Structure/Nerve Membrane Effect Relationships of Some Local Anaesthetics
|
|
|
- Roy Potter
- 5 years ago
- Views:
Transcription
1 Gen. Pbysigl. Biophys. (1986), 5, Structure/Nerve Membrane Effect Relationships of Some Local Anaesthetics M. TRIPSA 1, V. EM. SAHINI 2, C. NAE 2 and V. VASILESCU 1 1 Department of Biophysics, Faculty of Medicine, Bucharest, Romania 2 Laboratory of Physical Chemistry, Polytechnic Institute, Bucharest, Romania Abstract. Data on electron structure and octanol-water partition coefficients of certain local anaesthetics are compared with the capacity of reversibly block the ajstjpri potential of the frog sciatic nerve. Some active parts in the molecules of these substances have been shown to be primarily involved in the action of local anaesthetics. Key words: Local anaesthetics Nerve membrane Partition coefficient Nfpjecular orbitals Introduction Mechanisms underlying the action potential of the nerve membrane are ionic currents passing through the selective membrane channels. One of the present major lines of research concerns the molecular mechanisms by which ionic channels operate in excitable membranes (Hille 1978; Vasilescu 1982). Information concerning the nature and structure of these channels can be obtained by studying the action of local anaesthetics on nerve membranes (Seeman 1972; Strichartz 1973 ; Courtney 1975; Khodorov et al. 1974, 1976; Aimers and Cahalan 1977; Hille 1977a, b, 1980; Cahalan 1978; Borchard and Drouin 1980). Lgeal anaesthetics are drugs which reversibly block sodium channels in nerve membranes (Taylor 1959). Although numerous theories try to explain anaesthesia, the mojgcular mechanisms in the action of local anaesthetics still remain unclear. The present work analyses some data on the electron characteristics of the molecules of anaesthetics by comparing their structure with the ability to reversibly block agtion potential. Materials and Methods a) Capacity of Local Anaesthetics to Reversibly mock Action Potential We have studied the action of four local anaesthetics (procaine, lidocaine, tetracaine and benzocaine) on the sciatic nerve of the frog Rana ridibunda. The effects of the anaesthetics were estimated by determining standard electrophysiological parameters of the action potential (rheobase, chronaxy,
2 372 Tripsa et al conduction velocity of nerve impulse), which are modified by various concentrations of local anaesthetics: this allows to assess the abiiity of these substances to block action potential. Different concentrations of local anaesthetics in Ringer physiological solution at ph = 7.4 were used. The solution contained (in mol/1): 110 x 10" 3 NaCl; 2.5 x 10" 3 KC1; 1.8 x 10" 3 CaCl 2. 2 H 2 0; 4.5 x 10" 3 TRIS-CI. The ph of the solution of local anaesthetics in Ringer solution was adjusted to 7.4 with NaOH and checked using a Beckman ph-meter The concentration of local anaesthetic which induced a reversible block of action potential was taken as the threshold dose. The threshold doses obtained are listed in Table 1. Our data obtained by this method are in a good agreement with those reported by Strichariz (1976). b) Lipid/Phase/Water Partition of Local Anaesthetics The physiological activity of local anaesthetics depends on their ability to pass through the lipid membrane barrier (Singer 1980), a good measure of this ability is the partition coefficient (P). The Hansch fragment method was used to calculate the octanol/water partition coefficients of the four local anaesthetics studied. The values obtained are shown in Table 2. It can be seen that the Table 1. Relative potencies of some local anaesthetics Drug Threshold dose (reversible block of action potential) (mol/1) Effective* concentration (mol/1) Z (for N atom bound to aromatic ring) Lidocaine Tetracaine Procaine Benzocaine * Effective concentrations were obtained by evaluating the sodium permeability of Ranvier node of frog sciatic nerve under voltage-clamp reduced by 50 per cent (Strichartz 1976). Table 2. Octanol/water partition coefficients of some local anaesthetics log P Molecule Neutral form Protonated form Experimental Computed Experimental Computed Procaine Benzocaine Tetracaine Lidocaine 0.16 (ph = 7.3) 1.93'(pH = 7.4) (ph = 7.4) (ph = 3.8) ) Hansch and Leo (1979) 2) Reduced to octanol/water partition according to: Leo et al. (1971)
3 Membrane Effect of Local Anaesthetics 373 fragment method reflects in a rather exact way ph-dependent variations of partition coefficients. At standard temperature (25 C) the neutral anaesthetics are strongly hydrophobic. Actually, temperature has only negligible effects on P when mutually weakly miscible solvents are used, such as n-octanol and water. Results obtained with a variety of solutes in different solvent systems have shown that the effect usually is of the order of 0.01 log/unit/deg. (Leo et al. 1971). c) Electron Characteristics of Local Anaesthetics A self-consistent-technique, computation of the Hiickel n molecular orbital, was used to compare our experimental data concerning the action of local anaesthetics on the peripheral nerve with their electron structures. The chemical structure of the drugs studied is shown in Fig. 1. Procaine H 2 N-(_J>-C-0-CH 2 - CH^-N^ Tetracame \N -Ĺ }-C-0-CH 2 -CH 2 -N( 0 Benzocaine H 2 N 4 \-C-0 ~^2~ CH 3 Lidocaine, 3 o YH 3 Fig. 1. Chemical structure of some local anaesthetics. Following input parameters were used: ho= 1; fco = 2; h N = 1.5 (for tetracaine h N = 1); /IC_CH 3 = 0.5; fe N c= 1; k c -o= 1; kc~o-0.s with a self-consistency up to 1 % in total energy in each case. Following molecular parameters were calculated: charge densities (q), double bond order (p), free valence (F) (Fig. 2), Discussion Computation of n molecular orbitals of the anaesthetics studied provided some data on their structural characteristics which could be compared with those concerning their threshold effects and partition coefficients. The highest value of this parameter was obtained for the aromatic N charge in lidocaine (Z N = ), an intermediary value for tetracaine (Z N = ) and a lower value for procaine and benzocaine (Z N ). The results of our comparative study can be summarized as follows: 1. Aromatic nitrogen charge seems to be the major component responsible for the local anaesthetic activity. 2. The presence of a second amino-group in the structure of the molecule of
4 374 Tripsa et al. the anaesthetic raises its activity. This may be explained by the co-operative effects between nitrogen atoms. The co-operative effect of the second nitrogen atom increases the anaesthetic activity with the increase being proportionally inverse to the distance of the atom from the aromatic nitrogen atom. The lowest threshold dose was found for lidocaine which has its amino group closer to the aromatic nitrogen (see Fig. 1). tmu t n N "53245 H Z N ""TlSSS*^. 1-KS2 1B3S A30S T ff 7 2SM Fig. 2. Molecular parameters (q,, p ls and F) computed for local anaesthetics: A) procaine and benzocaine; B) tetracaine; C) lidocaine. Values of the parameters are shown near the corresponding atom i(q,), between the atoms i and /(P,,) and indicated by the corresponding arrow (F,). Our results suggest that the receptor for local anaesthetics has some functional groups which are able to interact with the aromatic nitrogen atom of these drugs. The aromatic nitrogen seems to induce the anaesthetic phenomenon itself while the second nitrogen atom and the alkyl group seem to determine by co-operative effects the intensity of anaesthesia. 3. Lidocaine, the most active one of the local anaesthetics studied, has both
5 Membrane Effect of Local Anaesthetics 375 a nitrogen atom bound to the aromatic ring and a hight value of the partition coefficient, indicating its capacity to pass easily from the aqueous to the lipid phase. As compared to lidocaine, tetracaine with a by one order higher partition coefficient has a higher threshold dose on account of 2 structural parameters (a N-butyl group bound to the aromatic nitrogen and a carboxyl instead of carbonyl group); it is less active, probably due to the large distance between the two nitrogen atoms co-operatively participating in the blocking action. Procaine, with a threshold dose equal to that of tetracaine, has a much smaller partition coefficient than tetracaine. Although the molecules of both drugs show the same distance between the co-operative nitrogen atoms, the aromatic nitrogen atom of procaine is not protected by N-butyl group as it is case in tetracaine. Due to this both drugs have equal threshold doses. The protected active centre of tetracaine is counter-balanced by its better partition capacity. The lowest anaesthetic potential was found for benzocaine which lacks the co-operative effect of the second nitrogen atom, though it has a partition coefficient by two orders higher than does procaine. 4. It is worth mentioning that for the sequence lidocaine-tetracaine-benzocaine the increase in the threshold doses is parallel to the decrease in charge of the nitrogen atom bound to the aromatic ring. Procaine represents an exception due to its easy interaction with the receptor. 5. The sequence of partition coefficients is the same for neutral and protonated forms. We only dealt with neutral forms, administering local anaesthetics in a chloride-water solution. Due to alkaline ph (about 7.35) of the body fluids, chloride is rapidly converted into its neutral form which is able to penetrate the membrane lipid phases. REFERENCES Aimers W., Cahalan M. D. (1977): Interaction between a local anaesthetic, the sodium channel.gates and tetrodotoxin. Biophys. J. 17, 205a Cahalan M. D. (1978): Local anaesthetic block of sodium channels in normal and pronase-treated squid giant axons. Biophys. J. 23, 285 Courtney K. R. (1975): Mechanism of frequency-dependent inhibition of sodium current in frog myelinated nerve by the lidocaine derivative GEA 968. J. Pharmacol. Exp. Ther. 195, 225 Hansch C, Leo A. J. (1979): Substituent Constants for Correlation Analysis in Chemistry and Biology. J. Wiley, New York Hille B. (1977a): The ph-dependent rate of local anaesthetic on the node of Ranvier. J. Gen. Physiol. 69, 475 Hille B. (1977b): Local anaesthetics; Hydrophilic and hydrophobic pathways for the drug-receptor reaction. J. Gen. Physiol. 69, 497 Hille B. (1978): Ionic channels in excitable membranes. Current problems and biophysical approaches. Biophys. J. 22, 283 Hille B. (1980): Theories of Anaesthesia; General Perturbation versus Specific Receptors in Molecular
6 376 Tripsa e t al. Mechanisms of Anaesthesia. In: Progress in Anesthesiology, vol. 2 (Ed. B. Raymond Fink), pp. 2, Raven Press, New York Khodorov B. I., Shishkova L. D., Peganov E. M. (1974): The effect of procaine and calcium ions on slow sodium inactivation in the membrane of Ranvier's node of frog. Bull. Exp. Biol. Med. 3, Khodorov B. I., Shiskhova L. D., Peganov E. M., Revenko S. (1976): Inhibition of sodium currents in frog Ranvier node treated with local anaesthetics. Role of siow sodium inactivation. Biochim. Biophys. Acta. 433, 409-^35 Leo A., Hansch A., Elkins D. (1971): Partition coefficients and their uses. Chem. Rev. 71, 525 Seeman P. (1972): The membrane actions of anaesthetics and tranquilizers. Pharmacol. Rev. 24, 583 Singer M. A., Jain M. K. (1980): Interaction of four local anaesthetics with phospholipid bilayer membranes; permeability effects and possible mechanisms. Can. J. Biochem. 58, 815 Strichartz G. (1976): Molecular mechanisms of nerve block by local anaesthetics. Anaesthesiology 45, 421 Strichartz G. R. (1973): The inhibition of sodium currents in myelinated nerve by quaternary deriváte of lidocaine. J. Gen. Physiol. 62, 37 Taylor R. E. (1959): Effect of procaine on electrical properties of squid axon membrane. Amer. J. Physiol. 196, 1071 Vasilescu V. (1981): Introduction to Neurobiophysics. Abacus Press, Tunbridge Wells, Kent Received November 14, 1984/Accepted September 25, 1985 f
LOCAL ANESTHETIC ALTERATION OF
 LOCAL ANESTHETIC ALTERATION OF MINIATURE ENDPLATE CURRENTS AND ENDPLATE CURRENT FLUCTUATIONS ROBERT L. RUFF From the Department of Physiology and Biophysics, University of Washington School of Medicine,
LOCAL ANESTHETIC ALTERATION OF MINIATURE ENDPLATE CURRENTS AND ENDPLATE CURRENT FLUCTUATIONS ROBERT L. RUFF From the Department of Physiology and Biophysics, University of Washington School of Medicine,
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 01
 Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Questions Patrick: An Introduction to Medicinal Chemistry 5e 01) Which of the following molecules is a phospholipid? a. i b. ii c. iii d. iv 02) Which of the following statements is false regarding the
Hole s Human Anatomy and Physiology Eleventh Edition. Chapter 2
 Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Hole s Human Anatomy and Physiology Eleventh Edition Shier Butler Lewis Chapter 2 1 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. CHAPTER 2 CHEMICAL BASIS OF
Lecture 10 : Neuronal Dynamics. Eileen Nugent
 Lecture 10 : Neuronal Dynamics Eileen Nugent Origin of the Cells Resting Membrane Potential: Nernst Equation, Donnan Equilbrium Action Potentials in the Nervous System Equivalent Electrical Circuits and
Lecture 10 : Neuronal Dynamics Eileen Nugent Origin of the Cells Resting Membrane Potential: Nernst Equation, Donnan Equilbrium Action Potentials in the Nervous System Equivalent Electrical Circuits and
CELL BIOLOGY - CLUTCH CH. 9 - TRANSPORT ACROSS MEMBRANES.
 !! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
!! www.clutchprep.com K + K + K + K + CELL BIOLOGY - CLUTCH CONCEPT: PRINCIPLES OF TRANSMEMBRANE TRANSPORT Membranes and Gradients Cells must be able to communicate across their membrane barriers to materials
EFFECTS OF CALCIUM AND MAGNESIUM ON PERIPHERAL NERVE CONDUCTION
 Copyright 2003 by Institute of Pharmacology Polish Academy of Sciences Polish Journal of Pharmacology Pol. J. Pharmacol., 2003, 55, 25 30 ISSN 1230-6002 EFFECTS OF CALCIUM AND MAGNESIUM ON PERIPHERAL NERVE
Copyright 2003 by Institute of Pharmacology Polish Academy of Sciences Polish Journal of Pharmacology Pol. J. Pharmacol., 2003, 55, 25 30 ISSN 1230-6002 EFFECTS OF CALCIUM AND MAGNESIUM ON PERIPHERAL NERVE
Potential, Structure, and Excitability of Giant Axon Membrane
 Potential, Structure, and Excitability of Giant Axon Membrane T. NARAHASHI From the Laboratory of Applied Entomology, Faculty of Agriculture, University of Tokyo, Tokyo, Japan Intracellular perfusion of
Potential, Structure, and Excitability of Giant Axon Membrane T. NARAHASHI From the Laboratory of Applied Entomology, Faculty of Agriculture, University of Tokyo, Tokyo, Japan Intracellular perfusion of
SAXITOXIN BINDING TO THE MAMMALIAN SODIUM CHANNEL. Competition by monovalent and divalent cations
 SAXITOXIN BINDING TO THE MAMMALIAN SODIUM CHANNEL Competition by monovalent and divalent cations J. B. WEIGELE and R. L. BARCHI* Departments of Neurology and of Biochemistiy and Biophysics, University
SAXITOXIN BINDING TO THE MAMMALIAN SODIUM CHANNEL Competition by monovalent and divalent cations J. B. WEIGELE and R. L. BARCHI* Departments of Neurology and of Biochemistiy and Biophysics, University
Interaction of H + Ions with Acid Groups in Normal Sodium Channels
 Gen. Physiol. Biophys. (1982). 1, 5 19 5 Interaction of H + Ions with Acid Groups in Normal Sodium Channels G. N. MOZHAYEVA, A. P. NAUMOV and Yu. A. NEGULYAEV Institute of Cytology, Academy of Sciences
Gen. Physiol. Biophys. (1982). 1, 5 19 5 Interaction of H + Ions with Acid Groups in Normal Sodium Channels G. N. MOZHAYEVA, A. P. NAUMOV and Yu. A. NEGULYAEV Institute of Cytology, Academy of Sciences
MEMBRANE POTENTIALS AND ACTION POTENTIALS:
 University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
University of Jordan Faculty of Medicine Department of Physiology & Biochemistry Medical students, 2017/2018 +++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++++ Review: Membrane physiology
6.3.4 Action potential
 I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
I ion C m C m dφ dt Figure 6.8: Electrical circuit model of the cell membrane. Normally, cells are net negative inside the cell which results in a non-zero resting membrane potential. The membrane potential
A Study of Properties of Batrachotoxin Modified Sodium Channels
 Gen. Physiol. Biophys. (1986), 5, 17 46 17 A Study of Properties of Batrachotoxin Modified Sodium Channels G. N. MOZHAYEVA 1, A. P. NAUMOV 1 and B. I. KHODOROV 2 1 Institute of Cytology, Academy of Sciences
Gen. Physiol. Biophys. (1986), 5, 17 46 17 A Study of Properties of Batrachotoxin Modified Sodium Channels G. N. MOZHAYEVA 1, A. P. NAUMOV 1 and B. I. KHODOROV 2 1 Institute of Cytology, Academy of Sciences
Chapter 2: Chemistry & Life
 Chapter 2: Chemistry & Life 1. Atoms 2. Molecules 3. Water 1. Atoms A Generic Atom All Matter is made of Atoms Atoms are the smallest indivisible unit of matter 92 different types of atoms: ELEMENTS Atoms
Chapter 2: Chemistry & Life 1. Atoms 2. Molecules 3. Water 1. Atoms A Generic Atom All Matter is made of Atoms Atoms are the smallest indivisible unit of matter 92 different types of atoms: ELEMENTS Atoms
Chapter 2: Chemistry & Life. 1. Atoms. 2. Molecules. 3. Water. 1. Atoms. A Generic Atom
 Chapter 2: Chemistry & Life 1. Atoms 2. Molecules 3. Water 1. Atoms A Generic Atom 1 All Matter is made of Atoms Atoms are the smallest indivisible unit of matter 92 different types of atoms: ELEMENTS
Chapter 2: Chemistry & Life 1. Atoms 2. Molecules 3. Water 1. Atoms A Generic Atom 1 All Matter is made of Atoms Atoms are the smallest indivisible unit of matter 92 different types of atoms: ELEMENTS
Hole s Human Anatomy and Physiology Tenth Edition. Chapter 2
 PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Outlines to accompany Hole s Human Anatomy and Physiology Tenth Edition Shier w Butler w Lewis Chapter 2 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Physiology Coloring Book: Panels 29, 32, 33,
 ELEC4623/ELEC9734: Semester 2, 2009 Dr Stephen Redmond School of Electrical Engineering & Telecommunications Email: s.redmond@unsw.edu.au Ph: 9385 6101 Rm: 458, ELECENG (G17) Physiology Coloring Book:
ELEC4623/ELEC9734: Semester 2, 2009 Dr Stephen Redmond School of Electrical Engineering & Telecommunications Email: s.redmond@unsw.edu.au Ph: 9385 6101 Rm: 458, ELECENG (G17) Physiology Coloring Book:
Membrane Physiology. Dr. Hiwa Shafiq Oct-18 1
 Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
Membrane Physiology Dr. Hiwa Shafiq 22-10-2018 29-Oct-18 1 Chemical compositions of extracellular and intracellular fluids. 29-Oct-18 2 Transport through the cell membrane occurs by one of two basic processes:
BRIEF COMMUNICATION OF ASYMMETRY CURRENT SQUID AXON MEMBRANE FREQUENCY DOMAIN ANALYSIS
 FREQUENCY DOMAIN ANALYSIS OF ASYMMETRY CURRENT IN SQUID AXON MEMBRANE SHIRo TAKASHIMA, Department ofbioengineering D2, University of Pennsylvania, Philadelphia, Pennsylvania 19104 U.S.A. ABSTRACT The change
FREQUENCY DOMAIN ANALYSIS OF ASYMMETRY CURRENT IN SQUID AXON MEMBRANE SHIRo TAKASHIMA, Department ofbioengineering D2, University of Pennsylvania, Philadelphia, Pennsylvania 19104 U.S.A. ABSTRACT The change
MEMBRANE STRUCTURE. Lecture 9. Biology Department Concordia University. Dr. S. Azam BIOL 266/
 MEMBRANE STRUCTURE Lecture 9 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University RED BLOOD CELL MEMBRANE PROTEINS The Dynamic Nature of the Plasma Membrane SEM of human erythrocytes
MEMBRANE STRUCTURE Lecture 9 BIOL 266/4 2014-15 Dr. S. Azam Biology Department Concordia University RED BLOOD CELL MEMBRANE PROTEINS The Dynamic Nature of the Plasma Membrane SEM of human erythrocytes
There are two main requirements for atoms to form a covalent bond and make a molecule:
 HOW ATOMS BOND TO EACH OTHER Covalent bonding Remember that a hydrogen atom has 1 proton and 1 electron and that the electron and the proton are attracted to each other. But if the atoms get close enough
HOW ATOMS BOND TO EACH OTHER Covalent bonding Remember that a hydrogen atom has 1 proton and 1 electron and that the electron and the proton are attracted to each other. But if the atoms get close enough
Channels can be activated by ligand-binding (chemical), voltage change, or mechanical changes such as stretch.
 1. Describe the basic structure of an ion channel. Name 3 ways a channel can be "activated," and describe what occurs upon activation. What are some ways a channel can decide what is allowed to pass through?
1. Describe the basic structure of an ion channel. Name 3 ways a channel can be "activated," and describe what occurs upon activation. What are some ways a channel can decide what is allowed to pass through?
Transmission of Nerve Impulses (see Fig , p. 403)
 How a nerve impulse works Transmission of Nerve Impulses (see Fig. 12.13, p. 403) 1. At Rest (Polarization) outside of neuron is positively charged compared to inside (sodium ions outside, chloride and
How a nerve impulse works Transmission of Nerve Impulses (see Fig. 12.13, p. 403) 1. At Rest (Polarization) outside of neuron is positively charged compared to inside (sodium ions outside, chloride and
Electrophysiology of the neuron
 School of Mathematical Sciences G4TNS Theoretical Neuroscience Electrophysiology of the neuron Electrophysiology is the study of ionic currents and electrical activity in cells and tissues. The work of
School of Mathematical Sciences G4TNS Theoretical Neuroscience Electrophysiology of the neuron Electrophysiology is the study of ionic currents and electrical activity in cells and tissues. The work of
Chapter 2. Introduction: Chapter Chemical Basis of Life. Structure of Matter:
 Chapter 2.1-2.2 Read text 2.1 and describe why chemistry is important in understanding life. Read text 2.2 and discuss how atomic structure determines how atoms interact. Also describe the types of chemical
Chapter 2.1-2.2 Read text 2.1 and describe why chemistry is important in understanding life. Read text 2.2 and discuss how atomic structure determines how atoms interact. Also describe the types of chemical
Membrane Potentials and Bioelectricity
 Membrane Potentials and Bioelectricity Hugh Purdy Honors University Physics II November 29, 2010 Most, if not all, cells in the human body have a net electric charge to some degree on either side of their
Membrane Potentials and Bioelectricity Hugh Purdy Honors University Physics II November 29, 2010 Most, if not all, cells in the human body have a net electric charge to some degree on either side of their
BRIEF COMMUNICATION 3,4-DIAMINOPYRIDINE A POTENT NEW POTASSIUM CHANNEL BLOCKER
 BRIEF COMMUNICATION 3,4-DIAMINOPYRIDINE A POTENT NEW POTASSIUM CHANNEL BLOCKER GLENN E. KIRSCH AND ToSHIo NARAHASHI, Department ofpharmacology, Northwestem University Medical School, Chicago, Illinois
BRIEF COMMUNICATION 3,4-DIAMINOPYRIDINE A POTENT NEW POTASSIUM CHANNEL BLOCKER GLENN E. KIRSCH AND ToSHIo NARAHASHI, Department ofpharmacology, Northwestem University Medical School, Chicago, Illinois
OT Exam 1, August 9, 2002 Page 1 of 8. Occupational Therapy Physiology, Summer Examination 1. August 9, 2002
 Page 1 of 8 Occupational Therapy Physiology, Summer 2002 Examination 1 August 9, 2002 Dr. Heckman's section is questions 1-6 and each question is worth 5 points for a total of 30 points. Dr. Driska's section
Page 1 of 8 Occupational Therapy Physiology, Summer 2002 Examination 1 August 9, 2002 Dr. Heckman's section is questions 1-6 and each question is worth 5 points for a total of 30 points. Dr. Driska's section
me239 mechanics of the cell - syllabus me239 mechanics of the cell me239 mechanics of the cell - grading me239 mechanics of the cell - overview
 6 mechanotransduction wong, goktepe, kuhl [2010] me239 mechanics of the cell add l information http://biomechanics.stanford.edu and coursework 1 me239 mechanics of the cell - syllabus favorite topics in
6 mechanotransduction wong, goktepe, kuhl [2010] me239 mechanics of the cell add l information http://biomechanics.stanford.edu and coursework 1 me239 mechanics of the cell - syllabus favorite topics in
BRIEF COMMUNICATIONS COMPUTATION OF AXON GATING CURRENTS FROM DIPOLE MOMENT CHANGES IN CHANNEL SUBUNITS
 BRIEF COMMUNICATIONS COMPUTATION OF AXON GATING CURRENTS FROM DIPOLE MOMENT CHANGES IN CHANNEL SUBUNITS E. LEVITANand Y. PALTI From the Department of Physiology and Biophysics, Technion School of Medicine,
BRIEF COMMUNICATIONS COMPUTATION OF AXON GATING CURRENTS FROM DIPOLE MOMENT CHANGES IN CHANNEL SUBUNITS E. LEVITANand Y. PALTI From the Department of Physiology and Biophysics, Technion School of Medicine,
Basic Chemistry. Chemistry Review. Bio 250: Anatomy & Physiology
 Basic Chemistry Bio 250: Anatomy & Physiology Chemistry Review It is going to be your responsibility to review the basic principles of chemistry you learned in BIO 101 This basic set of notes will help
Basic Chemistry Bio 250: Anatomy & Physiology Chemistry Review It is going to be your responsibility to review the basic principles of chemistry you learned in BIO 101 This basic set of notes will help
DYNAMICS OF POTASSIUM ION CURRENTS IN
 DYNAMICS OF POTASSIUM ION CURRENTS IN SQUID AXON MEMBRANE A RE-EXAMINATION J. W. MOORE AND STEVEN H. YOUNG, Department ofphysiology, Duke University Medical Center, Durham, North Carolina 27710, and Marine
DYNAMICS OF POTASSIUM ION CURRENTS IN SQUID AXON MEMBRANE A RE-EXAMINATION J. W. MOORE AND STEVEN H. YOUNG, Department ofphysiology, Duke University Medical Center, Durham, North Carolina 27710, and Marine
MULTIPLE CHOICE. Circle the one alternative that best completes the statement or answers the question.
 Summer Work Quiz - Molecules and Chemistry Name MULTIPLE CHOICE. Circle the one alternative that best completes the statement or answers the question. 1) The four most common elements in living organisms
Summer Work Quiz - Molecules and Chemistry Name MULTIPLE CHOICE. Circle the one alternative that best completes the statement or answers the question. 1) The four most common elements in living organisms
Hydrogen Bonding & Molecular Design Peter
 Hydrogen Bonding & Molecular Design Peter Kenny(pwk.pub.2008@gmail.com) Hydrogen Bonding in Drug Discovery & Development Interactions between drug and water molecules (Solubility, distribution, permeability,
Hydrogen Bonding & Molecular Design Peter Kenny(pwk.pub.2008@gmail.com) Hydrogen Bonding in Drug Discovery & Development Interactions between drug and water molecules (Solubility, distribution, permeability,
A THERMODYNAMIC MECHANISM BEHIND AN ACTION POTENTIAL AND BEHIND ANESTHESIA
 Biophysical Reviews and Letters Vol. 5, No. 1 (2010) 35 41 c World Scientific Publishing Company DOI: 10.1142/S1793048010001123 A THERMODYNAMIC MECHANISM BEHIND AN ACTION POTENTIAL AND BEHIND ANESTHESIA
Biophysical Reviews and Letters Vol. 5, No. 1 (2010) 35 41 c World Scientific Publishing Company DOI: 10.1142/S1793048010001123 A THERMODYNAMIC MECHANISM BEHIND AN ACTION POTENTIAL AND BEHIND ANESTHESIA
Curtis et al. Il nuovo Invito alla biologia.blu BIOLOGY HIGHLIGHTS KEYS
 BIOLOGY HIGHLIGHTS KEYS Watch the videos and download the transcripts of this section at: online.scuola.zanichelli.it/curtisnuovoinvitoblu/clil > THE HUMAN NERVOUS SYSTEM 2. WARM UP a) The structures that
BIOLOGY HIGHLIGHTS KEYS Watch the videos and download the transcripts of this section at: online.scuola.zanichelli.it/curtisnuovoinvitoblu/clil > THE HUMAN NERVOUS SYSTEM 2. WARM UP a) The structures that
Biomedical Instrumentation
 ELEC ENG 4BD4: Biomedical Instrumentation Lecture 5 Bioelectricity 1. INTRODUCTION TO BIOELECTRICITY AND EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
ELEC ENG 4BD4: Biomedical Instrumentation Lecture 5 Bioelectricity 1. INTRODUCTION TO BIOELECTRICITY AND EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
Neurons, Synapses, and Signaling
 Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
Chapter 48 Neurons, Synapses, and Signaling PowerPoint Lecture Presentations for Biology Eighth Edition Neil Campbell and Jane Reece Lectures by Chris Romero, updated by Erin Barley with contributions
5.65 g = kg m = mm 174 ml = L. 711 kg = g 3.79 km = m L = μl g = mg 745 μm = cm 127 μl = ml 302 C = K 185 K = C 100 C = K
 WLHS / AP Bio / UNIT 1 Chemistry of Life Name AP Biology Summer Assignment Use Campbell CH 2-4 Biology is the study of life and living things. Before we can study and understand many biological principles,
WLHS / AP Bio / UNIT 1 Chemistry of Life Name AP Biology Summer Assignment Use Campbell CH 2-4 Biology is the study of life and living things. Before we can study and understand many biological principles,
Membrane Protein Channels
 Membrane Protein Channels Potassium ions queuing up in the potassium channel Pumps: 1000 s -1 Channels: 1000000 s -1 Pumps & Channels The lipid bilayer of biological membranes is intrinsically impermeable
Membrane Protein Channels Potassium ions queuing up in the potassium channel Pumps: 1000 s -1 Channels: 1000000 s -1 Pumps & Channels The lipid bilayer of biological membranes is intrinsically impermeable
rate at varying temperatures according to the following equations (1, 2). The notation has the following significance: I,i reaction
 Proc. Nat. Acad. Sci. USA Vol. 73, No. 2, pp. 481-485, February 1976 Biophysics Molecular mechanism of inhibition of firefly luminescence by local anesthetics (molecular theory of anesthesia/lipoprotein
Proc. Nat. Acad. Sci. USA Vol. 73, No. 2, pp. 481-485, February 1976 Biophysics Molecular mechanism of inhibition of firefly luminescence by local anesthetics (molecular theory of anesthesia/lipoprotein
Quantitative Electrophysiology
 ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Tuesday, September 18, 2012 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Tuesday, September 18, 2012 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in 1780s
Membrane Potentials, Action Potentials, and Synaptic Transmission. Membrane Potential
 Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
Cl Cl - - + K + K+ K + K Cl - 2/2/15 Membrane Potentials, Action Potentials, and Synaptic Transmission Core Curriculum II Spring 2015 Membrane Potential Example 1: K +, Cl - equally permeant no charge
Voltage-clamp and Hodgkin-Huxley models
 Voltage-clamp and Hodgkin-Huxley models Read: Hille, Chapters 2-5 (best Koch, Chapters 6, 8, 9 See also Hodgkin and Huxley, J. Physiol. 117:500-544 (1952. (the source Clay, J. Neurophysiol. 80:903-913
Voltage-clamp and Hodgkin-Huxley models Read: Hille, Chapters 2-5 (best Koch, Chapters 6, 8, 9 See also Hodgkin and Huxley, J. Physiol. 117:500-544 (1952. (the source Clay, J. Neurophysiol. 80:903-913
STEIN IN-TERM EXAM -- BIOLOGY FEBRUARY 12, PAGE 1 of 7
 STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 12, 2009 -- PAGE 1 of 7 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
STEIN IN-TERM EXAM -- BIOLOGY 3058 -- FEBRUARY 12, 2009 -- PAGE 1 of 7 There are 25 questions in this Biology 3058 exam. All questions are "A, B, C, D, E, F, G, H" questions worth one point each. There
ph = -log[h+], [H+] = 10-pH ph + poh = 14
![ph = -log[h+], [H+] = 10-pH ph + poh = 14 ph = -log[h+], [H+] = 10-pH ph + poh = 14](/thumbs/89/98654987.jpg) You may remove this page. ph = -log[h+], [H+] = 10-pH McVc = MdVd ph + poh = 14 NA = 6.02 x 1023 mol-1 JBA 2017 Chemistry Exam 3 Name: Score: /100 = /80 Multiple choice questions are worth two points each.
You may remove this page. ph = -log[h+], [H+] = 10-pH McVc = MdVd ph + poh = 14 NA = 6.02 x 1023 mol-1 JBA 2017 Chemistry Exam 3 Name: Score: /100 = /80 Multiple choice questions are worth two points each.
Neuroscience: Exploring the Brain
 Slide 1 Neuroscience: Exploring the Brain Chapter 3: The Neuronal Membrane at Rest Slide 2 Introduction Action potential in the nervous system Action potential vs. resting potential Slide 3 Not at rest
Slide 1 Neuroscience: Exploring the Brain Chapter 3: The Neuronal Membrane at Rest Slide 2 Introduction Action potential in the nervous system Action potential vs. resting potential Slide 3 Not at rest
PRESENTATION TITLE. Chemistry. Chemistry
 PRESENTATION TITLE Chemistry Chemistry Chemistry is the study of the smallest forms of matter and their interactions. Matter is anything that has mass and takes up space. Generally, chemistry deals with
PRESENTATION TITLE Chemistry Chemistry Chemistry is the study of the smallest forms of matter and their interactions. Matter is anything that has mass and takes up space. Generally, chemistry deals with
POTASSIUM PERMEABILITY IN
 SLOW CHANGES OF POTASSIUM PERMEABILITY IN THE SQUID GIANT AXON GERALD EHRENSTEIN and DANIEL L. GILBERT From the National Institutes of Health, Bethesda, Maryland, and the Marine Biological Laboratory,
SLOW CHANGES OF POTASSIUM PERMEABILITY IN THE SQUID GIANT AXON GERALD EHRENSTEIN and DANIEL L. GILBERT From the National Institutes of Health, Bethesda, Maryland, and the Marine Biological Laboratory,
Quantitative Electrophysiology
 ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Wednesday, September 13, 2006 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in
ECE 795: Quantitative Electrophysiology Notes for Lecture #1 Wednesday, September 13, 2006 1. INTRODUCTION TO EXCITABLE CELLS Historical perspective: Bioelectricity first discovered by Luigi Galvani in
Ionic Blockage of Sodium Channels in Nerve
 Ionic Blockage of Sodium Channels in Nerve ANN M. WOODHULL From the Department of Physiology and Biophysics, University of Washington School of Medicine, Seattle, Washington 98195. Dr. Woodhull's present
Ionic Blockage of Sodium Channels in Nerve ANN M. WOODHULL From the Department of Physiology and Biophysics, University of Washington School of Medicine, Seattle, Washington 98195. Dr. Woodhull's present
Chapter 2 The Chemistry of Biology. Dr. Ramos BIO 370
 Chapter 2 The Chemistry of Biology Dr. Ramos BIO 370 2 Atoms, Bonds, and Molecules Matter - all materials that occupy space and have mass Matter is composed of atoms. Atom simplest form of matter not divisible
Chapter 2 The Chemistry of Biology Dr. Ramos BIO 370 2 Atoms, Bonds, and Molecules Matter - all materials that occupy space and have mass Matter is composed of atoms. Atom simplest form of matter not divisible
SECOND PUBLIC EXAMINATION. Honour School of Physics Part C: 4 Year Course. Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS
 2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
2757 SECOND PUBLIC EXAMINATION Honour School of Physics Part C: 4 Year Course Honour School of Physics and Philosophy Part C C7: BIOLOGICAL PHYSICS TRINITY TERM 2013 Monday, 17 June, 2.30 pm 5.45 pm 15
Chapter 2 Cellular Homeostasis and Membrane Potential
 Chapter 2 Cellular Homeostasis and Membrane Potential 2.1 Membrane Structure and Composition The human cell can be considered to consist of a bag of fluid with a wall that separates the internal, or intracellular,
Chapter 2 Cellular Homeostasis and Membrane Potential 2.1 Membrane Structure and Composition The human cell can be considered to consist of a bag of fluid with a wall that separates the internal, or intracellular,
Chapter 2: Neurons and Glia
 Chapter 2: Neurons and Glia The Prototypical Neuron The Soma Cytosol: watery fluid inside the cell Organelles: membrane-enclosed structures within the soma Cytoplasm: contents within a cell membrane, e.g.,
Chapter 2: Neurons and Glia The Prototypical Neuron The Soma Cytosol: watery fluid inside the cell Organelles: membrane-enclosed structures within the soma Cytoplasm: contents within a cell membrane, e.g.,
Physiology Unit 2. MEMBRANE POTENTIALS and SYNAPSES
 Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES In Physiology Today Ohm s Law I = V/R Ohm s law: the current through a conductor between two points is directly proportional to the voltage across the
Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES In Physiology Today Ohm s Law I = V/R Ohm s law: the current through a conductor between two points is directly proportional to the voltage across the
Garden City High School Science Department Honors Living Environment Summer Assignment
 Garden City High School Science Department Honors Living Environment Summer Assignment Each student anticipating enrollment in the Honors Living Environment course will be required to complete a summer
Garden City High School Science Department Honors Living Environment Summer Assignment Each student anticipating enrollment in the Honors Living Environment course will be required to complete a summer
Charges and Potentials Divalent ions and ph. at the Nerve Surface. BEl~TI L HILLE From The Rockefeller University, New York 10021
 Published Online: 1 February, 1968 Supp Info: http://doi.org/ Downloaded from jgp.rupress.org on November 17, 2018 Charges and Potentials Divalent ions and ph at the Nerve Surface BEl~TI L HILLE From The
Published Online: 1 February, 1968 Supp Info: http://doi.org/ Downloaded from jgp.rupress.org on November 17, 2018 Charges and Potentials Divalent ions and ph at the Nerve Surface BEl~TI L HILLE From The
Lecture 2. Excitability and ionic transport
 Lecture 2 Excitability and ionic transport Selective membrane permeability: The lipid barrier of the cell membrane and cell membrane transport proteins Chemical compositions of extracellular and intracellular
Lecture 2 Excitability and ionic transport Selective membrane permeability: The lipid barrier of the cell membrane and cell membrane transport proteins Chemical compositions of extracellular and intracellular
Water is one of the few compounds found in a liquid state over most of Earth s surface.
 The Water Molecule Water is one of the few compounds found in a liquid state over most of Earth s surface. Like other molecules, water (H2O) is neutral. The positive charges on its 10 protons balance out
The Water Molecule Water is one of the few compounds found in a liquid state over most of Earth s surface. Like other molecules, water (H2O) is neutral. The positive charges on its 10 protons balance out
Chemistry 6/15/2015. Outline. Why study chemistry? Chemistry is the basis for studying much of biology.
 Chemistry Biology 105 Lecture 2 Reading: Chapter 2 (pages 20-29) Outline Why study chemistry??? Elements Atoms Periodic Table Electrons Bonding Bonds Covalent bonds Polarity Ionic bonds Hydrogen bonding
Chemistry Biology 105 Lecture 2 Reading: Chapter 2 (pages 20-29) Outline Why study chemistry??? Elements Atoms Periodic Table Electrons Bonding Bonds Covalent bonds Polarity Ionic bonds Hydrogen bonding
Is the Site of Action of Grayanotoxin the Sodium Channel Gating of Squid Axon?
 Japanese Journal of Physiology, 35, 401-410,1985 Is the Site of Action of Grayanotoxin the Sodium Channel Gating of Squid Axon? Issei SEYAMA, Kaoru YAMAOKA, Masuhide YAKEHIRO, Yoshiake YOSHIOKA, and Kazuyuki
Japanese Journal of Physiology, 35, 401-410,1985 Is the Site of Action of Grayanotoxin the Sodium Channel Gating of Squid Axon? Issei SEYAMA, Kaoru YAMAOKA, Masuhide YAKEHIRO, Yoshiake YOSHIOKA, and Kazuyuki
Introduction and the Hodgkin-Huxley Model
 1 Introduction and the Hodgkin-Huxley Model Richard Bertram Department of Mathematics and Programs in Neuroscience and Molecular Biophysics Florida State University Tallahassee, Florida 32306 Reference:
1 Introduction and the Hodgkin-Huxley Model Richard Bertram Department of Mathematics and Programs in Neuroscience and Molecular Biophysics Florida State University Tallahassee, Florida 32306 Reference:
Chimica Farmaceutica
 Chimica Farmaceutica Drug Targets Why should chemicals, some of which have remarkably simple structures, have such an important effect «in such a complicated and large structure as a human being? The answer
Chimica Farmaceutica Drug Targets Why should chemicals, some of which have remarkably simple structures, have such an important effect «in such a complicated and large structure as a human being? The answer
Neurochemistry 1. Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906
 Neurochemistry 1 Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906 How Many Neurons Do We Have? The human brain contains ~86 billion neurons and
Neurochemistry 1 Nervous system is made of neurons & glia, as well as other cells. Santiago Ramon y Cajal Nobel Prize 1906 How Many Neurons Do We Have? The human brain contains ~86 billion neurons and
OXFORD H i g h e r E d u c a t i o n Oxford University Press, All rights reserved.
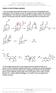 Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Chapter 002 The Chemistry of Biology
 Chapter 002 The Chemistry of Biology Multiple Choice Questions 1. Anything that occupies space and has mass is called A. Atomic B. Living C. Matter D. Energy E. Space 2. The electrons of an atom are A.
Chapter 002 The Chemistry of Biology Multiple Choice Questions 1. Anything that occupies space and has mass is called A. Atomic B. Living C. Matter D. Energy E. Space 2. The electrons of an atom are A.
Foundations in Microbiology Seventh Edition
 Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Lecture PowerPoint to accompany Foundations in Microbiology Seventh Edition Talaro Chapter 2 The Chemistry of Biology Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
Peripheral Nerve II. Amelyn Ramos Rafael, MD. Anatomical considerations
 Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Peripheral Nerve II Amelyn Ramos Rafael, MD Anatomical considerations 1 Physiologic properties of the nerve Irritability of the nerve A stimulus applied on the nerve causes the production of a nerve impulse,
Overview Organization: Central Nervous System (CNS) Peripheral Nervous System (PNS) innervate Divisions: a. Afferent
 Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
Overview Organization: Central Nervous System (CNS) Brain and spinal cord receives and processes information. Peripheral Nervous System (PNS) Nerve cells that link CNS with organs throughout the body.
Chapter 2 Basic Chemistry Outline
 Chapter 2 Basic Chemistry Outline 1.0 COMPOSITION OF MATTER 1.1 Atom 1.2 Elements 1.21 Isotopes 1.22 Radioisotopes 1.3 Compounds 1.31 Compounds Formed by Ionic Bonding 1.32 Compounds Formed by Covalent
Chapter 2 Basic Chemistry Outline 1.0 COMPOSITION OF MATTER 1.1 Atom 1.2 Elements 1.21 Isotopes 1.22 Radioisotopes 1.3 Compounds 1.31 Compounds Formed by Ionic Bonding 1.32 Compounds Formed by Covalent
IMMOBILIZATION FROM FAST Na INACTIVATION IN SQUID GIANT AXONS
 BATRACHOTOXIN UNCOUPLES GATING CHARGE IMMOBILIZATION FROM FAST Na INACTIVATION IN SQUID GIANT AXONS YEHt JOELLE TANGUY* AND JAY Z. *Laboratoire de Neurobiologie, Ecole Normale Superieure, F-755 Paris,
BATRACHOTOXIN UNCOUPLES GATING CHARGE IMMOBILIZATION FROM FAST Na INACTIVATION IN SQUID GIANT AXONS YEHt JOELLE TANGUY* AND JAY Z. *Laboratoire de Neurobiologie, Ecole Normale Superieure, F-755 Paris,
The Next Generation Science Standards (NGSS)
 The Next Generation Science Standards (NGSS) CHAPTER 4, LESSON 1: PROTONS, NEUTRONS, AND ELECTRONS HS-PS1-1. Use the periodic table as a model to predict the relative properties of elements based on the
The Next Generation Science Standards (NGSS) CHAPTER 4, LESSON 1: PROTONS, NEUTRONS, AND ELECTRONS HS-PS1-1. Use the periodic table as a model to predict the relative properties of elements based on the
Effect of Gramicidin A and Thallium Ions on Cation Effluxes in Frog Sartorius Muscle
 Gen. Physiol. Biophys. (1985), 4, 493-500 493 Effect of Gramicidin A and Thallium Ions on Cation Effluxes in Frog Sartorius Muscle N. E. SHVINKA and F. V. TOROPOVA Institute of Cytology, Academy of Sciences
Gen. Physiol. Biophys. (1985), 4, 493-500 493 Effect of Gramicidin A and Thallium Ions on Cation Effluxes in Frog Sartorius Muscle N. E. SHVINKA and F. V. TOROPOVA Institute of Cytology, Academy of Sciences
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS
 COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
COMPUTER AIDED DRUG DESIGN (CADD) AND DEVELOPMENT METHODS DRUG DEVELOPMENT Drug development is a challenging path Today, the causes of many diseases (rheumatoid arthritis, cancer, mental diseases, etc.)
CHEMICAL KINETICS. Collision theory and concepts, activation energy and its importance VERY SHORT ANSWER QUESTIONS
 Topic-3 CHEMICAL KINETICS Collision theory and concepts, activation energy and its importance 1. What is law of mass action? VERY SHORT ANSWER QUESTIONS This law relates rate of reaction with active mass
Topic-3 CHEMICAL KINETICS Collision theory and concepts, activation energy and its importance 1. What is law of mass action? VERY SHORT ANSWER QUESTIONS This law relates rate of reaction with active mass
A Brief Overview of Biochemistry. And I mean BRIEF!
 A Brief Overview of Biochemistry And I mean BRIEF! Introduction A. Chemistry deals with the composition of substances and how they change. B. A knowledge of chemistry is necessary for the understanding
A Brief Overview of Biochemistry And I mean BRIEF! Introduction A. Chemistry deals with the composition of substances and how they change. B. A knowledge of chemistry is necessary for the understanding
Lecture Notes 8C120 Inleiding Meten en Modelleren. Cellular electrophysiology: modeling and simulation. Nico Kuijpers
 Lecture Notes 8C2 Inleiding Meten en Modelleren Cellular electrophysiology: modeling and simulation Nico Kuijpers nico.kuijpers@bf.unimaas.nl February 9, 2 2 8C2 Inleiding Meten en Modelleren Extracellular
Lecture Notes 8C2 Inleiding Meten en Modelleren Cellular electrophysiology: modeling and simulation Nico Kuijpers nico.kuijpers@bf.unimaas.nl February 9, 2 2 8C2 Inleiding Meten en Modelleren Extracellular
9.01 Introduction to Neuroscience Fall 2007
 MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
MIT OpenCourseWare http://ocw.mit.edu 9.01 Introduction to Neuroscience Fall 2007 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms. 9.01 Recitation (R02)
Chemical and Physical Properties of Organic Molecules
 Chemical and Physical Properties of Organic Molecules I.Elements A. Chemical symbols: C H O P S N C=carbon, H=hydrogen, O=oxygen, P=phosphorus, S=sulfur, N=nitrogen B. Top 3 Earth s surface = O, Si, Al
Chemical and Physical Properties of Organic Molecules I.Elements A. Chemical symbols: C H O P S N C=carbon, H=hydrogen, O=oxygen, P=phosphorus, S=sulfur, N=nitrogen B. Top 3 Earth s surface = O, Si, Al
Buffered and Isotonic Solutions
 Physical Pharmacy Lecture 8 Buffered and Isotonic Solutions Assistant Lecturer in Pharmaceutics Overview Buffered Solutions Definition Buffer Equation Buffer Capacity Buffer in Biological Systems Pharmaceutical
Physical Pharmacy Lecture 8 Buffered and Isotonic Solutions Assistant Lecturer in Pharmaceutics Overview Buffered Solutions Definition Buffer Equation Buffer Capacity Buffer in Biological Systems Pharmaceutical
BIOL Week 5. Nervous System II. The Membrane Potential. Question : Is the Equilibrium Potential a set number or can it change?
 Collin County Community College BIOL 2401 Week 5 Nervous System II 1 The Membrane Potential Question : Is the Equilibrium Potential a set number or can it change? Let s look at the Nernst Equation again.
Collin County Community College BIOL 2401 Week 5 Nervous System II 1 The Membrane Potential Question : Is the Equilibrium Potential a set number or can it change? Let s look at the Nernst Equation again.
ACTION POTENTIAL. Dr. Ayisha Qureshi Professor MBBS, MPhil
 ACTION POTENTIAL Dr. Ayisha Qureshi Professor MBBS, MPhil DEFINITIONS: Stimulus: A stimulus is an external force or event which when applied to an excitable tissue produces a characteristic response. Subthreshold
ACTION POTENTIAL Dr. Ayisha Qureshi Professor MBBS, MPhil DEFINITIONS: Stimulus: A stimulus is an external force or event which when applied to an excitable tissue produces a characteristic response. Subthreshold
THE CHEMISTRY OF LIFE
 THE CHEMISTRY OF LIFE ATOMS All living things are made up of matter Atoms are the smallest unit of matter Made up of 3 subatomic particles: 1. Protons- positively charged, found in the nucleus, has mass
THE CHEMISTRY OF LIFE ATOMS All living things are made up of matter Atoms are the smallest unit of matter Made up of 3 subatomic particles: 1. Protons- positively charged, found in the nucleus, has mass
Some Predictions Concerning the Calcium Channel Model with Different Conformational States
 Gen. Physiol. Biophys. (1986), 6, 649 659 649 Short communication Some Predictions Concerning the Calcium Channel Model with Different Conformational States P. G. KOSTYUK and S. L. MIRONOV A. A. Bogomoletz
Gen. Physiol. Biophys. (1986), 6, 649 659 649 Short communication Some Predictions Concerning the Calcium Channel Model with Different Conformational States P. G. KOSTYUK and S. L. MIRONOV A. A. Bogomoletz
Structure and Function of Voltage-Gated Sodium Channels at Atomic Resolution
 Structure and Function of Voltage-Gated Sodium Channels at Atomic Resolution Royal Society of Chemistry Cambridge UK, March 2013 William A. Catterall, Department of Pharmacology, University of Washington
Structure and Function of Voltage-Gated Sodium Channels at Atomic Resolution Royal Society of Chemistry Cambridge UK, March 2013 William A. Catterall, Department of Pharmacology, University of Washington
b) What is the gradient at room temperature? Du = J/molK * 298 K * ln (1/1000) = kj/mol
 Chem350 Practice Problems Membranes 1. a) What is the chemical potential generated by the movement of glucose by passive diffusion established by a 1000 fold concentration gradient at physiological temperature?
Chem350 Practice Problems Membranes 1. a) What is the chemical potential generated by the movement of glucose by passive diffusion established by a 1000 fold concentration gradient at physiological temperature?
Chapter 2: Chemical Basis of Life
 Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
Chapter 2: Chemical Basis of Life Chemistry is the scientific study of the composition of matter and how composition changes. In order to understand human physiological processes, it is important to understand
Life is a chemical process
 CHEMISTRY FOR LIFE WHY STUDY CHEMISTRY? Chemistry is the ultimate (basic) cause of all physiological processes Interactions of atoms produce chemical changes Chemical reactions involve a transfer of energy
CHEMISTRY FOR LIFE WHY STUDY CHEMISTRY? Chemistry is the ultimate (basic) cause of all physiological processes Interactions of atoms produce chemical changes Chemical reactions involve a transfer of energy
Voltage-clamp and Hodgkin-Huxley models
 Voltage-clamp and Hodgkin-Huxley models Read: Hille, Chapters 2-5 (best) Koch, Chapters 6, 8, 9 See also Clay, J. Neurophysiol. 80:903-913 (1998) (for a recent version of the HH squid axon model) Rothman
Voltage-clamp and Hodgkin-Huxley models Read: Hille, Chapters 2-5 (best) Koch, Chapters 6, 8, 9 See also Clay, J. Neurophysiol. 80:903-913 (1998) (for a recent version of the HH squid axon model) Rothman
Learning Objectives. Learning Objectives (cont.) Chapter 2: Basic Chemistry 1. Lectures by Tariq Alalwan, Ph.D.
 Biology, 10e Mader Lectures by Tariq Alalwan, Ph.D. Learning Objectives Name the principal chemical elements in living things. Compare the physical properties (mass and charge) and locations of electrons,
Biology, 10e Mader Lectures by Tariq Alalwan, Ph.D. Learning Objectives Name the principal chemical elements in living things. Compare the physical properties (mass and charge) and locations of electrons,
Matter: Elements and Compounds
 Matter: Elements and Compounds Matter is defined as anything that takes up space and has mass. Matter exists in many diverse forms, each with its own characteristics. Rock, metals, and glass are just few
Matter: Elements and Compounds Matter is defined as anything that takes up space and has mass. Matter exists in many diverse forms, each with its own characteristics. Rock, metals, and glass are just few
AD-" IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..
 AD-"19 459 IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..,E NCLA SIFIE BIOPHYSIC S D C CHANG 6 i N 1988 Neg@14-85-K-6424
AD-"19 459 IONIC BASIS OF POTENTIAL REGULATION(U) BAYLOR COLLO / U U ijejmedicine L HOUSTON TX DEPT OF PHYSIOLOGY AND MOLECULAR 7 MEEE"..,E NCLA SIFIE BIOPHYSIC S D C CHANG 6 i N 1988 Neg@14-85-K-6424
Action Potentials & Nervous System. Bio 219 Napa Valley College Dr. Adam Ross
 Action Potentials & Nervous System Bio 219 Napa Valley College Dr. Adam Ross Review: Membrane potentials exist due to unequal distribution of charge across the membrane Concentration gradients drive ion
Action Potentials & Nervous System Bio 219 Napa Valley College Dr. Adam Ross Review: Membrane potentials exist due to unequal distribution of charge across the membrane Concentration gradients drive ion
CHAPTER 2. Life s Chemical Basis
 CHAPTER 2 Life s Chemical Basis The Chemistry of Life We are made up of elements. Atoms of one kind make up an element. Atoms are the smallest unit of an element still maintaing the element s properties.
CHAPTER 2 Life s Chemical Basis The Chemistry of Life We are made up of elements. Atoms of one kind make up an element. Atoms are the smallest unit of an element still maintaing the element s properties.
General Physics. Nerve Conduction. Newton s laws of Motion Work, Energy and Power. Fluids. Direct Current (DC)
 Newton s laws of Motion Work, Energy and Power Fluids Direct Current (DC) Nerve Conduction Wave properties of light Ionizing Radiation General Physics Prepared by: Sujood Alazzam 2017/2018 CHAPTER OUTLINE
Newton s laws of Motion Work, Energy and Power Fluids Direct Current (DC) Nerve Conduction Wave properties of light Ionizing Radiation General Physics Prepared by: Sujood Alazzam 2017/2018 CHAPTER OUTLINE
Chemical properties that affect binding of enzyme-inhibiting drugs to enzymes
 Introduction Chemical properties that affect binding of enzyme-inhibiting drugs to enzymes The production of new drugs requires time for development and testing, and can result in large prohibitive costs
Introduction Chemical properties that affect binding of enzyme-inhibiting drugs to enzymes The production of new drugs requires time for development and testing, and can result in large prohibitive costs
Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed
 Supplementary Text Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed state It is difficult to examine accessibility of cysteine-substituted mutants in the fully
Supplementary Text Inhibition of S532C by MTSET at intracellular ph 6.8 indicates accessibility in the closed state It is difficult to examine accessibility of cysteine-substituted mutants in the fully
Action potentials. Conductances channels
 Action potentials Conductances channels Cole and Curtis AC Wheatstone bridge resistance decreased during action potential R1 & R2 divide one path, Rv (variable) and Ru divide the other Galvanometer between
Action potentials Conductances channels Cole and Curtis AC Wheatstone bridge resistance decreased during action potential R1 & R2 divide one path, Rv (variable) and Ru divide the other Galvanometer between
The Action of Local Anesthetics on Myelin Structure and Nerve
 Biophysical Journal Volume 7 June 1997 2581-2587 2581 The Action of Local Anesthetics on Myelin Structure and Nerve Conduction in Toad Sciatic Nerve Leonardo Mateu,* Oscar Moran,* Rau'l Padr6n,* Maria
Biophysical Journal Volume 7 June 1997 2581-2587 2581 The Action of Local Anesthetics on Myelin Structure and Nerve Conduction in Toad Sciatic Nerve Leonardo Mateu,* Oscar Moran,* Rau'l Padr6n,* Maria
Physiology Unit 2. MEMBRANE POTENTIALS and SYNAPSES
 Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES Neuron Communication Neurons are stimulated by receptors on dendrites and cell bodies (soma) Ligand gated ion channels GPCR s Neurons stimulate cells
Physiology Unit 2 MEMBRANE POTENTIALS and SYNAPSES Neuron Communication Neurons are stimulated by receptors on dendrites and cell bodies (soma) Ligand gated ion channels GPCR s Neurons stimulate cells
