Supplemental Material
|
|
|
- Arlene McCoy
- 5 years ago
- Views:
Transcription
1 This journal is The Owner Societies 1 Supplemental Material Significant Role of DNA Backbone in Mediating the Transition Origin of Electronic Excitations of B-DNA Implication from Long Range Corrected TDDFT and Quantified NTO Analysis Jian-Hao Li a, b, Jeng-Da Chai a, c, *, Guang-Yu Guo a, c, d, Michitoshi Hayashi b, * a Department of Physics, Center for Theoretical Sciences, National Taiwan University, Taipei, 1617, Taiwan b Center for Condensed Matter Sciences, National Taiwan University, Taipei, 1617, Taiwan c Center for Quantum Science and Engineering, National Taiwan University, Taipei, 1617, Taiwan d Graduate Institute of Applied Physics, National Chengchi University, Taipei 1165, Taiwan jdchai@phys.ntu.edu.tw (J.-D. Chai); atmyh@ntu.edu.tw (M. Hayashi) Contents 1. Figure S1 for the various molecular segments extracted from X-ray diffraction determined B-DNA (PDB code 3BSE). S. Table S1 for the major internal coordinates of DNA backbone of non-relaxed and partially relaxed ideal and 3BSE dtpdt. S 3. Table S for the test of effects of solvent, geometry relaxation, and basis set on the excitation properties of ideal dtpdt. S3 4. Figure S for the percentage of backbone density contribution to the NTO1-H and calculated absorption cross sections of the first 5 electronic excitations of the ideal dtpdt discussed in Table S. S3 5. Table S3 for the standard-orbitals projection coefficients or coefficients square of NTO1()-H(E) of several SO-hosted excitations of ideal Thy, dt, dtpdt, dtpdt--dapda, and dapdtpdtpdg calculated by TD-ωB97X. S5 6. References S6
2 This journal is The Owner Societies 1 (a) (b) (c) (d) Figure S1. Various molecular segments extracted from X-ray diffraction determined B-DNA (PDB code 3BSE). The strucutres of (a) dt, (b)dtpdt, (c) dtpdt--dapda, and (d) dapdtpdtpdg under study are shown. TABLE S1: The major internal coordinates of DNA backbone nucleic-acid torsion angles of nonrelaxed and partially relaxed ideal and 3BSE dtpdt shown in Figure 1 and S1, adopted from the conventions defined in Chap. 5 of Ref. S1. All the torsion-angles of the two dtpdts, whether relaxation has been performed or not, are fairly within the common ranges of B-DNA structures (p.13 of Ref. S1). Non-relaxed ideal dtpdt dtpdt Non-relaxed 3BSE dtpdt 3BSE dtpdt Angle Sequence Value( ) Value( ) Value( ) Value( ) β T1 H*-O5 -C5 -C γ T1 O5 -C5 -C4 -C δ T1 C5 -C4 -C3 -O ε T1 C4 -C3 -O3 -P ζ T1 C3 -O3 -P-O α T O3 -P-O5 -C β T P-O5 -C5 -C γ T O5 -C5 -C4 -C δ T C5 -C4 -C3 -O ε T C4 -C3 -O3 -H* χ T1 O4 -C1 -N1-C χ T O4 -C1 -N1-C H* denotes the P atom that is replaced with H atom upon molecule extraction. S
3 This journal is The Owner Societies 1 TABLE S: The first 1 SO-hosted excitations predicted by TD-ωB97X S calculation of (a) nonrelaxed ideal dtpdt without optimizing hydrogen and backbone atoms first (see main text), (b) ideal dtpdt with large basis set G(df,p) S3, S4, and (c) ideal dtpdt put in water solvent of Polarisable Continuum Model (PCM) S5 around the optimized dtp(dapdtpdtpdg)pdc--adtp(dapda)pdcg (see main text). The basis set used in (a) and (c) is 6-31G(d). σ G/E/B stand for the variations of transition origin S6 caused by the change of conformation, surrounding environment, and used basis set, respectively. The results of (a) and (c) are referenced to those of the ideal dtpdt shown in Table 1(b), also adopted here in the lower-right corner for a guide to the eye. N; P λ (nm) f NTO1() σ G/E % Type N; P λ (nm) f NTO1() σ B % Type (a) Non-relaxed ideal dtpdt (b) dtpdt [6-311+G(df,p)] P1t - S1t B P1t - S1t.9 65 B - P1t1 - S1t1 3 - P1t1 - S1t P1t1 - S1t1. 67 B P1t1() - S1t1() B1 + P1t - S1t 3 + P1t(1) - S1t(1) N1t - S1t (St).4 98 (A) N1t1 - S1t A N1t1 - S1t1 (St1).5 98 (A1) N1t - S1t. 96 A N1t1 Nt1 - (S1t1) St C P1t1 (Nt1) - S1t St (E1B1) P1t1 - S1t St E1B (P1t1) (N1t1) Nt (C1) (S1t) St N1t Nt - (S1t) St C (N1t) Nt - (S1t) St (C) P1t B - S1t1 (St) (EB1) P1t - S1t1 St.7 81 EB P1t B - S1t1-87 (B1) Pt1 - S1t D P1t - S1t1 St - 9 EB1 + P1t1 - (S1t) St1 O Pt B - S1t (O) - 56 (D) - (Pt) B - O 1 (c) dtpdt with PCM solvent dtpdt (Pt1) B - S1t N1t - S1t (St) - 86 (A) (P1t1) (N1t1) B - S1t N1t1 - S1t1 (St1) - 87 (A1) (P1t) (N1t1) (B) - S1t P1t - S1t - 71 B (P1t1) (Pt1) B - S1t1() P1t1 - S1t1-7 B (P1t1) B - S1t(1) N1t1 Nt1 - St1-96 (C1) Pt1 B - S1t P1t1 (B) - S1t (St1) - 9 (E1B1) + Nt B - S1t(1) N1t Nt - (S1t) St - 93 C Nt (B) - S1t(1) P1t - S1t1 St - 89 (EB1) - Pt1 B - S1t1() Pt1 B - S1t1-7 (D1) P1t1 B - S1t P1t1() - (S1t) St1-54 (E1B Pt1 B - S1t(1) (St1) P1t(1) (Pt1) - S1t N1t1 B - (S1t1) St(1) Backbone-orbital contribution to NTO1-H (%) Non-relaxed ideal dtpdt (a) 1 dtpdt [6-311+G(df,p)] (b) 1 dtpdt with PCM solvent (c) Wavelength (nm) dtpdt 1 Epsilon/1 4 Figure S Percentage of backbone density contribution to the NTO1-H (red and black columns) and calculated absorption cross sections (curves) of the first 5 electronic excitations of dtpdt systems discussed in Table S. Red and black columns correspond to the SO-hosted and non-so-hosted excitations, respectively. The absorption cross sections are plotted by broadening the oscillator strength of each excitation with.3ev of Half-Width at Half-Height. S3
4 This journal is The Owner Societies 1 Relaxation and Basis Set Effect From panels a and b of Table S, it can be seen that up to some excitation reordering, the first several SO-hosted excitations remain in existence referencing the results of ideal dtpdt, namely, local Type-As, Type-Bs, Type-Cs, and Type-E1B1, Type-EB1, except the highest two of ideal dtpdt that are replaced by Type-B1, Type-EB1 in Table S(a) and Type-D1, Type-D in Table S(b). For the absorption energies of counterpart excitations, the two local Type-Bs, Type-E1B1, and Type-EB1 have appreciable red-shifts both in (a) and (b) while the local Type-As and Type-Cs are all rather intact. For the oscillator strength, more noticeable changes can be found in (a) for Type-E1B1 and Type- EB1, the former becoming stronger which should have something to do with the lifted backboneorbitals in its NTO1-H, whereas the latter becoming weaker due to the involvement of backboneorbitals in its NTO1-H. Finally, for the transition origin variations, most of the excitations in (a) and (b) have small values except the Type-EB1 in (a) and the Type-E1B1 and Type-C1 in (b) that have pronounced transition origin variations. From another aspect of view, Figure S shows that the absorption spectra for (a) and (b) are highly analogous to that of the ideal dtpdt, up to small energy-shifts. Taking a look at backbone-orbital contributions to NTO1-H, however, we see that the geometry relaxation lifts up the absorption energy of the lowest strong-backbone-involved excitation. It also makes several SO-hosted excitations around 17nm~18nm become strongly backbone-involved in their NTO1-Hs. Therefore, geometry relaxation effect is made clear only by analyzing the transition origins of excitations, giving another example that similar absorption spectrum can be borne by excitations of distinct transition origins. As the larger basis set 6-311G+(df,p) is used in calculation for the case (b), on the other hand, it happens that more backbone-dominant excitations are found around 17nm~19nm, which, accompanied with the results in Table S(b), suggests that for higher-lying excitations formed by local Type-D/E and CT Type-B transitions a larger basis set than the economic 6-31G(d) become more important for obtaining consistent results. For lowering-lying excitations, however, the economic basis set 6-31G(d) has been good enough to produce reliable results. PCM Solvent Effect Table S(c) shows the results of ideal dtpdt affected by PCM solvent built around the optimized dtp(dapdtpdtpdg)pdc--adtp(dapda)pdcg. Surprisingly, not like the case of Table S(a) and (b), the first several SO-hosted electronic excitations are qualitatively changed. Firstly, these SO-hosted excitations are generally located at higher positions with larger absorption energies. Secondly, the compositions of their transition origin are qualitatively different from those of the ideal dtpdt such that correspondences cannot be built. In addition, backbone orbital component is pervasively involved in their transition origins. Figure S provides a clearer demonstration of this situation, despite that the absorption spectrum does not bear so much change. However, since the solvent is only taken into account classically exhibiting polarization field, more quantum mechanical tests are still needed. In a detail, the used PCM model creates solute cavity via a set of overlapping spheres around atoms and employs a continuous surface charge whose field is determined self-consistently with the solute electrostatic potential. Therefore, a solvent, when included, is only classically taken into account providing polarization field. With actual solvent molecules surrounding solutes should be indispensible for correct description of excitation properties as, for instance, the CT contribution has been shown to play a role in the excitations of dtpdt--dapda and dapdtpdtpdg discussed in the main text. S4
5 This journal is The Owner Societies 1 TABLE S3: The detailed standard-orbitals projection coefficients or coefficients square of NTO1()- H(E) of several SO-hosted excitations of (a) ideal Thy, dt, di-thy, dtpdt, dtpdt--dapda, and dapdtpdtpdg and (b) 3BSE dt, dtpdt, dtpdt--dapda, and dapdtpdtpdg calculated by TDωB97X/6-31G(d). For orbital-fraction from backbone, the coefficients square which corresponds to electronic density contribution is shown instead. (c) The data for the coefficients square of orbitals from adenine and/or guanine in the ideal dtpdt--dapda and dapdtpdtpdg cases. (a) Thy λ (nm) h-orbital e-orbital P1 T1 P1 T N1 T1 N1 T P T1 P T N T1 N T (B) S1 T1 S1 T S T1 S T dt di- Thy dtpdt dtpdt-- dapda dapdtp dtpdg S5
6 This journal is The Owner Societies 1 (b) λ (nm) h-orbital e-orbital P1 T1 P1 T N1 T1 N1 T P T1 P T N T1 N T (B) S1 T1 S1 T S T1 S T 3BSE dt BSE dtpdt 3BSE dtpdt-- dapda 3BSE dapdtp dtpdg (c) dtpdt--dapda dapdtpdtpdg λ (nm) h-orbital e-orbital λ (nm) h-orbital e-orbital (A1) (A) (A1) (A) (G) (A) (G) (A) BSE References (S1) T. Schlick, Molecular Modeling and Simulation; Springer, New York,. (S) J.-D. Chai and M. Head-Gordon, J. Chem. Phys., 8, 18, (S3) A. D. McLean and G. S. Chandler, J. Chem. Phys., 198, 7, (S4) R. Krishnan, J. S. Binkley, R. Seeger and J. A. Pople, J. Chem. Phys., 198, 7, 65. (S5) J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 5, 15, 999. (S6) J.-H. Li, J.-D. Chai, G. Y. Guo and M. Hayashi, Chem. Phys. Lett., 11, 514, 36. S6
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Information (ESI) The correspondence between conformational
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Electronic Supplementary Information (ESI) The correspondence between conformational
Electronic Structure and Geometry Relaxation at Excited State
 Electronic Structure and Geometry Relaxation at Excited State Speaker: Chun I Wang ( 王俊壹 ) 2016.07.14 Structure-Performance Relationship Processing schemes Solvent quality Thermal annealing Blend composition
Electronic Structure and Geometry Relaxation at Excited State Speaker: Chun I Wang ( 王俊壹 ) 2016.07.14 Structure-Performance Relationship Processing schemes Solvent quality Thermal annealing Blend composition
Contrasting the optical properties of the different isomers of. oligophenylene
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Electronic supplementary information for: Contrasting the optical properties of
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Electronic supplementary information for: Contrasting the optical properties of
Solvatochromic shifts of a polarity probe implicit and explicit solvent modeling
 Solvatochromic shifts of a polarity probe implicit and explicit solvent modeling Andrzej Eilmes Faculty of Chemistry, Jagiellonian University, Kraków solvent effects in absorption spectra Absorption spectra
Solvatochromic shifts of a polarity probe implicit and explicit solvent modeling Andrzej Eilmes Faculty of Chemistry, Jagiellonian University, Kraków solvent effects in absorption spectra Absorption spectra
Prediction of absorption spectra: conjugated chains
 Prediction of absorption spectra: Exploration of TDDFT and PCM on conjugated chains John T. O Connor and Craig M. Teague* Cornell College Mount Vernon, IA MU3C Winter Conference, February 2014 Purpose
Prediction of absorption spectra: Exploration of TDDFT and PCM on conjugated chains John T. O Connor and Craig M. Teague* Cornell College Mount Vernon, IA MU3C Winter Conference, February 2014 Purpose
Supplementary Material for: Comparison of the. Marcus and Pekar partitions for non-equilibrium, polarizable-continuum reaction-field solvation.
 Supplementary Material for: Comparison of the Marcus and Pekar partitions for non-equilibrium, polarizable-continuum reaction-field solvation models Zhi-Qiang You, Jan-Michael Mewes, Andreas Dreuw, and
Supplementary Material for: Comparison of the Marcus and Pekar partitions for non-equilibrium, polarizable-continuum reaction-field solvation models Zhi-Qiang You, Jan-Michael Mewes, Andreas Dreuw, and
Relation between molecular electronic structure and nuclear spin-induced circular dichroism
 Relation between molecular electronic structure and nuclear spin-induced circular dichroism Petr Štěpánek, Sonia Coriani, Dage Sundholm, Vasily A. Ovchinnikov, Juha Vaara 1 Results Supporting Information
Relation between molecular electronic structure and nuclear spin-induced circular dichroism Petr Štěpánek, Sonia Coriani, Dage Sundholm, Vasily A. Ovchinnikov, Juha Vaara 1 Results Supporting Information
Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India. 1 st November, 2013
 Hydration of protein-rna recognition sites Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India 1 st November, 2013 Central Dogma of life DNA
Hydration of protein-rna recognition sites Ranjit P. Bahadur Assistant Professor Department of Biotechnology Indian Institute of Technology Kharagpur, India 1 st November, 2013 Central Dogma of life DNA
NUMERICAL SIMULATION OF THE IR SPECTRA OF DNA BASES
 BIOPHYSICS NUMERICAL SIMULATION OF THE IR SPECTRA OF DNA BASES C. I. MORARI, CRISTINA MUNTEAN National Institute of Research and Development for Isotopic and Molecular Technologies P.O. Box 700, R-400293
BIOPHYSICS NUMERICAL SIMULATION OF THE IR SPECTRA OF DNA BASES C. I. MORARI, CRISTINA MUNTEAN National Institute of Research and Development for Isotopic and Molecular Technologies P.O. Box 700, R-400293
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information Rational modifications on champion porphyrin
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information Rational modifications on champion porphyrin
Computational Study on the Dependence of Electronic Transition Energies of Porphin, Chlorin, Mg-Chlorin and Chlorophyll a on an External Charge
 Effect of External Charge on Electronic Energy of Chlorophyll Bull. Korean Chem. Soc. 2013, Vol. 34, No. 2 453 http://dx.doi.org/10.5012/bkcs.2013.34.2.453 Computational Study on the Dependence of Electronic
Effect of External Charge on Electronic Energy of Chlorophyll Bull. Korean Chem. Soc. 2013, Vol. 34, No. 2 453 http://dx.doi.org/10.5012/bkcs.2013.34.2.453 Computational Study on the Dependence of Electronic
Ab-initio modeling of opto-electronic properties of molecules in solvents and in proximity to a semiconductor nanoparticle
 Ab-initio modeling of opto-electronic properties of molecules in solvents and in proximity to a semiconductor nanoparticle Alain Delgado (a,b), Stefano Corni (b), Carlo Andrea Rozzi (b) Stefano Pittalis
Ab-initio modeling of opto-electronic properties of molecules in solvents and in proximity to a semiconductor nanoparticle Alain Delgado (a,b), Stefano Corni (b), Carlo Andrea Rozzi (b) Stefano Pittalis
CHEM6416 Theory of Molecular Spectroscopy 2013Jan Spectroscopy frequency dependence of the interaction of light with matter
 CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
CHEM6416 Theory of Molecular Spectroscopy 2013Jan22 1 1. Spectroscopy frequency dependence of the interaction of light with matter 1.1. Absorption (excitation), emission, diffraction, scattering, refraction
TDDFT as a tool in biophysics
 TDDFT as a tool in biophysics The primary event in vision Robert Send Universität Karlsruhe 09.09.08 Robert Send TDDFT as a tool in biophysics 09.09.08 1 / 28 Outline 1 Human vision 2 The methods 3 The
TDDFT as a tool in biophysics The primary event in vision Robert Send Universität Karlsruhe 09.09.08 Robert Send TDDFT as a tool in biophysics 09.09.08 1 / 28 Outline 1 Human vision 2 The methods 3 The
Supporting Information
 Supporting Information The Journal of Physical Chemistry A Determination of Binding Strengths of a Host-Guest Complex Using Resonance Raman Scattering Edward H. Witlicki, Stinne W. Hansen, Martin Christensen,
Supporting Information The Journal of Physical Chemistry A Determination of Binding Strengths of a Host-Guest Complex Using Resonance Raman Scattering Edward H. Witlicki, Stinne W. Hansen, Martin Christensen,
Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study
 pubs.acs.org/jpca Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study Tomoo Miyahara, Hiroshi Nakatsuji,*, and Hiroshi Sugiyama Quantum Chemistry Research Institute, JST, CREST,
pubs.acs.org/jpca Helical Structure and Circular Dichroism Spectra of DNA: A Theoretical Study Tomoo Miyahara, Hiroshi Nakatsuji,*, and Hiroshi Sugiyama Quantum Chemistry Research Institute, JST, CREST,
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes
 Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
Quantum chemical studies on the structures of some heterocyclic azo disperse dyes Nesrin Tokay, a* Zeynel Seferoğlu, b Cemil Öğretir, c and Nermin Ertan b a Hacettepe University, Faculty of Science, Chemistry
Quick Review. 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior.
 Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Quick Review 1. Kinetic Molecular Theory. 2. Average kinetic energy and average velocity. 3. Graham s Law of Effusion. 4. Real Gas Behavior. Emission spectra Every element has a unique emission spectrum
Supplementary information
 Supplementary information Vibrational coherence transfer in an electronically decoupled molecular dyad F. Schweighöfer 1, L. Dworak 1, M. Braun 1, M. Zastrow 2, J. Wahl 1, I. Burghardt 1, K. Rück-Braun
Supplementary information Vibrational coherence transfer in an electronically decoupled molecular dyad F. Schweighöfer 1, L. Dworak 1, M. Braun 1, M. Zastrow 2, J. Wahl 1, I. Burghardt 1, K. Rück-Braun
Manuel Díaz-Tinoco and J. V. Ortiz Department of Chemistry and Biochemistry Auburn University Auburn AL Abstract
 JCP Comment on Are polynuclear superhalogens without halogen atoms probable? A high level ab initio case study on triple bridged binuclear anions with cyanide ligands [J. Chem. Phys. 140, 094301 (2014)]
JCP Comment on Are polynuclear superhalogens without halogen atoms probable? A high level ab initio case study on triple bridged binuclear anions with cyanide ligands [J. Chem. Phys. 140, 094301 (2014)]
Polarizable Continuum Model Implementation in the Octopus code
 Polarizable Continuum Model Implementation in the Octopus code Alain Delgado, Carlo Andrea Rozzi, Stefano Corni S3 Center, CNR Institute of Nanoscience, Modena, Italy. Outline 1- The basics of the Polarizable
Polarizable Continuum Model Implementation in the Octopus code Alain Delgado, Carlo Andrea Rozzi, Stefano Corni S3 Center, CNR Institute of Nanoscience, Modena, Italy. Outline 1- The basics of the Polarizable
Supplementary information for. plasmonic nanorods interacting with J-aggregates.
 Supplementary information for Approaching the strong coupling limit in single plasmonic nanorods interacting with J-aggregates. by Gülis Zengin, Göran Johansson, Peter Johansson, Tomasz J. Antosiewicz,
Supplementary information for Approaching the strong coupling limit in single plasmonic nanorods interacting with J-aggregates. by Gülis Zengin, Göran Johansson, Peter Johansson, Tomasz J. Antosiewicz,
Absorption Spectra. ! Ti(H 2 O) 6 3+ appears purple (red + blue) because it absorbs green light at ~500 nm = ~20,000 cm 1.
 Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
5.74 Introductory Quantum Mechanics II
 MIT OpenCourseWare http://ocw.mit.edu 5.74 Introductory Quantum Mechanics II Spring 009 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Andrei Tokmakoff,
MIT OpenCourseWare http://ocw.mit.edu 5.74 Introductory Quantum Mechanics II Spring 009 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. Andrei Tokmakoff,
Trace Solvent as a Predominant Factor to Tune Dipeptide. Self-Assembly
 Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
Trace Solvent as a Predominant Factor to Tune Dipeptide Self-Assembly Juan Wang,, Kai Liu,,, Linyin Yan,, Anhe Wang, Shuo Bai, and Xuehai Yan *,, National Key Laboratory of Biochemical Engineering, Institute
Experiment 6: Vibronic Absorption Spectrum of Molecular Iodine
 Experiment 6: Vibronic Absorption Spectrum of Molecular Iodine We have already seen that molecules can rotate and bonds can vibrate with characteristic energies, each energy being associated with a particular
Experiment 6: Vibronic Absorption Spectrum of Molecular Iodine We have already seen that molecules can rotate and bonds can vibrate with characteristic energies, each energy being associated with a particular
Advanced TDDFT II. Double Excitations in TDDFT
 Advanced TDDFT II. Double Excitations in TDDFT f xc Neepa T. Maitra Hunter College and the Graduate Center of the City University of New York First, quick recall of how we get excitations in TDDFT: Linear
Advanced TDDFT II. Double Excitations in TDDFT f xc Neepa T. Maitra Hunter College and the Graduate Center of the City University of New York First, quick recall of how we get excitations in TDDFT: Linear
Electronic Selection Rules (II)
 Term Symbols Electronic Selection Rules (II) IMPORTANT now we are finally ready to clearly define our electronic states! microstates for a particular atomic configuration are grouped into what are called
Term Symbols Electronic Selection Rules (II) IMPORTANT now we are finally ready to clearly define our electronic states! microstates for a particular atomic configuration are grouped into what are called
X-Shaped Donor Molecules Based on Benzo[2,1-b:3,4-b ]dithiophene for Organic Solar Cells Devices with PDIs as Acceptors.
![X-Shaped Donor Molecules Based on Benzo[2,1-b:3,4-b ]dithiophene for Organic Solar Cells Devices with PDIs as Acceptors. X-Shaped Donor Molecules Based on Benzo[2,1-b:3,4-b ]dithiophene for Organic Solar Cells Devices with PDIs as Acceptors.](/thumbs/75/72361771.jpg) Supporting Information X-Shaped Donor Molecules Based on Benzo[2,1-b:3,4-b ]dithiophene for Organic Solar Cells Devices with PDIs as Acceptors. Shamsa Bibi, Ping Li, Jingping Zhang * Faculty of Chemistry,
Supporting Information X-Shaped Donor Molecules Based on Benzo[2,1-b:3,4-b ]dithiophene for Organic Solar Cells Devices with PDIs as Acceptors. Shamsa Bibi, Ping Li, Jingping Zhang * Faculty of Chemistry,
Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions
 Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions Michael Kamrath, Krista Cruse, Nathan Erickson, Molly Beernink Abstract We report results of an experimental
Computational and spectroscopic investigation of 7-azaindole: Solvation and intermolecular interactions Michael Kamrath, Krista Cruse, Nathan Erickson, Molly Beernink Abstract We report results of an experimental
Eran Rabani, S. A. Egorov, a) and B. J. Berne Department of Chemistry, Columbia University, 3000 Broadway, New York, New York 10027
 JOURNAL OF CHEMICAL PHYSICS VOLUME 109, NUMBER 15 15 OCTOBER 1998 A comparison of exact quantum mechanical and various semiclassical treatments for the vibronic absorption spectrum: The case of fast vibrational
JOURNAL OF CHEMICAL PHYSICS VOLUME 109, NUMBER 15 15 OCTOBER 1998 A comparison of exact quantum mechanical and various semiclassical treatments for the vibronic absorption spectrum: The case of fast vibrational
Supplementary Information
 Electronic Excited States of Tungsten(0) Arylisocyanides Hana Kvapilová, a,b Wesley Sattler, c Aaron Sattler, c Igor V. Sazanovich, d Ian P. Clark, d Michael Towrie, d Harry B. Gray, c, * Stanislav Záliš,
Electronic Excited States of Tungsten(0) Arylisocyanides Hana Kvapilová, a,b Wesley Sattler, c Aaron Sattler, c Igor V. Sazanovich, d Ian P. Clark, d Michael Towrie, d Harry B. Gray, c, * Stanislav Záliš,
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
CHM 111 Unit 7 Sample Questions
 Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Name: Class: Date: As you work these problems, consider and explain: A. What type of question is it? B. How do you know what type of question it is? C. What information are you looking for? D. What information
Near-infrared organic light-emitting diodes with very high external quantum efficiency and radiance
 In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION DOI: 10.1038/NPHOTON.2016.230 Near-infrared organic light-emitting diodes with very high external quantum efficiency and radiance
In the format provided by the authors and unedited. SUPPLEMENTARY INFORMATION DOI: 10.1038/NPHOTON.2016.230 Near-infrared organic light-emitting diodes with very high external quantum efficiency and radiance
Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic Dynamics
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Supplementary Data for Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2015 Supplementary Data for Modeling Ultrafast Deactivation in Oligothiophenes via Nonadiabatic
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Introduction to Franck-Condon Factors
 Introduction to Franck-Condon Factors Theresa Julia Zielinski Monmouth University Department of Chemistry, Medical Technology, and Physics West Long Branch, NJ 07764 tzielins@monmouth.edu and La Salle
Introduction to Franck-Condon Factors Theresa Julia Zielinski Monmouth University Department of Chemistry, Medical Technology, and Physics West Long Branch, NJ 07764 tzielins@monmouth.edu and La Salle
Prediction of Thymine Dimer Repair by Electron Transfer from Photoexcited 8 Aminoguanine or Its Deprotonated Anion
 pubs.acs.org/jpca Prediction of Thymine Dimer epair by Electron Transfer from Photoexcited 8 Aminoguanine or Its Deprotonated Anion Iwona Sieradzan, Marzena Marchaj, Iwona Anusiewicz, Piotr Skurski,, and
pubs.acs.org/jpca Prediction of Thymine Dimer epair by Electron Transfer from Photoexcited 8 Aminoguanine or Its Deprotonated Anion Iwona Sieradzan, Marzena Marchaj, Iwona Anusiewicz, Piotr Skurski,, and
2008 Brooks/Cole 2. Frequency (Hz)
 Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Electromagnetic Radiation and Matter Oscillating electric and magnetic fields. Magnetic field Electric field Chapter 7: Electron Configurations and the Periodic Table Traveling wave moves through space
Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia
 University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
Electron scattering in large water clusters from photoelectron imaging with high harmonic radiation
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 PCCP- Electronic Supplementary Information Electron scattering in large water clusters
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2018 PCCP- Electronic Supplementary Information Electron scattering in large water clusters
5.111 Lecture Summary #5 Friday, September 12, 2014
 5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
ATOM atomov - indivisible
 Structure of matter ATOM atomov - indivisible Greek atomists - Democrite and Leukip 300 b.c. R. Bošković - accepts the concept of atom and defines the force J. Dalton - accepts the concept of atom and
Structure of matter ATOM atomov - indivisible Greek atomists - Democrite and Leukip 300 b.c. R. Bošković - accepts the concept of atom and defines the force J. Dalton - accepts the concept of atom and
( ) x10 8 m. The energy in a mole of 400 nm photons is calculated by: ' & sec( ) ( & % ) 6.022x10 23 photons' E = h! = hc & 6.
 Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
Introduction to Spectroscopy Spectroscopic techniques are widely used to detect molecules, to measure the concentration of a species in solution, and to determine molecular structure. For proteins, most
All-atom Molecular Mechanics. Trent E. Balius AMS 535 / CHE /27/2010
 All-atom Molecular Mechanics Trent E. Balius AMS 535 / CHE 535 09/27/2010 Outline Molecular models Molecular mechanics Force Fields Potential energy function functional form parameters and parameterization
All-atom Molecular Mechanics Trent E. Balius AMS 535 / CHE 535 09/27/2010 Outline Molecular models Molecular mechanics Force Fields Potential energy function functional form parameters and parameterization
HL Chemistry. Thursday August 20th Thursday, August 20, 15
 HL Chemistry Thursday August 20th 2015 Agenda Warm Up: Electron Configuration Practice Review Topic 2.2 Electron Configuration and begin Topic 12.1 Atomic Structure Warm Up You have 10 minutes ONLY! GO!
HL Chemistry Thursday August 20th 2015 Agenda Warm Up: Electron Configuration Practice Review Topic 2.2 Electron Configuration and begin Topic 12.1 Atomic Structure Warm Up You have 10 minutes ONLY! GO!
SUPPLEMENTARY INFORMATION
 Fig. S1: High-Harmonic Interferometry of a Chemical Reaction A weak femtosecond laser pulse excites a molecule from its ground state (on the bottom) to its excited state (on top) in which it dissociates.
Fig. S1: High-Harmonic Interferometry of a Chemical Reaction A weak femtosecond laser pulse excites a molecule from its ground state (on the bottom) to its excited state (on top) in which it dissociates.
Crystal Violet as a Fluorescent Switch-On Probe for I-Motif: Label-Free DNA-Based Logic Gate
 Crystal Violet as a Fluorescent Switch-On Probe for I-Motif: Label-Free DNA-Based Logic Gate Dik-Lung Ma,* a Maria Hiu-Tung Kwan, a Daniel Shiu-Hin Chan, a Paul Lee, a Hui Yang, a Victor Pui-Yan Ma, a
Crystal Violet as a Fluorescent Switch-On Probe for I-Motif: Label-Free DNA-Based Logic Gate Dik-Lung Ma,* a Maria Hiu-Tung Kwan, a Daniel Shiu-Hin Chan, a Paul Lee, a Hui Yang, a Victor Pui-Yan Ma, a
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers
 Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
SUPPLEMENTAL MATERIAL Systematic Coarse-Grained Modeling of Complexation between Small Interfering RNA and Polycations Zonghui Wei 1 and Erik Luijten 1,2,3,4,a) 1 Graduate Program in Applied Physics, Northwestern
Lecture 5: the Hydrogen Atom
 Lecture 5: the Hydrogen Atom 1. Hydrogen atom: energy levels in the Bohr model 2. Emission lines: atoms releasing energy as electrons fall from level to level 3. Absorption lines: electrons being bumped
Lecture 5: the Hydrogen Atom 1. Hydrogen atom: energy levels in the Bohr model 2. Emission lines: atoms releasing energy as electrons fall from level to level 3. Absorption lines: electrons being bumped
Analysis of the simulation
 Analysis of the simulation Marcus Elstner and Tomáš Kubař January 7, 2014 Thermodynamic properties time averages of thermodynamic quantites correspond to ensemble averages (ergodic theorem) some quantities
Analysis of the simulation Marcus Elstner and Tomáš Kubař January 7, 2014 Thermodynamic properties time averages of thermodynamic quantites correspond to ensemble averages (ergodic theorem) some quantities
Supporting information for: Ultrafast Transient Absorption Spectroscopy of. the Sunscreen Constituent Ethylhexyl Triazone
 Supporting information for: Ultrafast Transient Absorption Spectroscopy of the Sunscreen Constituent Ethylhexyl Triazone Lewis A. Baker,,, Sarah L. Clark, Scott Habershon,, and Vasilios G. Stavros, Department
Supporting information for: Ultrafast Transient Absorption Spectroscopy of the Sunscreen Constituent Ethylhexyl Triazone Lewis A. Baker,,, Sarah L. Clark, Scott Habershon,, and Vasilios G. Stavros, Department
Evaluation of intramolecular charge transfer state of 4-N, N-dimethylamino cinnamaldehyde using time-dependent density functional theory
 J. Chem. Sci. Vol. 125, No. 4, July 2013, pp. 933 938. c Indian Academy of Sciences. Evaluation of intramolecular charge transfer state of 4-N, N-dimethylamino cinnamaldehyde using time-dependent density
J. Chem. Sci. Vol. 125, No. 4, July 2013, pp. 933 938. c Indian Academy of Sciences. Evaluation of intramolecular charge transfer state of 4-N, N-dimethylamino cinnamaldehyde using time-dependent density
1. Introduction T = T! " T +T! (1)
 Thymine dimer repair by electron transfer from photo-excited 2!,3!,5!-tri- -acetyl-8-oxo-7,8-dihydroguanosine or 2!,3!,5!-tri--acetyl-ribosyluric acid- a theoretical study Marzena Marchaj 1, Iwona Sieradzan
Thymine dimer repair by electron transfer from photo-excited 2!,3!,5!-tri- -acetyl-8-oxo-7,8-dihydroguanosine or 2!,3!,5!-tri--acetyl-ribosyluric acid- a theoretical study Marzena Marchaj 1, Iwona Sieradzan
Unveiling the role of hot charge-transfer states. in molecular aggregates via nonadiabatic. dynamics
 Unveiling the role of hot charge-transfer states in molecular aggregates via nonadiabatic dynamics Daniele Fazzi 1, Mario Barbatti 2, Walter Thiel 1 1 Max-Planck-Institut für Kohlenforschung Kaiser-Wilhelm-Platz
Unveiling the role of hot charge-transfer states in molecular aggregates via nonadiabatic dynamics Daniele Fazzi 1, Mario Barbatti 2, Walter Thiel 1 1 Max-Planck-Institut für Kohlenforschung Kaiser-Wilhelm-Platz
Analysis of Permanent Electric Dipole Moments of Aliphatic Amines.
 Analysis of Permanent Electric Dipole Moments of Aliphatic Amines. Boris Lakard* LPUB, UMR CNRS 5027, University of Bourgogne, F-21078, Dijon, France Internet Electronic Conference of Molecular Design
Analysis of Permanent Electric Dipole Moments of Aliphatic Amines. Boris Lakard* LPUB, UMR CNRS 5027, University of Bourgogne, F-21078, Dijon, France Internet Electronic Conference of Molecular Design
Supplementary information. Nitrogen backbone oligomers
 Supplementary information Nitrogen backbone oligomers Hongbo Wang 1,2, Mikhail I. Eremets 1 *, Ivan Troyan 1,3, Hanyu Liu 2, Yanming Ma 2, Luc Vereecken 1 1 Max Planck Institute for Chemistry, Biogeochemistry
Supplementary information Nitrogen backbone oligomers Hongbo Wang 1,2, Mikhail I. Eremets 1 *, Ivan Troyan 1,3, Hanyu Liu 2, Yanming Ma 2, Luc Vereecken 1 1 Max Planck Institute for Chemistry, Biogeochemistry
Atoms, Molecules and Solids. From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation of the wave function:
 Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Essay outline and Ref to main article due next Wed. HW 9: M Chap 5: Exercise 4 M Chap 7: Question A M Chap 8: Question A From Last Time Superposition of quantum states Philosophy of quantum mechanics Interpretation
Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca
 Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 509 514 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca DONG
Commun. Theor. Phys. (Beijing, China) 43 (2005) pp. 509 514 c International Academic Publishers Vol. 43, No. 3, March 15, 2005 Shape Coexistence and Band Termination in Doubly Magic Nucleus 40 Ca DONG
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
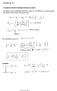 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
SERS and NMR Studies of Typical Aggregation-induced. Emission Molecules
 Supplemental information SERS and NMR Studies of Typical Aggregation-induced Emission Molecules Cheng Fang, Yujun Xie, Martin R. Johnston, Yinlan Ruan, Ben Zhong Tang 5, *, Qian Peng, *, Youhong Tang 6,
Supplemental information SERS and NMR Studies of Typical Aggregation-induced Emission Molecules Cheng Fang, Yujun Xie, Martin R. Johnston, Yinlan Ruan, Ben Zhong Tang 5, *, Qian Peng, *, Youhong Tang 6,
Supplementary Information
 Supplementary Information Supplementary Figure 1. Photographs show the titration experiments by dropwise adding ~5 times number of moles of (a) LiOH and LiOH+H 2 O, (b) H 2 O 2 and H 2 O 2 +LiOH, (c) Li
Supplementary Information Supplementary Figure 1. Photographs show the titration experiments by dropwise adding ~5 times number of moles of (a) LiOH and LiOH+H 2 O, (b) H 2 O 2 and H 2 O 2 +LiOH, (c) Li
An Initio Theoretical Study for the Electronic Spectra of β-thioxoketones
 American Journal of Applied Sciences 9 (1): 152-157, 2012 ISSN 1546-9239 2012 Science Publications An Initio Theoretical Study for the Electronic Spectra of β-thioxoketones 1 Rita S. Elias, 2 Bahjat A.
American Journal of Applied Sciences 9 (1): 152-157, 2012 ISSN 1546-9239 2012 Science Publications An Initio Theoretical Study for the Electronic Spectra of β-thioxoketones 1 Rita S. Elias, 2 Bahjat A.
Modeling in Solution. Important Observations. Energy Concepts
 Modeling in Solution 1 Important Observations Various electrostatic effects for a solvated molecule are often less important than for an isolated gaseous molecule when the molecule is dissolved in a solvent
Modeling in Solution 1 Important Observations Various electrostatic effects for a solvated molecule are often less important than for an isolated gaseous molecule when the molecule is dissolved in a solvent
ELECTRONIC SUPPLEMENTARY INFORMATION
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 S-1 ELECTRONIC SUPPLEMENTARY INFORMATION OCT. 1, 2017 Combined Quantum Mechanical
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 S-1 ELECTRONIC SUPPLEMENTARY INFORMATION OCT. 1, 2017 Combined Quantum Mechanical
Chapter 5 Predicted A-X Transition Frequencies and 2-Dimensional Torsion-Torsion Potential Energy Surfaces of HOCH 2 OO and HOC(CH 3 ) 2 OO
 119 Chapter 5 Predicted A-X Transition Frequencies and 2-Dimensional Torsion-Torsion Potential Energy Surfaces of HOCH 2 OO and HOC(CH 3 ) 2 OO Abstract In Chapter 4, we presented the 1 (OH stretch) vibrational
119 Chapter 5 Predicted A-X Transition Frequencies and 2-Dimensional Torsion-Torsion Potential Energy Surfaces of HOCH 2 OO and HOC(CH 3 ) 2 OO Abstract In Chapter 4, we presented the 1 (OH stretch) vibrational
Supporting Information
 Supporting Information Computational Evidence of Inversion of 1 L a and 1 L b -Derived Excited States in Naphthalene Excimer Formation from ab Initio Multireference Theory with Large Active Space: DMRG-CASPT2
Supporting Information Computational Evidence of Inversion of 1 L a and 1 L b -Derived Excited States in Naphthalene Excimer Formation from ab Initio Multireference Theory with Large Active Space: DMRG-CASPT2
A critical approach toward resonance-assistance in the intramolecular hydrogen bond
 Electronic Supplementary Material (ESI) for Photochemical & Photobiological Sciences. This journal is The Royal Society of Chemistry and Owner Societies 2015 Supplementary Information for A critical approach
Electronic Supplementary Material (ESI) for Photochemical & Photobiological Sciences. This journal is The Royal Society of Chemistry and Owner Societies 2015 Supplementary Information for A critical approach
Theoretical models for the solvent effect
 Theoretical models for the solvent effect Benedetta Mennucci Dipartimento di Chimica e Chimica Industriale Web: http://benedetta.dcci.unipi.it Email: bene@dcci.unipi.it 8. Excited electronic states in
Theoretical models for the solvent effect Benedetta Mennucci Dipartimento di Chimica e Chimica Industriale Web: http://benedetta.dcci.unipi.it Email: bene@dcci.unipi.it 8. Excited electronic states in
Supplementary Information for: Hydrogen abstraction by photoexcited benzophenone: consequences for DNA photosensitization
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Supplementary Information for: Hydrogen abstraction by photoexcited benzophenone:
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2016 Supplementary Information for: Hydrogen abstraction by photoexcited benzophenone:
Supporting Materials
 Supporting Materials Figure S1 Experimental Setup Page Figure S (a) (b) (c) Feynman Diagrams Page 3-6 Figure S3 D IR Spectra Page 7 Figure S4 Kinetic Model Page 8 Figure S5 Van t Hoff Plots Page 9 1 k
Supporting Materials Figure S1 Experimental Setup Page Figure S (a) (b) (c) Feynman Diagrams Page 3-6 Figure S3 D IR Spectra Page 7 Figure S4 Kinetic Model Page 8 Figure S5 Van t Hoff Plots Page 9 1 k
Application of electrostatic Solvent Models to the Electronic Spectrum of Mesityl Oxide
 Journal of Basrah Researches ((Sciences Volume 38. Number 1. ((2012 vailable online at: www.basra-science journal.org 2695 1817 ISSN pplication of electrostatic Solvent Models to the Electronic Spectrum
Journal of Basrah Researches ((Sciences Volume 38. Number 1. ((2012 vailable online at: www.basra-science journal.org 2695 1817 ISSN pplication of electrostatic Solvent Models to the Electronic Spectrum
State Key Laboratory of Molecular Reaction Dynamics, Dalian Institute of Chemical Physics, Chinese Academic of Sciences, Dalian , P. R. China.
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information for: Theoretical exploration of MgH 2 and graphene nano-flake in cyclohexane:
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Supplementary Information for: Theoretical exploration of MgH 2 and graphene nano-flake in cyclohexane:
Supporting Information
 Supporting Information Light emission near a gradient metasurface Leonard C. Kogos and Roberto Paiella Department of Electrical and Computer Engineering and Photonics Center, Boston University, Boston,
Supporting Information Light emission near a gradient metasurface Leonard C. Kogos and Roberto Paiella Department of Electrical and Computer Engineering and Photonics Center, Boston University, Boston,
SUPPLEMENTARY INFORMATION
 Atomic structure and dynamic behaviour of truly one-dimensional ionic chains inside carbon nanotubes Ryosuke Senga 1, Hannu-Pekka Komsa 2, Zheng Liu 1, Kaori Hirose-Takai 1, Arkady V. Krasheninnikov 2
Atomic structure and dynamic behaviour of truly one-dimensional ionic chains inside carbon nanotubes Ryosuke Senga 1, Hannu-Pekka Komsa 2, Zheng Liu 1, Kaori Hirose-Takai 1, Arkady V. Krasheninnikov 2
Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002
 Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002 More Quantum Physics We know now how to detect light (or photons) One possibility to detect
Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002 More Quantum Physics We know now how to detect light (or photons) One possibility to detect
Electronic Supplementary Information Band-Structure-Controlled BiO(ClBr) (1-x)/2 I x Solid Solutions for Visible-Light Photocatalysis
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Band-Structure-Controlled BiO(ClBr)
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Band-Structure-Controlled BiO(ClBr)
Coherent Lattice Vibrations in Mono- and Few-Layer. WSe 2. Supporting Information for. 749, Republic of Korea
 Supporting Information for Coherent Lattice Vibrations in Mono- and Few-Layer WSe 2 Tae Young Jeong, 1,2 Byung Moon Jin, 1 Sonny H. Rhim, 3 Lamjed Debbichi, 4 Jaesung Park, 2 Yu Dong Jang, 1 Hyang Rok
Supporting Information for Coherent Lattice Vibrations in Mono- and Few-Layer WSe 2 Tae Young Jeong, 1,2 Byung Moon Jin, 1 Sonny H. Rhim, 3 Lamjed Debbichi, 4 Jaesung Park, 2 Yu Dong Jang, 1 Hyang Rok
A Condition for Entropy Exchange Between Atom and Field
 Commun. Theor. Phys. 57 (2012) 209 213 Vol. 57, No. 2, February 15, 2012 A Condition for Entropy Exchange Between Atom and Field YAN Xue-Qun ( ) and LÜ Yu-Guang (ù ½) Institute of Physics and Department
Commun. Theor. Phys. 57 (2012) 209 213 Vol. 57, No. 2, February 15, 2012 A Condition for Entropy Exchange Between Atom and Field YAN Xue-Qun ( ) and LÜ Yu-Guang (ù ½) Institute of Physics and Department
Time-dependent density functional theory
 Time-dependent density functional theory E.K.U. Gross Max-Planck Institute for Microstructure Physics OUTLINE LECTURE I Phenomena to be described by TDDFT LECTURE II Review of ground-state DFT LECTURE
Time-dependent density functional theory E.K.U. Gross Max-Planck Institute for Microstructure Physics OUTLINE LECTURE I Phenomena to be described by TDDFT LECTURE II Review of ground-state DFT LECTURE
Copyright 2018 Dan Dill 1
 TP The wavelength of the electron wave in the ground state of H atom is about the size of the atom. In what region of the spectrum is light of similar wavelength? 1. Radio 2. Microwave 3. Infrared 4. Visible
TP The wavelength of the electron wave in the ground state of H atom is about the size of the atom. In what region of the spectrum is light of similar wavelength? 1. Radio 2. Microwave 3. Infrared 4. Visible
ONETEP PB/SA: Application to G-Quadruplex DNA Stability. Danny Cole
 ONETEP PB/SA: Application to G-Quadruplex DNA Stability Danny Cole Introduction Historical overview of structure and free energy calculation of complex molecules using molecular mechanics and continuum
ONETEP PB/SA: Application to G-Quadruplex DNA Stability Danny Cole Introduction Historical overview of structure and free energy calculation of complex molecules using molecular mechanics and continuum
Investigating excited state dynamics in 7 azaindole. Nathan Erickson, Molly Beernink, and Nathaniel Swenson
 Investigating excited state dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson 1 Background I 7AI Dimer Previous studies have shown that 7 azaindole (7AI) readily forms H bonded
Investigating excited state dynamics in 7 azaindole Nathan Erickson, Molly Beernink, and Nathaniel Swenson 1 Background I 7AI Dimer Previous studies have shown that 7 azaindole (7AI) readily forms H bonded
Electronic structure theory: Fundamentals to frontiers. 2. Density functional theory
 Electronic structure theory: Fundamentals to frontiers. 2. Density functional theory MARTIN HEAD-GORDON, Department of Chemistry, University of California, and Chemical Sciences Division, Lawrence Berkeley
Electronic structure theory: Fundamentals to frontiers. 2. Density functional theory MARTIN HEAD-GORDON, Department of Chemistry, University of California, and Chemical Sciences Division, Lawrence Berkeley
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015,
 Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score)
 IB PHYSICS Name: Period: Date: DEVIL PHYSICS BADDEST CLASS ON CAMPUS GETTING STARTED: PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score) Go to https://phet.colorado.edu/ Click
IB PHYSICS Name: Period: Date: DEVIL PHYSICS BADDEST CLASS ON CAMPUS GETTING STARTED: PhET Light Emission and Lasers (27 points available x 2/3 = 18 points max score) Go to https://phet.colorado.edu/ Click
Theoretical comparative study on hydrogen storage of BC 3 and carbon nanotubes
 J. At. Mol. Sci. doi: 10.4208/jams.121011.011412a Vol. 3, No. 4, pp. 367-374 November 2012 Theoretical comparative study on hydrogen storage of BC 3 and carbon nanotubes Xiu-Ying Liu a,, Li-Ying Zhang
J. At. Mol. Sci. doi: 10.4208/jams.121011.011412a Vol. 3, No. 4, pp. 367-374 November 2012 Theoretical comparative study on hydrogen storage of BC 3 and carbon nanotubes Xiu-Ying Liu a,, Li-Ying Zhang
NMR of Nucleic Acids. K.V.R. Chary Workshop on NMR and it s applications in Biological Systems November 26, 2009
 MR of ucleic Acids K.V.R. Chary chary@tifr.res.in Workshop on MR and it s applications in Biological Systems TIFR ovember 26, 2009 ucleic Acids are Polymers of ucleotides Each nucleotide consists of a
MR of ucleic Acids K.V.R. Chary chary@tifr.res.in Workshop on MR and it s applications in Biological Systems TIFR ovember 26, 2009 ucleic Acids are Polymers of ucleotides Each nucleotide consists of a
Quantum Physics 2: Homework #6
 Quantum Physics : Homework #6 [Total 10 points] Due: 014.1.1(Mon) 1:30pm Exercises: 014.11.5(Tue)/11.6(Wed) 6:30 pm; 56-105 Questions for problems: 민홍기 hmin@snu.ac.kr Questions for grading: 모도영 modori518@snu.ac.kr
Quantum Physics : Homework #6 [Total 10 points] Due: 014.1.1(Mon) 1:30pm Exercises: 014.11.5(Tue)/11.6(Wed) 6:30 pm; 56-105 Questions for problems: 민홍기 hmin@snu.ac.kr Questions for grading: 모도영 modori518@snu.ac.kr
6.2. Introduction to Spectroscopic states and term symbols
 Chemistry 3820 Lecture Notes Dr. M. Gerken Page62 6.2. Introduction to Spectroscopic states and term symbols From the number of absorption bands we have already seen that usually more d-d transitions are
Chemistry 3820 Lecture Notes Dr. M. Gerken Page62 6.2. Introduction to Spectroscopic states and term symbols From the number of absorption bands we have already seen that usually more d-d transitions are
Iridium Complexes Bearing a PNP Ligand, Favoring Facile C(sp 3 )- H Bond Cleavage
 Iridium Complexes Bearing a PNP Ligand, Favoring Facile C(sp 3 )- H Bond Cleavage Kapil S. Lokare,* a Robert J. Nielsen, *b Muhammed Yousufuddin, c William A. Goddard III, b and Roy A. Periana*,a a Scripps
Iridium Complexes Bearing a PNP Ligand, Favoring Facile C(sp 3 )- H Bond Cleavage Kapil S. Lokare,* a Robert J. Nielsen, *b Muhammed Yousufuddin, c William A. Goddard III, b and Roy A. Periana*,a a Scripps
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions
 Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION DOI: 10.1038/NPHYS2210 Femtosecond torsional relaxation Theoretical methodology: J. Clark, S. Tretiak, T. Nelson, G. Cirmi & G. Lanzani To model non-adiabatic excited state dynamics
SUPPLEMENTARY INFORMATION DOI: 10.1038/NPHYS2210 Femtosecond torsional relaxation Theoretical methodology: J. Clark, S. Tretiak, T. Nelson, G. Cirmi & G. Lanzani To model non-adiabatic excited state dynamics
Computational Molecular Modeling
 Computational Molecular Modeling Lecture 1: Structure Models, Properties Chandrajit Bajaj Today s Outline Intro to atoms, bonds, structure, biomolecules, Geometry of Proteins, Nucleic Acids, Ribosomes,
Computational Molecular Modeling Lecture 1: Structure Models, Properties Chandrajit Bajaj Today s Outline Intro to atoms, bonds, structure, biomolecules, Geometry of Proteins, Nucleic Acids, Ribosomes,
1 of 5 14/10/ :21
 X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
X-ray absorption s, characteristic X-ray lines... 4.2.1 Home About Table of Contents Advanced Search Copyright Feedback Privacy You are here: Chapter: 4 Atomic and nuclear physics Section: 4.2 Absorption
Coupling the Level-Set Method with Variational Implicit Solvent Modeling of Molecular Solvation
 Coupling the Level-Set Method with Variational Implicit Solvent Modeling of Molecular Solvation Bo Li Math Dept & CTBP, UCSD Li-Tien Cheng (Math, UCSD) Zhongming Wang (Math & Biochem, UCSD) Yang Xie (MAE,
Coupling the Level-Set Method with Variational Implicit Solvent Modeling of Molecular Solvation Bo Li Math Dept & CTBP, UCSD Li-Tien Cheng (Math, UCSD) Zhongming Wang (Math & Biochem, UCSD) Yang Xie (MAE,
X-Ray transitions to low lying empty states
 X-Ray Spectra: - continuous part of the spectrum is due to decelerated electrons - the maximum frequency (minimum wavelength) of the photons generated is determined by the maximum kinetic energy of the
X-Ray Spectra: - continuous part of the spectrum is due to decelerated electrons - the maximum frequency (minimum wavelength) of the photons generated is determined by the maximum kinetic energy of the
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
