( KS A ) (1) , vapour, vapor (USA) , saturation vapour pressure. , standard reference conditions for gases. , degree of saturation
|
|
|
- Vernon Kevin Holt
- 5 years ago
- Views:
Transcription
1 ( KS A ) (1), standard reference conditions for gases 0, Pa (1 =760mmHg ), vacuum, low ( rough ) vacuum 100Pa, medium vacuum Pa, high vacuum Pa, ultra high vacuum ( UHV ) Pa, extreme ultrahigh vacuum ( XHV ) 10 8, pressure, : p, P, Pa pascal Pa, 1 Pa = 1 N m - 2, Torr 1 ( Pa ) Torr = Pa, partial pressure : i p i, : Pa, total pressure : p, P, Pa, gas, :,,,, (2000 )p16, non- condensable gas, vapour, vapor (USA), saturation vapour pressure P L, Pa, degree of saturation,, saturated vapour,, unsaturated vapour, number density of molecules :n, m -3, mean free path ( ) (,,,,, ), :,l, : m, collision rate :, : s -1, volume collision rate, :, : m -3 s -1 pv, quantity of gas : Pa m 3, diffusion ( ), diffusion coefficient, diffusivity : D, : m 2 s -1, viscous flow, Poissuille flpw ( ), Reynolds number, (, ),,, molecular flow
2 ( ), Knudsen number, (, ), intermediate flow, molecular effusion,, thermal transpiration 2,, molecule flow rate, molecule flux : q N, :s -1, molecule flow rate density : m -2 s -1 ( ), throughput pv : Q, : Pa m 3 s -1, mass flow rate : Q m, : kg s -1, volume flow rate : Q v, : m 3 s -1, molar flow rate : Q, : mol s -1, Maxwellian velocity distribution,, transmission probability, Clausing factor, conductance, 2, : C, U, G, : m 3 s -1, absorption, (absorbate), (absorbent), adsorption ( ) ( ), physical adsorption, physisorption, chemical adsorption, chemisorption, sorption ( ) ( ) 2,, accommodation coefficient :, impingement rate :,, : m -2 s -1, impingement rate, condensation rate,, condensation coefficient : c, sticking rate, sticking probability : s ( ), ( ), residence time :, : s, migration, desorption, outgassing, degassing, evaporation rate, outgassing or desorption or degassing rate pv : q d, : p a m s -1, m -2 s -1, permeation,,,
3 (2), vacuum pump, positive displacement pump,,, 2, piston vacuum pump, liquid ring vacuum pump ( ), oil-sealed rotary vacuum pump,,, sliding vane rotary vacuum pump 2,,,, ( ), rotary piston vacuum pump ( ),, ( ),,, rotary plunger vacuum pump,,, ( ), Roots vacuum pump ( ), gas ballast (vacuum) pump ( ), dry-sealed vacuum pump, kinetic vacuum pump 2 ( ), turbine vacuum pump, molecular drag pump, turbo molecular pump ( ), ejector vacuum pump ( ), liquid jet vacuum pump
4 ( ) ( ), gas jet vacuum pump ( ) ( ), vapour jet vacuum pump (, ), diffusion pump,, diffusion ejector pump, fractionating diffusion pump,, self purifying diffusion pump ( ), vapour pump, entrapment vacuum pump ( ), adsorption pump (, ), getter pump (, ), sublimation pump, ( ), getter ion pump,,, evaporation ion pump,, sputter ion pump, cryopump, cryosorption pump ( ), pump case, inlet, outlet,, blade, vane, discharge valve, vacuum pump oil,,, pump fluid, nozzle, nozzle throat, nozzle clearance, nozzle clearance area, jet, diffuser, ( ), diffuser throat,, vapour tube, vapour pipe, nozzle assembly, trap,, cold trap, sorption trap, baffle
5 , oil separator, oil purifier, main pump, backing vacuum pump, roughing vacuum pump, holding vacuum pump,, high vacuum pump, rough(or low) vacuum pump ( ), booster vacuum pump, appendage vacuum pump, throughput of a vacuum pump : S, : m 3 s -1, pumping speed, volume flow rate of a vacuum pump : S, : m 3 s -1, inlet pressure, maximum working pressure, backing pressure : P b,:, : Pa, critical backing pressure : P bc, : Pa, starting pressure, ultimate pressure of a pump : P u, : Pa, compression ratio, back diffusion of gas, back streaming, back migration (3), pressure gauge,, vacuum gauge,,,, gauge head ( ), nude gauge, gauge control unit, absolute vacuum gauge, differential vacuum gauge
6 , total pressure vacuum gauge, partial pressure vacuum gauge, partial pressure analyzer,, sensitivity, relative sensitivity factor,: ( ), ionization gauge coefficient : K, : Pa -1 ( ), equivalent nitrogen pressure X, X ray limit, liquid level manometer U, U tube manometer U, elastic element gauge (, ), diaphragm gauge, Bourdon gauge, compression gauge,, Mcleod gauge, viscosity gauge, thermal conductivity gauge, Pirani gauge, thermister gauge, Knudsen gauge 2, ionization vacuum gauge,, cold cathode ionization gauge, Penning gauge, cold cathode magnetron gauge, discharge tube indicator,, hot cathode ionization gauge 3, triode ionization gauge 3 - (B-A), Bayard Alpert gauge X, modulator gauge -, extractor gauge X, suppressor gauge
7 2 X, bent beam gauge, hot cathode magnetron gauge, radio frequency mass spectrometer 4 (4 ), quadrupole mass spectrometer 4, magnetic deflection mass spectrometer, trochoidal focusing mass spectrometer, omegatron mass spectrometer, time of flight mass spectrometer
VACUUM PUMPING METHODS
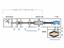 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
Vacuum I. G. Franchetti CAS - Bilbao. 30/5/2011 G. Franchetti 1
 Vacuum I G. Franchetti CAS - Bilbao 30/5/2011 G. Franchetti 1 Index Introduction to Vacuum Vacuum and the Beam Flow Regimes Creating Vacuum 30/5/2011 G. Franchetti 2 Vacuum in accelerators All beam dynamics
Vacuum I G. Franchetti CAS - Bilbao 30/5/2011 G. Franchetti 1 Index Introduction to Vacuum Vacuum and the Beam Flow Regimes Creating Vacuum 30/5/2011 G. Franchetti 2 Vacuum in accelerators All beam dynamics
Generation of vacuum (pumps) and measurements
 Generation of vacuum (pumps) and measurements Generation of vacuum (pumps): ᴥ pumping systems general considerations on their use and match with physical quantities introduced for the dimensioning of vacuum
Generation of vacuum (pumps) and measurements Generation of vacuum (pumps): ᴥ pumping systems general considerations on their use and match with physical quantities introduced for the dimensioning of vacuum
Lecture 3 Vacuum Science and Technology
 Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Lecture 4. Ultrahigh Vacuum Science and Technology
 Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Vacuum Pumps. Two general classes exist: Gas transfer physical removal of matter. Mechanical, diffusion, turbomolecular
 Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
Vacuum Technology Vacuum Pumps Two general classes exist: Gas transfer physical removal of matter Mechanical, diffusion, turbomolecular Adsorption entrapment of matter Cryo, sublimation, ion Mechanical
Generation of vacuum (pumps): Vacuum (pressure) measurements:
 Generation of vacuum (pumps) p and measurements Generation of vacuum (pumps): ᴥ pumping p systems general considerations on their use and match with physical quantities introduced for the dimensioning
Generation of vacuum (pumps) p and measurements Generation of vacuum (pumps): ᴥ pumping p systems general considerations on their use and match with physical quantities introduced for the dimensioning
To move a particle in a (straight) line over a large distance
 2.1 Introduction to Vacuum Technology 2.1.1 Importance of Vacuum Technology for Processing and Characterization Under partial vacuum conditions (pressures orders of magnitude below ambient atmospheric
2.1 Introduction to Vacuum Technology 2.1.1 Importance of Vacuum Technology for Processing and Characterization Under partial vacuum conditions (pressures orders of magnitude below ambient atmospheric
K n. III. Gas flow. 1. The nature of the gas : Knudsen s number. 2. Relative flow : Reynold s number R = ( dimensionless )
 III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
Physical Vapor Deposition
 Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Vacuum. Residual pressure can thwart the best cryogenic design. Each gas molecule collision carries ~kt from the hot exterior to the cold interior.
 Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
Vacuum II. G. Franchetti CAS - Bilbao. 30/5/2011 G. Franchetti 1
 Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
(1) E. & F. N. SPON London VACUUM MANUAL. Edited by. W. Steckelmacher. J. Yarwood. L. Holland
 Vacuum Manual VACUUM MANUAL Edited by L. Holland Formerly Director of Research Edwards High Vacuum; Associate Reader in Physics, Brunei University W. Steckelmacher Formerly Deputy Director of Research
Vacuum Manual VACUUM MANUAL Edited by L. Holland Formerly Director of Research Edwards High Vacuum; Associate Reader in Physics, Brunei University W. Steckelmacher Formerly Deputy Director of Research
PHYSICAL VAPOR DEPOSITION OF THIN FILMS
 PHYSICAL VAPOR DEPOSITION OF THIN FILMS JOHN E. MAHAN Colorado State University A Wiley-Interscience Publication JOHN WILEY & SONS, INC. New York Chichester Weinheim Brisbane Singapore Toronto CONTENTS
PHYSICAL VAPOR DEPOSITION OF THIN FILMS JOHN E. MAHAN Colorado State University A Wiley-Interscience Publication JOHN WILEY & SONS, INC. New York Chichester Weinheim Brisbane Singapore Toronto CONTENTS
Repetition: Physical Deposition Processes
 Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Previous Lecture. Electron beam lithoghraphy e - Electrons are generated in vacuum. Electron beams propagate in vacuum
 Previous Lecture Electron beam lithoghraphy e - Electrons are generated in vacuum Electron beams propagate in vacuum Lecture 6: Vacuum & plasmas Objectives From this vacuum lecture you will learn: What
Previous Lecture Electron beam lithoghraphy e - Electrons are generated in vacuum Electron beams propagate in vacuum Lecture 6: Vacuum & plasmas Objectives From this vacuum lecture you will learn: What
Vacuum techniques (down to 1 K)
 Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum. Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006
 Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
Physics,, Technology and Techniques of the Vacuum Kai Schwarzwälder, Institut für Physik Universität Basel October 6 th 2006 Outline Introduction and basics Defintion of Vacuum Vacuum A vacuum is a volume
COURSE CODE : 3072 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE
 COURSE TITLE : FLUID MECHANICS COURSE CODE : 307 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE MODULE TOPIC PERIOD 1 Properties of Fluids 0 Fluid Friction and Flow
COURSE TITLE : FLUID MECHANICS COURSE CODE : 307 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE MODULE TOPIC PERIOD 1 Properties of Fluids 0 Fluid Friction and Flow
Sputter Ion Pump (Ion Pump) By Biswajit
 Sputter Ion Pump (Ion Pump) By Biswajit 08-07-17 Sputter Ion Pump (Ion Pump) An ion pump is a type of vacuum pump capable of reaching pressures as low as 10 11 mbar under ideal conditions. An ion pump
Sputter Ion Pump (Ion Pump) By Biswajit 08-07-17 Sputter Ion Pump (Ion Pump) An ion pump is a type of vacuum pump capable of reaching pressures as low as 10 11 mbar under ideal conditions. An ion pump
Vacuum for Accelerators
 Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Experimental techniques of Surface Science and how a theorist understands them
 Experimental techniques of Surface Science and how a theorist understands them Karsten Reuter Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, D-14195 Berlin reuter@fhi-berlin.mpg.de 0.
Experimental techniques of Surface Science and how a theorist understands them Karsten Reuter Fritz-Haber-Institut der Max-Planck-Gesellschaft Faradayweg 4-6, D-14195 Berlin reuter@fhi-berlin.mpg.de 0.
Lecture 10. Vacuum Technology and Plasmas Reading: Chapter 10. ECE Dr. Alan Doolittle
 Lecture 10 Vacuum Technology and Plasmas Reading: Chapter 10 Vacuum Science and Plasmas In order to understand deposition techniques such as evaporation, sputtering,, plasma processing, chemical vapor
Lecture 10 Vacuum Technology and Plasmas Reading: Chapter 10 Vacuum Science and Plasmas In order to understand deposition techniques such as evaporation, sputtering,, plasma processing, chemical vapor
Rough (low) vacuum : Medium vacuum : High vacuum (HV) : Ultrahigh vacuum (UHV) :
 4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
Angular Correlation Experiments
 Angular Correlation Experiments John M. LoSecco April 2, 2007 Angular Correlation Experiments J. LoSecco Notre Dame du Lac Nuclear Spin In atoms one can use the Zeeman Effect to determine the spin state.
Angular Correlation Experiments John M. LoSecco April 2, 2007 Angular Correlation Experiments J. LoSecco Notre Dame du Lac Nuclear Spin In atoms one can use the Zeeman Effect to determine the spin state.
Earlier Lecture. In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide.
 41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
Introduction to Chemical Engineering Thermodynamics. Chapter 7. KFUPM Housam Binous CHE 303
 Introduction to Chemical Engineering Thermodynamics Chapter 7 1 Thermodynamics of flow is based on mass, energy and entropy balances Fluid mechanics encompasses the above balances and conservation of momentum
Introduction to Chemical Engineering Thermodynamics Chapter 7 1 Thermodynamics of flow is based on mass, energy and entropy balances Fluid mechanics encompasses the above balances and conservation of momentum
Vacuum Technology and film growth. Diffusion Resistor
 Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Conductance of an aperture
 n Vacuum φ A 4 How many molecules travel through A in a time t? P P A onductance of an aperture 3 q φa 3.64 0 ( T / M) na Volume of gas passing through A? Q Q Q / ΔP 3.64 ( T / M ) 3 dv/dt q/n 3.64 0 (
n Vacuum φ A 4 How many molecules travel through A in a time t? P P A onductance of an aperture 3 q φa 3.64 0 ( T / M) na Volume of gas passing through A? Q Q Q / ΔP 3.64 ( T / M ) 3 dv/dt q/n 3.64 0 (
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015. Solid. Liquid Commonly found on Gas Earth Plasma
 Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) STATES OF MATTER The Four States of Matter Solid } Liquid Commonly found on Gas Earth Plasma STATES OF MATTER Based upon particle
STATES OF MATTER STATES OF MATTER. The Four States of Matter 3/5/2015
 The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
The Four States of Matter Unit 10: States of Matter Lesson 10.1: States and Their Changes (Review) Solid } Liquid Commonly found on Gas Earth Plasma Based upon particle arrangement Based upon energy of
A Vacuum point of view
 Beam-Surface Interaction A Vacuum point of view F. Le Pimpec SLAC/NLC SSRL April 2004 1 Dynamic Vacuum You want to address the terms of this formula How to measure the Pressure? F. Le Pimpec - SLAC 2 Outline
Beam-Surface Interaction A Vacuum point of view F. Le Pimpec SLAC/NLC SSRL April 2004 1 Dynamic Vacuum You want to address the terms of this formula How to measure the Pressure? F. Le Pimpec - SLAC 2 Outline
CHAPTER EIGHT P U M P I N G O F L I Q U I D S
 CHAPTER EIGHT P U M P I N G O F L I Q U I D S Pupmps are devices for supplying energy or head to a flowing liquid in order to overcome head losses due to friction and also if necessary, to raise liquid
CHAPTER EIGHT P U M P I N G O F L I Q U I D S Pupmps are devices for supplying energy or head to a flowing liquid in order to overcome head losses due to friction and also if necessary, to raise liquid
Conductance of an aperture
 n Vacuum φ 4 How many molecules travel through in a time t? P P onductance of an aperture q φ.64 0 ( T / M) n Volume of gas passing through? Q Q dv/dt q/n.64 0 ( T / M) Q P Q / ΔP.64 (T / M ) l/s.64 0
n Vacuum φ 4 How many molecules travel through in a time t? P P onductance of an aperture q φ.64 0 ( T / M) n Volume of gas passing through? Q Q dv/dt q/n.64 0 ( T / M) Q P Q / ΔP.64 (T / M ) l/s.64 0
Hydrogen in the LCLS2 Beamline Vacuum
 Hydrogen in the LCLS2 Beamline Vacuum Anthony C. Crawford Fermilab Technical Div./SRF Development Dept. acc52@fnal.gov 13Oct15 This note demonstrates that the cold segments of the LCLS2 linac will cryopump
Hydrogen in the LCLS2 Beamline Vacuum Anthony C. Crawford Fermilab Technical Div./SRF Development Dept. acc52@fnal.gov 13Oct15 This note demonstrates that the cold segments of the LCLS2 linac will cryopump
Vacuum Technology for Particle Accelerators
 Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
CHEMISTRY Matter and Change. Chapter 12: States of Matter
 CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
Chemistry Instrumental Analysis Lecture 34. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Chemistry 4631 Instrumental Analysis Lecture 34 From molecular to elemental analysis there are three major techniques used for elemental analysis: Optical spectrometry Mass spectrometry X-ray spectrometry
Cryogenic Viscous Compressor Development and Modeling for the ITER Vacuum System
 Cryogenic Viscous Compressor Development and Modeling for the ITER Vacuum System L. R. Baylor, C.N. Barbier, S. K. Combs, R.C. Duckworth, T.D. Edgemon, S. J. Meitner, M.P. Hechler, D.A. Rasmussen, R. Kersevan*,
Cryogenic Viscous Compressor Development and Modeling for the ITER Vacuum System L. R. Baylor, C.N. Barbier, S. K. Combs, R.C. Duckworth, T.D. Edgemon, S. J. Meitner, M.P. Hechler, D.A. Rasmussen, R. Kersevan*,
CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES
 Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
MOLECULAR SURFACE PUMPING: CRYOPUMPING
 51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
 CE 6303 MECHANICS OF FLUIDS L T P C QUESTION BANK 3 0 0 3 UNIT I FLUID PROPERTIES AND FLUID STATICS PART - A 1. Define fluid and fluid mechanics. 2. Define real and ideal fluids. 3. Define mass density
CE 6303 MECHANICS OF FLUIDS L T P C QUESTION BANK 3 0 0 3 UNIT I FLUID PROPERTIES AND FLUID STATICS PART - A 1. Define fluid and fluid mechanics. 2. Define real and ideal fluids. 3. Define mass density
Ultra-High Vacuum Technology. Sputter Ion Pumps l/s
 Ultra-High Vacuum Technology 30-400 l/s 181.06.01 Excerpt from the Product Chapter C15 Edition November 2007 Contents General General..........................................................................
Ultra-High Vacuum Technology 30-400 l/s 181.06.01 Excerpt from the Product Chapter C15 Edition November 2007 Contents General General..........................................................................
Chapter 13 - States of Matter. Section 13.1 The nature of Gases
 Chapter 13 - States of Matter Section 13.1 The nature of Gases Kinetic energy and gases Kinetic energy: the energy an object has because of its motion Kinetic theory: all matter is made if particles in
Chapter 13 - States of Matter Section 13.1 The nature of Gases Kinetic energy and gases Kinetic energy: the energy an object has because of its motion Kinetic theory: all matter is made if particles in
CONCEPTS AND DEFINITIONS. Prepared by Engr. John Paul Timola
 CONCEPTS AND DEFINITIONS Prepared by Engr. John Paul Timola ENGINEERING THERMODYNAMICS Science that involves design and analysis of devices and systems for energy conversion Deals with heat and work and
CONCEPTS AND DEFINITIONS Prepared by Engr. John Paul Timola ENGINEERING THERMODYNAMICS Science that involves design and analysis of devices and systems for energy conversion Deals with heat and work and
UHV - Technology. Oswald Gröbner
 School on Synchrotron Radiation UHV - Technology Trieste, 20-21 April 2004 1) Introduction and some basics 2) Building blocks of a vacuum system 3) How to get clean ultra high vacuum 4) Desorption phenomena
School on Synchrotron Radiation UHV - Technology Trieste, 20-21 April 2004 1) Introduction and some basics 2) Building blocks of a vacuum system 3) How to get clean ultra high vacuum 4) Desorption phenomena
Index. b back diffusion, 392, 394 backflow, , 420 backing pressure critical, 398 backing pump, 445
 977 Index a absolute vacuum, 15 absorption, 223, 237, 463 absorption method, 659 AC motor, 256 accommodating time, 899 accommodation coefficient, 47, 171 173, 578 accommodation coefficient of tangential
977 Index a absolute vacuum, 15 absorption, 223, 237, 463 absorption method, 659 AC motor, 256 accommodating time, 899 accommodation coefficient, 47, 171 173, 578 accommodation coefficient of tangential
Applied Fluid Mechanics
 Applied Fluid Mechanics 1. The Nature of Fluid and the Study of Fluid Mechanics 2. Viscosity of Fluid 3. Pressure Measurement 4. Forces Due to Static Fluid 5. Buoyancy and Stability 6. Flow of Fluid and
Applied Fluid Mechanics 1. The Nature of Fluid and the Study of Fluid Mechanics 2. Viscosity of Fluid 3. Pressure Measurement 4. Forces Due to Static Fluid 5. Buoyancy and Stability 6. Flow of Fluid and
The Art of Cryogenics
 The Art of Cryogenics This page intentionally left blank The Art of Cryogenics Low-Temperature Experimental Techniques Guglielmo Ventura and Lara Risegari Amsterdam Boston Heidelberg London New York Oxford
The Art of Cryogenics This page intentionally left blank The Art of Cryogenics Low-Temperature Experimental Techniques Guglielmo Ventura and Lara Risegari Amsterdam Boston Heidelberg London New York Oxford
A SMALL GETTER ION-PUMP
 . 260 PHILIPS TECHNICAL REVIEW VOLUME 22 A SMALL GETTER ION-PUMP by A. KLOPFER *) and W. ERMRICH *). 621.528.5.6 The advances made in vacuum technique in the last ten years have given a new lease of life
. 260 PHILIPS TECHNICAL REVIEW VOLUME 22 A SMALL GETTER ION-PUMP by A. KLOPFER *) and W. ERMRICH *). 621.528.5.6 The advances made in vacuum technique in the last ten years have given a new lease of life
Compression and Expansion of Fluids
 CH2303 Chemical Engineering Thermodynamics I Unit V Compression and Expansion of Fluids Dr. M. Subramanian 26-Sep-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College
CH2303 Chemical Engineering Thermodynamics I Unit V Compression and Expansion of Fluids Dr. M. Subramanian 26-Sep-2011 Associate Professor Department of Chemical Engineering Sri Sivasubramaniya Nadar College
Chapter 7 The Energy Equation
 Chapter 7 The Energy Equation 7.1 Energy, Work, and Power When matter has energy, the matter can be used to do work. A fluid can have several forms of energy. For example a fluid jet has kinetic energy,
Chapter 7 The Energy Equation 7.1 Energy, Work, and Power When matter has energy, the matter can be used to do work. A fluid can have several forms of energy. For example a fluid jet has kinetic energy,
Industrial Instrumentation Prof. A. Barua Department of Electrical Engineering Indian Institute of Technology - Kharagpur
 Industrial Instrumentation Prof. A. Barua Department of Electrical Engineering Indian Institute of Technology - Kharagpur Lecture - 19 Low Pressure Measurement Welcome to the lesson 19 of Industrial Instrumentation.
Industrial Instrumentation Prof. A. Barua Department of Electrical Engineering Indian Institute of Technology - Kharagpur Lecture - 19 Low Pressure Measurement Welcome to the lesson 19 of Industrial Instrumentation.
Vacuum gas dynamics investigation and experimental results on the TRASCO-ADS Windowless Interface
 Vacuum gas dynamics investigation and experimental results on the TRASCO-ADS Windowless Interface P. Michelato, E. Bari, E. Cavaliere, L. Monaco, D. Sertore; INFN Milano - LASA - Italy A. Bonucci, R. Giannantonio,
Vacuum gas dynamics investigation and experimental results on the TRASCO-ADS Windowless Interface P. Michelato, E. Bari, E. Cavaliere, L. Monaco, D. Sertore; INFN Milano - LASA - Italy A. Bonucci, R. Giannantonio,
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators. Mauro Audi, Academic, Government & Research Marketing Manager
 ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
ION Pumps for UHV Systems, Synchrotrons & Particle Accelerators Mauro Audi, Academic, Government & Research Marketing Manager ION Pumps Agilent Technologies 1957-59 Varian Associates invents the first
CHAPTER 13. States of Matter. Kinetic = motion. Polar vs. Nonpolar. Gases. Hon Chem 13.notebook
 CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
Given Find Water Properties
 Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Influence of water on the total heat transfer in evacuated insulations
 Bavarian Center for Applied Energy Research Influence of water on the total heat transfer in evacuated insulations Ulrich Heinemann ZAE Bayern, Würzburg, Germany ulrich.heinemann@zae.uni-wuerzburg.de 7
Bavarian Center for Applied Energy Research Influence of water on the total heat transfer in evacuated insulations Ulrich Heinemann ZAE Bayern, Würzburg, Germany ulrich.heinemann@zae.uni-wuerzburg.de 7
6.5 Optical-Coating-Deposition Technologies
 92 Chapter 6 6.5 Optical-Coating-Deposition Technologies The coating process takes place in an evaporation chamber with a fully controlled system for the specified requirements. Typical systems are depicted
92 Chapter 6 6.5 Optical-Coating-Deposition Technologies The coating process takes place in an evaporation chamber with a fully controlled system for the specified requirements. Typical systems are depicted
SUMMER 14 EXAMINATION
 Important Instructions to examiners: 1) The answers should be examined by key words and not as word-to-word as given in the model answer scheme. 2) The model answer and the answer written by candidate
Important Instructions to examiners: 1) The answers should be examined by key words and not as word-to-word as given in the model answer scheme. 2) The model answer and the answer written by candidate
EXPERIMENT No.1 FLOW MEASUREMENT BY ORIFICEMETER
 EXPERIMENT No.1 FLOW MEASUREMENT BY ORIFICEMETER 1.1 AIM: To determine the co-efficient of discharge of the orifice meter 1.2 EQUIPMENTS REQUIRED: Orifice meter test rig, Stopwatch 1.3 PREPARATION 1.3.1
EXPERIMENT No.1 FLOW MEASUREMENT BY ORIFICEMETER 1.1 AIM: To determine the co-efficient of discharge of the orifice meter 1.2 EQUIPMENTS REQUIRED: Orifice meter test rig, Stopwatch 1.3 PREPARATION 1.3.1
Vacuum and Thin Film Technology
 1 Instructions for the Laboratory Course Vacuum and Thin Film Technology (Vakuum- und Dünnschichttechnik) Part of the Course 511.121 Advanced Laboratory Exercises (Praktikum für Fortgeschrittene) Graz
1 Instructions for the Laboratory Course Vacuum and Thin Film Technology (Vakuum- und Dünnschichttechnik) Part of the Course 511.121 Advanced Laboratory Exercises (Praktikum für Fortgeschrittene) Graz
Gases. Chapter 5. Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Gases Chapter 5 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Elements that exist as gases at 25 0 C and 1 atmosphere 2 3 Physical Characteristics of Gases
Gases Chapter 5 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Elements that exist as gases at 25 0 C and 1 atmosphere 2 3 Physical Characteristics of Gases
Unit Outline. I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI.
 Unit 10: Gases Unit Outline I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI. Real Gases I. Opening thoughts Have you ever: Seen a hot air balloon?
Unit 10: Gases Unit Outline I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI. Real Gases I. Opening thoughts Have you ever: Seen a hot air balloon?
Vacuum Solutions for Ion Thruster Testing
 Vacuum Solutions for Ion Thruster Testing Stefan Lausberg Application & Product Support VA 1.2 Mon 9:45 H25 DPG-Frühjahrstagung Regensburg 2016 Oerlikon Leybold Vacuum DPG-Frühjahrstagung Regensburg Stefan
Vacuum Solutions for Ion Thruster Testing Stefan Lausberg Application & Product Support VA 1.2 Mon 9:45 H25 DPG-Frühjahrstagung Regensburg 2016 Oerlikon Leybold Vacuum DPG-Frühjahrstagung Regensburg Stefan
7/16/2012. Characteristics of Gases. Chapter Five: Pressure is equal to force/unit area. Manometer. Gas Law Variables. Pressure-Volume Relationship
 7/6/0 Chapter Five: GASES Characteristics of Gases Uniformly fills any container. Mixes completely with any other gas. Exerts pressure on its surroundings. When subjected to pressure, its volume decreases.
7/6/0 Chapter Five: GASES Characteristics of Gases Uniformly fills any container. Mixes completely with any other gas. Exerts pressure on its surroundings. When subjected to pressure, its volume decreases.
General Chemistry I. Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University. Module 3: The Three States of Matter
 General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 3: The Three States of Matter Gas state (Equation of state: ideal gas and real gas). Liquid state
General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 3: The Three States of Matter Gas state (Equation of state: ideal gas and real gas). Liquid state
Roll No. :... Invigilator s Signature :.. CS/B.Tech (EIE)/SEM-5/EI-501/ INDUSTRIAL INSTRUMENTATION II
 Name : Roll No. :.... Invigilator s Signature :.. CS/B.Tech (EIE)/SEM-5/EI-501/2011-12 2011 INDUSTRIAL INSTRUMENTATION II Time Allotted : 3 Hours Full Marks : 70 The figures in the margin indicate full
Name : Roll No. :.... Invigilator s Signature :.. CS/B.Tech (EIE)/SEM-5/EI-501/2011-12 2011 INDUSTRIAL INSTRUMENTATION II Time Allotted : 3 Hours Full Marks : 70 The figures in the margin indicate full
A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings.
 Chapter 5 Gases Chapter 5 A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Copyright Cengage Learning. All rights reserved
Chapter 5 Gases Chapter 5 A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Copyright Cengage Learning. All rights reserved
Lecture 24. Ideal Gas Law and Kinetic Theory
 Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
SECOND ENGINEER REG. III/2 APPLIED HEAT
 SECOND ENGINEER REG. III/2 APPLIED HEAT LIST OF TOPICS A B C D E F G H I J K Pressure, Temperature, Energy Heat Transfer Internal Energy, Thermodynamic systems. First Law of Thermodynamics Gas Laws, Displacement
SECOND ENGINEER REG. III/2 APPLIED HEAT LIST OF TOPICS A B C D E F G H I J K Pressure, Temperature, Energy Heat Transfer Internal Energy, Thermodynamic systems. First Law of Thermodynamics Gas Laws, Displacement
States of Matter Unit
 Learning Target Notes Section 1: Matter and Energy What makes up matter? Matter is made of atoms and molecules that are in constant motion. Kinetic Theory of Matter A. Particles that make up matter are
Learning Target Notes Section 1: Matter and Energy What makes up matter? Matter is made of atoms and molecules that are in constant motion. Kinetic Theory of Matter A. Particles that make up matter are
Unit 8 Kinetic Theory of Gases. Chapter 13-14
 Unit 8 Kinetic Theory of Gases Chapter 13-14 This tutorial is designed to help students understand scientific measurements. Objectives for this unit appear on the next slide. Each objective is linked to
Unit 8 Kinetic Theory of Gases Chapter 13-14 This tutorial is designed to help students understand scientific measurements. Objectives for this unit appear on the next slide. Each objective is linked to
Chemical Vapor Deposition (CVD)
 Chemical Vapor Deposition (CVD) source chemical reaction film substrate More conformal deposition vs. PVD t Shown here is 100% conformal deposition ( higher temp has higher surface diffusion) t step 1
Chemical Vapor Deposition (CVD) source chemical reaction film substrate More conformal deposition vs. PVD t Shown here is 100% conformal deposition ( higher temp has higher surface diffusion) t step 1
Chapter 5. Mass and Energy Analysis of Control Volumes
 Chapter 5 Mass and Energy Analysis of Control Volumes Conservation Principles for Control volumes The conservation of mass and the conservation of energy principles for open systems (or control volumes)
Chapter 5 Mass and Energy Analysis of Control Volumes Conservation Principles for Control volumes The conservation of mass and the conservation of energy principles for open systems (or control volumes)
dmγ v dt = q E +q v B, (1)
 Vacuum I Giuliano Franchetti GSI Darmstadt, D-64291 Darmstadt, Germany Abstract This paper is an introduction to the basics of vacuum. It is intended for readers unfamiliar with the topic; more advanced
Vacuum I Giuliano Franchetti GSI Darmstadt, D-64291 Darmstadt, Germany Abstract This paper is an introduction to the basics of vacuum. It is intended for readers unfamiliar with the topic; more advanced
The Vacuum Case for KATRIN
 The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
10/16/2018. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 10/16/018 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 1 10/16/018 A Gas Uniformly fills any container.
10/16/018 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 1 10/16/018 A Gas Uniformly fills any container.
Warning!! Chapter 5 Gases. Chapter Objectives. Chapter Objectives. Chapter Objectives. Air Pollution
 Warning!! Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 5 Gases These slides contains visual aids for learning BUT they are NOT the actual lecture notes! Failure to attend to lectures most
Warning!! Larry Brown Tom Holme www.cengage.com/chemistry/brown Chapter 5 Gases These slides contains visual aids for learning BUT they are NOT the actual lecture notes! Failure to attend to lectures most
Cold / sticky system. Vincent Baglin. CERN AT-VAC, Geneva
 Cold / sticky system Vincent Baglin CERN AT-VAC, Geneva 1 Cold / sticky system?? Vacuum?? 2 Eureka?? 3 Cold / sticky system Outline 1. Cryopumping 2. Adsorption isotherms 3. Cryosorbers in cold systems
Cold / sticky system Vincent Baglin CERN AT-VAC, Geneva 1 Cold / sticky system?? Vacuum?? 2 Eureka?? 3 Cold / sticky system Outline 1. Cryopumping 2. Adsorption isotherms 3. Cryosorbers in cold systems
Why study gases? A Gas 10/17/2017. An understanding of real world phenomena. An understanding of how science works.
 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? n understanding of real world phenomena. n understanding of how science works. Gas Uniformly fills any container. Mixes completely
Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? n understanding of real world phenomena. n understanding of how science works. Gas Uniformly fills any container. Mixes completely
Part II. Pressure Measurements in the Range from 10 2 Pa to 10 8 Pa
 Part II Pressure Measurements in the Range from 10 2 Pa to 10 8 Pa Introduction The pressure range 10 2-10 8 Pa has been selected as it is the interval covering the highest variety of interests and applications
Part II Pressure Measurements in the Range from 10 2 Pa to 10 8 Pa Introduction The pressure range 10 2-10 8 Pa has been selected as it is the interval covering the highest variety of interests and applications
Part I. Temperature Measurements in the Range from 0.1 K to 300 K
 Part I Temperature Measurements in the Range from 0.1 K to 300 K Introduction Part I describes modem methods for measuring temperatures lower than O C based on the use of substances that are gaseous at
Part I Temperature Measurements in the Range from 0.1 K to 300 K Introduction Part I describes modem methods for measuring temperatures lower than O C based on the use of substances that are gaseous at
William В. Brower, Jr. A PRIMER IN FLUID MECHANICS. Dynamics of Flows in One Space Dimension. CRC Press Boca Raton London New York Washington, D.C.
 William В. Brower, Jr. A PRIMER IN FLUID MECHANICS Dynamics of Flows in One Space Dimension CRC Press Boca Raton London New York Washington, D.C. Table of Contents Chapter 1 Fluid Properties Kinetic Theory
William В. Brower, Jr. A PRIMER IN FLUID MECHANICS Dynamics of Flows in One Space Dimension CRC Press Boca Raton London New York Washington, D.C. Table of Contents Chapter 1 Fluid Properties Kinetic Theory
Given Find Water Properties
 Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Cryogenic Engineering Prof. M. D. Atrey Department of Mechanical Engineering Indian Institute of Technology, Bombay
 Cryogenic Engineering Prof. M. D. Atrey Department of Mechanical Engineering Indian Institute of Technology, Bombay Lecture No. # 41 Instrumentation in Cryogenics (Refer Slide Time: 00:31) So, welcome
Cryogenic Engineering Prof. M. D. Atrey Department of Mechanical Engineering Indian Institute of Technology, Bombay Lecture No. # 41 Instrumentation in Cryogenics (Refer Slide Time: 00:31) So, welcome
The First Law of Thermodynamics. By: Yidnekachew Messele
 The First Law of Thermodynamics By: Yidnekachew Messele It is the law that relates the various forms of energies for system of different types. It is simply the expression of the conservation of energy
The First Law of Thermodynamics By: Yidnekachew Messele It is the law that relates the various forms of energies for system of different types. It is simply the expression of the conservation of energy
Proudly serving laboratories worldwide since 1979 CALL for Refurbished & Certified Lab Equipment
 www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +1.847.913.0777 for Refurbished & Certified Lab Equipment Applied Biosystems QStar Pulsar i Features of the API QSTAR Pulsar i The
www.ietltd.com Proudly serving laboratories worldwide since 1979 CALL +1.847.913.0777 for Refurbished & Certified Lab Equipment Applied Biosystems QStar Pulsar i Features of the API QSTAR Pulsar i The
Lecture Presentation. Chapter 10. Gases. John D. Bookstaver St. Charles Community College Cottleville, MO Pearson Education, Inc.
 Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
EXECUTIVE SUMMARY. especially in last 50 years. Industries, especially power industry, are the large anthropogenic
 EXECUTIVE SUMMARY Introduction The concentration of CO 2 in atmosphere has increased considerably in last 100 years, especially in last 50 years. Industries, especially power industry, are the large anthropogenic
EXECUTIVE SUMMARY Introduction The concentration of CO 2 in atmosphere has increased considerably in last 100 years, especially in last 50 years. Industries, especially power industry, are the large anthropogenic
A 200 kev Electrostatic Accelerator
 A 200 kev Electrostatic Accelerator P.Brady, B. Winey, and M.Yuly Department of Physics Houghton College Houghton, NY 14744 I. Abstract A small 200 kev electrostatic electron accelerator is being constructed.
A 200 kev Electrostatic Accelerator P.Brady, B. Winey, and M.Yuly Department of Physics Houghton College Houghton, NY 14744 I. Abstract A small 200 kev electrostatic electron accelerator is being constructed.
Biological Process Engineering An Analogical Approach to Fluid Flow, Heat Transfer, and Mass Transfer Applied to Biological Systems
 Biological Process Engineering An Analogical Approach to Fluid Flow, Heat Transfer, and Mass Transfer Applied to Biological Systems Arthur T. Johnson, PhD, PE Biological Resources Engineering Department
Biological Process Engineering An Analogical Approach to Fluid Flow, Heat Transfer, and Mass Transfer Applied to Biological Systems Arthur T. Johnson, PhD, PE Biological Resources Engineering Department
Chapter 3 Phases of Matter Physical Science
 Chapter 3 Phases of Matter Physical Science CH 3- States of Matter 1 What makes up matter? What is the difference between a solid, a liquid, and a gas? What kind of energy do all particles of matter have?
Chapter 3 Phases of Matter Physical Science CH 3- States of Matter 1 What makes up matter? What is the difference between a solid, a liquid, and a gas? What kind of energy do all particles of matter have?
Chapter 10 Gases Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.
 Chapter 10 Gases 10.1 Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.1) Unlike liquids and solids, gases expand to fill their
Chapter 10 Gases 10.1 Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.1) Unlike liquids and solids, gases expand to fill their
Fluid Mechanics. du dy
 FLUID MECHANICS Technical English - I 1 th week Fluid Mechanics FLUID STATICS FLUID DYNAMICS Fluid Statics or Hydrostatics is the study of fluids at rest. The main equation required for this is Newton's
FLUID MECHANICS Technical English - I 1 th week Fluid Mechanics FLUID STATICS FLUID DYNAMICS Fluid Statics or Hydrostatics is the study of fluids at rest. The main equation required for this is Newton's
INSTITUTE OF AERONAUTICAL ENGINEERING Dundigal, Hyderabad AERONAUTICAL ENGINEERING QUESTION BANK : AERONAUTICAL ENGINEERING.
 Course Name Course Code Class Branch INSTITUTE OF AERONAUTICAL ENGINEERING Dundigal, Hyderabad - 00 0 AERONAUTICAL ENGINEERING : Mechanics of Fluids : A00 : II-I- B. Tech Year : 0 0 Course Coordinator
Course Name Course Code Class Branch INSTITUTE OF AERONAUTICAL ENGINEERING Dundigal, Hyderabad - 00 0 AERONAUTICAL ENGINEERING : Mechanics of Fluids : A00 : II-I- B. Tech Year : 0 0 Course Coordinator
Chapter 16 Simple Kinetic Theory of Matter
 Chapter 16 Simple Kinetic Theory of Matter MCQ 1: Air trapped inside a single-piston-cylinder exerts a pressure of 760 mmhg. If its volume is increased by 30% at a constant temperature, then the pressure
Chapter 16 Simple Kinetic Theory of Matter MCQ 1: Air trapped inside a single-piston-cylinder exerts a pressure of 760 mmhg. If its volume is increased by 30% at a constant temperature, then the pressure
Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources
 CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
10/15/2015. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 0/5/05 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 0/5/05 A Gas fills any container. completely
0/5/05 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 0/5/05 A Gas fills any container. completely
