Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila
|
|
|
- Philip Stokes
- 6 years ago
- Views:
Transcription
1 Baz, Par-6 and apkc are not required for axon or dendrite specification in Drosophila Melissa M. Rolls and Chris Q. Doe, Inst. Neurosci and Inst. Mol. Biol., HHMI, Univ. Oregon, Eugene, Oregon Correspondence should be addressed to CQD Abstract Par-3/Baz, Par-6, and apkc are evolutionarily-conserved regulators of cell polarity, and overexpression experiments implicate them as axon determinants in vertebrate hippocampal neurons. Here we assay their mutant and overexpression phenotypes in Drosophila neurons; we show that mutant neurons have normal axon and dendrite morphology, including axonal remodeling during metamorphosis, and overexpression does not affect axon or dendrite specification. Thus, Baz/Par- 6/aPKC are not essential for axon specification in Drosophila. Neurons are highly polarized cells that establish two distinct compartments axonal and somatodendritic that are thought to be distinct at many levels: from ability to synthesize proteins, to the cytoskeleton, to plasma membrane proteins 1. Recently, Par- 3 (Baz in Drosophila), Par-6 and apkcλ/apkcζ (atypical protein kinase C, apkc in Drosophila) have been proposed to be axon determinants in vertebrate neurons 2,3. In cultured hippocampal neurons, Par-3/Par-6/aPKC concentrate at tips of growing axons when they are growing beyond unpolarized processes 2,3. Overexpression of Par-3 or Par- 6 3 or expression of a truncated dominant-negative Par-3 protein 2 results in additional axon-like long processes at this stage. These results suggest Par complex proteins could be axon determinants. However, this hypothesis has not been directly tested by examining the mutant phenotype for any of these genes, nor by in vivo analysis of their neuronal function. If, as suggested by overexpression, Par-3 and Par-6 are crucial players
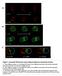
2 in generating axons, one might expect a failure of axon formation in their absence. Here we test use null mutants to eliminate Drosophila Baz, Par-6 and apkc from developing neurons in vivo and assay axon specification and outgrowth. We use previously characterized null alleles of baz, par-6, or apkc for our analysis 4-6, and we used the MARCM system to make GFP-marked homozygous mutant clones 7,8. We assayed the mushroom body neurons, a group of several hundred interneurons that are generated from just four precursors (called neuroblasts) that sequentially generate neurons from embryonic through pupal stages, and have been extensively characterized for axon and dendrite outgrowth and remodeling (Figure 1A) 7,8. We induced homozyogous mutant clones in mushroom body neuroblasts at first larval instar and assayed homozygous mutant neuronal progeny 3 days later in third larval instar brains, or 7-10 days later in adult brains (Figure 1B and C). Wild-type third larval instar neuronal clones showed the expected morphology, with dendrites filling the calyx adjacent to the cell bodies and branched dorsal and medial axon lobes (Figure 1B, left panel). Surprisingly, baz, par-6, or apkc null mutant neurons all showed normal axon and dendrite morphology (Figure 1B, right panels). We next assayed mutant clones in adults, so we could score development of the latest-born α β and αβ neurons (born during late third instar and pupal stages 7,8 ). Importantly, these neurons project into both dorsal and medial axon lobes in adults, while first-born γ neurons do not project into the adult dorsal lobe (Figure 1A), so we can be confident that we are scoring only the late-born α β and αβ axons. Wild type adult neuronal clones showed α β and αβ neurons extending into the dorsal axon lobe (Figure 1C, upper left panel). In mutant clones, α β neurons showed completely normal axon projections that reached the tip of the dorsal lobe (Figure 1C right panels). We observed fewer α β and αβ neurons, as expected due to failure of mutant neuroblasts to generate a full lineage. apkc mutant clones were about 1/4 the size of wild-type clones 4, consistent with proliferation arrest at about the time the neuroblast swiches from producing γ neurons to generating α β neurons. The small number of α β neurons has an unexpected advantage, however, in that it allows us to score axon morphology with single cell resolution. We can clearly see that individual mutant α β neurons have
3 normal axon projections in this experiment. Thus, we conclude that genetically null mutant baz, par-6, or apkc neurons show no defects in axon specification or outgrowth. Our results indicate that Baz/Par-6/aPKC are not required for axon specification, but we were concerned that Baz/Par-6/aPKC proteins might be so stable that protein present in the single parental neuroblast at the time of clone induction may persist for many days and be inherited about a hundred genetically mutant progeny neurons, where it might be at sufficiently high levels to promote axon specification. To address this possibility, we assayed Par-6 and apkc protein levels in mutant clones at third larval instar, three days after clone induction, and found no detectable protein in par-6 or apkc mutant neuroblasts or neurons (Supplementary Figure 1 and Reference 6), and thus α β and αβ neurons born subsequently must have initiated axon and dendrite formation in the absence of protein. In addition, we have observed that all three proteins (Baz, Par-6, apkc) are partitioned into the neuroblast and out of the differentiating neurons during each asymmetric neuroblast division 4. This would make it virtually impossible for residual protein to be passively inherited by the mutant neuronal progeny. We conclude that Baz, Par-6 or apkc are not required for axon or dendrite specification in mushroom body interneurons. We next tested whether Baz, Par-6 and apkc play a role in axon remodeling during metamorphosis. In wild type brains, γ neuron axons are pruned back early in pupal life and later regrow adult-specific axons into only the medial lobe 7 (Figure 1A). We find that baz, par-6, or apkc null mutant neurons show a normal pattern of γ axon remodeling, with adult-specific projections only into the medial lobe (Figure 1C). The remodeling event occurs many days after clone induction and after third larval instar stage where no protein is detectable in clones, so we can be sure there is no protein present in the neurons during the process of axon remodeling. We conclude that Baz, Par- 6 and apkc are not required for axon outgrowth during metamorphosis. Previous work has shown that overexpression of mammalian Par-3 (Baz) or Par-6 led to growth of extra axon-like processes in cultured hippocampal neurons 2,3. To determine if overexpression of Baz or Par-6 has similar effects on Drosophila neurons in vivo, we overexpressed them in alternating segments of the embryo, and assayed axon development of the acc motor neuron using an eve::tau-lacz transgene 9. Wild type acc
4 neurons had no detectable Baz or Par-6 during axon outgrowth (data not shown), whereas misexpression resulted in clearly detectable expression in the CNS (Figure 2A). Despite these high levels, overexpression of Baz or Par-6 showed no effect on the number, orientation, or growth of acc axons (arrowheads, Figure 2A). In addition, we overexpressed Baz or Par-6 proteins at higher than normal levels in larval mushroom body neurons using a Gal4 driver expressed at the time of axon initiation (OK107) or a Gal4 driver expressed slightly later (201Y); in both cases we found no defect in axon or dendrite morphology (Figure 2B). We conclude that overexpression of Baz or Par-6 in neurons does not promote axon specification or block dendrite formation. Although we have not found a role for Baz, Par-6 or apkc in axon specification, nor do we observe these proteins targeted to the tips of growing axons in vivo or in vitro (data not shown), we do observe very striking polarized localization of the proteins in mature sensory or central neurons. In sensory neurons, Baz is localized to the dendrite tips (Supplementary Figure 2A). In mushroom body interneurons, Baz is axon-specific; Par-6 is enriched in young axons as well as present at low levels throughout central interneurons; and apkc is perisynaptic (Supplementary Figure 2B). Interestingly, the three proteins have distinct subcellular distribution in these mature neurons, rather than being co-localized as observed for epithelia, Drosophila neuroblasts, and vertebrate neurons 10. We propose that all three proteins have subtype-specific functions in mature neurons. In this work we show that Drosophila Baz, Par-6, or apkc are not required for axon specification in vivo, and that their overexpression has no effect on axon specification or outgrowth. In contrast, in cultured mammalian hippocampal neurons overexpression of Par-3 or Par-6 results in multiple axon-like processes, leading to the hypothesis that these proteins are axon determinants 3. How can we reconcile these apparently paradoxical results? One possibility is that vertebrate neurons require Par complex proteins for axon specification, while Drosophila neurons do not. If this is the case it will be very interesting to learn how different molecular pathways in mammals and flies generate the same functional subcellular domain (the axon). Another possibility is that neither fly nor vertebrate neurons use Par proteins to specify axon identity when they are in their normal in vivo context. Cultured hippocampal neurons are separated
5 from normal external polarity cues, and may use a different mechanism for axon specification. Polarity cues from surrounding cells may also inhibit neurons in vivo from changing polarity in response to extra Par-3 or Par-6, explaining the different effects of overexpressing these proteins in Drosophila and hippocampal neurons. A third possibility is that the overexpression experiments may give artifactual results that are not relevant to the in vivo function of the proteins, as overexpressed proteins may be at dramatically higher levels compared to the in vivo situation, and mislocalization can result in the proteins taking on new functions in places they are not normally present. Loss of function and overexpression experiments that assay vertebrate neurons in vivo or in slice preparations will be crucial for fully understanding the role of Par complex proteins in vertebrate axon specification. Although Baz, Par-6 and apkc are not required for axon specification in Drosophila, nor do we find them at the tips of growing axons as reported for cultured hippocampal neurons 2,3, we find that they do have interesting polarized distributions in mature neurons. Baz is present at sensory dendrite tips and in interneuron axons. apkc is perisynaptic in interneurons, and Par-6 is present throughout interneurons, but enriched in young axons. These results raise the possibility that Par-6 and apkc have independent functions in mature neurons- in contrast to most other cell types where they act together as part of a protein complex to perform a common function. Our results raise the interesting possibility that Par proteins may govern aspects of axon or dendrite function in mature mammalian neurons. Materials and Methods The mutant alleles used have all been described as null 4,6,5 and were: baz 4, apkc K06403, and par-6 Δ226. The eve::tau-lacz line was provided by Miki Fujioka and Jim Jaynes (Thomas Jefferson Univ.). 201Y-Gal4 and OK107-Gal4 were obtained from Bloomington stock center. Clones were generated using standard methods 7. Brains were dissected from wandering third instar larvae, or from 2-5 day old adults using standard methods 7. Crosses to generate MARCM clones are available upon request. Antibody staining was performed as described 4, with the addition of guinea pig anti-baz (used at 1:500).
6 Acknowledgements We thank Howard Hughes Medical Institute (CQD) and the Damon Runyon Cancer Research Foundation (MMR) for funding, and the Bloomington Stock Center for fly stocks. Andreas Wodarz kindly sent us antibodies. We thank Sarah Siegrist for comments on the manuscript. Figure Legends Figure 1. baz, par-6, and apkc mutant neurons form normal axons and dendrites in vivo (A) Timeline and classes of mushroom body neurons. Four neuroblasts per hemisphere generate mushroom body neuroblasts continuously from the embryonic to pupal stage. γ neurons are produced first, with dendrites emerging close to the cell body and branched dorsal and medial axon lobes. At pupariation γ axons are pruned back to the branch point; they regrow only into the medial lobe (far right). α β neurons are produced at the end of the third larval instar, and αβ neurons are born last, at the beginning of the pupal stage. α β and αβ neurons always project axons to both dorsal and medial axon lobes. (B) GFP-marked neuronal clones made in first instar and scored 3 days later in late third instar larvae. Wild type clone (left panel) or baz, par-6, or apkc null mutant clones (right panels; n=8 for baz, n=7 for par-6 and n=24 for apkc). GFP-actin marks neurons in wild-type, baz and par-6 clones; mcd8-gfp marks the apkc clone. The dorsal axon lobes point up, and medial axon lobes to the right, except for the baz clone where it points to the left. Position of cell bodies (C), dendrites (D) and two axons lobes (A) are indicated in the wild-type clone. (C) GFP-marked neuronal clones made in first instar and scored in 2 to 5 day old adult brains. Wild type clone (left panel) or baz, par-6, or apkc null mutant clones (right panels; n=2 for baz, n=4 for par-6 and n=12 for apkc). Only dorsal and medial axon lobes are shown. GFP clone marker (upper panels); Scrib highlights the axon lobes and shows that the whole dorsal and medial lobes are present in these compressed confocal z- series (middle panels); merged images (lower panels).
7 Figure 2. Overexpression of Baz or Par-6 does not affect axon morphology or number. (A) Baz or Par-6 overexpression in alternating segments of the embryonic CNS generated no difference in acc pioneer motor neuron axon number and outgrowth (arrows; n=28 for Baz and n=14 for Par-6). Full-length par-6 or baz was expressed in alternating segments of the embryo before and during the time when the acc motor neuron extends its axon. The acc motor neuron was identified by an eve::tau-lacz transgene which is expressed in only three neurons per hemisegment. Embryos are paired-gal4/uas-baz or UAS-par-6; eve::tau-lacz/+ shown at stage 12. (B) Baz and Par-6 overexpression in developing larval mushroom body neurons does not affect axon or dendrite morphology. Panels show the Dlg axon/dendrite marker in wild type brains (left) and overexpression brains (right). Dendrites (D) and the dorsal and medial axon lobes (A) are indicated. Larvae are 201Y-Gal4 / UAS-Baz or UAS-Par-6 shown at third larval instar. Similar results were observed when the OK107-Gal4 driver was used to drive baz or par-6 expression (data not shown). Supplementary Figure 1. Par-6 is not detectable in par-6 mutant mushroom body neuroblasts. GFP-marked par-6 mutant clone in the mushroom body of a third instar brain, dotted lines. Neuroblasts, highlighted by Miranda (Mir), have high levels of Par-6 outside the clone and none inside the clone. In addition, apkc mutant clones have no detectable apkc protein at this stage 4. Supplementary Figure 2. Baz, Par-6 and apkc are polarized in mature sensory or CNS neurons (A) Baz and apkc localization in ciliated neurons of the embryonic chordotonal organ. 22C10 (green) reveals chordotonal neuron morphology. Baz (red) localized to two puncta; one is on the dendrite (data not shown). apkc (red) and Par-6 (not shown) localized in the scolopale support cell that surrounds the sensory dendrite. baz, par-6, and apkc mutant embryos or larvae show no defects in sensory dendrite morphology, but we cannot be sure all maternal protein is gone at the time of dendrite formation (data not shown).
8 (B) Baz, Par-6, and apkc polarized localization in third instar larval mushroom body interneurons. Dlg or GFP mark mushroom body architecture. Confocal sections are indicated by the red line (right schematic). Each of the single confocal sections shown contains mushroom body dendrites (D) and proximal axons (A) so that relative protein levels in dendrites and axons can be directly compared. The dendrite region contains synapses, the proximal axons do not. Arrows indicate young axons which extend down the middle of the axon bundle as they develop. References 1. Craig, A.M. & Banker, G. Annu Rev Neurosci 17, (1994). 2. Nishimura, T. et al. Nat Cell Biol 6, (2004). 3. Shi, S.H., Jan, L.Y. & Jan, Y.N. Cell 112, (2003). 4. Rolls, M.M., Albertson, R., Shih, H.P., Lee, C.Y. & Doe, C.Q. J Cell Biol 163, (2003). 5. Petronczki, M. & Knoblich, J.A. Nat Cell Biol 3, 43-9 (2001). 6. Wodarz, A., Ramrath, A., Kuchinke, U. & Knust, E. Nature 402, (1999). 7. Lee, T., Lee, A. & Luo, L. Development 126, (1999). 8. Jefferis, G.S., Marin, E.C., Watts, R.J. & Luo, L. Curr Opin Neurobiol 12, 80-6 (2002). 9. Fujioka, M., Emi-Sarker, Y., Yusibova, G.L., Goto, T. & Jaynes, J.B. Development 126, (1999). 10. Henrique, D. & Schweisguth, F. Curr Opin Genet Dev 13, (2003).
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
DOI: 10.1038/ncb3267 Supplementary Figure 1 A group of genes required for formation or orientation of annular F-actin bundles and aecm ridges: RNAi phenotypes and their validation by standard mutations.
Chapter 4 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays.
 Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Evaluating a potential interaction between deltex and git in Drosophila: genetic interaction, gene overexpression and cell biology assays. The data described in chapter 3 presented evidence that endogenous
Development of the Drosophila mushroom bodies: sequential generation of three distinct types of neurons from a neuroblast
 Development 126, 4065-4076 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8625 4065 Development of the Drosophila mushroom bodies: sequential generation of three distinct types
Development 126, 4065-4076 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV8625 4065 Development of the Drosophila mushroom bodies: sequential generation of three distinct types
Sang-Chul Nam a,1, Bibhash Mukhopadhyay b, Kwang-Wook Choi a,b,c,
 Developmental Biology 306 (2007) 624 635 www.elsevier.com/locate/ydbio Antagonistic functions of Par-1 kinase and protein phosphatase 2A are required for localization of Bazooka and photoreceptor morphogenesis
Developmental Biology 306 (2007) 624 635 www.elsevier.com/locate/ydbio Antagonistic functions of Par-1 kinase and protein phosphatase 2A are required for localization of Bazooka and photoreceptor morphogenesis
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo
 Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
Fig. S1. Expression pattern of moody-gal4 in third instar. Maximum projection illustrating a dissected moody-gal4>ngfp L3 larva stained for Repo (magenta), Fas2 (blue) and GFP (green) in overview (A) and
NN-A29932: Supplementary Data 1 Diversity and Wiring Variability of Olfactory Local Interneurons in the Drosophila Antennal Lobe
 NN-A29932: Supplementary Data 1 Diversity and Wiring Variability of Olfactory Local Interneurons in the Drosophila Antennal Lobe Ya-Hui Chou, Maria L. Spletter, Emre Yaksi, Jonathan C. S. Leong, Rachel
NN-A29932: Supplementary Data 1 Diversity and Wiring Variability of Olfactory Local Interneurons in the Drosophila Antennal Lobe Ya-Hui Chou, Maria L. Spletter, Emre Yaksi, Jonathan C. S. Leong, Rachel
Nature Neuroscience: doi: /nn.2662
 Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
Supplementary Figure 1 Atlastin phylogeny and homology. (a) Maximum likelihood phylogenetic tree based on 18 Atlastin-1 sequences using the program Quicktree. Numbers at internal nodes correspond to bootstrap
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
doi:10.1038/nature10923 Supplementary Figure 1 Ten-a and Ten-m antibody and cell type specificities. a c, Representative single confocal sections of a Drosophila NMJ stained with antibodies to Ten-a (red),
Midterm 1. Average score: 74.4 Median score: 77
 Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Midterm 1 Average score: 74.4 Median score: 77 NAME: TA (circle one) Jody Westbrook or Jessica Piel Section (circle one) Tue Wed Thur MCB 141 First Midterm Feb. 21, 2008 Only answer 4 of these 5 problems.
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
www.sciencesignaling.org/cgi/content/full/6/301/ra98/dc1 Supplementary Materials for Regulation of Epithelial Morphogenesis by the G Protein Coupled Receptor Mist and Its Ligand Fog Alyssa J. Manning,
Sensory/ Motor Systems March 10, 2008
 Sensory/ Motor Systems March 10, 2008 Greg Suh greg.suh@med.nyu.edu Title: Chemical Senses- periphery Richard Axel and Linda Buck Win Nobel Prize for Olfactory System Research 2 3 After Leslie Vosshal
Sensory/ Motor Systems March 10, 2008 Greg Suh greg.suh@med.nyu.edu Title: Chemical Senses- periphery Richard Axel and Linda Buck Win Nobel Prize for Olfactory System Research 2 3 After Leslie Vosshal
Supplementary Materials for
 www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
www.sciencemag.org/cgi/content/full/science.1244624/dc1 Supplementary Materials for Cytoneme-Mediated Contact-Dependent Transport of the Drosophila Decapentaplegic Signaling Protein Sougata Roy, Hai Huang,
Developmental Biology
 Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
Developmental Biology 366 (2012) 204 217 Contents lists available at SciVerse ScienceDirect Developmental Biology journal homepage: www.elsevier.com/locate/developmentalbiology Developmental changes in
MCDB 4777/5777 Molecular Neurobiology Lecture 29 Neural Development- In the beginning
 MCDB 4777/5777 Molecular Neurobiology Lecture 29 Neural Development- In the beginning Learning Goals for Lecture 29 4.1 Describe the contributions of early developmental events in the embryo to the formation
MCDB 4777/5777 Molecular Neurobiology Lecture 29 Neural Development- In the beginning Learning Goals for Lecture 29 4.1 Describe the contributions of early developmental events in the embryo to the formation
Follow this and additional works at: Part of the Medical Sciences Commons
 Bucknell University Bucknell Digital Commons Master s Theses Student Theses 2010 The overexpression of homeotic complex gene Ultrabithorax in the post-embryonic neuronal lineages of the ventral nervous
Bucknell University Bucknell Digital Commons Master s Theses Student Theses 2010 The overexpression of homeotic complex gene Ultrabithorax in the post-embryonic neuronal lineages of the ventral nervous
A mosaic genetic screen for genes necessary for Drosophila mushroom body neuronal morphogenesis
 Development 130, 1203-1213 2003 The Company of Biologists Ltd doi:10.1242/dev.00319 1203 A mosaic genetic screen for genes necessary for Drosophila mushroom body neuronal morphogenesis John E. Reuter 1,
Development 130, 1203-1213 2003 The Company of Biologists Ltd doi:10.1242/dev.00319 1203 A mosaic genetic screen for genes necessary for Drosophila mushroom body neuronal morphogenesis John E. Reuter 1,
Supplementary Information
 Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Supplementary Information Supplementary Figure 1. JAK/STAT in early wing development (a-f) Wing primordia of second instar larvae of the indicated genotypes labeled to visualize expression of upd mrna
Analysis of non-olfactory sensory pathways that are necessary for the olfactory associative learning of the Drosophila mushroom body
 15 12601 B 2009 2011 21300117 Analysis of non-olfactory sensory pathways that are necessary for the olfactory associative learning of the Drosophila mushroom body ITO, Kei 00311192 The mushroom body is
15 12601 B 2009 2011 21300117 Analysis of non-olfactory sensory pathways that are necessary for the olfactory associative learning of the Drosophila mushroom body ITO, Kei 00311192 The mushroom body is
Report. Functional Diversity of Robo Receptor Immunoglobulin Domains Promotes Distinct Axon Guidance Decisions
 Current Biology 20, 567 572, March 23, 2010 ª2010 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2010.02.021 Functional Diversity of Robo Receptor Immunoglobulin Domains Promotes Distinct Axon Guidance
Current Biology 20, 567 572, March 23, 2010 ª2010 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2010.02.021 Functional Diversity of Robo Receptor Immunoglobulin Domains Promotes Distinct Axon Guidance
Current Opinion in Neurobiology 2002, 12: /02/$ see front matter 2002 Elsevier Science Ltd. All rights reserved.
 80 Development of neuronal connectivity in Drosophila antennal lobes and mushroom bodies Gregory SXE Jefferis*, Elizabeth C Marin, Ryan J Watts and Liqun Luo* Recent advances in the study of the connectivity
80 Development of neuronal connectivity in Drosophila antennal lobes and mushroom bodies Gregory SXE Jefferis*, Elizabeth C Marin, Ryan J Watts and Liqun Luo* Recent advances in the study of the connectivity
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
doi:10.1038/nature11589 Supplementary Figure 1 Ciona intestinalis and Petromyzon marinus neural crest expression domain comparison. Cartoon shows dorsal views of Ciona mid gastrula (left) and Petromyzon
The Midline Protein Regulates Axon Guidance by Blocking the Reiteration of Neuroblast Rows within the Drosophila Ventral Nerve Cord
 The Midline Protein Regulates Axon Guidance by Blocking the Reiteration of Neuroblast Rows within the Drosophila Ventral Nerve Cord Mary Ann Manavalan, Ivana Gaziova, Krishna Moorthi Bhat* Department of
The Midline Protein Regulates Axon Guidance by Blocking the Reiteration of Neuroblast Rows within the Drosophila Ventral Nerve Cord Mary Ann Manavalan, Ivana Gaziova, Krishna Moorthi Bhat* Department of
Polarity and intracellular compartmentalization of Drosophila neurons
 Neural Development This Provisional PDF corresponds to the article as it appeared upon acceptance. Copyedited and fully formatted PDF and full text (HTML) versions will be made available soon. Polarity
Neural Development This Provisional PDF corresponds to the article as it appeared upon acceptance. Copyedited and fully formatted PDF and full text (HTML) versions will be made available soon. Polarity
Nature Biotechnology: doi: /nbt Supplementary Figure 1. Overexpression of YFP::GPR-1 in the germline.
 Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Supplementary Figure 1 Overexpression of YFP::GPR-1 in the germline. The pie-1 promoter and 3 utr were used to express yfp::gpr-1 in the germline. Expression levels from the yfp::gpr-1(cai 1.0)-expressing
Respective roles of the DRL receptor and its ligand WNT5 in Drosophila mushroom body development
 RESEARCH ARTICLE 3089 Development 134, 3089-3097 (2007) doi:10.1242/dev.02876 Respective roles of the DRL receptor and its ligand WNT5 in Drosophila mushroom body development Nicola Grillenzoni*, Adrien
RESEARCH ARTICLE 3089 Development 134, 3089-3097 (2007) doi:10.1242/dev.02876 Respective roles of the DRL receptor and its ligand WNT5 in Drosophila mushroom body development Nicola Grillenzoni*, Adrien
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
Supplementary Figure 1 Sns and Duf co-localise in embryonic nephrocytes a-c, Wild-type stage 11 (a),14 (b) and 16 (c) embryos stained with anti-duf (green) and anti-sns (red). Higher magnification images
10/2/2015. Chapter 4. Determination and Differentiation. Neuroanatomical Diversity
 Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Chapter 4 Determination and Differentiation Neuroanatomical Diversity 1 Neurochemical diversity: another important aspect of neuronal fate Neurotransmitters and their receptors Excitatory Glutamate Acetylcholine
Nature Neuroscience: doi: /nn.2717
 Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Supplementary Fig. 1. Dendrite length is not secondary to body length. Dendrite growth proceeds independently of the rate of body growth and decreases in rate in adults. n 20 on dendrite measurement, n
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae. BioNB4910 Cornell University.
 Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
Modulation of central pattern generator output by peripheral sensory cells in Drosophila larvae BioNB4910 Cornell University Goals 1) Observe the behavioral effects of remotely activating different populations
Drosophila homeodomain protein Nkx6 coordinates motoneuron
 Research article 5233 Drosophila homeodomain protein Nkx6 coordinates motoneuron subtype identity and axonogenesis Heather T. Broihier 1, *,,, Alexander Kuzin 2,, Yi Zhu 1, Ward Odenwald 2 and James B.
Research article 5233 Drosophila homeodomain protein Nkx6 coordinates motoneuron subtype identity and axonogenesis Heather T. Broihier 1, *,,, Alexander Kuzin 2,, Yi Zhu 1, Ward Odenwald 2 and James B.
Asymmetric Prospero localization is required to generate mixed neuronal/glial lineages in the Drosophila CNS
 Development 128, 4103-4112 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV3509 4103 Asymmetric Prospero localization is required to generate mixed neuronal/glial lineages in
Development 128, 4103-4112 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV3509 4103 Asymmetric Prospero localization is required to generate mixed neuronal/glial lineages in
Cell biology: Death drags down the neighbourhood
 Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Cell biology: Death drags down the neighbourhood The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published Publisher Vasquez,
Supplementary Figure 1. Nature Genetics: doi: /ng.3848
 Supplementary Figure 1 Phenotypes and epigenetic properties of Fab2L flies. A- Phenotypic classification based on eye pigment levels in Fab2L male (orange bars) and female (yellow bars) flies (n>150).
Supplementary Figure 1 Phenotypes and epigenetic properties of Fab2L flies. A- Phenotypic classification based on eye pigment levels in Fab2L male (orange bars) and female (yellow bars) flies (n>150).
Cdc42 acts downstream of Bazooka to regulate neuroblast polarity through Par-6 apkc
 3200 Research Article Cdc42 acts downstream of Bazooka to regulate neuroblast polarity through Par-6 apkc Scott X. Atwood 1, Chiswili Chabu 2, Rhiannon R. Penkert 1, Chris Q. Doe 2 and Kenneth E. Prehoda
3200 Research Article Cdc42 acts downstream of Bazooka to regulate neuroblast polarity through Par-6 apkc Scott X. Atwood 1, Chiswili Chabu 2, Rhiannon R. Penkert 1, Chris Q. Doe 2 and Kenneth E. Prehoda
Axon guidance I. Paul Garrity March 15, /9.013
 Axon guidance I Paul Garrity March 15, 2004 7.68/9.013 Neuronal Wiring: Functional Framework of the Nervous System Stretch reflex circuit Early theories of axonogenesis Schwann: many neurons link to form
Axon guidance I Paul Garrity March 15, 2004 7.68/9.013 Neuronal Wiring: Functional Framework of the Nervous System Stretch reflex circuit Early theories of axonogenesis Schwann: many neurons link to form
Essential Roles of Drosophila RhoA in the Regulation of Neuroblast Proliferation and Dendritic but Not Axonal Morphogenesis
 Neuron, Vol. 25, 307 316, February, 2000, Copyright 2000 by Cell Press Essential Roles of Drosophila RhoA in the Regulation of Neuroblast Proliferation and Dendritic but Not Axonal Morphogenesis Tzumin
Neuron, Vol. 25, 307 316, February, 2000, Copyright 2000 by Cell Press Essential Roles of Drosophila RhoA in the Regulation of Neuroblast Proliferation and Dendritic but Not Axonal Morphogenesis Tzumin
Article. The Conserved Discs-large Binding Partner Banderuola Regulates Asymmetric Cell Division in Drosophila
 Current Biology 24, 1811 1825, August 18, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.06.059 The Conserved Discs-large Binding Partner Banderuola Regulates Asymmetric
Current Biology 24, 1811 1825, August 18, 2014 ª2014 Elsevier Ltd All rights reserved http://dx.doi.org/10.1016/j.cub.2014.06.059 The Conserved Discs-large Binding Partner Banderuola Regulates Asymmetric
Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway
 Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway Justin Hogan, Meagan Valentine, Chris Cox, Kristy Doyle, Simon Collier* Department
Two Frizzled Planar Cell Polarity Signals in the Drosophila Wing Are Differentially Organized by the Fat/Dachsous Pathway Justin Hogan, Meagan Valentine, Chris Cox, Kristy Doyle, Simon Collier* Department
Reading. Lecture VI. Making Connections 9/17/12. Bio 3411 Lecture VI. Making Connections. Bio 3411 Monday September 17, 2012
 Lecture VI. Making Connections Bio 3411 Monday September 17, 2012!! 1! Reading NEUROSCIENCE: 5 th ed, pp!507?536! 4 th ed, pp 577-609 Bentley, D., & Caudy, M. (1983). Nature, 304(5921), 62-65. Dickson,
Lecture VI. Making Connections Bio 3411 Monday September 17, 2012!! 1! Reading NEUROSCIENCE: 5 th ed, pp!507?536! 4 th ed, pp 577-609 Bentley, D., & Caudy, M. (1983). Nature, 304(5921), 62-65. Dickson,
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Role of Mitochondrial Remodeling in Programmed Cell Death in
 Developmental Cell, Vol. 12 Supplementary Data Role of Mitochondrial Remodeling in Programmed Cell Death in Drosophila melanogaster Gaurav Goyal, Brennan Fell, Apurva Sarin, Richard J. Youle, V. Sriram.
Developmental Cell, Vol. 12 Supplementary Data Role of Mitochondrial Remodeling in Programmed Cell Death in Drosophila melanogaster Gaurav Goyal, Brennan Fell, Apurva Sarin, Richard J. Youle, V. Sriram.
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290
 Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Shavenbaby Couples Patterning to Epidermal Cell Shape Control. Chanut-Delalande H, Fernandes I, Roch F, Payre F, Plaza S (2006) PLoS Biol 4(9): e290 Question (from Introduction): How does svb control the
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction
 Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
Anterograde Activin Signaling Regulates Postsynaptic Membrane Potential and GluRIIA/B Abundance at the Drosophila Neuromuscular Junction Myung-Jun Kim, Michael B. O Connor* Department of Genetics, Cell
Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis
 Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis Marta Zlatic 1,2,3 *, Feng Li 1, Maura Strigini 4, Wesley Grueber 2, Michael Bate 1 * 1 Department of Zoology, University
Positional Cues in the Drosophila Nerve Cord: Semaphorins Pattern the Dorso-Ventral Axis Marta Zlatic 1,2,3 *, Feng Li 1, Maura Strigini 4, Wesley Grueber 2, Michael Bate 1 * 1 Department of Zoology, University
Different cell types exhibit different degrees of complexity in
 Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Distinct roles of Bazooka and Stardust in the specification of Drosophila photoreceptor membrane architecture Yang Hong, Larry Ackerman, Lily Yeh Jan, and Yuh-Nung Jan* Howard Hughes Medical Institute
Supporting Information
 Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Supporting Information Cao et al. 10.1073/pnas.1306220110 Gram - bacteria Hemolymph Cytoplasm PGRP-LC TAK1 signalosome Imd dfadd Dredd Dnr1 Ikk signalosome P Relish Nucleus AMP and effector genes Fig.
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila
 Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Varicose and cheerio collaborate with pebble to mediate semaphorin-1a reverse signaling in Drosophila Sangyun Jeong a,1, Da-som Yang a, Young Gi Hong a, Sarah P. Mitchell b,c, Matthew P. rown b,c, and
Green Fluorescent Protein (GFP) Today s Nobel Prize in Chemistry
 In the news: High-throughput sequencing using Solexa/Illumina technology The copy number of each fetal chromosome can be determined by direct sequencing of DNA in cell-free plasma from pregnant women Confession:
In the news: High-throughput sequencing using Solexa/Illumina technology The copy number of each fetal chromosome can be determined by direct sequencing of DNA in cell-free plasma from pregnant women Confession:
A Role for Kinesin Heavy Chain in Controlling Vesicle Transport into Dendrites in
 A Role for Kinesin Heavy Chain in Controlling Vesicle Transport into Dendrites in Drosophila Kristina Schimmelpfeng Henthorn* Meike Sabina Roux#, Cheryl Herrera* and Lawrence S.B. Goldstein* * Howard Hughes
A Role for Kinesin Heavy Chain in Controlling Vesicle Transport into Dendrites in Drosophila Kristina Schimmelpfeng Henthorn* Meike Sabina Roux#, Cheryl Herrera* and Lawrence S.B. Goldstein* * Howard Hughes
RPM-1 is localized to distinct subcellular compartments and regulates axon length in GABAergic motor neurons
 Opperman and Grill Neural Development 14, 9:1 RESERCH RTICLE Open ccess RPM-1 is localized to distinct subcellular compartments and regulates axon length in Gergic motor neurons Karla J Opperman and rock
Opperman and Grill Neural Development 14, 9:1 RESERCH RTICLE Open ccess RPM-1 is localized to distinct subcellular compartments and regulates axon length in Gergic motor neurons Karla J Opperman and rock
MCN. Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases Regulate Axon Guidance in Drosophila
 MCN Molecular and Cellular Neuroscience 17, 274 291 (2001) doi:10.1006/mcne.2000.0939, available online at http://www.idealibrary.com on Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases
MCN Molecular and Cellular Neuroscience 17, 274 291 (2001) doi:10.1006/mcne.2000.0939, available online at http://www.idealibrary.com on Complex Genetic Interactions among Four Receptor Tyrosine Phosphatases
The Control of Semaphorin-1a-Mediated Reverse Signaling by Opposing Pebble and RhoGAPp190 Functions in Drosophila
 Article The Control of Semaphorin-1a-Mediated Reverse Signaling by Opposing Pebble and RhoGAPp190 Functions in Drosophila Sangyun Jeong, 1 Katarina Juhaszova, 1 and Alex L. Kolodkin 1, 1 Solomon H. Snyder
Article The Control of Semaphorin-1a-Mediated Reverse Signaling by Opposing Pebble and RhoGAPp190 Functions in Drosophila Sangyun Jeong, 1 Katarina Juhaszova, 1 and Alex L. Kolodkin 1, 1 Solomon H. Snyder
Drosophila adult external sense organ lineage
 Development 126, 2063-2071 (1999) rinted in Great Britain The Company of Biologists Limited 1999 DEV8583 2063 rospero distinguishes sibling cell fate without asymmetric localization in the Drosophila adult
Development 126, 2063-2071 (1999) rinted in Great Britain The Company of Biologists Limited 1999 DEV8583 2063 rospero distinguishes sibling cell fate without asymmetric localization in the Drosophila adult
SUPPLEMENTARY INFORMATION
 Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
Supplementary Notes Downregulation of atlastin does not affect secretory traffic We investigated whether Atlastin might play a role in secretory traffic. Traffic impairment results in disruption of Golgi
Building the brain (1): Evolutionary insights
 Building the brain (1): Evolutionary insights Historical considerations! Initial insight into the general role of the brain in human behaviour was already attained in antiquity and formulated by Hippocrates
Building the brain (1): Evolutionary insights Historical considerations! Initial insight into the general role of the brain in human behaviour was already attained in antiquity and formulated by Hippocrates
Cell division genes promote asymmetric interaction between Numb and Notch in the Drosophila CNS
 Development 126, 2759-2770 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5300 2759 Cell division genes promote asymmetric interaction between Numb and Notch in the Drosophila
Development 126, 2759-2770 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV5300 2759 Cell division genes promote asymmetric interaction between Numb and Notch in the Drosophila
purpose of this Chapter is to highlight some problems that will likely provide new
 119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
119 Chapter 6 Future Directions Besides our contributions discussed in previous chapters to the problem of developmental pattern formation, this work has also brought new questions that remain unanswered.
Reading: Chapter 5, pp ; Reference chapter D, pp Problem set F
 Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
Mosaic Analysis Reading: Chapter 5, pp140-141; Reference chapter D, pp820-823 Problem set F Twin spots in Drosophila Although segregation and recombination in mitosis do not occur at the same frequency
 DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
DOI: 10.1038/ncb2819 Gαi3 / Actin / Acetylated Tubulin Gαi3 / Actin / Acetylated Tubulin a a Gαi3 a Actin Gαi3 WT Gαi3 WT Gαi3 WT b b Gαi3 b Actin Gαi3 KO Gαi3 KO Gαi3 KO # # Figure S1 Loss of protein
Segment boundary formation in Drosophila embryos
 Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Segment boundary formation in Drosophila embryos Development 130, August 2003 Camilla W. Larsen, Elizabeth Hirst, Cyrille Alexandre and Jean Paul Vincent 1. Introduction: - Segment boundary formation:
Patterning Axon Targeting of Olfactory Receptor Neurons by Coupled Hedgehog Signaling at Two Distinct Steps
 Patterning Axon Targeting of Olfactory Receptor Neurons by Coupled Hedgehog Signaling at Two Distinct Steps Ya-Hui Chou, 1,2 Xiaoyan Zheng, 1,3 Philip A. Beachy, 1,3 and Liqun Luo 1,2, * 1 Howard Hughes
Patterning Axon Targeting of Olfactory Receptor Neurons by Coupled Hedgehog Signaling at Two Distinct Steps Ya-Hui Chou, 1,2 Xiaoyan Zheng, 1,3 Philip A. Beachy, 1,3 and Liqun Luo 1,2, * 1 Howard Hughes
Linking Cell Fate, Trajectory Choice, and Target Selection: Genetic Analysis of Sema-2b in Olfactory Axon Targeting
 Article Linking Cell Fate, Trajectory Choice, and Target Selection: Genetic Analysis of Sema-2b in Olfactory Axon Targeting William J. Joo, 1,2 Lora B. Sweeney, 1,2,4 Liang Liang, 1,3 and Liqun Luo 1,2,
Article Linking Cell Fate, Trajectory Choice, and Target Selection: Genetic Analysis of Sema-2b in Olfactory Axon Targeting William J. Joo, 1,2 Lora B. Sweeney, 1,2,4 Liang Liang, 1,3 and Liqun Luo 1,2,
Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila
 Hereditas 147: 237 242 (2010) Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila ANJA C. NAGEL, JENS SCHMID, JASMIN S. AUER, ANETTE PREISS and
Hereditas 147: 237 242 (2010) Constitutively active Protein kinase D acts as negative regulator of the Slingshot-phosphatase in Drosophila ANJA C. NAGEL, JENS SCHMID, JASMIN S. AUER, ANETTE PREISS and
Diverse Functions of N-Cadherin in Dendritic and Axonal Terminal Arborization of Olfactory Projection Neurons
 Neuron, Vol. 42, 63 75, April 8, 2004, Copyright 2004 by Cell Press Diverse Functions of N-Cadherin in Dendritic and Axonal Terminal Arborization of Olfactory Projection Neurons Haitao Zhu and Liqun Luo*
Neuron, Vol. 42, 63 75, April 8, 2004, Copyright 2004 by Cell Press Diverse Functions of N-Cadherin in Dendritic and Axonal Terminal Arborization of Olfactory Projection Neurons Haitao Zhu and Liqun Luo*
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function
 Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
Drosophila Spastin Regulates Synaptic Microtubule Networks and Is Required for Normal Motor Function Nina Tang Sherwood 1, Qi Sun 1 1, Mingshan Xue 2 2, Bing Zhang 2, Kai Zinn 1 * Open access, freely available
Crk-associated substrate (Cas) signaling protein functions with integrins to specify axon guidance during development
 RESEARCH ARTICLE 2337 Development 134, 2337-2347 (2007) doi:10.1242/dev.004242 Crk-associated substrate (Cas) signaling protein functions with integrins to specify axon guidance during development Zhiyu
RESEARCH ARTICLE 2337 Development 134, 2337-2347 (2007) doi:10.1242/dev.004242 Crk-associated substrate (Cas) signaling protein functions with integrins to specify axon guidance during development Zhiyu
Drosophila IAP1-mediated ubiquitylation controls activation of the initiator caspase DRONC independent of protein degradation
 University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 9-1-2011 Drosophila IAP1-mediated ubiquitylation controls
University of Massachusetts Medical School escholarship@umms Molecular, Cell and Cancer Biology Publications Molecular, Cell and Cancer Biology 9-1-2011 Drosophila IAP1-mediated ubiquitylation controls
Conclusions. The experimental studies presented in this thesis provide the first molecular insights
 C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
C h a p t e r 5 Conclusions 5.1 Summary The experimental studies presented in this thesis provide the first molecular insights into the cellular processes of assembly, and aggregation of neural crest and
Intrinsic regulation of enteroendocrine fate by Numb
 Article Intrinsic regulation of enteroendocrine fate by Numb Jérémy Sallé 1,2, Louis Gervais 1,2, Benjamin Boumard 1,2,3, Marine Stefanutti 1,2, Katarzyna Siudeja 1,2 & Allison J Bardin 1,2,* Abstract
Article Intrinsic regulation of enteroendocrine fate by Numb Jérémy Sallé 1,2, Louis Gervais 1,2, Benjamin Boumard 1,2,3, Marine Stefanutti 1,2, Katarzyna Siudeja 1,2 & Allison J Bardin 1,2,* Abstract
C. elegans L1 cell adhesion molecule functions in axon guidance
 C. elegans L1 cell adhesion molecule functions in axon guidance Biorad Lihsia Chen Dept. of Genetics, Cell Biology & Development Developmental Biology Center C. elegans embryogenesis Goldstein lab, UNC-Chapel
C. elegans L1 cell adhesion molecule functions in axon guidance Biorad Lihsia Chen Dept. of Genetics, Cell Biology & Development Developmental Biology Center C. elegans embryogenesis Goldstein lab, UNC-Chapel
Cell-Type-Specific TEV Protease Cleavage Reveals Cohesin Functions in Drosophila Neurons
 Article Cell-Type-Specific TEV Protease Cleavage Reveals Cohesin Functions in Drosophila Neurons Andrea Pauli, 1 Friederike Althoff, 2,6 Raquel A. Oliveira, 1,6 Stefan Heidmann, 3 Oren Schuldiner, 4 Christian
Article Cell-Type-Specific TEV Protease Cleavage Reveals Cohesin Functions in Drosophila Neurons Andrea Pauli, 1 Friederike Althoff, 2,6 Raquel A. Oliveira, 1,6 Stefan Heidmann, 3 Oren Schuldiner, 4 Christian
09/30/2017. Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center
 09/30/2017 Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs
09/30/2017 Kyu-Sun Lee, Ph.D. Metabolism & Neurophysiology Research Group Hazard Monitoring BNT Res Center Conflict of interest disclosure None Committee of Scientific Affairs Committee of Scientific Affairs
Chapter 18 Lecture. Concepts of Genetics. Tenth Edition. Developmental Genetics
 Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Chapter 18 Lecture Concepts of Genetics Tenth Edition Developmental Genetics Chapter Contents 18.1 Differentiated States Develop from Coordinated Programs of Gene Expression 18.2 Evolutionary Conservation
Figure 1. Automated 4D detection and tracking of endosomes and polarity markers.
 A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
A t = 0 z t = 90 B t = 0 z t = 90 C t = 0-90 t = 0-180 t = 0-300 Figure 1. Automated 4D detection and tracking of endosomes and polarity markers. A: Three different z planes of a dividing SOP shown at
Drosophila Homeodomain Protein dhb9 Directs Neuronal Fate via Crossrepressive and Cell-Nonautonomous Mechanisms
 Neuron, Vol. 35, 39 50, July 3, 2002, Copyright 2002 by Cell Press Drosophila Homeodomain Protein dhb9 Directs Neuronal Fate via Crossrepressive and Cell-Nonautonomous Mechanisms Heather T. Broihier and
Neuron, Vol. 35, 39 50, July 3, 2002, Copyright 2002 by Cell Press Drosophila Homeodomain Protein dhb9 Directs Neuronal Fate via Crossrepressive and Cell-Nonautonomous Mechanisms Heather T. Broihier and
1
 http://photos1.blogger.com/img/13/2627/640/screenhunter_047.jpg 1 The Nose Knows http://graphics.jsonline.com/graphics/owlive/img/mar05/sideways.one0308_big.jpg 2 http://www.stlmoviereviewweekly.com/sitebuilder/images/sideways-253x364.jpg
http://photos1.blogger.com/img/13/2627/640/screenhunter_047.jpg 1 The Nose Knows http://graphics.jsonline.com/graphics/owlive/img/mar05/sideways.one0308_big.jpg 2 http://www.stlmoviereviewweekly.com/sitebuilder/images/sideways-253x364.jpg
Lineage, cell polarity and inscuteable function in the peripheral nervous system of the Drosophila embryo
 Development 128, 631-643 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV5434 631 Lineage, cell polarity and inscuteable function in the peripheral nervous system of the Drosophila
Development 128, 631-643 (2001) Printed in Great Britain The Company of Biologists Limited 2001 DEV5434 631 Lineage, cell polarity and inscuteable function in the peripheral nervous system of the Drosophila
Drosophila homologs of mammalian TNF/TNFRrelated molecules regulate segregation of Miranda/Prospero in neuroblasts
 The EMBO Journal (2006) 25, 5783 5793 & 2006 European Molecular Biology Organization All Rights Reserved 0261-4189/06 www.embojournal.org Drosophila homologs of mammalian TNF/TNFRrelated molecules regulate
The EMBO Journal (2006) 25, 5783 5793 & 2006 European Molecular Biology Organization All Rights Reserved 0261-4189/06 www.embojournal.org Drosophila homologs of mammalian TNF/TNFRrelated molecules regulate
Structure of the Vertical and Horizontal System Neurons of the Lobula Plate in Drosophila
 THE JOURNAL OF COMPARATIVE NEUROLOGY 454:470 481 (2002) Structure of the Vertical and Horizontal System Neurons of the Lobula Plate in Drosophila ETHAN K. SCOTT, 1 * THOMAS RAABE, 2 AND LIQUN LUO 1,3 1
THE JOURNAL OF COMPARATIVE NEUROLOGY 454:470 481 (2002) Structure of the Vertical and Horizontal System Neurons of the Lobula Plate in Drosophila ETHAN K. SCOTT, 1 * THOMAS RAABE, 2 AND LIQUN LUO 1,3 1
Axon Guidance. Multiple decision points along a growing axon s trajectory Different types of axon guidance cues:
 Axon Guidance Multiple decision points along a growing axon s trajectory Different types of axon guidance cues: Contact mediated - requires direct contact by growth cone Long range - growth cone responds
Axon Guidance Multiple decision points along a growing axon s trajectory Different types of axon guidance cues: Contact mediated - requires direct contact by growth cone Long range - growth cone responds
THE PROBLEMS OF DEVELOPMENT. Cell differentiation. Cell determination
 We emphasize these points from Kandel in Bi/CNS 150 Bi/CNS/NB 150: Neuroscience Read Lecture Lecture Friday, October 2, 2015 Development 1: pp 5-10 Introduction Brains evolved All higher animals have brains
We emphasize these points from Kandel in Bi/CNS 150 Bi/CNS/NB 150: Neuroscience Read Lecture Lecture Friday, October 2, 2015 Development 1: pp 5-10 Introduction Brains evolved All higher animals have brains
Asymmetry and cell fate in the Drosophila embryonic CNS
 Int. J. Dev. Biol. 42: 379-383 (1998) Asymmetry and cell fate in the Drosophila embryonic CNS SAL FUERSTENBERG, JULIE BROADUS and CHRIS Q. DOE Department of Cell and Structural Biology, University of Illinois,
Int. J. Dev. Biol. 42: 379-383 (1998) Asymmetry and cell fate in the Drosophila embryonic CNS SAL FUERSTENBERG, JULIE BROADUS and CHRIS Q. DOE Department of Cell and Structural Biology, University of Illinois,
Peripheral Glia Direct Axon Guidance across the CNS/PNS Transition Zone
 Developmental Biology 238, 47 63 (2001) doi:10.1006/dbio.2001.0411, available online at http://www.idealibrary.com on Peripheral Glia Direct Axon Guidance across the CNS/PNS Transition Zone Katharine J.
Developmental Biology 238, 47 63 (2001) doi:10.1006/dbio.2001.0411, available online at http://www.idealibrary.com on Peripheral Glia Direct Axon Guidance across the CNS/PNS Transition Zone Katharine J.
MCB 141 Midterm I Feb. 19, 2009
 Write your name and student ID# on EVERY PAGE of your exam MCB 141 Midterm I Feb. 19, 2009 Circle the name of your TA Jessica Lyons Alberto Stolfi Question #1 Question #2 Question #3 Question #4 TOTAL
Write your name and student ID# on EVERY PAGE of your exam MCB 141 Midterm I Feb. 19, 2009 Circle the name of your TA Jessica Lyons Alberto Stolfi Question #1 Question #2 Question #3 Question #4 TOTAL
Current address: Whitney Laboratory for Marine Bioscience. University of Florida, St. Augustine, FL 32080
 *Manuscript The eneuro atlas: a modifiable, searchable 3D atlas that individually identifies the majority of interneurons in the Drosophila embryonic CNS and provides tools for functional access Ellie
*Manuscript The eneuro atlas: a modifiable, searchable 3D atlas that individually identifies the majority of interneurons in the Drosophila embryonic CNS and provides tools for functional access Ellie
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles
 Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
Illegitimate translation causes unexpected gene expression from on-target out-of-frame alleles created by CRISPR-Cas9 Shigeru Makino, Ryutaro Fukumura, Yoichi Gondo* Mutagenesis and Genomics Team, RIKEN
Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila
 The Journal of Neuroscience, July 21, 2004 24(29):6573 6577 6573 Brief Communication Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila Bénédicte Franco,
The Journal of Neuroscience, July 21, 2004 24(29):6573 6577 6573 Brief Communication Shaggy, the Homolog of Glycogen Synthase Kinase 3, Controls Neuromuscular Junction Growth in Drosophila Bénédicte Franco,
SUPPLEMENTARY INFORMATION
 Supplementary Discussion Rationale for using maternal ythdf2 -/- mutants as study subject To study the genetic basis of the embryonic developmental delay that we observed, we crossed fish with different
Supplementary Discussion Rationale for using maternal ythdf2 -/- mutants as study subject To study the genetic basis of the embryonic developmental delay that we observed, we crossed fish with different
Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila trachea
 Development 129, 4941-4951 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7966 4941 Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila
Development 129, 4941-4951 (2002) Printed in Great Britain The Company of Biologists Limited 2002 DEV7966 4941 Attractive and repulsive functions of Slit are mediated by different receptors in the Drosophila
Chapter 11. Development: Differentiation and Determination
 KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
KAP Biology Dept Kenyon College Differential gene expression and development Mechanisms of cellular determination Induction Pattern formation Chapter 11. Development: Differentiation and Determination
Astrocytes Play a Key Role in Drosophila Mushroom Body Axon Pruning
 Astrocytes Play a Key Role in Drosophila Mushroom Body Axon Pruning Yaniv Hakim, Shiri P. Yaniv, Oren Schuldiner* Department of Molecular Cell Biology, Weizmann Institute of Sciences, Rehovot, Israel Abstract
Astrocytes Play a Key Role in Drosophila Mushroom Body Axon Pruning Yaniv Hakim, Shiri P. Yaniv, Oren Schuldiner* Department of Molecular Cell Biology, Weizmann Institute of Sciences, Rehovot, Israel Abstract
Neurite initiation. Neurite formation begins with a bud that sprouts from the cell body. One or several neurites can sprout at a time.
 Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Neurite initiation. Neuronal maturation initiation f-actin polarization and maturation tubulin stage 1: "spherical" neuron stage 2: neurons extend several neurites stage 3: one neurite accelerates its
Segment-specific prevention of pioneer neuron apoptosis by cellautonomous, postmitotic Hox gene activity
 First posted online on 10 November 2004 as 10.1242/dev.01521 Access Development the most recent epress version at online http://dev.biologists.org/lookup/doi/10.1242/dev.01521 publication date November
First posted online on 10 November 2004 as 10.1242/dev.01521 Access Development the most recent epress version at online http://dev.biologists.org/lookup/doi/10.1242/dev.01521 publication date November
Geert Geeven. April 14, 2010
 iction of Gene Regulatory Interactions NDNS+ Workshop April 14, 2010 Today s talk - Outline Outline Biological Background Construction of Predictors The main aim of my project is to better understand the
iction of Gene Regulatory Interactions NDNS+ Workshop April 14, 2010 Today s talk - Outline Outline Biological Background Construction of Predictors The main aim of my project is to better understand the
The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also
 c Indian Academy of Sciences RESEARCH ARTICLE The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also MUKULIKA RAY and SUBHASH
c Indian Academy of Sciences RESEARCH ARTICLE The commonly used eye-specific sev-gal4 and GMR-GAL4 drivers in Drosophila melanogaster are expressed in tissues other than eyes also MUKULIKA RAY and SUBHASH
The secreted cell signal Folded Gastrulation regulates glial morphogenesis and axon guidance in Drosophila
 Developmental Biology 308 (2007) 158 168 www.elsevier.com/locate/ydbio The secreted cell signal Folded Gastrulation regulates glial morphogenesis and axon guidance in Drosophila Anuradha Ratnaparkhi 1,
Developmental Biology 308 (2007) 158 168 www.elsevier.com/locate/ydbio The secreted cell signal Folded Gastrulation regulates glial morphogenesis and axon guidance in Drosophila Anuradha Ratnaparkhi 1,
Dap160/intersectin binds and activates apkc to regulate cell polarity and cell cycle progression
 Development epress online publication date 9 July 2008 RESEARCH ARTICLE 2739 Development 135, 2739-2746 (2008) doi:10.1242/dev.024059 Dap160/intersectin binds and activates apkc to regulate cell polarity
Development epress online publication date 9 July 2008 RESEARCH ARTICLE 2739 Development 135, 2739-2746 (2008) doi:10.1242/dev.024059 Dap160/intersectin binds and activates apkc to regulate cell polarity
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY METHODS Mef2 primary screen. RNAi hairpins from the VDRC collection were crossed to Mef2-GAL4 at 27 C. After 2 weeks lethality rate and stage was scored, and if possible 20-30 males containing
SUPPLEMENTARY METHODS Mef2 primary screen. RNAi hairpins from the VDRC collection were crossed to Mef2-GAL4 at 27 C. After 2 weeks lethality rate and stage was scored, and if possible 20-30 males containing
Why Flies? stages of embryogenesis. The Fly in History
 The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
The Fly in History 1859 Darwin 1866 Mendel c. 1890 Driesch, Roux (experimental embryology) 1900 rediscovery of Mendel (birth of genetics) 1910 first mutant (white) (Morgan) 1913 first genetic map (Sturtevant
Solutions to Problem Set 4
 Question 1 Solutions to 7.014 Problem Set 4 Because you have not read much scientific literature, you decide to study the genetics of garden peas. You have two pure breeding pea strains. One that is tall
Question 1 Solutions to 7.014 Problem Set 4 Because you have not read much scientific literature, you decide to study the genetics of garden peas. You have two pure breeding pea strains. One that is tall
