Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles: The Effects of Particle Size and Halide Ions on Aggregation
|
|
|
- Posy Watkins
- 6 years ago
- Views:
Transcription
1 Journal of Cluster Science, Vol. 16, No. 1, March 2005 (Ó 2005) DOI: /s x Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles: The Effects of Particle Size and Halide Ions on Aggregation Garry P. Glaspell, 1 Chen Zuo, 2 and Paul W. Jagodzinski 2,3 Received August 26, 2004 A surface enhanced Raman spectroscopy (SERS) investigation of the aggregation of silver nanoparticles formed via LVCC with diameters in the range 5 50 nm were studied. It was found that with nm excitation maximum enhancement is observed using particles with 11 nm diameters. Upon addition of sodium halides, enhancement is proportional to the polarizability of the anion. Maximum enhancement was observed when the concentration of the anion is approximately equal to the concentration of the adsorbate. KEY WORDS: SERS; silver; nanoparticles; aggregation; halide. INTRODUCTION The surface enhanced Raman (SER) effect is currently explained using chemical (CM) and electromagnetic (EM) mechanisms. [1 4] Chemical effects provide enhancement via an increase in the molecular polarizability of the adsorbate due to the interaction of the adsorbate with the metal surface. Maximum chemical enhancement is observed when the substrate forms a monolayer on the metal of interest. Atomic scale structural features produce varying degrees of enhancement. Electromagnetic effects provide enhancement due to an increase in the electromagnetic field experienced by the molecule on the metal surface. The latter effect is paramount when investigating nanoparticles [5]. The size and shape of the particles, the specific metal, and hence the nature of the surface plasmons, 1 Department of Chemistry, West Virginia University, Morgantown, WV Department of Chemistry & Geochemistry, Colorado School of Mines, Golden, CO To whom correspondence should be addressed. pwjag@mines.edu /05/ /0 Ó 2005 Springer Science+Business Media, Inc.
2 40 Glaspell, Zuo, and Jagodzinski which can be defined as a sea of electrons with a particular resonance, contribute to the EM mechanism. We have investigated the change in SER intensity of Rhodamine 6G (R6G) on silver nanoparticles as a function of initial particle size and as a function of aggregate formation with time. The latter work was performed both with and without the influence of halides ions. Faraday first reported that the addition of salts to metal sols resulted in aggregation of individual particles [6] and this approach has been applied to metal colloids in numerous SERS studies [7]. There are three mechanisms that describe the effect of adding salts to a metal colloid: activation, aggregation and desorption [8 10]. Activation is experienced at low concentrations of halides where the metal particles show an immediate increase in surface enhancement, but do not experience aggregation. Aggregation of the metal particles and the formation of hot spots is experienced at elevated halide concentrations. Desorption removes the adsorbate from the metal. A possible explanation provided by Hildebrandt [11] for the increased enhancement generated by activated and aggregated metal particles is a combination of the chemical and electromagnetic mechanisms. Kniepp et al. [12 14], Brus et al. [15], and Nie and Emory [16, 17] have shown that not all particles are SER active and have reported that the aggregation of individual particles can lead to intense SER enhancement through the formation of hot spots (an intense plasmon resonance that exists between two particles in close proximity). Xa et al. [18] and Van Duyne et al. [19 21] have reported that the enhancement factors for a hot spot can be as high as to and that this could be responsible for single molecule detection. Synthesis of various semiconductor and intermetallic nanoparticles via laser vaporization controlled condensation (LVCC) has been of particular interest [22]. This method utilizes well-defined conditions to produce nanoparticles of controlled size and composition without the need for chemical precursors or heat treatments. Specifically, a laser is used to produce a supersaturated metal vapor from a target inside a chamber. Convection currents, resulting from the top plate of the chamber being cooled and the bottom plate heated, affect the rate of nucleation of the metal vapor. The size of the particles can be controlled by the choice of laser power, chamber pressure, and temperature gradient [23 25]. Particles generated by this procedure are predominately spherical and have significantly less fluctuation in diameter (usually ±3 nm) compared to most reduction methods. Silver nanoparticles were generated using LVCC methodologies and SER data were collected using R6G since this adsorbate gives good enhancement on silver [26 28].
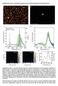
3 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles 41 EXPERIMENTAL Silver metal (99.98%) was purchased from Alpha Aesar (Ward Hill, MA). Nanoparticles of silver were generated via LVCC in a home-built chamber that consists of two metal plates modified to provide uniform heating and cooling. To provide convection, the top plate was cooled with liquid nitrogen and the bottom plate was heated with circulating water. A Laser Photonics (Orlando, Fl) Nd/YAG laser was used to ablate the metal target. Altering the laser power, chamber pressure, and temperature gradient between the two plates results in the formation of particles of various sizes [29]. The size and shape of the particles were confirmed via transmission electron microscopy (TEM) using a JEOL 1220 (Tokyo, Japan) operating at 80 Kev. Figure 1 shows a representative particle size Fig. 1. Representative particle size histogram and TEM of 11 nm Ag nanoparticles produced via LVCC.
4 42 Glaspell, Zuo, and Jagodzinski histogram and TEM micrograph for the 11 nm sample. Immediately after formation, the silver particles ( g of particles/ml of solution) were placed in a test tube and mixed with a stock solution of R6G ð3: M) in ethanol. The R6G (99%; Lamda Physik, Fort Lauderdale, FL) and ethanol (99.9%; Fisher Scientific, Pittsburgh, PA) were used without further purification. Stock solutions of aqueous NaF, NaCl, NaBr and NaI were prepared at various concentrations and added to solutions of particles alone and particles with R6G. The concentrations of the anions in the solutions that were interrogated ranged from 1: Mto1:710 1 M. The salts (99%) were purchased from Fisher Scientific (Pittsburgh, PA) and used without further purification. SER spectra were obtained using a SPEX Triplemate spectrometer (Edison, NJ) equipped with 1200 grooves/mm gratings and an Oxford Instruments CCD detector cooled to 133 K. A krypton ion laser (Lexel Laser, Fremont, CA) was used to provide excitation at nm. The laser power was set to 18 mw (at the sample) and the collection time was 5 s. The intensities of the SER signals were normalized relative to the well-separated ethanol peaks at 2931 cm 1 (internal intensity standard). No signals due to ethanol were observed below 2900 cm 1. Spectra were collected up to 46 h after sample preparation after which oxidation of the silver surface obviated the possibility of obtaining reproducible data. RESULTS AND DISCUSSION Stock solution of R6G (3: M) was added to the Ag particles until a maximum SER intensity was observed ( g/ml; due possibly to monolayer coverage since excess amounts of R6G decreased the SER intensity). Once the optimum intensity was reached SER spectra were collected at various times for each of the initial particle sizes. It is significant to note that at the concentration used the stock solution of R6G did not produce any observable peaks in the Raman spectrum. Over the range of particles tested (5 50 nm in diameter) it was determined that 11 nm particles exhibited maximum spectral enhancement during a 46 h period (vide infra). Figure 2 shows the spectra for 11 nm particles as a function of time through 46 h. Plots of normalized intensities versus time for 15, 11 and 8 nm particles for four selected aromatic ring-based normal modes (1650, 1523, 1365, 1310 cm 1 ) were generated. Figure 3 is an example of the plot generated from the data collected for the 1523 cm 1 stretching mode and shows a linear dependence of intensity with time. It is also significant to note that the increase in intensity as a function of time is not due to a lag in the R6G absorbing to the surface since the
5 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles 43 EtOH t=46 t=40 t=27 t=22 t=17 t=4 t= Wavenumbers / cm -1 Fig. 2. Raman spectra (647.1 nm excitation) of R6G ( M) on 11 nm Ag particles as a function of time (h). Ethanol peaks (EtOH) are only observed between 2900 and 3000 cm 1. Asterisks indicate which bands are being monitored. stock solution of R6G does not produce any observable peaks and enhancement is observed immediately after mixing the R6G with the silver particles. It was observed that the 11 nm particles exhibit the largest initial Normalized Intenstiy Time / (hours) 50 Fig. 3. Plot of normalized intensities of the R6G band at 1523 cm 1 as a function of time for 15 nm ðþ, 11nmð(Þ and 8 nm ð4þ diameter Ag particles. Error bars indicate the standard deviation of five trials.
6 44 Glaspell, Zuo, and Jagodzinski enhancement for each of the normal modes, although all three particle solutions do exhibit increased SER enhancement with time. Table I shows how the slopes differ among particle sizes for the four modes. The observed enhancements could be explained via the chemical and electromagnetic effects of individual particles, hot spots of preformed clusters, or any combination of these. We can deduce that the chemical effect is minor since particles in the range of nm do not exhibit a significant initial enhancement compared to the 11 nm particles, i.e., the initial spectra show a particle size dependent enhancement. We can also deduce that there are few preformed clusters since they would produce varying degrees of initial enhancement due to random numbers of hot spots which would be independent of particle size. We have observed reproducible initial enhancements and so preformed clusters are not a major contributor to the observed enhancements. Thus we can infer that the major contribution to the initial enhancement is the electromagnetic effect [18, 20]. In order to show that the intensity enhancements are related to aggregate formation we collected TEM images of the 11 nm particles throughout the period of SER spectra collection. These images are depicted in Fig. 4. Aggregation is also reflected in the red shift of the Raman signals with time (15 cm 1 shifts after 46 h) [26]. As aggregates form, it is possible that hot spots are generated where the particles come in close contact [18]. We believe that this aggregation is caused by the R6G binding the particles together as reported by Kall and coworkers [30] for silver colloid particles. The enhancement is caused by a coupling of the single particle resonances and the localization of the potential drop due to the confined region between particles [31]. These authors show that with small interparticle separations there exists a localized region of ultrahigh electromagnetic enhancement which explains the hot spots. Our Table I. Slopes of the Intensity Versus Time Plots for Four Raman Lines of R6G as a Function of Size of the Silver Nanoparticles SER Line (cm 1 ) Particle Size (/nm)
7 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles 45 Fig. 4. TEM images of 11 nm Ag particles as a function of time (h). Formation of aggregates is clearly visible. data indicate that the energies of the hot spots associated with all three initial particle sizes increase their overlap with the excitation energy as the aggregates form since we observe increasing enhancement. Clearly the aggregation of 11 nm particles does this more efficiently when compared to the aggregation of 8 nm and 15 nm silver particles as evidenced by the intensity versus time slopes presented in Table I. After 46 h the relative intensities of the SER signals of the 11 nm particles are significantly stronger than their initial intensities. Another observed trend is the difference in the slopes (see Table I) of the intensity versus time plots for the Raman bands arising from the four totally symmetric in-plane aromatic C C stretching normal modes [26]. The differences are attributed to the orientation of the R6G with respect to the hot spots where stretching modes with a significant dipole
8 46 Glaspell, Zuo, and Jagodzinski moment vector perpendicular to the hot spots should exhibit the greatest enhancement [12 21]. One possible explanation is that with increasing numbers of aggregates the contributions from adjacent particles perturb the orientation of the adsorbed molecule into a more organized pattern between the particles. We have also studied the effect of halide ions on the aggregation process. Figure 5 shows large intensity enhancements as a function of time for the four Raman bands after the addition of chloride ions (the final concentration of R6G and Cl are and M, respectively) over a 7 h-period after which the particles become oxidized and intensity diminishes [23]. Table II shows the slopes of intensity versus time plots for the various salt additions to solutions containing R6G and 11 nm particles. The slopes are typically much more positive with halide ions present. Again, one possible explanation of the enhancement as a function of time is the formation of hot spots due to aggregated particles. The significantly more positive slopes are attributed to the formation of R6Gadatom-anion surface complexes as proposed by Hildebrandt and Stockberger [26]. Comparing fluoride, chloride and bromide ions (omitting iodide because of the possibility of forming I 3 and other polyiodides due to trace amounts of water in the ethanol solution) enhancement increases proportionally with the polarizability of the anion. It is possible that at EtOH Wavenumbers / cm -1 t= 7 t= 6 t= 5 t= 4 t= 3 t= 2 t= 1 t= 0 Fig. 5. Raman spectra (647.1 nm excitation) of R6G (3: M) on 11 nm Ag particles with the addition of chloride ions (1: M) as a function of time (h). Ethanol peaks (EtOH) are only observed between 2900 and 3000 cm 1. Asterisks indicate which bands are being monitored.
9 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles 47 Table II. Slopes of the Intensity Versus Time Plots for Various Raman Lines for R6G (3: M) Adsorbed on 11 nm Ag Particles for Various Halides Salt Concentration (M) Raman Line 1310 cm ) cm ) cm ) cm )1 No salt F Cl Br I The molarities reported are the final concentrations of the halide. The addition of 0.17 M F induced precipitation of R6G. the hot spot oscillations of the plasmon resonance are able to permeate more effectively through the halide with the highest polarizability, thus extending the area providing the observed enhancement. To elucidate the effects of co-adsorption of the anions, the intensities of the Raman bands were observed immediately after the addition of each salt. The results are reported in Table III. Maximum enhancement is observed when the concentration of halide anions is approximately equal to the concentration of the adsorbate. This is different from previous results where a significant excess of anions were necessary for maximum enhancement using colloidal particle produced via a chemical reduction [32]. As shown in Table III, R6G Raman lines are observed immediately upon the addition of particles. This is contrast to results for typical particles prepared via chemical reduction processes in which the particles are allowed to age prior to
10 48 Glaspell, Zuo, and Jagodzinski Table III. Intensities of the Raman Peaks Measured, Relative to Ethanol, Immediately upon Addition of the Halide to a Solution of 3: M R6G and 11 nm Silver Nanoparticles Salt Concentration (M) Raman Line 1310 cm cm cm cm 1 No salt F Cl Br I observation of strong SER signals. In addition, the repeatability of the initial enhancement is attributed to the uniform size and shape of the particles produced via LVCC and not generally observed with particles produced via chemical reduction. This is most probably due to the absence of variable surface features that lead to variations in the observed SERS for the chemical reduction particles. Finally, the surfaces on the particles are clean and have no adsorbed species from the reduction process. The decrease in intensity when the halide concentration is high is assumed to be due to coverage of the SERS active regions by anions rather than R6G as described by the chemical and electromagnetic mechanisms. Considering the trend in the slopes for the intensity versus time plots for the four aromatic modes (shown in Table II), we find remarkable consistency for each anion at each concentration relative to particles without the addition of halides. Namely m 1523 > m 1365 > m 1650 > m This leads to the assumption that the addition of halides to the R6G/silver nanoparticle
11 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles 49 solution does not change the orientation of the R6G. One indication of aggregation is the red shift of the Raman signals as a function of time [21]. In the absence of halide ions the peaks shifted approximately 15 cm 1 after 46 h, whereas, the same red shift of 15 cm 1 was observed after 26 h with the addition of bromide ions ð1: M). CONCLUSION We have observed that the range of diameters produced for Ag particles needed for significant SERS enhancement is relatively narrow (11 ± 3 nm) for nm excitation for particles ranging in size from 5 to 50 nm. The main contribution to enhancement is the electromagnetic mechanism since particles outside the range exhibit little or no enhancement. Since the excitation wavelength was invariant, we attribute the increased SER enhancement with time to be due to particle aggregation which is supported by TEM images as a function of time. The interaction of nanoparticle aggregates could provide an increase in the number of hot spots. Thus the EM mechanism must contribute significantly to the intensity although more rigorous studies of the surface morphology need to be conducted to confirm the dependence of this effect. Future work will involve the determination of the optimum size of Ag particles for SER enhancement at various excitation wavelengths. We have also observed that the effect of halide ion concentration on SER enhancement is related to the concentration of adsorbate. Maximum enhancement is observed when the concentration of the halide is approximately equal to that of the adsorbate when monitoring the effects of aggregation and co-adsorption on the SER signal. In comparing the enhancement provided by various halides (fluoride, chloride and bromide) we have also observed a dependence on the polarizability of the anion involved. Furthermore, the invariance of the slopes of the intensity versus time plots with and without the presence of halide ions leads to the assumption that the addition of halides does not alter the orientation of R6G on the surface. In summary, we have shown that maximum SER enhancement for R6G adsorbed on silver nanoparticles produced via LVCC is obtained with 11 nm particles when using 647 nm excitation. We have observed an increase in enhancement as a function of time due to aggregation caused by the adsorbate and an increase in enhancement as a function of time due to the formation of R6G adatom anion surface complexes. We believe that the differences between our results and previously reported results are due to the differences in particles produced via LVCC and colloidal particles produced via chemical reduction methods.
12 50 Glaspell, Zuo, and Jagodzinski ACKNOWLEDGMENTS The authors express their gratitude to Dr. George Schatz (Northwestern University), Dr. Michael Kall (Chalmers University of Technology), Dr. Fred King (West Virginia University), Dr. Matthew Scanlon (Fairmont State University) and Michael Jacobs (Colorado School of Mines) for valuable consultations and helpful discussions. REFERENCES 1. C. V. Raman and K. S. Krishnan (1928). Nature 121, A. Skoog, F. J. Holler and T. A. Nieman, Principles of Instrumental Analysis, 5th edn. (Saunders College Publishing, Philadelphia, 1998). 3. M. Fleischmann, P. J. Hendra and A. McQuillan (1974). J. Chem. Phys. Lett. 26, R. K. Chang and T. E. Furtak Surface Enhanced Raman Scattering (Plenum Press, New York 1982). 5. M. Kerker, D. Wang and S. Chew (1980). Appl. Opt. 19, M. Faraday (1857). Trans. R. Soc. London 147, X. Mie (1908). Ann. Physik. 26, H. Wetzel and H. Gerischer (1980). Chem Phys. Lett. 76, E. J. Liang, C. Engert and W. Kiefer (1995). Vib. Spectrosc. 8, M. Kerker, O. Siiman and D. Wang (1984). J. Phys. Chem. 15 (88), P. Hildebrandt, S. Kellar, A. Hoffmann, F. Vanhecke and B. Schrader (1993). J. Raman Spectrosc. 24, K. Kneipp, H. Kneipp, I. Itzkan, R. R. Dasari and M. S. Feld (1999). Chem. Phys. 247 (1), K. Kneipp, H. Kneipp, R. Manoharan, I. Itzkan, R. R. Dasari and M. S. Feld (1998). J. Raman Spectrosc. 29 (8), K. Kneipp, H. Kneipp, G. Deinum, I. Itzkan, R. R. Dasari and M. S. Feld (1998). Appl. Spectrosc. 52 (2), A. M. Michaels, J. Jiang and L. Brus (2000). J. Phys. Chem. B 104 (50), S. R. Emory and S. Nie (1997). Anal. Chem. 69 (14), S. Nie and S. R. Emory (1997). Science 275 (5303), H. Xa, J. Aizpurua, M. Kall and P. Apell (2000). Phys. Rev. E:Stat. Phys., Plasmas, Fluids, Relat. Interdiscip. Top. 62 (3-B), J. C. Hulteen, D. A. Treichel, M. T. Smith, M. L. Duval, T. R. Jensen and R. P. Van Duyne (1999). J. Phys. Chem. B 103 (19), T. R. Jensen, M. L. Duval, K. L. Kelly, A. A. Lazarides, G. C. Schatz and R. P. Van Duyne (1999). J. Phys. Chem. B 103 (45), M. D. Malinsky, K. L. Kelly, G. C. Schatz and R. P. Van Duyne (2001). J. Phys. Chem. B 105 (12), S. S. Li, S. J. Silvers and M. S. El-Shall (1997). J. Phys. Chem. B 101, W. Z. Wang, I. N. Germanenko, G. Glaspell and M. S. El-Shall (2000). Clusters Nanostruct. Interfaces, Proc. Int. Symp M. S. El-Shall, NASA Conf. Publ., (NASA Microgravity Materials Science Conference, 1998) p M. S. El-Shall, S. Li, D. Graiver and U. Pernisz (1996). ACS Symp. Ser. 622 (Nanotechnology), 79.
13 Surface Enhanced Raman Spectroscopy Using Silver Nanoparticles P. Hildebrandt and M. Stockburger (1984). J. Phys. Chem. 88 (24), K. Kneipp, Y. Wang, R. R. Dasari and M. S. Feld (1995). Appl. Spectrosc. 49 (6), S. R. Emory, W. E. Haskins and S. Nie (1998). J. Am. Chem. Soc. 120 (31), M. S. El-Shall, W. Slack, W. Vann, D. Kane and D. Hanley (1994). J. Phys. Chem. 98 (12), H. Xu, E. J. Bjerneld, M. Kall and L. Borjesson (1999). Phys. Rev. Lett. 83, H. X. Xu, J. Aizpurua, M. Kall and P. Apell (2000). Phys. Rev. E 6, K. Kneipp, R. Roth, C. Engert and W. Kiefer (1993). Chem. Phys. Lett. 207, 450.
HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS
 www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
The Student Seminar Series. Jinseok Heo. Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry
 Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Surface-enhanced raman scattering from a layer of gold nanoparticles
 VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering
 Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Field enhancement and molecular response in surfaceenhanced Raman scattering and fluorescence spectroscopy
 JOURNAL OF RAMAN SPECTROSCOPY J. Raman Spectrosc. 25; 36: 51 514 Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 1.12/jrs.1357 Field enhancement and molecular response in surfaceenhanced
JOURNAL OF RAMAN SPECTROSCOPY J. Raman Spectrosc. 25; 36: 51 514 Published online in Wiley InterScience (www.interscience.wiley.com). DOI: 1.12/jrs.1357 Field enhancement and molecular response in surfaceenhanced
"Surface-Enhanced Raman Scattering
 SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
Nicolae Leopold, and Bernhard Lendl*,
 J. Phys. Chem. B 2003, 107, 5723-5727 5723 A New Method for Fast Preparation of Highly Surface-Enhanced Raman Scattering (SERS) Active Silver Colloids at Room Temperature by Reduction of Silver Nitrate
J. Phys. Chem. B 2003, 107, 5723-5727 5723 A New Method for Fast Preparation of Highly Surface-Enhanced Raman Scattering (SERS) Active Silver Colloids at Room Temperature by Reduction of Silver Nitrate
ECE280: Nano-Plasmonics and Its Applications. Week8
 ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
Nonlinear Raman Probe of Single Molecules Attached to Colloidal Silver and Gold Clusters
 Nonlinear Raman Probe of Single Molecules Attached to Colloidal Silver and Gold Clusters Katrin Kneipp 1,2, Harald Kneipp 2,IrvingItzkan 1, Ramachandra R. Dasari 1, Michael S. Feld 1, and Mildred S. Dresselhaus
Nonlinear Raman Probe of Single Molecules Attached to Colloidal Silver and Gold Clusters Katrin Kneipp 1,2, Harald Kneipp 2,IrvingItzkan 1, Ramachandra R. Dasari 1, Michael S. Feld 1, and Mildred S. Dresselhaus
Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering
 Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering By Hongyan Liang, Zhipeng Li, Wenzhong Wang, Youshi Wu, and Hongxing Xu*
Highly Surface-roughened Flower-like Silver Nanoparticles for Extremely Sensitive Substrates of Surface-enhanced Raman Scattering By Hongyan Liang, Zhipeng Li, Wenzhong Wang, Youshi Wu, and Hongxing Xu*
Temperature Dependence and Annealing Effects in Surface-Enhanced Raman Scattering on Chemically Prepared Silver Island Films
 9610 J. Phys. Chem. B 1999, 103, 9610-9615 Temperature Dependence and Annealing Effects in Surface-Enhanced Raman Scattering on Chemically Prepared Silver Island Films Chan Ho Kwon, Doo Wan Boo, Hyun Jin
9610 J. Phys. Chem. B 1999, 103, 9610-9615 Temperature Dependence and Annealing Effects in Surface-Enhanced Raman Scattering on Chemically Prepared Silver Island Films Chan Ho Kwon, Doo Wan Boo, Hyun Jin
Highly efficient SERS test strips
 Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
Electronic Supplementary Information (ESI) for Highly efficient SERS test strips 5 Ran Zhang, a Bin-Bin Xu, a Xue-Qing Liu, a Yong-Lai Zhang, a Ying Xu, a Qi-Dai Chen, * a and Hong-Bo Sun* a,b 5 10 Experimental
International Journal of Pure and Applied Sciences and Technology
 Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection
 Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Nanojet and Surface Enhanced Raman Spectroscopy (NASERS) for Highly Reproducible and Controllable Single Molecule Detection Te-Wei Chang, Manas Ranjan Gartia and Gang Logan Liu Department of Electrical
Supplemental Information for
 Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity
 Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity M. R. Barmi 1, B. D. Piorek 1, M. Moskovits 2, C. D. Meinhart 1* 1 Department of Mechanical Engineering, University
Aggregation Kinetics of Colloidal Nanoparticles in a Circulating Microfluidic Cavity M. R. Barmi 1, B. D. Piorek 1, M. Moskovits 2, C. D. Meinhart 1* 1 Department of Mechanical Engineering, University
Microfluidic/SERS Detection of Trace Explosives
 Microfluidic/SERS Detection of Trace Explosives Carl Meinhart, a Brian Piorek, a Seung Joon Lee, b Martin Moskovits b, Craig Cummings c a Department of Mechanical Engineering, University of California
Microfluidic/SERS Detection of Trace Explosives Carl Meinhart, a Brian Piorek, a Seung Joon Lee, b Martin Moskovits b, Craig Cummings c a Department of Mechanical Engineering, University of California
Polarization-Dependent Surface Enhanced Raman Scattering from Silver 1D Nanoparticle Arrays
 11609 2008, 112, 11609 11613 Published on Web 07/11/2008 Polarization-Dependent Surface Enhanced Raman Scattering from Silver 1D Nanoparticle Arrays Wei Luo,, Wytze van der Veer, Ping Chu, D. L. Mills,
11609 2008, 112, 11609 11613 Published on Web 07/11/2008 Polarization-Dependent Surface Enhanced Raman Scattering from Silver 1D Nanoparticle Arrays Wei Luo,, Wytze van der Veer, Ping Chu, D. L. Mills,
Raman Investigations of Adsorbate-Substrate Interactions on Composite Ag (or Cu)/TiO 2 Nanotubes/Ti Substrates
 WDS'09 Proceedings of Contributed Papers, Part III, 136 141, 2009. ISBN 978-80-7378-103-3 MATFYZPRESS Raman Investigations of Adsorbate-Substrate Interactions on Composite Ag (or Cu)/TiO 2 Nanotubes/Ti
WDS'09 Proceedings of Contributed Papers, Part III, 136 141, 2009. ISBN 978-80-7378-103-3 MATFYZPRESS Raman Investigations of Adsorbate-Substrate Interactions on Composite Ag (or Cu)/TiO 2 Nanotubes/Ti
Surface-Enhanced Raman Scattering of Benzenethiol Adsorbed on Silver-Exchanged Copper Powders
 SERS of Benzenethiol on Ag-Exchanged Cu Powders Bull. Korean Chem. Soc. 2008, Vol. 29, No. 2 445 Surface-Enhanced Raman Scattering of Benzenethiol Adsorbed on Silver-Exchanged Copper Powders Kuan Soo Shin,
SERS of Benzenethiol on Ag-Exchanged Cu Powders Bull. Korean Chem. Soc. 2008, Vol. 29, No. 2 445 Surface-Enhanced Raman Scattering of Benzenethiol Adsorbed on Silver-Exchanged Copper Powders Kuan Soo Shin,
Electromagnetic contributions to single-molecule sensitivity in surface-enhanced Raman scattering
 PHYSICAL REVIEW E VOLUME 62, NUMBER 3 SEPTEMBER 2000 Electromagnetic contributions to single-molecule sensitivity in surface-enhanced Raman scattering Hongxing Xu, 1 Javier Aizpurua, 2 Mikael Käll, 1 and
PHYSICAL REVIEW E VOLUME 62, NUMBER 3 SEPTEMBER 2000 Electromagnetic contributions to single-molecule sensitivity in surface-enhanced Raman scattering Hongxing Xu, 1 Javier Aizpurua, 2 Mikael Käll, 1 and
Electronic supplementary information for:
 Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy
 Raman Spectroscopy Nanomaterials Exposure? Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy Matthew Y. Chan, Weinan
Raman Spectroscopy Nanomaterials Exposure? Three-dimensional Visualization and Quantification of Gold Nanomaterial Deposition and Aggregation in Porous Media via Raman Spectroscopy Matthew Y. Chan, Weinan
Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation
 Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Prediction and Optimization of Surface-Enhanced Raman Scattering Geometries using COMSOL Multiphysics
 Excerpt from the Proceedings of the COMSOL Conference 2008 Hannover Prediction and Optimization of Surface-Enhanced Raman Scattering Geometries using COMSOL Multiphysics I. Knorr 1, K. Christou,2, J. Meinertz
Excerpt from the Proceedings of the COMSOL Conference 2008 Hannover Prediction and Optimization of Surface-Enhanced Raman Scattering Geometries using COMSOL Multiphysics I. Knorr 1, K. Christou,2, J. Meinertz
Supplementary Information for. Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings
 Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2019 Supporting Information for Halide-assisted activation of atomic hydrogen for photoreduction on
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2019 Supporting Information for Halide-assisted activation of atomic hydrogen for photoreduction on
Homogeneous surface-enhanced Raman scattering observed from self-assembled gold nanoparticle films deposited from the liquid liquid interface
 Vibrational Spectroscopy 37 (2005) 189 193 www.elsevier.com/locate/vibspec Homogeneous surface-enhanced Raman scattering observed from self-assembled gold nanoparticle films deposited from the liquid liquid
Vibrational Spectroscopy 37 (2005) 189 193 www.elsevier.com/locate/vibspec Homogeneous surface-enhanced Raman scattering observed from self-assembled gold nanoparticle films deposited from the liquid liquid
In the late 1970s, signal intensity in Raman spectroscopy was
 A Unified View of Surface-Enhanced Raman Scattering JOHN R. LOMBARDI* AND RONALD L. BIRKE Department of Chemistry, The City College of New York, New York, New York 10031 RECEIVED ON NOVEMBER 5, 2008 CON
A Unified View of Surface-Enhanced Raman Scattering JOHN R. LOMBARDI* AND RONALD L. BIRKE Department of Chemistry, The City College of New York, New York, New York 10031 RECEIVED ON NOVEMBER 5, 2008 CON
Surface-Enhanced Raman Spectroscopy
 GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
Electromagnetic Mechanism of SERS
 Electromagnetic Mechanism of SERS George C. Schatz, Matthew A. Young, and Richard P. Van Duyne Department of Chemistry, Northwestern University, Evanston, IL 60208-3113, USA {schatz,vanduyne}@chem.northwestern.edu
Electromagnetic Mechanism of SERS George C. Schatz, Matthew A. Young, and Richard P. Van Duyne Department of Chemistry, Northwestern University, Evanston, IL 60208-3113, USA {schatz,vanduyne}@chem.northwestern.edu
SERS enhancement dependence on the diameter of Au nanoparticles
 Journal of Physics: Conference Series PAPER OPEN ACCESS SERS enhancement dependence on the diameter of Au nanoparticles To cite this article: Yifei Yang 2017 J. Phys.: Conf. Ser. 844 012030 View the article
Journal of Physics: Conference Series PAPER OPEN ACCESS SERS enhancement dependence on the diameter of Au nanoparticles To cite this article: Yifei Yang 2017 J. Phys.: Conf. Ser. 844 012030 View the article
Heterodimer nanostructures induced energy focusing on metal
 Heterodimer nanostructures induced energy focusing on metal film Ting Liu a, Jingjing Hao a, Yingzhou Huang*,a, Xun Su a, Li Hu a and Yurui Fang*,b a Soft Matter and Interdisciplinary Research Center,
Heterodimer nanostructures induced energy focusing on metal film Ting Liu a, Jingjing Hao a, Yingzhou Huang*,a, Xun Su a, Li Hu a and Yurui Fang*,b a Soft Matter and Interdisciplinary Research Center,
Supporting Information for. Chad A. Mirkin* Department of Chemistry and Institute for Nanotechnology, Northwestern University,
 S1 Supporting Information for Observation of a Quadrupole Plasmon Mode for a Colloidal Solution of Gold Nanoprisms Jill E. Millstone, Sungho Park, Kevin L. Shuford, Lidong Qin, George C. Schatz, and Chad
S1 Supporting Information for Observation of a Quadrupole Plasmon Mode for a Colloidal Solution of Gold Nanoprisms Jill E. Millstone, Sungho Park, Kevin L. Shuford, Lidong Qin, George C. Schatz, and Chad
Probing the Kinetics of Ligand Exchange on Colloidal Gold. Nanoparticles by Surface-Enhanced Raman Scattering
 -Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
-Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using
 Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Headspace Raman Spectroscopy
 ELECTRONICALLY REPRINTED FROM SEPTEMBER 2014 Molecular Spectroscopy Workbench Raman Spectroscopy We examine vapor-phase Raman spectroscopy through the acquisition of spectra from gas molecules confined
ELECTRONICALLY REPRINTED FROM SEPTEMBER 2014 Molecular Spectroscopy Workbench Raman Spectroscopy We examine vapor-phase Raman spectroscopy through the acquisition of spectra from gas molecules confined
Supporting Information
 Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental Methods Instrumentation Zeta potentials and sizes of colloidal
Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental Methods Instrumentation Zeta potentials and sizes of colloidal
Near-field Raman spectroscopy using a sharp metal tip
 Journal of Microscopy, Vol. 210, Pt 3 June 2003, pp. 234 240 Received 10 August 2002; accepted 25 October 2002 Near-field Raman spectroscopy using a sharp metal tip Blackwell Publishing Ltd. A. HARTSCHUH,
Journal of Microscopy, Vol. 210, Pt 3 June 2003, pp. 234 240 Received 10 August 2002; accepted 25 October 2002 Near-field Raman spectroscopy using a sharp metal tip Blackwell Publishing Ltd. A. HARTSCHUH,
Drexel-SDP GK-12 ACTIVITY
 Drexel-SDP GK-12 ACTIVITY Subject Area(s) Chemistry, Physical Science, Science & Technology Associated Unit Nanotechnology Activity Title: A DNA biosensor Grade Level: 11th-12th Time Required: 3 hours
Drexel-SDP GK-12 ACTIVITY Subject Area(s) Chemistry, Physical Science, Science & Technology Associated Unit Nanotechnology Activity Title: A DNA biosensor Grade Level: 11th-12th Time Required: 3 hours
The use of diagrams is good and the enhancement factors seen are reasonable but not in the league of gold or silver nanoparticles.
 Reviewers' comments: Reviewer #1 (Remarks to the Author): This is a very interesting article and should be published. The authors demonstrate a method to produce metallic MoO2 nano-dumbbells as SERS substrate
Reviewers' comments: Reviewer #1 (Remarks to the Author): This is a very interesting article and should be published. The authors demonstrate a method to produce metallic MoO2 nano-dumbbells as SERS substrate
often display a deep green color due to where the SPR occurs (i.e., the wavelength of light that interacts with this specific morphology).
 Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
SERS Analysis of CMC on Gold-Assembled Micelle
 1392 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 9 Nak Han Jang SERS Analysis of CMC on Gold-Assembled Micelle Nak Han Jang Institute of Science Education, Kongju National University, Kongju, Chungnam 314-701,
1392 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 9 Nak Han Jang SERS Analysis of CMC on Gold-Assembled Micelle Nak Han Jang Institute of Science Education, Kongju National University, Kongju, Chungnam 314-701,
Supplementary information for. plasmonic nanorods interacting with J-aggregates.
 Supplementary information for Approaching the strong coupling limit in single plasmonic nanorods interacting with J-aggregates. by Gülis Zengin, Göran Johansson, Peter Johansson, Tomasz J. Antosiewicz,
Supplementary information for Approaching the strong coupling limit in single plasmonic nanorods interacting with J-aggregates. by Gülis Zengin, Göran Johansson, Peter Johansson, Tomasz J. Antosiewicz,
Flexible, Transparent and Highly Sensitive SERS. Substrates with Cross-nanoporous Structures for
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 supplementary information Flexible, Transparent and Highly Sensitive SERS Substrates with Cross-nanoporous
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2018 supplementary information Flexible, Transparent and Highly Sensitive SERS Substrates with Cross-nanoporous
Surface Enhanced Raman Scattering b.
 Surface Enhanced Raman Scattering b TitleMonobromopyridines Adsorbed on Gold Particles Author(s) Takenaka, Tohru; Eda, Kazuo; Mabuch Yoshinori; Uyeda, Natsu Citation Bulletin of the Institute for Chemi
Surface Enhanced Raman Scattering b TitleMonobromopyridines Adsorbed on Gold Particles Author(s) Takenaka, Tohru; Eda, Kazuo; Mabuch Yoshinori; Uyeda, Natsu Citation Bulletin of the Institute for Chemi
PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID
 PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID A.M. Polubotko *, V.P. Chelibanov** *A.F. Ioffe Physico-Technical Institute Russian Academy of Sciences, Politechnicheskaya
PECULIARITIES OF THE SERS SPECTRA OF THE HYDROQUINONE MOLECULE ADSORBED ON TITANIUM DIOXID A.M. Polubotko *, V.P. Chelibanov** *A.F. Ioffe Physico-Technical Institute Russian Academy of Sciences, Politechnicheskaya
Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters
 Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Size dependence of multipolar plasmon resonance frequencies and damping rates in simple metal spherical nanoparticles
 The original publication is available at www.eurphysj.org: http://www.epjst-journal.org/index.php?option=article&access=standard&itemid=9&url= /articles/epjst/pdf/7/5/st44.pdf EPJ manuscript No. (will
The original publication is available at www.eurphysj.org: http://www.epjst-journal.org/index.php?option=article&access=standard&itemid=9&url= /articles/epjst/pdf/7/5/st44.pdf EPJ manuscript No. (will
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries
 Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
ARTICLE. Surface-enhanced Raman scattering
 Double-Resonance Plasmon Substrates for Surface-Enhanced Raman Scattering with Enhancement at Excitation and Stokes Frequencies Yizhuo Chu, Mohamad G. Banaee, and Kenneth B. Crozier* School of Engineering
Double-Resonance Plasmon Substrates for Surface-Enhanced Raman Scattering with Enhancement at Excitation and Stokes Frequencies Yizhuo Chu, Mohamad G. Banaee, and Kenneth B. Crozier* School of Engineering
Application note. SERS study of EMImTFSI on gold surfaces. (c) rhd instruments GmbH & Co. KG Mareike Länger
 Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
Supporting Information
 Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
λmax = k d Supplementary Figures
 Supplementary Figures a b HQ CCD Transmission Grating Beam splitting lens Color CCD Objective Sample Dark-field Condenser Raw data Gaussian fit c λmax = k d k = 1.733 nm/pixel 53 nm 307 pixels d Supplementary
Supplementary Figures a b HQ CCD Transmission Grating Beam splitting lens Color CCD Objective Sample Dark-field Condenser Raw data Gaussian fit c λmax = k d k = 1.733 nm/pixel 53 nm 307 pixels d Supplementary
Plasmonic and Diffractive Coupling in 2D Arrays of Nanoparticles Produced by Electron Beam Lithography
 Mater. Res. Soc. Symp. Proc. Vol. 951 2007 Materials Research Society 0951-E09-20 Plasmonic and Diffractive Coupling in 2D Arrays of Nanoparticles Produced by Electron Beam Lithography Erin McLellan 1,
Mater. Res. Soc. Symp. Proc. Vol. 951 2007 Materials Research Society 0951-E09-20 Plasmonic and Diffractive Coupling in 2D Arrays of Nanoparticles Produced by Electron Beam Lithography Erin McLellan 1,
Identifying molecules at very low. Applications of Reproducible SERS Substrates for Trace Level Detection
 8 Raman Technology for Today s Spectroscopists June 2006 Applications of Reproducible SERS Substrates for Trace Level Detection Recent progress in photonic crystal design is transforming surface-enhanced
8 Raman Technology for Today s Spectroscopists June 2006 Applications of Reproducible SERS Substrates for Trace Level Detection Recent progress in photonic crystal design is transforming surface-enhanced
Fluorescent silver nanoparticles via exploding wire technique
 PRAMANA c Indian Academy of Sciences Vol. 65, No. 5 journal of November 2005 physics pp. 815 819 Fluorescent silver nanoparticles via exploding wire technique ALQUDAMI ABDULLAH and S ANNAPOORNI Department
PRAMANA c Indian Academy of Sciences Vol. 65, No. 5 journal of November 2005 physics pp. 815 819 Fluorescent silver nanoparticles via exploding wire technique ALQUDAMI ABDULLAH and S ANNAPOORNI Department
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics
 Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Evaluating nanogaps in Ag and Au nanoparticle clusters for SERS applications using COMSOL Multiphysics Ramesh Asapu 1, Radu-George Ciocarlan 2, Nathalie Claes 3, Natan Blommaerts 1, Sara Bals 3, Pegie
Novel Nanoparticles for Ultrasensitive Detection and Spectroscopy
 Final Technical Report (DOE-FG02-98ER14873) Project Officer: Dr. Richard Gordon / Dr. John Miller Novel Nanoparticles for Ultrasensitive Detection and Spectroscopy Shuming Nie Indiana University P. 0.
Final Technical Report (DOE-FG02-98ER14873) Project Officer: Dr. Richard Gordon / Dr. John Miller Novel Nanoparticles for Ultrasensitive Detection and Spectroscopy Shuming Nie Indiana University P. 0.
ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation
 S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
Supporting Information. Plasmon Ruler for Measuring Dielectric Thin Films
 Supporting Information Single Nanoparticle Based Hetero-Nanojunction as a Plasmon Ruler for Measuring Dielectric Thin Films Li Li, *a,b Tanya Hutter, c Wenwu Li d and Sumeet Mahajan *b a School of Chemistry
Supporting Information Single Nanoparticle Based Hetero-Nanojunction as a Plasmon Ruler for Measuring Dielectric Thin Films Li Li, *a,b Tanya Hutter, c Wenwu Li d and Sumeet Mahajan *b a School of Chemistry
Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering
 Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Supporting Information for: Nanoscale Chemical Imaging of a Dynamic Molecular Phase. Boundary with Ultrahigh Vacuum Tip-Enhanced Raman.
 Supporting Information for: Nanoscale Chemical Imaging of a Dynamic Molecular Phase Boundary with Ultrahigh Vacuum Tip-Enhanced Raman Spectroscopy Nan Jiang,,,* Naihao Chiang, Lindsey R. Madison, Eric
Supporting Information for: Nanoscale Chemical Imaging of a Dynamic Molecular Phase Boundary with Ultrahigh Vacuum Tip-Enhanced Raman Spectroscopy Nan Jiang,,,* Naihao Chiang, Lindsey R. Madison, Eric
Single Molecule Raman Scattering
 BY KATRIN KNEIPP AND HARALD KNEIPP WELLMAN CENTER FOR PHOTOMEDICINE HARVARD-MIT DIVISION OF HEALTH SCIENCES AND TECHNOLOGY HARVARD UNIVERSITY, MEDICAL SCHOOL BOSTON, MASSACHUSETTS 02114 Single Molecule
BY KATRIN KNEIPP AND HARALD KNEIPP WELLMAN CENTER FOR PHOTOMEDICINE HARVARD-MIT DIVISION OF HEALTH SCIENCES AND TECHNOLOGY HARVARD UNIVERSITY, MEDICAL SCHOOL BOSTON, MASSACHUSETTS 02114 Single Molecule
Preparation of Silver Nanoparticles and Their Characterization
 Preparation of Silver Nanoparticles and Their Characterization Abstract The preparation of stable, uniform silver nanoparticles by reduction of silver ions by ethanol is reported in the present paper.
Preparation of Silver Nanoparticles and Their Characterization Abstract The preparation of stable, uniform silver nanoparticles by reduction of silver ions by ethanol is reported in the present paper.
Supporting Information
 Supporting Information Precisely Controllable Core-Shell Ag@Carbon Dots Nanoparticles: Application to in Situ Super-Sensitive Monitoring of Catalytic Reactions Jing Jin, Shoujun Zhu, Yubin Song, Hongyue
Supporting Information Precisely Controllable Core-Shell Ag@Carbon Dots Nanoparticles: Application to in Situ Super-Sensitive Monitoring of Catalytic Reactions Jing Jin, Shoujun Zhu, Yubin Song, Hongyue
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.1655 Supplementary Information Initiation of Carbon Nanotube Growth by Well-Defined Carbon Nanorings Haruka Omachi 1, Takuya Nakayama 1, Eri Takahashi 2, Yasutomo Segawa 1, and Kenichiro
DOI: 10.1038/NCHEM.1655 Supplementary Information Initiation of Carbon Nanotube Growth by Well-Defined Carbon Nanorings Haruka Omachi 1, Takuya Nakayama 1, Eri Takahashi 2, Yasutomo Segawa 1, and Kenichiro
Synthesis of Colloidal Au-Cu 2 S Heterodimers via Chemically Triggered Phase Segregation of AuCu Nanoparticles
 SUPPORTING INFORMATION Synthesis of Colloidal Au-Cu 2 S Heterodimers via Chemically Triggered Phase Segregation of AuCu Nanoparticles Nathan E. Motl, James F. Bondi, and Raymond E. Schaak* Department of
SUPPORTING INFORMATION Synthesis of Colloidal Au-Cu 2 S Heterodimers via Chemically Triggered Phase Segregation of AuCu Nanoparticles Nathan E. Motl, James F. Bondi, and Raymond E. Schaak* Department of
24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability ellipsoid. Selection rules.
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 24/ Rayleigh and Raman scattering. Stokes and anti-stokes lines. Rotational Raman spectroscopy. Polarizability
Ultra-sensitive spectroscopy to detect single molecules is of
 The structural basis for giant enhancement enabling single-molecule Raman scattering Zhenjia Wang, Shanlin Pan, Todd D. Krauss, Hui Du, and Lewis J. Rothberg* Department of Chemistry, University of Rochester,
The structural basis for giant enhancement enabling single-molecule Raman scattering Zhenjia Wang, Shanlin Pan, Todd D. Krauss, Hui Du, and Lewis J. Rothberg* Department of Chemistry, University of Rochester,
Natallia Strekal. Plasmonic films of noble metals for nanophotonics
 Natallia Strekal Plasmonic films of noble metals for nanophotonics The aim of our investigation is the mechanisms of light interactions with nanostructure and High Tech application in the field of nanophotonics
Natallia Strekal Plasmonic films of noble metals for nanophotonics The aim of our investigation is the mechanisms of light interactions with nanostructure and High Tech application in the field of nanophotonics
Optics and Spectroscopy
 Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
Introduction to Optics and Spectroscopy beyond the diffraction limit Chi Chen 陳祺 Research Center for Applied Science, Academia Sinica 2015Apr09 1 Light and Optics 2 Light as Wave Application 3 Electromagnetic
- Supporting Information - Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles
 - Supporting Information - S1 Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles Tao Chen, Miaoxin Yang, Xinjiao Wang, Li Huey Tan, Hongyu Chen* Division of Chemistry and Biological Chemistry,
- Supporting Information - S1 Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles Tao Chen, Miaoxin Yang, Xinjiao Wang, Li Huey Tan, Hongyu Chen* Division of Chemistry and Biological Chemistry,
Vibrational Spectroscopy
 Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
Vibrational Spectroscopy In this part of the course we will look at the kind of spectroscopy which uses light to excite the motion of atoms. The forces required to move atoms are smaller than those required
Invited Paper ABSTRACT 1. INTRODUCTION
 Invited Paper Numerical Prediction of the Effect of Nanoscale Surface Roughness on Film-coupled Nanoparticle Plasmon Resonances Chatdanai Lumdee and Pieter G. Kik *,, CREOL, the College of Optics and Photonics;
Invited Paper Numerical Prediction of the Effect of Nanoscale Surface Roughness on Film-coupled Nanoparticle Plasmon Resonances Chatdanai Lumdee and Pieter G. Kik *,, CREOL, the College of Optics and Photonics;
6. Plasmon coupling between a flat gold interface and gold nanoparticles.
 6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
6. Plasmon coupling between a flat gold interface and gold nanoparticles. 6.1. Introduction In this outlook oriented chapter the applicability of the multilayered system used in chapter 4.1., for the study
Dichroic Optical Properties of Extended Nanostructures Fabricated Using Angle-Resolved Nanosphere Lithography
 Dichroic Optical Properties of Extended Nanostructures Fabricated Using Angle-Resolved Nanosphere Lithography NANO LETTERS 2003 Vol. 3, No. 7 939-943 Christy L. Haynes and Richard P. Van Duyne* Department
Dichroic Optical Properties of Extended Nanostructures Fabricated Using Angle-Resolved Nanosphere Lithography NANO LETTERS 2003 Vol. 3, No. 7 939-943 Christy L. Haynes and Richard P. Van Duyne* Department
PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES
 PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES Lucie ŠTOLCOVÁ a, Jan PROŠKA a, Filip NOVOTNÝ a, Marek PROCHÁZKA b, Ivan RICHTER a a Czech Technical University in Prague, Faculty of Nuclear
PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES Lucie ŠTOLCOVÁ a, Jan PROŠKA a, Filip NOVOTNÝ a, Marek PROCHÁZKA b, Ivan RICHTER a a Czech Technical University in Prague, Faculty of Nuclear
The inorganic chemistry of surface enhanced Raman scattering (SERS)
 The inorganic chemistry of surface enhanced Raman scattering (SERS) Karen Faulds, a Aaron Hernandez-Santana a and W. Ewen Smith a, b DOI: 10.1039/9781849730853-00001 1. Introduction Surface enhanced Raman
The inorganic chemistry of surface enhanced Raman scattering (SERS) Karen Faulds, a Aaron Hernandez-Santana a and W. Ewen Smith a, b DOI: 10.1039/9781849730853-00001 1. Introduction Surface enhanced Raman
IR Spectrography - Absorption. Raman Spectrography - Scattering. n 0 n M - Raman n 0 - Rayleigh
 RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
Toward a Thermally Robust Operando Surface-Enhanced Raman Spectroscopy Substrate
 J. Phys. Chem. C 2007, 111, 16827-16832 16827 Toward a Thermally Robust Operando Surface-Enhanced Raman Spectroscopy Substrate Alyson V. Whitney, Jeffrey W. Elam, Peter C. Stair,, and Richard P. Van Duyne*,
J. Phys. Chem. C 2007, 111, 16827-16832 16827 Toward a Thermally Robust Operando Surface-Enhanced Raman Spectroscopy Substrate Alyson V. Whitney, Jeffrey W. Elam, Peter C. Stair,, and Richard P. Van Duyne*,
ABSTRACT 1. INTRODUCTION
 Cascaded plasmon resonances multi-material nanoparticle trimers for extreme field enhancement S. Toroghi a, Chatdanai Lumdee a, and P. G. Kik* a CREOL, The College of Optics and Photonics, University of
Cascaded plasmon resonances multi-material nanoparticle trimers for extreme field enhancement S. Toroghi a, Chatdanai Lumdee a, and P. G. Kik* a CREOL, The College of Optics and Photonics, University of
Luminescent Blinking from Silver Nanostructures
 J. Phys. Chem. B 2003, 107, 9989-9993 9989 Luminescent Blinking from Silver Nanostructures Chris D. Geddes, Alex Parfenov, Ignacy Gryczynski, and Joseph R. Lakowicz*, Institute of Fluorescence and the
J. Phys. Chem. B 2003, 107, 9989-9993 9989 Luminescent Blinking from Silver Nanostructures Chris D. Geddes, Alex Parfenov, Ignacy Gryczynski, and Joseph R. Lakowicz*, Institute of Fluorescence and the
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Interaction between Single-walled Carbon Nanotubes and Water Molecules
 Workshop on Molecular Thermal Engineering Univ. of Tokyo 2013. 07. 05 Interaction between Single-walled Carbon Nanotubes and Water Molecules Shohei Chiashi Dept. of Mech. Eng., The Univ. of Tokyo, Japan
Workshop on Molecular Thermal Engineering Univ. of Tokyo 2013. 07. 05 Interaction between Single-walled Carbon Nanotubes and Water Molecules Shohei Chiashi Dept. of Mech. Eng., The Univ. of Tokyo, Japan
PCCP PAPER. Single-molecule surface-enhanced Raman spectroscopy with nanowatt excitation. 1 Introduction. 2 Experimental
 PAPER Cite this: Phys. Chem. Chem. Phys., 2014, 16, 23895 Single-molecule surface-enhanced Raman spectroscopy with nanowatt excitation Brendan L. Darby, Pablo G. Etchegoin and Eric C. Le Ru* Received 31st
PAPER Cite this: Phys. Chem. Chem. Phys., 2014, 16, 23895 Single-molecule surface-enhanced Raman spectroscopy with nanowatt excitation Brendan L. Darby, Pablo G. Etchegoin and Eric C. Le Ru* Received 31st
Multimodal multiplex Raman spectroscopy optimized for in vivo chemometrics
 Multimodal multiplex Raman spectroscopy optimized for in vivo chemometrics S. T. McCain, M. E. Gehm, Y. Wang, N. P. Pitsianis, and D. J. Brady Duke University Fitzpatrick Center for Photonics and Communication
Multimodal multiplex Raman spectroscopy optimized for in vivo chemometrics S. T. McCain, M. E. Gehm, Y. Wang, N. P. Pitsianis, and D. J. Brady Duke University Fitzpatrick Center for Photonics and Communication
Environmental modification of self-assembled plasmonic core-shell cluster (silica-gold nanoparticles) for surface enhanced Raman scattering (SERS)
 Environmental modification of self-assembled plasmonic core-shell cluster (silica-gold nanoparticles) for surface enhanced Raman scattering (SERS) Nadiah Ghazali 1,* and Mohd Rafie Johan 1,2 1 Nanomaterials
Environmental modification of self-assembled plasmonic core-shell cluster (silica-gold nanoparticles) for surface enhanced Raman scattering (SERS) Nadiah Ghazali 1,* and Mohd Rafie Johan 1,2 1 Nanomaterials
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Optical materials for surface enhanced Raman applications based on sol-gel encapsulated gold particles
 Optical materials for surface enhanced Raman applications based on sol-gel encapsulated gold particles Fatemeh Akbarian, Bruce S. Dunn and Jeffrey I. Zink Department of Chemistry and Biochemistry and Department
Optical materials for surface enhanced Raman applications based on sol-gel encapsulated gold particles Fatemeh Akbarian, Bruce S. Dunn and Jeffrey I. Zink Department of Chemistry and Biochemistry and Department
Characterisation of vibrational modes of adsorbed species
 17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
17.7.5 Characterisation of vibrational modes of adsorbed species Infrared spectroscopy (IR) See Ch.10. Infrared vibrational spectra originate in transitions between discrete vibrational energy levels of
In order to determine the energy level alignment of the interface between cobalt and
 SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
Surfactant-free exfoliation of graphite in aqueous solutions
 Surfactant-free exfoliation of graphite in aqueous solutions Karen B. Ricardo, Anne Sendecki, and Haitao Liu * Department of Chemistry, University of Pittsburgh, Pittsburgh, PA 15260, U.S.A 1. Materials
Surfactant-free exfoliation of graphite in aqueous solutions Karen B. Ricardo, Anne Sendecki, and Haitao Liu * Department of Chemistry, University of Pittsburgh, Pittsburgh, PA 15260, U.S.A 1. Materials
Supporting Information for. Shape Transformation of Gold Nanoplates and their Surface Plasmon. Characterization: Triangular to Hexagonal Nanoplates
 1 Supporting Information for Shape Transformation of Gold Nanoplates and their Surface Plasmon Characterization: Triangular to Hexagonal Nanoplates Soonchang Hong, Kevin L. Shuford *,, and Sungho Park
1 Supporting Information for Shape Transformation of Gold Nanoplates and their Surface Plasmon Characterization: Triangular to Hexagonal Nanoplates Soonchang Hong, Kevin L. Shuford *,, and Sungho Park
International Journal of Scientific & Engineering Research, Volume 5, Issue 3, March-2014 ISSN
 156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
156 Copper Nanoparticles: Green Synthesis Characterization Y.Suresh*1, S.Annapurna*2, G.Bhikshamaiah*3, A.K.Singh#4 Abstract Present work describes the synthesis nanoparticles using papaya extract as a
Molecular Interactions between Graphene and Biological Molecules
 Supporting Information: Molecular Interactions between Graphene and Biological Molecules Xinguan Zou, *,1 Shuai Wei, *,1 Joshua Jasensky, 1 Minyu Xiao, 1 Qiuming Wang, 1 Charles L. Brooks III **,1,2 and
Supporting Information: Molecular Interactions between Graphene and Biological Molecules Xinguan Zou, *,1 Shuai Wei, *,1 Joshua Jasensky, 1 Minyu Xiao, 1 Qiuming Wang, 1 Charles L. Brooks III **,1,2 and
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups
 Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
Infrared Spectroscopy An Instrumental Method for Detecting Functional Groups 1 The Electromagnetic Spectrum Infrared Spectroscopy I. Physics Review Frequency, υ (nu), is the number of wave cycles that
B.-Y. Lin et al., Opt. Express 17, (2009).
 !!!! The Ag nanoparticle array can be considered Ag nanorods arranged in hexagonal pattern with an inter-nanorod gap (W). The rod diameter (D) is 25 nm and the rod length (L) is 100 nm. A series of curved
!!!! The Ag nanoparticle array can be considered Ag nanorods arranged in hexagonal pattern with an inter-nanorod gap (W). The rod diameter (D) is 25 nm and the rod length (L) is 100 nm. A series of curved
The Origin of Relative Intensity Fluctuations in Single-Molecule Tip- Enhanced Raman Spectroscopy
 pubs.acs.org/jacs The Origin of Relative Intensity Fluctuations in Single-Molecule Tip- Enhanced Raman Spectroscopy Matthew. D. Sonntag, Dhabih Chulhai, Tamar Seideman, Lasse Jensen, and Richard P. Van
pubs.acs.org/jacs The Origin of Relative Intensity Fluctuations in Single-Molecule Tip- Enhanced Raman Spectroscopy Matthew. D. Sonntag, Dhabih Chulhai, Tamar Seideman, Lasse Jensen, and Richard P. Van
