Description of the three-phase contact line expansion
|
|
|
- Katrina Cobb
- 5 years ago
- Views:
Transcription
1 EPJ Web of Conferences 67, (2014) DOI: / epjconf/ C Owned by the authors, published by EDP Sciences, 2014 Description of the three-phase contact line expansion Tereza Váchová 1,a, Zuzana Brabcová 1 and Pavlína Basaová 1 1 Institute of Chemical Technology, Prague, Technická 5, Prague, Czech Republic Abstract. Knowledge of bubble-particle interaction is important in many industrial processes such as in flotation. While the collision (first interaction sub-process) between bubbles and particles is influenced only by hydrodynamic forces, the bubble behaviour during the attachment (second sub-process) is influenced both by hydrodynamic and surface forces. This work is focused on the study of the three-phase contact (TPC) line expansion during bubble adhesion on hydrophobic surface and on its experimental and mathematical description. The experiments were carried out in pure water where mobile bubble surface is expected. The rising bubble was studied in dynamic arrangement, whereas the stationary bubble was analysed in static arrangement. The attachment process was recorded using a high-speed digital camera and evaluated using image analysis. The diameter of the expanding TPC line as well as the dynamic contact angle was determined. Two approaches - the hydrodynamic and the molecular-kinetic - were used for mathematical description of the TPC line expansion. According to our results, the hydrodynamic model is suitable for the description of the initial fast phase of the expansion. The molecular-kinetic model was assessed as appropriate for almost whole range of TPC expansion. Parameters of the model were evaluated and compared for both types of arrangement. 1 Introduction Bubbles and drops are entities of enormous practical interest since their interfaces are encountered in numerous industrial processes and applications of everyday life. Froth flotation is just one of the examples in which bubble-particle attachment plays a vital role. Due to its high separation efficiency, cost effectiveness and simplicity in operation and maintenance, flotation has been extended to other industries dealing with solid-solid and solid-liquid separations. Although flotation phenomena have been known for centuries, the pioneering systematic analysis on the fundamental steps of bubble-particle interactions in flotation could only be traced back to about 50 years ago. In contrast with the mentioned efforts stemmed from the needs of mineral flotation aimed at particles much smaller than bubbles, this project is focused on the investigation of behaviour of bubbles smaller in size with particles, which has not been satisfactorily covered yet. It seems that a good starting point for the small particle large bubble interaction is the study of a bubble interaction with a flat wall, which represents a particle of infinite size. The bubble adhesion on the wall is controlled by the bubble ability to push out the liquid film, which separates them. For attachment, this film has to be thinned to the thickness of order of van der Waals and electrostatic forces range. These forces then destabilize the film, leading to its rupture and formation of the three-phase contact (TPC) line [1, 2]. Finally the three-phase contact line expands to the equilibrium. This project is focused on the study of the three-phase contact line during the bubble adhesion on the horizontal plane under two different experimental arrangements; stationary and dynamic. The experimental results are compared to model predictions, based on the hydrodynamic and molecular-kinetic theories. Fundamentals of the hydrodynamic model were given by Cox [3], Huh and Scriven [4] and Voinov [5] whose described the TPC line from viewpoint of fluid dynamics. The hydrodynamic approach assumes only the viscous friction as a dissipative force; dissipation at the TPC line is neglected. The first models break down in a close proximity to the TPC line. Due to the application of noslip condition of classical hydrodynamics the stress singularity occurs directly at the TPC line. To avoid such problem several models were proposed [4-6]. Generally, inner, intermediate and outer regions are distinguished for the description of the bubble close to the TPC line. The inner region applicable closest to the TPC line is determined by the slip length L s of order of molecular distance. At distance greater than L s the slip of the liquid along the solid surface occurs and stress singularity is removed. The TPC line motion in the inner region is influenced by molecular interactions and its shape could be represented by the microscopic contact angle m. The a Corresponding author: tereza.vachova@vscht.cz This is an Open Access article distributed under the terms of the Creative Commons Attribution License 2.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited. Article available at or
2 EPJ Web of Conferences size of the outer region is similar to bubble radius and is usually denoted as R. The outer region is also described by the macroscopic contact angle which is equal to the Young contact angle eq [6, 7]. The intermediate region where viscous forces and surface tension operate is characterized by the dynamic contact angle dyn. Such angle is dependent on the TPC line velocity and could be measured by available techniques [8]. After the equilibrium is established, the dynamic contact angle is equal to the Young contact angle. The stress influence is neither in the inner nor in the outer region so significant and contact angles eq and m remain during the TPC line motion almost constant. Phan [9] determined that the m is almost equal to eq for the glass/air bubble/pure water interface and this assumption is often used in the hydrodynamic model. Hocking, Rivers [7] and Cox [6] suggested the relation describing the difference between dynamic and microscopic contact angles. ηu ( ) ( ) dyn q θ m = ln R γ Ls q θ (1) Here represents the liquid viscosity, U is the TPC line velocity and is liquid surface tension. The expression q() for dyn < 3/4 could be simplified as 3 /9 [5]. Then one could obtain: 3 3 ηu R θ dyn θ m = 9 ln. (2) γ Ls When the microscopic contact angle is replaced by equilibrium contact angle [9], the combination of eq. (1) and (2) gives the following equation for the TPC line velocity and consequently for the radius of the TPC line r TPC : ( R L ) t 3 3 [ θ ( τ ) θ ] dτ r γ TPC () τ = 9η ln dyn eq. (3) s The term ln(r/l s ) appears here as an adjustable parameter. According to recent findings [9-11] the hydrodynamic model provides unreasonably large and thus physical unacceptable values of ln(r/l s ). It was concluded that the surface properties have to be considered to calculation of the TPC line velocity. The molecular-kinetic model follows up the principles of surface chemistry [3, 12]. The first description of the model was published by Eyring [13] and by Yarnold and Mason [12]. The theory is based on the description of molecules and ions transport from one site of the solid surface to another one. Blake and Haynes [3] assume an existence of a large number of identical adsorption sites at which molecules or ions of gas can adsorb. The adsorption sites determine the equilibrium position, activation energy and due to the hills and valleys also the energy barrier which has to be overcome. The molecules and ions displacement takes place in both directions (forward, which is denotes as +, and backward, denotes as - ) by jumping from one site to another [3, 14, 15]. According to the Eyring s [12, 13] 0 statistical treatment of the transport process the rate of displacements K ± for both direction is given as K ± k BT ε ± = exp, (4) h k BT where ε ± is the activation energy, k B Boltzmann constant, h Planck constant and T denotes the temperature. The work W done to the system to overcome the energy barrier could be expressed by equation: ( cosθ cosθ ) W = γ. (5) Here the work W represents the unbalanced capillary force in the case when the dynamic and equilibrium contact angle differs from each other. The TPC line velocity is expressed as dyn ( K ) + eq U = λ K (6) and according to [3] the radius of the TPC line is t 2 γλ ( τ ) 2 λ sinh ( cosθ () τ cosθ ) dτ 2 r TPC = K dyn eq. (7) 0 kbt Here K is the frequency of molecular displacement with a typical value s -1 [3, 9, 10, 16]. Parameter is the average jumping distance and its value should be of order of molecular dimensions ( 1 nm). It was found out that the molecular-kinetic model describes the TPC line motion very well. Agreement between calculated and experimental data is high and moreover, values of the adjustable parameters agree with the physical assumption [9, 11, 17]. Recently new models combining both approaches have been published [16, 18-20]. The basic assumption follows the idea that both the dynamic contact angle dyn and the microscopic contact angle m depend on the velocity. For the microscopic contact angle m one could obtain [18]: θ m ( U ) = arccoscosθ eq 2k BT U arcsinh. (8) 2 γλ 2Kλ After implementation of equation (8) into equation (1) and for dyn < 3/4 one will get following equation [16]: θ 3 dyn arccoscosθ 9ηU ln γ R L s = 0. eq 2k BT U arcsinh 2 γλ 2Kλ The velocity U and thus also the TPC line radius are related both to microscopic (K,, L s ) and macroscopic (, R,, ) properties of the system. Although the molecularkinetic model fits the experimental data very well, this approach gives more precise prediction of the TPC line velocity in its whole range [18]. 3 (9) p.2
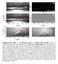
3 EFM 2013 diameter dtpc were determined. Contact angles were measured using the modified ADSA methodology [24]. 2 Experiments The experimental measurements were done in a special glass flotation cell (5.5 cm height, 8 cm width and 14 cm depth). The scheme of the experimental apparatus is given in figure 1. Single bubbles with diameters from 0.4 to 0.7 mm were created using a bubble generator built according to Vejražka [21]. The bubble collided with a microscope slide glass placed on a prism with a horizontal plane. The glass was silanized by dimethyldichlorsilan (CH3)2SiCl2 provided by SigmaAldrich as the Silanization solution I. According to Zisman s method the solid surface energy Ȗsg of silanised glass was 23.1 mn/m. Experiments were provided in pure distilled, de-ionised and de-mineralised water at 26 C. The surface tension was 71.6 mn/m, ph value was 6.13 and conductivity 1.6 ȝs/cm. Equilibrium contact angle 97.8 for a sessile drop of pure water on this surface was measured using the ADSA technique. 3 Results The typical experiment is illustrated in figure 2. The adhesion of a bubble with diameter 0.57 mm (dynamic arrangement) and 0.60 mm (stationary arrangement) is given here. The time when the rupture of thin liquid film occurs was set as a zero time. After this moment the TPC line expansion was observed. After 0.1 s the equilibrium was almost achieved. The capturing was interrupted after 0.3 s. At this time the bubble shape was stable and the diameter of the TPC line together with contact angles did not change for more then 0.1 s. Figure 2. Sequence of photos showing the bubble adhesion on horizontal plane under dynamic (a) and stationary (b) experimental arrangement. Figure 1. Scheme of experimental setup. Whole experiment took place under two arrangements. Dynamic arrangement is characterized by bubble rising throught stagnant liquid before its collision with solid particle. In stationary arrangement the stagnant bubble collides with a solid surface. Whereas in dynamic arrangement the kinetic energy of the bubble plays an important role, in static arrangement the bubble kinetic energy is negligible. The collision process and the bubble adhesion were recorded using the high-speed digital camera Redlake Motion Scope M2 (2000 fps, resolution 1280 x 256 pixels, image calibration µm/px). Around 1000 frames were taken for each sequence. The experimental terminal velocity of bubble rise was measured and compared with the prediction of Mei [22, 23] which is valid for pure water. The velocity calculated using this model is in close agreement with the experimental data, suggesting that the bubble interface is not contaminated with any additives or impurities. Altogether, 6 sets of experimental data were obtained. These sets differ in bubble size (db = about 0.4, 0.6 and 0.7 mm) and experimental arrangement (dynamic x stationary). For one experimental set, adhesion process of ten bubbles was captured. Within one set of data, the variation of bubble size was about 1% and the maximum deviation from the mean did not exceed 3%. All images were evaluated using the image analysis software NISElements when the bubble radius rb and the TPC line At first, the suitability of the hydrodynamic and the molecular-kinetic model was tested. The experimental radius of the TPC line rtpc together with contact angle on both sides of the bubble was measured. Due to the fact that the difference between these two contact angles does not exceed 2, their average value was taken as the dynamic contact angle șdyn. The equilibrium contact angle șeq was evaluated too. The average value was 94. Finally, the parameter ln(r/ls) of the hydrodynamic model (eq. 3) and parameters Ȝ and K of the molecularkinetic model (eq. 7) were evaluated using the regression analysis. The results for bubble with db = 0.44 mm are illustrated in figure 3, where the time dependence of radius of the TPC line rtpc is given. The experimental data are depicted as circles; rtpc calculated using the hydrodynamic model is illustrated as the dash line and rtpc calculated using the molecular-kinetic model is illustrated as the full line. The hydrodynamic model describes the motion of the liquid near the interface and it could be appropriate for description of the first fast phase of the expansion. However, its parameter ln(r/ls) is disproportionately high. If we consider the bubble radius rb smaller then 0.5 mm and the slip length Ls close to 1 nm, the parameter ln(r/ls) should reach the value close to 13. The value calculated from experimental data is about 100. Our observations are in accordance with previously published results [9-11] pointing out this drawback of the hydrodynamic model. On the other hand, the molecular-kinetic model fits experimental data very well. Both adjustable parameters Ȝ and K reached values which are in a good agreement with theoretical p.3
4 EPJ Web of Conferences predictions [3, 9, 10, 16]. Therefore only the molecularkinetic model was considered for next calculations. distance decreases. It could be concluded that the bubble motion preceding the bubble adhesion has an appreciable influence on the TPC line dynamics. Figure 3. Expansion of radius of the three-phase contact line during the bubble adhesion. Experimental data (d b = 0.44 mm) and comparison of molecular-kinetic and hydrodynamic model. The calculated parameters and K of the molecularkinetic model are summarized in table 1 together with sample variance and eq for each bubble size and equilibrium angle. Table 1. Molecular-kinetic model parameters values. d b (mm) K (s -1 ) (nm) R 2 eq ( ) Dynamic arrangement x x x Stationary arrangement x x x The comparison of experimental and calculated r TPC is illustrated in figure 4 for both experimental arrangements. Very good agreement between experimental and calculated TPC line radius r TPC is observed for both dynamic and stationary arrangement and moreover for all bubble diameters. Sample variances range from 0.97 to The fitted parameteres and K correspond with theoretical predictions. Average jumping distance ranges from 0.65 to 0.77 nm which is in agreement with assumption of the order of molecular dimension ( nm). The frequency of molecular displacement K moves within the order of 10 6 s -1 which is physically correct and similar to previously published data [19, 25]. The molecular-kinetic model was determined as an appropriate one for almost the whole range of TPC expansion and can be thus used for the description of the TPC line motion. When results for dynamic and stationary arrangement are compared one could observe the significant difference. For the stationary bubble the frequency of molecular displacement K is more than twice higher than for the rising bubble. Contrarily the average jumping Figure 4. Comparison of experimental radii of the TPC line with molecular-kinetic prediction for dynamic and stationary experimental arrangement. The parameters and K of the molecular-kinetic model are obtained by means of the regression analysis. The model should describe both the initial fast part and the following slow phase of bubble adhesion. Unfortunately the number of experimental points is significantly lower for the fast part of adhesion which is crutial for the TPC enlargement [26]. The influence of number of points (expressed as time dependence) on parameters and K is illustrated in figure 5 for both dynamic and stationary experimental arrangements. While parameter increases slightly with growing time interval, parameter K has decreasing trend. It declines considerabely, primarily in the first phase of the expansion. On the other hand, increase in points number results in substantial accuracy decrease in the crutial fast part. Therefore only the limited number of experimental points should be reccommended for the regression [26]. In this project the maximum time was s. Parametres given in table 1 were calculated under this condition. Authors believe the accuracy to be sufficient p.4
5 EFM 2013 Figure 5. Time dependence of adjustable parameters and K of the molecular-kinetic model for dynamic and stationary experimental arrangement. 4 Conclusions The expansion of the TPC line for a small bubble adhering on the horizontal hydrophobic surface was studied for the dynamic and the stationary experimental arrangements provided that the bubble surface is mobile. The experimental TPC radii and contact angles were obtained using image analysis. Mathematical models (hydrodynamic and molecular-kinetic) were tested for the theoretical description of the TPC line expansion. While the hydrodynamic model fails, the molecular-kinetic model describes experimental data with excellent agreement. Values of both parameters (the frequency of molecular displacement K and the average jumping distance ) correspond to physical conditions. These parameters depend on bubble size and also vary for different experimental arrangements. Therefore it was concluded that the bubble motion preceding the bubble adhesion has an appreciable influence on the TPC line dynamics. Acknowledgements This work was supported by the Czech Science Foundation (project 101/11/0806) and by the Ministry of Education, Youth and Sports (project LD 13025) and by specific university research (MSMT no. 20/2013). Likewise the support from COST action MP1106 was appreciated. References 1. A.V. Nguyen and H.J. Schulze, Colloidal science of flotation. first edn. (Marcel Dekker New York, 2004) 2. P.G. de Gennes, F. Brochard-Wyart, and D. Quéré, Capillarity and wetting phenomena: drops, bubbles, pearls, waves. first edn. (Springer New York, 2004) 3. T.D. Blake and J.M. Haynes, J Colloid Interf Sci. 30, 421 (1969) 4. C. Huh and L.E. Scriven, J Colloid Interf Sci. 35, 85 (1971) 5. O.V. Voinov, Fluid Dynamics 11, 714 (1976) 6. R.G. Cox, J Fluid Mech. 168, 169 (1986) 7. L.M. Hocking and A.D. Rivers, J Fluid Mech. 121, 425 (1982) 8. D.Y. Kwok and A.W. Neumann, Adv Colloid Interfac. 81, 167 (1999) 9. C.M. Phan, A.V. Nguyen, and G.M. Evans, Langmuir. 19, 6796 (2003) 10. R. Fetzer and J. Ralston, J Phys Chem C. 113, 8888 (2009) 11. A.V. Nguyen, et al., Miner Eng. 19, 651 (2006) 12. G.D. Yarnold and B.J. Mason, Proc. Phys. Soc. B. 62, 121 (1948) 13. S. Glasstone, K.J. Laidler, and H. Eyring, The theory of rate processes; the kinetics of chemical reactions, p.5
6 EPJ Web of Conferences viscosity, diffusion and electrochemical phenomena. first edn. (McGraw-Hill Book Company, inc. New York; London,, 1941) 14. B.W. Cherry and C.M. Holmes, J Colloid Interf Sci. 29, 174 (1969) 15. F. Brochard-Wyart and P.G. de Gennes, Adv Colloid Interfac. 39, 1 (1992) 16. S.R. Ranabothu, C. Karnezis, and L.L. Dai, J Colloid Interf Sci. 288, 213 (2005) 17. T. Váchová, P. Basaová, and Z. Brabcová, Procedia Engineering. 42, 1897 (2012) 18. P.G. Petrov and J.G. Petrov, Langmuir. 8, 1762 (1992) 19. C.M. Phan, A.V. Nguyen, and G.M. Evans, J Colloid Interf Sci. 296, 669 (2006) 20. J.G. Petrov and B.P. Radoev, Colloid Polym Sci. 259, 753 (1981) 21. J. Vejrazka, et al., Fluid Dyn Res. 40, 521 (2008) 22. R.W. Mei and R.J. Adrian, J Fluid Mech. 237, 323 (1992) 23. R.W. Mei, J.F. Klausner, and C.J. Lawrence, Phys Fluids. 6, 418 (1994) 24. Z. Brabcová, P. Basaová, and T. Váchová, Procedia Engineering. 42, 358 (2012) 25. M. Schneemilch, et al., Langmuir. 14, 7047 (1998) 26. T. Blake, Private comunication p.6
contact line dynamics
 contact line dynamics Jacco Snoeijer Physics of Fluids - University of Twente sliding drops flow near contact line static contact line Ingbrigtsen & Toxvaerd (2007) γ γ sv θ e γ sl molecular scales macroscopic
contact line dynamics Jacco Snoeijer Physics of Fluids - University of Twente sliding drops flow near contact line static contact line Ingbrigtsen & Toxvaerd (2007) γ γ sv θ e γ sl molecular scales macroscopic
Hydrodynamics of wetting phenomena. Jacco Snoeijer PHYSICS OF FLUIDS
 Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
Hydrodynamics of wetting phenomena Jacco Snoeijer PHYSICS OF FLUIDS Outline 1. Creeping flow: hydrodynamics at low Reynolds numbers (2 hrs) 2. Thin films and lubrication flows (3 hrs + problem session
ISCST shall not be responsible for statements or opinions contained in papers or printed in its publications.
 Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
Modeling of Drop Motion on Solid Surfaces with Wettability Gradients J. B. McLaughlin, Sp. S. Saravanan, N. Moumen, and R. S. Subramanian Department of Chemical Engineering Clarkson University Potsdam,
On the Landau-Levich Transition
 10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
10116 Langmuir 2007, 23, 10116-10122 On the Landau-Levich Transition Maniya Maleki Institute for AdVanced Studies in Basic Sciences (IASBS), Zanjan 45195, P.O. Box 45195-1159, Iran Etienne Reyssat and
Derivation of continuum models for the moving contact line problem based on thermodynamic principles. Abstract
 Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
Derivation of continuum models for the moving contact line problem based on thermodynamic principles Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 002, USA Weinan
the moving contact line
 Molecular hydrodynamics of the moving contact line Tiezheng Qian Mathematics Department Hong Kong University of Science and Technology in collaboration with Ping Sheng (Physics Dept, HKUST) Xiao-Ping Wang
Molecular hydrodynamics of the moving contact line Tiezheng Qian Mathematics Department Hong Kong University of Science and Technology in collaboration with Ping Sheng (Physics Dept, HKUST) Xiao-Ping Wang
Analysis of Two-Layered Journal Bearing Lubricated with Ferrofluid
 MATEC Web of Conferences 1, 41 (14) DOI: 1.151/ matecconf/ 141 41 C Owned by the authors, published by EDP Sciences, 14 Analysis of Two-Layered Journal Bearing Lubricated with Ferrofluid T. V. V. L. N.
MATEC Web of Conferences 1, 41 (14) DOI: 1.151/ matecconf/ 141 41 C Owned by the authors, published by EDP Sciences, 14 Analysis of Two-Layered Journal Bearing Lubricated with Ferrofluid T. V. V. L. N.
Contents. Preface XI Symbols and Abbreviations XIII. 1 Introduction 1
 V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
V Contents Preface XI Symbols and Abbreviations XIII 1 Introduction 1 2 Van der Waals Forces 5 2.1 Van der Waals Forces Between Molecules 5 2.1.1 Coulomb Interaction 5 2.1.2 Monopole Dipole Interaction
spreading of drops on soft surfaces
 Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Supplementary Material on Electrically modulated dynamic spreading of drops on soft surfaces Ranabir Dey 1, Ashish Daga 1, Sunando DasGupta 2,3, Suman Chakraborty 1,3 1 Department of Mechanical Engineering,
Shear Thinning Near the Rough Boundary in a Viscoelastic Flow
 Advanced Studies in Theoretical Physics Vol. 10, 2016, no. 8, 351-359 HIKARI Ltd, www.m-hikari.com http://dx.doi.org/10.12988/astp.2016.6624 Shear Thinning Near the Rough Boundary in a Viscoelastic Flow
Advanced Studies in Theoretical Physics Vol. 10, 2016, no. 8, 351-359 HIKARI Ltd, www.m-hikari.com http://dx.doi.org/10.12988/astp.2016.6624 Shear Thinning Near the Rough Boundary in a Viscoelastic Flow
Solvability condition for the moving contact line
 PHYSICAL REVIEW E 78, 564 28 Solvability condition for the moving contact line L. M. Pismen 1 and Jens Eggers 2 1 Department of Chemical Engineering and Minerva Center for Nonlinear Physics of Complex
PHYSICAL REVIEW E 78, 564 28 Solvability condition for the moving contact line L. M. Pismen 1 and Jens Eggers 2 1 Department of Chemical Engineering and Minerva Center for Nonlinear Physics of Complex
Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supplementary Information on Thermally Enhanced Self-Propelled Droplet Motion on Gradient Surfaces
Boundary Conditions for the Moving Contact Line Problem. Abstract
 Boundary Conditions for the Moving Contact Line Problem Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 10012, USA Weinan E Department of Mathematics and PACM,
Boundary Conditions for the Moving Contact Line Problem Weiqing Ren Courant Institute of Mathematical Sciences, New York University, New York, NY 10012, USA Weinan E Department of Mathematics and PACM,
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa.
 Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Supplementary Figure 1 Extracting process of wetting ridge profiles. a1-4, An extraction example of a ridge profile for E 16 kpa. An original image (a1) was binarized, as shown in a2, by Canny edge detector
Capillarity and Wetting Phenomena
 ? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
? Pierre-Gilles de Gennes Frangoise Brochard-Wyart David Quere Capillarity and Wetting Phenomena Drops, Bubbles, Pearls, Waves Translated by Axel Reisinger With 177 Figures Springer Springer New York Berlin
Lecture 7 Contact angle phenomena and wetting
 Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Lecture 7 Contact angle phenomena and Contact angle phenomena and wetting Young s equation Drop on the surface complete spreading Establishing finite contact angle γ cosθ = γ γ L S SL γ S γ > 0 partial
Kinetic Slip Condition, van der Waals Forces, and Dynamic. Contact Angle. Abstract
 Kinetic Slip Condition, van der Waals Forces, and Dynamic Contact Angle Len M. Pismen and Boris Y. Rubinstein Department of Chemical Engineering, Technion Israel Institute of Technology, Haifa 3000, Israel.
Kinetic Slip Condition, van der Waals Forces, and Dynamic Contact Angle Len M. Pismen and Boris Y. Rubinstein Department of Chemical Engineering, Technion Israel Institute of Technology, Haifa 3000, Israel.
Jacco Snoeijer PHYSICS OF FLUIDS
 Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Jacco Snoeijer PHYSICS OF FLUIDS dynamics dynamics freezing dynamics freezing microscopics of capillarity Menu 1. surface tension: thermodynamics & microscopics 2. wetting (statics): thermodynamics & microscopics
Optical Measurements of Cavitation in Tribological Contacts
 Journal of Physics: Conference Series PAPER OPEN ACCESS Optical Measurements of Cavitation in Tribological Contacts To cite this article: Tian Tang et al 2015 J. Phys.: Conf. Ser. 656 012119 View the article
Journal of Physics: Conference Series PAPER OPEN ACCESS Optical Measurements of Cavitation in Tribological Contacts To cite this article: Tian Tang et al 2015 J. Phys.: Conf. Ser. 656 012119 View the article
Boundary Conditions in Fluid Mechanics
 Boundary Conditions in Fluid Mechanics R. Shankar Subramanian Department of Chemical and Biomolecular Engineering Clarkson University The governing equations for the velocity and pressure fields are partial
Boundary Conditions in Fluid Mechanics R. Shankar Subramanian Department of Chemical and Biomolecular Engineering Clarkson University The governing equations for the velocity and pressure fields are partial
Generalized Wenzel equation for contact angle of droplets on spherical rough solid substrates
 Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Science Front Publishers Journal for Foundations and Applications of Physics, 3 (2), (2016) (sciencefront.org) ISSN 2394-3688 Generalized Wenzel equation for contact angle of droplets on spherical rough
Initial position, x p (0)/L
 .4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
.4 ) xp().2 ) ( 2L 2 xp Dc ( Displacement, /L.2.4.5.5 Initial position, x p ()/L Supplementary Figure Computed displacements of (red) positively- and (blue) negatively-charged particles at several CO 2
REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE
 REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE Hossein Azizinaghsh a, Hamid Ghaednia b a Sharif University of Technology, School of Computer Engineering, I. R. Iran Hossein.Azizi@gmail.com
REMOVING A PIECE OF ADHESIVE TAPE FROM A HORIZONTAL SURFACE Hossein Azizinaghsh a, Hamid Ghaednia b a Sharif University of Technology, School of Computer Engineering, I. R. Iran Hossein.Azizi@gmail.com
Physics and Chemistry of Interfaces
 Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Hans Jürgen Butt, Karlheinz Graf, and Michael Kappl Physics and Chemistry of Interfaces Second, Revised and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA Contents Preface XI 1 Introduction
Interfacial Flows of Contact Line Dynamics and Liquid Displacement in a Circular Microchannel
 Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Proceedings of the 3 rd World Congress on Mechanical, Chemical, and Material Engineering (MCM'17) Rome, Italy June 8 10, 2017 Paper No. HTFF 159 ISSN: 2369-8136 DOI: 10.11159/htff17.159 Interfacial Flows
Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate
 Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate Derrick O. Njobuenwu * Department of Chemical Engineering, Loughborough University Leicestershire LE11 3TU,
Complete Wetting of Acrylic Solid Substrate with Silicone Oil at the Center of the Substrate Derrick O. Njobuenwu * Department of Chemical Engineering, Loughborough University Leicestershire LE11 3TU,
Line Tension Effect upon Static Wetting
 Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
Line Tension Effect upon Static Wetting Pierre SEPPECHER Université de Toulon et du Var, BP 132 La Garde Cedex seppecher@univ tln.fr Abstract. Adding simply, in the classical capillary model, a constant
PERTURBATION ANALYSIS OF k ω AND k ɛ TURBULENT MODELS. WALL FUNCTIONS
 EPJ Web of Conferences 45, 01097 2013 DOI: 10.1051/ epjconf/ 20134501097 C Owned by the authors, published by EDP Sciences, 2013 PERTURBATION ANALYSIS OF k ω AND k ɛ TURBULENT MODELS. WALL FUNCTIONS Karel
EPJ Web of Conferences 45, 01097 2013 DOI: 10.1051/ epjconf/ 20134501097 C Owned by the authors, published by EDP Sciences, 2013 PERTURBATION ANALYSIS OF k ω AND k ɛ TURBULENT MODELS. WALL FUNCTIONS Karel
A General Equation for Fitting Contact Area and Friction vs Load Measurements
 Journal of Colloid and Interface Science 211, 395 400 (1999) Article ID jcis.1998.6027, available online at http://www.idealibrary.com on A General Equation for Fitting Contact Area and Friction vs Load
Journal of Colloid and Interface Science 211, 395 400 (1999) Article ID jcis.1998.6027, available online at http://www.idealibrary.com on A General Equation for Fitting Contact Area and Friction vs Load
Chapter 10. Solids and Fluids
 Chapter 10 Solids and Fluids Surface Tension Net force on molecule A is zero Pulled equally in all directions Net force on B is not zero No molecules above to act on it Pulled toward the center of the
Chapter 10 Solids and Fluids Surface Tension Net force on molecule A is zero Pulled equally in all directions Net force on B is not zero No molecules above to act on it Pulled toward the center of the
A rate-dependent Hosford-Coulomb model for predicting ductile fracture at high strain rates
 EPJ Web of Conferences 94, 01080 (2015) DOI: 10.1051/epjconf/20159401080 c Owned by the authors, published by EDP Sciences, 2015 A rate-dependent Hosford-Coulomb model for predicting ductile fracture at
EPJ Web of Conferences 94, 01080 (2015) DOI: 10.1051/epjconf/20159401080 c Owned by the authors, published by EDP Sciences, 2015 A rate-dependent Hosford-Coulomb model for predicting ductile fracture at
Analytical Prediction of Particle Detachment from a Flat Surface by Turbulent Air Flows
 Chiang Mai J. Sci. 2011; 38(3) 503 Chiang Mai J. Sci. 2011; 38(3) : 503-507 http://it.science.cmu.ac.th/ejournal/ Short Communication Analytical Prediction of Particle Detachment from a Flat Surface by
Chiang Mai J. Sci. 2011; 38(3) 503 Chiang Mai J. Sci. 2011; 38(3) : 503-507 http://it.science.cmu.ac.th/ejournal/ Short Communication Analytical Prediction of Particle Detachment from a Flat Surface by
This is a Gaussian probability centered around m = 0 (the most probable and mean position is the origin) and the mean square displacement m 2 = n,or
 Physics 7b: Statistical Mechanics Brownian Motion Brownian motion is the motion of a particle due to the buffeting by the molecules in a gas or liquid. The particle must be small enough that the effects
Physics 7b: Statistical Mechanics Brownian Motion Brownian motion is the motion of a particle due to the buffeting by the molecules in a gas or liquid. The particle must be small enough that the effects
An energy balance approach to modeling the hydrodynamically driven spreading of a liquid drop
 Colloids and Surfaces A: Physicochemical and Engineering Aspects 182 (2001) 109 122 www.elsevier.nl/locate/colsurfa An energy balance approach to modeling the hydrodynamically driven spreading of a liquid
Colloids and Surfaces A: Physicochemical and Engineering Aspects 182 (2001) 109 122 www.elsevier.nl/locate/colsurfa An energy balance approach to modeling the hydrodynamically driven spreading of a liquid
Slow viscous flow in a microchannel with similar and different superhydrophobic walls
 Journal of Physics: Conference Series PAPER OPEN ACCESS Slow viscous flow in a microchannel with similar and different superhydrophobic walls To cite this article: A I Ageev and A N Osiptsov 2018 J. Phys.:
Journal of Physics: Conference Series PAPER OPEN ACCESS Slow viscous flow in a microchannel with similar and different superhydrophobic walls To cite this article: A I Ageev and A N Osiptsov 2018 J. Phys.:
Particle removal in linear shear flow: model prediction and experimental validation
 Particle removal in linear shear flow: model prediction and experimental validation M.L. Zoeteweij, J.C.J. van der Donck and R. Versluis TNO Science and Industry, P.O. Box 155, 600 AD Delft, The Netherlands
Particle removal in linear shear flow: model prediction and experimental validation M.L. Zoeteweij, J.C.J. van der Donck and R. Versluis TNO Science and Industry, P.O. Box 155, 600 AD Delft, The Netherlands
Modelling and analysis for contact angle hysteresis on rough surfaces
 Modelling and analysis for contact angle hysteresis on rough surfaces Xianmin Xu Institute of Computational Mathematics, Chinese Academy of Sciences Collaborators: Xiaoping Wang(HKUST) Workshop on Modeling
Modelling and analysis for contact angle hysteresis on rough surfaces Xianmin Xu Institute of Computational Mathematics, Chinese Academy of Sciences Collaborators: Xiaoping Wang(HKUST) Workshop on Modeling
contact line dynamics
 contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
contact line dynamics part 2: hydrodynamics dynamic contact angle? lubrication: Cox-Voinov theory maximum speed for instability corner shape? dimensional analysis: speed U position r viscosity η pressure
arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000
![arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000 arxiv:cond-mat/ v1 [cond-mat.soft] 18 Sep 2000](/thumbs/81/83362442.jpg) arxiv:cond-mat/0009259v1 [cond-mat.soft] 18 Sep 2000 Two remarks on wetting and emulsions P. G. de Gennes Collège de France, 11 place M. Berthelot, 75231 Paris Cedex, France June 12, 2018 Abstract This
arxiv:cond-mat/0009259v1 [cond-mat.soft] 18 Sep 2000 Two remarks on wetting and emulsions P. G. de Gennes Collège de France, 11 place M. Berthelot, 75231 Paris Cedex, France June 12, 2018 Abstract This
Velocity profiles of fluid flow close to a hydrophobic surface
 Velocity profiles of fluid flow close to a hydrophobic surface Simona Fialová 1, František Pochylý 1, Michal Kotek 2, Darina Jašíková 2 1 Faculty of mechanical engineering, Brno University of Technology,
Velocity profiles of fluid flow close to a hydrophobic surface Simona Fialová 1, František Pochylý 1, Michal Kotek 2, Darina Jašíková 2 1 Faculty of mechanical engineering, Brno University of Technology,
BRIEF COMMUNICATION TO SURFACES ANALYSIS OF ADHESION OF LARGE VESICLES
 BRIEF COMMUNICATION ANALYSIS OF ADHESION OF LARGE VESICLES TO SURFACES EVAN A. EVANS, Department ofbiomedical Engineering, Duke University, Durham, North Carolina 27706 U.S.A. ABSTRACT An experimental
BRIEF COMMUNICATION ANALYSIS OF ADHESION OF LARGE VESICLES TO SURFACES EVAN A. EVANS, Department ofbiomedical Engineering, Duke University, Durham, North Carolina 27706 U.S.A. ABSTRACT An experimental
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE
 MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
Praktikum zur. Materialanalytik
 Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Praktikum zur Materialanalytik Functionalized Surfaces B510 Stand: 20.10.2017 Table of contents Introduction 2 Basics 2 Surface tension 2 From wettability to the contact angle 4 The Young equation 5 Wetting
Surface and Interfacial Tensions. Lecture 1
 Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
Surface and Interfacial Tensions Lecture 1 Surface tension is a pull Surfaces and Interfaces 1 Thermodynamics for Interfacial Systems Work must be done to increase surface area just as work must be done
The influence of axial orientation of spheroidal particles on the adsorption
 The influence of axial orientation of spheroidal particles on the adsorption rate in a granular porous medium F. A. Coutelieris National Center for Scientific Research Demokritos, 1510 Aghia Paraskevi
The influence of axial orientation of spheroidal particles on the adsorption rate in a granular porous medium F. A. Coutelieris National Center for Scientific Research Demokritos, 1510 Aghia Paraskevi
ASPHALT WETTING DYNAMICS
 ASPHALT WETTING DYNAMICS Troy Pauli, Fran Miknis, Appy Beemer, Julie Miller, Mike Farrar, and Will Wiser 5 TH Annual Pavement Performance Prediction Symposium Adhesion & Cohesion of Asphalt Pavements Cheyenne,
ASPHALT WETTING DYNAMICS Troy Pauli, Fran Miknis, Appy Beemer, Julie Miller, Mike Farrar, and Will Wiser 5 TH Annual Pavement Performance Prediction Symposium Adhesion & Cohesion of Asphalt Pavements Cheyenne,
Numerical study of the structure flow of the gas-vapor mixture in a channel with injection of water droplets
 EPJ Web of Conferences 76, 01027 (2014) DOI: 10.1051/epjconf/20147601027 C Owned by the authors, published by EDP Sciences, 2014 Numerical study of the structure flow of the gas-vapor mixture in a channel
EPJ Web of Conferences 76, 01027 (2014) DOI: 10.1051/epjconf/20147601027 C Owned by the authors, published by EDP Sciences, 2014 Numerical study of the structure flow of the gas-vapor mixture in a channel
Supplementary Material
 1 2 3 Topological defects in confined populations of spindle-shaped cells by G. Duclos et al. Supplementary Material 4 5 6 7 8 9 10 11 12 13 Supplementary Note 1: Characteristic time associated with the
1 2 3 Topological defects in confined populations of spindle-shaped cells by G. Duclos et al. Supplementary Material 4 5 6 7 8 9 10 11 12 13 Supplementary Note 1: Characteristic time associated with the
PHYSICS OF FLUID SPREADING ON ROUGH SURFACES
 INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
INTERNATIONAL JOURNAL OF NUMERICAL ANALYSIS AND MODELING Volume 5, Supp, Pages 85 92 c 2008 Institute for Scientific Computing and Information PHYSICS OF FLUID SPREADING ON ROUGH SURFACES K. M. HAY AND
Comparison of pool boiling heat transfer for different tunnel-pore surfaces
 EPJ Web of Conferences, 9 () DOI:./ epjconf/9 C Owned by the authors, published by EDP Sciences, Comparison of pool boiling heat transfer for different nel-pore surfaces Robert Pastuszko,a Kielce University
EPJ Web of Conferences, 9 () DOI:./ epjconf/9 C Owned by the authors, published by EDP Sciences, Comparison of pool boiling heat transfer for different nel-pore surfaces Robert Pastuszko,a Kielce University
Study of Rupture Directivity in a Foam Rubber Physical Model
 Progress Report Task 1D01 Study of Rupture Directivity in a Foam Rubber Physical Model Rasool Anooshehpoor and James N. Brune University of Nevada, Reno Seismological Laboratory (MS/174) Reno, Nevada 89557-0141
Progress Report Task 1D01 Study of Rupture Directivity in a Foam Rubber Physical Model Rasool Anooshehpoor and James N. Brune University of Nevada, Reno Seismological Laboratory (MS/174) Reno, Nevada 89557-0141
Influence of the Flow Direction on the Mass Transport of Vapors Through Membranes Consisting of Several Layers
 Influence of the Flow Direction on the Mass Transport of Vapors Through Membranes Consisting of Several Layers Thomas Loimer 1,a and Petr Uchytil 2,b 1 Institute of Fluid Mechanics and Heat Transfer, Vienna
Influence of the Flow Direction on the Mass Transport of Vapors Through Membranes Consisting of Several Layers Thomas Loimer 1,a and Petr Uchytil 2,b 1 Institute of Fluid Mechanics and Heat Transfer, Vienna
Differential criterion of a bubble collapse in viscous liquids
 PHYSICAL REVIEW E VOLUME 60, NUMBER 1 JULY 1999 Differential criterion of a bubble collapse in viscous liquids Vladislav A. Bogoyavlenskiy* Low Temperature Physics Department, Moscow State University,
PHYSICAL REVIEW E VOLUME 60, NUMBER 1 JULY 1999 Differential criterion of a bubble collapse in viscous liquids Vladislav A. Bogoyavlenskiy* Low Temperature Physics Department, Moscow State University,
DLVO interaction between the spheres
 DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
DLVO interaction between the spheres DL-interaction energy for two spheres: D w ( x) 64c π ktrϕ e λ DL 2 x λ 2 0 0 D DLVO interaction w ( x) 64πkTRϕ e λ DLVO AR /12x 2 x λd 2 0 D Lecture 11 Contact angle
The dynamics of small particles whose size is roughly 1 µmt or. smaller, in a fluid at room temperature, is extremely erratic, and is
 1 I. BROWNIAN MOTION The dynamics of small particles whose size is roughly 1 µmt or smaller, in a fluid at room temperature, is extremely erratic, and is called Brownian motion. The velocity of such particles
1 I. BROWNIAN MOTION The dynamics of small particles whose size is roughly 1 µmt or smaller, in a fluid at room temperature, is extremely erratic, and is called Brownian motion. The velocity of such particles
Experimentally determined distribution of granular-flow characteristics in collisional bed load transport
 Experimentally determined distribution of granular-flow characteristics in collisional bed load transport Václav Matoušek 1,*, Štěpán Zrostlík 1, Luigi Fraccarollo 2, Anna Prati 2, and Michele Larcher
Experimentally determined distribution of granular-flow characteristics in collisional bed load transport Václav Matoušek 1,*, Štěpán Zrostlík 1, Luigi Fraccarollo 2, Anna Prati 2, and Michele Larcher
On Relationship between PVT and Rheological Measurements of Polymer Melts
 ANNUAL TRANSACTIONS OF THE NORDIC RHEOLOGY SOCIETY, VOL. 3, 2005 On Relationship between PVT and Rheological Measurements of Polymer Melts Tomas Sedlacek, Peter Filip 2, Peter Saha Polymer Centre, Faculty
ANNUAL TRANSACTIONS OF THE NORDIC RHEOLOGY SOCIETY, VOL. 3, 2005 On Relationship between PVT and Rheological Measurements of Polymer Melts Tomas Sedlacek, Peter Filip 2, Peter Saha Polymer Centre, Faculty
Vibration of submillimeter-size supported droplets
 PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
PHYSICAL REVIEW E 73, 041602 2006 Vibration of submillimeter-size supported droplets Franck Celestini* and Richard Kofman Laboratoire de Physique de la Matière Condensée, UMR 6622, CNRS, Université de
Foundations of. Colloid Science SECOND EDITION. Robert J. Hunter. School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS
 Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Foundations of Colloid Science SECOND EDITION Robert J. Hunter School of Chemistry University of Sydney OXPORD UNIVERSITY PRESS CONTENTS 1 NATURE OF COLLOIDAL DISPERSIONS 1.1 Introduction 1 1.2 Technological
Digital Spectrometric System for Characterization of Mixed Neutron Gamma Field in the Experimental Reactor LR-0
 EPJ Web of Conferences 106, 05008 (2016) DOI: 10.1051/epjconf/201610605008 C Owned by the authors, published by EDP Sciences, 2016 Digital Spectrometric System for Characterization of Mixed Neutron Gamma
EPJ Web of Conferences 106, 05008 (2016) DOI: 10.1051/epjconf/201610605008 C Owned by the authors, published by EDP Sciences, 2016 Digital Spectrometric System for Characterization of Mixed Neutron Gamma
Lecture 6 Friction. Friction Phenomena Types of Friction
 Lecture 6 Friction Tangential forces generated between contacting surfaces are called friction forces and occur to some degree in the interaction between all real surfaces. whenever a tendency exists for
Lecture 6 Friction Tangential forces generated between contacting surfaces are called friction forces and occur to some degree in the interaction between all real surfaces. whenever a tendency exists for
PreClass Notes: Chapter 5, Sections 5.4,5.5
 PreClass Notes: Chapter 5, Sections 5.4,5.5 From Essential University Physics 3 rd Edition by Richard Wolfson, Middlebury College 2016 by Pearson Education, Inc. Narration and extra little notes by Jason
PreClass Notes: Chapter 5, Sections 5.4,5.5 From Essential University Physics 3 rd Edition by Richard Wolfson, Middlebury College 2016 by Pearson Education, Inc. Narration and extra little notes by Jason
The Origins of Surface and Interfacial Tension
 The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
The Origins of Surface and Interfacial Tension Imbalance of intermolecular forces exists at the liquid-air interface γ la= the surface tension that exists at the liquid-air interface Suppose we have a
Supplementary table I. Table of contact angles of the different solutions on the surfaces used here. Supplementary Notes
 1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
1 Supplementary Figure 1. Sketch of the experimental setup (not to scale) : it consists of a thin mylar sheet (0, 02 4 3cm 3 ) held fixed vertically. The spacing y 0 between the glass plate and the upper
arxiv: v1 [cond-mat.soft] 26 Jan 2016
![arxiv: v1 [cond-mat.soft] 26 Jan 2016 arxiv: v1 [cond-mat.soft] 26 Jan 2016](/thumbs/73/68299281.jpg) Relaxation of nonspherical sessile drops towards equilibrium Vadim S. Nikolayev and Daniel A. Beysens ESEME, Service des Basses Températures, CEA-Grenoble (France) (Dated: January 27, 2016) arxiv:1601.06957v1
Relaxation of nonspherical sessile drops towards equilibrium Vadim S. Nikolayev and Daniel A. Beysens ESEME, Service des Basses Températures, CEA-Grenoble (France) (Dated: January 27, 2016) arxiv:1601.06957v1
Colloidal Particles at Liquid Interfaces: An Introduction
 1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
1 Colloidal Particles at Liquid Interfaces: An Introduction Bernard P. Binks and Tommy S. Horozov Surfactant and Colloid Group, Department of Chemistry, University of Hull, Hull, HU6 7RX, UK 1.1 Some Basic
Changes of polymer material wettability by surface discharge
 Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
Changes of polymer material wettability by surface discharge Surface discharge and material treatment Surface treatment of materials in low temperature plasma belongs to the modern and very perspective
An Analytical Approach for Determination of Riverbank Erosion under Action of Capillary Cohesion, Viscous Force and Force due to Pore Pressure
 An Analytical Approach for Determination of Riverbank Erosion under Action of Capillary Cohesion, Viscous Force and Force due to Pore Pressure Sanchayan Mukherjee 1, Bimalendu Pal 2, Debasish Mandi 2,
An Analytical Approach for Determination of Riverbank Erosion under Action of Capillary Cohesion, Viscous Force and Force due to Pore Pressure Sanchayan Mukherjee 1, Bimalendu Pal 2, Debasish Mandi 2,
Fluid Mechanics Introduction
 Fluid Mechanics Introduction Fluid mechanics study the fluid under all conditions of rest and motion. Its approach is analytical, mathematical, and empirical (experimental and observation). Fluid can be
Fluid Mechanics Introduction Fluid mechanics study the fluid under all conditions of rest and motion. Its approach is analytical, mathematical, and empirical (experimental and observation). Fluid can be
Revision Guide for Chapter 14
 Revision Guide for Chapter 14 Contents Revision Checklist Revision Notes Values of the energy kt...4 The Boltzmann factor...4 Thermal activation processes...5 Summary Diagrams Climbing a ladder by chance...7
Revision Guide for Chapter 14 Contents Revision Checklist Revision Notes Values of the energy kt...4 The Boltzmann factor...4 Thermal activation processes...5 Summary Diagrams Climbing a ladder by chance...7
Modeling of Inflation of Liquid Spherical Layers. under Zero Gravity
 Applied Mathematical Sciences Vol. 7 013 no. 138 6867-6876 HIKAI Ltd www.m-hikari.com http://dx.doi.org/10.1988/ams.013.310570 Modeling of Inflation of Liquid Spherical Layers under Zero Gravity G. I.
Applied Mathematical Sciences Vol. 7 013 no. 138 6867-6876 HIKAI Ltd www.m-hikari.com http://dx.doi.org/10.1988/ams.013.310570 Modeling of Inflation of Liquid Spherical Layers under Zero Gravity G. I.
Contact angle hysteresis: a review of fundamentals and applications
 Contact angle hysteresis: a review of fundamentals and applications The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published
Contact angle hysteresis: a review of fundamentals and applications The MIT Faculty has made this article openly available. Please share how this access benefits you. Your story matters. Citation As Published
LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE
 Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
Proceedings of the ASME/JSME 2011 8th Thermal Engineering Joint Conference AJTEC2011 March 13-17, 2011, Honolulu, Hawaii, USA AJTEC2011-44190 LIQUID FILM THICKNESS OF OSCILLATING FLOW IN A MICRO TUBE Youngbae
Interfacial dynamics
 Interfacial dynamics Interfacial dynamics = dynamic processes at fluid interfaces upon their deformation Interfacial rheological properties: elasticity, viscosity, yield stress, Relation between macroscopic
Interfacial dynamics Interfacial dynamics = dynamic processes at fluid interfaces upon their deformation Interfacial rheological properties: elasticity, viscosity, yield stress, Relation between macroscopic
Micromechanics of Colloidal Suspensions: Dynamics of shear-induced aggregation
 : Dynamics of shear-induced aggregation G. Frungieri, J. Debona, M. Vanni Politecnico di Torino Dept. of Applied Science and Technology Lagrangian transport: from complex flows to complex fluids Lecce,
: Dynamics of shear-induced aggregation G. Frungieri, J. Debona, M. Vanni Politecnico di Torino Dept. of Applied Science and Technology Lagrangian transport: from complex flows to complex fluids Lecce,
Particle Characterization Laboratories, Inc.
 Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
Analytical services Particle size analysis Dynamic Light Scattering Static Light Scattering Sedimentation Diffraction Zeta Potential Analysis Single Point Titration Isoelectric point determination Aqueous
Effect of flow velocity on the process of air-steam condensation in a vertical tube condenser
 Effect of flow velocity on the process of air-steam condensation in a vertical tube condenser Jan Havlík 1,*, Tomáš Dlouhý 1 1 Czech Technical University in Prague, Faculty of Mechanical Engineering, Department
Effect of flow velocity on the process of air-steam condensation in a vertical tube condenser Jan Havlík 1,*, Tomáš Dlouhý 1 1 Czech Technical University in Prague, Faculty of Mechanical Engineering, Department
Influence of Eccentricity and Angular Velocity on Force Effects on Rotor of Magnetorheological Damper
 EPJ Web of Conferences 18, 291 (218) EFM 217 https://doi.org/1.151/epjconf/21818291 Influence of Eccentricity and Angular Velocity on Force Effects on Rotor of Magnetorheological Damper Dominik Šedivý
EPJ Web of Conferences 18, 291 (218) EFM 217 https://doi.org/1.151/epjconf/21818291 Influence of Eccentricity and Angular Velocity on Force Effects on Rotor of Magnetorheological Damper Dominik Šedivý
Temperature ( o C)
 Viscosity (Pa sec) Supplementary Information 10 8 10 6 10 4 10 2 150 200 250 300 Temperature ( o C) Supplementary Figure 1 Viscosity of fibre components (PC cladding blue; As 2 Se 5 red; CPE black) as
Viscosity (Pa sec) Supplementary Information 10 8 10 6 10 4 10 2 150 200 250 300 Temperature ( o C) Supplementary Figure 1 Viscosity of fibre components (PC cladding blue; As 2 Se 5 red; CPE black) as
Potential Energy & Conservation of Energy
 PHYS 101 Previous Exam Problems CHAPTER 8 Potential Energy & Conservation of Energy Potential energy Conservation of energy conservative forces Conservation of energy friction Conservation of energy external
PHYS 101 Previous Exam Problems CHAPTER 8 Potential Energy & Conservation of Energy Potential energy Conservation of energy conservative forces Conservation of energy friction Conservation of energy external
Coherent photo-production of ρ 0 mesons in ultra-peripheral Pb-Pb collisions at the LHC measured by ALICE
 EPJ Web of Conferences 8, 4) DOI:.5/ epjconf/ 4 8 C Owned by the authors, published by EDP Sciences, 4 Coherent photo-production of ρ mesons in ultra-peripheral Pb-Pb collisions at the LHC measured by
EPJ Web of Conferences 8, 4) DOI:.5/ epjconf/ 4 8 C Owned by the authors, published by EDP Sciences, 4 Coherent photo-production of ρ mesons in ultra-peripheral Pb-Pb collisions at the LHC measured by
Analysis of moist air unwatering by adsorbent in fluidized bed for thermal power stations units conservation
 EPJ Web of Conferences 76, 01011 (2014) DOI: 10.1051/epjconf/20147601011 C Owned by the authors, published by EDP Sciences, 2014 Analysis of moist air unwatering by adsorbent in fluidized bed for thermal
EPJ Web of Conferences 76, 01011 (2014) DOI: 10.1051/epjconf/20147601011 C Owned by the authors, published by EDP Sciences, 2014 Analysis of moist air unwatering by adsorbent in fluidized bed for thermal
Validation analyses of advanced turbulence model approaches for stratified two-phase flows
 Computational Methods in Multiphase Flow VIII 361 Validation analyses of advanced turbulence model approaches for stratified two-phase flows M. Benz & T. Schulenberg Institute for Nuclear and Energy Technologies,
Computational Methods in Multiphase Flow VIII 361 Validation analyses of advanced turbulence model approaches for stratified two-phase flows M. Benz & T. Schulenberg Institute for Nuclear and Energy Technologies,
(1) +0.2 m/s (2) +0.4 m/s (3) +0.6 m/s (4) +1 m/s (5) +0.8 m/s
 77777 77777 Instructor: Biswas/Ihas/Whiting PHYSICS DEPARTMENT PHY 2053 Exam 2, 120 minutes November 13, 2009 Name (print, last first): Signature: On my honor, I have neither given nor received unauthorized
77777 77777 Instructor: Biswas/Ihas/Whiting PHYSICS DEPARTMENT PHY 2053 Exam 2, 120 minutes November 13, 2009 Name (print, last first): Signature: On my honor, I have neither given nor received unauthorized
Thermocapillary Migration of a Drop
 Thermocapillary Migration of a Drop An Exact Solution with Newtonian Interfacial Rheology and Stretching/Shrinkage of Interfacial Area Elements for Small Marangoni Numbers R. BALASUBRAMANIAM a AND R. SHANKAR
Thermocapillary Migration of a Drop An Exact Solution with Newtonian Interfacial Rheology and Stretching/Shrinkage of Interfacial Area Elements for Small Marangoni Numbers R. BALASUBRAMANIAM a AND R. SHANKAR
THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE DISPLACEMENT MODEL
 International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
International Journal of Modern Physics B, Vol. 13, No. 7 (1999) 355 359 c World Scientific Publishing Company THE MODIFIED YOUNG S EQUATION FOR THE CONTACT ANGLE OF A SMALL SESSILE DROP FROM AN INTERFACE
Exam II. Spring 2004 Serway & Jewett, Chapters Fill in the bubble for the correct answer on the answer sheet. next to the number.
 Agin/Meyer PART I: QUALITATIVE Exam II Spring 2004 Serway & Jewett, Chapters 6-10 Assigned Seat Number Fill in the bubble for the correct answer on the answer sheet. next to the number. NO PARTIAL CREDIT:
Agin/Meyer PART I: QUALITATIVE Exam II Spring 2004 Serway & Jewett, Chapters 6-10 Assigned Seat Number Fill in the bubble for the correct answer on the answer sheet. next to the number. NO PARTIAL CREDIT:
Numerical Investigation of Boundary Conditions for Moving Contact Line Problems
 University of Rhode Island DigitalCommons@URI Chemical Engineering Faculty Publications Chemical Engineering 2000 Numerical Investigation of Boundary Conditions for Moving Contact Line Problems Sandesh
University of Rhode Island DigitalCommons@URI Chemical Engineering Faculty Publications Chemical Engineering 2000 Numerical Investigation of Boundary Conditions for Moving Contact Line Problems Sandesh
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen
 Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Experimental and Theoretical Study of Motion of Drops on Horizontal Solid Surfaces with a Wettability Gradient Nadjoua Moumen Department of Chemical and Biomolecular Engineering Clarkson University Outline
Interfacial forces and friction on the nanometer scale: A tutorial
 Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Interfacial forces and friction on the nanometer scale: A tutorial M. Ruths Department of Chemistry University of Massachusetts Lowell Presented at the Nanotribology Tutorial/Panel Session, STLE/ASME International
Colloidal Suspension Rheology Chapter 1 Study Questions
 Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Colloidal Suspension Rheology Chapter 1 Study Questions 1. What forces act on a single colloidal particle suspended in a flowing fluid? Discuss the dependence of these forces on particle radius. 2. What
Early stages of dewetting of microscopically thin polymer films: A molecular dynamics study
 JOURNAL OF CHEMICAL PHYSICS VOLUME 109, NUMBER 19 15 NOVEMBER 1998 Early stages of dewetting of microscopically thin polymer films: A molecular dynamics study Hong Liu Department of Physics, Kansas State
JOURNAL OF CHEMICAL PHYSICS VOLUME 109, NUMBER 19 15 NOVEMBER 1998 Early stages of dewetting of microscopically thin polymer films: A molecular dynamics study Hong Liu Department of Physics, Kansas State
Sliding Contact Bearings
 Sliding Contact Bearings Classification of Bearings 1. According to the direction of load to be supported. The bearings under this group are classified as: (a) Radial bearings (b) Thrust bearings. In radial
Sliding Contact Bearings Classification of Bearings 1. According to the direction of load to be supported. The bearings under this group are classified as: (a) Radial bearings (b) Thrust bearings. In radial
Capillary rise between closely spaced plates : effect of Van der Waals forces
 Capillary rise between closely spaced plates : effect of Van der Waals forces B. Legait, P.G. De Gennes To cite this version: B. Legait, P.G. De Gennes. Capillary rise between closely spaced plates : effect
Capillary rise between closely spaced plates : effect of Van der Waals forces B. Legait, P.G. De Gennes To cite this version: B. Legait, P.G. De Gennes. Capillary rise between closely spaced plates : effect
Measurement and Prediction of Fluid Viscosities at High Shear Rates
 Chapter 5 Measurement and Prediction of Fluid Viscosities at High Shear Rates Jeshwanth K. Rameshwaram and Tien T. Dao Additional information is available at the end of the chapter http://dx.doi.org/10.5772/54282
Chapter 5 Measurement and Prediction of Fluid Viscosities at High Shear Rates Jeshwanth K. Rameshwaram and Tien T. Dao Additional information is available at the end of the chapter http://dx.doi.org/10.5772/54282
Stability of colloidal systems
 Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
Stability of colloidal systems Colloidal stability DLVO theory Electric double layer in colloidal systems Processes to induce charges at surfaces Key parameters for electric forces (ζ-potential, Debye
V. Electrostatics. MIT Student
 V. Electrostatics Lecture 26: Compact Part of the Double Layer MIT Student 1 Double-layer Capacitance 1.1 Stern Layer As was discussed in the previous lecture, the Gouy-Chapman model predicts unphysically
V. Electrostatics Lecture 26: Compact Part of the Double Layer MIT Student 1 Double-layer Capacitance 1.1 Stern Layer As was discussed in the previous lecture, the Gouy-Chapman model predicts unphysically
J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France Paris Cedex 05, France
 EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
EUROPHYSICS LETTERS 15 July 2001 Europhys. Lett., 55 (2), pp. 214 220 (2001) Rough wetting J. Bico, C. Tordeux and D. Quéré Laboratoire de Physique de la Matière Condensée, URA 792 du CNRS Collège de France
Multiscale simulations of sliding droplets
 Acta Mech https://doi.org/10.1007/s00707-018-2264-6 ORIGINAL PAPER J. J. Derksen A. E. Komrakova Multiscale simulations of sliding droplets Received: 15 February 2018 / Revised: 15 June 2018 The Author(s)
Acta Mech https://doi.org/10.1007/s00707-018-2264-6 ORIGINAL PAPER J. J. Derksen A. E. Komrakova Multiscale simulations of sliding droplets Received: 15 February 2018 / Revised: 15 June 2018 The Author(s)
Conduction Modeling in Mixed Alkali Borate Glasses
 International Journal of Pure & Applied Physics ISSN 0973-1776 Vol.1 No.2 (2005), pp. 191-197 Research India Publications http://www.ripub lication.com/ijpap.htm Conduction Modeling in Mixed Alkali Borate
International Journal of Pure & Applied Physics ISSN 0973-1776 Vol.1 No.2 (2005), pp. 191-197 Research India Publications http://www.ripub lication.com/ijpap.htm Conduction Modeling in Mixed Alkali Borate
