MultiMax Application Note
|
|
|
- Pierce Hopkins
- 6 years ago
- Views:
Transcription
1 MultiMax Application Note Investigation of an Imine Formation 1. Introduction Today s Research and Development world demands faster results to be able to deliver more products on time. Automation techniques coupled with in-situ analytics offer the ability to obtain physical and chemical information at the same time. Rapid process development is achieved by incorporating auto - mation tools and instrumen tation 2. Reaction The reaction studied is an imine formation in methanol (Schiff base). Recipe: Ramp Isobutylamine in methanol to desired temperature Add Benzaldehyde expertise into traditional organic chemistry. Furthermore the processes can be maximized if they are used in hand with statis tical design and screening. In this study, we focused on the formation of an Imine. The goal was to in vestigate the progress of the reaction as well as the end point. The work consisted of four No batch experiments to gather initial information on the reaction. At a later stage four semi-batch experiments were performed. The reaction was monitored by: The temperatures of the reaction mass and jacket (Tr-Tj) Mid-IR signal Heat of the Reaction () Thermal Conversion Reagent Quality Mol Ratio Amount Benzaldehyde 99.5+% 10 mmol g Isobutylamine 99% 10 mmol g Methanol 99.93% 15 ml Table 1 Variables: xperimental parameters Temperature (20 C or 40 C) Dosing time (5 min) or batch For the batch reactions 10 ml were while the semi-batch ones were done reaction added in one shot using a pipette automatically using a dispenser box.
2 3. Apparatus Figure 1 The MultiMax multiple reactor system is designed for process screening, optimization and safety studies. It delivers precise and reproducible control of critical reaction variables such as temperature, stirrer speed, etc. and automation of routine procedures such as dosing, ph measurement, and ph control. The high quality of the temperature control and measurements allows getting valuable reaction and process information such as reaction initiation, reaction end-point, and thermal data. The MultiMaxIR is a multiple automated lab reactor with in-situ Mid Infrared Technology. The system uses a MultiMax reactor box RB 04x50; part of the MultiMax family; which consists of four 50ml tank style vessels with independent control and built-in infrared sampling technology. The reactor box is coupled to an optical docking station with an automated light conduit and a ReactIR system. Figure 1 Apparatus utilized for this application 4. Considerations 4.1. Calorimetry Figure 2 Reaction Calorimetry measures the instantaneous heat generated by a given reaction. The measured heat is related to the reaction rate (kinetics), which makes rate the primary measured parameter of calorimetry. By contrast, conventional techniques such as FTIR, NMR and chromatographic methods yield concentration as a function of time. Calorimetry is a complementary method that provides thermal information. This makes calorimetry an ideal complementary analytical method. Tr Heat 4.2. Reaction nthalpy 4.3. Thermal conversion Figure 3 Reaction nthalpy (ΔH r ) Figure 4 & 5 The thermal conversion is the curve that describes by definition is the integral of heat generated (Q r ) minus a baseline (Q b ), the partial integral related to the over the time of the reaction. total heat of reaction. A suitable baseline means that under It is displayed in function of time the same experimental conditions and varies between 0 and 100%. there is no heat generated. Tj Figure 2 Calorimetry concept S Figure 3 Reaction enthalpy Q t Xr S t 1 S t 2 Figure 4 Thermal conversion concept: Thermal conversion Xr (t 1 ) = ΔH r (t 1 ) / ΔH r Thermal conversion Xr (t 2 ) = ΔH r (t 2 ) / ΔH r ΔHr (t 1 ) Thermal Conversion S Figure 5 Thermal conversion profile ΔHr (t 2 )
3 5. xperiments Screening Calorimetry in MultiMax Figure 6 In order to make the thermal information quantifiable, we performed screening calorimetry at the reaction temperature, by activating a calibration heater inside the reactor. This heater is connected to the MultiMax by the Universal Control Box. The calibration steps are performed before and after the reaction takes place, allowing us to determine the calibration factors necessary for the calorimetric calculations. Figure 8 After evaluating the calibration factors, we selected the heat terms that were in voived in the heat of reaction such as heat flow (Q flow ), heat accu mulated (Q accu ) and heat due to dosings (Q dos ). We observed the numeric values for the start and end time of the reaction, and the selected baseline type. The specific heat of the reaction mass (cp) and the temperature of the material dosed (T dos ) are presented too. Ramp to desired Temperature Wait (until stable) Figure 6 MultiMax screening calorimetry method Calibration heater Wait (until stable) Dosing/reaction and wait Calibration heater Wait (stable) Figure 7 Once the experiment with the corresponding calorimetry steps was completed, the trends of the calibration heater (Q c ), the reaction mass (mr) and the temperature difference between the reaction mass and the jacket temperature (T r T j ) were displayed. Subsequently, the results such as heat flow and thermal conversion were evaluated. Figure 9 Once the corresponding selections were made in the dialogue box for heat integration, the software calculated the enthalpy of the respective reactions and displayed them together with the start and end times of the integration ranges. Figure 10 Shows the graphical representation of the evaluated heat flow curve (Q r ) together with the calibration signal and the mass balance. UA1 Tr-Tj Figure 7 MultiMax screening calorimetry method UA2 Figure 8 MultiMax dialogue box for heat integration Figure 9 MultiMax results box for the screening calorimetry method Figure 10 Graphical display for the heat of the reaction (Q r )
4 6. Results 6.1. Graphical Analysis Figure 11 By using the experimental approach described in the previous section, we determined the thermal conversion and end point for the given reaction. 6.2 Batch Reaction Results Table 2 After performing the analysis it was possible to tabulate the results with the reaction temperature and mixing speed. The results showed that the reation proceeded fast, but it was difficult to determine whether the reaction rate was limited by mixing or temperature, or both. 6.3 Semi-batch Reaction Results Table 3 Similarly to the data analysis for batch reactions, we performed the analysis for the semi-batch ones. In this case we focused on the thermal conversion after dosing was completed and tabulated it with the reaction temperature and mixing speed. We observed that at 20 C the results were the same indepedently of the mixing speed. When the reaction was performed at 40 C the thermal conversion was identical. These results excluded mixing as the cause for rate limitation and identified the reaction temperature as a rate limiting factor. 6.4 nthalpy Results Tables 4 & 5 summarize the enthalpies calculated for the eight experiments performed. In average, the result was 3.98 kj; the individual results spread within ±20% of what we expected for a screening calorimetry experiment. Average 3.98 kj Average + 20% = 4.78 kj Average 20% = 3.19 kj Figure 11 Thermal conversion with mass and heat of reaction Temperature Stirrer speed to reaction completion 20 C 1000 rpm 7 min 20 C 200 rpm 7.5 min 40 C 1000 rpm 5 min 40 C 200 rpm 5.5 min Table 2 Batch results Temperature Stirrer speed Thermal conversion after dosing was completed 20 C 1000 rpm 71.6% * 20 C 200 rpm 68.5% * 40 C 1000 rpm 85% 40 C 200 rpm 85% Table 3 Semi-batch results Thermal conversion *No significant difference between 71.6 and 68.5% Temperature Stirrer speed nthalpy 20 C 1000 rpm 3.78 kj 20 C 200 rpm 3.58 kj 40 C 1000 rpm 4.44 kj 40 C 200 rpm 3.62 kj Table 4 Semi-batch enthalpy results Temperature Stirrer speed nthalpy 20 C 1000 rpm 4.38 kj 20 C 200 rpm 4.34 kj 40 C 1000 rpm 3.65 kj 40 C 200 rpm 4.10 kj Table 5 Batch enthalpy results
5 6.5 Validation of the Information Figure 12 & 13 In order to validate the thermal information, we utilized Mid-IR data as a complementary measurement. The measurement was performed with a MultiMaxIR. It scans all reactor vessels simultaneously while the reaction is performed. The Mid-Infrared profiles can be presented together with the Multi- Max data in the same trend graph which allows us to compare them directly with the heat flow trends. Figure 12 Mid-IR progress of the reaction using MultiMaxIR PRODUCT Figure 13 shows that the thermal as well as the chemical conversions are in agreement. ReactIR product profile Thermal conversion Figure 13 Comparison of the chemical and thermal conversions 7. Conclusions Reaction calorimetry allows the determination of thermal conversion. Thermal conversion can be used as an in-situ analytical technique that allows to study: Factors that have an impact on the reaction rate such as: Mixing Reaction temperature Addition rate Concentration Reagent/substrate accumulation during a reaction Reaction dynamics and kinetics studies Troubleshoot scale up issues With only a few experiments we gained a better understanding of the imine formation and identified the driving factors of the process. Temperature was identified as the decisive factor whereas mixing only plays a secondary role. Using MultiMax calorimetry and ReactIR as analytical tools increased the speed of development, but also supported the validation of the results assuring its correctness and integrity. Mettler-Toledo AutoChem Inc Samuel Morse Drive Columbia, MD 21046, USA Phone Fax Mail: autochem.marketing@mt.com Mettler-Toledo AG, AutoChem Sonnenbergstrasse 74 CH-8603 Schwerzenbach, Switzerland Phone Fax Subject to technical changes. 10/2007 Mettler-Toledo AG Printed in Switzerland, Marketing ALR For more Information
Calorimetry Guide. Safety by Design What do we Learn from Reaction Calorimetry?
 Calorimetry Guide What do we Learn from Reaction Calorimetry? Developing new compounds and transferring them to manufacturing requires an understanding of the chemical route, process and all its parameters.
Calorimetry Guide What do we Learn from Reaction Calorimetry? Developing new compounds and transferring them to manufacturing requires an understanding of the chemical route, process and all its parameters.
Process Safety. Process Safety and Hazard Assessment Avoiding Incidents in the Lab and in the Plant
 Process Safety Process Safety and Hazard Assessment Avoiding Incidents in the Lab and in the Plant Process Safety Process Safety and Hazard Assessment From Early Development to Manufacturing The importance
Process Safety Process Safety and Hazard Assessment Avoiding Incidents in the Lab and in the Plant Process Safety Process Safety and Hazard Assessment From Early Development to Manufacturing The importance
Recent Advances to Improve Chemical Development
 Recent Advances to Improve Chemical Development Agenda Challenges in Today s Pharma Industry Industry Drivers for Change Four Ways to Improve Organic Synthesis Enabling Technology Impact of New Tools on
Recent Advances to Improve Chemical Development Agenda Challenges in Today s Pharma Industry Industry Drivers for Change Four Ways to Improve Organic Synthesis Enabling Technology Impact of New Tools on
LabMax ReactIR FBRM. Relative Supersaturation Control in Batch Cooling Crystallization. Application Note. 1 Abstract.
 Jörg Worlitschek Terry Redman Herbert Briggeler Olivier Ubrich Paul Barrett Brian O Sullivan Claude Didierjean Mettler Toledo GmbH, AutoChem Address Sonnenbergstrasse 74 PO Box, CH-8603 Schwerzenbach Phone
Jörg Worlitschek Terry Redman Herbert Briggeler Olivier Ubrich Paul Barrett Brian O Sullivan Claude Didierjean Mettler Toledo GmbH, AutoChem Address Sonnenbergstrasse 74 PO Box, CH-8603 Schwerzenbach Phone
calorimeter heat flow calorimetry, power compensation calorimetry
 calorimeter heat flow calorimetry, power compensation calorimetry Atlas simply does it all What is the Atlas Reaction Calorimeter? The Atlas Reaction Calorimeter accurately measures the power and enthalpy
calorimeter heat flow calorimetry, power compensation calorimetry Atlas simply does it all What is the Atlas Reaction Calorimeter? The Atlas Reaction Calorimeter accurately measures the power and enthalpy
Perseverance. Experimentation. Knowledge.
 2410 Intuition. Perseverance. Experimentation. Knowledge. All are critical elements of the formula leading to breakthroughs in chemical development. Today s process chemists face increasing pressure to
2410 Intuition. Perseverance. Experimentation. Knowledge. All are critical elements of the formula leading to breakthroughs in chemical development. Today s process chemists face increasing pressure to
Atlas Syringe Pump XL
 Atlas Syringe Pump XL Batch chemistry application note control of an un-buffered solution using the Atlas Syringe Pump XL Version: 1.0 Issue Date: 21/03/2018 Author: SJH 1 Summary 2 Introduction 2.1 Equipment
Atlas Syringe Pump XL Batch chemistry application note control of an un-buffered solution using the Atlas Syringe Pump XL Version: 1.0 Issue Date: 21/03/2018 Author: SJH 1 Summary 2 Introduction 2.1 Equipment
In Situ Spectroscopy. Reaction Analysis and PAT Tools From Research to Manufacturing
 In Situ Spectroscopy Reaction Analysis and PAT Tools From Research to Manufacturing Chemical Synthesis, Engineering and PAT Understanding Reaction Chemistry with ReactIR In Situ Analytical Tools ReactIR
In Situ Spectroscopy Reaction Analysis and PAT Tools From Research to Manufacturing Chemical Synthesis, Engineering and PAT Understanding Reaction Chemistry with ReactIR In Situ Analytical Tools ReactIR
Avoid Batch Failures via Scale Down Simulation of Exothermic Reactions in the Lab
 Avoid Batch Failures via Scale Down Simulation of Exothermic Reactions in the Lab Leen Schellekens Applications & Technology Consultants Manager Americas VisiMix - The Influence of Mixing In Your Process
Avoid Batch Failures via Scale Down Simulation of Exothermic Reactions in the Lab Leen Schellekens Applications & Technology Consultants Manager Americas VisiMix - The Influence of Mixing In Your Process
Optimization of a Nitration Reaction
 CBC-PROCOS S.p.A. - Research and Development Laboratory - Calorimetry Optimization of a Nitration Reaction Alessandro Barozza*, Paolo Paissoni, Jacopo Roletto barozza@procos.it Giornata di Studio sullo
CBC-PROCOS S.p.A. - Research and Development Laboratory - Calorimetry Optimization of a Nitration Reaction Alessandro Barozza*, Paolo Paissoni, Jacopo Roletto barozza@procos.it Giornata di Studio sullo
AGrignard reaction, in which a reactant, A, is coupled with phenyl magnesium chloride, is used to
 DEVELOPMENT OF AN EFFICIENT AND SAFE PROCESS FOR A GRIGNARD REACTION VIA REACTION CALORIMETRY H. Ferguson and Y. M. Puga The Dow Chemical Co., Inc. Engineering Sciences/Market Development A paper from
DEVELOPMENT OF AN EFFICIENT AND SAFE PROCESS FOR A GRIGNARD REACTION VIA REACTION CALORIMETRY H. Ferguson and Y. M. Puga The Dow Chemical Co., Inc. Engineering Sciences/Market Development A paper from
Scale-up problems are often perceived as difficult. Here the reaction calorimetry has proven to be
 APPLICATION OF REACTION CALORIMETRY FOR THE SOLUTION OF SCALE-UP PROBLEMS A paper from the RC User Forum Europe, Interlaken, 1995 Francis Stoessel, Ciba AG, Basel, Switzerland. Scale-up problems are often
APPLICATION OF REACTION CALORIMETRY FOR THE SOLUTION OF SCALE-UP PROBLEMS A paper from the RC User Forum Europe, Interlaken, 1995 Francis Stoessel, Ciba AG, Basel, Switzerland. Scale-up problems are often
Compact Titration. Compact Titrator G20. One Click Titration Simple & Dependable
 Compact Titration Compact Titrator G20 One Click Titration Simple & Dependable Simplicity The Intelligent Helper for Simple Titrations One Click Titration Intuitive method adaptation Accurate results simply
Compact Titration Compact Titrator G20 One Click Titration Simple & Dependable Simplicity The Intelligent Helper for Simple Titrations One Click Titration Intuitive method adaptation Accurate results simply
ISoTherMal TITraTIon Calorimetry
 ISoTherMal TITraTIon Calorimetry With the Nano ITC, heat effects as small as 1 nanojoules are detectable using one nanomole or less of biopolymer. The Nano ITC uses a solid-state thermoelectric heating
ISoTherMal TITraTIon Calorimetry With the Nano ITC, heat effects as small as 1 nanojoules are detectable using one nanomole or less of biopolymer. The Nano ITC uses a solid-state thermoelectric heating
Spectroscopy tools for PAT applications in the Pharmaceutical Industry
 Spectroscopy tools for PAT applications in the Pharmaceutical Industry Claude Didierjean Sr. Technology and Applications Consultant Real Time Analytics Mettler Toledo AutoChem, Inc. claude.didierjean@mt.com
Spectroscopy tools for PAT applications in the Pharmaceutical Industry Claude Didierjean Sr. Technology and Applications Consultant Real Time Analytics Mettler Toledo AutoChem, Inc. claude.didierjean@mt.com
The THT micro reaction calorimeter μrc
 The THT micro reaction calorimeter μrc Titration calorimetry isothermal calorimetry and scanning calorimetry - all in one instrument CHEMICAL APPLICATIONS BROCHURE Introduction and reaction kinetics The
The THT micro reaction calorimeter μrc Titration calorimetry isothermal calorimetry and scanning calorimetry - all in one instrument CHEMICAL APPLICATIONS BROCHURE Introduction and reaction kinetics The
Analysing Phenol-Formaldehyde Resin Reaction For Safe Process Scale Up
 SYMPOSIUM SERIES NO 16 HAZARDS 25 215 IChemE Analysing Phenol-Formaldehyde Resin Reaction For Safe Process Scale Up David Dale, Process Safety Manager, SciMed/Fauske and Associates, Unit B4, The Embankment
SYMPOSIUM SERIES NO 16 HAZARDS 25 215 IChemE Analysing Phenol-Formaldehyde Resin Reaction For Safe Process Scale Up David Dale, Process Safety Manager, SciMed/Fauske and Associates, Unit B4, The Embankment
Toluene Mono-nitration in a Semi-batch Reactor
 Toluene Mono-nitration in a Semi-batch Reactor 37 Central European Journal of Energetic Materials, 2008, 5(2), 37-47. ISSN 1733-7178 Toluene Mono-nitration in a Semi-batch Reactor Li-Ping CHEN, Wang-Hua
Toluene Mono-nitration in a Semi-batch Reactor 37 Central European Journal of Energetic Materials, 2008, 5(2), 37-47. ISSN 1733-7178 Toluene Mono-nitration in a Semi-batch Reactor Li-Ping CHEN, Wang-Hua
Applications of the Micro Reaction Calorimeter
 Applications of the Micro Reaction Calorimeter Power-compensation method. Versatility Automated sample addition, repeated or timed. Wide temperature range Pressure measurement Stirring Highly sensitive
Applications of the Micro Reaction Calorimeter Power-compensation method. Versatility Automated sample addition, repeated or timed. Wide temperature range Pressure measurement Stirring Highly sensitive
Manufacturing process control with PAT Substitution of off-line HPLC & GC by in-line IR spectroscopy
 Manufacturing process control with PAT Substitution of off-line HPLC & GC by in-line IR spectroscopy Christian Lautz, F. Hoffmann-La Roche Ltd, Basel, Switzerland IFPAC-2015, January 25-28, Arlington,USA
Manufacturing process control with PAT Substitution of off-line HPLC & GC by in-line IR spectroscopy Christian Lautz, F. Hoffmann-La Roche Ltd, Basel, Switzerland IFPAC-2015, January 25-28, Arlington,USA
Reaction Calorimetry as a Tool for Thermal Risk Assessment and Improvement of Safe Scalable Chemical Processes
 Reaction Calorimetry as a Tool for Thermal Risk Assessment and Improvement of Safe Scalable Chemical Processes Kamala Jyotsna G, Sindhanur Srikanth, Vinay Ratnaparkhi, and Rakeshwar Bandichhora * Research
Reaction Calorimetry as a Tool for Thermal Risk Assessment and Improvement of Safe Scalable Chemical Processes Kamala Jyotsna G, Sindhanur Srikanth, Vinay Ratnaparkhi, and Rakeshwar Bandichhora * Research
LABOTRON EXTRACTION & SYNTHESIS 2450 MHz
 LABOTRON EXTRACTION & SYNTHESIS 2450 MHz The LABOTRON TM series is the generic name for a ground breaking range of integrated reactor and microwave transmission systems especially designed to carry out
LABOTRON EXTRACTION & SYNTHESIS 2450 MHz The LABOTRON TM series is the generic name for a ground breaking range of integrated reactor and microwave transmission systems especially designed to carry out
Supporting Information
 1 Supporting Information 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Materials and Methods Experiment In this study, an alloy IR probe which allowed us to get access to spectral
1 Supporting Information 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 Materials and Methods Experiment In this study, an alloy IR probe which allowed us to get access to spectral
The THT micro reaction calorimeter RC
 The THT micro reaction calorimeter RC Titration calorimetry isothermal calorimetry and scanning calorimetry - all in one instrument C H E M I C A L A P P L I C A T I O N S B R O C H U R E Introduction
The THT micro reaction calorimeter RC Titration calorimetry isothermal calorimetry and scanning calorimetry - all in one instrument C H E M I C A L A P P L I C A T I O N S B R O C H U R E Introduction
VISIMIX TURBULENT. BATCH REACTOR SCALING UP.
 Introduction. VISIMIX TURBULENT. BATCH REACTOR SCALING UP. This example is based on a practical case application of VisiMix Turbulent by one of our clients. It is related to development of a new process
Introduction. VISIMIX TURBULENT. BATCH REACTOR SCALING UP. This example is based on a practical case application of VisiMix Turbulent by one of our clients. It is related to development of a new process
SIFT-MS SELECTED ION FLOW TUBE MASS SPECTROMETRY TECHNOLOGY OVERVIEW \ SYFT TECHNOLOGIES
 SIFT-MS SELECTED ION FLOW TUBE MASS SPECTROMETRY TECHNOLOGY OVERVIEW \ SYFT TECHNOLOGIES CONTENTS 1. An overview of SIFT-MS 2. Principles of SIFT-MS a. Why chemical ionization? b. Why multiple reagent
SIFT-MS SELECTED ION FLOW TUBE MASS SPECTROMETRY TECHNOLOGY OVERVIEW \ SYFT TECHNOLOGIES CONTENTS 1. An overview of SIFT-MS 2. Principles of SIFT-MS a. Why chemical ionization? b. Why multiple reagent
Automatic titrators, TitroMatic
 112 TitroMatic These are the fourth generation of CRISON titrators. CRISON has over 25 years experience in this field and knows that, for certain applications, the performance and cost of large titrator
112 TitroMatic These are the fourth generation of CRISON titrators. CRISON has over 25 years experience in this field and knows that, for certain applications, the performance and cost of large titrator
Quantos GraviPrep. Bye Bye, ml Hello, Quantos
 Quantos GraviPrep Bye Bye, ml Hello, Quantos From Volumetric to Gravimetric 75 Years of the Volumetric Flask Remove the Errors in Sample Preparation History of the Volumetric Flask Volumetric flasks have
Quantos GraviPrep Bye Bye, ml Hello, Quantos From Volumetric to Gravimetric 75 Years of the Volumetric Flask Remove the Errors in Sample Preparation History of the Volumetric Flask Volumetric flasks have
The Benefits of Gravimetric Sample Preparation
 The Benefits of Gravimetric Sample Preparation Improving the Efficiency and Quality of Analytical Workflows Dr. Joanne Ratcliff and Dr. Jan Prochnow, Mettler-Toledo AG Analytical sample and standard preparation
The Benefits of Gravimetric Sample Preparation Improving the Efficiency and Quality of Analytical Workflows Dr. Joanne Ratcliff and Dr. Jan Prochnow, Mettler-Toledo AG Analytical sample and standard preparation
News. Testing Labs Weighing and Analysis in the Laboratory. EMPA Scientists Beyond Scratching the Surface
 Testing Labs Weighing and Analysis in the Laboratory 3 News EMPA Scientists Beyond Scratching the Surface EMPA is an internationally recognized research, development and service institution for materials
Testing Labs Weighing and Analysis in the Laboratory 3 News EMPA Scientists Beyond Scratching the Surface EMPA is an internationally recognized research, development and service institution for materials
POLYMERIZATION REACTION MONITORING FOR PSA PRODUCTION USING AN ATR-FTIR PROBE
 POLYMERIZATION REACTION MONITORING FOR PSA PRODUCTION USING AN ATR-FTIR PROBE Renata Jovanović, Doctoral student, Department of Chemical Engineering, University of Ottawa, Ottawa, Canada, (jovanovi@genie.uottawa.ca)
POLYMERIZATION REACTION MONITORING FOR PSA PRODUCTION USING AN ATR-FTIR PROBE Renata Jovanović, Doctoral student, Department of Chemical Engineering, University of Ottawa, Ottawa, Canada, (jovanovi@genie.uottawa.ca)
THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS
 THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS R.L. ROGERS* Dewar Calorimetry is one of the simplest and most useful techniques used in the assessment of chemical reaction
THE USE OF DEWAR CALORIMETRY IN THE ASSESSMENT OF CHEMICAL REACTION HAZARDS R.L. ROGERS* Dewar Calorimetry is one of the simplest and most useful techniques used in the assessment of chemical reaction
Experimental Procedure
 Experimental Procedure Overview The ph meter is used in conjunction with a titration apparatus and a standardized sodium hydroxide solution to determine the molar concentration of a weak acid solution
Experimental Procedure Overview The ph meter is used in conjunction with a titration apparatus and a standardized sodium hydroxide solution to determine the molar concentration of a weak acid solution
Level. Automate your processes Automate your lab Innovate with. sensing. EasyPREP. sample handler. Automated Sample Preparation.
 Automate your processes Automate your lab Innovate with EasyPREP sample handler Automated Sample Preparation Dispense up to 7 reagents Dilute Samples Aliquote Samples Transfer Samples Normalize Samples
Automate your processes Automate your lab Innovate with EasyPREP sample handler Automated Sample Preparation Dispense up to 7 reagents Dilute Samples Aliquote Samples Transfer Samples Normalize Samples
A kinetically controlled crystallization process for identifying new co-crystal forms: Fast evaporation of solvent from solutions to dryness
 A kinetically controlled crystallization process for identifying new co-crystal forms: Fast evaporation of solvent from solutions to dryness Partha Pratim Bag, a Mohit Patni, ab C. Malla Reddy* a Department
A kinetically controlled crystallization process for identifying new co-crystal forms: Fast evaporation of solvent from solutions to dryness Partha Pratim Bag, a Mohit Patni, ab C. Malla Reddy* a Department
Thermochemistry/Calorimetry. Determination of the enthalpy of vaporization of liquids LEC 02. What you need: What you can learn about
 LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
LEC 02 Thermochemistry/Calorimetry Determination of the enthalpy of vaporization of liquids What you can learn about Enthalpy of vaporisation Entropy of vaporisation Trouton s rule Calorimetry Heat capacity
Micro Reaction Calorimeter
 C3 Prozess- und Analysentechnik GmbH Produktinformation µrc Micro Reaction Calorimeter Tel. (089) 45 60 06 70 info@c3-analysentechnik.de www.c3-analysentechnik.de The THT micro reaction calorimeter µrc
C3 Prozess- und Analysentechnik GmbH Produktinformation µrc Micro Reaction Calorimeter Tel. (089) 45 60 06 70 info@c3-analysentechnik.de www.c3-analysentechnik.de The THT micro reaction calorimeter µrc
AUTO-CATALYTIC EFFECT OF ACETIC ACID ON THE KINETICS OF THE METHANOL / ACETIC ANHYDRIDE ESTERIFICATION
 AUTO-CATALYTIC EFFECT OF ACETIC ACID ON THE KINETICS OF THE METHANOL / ACETIC ANHYDRIDE ESTERIFICATION 1. Introduction Stefan Bohm, Günther Hessel, Holger Kryk, Horst-Michael Prasser, and Wilfried Schmitt
AUTO-CATALYTIC EFFECT OF ACETIC ACID ON THE KINETICS OF THE METHANOL / ACETIC ANHYDRIDE ESTERIFICATION 1. Introduction Stefan Bohm, Günther Hessel, Holger Kryk, Horst-Michael Prasser, and Wilfried Schmitt
High Pressure DSC Differential Scanning Calorimeter
 High Pressure DSC Differential Scanning Calorimeter Introduction The Differential Scanning Calorimetry (DSC) is the most popular thermal analysis technique to measure endothermic and exothermic transitions
High Pressure DSC Differential Scanning Calorimeter Introduction The Differential Scanning Calorimetry (DSC) is the most popular thermal analysis technique to measure endothermic and exothermic transitions
Automating Gradient Method Development in Flash Chromatography For Greater Productivity and Minimizing Solvent Use
 Automating Gradient in Flash For Greater Productivity and Minimizing Solvent Use Melissa Wilcox, Rakesh Bose, Scott Anderson, Kathy Lawrence Grace Davison Discovery Sciences 251 Waukegan Rd. Deerfield
Automating Gradient in Flash For Greater Productivity and Minimizing Solvent Use Melissa Wilcox, Rakesh Bose, Scott Anderson, Kathy Lawrence Grace Davison Discovery Sciences 251 Waukegan Rd. Deerfield
JAPAN QUALITY. Automated Clinical Analyzer
 JAPAN QUALITY Automated Clinical Analyzer Higher test performance and operability with new functions The clinical analyzer in the next stage 1 The best to the requirements for functionality and usability
JAPAN QUALITY Automated Clinical Analyzer Higher test performance and operability with new functions The clinical analyzer in the next stage 1 The best to the requirements for functionality and usability
3.2 Calorimetry and Enthalpy
 3.2 Calorimetry and Enthalpy Heat Capacity Specific heat capacity (c) is the quantity of thermal energy required to raise the temperature of 1 g of a substance by 1 C. The SI units for specific heat capacity
3.2 Calorimetry and Enthalpy Heat Capacity Specific heat capacity (c) is the quantity of thermal energy required to raise the temperature of 1 g of a substance by 1 C. The SI units for specific heat capacity
A NEW MEASUREMENT AND EVALUATION METHOD FOR DSC OF PCM SAMPLES
 A NEW MEASUREMENT AND EVALUATION METHOD FOR DSC OF PCM SAMPLES H Mehling, E Günther, S Hiebler, Bavarian Center for Applied Energy Research (ZAE Bayern), Walther-Meißner-Str. 6, D-85748 Garching, Germany.
A NEW MEASUREMENT AND EVALUATION METHOD FOR DSC OF PCM SAMPLES H Mehling, E Günther, S Hiebler, Bavarian Center for Applied Energy Research (ZAE Bayern), Walther-Meißner-Str. 6, D-85748 Garching, Germany.
High Pressure Chemistry. Hydrogenation & Catalysis Tools.
 High Pressure Chemistry Hydrogenation & Catalysis Tools www.helgroup.com HYDROGENATION AND CATALYSIS Product Selection Guide HEL Group s top quality high pressure reactors and systems offer innovative,
High Pressure Chemistry Hydrogenation & Catalysis Tools www.helgroup.com HYDROGENATION AND CATALYSIS Product Selection Guide HEL Group s top quality high pressure reactors and systems offer innovative,
JAPAN QUALITY. Automated Clinical Analyzer
 JAPAN QUALITY Automated Clinical Analyzer Higher test performance and operability with new functions The clinical analyzer in the next stage 1 The best to the requirements for functionality and usability
JAPAN QUALITY Automated Clinical Analyzer Higher test performance and operability with new functions The clinical analyzer in the next stage 1 The best to the requirements for functionality and usability
Automated Clinical Analyzer
 Automated Clinical Analyzer Improved ease of operation and better test efficiency, and provides the optimal solution for routine operations, STAT operation and a broad range of clinical analysis requirements.
Automated Clinical Analyzer Improved ease of operation and better test efficiency, and provides the optimal solution for routine operations, STAT operation and a broad range of clinical analysis requirements.
Thermal Analysis Premium
 Thermal Analysis Premium HP DSC 2+ STAR e System Innovative Technology Versatile Modularity Swiss Quality DSC Measurements under Pressure for Accelerated Materials Testing Double Safety System The Right
Thermal Analysis Premium HP DSC 2+ STAR e System Innovative Technology Versatile Modularity Swiss Quality DSC Measurements under Pressure for Accelerated Materials Testing Double Safety System The Right
96 Well? Done! Eppendorf epmotion 96 semi-automated electronic pipette for fast and precise 96 channel microplate processing
 96 Well? Done! Eppendorf epmotion 96 semi-automated electronic pipette for fast and precise 96 channel microplate processing Try it and you will see how easy and fast your 96-well and 384-well plates get
96 Well? Done! Eppendorf epmotion 96 semi-automated electronic pipette for fast and precise 96 channel microplate processing Try it and you will see how easy and fast your 96-well and 384-well plates get
C80. Calvet Calorimeter From ambient to 300 C. A trademark of KEP Technologies group
 C80 Calvet Calorimeter From ambient to 300 C A trademark of KEP Technologies group C80 SENSOR The C80 Calorimeter is one of the most powerful, yet flexible calorimeters available. The high precision Calvet
C80 Calvet Calorimeter From ambient to 300 C A trademark of KEP Technologies group C80 SENSOR The C80 Calorimeter is one of the most powerful, yet flexible calorimeters available. The high precision Calvet
From 1 ppm to 100% Karl Fischer water content determination
 METTLER TOLEDO Karl Fischer Titrators DL39 & DL32 Coulometers DL38 & DL31 Volumeters Oven sample changer Stromboli From 1 ppm to 100% Karl Fischer water content determination Karl Fischer water content
METTLER TOLEDO Karl Fischer Titrators DL39 & DL32 Coulometers DL38 & DL31 Volumeters Oven sample changer Stromboli From 1 ppm to 100% Karl Fischer water content determination Karl Fischer water content
Thermal Safety Software (TSS) series
 Thermal Safety Software (TSS) series the analog-free methodology and software for reaction hazard assessment of Chemical Processes and Products. TSS is the integrated system which covers the entire spectrum
Thermal Safety Software (TSS) series the analog-free methodology and software for reaction hazard assessment of Chemical Processes and Products. TSS is the integrated system which covers the entire spectrum
Easy and reliable KF titrations From 1 ppm to 100%
 AGC Karl Book Fischer 20% Titrators Cyan Volumetric KF Titrators DL31, DL38 Coulometric KF Titrators DL32, DL39 Oven sample changer Stromboli Easy and reliable KF titrations From 1 ppm to 100% Karl Fischer
AGC Karl Book Fischer 20% Titrators Cyan Volumetric KF Titrators DL31, DL38 Coulometric KF Titrators DL32, DL39 Oven sample changer Stromboli Easy and reliable KF titrations From 1 ppm to 100% Karl Fischer
Calorimetric Determination of Reaction Enthalpies
 H + (aq) + OH - q H 2 O Calorimetric Determination of Reaction Enthalpies Purpose: Determine the enthalpy of dissociation of CH 3 COOH CH 3 COOH (aq) CH 3 COO - (aq) + H + (aq) Techniques: Calorimetry
H + (aq) + OH - q H 2 O Calorimetric Determination of Reaction Enthalpies Purpose: Determine the enthalpy of dissociation of CH 3 COOH CH 3 COOH (aq) CH 3 COO - (aq) + H + (aq) Techniques: Calorimetry
1. Starting of a project and entering of basic initial data.
 PROGRAM VISIMIX TURBULENT SV. Example 1. Contents. 1. Starting of a project and entering of basic initial data. 1.1. Opening a Project. 1.2. Entering dimensions of the tank. 1.3. Entering baffles. 1.4.
PROGRAM VISIMIX TURBULENT SV. Example 1. Contents. 1. Starting of a project and entering of basic initial data. 1.1. Opening a Project. 1.2. Entering dimensions of the tank. 1.3. Entering baffles. 1.4.
IMPROVED ADIABATIC CALORIMETRY IN THE PHI-TEC APPARATUS USING AUTOMATED ON-LINE HEAT LOSS COMPENSATION
 # 27 IChemE IMPROVED ADIABATIC CALORIMETRY IN THE PHI-TEC APPARATUS USING AUTOMATED ON-LINE HEAT LOSS COMPENSATION B Kubascikova, D.G. Tee and S.P. Waldram HEL Ltd, 5 Moxon Street, Barnet, Hertfordshire,
# 27 IChemE IMPROVED ADIABATIC CALORIMETRY IN THE PHI-TEC APPARATUS USING AUTOMATED ON-LINE HEAT LOSS COMPENSATION B Kubascikova, D.G. Tee and S.P. Waldram HEL Ltd, 5 Moxon Street, Barnet, Hertfordshire,
881 Compact IC pro / 882 Compact IC plus for ion chromatography. Intelligent. Reliable. Precise.
 Ion analysis 881 Compact IC pro / 882 Compact IC plus for ion chromatography. Intelligent. Reliable. Precise. 881 Compact IC pro and 882 Compact IC plus. Intelligent. Reliable. Precise. Metrohm s top ion
Ion analysis 881 Compact IC pro / 882 Compact IC plus for ion chromatography. Intelligent. Reliable. Precise. 881 Compact IC pro and 882 Compact IC plus. Intelligent. Reliable. Precise. Metrohm s top ion
White Paper. Particle Size Analysis for Process Optimization
 Particle Size Analysis for Process Optimization Particles, crystals, and droplets occur in many chemical processes, across a range of industries, and often pose challenges for scientists and engineers
Particle Size Analysis for Process Optimization Particles, crystals, and droplets occur in many chemical processes, across a range of industries, and often pose challenges for scientists and engineers
HPLC Sample Preparation
 HPLC Sample Preparation Rely on Sartorius solutions and benefit from optimized workflows for clean results mau 400 350 300 250 200 150 100 50 0 50 0.0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 11.0 12.0
HPLC Sample Preparation Rely on Sartorius solutions and benefit from optimized workflows for clean results mau 400 350 300 250 200 150 100 50 0 50 0.0 1.0 2.0 3.0 4.0 5.0 6.0 7.0 8.0 9.0 10.0 11.0 12.0
Reaction Calorimetry as a Tool in Process Development, Research and Safety Analysis
 Reaction Calorimetry as a Tool in Process Development, Research and Safety nalysis Rikard Widell Department of Chemical Engineering II, Lund University, P. O. Box 14, SE-1 Lund, Sweden bstract Reaction
Reaction Calorimetry as a Tool in Process Development, Research and Safety nalysis Rikard Widell Department of Chemical Engineering II, Lund University, P. O. Box 14, SE-1 Lund, Sweden bstract Reaction
PC Control / Touch Control
 PC Control / Touch Control Version 6.0 / 5.840.0150 New Features Manual 8.840.8007EN Metrohm AG CH-9101 Herisau Switzerland Phone +41 71 353 85 85 Fax +41 71 353 89 01 info@metrohm.com www.metrohm.com
PC Control / Touch Control Version 6.0 / 5.840.0150 New Features Manual 8.840.8007EN Metrohm AG CH-9101 Herisau Switzerland Phone +41 71 353 85 85 Fax +41 71 353 89 01 info@metrohm.com www.metrohm.com
SIFT-MS. technology overview
 SIFT-MS SELECTED ION FLOW TUBE MASS SPECTROMETRY technology overview contents 1. An overview of SIFT-MS 2. Principles of SIFT-MS a. Why chemical ionization? b. Why three reagent ions? c. How does SIFT-MS
SIFT-MS SELECTED ION FLOW TUBE MASS SPECTROMETRY technology overview contents 1. An overview of SIFT-MS 2. Principles of SIFT-MS a. Why chemical ionization? b. Why three reagent ions? c. How does SIFT-MS
- Joule (J): SI unit for energy. It's defined based on the equation for kinetic energy. from. mass. velocity
 153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
153 ENERGY UNITS - calorie (cal): the amount of energy required to change the temperature of one gram of water by one degree Celsius (or Kelvin) 1g 1g add one calorie of energy - Calories in food? The
Applications of Reaction Calorimetry in the Chemical Process Industry Reinaldo M. Machado 20 May 2105
 rm 2 technologies LLC Applications of Reaction Calorimetry in the Chemical Process Industry Reinaldo M. Machado 20 May 2105 fundamentals of scale-up Numerous physical and chemical processes interact during
rm 2 technologies LLC Applications of Reaction Calorimetry in the Chemical Process Industry Reinaldo M. Machado 20 May 2105 fundamentals of scale-up Numerous physical and chemical processes interact during
Real-time In Situ FTIR Reaction Analysis Evolution of Technology Current State-of-the-Art. July 23, 2009 CPAC Summer Institute Seattle, WA
 Real-time In Situ FTIR Reaction Analysis Evolution of Technology Current State-of-the-Art July 23, 2009 CPAC Summer Institute Seattle, WA Evolution of Reaction Analysis Technologies Optical modules - Interferometer
Real-time In Situ FTIR Reaction Analysis Evolution of Technology Current State-of-the-Art July 23, 2009 CPAC Summer Institute Seattle, WA Evolution of Reaction Analysis Technologies Optical modules - Interferometer
Ti-Pure TITANIUM DIOXIDE. DETERMINATION OF PARTICLE SIZE DISTRIBUTION OF RPS ( Horiba (LA-900) Procedure) METHOD: T WP
 Ti-Pure TITANIUM DIOXIDE DETERMINATION OF PARTICLE SIZE DISTRIBUTION OF RPS ( Horiba (LA-900) Procedure) METHOD: T4400.274.03WP 1 TITANIUM DIOXIDE (Ti-Pure ) Determination of Particle Size Distribution
Ti-Pure TITANIUM DIOXIDE DETERMINATION OF PARTICLE SIZE DISTRIBUTION OF RPS ( Horiba (LA-900) Procedure) METHOD: T4400.274.03WP 1 TITANIUM DIOXIDE (Ti-Pure ) Determination of Particle Size Distribution
RUNAWAY REACTIONS Experimental Characterization and Vent Sizing
 RUNAWAY REACTIONS Experimental Characterization and Vent Sizing Ron Darby Professor of Chemical Engineering Texas A&M University College Station, TX 77843-3122 (979) 845-3301 r-darby@tamu.edu ARSST CALORIMETER
RUNAWAY REACTIONS Experimental Characterization and Vent Sizing Ron Darby Professor of Chemical Engineering Texas A&M University College Station, TX 77843-3122 (979) 845-3301 r-darby@tamu.edu ARSST CALORIMETER
Comparing the MICROLAB 600 to Volumetric Glassware and Air Displacement Pipettes. June 2011
 Comparing the MICROLAB 600 to tric Glassware and Air Displacement Pipettes June 2011 tric Glassware and Air Displacement Pipettes Table of Contents ``Abstract...3 ``Introduction...4 ``Method & Results...5
Comparing the MICROLAB 600 to tric Glassware and Air Displacement Pipettes June 2011 tric Glassware and Air Displacement Pipettes Table of Contents ``Abstract...3 ``Introduction...4 ``Method & Results...5
ECS8020 ORGANIC ELEMENTAL ANALYZER CHNS-O Analyzer
 ECS8020 ORGANIC ELEMENTAL ANALYZER CHNS-O Analyzer ECS 8020 CHNS-O Analyzer Organic Elemental Analysis ECS 8020 is a C-H-N-S-O Elemental Analyzer Model based on the Dumas combustion method. ECS 8020 is
ECS8020 ORGANIC ELEMENTAL ANALYZER CHNS-O Analyzer ECS 8020 CHNS-O Analyzer Organic Elemental Analysis ECS 8020 is a C-H-N-S-O Elemental Analyzer Model based on the Dumas combustion method. ECS 8020 is
SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS
 SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS ASAP 2020 Plus: Accelerated Surface Area and Porosimetry System The Micromeritics ASAP 2020 Plus integrates a variety of automated gas
SOPHISTICATED DESIGN, INTUITIVE OPERATION, RESEARCH-GRADE RESULTS ASAP 2020 Plus: Accelerated Surface Area and Porosimetry System The Micromeritics ASAP 2020 Plus integrates a variety of automated gas
Assessing Mixing Sensitivities for Scale-up. Matthew Jörgensen Atlanta, 10/20/2014
 Assessing Mixing Sensitivities for Scale-up Matthew Jörgensen Atlanta, 10/20/2014 Nalas: Solutions that Scale! Engineering Services Broad range of services & capabilities supporting chemical process development
Assessing Mixing Sensitivities for Scale-up Matthew Jörgensen Atlanta, 10/20/2014 Nalas: Solutions that Scale! Engineering Services Broad range of services & capabilities supporting chemical process development
PROPULSION LAB MANUAL
 PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
PROPULSION LAB MANUAL Measurement of Calorific Value of a Solid Fuel Sample using a Bomb Calorimeter DEPARTMENT OF AEROSPACE ENGINEERING Indian Institute of Technology Kharagpur CONTENTS 1. Introduction
Color Compensation Set 1
 Color Compensation Set 1 Version 2, March 2012 Solutions for the generation of color compensation objects for the LightCycler System. Order No. A 500 08 For 5 calibration runs. Store in dark at -15 to
Color Compensation Set 1 Version 2, March 2012 Solutions for the generation of color compensation objects for the LightCycler System. Order No. A 500 08 For 5 calibration runs. Store in dark at -15 to
An Introduction to Reaction Calorimetry
 An Introduction to Reaction Calorimetry Syrris A brief history Driven by Productizing Science Founded in 2001 to address the challenges faced by the pharmaceutical industry Syrris is the longest established
An Introduction to Reaction Calorimetry Syrris A brief history Driven by Productizing Science Founded in 2001 to address the challenges faced by the pharmaceutical industry Syrris is the longest established
IN QUALITATIVE ANALYSIS,
 IN QUALITATIVE ANALYSIS, YOU VE ALWAYS HAD TO CHOOSE BETWEEN ACCURACY AND THROUGHPUT. NOW THERE S NO REASON TO ever have to. WATERS QUALITATIVE ANALYSIS solutions Waters ACQUITY UPLC System with the Waters
IN QUALITATIVE ANALYSIS, YOU VE ALWAYS HAD TO CHOOSE BETWEEN ACCURACY AND THROUGHPUT. NOW THERE S NO REASON TO ever have to. WATERS QUALITATIVE ANALYSIS solutions Waters ACQUITY UPLC System with the Waters
How to Safely Quantify a Reaction Exotherm
 How to Safely Quantify a Reaction Exotherm One of the well publicised benefits of Flow Chemistry is the high available surface area and the superior heat transfer. This is particularly important with an
How to Safely Quantify a Reaction Exotherm One of the well publicised benefits of Flow Chemistry is the high available surface area and the superior heat transfer. This is particularly important with an
Focus XC + 6 Microwave capability = Infinity Explore Infinite Possibilities. with the Infinity Microwave Peptide Synthesizer
 Focus XC + 6 Microwave capability = Infinity 2400 Explore Infinite Possibilities with the Infinity 2400 In 1995, when Mr. Hans Tanner began testing microwave peptide synthesis with Dr. Hossain Saneii,
Focus XC + 6 Microwave capability = Infinity 2400 Explore Infinite Possibilities with the Infinity 2400 In 1995, when Mr. Hans Tanner began testing microwave peptide synthesis with Dr. Hossain Saneii,
DYNAMIC VAPOR SORPTION ANALYZER FULLY AUTOMATED COMPACT BENCH-TOP DVS ANALYZER, FOR FAST AND ACCURATE SORPTION MEASUREMENTS.
 DYNAMIC VAPOR SORPTION ANALYZER FULLY AUTOMATED COMPACT BENCH-TOP DVS ANALYZER, FOR FAST AND ACCURATE SORPTION MEASUREMENTS. www.hidenisochema.com The IGAsorp is a fully automated compact bench-top DVS
DYNAMIC VAPOR SORPTION ANALYZER FULLY AUTOMATED COMPACT BENCH-TOP DVS ANALYZER, FOR FAST AND ACCURATE SORPTION MEASUREMENTS. www.hidenisochema.com The IGAsorp is a fully automated compact bench-top DVS
T h e r m a l a n a l y s i s a n d c a l o r i m e t r y s o l u t i o n s
 T h e r m a l a n a l y s i s a n d c a l o r i m e t r y s o l u t i o n s calvet 2 3 Process Safety Our Products The DSC131 evo is a highly robust and flexible DSC designed for busy laboratories with
T h e r m a l a n a l y s i s a n d c a l o r i m e t r y s o l u t i o n s calvet 2 3 Process Safety Our Products The DSC131 evo is a highly robust and flexible DSC designed for busy laboratories with
British American Tobacco Group Research & Development. Method - Determination of ammonia in mainstream smoke
 British American Tobacco Group Research & Development Method - Determination of ammonia in mainstream smoke 1 SCOPE OF APPLICATION The method is applicable to quantitative determination of the yields of
British American Tobacco Group Research & Development Method - Determination of ammonia in mainstream smoke 1 SCOPE OF APPLICATION The method is applicable to quantitative determination of the yields of
This reaction is ENDOTHERMIC. Energy is being transferred from the room/flask/etc. (the SURROUNDINGS) to the reaction itself (the SYSTEM).
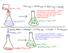 151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
151 This reaction is EXOTHERMIC. Energy is transferred from the reactants and products (the SYSTEM) to the water in the flask, the flask, etc. (the SURROUNDINGS) This reaction is ENDOTHERMIC. Energy is
On the use of Deming and quadratic calibrations in analytical chemistry
 On the use of Deming and quadratic calibrations in analytical chemistry Henry Foden Rand Water October 09-2013 PO Box 3526, Vereeniging, 1930, Republic of South Africa e-mail: hfoden@randwater.co.za Phone:
On the use of Deming and quadratic calibrations in analytical chemistry Henry Foden Rand Water October 09-2013 PO Box 3526, Vereeniging, 1930, Republic of South Africa e-mail: hfoden@randwater.co.za Phone:
(Refer Slide Time: 00:10)
 Chemical Reaction Engineering 1 (Homogeneous Reactors) Professor R. Krishnaiah Department of Chemical Engineering Indian Institute of Technology Madras Lecture No 10 Design of Batch Reactors Part 1 (Refer
Chemical Reaction Engineering 1 (Homogeneous Reactors) Professor R. Krishnaiah Department of Chemical Engineering Indian Institute of Technology Madras Lecture No 10 Design of Batch Reactors Part 1 (Refer
- Kinetic energy: energy of matter in motion. gravity
 148 2500 L of chlorine gas at 25.0 C and 1.00 atm are used to make hydrochloric acid. How many grams of hydrochloric acid could be produced if all the chlorine reacts? 1 - Convert 2500 L chlorine gas to
148 2500 L of chlorine gas at 25.0 C and 1.00 atm are used to make hydrochloric acid. How many grams of hydrochloric acid could be produced if all the chlorine reacts? 1 - Convert 2500 L chlorine gas to
Petrochemical. Transformer Oil Gas Analysis - TOGA
 Petrochemical Transformer Oil Gas Analysis - TOGA www.dps-instruments.com The DPS TOGA GC Systems are designed to analyze oil from electrical insulation materials that may have decomposed under thermal,
Petrochemical Transformer Oil Gas Analysis - TOGA www.dps-instruments.com The DPS TOGA GC Systems are designed to analyze oil from electrical insulation materials that may have decomposed under thermal,
Process Analytical Technology Diagnosis, Optimization and Monitoring of Chemical Processes
 FRAUNHOFER INSTITUTe FoR Chemical Technology ICT Process Analytical Technology Diagnosis, Optimization and Monitoring of Chemical Processes Process Analytical Technology Diagnosis, Optimization and Monitoring
FRAUNHOFER INSTITUTe FoR Chemical Technology ICT Process Analytical Technology Diagnosis, Optimization and Monitoring of Chemical Processes Process Analytical Technology Diagnosis, Optimization and Monitoring
PARTICLE VELOCITY SENSOR FOR IMPROVING SHOT PEENING PROCESS CONTROL
 PARTICLE VELOCITY SENSOR FOR IMPROVING SHOT PEENING PROCESS CONTROL Bill Barker, Kevin Young - Progressive Technologies, Inc. - Grand Rapids, MI U!S Luc Pouliot - Tecnar Automation, Ltee - St-Bruno, Quebec
PARTICLE VELOCITY SENSOR FOR IMPROVING SHOT PEENING PROCESS CONTROL Bill Barker, Kevin Young - Progressive Technologies, Inc. - Grand Rapids, MI U!S Luc Pouliot - Tecnar Automation, Ltee - St-Bruno, Quebec
Use the data in the table to calculate the standard enthalpy of formation of liquid methylbenzene, C 7 H 8. Substance C(s) H 2 (g) C 7 H 8 (l)
 Q1.(a) Define the term standard enthalpy of formation, H f ο (3) (b) Use the data in the table to calculate the standard enthalpy of formation of liquid methylbenzene, C 7 H 8 Substance C(s) H 2 (g) C
Q1.(a) Define the term standard enthalpy of formation, H f ο (3) (b) Use the data in the table to calculate the standard enthalpy of formation of liquid methylbenzene, C 7 H 8 Substance C(s) H 2 (g) C
4 BOMB CALORIMETRY. Chapter 4. Bomb Calorimetry 24 I. PREPARATION
 Chapter 4. Bomb Calorimetry 24 4 BOMB CALORIMETRY I. PREPARATION Read "Principles of Calorimetry" in Ch. VI of Shoemaker, Garland, and Nibler (SGN, pp. 145-151), as well as Experiment 6 (pp. 152-158).
Chapter 4. Bomb Calorimetry 24 4 BOMB CALORIMETRY I. PREPARATION Read "Principles of Calorimetry" in Ch. VI of Shoemaker, Garland, and Nibler (SGN, pp. 145-151), as well as Experiment 6 (pp. 152-158).
DETERMINATION OF REACTION KINETICS USING ONLINE X-RAY DIFFRACTION
 337 DETERMINTION OF RECTION KINETICS USING ONLINE X-RY DIFFRCTION Department of Chemical Engineering, Faculty of Engineering, Lampung University, Bandar Lampung, Indonesia 35145 Received 30 pril 2008;
337 DETERMINTION OF RECTION KINETICS USING ONLINE X-RY DIFFRCTION Department of Chemical Engineering, Faculty of Engineering, Lampung University, Bandar Lampung, Indonesia 35145 Received 30 pril 2008;
News. Flavors & Ingredients. Optimal Moisture Control Guarantees Excellent Quality. Weighing and Analysis in the Laboratory
 Flavors & Ingredients Weighing and Analysis in the Laboratory 6 News Optimal Moisture Control Guarantees Excellent Quality Shanghai Apple Flavour & Fragrance Co., Ltd. is one of the industry leaders in
Flavors & Ingredients Weighing and Analysis in the Laboratory 6 News Optimal Moisture Control Guarantees Excellent Quality Shanghai Apple Flavour & Fragrance Co., Ltd. is one of the industry leaders in
Introduction to Fourier Transform Infrared Spectroscopy
 Introduction to Fourier Transform Infrared Spectroscopy Introduction What is FTIR? FTIR stands for Fourier transform infrared, the preferred method of infrared spectroscopy. In infrared spectroscopy, IR
Introduction to Fourier Transform Infrared Spectroscopy Introduction What is FTIR? FTIR stands for Fourier transform infrared, the preferred method of infrared spectroscopy. In infrared spectroscopy, IR
DI 3500 Discrete Aoalvzer
 DI 3500 Discrete Aoalvzer Automated Chemistry and Ion Analysis ':t O+Analytical:!, A World ofsolutions. - Discrete Analysis Efficient Microscale Chemistry Increasingly laboratories are being challenged
DI 3500 Discrete Aoalvzer Automated Chemistry and Ion Analysis ':t O+Analytical:!, A World ofsolutions. - Discrete Analysis Efficient Microscale Chemistry Increasingly laboratories are being challenged
CHEMICAL ENGINEERING LABORATORY CHEG 4137W/4139W. Reaction Kinetics Saponification of Isopropyl Acetate with Sodium Hydroxide
 CHEMICAL ENGINEERING LABORATORY CHEG 4137W/4139W Reaction Kinetics Saponification of Isopropyl Acetate with Sodium Hydroxide Objective: The purpose of this experiment is to examine and determine the reaction
CHEMICAL ENGINEERING LABORATORY CHEG 4137W/4139W Reaction Kinetics Saponification of Isopropyl Acetate with Sodium Hydroxide Objective: The purpose of this experiment is to examine and determine the reaction
New Dynamic MRM Mode Improves Data Quality and Triple Quad Quantification in Complex Analyses
 New Dynamic MRM Mode Improves Data Quality and Triple Quad Quantification in Complex Analyses Technical Overview Authors Abstract Peter Stone, Thomas Glauner, Frank Kuhlmann, Tim Schlabach and Ken Miller
New Dynamic MRM Mode Improves Data Quality and Triple Quad Quantification in Complex Analyses Technical Overview Authors Abstract Peter Stone, Thomas Glauner, Frank Kuhlmann, Tim Schlabach and Ken Miller
TraC A disruptive new hand-held rapid measurement system to verify pharma equipment surface cleanliness
 TraC A disruptive new hand-held rapid measurement system to verify pharma equipment surface cleanliness Presented by: Ray Reid, President, Photon Systems Come see this at booth # 514 Agenda Present method
TraC A disruptive new hand-held rapid measurement system to verify pharma equipment surface cleanliness Presented by: Ray Reid, President, Photon Systems Come see this at booth # 514 Agenda Present method
CHEM*3440. Thermal Methods. Thermogravimetry. Instrumental Components. Chemical Instrumentation. Thermal Analysis. Topic 14
 Thermal Methods We will examine three thermal analytical techniques: Thermogravimetric Analysis (TGA) CHEM*3440 Chemical Instrumentation Topic 14 Thermal Analysis Differential Thermal Analysis (DTA) Differential
Thermal Methods We will examine three thermal analytical techniques: Thermogravimetric Analysis (TGA) CHEM*3440 Chemical Instrumentation Topic 14 Thermal Analysis Differential Thermal Analysis (DTA) Differential
The New Eppendorf X-Drive - Maximum Performance, Flexibility and Longevity for Demanding Shaker Tasks
 WHITE PAPER No. 47 The New Eppendorf X-Drive - Maximum Performance, Flexibility and Longevity for Demanding Shaker Tasks Ines Hartmann¹, James Jarvis² ¹Eppendorf AG, ²Eppendorf Inc. Executive Summary Temperature
WHITE PAPER No. 47 The New Eppendorf X-Drive - Maximum Performance, Flexibility and Longevity for Demanding Shaker Tasks Ines Hartmann¹, James Jarvis² ¹Eppendorf AG, ²Eppendorf Inc. Executive Summary Temperature
The first address for Titration. TITRONIC 300 and TitroLine 5000
 The first address for Titration TITRONIC 300 and TitroLine 5000 Selection table titration piston burettes TITRONIC and automatic titrators TitroLine The most important features of titrators TitroLine and
The first address for Titration TITRONIC 300 and TitroLine 5000 Selection table titration piston burettes TITRONIC and automatic titrators TitroLine The most important features of titrators TitroLine and
CHEMICAL REACTION ENGINEERING LAB
 CHEMICAL REACTION ENGINEERING LAB EQUIPMENTS 1.CHEMICAL REACTORS SERVICE UNIT The chemical reactors service unit consists of a moulded ABS plinth which is used as a mounting for the chemical reactor to
CHEMICAL REACTION ENGINEERING LAB EQUIPMENTS 1.CHEMICAL REACTORS SERVICE UNIT The chemical reactors service unit consists of a moulded ABS plinth which is used as a mounting for the chemical reactor to
DMA-80L TECHNICAL SPECIFICATIONS
 DMA-80L Advantages Determination of Mercury in water and digestates Unique atomic absorption spectrometer, optical paths with three independent signals Short analysis time, typically less than 2 minutes
DMA-80L Advantages Determination of Mercury in water and digestates Unique atomic absorption spectrometer, optical paths with three independent signals Short analysis time, typically less than 2 minutes
