Industrial Surfaces Behaviour Related to the Adsorption and Desorption of Hydrogen at Cryogenic Temperature
|
|
|
- Abigayle Hicks
- 6 years ago
- Views:
Transcription
1 EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 375 Industrial Surfaces Behaviour Related to the Adsorption and Desorption of Hydrogen at Cryogenic Temperature G. Moulard 1, B. Jenninger 1 and Y. Saito 2 Abstract The determination of the hydrogen adsorption capacity on different industrial surfaces has been carried out by measuring isothermal adsorption. First results show that the adsorption capacity is mainly determined by surface porosity. Therefore, the samples may be classified in two categories: smooth surfaces and porous surfaces. Thermal desorption spectra reveal two adsorption energy levels for hydrogen physisorbed on porous materials, but only a single one on smooth samples. The value of the lowest energy level seems to be independent on the substrate nature. The physisorption process studied at low coverage, well below a monolayer, shows that these two levels are not well defined but an energy distribution exists for each of them. The influences of the isotherm temperature and an annealing at 7 K of an adsorbed monolayer on hydrogen adsorption capacity have been studied. 1 CERN, LHC Division, Geneva, Switzerland 2 KEK, Tsukuba, Japan. Member of the CERN-Japan Collaboration to the LHC Project 6th European Vacuum Conference (EVC-6) 7-12 December 1999, Villeurbanne, France Administrative Secretariat LHC Division CERN CH Geneva 23 Switzerland Geneva, 11 April 2000
2 I Introduction The use of superconducting magnets in the LHC Project provides cold bores at 1.9 K. The accelerator vacuum chamber becomes an efficient cryopump. However, specific parts will only work at a temperature of 4.5 K. In this region, the choice of a cryosorber material is of great importance. It must supply a large adsorption capacity, especially for hydrogen because its saturated vapour pressure at this temperature is above the maximum allowed vacuum pressure of the LHC. In this study, two kinds of materials have been tested with smooth and porous surfaces. Hydrogen adsorption capacities, V m, and hydrogen desorption energies, E d, of all samples have been determined by recording respectively adsorption isotherms and thermal desorption spectra (T.D.S). II Principles a)determination of the adsorption capacity The adsorption capacity, V m in molecules/cm 2, is estimated from adsorption isotherm measurements. The adsorption isotherm describes the equilibrium pressure evolution obtained by progressively increasing the gas coverage on a given surface which is maintained at a given temperature 1. The sample is pumped under ultra-high vacuum before being cooled to the working temperature, between 3.7 K and 9 K. The system is then isolated from the external pumping. The volumetric method is used 2. Small amounts of hydrogen are injected and the equilibrium pressure is measured. A steep pressure increase indicates the completion of the first monolayer on the surface and defines the adsorption capacity of the sample, indicating that the surface is saturated. The surface coverage, defined as the ratio between the number of adsorbed molecules and the adsorption capacity, is then equal to unity. The saturated vapour pressure p o, of the gas at the working temperature is reached with additional injections. b)determination of the adsorption energy Adsorption energies of physisorbed molecules are determined by increasing the sample temperature at a constant rate : T T o Et (1) The rate of the temperature change in our measurement, E, is in the range 10-3 K/s; T o being the isotherm temperature. During the recording of the T.D.S, the desorbed molecules are evacuated by the external pumping system. Desorption peaks appear on the graph: pressure = f(t) (Fig. 1). Assuming a first order desorption, i.e. non associative desorption and no re-adsorption, the desorption energy is determined from the following equation 3 : 1
3 E d 1 E d exp (2) 2 kt p W oe ktp ¹ where T P is the temperature at the maximum of the desorption peak, W o is a constant equal to s 4, E d the desorption energy and k the Boltzmann constant. III Experimental set-up The experimental set-up is shown in Fig. 2. All samples consist of a tube inserted into a cryostat and the inner surface is studied. Two copper shields assure a homogeneous temperature along the sample, independent on the liquid helium. The non-uniform sample temperature between the top and the bottom when heating up does not exceed 1.5 K. The cryostat can be filled either with liquid helium or with gaseous helium, depending on the working temperature. Samples have been tested in the range 3.7 K 9 K. Carbon resistance sensors are placed at the top, middle, and bottom of the sample to measure its temperature. The number of injected hydrogen molecules is deduced from the pressure drop in a 2 litre reference volume, measured with a capacitance gauge. The ultra-high vacuum is monitored with a Bayart Alpert gauge, while for pressure values higher than Torr, a combined Pirani Penning gauge is used. The transpiration factor is applied on pressure values. Desorbed species during T.D.S are recorded with a residual gas analyser. It is found that the amounts of O 2, CO, CO 2, CH 4 and H 2 O are insufficient to cause the cryotrapping of hydrogen 5. IV Samples studied Five different materials have been tested. The adsorption capacity is determined before and after an in situ bake out between 150qC and 250qC, for 48 hours. The sample characteristics are presented below. Surfaces of samples (1) to (3) are considered as smooth while (4) and (5) exhibit a porous surface due to the anodization process. 1) Copper surface 5 : 100 Pm thick copper colaminated stainless steel with a geometrical surface area of 2375 cm 2. It is baked at 150qC. 2) Electrochemical buffed SUS316L 6 with a maximum roughness of 0.2 Pm and a geometrical surface area of 2050 cm 2. The baking temperature is 200qC. 3) TiZrV film: a film of 1 Pm thick TiZrV coated on sample (2) with a geometrical surface area of 770 cm 2, supplied by CERN /EST Division. It is baked at 250qC. 4) Anodised aluminium 7 with a porous oxide layer of 40 Pm thick and a geometrical surface area of 631 cm 2, delivered by Jefferson Lab (U.S.A). The baking temperature is 150qC. 2
4 5) Anodised aluminium : a porous oxide layer with a real surface over geometrical surface ratio of 240, and a geometrical surface of 560 cm 2, provided by KEK laboratory (Japan). The baking temperature is 150qC. V Results a)adsorption capacity The adsorption capacity for hydrogen has been determined on unbaked and baked samples. Figure 3 shows the adsorption isotherms at 4.2 K. The graphical representation is not the one commonly used in the literature, but it allows us to compare, for a given pressure, the corresponding surface coverage of each material. The graph shows two distinct families of curves: smooth substrates with a capacity lower than molecules/cm 2, and porous surfaces that present a capacity larger than molecules/cm 2. The corresponding V m values are given in Table 1. Once the completion of the first hydrogen monolayer is obtained on smooth materials, the pressure rises steeply up to the saturated pressure at a value which is approximately twice V m. On a porous substrate, the pressure rises abruptly after saturation, but the saturated vapour pressure is not reached. Table 1 Hydrogen adsorption capacity at 4.2 K molecules/cm 2 at molecules/cm 2 at Ratio saturation : V m P sat (10-6 Torr) : V sat V sat /V m SMOOTH SURFACES Copper film unbaked Electrochemical buffed stainless steel unbaked Electrochemical buffed stainless steel baked TiZrV film POROUS SURFACES Al anodized unbaked (USA) / / Al anodized baked (USA) / / Al anodized (KEK) The influence of the in-situ baking process on surface capacity is deduced from Figure 3. This cleaning procedure enhances the adsorption capacity, but its efficiency is better on porous samples than on smooth ones. Bakeout also reduces the system background pressure. 3
5 The influence of an annealing process on the adsorbed molecules distribution on a porous surface has been examined. At first, hydrogen is injected on an anodised aluminium surface at 4.2 K to obtain a surface coverage slightly higher than unity but with an equilibrium pressure lower than the saturation pressure. The measured pressure is then Torr. After an annealing at 7 K for 2 hours the pressure reading at 4.2 K is Torr. b)adsorption energy Table 2 presents the hydrogen binding energies for all the samples, determined from their respective T.D.S. Due to the measurement method used, the given binding energies correspond to average values. The values in brackets refer to small desorption peaks partially hidden inside larger ones. Two general considerations can be made : - A varying amount of hydrogen is captured with an average energy, E d1, ranging from 17 mev to 24 mev, independent of the substrate. - On porous samples, a second adsorption energy level, between 60 mev and 76 mev exists. This second peak can be observed only after the baking process. Table 2 Hydrogen adsorption energy at 4.2 K Sample Preparation Ed 1 (mev) Ed 2 (mev) SMOOTH SURFACES Copper film Baked No peak Electrochemical buffed Unbaked 15.5 No peak stainless steel Electrochemical buffed Baked 17.5 (43.5) 62.5 stainless steel TiZrV film activated 21 No peak POROUS SURFACES Al anodized (USA) Unbaked 17 No peak Al anodized (USA) Unbaked 23 No peak Al anodized (USA) Baked (83) Al anodized (KEK) Unbaked When the isotherm is measured at higher temperature or when the surface is not fully saturated at 4.2 K, higher values of E d1 and of E d2 are observed, as shown in Table 3. T.D.S after an annealing at 7 K, shows only one broad desorption peak extending from 30 mev to 108 mev. 4
6 Table 3 Hydrogen adsorption energy at 4.2 K for non saturated surfaces and at 7 K for saturated surfaces Sample Preparation Ed 1 (mev) Ed 2 (mev) Electrochemical Not saturated 16.5 (45) No peak buffed stainless steel Al anodized (USA) 7 K baked Al anodized (USA) Not saturated The T.D.S of porous surfaces also gives the relative adsorption capacity corresponding to each adsorption energy level. Different growing modes of the first monolayer can be observed, depending on the isotherm temperature and on the surface coverage. At 7 K, 99% of trapped hydrogen molecules are captured with an energy of 70 mev, while at 4.2 K, 60% are bound with approximately the same energy. For this latter temperature, at low coverage (T = 0.2), the T.D.S shows up a first and a second peak, respectively, at 47 mev and 106 mev instead of 18 mev and 76 mev as for a saturated surface (T =1) (Fig. 4 and Table 3). The isotherm obtained at 3.7 K, with the same sample shows a second desorption peak corresponding to an average binding energy lower than the one at 4.7 K (Fig. 5). Moreover, the smaller area of this peak, as compared to the one observed at 4.2 K, indicates a lower capacity than at 4.2 K. V Discussion At 4.2 K, the sorption capacity of hydrogen is higher on porous surfaces than on smooth ones by at least one order of magnitude. From the T.D.S, one concludes that the large adsorption capacity is due to the porosity. The ratio between the real surface area and the geometrical area (S r /S g ) on smooth samples is close to unity. The saturated vapour pressure is reached at a coverage V sat equal to twice the value of V m, supposing that the adsorption capacity of the first and second monolayers for H 2 are the same. The large S r /S g ratio of a porous substrate is attributed to the surface porosity. A number of molecules, more than two orders of magnitude higher than the one needed for a smooth surface, is necessary to fill the pores and to form the first monolayer. The second monolayer corresponds to the one necessary to saturate a smooth surface. The steep pressure rise after the saturation is characteristic of a porous surface 8. The saturated H 2 vapour pressure, p o, at 4.2 K, is not reached with the porous sample. This phenomenon, called the capillary condensation is expressed mathematically by Kelvin s equation : the smaller the pore diameter, the lower the relative vapour pressure, p/ p o 9. Since electrochemical buffing is carried out by adding small abrasives to electrolyte liquid, this method may cause not only smoothness but also small cracks which are possible adsorption sites with higher energy than E d1. 5
7 Independent of the surface structure, on a surface saturated at 4.2 K, some hydrogen molecules are physisorbed with an energy ranging from 15.5 mev to 24.5 mev, in good agreement with the 22 mev usually found in the literature 10,11. It seems that the nature of the material does not strongly affect the lower energy level. One may therefore consider this energy level as a characteristic of hydrogen physisorption. On smooth surfaces, no hydrogen is adsorbed at other energy levels, indicating that this energy is characteristic for hydrogen adsorption on top of the surface, i.e. a surface without pores. The adsorption energy inside a pore is between 60 mev and 76 mev; slightly lower than on charcoal (80 mev) and on porous glass (85 mev) 4. The large width of the peaks shown on the T.D.S is related to an adsorption energy distribution on the top surface and within the pores. In both cases, a local distortion of the interaction potential between the sample and the gas molecule exists as follows: - The top of the surface is liable to exhibit imperfections which include cleavage steps, dislocations and point defects. The defects produce heterogeneity on the surface 12 leading to an adsorption energy distribution. Adsorption energy on such defects is usually high (47 mev) in comparison with the value on a perfectly flat substrate (22 mev). Nevertheless, the number of H 2 molecules adsorbed on such defects is small compared to the total quantity adsorbed on the top surface. - Within a pore, the interaction potential between an adsorbed molecule and the wall is strongly modified. The potential well is deeper than on the top surface, leading to higher adsorption energies. According to Zsigmondy s model, 13 as the pressure is progressively increased, wider and wider pores are filled until, at the saturation pressure, the entire system is filled with condensate. The adsorption energy of a molecule inside a pore depends on the dimension of the adsorbate relative to the pore 14 and the smaller the pore diameter, the higher the binding energy. The T.D.S of an isotherm at 4.2 K with a relative coverage of0.2 on a porous surface suggest the tendency for adsorbed molecules to migrate to the various defects. Hence, molecules come in contact with a greater number of substrate atoms, leading to an increase of the adsorption energy. At this low coverage, H 2 molecules fill the high energy sites i.e. the smallest pores and surface defects, leading to high binding energies (106 mev within the pores and 47 mev on the top surface). As the coverage increases, the mean binding energy decreases. On such heterogeneous surfaces, interactions with hydrogen molecules cannot be characterised by a single energy value 4. Nevertheless, we can consider that two kinds of adsorption sites exist: on the top surface, where sites are defined by an average energy of 20 mev for H 2 ; and within the porosity, where the largest capacity is available on sites with an average energy of 70 mev. It follows from this analysis that, as long as the interaction between adsorbed molecules can be neglected, an adsorption site can be defined as a definite position on the surface which will always physisorb a specific gas molecule with the same energy. The sorption of a molecule is considered as effective if its sojourn time on the site is long enough. This parameter strongly depends on the temperature. Therefore, sites at 20 mev cannot be considered as effective adsorption sites for an adsorption at 6
8 7 K since the mean sojourn time is 25 seconds. The adsorption capacity is reduced when the isotherm temperature increases because some adsorption sites become unavailable (Fig. 6). The number of available sites depends on the energy distribution of the sites. Adsorption on defects and into small pores involves a diffusion mechanism of molecules, from low to higher energy sites. The diffusion process of H 2 molecules is efficient at 7 K and molecules on the top surface can diffuse to sites corresponding to the surface defects or to porosity. At 3.7 K, the diffusion is strongly reduced and H 2 molecules are adsorbed where they impinge the surface. The rate of diffusion into the smallest pores is low and the porosity is not completely filled. As a consequence, the T.D.S shows a second desorption peak smaller in height than for the isotherm at 4.2 K, and the number of occupied sites with energy higher than 80 mev is considerably reduced. The subsequent annealing at 7 K of the monolayer deposited at 4.2 K activates the diffusion such that all the pores are filled. The adsorption capacity is optimised which results in a reduction of the pressure. The second desorption peak observed with an isotherm at 4.2 K, on a porous substrate and with a coverage of T = 0.2 and T = 1 respectively, have been compared in Fig. 4. These peaks overlap at high temperature [35 K - 45 K]. At low coverage, high energy sites are filled first if the diffusion is effective. One assumption would be to consider in first approximation, this second desorption peak for T 1, as the sum of three peaks as shown in Fig. 7. Then, the peak with the highest energy (106 mev) should correspond to micropores with the lowest diameters; the second one has an energy of 82 mev and the largest one, corresponds to the widest micropores and has an energy of 70 mev. Widest micropores also have the largest capacity 15. The shape of this second desorption peak is linked to the micropore size distribution, i.e, pores with a diameter less than 20 Å 16. VI Conclusion Measurements of isotherm adsorption and of thermal desorption spectra of hydrogen at, and close to liquid helium temperature on different kinds of samples have been carried out. In this temperature range, hydrogen physisorption does not depend on the sample nature but depends on the surface structure. A binding energy around 20 mev is found for all the samples; it corresponds to the hydrogen adsorption on the top surface. The energy distribution around the 70 mev peak is linked to the pore size distribution. The annealing at 7 K of the adsorbed layer has revealed the importance of the surface diffusion process. The maximum sorption capacity is reached when the smallest pores are filled. Therefore, the concept of adsorption capacity at a given temperature, is closely related to the distribution of the first adsorbed monolayer into the pores. 7
9 References 1 : D.M. Young, A.D. Crowell, Physical adsorption of Gases, (Butterworths, London, 1962) p.3. 2 J.P. Hobson, The Journal of Physical Chemistry, Vol 73, N 8 (1969). 3 D.P. Woodruff, T.A. Delchar, Modern Techniques of Surface Science 2 nd edition, p P. A. Redhead, J. P. Hobson, E. V. Kornelsen, The Physical Basis of Ultrahigh Vacuum, American Vacuum society classics. 5 E. Wallén, J. Vac. Sci. Technol. A 14(5) (1996) p M. Baba et al. Proc. 15 th IS DEIV, Darmstadt,1992 p.3. 7 M. G. Rao, P. Kneisel and J. Susta, Cryogenics, vol 34, (1994) p E: Wallén, J. Vac. Sci. Technol. A 15(2), (1997), p S. J. Gregg and K. S. W. Sing Adsorption, Surface area and Porosity, 2 nd edition 1982 Academic press, p S. Andersson, L. Wilzén, M. Persson, Phys. Rev. B, 38(5), (1988), p J. P. Hobson, J. Vac. Sci. Technol. A 13(3), (1995), p S. Ross and J. P. Oliver, Physical Adsorption, Interscience, New-York (1964). 13 A. Zsigmondy, Z. Anorg. Chem. 71, (1911), p D. H. Everett and J. C. Powl, J. Chem. Soc., Faraday Trans. I, 72, 619 (1976). 15 M. J. Sparnaay, Surface Science, 9 (1968), p IUPAC Manual of Symbols and Terminology, appendix 2, Pt. 1, Colloid and Surface Chemistry. Pure Appl. Chem. 31, 578 (1972). 8
10 Temperature Tp Pressure Time Figure 1 : Thermal Desorption Spectra (T.D.S) 9
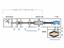
11 P UHV Reference Volume 2 liters H2 CH4 CO CO2 P LHe supplied by a 500 L dewar M Vacuum insulation Thermal shield Thermal shields (copper) Sample Injection Line (perforated) Liquid Helium Aluminium block P M Vacuum gauge Gas analyser Temperature sensor Lhe indicator Heating Figure 2 : Experimental set-up for isotherm adsorption and thermal desorption spectra 10
12 1.E-05 Coverage (molecules/cm 2 ) 1.E+15 1.E+16 1.E+17 1.E+18 1.E-06 1.E-07 Pressure (Torr) 1.E-08 1.E-09 1.E-10 1.E-11 Electo-chemical buffed stainless steel, unbaked Electo-chemical buffed stainless steel, baked TiZrV (coating), unactivated TiZrV (coating), activated Cu coated stainless steel, baked Anodized Al (USA), unbaked Anodized Al (USA), baked Anodized Al (Japan) unbaked Anodized Al(Japan) baked Figure 3 : Hydrogen adsorption isotherm at 4.2K for various samples 11
13 1.E-05 Temperature (K) Pressure (Torr) 1.E-06 1.E-07 1.E-08 1.E-09 1.E-10 T T Figure 4 : T.D.S for anodized Al (sample 4) following an isotherm measurement at 4.2 K, with 2 different coverages 7.E-06 Temperature (K) Pressure (Torr) 6.E-06 5.E-06 4.E-06 3.E-06 2.E-06 1.E-06 0.E K 4.2K Figure 5 : T.D.S for anodized Al (sample 4) following an isotherm measurements at 3.7 K and 4.2 K 12
14 1.E-03 Coverage (molecules/cm 2 ) 1.E+16 1.E+17 1.E+18 1.E-04 Pressure (Torr) 1.E-05 1.E-06 1.E-07 1.E-08 8 K 7 K 4.2 K 1.E-09 Figure 6 : Adsorption isotherms on anodised aluminium at 3 different temperatures Temperature (K) Pressure (Torr) 6.E-06 4.E-06 2.E-06 T T 0.E Figure 7 : Energy distribution within the microporosity 13
MOLECULAR SURFACE PUMPING: CRYOPUMPING
 51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
Cold / sticky system. Vincent Baglin. CERN AT-VAC, Geneva
 Cold / sticky system Vincent Baglin CERN AT-VAC, Geneva 1 Cold / sticky system?? Vacuum?? 2 Eureka?? 3 Cold / sticky system Outline 1. Cryopumping 2. Adsorption isotherms 3. Cryosorbers in cold systems
Cold / sticky system Vincent Baglin CERN AT-VAC, Geneva 1 Cold / sticky system?? Vacuum?? 2 Eureka?? 3 Cold / sticky system Outline 1. Cryopumping 2. Adsorption isotherms 3. Cryosorbers in cold systems
λ = 5 10 p (i.e. 5 cm at 10 3 Torr, or ( )
 Getter pumping C. Benvenuti R&B Energy Research, Geneva, Switzerland Abstract A surface may provide a useful pumping action when able to retain adsorbed gas molecules for the duration of a given experiment.
Getter pumping C. Benvenuti R&B Energy Research, Geneva, Switzerland Abstract A surface may provide a useful pumping action when able to retain adsorbed gas molecules for the duration of a given experiment.
An Investigative Review on Cryosorption Pumps - Development and Application
 An Investigative Review on Cryosorption Pumps - Development and Application Vismay.K.G #1, Vinay.K.B *2 # PG Student, Department of Mechanical Engineering, Vidyavardhaka College of Engineering, Mysuru
An Investigative Review on Cryosorption Pumps - Development and Application Vismay.K.G #1, Vinay.K.B *2 # PG Student, Department of Mechanical Engineering, Vidyavardhaka College of Engineering, Mysuru
Vacuum for Accelerators
 Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Cryopumping & Vacuum Systems
 Cryopumping & Vacuum Systems V. Baglin CERN TE-VSC, Geneva 2 Cryopumping: do you wonder? What is this white stain? Why it is on the LHC beam screen? What is then the expected gas density in this expensive
Cryopumping & Vacuum Systems V. Baglin CERN TE-VSC, Geneva 2 Cryopumping: do you wonder? What is this white stain? Why it is on the LHC beam screen? What is then the expected gas density in this expensive
High-Pressure Volumetric Analyzer
 High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
Vacuum. Residual pressure can thwart the best cryogenic design. Each gas molecule collision carries ~kt from the hot exterior to the cold interior.
 Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
UHV - Technology. Oswald Gröbner
 School on Synchrotron Radiation UHV - Technology Trieste, 20-21 April 2004 1) Introduction and some basics 2) Building blocks of a vacuum system 3) How to get clean ultra high vacuum 4) Desorption phenomena
School on Synchrotron Radiation UHV - Technology Trieste, 20-21 April 2004 1) Introduction and some basics 2) Building blocks of a vacuum system 3) How to get clean ultra high vacuum 4) Desorption phenomena
THE LARGE HADRON COLLIDER VACUUM SYSTEM
 THE LARGE HADRON COLLIDER VACUUM SYSTEM B. Angerth, F. Bertinelli, J.-C. Brunet, R. Calder, F. Caspers, P. Cruikshank, J-M. Dalin, O. Gröbner, N. Kos, A. Mathewson, A. Poncet, C. Reymermier, F. Ruggiero,
THE LARGE HADRON COLLIDER VACUUM SYSTEM B. Angerth, F. Bertinelli, J.-C. Brunet, R. Calder, F. Caspers, P. Cruikshank, J-M. Dalin, O. Gröbner, N. Kos, A. Mathewson, A. Poncet, C. Reymermier, F. Ruggiero,
THERMOHYDRAULIC BEHAVIOUR OF HEII IN STRATIFIED CO-CURRENT TWO-PHASE FLOW AT HIGH VAPOR VELOCITIES
 EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 67 THERMOHYDRAULIC BEHAVIOUR OF HEII IN STRATIFIED CO-CURRENT TWO-PHASE
EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 67 THERMOHYDRAULIC BEHAVIOUR OF HEII IN STRATIFIED CO-CURRENT TWO-PHASE
A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC
 A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC A. Rossi, G. Rumolo and F. Ziermann, CERN, Geneva, Switzerland Abstract The LHC experimental regions (ATLAS, ALICE, CMS and LHC
A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC A. Rossi, G. Rumolo and F. Ziermann, CERN, Geneva, Switzerland Abstract The LHC experimental regions (ATLAS, ALICE, CMS and LHC
Experimental Results of a LHC Type Cryogenic Vacuum System Subjected to an Electron Cloud
 Experimental Results of a LHC Type Cryogenic Vacuum System Subjected to an Electron Cloud V. Baglin, B. Jenninger CERN AT-VAC, Geneva 1. Introduction LHC & Electron Cloud LHC cryogenic vacuum system 2.
Experimental Results of a LHC Type Cryogenic Vacuum System Subjected to an Electron Cloud V. Baglin, B. Jenninger CERN AT-VAC, Geneva 1. Introduction LHC & Electron Cloud LHC cryogenic vacuum system 2.
Non-Evaporable Getters
 Non-Evaporable Getters Dr. Oleg B. Malyshev Senior Vacuum Scientist ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK VS-2, 18-19 October 2011, Ricoh Arena, Coventry 1 Sorption and diffusion Gas
Non-Evaporable Getters Dr. Oleg B. Malyshev Senior Vacuum Scientist ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK VS-2, 18-19 October 2011, Ricoh Arena, Coventry 1 Sorption and diffusion Gas
Lecture 4. Ultrahigh Vacuum Science and Technology
 Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
( KS A ) (1) , vapour, vapor (USA) , saturation vapour pressure. , standard reference conditions for gases. , degree of saturation
 ( KS A 3014-91 ) (1), standard reference conditions for gases 0, 101325 Pa (1 =760mmHg ), vacuum, low ( rough ) vacuum 100Pa, medium vacuum 100 01 Pa, high vacuum 01 10 5 Pa, ultra high vacuum ( UHV )
( KS A 3014-91 ) (1), standard reference conditions for gases 0, 101325 Pa (1 =760mmHg ), vacuum, low ( rough ) vacuum 100Pa, medium vacuum 100 01 Pa, high vacuum 01 10 5 Pa, ultra high vacuum ( UHV )
Supercritical Helium Cooling of the LHC Beam Screens
 EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report Supercritical Helium Cooling of the LHC Beam Screens Emmanuel Hatchadourian,,
EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report Supercritical Helium Cooling of the LHC Beam Screens Emmanuel Hatchadourian,,
Hydrogen in the LCLS2 Beamline Vacuum
 Hydrogen in the LCLS2 Beamline Vacuum Anthony C. Crawford Fermilab Technical Div./SRF Development Dept. acc52@fnal.gov 13Oct15 This note demonstrates that the cold segments of the LCLS2 linac will cryopump
Hydrogen in the LCLS2 Beamline Vacuum Anthony C. Crawford Fermilab Technical Div./SRF Development Dept. acc52@fnal.gov 13Oct15 This note demonstrates that the cold segments of the LCLS2 linac will cryopump
Supporting Information
 Supporting Information Yao et al. 10.1073/pnas.1416368111 Fig. S1. In situ LEEM imaging of graphene growth via chemical vapor deposition (CVD) on Pt(111). The growth of graphene on Pt(111) via a CVD process
Supporting Information Yao et al. 10.1073/pnas.1416368111 Fig. S1. In situ LEEM imaging of graphene growth via chemical vapor deposition (CVD) on Pt(111). The growth of graphene on Pt(111) via a CVD process
Experimental techniques of Surface Science and how a theorist understands them
 Experimental techniques of Surface Science and how a theorist understands them Karsten Reuter Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, D-14195 Berlin reuter@fhi-berlin.mpg.de 0.
Experimental techniques of Surface Science and how a theorist understands them Karsten Reuter Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, D-14195 Berlin reuter@fhi-berlin.mpg.de 0.
H. J. Halama, H. C. Hseuh and T. S. Chou Accelerator Department Brookhaven National Laboratory Upton, New York 11973
 Presented at the 1981 Cryogenic Engineering Conference, San Diego, California, August 10-14, 1981 BNL 30012 CRYOPUMPING OF HYDROGEN AMD HELIUM* H. J. Halama, H. C. Hseuh and T. S. Chou Accelerator Department
Presented at the 1981 Cryogenic Engineering Conference, San Diego, California, August 10-14, 1981 BNL 30012 CRYOPUMPING OF HYDROGEN AMD HELIUM* H. J. Halama, H. C. Hseuh and T. S. Chou Accelerator Department
The Vacuum Case for KATRIN
 The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
Earlier Lecture. In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide.
 41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
Vacuum Pumps. Two general classes exist: Gas transfer physical removal of matter. Mechanical, diffusion, turbomolecular
 Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness
 JET P(98)57 P A eladarakis W Obert On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness This document is intended for publication in the open literature.
JET P(98)57 P A eladarakis W Obert On the Emissivity of Silver Coated Panels, Effect of Long Term Stability and Effect of Coating Thickness This document is intended for publication in the open literature.
SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS
 SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS ASAP 2020 Plus: Accelerated Surface Area and Porosimetry System The Micromeritics ASAP 2020 Plus integrates a variety of automated gas
SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS ASAP 2020 Plus: Accelerated Surface Area and Porosimetry System The Micromeritics ASAP 2020 Plus integrates a variety of automated gas
DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD
 Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
Chapter 4 DEPOSITION OF THIN TiO 2 FILMS BY DC MAGNETRON SPUTTERING METHOD 4.1 INTRODUCTION Sputter deposition process is another old technique being used in modern semiconductor industries. Sputtering
Module 5: "Adsoption" Lecture 25: The Lecture Contains: Definition. Applications. How does Adsorption occur? Physisorption Chemisorption.
 The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
Beam heat load due to geometrical and resistive wall impedance in COLDDIAG
 Beam heat load due to geometrical and resistive wall impedance in COLDDIAG Sara Casalbuoni, Mauro Migliorati, Andrea Mostacci, Luigi Palumbo, Bruno Spataro 2012 JINST 7 P11008, http://iopscience.iop.org/17480221/7/11/p11008
Beam heat load due to geometrical and resistive wall impedance in COLDDIAG Sara Casalbuoni, Mauro Migliorati, Andrea Mostacci, Luigi Palumbo, Bruno Spataro 2012 JINST 7 P11008, http://iopscience.iop.org/17480221/7/11/p11008
Vacuum System of Synchrotron radiation sources
 3 rd ILSF Advanced School on Synchrotron Radiation and Its Applications September 14-16, 2013 Vacuum System of Synchrotron radiation sources Prepared by: Omid Seify, Vacuum group, ILSF project Institute
3 rd ILSF Advanced School on Synchrotron Radiation and Its Applications September 14-16, 2013 Vacuum System of Synchrotron radiation sources Prepared by: Omid Seify, Vacuum group, ILSF project Institute
Specific Surface Area and Porosity Measurements of Aluminosilicate Adsorbents
 ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2016, Vol. 32, No. (5): Pg. 2401-2406 Specific Surface
ORIENTAL JOURNAL OF CHEMISTRY An International Open Free Access, Peer Reviewed Research Journal www.orientjchem.org ISSN: 0970-020 X CODEN: OJCHEG 2016, Vol. 32, No. (5): Pg. 2401-2406 Specific Surface
A Vacuum point of view
 Beam-Surface Interaction A Vacuum point of view F. Le Pimpec SLAC/NLC SSRL April 2004 1 Dynamic Vacuum You want to address the terms of this formula How to measure the Pressure? F. Le Pimpec - SLAC 2 Outline
Beam-Surface Interaction A Vacuum point of view F. Le Pimpec SLAC/NLC SSRL April 2004 1 Dynamic Vacuum You want to address the terms of this formula How to measure the Pressure? F. Le Pimpec - SLAC 2 Outline
Vacuum techniques (down to 1 K)
 Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Magnetic and Electric Field Effects on the Photoelectron Emission from Prototype LHC Beam Screen Material
 EUOPEAN OGANIZATION FO NUCLEA ESEACH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project eport 373 Magnetic and Electric Field Effects on the Photoelectron Emission from
EUOPEAN OGANIZATION FO NUCLEA ESEACH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project eport 373 Magnetic and Electric Field Effects on the Photoelectron Emission from
Repetition: Physical Deposition Processes
 Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Physical Vapor Deposition
 Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Optimized Annular Triode Ion Pump for Experimental Areas in the LHC
 See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/41059758 Optimized Annular Triode Ion Pump for Experimental Areas in the LHC Article in Vacuum
See discussions, stats, and author profiles for this publication at: https://www.researchgate.net/publication/41059758 Optimized Annular Triode Ion Pump for Experimental Areas in the LHC Article in Vacuum
BET Surface Area Analysis of Nanoparticles *
 OpenStax-CNX module: m38278 1 BET Surface Area Analysis of Nanoparticles * Nina Hwang Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
OpenStax-CNX module: m38278 1 BET Surface Area Analysis of Nanoparticles * Nina Hwang Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators. Mauro Audi, Academic, Government & Research Marketing Manager
 ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
Beam induced desorption
 Accelerators in a new light Beam induced desorption Dr. Oleg B. Malyshev, ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK oleg.malyshev@stfc.ac.uk CAS on Vacuum for Particle Accelerators 2017
Accelerators in a new light Beam induced desorption Dr. Oleg B. Malyshev, ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK oleg.malyshev@stfc.ac.uk CAS on Vacuum for Particle Accelerators 2017
Distinguish quantitatively between the adsorption isotherms of Gibbs, Freundlich and Langmuir.
 Module 8 : Surface Chemistry Lecture 36 : Adsorption Objectives After studying this lecture, you will be able to Distinguish between physisorption and chemisorption. Distinguish between monolayer adsorption
Module 8 : Surface Chemistry Lecture 36 : Adsorption Objectives After studying this lecture, you will be able to Distinguish between physisorption and chemisorption. Distinguish between monolayer adsorption
Electron Cloud Studies made at CERN in the SPS
 Electron Cloud Studies made at CERN in the SPS J.M. Jimenez On behalf of the Electron Cloud Study Team, a Collaboration between AT and AB Departments Main Topics Introduction LHC Injectors SPS Running
Electron Cloud Studies made at CERN in the SPS J.M. Jimenez On behalf of the Electron Cloud Study Team, a Collaboration between AT and AB Departments Main Topics Introduction LHC Injectors SPS Running
Results on a-c tubes subjected to synchrotron irradiation
 Results on a-c tubes subjected to synchrotron irradiation V. Baglin, P. Chiggiato, P. Costa-Pinto, B. Henrist (CERN, Geneva) V. Anashin, D. Dorokhov. A. Semenov, A. Krasnov, D. Shwartz, A. Senchenko (,
Results on a-c tubes subjected to synchrotron irradiation V. Baglin, P. Chiggiato, P. Costa-Pinto, B. Henrist (CERN, Geneva) V. Anashin, D. Dorokhov. A. Semenov, A. Krasnov, D. Shwartz, A. Senchenko (,
Effects of methanol on crystallization of water in the deeply super cooled region
 Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
The Low Temperature Physics of Thin Films Superconducting Tin and Monolayer Graphene
 The Low Temperature Physics of Thin Films Superconducting Tin and Monolayer Graphene Abstract: The aim of this project was to investigate how the electrical resistance of a conductor changes if it is deposited
The Low Temperature Physics of Thin Films Superconducting Tin and Monolayer Graphene Abstract: The aim of this project was to investigate how the electrical resistance of a conductor changes if it is deposited
Experience from the LEP Vacuum System
 Experience from the LEP Vacuum System O. Gröbner CERN, LHC-VAC Workshop on an e + e - Ring at VLHC ITT, 9-11 March 2001 3/4/01 O. Gröbner, CERN-LHC/VAC References 1) LEP Design Report, Vol.II, CERN-LEP/84-01,
Experience from the LEP Vacuum System O. Gröbner CERN, LHC-VAC Workshop on an e + e - Ring at VLHC ITT, 9-11 March 2001 3/4/01 O. Gröbner, CERN-LHC/VAC References 1) LEP Design Report, Vol.II, CERN-LEP/84-01,
PHYSICAL VAPOR DEPOSITION OF THIN FILMS
 PHYSICAL VAPOR DEPOSITION OF THIN FILMS JOHN E. MAHAN Colorado State University A Wiley-Interscience Publication JOHN WILEY & SONS, INC. New York Chichester Weinheim Brisbane Singapore Toronto CONTENTS
PHYSICAL VAPOR DEPOSITION OF THIN FILMS JOHN E. MAHAN Colorado State University A Wiley-Interscience Publication JOHN WILEY & SONS, INC. New York Chichester Weinheim Brisbane Singapore Toronto CONTENTS
Vacuum II. G. Franchetti CAS - Bilbao. 30/5/2011 G. Franchetti 1
 Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
TPD-MS. Photocatalytic Studies Using Temperature Programmed Desorption Mass Spectrometry (TPD-MS) APPLICATION NOTE NOTE
 TPD-MS APPLICATION NOTE NOTE Photocatalytic Studies Using Temperature Programmed Desorption Mass Spectrometry (TPD-MS) Thermal analysis consists of many techniques for the exploration of the physical properties
TPD-MS APPLICATION NOTE NOTE Photocatalytic Studies Using Temperature Programmed Desorption Mass Spectrometry (TPD-MS) Thermal analysis consists of many techniques for the exploration of the physical properties
Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources
 CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
1. SYNCHROTRON RADIATION-INDUCED DESORPTION
 127 DYNAMIC OUTGASSING Oswald Gröbner CERN, Geneva, Switzerland Abstract Outgassing stimulated by photons, ions and electrons created by highenergy and high-intensity particle beams in accelerators and
127 DYNAMIC OUTGASSING Oswald Gröbner CERN, Geneva, Switzerland Abstract Outgassing stimulated by photons, ions and electrons created by highenergy and high-intensity particle beams in accelerators and
He II Heat transfer through a Corrugated Tube - Test Report
 He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
He II Heat transfer through a Corrugated Tube - Test Report Authors: Ch. Darve, Y. Huang, T. Nicol, T. Peterson Keywords: LHC inner triplet, heat exchanger, He II heat transfer, Kapitza resistance. Abstract
The first three categories are considered a bottom-up approach while lithography is a topdown
 Nanowires and Nanorods One-dimensional structures have been called in different ways: nanowires, nanorod, fibers of fibrils, whiskers, etc. The common characteristic of these structures is that all they
Nanowires and Nanorods One-dimensional structures have been called in different ways: nanowires, nanorod, fibers of fibrils, whiskers, etc. The common characteristic of these structures is that all they
Lecture 1: Vapour Growth Techniques
 PH3EC2 Vapour Growth and Epitaxial Growth Lecturer: Dr. Shinoj V K Lecture 1: Vapour Growth Techniques 1.1 Vapour growth The growth of single crystal materials from the vapour phase. Deposition from the
PH3EC2 Vapour Growth and Epitaxial Growth Lecturer: Dr. Shinoj V K Lecture 1: Vapour Growth Techniques 1.1 Vapour growth The growth of single crystal materials from the vapour phase. Deposition from the
ROLE OF SURFACE CHEMISTRY IN ADSORPTION OF ETHYLMETHYLAMINE ON ACTIVATED CARBONS
 ROLE OF SURFACE CHEMISTRY IN ADSORPTION OF ETHYLMETHYLAMINE ON ACTIVATED CARBONS Yehya El-Sayed, and Teresa J. Bandosz Department of Chemistry, The City College and The Graduate School of the City University
ROLE OF SURFACE CHEMISTRY IN ADSORPTION OF ETHYLMETHYLAMINE ON ACTIVATED CARBONS Yehya El-Sayed, and Teresa J. Bandosz Department of Chemistry, The City College and The Graduate School of the City University
Influence of water on the total heat transfer in evacuated insulations
 Bavarian Center for Applied Energy Research Influence of water on the total heat transfer in evacuated insulations Ulrich Heinemann ZAE Bayern, Würzburg, Germany ulrich.heinemann@zae.uni-wuerzburg.de 7
Bavarian Center for Applied Energy Research Influence of water on the total heat transfer in evacuated insulations Ulrich Heinemann ZAE Bayern, Würzburg, Germany ulrich.heinemann@zae.uni-wuerzburg.de 7
Light-Induced Atom Desorption in Alkali Vapor Cells
 Fundamental Physics Using Atoms, 2010/08/09, Osaka Light-Induced Atom Desorption in Alkali Vapor Cells A. Hatakeyama (Tokyo Univ. Agr. Tech.) K. Hosumi K. Kitagami Alkali vapor cells UHV cell for laser
Fundamental Physics Using Atoms, 2010/08/09, Osaka Light-Induced Atom Desorption in Alkali Vapor Cells A. Hatakeyama (Tokyo Univ. Agr. Tech.) K. Hosumi K. Kitagami Alkali vapor cells UHV cell for laser
Vacuum Technology and film growth. Diffusion Resistor
 Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Lecture 5. Solid surface: Adsorption and Catalysis
 Lecture 5 Solid surface: Adsorption and Catalysis Adsorbtion G = H T S DG ads should be negative (spontaneous process) DS ads is negative (reduced freedom) DH should be negative for adsorption processes
Lecture 5 Solid surface: Adsorption and Catalysis Adsorbtion G = H T S DG ads should be negative (spontaneous process) DS ads is negative (reduced freedom) DH should be negative for adsorption processes
Experimental Methods and Analysis
 Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
Chapter 3 28 Experimental Methods and Analysis 1. Equipment The fundamental basis of quantitative adsorption analysis is the measurement of excess adsorption isotherms. Each isotherm comprises a series
Vacuum and mechanical design of ILC DR
 Vacuum and mechanical design of ILC DR O. B. Malyshev ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK Low Emittance Ring Workshop 2010 12-15 January 2010 Integration design: usual consideration
Vacuum and mechanical design of ILC DR O. B. Malyshev ASTeC Vacuum Science Group, STFC Daresbury Laboratory, UK Low Emittance Ring Workshop 2010 12-15 January 2010 Integration design: usual consideration
Characterisation of Microporous Materials by Finite Concentration Inverse Gas Chromatography
 Characterisation of Microporous Materials by Finite Concentration Inverse Gas Chromatography Surface Measurement Systems Ltd. Finite concentration IGC SEA is a useful tool for the investigation of surface
Characterisation of Microporous Materials by Finite Concentration Inverse Gas Chromatography Surface Measurement Systems Ltd. Finite concentration IGC SEA is a useful tool for the investigation of surface
GAS-SURFACE INTERACTIONS
 Page 1 of 16 GAS-SURFACE INTERACTIONS In modern surface science, important technological processes often involve the adsorption of molecules in gaseous form onto a surface. We can treat this adsorption
Page 1 of 16 GAS-SURFACE INTERACTIONS In modern surface science, important technological processes often involve the adsorption of molecules in gaseous form onto a surface. We can treat this adsorption
Vacuum. Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006
 Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
28 Processes at solid surfaces
 28 Processes at solid surfaces Solutions to exercises E28.b E28.2b E28.3b Discussion questions The motion of one section of a crystal past another a dislocation results in steps and terraces. See Figures
28 Processes at solid surfaces Solutions to exercises E28.b E28.2b E28.3b Discussion questions The motion of one section of a crystal past another a dislocation results in steps and terraces. See Figures
ADSORPTION ON SURFACES. Kinetics of small molecule binding to solid surfaces
 ADSORPTION ON SURFACES Kinetics of small molecule binding to solid surfaces When the reactants arrive at the catalyst surface, reactions are accelerated Physisorption and Chemisorption 1- diffusion to
ADSORPTION ON SURFACES Kinetics of small molecule binding to solid surfaces When the reactants arrive at the catalyst surface, reactions are accelerated Physisorption and Chemisorption 1- diffusion to
. VOLUME 28, 1967, No.12
 PH I LI PS TECHNIC'AL REVIEW '_...,. VOLUME 28, 1967, No.12 A fast, cryopump system for ultra-high vacuum A. Venema Now that pumps which have a high pumping speed at low pressure have been developed it
PH I LI PS TECHNIC'AL REVIEW '_...,. VOLUME 28, 1967, No.12 A fast, cryopump system for ultra-high vacuum A. Venema Now that pumps which have a high pumping speed at low pressure have been developed it
Table 1: Residence time (τ) in seconds for adsorbed molecules
 1 Surfaces We got our first hint of the importance of surface processes in the mass spectrum of a high vacuum environment. The spectrum was dominated by water and carbon monoxide, species that represent
1 Surfaces We got our first hint of the importance of surface processes in the mass spectrum of a high vacuum environment. The spectrum was dominated by water and carbon monoxide, species that represent
Chemistry Instrumental Analysis Lecture 34. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Critical Current Properties of HTS Twisted Stacked-Tape Cable in Subcooled- and Pressurized-Liquid Nitrogen
 IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Critical Current Properties of HTS Twisted Stacked-Tape Cable in Subcooled- and Pressurized-Liquid Nitrogen To cite this article:
IOP Conference Series: Materials Science and Engineering PAPER OPEN ACCESS Critical Current Properties of HTS Twisted Stacked-Tape Cable in Subcooled- and Pressurized-Liquid Nitrogen To cite this article:
Powder Surface Area and Porosity
 Powder Surface Area and Porosity Powder Technology Series Edited by B. Scarlett Department of Chemical Engineering University of Technology Loughborough Powder Surface Area and Porosity S. Lowell PhD Quantachrome
Powder Surface Area and Porosity Powder Technology Series Edited by B. Scarlett Department of Chemical Engineering University of Technology Loughborough Powder Surface Area and Porosity S. Lowell PhD Quantachrome
Electronic Supporting information (ESI) for
 Electronic Supporting information (ESI) for Experimental assessment of physical upper limit for hydrogen storage capacity at 20 K in densified MIL-101 monoliths Hyunchul Oh a, Dan Lupu b, Gabriela Blanita
Electronic Supporting information (ESI) for Experimental assessment of physical upper limit for hydrogen storage capacity at 20 K in densified MIL-101 monoliths Hyunchul Oh a, Dan Lupu b, Gabriela Blanita
Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces
 6 February 2002 Chemical Physics Letters 352 (2002) 318 322 www.elsevier.com/locate/cplett Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces Y.D. Kim a, R.M. Lynden-Bell b, *, A. Alavi
6 February 2002 Chemical Physics Letters 352 (2002) 318 322 www.elsevier.com/locate/cplett Evidence for partial dissociation of water on flat MgO(1 0 0) surfaces Y.D. Kim a, R.M. Lynden-Bell b, *, A. Alavi
Particle Size Determinations: Dynamic Light Scattering: page 161 text
 Particle Size Determinations: Dynamic Light Scattering: page 161 text Dynamic light scattering (also known as Photon Correlation Spectroscopy or Quasi- Elastic Light Scattering) is a technique which can
Particle Size Determinations: Dynamic Light Scattering: page 161 text Dynamic light scattering (also known as Photon Correlation Spectroscopy or Quasi- Elastic Light Scattering) is a technique which can
Surface Chemistry & States of Matter
 Surface Chemistry & States of Matter S. Sunil Kumar Lecturer in Chemistry 1. Adsorption is a. Colligative property b. Oxidation process c. Reduction process d. Surface phenomenon Ans. d 2. When adsorption
Surface Chemistry & States of Matter S. Sunil Kumar Lecturer in Chemistry 1. Adsorption is a. Colligative property b. Oxidation process c. Reduction process d. Surface phenomenon Ans. d 2. When adsorption
Diffusion and Adsorption in porous media. Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad
 Diffusion and Adsorption in porous media Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Devices used to Measure Diffusion in Porous Solids Modes of transport in
Diffusion and Adsorption in porous media Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Devices used to Measure Diffusion in Porous Solids Modes of transport in
WORK EXPRERIENCE EDUCATION:
 Curriculum Vitae of Dinesh Singh Rawat 410 West Freeman Apt#4, IL-62901 Ph: (618) 529-3637 Emails: (a) dines_rawat@hotmail.com (b) dinesh1_rawat@yahoo.com WORK EXPRERIENCE Research Assistant and Teaching
Curriculum Vitae of Dinesh Singh Rawat 410 West Freeman Apt#4, IL-62901 Ph: (618) 529-3637 Emails: (a) dines_rawat@hotmail.com (b) dinesh1_rawat@yahoo.com WORK EXPRERIENCE Research Assistant and Teaching
Inner-Triplet Quadrupole Magnet. Inner Support Structure. Cross Section at One Support Point
 Insulation--FermiLab Failure at CERN On Tuesday, March 27,2007 there was a serious failure in a highpressure test at CERN of a Fermilab-built inner-triplet series of three quadrupole magnets in the tunnel
Insulation--FermiLab Failure at CERN On Tuesday, March 27,2007 there was a serious failure in a highpressure test at CERN of a Fermilab-built inner-triplet series of three quadrupole magnets in the tunnel
CB FDHMT. Contract No GSI Helmholzzentrum für Schwerionenforschung GmbH Planckstraße Darmstadt. 15. December /11
 Contract No. 4500109716 GSI Helmholzzentrum für Schwerionenforschung GmbH Planckstraße 1 64291 Darmstadt 15. December 2013 1/11 CONTENTS 1. Introduction 2. Single Panda electrically cooled Detector 2.1.
Contract No. 4500109716 GSI Helmholzzentrum für Schwerionenforschung GmbH Planckstraße 1 64291 Darmstadt 15. December 2013 1/11 CONTENTS 1. Introduction 2. Single Panda electrically cooled Detector 2.1.
THE CERN CRYOGENIC TEST FACILITY FOR THE ATLAS BARREL TOROID MAGNETS
 EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 365 THE CERN CRYOGENIC TEST FACILITY FOR THE ATLAS BARREL TOROID MAGNETS
EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 365 THE CERN CRYOGENIC TEST FACILITY FOR THE ATLAS BARREL TOROID MAGNETS
Vacuum Technology for Particle Accelerators
 Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
Magnetic Property Measurement System
 Magnetic Property Measurement System Product Description Quantum Design's MPMS 3 represents the culmination of more than 3 years of development and design in the world of SQUID Magnetometry. Providing
Magnetic Property Measurement System Product Description Quantum Design's MPMS 3 represents the culmination of more than 3 years of development and design in the world of SQUID Magnetometry. Providing
Rough (low) vacuum : Medium vacuum : High vacuum (HV) : Ultrahigh vacuum (UHV) :
 4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
Helium two-phase flow in a thermosiphon open loop
 Presented at the COMSOL Conference 2009 Milan COMSOL Conference 2009 Milan October 14-16 2009 Helium two-phase flow in a thermosiphon open loop Florian Visentin, Bertrand Baudouy CEA Saclay Accelerator,
Presented at the COMSOL Conference 2009 Milan COMSOL Conference 2009 Milan October 14-16 2009 Helium two-phase flow in a thermosiphon open loop Florian Visentin, Bertrand Baudouy CEA Saclay Accelerator,
A PHOTON-STOP FOR THE VLHC-2 ENGINEERING DESIGN PART 1
 TD-01-023 04/01 A PHOTON-STOP FOR THE VLHC-2 ENGINEERING DESIGN PART 1 P. Bauer, K. Ewald, C. Darve, P. Limon, J.M. Rey, I. Terechkine, L. Imbasciati Fermilab, Technical Division Keywords: VLHC, photon-stop,
TD-01-023 04/01 A PHOTON-STOP FOR THE VLHC-2 ENGINEERING DESIGN PART 1 P. Bauer, K. Ewald, C. Darve, P. Limon, J.M. Rey, I. Terechkine, L. Imbasciati Fermilab, Technical Division Keywords: VLHC, photon-stop,
To move a particle in a (straight) line over a large distance
 2.1 Introduction to Vacuum Technology 2.1.1 Importance of Vacuum Technology for Processing and Characterization Under partial vacuum conditions (pressures orders of magnitude below ambient atmospheric
2.1 Introduction to Vacuum Technology 2.1.1 Importance of Vacuum Technology for Processing and Characterization Under partial vacuum conditions (pressures orders of magnitude below ambient atmospheric
ISO 9277 INTERNATIONAL STANDARD. Determination of the specific surface area of solids by gas adsorption BET method
 INTERNATIONAL STANDARD ISO 9277 Second edition 2010-09-01 Determination of the specific surface area of solids by gas adsorption BET method Détermination de l'aire massique (surface spécifique) des solides
INTERNATIONAL STANDARD ISO 9277 Second edition 2010-09-01 Determination of the specific surface area of solids by gas adsorption BET method Détermination de l'aire massique (surface spécifique) des solides
Evidence for structure sensitivity in the high pressure CO NO reaction over Pd(111) and Pd(100)
 Evidence for structure sensitivity in the high pressure CO NO reaction over Pd(111) and Pd(100) Scott M. Vesecky, Peijun Chen, Xueping Xu, and D. Wayne Goodman a) Department of Chemistry, Texas A&M University,
Evidence for structure sensitivity in the high pressure CO NO reaction over Pd(111) and Pd(100) Scott M. Vesecky, Peijun Chen, Xueping Xu, and D. Wayne Goodman a) Department of Chemistry, Texas A&M University,
Gas Chromatography. Presented By Mr. Venkateswarlu Mpharm KTPC
 Gas Chromatography Gas Chromatography Presented By Mr. Venkateswarlu Mpharm KTPC What is Gas Chromatography? It is also known as Gas-Liquid Chromatography (GLC) GAS CHROMATOGRAPHY Separation of gaseous
Gas Chromatography Gas Chromatography Presented By Mr. Venkateswarlu Mpharm KTPC What is Gas Chromatography? It is also known as Gas-Liquid Chromatography (GLC) GAS CHROMATOGRAPHY Separation of gaseous
Generation of vacuum (pumps) and measurements
 Generation of vacuum (pumps) and measurements Generation of vacuum (pumps): ᴥ pumping systems general considerations on their use and match with physical quantities introduced for the dimensioning of vacuum
Generation of vacuum (pumps) and measurements Generation of vacuum (pumps): ᴥ pumping systems general considerations on their use and match with physical quantities introduced for the dimensioning of vacuum
Report Liquid Hydrogen Target for the COMPASS Experiment
 CERN-ACC-2014-0290 Johan.Bremer@cern.ch Report Liquid Hydrogen Target for the COMPASS Experiment J. Bremer 1, N. Doshita 2, L. Dufay-Chanat 1, R. Geyer 3, G.K. Mallot 1, O. Pirotte 1, and B. Vullierme
CERN-ACC-2014-0290 Johan.Bremer@cern.ch Report Liquid Hydrogen Target for the COMPASS Experiment J. Bremer 1, N. Doshita 2, L. Dufay-Chanat 1, R. Geyer 3, G.K. Mallot 1, O. Pirotte 1, and B. Vullierme
TPD and FT-IRAS Investigation of Ethylene Oxide (EtO) Adsorption on a Au(211) Stepped Surface
 3886 Langmuir 2005, 21, 3886-3891 TPD and FT-IRAS Investigation of Ethylene Oxide (EtO) Adsorption on a Au(211) Stepped Surface Jooho Kim and Bruce E. Koel* Department of Chemistry, University of Southern
3886 Langmuir 2005, 21, 3886-3891 TPD and FT-IRAS Investigation of Ethylene Oxide (EtO) Adsorption on a Au(211) Stepped Surface Jooho Kim and Bruce E. Koel* Department of Chemistry, University of Southern
Supporting Information
 Supporting Information Highly Selective Carbon Dioxide Sorption in an Organic Molecular Porous Material Hyunuk Kim, Yonghwi Kim, Minyoung Yoon, Soyoung Lim, Se Min Park, Gon Seo, Kimoon Kim*, National
Supporting Information Highly Selective Carbon Dioxide Sorption in an Organic Molecular Porous Material Hyunuk Kim, Yonghwi Kim, Minyoung Yoon, Soyoung Lim, Se Min Park, Gon Seo, Kimoon Kim*, National
Generation of vacuum (pumps): Vacuum (pressure) measurements:
 Generation of vacuum (pumps) p and measurements Generation of vacuum (pumps): ᴥ pumping p systems general considerations on their use and match with physical quantities introduced for the dimensioning
Generation of vacuum (pumps) p and measurements Generation of vacuum (pumps): ᴥ pumping p systems general considerations on their use and match with physical quantities introduced for the dimensioning
VACUUM PUMPING METHODS
 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
The LHC: the energy, cooling, and operation. Susmita Jyotishmati
 The LHC: the energy, cooling, and operation Susmita Jyotishmati LHC design parameters Nominal LHC parameters Beam injection energy (TeV) 0.45 Beam energy (TeV) 7.0 Number of particles per bunch 1.15
The LHC: the energy, cooling, and operation Susmita Jyotishmati LHC design parameters Nominal LHC parameters Beam injection energy (TeV) 0.45 Beam energy (TeV) 7.0 Number of particles per bunch 1.15
Characterisation of Porous Hydrogen Storage Materials: Carbons, Zeolites, MOFs and PIMs
 Characterisation of Porous Hydrogen Storage Materials: Carbons, Zeolites, MOFs and PIMs Steven Tedds, a * Allan Walton, a Darren P. Broom, b and David Book a DOI:.39/c0fd00022a Electronic Supplementary
Characterisation of Porous Hydrogen Storage Materials: Carbons, Zeolites, MOFs and PIMs Steven Tedds, a * Allan Walton, a Darren P. Broom, b and David Book a DOI:.39/c0fd00022a Electronic Supplementary
Ultra-High Vacuum Technology. Sputter Ion Pumps l/s
 Ultra-High Vacuum Technology 30-400 l/s 181.06.01 Excerpt from the Product Chapter C15 Edition November 2007 Contents General General..........................................................................
Ultra-High Vacuum Technology 30-400 l/s 181.06.01 Excerpt from the Product Chapter C15 Edition November 2007 Contents General General..........................................................................
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Vacuum System in Accelerator
 Vacuum System in Accelerator -- geometrical structure effects on the pumping delay time -- Y. Saito, KEK, Tsukuba, Japan N. Matuda, Tokyo Denki Univ., Tokyo, Japan 1. Required vacuum condition; what kind
Vacuum System in Accelerator -- geometrical structure effects on the pumping delay time -- Y. Saito, KEK, Tsukuba, Japan N. Matuda, Tokyo Denki Univ., Tokyo, Japan 1. Required vacuum condition; what kind
