclusions contained
THERMAL PROPERTY MEASUREMENT TECHNIQUES AND SOME RESULTS FOR CdTe H. F. Poppendiek D. J. Connelly R. K. Livett QUARTERLY REPORT NO. 1 PERIOD: MARCH 10 TO JUNE 10, 1983 "The views and conclusions contained
CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS
 50 CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS 4.1 INTRODUCTION In the development of any energy-efficient heat transfer fluids for enhanced heat transfer performance, in practical applications,
50 CHAPTER 4 THERMAL CONDUCTIVITY AND VISCOSITY MEASUREMENTS 4.1 INTRODUCTION In the development of any energy-efficient heat transfer fluids for enhanced heat transfer performance, in practical applications,
OPERATION MANUAL. 3 in 1, IR, Type K/J/R/E/T, Pt 100 ohm THERMOMETER. Model : TM-2000
 3 in 1, IR, Type K/J/R/E/T, Pt 100 ohm THERMOMETER Model : TM-2000 Your purchase of this THERMOMETER marks a step forward for you into the field of precision measurement. Although this THERMOMETER is a
3 in 1, IR, Type K/J/R/E/T, Pt 100 ohm THERMOMETER Model : TM-2000 Your purchase of this THERMOMETER marks a step forward for you into the field of precision measurement. Although this THERMOMETER is a
CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR
 CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR 1. The diagram shows a simple mercury barometer. If atmospheric pressure increases, what happens to level X and to level Y? 2. Four flower vases have
CLASSIFIED 2 PRESSURE THERMAL PHYSICS MR. HUSSAM SAMIR 1. The diagram shows a simple mercury barometer. If atmospheric pressure increases, what happens to level X and to level Y? 2. Four flower vases have
I m. R s. Digital. R x. OhmmetersxSeries Shunt Digital. R m
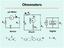 µa Meter I I s I m s E Digital x I Voltmeter x x E µa Meter m Is OhmmetersxSeries Shunt Digital EIx= = ()E sm x mxvi= x Shunt Ohmmeter Shunt s x E µa Meter I m I m V m E ) ( v I E ) ( E v E v E I When
µa Meter I I s I m s E Digital x I Voltmeter x x E µa Meter m Is OhmmetersxSeries Shunt Digital EIx= = ()E sm x mxvi= x Shunt Ohmmeter Shunt s x E µa Meter I m I m V m E ) ( v I E ) ( E v E v E I When
Ideal Gas Law and Absolute Zero
 Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Experiment IX Ideal Gas Law and Absolute Zero I. Purpose The purpose of this lab is to examine the relationship between the pressure, volume and temperature of air in a closed chamber. To do this, you
Chapter 11. Important to distinguish between them. They are not interchangeable. They mean very different things when used in physics Internal Energy
 Chapter 11 Energy in Thermal Processes Energy Transfer When two objects of different temperatures are placed in thermal contact, the temperature of the warmer decreases and the temperature of the cooler
Chapter 11 Energy in Thermal Processes Energy Transfer When two objects of different temperatures are placed in thermal contact, the temperature of the warmer decreases and the temperature of the cooler
11B, 11E Temperature and heat are related but not identical.
 Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
Thermochemistry Key Terms thermochemistry heat thermochemical equation calorimeter specific heat molar enthalpy of formation temperature enthalpy change enthalpy of combustion joule enthalpy of reaction
PROGRESS IN GAS-LOADING D/Pd SYSTEM. The feasibility of a self-sustaining heat generator
 Li, X., et al. PROGRESS IN GAS-LOADING D/Pd SYSTEM -- The feasibility of a self-sustaining heat generator --. in Tenth International Conference on Cold Fusion. 2003. Cambridge, MA: LENR-CANR.org. This
Li, X., et al. PROGRESS IN GAS-LOADING D/Pd SYSTEM -- The feasibility of a self-sustaining heat generator --. in Tenth International Conference on Cold Fusion. 2003. Cambridge, MA: LENR-CANR.org. This
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *4400796608* PHYSICS 0625/32 Paper 3 Extended May/June 2014 1 hour 15 minutes Candidates answer on
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *4400796608* PHYSICS 0625/32 Paper 3 Extended May/June 2014 1 hour 15 minutes Candidates answer on
Chapter 18. Temperature, Heat, and the First Law of Thermodynamics Temperature
 Chapter 18 Temperature, Heat, and the First Law of Thermodynamics 18.2 Temperature 18.3: The Zeroth aw of Thermodynamics If bodies A and B are each in thermal equilibrium with a third body T, then A and
Chapter 18 Temperature, Heat, and the First Law of Thermodynamics 18.2 Temperature 18.3: The Zeroth aw of Thermodynamics If bodies A and B are each in thermal equilibrium with a third body T, then A and
gskin Application Note: Calorimetry
 1 / 7 gskin Application Note: Calorimetry Direct and precise calorimetric measurements with the gskin Sensor The gskin Heat Flux Sensor enables direct and highly precise measurements of reaction enthalpies
1 / 7 gskin Application Note: Calorimetry Direct and precise calorimetric measurements with the gskin Sensor The gskin Heat Flux Sensor enables direct and highly precise measurements of reaction enthalpies
DEVICE FOR MEASUREMENT OF THERMAL EMISSIVITY AT CRYOGENIC TEMPERATURES
 DEVICE FOR MEASUREMENT OF THERMAL EMISSIVITY AT CRYOGENIC TEMPERATURES Tomas Kralik, Pavel Hanzelka, Vera Musilova, Ales Srnka Institute of Scientific Instruments ASCR, Kralovopolska 47, Brno, CZ 62 64,
DEVICE FOR MEASUREMENT OF THERMAL EMISSIVITY AT CRYOGENIC TEMPERATURES Tomas Kralik, Pavel Hanzelka, Vera Musilova, Ales Srnka Institute of Scientific Instruments ASCR, Kralovopolska 47, Brno, CZ 62 64,
RESULTS OF ICARUS 9 EXPERIMENTS RUN AT IMRA EUROPE
 Roulette, T., J. Roulette, and S. Pons. Results of ICARUS 9 Experiments Run at IMRA Europe. in Sixth International Conference on Cold Fusion, Progress in New Hydrogen Energy. 1996. Lake Toya, Hokkaido,
Roulette, T., J. Roulette, and S. Pons. Results of ICARUS 9 Experiments Run at IMRA Europe. in Sixth International Conference on Cold Fusion, Progress in New Hydrogen Energy. 1996. Lake Toya, Hokkaido,
International Journal of Advance Engineering and Research Development
 Scientific Journal of Impact Factor (SJIF): 4.72 International Journal of Advance Engineering and Research Development Volume 4, Issue 4, April -2017 e-issn (O): 2348-4470 p-issn (P): 2348-6406 An Experimental
Scientific Journal of Impact Factor (SJIF): 4.72 International Journal of Advance Engineering and Research Development Volume 4, Issue 4, April -2017 e-issn (O): 2348-4470 p-issn (P): 2348-6406 An Experimental
Waves Final Review. Name: Date: 1. On which one of the following graphs is the wavelength λ and the amplitude a of a wave correctly represented?
 Name: Date: Waves Final Review 1. On which one of the following graphs is the wavelength λ and the amplitude a of a wave correctly represented? A. Displacement λ a Distance along wave B. Displacement λ
Name: Date: Waves Final Review 1. On which one of the following graphs is the wavelength λ and the amplitude a of a wave correctly represented? A. Displacement λ a Distance along wave B. Displacement λ
Tick the box next to those resources for which the Sun is also the source of energy.
 1 (a) The source of solar energy is the Sun. Tick the box next to those resources for which the Sun is also the source of energy. coal geothermal hydroelectric nuclear wind [2] (b) Fig. 4.1 shows a solar
1 (a) The source of solar energy is the Sun. Tick the box next to those resources for which the Sun is also the source of energy. coal geothermal hydroelectric nuclear wind [2] (b) Fig. 4.1 shows a solar
Anomalous Heat Generation in Charging of Pd Powders with High Density Hydrogen Isotopes (I) Results of absorption experiments using Pd powders
 Material Science Session 7 O_5 Anomalous Heat Generation in Charging of Pd Powders with High Density Hydrogen Isotopes (I) Results of absorption experiments using Pd powders Y. Sasaki 1, A. Kitamura 1,
Material Science Session 7 O_5 Anomalous Heat Generation in Charging of Pd Powders with High Density Hydrogen Isotopes (I) Results of absorption experiments using Pd powders Y. Sasaki 1, A. Kitamura 1,
PHYS102 Previous Exam Problems. Temperature, Heat & The First Law of Thermodynamics
 PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
PHYS102 Previous Exam Problems CHAPTER 18 Temperature, Heat & The First Law of Thermodynamics Equilibrium & temperature scales Thermal expansion Exchange of heat First law of thermodynamics Heat conduction
Using a Mercury itc with thermocouples
 Technical Note Mercury Support Using a Mercury itc with thermocouples Abstract and content description This technical note describes how to make accurate and reliable temperature measurements using an
Technical Note Mercury Support Using a Mercury itc with thermocouples Abstract and content description This technical note describes how to make accurate and reliable temperature measurements using an
Franck-Hertz Experiment
 Franck-Hertz Experiment Introduction: In 1914, James Franck and Gustav Hertz discovered in the course of their investigations an energy loss in distinct steps for electrons passing through mercury vapor,
Franck-Hertz Experiment Introduction: In 1914, James Franck and Gustav Hertz discovered in the course of their investigations an energy loss in distinct steps for electrons passing through mercury vapor,
Determine the Inside Wall Temperature of DSTs using an Infrared Temperature Sensor
 SUMMARY DOCUMENT Determine the Inside Wall Temperature of DSTs using an Infrared Temperature Sensor Date submitted: April 14, 2017 Prepared by: Aparna Aravelli, Ph.D. Florida International University Collaborators:
SUMMARY DOCUMENT Determine the Inside Wall Temperature of DSTs using an Infrared Temperature Sensor Date submitted: April 14, 2017 Prepared by: Aparna Aravelli, Ph.D. Florida International University Collaborators:
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* PHYSICS 0625/04 Paper 4 Theory (Extended) For Examination from 2016 SPECIMEN PAPER 1
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *0123456789* PHYSICS 0625/04 Paper 4 Theory (Extended) For Examination from 2016 SPECIMEN PAPER 1
The student s results are shown in the table below. Time / minutes Time / minutes
 Q1.(a) Anhydrous calcium chloride is not used as a commercial de-icer because it reacts with water. The reaction with water is exothermic and causes handling problems. A student weighed out 1.00 g of anhydrous
Q1.(a) Anhydrous calcium chloride is not used as a commercial de-icer because it reacts with water. The reaction with water is exothermic and causes handling problems. A student weighed out 1.00 g of anhydrous
Embedded Control System of Apparatus for Measurement of Thermal Conductivity
 Embedded Control System of Apparatus for Measurement of Thermal Conductivity RENÉ PYSZKO 1, LUDVÍK KOVAL 2, ADÉLA MACHÁČKOVÁ 1 ZUZANA KLEČKOVÁ 1 and ZDENĚK JEDLIČKA 1 1 Department of Thermal Engineering
Embedded Control System of Apparatus for Measurement of Thermal Conductivity RENÉ PYSZKO 1, LUDVÍK KOVAL 2, ADÉLA MACHÁČKOVÁ 1 ZUZANA KLEČKOVÁ 1 and ZDENĚK JEDLIČKA 1 1 Department of Thermal Engineering
DETECTION OF ANOMALOUS ELEMENTS, X-RAY AND EXCESS HEAT INDUCED BY CONTINUOUS DIFFUSION OF DEUTERIUM THROUGH MULTI-LAYER CATHODE (Pd/CaO/Pd)
 The Seventh International Conference on Cold Fusion. 1998. Vancouver, Canada:, ENECO, Inc., Salt Lake City, UT. : p. 7. DETECTION OF ANOMALOUS ELEMENTS, X-RAY AND EXCESS HEAT INDUCED BY CONTINUOUS DIFFUSION
The Seventh International Conference on Cold Fusion. 1998. Vancouver, Canada:, ENECO, Inc., Salt Lake City, UT. : p. 7. DETECTION OF ANOMALOUS ELEMENTS, X-RAY AND EXCESS HEAT INDUCED BY CONTINUOUS DIFFUSION
CRYOGENIC CONDUCTION COOLING TEST OF REMOVABLE PANEL MOCK-UP FOR ITER CRYOSTAT THERMAL SHIELD
 CRYOGENIC CONDUCTION COOLING TEST OF REMOVABLE PANEL MOCK-UP FOR ITER CRYOSTAT THERMAL SHIELD K. Nam, a D. K. Kang, a W. Chung, a C. H. Noh, a J. Yu, b N. I. Her, b C. Hamlyn-Harris, b Y. Utin, b and K.
CRYOGENIC CONDUCTION COOLING TEST OF REMOVABLE PANEL MOCK-UP FOR ITER CRYOSTAT THERMAL SHIELD K. Nam, a D. K. Kang, a W. Chung, a C. H. Noh, a J. Yu, b N. I. Her, b C. Hamlyn-Harris, b Y. Utin, b and K.
