Physics 127c: Statistical Mechanics. Weakly Interacting Fermi Gas. The Electron Gas
|
|
|
- Raymond Merritt
- 5 years ago
- Views:
Transcription
1 Physics 7c: Statistical Mechanics Wealy Interacting Fermi Gas Unlie the Boson case, there is usually no ualitative change in behavior going from the noninteracting to the wealy interacting Fermi gas for repulsive interactions. For example the excitation spectrum for adding a particle of momentum h with > F or for removing a particle with < F creating a hole has the same ualitative form ε µ = hv F F () with µ the chemical potential or Fermi energy and F the Fermi wave vector (which can be shown to unchanged by the interactions Luttinger s theorem). Here v F is called the Fermi velocity v F = ε () =F and defines the spectrum. The value of v F is altered by interactions. The familiar spectrum leads to the usual temperature dependences at low temperature, e.g. specific heat C T, susceptibility χ independent of temperature. Furthermore, since there is no ualitative change of behavior, unlie for the Boson case (where the canonical transformation was necessary) uantitative results can be calculated using perturbation theory. When are the interactions wea? For a short range pair interaction potential this is small potential or low density. On the other hand for a Coulomb interaction, as for the electron gas, interactions are wea compared with the inetic terms at high density The Electron Gas We write the interaction potential as u(r) = e r ; ũ() =4πe, () where e is the electron charge in CGS units, and e = e /4πε with e the electron charge in SI units. An estimate of the interaction energy per particle is V N e, (4) r with r a measure of the interelectron spacing given by 4 πr = N, (5) i.e. a sphere of radius r contains on average one electron. Since F is related to N by the usual counting of states 4. (π) π F = N (6) we have F r = ( ) 9π / (7) 4
2 It is conventional to write r in terms of the Bohr radius a = h /me r = r s a (8) and r s the interelectron spacing in units of a is the dimensionless measure of the density of the electron gas. The estimate of the potential energy becomes The total inetic energy is V N e a r s. (9) E in N = 5 ε F = h F 5 m = e a 5 ( ) 9π / 4 rs e.. () a r s Thus the potential energy is relatively small for small r s, i.e. large density. For typical metals r s 6 (e.g. for sodium r s 4) and so the interactions are not wea. Perturbation Theory Potential Energy The ground state energy correction to lowest order in the interactions is given by the expectation value of the potential energy in the noninteracting ground state. In conventional wavefunction notation we would just write the wavefunction as an antisymmetrized product (Slater determinant) of single particle wavefunctions (functions of coordinate and spin) ψ N (r,...r N ; σ...σ N ) = A,, N χ i (r i ; σ i ), () with A the antisymmetrization operator (giving the Slater determinant form). In chemistry, where we are dealing with electrons in an external potential as in the trapped Bose gases, the best single particle states χ i would be found self consistently by minimizing the resulting energy expression. In a homogeneous fermion gas the χ i remain spin up or down plane wave states, and the wavefunction reduces to the noninteracting one. Thus E E = ũ()a + +,σ a+,σ a,σ a,σ. () In the noninteracting ground state the four particle expectation value factorizes into the product of the nonzero pairwise averages, and as you proved in Homewor a + +,σ a+,σ a,σ a,σ = a + +,σ a,σ a +,σ a,σ a ++,σ a,σ a +,σ a,σ (a) i= = n,σ n,σ δ, n +,σ n,σ δ,+δ σσ (b) where the minus sign for the second term comes because we have changed the order of the middle pair of Fermi operators (remember the anticommutation rules!), and n,σ is the noninteracting ground state occupation number { for <F n,σ =. (4) for > F
3 This gives ( ) E E = ũ() n.σ ũ()n +,σ n,σ. (5),σ,,σ The first term in E. (5) can be written as N ũ()/ and is just the mean potential interaction between all the particles. In this context it is nown as the Hartree term. You might have expected this to be the complete result since we are just using the noninteracting wavefunction, and indeed this would be the case for distinguishable particles. The second term arises from the antisymmetrization of the wavefunction, and is nown as the exchange or Foch term. Together we have Hartree-Foch theory. Radial Distribution Function The Foch term can be understood better by remembering the result for the potential energy from our study of the classical interacting gas. For a pairwise interaction the potential energy just depends on the pair correlation function, conveniently expressed in terms of the radial distribution function g(r) V = N u(r)g(r)d r = N ũ()g( ). (6) The radial distribution function g(r) is proportional to the probability of finding a particle at r given that there is one at the origin. It is normalized so that g(r ) =. Also, remember the convention for Fourier transforms I am using f(r) = f()e i r (7) f() = f(r)e i r (8) where at the end of the calculation we evaluate the wavevector sum as... d... (9) (π) () Comparing with E. () we can introduce the spin dependent radial distribution function g σσ and write g() = g σσ () 4 σσ with g σσ () = a + (N/),σ a+ +,σ a,σ a,σ,, () or Fourier transforming g σσ (R) = e i R a + (N/),σ a+ +,σ a,σ a,σ,. () Using the same factorization procedure gives (writing the last term in terms of, rather than, ) ( )( ) g σσ (R) = 4 n N,σ n,σ δ σσ e i( ) R n,σ n,σ. (),
4 This can be written in terms of the real function φ(r) φ(r) = e i R n σ. (4) N as g σσ = δ σσ φ (R). (5) The function φ is easily evaluated, since n σ is for < F and for > F as F φ(r) = 4π d d(cos θ)e ir cos θ (6) n (π) = F r dy y sin y (7) ( F r) (writing the density n in terms of F and introducing y = F r), or φ(r) = ( F r) [sin( F r) ( F r)cos( F r)]. (8) g F r Figure : Radial distribution function for parallel spin fermions plotted from Es. (5) and (8). These results show that g = g = as might be expected for the noninteracting wave function. However g = g go to zero as r, and there is a correlation hole of radius F because of the antisymmetrization of the wavefunction, the nonlocal ramification of the Pauli exclusion principle, see Fig.. Note that the recovery to g = g = for r is oscillatory and with a power law tail g (r ) cos( F r). (9) r 4 These slowly decaying oscillations, appearing here and in other uantities, and resulting from the sharp discontinuity at the Fermi surface are nown as Friedel oscillations. 4
5 + Figure : Shaded volume gives the integral in the Foch term of the ground state energy. Potential Energy for the Electron Gas For the electron gas with a uniform positive bacground (the jellium model) the first term in E. (5) is cancelled by the interaction with the positive bacground. This leaves the Foch term. There is a lowering of the energy because as we have seen from g lie spins tend to stay apart, reducing the repulsive interaction. To evaluate E. (5)or(??) we need to calculate n +,σ n,σ. () This is easily evaluated geometrically, since the product n + n is unity inside the intersection of the Fermi sea and a Fermi sea displaced by (shaded in Fig. ()) and zero outside. The shaded volume is [ F π sin θd(cos θ) = 4 / F π F + ( ) ] () F F for < F and zero otherwise. Thus [ n +,σ n,σ = 4 (π) π F + ( ) ] ( F F ) () F F [ = N + ( ) ] ( F ). () F F The Foch term to the perturbation of the energy is then E E = ( ) [ F 4π 4πe N + ( ) ] 8π F F = Ne 4 F π dx[ x + x ] Ne F π d. (4) (5) or in terms of r s E E = Ne α π ( ) 9π /. (6) 4 r s 5
6 This gives for the total energy [ E = e..96 ] + (7) a rs r s where the inetic energy is given by summing over the Fermi sphere in the usual way. Evaluating the higher order terms was a major focus of many in the early days of many body physics, using diagrammatic perturbation theory or other approaches. We can also calculate the excitation energy ε,σ the energy reuired to add an additional particle within the same approximation: ε,σ = E n,σ ε +ũ() n,σ ũ( )n,σ (8),σ with inetic, Hartree and Foch terms. (Note in the later the interaction is only with the same spin particles there is no spin sum in this term.). It turns out the for the Coulomb interaction, the last term gives a logarithmic singularity at = F, so that v F = ε / is infinite. This is an unphysical result, and results from the diverging potential at small. In this limit we must tae screening into account an electron repels its neighbors via the Coulomb interaction giving an effective positive screening cloud, so that the inter-electron interaction is reduced. Using the Thomas-Fermi approximation, the same method we used in the calculation of white dwarf stars (see the Appendix below), leads to the replacement 4πe 4πe + TF (9) with the Thomas-Fermi wavevector given by TF = 6πne ε F = 4 π F /a. (4) We can thin of this as replacing the screening the / potential with a dielectric constant ũ eff () = 4πe ε() (4) with the Thomas-Fermi approximation to the dielectric constant In real space this corresponds to a Yuawa potential ε TF () = + TF. (4) u eff (r) = e r e TFr (4) with an exponential decay at large separations. This screened potential leads to a finite Fermi velocity. The corresponding corrections to the ground state energy are in the + in E. (7). Diagrammatic Language The approximation we have done can be expressed (if you lie such things!) in diagrammatic language as shown in Fig. (). The diagrams for the screening of the interaction (the last line of the figure) can be 6
7 = + Σ Self energy Σ = + Hartree Foch Screened interaction = + Π Figure : Diagrams for the Hartree-Foch calculation of the single particle propagator using the screened interaction, with the screening calculated to lowest order. expressed algebraically as u eff () = 4πe + 4πe ()u eff () (44) 4πe u eff () = (45) + 4πe [ ()] where () is given (to lowest order) by the loop or bubble diagram, and corresponds to an approximate evaluation of the polarizability of the electron gas. Actually is freuency dependent ( represents,ω), and the full dielectric constant screening the interaction ε() = 4πe () (46) is also freuency dependent. In the static and long wavelength limit, the expression reduces to the Thomas- Fermi result. More generally we could use the expression for the bubble to give a better approximation to. This gives the Lindhard approximation to the dielectric constant. Using this interaction to evaluate the properties of the electron gas is nown as the Random Phase Approximation or RPA. Appendix: Thomas-Fermi Calculation of Dielectric Constant The Thomas-Fermi approach balances the degeneracy pressure against the forces from the electrostatic potential in a local approximation: at each point the degeneracy pressure is calculated for a gas at the local density as for a homogeneous gas. We used this before in the physics of white dwarf stars in Lecture 7 of Ph7a and trapped-atom Bose systems in Lecture 7 this term. Suppose that there is a net potential V(r) acting on the electrons, coming from some fixed set of charges and the electrons themselves. The chemical potential must be constant, and is the sum of potential and inetic terms V(r) + ε F (n(r)) = const, (47) 7
8 where n is the local electron density, ε F F n/ is the local Fermi energy, and we are considering low temperatures B T ε F. To calculate the dielectric constant we assume a small external potential, and may then linearize E. (47) δv (r) + ε F δn =, (48) n with n,ε F the values without the perturbation. The potential derives from the sum of external charges and eδn from the electrons Euations (48) and (49) are easily solved by Fourier transforming with the Thomas-Fermi wave vector given by δv = 4π(ρ ext eδn). (49) δṽ() = 4π ρ ext + TF, (5) TF = 6πn e ε F. (5) The potential without the screening would be δṽ ext = 4πρ ext / and so we may introduce the dielectric constant δṽ = δṽ ext /ε() with ε() = + TF. (5) This is singular at small due to the efficient screening at long wavelengths. For the interaction between electrons, the external charge is provided by another electron. The effective interaction in the Thomas-Fermi approximation is ũ() = 4πe + TF, u(r) = e r e TFr, (5) a screened potential finite as in Fourier representation, and a Yuawa potential decaying exponentially at large separations in real space. The Thomas-Fermi wavevector is typically comparable to the inverse spacing for the electron gas in metals, although it actually scales as the inverse of the suare root of the spacing TF = 4 π ( F a ) /.56 r / s a. (54) April 5, 4 8
Preface Introduction to the electron liquid
 Table of Preface page xvii 1 Introduction to the electron liquid 1 1.1 A tale of many electrons 1 1.2 Where the electrons roam: physical realizations of the electron liquid 5 1.2.1 Three dimensions 5 1.2.2
Table of Preface page xvii 1 Introduction to the electron liquid 1 1.1 A tale of many electrons 1 1.2 Where the electrons roam: physical realizations of the electron liquid 5 1.2.1 Three dimensions 5 1.2.2
with a proper choice of the potential U(r). Clearly, we should include the potential of the ions, U ion (r):.
 The Hartree Equations So far we have ignored the effects of electron-electron (e-e) interactions by working in the independent electron approximation. In this lecture, we shall discuss how this effect
The Hartree Equations So far we have ignored the effects of electron-electron (e-e) interactions by working in the independent electron approximation. In this lecture, we shall discuss how this effect
Physics 127a: Class Notes
 Physics 127a: Class Notes Lecture 15: Statistical Mechanics of Superfluidity Elementary excitations/quasiparticles In general, it is hard to list the energy eigenstates, needed to calculate the statistical
Physics 127a: Class Notes Lecture 15: Statistical Mechanics of Superfluidity Elementary excitations/quasiparticles In general, it is hard to list the energy eigenstates, needed to calculate the statistical
Interacting Fermi Gases
 Interacting Fermi Gases Mike Hermele (Dated: February 11, 010) Notes on Interacting Fermi Gas for Physics 7450, Spring 010 I. FERMI GAS WITH DELTA-FUNCTION INTERACTION Since it is easier to illustrate
Interacting Fermi Gases Mike Hermele (Dated: February 11, 010) Notes on Interacting Fermi Gas for Physics 7450, Spring 010 I. FERMI GAS WITH DELTA-FUNCTION INTERACTION Since it is easier to illustrate
v(r i r j ) = h(r i )+ 1 N
 Chapter 1 Hartree-Fock Theory 1.1 Formalism For N electrons in an external potential V ext (r), the many-electron Hamiltonian can be written as follows: N H = [ p i i=1 m +V ext(r i )]+ 1 N N v(r i r j
Chapter 1 Hartree-Fock Theory 1.1 Formalism For N electrons in an external potential V ext (r), the many-electron Hamiltonian can be written as follows: N H = [ p i i=1 m +V ext(r i )]+ 1 N N v(r i r j
V. ELECTRON GAS, RANDOM PHASE APPROXIMATION
 V. ELECTRON GAS, RANDOM PHASE APPROXIMATION Marus Holzmann LPMMC, Maison de Magistère, Grenoble, and LPTMC, Jussieu, Paris marus@lptl.jussieu.fr http://www.lptl.jussieu.fr/users/marus/cours.html (Dated:
V. ELECTRON GAS, RANDOM PHASE APPROXIMATION Marus Holzmann LPMMC, Maison de Magistère, Grenoble, and LPTMC, Jussieu, Paris marus@lptl.jussieu.fr http://www.lptl.jussieu.fr/users/marus/cours.html (Dated:
1 GW+DMFT. KH Computational Physics QMC. Dynamical Mean Field Theory + Band Structure Method. Γ[G] = Trlog G Tr(ΣG) + Φ[G] (1)
![1 GW+DMFT. KH Computational Physics QMC. Dynamical Mean Field Theory + Band Structure Method. Γ[G] = Trlog G Tr(ΣG) + Φ[G] (1) 1 GW+DMFT. KH Computational Physics QMC. Dynamical Mean Field Theory + Band Structure Method. Γ[G] = Trlog G Tr(ΣG) + Φ[G] (1)](/thumbs/82/86200337.jpg) Dynamical Mean Field Theory + Band Structure Method 1 GW+DMFT We will express the various types of approximations in language of Luttinger-Ward functionals. The exact Luttinger Ward functional takes the
Dynamical Mean Field Theory + Band Structure Method 1 GW+DMFT We will express the various types of approximations in language of Luttinger-Ward functionals. The exact Luttinger Ward functional takes the
Dept of Mechanical Engineering MIT Nanoengineering group
 1 Dept of Mechanical Engineering MIT Nanoengineering group » To calculate all the properties of a molecule or crystalline system knowing its atomic information: Atomic species Their coordinates The Symmetry
1 Dept of Mechanical Engineering MIT Nanoengineering group » To calculate all the properties of a molecule or crystalline system knowing its atomic information: Atomic species Their coordinates The Symmetry
Physics 221B Spring 2018 Notes 30 The Thomas-Fermi Model
 Copyright c 217 by Robert G. Littlejohn Physics 221B Spring 218 Notes 3 The Thomas-Fermi Model 1. Introduction The Thomas-Fermi model is a relatively crude model of multi-electron atoms that is useful
Copyright c 217 by Robert G. Littlejohn Physics 221B Spring 218 Notes 3 The Thomas-Fermi Model 1. Introduction The Thomas-Fermi model is a relatively crude model of multi-electron atoms that is useful
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
2 Electronic structure theory
 Electronic structure theory. Generalities.. Born-Oppenheimer approximation revisited In Sec..3 (lecture 3) the Born-Oppenheimer approximation was introduced (see also, for instance, [Tannor.]). We are
Electronic structure theory. Generalities.. Born-Oppenheimer approximation revisited In Sec..3 (lecture 3) the Born-Oppenheimer approximation was introduced (see also, for instance, [Tannor.]). We are
Fermi liquid theory Can we apply the free fermion approximation to a real metal? Phys540.nb Strong interaction vs.
 Phys540.nb 7 Fermi liquid theory.. Can we apply the free fermion approximation to a real metal? Can we ignore interactions in a real metal? Experiments says yes (free fermion models work very well), but
Phys540.nb 7 Fermi liquid theory.. Can we apply the free fermion approximation to a real metal? Can we ignore interactions in a real metal? Experiments says yes (free fermion models work very well), but
The general solution of Schrödinger equation in three dimensions (if V does not depend on time) are solutions of time-independent Schrödinger equation
 Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Fermi gas model. Introduction to Nuclear Science. Simon Fraser University Spring NUCS 342 February 2, 2011
 Fermi gas model Introduction to Nuclear Science Simon Fraser University Spring 2011 NUCS 342 February 2, 2011 NUCS 342 (Lecture 9) February 2, 2011 1 / 34 Outline 1 Bosons and fermions NUCS 342 (Lecture
Fermi gas model Introduction to Nuclear Science Simon Fraser University Spring 2011 NUCS 342 February 2, 2011 NUCS 342 (Lecture 9) February 2, 2011 1 / 34 Outline 1 Bosons and fermions NUCS 342 (Lecture
Quantum Theory of Many-Particle Systems, Phys. 540
 Quantum Theory of Many-Particle Systems, Phys. 540 Questions about organization Second quantization Questions about last class? Comments? Similar strategy N-particles Consider Two-body operators in Fock
Quantum Theory of Many-Particle Systems, Phys. 540 Questions about organization Second quantization Questions about last class? Comments? Similar strategy N-particles Consider Two-body operators in Fock
Lecture 5. Hartree-Fock Theory. WS2010/11: Introduction to Nuclear and Particle Physics
 Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
Lecture 5 Hartree-Fock Theory WS2010/11: Introduction to Nuclear and Particle Physics Particle-number representation: General formalism The simplest starting point for a many-body state is a system of
e L 2m e the Bohr magneton
 e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
e L μl = L = μb 2m with : μ B e e 2m e the Bohr magneton Classical interation of magnetic moment and B field: (Young and Freedman, Ch. 27) E = potential energy = μ i B = μbcosθ τ = torque = μ B, perpendicular
Exchange-Correlation Functional
 Exchange-Correlation Functional Aiichiro Nakano Collaboratory for Advanced Computing & Simulations Depts. of Computer Science, Physics & Astronomy, Chemical Engineering & Materials Science, and Biological
Exchange-Correlation Functional Aiichiro Nakano Collaboratory for Advanced Computing & Simulations Depts. of Computer Science, Physics & Astronomy, Chemical Engineering & Materials Science, and Biological
Chapter 12: Phenomena
 Chapter 12: Phenomena K Fe Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected
Chapter 12: Phenomena K Fe Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected
1. Thomas-Fermi method
 1. Thomas-Fermi method We consider a system of N electrons in a stationary state, that would obey the stationary Schrödinger equation: h i m + 1 v(r i,r j ) Ψ(r 1,...,r N ) = E i Ψ(r 1,...,r N ). (1.1)
1. Thomas-Fermi method We consider a system of N electrons in a stationary state, that would obey the stationary Schrödinger equation: h i m + 1 v(r i,r j ) Ψ(r 1,...,r N ) = E i Ψ(r 1,...,r N ). (1.1)
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation
 Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
Chapter 9: Multi- Electron Atoms Ground States and X- ray Excitation Up to now we have considered one-electron atoms. Almost all atoms are multiple-electron atoms and their description is more complicated
Electrons in a periodic potential
 Chapter 3 Electrons in a periodic potential 3.1 Bloch s theorem. We consider in this chapter electrons under the influence of a static, periodic potential V (x), i.e. such that it fulfills V (x) = V (x
Chapter 3 Electrons in a periodic potential 3.1 Bloch s theorem. We consider in this chapter electrons under the influence of a static, periodic potential V (x), i.e. such that it fulfills V (x) = V (x
Chemistry 120A 2nd Midterm. 1. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (1-electron):
 April 6th, 24 Chemistry 2A 2nd Midterm. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (-electron): E n = m e Z 2 e 4 /2 2 n 2 = E Z 2 /n 2, n =, 2, 3,... where Ze is
April 6th, 24 Chemistry 2A 2nd Midterm. (36 pts) For this question, recall the energy levels of the Hydrogenic Hamiltonian (-electron): E n = m e Z 2 e 4 /2 2 n 2 = E Z 2 /n 2, n =, 2, 3,... where Ze is
only two orbitals, and therefore only two combinations to worry about, but things get
 131 Lecture 1 It is fairly easy to write down an antisymmetric wavefunction for helium since there are only two orbitals, and therefore only two combinations to worry about, but things get complicated
131 Lecture 1 It is fairly easy to write down an antisymmetric wavefunction for helium since there are only two orbitals, and therefore only two combinations to worry about, but things get complicated
Electromagnetic Radiation. Chapter 12: Phenomena. Chapter 12: Quantum Mechanics and Atomic Theory. Quantum Theory. Electromagnetic Radiation
 Chapter 12: Phenomena Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected and
Chapter 12: Phenomena Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected and
We can instead solve the problem algebraically by introducing up and down ladder operators b + and b
 Physics 17c: Statistical Mechanics Second Quantization Ladder Operators in the SHO It is useful to first review the use of ladder operators in the simple harmonic oscillator. Here I present the bare bones
Physics 17c: Statistical Mechanics Second Quantization Ladder Operators in the SHO It is useful to first review the use of ladder operators in the simple harmonic oscillator. Here I present the bare bones
Symmetry properties, density profiles and momentum distribution of multicomponent mixtures of strongly interacting 1D Fermi gases
 Symmetry properties, density profiles and momentum distribution of multicomponent mixtures of strongly interacting 1D Fermi gases Anna Minguzzi LPMMC Université Grenoble Alpes and CNRS Multicomponent 1D
Symmetry properties, density profiles and momentum distribution of multicomponent mixtures of strongly interacting 1D Fermi gases Anna Minguzzi LPMMC Université Grenoble Alpes and CNRS Multicomponent 1D
lim = F F = F x x + F y y + F z
 Physics 361 Summary of Results from Lecture Physics 361 Derivatives of Scalar and Vector Fields The gradient of a scalar field f( r) is given by g = f. coordinates f g = ê x x + ê f y y + ê f z z Expressed
Physics 361 Summary of Results from Lecture Physics 361 Derivatives of Scalar and Vector Fields The gradient of a scalar field f( r) is given by g = f. coordinates f g = ê x x + ê f y y + ê f z z Expressed
Strongly correlated systems in atomic and condensed matter physics. Lecture notes for Physics 284 by Eugene Demler Harvard University
 Strongly correlated systems in atomic and condensed matter physics Lecture notes for Physics 284 by Eugene Demler Harvard University January 25, 2011 2 Chapter 12 Collective modes in interacting Fermi
Strongly correlated systems in atomic and condensed matter physics Lecture notes for Physics 284 by Eugene Demler Harvard University January 25, 2011 2 Chapter 12 Collective modes in interacting Fermi
8.3. Feynman diagram. Phys620.nb multi-particle Green s functions the equations of motion of the Green s functions
 Phys60.nb 0 8.3. Feynman diagram 8.3.. multi-particle Green s functions Single particle Green s function: G, ' ' (8.7) Two-particle Green s function: G, ; ', ' ' ' (8.73) three-particle Green s function:
Phys60.nb 0 8.3. Feynman diagram 8.3.. multi-particle Green s functions Single particle Green s function: G, ' ' (8.7) Two-particle Green s function: G, ; ', ' ' ' (8.73) three-particle Green s function:
Probability and Normalization
 Probability and Normalization Although we don t know exactly where the particle might be inside the box, we know that it has to be in the box. This means that, ψ ( x) dx = 1 (normalization condition) L
Probability and Normalization Although we don t know exactly where the particle might be inside the box, we know that it has to be in the box. This means that, ψ ( x) dx = 1 (normalization condition) L
Workshop on: ATOMIC STRUCTURE AND TRANSITIONS: THEORY IN USING SUPERSTRUCTURE PROGRAM
 Workshop on: ATOMIC STRUCTURE AND TRANSITIONS: THEORY IN USING SUPERSTRUCTURE PROGRAM PROF. SULTANA N. NAHAR Astronomy, Ohio State U, Columbus, Ohio, USA Email: nahar.1@osu.edu http://www.astronomy.ohio-state.edu/
Workshop on: ATOMIC STRUCTURE AND TRANSITIONS: THEORY IN USING SUPERSTRUCTURE PROGRAM PROF. SULTANA N. NAHAR Astronomy, Ohio State U, Columbus, Ohio, USA Email: nahar.1@osu.edu http://www.astronomy.ohio-state.edu/
Brief review of Quantum Mechanics (QM)
 Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
Brief review of Quantum Mechanics (QM) Note: This is a collection of several formulae and facts that we will use throughout the course. It is by no means a complete discussion of QM, nor will I attempt
Condensed matter theory Lecture notes and problem sets 2012/2013
 Condensed matter theory Lecture notes and problem sets 2012/2013 Dmitri Ivanov Recommended books and lecture notes: [AM] N. W. Ashcroft and N. D. Mermin, Solid State Physics. [Mar] M. P. Marder, Condensed
Condensed matter theory Lecture notes and problem sets 2012/2013 Dmitri Ivanov Recommended books and lecture notes: [AM] N. W. Ashcroft and N. D. Mermin, Solid State Physics. [Mar] M. P. Marder, Condensed
f lmn x m y n z l m n (1) 3/πz/r and Y 1 ±1 = 3/2π(x±iy)/r, and also gives a formula for Y
 Problem Set 6: Group representation theory and neutron stars Graduate Quantum I Physics 6572 James Sethna Due Monday Nov. 12 Last correction at November 14, 2012, 2:32 pm Reading Sakurai and Napolitano,
Problem Set 6: Group representation theory and neutron stars Graduate Quantum I Physics 6572 James Sethna Due Monday Nov. 12 Last correction at November 14, 2012, 2:32 pm Reading Sakurai and Napolitano,
We already came across a form of indistinguishably in the canonical partition function: V N Q =
 Bosons en fermions Indistinguishability We already came across a form of indistinguishably in the canonical partition function: for distinguishable particles Q = Λ 3N βe p r, r 2,..., r N ))dτ dτ 2...
Bosons en fermions Indistinguishability We already came across a form of indistinguishably in the canonical partition function: for distinguishable particles Q = Λ 3N βe p r, r 2,..., r N ))dτ dτ 2...
Theoretische Physik 2: Elektrodynamik (Prof. A-S. Smith) Home assignment 11
 WiSe 22..23 Prof. Dr. A-S. Smith Dipl.-Phys. Matthias Saba am Lehrstuhl für Theoretische Physik I Department für Physik Friedrich-Alexander-Universität Erlangen-Nürnberg Problem. Theoretische Physik 2:
WiSe 22..23 Prof. Dr. A-S. Smith Dipl.-Phys. Matthias Saba am Lehrstuhl für Theoretische Physik I Department für Physik Friedrich-Alexander-Universität Erlangen-Nürnberg Problem. Theoretische Physik 2:
Lecture 3: Propagators
 Lecture 3: Propagators 0 Introduction to current particle physics 1 The Yukawa potential and transition amplitudes 2 Scattering processes and phase space 3 Feynman diagrams and QED 4 The weak interaction
Lecture 3: Propagators 0 Introduction to current particle physics 1 The Yukawa potential and transition amplitudes 2 Scattering processes and phase space 3 Feynman diagrams and QED 4 The weak interaction
MODEL WITH SPIN; CHARGE AND SPIN EXCITATIONS 57
 56 BOSONIZATION Note that there seems to be some arbitrariness in the above expressions in terms of the bosonic fields since by anticommuting two fermionic fields one can introduce a minus sine and thus
56 BOSONIZATION Note that there seems to be some arbitrariness in the above expressions in terms of the bosonic fields since by anticommuting two fermionic fields one can introduce a minus sine and thus
E = 2 (E 1)+ 2 (4E 1) +1 (9E 1) =19E 1
 Quantum Mechanics and Atomic Physics Lecture 22: Multi-electron Atoms http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last Time Multi-electron atoms and Pauli s exclusion principle Electrons
Quantum Mechanics and Atomic Physics Lecture 22: Multi-electron Atoms http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Last Time Multi-electron atoms and Pauli s exclusion principle Electrons
VII.B Canonical Formulation
 VII.B Canonical Formulation Using the states constructed in the previous section, we can calculate the canonical density matrix for non-interacting identical particles. In the coordinate representation
VII.B Canonical Formulation Using the states constructed in the previous section, we can calculate the canonical density matrix for non-interacting identical particles. In the coordinate representation
1.6. Quantum mechanical description of the hydrogen atom
 29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
S j H o = gµ o H o. j=1
 LECTURE 17 Ferromagnetism (Refs.: Sections 10.6-10.7 of Reif; Book by J. S. Smart, Effective Field Theories of Magnetism) Consider a solid consisting of N identical atoms arranged in a regular lattice.
LECTURE 17 Ferromagnetism (Refs.: Sections 10.6-10.7 of Reif; Book by J. S. Smart, Effective Field Theories of Magnetism) Consider a solid consisting of N identical atoms arranged in a regular lattice.
The free electron gas: Sommerfeld model
 The free electron gas: Sommerfeld model Valence electrons: delocalized gas of free electrons (selectrons of alkalies) which is not perturbed by the ion cores (ionic radius of Na + equals 0.97 Å, internuclear
The free electron gas: Sommerfeld model Valence electrons: delocalized gas of free electrons (selectrons of alkalies) which is not perturbed by the ion cores (ionic radius of Na + equals 0.97 Å, internuclear
Conductors and Insulators
 Conductors and Insulators Lecture 11: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay Self Energy of a Charge Distribution : In Lecture 1 we briefly discussed what we called
Conductors and Insulators Lecture 11: Electromagnetic Theory Professor D. K. Ghosh, Physics Department, I.I.T., Bombay Self Energy of a Charge Distribution : In Lecture 1 we briefly discussed what we called
221B Lecture Notes Quantum Field Theory II (Fermi Systems)
 1B Lecture Notes Quantum Field Theory II (Fermi Systems) 1 Statistical Mechanics of Fermions 1.1 Partition Function In the case of fermions, we had learnt that the field operator satisfies the anticommutation
1B Lecture Notes Quantum Field Theory II (Fermi Systems) 1 Statistical Mechanics of Fermions 1.1 Partition Function In the case of fermions, we had learnt that the field operator satisfies the anticommutation
The non-interacting Bose gas
 Chapter The non-interacting Bose gas Learning goals What is a Bose-Einstein condensate and why does it form? What determines the critical temperature and the condensate fraction? What changes for trapped
Chapter The non-interacting Bose gas Learning goals What is a Bose-Einstein condensate and why does it form? What determines the critical temperature and the condensate fraction? What changes for trapped
Quantum mechanics of many-fermion systems
 Quantum mechanics of many-fermion systems Kouichi Hagino Tohoku University, Sendai, Japan 1. Identical particles: Fermions and Bosons 2. Simple examples: systems with two identical particles 3. Pauli principle
Quantum mechanics of many-fermion systems Kouichi Hagino Tohoku University, Sendai, Japan 1. Identical particles: Fermions and Bosons 2. Simple examples: systems with two identical particles 3. Pauli principle
Landau s Fermi Liquid Theory
 Thors Hans Hansson Stockholm University Outline 1 Fermi Liquids Why, What, and How? Why Fermi liquids? What is a Fermi liquids? Fermi Liquids How? 2 Landau s Phenomenological Approach The free Fermi gas
Thors Hans Hansson Stockholm University Outline 1 Fermi Liquids Why, What, and How? Why Fermi liquids? What is a Fermi liquids? Fermi Liquids How? 2 Landau s Phenomenological Approach The free Fermi gas
Advanced Quantum Mechanics
 Advanced Quantum Mechanics University of York Lecturer: Rex Godby Notes by Victor Naden Robinson Lecture 1: TDSE Lecture 2: TDSE Lecture 3: FMG Lecture 4: FMG Lecture 5: Ehrenfest s Theorem and the Classical
Advanced Quantum Mechanics University of York Lecturer: Rex Godby Notes by Victor Naden Robinson Lecture 1: TDSE Lecture 2: TDSE Lecture 3: FMG Lecture 4: FMG Lecture 5: Ehrenfest s Theorem and the Classical
1. Electricity and Magnetism (Fall 1995, Part 1) A metal sphere has a radius R and a charge Q.
 1. Electricity and Magnetism (Fall 1995, Part 1) A metal sphere has a radius R and a charge Q. (a) Compute the electric part of the Maxwell stress tensor T ij (r) = 1 {E i E j 12 } 4π E2 δ ij both inside
1. Electricity and Magnetism (Fall 1995, Part 1) A metal sphere has a radius R and a charge Q. (a) Compute the electric part of the Maxwell stress tensor T ij (r) = 1 {E i E j 12 } 4π E2 δ ij both inside
Multielectron Atoms and Periodic Table
 GRE Question Multielectron Atoms and Periodic Table Helium Atom 2 2m e ( 2 1 + 2 2) + 2ke 2 2ke 2 + ke2 r 1 r 2 r 2 r 1 Electron-electron repulsion term destroys spherical symmetry. No analytic solution
GRE Question Multielectron Atoms and Periodic Table Helium Atom 2 2m e ( 2 1 + 2 2) + 2ke 2 2ke 2 + ke2 r 1 r 2 r 2 r 1 Electron-electron repulsion term destroys spherical symmetry. No analytic solution
Summary Sec. 1.6: The dielectric function of jellium in the RPA
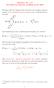 Summary Sec 6: The dielectric function of jellium in the RPA Starting with the diagram that describes the simplest approximation of a polarisation insertion into a bare Coulomb interaction line and translating
Summary Sec 6: The dielectric function of jellium in the RPA Starting with the diagram that describes the simplest approximation of a polarisation insertion into a bare Coulomb interaction line and translating
1 Density functional theory (DFT)
 1 Density functional theory (DFT) 1.1 Introduction Density functional theory is an alternative to ab initio methods for solving the nonrelativistic, time-independent Schrödinger equation H Φ = E Φ. The
1 Density functional theory (DFT) 1.1 Introduction Density functional theory is an alternative to ab initio methods for solving the nonrelativistic, time-independent Schrödinger equation H Φ = E Φ. The
Section 10: Many Particle Quantum Mechanics Solutions
 Physics 143a: Quantum Mechanics I Section 10: Many Particle Quantum Mechanics Solutions Spring 015, Harvard Here is a summary of the most important points from this week (with a few of my own tidbits),
Physics 143a: Quantum Mechanics I Section 10: Many Particle Quantum Mechanics Solutions Spring 015, Harvard Here is a summary of the most important points from this week (with a few of my own tidbits),
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
14. Structure of Nuclei
 14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
1 Superfluidity and Bose Einstein Condensate
 Physics 223b Lecture 4 Caltech, 04/11/18 1 Superfluidity and Bose Einstein Condensate 1.6 Superfluid phase: topological defect Besides such smooth gapless excitations, superfluid can also support a very
Physics 223b Lecture 4 Caltech, 04/11/18 1 Superfluidity and Bose Einstein Condensate 1.6 Superfluid phase: topological defect Besides such smooth gapless excitations, superfluid can also support a very
Noncollinear spins in QMC: spiral Spin Density Waves in the HEG
 Noncollinear spins in QMC: spiral Spin Density Waves in the HEG Zoltán Radnai and Richard J. Needs Workshop at The Towler Institute July 2006 Overview What are noncollinear spin systems and why are they
Noncollinear spins in QMC: spiral Spin Density Waves in the HEG Zoltán Radnai and Richard J. Needs Workshop at The Towler Institute July 2006 Overview What are noncollinear spin systems and why are they
Lecture 8: Radial Distribution Function, Electron Spin, Helium Atom
 Lecture 8: Radial Distribution Function, Electron Spin, Helium Atom Radial Distribution Function The interpretation of the square of the wavefunction is the probability density at r, θ, φ. This function
Lecture 8: Radial Distribution Function, Electron Spin, Helium Atom Radial Distribution Function The interpretation of the square of the wavefunction is the probability density at r, θ, φ. This function
Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015)
 Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015) Interaction of x-ray with matter: - Photoelectric absorption - Elastic (coherent) scattering (Thomson Scattering) - Inelastic (incoherent) scattering
Notes on x-ray scattering - M. Le Tacon, B. Keimer (06/2015) Interaction of x-ray with matter: - Photoelectric absorption - Elastic (coherent) scattering (Thomson Scattering) - Inelastic (incoherent) scattering
Quantum Theory of Matter
 Quantum Theory of Matter Revision Lecture Derek Lee Imperial College London May 2006 Outline 1 Exam and Revision 2 Quantum Theory of Matter Microscopic theory 3 Summary Outline 1 Exam and Revision 2 Quantum
Quantum Theory of Matter Revision Lecture Derek Lee Imperial College London May 2006 Outline 1 Exam and Revision 2 Quantum Theory of Matter Microscopic theory 3 Summary Outline 1 Exam and Revision 2 Quantum
Chapter 10 Dyson s equation, RPA and Ladder Approximations
 Chapter 1 Dyson s euation RPA and Ladder Approximations Dyson s euation low-high density fermion gases Summation tris to deal with divergent series. Conept of renormalization i.e. dressed partile is bare
Chapter 1 Dyson s euation RPA and Ladder Approximations Dyson s euation low-high density fermion gases Summation tris to deal with divergent series. Conept of renormalization i.e. dressed partile is bare
LS coupling. 2 2 n + H s o + H h f + H B. (1) 2m
 LS coupling 1 The big picture We start from the Hamiltonian of an atomic system: H = [ ] 2 2 n Ze2 1 + 1 e 2 1 + H s o + H h f + H B. (1) 2m n e 4πɛ 0 r n 2 4πɛ 0 r nm n,m Here n runs pver the electrons,
LS coupling 1 The big picture We start from the Hamiltonian of an atomic system: H = [ ] 2 2 n Ze2 1 + 1 e 2 1 + H s o + H h f + H B. (1) 2m n e 4πɛ 0 r n 2 4πɛ 0 r nm n,m Here n runs pver the electrons,
Chapter 2 Electron Phonon Interaction
 Chapter Electron Phonon Interaction The electron phonon interaction is important not only in creating the phonon scattering of the electrons but also in the formation of Cooper pairs. This interaction
Chapter Electron Phonon Interaction The electron phonon interaction is important not only in creating the phonon scattering of the electrons but also in the formation of Cooper pairs. This interaction
Atoms 09 update-- start with single electron: H-atom
 Atoms 09 update-- start with single electron: H-atom VII 33 x z φ θ e -1 y 3-D problem - free move in x, y, z - handy to change systems: Cartesian Spherical Coordinate (x, y, z) (r, θ, φ) Reason: V(r)
Atoms 09 update-- start with single electron: H-atom VII 33 x z φ θ e -1 y 3-D problem - free move in x, y, z - handy to change systems: Cartesian Spherical Coordinate (x, y, z) (r, θ, φ) Reason: V(r)
Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures
 Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures Jeong Won Kang Department of Chemical Engineering Korea University Subjects Three Basic Types of Motions
Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures Jeong Won Kang Department of Chemical Engineering Korea University Subjects Three Basic Types of Motions
Physics 342 Lecture 22. The Hydrogen Atom. Lecture 22. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 22 The Hydrogen Atom Lecture 22 Physics 342 Quantum Mechanics I Friday, March 28th, 2008 We now begin our discussion of the Hydrogen atom. Operationally, this is just another choice
Physics 342 Lecture 22 The Hydrogen Atom Lecture 22 Physics 342 Quantum Mechanics I Friday, March 28th, 2008 We now begin our discussion of the Hydrogen atom. Operationally, this is just another choice
Chapter 19 - Second Quantization. 1. Identical Particles Revisited
 Chapter 19 - Second Quantization 1. Identical Particles Revisited When dealing with a system that contains only a few identical particles, we were easily able to explicitly construct appropriately symmetrized
Chapter 19 - Second Quantization 1. Identical Particles Revisited When dealing with a system that contains only a few identical particles, we were easily able to explicitly construct appropriately symmetrized
Preliminary Quantum Questions
 Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
INTRODUCTION À LA PHYSIQUE MÉSOSCOPIQUE: ÉLECTRONS ET PHOTONS INTRODUCTION TO MESOSCOPIC PHYSICS: ELECTRONS AND PHOTONS
 Chaire de Physique Mésoscopique Michel Devoret Année 2007, Cours des 7 et 14 juin INTRODUCTION À LA PHYSIQUE MÉSOSCOPIQUE: ÉLECTRONS ET PHOTONS INTRODUCTION TO MESOSCOPIC PHYSICS: ELECTRONS AND PHOTONS
Chaire de Physique Mésoscopique Michel Devoret Année 2007, Cours des 7 et 14 juin INTRODUCTION À LA PHYSIQUE MÉSOSCOPIQUE: ÉLECTRONS ET PHOTONS INTRODUCTION TO MESOSCOPIC PHYSICS: ELECTRONS AND PHOTONS
Quantum Physics II (8.05) Fall 2002 Outline
 Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Chapter 9. Atomic structure and atomic spectra
 Chapter 9. Atomic structure and atomic spectra -The structure and spectra of hydrogenic atom -The structures of many e - atom -The spectra of complex atoms The structure and spectra of hydrogenic atom
Chapter 9. Atomic structure and atomic spectra -The structure and spectra of hydrogenic atom -The structures of many e - atom -The spectra of complex atoms The structure and spectra of hydrogenic atom
Lecture 4 Recap: normal metals and the clectron-phonon interaction *
 Phys. 598SC Fall 2015 Prof. A. J. Leggett Lecture 4 Recap: normal metals and the clectron-phonon interaction * 1. Normal metals: Sommerfeld-Bloch picture 2. Screening 3. Fermi liquid theory 4. Electron-phonon
Phys. 598SC Fall 2015 Prof. A. J. Leggett Lecture 4 Recap: normal metals and the clectron-phonon interaction * 1. Normal metals: Sommerfeld-Bloch picture 2. Screening 3. Fermi liquid theory 4. Electron-phonon
Introduction to Cold Atoms and Bose-Einstein Condensation. Randy Hulet
 Introduction to Cold Atoms and Bose-Einstein Condensation Randy Hulet Outline Introduction to methods and concepts of cold atom physics Interactions Feshbach resonances Quantum Gases Quantum regime nλ
Introduction to Cold Atoms and Bose-Einstein Condensation Randy Hulet Outline Introduction to methods and concepts of cold atom physics Interactions Feshbach resonances Quantum Gases Quantum regime nλ
Development of atomic theory
 Development of atomic theory The chapter presents the fundamentals needed to explain and atomic & molecular structures in qualitative or semiquantitative terms. Li B B C N O F Ne Sc Ti V Cr Mn Fe Co Ni
Development of atomic theory The chapter presents the fundamentals needed to explain and atomic & molecular structures in qualitative or semiquantitative terms. Li B B C N O F Ne Sc Ti V Cr Mn Fe Co Ni
Journal of Theoretical Physics
 1 Journal of Theoretical Physics Founded and Edited by M. Apostol 53 (2000) ISSN 1453-4428 Ionization potential for metallic clusters L. C. Cune and M. Apostol Department of Theoretical Physics, Institute
1 Journal of Theoretical Physics Founded and Edited by M. Apostol 53 (2000) ISSN 1453-4428 Ionization potential for metallic clusters L. C. Cune and M. Apostol Department of Theoretical Physics, Institute
The Overhauser Instability
 The Overhauser Instability Zoltán Radnai and Richard Needs TCM Group ESDG Talk 14th February 2007 Typeset by FoilTEX Introduction Hartree-Fock theory and Homogeneous Electron Gas Noncollinear spins and
The Overhauser Instability Zoltán Radnai and Richard Needs TCM Group ESDG Talk 14th February 2007 Typeset by FoilTEX Introduction Hartree-Fock theory and Homogeneous Electron Gas Noncollinear spins and
ECE440 Nanoelectronics. Lecture 07 Atomic Orbitals
 ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
ECE44 Nanoelectronics Lecture 7 Atomic Orbitals Atoms and atomic orbitals It is instructive to compare the simple model of a spherically symmetrical potential for r R V ( r) for r R and the simplest hydrogen
Quasiparticle properties and the dynamics of high-density nuclear matter
 Quasiparticle properties and the dynamics of high-density nuclear matter Kazuhiro TANAKA Radiation Laboratory The Institute of Physical and Chemical Research (RIKEN) Hirosawa 2-1, Wako-shi, Saitama, 351-01
Quasiparticle properties and the dynamics of high-density nuclear matter Kazuhiro TANAKA Radiation Laboratory The Institute of Physical and Chemical Research (RIKEN) Hirosawa 2-1, Wako-shi, Saitama, 351-01
CHAPTER 9 Statistical Physics
 CHAPTER 9 Statistical Physics 9.1 Historical Overview 9.2 Maxwell Velocity Distribution 9.3 Equipartition Theorem 9.4 Maxwell Speed Distribution 9.5 Classical and Quantum Statistics 9.6 Fermi-Dirac Statistics
CHAPTER 9 Statistical Physics 9.1 Historical Overview 9.2 Maxwell Velocity Distribution 9.3 Equipartition Theorem 9.4 Maxwell Speed Distribution 9.5 Classical and Quantum Statistics 9.6 Fermi-Dirac Statistics
Roton Mode in Dipolar Bose-Einstein Condensates
 Roton Mode in Dipolar Bose-Einstein Condensates Sandeep Indian Institute of Science Department of Physics, Bangalore March 14, 2013 BECs vs Dipolar Bose-Einstein Condensates Although quantum gases are
Roton Mode in Dipolar Bose-Einstein Condensates Sandeep Indian Institute of Science Department of Physics, Bangalore March 14, 2013 BECs vs Dipolar Bose-Einstein Condensates Although quantum gases are
Supporting Information
 Supporting Information A: Calculation of radial distribution functions To get an effective propagator in one dimension, we first transform 1) into spherical coordinates: x a = ρ sin θ cos φ, y = ρ sin
Supporting Information A: Calculation of radial distribution functions To get an effective propagator in one dimension, we first transform 1) into spherical coordinates: x a = ρ sin θ cos φ, y = ρ sin
Quantum statistics: properties of the Fermi-Dirac distribution.
 Statistical Mechanics Phys54 Fall 26 Lecture #11 Anthony J. Leggett Department of Physics, UIUC Quantum statistics: properties of the Fermi-Dirac distribution. In the last lecture we discussed the properties
Statistical Mechanics Phys54 Fall 26 Lecture #11 Anthony J. Leggett Department of Physics, UIUC Quantum statistics: properties of the Fermi-Dirac distribution. In the last lecture we discussed the properties
Advanced Solid State Theory SS Roser Valentí and Harald Jeschke Institut für Theoretische Physik, Goethe-Universität Frankfurt
 Advanced Solid State Theory SS 2010 Roser Valentí and Harald Jeschke Institut für Theoretische Physik, Goethe-Universität Frankfurt i 0. Literatur R. M. Martin, Electronic Structure: Basic Theory and
Advanced Solid State Theory SS 2010 Roser Valentí and Harald Jeschke Institut für Theoretische Physik, Goethe-Universität Frankfurt i 0. Literatur R. M. Martin, Electronic Structure: Basic Theory and
Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization"
 Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization" Outline 1 Bosons and Fermions 2 N-particle wave functions ("first quantization" 3 The method of quantized
Next topic: Quantum Field Theories for Quantum Many-Particle Systems; or "Second Quantization" Outline 1 Bosons and Fermions 2 N-particle wave functions ("first quantization" 3 The method of quantized
Lecture 3: Quantum Satis*
 Lecture 3: Quantum Satis* Last remarks about many-electron quantum mechanics. Everything re-quantized! * As much as needed, enough. Electron correlation Pauli principle Fermi correlation Correlation energy
Lecture 3: Quantum Satis* Last remarks about many-electron quantum mechanics. Everything re-quantized! * As much as needed, enough. Electron correlation Pauli principle Fermi correlation Correlation energy
Orbital-free density functional theory: Kinetic potentials and ab initio local pseudopotentials
 Orbital-free density functional theory: Kinetic potentials and ab initio local pseudopotentials Jeng-Da Chai 1, * and John D. Weeks 1,2 1 Institute for Physical Science and Technology, University of Maryland,
Orbital-free density functional theory: Kinetic potentials and ab initio local pseudopotentials Jeng-Da Chai 1, * and John D. Weeks 1,2 1 Institute for Physical Science and Technology, University of Maryland,
1 Schroenger s Equation for the Hydrogen Atom
 Schroenger s Equation for the Hydrogen Atom Here is the Schroedinger equation in D in spherical polar coordinates. Note that the definitions of θ and φ are the exact reverse of what they are in mathematics.
Schroenger s Equation for the Hydrogen Atom Here is the Schroedinger equation in D in spherical polar coordinates. Note that the definitions of θ and φ are the exact reverse of what they are in mathematics.
Approximation Methods in QM
 Chapter 3 Approximation Methods in QM Contents 3.1 Time independent PT (nondegenerate)............... 5 3. Degenerate perturbation theory (PT)................. 59 3.3 Time dependent PT and Fermi s golden
Chapter 3 Approximation Methods in QM Contents 3.1 Time independent PT (nondegenerate)............... 5 3. Degenerate perturbation theory (PT)................. 59 3.3 Time dependent PT and Fermi s golden
PHY4604 Introduction to Quantum Mechanics Fall 2004 Final Exam SOLUTIONS December 17, 2004, 7:30 a.m.- 9:30 a.m.
 PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
Neighbor Tables Long-Range Potentials
 Neighbor Tables Long-Range Potentials Today we learn how we can handle long range potentials. Neighbor tables Long-range potential Ewald sums MSE485/PHY466/CSE485 1 Periodic distances Minimum Image Convention:
Neighbor Tables Long-Range Potentials Today we learn how we can handle long range potentials. Neighbor tables Long-range potential Ewald sums MSE485/PHY466/CSE485 1 Periodic distances Minimum Image Convention:
 Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
Math Questions for the 2011 PhD Qualifier Exam 1. Evaluate the following definite integral 3" 4 where! ( x) is the Dirac! - function. # " 4 [ ( )] dx x 2! cos x 2. Consider the differential equation dx
Supplementary Figure 2 Photoluminescence in 1L- (black line) and 7L-MoS 2 (red line) of the Figure 1B with illuminated wavelength of 543 nm.
 PL (normalized) Intensity (arb. u.) 1 1 8 7L-MoS 1L-MoS 6 4 37 38 39 4 41 4 Raman shift (cm -1 ) Supplementary Figure 1 Raman spectra of the Figure 1B at the 1L-MoS area (black line) and 7L-MoS area (red
PL (normalized) Intensity (arb. u.) 1 1 8 7L-MoS 1L-MoS 6 4 37 38 39 4 41 4 Raman shift (cm -1 ) Supplementary Figure 1 Raman spectra of the Figure 1B at the 1L-MoS area (black line) and 7L-MoS area (red
Attempts at relativistic QM
 Attempts at relativistic QM based on S-1 A proper description of particle physics should incorporate both quantum mechanics and special relativity. However historically combining quantum mechanics and
Attempts at relativistic QM based on S-1 A proper description of particle physics should incorporate both quantum mechanics and special relativity. However historically combining quantum mechanics and
BEC in one dimension
 BEC in one dimension Tilmann John 11. Juni 2013 Outline 1 one-dimensional BEC 2 theoretical description Tonks-Girardeau gas Interaction exact solution (Lieb and Liniger) 3 experimental realization 4 conclusion
BEC in one dimension Tilmann John 11. Juni 2013 Outline 1 one-dimensional BEC 2 theoretical description Tonks-Girardeau gas Interaction exact solution (Lieb and Liniger) 3 experimental realization 4 conclusion
Origin of the first Hund rule in He-like atoms and 2-electron quantum dots
 in He-like atoms and 2-electron quantum dots T Sako 1, A Ichimura 2, J Paldus 3 and GHF Diercksen 4 1 Nihon University, College of Science and Technology, Funabashi, JAPAN 2 Institute of Space and Astronautical
in He-like atoms and 2-electron quantum dots T Sako 1, A Ichimura 2, J Paldus 3 and GHF Diercksen 4 1 Nihon University, College of Science and Technology, Funabashi, JAPAN 2 Institute of Space and Astronautical
Luttinger Liquid at the Edge of a Graphene Vacuum
 Luttinger Liquid at the Edge of a Graphene Vacuum H.A. Fertig, Indiana University Luis Brey, CSIC, Madrid I. Introduction: Graphene Edge States (Non-Interacting) II. III. Quantum Hall Ferromagnetism and
Luttinger Liquid at the Edge of a Graphene Vacuum H.A. Fertig, Indiana University Luis Brey, CSIC, Madrid I. Introduction: Graphene Edge States (Non-Interacting) II. III. Quantum Hall Ferromagnetism and
Spring /2/ pts 1 point per minute
 Physics 519 MIDTERM Name: Spring 014 6//14 80 pts 1 point per minute Exam procedures. Please write your name above. Please sit away from other students. If you have a question about the exam, please ask.
Physics 519 MIDTERM Name: Spring 014 6//14 80 pts 1 point per minute Exam procedures. Please write your name above. Please sit away from other students. If you have a question about the exam, please ask.
N independent electrons in a volume V (assuming periodic boundary conditions) I] The system 3 V = ( ) k ( ) i k k k 1/2
![N independent electrons in a volume V (assuming periodic boundary conditions) I] The system 3 V = ( ) k ( ) i k k k 1/2 N independent electrons in a volume V (assuming periodic boundary conditions) I] The system 3 V = ( ) k ( ) i k k k 1/2](/thumbs/90/101212070.jpg) Lecture #6. Understanding the properties of metals: the free electron model and the role of Pauli s exclusion principle.. Counting the states in the E model.. ermi energy, and momentum. 4. DOS 5. ermi-dirac
Lecture #6. Understanding the properties of metals: the free electron model and the role of Pauli s exclusion principle.. Counting the states in the E model.. ermi energy, and momentum. 4. DOS 5. ermi-dirac
