UHV - Technology. Oswald Gröbner
|
|
|
- Raymond Fisher
- 5 years ago
- Views:
Transcription
1 School on Synchrotron Radiation UHV - Technology Trieste, April ) Introduction and some basics 2) Building blocks of a vacuum system 3) How to get clean ultra high vacuum 4) Desorption phenomena 5) Practical examples, Retired from CERN Vacuum Group. Present Address: Schmiedgasse 5, Innsbruck, AT-6020, Austria Oswald.Grobner@cern.ch, Oswald.Groebner@CHELLO.at
2 Books UHV-Technology Literature The Physical Basis of Ultrahigh Vacuum, P.A. Redhead, J.P. Hobson, E.V. Kornelsen, American Vacuum Society Classics, American Institute of Physics, 1993 Foundations of Vacuum Science and Technology, Ed. J.M. Lafferty, John Wiley & Sons, 1998 Handbook of Accelerator Physics and Engineering, A. W. Chao, M. Tigner, World Scientific, 1998 CAS CERN Accelerator School : Vacuum Technology, Ed. : S. Turner. CERN 99-05, 19 August 1999 Handbuch Vakuumtechnik, M. Wutz et. al, Vieweg, Braunschweig/Wiesbaden, 2000 Journals: VACUUM Journal of Vacuum Science and Technology (A) Nuclear Instruments and Methods (Section A)
3 Accelerators at CERN Historical map from 1996 Nearly all CERN accelerators require ultrahigh vacuum Total length of vacuum systems over 60 km LEP to be replaced by LHC (cryogenic vacuum system)
4 Pressure and Molecular Density Ideal gas law: P V = N No R T P pressure, V volume, T temperature N number of molecules R gas constant = 8.31 kj kmol -1 K -1, No = molecules kmol -1 Molecular density n = N V Pressure : P = n k T Boltzmann constant k = J/K Note : R = No k Note: In nearly all cases, it is the gas density rather than the pressure which matters. Units : Pressure : Pa (N/m 2 ), mbar = 100 Pa, Torr = 133 Pa Gas load : Pa m 3 = 7.5 Torr l, mbar l ~ molecules at RT Specific outgassing rate : Gas release from the walls Pa m 3 /s/m 2 ~ Torr l/s/cm 2 Leak rate : Pa m 3 /s or W, mbar l/s or Torr l/s
5 Wall collisions Frequency of wall collisions n = 1 4 n v proportional to the number density n and to the average molecular velocity v. m,v v Momentum transfer to the walls is 2 m v, hence the pressure is proportional to m n v
6 Distribution of Molecular Velocities Maxwell-Boltzmann distribution of molecular velocities at the temperature T 3 1 dn N dv = 4 - m v 2 Ê Á m ˆ 2 v 2 2 kt e p Ë 2 kt 8 kt The average velocity is given by (m = M m o ): v =, numerically ~146 p M m o T M (m s-1 ) Molecular velocities for N 2 at 50, 100, 300 and 500K. Mean molecular velocities at 20 C (m/s) H 2 N 2 Air A Kr
7 Pressure p= const m v n m mass of a molecule v average molecular velocity n rate of wall collisions A complete calculation gives const = p 2 Thus one finds and the final result Considering a molar quantity, n = N o V o One finds again the familiar result with the gas constant R p=p 2 m 8kT pm n p= n kt kt 2pm pv o = N o kt = RT
8 Mean Kinetic Energy The kinetic energy : E = 1 kin 2 m v 2 = 1 2 M m Ê 8kT ˆ oá = 4 Ë p M m o p kt M molecular weight, m o = kg does not depend on the molecular mass, M, but only on temperature T. In thermal equilibrium heavy molecules move sufficiently slowly and light molecules move sufficiently fast to carry on average the same kinetic energy. Total and Partial Pressures For each gas component n 1, n 2, n 3, the contribution to the total pressure :P i = n i kt The total pressure is therefore the sum of the partial pressures: P = Â P = k T Ân i i i i Partial pressures for atmospheric air Gas % Pi (Pa) N O Ar CO Ne He
9 Mean Free Path 1 l = 2 p D 2 n D molecular diameter (~ m) r v D Volume traversed by a molecule per second : p D 2 v Molecule collides with all other molecules contained within the cylinder of radius D. Number of collisions: Z ª p D 2 v n. The mean free path l = v Z = 1 2 p D 2 n It also follows that n l µ P l ª const. Note: 2 accounts for the fact that molecules in the cylinder are not stationary. For air the product n l ª const is ~ m -2. For N 2 at 20 deg. C and at a pressure of 1 Pa -> l ~ 9 mm
10 Molecular Flow Conditions Knudsen relation: gas flow Q µ DP applies if the mean free path >> relevant dimensions of system Molecular flow conductance c = 4 v L (m 3 H 3 /s) Ú A 2 dl 0 L length of the element (L >> transverse dimensions). H perimeter, A cross section of the element. The conductance is proportional to the mean molecular velocity, i.e. to T M. A cylindrical duct with uniform section and radius r : Ê Ë ˆ c= 4 3 v r3 Á L ~ 306 r3 Á L An orifice (pumping orifice, L~0) : c= 1 4 v A ~ 36.5 A T M. Conductance of elements in series or in parallel add the same as for electric circuits Series : 1 c = 1 c c 2 and parallel: c = c 1 +c 2 For complicated geometries it is often necessary to use Monte Carlo calculations for the molecular flow. Ê Ë ˆ T M.
11 Molecular conductance of circular tubes
12 Summary expressions: n = P kt kt = Joule r = M mo n M mo = kg mo = kg n = 1 T n v and v = l = 2p D 2 n D (N 2 ) = m M UHV-Technology Vacuum characteristics gas : Nitrogen, N 2, 20 C, M = 28 Pressure P Pa n r n l m -3 kg m -3 m -2 s -1 m atm primary vacuum high vacuum uhv xhv <10-11
13 Thermal transpiration T 1 T 2 n 1 n 2 At high pressure, the pressures on both sides are equal. In molecular flow, the net number of molecules traversing the separating wall must be zero. n 1 =n 2 Since n = 1 4 v n and v = 8kT p m n 1 T 1 = n 2 T 2 or P 1 T 1 = P 2 T 2
14 Thermal Conductivity Thermal conductivity of a gas is independent of the pressure when the pressure is well above the molecular flow regime. In the transition regime, the heat transfer is proportional to the pressure and to the temperature difference. Principle of pressure measurement with a Pirani gauge Torr < P < 10 Torr At very low pressures, the heat transfer by conduction is negligible : vacuum for thermal insulation in cryogenics. LHC Cryodipole is
15 Basic Vacuum System V volume (m 3 ), F surface (m 2 ) P pressure (Pa), S pumping speed (m 3 /s) q specific outgassing rate (Pa m 3 /s/ m 2 ) P q F Stationary conditions (P is independent of volume) P = q F S Dynamic pressure V P = q F - S P t S V Solution (constant K depends on initial conditions) P(t)= K e - S V t + q F S The time constant of the pump down: V S To obtain a low pressure : Low outgassing rate of the surface, No leaks! Large pumping speed
16 Linear Vacuum System (Accelerators) x x+dx x Q(x) q 2S 2L 2S Gas flow: Q(x) [Pa m 3 s -1 ], specific outgassing rate: q(x) ) [Pa m s -1 ] Specific surface area per unit length: A[m], molecular conductance per unit length: c [m 4 s -1 ] dq dx = Aq and Q(x) = - c dp dx c d 2 P dx 2 = - A q
17 Boundary conditions for this configuration: dp By symmetry È = 0 and the pressure at x = 0 Î dx x =± L P(x = 0) = Q(0) = 2 A q L Q (x = 0) 2 S Ê One obtains a parabolic pressure distribution: P(x) = A qá Ë 2L x - x2 2 c + L ˆ S The average pressure, relevant for the beam: P av = 1 2L 2L Ú P( x)dx 0 Ê = A q L2 3c + L ˆ Á Ë S Note: the pressure is limited by the molecular conductance of the system. In spite of an increase of the pumping speed S, the average pressure is limited to the value : P av min = A q L2 3c Two important requirements : Large diameter of the vacuum chamber Close spacing of pumps
18 Time to form one monolayer t= Q 1 4 v sn Mono layer coverage: Q (~ molecules m -2 ) Molecular velocity v (m s -1 ) Gas density n (molecules m -3 ) Sticking probability s < 1 Time (h) Monolayer time 10-2 UHV becomes indispensable for surface analysis and for thin film technology -> Historically the main motivation to develop uhv techniques Pressure (Pa) Note: Area occupied per molecule A ~ 2 3r 2
19 Beam loss by Bremsstrahlung : UHV-Technology Beam Lifetime due to Vacuum - de dx = E X o Lifetime 1 t = - 1 N dn dt = cr X o W X o radiation length, c speed of particles and r = m 0M kt P is the density of the residual gas at the pressure P. Here W = log(e DE ) Represents the probability per radiation length to emit a photon with an energy larger than the energy acceptance of the machine so that the particle will be lost. The lifetime t = X o crw µ X o P For nitrogen or CO one finds typically tp = (Torr hours) Consequence : UHV is required for storage rings. Heavy molecules with short radiation length must be avoided.
Vacuum for Accelerators
 Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Vacuum for Accelerators Oswald Gröbner Baden, 22 September 2004 1) Introduction and some basics 2) Pumps used in accelerators 3) Desorption phenomena 4) Practical examples Oswald Gröbner, Retired from
Vacuum I. G. Franchetti CAS - Bilbao. 30/5/2011 G. Franchetti 1
 Vacuum I G. Franchetti CAS - Bilbao 30/5/2011 G. Franchetti 1 Index Introduction to Vacuum Vacuum and the Beam Flow Regimes Creating Vacuum 30/5/2011 G. Franchetti 2 Vacuum in accelerators All beam dynamics
Vacuum I G. Franchetti CAS - Bilbao 30/5/2011 G. Franchetti 1 Index Introduction to Vacuum Vacuum and the Beam Flow Regimes Creating Vacuum 30/5/2011 G. Franchetti 2 Vacuum in accelerators All beam dynamics
Conductance of an aperture
 n Vacuum φ A 4 How many molecules travel through A in a time t? P P A onductance of an aperture 3 q φa 3.64 0 ( T / M) na Volume of gas passing through A? Q Q Q / ΔP 3.64 ( T / M ) 3 dv/dt q/n 3.64 0 (
n Vacuum φ A 4 How many molecules travel through A in a time t? P P A onductance of an aperture 3 q φa 3.64 0 ( T / M) na Volume of gas passing through A? Q Q Q / ΔP 3.64 ( T / M ) 3 dv/dt q/n 3.64 0 (
MOLECULAR SURFACE PUMPING: CRYOPUMPING
 51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
51 MOLECULAR SURFACE PUMPING: CRYOPUMPING C. Benvenuti CERN, Geneva, Switzerland Abstract Weak van der Waals attractive forces may provide molecular gas pumping on surfaces cooled at sufficiently low temperatures.
Lecture 4. Ultrahigh Vacuum Science and Technology
 Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Lecture 4 Ultrahigh Vacuum Science and Technology Why do we need UHV? 1 Atmosphere = 760 torr; 1 torr = 133 Pa; N ~ 2.5 10 19 molecules/cm 3 Hertz-Knudsen equation p ZW 1/ 2 ( 2mk T) At p = 10-6 Torr it
Vacuum System of Synchrotron radiation sources
 3 rd ILSF Advanced School on Synchrotron Radiation and Its Applications September 14-16, 2013 Vacuum System of Synchrotron radiation sources Prepared by: Omid Seify, Vacuum group, ILSF project Institute
3 rd ILSF Advanced School on Synchrotron Radiation and Its Applications September 14-16, 2013 Vacuum System of Synchrotron radiation sources Prepared by: Omid Seify, Vacuum group, ILSF project Institute
Vacuum Science and Technology in Accelerators
 Vacuum Science and Technology in Accelerators Ron Reid Consultant ASTeC Vacuum Science Group (ron.reid@stfc.ac.uk) Lecture 1: 1 of 25 Aims To give a basic understanding of vacuum Underlying physical principles
Vacuum Science and Technology in Accelerators Ron Reid Consultant ASTeC Vacuum Science Group (ron.reid@stfc.ac.uk) Lecture 1: 1 of 25 Aims To give a basic understanding of vacuum Underlying physical principles
Conductance measurement of a conical tube and calculation of the pressure distribution
 Conductance measurement of a conical tube and calculation of the pressure distribution B. Mercier a Laboratoire de l Accélérateur Linéaire (LAL), Université Paris-Sud, Batiment 200, BP34, 9898 Orsay Cedex,
Conductance measurement of a conical tube and calculation of the pressure distribution B. Mercier a Laboratoire de l Accélérateur Linéaire (LAL), Université Paris-Sud, Batiment 200, BP34, 9898 Orsay Cedex,
Ⅰ. Vacuum Technology. 1. Introduction Vacuum empty in Greek Torricelli Vacuum 2 ) 760 mm. mbar 2 ) Enclosure. To pump. Thin Film Technology
 Ⅰ. Vacuum Technology. Introduction Vacuum empty in Greek 643 Torricelli Vacuum 76 mm Hg Enclosure F ma 76mmHg 76Torr atm (76cm) (3.6g / cm 3 ) (98.665cm/sec ).33 6 dynes/ cm.33bar.33 5 ascal( N / m ).4696
Ⅰ. Vacuum Technology. Introduction Vacuum empty in Greek 643 Torricelli Vacuum 76 mm Hg Enclosure F ma 76mmHg 76Torr atm (76cm) (3.6g / cm 3 ) (98.665cm/sec ).33 6 dynes/ cm.33bar.33 5 ascal( N / m ).4696
Homework: 13, 14, 18, 20, 24 (p )
 Homework: 13, 14, 18, 0, 4 (p. 531-53) 13. A sample of an ideal gas is taken through the cyclic process abca shown in the figure below; at point a, T=00 K. (a) How many moles of gas are in the sample?
Homework: 13, 14, 18, 0, 4 (p. 531-53) 13. A sample of an ideal gas is taken through the cyclic process abca shown in the figure below; at point a, T=00 K. (a) How many moles of gas are in the sample?
Repetition: Physical Deposition Processes
 Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Repetition: Physical Deposition Processes PVD (Physical Vapour Deposition) Evaporation Sputtering Diode-system Triode-system Magnetron-system ("balanced/unbalanced") Ion beam-system Ionplating DC-glow-discharge
Conductance of an aperture
 n Vacuum φ 4 How many molecules travel through in a time t? P P onductance of an aperture q φ.64 0 ( T / M) n Volume of gas passing through? Q Q dv/dt q/n.64 0 ( T / M) Q P Q / ΔP.64 (T / M ) l/s.64 0
n Vacuum φ 4 How many molecules travel through in a time t? P P onductance of an aperture q φ.64 0 ( T / M) n Volume of gas passing through? Q Q dv/dt q/n.64 0 ( T / M) Q P Q / ΔP.64 (T / M ) l/s.64 0
K n. III. Gas flow. 1. The nature of the gas : Knudsen s number. 2. Relative flow : Reynold s number R = ( dimensionless )
 III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
III. Gas flow. The nature of the gas : Knudsen s number K n λ d 2. Relative flow : U ρ d η U : stream velocity ρ : mass density Reynold s number R ( dimensionless ) 3. Flow regions - turbulent : R > 2200
PV = n R T = N k T. Measured from Vacuum = 0 Gauge Pressure = Vacuum - Atmospheric Atmospheric = 14.7 lbs/sq in = 10 5 N/m
 PV = n R T = N k T P is the Absolute pressure Measured from Vacuum = 0 Gauge Pressure = Vacuum - Atmospheric Atmospheric = 14.7 lbs/sq in = 10 5 N/m V is the volume of the system in m 3 often the system
PV = n R T = N k T P is the Absolute pressure Measured from Vacuum = 0 Gauge Pressure = Vacuum - Atmospheric Atmospheric = 14.7 lbs/sq in = 10 5 N/m V is the volume of the system in m 3 often the system
Lecture 24. Ideal Gas Law and Kinetic Theory
 Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Particle Accelerators
 Experimental Methods of Particle Physics Particle Accelerators Andreas Streun, PSI andreas.streun@psi.ch https://ados.web.psi.ch/empp-streun Andreas Streun, PSI 1 Particle Accelerators 1. Introduction
Experimental Methods of Particle Physics Particle Accelerators Andreas Streun, PSI andreas.streun@psi.ch https://ados.web.psi.ch/empp-streun Andreas Streun, PSI 1 Particle Accelerators 1. Introduction
Gases. T boil, K. 11 gaseous elements. Rare gases. He, Ne, Ar, Kr, Xe, Rn Diatomic gaseous elements H 2, N 2, O 2, F 2, Cl 2
 Gases Gas T boil, K Rare gases 11 gaseous elements He, Ne, Ar, Kr, Xe, Rn 165 Rn 211 N 2 O 2 77 F 2 90 85 Diatomic gaseous elements Cl 2 238 H 2, N 2, O 2, F 2, Cl 2 H 2 He Ne Ar Kr Xe 20 4.4 27 87 120
Gases Gas T boil, K Rare gases 11 gaseous elements He, Ne, Ar, Kr, Xe, Rn 165 Rn 211 N 2 O 2 77 F 2 90 85 Diatomic gaseous elements Cl 2 238 H 2, N 2, O 2, F 2, Cl 2 H 2 He Ne Ar Kr Xe 20 4.4 27 87 120
Chapter 18 Thermal Properties of Matter
 Chapter 18 Thermal Properties of Matter In this section we define the thermodynamic state variables and their relationship to each other, called the equation of state. The system of interest (most of the
Chapter 18 Thermal Properties of Matter In this section we define the thermodynamic state variables and their relationship to each other, called the equation of state. The system of interest (most of the
Rough (low) vacuum : Medium vacuum : High vacuum (HV) : Ultrahigh vacuum (UHV) :
 4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
4. UHV & Effects of Gas Pressure 4.1 What is Ultra-high Vacuum (UHV)? 4.2 Why is UHV required for Surface Studies? 4.3 Exercises - Effects of Gas Pressure Vacuum technology has advanced considerably over
Physical Vapor Deposition
 Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
Physical Vapor Deposition EVAPORATION SPUTTERING Typically used for metallization of semiconductors. Both Evaporation & Sputtering are done in vacuum environments. Typically: y Evaporation Pressures are
C p = (3/2) R + R, KE change + work. Also Independent of T
 Heat Capacity Summary for Ideal Gases: C v = (3/2) R, KE change only. Note, C v independent of T. C p = (3/2) R + R, KE change + work. Also Independent of T C p /C v = [(5/2)R]/[(3/2)R] = 5/3 C p /C v
Heat Capacity Summary for Ideal Gases: C v = (3/2) R, KE change only. Note, C v independent of T. C p = (3/2) R + R, KE change + work. Also Independent of T C p /C v = [(5/2)R]/[(3/2)R] = 5/3 C p /C v
( KS A ) (1) , vapour, vapor (USA) , saturation vapour pressure. , standard reference conditions for gases. , degree of saturation
 ( KS A 3014-91 ) (1), standard reference conditions for gases 0, 101325 Pa (1 =760mmHg ), vacuum, low ( rough ) vacuum 100Pa, medium vacuum 100 01 Pa, high vacuum 01 10 5 Pa, ultra high vacuum ( UHV )
( KS A 3014-91 ) (1), standard reference conditions for gases 0, 101325 Pa (1 =760mmHg ), vacuum, low ( rough ) vacuum 100Pa, medium vacuum 100 01 Pa, high vacuum 01 10 5 Pa, ultra high vacuum ( UHV )
Lecture 24. Ideal Gas Law and Kinetic Theory
 Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Lecture 4 Ideal Gas Law and Kinetic Theory Today s Topics: Ideal Gas Law Kinetic Theory of Gases Phase equilibria and phase diagrams Ideal Gas Law An ideal gas is an idealized model for real gases that
Gases. Properties of Gases Kinetic Molecular Theory of Gases Pressure Boyle s and Charles Law The Ideal Gas Law Gas reactions Partial pressures.
 Gases Properties of Gases Kinetic Molecular Theory of Gases Pressure Boyle s and Charles Law The Ideal Gas Law Gas reactions Partial pressures Gases Properties of Gases All elements will form a gas at
Gases Properties of Gases Kinetic Molecular Theory of Gases Pressure Boyle s and Charles Law The Ideal Gas Law Gas reactions Partial pressures Gases Properties of Gases All elements will form a gas at
Lecture 3 Vacuum Science and Technology
 Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Statistical Mechanics
 Statistical Mechanics Newton's laws in principle tell us how anything works But in a system with many particles, the actual computations can become complicated. We will therefore be happy to get some 'average'
Statistical Mechanics Newton's laws in principle tell us how anything works But in a system with many particles, the actual computations can become complicated. We will therefore be happy to get some 'average'
Vacuum. Residual pressure can thwart the best cryogenic design. Each gas molecule collision carries ~kt from the hot exterior to the cold interior.
 Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
Vacuum Residual pressure can thwart the best cryogenic design Each gas molecule collision carries ~kt from the hot exterior to the cold interior. 1 millitorr = 3.5x10¹³/cm³ Gas atoms leaving the hot surfaces
Speed Distribution at CONSTANT Temperature is given by the Maxwell Boltzmann Speed Distribution
 Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature
 Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature Any physical property that changes with temperature is called a thermometric property and can be used to measure
Chapter 17 Temperature & Kinetic Theory of Gases 1. Thermal Equilibrium and Temperature Any physical property that changes with temperature is called a thermometric property and can be used to measure
LECTURE 5 SUMMARY OF KEY IDEAS
 LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
LECTURE 5 SUMMARY OF KEY IDEAS Etching is a processing step following lithography: it transfers a circuit image from the photoresist to materials form which devices are made or to hard masking or sacrificial
Vacuum System in Accelerator
 Vacuum System in Accelerator -- geometrical structure effects on the pumping delay time -- Y. Saito, KEK, Tsukuba, Japan N. Matuda, Tokyo Denki Univ., Tokyo, Japan 1. Required vacuum condition; what kind
Vacuum System in Accelerator -- geometrical structure effects on the pumping delay time -- Y. Saito, KEK, Tsukuba, Japan N. Matuda, Tokyo Denki Univ., Tokyo, Japan 1. Required vacuum condition; what kind
12/21/2014 7:39 PM. Chapter 2. Energy and the 1st Law of Thermodynamics. Dr. Mohammad Suliman Abuhaiba, PE
 Chapter 2 Energy and the 1st Law of Thermodynamics 1 2 Homework Assignment # 2 Problems: 1, 7, 14, 20, 30, 36, 42, 49, 56 Design and open end problem: 2.1D Due Monday 22/12/2014 3 Work and Kinetic Energy
Chapter 2 Energy and the 1st Law of Thermodynamics 1 2 Homework Assignment # 2 Problems: 1, 7, 14, 20, 30, 36, 42, 49, 56 Design and open end problem: 2.1D Due Monday 22/12/2014 3 Work and Kinetic Energy
KINETIC THEORY. was the original mean square velocity of the gas. (d) will be different on the top wall and bottom wall of the vessel.
 Chapter Thirteen KINETIC THEORY MCQ I 13.1 A cubic vessel (with faces horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of 500m s 1
Chapter Thirteen KINETIC THEORY MCQ I 13.1 A cubic vessel (with faces horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of 500m s 1
Vacuum Technology for Particle Accelerators
 Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
Vacuum Technology for Particle Accelerators (Max IV laboratory) CERN Accelerator School: Introduction to Accelerator Physics 8 th October 2016, Budapest, Hungary 1 Contents What is vacuum and why do we
First Name: Last Name: Section: 1 December 20, 2004 Physics 201 FINAL EXAM
 First Name: Last Name: Section: 1 December 20, 2004 Physics 201 FINAL EXAM Print your name and section clearly on all nine pages. (If you do not know your section number, write your TA s name.) Show all
First Name: Last Name: Section: 1 December 20, 2004 Physics 201 FINAL EXAM Print your name and section clearly on all nine pages. (If you do not know your section number, write your TA s name.) Show all
Statistical Physics. Problem Set 4: Kinetic Theory
 Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
Statistical Physics xford hysics Second year physics course Dr A. A. Schekochihin and Prof. A. Boothroyd (with thanks to Prof. S. J. Blundell) Problem Set 4: Kinetic Theory PROBLEM SET 4: Collisions and
Downloaded from
 Chapter 13 (Kinetic Theory) Q1. A cubic vessel (with face horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of500 ms in vertical direction.
Chapter 13 (Kinetic Theory) Q1. A cubic vessel (with face horizontal + vertical) contains an ideal gas at NTP. The vessel is being carried by a rocket which is moving at a speed of500 ms in vertical direction.
Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources
 CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
CERN-ACC-2014-0259 marton.ady@cern.ch Monte Carlo Simulations of Synchrotron Radiation and Vacuum Performance of the MAX IV Light Sources M. Ady, R. Kersevan CERN, Geneva, Switzerland M. Grabski MAX IV,
Vacuum at the ESRF. current activities that benefit from simulation models
 Vacuum at the ESRF current activities that benefit from simulation models H.P. Marques - 64th IUVSTA Workshop Leinsweiler 2011 Vacuum at the ESRF Overview of the ESRF Vacuum group activities MC simulation
Vacuum at the ESRF current activities that benefit from simulation models H.P. Marques - 64th IUVSTA Workshop Leinsweiler 2011 Vacuum at the ESRF Overview of the ESRF Vacuum group activities MC simulation
Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov
 Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov 18-23 2015 MSU Physics 231 Fall 2015 1 homework 3 rd midterm final Thursday 8-10 pm makeup Friday final 9-11 am MSU
Physics 231 Topic 12: Temperature, Thermal Expansion, and Ideal Gases Alex Brown Nov 18-23 2015 MSU Physics 231 Fall 2015 1 homework 3 rd midterm final Thursday 8-10 pm makeup Friday final 9-11 am MSU
Vacuum II. G. Franchetti CAS - Bilbao. 30/5/2011 G. Franchetti 1
 Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
Vacuum II G. Franchetti CAS Bilbao 30/5/2011 G. Franchetti 1 Index Creating Vacuum (continuation) Measuring Vacuum Partial Pressure Measurements 30/5/2011 G. Franchetti 2 Laminar flow Cold surface Diffusion
Vacuum Technology and film growth. Diffusion Resistor
 Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Vacuum Technology and film growth Poly Gate pmos Polycrystaline Silicon Source Gate p-channel Metal-Oxide-Semiconductor (MOSFET) Drain polysilicon n-si ion-implanted Diffusion Resistor Poly Si Resistor
Example Problems: 1.) What is the partial pressure of: Total moles = 13.2 moles 5.0 mol A 7.0 mol B 1.2 mol C Total Pressure = 3.
 5.6 Dalton s Law of Partial Pressures Dalton s Law of Partial Pressure; The total pressure of a gas is the sum of all its parts. P total = P 1 + P + P 3 + P n Pressures are directly related to moles: n
5.6 Dalton s Law of Partial Pressures Dalton s Law of Partial Pressure; The total pressure of a gas is the sum of all its parts. P total = P 1 + P + P 3 + P n Pressures are directly related to moles: n
Study of Distributed Ion-Pumps in CESR 1
 Study of Distributed Ion-Pumps in CESR 1 Yulin Li, Roberto Kersevan, Nariman Mistry Laboratory of Nuclear Studies, Cornell University Ithaca, NY 153-001 Abstract It is desirable to reduce anode voltage
Study of Distributed Ion-Pumps in CESR 1 Yulin Li, Roberto Kersevan, Nariman Mistry Laboratory of Nuclear Studies, Cornell University Ithaca, NY 153-001 Abstract It is desirable to reduce anode voltage
Vacuum techniques (down to 1 K)
 Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Vacuum techniques (down to 1 K) For isolation (deep Knudsen regime) liquid helium dewar / inner vacuum jacket Leak testing at level 10-11 Pa m3/s (10-10 mbar l/s) liquid helium dewar & transfer syphon
Experience from the LEP Vacuum System
 Experience from the LEP Vacuum System O. Gröbner CERN, LHC-VAC Workshop on an e + e - Ring at VLHC ITT, 9-11 March 2001 3/4/01 O. Gröbner, CERN-LHC/VAC References 1) LEP Design Report, Vol.II, CERN-LEP/84-01,
Experience from the LEP Vacuum System O. Gröbner CERN, LHC-VAC Workshop on an e + e - Ring at VLHC ITT, 9-11 March 2001 3/4/01 O. Gröbner, CERN-LHC/VAC References 1) LEP Design Report, Vol.II, CERN-LEP/84-01,
λ = 5 10 p (i.e. 5 cm at 10 3 Torr, or ( )
 Getter pumping C. Benvenuti R&B Energy Research, Geneva, Switzerland Abstract A surface may provide a useful pumping action when able to retain adsorbed gas molecules for the duration of a given experiment.
Getter pumping C. Benvenuti R&B Energy Research, Geneva, Switzerland Abstract A surface may provide a useful pumping action when able to retain adsorbed gas molecules for the duration of a given experiment.
Theory English (Official)
 Q3-1 Large Hadron Collider (10 points) Please read the general instructions in the separate envelope before you start this problem. In this task, the physics of the particle accelerator LHC (Large Hadron
Q3-1 Large Hadron Collider (10 points) Please read the general instructions in the separate envelope before you start this problem. In this task, the physics of the particle accelerator LHC (Large Hadron
Speed Distribution at CONSTANT Temperature is given by the Maxwell Boltzmann Speed Distribution
 Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Temperature ~ Average KE of each particle Particles have different speeds Gas Particles are in constant RANDOM motion Average KE of each particle is: 3/2 kt Pressure is due to momentum transfer Speed Distribution
Preliminary Examination - Day 2 Friday, August 12, 2016
 UNL - Department of Physics and Astronomy Preliminary Examination - Day Friday, August 1, 016 This test covers the topics of Thermodynamics and Statistical Mechanics (Topic 1) and Mechanics (Topic ). Each
UNL - Department of Physics and Astronomy Preliminary Examination - Day Friday, August 1, 016 This test covers the topics of Thermodynamics and Statistical Mechanics (Topic 1) and Mechanics (Topic ). Each
Preliminary Examination - Day 2 August 16, 2013
 UNL - Department of Physics and Astronomy Preliminary Examination - Day August 16, 13 This test covers the topics of Quantum Mechanics (Topic 1) and Thermodynamics and Statistical Mechanics (Topic ). Each
UNL - Department of Physics and Astronomy Preliminary Examination - Day August 16, 13 This test covers the topics of Quantum Mechanics (Topic 1) and Thermodynamics and Statistical Mechanics (Topic ). Each
KINETIC MOLECULAR DESCRIPTION OF THE STATES OF MATTER
 KINETIC MOLECULAR DESCRIPTION OF THE STATES OF MATTER CHAPTER 9 The Gaseous State CHAPTER 10 Solids, Liquids, and Phase Transitions CHAPTER 11 Solutions 392 Gas Liquid Solid 9 THE GASEOUS STATE 9.1 The
KINETIC MOLECULAR DESCRIPTION OF THE STATES OF MATTER CHAPTER 9 The Gaseous State CHAPTER 10 Solids, Liquids, and Phase Transitions CHAPTER 11 Solutions 392 Gas Liquid Solid 9 THE GASEOUS STATE 9.1 The
ε tran ε tran = nrt = 2 3 N ε tran = 2 3 nn A ε tran nn A nr ε tran = 2 N A i.e. T = R ε tran = 2
 F1 (a) Since the ideal gas equation of state is PV = nrt, we can equate the right-hand sides of both these equations (i.e. with PV = 2 3 N ε tran )and write: nrt = 2 3 N ε tran = 2 3 nn A ε tran i.e. T
F1 (a) Since the ideal gas equation of state is PV = nrt, we can equate the right-hand sides of both these equations (i.e. with PV = 2 3 N ε tran )and write: nrt = 2 3 N ε tran = 2 3 nn A ε tran i.e. T
Similarities and differences:
 How does the system reach equilibrium? I./9 Chemical equilibrium I./ Equilibrium electrochemistry III./ Molecules in motion physical processes, non-reactive systems III./5-7 Reaction rate, mechanism, molecular
How does the system reach equilibrium? I./9 Chemical equilibrium I./ Equilibrium electrochemistry III./ Molecules in motion physical processes, non-reactive systems III./5-7 Reaction rate, mechanism, molecular
LECTURE 18. Beam loss and beam emittance growth. Mechanisms for beam loss. Mechanisms for emittance growth and beam loss Beam lifetime:
 LCTUR 18 Beam loss and beam emittance growth Mechanisms for emittance growth and beam loss Beam lifetime: from residual gas interactions; Touschek effect; quantum lifetimes in electron machines; Beam lifetime
LCTUR 18 Beam loss and beam emittance growth Mechanisms for emittance growth and beam loss Beam lifetime: from residual gas interactions; Touschek effect; quantum lifetimes in electron machines; Beam lifetime
Lecture 18 Molecular Motion and Kinetic Energy
 Physical Principles in Biology Biology 3550 Fall 2017 Lecture 18 Molecular Motion and Kinetic Energy Monday, 2 October c David P. Goldenberg University of Utah goldenberg@biology.utah.edu Fick s First
Physical Principles in Biology Biology 3550 Fall 2017 Lecture 18 Molecular Motion and Kinetic Energy Monday, 2 October c David P. Goldenberg University of Utah goldenberg@biology.utah.edu Fick s First
Industrial Surfaces Behaviour Related to the Adsorption and Desorption of Hydrogen at Cryogenic Temperature
 EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 375 Industrial Surfaces Behaviour Related to the Adsorption and Desorption
EUROPEAN ORGANIZATION FOR NUCLEAR RESEARCH European Laboratory for Particle Physics Large Hadron Collider Project LHC Project Report 375 Industrial Surfaces Behaviour Related to the Adsorption and Desorption
Ay Fall 2004 Lecture 6 (given by Tony Travouillon)
 Ay 122 - Fall 2004 Lecture 6 (given by Tony Travouillon) Stellar atmospheres, classification of stellar spectra (Many slides c/o Phil Armitage) Formation of spectral lines: 1.excitation Two key questions:
Ay 122 - Fall 2004 Lecture 6 (given by Tony Travouillon) Stellar atmospheres, classification of stellar spectra (Many slides c/o Phil Armitage) Formation of spectral lines: 1.excitation Two key questions:
The Vacuum Case for KATRIN
 The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
The Vacuum Case for KATRIN Institute of Nuclear Physics, Forschungszentrum Karlsruhe,Germany, for the KATRIN Collaboration Lutz.Bornschein@ik.fzk.de The vacuum requirements of the KATRIN experiment have
Diffusion and Adsorption in porous media. Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad
 Diffusion and Adsorption in porous media Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Devices used to Measure Diffusion in Porous Solids Modes of transport in
Diffusion and Adsorption in porous media Ali Ahmadpour Chemical Eng. Dept. Ferdowsi University of Mashhad Contents Introduction Devices used to Measure Diffusion in Porous Solids Modes of transport in
Chemical Thermodynamics : Georg Duesberg
 The Properties of Gases Kinetic gas theory Maxwell Boltzman distribution, Collisions Real (non-ideal) gases fugacity, Joule Thomson effect Mixtures of gases Entropy, Chemical Potential Liquid Solutions
The Properties of Gases Kinetic gas theory Maxwell Boltzman distribution, Collisions Real (non-ideal) gases fugacity, Joule Thomson effect Mixtures of gases Entropy, Chemical Potential Liquid Solutions
Molecular Motion and Gas Laws
 Molecular Motion and Gas Laws What is the connection between the motion of molecules (F = ma and K = mv 2 /2) and the thermodynamics of gases (pv = nrt and U = 3nRT/2)? In this lab, you will discover how
Molecular Motion and Gas Laws What is the connection between the motion of molecules (F = ma and K = mv 2 /2) and the thermodynamics of gases (pv = nrt and U = 3nRT/2)? In this lab, you will discover how
Kinetic Theory. 84 minutes. 62 marks. theonlinephysicstutor.com. facebook.com/theonlinephysicstutor. Name: Class: Date: Time: Marks: Comments:
 Kinetic Theory Name: Class: Date: Time: 84 minutes Marks: 62 marks Comments: Page 1 of 19 1 Which one of the following is not an assumption about the properties of particles in the simple kinetic theory?
Kinetic Theory Name: Class: Date: Time: 84 minutes Marks: 62 marks Comments: Page 1 of 19 1 Which one of the following is not an assumption about the properties of particles in the simple kinetic theory?
KINETIC THEORY OF GASES
 KINETIC THEORY OF GASES VERY SHORT ANSWER TYPE QUESTIONS ( MARK). Write two condition when real gases obey the ideal gas equation ( nrt). n number of mole.. If the number of molecule in a container is
KINETIC THEORY OF GASES VERY SHORT ANSWER TYPE QUESTIONS ( MARK). Write two condition when real gases obey the ideal gas equation ( nrt). n number of mole.. If the number of molecule in a container is
BCIT Fall Chem Exam #1
 BCIT Fall 2012 Chem 3615 Exam #1 Name: Attempt all questions in this exam. Read each question carefully and give a complete answer in the space provided. Part marks given for wrong answers with partially
BCIT Fall 2012 Chem 3615 Exam #1 Name: Attempt all questions in this exam. Read each question carefully and give a complete answer in the space provided. Part marks given for wrong answers with partially
Graduate Written Examination Spring 2014 Part I Thursday, January 16th, :00am to 1:00pm
 Graduate Written Examination Spring 2014 Part I Thursday, January 16th, 2014 9:00am to 1:00pm University of Minnesota School of Physics and Astronomy Examination Instructions Part 1 of this exam consists
Graduate Written Examination Spring 2014 Part I Thursday, January 16th, 2014 9:00am to 1:00pm University of Minnesota School of Physics and Astronomy Examination Instructions Part 1 of this exam consists
PHYSICS - CLUTCH CH 19: KINETIC THEORY OF IDEAL GASSES.
 !! www.clutchprep.com CONCEPT: ATOMIC VIEW OF AN IDEAL GAS Remember! A gas is a type of fluid whose volume can change to fill a container - What makes a gas ideal? An IDEAL GAS is a gas whose particles
!! www.clutchprep.com CONCEPT: ATOMIC VIEW OF AN IDEAL GAS Remember! A gas is a type of fluid whose volume can change to fill a container - What makes a gas ideal? An IDEAL GAS is a gas whose particles
Accelerator Physics Weak Focussing. A. Bogacz, G. A. Krafft, and T. Zolkin Jefferson Lab Colorado State University Lecture 2
 Accelerator Physics Weak Focussing A. Bogacz, G. A. Krafft, and T. Zolkin Jefferson Lab Colorado State University Lecture 2 Betatrons 25 MeV electron accelerator with its inventor: Don Kerst. The earliest
Accelerator Physics Weak Focussing A. Bogacz, G. A. Krafft, and T. Zolkin Jefferson Lab Colorado State University Lecture 2 Betatrons 25 MeV electron accelerator with its inventor: Don Kerst. The earliest
Chapter 14 Kinetic Theory
 Chapter 14 Kinetic Theory Kinetic Theory of Gases A remarkable triumph of molecular theory was showing that the macroscopic properties of an ideal gas are related to the molecular properties. This is the
Chapter 14 Kinetic Theory Kinetic Theory of Gases A remarkable triumph of molecular theory was showing that the macroscopic properties of an ideal gas are related to the molecular properties. This is the
Tutorial on Gas Flow, Conductance, Pressure Profiles R. Kersevan, TE/VSC-VSM CERN, Geneva
 JUA 207, Archamps, France R. Kersevan, TE/VC-VM CERN, Geneva Content: Concepts of gas flow, conductance, pressure profile as relevant to the design of the vacuum system of modern accelerators; A quick
JUA 207, Archamps, France R. Kersevan, TE/VC-VM CERN, Geneva Content: Concepts of gas flow, conductance, pressure profile as relevant to the design of the vacuum system of modern accelerators; A quick
Chapter 15 Thermal Properties of Matter
 Chapter 15 Thermal Properties of Matter To understand the mole and Avogadro's number. To understand equations of state. To study the kinetic theory of ideal gas. To understand heat capacity. To learn and
Chapter 15 Thermal Properties of Matter To understand the mole and Avogadro's number. To understand equations of state. To study the kinetic theory of ideal gas. To understand heat capacity. To learn and
Accelerator Physics Weak Focusing. S. A. Bogacz, G. A. Krafft, S. DeSilva, R. Gamage Jefferson Lab Old Dominion University Lecture 2
 Accelerator Physics Weak Focusing S. A. Bogacz, G. A. Krafft, S. DeSilva, R. Gamage Jefferson Lab Old Dominion University Lecture 2 Betatrons 25 MeV electron accelerator with its inventor: Don Kerst. The
Accelerator Physics Weak Focusing S. A. Bogacz, G. A. Krafft, S. DeSilva, R. Gamage Jefferson Lab Old Dominion University Lecture 2 Betatrons 25 MeV electron accelerator with its inventor: Don Kerst. The
Thermodynamics and Statistical Physics Exam
 Thermodynamics and Statistical Physics Exam You may use your textbook (Thermal Physics by Schroeder) and a calculator. 1. Short questions. No calculation needed. (a) Two rooms A and B in a building are
Thermodynamics and Statistical Physics Exam You may use your textbook (Thermal Physics by Schroeder) and a calculator. 1. Short questions. No calculation needed. (a) Two rooms A and B in a building are
Note. Performance limitations of circular colliders: head-on collisions
 2014-08-28 m.koratzinos@cern.ch Note Performance limitations of circular colliders: head-on collisions M. Koratzinos University of Geneva, Switzerland Keywords: luminosity, circular, collider, optimization,
2014-08-28 m.koratzinos@cern.ch Note Performance limitations of circular colliders: head-on collisions M. Koratzinos University of Geneva, Switzerland Keywords: luminosity, circular, collider, optimization,
Relativity II. Home Work Solutions
 Chapter 2 Relativity II. Home Work Solutions 2.1 Problem 2.4 (In the text book) A charged particle moves along a straight line in a uniform electric field E with a speed v. If the motion and the electric
Chapter 2 Relativity II. Home Work Solutions 2.1 Problem 2.4 (In the text book) A charged particle moves along a straight line in a uniform electric field E with a speed v. If the motion and the electric
D. Brandt, CERN. CAS Frascati 2008 Accelerators for Newcomers D. Brandt 1
 Accelerators for Newcomers D. Brandt, CERN D. Brandt 1 Why this Introduction? During this school, you will learn about beam dynamics in a rigorous way but some of you are completely new to the field of
Accelerators for Newcomers D. Brandt, CERN D. Brandt 1 Why this Introduction? During this school, you will learn about beam dynamics in a rigorous way but some of you are completely new to the field of
Fundamental Concepts of Particle Accelerators V : Future of the High Energy Accelerators. Koji TAKATA KEK. Accelerator Course, Sokendai
 .... Fundamental Concepts of Particle Accelerators V : Future of the High Energy Accelerators Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator Course, Sokendai
.... Fundamental Concepts of Particle Accelerators V : Future of the High Energy Accelerators Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator Course, Sokendai
UNIVERSITY OF SOUTHAMPTON
 UNIVERSITY OF SOUTHAMPTON PHYS1013W1 SEMESTER 2 EXAMINATION 2014-2015 ENERGY AND MATTER Duration: 120 MINS (2 hours) This paper contains 8 questions. Answers to Section A and Section B must be in separate
UNIVERSITY OF SOUTHAMPTON PHYS1013W1 SEMESTER 2 EXAMINATION 2014-2015 ENERGY AND MATTER Duration: 120 MINS (2 hours) This paper contains 8 questions. Answers to Section A and Section B must be in separate
ADIABATIC PROCESS Q = 0
 THE KINETIC THEORY OF GASES Mono-atomic Fig.1 1 3 Average kinetic energy of a single particle Fig.2 INTERNAL ENERGY U and EQUATION OF STATE For a mono-atomic gas, we will assume that the total energy
THE KINETIC THEORY OF GASES Mono-atomic Fig.1 1 3 Average kinetic energy of a single particle Fig.2 INTERNAL ENERGY U and EQUATION OF STATE For a mono-atomic gas, we will assume that the total energy
Rate of Heating and Cooling
 Rate of Heating and Cooling 35 T [ o C] Example: Heating and cooling of Water E 30 Cooling S 25 Heating exponential decay 20 0 100 200 300 400 t [sec] Newton s Law of Cooling T S > T E : System S cools
Rate of Heating and Cooling 35 T [ o C] Example: Heating and cooling of Water E 30 Cooling S 25 Heating exponential decay 20 0 100 200 300 400 t [sec] Newton s Law of Cooling T S > T E : System S cools
The Gaseous State of Matter
 The Gaseous State of Matter Chapter 12 Hein and Arena Version 1.1 Dr. Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Company The Kinetic- Molecular Theory 2 The Kinetic-Molecular
The Gaseous State of Matter Chapter 12 Hein and Arena Version 1.1 Dr. Eugene Passer Chemistry Department Bronx Community 1 College John Wiley and Company The Kinetic- Molecular Theory 2 The Kinetic-Molecular
Kinetic theory of the ideal gas
 Appendix H Kinetic theory of the ideal gas This Appendix contains sketchy notes, summarizing the main results of elementary kinetic theory. The students who are not familiar with these topics should refer
Appendix H Kinetic theory of the ideal gas This Appendix contains sketchy notes, summarizing the main results of elementary kinetic theory. The students who are not familiar with these topics should refer
- Apply closed system energy balances, observe sign convention for work and heat transfer.
 CHAPTER : ENERGY AND THE FIRST LAW OF THERMODYNAMICS Objectives: - In this chapter we discuss energy and develop equations for applying the principle of conservation of energy. Learning Outcomes: - Demonstrate
CHAPTER : ENERGY AND THE FIRST LAW OF THERMODYNAMICS Objectives: - In this chapter we discuss energy and develop equations for applying the principle of conservation of energy. Learning Outcomes: - Demonstrate
BALKAN PHYSICS LETTERS Bogazici University Press 15 November 2016 BPL, 24, , pp , (2016)
 139 BALKAN PHYSICS LETTERS Bogazici University Press 15 November 2016 BPL, 24, 241016, pp. 139 145, (2016) VACUUM STUDIES ON ELECTRON GUN AND INJECTOR LINE OF TARLA FACILITY E. COŞGUN, E.P. DEMİRCİ, Ö.
139 BALKAN PHYSICS LETTERS Bogazici University Press 15 November 2016 BPL, 24, 241016, pp. 139 145, (2016) VACUUM STUDIES ON ELECTRON GUN AND INJECTOR LINE OF TARLA FACILITY E. COŞGUN, E.P. DEMİRCİ, Ö.
and another with a peak frequency ω 2
 Physics Qualifying Examination Part I 7-Minute Questions September 13, 2014 1. A sealed container is divided into two volumes by a moveable piston. There are N A molecules on one side and N B molecules
Physics Qualifying Examination Part I 7-Minute Questions September 13, 2014 1. A sealed container is divided into two volumes by a moveable piston. There are N A molecules on one side and N B molecules
VACUUM PUMPING METHODS
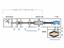 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
Earlier Lecture. In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide.
 41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
41 1 Earlier Lecture In the earlier lecture, we have seen non metallic sensors like Silicon diode, Cernox and Ruthenium Oxide. Silicon diodes have negligible i 2 R losses. Cernox RTDs offer high response
Introduction to Accelerators
 Introduction to Accelerators D. Brandt, CERN CAS Platja d Aro 2006 Introduction to Accelerators D. Brandt 1 Why an Introduction? The time where each accelerator sector was working alone in its corner is
Introduction to Accelerators D. Brandt, CERN CAS Platja d Aro 2006 Introduction to Accelerators D. Brandt 1 Why an Introduction? The time where each accelerator sector was working alone in its corner is
Lecture 25 Goals: Chapter 18 Understand the molecular basis for pressure and the idealgas
 Lecture 5 Goals: Chapter 18 Understand the molecular basis for pressure and the idealgas law. redict the molar specific heats of gases and solids. Understand how heat is transferred via molecular collisions
Lecture 5 Goals: Chapter 18 Understand the molecular basis for pressure and the idealgas law. redict the molar specific heats of gases and solids. Understand how heat is transferred via molecular collisions
A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC
 A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC A. Rossi, G. Rumolo and F. Ziermann, CERN, Geneva, Switzerland Abstract The LHC experimental regions (ATLAS, ALICE, CMS and LHC
A SIMULATION STUDY OF THE ELECTRON CLOUD IN THE EXPERIMENTAL REGIONS OF LHC A. Rossi, G. Rumolo and F. Ziermann, CERN, Geneva, Switzerland Abstract The LHC experimental regions (ATLAS, ALICE, CMS and LHC
Fundamentals of Accelerators
 Fundamentals of Accelerators - 2015 Lecture 9 - Synchrotron Radiation William A. Barletta Director, Dept. of Physics, MIT Economics Faculty, University of Ljubljana Photon energy to frequency conversion
Fundamentals of Accelerators - 2015 Lecture 9 - Synchrotron Radiation William A. Barletta Director, Dept. of Physics, MIT Economics Faculty, University of Ljubljana Photon energy to frequency conversion
Contents. LC : Linear Collider. µ-µ Collider. Laser-Plasma Wave Accelerator. Livingston Chart 6 References
 .... Fundamental Concepts of Particle Accelerators V : Future of the High Energy Accelerators VI : References Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator
.... Fundamental Concepts of Particle Accelerators V : Future of the High Energy Accelerators VI : References Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator
Chapter 10. Chapter 10 Gases
 Chapter 10 Gases Earth is surrounded by a layer of gaseous molecules - the atmosphere - extending out to about 50 km. 10.1 Characteristics of Gases Gases low density; compressible volume and shape of container
Chapter 10 Gases Earth is surrounded by a layer of gaseous molecules - the atmosphere - extending out to about 50 km. 10.1 Characteristics of Gases Gases low density; compressible volume and shape of container
Chapter 12. Temperature and Heat. continued
 Chapter 12 Temperature and Heat continued 12.3 The Ideal Gas Law THE IDEAL GAS LAW The absolute pressure of an ideal gas is directly proportional to the Kelvin temperature and the number of moles (n) of
Chapter 12 Temperature and Heat continued 12.3 The Ideal Gas Law THE IDEAL GAS LAW The absolute pressure of an ideal gas is directly proportional to the Kelvin temperature and the number of moles (n) of
Gas Density. Standard T & P (STP) 10/29/2011. At STP, 1 mol of any ideal gas occupies 22.4 L. T = 273 K (0 o C) P = 1 atm = kpa = 1.
 Standard T & P (STP) T = 73 K (0 o C) P = 1 atm = 101.35 kpa = 1.0135 bar At STP, 1 mol of any ideal gas occupies.4 L.4 L Gas Density We can use PV = nrt to determine the density of gases. What are the
Standard T & P (STP) T = 73 K (0 o C) P = 1 atm = 101.35 kpa = 1.0135 bar At STP, 1 mol of any ideal gas occupies.4 L.4 L Gas Density We can use PV = nrt to determine the density of gases. What are the
Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths
 Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths 4.1 The Natural Line Shape 4.2 Collisional Broadening 4.3 Doppler Broadening 4.4 Einstein Treatment of Stimulated Processes Width
Quantum Electronics/Laser Physics Chapter 4 Line Shapes and Line Widths 4.1 The Natural Line Shape 4.2 Collisional Broadening 4.3 Doppler Broadening 4.4 Einstein Treatment of Stimulated Processes Width
Gases and Kinetic Theory
 Gases and Kinetic Theory Chemistry 35 Fall 2000 Gases One of the four states of matter Simplest to understand both physically and chemically Gas Properties Low density Fluid Can be defined by their: 1.
Gases and Kinetic Theory Chemistry 35 Fall 2000 Gases One of the four states of matter Simplest to understand both physically and chemically Gas Properties Low density Fluid Can be defined by their: 1.
Fundamental Concepts of Particle Accelerators V: Future of the High Energy Accelerators VI: References. Koji TAKATA KEK. Accelerator Course, Sokendai
 .... Fundamental Concepts of Particle Accelerators V: Future of the High Energy Accelerators VI: References Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator
.... Fundamental Concepts of Particle Accelerators V: Future of the High Energy Accelerators VI: References Koji TAKATA KEK koji.takata@kek.jp http://research.kek.jp/people/takata/home.html Accelerator
Spring Not-Break Review Assignment
 Name AP Physics B Spring Not-Break Review Assignment Date Mrs. Kelly. A kilogram block is released from rest at the top of a curved incline in the shape of a quarter of a circle of radius R. The block
Name AP Physics B Spring Not-Break Review Assignment Date Mrs. Kelly. A kilogram block is released from rest at the top of a curved incline in the shape of a quarter of a circle of radius R. The block
1. SYNCHROTRON RADIATION-INDUCED DESORPTION
 127 DYNAMIC OUTGASSING Oswald Gröbner CERN, Geneva, Switzerland Abstract Outgassing stimulated by photons, ions and electrons created by highenergy and high-intensity particle beams in accelerators and
127 DYNAMIC OUTGASSING Oswald Gröbner CERN, Geneva, Switzerland Abstract Outgassing stimulated by photons, ions and electrons created by highenergy and high-intensity particle beams in accelerators and
