Mean Activity Coefficients of Peroxodisulfates in Saturated Solutions of the Conversion System 2NH 4. H 2 O at 20 C and 30 C
|
|
|
- Annice Chapman
- 5 years ago
- Views:
Transcription
1 Mean Ativity Coeffiients of Peroxodisulfates in Saturated Solutions of the Conversion System NH 4 Na S O 8 H O at 0 C and 0 C Jan Balej Heřmanova 5, Prague 7, Czeh Republi balejan@seznam.z Abstrat: Solubility data of the onversion system NH 4 Na S O 8 H O at 0 C and 0 C for the partial pseudoternary systems Na S O 8 (NH 4 ) SO 4 H O, (NH 4 ) S O 8 Na SO 4 H O, Na S O 8 NaNH 4 SO 4 H O and (NH 4 ) S O 8 NaNH 4 SO 4 H O have been orrelated using the relative ativity oeffiient expansion of the rystallizing omponent. The obtained orrelation equations have been used for the alulation of mean ativity oeffiients of ammonium and sodium peroxodisulfates in their saturated solutions of the mentioned pseudoternary systems. Keywords: mean ativity oeffiient, ammonium peroxodisulfate, sodium peroxodisulfate, onversion system Introdution In the previous ontribution (Balej 011a), mean ativity oeffiients (further, term mean is mostly omitted) of sodium, potassium and ammonium peroxodisulfates in pure aqueous solutions in the temperature region (0 50) C have been presented. Another ontribution (Balej 011b) has brought ativity oeffiients of peroxodisulfates in saturated aqueous polyomponent solutions of ternary systems Na S O 8 Na SO 4 H O, K S O 8 K SO 4 H O, (NH 4 ) S O 8 (NH 4 ) SO 4 H O, Na S O 8 NaOH H O, K S O 8 KOH H O, Na S O 8 K S O 8 H O, Na S O 8 (NH 4 ) S O 8 H O, Na - S O 8 H SO 4 H O, K S O 8 H SO 4 H O, (NH 4 ) S O 8 H SO 4 H O, and quaternary systems (NH 4 ) S O 8 (NH 4 ) SO 4 H SO 4 H O, Na S O 8 Na SO 4 H SO 4 H O, and (NH 4 ) S O 8 Na - S O 8 H SO 4 H O. The aim of the present ommuniation is to bring values of ativity oeffiients of ammonium or sodium peroxodisulfate in seleted saturated solutions of the onversion system NH 4 Na S O 8 H O based on the solubility data at 0 C (Balej, Čížek and Thumová 1976) and at 0 C (Balej and Thumová 1976). From the isothermal solubility diagrams of this system, it followed that besides the four solid main substanes, (NH 4 ) S O 8, Na S O 8, (NH 4 ) SO 4 and Na SO 4, the double sulfate NaNH 4 SO 4 H O also exists in the equilibrium with orresponding saturated solutions. Consequently, five different rystallization regions of the given solid substanes are separated by seven binary eutonia lines rossing in three ternary eutoni points at the given temperature. It followed further that no of the rystallization regions of the four mean omponents touhes the rystallization region of the opposite omponent, beause they are always separated by a quite large rystallization area of the fifth omponent, NaNH 4 SO 4 H O. Due to a high number of the original solubility data (totally 05 measurements) bringing results of four diagonal setions, seven binary eutonia lines and three ternary eutoni points at the given temperature, this ommuniation is only limited to the orrelation of the previous solubility data of orresponding peroxodisulfates in four diagonal setions, i.e. (NH 4 ) S O 8 Na SO 4 H O, Na S O 8 (NH 4 ) SO 4 H O, (NH 4 ) S O 8 NaNH 4 SO 4 H O, and Na S O 8 NaNH 4 SO 4 H O at both onsidered temperatures. The first two systems represent pseudoternary systems without any ommon ion, whereas the last two belong to suh ones with a ommon and an unommon ation. The obtained results have been used for the alulation of ativity oeffiients of the orresponding peroxodisulfates in their saturated polyomponent solutions. The solubility orrelation of the onsidered systems was based on the relative ativity oeffiient expansion (Nývlt 1977, Balej 198) (1) Here, so and s denotes the ativity oeffiient of the rystallizing substane in the saturated solution in pure water and in the polyomponent solution, respetively, m C, S and m A, S denotes the molality of Ata Chimia Slovaa, Vol. 6, No., 01, pp , DOI: /as
2 the ation and anion of the rystallizing substane in the saturated polyomponent solution, and m 0, S represents the solubility of the rystallizing substane in pure water, all at the given temperature. The ativity oeffiients of the onsidered peroxodisulfate in the saturated solutions of the onsidered pseudoternary systems have been alulated by two different methods. The first one onsists on the mentioned method of the relative ativity oeffiient expansion of the rystallizing omponent defined by Eq. (1), aording to whih the ativity oeffiient of the onsidered substane in its saturated polyomponent solution is given by the equation 0 exp { () 1 The seond method is based on the thermodynami ondition of the equilibrium between the solid anhydrous eletrolyte of the type 1-, A B, and its saturated solution formulated by the expression (Balej 010a) S % D f G ( C A, r) - D G % f ^C A, aqh f RT - ^m C, S A, S m h p () The equation is valid under the assumption of a omplete dissoiation of the onsidered strong eletrolyte. Here, D f G (C A, r) denotes the standard formation Gibbs energy of the anhydrous solid eletrolyte and D f G (C A, aq) denotes the same quantity of the same eletrolyte in its aqueous solution, respetively. Values of D f G r and D f G aq of the peroxodisulfates at the given temperatures have been taken from the previous paper (Balej 010b), molalities of the appropriate ions in saturated polyomponent solutions have been alulated from the mentioned solubility data originally given in wt. %. Results and Disussion The omposition (in mol kg 1 ) of the saturated solutions of the onsidered ternary systems without a ommon ion, (NH 4 ) S O 8 Na SO 4 H O, at both temperatures is presented in Table 1. The last data at every temperature orrespond to the eutoni solution at whih two solid substanes oexist with the saturated solution. The evaluation of solubility data aording to Eq. (1) led to the expression 0 m { 1 a k ` j 0 ^ thm (4) so that the solubility dependene of the ammonium peroxodisulfate ( ) on the ontent of sodium sulfate (m ) is given by the equation ( t)exp(( t)m ) (5) Here, 0 denotes the solubility of the rystallizing omponen in pure water, t the temperature in C. The same dependene with almost the same auray ould be expressed by an empirial equation Tab. 1. Solubility data and ativity oeffiients in the system (NH 4 ) S O 8 Na SO 4 H O. m solid exp Eq. (5) Eq. (6) Eq. () Eq. () 0 C (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O 8 NaNH 4 SO 4 H O 0 C (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O 8 NaNH 4 SO 4 H O 164
3 t ( t)m (6) Results of the orresponding alulations are given in Tab. 1 as well. As an be seen, the agreement between the measured and alulated values aording to different equations is very good and the mean deviation is always below 0.1 rel. %. Solubility data of the pseudoternary system Na S O 8 (NH 4 ) SO 4 H O at the both onsidered temperatures are given in Table, from whih the following orrelation equation has been obtained: so that 0 m { 1 a k ` j 0 ^ thm (7) ( t)exp(( t)m ) (8) The solubility ould also be expressed by a simpler empiri equation t ( t)m (9) Results of the orresponding alulations are given in Tab. as well. As an be seen, the agreement between the measured and alulated values aording to different equations is in this ase also very good and the mean deviation is always below 0.1 rel. %. At the other two onsidered pseudoternary systems with a ommon ation, the solubility orrelation equation has the following ommon form ^m mh m 4m0 { 1 1 (10) Aordingly, the alulation of the solubility of rystallizing peroxodisulfate ( ) must be performed by the solution of the polynomi equation of the following form m m m 0.5m m 0exp( 1 ) 0 (11) The solubility data of the system (NH 4 ) S O 8 NaNH 4 SO 4 H O are given in Table. The evaluation of solubility data aording to Eq. (1) led to the following expression for the solubility dependene of the ammonium peroxodisulfate ( ) on the ontent of sodium ammonium sulfate (m ) ^m1 mh m1 4m0 { 1 ^ thm ^ thm (1) so that the molality of ammonium peroxodisulfate in the saturated polyomponent solutions has been alulated by the solvation of the polynomi equation m m m 0.5m m 0exp(( t)m (1) ( t)m ) 0 Values of ould also be obtained using a simpler empiri equation t ( t)m (14) The results of alulations are given in Table as well. As an be seen, the agreement between the measured and alulated values of is very good and the mean deviation 0.1 rel. %. The solubility data of the system Na S O 8 NaNH 4 SO 4 H O are given in Table 4. The evaluation of solubility data aording to Eq. (1) led to the following ommon expression Tab.. Solubility data and ativity oeffiients in the system Na S O 8 (NH 4 ) SO 4 H O. m oexisting solid substane exp Eq. (8) Eq. (9) Eq. () Eq. () 0 C Na S O Na S O Na S O Na S O 8 NaNH 4 SO 4 H O 0 C Na S O Na S O Na S O Na S O Na S O 8 NaNH 4 SO 4 H O 165
4 Tab.. Solubility data and ativity oeffiients in the system (NH 4 ) S O 8 NaNH 4 SO 4 H O. m oexisting solid substane exp Eq. (1) Eq. (14) Eq. () Eq. () 0 C (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O 8 NaNH 4 SO 4 H O 0 C (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O (NH 4 ) S O 8 NaNH 4 SO 4 H O ^m1 mh m1 4m0 { 1 ^ thm ^ thm (15) so that the molality of sodium peroxodisulfate in the saturated polyomponent solutions has been alulated by the solvation of the polynomi equation m m m 0.5m m 0exp(( t)m (16) ( t)m ) 0 The same quantity ould be alulated using a simpler empiri equation t ( t)m (17) with a mean deviation 0.08 rel. %. Mean ativity oeffiients of the orresponding peroxodisulfates in their saturated polyomponent solutions have been alulated using Eq. () and Eq. (). The obtained results are given in Tab. 1 4 as well. As an be seen, the individual values of the mean ativity oeffiients of the onsidered peroxodisulfates omputed by both mentioned methods are pratially idential in all ases. From the results, it further follows that in the both pseudoternary systems without a ommon ation, the added omponents enlarge the solubility of the rystallizing peroxodisulfate and onsequently derease its ativity oeffiient, beause the produt onst. (see Eq. 4). This behaviour is totally opposite to that observed at the systems M S O 8 H SO 4 H O with M Na or NH 4 (Balej 011b) where the inreasing ontent of H SO 4 aused a derease of the solubility and onsequently an inrease of the ativity oeffiients of the orresponding peroxodisulfate. Very interesting is the mutual omparison of the influene of individual additives on the final effets expressed as the speifi inrease or derease of the Tab. 4. Solubility data and ativity oeffiients in the system Na S O 8 NaNH 4 SO 4 H O. m oexisting solid substane exp Eq. (16) Eq. (17) Eq. () Eq. () 0 C Na S O Na S O Na S O 8 NaNH 4 SO 4 H O 0 C Na S O Na S O Na S O Na S O Na S O Na S O 8 NaNH 4 SO 4 H O 166
5 Tab. 5. Speifi hange of the solubility D and ativity oeffiient D of the rystallizing omponent of the onsidered systems aused by the added substane. system D D 0 C 0 C 0 C 0 C (NH 4 ) S O 8 Na SO 4 H O Na S O 8 (NH 4 ) SO 4 H O (NH 4 ) S O 8 NaNH 4 SO 4 H O ~ 0 ~ 0 Na S O 8 NaNH 4 SO 4 H O solubility Δ and ativity oeffiients Δ of the rystallizing substane formulated as followed and m Se - m D m me D me e 0 (18) (19) where index e denotes eutoni solution. From the obtained results given in Table 5 it follows that the influene of added ammonium sulfate on the solubility of sodium peroxodisulfate is totally different from that of sodium sulfate addition on the solubility of ammonium peroxodisulfate. While in the first ase the solubility inrease of sodium peroxodisulfate with ammonium sulfate addition at 0 C was more than twie higher than at 0 C, the solubility inrease of ammonium peroxodisulfate with sodium sulfate addition was about twie lower at the same onditions. At systems with a ommon ation, the addition of sodium ammonium sulfate to the rystallizing ammonium peroxodisulfate solution aused a quite distint derease of its solubility, with an almost negligible influene of temperature. The addition of the same double sulfate to the rystallizing sodium peroxodisulfate solution also aused its solubility derease, however, in this ase, the effet was more distint and higher at a higher temperature. What about the influene of the added sulfates on the speifi hange of ativity oeffiients of the rystallizing peroxodisulfates, the final effet was always opposite in omparison to their influene on the solubility hange, in aordane with the theoretial expetation. However, it an ommonly be stated that the values of ativity oeffiients of ammonium or sodium peroxodisulfate in their saturated polyomponent solution differ not too muh from those in pure water at the same temperature, the maximum deviation 1 rel. % has been observed at the eutoni solution of the system Na S O 8 (NH 4 ) SO 4 H O at 0 C. Conlusion The presented results brought the evaluation of the previous solubility data of the onversion system NH 4 Na S O 8 H O at 0 C and 0 C for four seleted pseudoternary systems Na S O 8 (NH 4 ) SO 4 H O, (NH 4 ) S O 8 Na SO 4 H O, Na S O 8 NaNH 4 SO 4 H O and (NH 4 ) S O 8 NaNH 4 SO 4 H O in the form of suitable orrelation equations using the relative ativity oeffiient expansion of the rystallizing omponent. The obtained equations have been used for the alulation of mean ativity oeffiients of sodium and ammonium peroxodisulfates in their saturated solutions of the mentioned pseudoternary systems. It was observed that the values of ativity oeffiients of ammonium or sodium peroxodisulfate in their saturated polyomponent solutions differ not too muh from those in pure water. The presented results represent a supplement of previous papers about the same theme (Balej 198, 011a, b). Referenes Balej J (198) Collet. Czeh. Chem. Commun. 47: Balej J (010a) Collet. Czeh. Chem. Commun. 75: 1 1. Balej J (010b) Z. Phys. Chem. 4: Balej J (011a) Ata Chim. Slov. 4: Balej J (011b) Ata Chim. Slov. 4: Balej J, Čížek M, Thumová M (1976) Collet. Czeh. Chem. Commun. 41: Balej J, Thumová M (1976) Collet. Czeh. Chem. Commun. 41: Nývlt J (1977) Solid-Liquid Phase Equilibria, p Aademia, Prague. 167
General Equilibrium. What happens to cause a reaction to come to equilibrium?
 General Equilibrium Chemial Equilibrium Most hemial reations that are enountered are reversible. In other words, they go fairly easily in either the forward or reverse diretions. The thing to remember
General Equilibrium Chemial Equilibrium Most hemial reations that are enountered are reversible. In other words, they go fairly easily in either the forward or reverse diretions. The thing to remember
Millennium Relativity Acceleration Composition. The Relativistic Relationship between Acceleration and Uniform Motion
 Millennium Relativity Aeleration Composition he Relativisti Relationship between Aeleration and niform Motion Copyright 003 Joseph A. Rybzyk Abstrat he relativisti priniples developed throughout the six
Millennium Relativity Aeleration Composition he Relativisti Relationship between Aeleration and niform Motion Copyright 003 Joseph A. Rybzyk Abstrat he relativisti priniples developed throughout the six
arxiv:gr-qc/ v2 6 Feb 2004
 Hubble Red Shift and the Anomalous Aeleration of Pioneer 0 and arxiv:gr-q/0402024v2 6 Feb 2004 Kostadin Trenčevski Faulty of Natural Sienes and Mathematis, P.O.Box 62, 000 Skopje, Maedonia Abstrat It this
Hubble Red Shift and the Anomalous Aeleration of Pioneer 0 and arxiv:gr-q/0402024v2 6 Feb 2004 Kostadin Trenčevski Faulty of Natural Sienes and Mathematis, P.O.Box 62, 000 Skopje, Maedonia Abstrat It this
JF Physical Chemistry JF CH 1101: Introduction to Physical Chemistry.
 JF Physial Chemistry 010-011. JF CH 1101: Introdution to Physial Chemistry. Dr Mike Lyons. Shool of Chemistry Trinity College Dublin. melyons@td.ie A ompendium of past examination questions set on Physial
JF Physial Chemistry 010-011. JF CH 1101: Introdution to Physial Chemistry. Dr Mike Lyons. Shool of Chemistry Trinity College Dublin. melyons@td.ie A ompendium of past examination questions set on Physial
ELECTROCHEMISTRY Lecture/Lession Plan -1
 Chapter 4 ELECTROCHEMISTRY Leture/Lession Plan -1 ELECTROCHEMISTRY 4.1 Conept of eletrohemistry Eletrohemistry is a branh of hemistry where we will study how hemial energy an be transformed into eletrial
Chapter 4 ELECTROCHEMISTRY Leture/Lession Plan -1 ELECTROCHEMISTRY 4.1 Conept of eletrohemistry Eletrohemistry is a branh of hemistry where we will study how hemial energy an be transformed into eletrial
DIGITAL DISTANCE RELAYING SCHEME FOR PARALLEL TRANSMISSION LINES DURING INTER-CIRCUIT FAULTS
 CHAPTER 4 DIGITAL DISTANCE RELAYING SCHEME FOR PARALLEL TRANSMISSION LINES DURING INTER-CIRCUIT FAULTS 4.1 INTRODUCTION Around the world, environmental and ost onsiousness are foring utilities to install
CHAPTER 4 DIGITAL DISTANCE RELAYING SCHEME FOR PARALLEL TRANSMISSION LINES DURING INTER-CIRCUIT FAULTS 4.1 INTRODUCTION Around the world, environmental and ost onsiousness are foring utilities to install
COMBINED PROBE FOR MACH NUMBER, TEMPERATURE AND INCIDENCE INDICATION
 4 TH INTERNATIONAL CONGRESS OF THE AERONAUTICAL SCIENCES COMBINED PROBE FOR MACH NUMBER, TEMPERATURE AND INCIDENCE INDICATION Jiri Nozika*, Josef Adame*, Daniel Hanus** *Department of Fluid Dynamis and
4 TH INTERNATIONAL CONGRESS OF THE AERONAUTICAL SCIENCES COMBINED PROBE FOR MACH NUMBER, TEMPERATURE AND INCIDENCE INDICATION Jiri Nozika*, Josef Adame*, Daniel Hanus** *Department of Fluid Dynamis and
A simple expression for radial distribution functions of pure fluids and mixtures
 A simple expression for radial distribution funtions of pure fluids and mixtures Enrio Matteoli a) Istituto di Chimia Quantistia ed Energetia Moleolare, CNR, Via Risorgimento, 35, 56126 Pisa, Italy G.
A simple expression for radial distribution funtions of pure fluids and mixtures Enrio Matteoli a) Istituto di Chimia Quantistia ed Energetia Moleolare, CNR, Via Risorgimento, 35, 56126 Pisa, Italy G.
Methods of evaluating tests
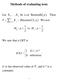 Methods of evaluating tests Let X,, 1 Xn be i.i.d. Bernoulli( p ). Then 5 j= 1 j ( 5, ) T = X Binomial p. We test 1 H : p vs. 1 1 H : p>. We saw that a LRT is 1 if t k* φ ( x ) =. otherwise (t is the observed
Methods of evaluating tests Let X,, 1 Xn be i.i.d. Bernoulli( p ). Then 5 j= 1 j ( 5, ) T = X Binomial p. We test 1 H : p vs. 1 1 H : p>. We saw that a LRT is 1 if t k* φ ( x ) =. otherwise (t is the observed
Measuring & Inducing Neural Activity Using Extracellular Fields I: Inverse systems approach
 Measuring & Induing Neural Ativity Using Extraellular Fields I: Inverse systems approah Keith Dillon Department of Eletrial and Computer Engineering University of California San Diego 9500 Gilman Dr. La
Measuring & Induing Neural Ativity Using Extraellular Fields I: Inverse systems approah Keith Dillon Department of Eletrial and Computer Engineering University of California San Diego 9500 Gilman Dr. La
Assessing the Performance of a BCI: A Task-Oriented Approach
 Assessing the Performane of a BCI: A Task-Oriented Approah B. Dal Seno, L. Mainardi 2, M. Matteui Department of Eletronis and Information, IIT-Unit, Politenio di Milano, Italy 2 Department of Bioengineering,
Assessing the Performane of a BCI: A Task-Oriented Approah B. Dal Seno, L. Mainardi 2, M. Matteui Department of Eletronis and Information, IIT-Unit, Politenio di Milano, Italy 2 Department of Bioengineering,
RESEARCH ON RANDOM FOURIER WAVE-NUMBER SPECTRUM OF FLUCTUATING WIND SPEED
 The Seventh Asia-Paifi Conferene on Wind Engineering, November 8-1, 9, Taipei, Taiwan RESEARCH ON RANDOM FORIER WAVE-NMBER SPECTRM OF FLCTATING WIND SPEED Qi Yan 1, Jie Li 1 Ph D. andidate, Department
The Seventh Asia-Paifi Conferene on Wind Engineering, November 8-1, 9, Taipei, Taiwan RESEARCH ON RANDOM FORIER WAVE-NMBER SPECTRM OF FLCTATING WIND SPEED Qi Yan 1, Jie Li 1 Ph D. andidate, Department
QCLAS Sensor for Purity Monitoring in Medical Gas Supply Lines
 DOI.56/sensoren6/P3. QLAS Sensor for Purity Monitoring in Medial Gas Supply Lines Henrik Zimmermann, Mathias Wiese, Alessandro Ragnoni neoplas ontrol GmbH, Walther-Rathenau-Str. 49a, 7489 Greifswald, Germany
DOI.56/sensoren6/P3. QLAS Sensor for Purity Monitoring in Medial Gas Supply Lines Henrik Zimmermann, Mathias Wiese, Alessandro Ragnoni neoplas ontrol GmbH, Walther-Rathenau-Str. 49a, 7489 Greifswald, Germany
Application of the Dyson-type boson mapping for low-lying electron excited states in molecules
 Prog. Theor. Exp. Phys. 05, 063I0 ( pages DOI: 0.093/ptep/ptv068 Appliation of the Dyson-type boson mapping for low-lying eletron exited states in moleules adao Ohkido, and Makoto Takahashi Teaher-training
Prog. Theor. Exp. Phys. 05, 063I0 ( pages DOI: 0.093/ptep/ptv068 Appliation of the Dyson-type boson mapping for low-lying eletron exited states in moleules adao Ohkido, and Makoto Takahashi Teaher-training
University of Washington Department of Chemistry Chemistry 452/456 Summer Quarter 2011
 Homework Assignment #4: Due at 500 pm Monday 8 July,. University of Washington Department of Chemistry Chemistry 45/456 Summer Quarter 0 ) o a very good approximation, ammonia obeys the Bertholet equation
Homework Assignment #4: Due at 500 pm Monday 8 July,. University of Washington Department of Chemistry Chemistry 45/456 Summer Quarter 0 ) o a very good approximation, ammonia obeys the Bertholet equation
Modeling of Threading Dislocation Density Reduction in Heteroepitaxial Layers
 A. E. Romanov et al.: Threading Disloation Density Redution in Layers (II) 33 phys. stat. sol. (b) 99, 33 (997) Subjet lassifiation: 6.72.C; 68.55.Ln; S5.; S5.2; S7.; S7.2 Modeling of Threading Disloation
A. E. Romanov et al.: Threading Disloation Density Redution in Layers (II) 33 phys. stat. sol. (b) 99, 33 (997) Subjet lassifiation: 6.72.C; 68.55.Ln; S5.; S5.2; S7.; S7.2 Modeling of Threading Disloation
Chemical Engineering Thermodynamics II ( ) 02 - The Molar Gibbs Free Energy & Fugacity of a Pure Component
 Chemial Engineering Thermodynamis II (090533) 0 - The Molar Gibbs Free Energy & Fugaity of a ure Component Dr. Ali Khalaf Al-matar Chemial Engineering Department University of Jordan banihaniali@yahoo.om
Chemial Engineering Thermodynamis II (090533) 0 - The Molar Gibbs Free Energy & Fugaity of a ure Component Dr. Ali Khalaf Al-matar Chemial Engineering Department University of Jordan banihaniali@yahoo.om
COMPLEX INDUCTANCE AND ITS COMPUTER MODELLING
 Journal of ELECTRICAL ENGINEERING, VOL. 53, NO. 1-2, 22, 24 29 COMPLEX INDUCTANCE AND ITS COMPUTER MODELLING Daniel Mayer Bohuš Ulryh The paper introdues the onept of the omplex indutane as a parameter
Journal of ELECTRICAL ENGINEERING, VOL. 53, NO. 1-2, 22, 24 29 COMPLEX INDUCTANCE AND ITS COMPUTER MODELLING Daniel Mayer Bohuš Ulryh The paper introdues the onept of the omplex indutane as a parameter
CRITICAL EXPONENTS TAKING INTO ACCOUNT DYNAMIC SCALING FOR ADSORPTION ON SMALL-SIZE ONE-DIMENSIONAL CLUSTERS
 Russian Physis Journal, Vol. 48, No. 8, 5 CRITICAL EXPONENTS TAKING INTO ACCOUNT DYNAMIC SCALING FOR ADSORPTION ON SMALL-SIZE ONE-DIMENSIONAL CLUSTERS A. N. Taskin, V. N. Udodov, and A. I. Potekaev UDC
Russian Physis Journal, Vol. 48, No. 8, 5 CRITICAL EXPONENTS TAKING INTO ACCOUNT DYNAMIC SCALING FOR ADSORPTION ON SMALL-SIZE ONE-DIMENSIONAL CLUSTERS A. N. Taskin, V. N. Udodov, and A. I. Potekaev UDC
Bending resistance of high performance concrete elements
 High Performane Strutures and Materials IV 89 Bending resistane of high performane onrete elements D. Mestrovi 1 & L. Miulini 1 Faulty of Civil Engineering, University of Zagreb, Croatia Faulty of Civil
High Performane Strutures and Materials IV 89 Bending resistane of high performane onrete elements D. Mestrovi 1 & L. Miulini 1 Faulty of Civil Engineering, University of Zagreb, Croatia Faulty of Civil
Determination of the reaction order
 5/7/07 A quote of the wee (or amel of the wee): Apply yourself. Get all the eduation you an, but then... do something. Don't just stand there, mae it happen. Lee Iaoa Physial Chemistry GTM/5 reation order
5/7/07 A quote of the wee (or amel of the wee): Apply yourself. Get all the eduation you an, but then... do something. Don't just stand there, mae it happen. Lee Iaoa Physial Chemistry GTM/5 reation order
Chapter 13, Chemical Equilibrium
 Chapter 13, Chemial Equilibrium You may have gotten the impression that when 2 reatants mix, the ensuing rxn goes to ompletion. In other words, reatants are onverted ompletely to produts. We will now learn
Chapter 13, Chemial Equilibrium You may have gotten the impression that when 2 reatants mix, the ensuing rxn goes to ompletion. In other words, reatants are onverted ompletely to produts. We will now learn
properties via a simple hydrolysis-based approach
 Eletroni Supplementary Material (ESI) for Journal of Materials Chemistry A This journal is The Royal Soiety of Chemistry 01 Supporting Information Salable synthesis of hollow O nanoubes with unique optial
Eletroni Supplementary Material (ESI) for Journal of Materials Chemistry A This journal is The Royal Soiety of Chemistry 01 Supporting Information Salable synthesis of hollow O nanoubes with unique optial
COPYRIGHTED MATERIAL. Gravimetrics. Key Terms. Key Concepts. Words that can be used as topics in essays: accuracy atomic theory
 8684-X Ch02.F 2/9/01 7:49 AM Page 23 Gravimetris Key Terms Words that an be used as topis in essays: auray atomi theory density empirial formula extensive property frational rystallization heterogeneous
8684-X Ch02.F 2/9/01 7:49 AM Page 23 Gravimetris Key Terms Words that an be used as topis in essays: auray atomi theory density empirial formula extensive property frational rystallization heterogeneous
III. SURFACE PROPERTIES III.A. SURFACE TENSION SURFACE PROPERTIES
 III. SURFACE PROPERTIES III.A. SURFACE TENSION GOAL: To investigate the influene of the solution onentration and/or the kind of the solute on the surfae tension INTRODUCTION Liquids tend to adopt shapes
III. SURFACE PROPERTIES III.A. SURFACE TENSION GOAL: To investigate the influene of the solution onentration and/or the kind of the solute on the surfae tension INTRODUCTION Liquids tend to adopt shapes
Four-dimensional equation of motion for viscous compressible substance with regard to the acceleration field, pressure field and dissipation field
 Four-dimensional equation of motion for visous ompressible substane with regard to the aeleration field, pressure field and dissipation field Sergey G. Fedosin PO box 6488, Sviazeva str. -79, Perm, Russia
Four-dimensional equation of motion for visous ompressible substane with regard to the aeleration field, pressure field and dissipation field Sergey G. Fedosin PO box 6488, Sviazeva str. -79, Perm, Russia
PHASE EQUILIBRIA OF CARBON DIOXIDE AND METHANE GAS-HYDRATES PREDICTED WITH THE MODIFIED ANALYTICAL S-L-V EQUATION OF STATE
 EPJ Web of Conferenes, 010 (01) DOI: 10.1051/epjonf/015010 Owned by the authors, published by EDP Sienes, 01 PHASE EQUILIBRIA OF CARBON DIOXIDE AND METHANE GAS-HYDRATES PREDICTED WITH THE MODIFIED ANALYTICAL
EPJ Web of Conferenes, 010 (01) DOI: 10.1051/epjonf/015010 Owned by the authors, published by EDP Sienes, 01 PHASE EQUILIBRIA OF CARBON DIOXIDE AND METHANE GAS-HYDRATES PREDICTED WITH THE MODIFIED ANALYTICAL
Probabilistic interpretation of «Bath s Law»
 AALS OF GEOPHYSICS, VOL. 45,. 3/4, June/August 2002 Probabilisti interpretation of «Bath s Law» Anna aria Lombardi Istituto azionale di Geofisia e Vulanologia, Roma, Italy Abstrat Assuming that, in a atalog,
AALS OF GEOPHYSICS, VOL. 45,. 3/4, June/August 2002 Probabilisti interpretation of «Bath s Law» Anna aria Lombardi Istituto azionale di Geofisia e Vulanologia, Roma, Italy Abstrat Assuming that, in a atalog,
Improvements in the Modeling of the Self-ignition of Tetrafluoroethylene
 Exerpt from the Proeedings of the OMSOL onferene 010 Paris Improvements in the Modeling of the Self-ignition of Tetrafluoroethylene M. Bekmann-Kluge 1 *,. errero 1, V. Shröder 1, A. Aikalin and J. Steinbah
Exerpt from the Proeedings of the OMSOL onferene 010 Paris Improvements in the Modeling of the Self-ignition of Tetrafluoroethylene M. Bekmann-Kluge 1 *,. errero 1, V. Shröder 1, A. Aikalin and J. Steinbah
Possibility of Refuse Derived Fuel Fire Inception by Spontaneous Ignition
 Possibility of Refuse Derived Fuel Fire Ineption by Spontaneous Ignition Lijing Gao 1, Takashi Tsuruda 1, Takeshi Suzuki 1, Yoshio Ogawa 1, Chihong Liao 1 and Yuko Saso 1 1 National Researh Institute of
Possibility of Refuse Derived Fuel Fire Ineption by Spontaneous Ignition Lijing Gao 1, Takashi Tsuruda 1, Takeshi Suzuki 1, Yoshio Ogawa 1, Chihong Liao 1 and Yuko Saso 1 1 National Researh Institute of
Effect of Different Types of Promoters on Bed Expansion in a Gas-Solid Fluidized Bed with Varying Distributor Open Areas
 Journal of Chemial Engineering of Japan, Vol. 35, No. 7, pp. 681 686, 2002 Short Communiation Effet of Different Types of Promoters on Bed Expansion in a Gas-Solid Fluidized Bed with Varying Distributor
Journal of Chemial Engineering of Japan, Vol. 35, No. 7, pp. 681 686, 2002 Short Communiation Effet of Different Types of Promoters on Bed Expansion in a Gas-Solid Fluidized Bed with Varying Distributor
KINETICS OF IRON OXIDE DIRECT REDUCTION BY COAL E.R. ABRIL 1
 KINETICS OF IRON OXIDE DIRECT REDUCTION BY COAL E.R. ABRIL 1 CIMM- Av.Velez Sarsfield 1561 C.P.5000 Córdoba, Argentina. aabril@intiemor.gov.ar Abstrat - A new interpretation to the kinetis of iron oxide
KINETICS OF IRON OXIDE DIRECT REDUCTION BY COAL E.R. ABRIL 1 CIMM- Av.Velez Sarsfield 1561 C.P.5000 Córdoba, Argentina. aabril@intiemor.gov.ar Abstrat - A new interpretation to the kinetis of iron oxide
The Complete Energy Translations in the Detailed. Decay Process of Baryonic Sub-Atomic Particles. P.G.Bass.
 The Complete Energy Translations in the Detailed Deay Proess of Baryoni Su-Atomi Partiles. [4] P.G.Bass. PGBass P12 Version 1..3 www.relativitydomains.om August 218 Astrat. This is the final paper on the
The Complete Energy Translations in the Detailed Deay Proess of Baryoni Su-Atomi Partiles. [4] P.G.Bass. PGBass P12 Version 1..3 www.relativitydomains.om August 218 Astrat. This is the final paper on the
Determination of the Aerodynamic Characteristics of Flying Vehicles Using Method Large Eddy Simulation with Software ANSYS
 Automation, Control and Intelligent Systems 15; 3(6): 118-13 Published online Deember, 15 (http://www.sienepublishinggroup.om//ais) doi: 1.11648/.ais.1536.14 ISSN: 38-5583 (Print); ISSN: 38-5591 (Online)
Automation, Control and Intelligent Systems 15; 3(6): 118-13 Published online Deember, 15 (http://www.sienepublishinggroup.om//ais) doi: 1.11648/.ais.1536.14 ISSN: 38-5583 (Print); ISSN: 38-5591 (Online)
Copyright 2018 Society of Photo-Optical Instrumentation Engineers (SPIE). One print or electronic copy may be made for personal use only.
 Copyright 018 Soiety of Photo-Optial Instrumentation Engineers (SPIE) One print or eletroni opy may be made for personal use only Systemati reprodution and distribution, dupliation of any material in this
Copyright 018 Soiety of Photo-Optial Instrumentation Engineers (SPIE) One print or eletroni opy may be made for personal use only Systemati reprodution and distribution, dupliation of any material in this
MultiPhysics Analysis of Trapped Field in Multi-Layer YBCO Plates
 Exerpt from the Proeedings of the COMSOL Conferene 9 Boston MultiPhysis Analysis of Trapped Field in Multi-Layer YBCO Plates Philippe. Masson Advaned Magnet Lab *7 Main Street, Bldg. #4, Palm Bay, Fl-95,
Exerpt from the Proeedings of the COMSOL Conferene 9 Boston MultiPhysis Analysis of Trapped Field in Multi-Layer YBCO Plates Philippe. Masson Advaned Magnet Lab *7 Main Street, Bldg. #4, Palm Bay, Fl-95,
MATHEMATICAL AND NUMERICAL BASIS OF BINARY ALLOY SOLIDIFICATION MODELS WITH SUBSTITUTE THERMAL CAPACITY. PART II
 Journal of Applied Mathematis and Computational Mehanis 2014, 13(2), 141-147 MATHEMATICA AND NUMERICA BAI OF BINARY AOY OIDIFICATION MODE WITH UBTITUTE THERMA CAPACITY. PART II Ewa Węgrzyn-krzypzak 1,
Journal of Applied Mathematis and Computational Mehanis 2014, 13(2), 141-147 MATHEMATICA AND NUMERICA BAI OF BINARY AOY OIDIFICATION MODE WITH UBTITUTE THERMA CAPACITY. PART II Ewa Węgrzyn-krzypzak 1,
The Second Postulate of Euclid and the Hyperbolic Geometry
 1 The Seond Postulate of Eulid and the Hyperboli Geometry Yuriy N. Zayko Department of Applied Informatis, Faulty of Publi Administration, Russian Presidential Aademy of National Eonomy and Publi Administration,
1 The Seond Postulate of Eulid and the Hyperboli Geometry Yuriy N. Zayko Department of Applied Informatis, Faulty of Publi Administration, Russian Presidential Aademy of National Eonomy and Publi Administration,
23.1 Tuning controllers, in the large view Quoting from Section 16.7:
 Lesson 23. Tuning a real ontroller - modeling, proess identifiation, fine tuning 23.0 Context We have learned to view proesses as dynami systems, taking are to identify their input, intermediate, and output
Lesson 23. Tuning a real ontroller - modeling, proess identifiation, fine tuning 23.0 Context We have learned to view proesses as dynami systems, taking are to identify their input, intermediate, and output
The Effectiveness of the Linear Hull Effect
 The Effetiveness of the Linear Hull Effet S. Murphy Tehnial Report RHUL MA 009 9 6 Otober 009 Department of Mathematis Royal Holloway, University of London Egham, Surrey TW0 0EX, England http://www.rhul.a.uk/mathematis/tehreports
The Effetiveness of the Linear Hull Effet S. Murphy Tehnial Report RHUL MA 009 9 6 Otober 009 Department of Mathematis Royal Holloway, University of London Egham, Surrey TW0 0EX, England http://www.rhul.a.uk/mathematis/tehreports
An Improved Model for Calculating Heats of Dilution and Equilibrium Constants for High Temperature Aqueous Electrolyte Solutions
 Brigham Young University BYU SholarsArhive All Theses and Dissertations 2007-01-08 An Improved Model for Calulating Heats of Dilution and Equilibrium Constants for High Temperature Aqueous Eletrolyte Solutions
Brigham Young University BYU SholarsArhive All Theses and Dissertations 2007-01-08 An Improved Model for Calulating Heats of Dilution and Equilibrium Constants for High Temperature Aqueous Eletrolyte Solutions
Wavetech, LLC. Ultrafast Pulses and GVD. John O Hara Created: Dec. 6, 2013
 Ultrafast Pulses and GVD John O Hara Created: De. 6, 3 Introdution This doument overs the basi onepts of group veloity dispersion (GVD) and ultrafast pulse propagation in an optial fiber. Neessarily, it
Ultrafast Pulses and GVD John O Hara Created: De. 6, 3 Introdution This doument overs the basi onepts of group veloity dispersion (GVD) and ultrafast pulse propagation in an optial fiber. Neessarily, it
Taste for variety and optimum product diversity in an open economy
 Taste for variety and optimum produt diversity in an open eonomy Javier Coto-Martínez City University Paul Levine University of Surrey Otober 0, 005 María D.C. Garía-Alonso University of Kent Abstrat We
Taste for variety and optimum produt diversity in an open eonomy Javier Coto-Martínez City University Paul Levine University of Surrey Otober 0, 005 María D.C. Garía-Alonso University of Kent Abstrat We
FINITE WORD LENGTH EFFECTS IN DSP
 FINITE WORD LENGTH EFFECTS IN DSP PREPARED BY GUIDED BY Snehal Gor Dr. Srianth T. ABSTRACT We now that omputers store numbers not with infinite preision but rather in some approximation that an be paed
FINITE WORD LENGTH EFFECTS IN DSP PREPARED BY GUIDED BY Snehal Gor Dr. Srianth T. ABSTRACT We now that omputers store numbers not with infinite preision but rather in some approximation that an be paed
Complexity of Regularization RBF Networks
 Complexity of Regularization RBF Networks Mark A Kon Department of Mathematis and Statistis Boston University Boston, MA 02215 mkon@buedu Leszek Plaskota Institute of Applied Mathematis University of Warsaw
Complexity of Regularization RBF Networks Mark A Kon Department of Mathematis and Statistis Boston University Boston, MA 02215 mkon@buedu Leszek Plaskota Institute of Applied Mathematis University of Warsaw
Controller Design Based on Transient Response Criteria. Chapter 12 1
 Controller Design Based on Transient Response Criteria Chapter 12 1 Desirable Controller Features 0. Stable 1. Quik responding 2. Adequate disturbane rejetion 3. Insensitive to model, measurement errors
Controller Design Based on Transient Response Criteria Chapter 12 1 Desirable Controller Features 0. Stable 1. Quik responding 2. Adequate disturbane rejetion 3. Insensitive to model, measurement errors
Angular Distribution of Photoelectrons during Irradiation of Metal Surface by Electromagnetic Waves
 Journal of Modern Physis, 0,, 780-786 doi:0436/jmp0809 Published Online August 0 (http://wwwsirporg/journal/jmp) Angular Distribution of Photoeletrons during Irradiation of Metal Surfae by letromagneti
Journal of Modern Physis, 0,, 780-786 doi:0436/jmp0809 Published Online August 0 (http://wwwsirporg/journal/jmp) Angular Distribution of Photoeletrons during Irradiation of Metal Surfae by letromagneti
Duct Acoustics. Chap.4 Duct Acoustics. Plane wave
 Chap.4 Dut Aoustis Dut Aoustis Plane wave A sound propagation in pipes with different ross-setional area f the wavelength of sound is large in omparison with the diameter of the pipe the sound propagates
Chap.4 Dut Aoustis Dut Aoustis Plane wave A sound propagation in pipes with different ross-setional area f the wavelength of sound is large in omparison with the diameter of the pipe the sound propagates
Sensitivity Analysis in Markov Networks
 Sensitivity Analysis in Markov Networks Hei Chan and Adnan Darwihe Computer Siene Department University of California, Los Angeles Los Angeles, CA 90095 {hei,darwihe}@s.ula.edu Abstrat This paper explores
Sensitivity Analysis in Markov Networks Hei Chan and Adnan Darwihe Computer Siene Department University of California, Los Angeles Los Angeles, CA 90095 {hei,darwihe}@s.ula.edu Abstrat This paper explores
Chapter 14. The Concept of Equilibrium and the Equilibrium Constant. We have for the most part depicted reactions as going one way.
 Chapter 14 The Conept of Equilibrium and the Equilibrium Constant In hapter 1 we dealt with Physial Equilibrium Physial Changes HO 2 (l) HO 2 (g) In hapter 14 we will learn about Chemial Equilibrium. We
Chapter 14 The Conept of Equilibrium and the Equilibrium Constant In hapter 1 we dealt with Physial Equilibrium Physial Changes HO 2 (l) HO 2 (g) In hapter 14 we will learn about Chemial Equilibrium. We
Process engineers are often faced with the task of
 Fluids and Solids Handling Eliminate Iteration from Flow Problems John D. Barry Middough, In. This artile introdues a novel approah to solving flow and pipe-sizing problems based on two new dimensionless
Fluids and Solids Handling Eliminate Iteration from Flow Problems John D. Barry Middough, In. This artile introdues a novel approah to solving flow and pipe-sizing problems based on two new dimensionless
An iterative least-square method suitable for solving large sparse matrices
 An iteratie least-square method suitable for soling large sparse matries By I. M. Khabaza The purpose of this paper is to report on the results of numerial experiments with an iteratie least-square method
An iteratie least-square method suitable for soling large sparse matries By I. M. Khabaza The purpose of this paper is to report on the results of numerial experiments with an iteratie least-square method
Final Exam: know your section, bring your ID!
 Chapter 15: Equilibrium Part 1 Read: BLB 15.1 3 HW: BLB 15:13,14, 21 Supplemental 15:1 4 Know: Chemial Equilibrium Catalysts Equilibrium Constant Equilibrium onstant expression Homogeneous/Heterogeneous
Chapter 15: Equilibrium Part 1 Read: BLB 15.1 3 HW: BLB 15:13,14, 21 Supplemental 15:1 4 Know: Chemial Equilibrium Catalysts Equilibrium Constant Equilibrium onstant expression Homogeneous/Heterogeneous
Supporting Information
 Supporting Information Olsman and Goentoro 10.1073/pnas.1601791113 SI Materials Analysis of the Sensitivity and Error Funtions. We now define the sensitivity funtion Sð, «0 Þ, whih summarizes the steepness
Supporting Information Olsman and Goentoro 10.1073/pnas.1601791113 SI Materials Analysis of the Sensitivity and Error Funtions. We now define the sensitivity funtion Sð, «0 Þ, whih summarizes the steepness
Temperature Control of Batch Suspension Polyvinyl Chloride Reactors
 1285 A publiation of CHEMICAL ENGINEERING TRANSACTIONS VOL. 39, 2014 Guest Editors: Petar Sabev Varbanov, Jiří Jaromír Klemeš, Peng Yen Liew, Jun Yow Yong Copyright 2014, AIDIC Servizi S.r.l., ISBN 978-88-95608-30-3;
1285 A publiation of CHEMICAL ENGINEERING TRANSACTIONS VOL. 39, 2014 Guest Editors: Petar Sabev Varbanov, Jiří Jaromír Klemeš, Peng Yen Liew, Jun Yow Yong Copyright 2014, AIDIC Servizi S.r.l., ISBN 978-88-95608-30-3;
The First Principle of Thermodynamics under Relativistic Conditions and Temperature
 New Horizons in Mathematial Physis, Vol., No., September 7 https://dx.doi.org/.66/nhmp.7. 37 he First Priniple of hermodynamis under Relativisti Conditions and emperature Emil Veitsman Independent Researher
New Horizons in Mathematial Physis, Vol., No., September 7 https://dx.doi.org/.66/nhmp.7. 37 he First Priniple of hermodynamis under Relativisti Conditions and emperature Emil Veitsman Independent Researher
UTC. Engineering 329. Proportional Controller Design. Speed System. John Beverly. Green Team. John Beverly Keith Skiles John Barker.
 UTC Engineering 329 Proportional Controller Design for Speed System By John Beverly Green Team John Beverly Keith Skiles John Barker 24 Mar 2006 Introdution This experiment is intended test the variable
UTC Engineering 329 Proportional Controller Design for Speed System By John Beverly Green Team John Beverly Keith Skiles John Barker 24 Mar 2006 Introdution This experiment is intended test the variable
Coastal Engineering 64 (2012) Contents lists available at SciVerse ScienceDirect. Coastal Engineering
 Coastal Engineering 64 (2012) 87 101 Contents lists available at SiVerse SieneDiret Coastal Engineering journal homepage: www.elsevier.om/loate/oastaleng Probability distribution of individual wave overtopping
Coastal Engineering 64 (2012) 87 101 Contents lists available at SiVerse SieneDiret Coastal Engineering journal homepage: www.elsevier.om/loate/oastaleng Probability distribution of individual wave overtopping
THEORETICAL PROBLEM No. 3 WHY ARE STARS SO LARGE?
 THEORETICAL PROBLEM No. 3 WHY ARE STARS SO LARGE? The stars are spheres of hot gas. Most of them shine beause they are fusing hydrogen into helium in their entral parts. In this problem we use onepts of
THEORETICAL PROBLEM No. 3 WHY ARE STARS SO LARGE? The stars are spheres of hot gas. Most of them shine beause they are fusing hydrogen into helium in their entral parts. In this problem we use onepts of
EFFECTIVE STRESS LAW FOR THE PERMEABILITY OF CLAY-RICH SANDSTONES
 SCA22-5 1/6 EFFECTIVE STRESS LAW FOR THE PERMEABILITY OF CLAY-RICH SANDSTONES Widad Al-Wardy and Robert W. Zimmerman Department of Earth Siene and Engineering Imperial College of Siene, Tehnology and Mediine
SCA22-5 1/6 EFFECTIVE STRESS LAW FOR THE PERMEABILITY OF CLAY-RICH SANDSTONES Widad Al-Wardy and Robert W. Zimmerman Department of Earth Siene and Engineering Imperial College of Siene, Tehnology and Mediine
MODELING MATTER AT NANOSCALES. 4. Introduction to quantum treatments Eigenvectors and eigenvalues of a matrix
 MODELING MATTER AT NANOSCALES 4 Introdution to quantum treatments 403 Eigenvetors and eigenvalues of a matrix Simultaneous equations in the variational method The problem of simultaneous equations in the
MODELING MATTER AT NANOSCALES 4 Introdution to quantum treatments 403 Eigenvetors and eigenvalues of a matrix Simultaneous equations in the variational method The problem of simultaneous equations in the
( ) ( ) Volumetric Properties of Pure Fluids, part 4. The generic cubic equation of state:
 CE304, Spring 2004 Leture 6 Volumetri roperties of ure Fluids, part 4 The generi ubi equation of state: There are many possible equations of state (and many have been proposed) that have the same general
CE304, Spring 2004 Leture 6 Volumetri roperties of ure Fluids, part 4 The generi ubi equation of state: There are many possible equations of state (and many have been proposed) that have the same general
Line Radiative Transfer
 http://www.v.nrao.edu/ourse/astr534/ineradxfer.html ine Radiative Transfer Einstein Coeffiients We used armor's equation to estimate the spontaneous emission oeffiients A U for À reombination lines. A
http://www.v.nrao.edu/ourse/astr534/ineradxfer.html ine Radiative Transfer Einstein Coeffiients We used armor's equation to estimate the spontaneous emission oeffiients A U for À reombination lines. A
Inter-fibre contacts in random fibrous materials: experimental verification of theoretical dependence on porosity and fibre width
 J Mater Si (2006) 41:8377 8381 DOI 10.1007/s10853-006-0889-7 LETTER Inter-fibre ontats in random fibrous materials: experimental verifiation of theoretial dependene on porosity and fibre width W. J. Bathelor
J Mater Si (2006) 41:8377 8381 DOI 10.1007/s10853-006-0889-7 LETTER Inter-fibre ontats in random fibrous materials: experimental verifiation of theoretial dependene on porosity and fibre width W. J. Bathelor
Lab.6. Extraction. Upper phase. water. Bottom phase. chloroform
 Lab.6. Extration Key words: Solubility, immisible solvent, partition solutes in two phase system, distribution onstant, partition neutral ompound, partition aids and basis, fators (ph, temperature), partition
Lab.6. Extration Key words: Solubility, immisible solvent, partition solutes in two phase system, distribution onstant, partition neutral ompound, partition aids and basis, fators (ph, temperature), partition
A Time-Dependent Model For Predicting The Response Of A Horizontally Loaded Pile Embedded In A Layered Transversely Isotropic Saturated Soil
 IOSR Journal of Mehanial and Civil Engineering (IOSR-JMCE) e-issn: 2278-1684,p-ISSN: 232-334X, Volume 16, Issue 2 Ser. I (Mar. - Apr. 219), PP 48-53 www.iosrjournals.org A Time-Dependent Model For Prediting
IOSR Journal of Mehanial and Civil Engineering (IOSR-JMCE) e-issn: 2278-1684,p-ISSN: 232-334X, Volume 16, Issue 2 Ser. I (Mar. - Apr. 219), PP 48-53 www.iosrjournals.org A Time-Dependent Model For Prediting
Wave Propagation through Random Media
 Chapter 3. Wave Propagation through Random Media 3. Charateristis of Wave Behavior Sound propagation through random media is the entral part of this investigation. This hapter presents a frame of referene
Chapter 3. Wave Propagation through Random Media 3. Charateristis of Wave Behavior Sound propagation through random media is the entral part of this investigation. This hapter presents a frame of referene
Simplified Buckling Analysis of Skeletal Structures
 Simplified Bukling Analysis of Skeletal Strutures B.A. Izzuddin 1 ABSRAC A simplified approah is proposed for bukling analysis of skeletal strutures, whih employs a rotational spring analogy for the formulation
Simplified Bukling Analysis of Skeletal Strutures B.A. Izzuddin 1 ABSRAC A simplified approah is proposed for bukling analysis of skeletal strutures, whih employs a rotational spring analogy for the formulation
Non-Markovian study of the relativistic magnetic-dipole spontaneous emission process of hydrogen-like atoms
 NSTTUTE OF PHYSCS PUBLSHNG JOURNAL OF PHYSCS B: ATOMC, MOLECULAR AND OPTCAL PHYSCS J. Phys. B: At. Mol. Opt. Phys. 39 ) 7 85 doi:.88/953-75/39/8/ Non-Markovian study of the relativisti magneti-dipole spontaneous
NSTTUTE OF PHYSCS PUBLSHNG JOURNAL OF PHYSCS B: ATOMC, MOLECULAR AND OPTCAL PHYSCS J. Phys. B: At. Mol. Opt. Phys. 39 ) 7 85 doi:.88/953-75/39/8/ Non-Markovian study of the relativisti magneti-dipole spontaneous
Supplementary information for: All-optical signal processing using dynamic Brillouin gratings
 Supplementary information for: All-optial signal proessing using dynami Brillouin gratings Maro Santagiustina, Sanghoon Chin 2, Niolay Primerov 2, Leonora Ursini, Lu Thévena 2 Department of Information
Supplementary information for: All-optial signal proessing using dynami Brillouin gratings Maro Santagiustina, Sanghoon Chin 2, Niolay Primerov 2, Leonora Ursini, Lu Thévena 2 Department of Information
A model for measurement of the states in a coupled-dot qubit
 A model for measurement of the states in a oupled-dot qubit H B Sun and H M Wiseman Centre for Quantum Computer Tehnology Centre for Quantum Dynamis Griffith University Brisbane 4 QLD Australia E-mail:
A model for measurement of the states in a oupled-dot qubit H B Sun and H M Wiseman Centre for Quantum Computer Tehnology Centre for Quantum Dynamis Griffith University Brisbane 4 QLD Australia E-mail:
The gravitational phenomena without the curved spacetime
 The gravitational phenomena without the urved spaetime Mirosław J. Kubiak Abstrat: In this paper was presented a desription of the gravitational phenomena in the new medium, different than the urved spaetime,
The gravitational phenomena without the urved spaetime Mirosław J. Kubiak Abstrat: In this paper was presented a desription of the gravitational phenomena in the new medium, different than the urved spaetime,
Electromagnetic radiation of the travelling spin wave propagating in an antiferromagnetic plate. Exact solution.
 arxiv:physis/99536v1 [physis.lass-ph] 15 May 1999 Eletromagneti radiation of the travelling spin wave propagating in an antiferromagneti plate. Exat solution. A.A.Zhmudsky November 19, 16 Abstrat The exat
arxiv:physis/99536v1 [physis.lass-ph] 15 May 1999 Eletromagneti radiation of the travelling spin wave propagating in an antiferromagneti plate. Exat solution. A.A.Zhmudsky November 19, 16 Abstrat The exat
Likelihood-confidence intervals for quantiles in Extreme Value Distributions
 Likelihood-onfidene intervals for quantiles in Extreme Value Distributions A. Bolívar, E. Díaz-Franés, J. Ortega, and E. Vilhis. Centro de Investigaión en Matemátias; A.P. 42, Guanajuato, Gto. 36; Méxio
Likelihood-onfidene intervals for quantiles in Extreme Value Distributions A. Bolívar, E. Díaz-Franés, J. Ortega, and E. Vilhis. Centro de Investigaión en Matemátias; A.P. 42, Guanajuato, Gto. 36; Méxio
Physics 218, Spring February 2004
 Physis 8 Spring 004 8 February 004 Today in Physis 8: dispersion Motion of bound eletrons in matter and the frequeny dependene of the dieletri onstant Dispersion relations Ordinary and anomalous dispersion
Physis 8 Spring 004 8 February 004 Today in Physis 8: dispersion Motion of bound eletrons in matter and the frequeny dependene of the dieletri onstant Dispersion relations Ordinary and anomalous dispersion
Counting Idempotent Relations
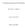 Counting Idempotent Relations Beriht-Nr. 2008-15 Florian Kammüller ISSN 1436-9915 2 Abstrat This artile introdues and motivates idempotent relations. It summarizes haraterizations of idempotents and their
Counting Idempotent Relations Beriht-Nr. 2008-15 Florian Kammüller ISSN 1436-9915 2 Abstrat This artile introdues and motivates idempotent relations. It summarizes haraterizations of idempotents and their
POROUS CARBON PARTICLE COMBUSTION IN AIR
 MCS 7 Chia Laguna, Cagliari, Sardinia, taly, 11-15, 11 POOUS CABON PATCLE COMBUSTON N A V. M. Gremyahkin grema@ipmnet.ru nstitute for Problems in Mehanis, AS, Mosow, ussia Abstrat Theoretial investigation
MCS 7 Chia Laguna, Cagliari, Sardinia, taly, 11-15, 11 POOUS CABON PATCLE COMBUSTON N A V. M. Gremyahkin grema@ipmnet.ru nstitute for Problems in Mehanis, AS, Mosow, ussia Abstrat Theoretial investigation
Part G-4: Sample Exams
 Part G-4: Sample Exams 1 Cairo University M.S.: Eletronis Cooling Faulty of Engineering Final Exam (Sample 1) Mehanial Power Engineering Dept. Time allowed 2 Hours Solve as muh as you an. 1. A heat sink
Part G-4: Sample Exams 1 Cairo University M.S.: Eletronis Cooling Faulty of Engineering Final Exam (Sample 1) Mehanial Power Engineering Dept. Time allowed 2 Hours Solve as muh as you an. 1. A heat sink
The universal model of error of active power measuring channel
 7 th Symposium EKO TC 4 3 rd Symposium EKO TC 9 and 5 th WADC Workshop nstrumentation for the CT Era Sept. 8-2 Kosie Slovakia The universal model of error of ative power measuring hannel Boris Stogny Evgeny
7 th Symposium EKO TC 4 3 rd Symposium EKO TC 9 and 5 th WADC Workshop nstrumentation for the CT Era Sept. 8-2 Kosie Slovakia The universal model of error of ative power measuring hannel Boris Stogny Evgeny
Discrete Bessel functions and partial difference equations
 Disrete Bessel funtions and partial differene equations Antonín Slavík Charles University, Faulty of Mathematis and Physis, Sokolovská 83, 186 75 Praha 8, Czeh Republi E-mail: slavik@karlin.mff.uni.z Abstrat
Disrete Bessel funtions and partial differene equations Antonín Slavík Charles University, Faulty of Mathematis and Physis, Sokolovská 83, 186 75 Praha 8, Czeh Republi E-mail: slavik@karlin.mff.uni.z Abstrat
The simulation analysis of the bridge rectifier continuous operation in AC circuit
 Computer Appliations in Eletrial Engineering Vol. 4 6 DOI 8/j.8-448.6. The simulation analysis of the bridge retifier ontinuous operation in AC iruit Mirosław Wiślik, Paweł Strząbała Kiele University of
Computer Appliations in Eletrial Engineering Vol. 4 6 DOI 8/j.8-448.6. The simulation analysis of the bridge retifier ontinuous operation in AC iruit Mirosław Wiślik, Paweł Strząbała Kiele University of
What s New in ChemSep TM 6.8
 What s New in ChemSep TM 6.8 January 2011 (Updated Marh 2011) Harry Kooijman and Ross Taylor In this doument we identify and desribe the most important new features in ChemSep. 1. New: GUI an diretly load
What s New in ChemSep TM 6.8 January 2011 (Updated Marh 2011) Harry Kooijman and Ross Taylor In this doument we identify and desribe the most important new features in ChemSep. 1. New: GUI an diretly load
Model-based mixture discriminant analysis an experimental study
 Model-based mixture disriminant analysis an experimental study Zohar Halbe and Mayer Aladjem Department of Eletrial and Computer Engineering, Ben-Gurion University of the Negev P.O.Box 653, Beer-Sheva,
Model-based mixture disriminant analysis an experimental study Zohar Halbe and Mayer Aladjem Department of Eletrial and Computer Engineering, Ben-Gurion University of the Negev P.O.Box 653, Beer-Sheva,
A Cubic Equation of State for Reservoir Fluids
 6th IASME/WSEAS International Conferene on HEAT TRANSFER, THERMAL ENGINEERING and ENVIRONMENT (HTE'08) Rhodes, Greee, August 0-, 008 A Cubi Equation of State for Reservoir Fluids AMIR AHMAD SHIRAZIMANESH,
6th IASME/WSEAS International Conferene on HEAT TRANSFER, THERMAL ENGINEERING and ENVIRONMENT (HTE'08) Rhodes, Greee, August 0-, 008 A Cubi Equation of State for Reservoir Fluids AMIR AHMAD SHIRAZIMANESH,
I. Aim of the experiment
 Task VIII TRAUBE S RULE I. Aim of the eperiment The purpose of this task is to verify the Traube s rule for a homologous series of apillary ative substane solutions (i.e. alohols or arboyli aids) on the
Task VIII TRAUBE S RULE I. Aim of the eperiment The purpose of this task is to verify the Traube s rule for a homologous series of apillary ative substane solutions (i.e. alohols or arboyli aids) on the
Simple Considerations on the Cosmological Redshift
 Apeiron, Vol. 5, No. 3, July 8 35 Simple Considerations on the Cosmologial Redshift José Franiso Garía Juliá C/ Dr. Maro Mereniano, 65, 5. 465 Valenia (Spain) E-mail: jose.garia@dival.es Generally, the
Apeiron, Vol. 5, No. 3, July 8 35 Simple Considerations on the Cosmologial Redshift José Franiso Garía Juliá C/ Dr. Maro Mereniano, 65, 5. 465 Valenia (Spain) E-mail: jose.garia@dival.es Generally, the
Sensitivity analysis for linear optimization problem with fuzzy data in the objective function
 Sensitivity analysis for linear optimization problem with fuzzy data in the objetive funtion Stephan Dempe, Tatiana Starostina May 5, 2004 Abstrat Linear programming problems with fuzzy oeffiients in the
Sensitivity analysis for linear optimization problem with fuzzy data in the objetive funtion Stephan Dempe, Tatiana Starostina May 5, 2004 Abstrat Linear programming problems with fuzzy oeffiients in the
Chapter 15 Equilibrium. Reversible Reactions & Equilibrium. Reversible Reactions & Equilibrium. Reversible Reactions & Equilibrium 2/3/2014
 Amount of reatant/produt //01 quilibrium in Chemial Reations Lets look bak at our hypothetial reation from the kinetis hapter. A + B C Chapter 15 quilibrium [A] Why doesn t the onentration of A ever go
Amount of reatant/produt //01 quilibrium in Chemial Reations Lets look bak at our hypothetial reation from the kinetis hapter. A + B C Chapter 15 quilibrium [A] Why doesn t the onentration of A ever go
Coding for Random Projections and Approximate Near Neighbor Search
 Coding for Random Projetions and Approximate Near Neighbor Searh Ping Li Department of Statistis & Biostatistis Department of Computer Siene Rutgers University Pisataay, NJ 8854, USA pingli@stat.rutgers.edu
Coding for Random Projetions and Approximate Near Neighbor Searh Ping Li Department of Statistis & Biostatistis Department of Computer Siene Rutgers University Pisataay, NJ 8854, USA pingli@stat.rutgers.edu
Case I: 2 users In case of 2 users, the probability of error for user 1 was earlier derived to be 2 A1
 MUTLIUSER DETECTION (Letures 9 and 0) 6:33:546 Wireless Communiations Tehnologies Instrutor: Dr. Narayan Mandayam Summary By Shweta Shrivastava (shwetash@winlab.rutgers.edu) bstrat This artile ontinues
MUTLIUSER DETECTION (Letures 9 and 0) 6:33:546 Wireless Communiations Tehnologies Instrutor: Dr. Narayan Mandayam Summary By Shweta Shrivastava (shwetash@winlab.rutgers.edu) bstrat This artile ontinues
Berry s phase for coherent states of Landau levels
 Berry s phase for oherent states of Landau levels Wen-Long Yang 1 and Jing-Ling Chen 1, 1 Theoretial Physis Division, Chern Institute of Mathematis, Nankai University, Tianjin 300071, P.R.China Adiabati
Berry s phase for oherent states of Landau levels Wen-Long Yang 1 and Jing-Ling Chen 1, 1 Theoretial Physis Division, Chern Institute of Mathematis, Nankai University, Tianjin 300071, P.R.China Adiabati
Chapter 15: Chemical Equilibrium
 Chapter 5: Chemial Equilibrium ahoot!. At eq, the rate of the forward reation is the rate of the reverse reation. equal to, slower than, faster than, the reverse of. Selet the statement that BEST desribes
Chapter 5: Chemial Equilibrium ahoot!. At eq, the rate of the forward reation is the rate of the reverse reation. equal to, slower than, faster than, the reverse of. Selet the statement that BEST desribes
Part II SECTION I : One or more options correct Type
 [1] JEE Advaned 2013/ Paper -2 Part II SECTION I : One or more options orret Type This setion ontains 8 multiple hoie questions. Eah question has four hoies (A), (B), (C) and (D), out of whih ONE or MORE
[1] JEE Advaned 2013/ Paper -2 Part II SECTION I : One or more options orret Type This setion ontains 8 multiple hoie questions. Eah question has four hoies (A), (B), (C) and (D), out of whih ONE or MORE
FNSN 2 - Chapter 11 Searches and limits
 FS 2 - Chapter 11 Searhes and limits Paolo Bagnaia last mod. 19-May-17 11 Searhes and limits 1. Probability 2. Searhes and limits 3. Limits 4. Maximum likelihood 5. Interpretation of results methods ommonly
FS 2 - Chapter 11 Searhes and limits Paolo Bagnaia last mod. 19-May-17 11 Searhes and limits 1. Probability 2. Searhes and limits 3. Limits 4. Maximum likelihood 5. Interpretation of results methods ommonly
Critical Reflections on the Hafele and Keating Experiment
 Critial Refletions on the Hafele and Keating Experiment W.Nawrot In 1971 Hafele and Keating performed their famous experiment whih onfirmed the time dilation predited by SRT by use of marosopi loks. As
Critial Refletions on the Hafele and Keating Experiment W.Nawrot In 1971 Hafele and Keating performed their famous experiment whih onfirmed the time dilation predited by SRT by use of marosopi loks. As
3 Tidal systems modelling: ASMITA model
 3 Tidal systems modelling: ASMITA model 3.1 Introdution For many pratial appliations, simulation and predition of oastal behaviour (morphologial development of shorefae, beahes and dunes) at a ertain level
3 Tidal systems modelling: ASMITA model 3.1 Introdution For many pratial appliations, simulation and predition of oastal behaviour (morphologial development of shorefae, beahes and dunes) at a ertain level
Investigation of the de Broglie-Einstein velocity equation s. universality in the context of the Davisson-Germer experiment. Yusuf Z.
 Investigation of the de Broglie-instein veloity equation s universality in the ontext of the Davisson-Germer experiment Yusuf Z. UMUL Canaya University, letroni and Communiation Dept., Öğretmenler Cad.,
Investigation of the de Broglie-instein veloity equation s universality in the ontext of the Davisson-Germer experiment Yusuf Z. UMUL Canaya University, letroni and Communiation Dept., Öğretmenler Cad.,
22.54 Neutron Interactions and Applications (Spring 2004) Chapter 6 (2/24/04) Energy Transfer Kernel F(E E')
 22.54 Neutron Interations and Appliations (Spring 2004) Chapter 6 (2/24/04) Energy Transfer Kernel F(E E') Referenes -- J. R. Lamarsh, Introdution to Nulear Reator Theory (Addison-Wesley, Reading, 1966),
22.54 Neutron Interations and Appliations (Spring 2004) Chapter 6 (2/24/04) Energy Transfer Kernel F(E E') Referenes -- J. R. Lamarsh, Introdution to Nulear Reator Theory (Addison-Wesley, Reading, 1966),
Physical Laws, Absolutes, Relative Absolutes and Relativistic Time Phenomena
 Page 1 of 10 Physial Laws, Absolutes, Relative Absolutes and Relativisti Time Phenomena Antonio Ruggeri modexp@iafria.om Sine in the field of knowledge we deal with absolutes, there are absolute laws that
Page 1 of 10 Physial Laws, Absolutes, Relative Absolutes and Relativisti Time Phenomena Antonio Ruggeri modexp@iafria.om Sine in the field of knowledge we deal with absolutes, there are absolute laws that
Charmed Hadron Production in Polarized pp Collisions
 Charmed Hadron Prodution in Polarized pp Collisions oshiyuki Morii Λ and Kazumasa Ohkuma Λ Division of Sienes for Natural Environment, Faulty of Human Development, Kobe University, Nada, Kobe 657-851,
Charmed Hadron Prodution in Polarized pp Collisions oshiyuki Morii Λ and Kazumasa Ohkuma Λ Division of Sienes for Natural Environment, Faulty of Human Development, Kobe University, Nada, Kobe 657-851,
