McKetta Department of Chemical Engineering, University of Texas at Austin, 1 University Station, C0400 Austin, USA b
|
|
|
- Sheryl Thompson
- 5 years ago
- Views:
Transcription
1 Electronic Supplementary Material (ESI) for Reaction Chemistry & Engineering. This journal is The Royal Society of Chemistry 2017 Supplemental Information for Apparatus for Efficient Utilization of Isotopically- Labeled Gases in Pulse Transient Studies of Heterogeneously Catalyzed Gas Phase Reactions Adrianna Brush, a Shallaco McDonald, a Robin Dupré, Shruti Kota, Gregory M. Mullen, a C. Buddie Mullins* abcd *Corresponding author; mullins@che.utexas.edu a McKetta Department of Chemical Engineering, University of Texas at Austin, 1 University Station, C0400 Austin, USA b Department of Chemistry, University of Texas at Austin, 1 University Station, C0400 Austin, USA c Texas Materials Institute and Center for Nano- and Molecular Science, University of Texas at Austin, 1 University Station, C0400 Austin, USA d Center for Electrochemistry, University of Texas at Austin, 1 University Station, C0400 Austin, USA
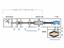
2 Cost Breakdown of Pulse Injection Apparatus versus Typical SSITKA System Figure S1: Block diagram of gas delivery systems for a) Typical SSITKA system, and b) Pulse Injection Apparatus described in this manuscript. The components considered in cost comparisons are circled with a purple dotted line. Item Size Connections Supplier Price # Cost VCR Gland 1/4" Socket Weld Swagelok $ $49.80 VCR Female Nut 1/4" Nut Swagelok $ $21.60 VCR Male Nut 1/4" Nut Swagelok $ $9.20 VCR Gaskets 1/4" VCR Swagelok $ $8.40 NTP-Socket Weld 1/4" MNTP-Weld Swagelok $ $25.60 SS Tubing 5ft. 1/4" N/A Swagelok $ $ Port Valve 1/4" 4x FNPT Sizto $ $37.74 Mass Flow Controller 1/4" 2x MVCR MKS $1, $1, Table S1: Cost estimate of typical SSITKA system. Total: $1,706.04
3 Item Size Connections Supplier Price # Cost Fittings, tubing, and valves Ball Valve 1/4" 2x MVCR Swagelok $ $ Tee 1/4" 3x MVCR Swagelok $ $ way Cross 1/4" 4x MVCR Swagelok $ $48.90 Female Union 1/4" 2x FVCR Swagelok $ $ Pressure Gauge Adapter 1/4" MVCR - FNPT Swagelok $ $16.30 NTP-Socket Weld 1/4" MNTP-Weld Swagelok $ $32.00 VCR Gland 1/4" Socket Weld Swagelok $ $91.30 VCR Female Nut 1/4" Nut Swagelok $ $48.60 VCR Male Nut 1/4" Nut Swagelok $ $9.20 SS Tubing - 10ft. 1/4" N/A Swagelok $ $33.20 VCR Gaskets 1/4" VCR Swagelok $ $30.80 Pressure Gauge 1/4" MNPT McMaster-Carr $ $ Port Valve 1/4" 5x FNPT Sizto $ $49.32 Subtotal: $ Bellows Pump Component Ball Screw + Handle N/A N/A McMaster-Carr $ $58.55 Ball Screw modification and fitting (labor) N/A N/A N/A $ $ Metal Supports (materials + labor) N/A N/A McMaster-Carr $ $ Linear guide bars and bushings N/A N/A McMaster-Carr $ $ Misc. Hardware N/A N/A McMaster-Carr $ $15.00 Volume Reducing insert N/A N/A McMaster-Carr $ $28.00 Bellows 2.75"x2.04" CF-CF Lesker $ $ Movable End Cap 2.75" CF Lesker $ $15.50 Fixed End Cap /4" CF-FVCR Lesker $ $86.00 Subtotal: $1, Table S2: Cost breakdown of Pulse Injection Apparatus system. Total: $2, Based on these comparisons, the Pulse Injection Apparatus costs ~$457 more than a typical SSITKA system. These calculations do not include: labor for assembling system, isotopically labeled gas cylinder, isotopically labeled gas regulator, or components common to both systems. If a higher quality 4- or 6- port valve is desired, a Valco 4-port valve (for the typical SSITKA system) would cost $1,220 (new total cost: $2,888.30), and a Valco 6-port valve (for Pulse Injection Apparatus system) would cost $1,275 (new total cost: $ ), bringing the difference between the Pulse Injection Apparatus and a typical SSITKA system to $500. Using a pre-fabricated linear shift mechanism from Lesker ($1770) instead of fabricating the Bellows Pump Component in-house ($1,217), adds an additional $553 to the cost of the Pulse Injection
4 Apparatus). Finally, we did not include the price of the mechanical pump, since many facilities have existing vacuum systems that can be utilized. Buying a new mechanical pump dedicated to the Pulse Injection Apparatus would cost an additional $700-$3,000, depending on the pump selected. Estimates on Gas Use Efficiency Using STP of isotopically labeled gas in a 440mL sized lecture bottle, pressurized to psig (according to Cambridge Isotope Laboratories, a common supplier of isotopically labeled gases). Assuming reaction is running at atmospheric pressure, using ideal gas law: n total = PV RT = (1bar)(1L) ( bar L mol 1 K 1 = mols )(273.15K) n recoverable = PV RT = (1.433bar)(0.440L) ( bar L mol 1 K 1 = mols )(298.15K) % recoverable = n recoverable = (100%) = 57.78% n total This is close to the rough approximation of 56% recoverable gas by assuming volume of lecture bottle is unrecoverable (440mL), leaving ~560mL out of 1L (i.e. 56%) recoverable. If we assume ~10% gas loss due to venting and bottle change-out, the gas use efficiency becomes (0.9)(57.78%) = 52.00% If we assume ~10% gas loss using the Pulse Injection Apparatus from vacuuming and bottle change-out, the gas use efficiency of the Pulse Injection Apparatus becomes 90% This represents an improvement of ~1.7x in gas use efficiency. This increases the amount of gas available to the researcher by 73%. Not including shipping costs for refilling the lecture bottle, 99% 13 CH4 is $ for a 1L bottle. Cost of unused gas in typical SSITKA system: (0.48)($264.00) = $ Cost of unused gas in Pulse Injection Apparatus system: (0.1)($264.00) = $26.40 Difference per bottle = $ Assuming the Pulse Injection Apparatus costs ~$500 more than a typical SSITKA system, the return on investment for building the Pulse Injection Apparatus is ~5 Bottles of 13 CH4 (or another equally priced isotopically labeled gas)
5 Theoretical Operation for VOC reactants Figure S2: Theoretical operation for using VOC isotopically labeled reactants; A) Sample loop evacuation; B) Carrier gas filling; C) VOC reactant filling; Labeled components: 1) 5-port valve, 2) Sample loop, 3) Bellows pump; 4) Vacuum pump, 5) Carrier gas, 6) Isotopically labeled VOC, 7) Valve manifold Theoretically, this apparatus could be used to inject a pulse of isotopically labeled VOC + carrier gas, that could be placed in line after a VOC saturator. Similar to a saturator, the partial pressure of the VOC is controlled by heating the VOC to a temperature such that: X VOC = VP VOC P reactor, where XVOC is the mole fraction of the VOC in the saturator line, VPVOC is the vapor pressure of the VOC at the temperature the vial of VOC is heated to, and Preactor is the pressure of the reaction (or pressure of the saturator line). In our design, Figure S1 depicts the way to obtain a pulse of isotopically labeled VOC: a) Evacuate the sample loop with the vacuum pump b) With the bellows pump partially compressed, fill the bellows pump and sample loop with a low pressure of the carrier gas used in the saturator line c) Open the vial of VOC to the bellows pump and sample loop. Expand and adjust the bellows pump until the total pressure equals Preactor. This requires waiting for the VOC to reach vapor-liquid equilibrium, and iterating the bellows pump compression to reach Preactor. We stress that this is merely a theoretical design, and have not explored the practical feasibility of this design. The biggest impediment will likely be the time required to let the VOC reach vapor-liquid equilibrium at Preactor as well as time required to let the VOC and carrier gases mix thoroughly. Since the main benefit of the Pulse Injection Apparatus is the ability to recover the sub-atmosphere gas in the isotopically labeled gas cylinder, the same benefit may not extend to VOC samples. However, this design does not have a practical minimum of isotopically labeled VOC requirement, the way a saturator may (for example, a minimum liquid level required to let the carrier gas sparge effectively). Finally, this design would require the bellows pump, valve manifold, sample loop, and 5-port valve to be heated to a temperature above the VOC saturator temperature in order to avoid VOC condensation in other parts of the system.
6 Pulse Functions at Different Flowrates Figure S3: Shape of the pulse functions at various flowrates of the isotopically-labeled/nonisotopically-labeled reactant gas ( R ) and the total flowrate of the remaining gases ( C for generic carrier gas ). a) 5.5 Standard Cubic Centimeters (SCCM) reactant gas, 0 SCCM Carrier Gases; b) 2 SCCM reactant gas, 18 SCCM carrier gases; c) 3 SCCM reactant gas, 30 SCCM carrier gases; d) 9 SCCM reactant gas, 91 SCCM carrier gases. Figure S3 demonstrates the effect on flowrate (and therefore time for diffusion and other mass transfer effects to occur) on the pulse function. The ¼ tubing we used has a volume of ~16.4mL per meter. Relevant reactor system tubing lengths: Pulse Injection Apparatus to connection joining with other gases (i.e. connection where R and C join): 99cm Connection joining R and C to reactor: 286cm Reactor to Mass Spectrometer: 51cm
Muliple Gas#3 plus Sulfur GC Configuration
 The SRI Model 8610C Gas Chromatograph ( GC ) configured as a MultipleGas#3 plus Sulfur is designed to measure H2, O2, N2, CO, CO2, H2O, C1 through C5 hydrocarbons and also H2S, COS/SO2, and other sulfur
The SRI Model 8610C Gas Chromatograph ( GC ) configured as a MultipleGas#3 plus Sulfur is designed to measure H2, O2, N2, CO, CO2, H2O, C1 through C5 hydrocarbons and also H2S, COS/SO2, and other sulfur
Chapter 10 Practice. Name: Multiple Choice Identify the choice that best completes the statement or answers the question.
 Name: Score: 0 / 18 points (0%) [3 open ended questions not graded] Chapter 10 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A sample of gas
Name: Score: 0 / 18 points (0%) [3 open ended questions not graded] Chapter 10 Practice Multiple Choice Identify the choice that best completes the statement or answers the question. 1. A sample of gas
ProPress (304 FKM) 1.3
 ProPress (304 FKM) 41 ProPress (304 FKM) Viega ProPress for stainless is engineered for harsh industrial environments where the piping systems may come into contact with corrosive cleaning liquids. A complete
ProPress (304 FKM) 41 ProPress (304 FKM) Viega ProPress for stainless is engineered for harsh industrial environments where the piping systems may come into contact with corrosive cleaning liquids. A complete
Thermodynamics. Atoms are in constant motion, which increases with temperature.
 Thermodynamics SOME DEFINITIONS: THERMO related to heat DYNAMICS the study of motion SYSTEM an object or set of objects ENVIRONMENT the rest of the universe MICROSCOPIC at an atomic or molecular level
Thermodynamics SOME DEFINITIONS: THERMO related to heat DYNAMICS the study of motion SYSTEM an object or set of objects ENVIRONMENT the rest of the universe MICROSCOPIC at an atomic or molecular level
A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings.
 Chapter 5 Gases Chapter 5 A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Copyright Cengage Learning. All rights reserved
Chapter 5 Gases Chapter 5 A Gas Uniformly fills any container. Easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Copyright Cengage Learning. All rights reserved
High-Pressure Volumetric Analyzer
 High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
High-Pressure Volumetric Analyzer High-Pressure Volumetric Analysis HPVA II Benefits Dual free-space measurement for accurate isotherm data Free space can be measured or entered Correction for non-ideality
INDEX OF ILLUSTRATIONS
 INDEX OF ILLUSTRATIONS FIG 3.1.1 FIG 3.1.2 FIG 3.1.3 FIG 3.2.1 FIG 3.2.2 FIG 3.2.3 FIG 3.2.4 FIG 3.3.1 FIG 3.4.1 FIG 3.4.1.1 ENVELOPE TYPES ENVELOPE NOMENCLATURE OVERVIEW VELCRO RIP & Q-VENT BASKET NOMENCLATURE
INDEX OF ILLUSTRATIONS FIG 3.1.1 FIG 3.1.2 FIG 3.1.3 FIG 3.2.1 FIG 3.2.2 FIG 3.2.3 FIG 3.2.4 FIG 3.3.1 FIG 3.4.1 FIG 3.4.1.1 ENVELOPE TYPES ENVELOPE NOMENCLATURE OVERVIEW VELCRO RIP & Q-VENT BASKET NOMENCLATURE
Some Important Industrial Gases
 Gaseous state Table 5.1 Some Important Industrial Gases Name (Formula) Methane (CH 4 ) Ammonia (NH 3 ) Chlorine (Cl 2 ) Oxygen (O 2 ) Ethylene (C 2 H 4 ) Origin and Use natural deposits; domestic fuel
Gaseous state Table 5.1 Some Important Industrial Gases Name (Formula) Methane (CH 4 ) Ammonia (NH 3 ) Chlorine (Cl 2 ) Oxygen (O 2 ) Ethylene (C 2 H 4 ) Origin and Use natural deposits; domestic fuel
1.0 Reactive Gas Chromatography Design and Setup Procedure for Catalyst Pelletization & Inlet Packing Software and Method.
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 207 Supplementary Information Simple Quantification of Zeolite Acid Site Density
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 207 Supplementary Information Simple Quantification of Zeolite Acid Site Density
Chapter 11 Gases 1 Copyright McGraw-Hill 2009
 Chapter 11 Gases Copyright McGraw-Hill 2009 1 11.1 Properties of Gases The properties of a gas are almost independent of its identity. (Gas molecules behave as if no other molecules are present.) Compressible
Chapter 11 Gases Copyright McGraw-Hill 2009 1 11.1 Properties of Gases The properties of a gas are almost independent of its identity. (Gas molecules behave as if no other molecules are present.) Compressible
Chemistry Instrumental Analysis Lecture 31. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 31 High Performance Liquid Chromatography (HPLC) High Performance Liquid Chromatography (HPLC) High Performance Liquid Chromatography (HPLC) Solvent Delivery
Chemistry 4631 Instrumental Analysis Lecture 31 High Performance Liquid Chromatography (HPLC) High Performance Liquid Chromatography (HPLC) High Performance Liquid Chromatography (HPLC) Solvent Delivery
THE VERSATILE VSP2: A TOOL FOR ADIABATIC THERMAL ANALYSIS AND VENT SIZING APPLICATIONS
 THE VERSATILE VSP2: A TOOL FOR ADIABATIC THERMAL ANALYSIS AND VENT SIZING APPLICATIONS Charles F. Askonas, Dr. James P. Burelbach, and Dr. Joseph C. Leung Fauske and Associates, Inc. 16W070 W. 83 rd Street
THE VERSATILE VSP2: A TOOL FOR ADIABATIC THERMAL ANALYSIS AND VENT SIZING APPLICATIONS Charles F. Askonas, Dr. James P. Burelbach, and Dr. Joseph C. Leung Fauske and Associates, Inc. 16W070 W. 83 rd Street
CHEM 254 EXPERIMENT 7. Phase Diagrams - Liquid Vapour Equilibrium for two component solutions
 pressure CHEM 254 EXPERIMENT 7 Phase Diagrams - Liquid Vapour Equilibrium for two component solutions The partial pressures of the components of an ideal solution of two volatile liquids are related to
pressure CHEM 254 EXPERIMENT 7 Phase Diagrams - Liquid Vapour Equilibrium for two component solutions The partial pressures of the components of an ideal solution of two volatile liquids are related to
Physics 5D PRACTICE FINAL EXAM Fall 2013
 Print your name: Physics 5D PRACTICE FINAL EXAM Fall 2013 Real Exam is Wednesday December 11 Thimann Lecture 3 4:00-7:00 pm Closed book exam two 8.5x11 sheets of notes ok Note: Avogadro s number N A =
Print your name: Physics 5D PRACTICE FINAL EXAM Fall 2013 Real Exam is Wednesday December 11 Thimann Lecture 3 4:00-7:00 pm Closed book exam two 8.5x11 sheets of notes ok Note: Avogadro s number N A =
Summary of Gas Laws V T. Boyle s Law (T and n constant) Charles Law (p and n constant) Combined Gas Law (n constant) 1 =
 Summary of Gas Laws Boyle s Law (T and n constant) p 1 V 1 = p 2 V 2 Charles Law (p and n constant) V 1 = T 1 V T 2 2 Combined Gas Law (n constant) pv 1 T 1 1 = pv 2 T 2 2 1 Ideal Gas Equation pv = nrt
Summary of Gas Laws Boyle s Law (T and n constant) p 1 V 1 = p 2 V 2 Charles Law (p and n constant) V 1 = T 1 V T 2 2 Combined Gas Law (n constant) pv 1 T 1 1 = pv 2 T 2 2 1 Ideal Gas Equation pv = nrt
Ideal Gas & Gas Stoichiometry
 Ideal Gas & Gas Stoichiometry Avogadro s Law V a number of moles (n) V = constant x n Constant temperature Constant pressure V 1 /n 1 = V 2 /n 2 Ammonia burns in oxygen to form nitric oxide (NO) and water
Ideal Gas & Gas Stoichiometry Avogadro s Law V a number of moles (n) V = constant x n Constant temperature Constant pressure V 1 /n 1 = V 2 /n 2 Ammonia burns in oxygen to form nitric oxide (NO) and water
Entrance Exam Chemistry Time: 1 hour July 12, 2009
 Entrance Exam 009-010 Chemistry Time: 1 hour July 1, 009 First exercise (10 points) Kinetic Study by Following the Pressure The aim of this exercise is to determine the half-life, t½, of the reaction between
Entrance Exam 009-010 Chemistry Time: 1 hour July 1, 009 First exercise (10 points) Kinetic Study by Following the Pressure The aim of this exercise is to determine the half-life, t½, of the reaction between
AE 3051, Lab #16. Investigation of the Ideal Gas State Equation. By: George P. Burdell. Group E3
 AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
AE 3051, Lab #16 Investigation of the Ideal Gas State Equation By: George P. Burdell Group E3 Summer Semester 000 Abstract The validity of the ideal gas equation of state was experimentally tested for
PVC - WHITE PRESSURE FITTINGS PRODUCTS PRICE LIST INCLUDES: PVC SCHEDULE 40, CLASS 125, POOL/SPA, LASCOTITE, ULTRAFIX REPAIR COUPLINGS PRICE GROUP
 PRICE GROUP DESCRIPTION 01 SCH 40 PVC through 8 91 SCH 40 PVC 10" AND LARGER 96 CLASS 125 10 & 12 09 POOL AND SPA 14 LASCOTITE 15 ULTRAFIX REPAIR COUPLING DISCOUNT MULTIPLIER PRODUCTS PVC - WHITE PRESSURE
PRICE GROUP DESCRIPTION 01 SCH 40 PVC through 8 91 SCH 40 PVC 10" AND LARGER 96 CLASS 125 10 & 12 09 POOL AND SPA 14 LASCOTITE 15 ULTRAFIX REPAIR COUPLING DISCOUNT MULTIPLIER PRODUCTS PVC - WHITE PRESSURE
Exercise 4-4. Titration of a Buffer Solution EXERCISE OBJECTIVE DISCUSSION OUTLINE DISCUSSION. Buffer solutions
 Exercise 4-4 Titration of a Buffer Solution EXERCISE OBJECTIVE Titrate a buffer solution, plot a graph using the titration data, and analyze the titration curve. DISCUSSION OUTLINE The Discussion of this
Exercise 4-4 Titration of a Buffer Solution EXERCISE OBJECTIVE Titrate a buffer solution, plot a graph using the titration data, and analyze the titration curve. DISCUSSION OUTLINE The Discussion of this
Apply the ideal gas law (PV = nrt) to experimentally determine the number of moles of carbon dioxide gas generated
 Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
Teacher Information Ideal Gas Law Objectives Determine the number of moles of carbon dioxide gas generated during a reaction between hydrochloric acid and sodium bicarbonate. Through this investigation,
AC : A DIMENSIONAL ANALYSIS EXPERIMENT FOR THE FLUID MECHANICS CLASSROOM
 AC 2011-1108: A DIMENSIONAL ANALYSIS EXPERIMENT FOR THE FLUID MECHANICS CLASSROOM Charles Forsberg, Hofstra University Charles H. Forsberg is an Associate Professor of Engineering at Hofstra University,
AC 2011-1108: A DIMENSIONAL ANALYSIS EXPERIMENT FOR THE FLUID MECHANICS CLASSROOM Charles Forsberg, Hofstra University Charles H. Forsberg is an Associate Professor of Engineering at Hofstra University,
hydrideicp Hydride Generation System
 HG-MP2-4-A hydrideicp Generation System hydrideicp Hydride Generation System User Manual MP 2 hydrideicp Generation System Elemental Scientific Phone: 1.402.991.7800 Email: esi@icpms.com 1500 North 24th
HG-MP2-4-A hydrideicp Generation System hydrideicp Hydride Generation System User Manual MP 2 hydrideicp Generation System Elemental Scientific Phone: 1.402.991.7800 Email: esi@icpms.com 1500 North 24th
1,2,8,9,11,13,14,17,19,20,22,24,26,28,30,33,38,40,43,45,46,51,53,55,57,62,63,80,82,88,94
 CHAPTER 5GASES 1,,8,9,11,1,14,17,19,0,,4,6,8,0,,8,40,4,45,46,51,5,55,57,6,6,80,8,88,94 5.1 a) The volume of the liquid remains constant, but the volume of the gas increases to the volume of the larger
CHAPTER 5GASES 1,,8,9,11,1,14,17,19,0,,4,6,8,0,,8,40,4,45,46,51,5,55,57,6,6,80,8,88,94 5.1 a) The volume of the liquid remains constant, but the volume of the gas increases to the volume of the larger
PVC - WHITE PRESSURE FITTINGS L40P EFFECTIVE OCTOBER 1, 2018 REVISED PRODUCTS PRICE LIST
 PRICE GROUP DESCRIPTION 01 SCH 40 PVC through 8 91 SCH 40 PVC 10" AND LARGER 96 CLASS 125 10 & 12 09 POOL AND SPA 14 LASCOTITE 15 ULTRAFIX REPAIR COUPLING DISCOUNT MULTIPLIER PRODUCTS PVC - WHITE PRESSURE
PRICE GROUP DESCRIPTION 01 SCH 40 PVC through 8 91 SCH 40 PVC 10" AND LARGER 96 CLASS 125 10 & 12 09 POOL AND SPA 14 LASCOTITE 15 ULTRAFIX REPAIR COUPLING DISCOUNT MULTIPLIER PRODUCTS PVC - WHITE PRESSURE
Spark combustion reactor for 13-carbon isotope enrichment analysis of gases
 REVIEW OF SCIENTIFIC INSTRUMENTS VOLUME 74, NUMBER 4 APRIL 2003 Spark combustion reactor for 13-carbon isotope enrichment analysis of gases Michael May, a) Michael Gray, John E. Kuo, and C. T. Tan Quality
REVIEW OF SCIENTIFIC INSTRUMENTS VOLUME 74, NUMBER 4 APRIL 2003 Spark combustion reactor for 13-carbon isotope enrichment analysis of gases Michael May, a) Michael Gray, John E. Kuo, and C. T. Tan Quality
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
Properties of Gases. Molecular interactions van der Waals equation Principle of corresponding states
 Properties of Gases Chapter 1 of Atkins and de Paula The Perfect Gas States of gases Gas laws Real Gases Molecular interactions van der Waals equation Principle of corresponding states Kinetic Model of
Properties of Gases Chapter 1 of Atkins and de Paula The Perfect Gas States of gases Gas laws Real Gases Molecular interactions van der Waals equation Principle of corresponding states Kinetic Model of
Exercise 4-3. Titration of Weak Acids EXERCISE OBJECTIVE DISCUSSION OUTLINE. The 5% rule DISCUSSION
 Exercise 4-3 Titration of Weak Acids EXERCISE OBJECTIVE Titrate both a weak acid solution and a weak polyprotic acid solution with a strong base solution. Plot a graph using the titration data, analyze
Exercise 4-3 Titration of Weak Acids EXERCISE OBJECTIVE Titrate both a weak acid solution and a weak polyprotic acid solution with a strong base solution. Plot a graph using the titration data, analyze
Chapter 5 The Gaseous State
 Chapter 5 The Gaseous State Contents and Concepts Gas Laws We will investigate the quantitative relationships that describe the behavior of gases. 1. Gas Pressure and Its Measurement 2. Empirical Gas Laws
Chapter 5 The Gaseous State Contents and Concepts Gas Laws We will investigate the quantitative relationships that describe the behavior of gases. 1. Gas Pressure and Its Measurement 2. Empirical Gas Laws
Atmospheric Analysis Gases. Sampling and analysis of gaseous compounds
 Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
Atmospheric Analysis Gases Sampling and analysis of gaseous compounds Introduction - External environment (ambient air) ; global warming, acid rain, introduction of pollutants, etc - Internal environment
REMOVE THIS PAGE PRIOR TO STARTING EXAM.
 REMOVE THIS PAGE PRIOR TO STARTING EXAM. 1 2 ANSWER KEY CHEMISTRY F14O3 SECOND EXAM 11/10/99 PROFESSOR J. MORROW PRINT NAME, LAST: FIRST: I.D.# : MAXIMUM POINT VALUE IS IN PARENTHESES 1. (15) 9. (15) 17.
REMOVE THIS PAGE PRIOR TO STARTING EXAM. 1 2 ANSWER KEY CHEMISTRY F14O3 SECOND EXAM 11/10/99 PROFESSOR J. MORROW PRINT NAME, LAST: FIRST: I.D.# : MAXIMUM POINT VALUE IS IN PARENTHESES 1. (15) 9. (15) 17.
Chemistry 11. Unit 11 Ideal Gas Law (Special Topic)
 Chemistry 11 Unit 11 Ideal Gas Law (Special Topic) 2 1. States of substances It has been studied in Unit 3 that there exist 3 states of matter in nature: gas, liquid and solid. (Technically there is the
Chemistry 11 Unit 11 Ideal Gas Law (Special Topic) 2 1. States of substances It has been studied in Unit 3 that there exist 3 states of matter in nature: gas, liquid and solid. (Technically there is the
Practice Problems. Unit 11 - Gas Laws. CRHS Academic Chemistry. Due Date Assignment On-Time (100) Late (70)
 Name Period CRHS Academic Chemistry Unit 11 - Gas Laws Practice Problems Due Date Assignment On-Time (100) Late (70) 11.1 11.2 11.3 11.4 Warm-Up EC Notes, Homework, Exam Reviews and Their KEYS located
Name Period CRHS Academic Chemistry Unit 11 - Gas Laws Practice Problems Due Date Assignment On-Time (100) Late (70) 11.1 11.2 11.3 11.4 Warm-Up EC Notes, Homework, Exam Reviews and Their KEYS located
Thermodynamics. Joule-Thomson effect Ideal and Real Gases. What you need: Complete Equipment Set, Manual on CD-ROM included
 Ideal Real Gases Thermodynamics 30600 What you can learn about Real gas Intrinsic energy Gay-Lussac theory Throttling Van der Waals equation Van der Waals force Inverse Inversion temperature Principle:
Ideal Real Gases Thermodynamics 30600 What you can learn about Real gas Intrinsic energy Gay-Lussac theory Throttling Van der Waals equation Van der Waals force Inverse Inversion temperature Principle:
DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications
 DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications The Vaisala DRYCAP Hand-Held Dewpoint Meter DM70 offers accurate and fast measurement for industrial dewpoint applications, such as compressed
DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications The Vaisala DRYCAP Hand-Held Dewpoint Meter DM70 offers accurate and fast measurement for industrial dewpoint applications, such as compressed
Freezing point depression (Item No.: P )
 Freezing point depression (Item No.: P3021101) Curricular Relevance Area of Expertise: Chemistry Education Level: University Topic: General Chemistry Subtopic: Solutions and Mixtures Experiment: Freezing
Freezing point depression (Item No.: P3021101) Curricular Relevance Area of Expertise: Chemistry Education Level: University Topic: General Chemistry Subtopic: Solutions and Mixtures Experiment: Freezing
10/16/2018. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 10/16/018 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 1 10/16/018 A Gas Uniformly fills any container.
10/16/018 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 1 10/16/018 A Gas Uniformly fills any container.
Vapor Intrusion Sampling Options: Performance Data for Canisters, Badges, and Sorbent Tubes for VOCs
 Vapor Intrusion Sampling Options: Performance Data for s, Badges, and Sorbent Tubes for VOCs Linda S. Coyne SKC Inc., 863 Valley View Road, Eighty Four, PA 1533 George Havalias, Maria C. Echarte American
Vapor Intrusion Sampling Options: Performance Data for s, Badges, and Sorbent Tubes for VOCs Linda S. Coyne SKC Inc., 863 Valley View Road, Eighty Four, PA 1533 George Havalias, Maria C. Echarte American
Lecture 3 Vacuum Science and Technology
 Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Lecture 3 Vacuum Science and Technology Chapter 3 - Wolf and Tauber 1/56 Announcements Homework will be online from noon today. This is homework 1 of 4. 25 available marks (distributed as shown). This
Supporting information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting information From alcohol to 1,2,3-triazole via multi-step continuous-flow synthesis
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Supporting information From alcohol to 1,2,3-triazole via multi-step continuous-flow synthesis
Electronic Supplementary Material Experimentally Validated Mathematical Model of Analyte Uptake by Permeation Passive Samplers
 Electronic Supplementary Material (ESI) for Environmental Science: Processes & Impacts. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Material Experimentally Validated Mathematical
Electronic Supplementary Material (ESI) for Environmental Science: Processes & Impacts. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Material Experimentally Validated Mathematical
Given Find Water Properties
 Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Why study gases? A Gas 10/17/2017. An understanding of real world phenomena. An understanding of how science works.
 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? n understanding of real world phenomena. n understanding of how science works. Gas Uniformly fills any container. Mixes completely
Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? n understanding of real world phenomena. n understanding of how science works. Gas Uniformly fills any container. Mixes completely
405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Data Book and Flow Handbook
 405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book and Flow Handbook www.rosemount.com 405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book NOTICE Read
405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book and Flow Handbook www.rosemount.com 405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book NOTICE Read
Review Unit #11. Review Unit # H 2 O (g) + CO (g) H 2(g) + CO 2(g) H>1
 Review Unit #11 1. H 2 O (g) + CO (g) H 2(g) + CO 2(g) H>1 K c = 1.6 What effect would these changes have on the equilibrium position? a. Cool the mixture b. Increase the volume of the flask c. Add H 2(g)
Review Unit #11 1. H 2 O (g) + CO (g) H 2(g) + CO 2(g) H>1 K c = 1.6 What effect would these changes have on the equilibrium position? a. Cool the mixture b. Increase the volume of the flask c. Add H 2(g)
Continuous Flow Reactions. From idea to production size scale up in 3 steps
 Continuous Flow Reactions From idea to production size scale up in 3 steps PIN NL Spring Session 13 04 2017 13 4 2017 PIN NL Spring Session 1 Topics Introduction Flowlink Continuous Flow Reactions What
Continuous Flow Reactions From idea to production size scale up in 3 steps PIN NL Spring Session 13 04 2017 13 4 2017 PIN NL Spring Session 1 Topics Introduction Flowlink Continuous Flow Reactions What
Chapter 5. Gases and the Kinetic-Molecular Theory
 Chapter 5 Gases and the Kinetic-Molecular Theory Macroscopic vs. Microscopic Representation Kinetic Molecular Theory of Gases 1. Gas molecules are in constant motion in random directions. Collisions among
Chapter 5 Gases and the Kinetic-Molecular Theory Macroscopic vs. Microscopic Representation Kinetic Molecular Theory of Gases 1. Gas molecules are in constant motion in random directions. Collisions among
A Laboratory System for Simulation of Extreme Atmospheric Conditions in the Deep Atmospheres of Venus, Jupiter, and Beyond
 A Laboratory System for Simulation of Extreme Atmospheric Conditions in the Deep Atmospheres of Venus, Jupiter, and Beyond Bryan M. Karpowicz *, Paul G. Steffes +, and Thomas R. Hanley + * School of Earth
A Laboratory System for Simulation of Extreme Atmospheric Conditions in the Deep Atmospheres of Venus, Jupiter, and Beyond Bryan M. Karpowicz *, Paul G. Steffes +, and Thomas R. Hanley + * School of Earth
Gases. Chapter 5. Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Gases Chapter 5 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Elements that exist as gases at 25 0 C and 1 atmosphere 2 3 Physical Characteristics of Gases
Gases Chapter 5 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Elements that exist as gases at 25 0 C and 1 atmosphere 2 3 Physical Characteristics of Gases
Section Using Gas Laws to Solve Problems
 Gases and Gas Laws Section 13.2 Using Gas Laws to Solve Problems Kinetic Molecular Theory Particles of matter are ALWAYS in motion Volume of individual particles is zero. Consists of large number of particles
Gases and Gas Laws Section 13.2 Using Gas Laws to Solve Problems Kinetic Molecular Theory Particles of matter are ALWAYS in motion Volume of individual particles is zero. Consists of large number of particles
Thermochemistry/Calorimetry. Determination of the enthalpy of combustion with a calorimetric bomb LEC 02. What you need:
 LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
LEC 02 Thermochemistry/Calorimetry with a calorimetric bomb What you can learn about 1st law of thermodynamics Hess law Enthalpy of combustion Enthalpy of formation Heat capacity Principle and tasks The
Ch 9 Practice Problems
 Ch 9 Practice Problems 1. One mole of an ideal gas is expanded from a volume of 1.50 L to a volume of 10.18 L against a constant external pressure of 1.03 atm. Calculate the work. (1 L atm = 101.3 J) A)
Ch 9 Practice Problems 1. One mole of an ideal gas is expanded from a volume of 1.50 L to a volume of 10.18 L against a constant external pressure of 1.03 atm. Calculate the work. (1 L atm = 101.3 J) A)
Chapter 11. = 14.1 psi. = 28.7 in. Hg. 1 bar 1 atm 101, 325 Pascal 1000 mbar bar 1 atm 1 bar 1 atm 760 torr 1000 mbar 1.
 ractice Exercises. psi = 70 mm Hg in. Hg = 70 mm Hg 4.7 psi 760 mm Hg 9.9 in. Hg 760 mm Hg = 4. psi = 8.7 in. Hg. ascals = 888 mbar torr = 888 mbar bar 0, 5 ascal 000 mbar.0 bar bar 000 mbar.0 bar = 666
ractice Exercises. psi = 70 mm Hg in. Hg = 70 mm Hg 4.7 psi 760 mm Hg 9.9 in. Hg 760 mm Hg = 4. psi = 8.7 in. Hg. ascals = 888 mbar torr = 888 mbar bar 0, 5 ascal 000 mbar.0 bar bar 000 mbar.0 bar = 666
DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications
 DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications The Vaisala DRYCAP Hand-Held Dewpoint Meter DM70 offers accurate and fast measurement for industrial dewpoint applications, such as compressed
DM70 Hand-Held Dewpoint Meter for Spot-Checking Applications The Vaisala DRYCAP Hand-Held Dewpoint Meter DM70 offers accurate and fast measurement for industrial dewpoint applications, such as compressed
Gases. A gas. Difference between gas and vapor: Why Study Gases?
 Gases Chapter 5 Gases A gas Uniformly fills any container. Is easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Difference between gas and vapor: A gas is a substance
Gases Chapter 5 Gases A gas Uniformly fills any container. Is easily compressed. Mixes completely with any other gas. Exerts pressure on its surroundings. Difference between gas and vapor: A gas is a substance
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten. Chapter 10. Gases.
 Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 10 Characteristics of Unlike liquids and solids, they Expand to fill their containers.
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 10 Characteristics of Unlike liquids and solids, they Expand to fill their containers.
Some Fundamental Definitions:
 Lecture 2. The GAS LAWS Some Fundamental Definitions: SYSTEM: the part of the universe being the subject of study 1 Some Fundamental Definitions: State of the System: condition of a system at any given
Lecture 2. The GAS LAWS Some Fundamental Definitions: SYSTEM: the part of the universe being the subject of study 1 Some Fundamental Definitions: State of the System: condition of a system at any given
*Correspondence to:
 Supporting Information for Carbonate-promoted hydrogenation of carbon dioxide to multi-carbon carboxylates Aanindeeta Banerjee 1 and Matthew W. Kanan 1 * 1 Department of Chemistry, Stanford University,
Supporting Information for Carbonate-promoted hydrogenation of carbon dioxide to multi-carbon carboxylates Aanindeeta Banerjee 1 and Matthew W. Kanan 1 * 1 Department of Chemistry, Stanford University,
CHEM 254 EXPERIMENT 2 Critical point determination for SF 6
 CHEM 254 EXPERIMENT 2 Critical point determination for SF 6 The equation of state of a gas defines the relationship between the pressure, temperature and volume of the gas. For ideal gases the equation
CHEM 254 EXPERIMENT 2 Critical point determination for SF 6 The equation of state of a gas defines the relationship between the pressure, temperature and volume of the gas. For ideal gases the equation
Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji
 University of Groningen Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
Unit Outline. I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI.
 Unit 10: Gases Unit Outline I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI. Real Gases I. Opening thoughts Have you ever: Seen a hot air balloon?
Unit 10: Gases Unit Outline I. Introduction II. Gas Pressure III. Gas Laws IV. Gas Law Problems V. Kinetic-Molecular Theory of Gases VI. Real Gases I. Opening thoughts Have you ever: Seen a hot air balloon?
Cryogenic Target Handling System Operations Manual Volume IV CTHS Description Chapter 5: Tritium Fill Station (TFS)
 CHATER 5: TRITIUM FILL STATION (TFS) REVISION B (ARIL 2004) S-AA-M-31 Cryogenic Target Handling System Operations Manual Volume IV CTHS Description Chapter 5: Tritium Fill Station (TFS) Table of Contents
CHATER 5: TRITIUM FILL STATION (TFS) REVISION B (ARIL 2004) S-AA-M-31 Cryogenic Target Handling System Operations Manual Volume IV CTHS Description Chapter 5: Tritium Fill Station (TFS) Table of Contents
Simpo PDF Merge and Split Unregistered Version -
 74. The rate of heat flow by conduction through a slab does NOT depend upon the: A. temperature difference between opposite faces of the slab B. thermal conductivity of the slab C. slab thickness D. cross-sectional
74. The rate of heat flow by conduction through a slab does NOT depend upon the: A. temperature difference between opposite faces of the slab B. thermal conductivity of the slab C. slab thickness D. cross-sectional
Development of Safe and Scalable Continuous-Flow Methods for. Palladium-Catalyzed Aerobic Oxidation Reactions
 Supplementary Data for Development of Safe and Scalable Continuous-Flow Methods for Palladium-Catalyzed Aerobic Oxidation Reactions Xuan Ye, a Martin D. Johnson, *b Tianning Diao, a Matthew H. Yates *b
Supplementary Data for Development of Safe and Scalable Continuous-Flow Methods for Palladium-Catalyzed Aerobic Oxidation Reactions Xuan Ye, a Martin D. Johnson, *b Tianning Diao, a Matthew H. Yates *b
Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium Storage Material
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Sub-Stoichiometric Germanium Sulfide Thin-Films as a High-Rate Lithium
Accurate Analysis of Fuel Ethers and Oxygenates in a Single Injection without Calibration Standards using GC- Polyarc/FID. Application Note.
 Accurate Analysis of Fuel Ethers and Oxygenates in a Single Injection without Calibration Standards using GC- Polyarc/FID Application Note Volatile Organic Compounds (VOCs) Author Andrew Jones Activated
Accurate Analysis of Fuel Ethers and Oxygenates in a Single Injection without Calibration Standards using GC- Polyarc/FID Application Note Volatile Organic Compounds (VOCs) Author Andrew Jones Activated
INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin
 Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Gases by Christopher G. Hamaker Illinois State University Properties of Gases There are five important
Lecture INTRODUCTORY CHEMISTRY Concepts and Critical Thinking Seventh Edition by Charles H. Corwin Gases by Christopher G. Hamaker Illinois State University Properties of Gases There are five important
Given Find Water Properties
 Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
Venturi Example Given: A venturi is to be used to measure a 50 gpm flow of 70 F water in a 4-in ID pipe. Find: Select a throat diameter that provides Re d > 00,000 in the throat, and determine what differential
FDB1050REB2 FDB1050REC2 FDB1050REM2 FDB1050REQ2 FDB1050RES2
 Product No. B2 C2 M2 Q2 S2 Series Built-in 24" Built-in 24" Built-in 24" Built-in 24" Built-in 24" Color black stainless silver mist bisque white Owner's Guide English 154591101 154591101 154591101 154591101
Product No. B2 C2 M2 Q2 S2 Series Built-in 24" Built-in 24" Built-in 24" Built-in 24" Built-in 24" Color black stainless silver mist bisque white Owner's Guide English 154591101 154591101 154591101 154591101
B 2, C 2, N 2. O 2, F 2, Ne 2. Energy order of the p 2p and s 2p orbitals changes across the period.
 Chapter 11 Gases Energy order of the p p and s p orbitals changes across the period. Due to lower nuclear charge of B, C & N there is no s-p orbitals interaction Due to high nuclear charge of O, F& Ne
Chapter 11 Gases Energy order of the p p and s p orbitals changes across the period. Due to lower nuclear charge of B, C & N there is no s-p orbitals interaction Due to high nuclear charge of O, F& Ne
Apparatus for Studying the Relationship Between Pressure and Volume of a Gas
 Apparatus for Studying the Relationship Between Pressure and Volume of a Gas As P (h) increases V decreases 1 Boyle s Law P α 1/V P x V = constant P 1 x V 1 = P 2 x V 2 Constant temperature Constant amount
Apparatus for Studying the Relationship Between Pressure and Volume of a Gas As P (h) increases V decreases 1 Boyle s Law P α 1/V P x V = constant P 1 x V 1 = P 2 x V 2 Constant temperature Constant amount
Name: Regents Review Quiz #1 2016
 Name: Regents Review Quiz #1 2016 1. Which two particle diagrams represent mixtures of diatomic elements? A) A and B B) A and C C) B and C D) B and D 2. At STP, which physical property of aluminum always
Name: Regents Review Quiz #1 2016 1. Which two particle diagrams represent mixtures of diatomic elements? A) A and B B) A and C C) B and C D) B and D 2. At STP, which physical property of aluminum always
VACUUM PUMPING METHODS
 VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Positive Displacement Vacuum Gas Transfer Vacuum Kinetic Vacuum Entrapment Vacuum Adsorption Reciprocating Displacement Rotary Drag Fluid Entrainment Ion Transfer
TO-15 Checklist Determination of VOCs in Air by GC-MS
 LAB ID: DATE: LAB NAME: ASSESSOR NAME: Method Number: TO-15 Checklist Determination of VOCs in Air by GC-MS SOP Number: Revision Number: SOP Date: Personnel records observed: Data records observed: Revision
LAB ID: DATE: LAB NAME: ASSESSOR NAME: Method Number: TO-15 Checklist Determination of VOCs in Air by GC-MS SOP Number: Revision Number: SOP Date: Personnel records observed: Data records observed: Revision
10/15/2015. Why study gases? An understanding of real world phenomena. An understanding of how science works.
 0/5/05 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 0/5/05 A Gas fills any container. completely
0/5/05 Kinetic Theory and the Behavior of Ideal & Real Gases Why study gases? An understanding of real world phenomena. An understanding of how science works. 0/5/05 A Gas fills any container. completely
Technique to measure CO 2 mixing ratio in small flasks with a bellows/irga system
 Agricultural and Forest Meteorology 109 (2001) 61 65 Short communication Technique to measure CO 2 mixing ratio in small flasks with a bellows/irga system D.R. Bowling, C.S. Cook, J.R. Ehleringer Stable
Agricultural and Forest Meteorology 109 (2001) 61 65 Short communication Technique to measure CO 2 mixing ratio in small flasks with a bellows/irga system D.R. Bowling, C.S. Cook, J.R. Ehleringer Stable
The FLUX F 430 with mechanical seal
 The FLUX F 0 with mechanical seal Flexible coupling between motor and pump The pump must be used with many varying liquids, consequently the smallest residual left within the pump is unacceptable. You
The FLUX F 0 with mechanical seal Flexible coupling between motor and pump The pump must be used with many varying liquids, consequently the smallest residual left within the pump is unacceptable. You
Chapter 3 PROPERTIES OF PURE SUBSTANCES
 Thermodynamics: An Engineering Approach Seventh Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 3 PROPERTIES OF PURE SUBSTANCES Copyright The McGraw-Hill Companies, Inc. Permission
Thermodynamics: An Engineering Approach Seventh Edition Yunus A. Cengel, Michael A. Boles McGraw-Hill, 2011 Chapter 3 PROPERTIES OF PURE SUBSTANCES Copyright The McGraw-Hill Companies, Inc. Permission
EXPT 6. Boyle's Law and Graham's Law
 EXPT 6. Boyle's Law and Graham's Law [Key Contents] pressure-volume relation in gases, atomic model, ideal gas law kinetic theory of gases, effusion rate, diffusion rate, molecular weight [References]
EXPT 6. Boyle's Law and Graham's Law [Key Contents] pressure-volume relation in gases, atomic model, ideal gas law kinetic theory of gases, effusion rate, diffusion rate, molecular weight [References]
Thermal Soil Desorber Instructions September 2006
 The SRI Thermal Soil Desorber is mounted in the valve oven section of the 8610C GC chassis. The 8610C GC shown in the photo is sitting next to the H2-40XR Hydrogen generator on the benchtop. With both
The SRI Thermal Soil Desorber is mounted in the valve oven section of the 8610C GC chassis. The 8610C GC shown in the photo is sitting next to the H2-40XR Hydrogen generator on the benchtop. With both
Chapter 5 Gases and the Kinetic-Molecular Theory
 Chapter 5 Gases and the Kinetic-Molecular Theory Name (Formula) Methane (CH 4 ) Ammonia (NH 3 ) Chlorine (Cl 2 ) Oxygen (O 2 ) Ethylene (C 2 H 4 ) Origin and Use natural deposits; domestic fuel from N
Chapter 5 Gases and the Kinetic-Molecular Theory Name (Formula) Methane (CH 4 ) Ammonia (NH 3 ) Chlorine (Cl 2 ) Oxygen (O 2 ) Ethylene (C 2 H 4 ) Origin and Use natural deposits; domestic fuel from N
The 505F Series 3/4/5-Way Ball Valve. Standard Features and Benefits. Operating Conditions. Specifications. Options
 The 505F Series //5-Way Ball Valve The 505F is a true multi-way ball valve with balanced fourseat construction. This full port valve is available in a variety of flow plans which can replace multiple two-way
The 505F Series //5-Way Ball Valve The 505F is a true multi-way ball valve with balanced fourseat construction. This full port valve is available in a variety of flow plans which can replace multiple two-way
CF (Conflat Flange) Fittings - Adapters. (505)
 F (onflat ) 1 Fittings - dapters www.idealvac.com (505)72-0037 1 Vacuum Fittings - dapters - olt Kits F - onflat F onflat flanges (F) are used for ultra- Vin elasmer gaskets can be used over and over but
F (onflat ) 1 Fittings - dapters www.idealvac.com (505)72-0037 1 Vacuum Fittings - dapters - olt Kits F - onflat F onflat flanges (F) are used for ultra- Vin elasmer gaskets can be used over and over but
CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES
 Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Thermodynamics: An Engineering Approach 8th Edition in SI Units Yunus A. Çengel, Michael A. Boles McGraw-Hill, 2015 CHAPTER 5 MASS AND ENERGY ANALYSIS OF CONTROL VOLUMES Lecture slides by Dr. Fawzi Elfghi
Lecture 1: Vapour Growth Techniques
 PH3EC2 Vapour Growth and Epitaxial Growth Lecturer: Dr. Shinoj V K Lecture 1: Vapour Growth Techniques 1.1 Vapour growth The growth of single crystal materials from the vapour phase. Deposition from the
PH3EC2 Vapour Growth and Epitaxial Growth Lecturer: Dr. Shinoj V K Lecture 1: Vapour Growth Techniques 1.1 Vapour growth The growth of single crystal materials from the vapour phase. Deposition from the
Chapter 10 Gases Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.
 Chapter 10 Gases 10.1 Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.1) Unlike liquids and solids, gases expand to fill their
Chapter 10 Gases 10.1 Characteristics of Gases Elements that exist as gases: Noble gases, O 2, N 2,H 2, F 2 and Cl 2. (For compounds see table 10.1) Unlike liquids and solids, gases expand to fill their
405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Data Book and Flow Handbook
 Reference Manual 405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book and Flow Handbook www.rosemount.com Reference Manual 405 and 1595 405 Compact Orifice Series and 1595 Conditioning
Reference Manual 405 Compact Orifice Series and 1595 Conditioning Orifice Plate Flow Test Book and Flow Handbook www.rosemount.com Reference Manual 405 and 1595 405 Compact Orifice Series and 1595 Conditioning
COURSE CODE : 3072 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE
 COURSE TITLE : FLUID MECHANICS COURSE CODE : 307 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE MODULE TOPIC PERIOD 1 Properties of Fluids 0 Fluid Friction and Flow
COURSE TITLE : FLUID MECHANICS COURSE CODE : 307 COURSE CATEGORY : B PERIODS/ WEEK : 5 PERIODS/ SEMESTER : 75 CREDIT : 5 TIME SCHEDULE MODULE TOPIC PERIOD 1 Properties of Fluids 0 Fluid Friction and Flow
Chapter 10 Notes: Gases
 Chapter 10 Notes: Gases Watch Bozeman Videos & other videos on my website for additional help: Big Idea 2: Gases 10.1 Characteristics of Gases Read p. 398-401. Answer the Study Guide questions 1. Earth
Chapter 10 Notes: Gases Watch Bozeman Videos & other videos on my website for additional help: Big Idea 2: Gases 10.1 Characteristics of Gases Read p. 398-401. Answer the Study Guide questions 1. Earth
Lecture Presentation. Chapter 10. Gases. John D. Bookstaver St. Charles Community College Cottleville, MO Pearson Education, Inc.
 Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
Lecture Presentation Chapter 10 John D. Bookstaver St. Charles Community College Cottleville, MO Characteristics of Unlike liquids and solids, gases Expand to fill their containers. Are highly compressible.
Anomalous production of gaseous 4 He at the inside of DScathode during D 2 O-electrolysis
 Arata, Y. and Y.C. Zhang, Anomalous production of gaseous 4 He at the inside of 'DS cathode' during D 2 O- electrolysis. Proc. Jpn. Acad., Ser. B, 1999. 75: p. 281. Anomalous production of gaseous 4 He
Arata, Y. and Y.C. Zhang, Anomalous production of gaseous 4 He at the inside of 'DS cathode' during D 2 O- electrolysis. Proc. Jpn. Acad., Ser. B, 1999. 75: p. 281. Anomalous production of gaseous 4 He
Chemistry 102 Spring 2019 Discussion #4 Chapters 11 and 12 Student name TA name Section
 Chemistry 102 Spring 2019 Discussion #4 Chapters 11 and 12 Student name TA name Section Things you should know when you finish the Discussion hand out: Average molar kinetic energy = E = M u 2 rms 2 =
Chemistry 102 Spring 2019 Discussion #4 Chapters 11 and 12 Student name TA name Section Things you should know when you finish the Discussion hand out: Average molar kinetic energy = E = M u 2 rms 2 =
Fluid Flow Analysis Penn State Chemical Engineering
 Fluid Flow Analysis Penn State Chemical Engineering Revised Spring 2015 Table of Contents LEARNING OBJECTIVES... 1 EXPERIMENTAL OBJECTIVES AND OVERVIEW... 1 PRE-LAB STUDY... 2 EXPERIMENTS IN THE LAB...
Fluid Flow Analysis Penn State Chemical Engineering Revised Spring 2015 Table of Contents LEARNING OBJECTIVES... 1 EXPERIMENTAL OBJECTIVES AND OVERVIEW... 1 PRE-LAB STUDY... 2 EXPERIMENTS IN THE LAB...
PhysicsAndMathsTutor.com 1
 PhysicsAndMathsTutor.com 1 Q1. In an experiment to measure the temperature of the flame of a Bunsen burner, a lump of copper of mass 0.12 kg is heated in the flame for several minutes. The copper is then
PhysicsAndMathsTutor.com 1 Q1. In an experiment to measure the temperature of the flame of a Bunsen burner, a lump of copper of mass 0.12 kg is heated in the flame for several minutes. The copper is then
The first law of thermodynamics. U = internal energy. Q = amount of heat energy transfer
 Thermodynamics Investigation of the energy transfer by heat and work and how natural systems behave (Q) Heat transfer of energy due to temp differences. (W) Work transfer of energy through mechanical means.
Thermodynamics Investigation of the energy transfer by heat and work and how natural systems behave (Q) Heat transfer of energy due to temp differences. (W) Work transfer of energy through mechanical means.
PARTS LIST GAS BOILERS MODELS: CG320 (E) CG325 (E) CG330 (E)
 PARTS LIST GAS BOILERS MODELS: CG320 (E) CG325 (E) CG330 (E) CROWN FOOD SERVICE EQUIPMENT LTD. 70 OAKDALE ROAD, DOWNSVIEW, (TORONTO), ONTARIO, CANADA, M3N 1V9 TELEPHONE: (416) 746-2358, FAX: (416) 746-8324
PARTS LIST GAS BOILERS MODELS: CG320 (E) CG325 (E) CG330 (E) CROWN FOOD SERVICE EQUIPMENT LTD. 70 OAKDALE ROAD, DOWNSVIEW, (TORONTO), ONTARIO, CANADA, M3N 1V9 TELEPHONE: (416) 746-2358, FAX: (416) 746-8324
Russell B. Schweickart and Gary Mills
 ANALYSIS AND TEST VERIFICATION OF TRANSITIONAL FLOW IN A DEWAR VENT Russell B. Schweickart and Gary Mills Ball Aerospace & Technologies Corp. Boulder, CO 80301 USA ABSTRACT The pressure of the cryogen
ANALYSIS AND TEST VERIFICATION OF TRANSITIONAL FLOW IN A DEWAR VENT Russell B. Schweickart and Gary Mills Ball Aerospace & Technologies Corp. Boulder, CO 80301 USA ABSTRACT The pressure of the cryogen
Performance Investigation on Electrochemical Compressor with Ammonia
 Purdue University Purdue e-pubs International Compressor Engineering Conference School of Mechanical Engineering 2016 Performance Investigation on Electrochemical Compressor with Ammonia Ye Tao University
Purdue University Purdue e-pubs International Compressor Engineering Conference School of Mechanical Engineering 2016 Performance Investigation on Electrochemical Compressor with Ammonia Ye Tao University
Chapter 10. Gases THREE STATES OF MATTER. Chapter 10 Problems 6/29/2012. Problems 16, 19, 26, 33, 39,49, 57, 61
 Chemistry, The Central Science, 11th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 10 John Bookstaver St. Charles Community College Cottleville, MO Chapter 10 Problems Problems
Chemistry, The Central Science, 11th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 10 John Bookstaver St. Charles Community College Cottleville, MO Chapter 10 Problems Problems
