Electronic Spectra of Coordination Compounds
|
|
|
- Imogene Burns
- 5 years ago
- Views:
Transcription
1 Electronic Spectra of Coordination Compounds Microstates and free-ion terms for electron configurations Identify the lowest-energy term
2 Electronic Spectra of Coordination Compounds Identify the lowest-energy term 1. Sketch the energy levels, showing the d electrons. 2. Spin multiplicity of lowest-energy state = number of unpaired electrons Determine the maximum possible value of M L for the configuration as shown. This determines the type of free-ion term. 4. Combine results of steps 2 and 3 to get ground term. Spin multiplicity = 3+1=4 Max. of M L : =3 4 F
3 Electronic Spectra of Coordination Compounds: Selection Rules On the basis of the symmetry and spin multiplicity of ground and excited electronic states 1. Transitions between states of the same parity are forbidden.: Laporte selection rule Between d orbitals are forbidde Between d and p orbitals are allowed 2. Transitions between states of different spin multiplicities are forbidden: spin selection rule 4 A 2 and 4 T 1 : spin-allowed 4 A 2 and 2 T 2 : spin-forbidden
4 Electronic Spectra of Coordination Compounds: Selection Rules Some rules for relaxation of selection rules 1. Vibrations may temporarily change the symmetry(the center of symmetry is temporarily lost: vibronic coupling relax the first selection rule:d-d transition 2. Tetrahedral complexes often absorb more strongly than O h complexes. Metal-ligand sigma bonds can be described as involving a combination of sp 3 and sd 3 hybridization of the metal orbitals: relax the first selection rule 3. spin-orbit coupling provides a mechanism of relaxing the second selection rule
5 Electronic Spectra of Coordination Compounds: correlation diagrams To relate the electronic spectra of transition metal complexes to the ligand field splitting: correlation diagrams and Tanabe-Sugano diagrams 1. Free ions (no ligand field): d 2 ; 3 F, 3 P, 1 G, 1 D, 1 S. 2. Strong ligand field. t 2 t 2g e e 2 2g g g
6 Electronic Spectra of Coordination Compounds: correlation diagrams
7 Electronic Spectra of Coordination Compounds: correlation diagrams The free-ion terms will be split into states corresponding to the irreducible representation.
8 Electronic Spectra of Coordination Compounds: correlation diagrams
9 Electronic Spectra of Coordination Compounds: correlation diagrams Irreducible representations may be obtained for the strong-field limit configurations. Each free-ion irreducible representation is matched with a strongfield irreducible representation. The spin multiplicity of the ground state.
10 Electronic Spectra of Coordination Compounds: correlation diagrams
11 Electronic Spectra of Coordination Compounds: Tanabe-Sugano diagrams B = Racah parameter, a measure of the repulsion between terms of the same multiplicity; the energy difference between 3F and 3P is 15B. E is the energy above the ground state.
12 Electronic Spectra of Coordination Compounds: Tanabe-Sugano diagrams
13 Electronic Spectra of Coordination Compounds: Tanabe-Sugano diagrams High spin vs low spin High spin Low spin Ground state and spin multiplicity changed
14 Electronic Spectra of Coordination Compounds: Tanabe-Sugano diagrams
15 Jahn-Teller Distortions and Spectra d 1 d 9 complexes: might expect each to exhibit one absorption band: excitation from the t 2g to the e g levels. e g e g t 2g t 2g Two closely overlapping absorption bands.
16 Jahn-Teller Distortions and Spectra To lower the symmetry of the molecule and to reduce the degeneracy. Distortion from O h to D 4h : results in stabilization of the molecule. The most common distortion observed is elongation along z axis.
17 Jahn-Teller Distortions and Spectra : Symmetry labels for configurations Electron configurations have symmetry labels that match their degeneracies. T E A or B
18 Jahn-Teller Distortions and Spectra : Symmetry labels for configurations 2 D term for d 9 Lower energy Higher energy the opposite of the order of energies of the orbitals 2 E g 2 T 2g Distortions can be splitting of bands. Too weak
19 Tanabe-Sugano Diagrams: Determining o from Spectra;d 1, d 4, d 6, d 9
20 Tanabe-Sugano Diagrams: Determining o from Spectra
21 Tanabe-Sugano Diagrams: Determining o from Spectra;d 3, d 8 The lowest energy
22 Tanabe-Sugano Diagrams: Determining o from Spectra;d 2, d 7 (high spin)
23 Tetrahedral Complexes The lack of a center of symmetry: makes transitions between d orbitals more allowed; much more intense absorption bands. Hole formalism: d 1 O h configuration is analogous to the d 9 T d configuration: the hole in d 9 results in the same symmetry as the single electron in d 1. We can use the correlation diagram for d 10-n configuration in O h geometry e g t 2 hole octahedral t 2g e tetrahedral
24 Charge-Transfer Spectra Charge-transfer absorptions is much more intense than d-d transitions. Involve the transfer of electrons from molecular orbitals that are primarily ligand in character to orbitals that are primarily metal in character (or vice versa) Formal reduction of the metal: Co(III) to Co(II) LMCT
25 Charge-Transfer Spectra IrBr 6 2- (d 5 ): two band IrBr 6 3- (d 6 ): one band Why? Formal reduction of the metal: Co(III) to Co(II) LMCT
26 Charge-Transfer Spectra MLCT π-acceptor ligand (π* orbitals): CO, CN -, SCN -, bipyridine.. Oxidation of the metal d-d transitions may be completely overwhelmed and essentially impossible to observe. Formal oxidation of the metal: Fe(III) to Fe(IV) MLCT
11-1 Absorption of Light Quantum Numbers of Multielectron Atoms Electronic Spectra of Coordination Compounds
 Chapter 11 Coordination Chemistry III: Electronic Spectra 11-1 Absorption of Light 11-2 Quantum Numbers of Multielectron Atoms 11-3 Electronic Spectra of Coordination Compounds Chapter 11 Coordination
Chapter 11 Coordination Chemistry III: Electronic Spectra 11-1 Absorption of Light 11-2 Quantum Numbers of Multielectron Atoms 11-3 Electronic Spectra of Coordination Compounds Chapter 11 Coordination
Perhaps the most striking aspect of many coordination compounds of transition metals is that they have vivid colors. The UV-vis spectra of
 1 Perhaps the most striking aspect of many coordination compounds of transition metals is that they have vivid colors. The UV-vis spectra of coordination compounds of transition metals involve transitions
1 Perhaps the most striking aspect of many coordination compounds of transition metals is that they have vivid colors. The UV-vis spectra of coordination compounds of transition metals involve transitions
Electronic Selection Rules (II)
 Term Symbols Electronic Selection Rules (II) IMPORTANT now we are finally ready to clearly define our electronic states! microstates for a particular atomic configuration are grouped into what are called
Term Symbols Electronic Selection Rules (II) IMPORTANT now we are finally ready to clearly define our electronic states! microstates for a particular atomic configuration are grouped into what are called
How to identify types of transition in experimental spectra
 17 18 19 How to identify types of transition in experimental spectra 1. intensity 2. Band width 3. polarization Intensities are governed by how well the selection rules can be applied to the molecule under
17 18 19 How to identify types of transition in experimental spectra 1. intensity 2. Band width 3. polarization Intensities are governed by how well the selection rules can be applied to the molecule under
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
CHEMISTRY. Electronic Spectra and Magnetic Properties of Transition Metal Complexes)
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 7: Inorganic Chemistry-II (Metal-Ligand Bonding, Electronic Spectra and Magnetic Properties of Transition Metal Complexes) 16.
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 7: Inorganic Chemistry-II (Metal-Ligand Bonding, Electronic Spectra and Magnetic Properties of Transition Metal Complexes) 16.
Transition Metal Complexes Electronic Spectra 2
 Transition Metal Complexes Electronic Spectra 2 Electronic Spectra of Transition Metal Complexes Cr[(NH 3 ) 6 ] 3+ d 3 complex Molecular Term Symbols Quartet states Doublet state Different Ways of Transitions
Transition Metal Complexes Electronic Spectra 2 Electronic Spectra of Transition Metal Complexes Cr[(NH 3 ) 6 ] 3+ d 3 complex Molecular Term Symbols Quartet states Doublet state Different Ways of Transitions
Bonding in Octahedral and Tetrahedral Metal Complexes. Predict how the d orbitals are affected by the Metal- Ligand Bonding
 Bonding in Octahedral and Tetrahedral Metal Complexes 327 Molecular Orbital Theory and Crystal Field/Ligand Field Theory Predict how the d orbitals are affected by the Metal- Ligand Bonding d z 2, d x
Bonding in Octahedral and Tetrahedral Metal Complexes 327 Molecular Orbital Theory and Crystal Field/Ligand Field Theory Predict how the d orbitals are affected by the Metal- Ligand Bonding d z 2, d x
indicating the configuration they correspond to and predict their relative energy.
 Problem 1 (1 point) Three center four electron (3c/4e) bonds were introduced in class. John F. Berry (Dalton Trans. 2012, 41, 700-713) discusses the effect of the larger density of states for the 3c/4e
Problem 1 (1 point) Three center four electron (3c/4e) bonds were introduced in class. John F. Berry (Dalton Trans. 2012, 41, 700-713) discusses the effect of the larger density of states for the 3c/4e
Advanced Inorganic Chemistry
 Advanced Inorganic Chemistry Orgel Diagrams Correlation of spectroscopic terms for d n configuration in O h complexes Atomic Term Splitting of the weak field d n ground state terms in an octahedral ligand
Advanced Inorganic Chemistry Orgel Diagrams Correlation of spectroscopic terms for d n configuration in O h complexes Atomic Term Splitting of the weak field d n ground state terms in an octahedral ligand
6.2. Introduction to Spectroscopic states and term symbols
 Chemistry 3820 Lecture Notes Dr. M. Gerken Page62 6.2. Introduction to Spectroscopic states and term symbols From the number of absorption bands we have already seen that usually more d-d transitions are
Chemistry 3820 Lecture Notes Dr. M. Gerken Page62 6.2. Introduction to Spectroscopic states and term symbols From the number of absorption bands we have already seen that usually more d-d transitions are
Absorption Spectra. ! Ti(H 2 O) 6 3+ appears purple (red + blue) because it absorbs green light at ~500 nm = ~20,000 cm 1.
 Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
Absorption Spectra! Colors of transition metal complexes result from absorption of a small portion of the visible spectrum with transmission of the unabsorbed frequencies. Visible Spectra of [M(H 2 O)
Chapter 21 d-block metal chemistry: coordination complexes
 Chapter 21 d-block metal chemistry: coordination complexes Bonding: valence bond, crystal field theory, MO Spectrochemical series Crystal field stabilization energy (CFSE) Electronic Spectra Magnetic Properties
Chapter 21 d-block metal chemistry: coordination complexes Bonding: valence bond, crystal field theory, MO Spectrochemical series Crystal field stabilization energy (CFSE) Electronic Spectra Magnetic Properties
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
b) For this ground state, obtain all possible J values and order them from lowest to highest in energy.
 Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Problem 1 (2 points) Part A Consider a free ion with a d 3 electronic configuration. a) By inspection, obtain the term symbol ( 2S+1 L) for the ground state. 4 F b) For this ground state, obtain all possible
Chapter 21. d-block metal chemistry: coordination complexes
 Inorganic Chemistry B Chapter 21 d-block metal chemistry: coordination complexes Dr. Said El-Kurdi 1 21.1 Introduction In this chapter, we discuss complexes of the d-block metals and we consider bonding
Inorganic Chemistry B Chapter 21 d-block metal chemistry: coordination complexes Dr. Said El-Kurdi 1 21.1 Introduction In this chapter, we discuss complexes of the d-block metals and we consider bonding
Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy
 Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields 2. Term symbols and the method of microstates
Inorganic Chemistry with Doc M. Day 19. Transition Metals Complexes IV: Spectroscopy Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields 2. Term symbols and the method of microstates
Colors of Co(III) solutions. Electronic-Vibrational Coupling. Vibronic Coupling
 Colors of Co(III) solutions Electronic-Vibrational Coupling Vibronic Coupling Because they have g g character, the d-d transitions of complees of the transition metals are forbidden (LaPorte forbidden).
Colors of Co(III) solutions Electronic-Vibrational Coupling Vibronic Coupling Because they have g g character, the d-d transitions of complees of the transition metals are forbidden (LaPorte forbidden).
Chapter 20 d-block metal chemistry: coordination complexes
 Chapter 20 d-block metal chemistry: coordination complexes Bonding: valence bond, crystal field theory, MO Spectrochemical series Crystal field stabilization energy (CFSE) Electronic Spectra Magnetic Properties
Chapter 20 d-block metal chemistry: coordination complexes Bonding: valence bond, crystal field theory, MO Spectrochemical series Crystal field stabilization energy (CFSE) Electronic Spectra Magnetic Properties
d 2 Correlation Diagram
 d 2 Correlation Diagram 1 S 1A 1g 1A 1g 1E g e g 2 3A 2g 1E g 1 G 1T 1g 1T 2g 1A 1g 1T 1g 3 P 3T 1g 1T 2g 3T 1g t 2g 1e g 1 3T 2g 1 D 1E g 1T 2g 3A 2g 3 F 3T 2g 3T 1g 1A 1g 1E g 1T 2g t 2g 2 3T 1g Free
d 2 Correlation Diagram 1 S 1A 1g 1A 1g 1E g e g 2 3A 2g 1E g 1 G 1T 1g 1T 2g 1A 1g 1T 1g 3 P 3T 1g 1T 2g 3T 1g t 2g 1e g 1 3T 2g 1 D 1E g 1T 2g 3A 2g 3 F 3T 2g 3T 1g 1A 1g 1E g 1T 2g t 2g 2 3T 1g Free
light is absorbed, the complex appears green; If
 Color of Transition Metal Complexes The variety of color among transition metal complexes has long fascinated the chemists. For example, aqueous solutions of [Fe(H 2 O) 6 ] 3+ are red, [Co(H 2 O) 6 ] 2+
Color of Transition Metal Complexes The variety of color among transition metal complexes has long fascinated the chemists. For example, aqueous solutions of [Fe(H 2 O) 6 ] 3+ are red, [Co(H 2 O) 6 ] 2+
Electronic structure Crystal-field theory Ligand-field theory. Electronic-spectra electronic spectra of atoms
 Chapter 19 d-metal complexes: electronic structure and spectra Electronic structure 19.1 Crystal-field theory 19.2 Ligand-field theory Electronic-spectra 19.3 electronic spectra of atoms 19.4 electronic
Chapter 19 d-metal complexes: electronic structure and spectra Electronic structure 19.1 Crystal-field theory 19.2 Ligand-field theory Electronic-spectra 19.3 electronic spectra of atoms 19.4 electronic
In the fourth problem set, you derived the MO diagrams for two complexes containing Cr-Cr bonds:
 Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Problem 1 (2 points) Part 1 a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions
Template for the d 2 Correlation Diagram
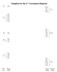 S A g Template for the d Correlation Diagram (,3)A g e g (,3)A g G T g T g A g T g 3 P 3T g T g 3T g t g e g 3T g D T g 3A g 3F 3T g 3T g A g (,3)T g t g (,3)T g Free Ion Weak Strong Extreme Making the
S A g Template for the d Correlation Diagram (,3)A g e g (,3)A g G T g T g A g T g 3 P 3T g T g 3T g t g e g 3T g D T g 3A g 3F 3T g 3T g A g (,3)T g t g (,3)T g Free Ion Weak Strong Extreme Making the
Chm 363. Spring 2017, Exercise Set 3 Transition Metal Bonding and Spectra. Mr. Linck. Version 1.5 March 9, 2017
 Chm 363 Spring 2017, Exercise Set 3 Transition Metal Bonding and Spectra Mr. Linck Version 1.5 March 9, 2017 3.1 Transition Metal Bonding in Octahedral Compounds How do the metal 3d, 4s, and 4p orbitals
Chm 363 Spring 2017, Exercise Set 3 Transition Metal Bonding and Spectra Mr. Linck Version 1.5 March 9, 2017 3.1 Transition Metal Bonding in Octahedral Compounds How do the metal 3d, 4s, and 4p orbitals
2018 Ch112 problem set 6 Due: Thursday, Dec. 6th. Problem 1 (2 points)
 Problem 1 (2 points) a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions (υ1 and
Problem 1 (2 points) a. Consider the following V III complexes: V(H2O)6 3+, VF6 3-, and VCl6 3-. The table below contains the energies corresponding to the two lowest spin-allowed d-d transitions (υ1 and
Coordination Chemistry: Bonding Theories. Crystal Field Theory. Chapter 20
 Coordination Chemistry: Bonding Theories Crystal Field Theory Chapter 0 Review of the Previous Lecture 1. We discussed different types of isomerism in coordination chemistry Structural or constitutional
Coordination Chemistry: Bonding Theories Crystal Field Theory Chapter 0 Review of the Previous Lecture 1. We discussed different types of isomerism in coordination chemistry Structural or constitutional
Inorganic Chemistry with Doc M. Fall Semester, 2011 Day 19. Transition Metals Complexes IV: Spectroscopy
 Inorganic Chemistry with Doc M. Fall Semester, 011 Day 19. Transition Metals Complexes IV: Spectroscopy Name(s): lement: Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields. Term symbols
Inorganic Chemistry with Doc M. Fall Semester, 011 Day 19. Transition Metals Complexes IV: Spectroscopy Name(s): lement: Topics: 1. The visible spectrum and the d-orbitals 3. Octahedral fields. Term symbols
Electronic Spectroscopy of Transition Metal Ions (continued)
 Electronic Spectroscopy of Transition Metal Ions (continued) What about the spectroscopy! First some selection rules are found to apply: 1) Spin selection rule: S = 0 theory: transitions can only occur
Electronic Spectroscopy of Transition Metal Ions (continued) What about the spectroscopy! First some selection rules are found to apply: 1) Spin selection rule: S = 0 theory: transitions can only occur
Orbitals and energetics
 Orbitals and energetics Bonding and structure Molecular orbital theory Crystal field theory Ligand field theory Provide fundamental understanding of chemistry dictating radionuclide complexes Structure
Orbitals and energetics Bonding and structure Molecular orbital theory Crystal field theory Ligand field theory Provide fundamental understanding of chemistry dictating radionuclide complexes Structure
RDCH 702 Lecture 4: Orbitals and energetics
 RDCH 702 Lecture 4: Orbitals and energetics Molecular symmetry Bonding and structure Molecular orbital theory Crystal field theory Ligand field theory Provide fundamental understanding of chemistry dictating
RDCH 702 Lecture 4: Orbitals and energetics Molecular symmetry Bonding and structure Molecular orbital theory Crystal field theory Ligand field theory Provide fundamental understanding of chemistry dictating
Free-Ion Terms to Ligand-field Terms
 Free-Ion Terms to Ligand-field Terms! Orbital term symbols for free atoms and ions are identical to symbols for irreducible representations in R 3. " The irreducible representations of R 3 include all
Free-Ion Terms to Ligand-field Terms! Orbital term symbols for free atoms and ions are identical to symbols for irreducible representations in R 3. " The irreducible representations of R 3 include all
CHAPTER TEN MOLECULAR GEOMETRY MOLECULAR GEOMETRY V S E P R CHEMICAL BONDING II: MOLECULAR GEOMETRY AND HYBRIDIZATION OF ATOMIC ORBITALS
 CHAPTER TEN CHEMICAL BONDING II: AND HYBRIDIZATION O ATOMIC ORBITALS V S E P R VSEPR Theory In VSEPR theory, multiple bonds behave like a single electron pair Valence shell electron pair repulsion (VSEPR)
CHAPTER TEN CHEMICAL BONDING II: AND HYBRIDIZATION O ATOMIC ORBITALS V S E P R VSEPR Theory In VSEPR theory, multiple bonds behave like a single electron pair Valence shell electron pair repulsion (VSEPR)
Excited States in Organic Light-Emitting Diodes
 Excited States in Organic Light-Emitting Diodes The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for solar harvesting and sensitization
Excited States in Organic Light-Emitting Diodes The metal-to-ligand charge transfer (MLCT) excited states of d 6 π coordination compounds have emerged as the most efficient for solar harvesting and sensitization
Chapter 20 d-metal complexes: electronic structures and properties
 CHEM 511 Chapter 20 page 1 of 21 Chapter 20 d-metal complexes: electronic structures and properties Recall the shape of the d-orbitals... Electronic structure Crystal Field Theory: an electrostatic approach
CHEM 511 Chapter 20 page 1 of 21 Chapter 20 d-metal complexes: electronic structures and properties Recall the shape of the d-orbitals... Electronic structure Crystal Field Theory: an electrostatic approach
Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory
 Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory Electronic Structure of Six and Four-Coordinate Complexes Using Crystal Field Theory, we can generate energy level
Chemistry 3211 Coordination Chemistry Part 3 Ligand Field and Molecular Orbital Theory Electronic Structure of Six and Four-Coordinate Complexes Using Crystal Field Theory, we can generate energy level
Chemistry Instrumental Analysis Lecture 11. Chem 4631
 Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
Chemistry 4631 Instrumental Analysis Lecture 11 Molar Absorptivities Range 0 to 10 5 Magnitude of e depends on capture cross section of the species and probability of the energy-absorbing transition. e
If you put an electron into the t 2g, like that for Ti 3+, then you stabilize the barycenter of the d orbitals by 0.4 D o.
 Crystal Field Stabilization Energy Week 2-1 Octahedral Symmetry (O h ) If you put an electron into the t 2g, like that for Ti 3+, then you stabilize the barycenter of the d orbitals by 0.4 D o. Each additional
Crystal Field Stabilization Energy Week 2-1 Octahedral Symmetry (O h ) If you put an electron into the t 2g, like that for Ti 3+, then you stabilize the barycenter of the d orbitals by 0.4 D o. Each additional
Chem 673, Problem Set 5 Due Thursday, December 1, 2005
 otton, Problem 9.3 (assume D 4h symmetry) Additional Problems: hem 673, Problem Set 5 Due Thursday, December 1, 2005 (1) Infrared and Raman spectra of Benzene (a) Determine the symmetries (irreducible
otton, Problem 9.3 (assume D 4h symmetry) Additional Problems: hem 673, Problem Set 5 Due Thursday, December 1, 2005 (1) Infrared and Raman spectra of Benzene (a) Determine the symmetries (irreducible
Electronic Spectroscopy Application of Group Theory
 Electronic Spectroscopy Application of Group Theory Ψ Tot assumed to be separable Ψ tttttt = ψψ eeeeeeee χχ vvvvvv = n v If a transition is not allowed by symmetry then vibronic coupling can be invoked
Electronic Spectroscopy Application of Group Theory Ψ Tot assumed to be separable Ψ tttttt = ψψ eeeeeeee χχ vvvvvv = n v If a transition is not allowed by symmetry then vibronic coupling can be invoked
Advanced Analytical Chemistry
 84.514 Advanced Analytical Chemistry Part III Molecular Spectroscopy (continued) Website http://faculty.uml.edu/david_ryan/84.514 http://www.cem.msu.edu/~reusch/virtualtext/ Spectrpy/UV-Vis/spectrum.htm
84.514 Advanced Analytical Chemistry Part III Molecular Spectroscopy (continued) Website http://faculty.uml.edu/david_ryan/84.514 http://www.cem.msu.edu/~reusch/virtualtext/ Spectrpy/UV-Vis/spectrum.htm
Ultraviolet-Visible Spectroscopy
 Ultraviolet-Visible Spectroscopy Introduction to UV-Visible Absorption spectroscopy from 160 nm to 780 nm Measurement of transmittance Conversion to absorbance * A=-logT=εbc Measurement of transmittance
Ultraviolet-Visible Spectroscopy Introduction to UV-Visible Absorption spectroscopy from 160 nm to 780 nm Measurement of transmittance Conversion to absorbance * A=-logT=εbc Measurement of transmittance
A Rigorous Introduction to Molecular Orbital Theory and its Applications in Chemistry. Zachary Chin, Alex Li, Alex Liu
 A Rigorous Introduction to Molecular Orbital Theory and its Applications in Chemistry Zachary Chin, Alex Li, Alex Liu Quantum Mechanics Atomic Orbitals and Early Bonding Theory Quantum Numbers n: principal
A Rigorous Introduction to Molecular Orbital Theory and its Applications in Chemistry Zachary Chin, Alex Li, Alex Liu Quantum Mechanics Atomic Orbitals and Early Bonding Theory Quantum Numbers n: principal
Chem 673, Problem Sets 4 & 5 Due Tuesday, December 3, Problems from Carter: Chapter 6: 6.1, 6.3, 6.7, 6.9 Chapter 7: 7.2a,b,e,g,i,j, 7.
 Chem 673, Problem Sets 4 & 5 Due Tuesday, December 3, 2013 Problems from Carter: Chapter 6: 6.1, 6.3, 6.7, 6.9 Chapter 7: 7.2a,b,e,g,i,j, 7.6, (1) Use the angular overlap table given on the back page of
Chem 673, Problem Sets 4 & 5 Due Tuesday, December 3, 2013 Problems from Carter: Chapter 6: 6.1, 6.3, 6.7, 6.9 Chapter 7: 7.2a,b,e,g,i,j, 7.6, (1) Use the angular overlap table given on the back page of
Valence Bond Theory - Description
 Bonding and Molecular Structure - PART 2 - Valence Bond Theory and Hybridization 1. Understand and be able to describe the Valence Bond Theory description of covalent bond formation. 2. Understand and
Bonding and Molecular Structure - PART 2 - Valence Bond Theory and Hybridization 1. Understand and be able to describe the Valence Bond Theory description of covalent bond formation. 2. Understand and
- an approach to bonding that is useful for making estimates of E of orbitals in coordination complexes
 10.4 Angular Overlap - an approach to bonding that is useful for making estimates of E of orbitals in coordination complexes - estimate the strength of interaction b/w ligand orbitals & metal d orbitals
10.4 Angular Overlap - an approach to bonding that is useful for making estimates of E of orbitals in coordination complexes - estimate the strength of interaction b/w ligand orbitals & metal d orbitals
Coordination and Special Materials Chemistry. Elective I/II: WS 2005/6 (Lecture) H.J. Deiseroth. Part 2
 Coordination and Special Materials Chemistry Elective I/II: WS 2005/6 (Lecture) H.J. Deiseroth Part 2 Coordination Chemistry: Spectroscopy -microstates and spectroscopic symbols (RS and jj coupling), see
Coordination and Special Materials Chemistry Elective I/II: WS 2005/6 (Lecture) H.J. Deiseroth Part 2 Coordination Chemistry: Spectroscopy -microstates and spectroscopic symbols (RS and jj coupling), see
ECEN 5005 Crystals, Nanocrystals and Device Applications Class 20 Group Theory For Crystals
 ECEN 5005 Crystals, Nanocrystals and Device Applications Class 20 Group Theory For Crystals Laporte Selection Rule Polarization Dependence Spin Selection Rule 1 Laporte Selection Rule We first apply this
ECEN 5005 Crystals, Nanocrystals and Device Applications Class 20 Group Theory For Crystals Laporte Selection Rule Polarization Dependence Spin Selection Rule 1 Laporte Selection Rule We first apply this
Manganese Chemistry. Mn : 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5
 Group 7 : Manganese Chemistry Mn : 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5 Crystal Field Splitting in Octahedral Transition Metal Complexes d Subshell Splitting in an O h Field In the octahedral (O h ) environment
Group 7 : Manganese Chemistry Mn : 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 5 Crystal Field Splitting in Octahedral Transition Metal Complexes d Subshell Splitting in an O h Field In the octahedral (O h ) environment
Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR. St. Olaf College Northfield, Minnesota
 Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR St. Olaf College Northfield, Minnesota Contents PREFACE xiii 1 INTRODUCTION TO INORGANIC CHEMISTRY 1 1-1 What Is Inorganic Chemistry? 1 1-2 Contrasts
Inorganic Chemistry GARY L. MIESSLER DONALD A. TARR St. Olaf College Northfield, Minnesota Contents PREFACE xiii 1 INTRODUCTION TO INORGANIC CHEMISTRY 1 1-1 What Is Inorganic Chemistry? 1 1-2 Contrasts
Other Crystal Fields
 Other Crystal Fields! We can deduce the CFT splitting of d orbitals in virtually any ligand field by " Noting the direct product listings in the appropriate character table to determine the ways in which
Other Crystal Fields! We can deduce the CFT splitting of d orbitals in virtually any ligand field by " Noting the direct product listings in the appropriate character table to determine the ways in which
Chem 673, Problem Set 5 Due Thursday, November 29, 2007
 Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
Chem 673, Problem Set 5 Due Thursday, November 29, 2007 (1) Trigonal prismatic coordination is fairly common in solid-state inorganic chemistry. In most cases the geometry of the trigonal prism is such
Chapter 25 Transition Metals and Coordination Compounds Part 2
 Chapter 25 Transition Metals and Coordination Compounds Part 2 Bonding in Coordination Compounds Valence Bond Theory Coordinate covalent bond is between: completely filled atomic orbital and an empty atomic
Chapter 25 Transition Metals and Coordination Compounds Part 2 Bonding in Coordination Compounds Valence Bond Theory Coordinate covalent bond is between: completely filled atomic orbital and an empty atomic
Lecture 6: Physical Methods II. UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy
 Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 21. Transition Metals Complexes V: Reaction Mechanisms
 Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 21. Transition Metals Complexes V: Reaction Mechanisms Name(s): Element: Topics: 1. Substitution reactions: dissociative v. associative 4. Pseudorotation
Inorganic Chemistry with Doc M. Fall Semester, 2012 Day 21. Transition Metals Complexes V: Reaction Mechanisms Name(s): Element: Topics: 1. Substitution reactions: dissociative v. associative 4. Pseudorotation
Organic Chemistry. Review Information for Unit 1. VSEPR Hybrid Orbitals Polar Molecules
 rganic hemistry Review Information for Unit 1 VSEPR ybrid rbitals Polar Molecules VSEPR The valence shell electron pair repulsion model (VSEPR) can be used to predict the geometry around a particular atom
rganic hemistry Review Information for Unit 1 VSEPR ybrid rbitals Polar Molecules VSEPR The valence shell electron pair repulsion model (VSEPR) can be used to predict the geometry around a particular atom
Chemistry 543--Final Exam--Keiderling May 5, pm SES
 Chemistry 543--Final Exam--Keiderling May 5,1992 -- 1-5pm -- 174 SES Please answer all questions in the answer book provided. Make sure your name is clearly indicated and that the answers are clearly numbered,
Chemistry 543--Final Exam--Keiderling May 5,1992 -- 1-5pm -- 174 SES Please answer all questions in the answer book provided. Make sure your name is clearly indicated and that the answers are clearly numbered,
Chapter Molecules are 3D. Shapes and Bonds. Chapter 9 1. Chemical Bonding and Molecular Structure
 Chapter 9 Chemical Bonding and Molecular Structure 1 Shape 9.1 Molecules are 3D Angle Linear 180 Planar triangular (trigonal planar) 120 Tetrahedral 109.5 2 Shapes and Bonds Imagine a molecule where the
Chapter 9 Chemical Bonding and Molecular Structure 1 Shape 9.1 Molecules are 3D Angle Linear 180 Planar triangular (trigonal planar) 120 Tetrahedral 109.5 2 Shapes and Bonds Imagine a molecule where the
FACULTY OF SCIENCE AND FACULTY OF ETERNAL STUDIES BACHELOR OF EDUCATION (BED SCI) SCH 304: INORGANIC CHEMISTRY 4 CO-ORDINATION CHEMISTRY.
 FACULTY OF SCIENCE AND FACULTY OF ETERNAL STUDIES BACHELOR OF EDUCATION (BED SCI) SCH 304: INORGANIC CHEMISTRY 4 CO-ORDINATION CHEMISTRY Written by Dr Lydia W. Njenga Department of chemistry Reviewed by
FACULTY OF SCIENCE AND FACULTY OF ETERNAL STUDIES BACHELOR OF EDUCATION (BED SCI) SCH 304: INORGANIC CHEMISTRY 4 CO-ORDINATION CHEMISTRY Written by Dr Lydia W. Njenga Department of chemistry Reviewed by
Molecular Orbital Theory (MOT)
 Molecular Orbital Theory (MOT) In this section, There are another approach to the bonding in metal complexes: the use of molecular orbital theory (MOT). In contrast to crystal field theory, the molecular
Molecular Orbital Theory (MOT) In this section, There are another approach to the bonding in metal complexes: the use of molecular orbital theory (MOT). In contrast to crystal field theory, the molecular
Coordination Chemistry II: Bonding
 d x2-y2 b 1g e g d x2-y2 b 1g D 1 t 2 d xy, d yz, d zx D t d d z2, d x2-y2 D o d z2 a 1g d xy D 2 d z2 b 2g a 1g e d z2, d x2-y2 d xy, d yz, d zx d xy b 2g D 3 t 2g e g d yz, d zx e g d yz, d zx 10 Coordination
d x2-y2 b 1g e g d x2-y2 b 1g D 1 t 2 d xy, d yz, d zx D t d d z2, d x2-y2 D o d z2 a 1g d xy D 2 d z2 b 2g a 1g e d z2, d x2-y2 d xy, d yz, d zx d xy b 2g D 3 t 2g e g d yz, d zx e g d yz, d zx 10 Coordination
VSEPR Model. Valence-Shell Electron-Pair Repulsion Bonds (single or multiple) and lone pairs are thought of as charge clouds
 Molecular Shapes VSEPR Model Valence-Shell Electron-Pair Repulsion Bonds (single or multiple) and lone pairs are thought of as charge clouds They repel each other and stay as far away from each other as
Molecular Shapes VSEPR Model Valence-Shell Electron-Pair Repulsion Bonds (single or multiple) and lone pairs are thought of as charge clouds They repel each other and stay as far away from each other as
LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES
 SYMMETRY II. J. M. GOICOECHEA. LECTURE 3 1 LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES 3.1 Direct products and many electron states Consider the problem of deciding upon the symmetry of
SYMMETRY II. J. M. GOICOECHEA. LECTURE 3 1 LECTURE 3 DIRECT PRODUCTS AND SPECTROSCOPIC SELECTION RULES 3.1 Direct products and many electron states Consider the problem of deciding upon the symmetry of
Electronic Spectroscopy of Polyatomics
 Electronic Spectroscopy of Polyatomics We shall discuss the electronic spectroscopy of the following types of polyatomic molecules: 1. general AH 2 molecules, A = first-row element 2. formaldehyde 3. benzene
Electronic Spectroscopy of Polyatomics We shall discuss the electronic spectroscopy of the following types of polyatomic molecules: 1. general AH 2 molecules, A = first-row element 2. formaldehyde 3. benzene
Molecular Shape and Molecular Polarity. Molecular Shape and Molecular Polarity. Molecular Shape and Molecular Polarity
 Molecular Shape and Molecular Polarity When there is a difference in electronegativity between two atoms, then the bond between them is polar. It is possible for a molecule to contain polar bonds, but
Molecular Shape and Molecular Polarity When there is a difference in electronegativity between two atoms, then the bond between them is polar. It is possible for a molecule to contain polar bonds, but
Chapter 19: d-metal Complexes: Electronic Structure and Spectra 121
 Chapter 19: d-metal Complexes: Electronic Structure and Spectra 121 S19.9 Assign the bands in the spectrum of [Cr(NCS)613-? This six-coordinate d 3 complex undoubtedly has Oh symmetry, so the general features
Chapter 19: d-metal Complexes: Electronic Structure and Spectra 121 S19.9 Assign the bands in the spectrum of [Cr(NCS)613-? This six-coordinate d 3 complex undoubtedly has Oh symmetry, so the general features
B. Electron Deficient (less than an octet) H-Be-H. Be does not need an octet Total of 4 valence electrons
 B. Electron Deficient (less than an octet) e.g. BeH 2 H-Be-H Be does not need an octet Total of 4 valence electrons Not the same as unsaturated systems that achieve the 8e - (octet) through the formation
B. Electron Deficient (less than an octet) e.g. BeH 2 H-Be-H Be does not need an octet Total of 4 valence electrons Not the same as unsaturated systems that achieve the 8e - (octet) through the formation
Spectra of Atoms and Molecules. Peter F. Bernath
 Spectra of Atoms and Molecules Peter F. Bernath New York Oxford OXFORD UNIVERSITY PRESS 1995 Contents 1 Introduction 3 Waves, Particles, and Units 3 The Electromagnetic Spectrum 6 Interaction of Radiation
Spectra of Atoms and Molecules Peter F. Bernath New York Oxford OXFORD UNIVERSITY PRESS 1995 Contents 1 Introduction 3 Waves, Particles, and Units 3 The Electromagnetic Spectrum 6 Interaction of Radiation
Andrew Rosen *Note: If you can rotate a molecule to have one isomer equal to another, they are both the same
 *Note: If you can rotate a molecule to have one isomer equal to another, they are both the same *Note: For hybridization, if an SP 2 is made, there is one unhybridized p orbital (because p usually has
*Note: If you can rotate a molecule to have one isomer equal to another, they are both the same *Note: For hybridization, if an SP 2 is made, there is one unhybridized p orbital (because p usually has
ORBITAL INTERACTIONS IN CHEMISTRY
 ORBITAL INTERACTIONS IN CHEMISTRY THOMAS A. ALBRIGHT Department of Chemistry University of Houston JEREMY K. BURDETT Department of Chemistry The University of Chicago MYUNG-HWAN WHANGBO Department of Chemistry
ORBITAL INTERACTIONS IN CHEMISTRY THOMAS A. ALBRIGHT Department of Chemistry University of Houston JEREMY K. BURDETT Department of Chemistry The University of Chicago MYUNG-HWAN WHANGBO Department of Chemistry
1. For both these compounds, the formal oxidation state is Mn(II), which is d 5.
 Sample final exam answers 1. For both these compounds, the formal oxidation state is Mn(II), which is d 5. Since NCS is a weak-field, high-spin ligand (from the spectrochemical series), the electron configuration
Sample final exam answers 1. For both these compounds, the formal oxidation state is Mn(II), which is d 5. Since NCS is a weak-field, high-spin ligand (from the spectrochemical series), the electron configuration
EXAM II Material. Part I Chemical Bonding I Lewis Theory Chapter 9 pages A. Drawing electron dot structures HOW TO:
 CHEMISTRY 112 LECTURE EXAM II Material Part I Chemical Bonding I Lewis Theory Chapter 9 pages 376-386 A. Drawing electron dot structures HOW TO: 1. Write e- dot structure for the individual atoms. 2. a)
CHEMISTRY 112 LECTURE EXAM II Material Part I Chemical Bonding I Lewis Theory Chapter 9 pages 376-386 A. Drawing electron dot structures HOW TO: 1. Write e- dot structure for the individual atoms. 2. a)
Chapter 5 Equations for Wave Function
 Chapter 5 Equations for Wave Function In very simple cases, the explicit expressions for the SALCs could be deduced by inspection, but not for complicated system. It would be useful for cases like these
Chapter 5 Equations for Wave Function In very simple cases, the explicit expressions for the SALCs could be deduced by inspection, but not for complicated system. It would be useful for cases like these
Crystal Field Theory
 Crystal Field Theory It is not a bonding theory Method of explaining some physical properties that occur in transition metal complexes. Involves a simple electrostatic argument which can yield reasonable
Crystal Field Theory It is not a bonding theory Method of explaining some physical properties that occur in transition metal complexes. Involves a simple electrostatic argument which can yield reasonable
Nuclear Quadrupole Resonance Spectroscopy. Some examples of nuclear quadrupole moments
 Nuclear Quadrupole Resonance Spectroscopy Review nuclear quadrupole moments, Q A negative value for Q denotes a distribution of charge that is "football-shaped", i.e. a sphere elongated at the poles; a
Nuclear Quadrupole Resonance Spectroscopy Review nuclear quadrupole moments, Q A negative value for Q denotes a distribution of charge that is "football-shaped", i.e. a sphere elongated at the poles; a
Chemical bonding in complexes of transition metals
 Chemical bonding in complexes of transition metals Chem 202, Sept. 28, 2010 What are transition elements? Electronic structure of atoms Naming delocalized molecular orbitals: tetrahedral and octahedral
Chemical bonding in complexes of transition metals Chem 202, Sept. 28, 2010 What are transition elements? Electronic structure of atoms Naming delocalized molecular orbitals: tetrahedral and octahedral
Dr. Fred O. Garces Chemistry 201
 23.4 400! 500! 600! 800! The relationship between Colors, Metal Complexes and Gemstones Dr. Fred O. Garces Chemistry 201 Miramar College 1 Transition Metal Gems Gemstone owe their color from trace transition-metal
23.4 400! 500! 600! 800! The relationship between Colors, Metal Complexes and Gemstones Dr. Fred O. Garces Chemistry 201 Miramar College 1 Transition Metal Gems Gemstone owe their color from trace transition-metal
Crystal Field Theory History
 Crystal Field Theory History 1929 Hans Bethe - Crystal Field Theory (CFT) Developed to interpret color, spectra, magnetism in crystals 1932 J. H. Van Vleck - CFT of Transition Metal Complexes Champions
Crystal Field Theory History 1929 Hans Bethe - Crystal Field Theory (CFT) Developed to interpret color, spectra, magnetism in crystals 1932 J. H. Van Vleck - CFT of Transition Metal Complexes Champions
Group Theory and Its Applications in Physics
 T. Inui Y Tanabe Y. Onodera Group Theory and Its Applications in Physics With 72 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Contents Sections marked with
T. Inui Y Tanabe Y. Onodera Group Theory and Its Applications in Physics With 72 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Contents Sections marked with
1 Inorganic and Bioinorganic COPYRIGHTED MATERIAL. Spectroscopy. EDWARD I. SOLOMON and CALEB B. BELL III 1.1 INTRODUCTION
 1 Inorganic and Bioinorganic Spectroscopy EDWARD I. SOLOMON and CALEB B. BELL III 1.1 INTRODUCTION Spectroscopic methods have played a critical and symbiotic role in the development of our understanding
1 Inorganic and Bioinorganic Spectroscopy EDWARD I. SOLOMON and CALEB B. BELL III 1.1 INTRODUCTION Spectroscopic methods have played a critical and symbiotic role in the development of our understanding
CH103 General Chemistry II 2018 Fall semester Quiz 4
 CH103 General Chemistry II 2018 Fall semester Quiz 4 Date: Dec. 3 rd (Mon) Time: 19:00~19:45 Professor Name Class Student I.D. Number Name 1. Circle on the correct answer in underlined parentheses. (1
CH103 General Chemistry II 2018 Fall semester Quiz 4 Date: Dec. 3 rd (Mon) Time: 19:00~19:45 Professor Name Class Student I.D. Number Name 1. Circle on the correct answer in underlined parentheses. (1
Chapter 10 Chemical Bonding II
 Chapter 10 Chemical Bonding II Valence Bond Theory Valence Bond Theory: A quantum mechanical model which shows how electron pairs are shared in a covalent bond. Bond forms between two atoms when the following
Chapter 10 Chemical Bonding II Valence Bond Theory Valence Bond Theory: A quantum mechanical model which shows how electron pairs are shared in a covalent bond. Bond forms between two atoms when the following
7.2 Dipolar Interactions and Single Ion Anisotropy in Metal Ions
 7.2 Dipolar Interactions and Single Ion Anisotropy in Metal Ions Up to this point, we have been making two assumptions about the spin carriers in our molecules: 1. There is no coupling between the 2S+1
7.2 Dipolar Interactions and Single Ion Anisotropy in Metal Ions Up to this point, we have been making two assumptions about the spin carriers in our molecules: 1. There is no coupling between the 2S+1
Molecular orbitals for σbonding in T d complexes
 Molecular orbitals for σbonding in T d complexes The set of n A B σ bonds in AB n (T d n = 4) molecules are often thought of as independent entities. The concept of MO s allows us to begin with a very
Molecular orbitals for σbonding in T d complexes The set of n A B σ bonds in AB n (T d n = 4) molecules are often thought of as independent entities. The concept of MO s allows us to begin with a very
Schedule. Lecture 7: M-M bonds δ-bonds and bonding in metal clusters
 Schedule Lecture 7: M-M bonds δ-bonds and bonding in metal clusters Lecture 8: Rates of reaction Ligand-exchange reactions, labile and inert metal ions Lecture 9: Redox reactions Inner and outer-sphere
Schedule Lecture 7: M-M bonds δ-bonds and bonding in metal clusters Lecture 8: Rates of reaction Ligand-exchange reactions, labile and inert metal ions Lecture 9: Redox reactions Inner and outer-sphere
Molecular Orbitals. Chapter 9. Sigma bonding orbitals. Sigma bonding orbitals. Pi bonding orbitals. Sigma and pi bonds
 Molecular Orbitals Chapter 9 Orbitals and Covalent Bond The overlap of atomic orbitals from separate atoms makes molecular orbitals Each molecular orbital has room for two electrons Two types of MO Sigma
Molecular Orbitals Chapter 9 Orbitals and Covalent Bond The overlap of atomic orbitals from separate atoms makes molecular orbitals Each molecular orbital has room for two electrons Two types of MO Sigma
What Should a Bonding Theory Explain? What Should a Bonding Theory Explain?
 What Should a Bonding Theory Explain? In our intro have already outlined some of the properties of transition metal complexes. For a bonding theory to be effective it must address these points. You already
What Should a Bonding Theory Explain? In our intro have already outlined some of the properties of transition metal complexes. For a bonding theory to be effective it must address these points. You already
Electronic structure / bonding in d-block complexes
 LN05-1 Electronic structure / bonding in d-block complexes Many, many properties of transition metal complexes (coordination number, structure, colour, magnetism, reactivity) are very sensitive to the
LN05-1 Electronic structure / bonding in d-block complexes Many, many properties of transition metal complexes (coordination number, structure, colour, magnetism, reactivity) are very sensitive to the
Practice sheet #6: Molecular Shape, Hybridization, Molecular Orbitals.
 CH 101/103 - Practice sheet 3/17/2014 Practice sheet #6: Molecular Shape, Hybridization, Molecular Orbitals. 1. Draw the 3D structures for the following molecules. You can omit the lone pairs on peripheral
CH 101/103 - Practice sheet 3/17/2014 Practice sheet #6: Molecular Shape, Hybridization, Molecular Orbitals. 1. Draw the 3D structures for the following molecules. You can omit the lone pairs on peripheral
Find the difference in electronegativity between the hydrogen and chlorine atoms
 Answers Questions 16.2 Molecular polarity 1. Write a dot diagram for the HCl molecule. Find the difference in electronegativity between the hydrogen and chlorine atoms Difference in electronegativity =
Answers Questions 16.2 Molecular polarity 1. Write a dot diagram for the HCl molecule. Find the difference in electronegativity between the hydrogen and chlorine atoms Difference in electronegativity =
CHEMISTRY 112 LECTURE EXAM II Material
 CHEMISTRY 112 LECTURE EXAM II Material Part I Chemical Bonding I Lewis Theory Chapter 9 pages 376-386 A. Drawing electron dot structures HOW TO: 1. Write e- dot structure for the individual atoms. 2. a)
CHEMISTRY 112 LECTURE EXAM II Material Part I Chemical Bonding I Lewis Theory Chapter 9 pages 376-386 A. Drawing electron dot structures HOW TO: 1. Write e- dot structure for the individual atoms. 2. a)
Chapter 9: Molecular Geometries and Bonding Theories Learning Outcomes: Predict the three-dimensional shapes of molecules using the VSEPR model.
 Chapter 9: Molecular Geometries and Bonding Theories Learning Outcomes: Predict the three-dimensional shapes of molecules using the VSEPR model. Determine whether a molecule is polar or nonpolar based
Chapter 9: Molecular Geometries and Bonding Theories Learning Outcomes: Predict the three-dimensional shapes of molecules using the VSEPR model. Determine whether a molecule is polar or nonpolar based
wbt Λ = 0, 1, 2, 3, Eq. (7.63)
 7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
7.2.2 Classification of Electronic States For all diatomic molecules the coupling approximation which best describes electronic states is analogous to the Russell- Saunders approximation in atoms The orbital
B7 Symmetry : Questions
 B7 Symmetry 009-10: Questions 1. Using the definition of a group, prove the Rearrangement Theorem, that the set of h products RS obtained for a fixed element S, when R ranges over the h elements of the
B7 Symmetry 009-10: Questions 1. Using the definition of a group, prove the Rearrangement Theorem, that the set of h products RS obtained for a fixed element S, when R ranges over the h elements of the
Chapter 9. Molecular Geometries and Bonding Theories. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 9 Theories John D. Bookstaver St. Charles Community College Cottleville, MO Shapes The shape of a molecule plays an important role in its reactivity. By noting the number of
Lecture Presentation Chapter 9 Theories John D. Bookstaver St. Charles Community College Cottleville, MO Shapes The shape of a molecule plays an important role in its reactivity. By noting the number of
Chapter 9. Molecular Geometry and Bonding Theories
 Chapter 9. Molecular Geometry and Bonding Theories 9.1 Molecular Shapes Lewis structures give atomic connectivity: they tell us which atoms are physically connected to which atoms. The shape of a molecule
Chapter 9. Molecular Geometry and Bonding Theories 9.1 Molecular Shapes Lewis structures give atomic connectivity: they tell us which atoms are physically connected to which atoms. The shape of a molecule
Chemistry 324 Midterm 1 KEY Wednesday, October 19, 2011 Instructor: D. J. Berg
 Chem 324 Midterm 1 Fall 2011 Version 1 Page 1 of 9 Chemistry 324 Midterm 1 KEY Wednesday, October 19, 2011 Instructor: D. J. Berg Name: Answer all questions on the paper (use the back if necessary). There
Chem 324 Midterm 1 Fall 2011 Version 1 Page 1 of 9 Chemistry 324 Midterm 1 KEY Wednesday, October 19, 2011 Instructor: D. J. Berg Name: Answer all questions on the paper (use the back if necessary). There
Valence Bond Theory. Localized Electron Model. Hybridize the Orbitals! Overlap and Bonding. Atomic Orbitals are. mmmkay. Overlap and Bonding
 Valence Bond Theory Atomic Orbitals are bad mmmkay Overlap and Bonding Lewis taught us to think of covalent bonds forming through the sharing of electrons by adjacent atoms. In such an approach this can
Valence Bond Theory Atomic Orbitals are bad mmmkay Overlap and Bonding Lewis taught us to think of covalent bonds forming through the sharing of electrons by adjacent atoms. In such an approach this can
Downloaded from
 1 Class XII: Chemistry Chapter 9: Coordination Compounds 1. Difference between coordination compound and double bond: Coordination compound A coordination compound contains a central metal atom or ion
1 Class XII: Chemistry Chapter 9: Coordination Compounds 1. Difference between coordination compound and double bond: Coordination compound A coordination compound contains a central metal atom or ion
Assignment 3 Due Tuesday, March 31, 2009
 Assignment 3 Due Tuesday, March 31, 2009 Download and read the Math_techniques.pdf file from the Handouts section of the class web page. Do problems 1, 2, and 4 following section C (for problem 1, you
Assignment 3 Due Tuesday, March 31, 2009 Download and read the Math_techniques.pdf file from the Handouts section of the class web page. Do problems 1, 2, and 4 following section C (for problem 1, you
