University Chemistry
|
|
|
- Marsha Franklin
- 6 years ago
- Views:
Transcription
1 University Chemistry 1
2 Electromagnetic Radiation Electromagnetic radiation has some of the properties of both a particle and a wave. Particles: have a definite mass and they occupy space. Wave have no mass and yet they carry energy as they travel through space. Waves have four other characteristic properties: speed, frequency, wavelength, and amplitude. The frequency (v) is the number of waves (or cycles) per unit of time and its units of cycles per second (s-1) or hertz (Hz). The wavelength (l) is the smallest distance between repeating points on the wave. The product of the frequency (v) times the wavelength (l) of a wave is therefore the speed (s) at which the wave travels through space. 2
3 The product of the frequency (ν) times the wavelength (λ) of a wave is therefore the speed (s) at which the wave travels through space. s = ν λ or ν = s /λ S ν λ What is the speed of a wave that has a wavelength of 1 meter and a frequency of 60 cycles per second? λ = 1 m and ν = 60 cycles per second (Hz) s = ν λ = 60 (Hz) x 1 m = 60 m per second 3
4 Light is a wave with both electric and magnetic components. It is therefore a form of electromagnetic radiation. The product of the frequency times the wavelength of electromagnetic radiation is always equal to the speed of light (c), 3.00 x 10 8 m/s. c = ν λ or ν = c /λ ν c λ 4
5 Calculate the frequency of red light that has a wavelength of nm if the speed of light is 3.00 x 10 8 m/s. λ = nm = x 10-9 m and c = 3.00 x 10 8 m/s c = ν λ or ν = c / λ ν = 3.00 x 10 8 (m/s) / x 10-9 (m) ν = 4.29 x (s -1 ) or Hz ν c λ Particle-Like Behavior of Light and Planck s Equation Light is composed of particles called photons. The energy of this photon is equal to the frequency of the light times a constant and can be calculated using the formula: E photon = h ν h E ν where E is the energy of the photon, ν is the frequency and h is called Plank's constant, h = J s 5
6 What is the energy (in kilojoules) of photons of radar waves with a frequency equal 4.00 x 10 8 Hz? Using the formula E photon = h ν E = ( J s) x (4.00 x 10 8 s -1 ) = 2.65 x J = 2.65 x kj h E ν What is the wavelength, in nanometers, of light that has an energy content of 508 kj/mol photons? 1000J 1molephoton Energy = 508 kj/mol photons x x = 8.44 x kJ -19 J/photon 6.023x10 photons E = hν = h c/λ λ = hc/e 34 8 (6.626x10 J s)(3.00x10 λ = x 10 J λ = 236 x 10-9 m = 236 x 10-9 x 10 9 nm = 236 nm m ) s = 236 x 10-9 m 6
7 Development of Current Atomic Theory Atomic spectrum can be used as a "fingerprint" for an element and the scientists conclude that if atoms emit only discrete wavelengths, may be atoms can have only discrete energies. The discrete amounts of energy are absorbed or released (energy is said to be quantized). Atoms absorb and emit electromagnetic radiation as the energies of their electrons change. Bohr s Theory of the hydrogen atom The Bohr model of the hydrogen atom uses the assumptions that: - the light negatively charged electron circulates around the much heavier positively charged. - the electron, although often acting like a particle, is actually a wave. - the electron fits in a circular orbit around the proton. 7
8 In the case of hydrogen atom spectra, the energies that the electron can possess are given by: E n = -R H (1/n 2 ) where R H, the Rydberg constant, has the value 2.18 x J. The number n is the integer called the principal quantum number; it has the values n = 1, 2, 3, 4, Example Calculate the value of the energy level (n = 3) of the hydrogen atom according to Bohr-Theory. Answer According to the equation: E n = -R H (1/n 2 ) E 3 = x J (1/3 2 ) = x J 8
9 E i = -R H (1/n i2 ) E f = -R H (1/n f2 ) E = hν =E f E i = R H (1/n i2-1/n f2 ) Lyman series is due to the transfer of electrons from excited state to n =1 Balmer series is due to the transfer of electrons from excited state to n =2 Paschen series is due to the transfer of electrons from excited state to n =3 Brackett series is due to the transfer of electrons from excited state to n =4 Pfund series is due to the transfer of electrons from excited state to n =5 9
10 What is the wavelength of a photon (in nanometers) emitted during a transition from the ni = 5 state to the nf = 1 state in the hydrogen atom? Step 1: Calculate E using the following equation: E = hν =E f E i = R H (1/n i2-1/n f2 ) = 2.18 x J (1/5 2-1/1 2 ) = x J The negative sign (-ve) indicates that this energy associated with an emission process. Step 2: calculate the wavelength, (omit the ve sign of E). E = 2.09 x J = hν = h x c/ λ λ = c h/ E = (3.00 x 10 8 m/s) (6.63 x J.s) / (2.09 x J) Wavelength λ = 9.52 x 10-8 m = 9.52 x 10-8 x 10 9 nm = 94.2 nm 10
11 What is the wavelength of a photon (in nanometers) emitted during a transition from the n i = 5 state to the n f = 1 state in the hydrogen atom? OR in one step: λ = 9.12 x ( - 2 n n i -8 m 2 f ) λ = x ( m ) = x ( ) m = 9.12 x m Wavelength λ = 9.52 x 10-8 m = 9.52 x 10-8 x 10 9 nm = 94.2 nm 11
12 Duality of the Electron Electron can be described as if it were a wave and particle. Since waves are described by their wavelength λ and particles are described by their momentum, p, E = h ν = hc/λ (wave) E = m c 2 = p c (particle) The DeBroglie relationship:between momentum (a particle property) and wavelength (a wave property) p = m c = h/λ or m s = h/ λ m s = h/ λ (m is the mass in kg and s is the speed in m/s), 12
13 What is the DeBroglie wavelength (in meter) of an electron of a mass 9.11 x kg and a speed of 2.2 x 10 6 m/s. (Notice that: J.s = kg.m 2.s -1 ) Using the formula: m s = h/ λ or λ = h/ m s λ = h/ m s = (6.63 x kg.m 2.s -1 )/(9.11 x x 2.2 x 10 6 m s -1 ) = 3.3 x m Calculate the energy (in joules) of a photon with a wavelength of 6.00 x 10 4 nm. Using the following formula: E photon = h ν and ν = c/ λ Wavelength (λ) = 6.00 x 10 4 nm = 6.00 x 10 4 x 10-9 m = 6.00 x 10-5 m, Speed of light (c) = 3.00 x 10 8 m/s, Planck s constant (h) = 6.63 x J.s ν = 0.5 x s -1 and E photon = x J 13
14 لمزيد من التمارين و الشرح أحصل على نسختك من آتاب University Chemistry من مكتبة خوارزم 14
Atomic Structure. Standing Waves x10 8 m/s. (or Hz or 1/s) λ Node
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE Brooks/Cole - Thomson
 Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Lecture 11 Atomic Structure
 Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
Lecture 11 Atomic Structure Earlier in the semester, you read about the discoveries that lead to the proposal of the nuclear atom, an atom of atomic number Z, composed of a positively charged nucleus surrounded
I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited
 NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
NCCS 1.1.2 & 1.1.3 I understand the relationship between energy and a quanta I understand the difference between an electron s ground state and an electron s excited state I will describe how an electron
Atomic Structure 11/21/2011
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
The Bohr Model of the Atom
 Unit 4: The Bohr Model of the Atom Properties of light Before the 1900 s, light was thought to behave only as a wave. Light is a type of electromagnetic radiation - a form of energy that exhibits wave
Unit 4: The Bohr Model of the Atom Properties of light Before the 1900 s, light was thought to behave only as a wave. Light is a type of electromagnetic radiation - a form of energy that exhibits wave
QUANTUM MECHANICS Chapter 12
 QUANTUM MECHANICS Chapter 12 Colours which appear through the Prism are to be derived from the Light of the white one Sir Issac Newton, 1704 Electromagnetic Radiation (prelude) FIG Electromagnetic Radiation
QUANTUM MECHANICS Chapter 12 Colours which appear through the Prism are to be derived from the Light of the white one Sir Issac Newton, 1704 Electromagnetic Radiation (prelude) FIG Electromagnetic Radiation
Chapter 7. Quantum Theory and Atomic Structure
 Chapter 7 Quantum Theory and Atomic Structure Outline 1. The Nature of Light 2. Atomic Spectra 3. The Wave-Particle Duality of Matter and Energy 4. The Quantum-Mechanical Model of the Atom 3 September
Chapter 7 Quantum Theory and Atomic Structure Outline 1. The Nature of Light 2. Atomic Spectra 3. The Wave-Particle Duality of Matter and Energy 4. The Quantum-Mechanical Model of the Atom 3 September
Properties of Light and Atomic Structure. Chapter 7. So Where are the Electrons? Electronic Structure of Atoms. The Wave Nature of Light!
 Properties of Light and Atomic Structure Chapter 7 So Where are the Electrons? We know where the protons and neutrons are Nuclear structure of atoms (Chapter 2) The interaction of light and matter helps
Properties of Light and Atomic Structure Chapter 7 So Where are the Electrons? We know where the protons and neutrons are Nuclear structure of atoms (Chapter 2) The interaction of light and matter helps
Quick Review. 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior.
 Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Chapter 7 Atomic Structure -1 Quantum Model of Atom. Dr. Sapna Gupta
 Chapter 7 Atomic Structure -1 Quantum Model of Atom Dr. Sapna Gupta The Electromagnetic Spectrum The electromagnetic spectrum includes many different types of radiation which travel in waves. Visible light
Chapter 7 Atomic Structure -1 Quantum Model of Atom Dr. Sapna Gupta The Electromagnetic Spectrum The electromagnetic spectrum includes many different types of radiation which travel in waves. Visible light
Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum. All EM radiation travels at the speed of light, c = 3 x 10 8 m/s
 Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum V I B G Y O R All EM radiation travels at the speed of light, c = 3 x 10 8 m/s Electromagnetic radiation is a wave with a wavelength
Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum V I B G Y O R All EM radiation travels at the speed of light, c = 3 x 10 8 m/s Electromagnetic radiation is a wave with a wavelength
Energy and the Quantum Theory
 Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
Energy and the Quantum Theory Light electrons are understood by comparing them to light 1. radiant energy 2. travels through space 3. makes you feel warm Light has properties of waves and particles Amplitude:
Chapter 6. Quantum Theory and the Electronic Structure of Atoms Part 1
 Chapter 6 Quantum Theory and the Electronic Structure of Atoms Part 1 The nature of light Quantum theory Topics Bohr s theory of the hydrogen atom Wave properties of matter Quantum mechanics Quantum numbers
Chapter 6 Quantum Theory and the Electronic Structure of Atoms Part 1 The nature of light Quantum theory Topics Bohr s theory of the hydrogen atom Wave properties of matter Quantum mechanics Quantum numbers
Electronic Structure of Atoms. Chapter 6
 Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
WAVE NATURE OF LIGHT
 WAVE NATURE OF LIGHT Light is electromagnetic radiation, a type of energy composed of oscillating electric and magnetic fields. The fields oscillate perpendicular to each other. In vacuum, these waves
WAVE NATURE OF LIGHT Light is electromagnetic radiation, a type of energy composed of oscillating electric and magnetic fields. The fields oscillate perpendicular to each other. In vacuum, these waves
Quantum Theory and Atomic Structure. Quantum Mechanics. Quantum Theory and Atomic Structure. 7.3 The Wave-Particle Duality of Matter and Energy
 Chapter 7 Quantum Theory and Atomic Structure Chap 7-1 Quantum Theory and Atomic Structure 7.1 The Nature of Light 7.2 Atomic Spectra 7.3 The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical
Chapter 7 Quantum Theory and Atomic Structure Chap 7-1 Quantum Theory and Atomic Structure 7.1 The Nature of Light 7.2 Atomic Spectra 7.3 The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical
Chapter 7. Quantum Theory and Atomic Structure. Quantum Mechanics. Chap 7-1
 Chapter 7 Quantum Theory and Atomic Structure Chap 7-1 Quantum Theory and Atomic Structure 7.1 The Nature of Light 7.2 Atomic Spectra 7.3 The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical
Chapter 7 Quantum Theory and Atomic Structure Chap 7-1 Quantum Theory and Atomic Structure 7.1 The Nature of Light 7.2 Atomic Spectra 7.3 The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical
Electron Arrangement - Part 1
 Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
Problems with the atomic model?
 Modern Atomic Theory- Electronic Structure of Atoms DR HNIMIR-CH7 Where should (-) electrons be found? Problems with the atomic model? First, a Little About Electromagnetic Radiation- Waves Another Look
Modern Atomic Theory- Electronic Structure of Atoms DR HNIMIR-CH7 Where should (-) electrons be found? Problems with the atomic model? First, a Little About Electromagnetic Radiation- Waves Another Look
Modern Atomic Theory and Electron Configurations
 Chem 101 Modern Atomic Theory and Electron Configurations Lectures 8 and 9 Types of Electromagnetic Radiation Electromagnetic radiation is given off by atoms when they have been excited by any form of
Chem 101 Modern Atomic Theory and Electron Configurations Lectures 8 and 9 Types of Electromagnetic Radiation Electromagnetic radiation is given off by atoms when they have been excited by any form of
The early periodic table based on atomic weight. (Section 5.1) Lets review: What is a hydrogen atom? 1 electron * nucleus H 1 proton
 PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
Chp 6: Atomic Structure
 Chp 6: Atomic Structure 1. Electromagnetic Radiation 2. Light Energy 3. Line Spectra & the Bohr Model 4. Electron & Wave-Particle Duality 5. Quantum Chemistry & Wave Mechanics 6. Atomic Orbitals Overview
Chp 6: Atomic Structure 1. Electromagnetic Radiation 2. Light Energy 3. Line Spectra & the Bohr Model 4. Electron & Wave-Particle Duality 5. Quantum Chemistry & Wave Mechanics 6. Atomic Orbitals Overview
Bohr. Electronic Structure. Spectroscope. Spectroscope
 Bohr Electronic Structure Bohr proposed that the atom has only certain allowable energy states. Spectroscope Using a device called a it was found that gaseous elements emitted electromagnetic radiation
Bohr Electronic Structure Bohr proposed that the atom has only certain allowable energy states. Spectroscope Using a device called a it was found that gaseous elements emitted electromagnetic radiation
Part One: Light Waves, Photons, and Bohr Theory. 2. Beyond that, nothing was known of arrangement of the electrons.
 CHAPTER SEVEN: QUANTUM THEORY AND THE ATOM Part One: Light Waves, Photons, and Bohr Theory A. The Wave Nature of Light (Section 7.1) 1. Structure of atom had been established as cloud of electrons around
CHAPTER SEVEN: QUANTUM THEORY AND THE ATOM Part One: Light Waves, Photons, and Bohr Theory A. The Wave Nature of Light (Section 7.1) 1. Structure of atom had been established as cloud of electrons around
ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 PROJECT DATE. Tuesday, October 13, 15
 PROJECT DATE ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 Agenda Begin Topic 2.2 Electron Configuration Homework: Energy of waves calculations Due tomorrow! Yes - tomorrow Atomic Structure The nuclear
PROJECT DATE ATOMIC STRUCTURE ELECTRON CONFIGURATION 10/13/15 Agenda Begin Topic 2.2 Electron Configuration Homework: Energy of waves calculations Due tomorrow! Yes - tomorrow Atomic Structure The nuclear
Chapter 6 Electronic Structure of Atoms. 許富銀 ( Hsu Fu-Yin)
 Chapter 6 Electronic Structure of Atoms 許富銀 ( Hsu Fu-Yin) 1 The Wave Nature of Light The light we see with our eyes, visible light, is one type of electromagnetic radiation. electromagnetic radiation carries
Chapter 6 Electronic Structure of Atoms 許富銀 ( Hsu Fu-Yin) 1 The Wave Nature of Light The light we see with our eyes, visible light, is one type of electromagnetic radiation. electromagnetic radiation carries
An element Is a substance that cannot be split into simpler substance. It is composed of discrete particles called atoms.
 digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
Physics 1C Lecture 29B
 Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
The relationship between these aspects is described by the following equation: E = hν =
 1 Learning Outcomes EXPERIMENT A10: LINE SPECTRUM Upon completion of this lab, the student will be able to: 1) Examine the line spectrum of the hydrogen atom. 2) Calculate the frequency and energy of the
1 Learning Outcomes EXPERIMENT A10: LINE SPECTRUM Upon completion of this lab, the student will be able to: 1) Examine the line spectrum of the hydrogen atom. 2) Calculate the frequency and energy of the
Atomic Structure and the Periodic Table
 Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
Atomic Structure and the Periodic Table The electronic structure of an atom determines its characteristics Studying atoms by analyzing light emissions/absorptions Spectroscopy: analysis of light emitted
The Photoelectric Effect
 The Photoelectric Effect Light can strike the surface of some metals causing an electron to be ejected No matter how brightly the light shines, electrons are ejected only if the light has sufficient energy
The Photoelectric Effect Light can strike the surface of some metals causing an electron to be ejected No matter how brightly the light shines, electrons are ejected only if the light has sufficient energy
3.03 EMR and the H-atom
 3.03 EMR and the H-atom What are the component of light? How are the electrons arranged in the atom?! What is the relationship between light and the atom? How does light give clues about the structure
3.03 EMR and the H-atom What are the component of light? How are the electrons arranged in the atom?! What is the relationship between light and the atom? How does light give clues about the structure
Optical Spectroscopy and Atomic Structure. PHYS 0219 Optical Spectroscopy and Atomic Structure 1
 Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
2.1- CLASSICAL CONCEPTS; Dr. A. DAYALAN, Former Prof & Head 1
 2.1- CLASSICAL CONCEPTS; Dr. A. DAYALAN, Former Prof & Head 1 QC-2 QUANTUM CHEMISTRY (Classical Concept) Dr. A. DAYALAN,Former Professor & Head, Dept. of Chemistry, LOYOLA COLLEGE (Autonomous), Chennai
2.1- CLASSICAL CONCEPTS; Dr. A. DAYALAN, Former Prof & Head 1 QC-2 QUANTUM CHEMISTRY (Classical Concept) Dr. A. DAYALAN,Former Professor & Head, Dept. of Chemistry, LOYOLA COLLEGE (Autonomous), Chennai
Unit 3. Chapter 4 Electrons in the Atom. Niels Bohr s Model. Recall the Evolution of the Atom. Bohr s planetary model
 Unit 3 Chapter 4 Electrons in the Atom Electrons in the Atom (Chapter 4) & The Periodic Table/Trends (Chapter 5) Niels Bohr s Model Recall the Evolution of the Atom He had a question: Why don t the electrons
Unit 3 Chapter 4 Electrons in the Atom Electrons in the Atom (Chapter 4) & The Periodic Table/Trends (Chapter 5) Niels Bohr s Model Recall the Evolution of the Atom He had a question: Why don t the electrons
Georgia Institute of Technology CHEM 1310 revised 10/8/09 Spring The Development of Quantum Mechanics. ν (nu) = frequency (in s -1 or hertz)
 The Development of Quantum Mechanics Early physicists used the properties of electromagnetic radiation to develop fundamental ideas about the structure of the atom. A fundamental assumption for their work
The Development of Quantum Mechanics Early physicists used the properties of electromagnetic radiation to develop fundamental ideas about the structure of the atom. A fundamental assumption for their work
Mystery #3 Emission Spectra of Elements. Tube filled with elemental gas. Voltage can be applied across both ends, this causes the gas to emit light
 Mystery #3 Emission Spectra of Elements Tube filled with elemental gas. Voltage can be applied across both ends, this causes the gas to emit light Line Spectra Copyright The McGraw-Hill Companies, Inc.
Mystery #3 Emission Spectra of Elements Tube filled with elemental gas. Voltage can be applied across both ends, this causes the gas to emit light Line Spectra Copyright The McGraw-Hill Companies, Inc.
Electrons! Chapter 5
 Electrons! Chapter 5 I.Light & Quantized Energy A.Background 1. Rutherford s nuclear model: nucleus surrounded by fast-moving electrons; no info on how electrons move, how they re arranged, or differences
Electrons! Chapter 5 I.Light & Quantized Energy A.Background 1. Rutherford s nuclear model: nucleus surrounded by fast-moving electrons; no info on how electrons move, how they re arranged, or differences
Quantum Theory of the Atom
 The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
Chapter 6. Electronic Structure. Electronic Structure and the Periodic Table ( 전자구조와주기율표 ) 강의개요. 2) The Particle Nature of Light: Photon Energies
 Chapter 6 Electronic Structure and the Periodic Table ( 전자구조와주기율표 ) 강의개요 Electronic Structure Electron arrangement in atoms energy levels orbitals electron configurations orbital diagram Periodic Table
Chapter 6 Electronic Structure and the Periodic Table ( 전자구조와주기율표 ) 강의개요 Electronic Structure Electron arrangement in atoms energy levels orbitals electron configurations orbital diagram Periodic Table
CHEMISTRY - TRO 4E CH.7 - THE QUANTUM-MECHANICAL MODEL OF THE ATOM
 !! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
!! www.clutchprep.com CONCEPT: THE NATURE OF LIGHT Visible light represents a small portion of the continuum of radiant energy known as. The visible light spectrum ranges from to. Its wave properties of
Electromagnetic spectrum Electromagnetic radiation
 Chapter 4 Section 1 Electromagnetic spectrum includes all the different wave lengths of radiation. Electromagnetic radiation is a form of energy that exhibits wave like behavior as it travels through space.
Chapter 4 Section 1 Electromagnetic spectrum includes all the different wave lengths of radiation. Electromagnetic radiation is a form of energy that exhibits wave like behavior as it travels through space.
Ch 7 Quantum Theory of the Atom (light and atomic structure)
 Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Chapter 7 Atomic Structure and Orbitals
 Chapter 7 Atomic Structure and Orbitals Alpha Scattering Experiment: Rutherford s observations Light as Waves or Particles Wavelength (λ) is the distance between any two identical points in consecutive
Chapter 7 Atomic Structure and Orbitals Alpha Scattering Experiment: Rutherford s observations Light as Waves or Particles Wavelength (λ) is the distance between any two identical points in consecutive
Who am I? CHEM 110 General Chemistry King Abdul Aziz University Jeddah KSA. How to reach me?
 CHEM 110 General Chemistry King Abdul Aziz University Jeddah KSA Who am I? Dr. Mohamed Abdel Salam Assistant Professor of Physical Chemistry PhD in Nano Chemistry, Canada (006) M Sc in Electrochemistry,
CHEM 110 General Chemistry King Abdul Aziz University Jeddah KSA Who am I? Dr. Mohamed Abdel Salam Assistant Professor of Physical Chemistry PhD in Nano Chemistry, Canada (006) M Sc in Electrochemistry,
ATOMIC STRUCTURE. Kotz Ch 7 & Ch 22 (sect 4,5)
 ATOMIC STRUCTURE Kotz Ch 7 & Ch 22 (sect 4,5) properties of light spectroscopy quantum hypothesis hydrogen atom Heisenberg Uncertainty Principle orbitals ELECTROMAGNETIC RADIATION subatomic particles (electron,
ATOMIC STRUCTURE Kotz Ch 7 & Ch 22 (sect 4,5) properties of light spectroscopy quantum hypothesis hydrogen atom Heisenberg Uncertainty Principle orbitals ELECTROMAGNETIC RADIATION subatomic particles (electron,
White Light. Chapter 7 Electron Structure of the Atom
 Chapter 7 Electron Structure of the Atom Electromagnetic Radiation and Energy The Bohr Model of the Hydrogen Atom The Modern Model of the Atom Periodicity of Electron Configurations Valence Electrons for
Chapter 7 Electron Structure of the Atom Electromagnetic Radiation and Energy The Bohr Model of the Hydrogen Atom The Modern Model of the Atom Periodicity of Electron Configurations Valence Electrons for
Energy levels and atomic structures lectures chapter one
 Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
The ELECTRON: Wave Particle Duality. chapter 4
 The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
The ELECTRON: Wave Particle Duality chapter 4 What do we know about light? Before 1900 s scientists thought light behaved as a wave. This belief changed when it was discovered that light also has particle
Chem 115 POGIL Worksheet - Week 8 - Answers Thermochemistry (Continued), Electromagnetic Radiation, and Line Spectra. ΔH o = 285.
 Chem 115 POGIL Worksheet - Week 8 - Answers Thermochemistry (Continued), Electromagnetic Radiation, and Line Spectra Key Questions & Exercises 1. Calculate ΔH o for the reaction, Given: C 2 H 2 (g) + H
Chem 115 POGIL Worksheet - Week 8 - Answers Thermochemistry (Continued), Electromagnetic Radiation, and Line Spectra Key Questions & Exercises 1. Calculate ΔH o for the reaction, Given: C 2 H 2 (g) + H
WEEK 2: 4 SEP THRU 10 SEP; LECTURES 4-6
 Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
Learning Objectives Energy: Light as energy Describe the wave nature of light, wavelength, and frequency using the equation c = λν What is meant by the particle nature of light? Calculate the energy of
10/17/11. Chapter 7. Quantum Theory and Atomic Structure. Amplitude (intensity) of a wave. Quantum Theory and Atomic Structure
 Quantum Theory and Atomic Structure Chapter 7 7. The Nature of Light Quantum Theory and Atomic Structure 7. Atomic Spectra 7. The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical Model
Quantum Theory and Atomic Structure Chapter 7 7. The Nature of Light Quantum Theory and Atomic Structure 7. Atomic Spectra 7. The Wave-Particle Duality of Matter and Energy 7.4 The Quantum-Mechanical Model
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
6.1.5 Define frequency and know the common units of frequency.
 CHM 111 Chapter 6 Worksheet and Study Guide Purpose: This is a guide for your as you work through the chapter. The major topics are provided so that you can write notes on each topic and work the corresponding
CHM 111 Chapter 6 Worksheet and Study Guide Purpose: This is a guide for your as you work through the chapter. The major topics are provided so that you can write notes on each topic and work the corresponding
Chapter 7: The Quantum-Mechanical Model of the Atom
 C h e m i s t r y 1 A : C h a p t e r 7 P a g e 1 Chapter 7: The Quantum-Mechanical Model of the Atom Homework: Read Chapter 7. Work out sample/practice exercises Check for the MasteringChemistry.com assignment
C h e m i s t r y 1 A : C h a p t e r 7 P a g e 1 Chapter 7: The Quantum-Mechanical Model of the Atom Homework: Read Chapter 7. Work out sample/practice exercises Check for the MasteringChemistry.com assignment
5.111 Lecture Summary #5 Friday, September 12, 2014
 5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
Structure of the atom
 Structure of the atom What IS the structure of an atom? What are the properties of atoms? REMEMBER: structure affects function! Important questions: Where are the electrons? What is the energy of an electron?
Structure of the atom What IS the structure of an atom? What are the properties of atoms? REMEMBER: structure affects function! Important questions: Where are the electrons? What is the energy of an electron?
Quantum Theory and the Electronic Structure of Atoms
 Quantum Theory and the Electronic Structure of Atoms Chapter 7 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Properties of Waves Wavelength ( ) is the distance
Quantum Theory and the Electronic Structure of Atoms Chapter 7 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Properties of Waves Wavelength ( ) is the distance
Chemistry (
 Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
Atomic Structure Part II Electrons in Atoms
 Atomic Structure Part II Electrons in Atoms Radiant energy travels in the form of waves that have both electrical and magnetic properties. These electromagnetic waves can travel through empty space, as
Atomic Structure Part II Electrons in Atoms Radiant energy travels in the form of waves that have both electrical and magnetic properties. These electromagnetic waves can travel through empty space, as
The Development of Atomic Theory
 The Development of Atomic Theory Democritus (400 BC) John Dalton (1803) J.J. Thomson (1897) Ernest Rutherford (1911) James Chadwick (1932) - suggested that matter is composed of indivisible particles called
The Development of Atomic Theory Democritus (400 BC) John Dalton (1803) J.J. Thomson (1897) Ernest Rutherford (1911) James Chadwick (1932) - suggested that matter is composed of indivisible particles called
Chemistry is in the electrons
 Chemistry is in the electrons Electronic structure arrangement of electrons in atom Two parameters: Energy Position The popular image of the atom is incorrect: electrons are not miniature planets orbiting
Chemistry is in the electrons Electronic structure arrangement of electrons in atom Two parameters: Energy Position The popular image of the atom is incorrect: electrons are not miniature planets orbiting
I. Multiple Choice Questions (Type-I)
 I. Multiple Choice Questions (Type-I) 1. Which of the following conclusions could not be derived from Rutherford s α -particle scattering experiement? (i) Most of the space in the atom is empty. (ii) The
I. Multiple Choice Questions (Type-I) 1. Which of the following conclusions could not be derived from Rutherford s α -particle scattering experiement? (i) Most of the space in the atom is empty. (ii) The
Astronomy The Nature of Light
 Astronomy The Nature of Light A. Dayle Hancock adhancock@wm.edu Small 239 Office hours: MTWR 10-11am Measuring the speed of light Light is an electromagnetic wave The relationship between Light and temperature
Astronomy The Nature of Light A. Dayle Hancock adhancock@wm.edu Small 239 Office hours: MTWR 10-11am Measuring the speed of light Light is an electromagnetic wave The relationship between Light and temperature
 Question 12.1: Choose the correct alternative from the clues given at the end of the each statement: (a) The size of the atom in Thomson s model is... the atomic size in Rutherford s model. (much greater
Question 12.1: Choose the correct alternative from the clues given at the end of the each statement: (a) The size of the atom in Thomson s model is... the atomic size in Rutherford s model. (much greater
Accounts for certain objects being colored. Used in medicine (examples?) Allows us to learn about structure of the atom
 1.1 Interaction of Light and Matter Accounts for certain objects being colored Used in medicine (examples?) 1.2 Wavelike Properties of Light Wavelength, : peak to peak distance Amplitude: height of the
1.1 Interaction of Light and Matter Accounts for certain objects being colored Used in medicine (examples?) 1.2 Wavelike Properties of Light Wavelength, : peak to peak distance Amplitude: height of the
Chapter 27 Early Quantum Theory and Models of the Atom Discovery and Properties of the electron
 Chapter 27 Early Quantum Theory and Models of the Atom 27-1 Discovery and Properties of the electron Measure charge to mass ratio e/m (J. J. Thomson, 1897) When apply magnetic field only, the rays are
Chapter 27 Early Quantum Theory and Models of the Atom 27-1 Discovery and Properties of the electron Measure charge to mass ratio e/m (J. J. Thomson, 1897) When apply magnetic field only, the rays are
Electrons in Atoms. Section 5.1 Light and Quantized Energy Section 5.2 Quantum Theory and the Atom Section 5.3 Electron Configuration
 Electrons in Atoms Section 5.1 Light and Quantized Energy Section 5.2 Quantum Theory and the Atom Section 5.3 Electron Configuration Click a hyperlink or folder tab to view the corresponding slides. Exit
Electrons in Atoms Section 5.1 Light and Quantized Energy Section 5.2 Quantum Theory and the Atom Section 5.3 Electron Configuration Click a hyperlink or folder tab to view the corresponding slides. Exit
Electronic Structure. of Atoms. 2009, Prentice-Hall, Inc. Electronic Structure. of Atoms. 2009, Prentice-Hall, Inc. Electronic Structure.
 Chemistry, The Central Science, 11th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 Section 1 6: The Marathon Adapted from: John D. Bookstaver St. Charles Community College
Chemistry, The Central Science, 11th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 Section 1 6: The Marathon Adapted from: John D. Bookstaver St. Charles Community College
The ELECTRON: Wave Particle Duality
 The ELECTRON: Wave Particle Duality No familiar conceptions can be woven around the electron. Something unknown is doing we don t know what. -Sir Arthur Eddington The Nature of the Physical World (1934)
The ELECTRON: Wave Particle Duality No familiar conceptions can be woven around the electron. Something unknown is doing we don t know what. -Sir Arthur Eddington The Nature of the Physical World (1934)
Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師
 Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師 2018-10-2 1 2 Light and the Electromagnetic Spectrum Electromagnetic energy ( light ) is characterized by wavelength, frequency, and amplitude.
Periodicity and the Electronic Structure of Atoms 國防醫學院生化學科王明芳老師 2018-10-2 1 2 Light and the Electromagnetic Spectrum Electromagnetic energy ( light ) is characterized by wavelength, frequency, and amplitude.
What are the component of light? How are the electrons arranged in the atom? What is the relationship between light and the atom?
 What are the component of light? How are the electrons arranged in the atom? What is the relationship between light and the atom? How does light give clues about the structure of the atom? 1 The Electro-
What are the component of light? How are the electrons arranged in the atom? What is the relationship between light and the atom? How does light give clues about the structure of the atom? 1 The Electro-
Test bank chapter (7)
 Test bank chapter (7) Choose the most correct answer 1. The lowest energy state of an atom is referred to as its a) bottom state. b) ground state. c) fundamental state. d) original state. 2. All s orbitals
Test bank chapter (7) Choose the most correct answer 1. The lowest energy state of an atom is referred to as its a) bottom state. b) ground state. c) fundamental state. d) original state. 2. All s orbitals
SCH4U: History of the Quantum Theory
 SCH4U: History of the Quantum Theory Black Body Radiation When an object is heated, it initially glows red hot and at higher temperatures becomes white hot. This white light must consist of all of the
SCH4U: History of the Quantum Theory Black Body Radiation When an object is heated, it initially glows red hot and at higher temperatures becomes white hot. This white light must consist of all of the
CVB102 Lecture 1 - Chemical Structure and Reactivity. Contact Information: Dr. Bill Lot Electronic Structure of Atoms
 CVB102 Lecture 1 - Chemical Structure and Reactivity Contact Information: Dr. Bill Lot b.lott@qut.edu.au Electronic Structure of Atoms Text: Blackman, et al Pp. 127-147 (Pp. 148-159 recommended) The periodic
CVB102 Lecture 1 - Chemical Structure and Reactivity Contact Information: Dr. Bill Lot b.lott@qut.edu.au Electronic Structure of Atoms Text: Blackman, et al Pp. 127-147 (Pp. 148-159 recommended) The periodic
CHEMISTRY Matter and Change
 CHEMISTRY Matter and Change Chapter 5: Electrons in Atoms 5 Section 5.1 Section Section 5.3 Table Of Contents Light and Quantized Energy Electron Configuration Compare the wave and particle natures of
CHEMISTRY Matter and Change Chapter 5: Electrons in Atoms 5 Section 5.1 Section Section 5.3 Table Of Contents Light and Quantized Energy Electron Configuration Compare the wave and particle natures of
Chapter 6. Electronic. Electronic Structure of Atoms Pearson Education
 Chapter 6 Laser: step-like energy transition 6.1 The Wave Nature of Light 6.2 Quantized Energy and Photons 6.3 Line Spectra and the Bohr Model 6.4 The Wave Behavior of Matter 6.5 Quantum Mechanics and
Chapter 6 Laser: step-like energy transition 6.1 The Wave Nature of Light 6.2 Quantized Energy and Photons 6.3 Line Spectra and the Bohr Model 6.4 The Wave Behavior of Matter 6.5 Quantum Mechanics and
Atomic Spectroscopy and Energy Levels
 Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
Activity 18 Atomic Spectroscopy and Energy Levels Why? The emission of light by the hydrogen atom and other atoms played a key role in helping scientists to understand the electronic structure of atoms.
QUANTUM THEORY & ATOMIC STRUCTURE
 QUANTUM THEORY & ATOMIC STRUCTURE GENERAL CHEMISTRY by Dr. Istadi 1 THE NATURE OF LIGHT Visible light is one type of electromagnetic radiation ( radiation (electromagnetic The electromagnetic radiation
QUANTUM THEORY & ATOMIC STRUCTURE GENERAL CHEMISTRY by Dr. Istadi 1 THE NATURE OF LIGHT Visible light is one type of electromagnetic radiation ( radiation (electromagnetic The electromagnetic radiation
Ch. 7 The Quantum Mechanical Atom. Brady & Senese, 5th Ed.
 Ch. 7 The Quantum Mechanical Atom Brady & Senese, 5th Ed. Index 7.1. Electromagnetic radiation provides the clue to the electronic structures of atoms 7.2. Atomic line spectra are evidence that electrons
Ch. 7 The Quantum Mechanical Atom Brady & Senese, 5th Ed. Index 7.1. Electromagnetic radiation provides the clue to the electronic structures of atoms 7.2. Atomic line spectra are evidence that electrons
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS
 1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
1) Introduction 2) Photo electric effect 3) Dual nature of matter 4) Bohr s atom model 5) LASERS 1. Introduction Types of electron emission, Dunnington s method, different types of spectra, Fraunhoffer
UNIT 4 Electrons in Atoms. Advanced Chemistry 235 Lanphier High School Mr. David Peeler
 UNIT 4 Electrons in Atoms Advanced Chemistry 235 Lanphier High School Mr. David Peeler Section 4.1 Models of the Atom OBJECTIVES: Identify the inadequacies in the Rutherford atomic model. Section 4.1 Models
UNIT 4 Electrons in Atoms Advanced Chemistry 235 Lanphier High School Mr. David Peeler Section 4.1 Models of the Atom OBJECTIVES: Identify the inadequacies in the Rutherford atomic model. Section 4.1 Models
QUANTUM THEORY & ATOMIC STRUCTURE. GENERAL CHEMISTRY by Dr. Istadi
 QUANTUM THEORY & ATOMIC STRUCTURE GENERAL CHEMISTRY by Dr. Istadi 1 THE NATURE OF LIGHT Visible light is one type of electromagnetic radiation ( radiation (electromagnetic The electromagnetic radiation
QUANTUM THEORY & ATOMIC STRUCTURE GENERAL CHEMISTRY by Dr. Istadi 1 THE NATURE OF LIGHT Visible light is one type of electromagnetic radiation ( radiation (electromagnetic The electromagnetic radiation
CHAPTER STRUCTURE OF ATOM
 12 CHAPTER STRUCTURE OF ATOM 1. The spectrum of He is expected to be similar to that [1988] H Li + Na He + 2. The number of spherical nodes in 3p orbitals are [1988] one three none two 3. If r is the radius
12 CHAPTER STRUCTURE OF ATOM 1. The spectrum of He is expected to be similar to that [1988] H Li + Na He + 2. The number of spherical nodes in 3p orbitals are [1988] one three none two 3. If r is the radius
10/4/2011. Tells you the number of protons
 Atomic Structure The arrangement of the subatomic particles within the atom determines the chemical properties of the elements How they interact with one another The types of ions and structures that they
Atomic Structure The arrangement of the subatomic particles within the atom determines the chemical properties of the elements How they interact with one another The types of ions and structures that they
Physics 1C Lecture 29A. Finish off Ch. 28 Start Ch. 29
 Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
AH Chemistry Unit 1. Electromagnetic Radiation and Atomic Spectra
 AH Chemistry Unit 1 Electromagnetic Radiation and Atomic Spectra Introduction This topic describes the Electromagnetic Spectrum and how it can interact with atoms, Spectroscopy. Much information about
AH Chemistry Unit 1 Electromagnetic Radiation and Atomic Spectra Introduction This topic describes the Electromagnetic Spectrum and how it can interact with atoms, Spectroscopy. Much information about
PHYS 202. Lecture 23 Professor Stephen Thornton April 25, 2005
 PHYS 202 Lecture 23 Professor Stephen Thornton April 25, 2005 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
PHYS 202 Lecture 23 Professor Stephen Thornton April 25, 2005 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration
 Electrons in Atoms October 20, 2014 Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration 1 Electromagnetic Spectrum Electromagnetic radiation
Electrons in Atoms October 20, 2014 Table of Contents Electrons in Atoms > Light and Quantized Energy > Quantum Theory and the Atom > Electron Configuration 1 Electromagnetic Spectrum Electromagnetic radiation
Key Equations. Determining the smallest change in an atom's energy.
 ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
ATOMIC STRUCTURE AND PERIODICITY Matter and Energy Key Equations λν = c ΔE = hν Relating speed of a wave to its wavelength and frequency. Determining the smallest change in an atom's energy. H( λ =R n
Atomic Structure Part II. Electrons in Atoms
 Atomic Structure Part II Electrons in Atoms Radiant energy travels in the form of waves that have both electrical and magnetic properties. These electromagnetic waves can travel through empty space, as
Atomic Structure Part II Electrons in Atoms Radiant energy travels in the form of waves that have both electrical and magnetic properties. These electromagnetic waves can travel through empty space, as
Different energy levels
 Different energy levels In the microscopic world energy is discrete www.cgrahamphysics.com Review Atomic electrons can only exist in certain discrete energy levels Light is made of photons When e s lose
Different energy levels In the microscopic world energy is discrete www.cgrahamphysics.com Review Atomic electrons can only exist in certain discrete energy levels Light is made of photons When e s lose
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
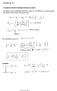 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY
 THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell?
 AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
AP Chemistry Ms. Ye Name Date Block Bohr Diagram, Lewis Structures, Valence Electrons Review 1. What is the maximum number of electrons you can fit in each energy level or shell? 1 st shell 2 nd shell
Because light behaves like a wave, we can describe it in one of two ways by its wavelength or by its frequency.
 Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
Light We can use different terms to describe light: Color Wavelength Frequency Light is composed of electromagnetic waves that travel through some medium. The properties of the medium determine how light
