SERS Analysis of CMC on Gold-Assembled Micelle
|
|
|
- Scott Doyle
- 5 years ago
- Views:
Transcription
1 1392 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 9 Nak Han Jang SERS Analysis of CMC on Gold-Assembled Micelle Nak Han Jang Institute of Science Education, Kongju National University, Kongju, Chungnam , Korea Received June 25, 2004 The micellization of dodecylpyridinum chloride (DPC) assembled on aqueous gold nanoparticles has been studied as a function of concentration using Surface-Enhanced Raman Scattering (SERS). At the low concentration, the strong SERS band of the benzene ring moiety was observed at 1025 cm 1, and assigned to trigonal ring breathing. According to high concentration of DPC, a new strong band was also appeared at 1012 cm 1, which was assigned to totally symmetry ring breathing. The difference of two spectra seems to ascribe to the geometry of polar head group, i.e., pyridinium cation. These geometry exist flat-down at low concentration, whereas standing-up or tilted geometry at high concentration. The critical micelle concentration (CMC) was first obtained from the ratio of intensities of the two bands related to the benzene ring moiety by vibrational spectroscopy, and was about 28 mm. After the CMC, the benzene ring moiety in the micelle state was more restricted than in monomer state because there is no more change of intensities at 1012 cm 1. In addition, the size of gold-assembled micelle was estimated using light scattering and it was about nm. Key Words : SERS, Cationic surfactant, CMC, Gold nanoparticles Introduction Surface-Enhanced Raman Scattering (SERS) is a very versatile technique to obtain the powerful information of characterizing the absorption and orientation of adsorbate on molecule-surface interactions. 1,2 The SERS is a process in which the Raman scattering intensity of molecules adsorbed on certain rough metal surface, (e.g., Ag, Au, Cu) is enhanced by factors of compared to the intensity expected for unadsored molecules of a comparable concentration. 3,4 This enormous sensitivity enhancement easily allowed adsorbates of a submonolayer coverage to be readily detected by Raman spectroscopy. Although not all molecules show enhancement at each interface, molecules which typically show enhancement at the solution-solid interface have unshared pairs of electrons, e.g. pyridine, piperidine and CN. Some important contradictions were attributed to a SERS spectrum for pyridinium ion which has unshared pair of electrons and the lack of a SERS spectrum of pyridine at low ph due to formation of pyridinium ion on silver surface. 4,5 However, because the pyridinium ion may not be able to adsorb on the surface itself, the SERS spectrum of pyridinium ion can be observed only when anion is present may be a consequence of greater enhancement due to anion. 6 The anion has a much greater affinity for the surface and is more polarizable. The anion also interacts strongly with the pyridinium cation. The affinity of anion for both the metal and the pyridinium ion may produce greater molecule-metal coupling. Thus, the enhancement may arise from a surface species of metalanion-pyridinium cation. Here, in cationic surfactant, dodecylpyridinum chloride (DPC), there is no unshared pair of electrons on the nitrogen, as there is in pyridine, and thus there is little interaction with the gold surface. However, because pyridinium cation also adsorb more strongly to Cl, the SERS spectrum is observed, indicating an enhanced coupling between the pyridinium cation and the gold surface via an adsorbed Cl complex. Cationic surfactants are amphiphilic molecules that contain a hydrophobic (nonpolar) alkyl chain, commonly called the tail, and a hydrophilic (polar) cation segment, called the head. This characteristic of being an amphiphilic molecule leads to aggregation. They reduce the surface tension by forming an extremely small aggregate that is called micelles. At low concentrations, surfactants are soluble in water, and increasing their concentration, the molecules of surfactants form micelles in solution. The smaller concentration at which micelles begin to form is called as the Critical Micelle Concentration (CMC). 7,8 Because micelles is consisted of polar head groups on each opposing surface with hydrophobic material in molecule, they are a form of a continuous bilayer of surfactant molecules. In other words, at or near the CMC micelles form in solution and bilayers form on surfaces. Until now, various methods are reported to determine the CMC of micelle on cationic surfactant using fluorescence, 9 electrical conductivity, 9 light scattering, 10 density, 11 viscosity, 11 ultrasonic relaxation, 12 and NMR 13 but is not reported any method to use vibrational spectroscopy. In this paper, the method to characterize the CMC of micellization is first reported on gold nanoparticles-assembled cationic surfactant using Raman spectroscopy and is elucidated the orientation of pyridinium moiety with a comparative analysis during micellization. In addition, the bulk size of gold-assembled micelle is proposed using the particle analyzer. * nhjang@kongju.ac.kr
2 SERS Analysis of CMC on Gold-Assembled Micelle Bull. Korean Chem. Soc. 2004, Vol. 25, No Experimental Section Materials: Dodecypyridinium chloride (DPC) was of analytical quality and was purchased from Aldrich Chemical Company and used without any more purification. All solutions were prepared from the stock solution. For all experiments, purified H 2 O (18.1 MΩ) was used and all glassware was cleaned in aqua regia (3 parts HCl, 1 parts HNO 3 ). Approximately 13 nm diameter gold particles were prepared by the citrate reduction of HAuCl 4 as described previously. 14 Stock solutions for DPC were prepared from 10 3 M to M concentration. The adsorbate was introduced in 1 ml gold nanoparticles by adding one drop (about ml) by drop of the above stock solutions, which resulted in a change of solution color from deep red to blue. A few drop of 3% poly(vinylpyrrolidone) (PVP, MW 10000) was added to the sample as a stabilizer, preventing further aggregation and eventual flocculation of the nanoparticle. The overall concentration of each DPC was from about M to M in surfactant/au sample. UV Spectra and Micelle Size: UV-Visible spectra were recorded on a Hewlett-Packard 8452A diode array spectrophotometer. A typical solution of 13 nm diameter gold particles exhibited a characteristic surface plasmon absorption maximum at 520 nm. After modifying with DPC, the aggregation of particles is also monitored by UV-Visible spectroscopy. Dynamic Light Scattering measurements were performed to characterize the size of gold-assembled micelle using a Brookhaven Instruments Corporation model BI- 9000AT digital autocorrelator and photon counter with a model BI-200SM goniometer. Incident light was provided by an argon ion laser (λ ex = nm) operating at 300 mw (Lexel Corp). Scattered light was collected at a fixed angle of 90. The DPC-linked gold nanoparticle aggregate samples were prepared as described above. The gold-assembled micelles were filtered through a 0.22 µm acetate membrane filter prior to mixing to remove any dust particles. Raman Spectroscopy: The Raman spectra were taken as described previously. 14 Raman spectrum of solid sample were achieved by using FT-Raman spectrometer (BioRad) equipped with 1064 nm Nd:YAG laser(spectra Physics). For the SERS spectra, the Ar + laser was used to pump a Spectra Physics Tsunami model locked Ti:Al 2O 3 laser to obtain λ ex = 710 nm. A band pass filter (Oriel Corporation, Stratford, CT) centered at 710 nm with a 10 nm range was utilized for removing extra lines. Spectra were recorded at λ ex = 710 nm using a SPEX Model 1877 Triplemate triple grating monochromator, and a SPEX Spectrum One charge-coupled device (CCD) detector. Figure 1. Schematic diagram of micellization on gold nanopariclebased DPC. gold surface and directly adsorb on the gold surface. Cl also interacts strongly with the pyridinium cation. Consequently, the affinity of Cl for both the gold and the pyridinium cation produce greater molecule-metal coupling to enhance the SERS effect and the DPC spectrum is observed, indicating an enhanced coupling between the pyridinium cation and the gold surface via an adsorbed Cl complex. After the DPC monolayer adsorbs to gold surface, the DPC-based gold nanoparticle is aggregating hugely, shifting the red to blue Results and Discussion The processes of adsorption on DPC and micellization on gold-based DPC were proposed in Figure 1. Basically, DPC is a cationic surfactant and it cannot directly adsorb to gold surface because pyridinium cation has no unshared pair of electrons. However, Cl has a much greater affinity for the Figure 2. Plots of UV-Visible absorption spectra of gold nanoparticles: (a) in the absence of DPC (the solid line), (b) 5 minutes after modifying with M DPC (dotted line), and (c) M DPC (the dash-dotted line).
3 1394 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 9 Nak Han Jang color. The UV-Visible absorption spectra were used to monitor the binding affinity of DPC to 13 nm diameter gold nanoparticles in Figure 2. The UV-Visible spectrum (spectrum a) of 13 nm diameter gold nanoparticles is shown for reference, which has a maximum at about 520 nm in Figure 2(a). Figures 2(b) and 2(c) show the changes in the plasmon band when DPC was added to a solution of 13 nm diameter gold nanoparicles, M and M DPC, respectively. To prevent further aggregation and eventual flocculation of the gold nanoparticles, the stabilizer was added in drop by drop and samples were waited for five minutes. At the low concentration (b) below CMC, all of DPC monolayer exhibited major changes in the maximum plasmon region at 615 nm. Furthermore, at a high concentration (c) over CMC, the gold-based DPC was micellized and showed major changes in the maximum plasmon region at 716 nm. This state of aggregation was stable existing further no more change for an hour. Figure 3 shows the Raman spectra of pyridine and DPC at several states. The Raman spectra of pyridine were introduced to compare with the main peak of DPC because it bears the pyridinium ring structure in the head chain. Assignments of Raman band frequencies for DPC were determined by comparing the frequencies of the observed Raman data with the vibrational data of references. 15,16 These tentatively assignments for SERS bands are listed in Table 1 for comparison the measured normal and FT-Raman frequencies in solution and in the solid. In Figure 3(g), the SERS band at 1012 cm 1 is due to the ring-breathing mode (Wilson mode 1 of benzene ring) of DPC adsorbed on gold nanoparticle surface, which is from mono-substituted benzene mode. This band is compared to the band (ring-breathing mode) at 998 cm 1 of pyridine adsorbed on gold nanoparticle surface in Figure 3(b). In Figure 3(e), this band is also not observed in the low DPC concentration. When DPC is dissolved at low concentrations, the molecules exist as individual entities. It is tentatively proposed a flat-on arrangement of DPC that the benzene ring moiety is parallel to the gold surface. That is, DPC is lying down on the surface because of the hydrophobic alkyl chain. Depending on the function of DPC concentration, this band is increased up to the maximum. This phenomenon can be supposed by the change of environment in pyridinium cation on the gold surface. At a high concentration of DPC, the pyridinium cation adsorbs on perpendicular to the gold surface, meaning that the benzene ring moiety is standing up to the gold Figure 3. Comparison of pyridine and DPC Raman spectra: (a) FT- Raman spectrum of pure pyridine. (b) SERS spectrum of M pyridine on gold nanoparticles. (c) FT-Raman spectrum of solid DPC. (d) M DPC in aqueous solution. (e) SERS spectrum of M. (f) M DPC (g) M DPC on gold nanoparticles. surface. Increasing the concentration of DPC, the hydrophobic effect is also increased by the alkyl chain and the micelle is formed to reduce the hydrophobicity. Consequently, DPC is restricted by gold nanoparticles and the benzene ring moiety of pyridinium cation has to stand up to the gold surface. The band at 1025 cm 1 corresponds to the trigonal ring-breathing mode (Wilson mode 12 of benzene ring) of DPC adsorbed on gold surface. This band is typically appearing at the ring of benzene derivatives. The position of this band is in a good agreement with the adsorption of neutral pyridine on gold nanoparticle surface and the result Table 1. Assignments of Raman spectra for DPC Pyridine DPC Liquid SERS Solid Aqueous SERS Tentative Assignment Ring Deformation Ring Vibration Ring Breathing Trigonal Ring Breathing C-C stretching Ring-C vibration
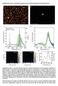
4 SERS Analysis of CMC on Gold-Assembled Micelle Bull. Korean Chem. Soc. 2004, Vol. 25, No Figure 5. The ratio of ring breathing band intensities (1012 cm 1 / 1025 cm 1 ) as a function of concentration. After the baseline is referred near 950 cm 1, each of intensity is subtracted by the baseline. Figure 4. SERS spectra of DPC as a function of concentration: (a) M (b) M (c) M (d) M (e) M (f) M (g) M (h) M (i) M. All spectra were normalized by the reference (a). For clarity of presentation, spectra were offset, respectively. reported in previous SERS work. 17 As the concentration of the DPC increases the molecules tend to associate to form aggregates. In aqueous solutions, the hydrophobic tails of the DPC associate, leaving the (hydrophilic) headgroups exposed to the solvent. Therefore, the environment of benzene ring moiety is different from the function of DPC concentration. The SERS spectra of benzene ring moiety are displayed as the function of DPC concentration in Figure 4. As is mentioned before, there is no band at 1012 cm 1 at very low DPC concentration because the benzene ring moiety is parallel to the surface. However, this band increases up because the benzene ring moiety is standing up to the surface as the concentration of DPC increases. After the CMC, the intensities of this band do not change although the concentration increases further. This reason is why once the micelles are formed, further increase of the total DPC concentration does not change the concentration of gold-assembled molecules on micelle. The DPC added is completely incorporated into the micelles without any assembly with gold nanoparticles. In other words, the concentration of the gold-assembled DPC molecules remains constant after the micelles are formed. After the CMC, the intensities of band at 1025 cm 1 are increased higher than that of the band at 1012 cm 1. Because the ratio of relative intensity changes between 1012 cm 1 and 1025 cm 1 related to the benzene ring moiety of DPC, the transition from a monomeric solution to a micelle form can be seen as a change in the slope of plots against DPC concentration of the ratio of relative intensity related to the benzene ring moiety. Here the new method was proposed to determine the CMC of gold assembled micelle using SERS. Figure 5 was plotted against the reciprocal concentration of DPC to the ratio of relative intensities between 1012 cm 1 and 1025 cm 1. The slope is changing before and after the CMC. Therefore, the CMC is determined at the maximum which shows the inversion of sign in slope. In the result, the CMC was about 28 mm on gold-assembled DPC micelle. This value is pretty larger than the CMC (17 mm) of pure DPC micelle reported 11,18 because DPC is assembled with the 13 nm gold nanoparticle. This study first shows the possibility of determination of CMC by vibrational spectroscopy. In addition, this method is valuable for the other CMC which is very small because SERS is very sensitive. Finally, the size of gold-assembled micelle was estimated using light scattering because typically the micelles have a closely spherical shape in a rather wide concentration range above the CMC (Figure 6). The size of gold-assembled micelle was about nm from multimodal size distribution during the average of four runs. This size is pretty huge compared with the size (50 nm) estimated from the general alkyl chain micelle. 7 This reason can be considered as the aggregation of gold particle and the repulsion of positive charge because Cl is first tethered to the gold surface to adsorb the pyridinium cation. In formation of the pure micelle, the small amount of added NaCl decreases the CMC of DPC. 18 This is due to the decrease in the electrical repulsion between the positively charged head groups in the micelle in the electrolyte solution. The effect of added NaCl on the CMC is much pronounced for the high hydrophobicity. Here the concentration of Cl reduces in goldbased DPC because chloride anion has a much greater
5 1396 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 9 Nak Han Jang bands (1012 cm 1 /1025 cm 1 ) related to the benzene ring moiety by vibrational spectroscopy, and was about 28 mm. After the CMC, the benzene ring moiety in the micelle state was more restricted than in monomer state because there is no more change of intensities at 1012 cm 1. In addition, the size of gold-assembled micelle was determined using light scattering and it was about nm. The large size of goldassembled micelle is due to the decrease in the electrical repulsion between the positively charged head groups in the micelle Acknowledgment. Author thanks for the partially financial support of Korean Research Foundation (KRF C00033) and also expresses his deepest appreciation to Professor Mirkin at Northwestern University. References Figure 6. Multimodal size distribution of DPC-assembled micelle measured by light scattering: (a) Before CMC (b) After CMC. Effective Diameter: nm, Polydispersity: 0.121, Sample Quality: 7.3. affinity for the gold surface and the hydrophobicity also reduces because the gold-based DPC is increasing the hydrophilicity. Moreover, the repulsion of positively charged head groups increases as the concentration of DPC increases. Consequently, more aggregation number need to form the micelle and this induces to increase the size of goldassembled micelle. Conclusions Good-quality SERS spectra of DPC were first acquired on gold nanoparticles and the CMC of gold-assembled micelle was determined although it is well-known to difficulty of the cationic surfactant on SERS because it cannot adsorb to the surface. Because the Cl has a much greater affinity for the gold surface and it also interacts strongly with pyridinium cation, it would be observed the SERS spectra of DPC on gold nanoparticles. At the low DPC concentration, the SERS band at 1025 cm 1 was only predominant, which was related to the benzene ring moiety, assigned to trigonal ring breathing. Increasing the concentration of DPC, a new strong band was strongly appeared at 1012 cm 1, which was assigned to totally symmetry ring breathing. This difference seems to ascribe to the geometry of polar head group, i.e., pyridinium cation. These geometry exist flat-down at low concentration, whereas standing-up or tilted geometry at high concentration. The critical micelle concentration (CMC) was first obtained from the ratio of intensities of the two 1. (a) Koglin, E.; Séquaris, J. M.; Valenta, P. J. Mol. Struct. 1980, 60, (b) Ervin, K. M.; Koglin, E.; Séquaris, J. M.; Valenta, P.; Nürnberg, H. W. J. Electroanal. Chem. 1980, 114, (c) Creighton, J. A. Surf. Sci. 1985, 158, (a) Angebranndt, M. J.; Winefordner, J. D. Talanta 1992, 39, (b) Jordan, C. E.; Frutos, A. G.; Thiel, A. J.; Corn, R. M. Anal. Chem. 1997, 69, (c) Angel, S. M.; Katz, L. F.; Archibald, D. D.; Honigs, D. E. Appl. Spectrosc. 1989, 43, (d) Pate, J.; Leiden, A.; Bozlee, B. J.; Garrell, R. L. J. Raman Spectrosc. 1991, 22, (e) Szafranski, C. A.; Tanner, W.; Laibinis, P. E.; Garrell, R. L. Langmuir 1998, 14, Fleischmann, M.; Hendra, P. J.; McQuillan, A. Chem. Phys. Lett. 1974, 26, Jeanmarie, D. L.; Van Duyne, R. P. J. Electroanal. Chem. 1977, 84, Regis, A.; Corset, J. Chem. Phys. Lett. 1980, 70, Bunding, K. A.; Bell, M. I.; Durst, R. A. Chem. Phys. Lett. 1982, 89, (a) Tnaford, C. The Hydrophobic Effect: Formation of Micelles and Biological Membranes, 2nd Ed.; John Wiley & Sons: New York, (b) Fendler, J. H. Membrane-Mimetic Approach to Advanced Materials; Springer-Verlag: Berlin, Voutsas, E. C.; Flores, M. V.; Spiliotis, N.; Bell, G.; Halling, P. J.; Tassios, D. P. Ind. Eng. Chem. Res. 2001, 40, Jang, N, H, M. Ed. Thesis, Seoul National University: Seoul, Korea, Chung, M. I.; Tak, I. J.; Lee, K. M. J. Korean Chem. Soc. 1975, 19, (a) Youn, Y. W.; Lee, K. M. J. Korean Chem. Soc. 1975, 19, (b) Lee, K. M. J. Korean Chem. Soc. 1976, 20, Lee, K. M. J. Korean Chem. Soc. 1973, 17, (a) Lee, Y. S.; Woo, K. W. Bull. Korean Chem. Soc. 1993, 14, (b) Lee, Y. S.; Woo, K. W. Bull. Korean Chem. Soc. 1993, 14, (c) Lee, Y. S.; Woo, K. W. J. Colloid Interface Sci. 1995, 169, Jang, N. H. Bull. Korean Chem. Soc. 2002, 23, Varsanyi, G. Assignments for Vibrational Spectra of Seven Hundred Benzene Derivatives; John Wiley & Sons: New York, 1974, and reference therein. 16. Lin-Vien, D.; Colthup, N. B.; Fately, W. G.; Grasselli, J. G. The Handbook of Infrared and Raman Characteristic Frequencies of Organic Molecules; Academic Press: London, 1991, and reference therein. 17. Brolo, A. G.; Irish, D. E.; Lipkowski, J. J. Phys. Chem. B 1997, 101, Simoncc i, B.; Špan, J. Acta Chim. Slov. 1998, 45,
HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS
 www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
www.arpapress.com/volumes/vol19issue1/ijrras_19_1_06.pdf HYPER-RAYLEIGH SCATTERING AND SURFACE-ENHANCED RAMAN SCATTERING STUDIES OF PLATINUM NANOPARTICLE SUSPENSIONS M. Eslamifar Physics Department, BehbahanKhatamAl-Anbia
Supplementary Information for. Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings
 Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Supplementary Information for Vibrational Spectroscopy at Electrolyte Electrode Interfaces with Graphene Gratings Supplementary Figure 1. Simulated from pristine graphene gratings at different Fermi energy
Critical Micellization Concentration Determination using Surface Tension Phenomenon
 Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Critical Micellization Concentration Determination using Phenomenon 1. Introduction Surface-active agents (surfactants) were already known in ancient times, when their properties were used in everyday
Supporting Information for. Chad A. Mirkin* Department of Chemistry and Institute for Nanotechnology, Northwestern University,
 S1 Supporting Information for Observation of a Quadrupole Plasmon Mode for a Colloidal Solution of Gold Nanoprisms Jill E. Millstone, Sungho Park, Kevin L. Shuford, Lidong Qin, George C. Schatz, and Chad
S1 Supporting Information for Observation of a Quadrupole Plasmon Mode for a Colloidal Solution of Gold Nanoprisms Jill E. Millstone, Sungho Park, Kevin L. Shuford, Lidong Qin, George C. Schatz, and Chad
Surface-Enhanced Raman Spectroscopy of Peptides Adsorbed on Silver and Gold Nanoparticles in Aqueous Solutions
 Surface-Enhanced Raman Spectroscopy of Peptides Adsorbed on Silver and Gold Nanoparticles in Aqueous Solutions Stephanie L. Stovall Linfield College Chemistry Department Abstract Distinguished from standard
Surface-Enhanced Raman Spectroscopy of Peptides Adsorbed on Silver and Gold Nanoparticles in Aqueous Solutions Stephanie L. Stovall Linfield College Chemistry Department Abstract Distinguished from standard
Aqueous Self-Assembly of Fp Derivatives: Multifunctional Metal-Carbonyl Nanovesicles (MCsomes)
 Aqueous Self-Assembly of Fp Derivatives: Multifunctional Metal-Carbonyl Nanovesicles (MCsomes) 38 th IPR Symposium-May 4 th 2016 Nimer Murshid and Xiaosong Wang Nimer Murshid Department of Chemistry Waterloo
Aqueous Self-Assembly of Fp Derivatives: Multifunctional Metal-Carbonyl Nanovesicles (MCsomes) 38 th IPR Symposium-May 4 th 2016 Nimer Murshid and Xiaosong Wang Nimer Murshid Department of Chemistry Waterloo
Interaction of Gold Nanoparticle with Proteins
 Chapter 7 Interaction of Gold Nanoparticle with Proteins 7.1. Introduction The interfacing of nanoparticle with biomolecules such as protein is useful for applications ranging from nano-biotechnology (molecular
Chapter 7 Interaction of Gold Nanoparticle with Proteins 7.1. Introduction The interfacing of nanoparticle with biomolecules such as protein is useful for applications ranging from nano-biotechnology (molecular
Probing the Kinetics of Ligand Exchange on Colloidal Gold. Nanoparticles by Surface-Enhanced Raman Scattering
 -Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
-Supporting Information- Probing the Kinetics of Ligand Exchange on Colloidal Gold Nanoparticles by Surface-Enhanced Raman Scattering Yuhua Feng, Shuangxi Xing, Jun Xu, Hong Wang, Jun Wei Lim, and Hongyu
Colloidal dispersion
 Dispersed Systems Dispersed systems consist of particulate matter, known as the dispersed phase, distributed throughout a continuous or dispersion medium. The dispersed material may range in size from
Dispersed Systems Dispersed systems consist of particulate matter, known as the dispersed phase, distributed throughout a continuous or dispersion medium. The dispersed material may range in size from
Identifying molecules at very low. Applications of Reproducible SERS Substrates for Trace Level Detection
 8 Raman Technology for Today s Spectroscopists June 2006 Applications of Reproducible SERS Substrates for Trace Level Detection Recent progress in photonic crystal design is transforming surface-enhanced
8 Raman Technology for Today s Spectroscopists June 2006 Applications of Reproducible SERS Substrates for Trace Level Detection Recent progress in photonic crystal design is transforming surface-enhanced
Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation
 Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supplementary Information Localized and Propagating Surface Plasmon Co-Enhanced Raman Spectroscopy Based on Evanescent Field Excitation Yu Liu, Shuping Xu, Haibo Li, Xiaoguang Jian, Weiqing Xu* State Key
Supporting Information
 Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Supporting Information Highly Sensitive, Reproducible, and Stable SERS Sensors Based on Well-Controlled Silver Nanoparticles Decorated Silicon Nanowire Building Blocks Xue Mei Han, Hui Wang, Xue Mei Ou,
Module 4: "Surface Thermodynamics" Lecture 22: "" The Lecture Contains: Examples on Effect of surfactant on interfacial tension. Objectives_template
 The Lecture Contains: Examples on Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture22/22_1.htm[6/16/2012 1:10:07 PM] Example Consider liquid, its vapors and
The Lecture Contains: Examples on Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture22/22_1.htm[6/16/2012 1:10:07 PM] Example Consider liquid, its vapors and
"Surface-Enhanced Raman Scattering
 SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
SMR: 1643/11 WINTER COLLEGE ON OPTICS ON OPTICS AND PHOTONICS IN NANOSCIENCE AND NANOTECHNOLOGY ( 7-18 February 2005) "Surface-Enhanced Raman Scattering presented by: Martin Moskovits University of California,
Supporting Information
 Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental Methods Instrumentation Zeta potentials and sizes of colloidal
Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Experimental Methods Instrumentation Zeta potentials and sizes of colloidal
SUPPORTING INFORMATION. A New Approach for the Surface Enhanced Resonance Raman Scattering (SERRS)
 SUPPORTING INFORMATION A New Approach for the Surface Enhanced Resonance Raman Scattering (SERRS) Detection of Dopamine at Picomolar (pm) Levels in the Presence of Ascorbic Acid Murat Kaya, Mürvet Volkan
SUPPORTING INFORMATION A New Approach for the Surface Enhanced Resonance Raman Scattering (SERRS) Detection of Dopamine at Picomolar (pm) Levels in the Presence of Ascorbic Acid Murat Kaya, Mürvet Volkan
Supporting Information
 Supporting Information Wiley-VCH 2011 69451 Weinheim, Germany Silver Nanocrystals with Concave Surfaces and Their Optical and Surface-Enhanced Raman Scattering Properties** Xiaohu Xia, Jie Zeng, Brenden
Supporting Information Wiley-VCH 2011 69451 Weinheim, Germany Silver Nanocrystals with Concave Surfaces and Their Optical and Surface-Enhanced Raman Scattering Properties** Xiaohu Xia, Jie Zeng, Brenden
Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters
 Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Acta Physica Sinica (Overseas Edition) Surface-enhanced Raman scattering study on non-activity of Ag-Cl clusters To cite this article: Fang Yan and Li Qin 1997 Acta Phys. Sin. (Overseas Edn) 6 183 View
Analysis on the birefringence property of lyotropic liquid crystals below Krafft temperature
 Analysis on the birefringence property of lyotropic liquid crystals below Krafft temperature Radhakrishnan Ranjini, Murukeshan Vadakke Matham *, Nam-Trung Nguyen Department of Mechanical and Aerospace
Analysis on the birefringence property of lyotropic liquid crystals below Krafft temperature Radhakrishnan Ranjini, Murukeshan Vadakke Matham *, Nam-Trung Nguyen Department of Mechanical and Aerospace
often display a deep green color due to where the SPR occurs (i.e., the wavelength of light that interacts with this specific morphology).
 Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
Synthesis-Dependent Catalytic Properties of Gold Nanoparticles Nanoscience is the study of materials that have dimensions, intuitively, on the nanoscale, typically between 1 100 nm. This field has received
Formation of complex films with water-soluble CTAB molecules
 Formation of complex films with water-soluble CTAB molecules S. Biswas a, b, S. A. Hussain a, S. Deb a, R. K. Nath b, D. Bhattacharjee a Department of Physics a and Department of chemistry b, Tripura University,
Formation of complex films with water-soluble CTAB molecules S. Biswas a, b, S. A. Hussain a, S. Deb a, R. K. Nath b, D. Bhattacharjee a Department of Physics a and Department of chemistry b, Tripura University,
Hierarchical Host-Guest Assemblies Formed on Dodecaborate-Coated Gold Nanoparticles
 Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 17 Hierarchical Host-Guest Assemblies Formed on Dodecaborate-Coated Gold Nanoparticles
Electronic Supplementary Material (ESI) for Chemical Communications. This journal is The Royal Society of Chemistry 17 Hierarchical Host-Guest Assemblies Formed on Dodecaborate-Coated Gold Nanoparticles
Formation of complex films with water-soluble CTAB molecules
 Spectrochimica Acta Part A 65 (2006) 628 632 Formation of complex films with water-soluble CTAB molecules S. Biswas a,b, S.A. Hussain a,s.deb a, R.K. Nath b, D. Bhattacharjee a, a Department of Physics,
Spectrochimica Acta Part A 65 (2006) 628 632 Formation of complex films with water-soluble CTAB molecules S. Biswas a,b, S.A. Hussain a,s.deb a, R.K. Nath b, D. Bhattacharjee a, a Department of Physics,
Bottom-up Optimization of SERS Hot Spots. Supplementary Information
 Bottom-up Optimization of SERS Hot Spots Laura Fabris, * Department of Materials Science and Engineering, Institute for Advanced Materials Devices ad Nanotechnology, Rutgers, The State University of New
Bottom-up Optimization of SERS Hot Spots Laura Fabris, * Department of Materials Science and Engineering, Institute for Advanced Materials Devices ad Nanotechnology, Rutgers, The State University of New
Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering
 Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Supporting Information Cyclic Electroplating and Stripping of Silver on Au@SiO 2 Core/Shell Nanoparticles for Sensitive and Recyclable Substrates of Surface-enhanced Raman Scattering Dan Li a, Da-Wei Li
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering
 Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Nanostructured substrate with nanoparticles fabricated by femtosecond laser for surface-enhanced Raman scattering Yukun Han, 1 Hai Xiao, 2 and Hai-Lung Tsai 1, * 1 Department of Mechanical and Aerospace
Vibrational Spectroscopies. C-874 University of Delaware
 Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Vibrational Spectroscopies C-874 University of Delaware Vibrational Spectroscopies..everything that living things do can be understood in terms of the jigglings and wigglings of atoms.. R. P. Feymann Vibrational
Monolayers. Factors affecting the adsorption from solution. Adsorption of amphiphilic molecules on solid support
 Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Monolayers Adsorption as process Adsorption of gases on solids Adsorption of solutions on solids Factors affecting the adsorption from solution Adsorption of amphiphilic molecules on solid support Adsorption
Module 4: "Surface Thermodynamics" Lecture 21: "" The Lecture Contains: Effect of surfactant on interfacial tension. Objectives_template
 The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
The Lecture Contains: Effect of surfactant on interfacial tension file:///e /courses/colloid_interface_science/lecture21/21_1.htm[6/16/2012 1:10:36 PM] Surface Thermodynamics: Roles of Surfactants and
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2019 Supporting Information for Halide-assisted activation of atomic hydrogen for photoreduction on
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2019 Supporting Information for Halide-assisted activation of atomic hydrogen for photoreduction on
Lecture 3. Phenomena at Liquid-gas and Liquid-Liquid interfaces. I
 Lecture 3 Phenomena at Liquid-gas and Liquid-Liquid interfaces. I Adsorption at Gas-Liquid interface Measurements of equilibrium adsorption surface tension measurements (Wilhelmy plate) surface analysis
Lecture 3 Phenomena at Liquid-gas and Liquid-Liquid interfaces. I Adsorption at Gas-Liquid interface Measurements of equilibrium adsorption surface tension measurements (Wilhelmy plate) surface analysis
Surface-enhanced raman scattering from a layer of gold nanoparticles
 VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
VNU Journal of Science, Mathematics - Physics 26 (2010) 187-192 Surface-enhanced raman scattering from a layer of gold nanoparticles Nguyen The Binh *, Nguyen Thanh Dinh, Nguyen Quang Dong, Vu Thi Khanh
Nicolae Leopold, and Bernhard Lendl*,
 J. Phys. Chem. B 2003, 107, 5723-5727 5723 A New Method for Fast Preparation of Highly Surface-Enhanced Raman Scattering (SERS) Active Silver Colloids at Room Temperature by Reduction of Silver Nitrate
J. Phys. Chem. B 2003, 107, 5723-5727 5723 A New Method for Fast Preparation of Highly Surface-Enhanced Raman Scattering (SERS) Active Silver Colloids at Room Temperature by Reduction of Silver Nitrate
We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions.
 CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
Raman spectroscopy of phthalocyanines and their sulfonated derivatives
 Journal of Molecular Structure 744 747 (2005) 481 485 www.elsevier.com/locate/molstruc Raman spectroscopy of phthalocyanines and their sulfonated derivatives B. Brożek-Płuska*, I. Szymczyk, H. Abramczyk
Journal of Molecular Structure 744 747 (2005) 481 485 www.elsevier.com/locate/molstruc Raman spectroscopy of phthalocyanines and their sulfonated derivatives B. Brożek-Płuska*, I. Szymczyk, H. Abramczyk
Synthesis of ternary chalcogenide colloidal nanocrystals in aqueous medium
 Journal of Physics: Conference Series PAPER OPEN ACCESS Synthesis of ternary chalcogenide colloidal nanocrystals in aqueous medium To cite this article: D S Mazing et al 28 J. Phys.: Conf. Ser. 38 25 View
Journal of Physics: Conference Series PAPER OPEN ACCESS Synthesis of ternary chalcogenide colloidal nanocrystals in aqueous medium To cite this article: D S Mazing et al 28 J. Phys.: Conf. Ser. 38 25 View
6 Hydrophobic interactions
 The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
Supplemental Information for
 Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Supplemental Information for Densely arranged two-dimensional silver nanoparticle assemblies with optical uniformity over vast areas as excellent surface-enhanced Raman scattering substrates Yoshimasa
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries
 Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Supplementary Note 1: Dark field measurements and Scattering properties of NPoM geometries Supplementary Figure 1: Dark field scattering properties of individual nanoparticle on mirror geometries separated
Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly
 SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
Electronic supplementary information for:
 Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
Electronic supplementary information for: Charge-transfer-induced suppression of galvanic replacement and synthesis of (Au@Ag)@Au double shell nanoparticles for highly uniform, robust and sensitive bioprobes
- Supporting Information - Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles
 - Supporting Information - S1 Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles Tao Chen, Miaoxin Yang, Xinjiao Wang, Li Huey Tan, Hongyu Chen* Division of Chemistry and Biological Chemistry,
- Supporting Information - S1 Controlled Assembly of Eccentrically Encapsulated Gold Nanoparticles Tao Chen, Miaoxin Yang, Xinjiao Wang, Li Huey Tan, Hongyu Chen* Division of Chemistry and Biological Chemistry,
Metal-Catalyzed Chemical Reaction of. Single Molecules Directly Probed by. Vibrational Spectroscopy
 Supporting Information to: Metal-Catalyzed Chemical Reaction of Single Molecules Directly Probed by Vibrational Spectroscopy Han-Kyu Choi, Won-Hwa Park, Chan Gyu Park, Hyun-Hang Shin, Kang Sup Lee and
Supporting Information to: Metal-Catalyzed Chemical Reaction of Single Molecules Directly Probed by Vibrational Spectroscopy Han-Kyu Choi, Won-Hwa Park, Chan Gyu Park, Hyun-Hang Shin, Kang Sup Lee and
Supporting information
 Supporting information Polymer-Single-Crystal@Nanoparticle Nanosandwich for Surface Enhanced Raman Spectroscopy Bin Dong, Wenda Wang, David L. Miller, Christopher Y. Li* Department of Material Science
Supporting information Polymer-Single-Crystal@Nanoparticle Nanosandwich for Surface Enhanced Raman Spectroscopy Bin Dong, Wenda Wang, David L. Miller, Christopher Y. Li* Department of Material Science
Surface-Enhanced Raman Spectroscopy
 GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
GENERAL ARTICLE Surface-Enhanced Raman Spectroscopy Recent Advancement of Raman Spectroscopy Ujjal Kumar Sur Ujjal Kumar Sur works in the Netaji Nagar Day College, Kolkata, West Bengal. He is also a visiting
Thermodynamics of Micellization of Nonionic Surfactant Tween-40 in Presence of Additive Chloramine-T Using Clouding Phenomenon
 http://www.e-journals.net ISSN: 973-4945; CDEN ECJHA E- Chemistry 21, 7(S1), S33-S334 Thermodynamics of Micellization of Nonionic Surfactant Tween-4 in Presence of Additive Chloramine-T Using Clouding
http://www.e-journals.net ISSN: 973-4945; CDEN ECJHA E- Chemistry 21, 7(S1), S33-S334 Thermodynamics of Micellization of Nonionic Surfactant Tween-4 in Presence of Additive Chloramine-T Using Clouding
Electrochemical SERS at a structured gold surface
 Electrochemistry Communications 7 (2005) 740 744 www.elsevier.com/locate/elecom Electrochemical SERS at a structured gold surface Mamdouh E. Abdelsalam a, Philip N. Bartlett a, *, Jeremy J. Baumberg b,
Electrochemistry Communications 7 (2005) 740 744 www.elsevier.com/locate/elecom Electrochemical SERS at a structured gold surface Mamdouh E. Abdelsalam a, Philip N. Bartlett a, *, Jeremy J. Baumberg b,
Colorimetric and Fluorescent Dual Detection of Paraquat and Diquat Based on a Novel Anionic Polythiophene Derivative
 This journal is The Royal Society of Chemistry 213 Supplementary information for: Colorimetric and Fluorescent Dual Detection of Paraquat and Diquat Based on a Novel Anionic Polythiophene Derivative Zhiyi
This journal is The Royal Society of Chemistry 213 Supplementary information for: Colorimetric and Fluorescent Dual Detection of Paraquat and Diquat Based on a Novel Anionic Polythiophene Derivative Zhiyi
Application Note: Ocean Optics in the Teaching and Research Laboratories Susquehanna University Department of Chemistry July 2014
 Application Note: Ocean Optics in the Teaching and Research Laboratories Susquehanna University Department of Chemistry July 2014 Authors: Dr. Swarna Basu (Associate Professor of Chemistry), Dr. Wade Johnson
Application Note: Ocean Optics in the Teaching and Research Laboratories Susquehanna University Department of Chemistry July 2014 Authors: Dr. Swarna Basu (Associate Professor of Chemistry), Dr. Wade Johnson
Supporting Information
 Block Copolymer Mimetic Self-Assembly of Inorganic Nanoparticles Yunyong Guo, Saman Harirchian-Saei, Celly M. S. Izumi and Matthew G. Moffitt* Department of Chemistry, University of Victoria, P.O. Box
Block Copolymer Mimetic Self-Assembly of Inorganic Nanoparticles Yunyong Guo, Saman Harirchian-Saei, Celly M. S. Izumi and Matthew G. Moffitt* Department of Chemistry, University of Victoria, P.O. Box
What happens when light falls on a material? Transmission Reflection Absorption Luminescence. Elastic Scattering Inelastic Scattering
 Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Raman Spectroscopy What happens when light falls on a material? Transmission Reflection Absorption Luminescence Elastic Scattering Inelastic Scattering Raman, Fluorescence and IR Scattering Absorption
Resonance Raman measurements utilizing a deep UV source
 Resonance Raman measurements utilizing a deep UV source Adam Willitsford a, C. Todd Chadwick b, Hans Hallen b, and C. Russell Philbrick a a The Pennsylvania State University, Department of Electrical Engineering,
Resonance Raman measurements utilizing a deep UV source Adam Willitsford a, C. Todd Chadwick b, Hans Hallen b, and C. Russell Philbrick a a The Pennsylvania State University, Department of Electrical Engineering,
Tetrathiafulvalene radical cation (TTF +. ) Charge Transfer aggregates included in PMMA matrix, a Resonance Raman Study
 Tetrathiafulvalene radical cation (TTF +. ) Charge Transfer aggregates included in PMMA matrix, a Resonance Raman Study B. F. Scremin 1 1-IOM CNR, Institute Officina dei Materiali of the National Research
Tetrathiafulvalene radical cation (TTF +. ) Charge Transfer aggregates included in PMMA matrix, a Resonance Raman Study B. F. Scremin 1 1-IOM CNR, Institute Officina dei Materiali of the National Research
Shell-isolated nanoparticle-enhanced Raman spectroscopy
 Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
Shell-isolated nanoparticle-enhanced Raman spectroscopy Jian Feng Li, Yi Fan Huang, Yong Ding, Zhi Lin Yang, Song Bo Li, Xiao Shun Zhou, Feng Ru Fan, Wei Zhang, Zhi You Zhou, De Yin Wu, Bin Ren, Zhong
I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically?
 Name I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically? Internal standards are standards added intentionally to all samples, standards and blanks.
Name I. Proteomics by Mass Spectrometry 1. What is an internal standard and what does it accomplish analytically? Internal standards are standards added intentionally to all samples, standards and blanks.
Optical properties of spherical and anisotropic gold shell colloids
 8 Optical properties of spherical and anisotropic gold shell colloids Core/shell colloids consisting of a metal shell and a dielectric core are known for their special optical properties. The surface plasmon
8 Optical properties of spherical and anisotropic gold shell colloids Core/shell colloids consisting of a metal shell and a dielectric core are known for their special optical properties. The surface plasmon
A NEW SIGHT ON HYDROPHOBIC BEHAVIOUR OF OXYETHYLENE GROUPS LOCATED BETWEEN ALKYL CHAIN AND IONIC GROUP IN CATIONIC SURFACTANTS
 A NEW SIGHT ON HYDROPHOBIC BEHAVIOUR OF OXYETHYLENE GROUPS LOCATED BETWEEN ALKYL CHAIN AND IONIC GROUP IN CATIONIC SURFACTANTS I. Mândru, Mihaela Olteanu, Otilia Cintez and Manuela Dud u abstract: The
A NEW SIGHT ON HYDROPHOBIC BEHAVIOUR OF OXYETHYLENE GROUPS LOCATED BETWEEN ALKYL CHAIN AND IONIC GROUP IN CATIONIC SURFACTANTS I. Mândru, Mihaela Olteanu, Otilia Cintez and Manuela Dud u abstract: The
Permeable Silica Shell through Surface-Protected Etching
 Permeable Silica Shell through Surface-Protected Etching Qiao Zhang, Tierui Zhang, Jianping Ge, Yadong Yin* University of California, Department of Chemistry, Riverside, California 92521 Experimental Chemicals:
Permeable Silica Shell through Surface-Protected Etching Qiao Zhang, Tierui Zhang, Jianping Ge, Yadong Yin* University of California, Department of Chemistry, Riverside, California 92521 Experimental Chemicals:
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using
 Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
IR Spectrography - Absorption. Raman Spectrography - Scattering. n 0 n M - Raman n 0 - Rayleigh
 RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
RAMAN SPECTROSCOPY Scattering Mid-IR and NIR require absorption of radiation from a ground level to an excited state, requires matching of radiation from source with difference in energy states. Raman
Digitized single scattering nanoparticles for probing molecular binding
 Electronic Supplementary Information (ESI) Digitized single scattering nanoparticles for probing molecular binding Yue Liu a, Cheng Zhi Huang a,b* a Education Ministry Key Laboratory on Luminescence and
Electronic Supplementary Information (ESI) Digitized single scattering nanoparticles for probing molecular binding Yue Liu a, Cheng Zhi Huang a,b* a Education Ministry Key Laboratory on Luminescence and
Dispersion-Flocculation Behavior of Fine Lead Particles in an Organic Solvent
 Materials Transactions, Vol. 49, No. 9 (2) pp. 2119 to 2123 #2 The Mining and Materials Processing Institute of Japan Dispersion-Flocculation Behavior of Fine Lead Particles in an Organic Solvent Masami
Materials Transactions, Vol. 49, No. 9 (2) pp. 2119 to 2123 #2 The Mining and Materials Processing Institute of Japan Dispersion-Flocculation Behavior of Fine Lead Particles in an Organic Solvent Masami
Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol
 Supporting Information Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol Sho Suzuki, Satoshi Kaneko*, Shintaro Fujii, Santiago Marqués-González, Tomoaki
Supporting Information Effect of the Molecule-Metal Interface on the Surface Enhanced Raman Scattering of 1,4-Benzenedithiol Sho Suzuki, Satoshi Kaneko*, Shintaro Fujii, Santiago Marqués-González, Tomoaki
The Student Seminar Series. Jinseok Heo. Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry
 Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Abstract The Student Seminar Series Presents a seminar by Jinseok Heo Department of Chemistry Texas A&M University Recent Applications of Surface Enhanced Raman Scattering in Analytical Chemistry 4:00
Solvent Isotopic Effects on a Surfactant Headgroup at the Air-Liquid Interface
 S1 SUPPORTING INFORMATION Solvent Isotopic Effects on a Surfactant Headgroup at the Air-Liquid Interface Uvinduni I. Premadasa, a Negar Moradighadi, b Kondalarao Kotturi, a Jeeranan Nonkumwong, a,c Md.
S1 SUPPORTING INFORMATION Solvent Isotopic Effects on a Surfactant Headgroup at the Air-Liquid Interface Uvinduni I. Premadasa, a Negar Moradighadi, b Kondalarao Kotturi, a Jeeranan Nonkumwong, a,c Md.
Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA
 Supporting Information for Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA Xu Zhang ab, Mark R. Servos b, and Juewen Liu* a a Department of Chemistry and Waterloo Institute
Supporting Information for Fast ph-assisted functionalization of silver nanoparticles with monothiolated DNA Xu Zhang ab, Mark R. Servos b, and Juewen Liu* a a Department of Chemistry and Waterloo Institute
Pramfina-J. Phys., Vol. 30, No. 2, February 1988, pp Printed in India.
 Pramfina-J. Phys., Vol. 30, No. 2, February 1988, pp. 135-141. Printed in India. Photoacoustic spectra of some laser dyes R S RAM and OM PRAKASH Division of Standards, National Physical Laboratory, New
Pramfina-J. Phys., Vol. 30, No. 2, February 1988, pp. 135-141. Printed in India. Photoacoustic spectra of some laser dyes R S RAM and OM PRAKASH Division of Standards, National Physical Laboratory, New
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions
 Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
Electronic and vibrational spectra of aniline benzene heterodimer and aniline homo-dimer ions Kazuhiko Ohashi a,*, Yoshiya Inokuchi b, Hironobu Izutsu a, Kazuyuki Hino a, Norifumi Yamamoto a, Nobuyuki
Electronic Supplementary Information. Selective detection of Al 3+ and citric acid with a fluorescent amphiphile
 Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Selective detection of Al 3+ and citric
Electronic Supplementary Material (ESI) for Organic & Biomolecular Chemistry. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Selective detection of Al 3+ and citric
Supporting Information
 Supporting Information Precisely Controllable Core-Shell Ag@Carbon Dots Nanoparticles: Application to in Situ Super-Sensitive Monitoring of Catalytic Reactions Jing Jin, Shoujun Zhu, Yubin Song, Hongyue
Supporting Information Precisely Controllable Core-Shell Ag@Carbon Dots Nanoparticles: Application to in Situ Super-Sensitive Monitoring of Catalytic Reactions Jing Jin, Shoujun Zhu, Yubin Song, Hongyue
Solvent Extraction Research and Development, Japan, Vol. 21, No 1, (2014)
 Solvent Extraction Research and Development, Japan, Vol. 1, No 1, 71 76 (14) Notes Salting-out Phase Separation System of Water Tetrahydrofuran with Co-using 1-Butyl-3-methylimidazolium Chloride and Sodium
Solvent Extraction Research and Development, Japan, Vol. 1, No 1, 71 76 (14) Notes Salting-out Phase Separation System of Water Tetrahydrofuran with Co-using 1-Butyl-3-methylimidazolium Chloride and Sodium
Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces
 748 Bull. Korean Chem. Soc. 2001, Vol. 22, No. 7 Myung M. Sung and Yunsoo Kim Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces Myung M. Sung * and Yunsoo Kim Department of Chemistry,
748 Bull. Korean Chem. Soc. 2001, Vol. 22, No. 7 Myung M. Sung and Yunsoo Kim Self-Assembled Monolayers of Alkanethiols on Clean Copper Surfaces Myung M. Sung * and Yunsoo Kim Department of Chemistry,
In-situ SERS Study of Ionic Transport and Joule Heating Effect in Plasmonic Nanopores
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information In-situ SERS Study of Ionic Transport and Joule Heating Effect in Plasmonic
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information In-situ SERS Study of Ionic Transport and Joule Heating Effect in Plasmonic
Development of NIR Bioimaging Systems
 Journal of Physics: Conference Series 16 (28) 1223 doi:1.188/1742-6596/16/1/1223 Development of NIR Bioimaging Systems Kohei SOGA 1, Takashi TSUJI 1, Fumio TASHIRO 1, Joe CHIBA 1 Motoi OISHI 2, Keitaro
Journal of Physics: Conference Series 16 (28) 1223 doi:1.188/1742-6596/16/1/1223 Development of NIR Bioimaging Systems Kohei SOGA 1, Takashi TSUJI 1, Fumio TASHIRO 1, Joe CHIBA 1 Motoi OISHI 2, Keitaro
QCM Study of β-casein Adsorption on the Hydrophobic Surface: Effect of Ionic Strength and Cations
 QCM Study of β-casein Adsorption on the Hydrophobic Surface Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 1031 QCM Study of β-casein Adsorption on the Hydrophobic Surface: Effect of Ionic Strength and Cations
QCM Study of β-casein Adsorption on the Hydrophobic Surface Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 1031 QCM Study of β-casein Adsorption on the Hydrophobic Surface: Effect of Ionic Strength and Cations
Preparation of Silver Nanoparticles through Alcohol Reduction with Organoalkoxysilanes
 Journal of Sol-Gel Science and Technology 26, 467 471, 2003 c 2003 Kluwer Academic Publishers. Manufactured in The Netherlands. Preparation of Silver Nanoparticles through Alcohol Reduction with Organoalkoxysilanes
Journal of Sol-Gel Science and Technology 26, 467 471, 2003 c 2003 Kluwer Academic Publishers. Manufactured in The Netherlands. Preparation of Silver Nanoparticles through Alcohol Reduction with Organoalkoxysilanes
Avidin Induced Silver Aggregation for SERS-based Bioassay
 Avidin Induced Silver Aggregation for SERS-based Bioassay Bull. Korean Chem. Soc. 2012, Vol. 33, No. 11 3681 http://dx.doi.org/10.5012/bkcs.2012.33.11.3681 Avidin Induced Silver Aggregation for SERS-based
Avidin Induced Silver Aggregation for SERS-based Bioassay Bull. Korean Chem. Soc. 2012, Vol. 33, No. 11 3681 http://dx.doi.org/10.5012/bkcs.2012.33.11.3681 Avidin Induced Silver Aggregation for SERS-based
Application note. SERS study of EMImTFSI on gold surfaces. (c) rhd instruments GmbH & Co. KG Mareike Länger
 Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
Application note SERS study of EMImTFSI on gold surfaces (c) 2013-2018 rhd instruments GmbH & Co. KG Mareike Länger Introduction Vibrational spectroscopy techniques like infrared or Raman spectroscopy
FT-Raman Spectroscopy of Heavy Metal-Nucleotide Complexes
 Asian Journal of Chemistry Vol. 21, No. 6 (2009), 4241-4245 FT-Raman Spectroscopy of Heavy Metal-Nucleotide Complexes N. IYANDURAI, K. SENTHIL* and R. SAROJINI Department of Physics, Kongunadu Arts and
Asian Journal of Chemistry Vol. 21, No. 6 (2009), 4241-4245 FT-Raman Spectroscopy of Heavy Metal-Nucleotide Complexes N. IYANDURAI, K. SENTHIL* and R. SAROJINI Department of Physics, Kongunadu Arts and
RAMAN INTENSITIES INVESTIGATING THE ENVIRONMENT- DEPENDENT PHOTOPHYSICS OF CHLORINE DIOXIDE WITH RESONANCE
 Laser Chem., 1999, Vol. 19, pp. 305-309 Reprints available directly from the publisher Photocopying permitted by license only (C) 1999 OPA (Overseas Publishers Association) N.V. Published by license under
Laser Chem., 1999, Vol. 19, pp. 305-309 Reprints available directly from the publisher Photocopying permitted by license only (C) 1999 OPA (Overseas Publishers Association) N.V. Published by license under
High-Purity Separation of Gold Nanoparticle Dimers and Trimers
 -Supporting Information- High-Purity Separation of Gold Nanoparticle Dimers and Trimers Gang Chen, Yong Wang, Li Huey Tan, Miaoxin Yang, Lee Siew Tan, Yuan Chen and Hongyu Chen* Division of Chemistry and
-Supporting Information- High-Purity Separation of Gold Nanoparticle Dimers and Trimers Gang Chen, Yong Wang, Li Huey Tan, Miaoxin Yang, Lee Siew Tan, Yuan Chen and Hongyu Chen* Division of Chemistry and
PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES
 PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES Lucie ŠTOLCOVÁ a, Jan PROŠKA a, Filip NOVOTNÝ a, Marek PROCHÁZKA b, Ivan RICHTER a a Czech Technical University in Prague, Faculty of Nuclear
PERIODIC ARRAYS OF METAL NANOBOWLS AS SERS-ACTIVE SUBSTRATES Lucie ŠTOLCOVÁ a, Jan PROŠKA a, Filip NOVOTNÝ a, Marek PROCHÁZKA b, Ivan RICHTER a a Czech Technical University in Prague, Faculty of Nuclear
Gold Nanoparticle-Based Detection of Hg(II) in an Aqueous Solution: Fluorescence Quenching and Surface-Enhanced Raman Scattering Study
 Gold Nanoparticle-Based Detection of Hg(II) in an Aqueous Solution Bull. Korean Chem. Soc. 2011, Vol. 32, No. 2 519 DOI 1012/bkcs.2011.32.2.519 Gold Nanoparticle-Based Detection of Hg(II) in an Aqueous
Gold Nanoparticle-Based Detection of Hg(II) in an Aqueous Solution Bull. Korean Chem. Soc. 2011, Vol. 32, No. 2 519 DOI 1012/bkcs.2011.32.2.519 Gold Nanoparticle-Based Detection of Hg(II) in an Aqueous
International Journal of Pure and Applied Sciences and Technology
 Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Int. J. Pure Appl. Sci. Technol., 9(1) (2012), pp. 1-8 International Journal of Pure and Applied Sciences and Technology ISSN 2229-6107 Available online at www.ijopaasat.in Research Paper Preparation,
Instantaneous and Quantitative Functionalization of Gold Nanoparticles with Thiolated DNA Using a ph-assisted and Surfactant-Free Route
 Supporting Information Instantaneous and Quantitative Functionalization of Gold Nanoparticles with Thiolated DNA Using a ph-assisted and Surfactant-Free Route Xu Zhang,, Mark R. Servos and Juewen Liu *
Supporting Information Instantaneous and Quantitative Functionalization of Gold Nanoparticles with Thiolated DNA Using a ph-assisted and Surfactant-Free Route Xu Zhang,, Mark R. Servos and Juewen Liu *
Electronic supplementary information
 Electronic supplementary information Surface plasmon resonance enhanced upconversion luminescence in aqueous media for TNT selective detection Nina Tu and Leyu Wang* State Key Laboratory of Chemical Resource
Electronic supplementary information Surface plasmon resonance enhanced upconversion luminescence in aqueous media for TNT selective detection Nina Tu and Leyu Wang* State Key Laboratory of Chemical Resource
Chem Homework Set Answers
 Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Chem 310 th 4 Homework Set Answers 1. Cyclohexanone has a strong infrared absorption peak at a wavelength of 5.86 µm. (a) Convert the wavelength to wavenumber.!6!1 8* = 1/8 = (1/5.86 µm)(1 µm/10 m)(1 m/100
Facile Phase Transfer of Gold Nanoparticles From Aqueous. Solution to Organic Solvents with Thiolated Poly(ethylene glycol)
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information for: Facile Phase Transfer of Gold Nanoparticles From Aqueous Solution
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information for: Facile Phase Transfer of Gold Nanoparticles From Aqueous Solution
Micellar Effect on The Reaction of Acetophenone with Phenylhydrazine
 Asian Journal of Chemistry Vol. 22, No. 2 (1), 829-833 Micellar Effect on The Reaction of Acetophenone with Phenylhydrazine K. NAGAJYOTHI*, P.S. RAGHAVAN and R. GOPALAN Department of Chemistry, Aarupadai
Asian Journal of Chemistry Vol. 22, No. 2 (1), 829-833 Micellar Effect on The Reaction of Acetophenone with Phenylhydrazine K. NAGAJYOTHI*, P.S. RAGHAVAN and R. GOPALAN Department of Chemistry, Aarupadai
Raman and infrared spectra of tere-phthalaldehyde
 Pram[qa, Vol. 18, No. 4, April 1982, pp. 311-315. ~) Printed in India. Raman and infrared spectra of tere-phthalaldehyde R A YADAV, RAMAKANT, P C MISHRA and I S SINGH Spectroscopy Laboratory, Department
Pram[qa, Vol. 18, No. 4, April 1982, pp. 311-315. ~) Printed in India. Raman and infrared spectra of tere-phthalaldehyde R A YADAV, RAMAKANT, P C MISHRA and I S SINGH Spectroscopy Laboratory, Department
Supporting Information s for
 Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Supporting Information s for # Self-assembling of DNA-templated Au Nanoparticles into Nanowires and their enhanced SERS and Catalytic Applications Subrata Kundu* and M. Jayachandran Electrochemical Materials
Dendritic Star Polymer of Polyacrylamide Based on β-cyclodextrin Trimer: A. Flocculant and Drug Vehicle
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water
 Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Kang, Lin-Woo, Ph.D. Professor Department of Biological Sciences Konkuk University Seoul, Korea nd Semester
 Kang, Lin-Woo, Ph.D. Professor Department of Biological Sciences Konkuk University Seoul, Korea 2018. 2 nd Semester Absorbance Assay (280 nm) Considerations for use Absorbance assays are fast and
Kang, Lin-Woo, Ph.D. Professor Department of Biological Sciences Konkuk University Seoul, Korea 2018. 2 nd Semester Absorbance Assay (280 nm) Considerations for use Absorbance assays are fast and
Supporting Information
 Gold Nanoparticle-Modified ITO Electrode for Electrogenerated Chemiluminescence: Well-Preserved Transparency and Highly-Enhanced Activity Zuofeng Chen and Yanbing Zu * Department of Chemistry, The University
Gold Nanoparticle-Modified ITO Electrode for Electrogenerated Chemiluminescence: Well-Preserved Transparency and Highly-Enhanced Activity Zuofeng Chen and Yanbing Zu * Department of Chemistry, The University
Self-assembly of PEGylated Gold Nanoparticles. with Satellite Structures as Seeds
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 216 Electronic Supplementary Information for Self-assembly of PEGylated Gold Nanoparticles with Satellite
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 216 Electronic Supplementary Information for Self-assembly of PEGylated Gold Nanoparticles with Satellite
Spectroscopy Problem Set February 22, 2018
 Spectroscopy Problem Set February, 018 4 3 5 1 6 7 8 1. In the diagram above which of the following represent vibrational relaxations? 1. Which of the following represent an absorbance? 3. Which of following
Spectroscopy Problem Set February, 018 4 3 5 1 6 7 8 1. In the diagram above which of the following represent vibrational relaxations? 1. Which of the following represent an absorbance? 3. Which of following
ECE280: Nano-Plasmonics and Its Applications. Week8
 ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
ECE280: Nano-Plasmonics and Its Applications Week8 Surface Enhanced Raman Scattering (SERS) and Surface Plasmon Amplification by Stimulated Emission of Radiation (SPASER) Raman Scattering Chandrasekhara
Headspace Raman Spectroscopy
 ELECTRONICALLY REPRINTED FROM SEPTEMBER 2014 Molecular Spectroscopy Workbench Raman Spectroscopy We examine vapor-phase Raman spectroscopy through the acquisition of spectra from gas molecules confined
ELECTRONICALLY REPRINTED FROM SEPTEMBER 2014 Molecular Spectroscopy Workbench Raman Spectroscopy We examine vapor-phase Raman spectroscopy through the acquisition of spectra from gas molecules confined
NANO 243/CENG 207 Course Use Only
 L12: Drug Loading & Quantification May 15, 2018 1. Drug loading techniques 1.1 Physical approaches Nanprecipitation Single emulsion Double emulsion Encapsulation Remote loading 1.2 Chemical approaches
L12: Drug Loading & Quantification May 15, 2018 1. Drug loading techniques 1.1 Physical approaches Nanprecipitation Single emulsion Double emulsion Encapsulation Remote loading 1.2 Chemical approaches
