Supplementary Figure 1 SEM image for the bulk LCO.
|
|
|
- Jemima Allison
- 5 years ago
- Views:
Transcription
1 Supplementary Figure 1 SEM image for the bulk LCO. S1
2 Supplementary Figure 2 TEM and HRTEM images of LCO nanoparticles. (a)-(c) TEM, HRTEM images, and SAED pattern for the 60 nm LCO, respectively. (d)-(f) TEM, HRTEM images, and SAED pattern for the 80 nm LCO, respectively. S2
3 Supplementary Figure 3 Structure of LCO nanoparticles. (a) and (b) The XRD patterns together with the Rietveld refinements for the 200 and 60 nm LCO, respectively. S3
4 M (emu/g) (a) 60 nm 80 nm 200 nm bulk μ eff (μ B ) (b) T (K) nm 80 nm 200 nm bulk Supplementary Figure 4 Magnetic properties of the bulk and nanosized LCO. (a) Temperature dependent magnetization under H = 1 koe and (b) the calculated effective magnetic moment for the nanosized and bulk LCO. S4
5 Supplementary Figure 5 EELS of LCO nanoparticles. (a) Representative positions of EELS acquisition and (b) the corresponding EELS spectra for 60 nm LCO at O-K edge. S5
6 Supplementary Figure 6 Core-shell model. (a) Core-shell model for the spin configurations of LCO nanoparticles. (b) Particle size-dependent volume fraction of shell (surface) layer for LCO nanoparticles. S6
7 Supplementary Figure 7 Long-term stability tests. Chronopotentiometric measurements at J = 10 A g -1 for the bulk and 80 nm LCO. S7
8 Supplementary Figure 8 XPS spectra. (a) Co 2p and (b) O 1s spectra before and after the durability tests for the 80 nm LCO. During the tests, no visible change can be found in the Co 2p spectra, whereas the oxygenated surface species can be found in O 1s spectra. S8
9 V A (cm 3 g -1 STP) (a) S BET = 1.94 m 2 g -1 V A (cm 3 g -1 STP) 10 5 (b) S BET = 2.47 m 2 g V A (cm 3 g -1 STP) (c) P/P 0 S BET = 4.44 m 2 g -1 V A (cm 3 g -1 STP) (d) P/P 0 S BET = m 2 g P/P P/P 0 Supplementary Figure 9 (a)-(d) Nitrogen adsorption-desorption isotherm curves for the bulk, 200, 80, and 60 nm LCO, respectively. S9
10 (a) (e) disk j /ma cm mv s Potential (V vs RHE) disk Δj 1.09V vsrhe /ma cm mf cm Scan rate /mv s -1 (b) (f) disk j /ma cm mv s Potential (V vs RHE) disk Δj 1.09V vsrhe /ma cm mf cm Scan rate /mv s -1 (c) (g) disk j /ma cm mv s Potential (V vs RHE) disk Δj 1.09V vsrhe /ma cm mf cm Scan rate /mv s -1 disk j /ma cm (d) mv s Potential (V vs RHE) disk Δj 1.09V vsrhe /ma cm (h) 1.24 mf cm Scan rate /mv s -1 Supplementary Figure 10 ECSA analyses of the bulk and nanosized LCO. (a)-(d) CVs at different scan rates of in a potential window where no Faradaic processes occur ( V vs. RHE) for the bulk, 200, 80 and 60 nm LCO, respectively. (e)-(h) Charging current density differences ( J = (J + - J - )/2) plotted against scan rates for the bulk, 200, 80 and 60 nm LCO, respectively. The linear slope is equivalent to the double-layer capacitance (C dl ). S10
11 1.0 (b) (a) 0.8 j (ma cm -2 BET ) j (ma cm -2 ) ECSA e g electron e g electron Supplementary Figure 11 OER specific activity as a function of e g electron filling. (a) and (b) OER specific activities at η = 0.49 V by normalized with the BET area and ECSA for the LCO with different e g filings, respectively. Error bars represent s.d. from at least three independent measurements. S11
12 Supplementary Figure 12 Model of water adsorbed on LCO surface. (a) Water adsorbed on top of La on LaO-terminated (001) surface, and (b) water adsorbed on top of Co on CoO 2 -terminated (001) surface. Light green frames represent the periodic boundaries. S12
13 Supplementary Table 1 Lattice parameters a and c, volume of the unit cell (V), x O for oxygen site position (x,0,1/4), and agreement factors R for LCO samples from the Rietveld refinement. LCO-bulk 200 nm 80 nm 60 nm Space group R 3c R 3c R 3c R 3c a (Å) (1) (1) (2) (2) c (Å) (1) (1) (4) (6) V (Å 3 ) (5) (14) (18) (21) x O (9) (12) (11) (12) R Bragg 3.48% 4.50% 4.84% 10.6% R wp 12.8% 13.6% 14.6% 14.8% S13
14 Supplementary Table 2 Effective magnetic moment (μ eff ) and e g orbital occupation for different spin states of Co 3+ ions in LaCoO 3 (LS: low spin; HS: high spin). Spin state LS HS 50% HS + 50% LS 55% HS + 45% LS 60.5% HS +39.5% LS 63.7% HS % LS μ eff (μ B ) e g occupation e g 0 e g 2 e g ~1.0 e g ~1.1 e g ~1.2 e g ~1.27 S14
15 Supplementary Table 3 Adsorption energy and oxygen-metal distance for water molecule adsorbed on LaCoO 3 (001) surfaces with LS and HS surface Co 3+ (minus for exothermic). Termination Adsorption energy (ev) Oxygen-metal distance (Å) LS HS LS HS LaO (001) CoO 2 (001) S15
16 Supplementary Note 1. The temperature dependent magnetizations (M) for the nanosized and bulk LCO under H = 1 koe are shown in Supplementary Fig. 2a. The effective magnetic moments (µ eff ) for all the samples are obtained by 8, where C is Curie constant and obtained from the fittings on the susceptibility (χ = M/H) above 150 K by a Curie-Wiess law (see Main Text). The obtained values are plotted in Supplementary Fig. 2b. Using these values, the volume fractions of Co ions in HS and LS states can be calculated from the relationship: μ gμ B S HS S HS 1 V HS S LS S LS 1 V LS, where S HS (= 2) and S LS (=0) are the S values, and V HS and V LS (= 1 - V HS ) are the volume fractions for Co 3+ ions in HS and LS states, respectively. Consequently, the e g electron (x) can be further calculated by x = S HS x V HS + S LS x V LS = 2 V HS. The calculated results are shown in Supplementary Tab. 2. Supplementary Note 2. A simple core-shell model is shown in Supplementary Fig. 3a. The volume fraction of the shell (surface) layer is roughly estimated by: V S (%) = 100 x [1- (1-2R/D) 3 ], where R and D is the average thickness of surface layer and the diameter of the particle (simplified as a ball), respectively. Using the thickness of surface layer R = 3 nm, the dependence of volume fraction of surface layer (V S ) with particle size (D) is shown in Supplementary Fig. 3b. Assuming that the spin states of Co 3+ ions in the core remain in 50% HS + 50 % LS and that in the shell is in 100% HS, the e g electron (x) can be roughly estimated by: x = (1 V S /100) x (50% x S HS + 50% x S LS ) + V S /100 x 100% x S HS = 1 + V S /100. Based on this model, the estimated e g fillings are about 1.09, 1.21, and 1.27 for the 200, 80, and 60 nm LCO, which are close to those values obtained from the magnetization (Supplementary Tab. 2). Supplementary Note 3. The ECSA was determined by measuring the capacitive current associated with double-layer charging from the scan-rate dependence of cyclic voltammetry (CV) 1-3. For this, the potential window of CV was V vs. RHE. The scan rates were 2, 4, 6, 8, and 10 mv s -1. The double layer capacitance (C dl ) was estimated by plotting the J = (J + - J - )/2 at 1.09 V vs. RHE against the scan rate. The linear slope is C dl, which is about 0.28, 0.47, 0.94 and 1.24 mf cm -2 for the bulk, 200, 80, and 60 nm LCO, respectively. Using a value of 60 μf cm -2 as a common estimate for oxide surfaces 1, 4 and a catalyst loading of 0.25 mg cm -2, the ECSA is estimated to be about 1.9, 3.1, 6.3 and 8.3 m 2 g -1 for the bulk, 200, 80, and 60 nm LCO, respectively. S16
17 Supplementary References 1. Hong, W. T., Risch, M., Stoerzinger, K. A., Grimaud, A., Suntivich, J. & Shao-Horn, Y. Toward the rational design of non-precious transition metal oxides for oxygen electrocatalysis. Energy Environ. Sci. 8, (2015). 2. Merki, D., Vrubel, H., Rovelli, L., Fierro, S. & Hu, X. L. Fe, Co, and Ni ions promote the catalytic activity of amorphous molybdenum sulfide films for hydrogen evolution. Chem. Sci. 3, (2012). 3. Bao, J., Zhang, X. D., Fan, B., Zhang, J. J., Zhou, M., Yang, W. L., Hu, X., Wang, H., Pan, B. C. & Xie, Y. Ultrathin spinel-structured nanosheets rich in oxygen deficiencies for enhanced electrocatalytic water oxidation. Angew. Chem. Int. Ed. 127, (2015). 4. Bockris, J. O. & Otagawa, T. The electrocatalysis of oxygen evolution on perovskites. J. Electrochem. Soc. 131, 290 (1984). S17
Supplementary Figure 1. (a-b) EDX of Mo 2 and Mo 2
 Supplementary Figure 1. (a-b) EDX of Mo 2 C@NPC/NPRGO and Mo 2 C@NPC. Supplementary Figure 2. (a) SEM image of PMo 12 2-PPy, (b) TEM, (c) HRTEM, (d) STEM image and EDX elemental mapping of C, N, P, and
Supplementary Figure 1. (a-b) EDX of Mo 2 C@NPC/NPRGO and Mo 2 C@NPC. Supplementary Figure 2. (a) SEM image of PMo 12 2-PPy, (b) TEM, (c) HRTEM, (d) STEM image and EDX elemental mapping of C, N, P, and
Supporting information
 Supporting information Enhancing electrocatalytic activity of perovskite oxides by tuning cation deficiency for oxygen reduction and evolution reactions Yinlong Zhu, Wei Zhou*, Jie Yu, Yubo Chen, Meilin
Supporting information Enhancing electrocatalytic activity of perovskite oxides by tuning cation deficiency for oxygen reduction and evolution reactions Yinlong Zhu, Wei Zhou*, Jie Yu, Yubo Chen, Meilin
Supplementary Figure 1. (a) XRD pattern of NCUNs. The red lines present the standard nickel hydroxide hydrate (JCPDS No ) peaks, the blue
 Supplementary Figure 1. (a) XRD pattern of NCUNs. The red lines present the standard nickel hydroxide hydrate (JCPDS No. 22-0444) peaks, the blue lines demonstrate the standard cobalt hydroxide (JCPDS
Supplementary Figure 1. (a) XRD pattern of NCUNs. The red lines present the standard nickel hydroxide hydrate (JCPDS No. 22-0444) peaks, the blue lines demonstrate the standard cobalt hydroxide (JCPDS
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Fig. S1 XRD patterns of a-nifeo x
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Fig. S1 XRD patterns of a-nifeo x
Supporting Information for:
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information for: A Highly Efficient Electrocatalyst Based on
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information for: A Highly Efficient Electrocatalyst Based on
Supplementary Information for. High-performance bifunctional porous non-noble metal phosphide catalyst for overall
 Supplementary Information for High-performance bifunctional porous non-noble metal phosphide catalyst for overall water splitting Yu et al. Supplementary Figure 1. A typical TEM image of as-prepared FeP/Ni
Supplementary Information for High-performance bifunctional porous non-noble metal phosphide catalyst for overall water splitting Yu et al. Supplementary Figure 1. A typical TEM image of as-prepared FeP/Ni
Supporting Information
 Supporting Information A General Strategy for the Synthesis of Transition-Metal Phosphide/N-doped Carbon Frameworks for Hydrogen and Oxygen Evolution Zonghua Pu, Chengtian Zhang, Ibrahim Saana Amiinu,
Supporting Information A General Strategy for the Synthesis of Transition-Metal Phosphide/N-doped Carbon Frameworks for Hydrogen and Oxygen Evolution Zonghua Pu, Chengtian Zhang, Ibrahim Saana Amiinu,
Co-vacancy-rich Co 1 x S nanosheets anchored on rgo for high-efficiency oxygen evolution
 Electronic Supplementary Material Co-vacancy-rich Co 1 x S nanosheets anchored on rgo for high-efficiency oxygen evolution Jiaqing Zhu 1, Zhiyu Ren 1 ( ), Shichao Du 1, Ying Xie 1, Jun Wu 1,2, Huiyuan
Electronic Supplementary Material Co-vacancy-rich Co 1 x S nanosheets anchored on rgo for high-efficiency oxygen evolution Jiaqing Zhu 1, Zhiyu Ren 1 ( ), Shichao Du 1, Ying Xie 1, Jun Wu 1,2, Huiyuan
Supporting Information
 Supporting Information Ultrathin Spinel-Structured Nanosheets Rich in Oxygen Deficiencies for Enhanced Electrocatalytic Water Oxidation** Jian Bao, Xiaodong Zhang,* Bo Fan, Jiajia Zhang, Min Zhou, Wenlong
Supporting Information Ultrathin Spinel-Structured Nanosheets Rich in Oxygen Deficiencies for Enhanced Electrocatalytic Water Oxidation** Jian Bao, Xiaodong Zhang,* Bo Fan, Jiajia Zhang, Min Zhou, Wenlong
unique electronic structure for efficient hydrogen evolution
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information Atom-scale dispersed palladium in conductive
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supplementary Information Atom-scale dispersed palladium in conductive
Ni-Mo Nanocatalysts on N-Doped Graphite Nanotubes for Highly Efficient Electrochemical Hydrogen Evolution in Acid
 Supporting Information Ni-Mo Nanocatalysts on N-Doped Graphite Nanotubes for Highly Efficient Electrochemical Hydrogen Evolution in Acid Teng Wang, Yanru Guo, Zhenxing Zhou, Xinghua Chang, Jie Zheng *,
Supporting Information Ni-Mo Nanocatalysts on N-Doped Graphite Nanotubes for Highly Efficient Electrochemical Hydrogen Evolution in Acid Teng Wang, Yanru Guo, Zhenxing Zhou, Xinghua Chang, Jie Zheng *,
Supplementary Information
 Supplementary Information Supplementary Figure 1. a) SEM image of Cu foil after electropolishing (5 µm scale bar). SEM images of Cu foils treated with H 2 plasma at 100W for 2 minutes b) as prepared and
Supplementary Information Supplementary Figure 1. a) SEM image of Cu foil after electropolishing (5 µm scale bar). SEM images of Cu foils treated with H 2 plasma at 100W for 2 minutes b) as prepared and
Supporting Information
 Supporting Information Stabilizing double perovskite for effective bifunctional oxygen electrocatalysis in alkaline conditions Bin Hua a, Yi-Fei Sun a, Meng Li a, Ning Yan b, *, Jian Chen c, Ya-Qian Zhang
Supporting Information Stabilizing double perovskite for effective bifunctional oxygen electrocatalysis in alkaline conditions Bin Hua a, Yi-Fei Sun a, Meng Li a, Ning Yan b, *, Jian Chen c, Ya-Qian Zhang
Supporting Information
 Supporting Information Ultra-thin Alumina Masks Assisted Nanopore Patterning on Monolayer MoS 2 for Highly Catalytic Efficiency in Hydrogen Evolution Reaction Shaoqiang Su, Qingwei Zhou, Zhiqiang Zeng,
Supporting Information Ultra-thin Alumina Masks Assisted Nanopore Patterning on Monolayer MoS 2 for Highly Catalytic Efficiency in Hydrogen Evolution Reaction Shaoqiang Su, Qingwei Zhou, Zhiqiang Zeng,
Supporting Information
 Supporting Information Hierarchical FeNiP @ Ultrathin Carbon Nanoflakes as Alkaline Oxygen Evolution and Acidic Hydrogen Evolution Catalyst for Efficient Water Electrolysis and Organic Decomposition Bowei
Supporting Information Hierarchical FeNiP @ Ultrathin Carbon Nanoflakes as Alkaline Oxygen Evolution and Acidic Hydrogen Evolution Catalyst for Efficient Water Electrolysis and Organic Decomposition Bowei
Supporting Information
 Supporting Information A Superlattice of Alternately Stacked Ni-Fe Hydroxide Nanosheets and Graphene for Efficient Splitting of Water Wei Ma,,, Renzhi Ma,, * Chengxiang Wang, Jianbo Liang, Xiaohe Liu,,
Supporting Information A Superlattice of Alternately Stacked Ni-Fe Hydroxide Nanosheets and Graphene for Efficient Splitting of Water Wei Ma,,, Renzhi Ma,, * Chengxiang Wang, Jianbo Liang, Xiaohe Liu,,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Three-dimensional amorphous tungsten-doped
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Three-dimensional amorphous tungsten-doped
Supporting Information for. Highly active catalyst derived from a 3D foam of Fe(PO 3 ) 2 /Ni 2 P for extremely efficient water oxidation
 Supporting Information for Highly active catalyst derived from a 3D foam of Fe(PO 3 ) 2 /Ni 2 P for extremely efficient water oxidation Haiqing Zhou a,1, Fang Yu a,1, Jingying Sun a, Ran He a, Shuo Chen
Supporting Information for Highly active catalyst derived from a 3D foam of Fe(PO 3 ) 2 /Ni 2 P for extremely efficient water oxidation Haiqing Zhou a,1, Fang Yu a,1, Jingying Sun a, Ran He a, Shuo Chen
Supporting Information. Electronic Modulation of Electrocatalytically Active. Highly Efficient Oxygen Evolution Reaction
 Supporting Information Electronic Modulation of Electrocatalytically Active Center of Cu 7 S 4 Nanodisks by Cobalt-Doping for Highly Efficient Oxygen Evolution Reaction Qun Li, Xianfu Wang*, Kai Tang,
Supporting Information Electronic Modulation of Electrocatalytically Active Center of Cu 7 S 4 Nanodisks by Cobalt-Doping for Highly Efficient Oxygen Evolution Reaction Qun Li, Xianfu Wang*, Kai Tang,
Supporting Information. Bi-functional Catalyst with Enhanced Activity and Cycle Stability for. Rechargeable Lithium Oxygen Batteries
 Supporting Information Hierarchical Mesoporous/Macroporous Perovskite La 0.5 Sr 0.5 CoO 3-x Nanotubes: a Bi-functional Catalyst with Enhanced Activity and Cycle Stability for Rechargeable Lithium Oxygen
Supporting Information Hierarchical Mesoporous/Macroporous Perovskite La 0.5 Sr 0.5 CoO 3-x Nanotubes: a Bi-functional Catalyst with Enhanced Activity and Cycle Stability for Rechargeable Lithium Oxygen
Supplementary Figure 1 Morpholigical properties of TiO 2-x SCs. The statistical particle size distribution (a) of the defective {001}-TiO 2-x SCs and
 Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Supplementary Figure 1. SEM characterization. SEM image shows the freshly made CoSe 2 /DETA nanobelt substrates possess widths of nm and
 Supplementary Figure 1. SEM characterization. SEM image shows the freshly made CoSe 2 /DETA nanobelt substrates possess widths of 100-800 nm and lengths up to several tens of micrometers with flexible,
Supplementary Figure 1. SEM characterization. SEM image shows the freshly made CoSe 2 /DETA nanobelt substrates possess widths of 100-800 nm and lengths up to several tens of micrometers with flexible,
Engineering NiS/Ni 2 P Heterostructures for Efficient Electrocatalytic Water Splitting
 Supporting Information Engineering NiS/Ni 2 P Heterostructures for Efficient Electrocatalytic Water Splitting Xin Xiao, Dekang Huang, Yongqing Fu, Ming Wen, Xingxing Jiang, Xiaowei Lv, Man Li, Lin Gao,
Supporting Information Engineering NiS/Ni 2 P Heterostructures for Efficient Electrocatalytic Water Splitting Xin Xiao, Dekang Huang, Yongqing Fu, Ming Wen, Xingxing Jiang, Xiaowei Lv, Man Li, Lin Gao,
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Adding refractory 5d transition metal W into PtCo
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Adding refractory 5d transition metal W into PtCo
Supplemental Information. In Situ Electrochemical Production. of Ultrathin Nickel Nanosheets. for Hydrogen Evolution Electrocatalysis
 Chem, Volume 3 Supplemental Information In Situ Electrochemical Production of Ultrathin Nickel Nanosheets for Hydrogen Evolution Electrocatalysis Chengyi Hu, Qiuyu Ma, Sung-Fu Hung, Zhe-Ning Chen, Daohui
Chem, Volume 3 Supplemental Information In Situ Electrochemical Production of Ultrathin Nickel Nanosheets for Hydrogen Evolution Electrocatalysis Chengyi Hu, Qiuyu Ma, Sung-Fu Hung, Zhe-Ning Chen, Daohui
Carbon-encapsulated heazlewoodite nanoparticles as highly efficient and durable electrocatalysts for oxygen evolution reactions
 Electronic Supplementary Material Carbon-encapsulated heazlewoodite nanoparticles as highly efficient and durable electrocatalysts for oxygen evolution reactions Mohammad Al-Mamun 1, Huajie Yin 1, Porun
Electronic Supplementary Material Carbon-encapsulated heazlewoodite nanoparticles as highly efficient and durable electrocatalysts for oxygen evolution reactions Mohammad Al-Mamun 1, Huajie Yin 1, Porun
Self-Supported Three-Dimensional Mesoporous Semimetallic WP 2. Nanowire Arrays on Carbon Cloth as a Flexible Cathode for
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Electronic supplementary information Self-Supported Three-Dimensional Mesoporous Semimetallic
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2016 Electronic supplementary information Self-Supported Three-Dimensional Mesoporous Semimetallic
Supporting Information. Engineering Two-Dimensional Mass-Transport Channels
 Supporting Information Engineering Two-Dimensional Mass-Transport Channels of MoS 2 Nanocatalyst towards Improved Hydrogen Evolution Performance Ge Wang a, Jingying Tao a, Yijie Zhang a, Shengping Wang
Supporting Information Engineering Two-Dimensional Mass-Transport Channels of MoS 2 Nanocatalyst towards Improved Hydrogen Evolution Performance Ge Wang a, Jingying Tao a, Yijie Zhang a, Shengping Wang
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Construction of hierarchical Ni-Co-P
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Construction of hierarchical Ni-Co-P
η (mv) J (ma cm -2 ) ma cm
 J (ma cm -2 ) 250 200 150 100 50 0 253 mv@10 ma cm -2-50 0.00 0.05 0.10 0.15 0.20 0.25 0.30 0.35 η (mv) Supplementary Figure 1 Polarization curve of NiSe. S1 FeO x Fe-Se Intensity (a. u.) 720 717 714 711
J (ma cm -2 ) 250 200 150 100 50 0 253 mv@10 ma cm -2-50 0.00 0.05 0.10 0.15 0.20 0.25 0.30 0.35 η (mv) Supplementary Figure 1 Polarization curve of NiSe. S1 FeO x Fe-Se Intensity (a. u.) 720 717 714 711
Supporting Information
 Supporting Information Nest-like NiCoP for Highly Efficient Overall Water Splitting Cheng Du, a Lan Yang, a Fulin Yang, a Gongzhen Cheng a and Wei Luo a,b* a College of Chemistry and Molecular Sciences,
Supporting Information Nest-like NiCoP for Highly Efficient Overall Water Splitting Cheng Du, a Lan Yang, a Fulin Yang, a Gongzhen Cheng a and Wei Luo a,b* a College of Chemistry and Molecular Sciences,
Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (b-f) bright-field images of the microstructure. Only two broad rings
 Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (bf) brightfield images of the microstructure. Only two broad rings were observed in the SAED pattern, as expected for amorphous
Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (bf) brightfield images of the microstructure. Only two broad rings were observed in the SAED pattern, as expected for amorphous
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for. Flexible Zn-Air Batteries
 Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Nickel Sulfides Freestanding Holey Films as Air-Breathing Electrodes for Flexible Zn-Air Batteries Kyle Marcus, 1,# Kun Liang, 1,# Wenhan Niu, 1,# Yang Yang 1,* 1 NanoScience Technology Center, Department
Supporting Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis and Application of Hexagonal Perovskite BaNiO 3 with Quadrivalent
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Synthesis and Application of Hexagonal Perovskite BaNiO 3 with Quadrivalent
Hexagonal-Phase Cobalt Monophosphosulfide for. Highly Efficient Overall Water Splitting
 Supporting Information for Hexagonal-Phase Cobalt Monophosphosulfide for Highly Efficient Overall Water Splitting Zhengfei Dai,,, Hongbo Geng,,, Jiong Wang, Yubo Luo, Bing Li, ǁ Yun Zong, ǁ Jun Yang, Yuanyuan
Supporting Information for Hexagonal-Phase Cobalt Monophosphosulfide for Highly Efficient Overall Water Splitting Zhengfei Dai,,, Hongbo Geng,,, Jiong Wang, Yubo Luo, Bing Li, ǁ Yun Zong, ǁ Jun Yang, Yuanyuan
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/8/e1700732/dc1 This PDF file includes: Supplementary Materials for Oriented assembly of anisotropic nanoparticles into frame-like superstructures Jianwei Nai,
advances.sciencemag.org/cgi/content/full/3/8/e1700732/dc1 This PDF file includes: Supplementary Materials for Oriented assembly of anisotropic nanoparticles into frame-like superstructures Jianwei Nai,
Supporting Information
 Supporting Information Tuning the Electrocatalytic Oxygen Reduction Reaction Activity and Stability of Shaped-Controlled Pt-Ni Nanoparticles by Thermal Annealing Elucidating the Surface Atomic Structural
Supporting Information Tuning the Electrocatalytic Oxygen Reduction Reaction Activity and Stability of Shaped-Controlled Pt-Ni Nanoparticles by Thermal Annealing Elucidating the Surface Atomic Structural
State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing , China
 Electronic Supplementary Material A Co-N/C hollow-sphere electrocatalyst derived from a metanilic CoAl layered double hydroxide for the oxygen reduction reaction, and its active sites in various ph media
Electronic Supplementary Material A Co-N/C hollow-sphere electrocatalyst derived from a metanilic CoAl layered double hydroxide for the oxygen reduction reaction, and its active sites in various ph media
Supporting Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Ultrathin Molybdenum Boride Films for Highly Efficient
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Ultrathin Molybdenum Boride Films for Highly Efficient
Electronic supplementary information. Amorphous carbon supported MoS 2 nanosheets as effective catalyst for electrocatalytic hydrogen evolution
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Amorphous carbon supported MoS 2 nanosheets as effective
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2014 Electronic supplementary information Amorphous carbon supported MoS 2 nanosheets as effective
B.E. (ev)
 a C 1s C=C b O 1s C-O C-O/C=N C=O/C-N O-C=O C=O Co-O 291 289 287 285 283 B.E. (ev) 540 538 536 534 532 530 528 B.E. (ev) Supplementary Figure 1. XPS C 1s and O 1s spectra of the Co-NG. Supplementary Figure
a C 1s C=C b O 1s C-O C-O/C=N C=O/C-N O-C=O C=O Co-O 291 289 287 285 283 B.E. (ev) 540 538 536 534 532 530 528 B.E. (ev) Supplementary Figure 1. XPS C 1s and O 1s spectra of the Co-NG. Supplementary Figure
Nitrogen and sulfur co-doped porous carbon derived from human hair as. highly efficient metal-free electrocatalyst for hydrogen evolution reaction
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Nitrogen and sulfur co-doped porous
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Nitrogen and sulfur co-doped porous
Supporting Information. Direct Observation of Structural Evolution of Metal Chalcogenide in. Electrocatalytic Water Oxidation
 Supporting Information Direct Observation of Structural Evolution of Metal Chalcogenide in Electrocatalytic Water Oxidation Ke Fan *,, Haiyuan Zou, Yue Lu *,, Hong Chen, Fusheng Li, Jinxuan Liu, Licheng
Supporting Information Direct Observation of Structural Evolution of Metal Chalcogenide in Electrocatalytic Water Oxidation Ke Fan *,, Haiyuan Zou, Yue Lu *,, Hong Chen, Fusheng Li, Jinxuan Liu, Licheng
Self-Growth-Templating Synthesis of 3D N,P,Co-Doped. Mesoporous Carbon Frameworks for Efficient Bifunctional
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Self-Growth-Templating Synthesis of
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Self-Growth-Templating Synthesis of
Hot Electron of Au Nanorods Activates the Electrocatalysis of Hydrogen Evolution on MoS 2 Nanosheets
 Supporting Information Available ot Electron of Au Nanorods Activates the Electrocatalysis of ydrogen Evolution on MoS Nanosheets Yi Shi, Jiong Wang, Chen Wang, Ting-Ting Zhai, Wen-Jing Bao, Jing-Juan
Supporting Information Available ot Electron of Au Nanorods Activates the Electrocatalysis of ydrogen Evolution on MoS Nanosheets Yi Shi, Jiong Wang, Chen Wang, Ting-Ting Zhai, Wen-Jing Bao, Jing-Juan
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Experimental section Synthesis of Ni-Co Prussian
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information Experimental section Synthesis of Ni-Co Prussian
Effect of Ball Milling on Electrocatalytic Activity of Ba 0.5 Sr 0.5 Co 0.8 Fe 0.2 O 3 toward Oxygen Evolution Reaction
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry
Supporting Information for
 Supporting Information for Iridium-tungsten Alloy Nanodendrites as ph-universal Water Splitting Electrocatalysts Fan Lv, Jianrui Feng, Kai Wang, Zhipeng Dou, Weiyu Zhang, Jinhui Zhou, Chao Yang, Mingchuan
Supporting Information for Iridium-tungsten Alloy Nanodendrites as ph-universal Water Splitting Electrocatalysts Fan Lv, Jianrui Feng, Kai Wang, Zhipeng Dou, Weiyu Zhang, Jinhui Zhou, Chao Yang, Mingchuan
Carbon Quantum Dots/NiFe Layered Double Hydroxide. Composite as High Efficient Electrocatalyst for Water
 Supplementary Information Carbon Quantum Dots/NiFe Layered Double Hydroxide Composite as High Efficient Electrocatalyst for Water Oxidation Di Tang, Juan Liu, Xuanyu Wu, Ruihua Liu, Xiao Han, Yuzhi Han,
Supplementary Information Carbon Quantum Dots/NiFe Layered Double Hydroxide Composite as High Efficient Electrocatalyst for Water Oxidation Di Tang, Juan Liu, Xuanyu Wu, Ruihua Liu, Xiao Han, Yuzhi Han,
Supporting Information
 Supporting Information A new family of perovskite catalysts for oxygen-evolution reaction in alkaline media: BaNiO 3 and BaNi 0.83 O 2.5 Jin Goo Lee, 1 Jeemin Hwang, 1 Ho Jung Hwang, 2 Ok Sung Jeon, 1
Supporting Information A new family of perovskite catalysts for oxygen-evolution reaction in alkaline media: BaNiO 3 and BaNi 0.83 O 2.5 Jin Goo Lee, 1 Jeemin Hwang, 1 Ho Jung Hwang, 2 Ok Sung Jeon, 1
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information NiSe 2 Pyramids Deposited on N-doped Graphene Encapsulated
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting Information NiSe 2 Pyramids Deposited on N-doped Graphene Encapsulated
Reviewers' comments: Reviewer #1 (Remarks to the Author):
 Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors reported RuCo nanoalloy@n doped graphene electrocatalysts for HER with excellent performance: low overpotentials of 28 mv at 10 ma/cm2,
Reviewers' comments: Reviewer #1 (Remarks to the Author): The authors reported RuCo nanoalloy@n doped graphene electrocatalysts for HER with excellent performance: low overpotentials of 28 mv at 10 ma/cm2,
Supporting Information. High Wettable and Metallic NiFe-Phosphate/Phosphide Catalyst Synthesized by
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information High Wettable and Metallic NiFe-Phosphate/Phosphide
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information High Wettable and Metallic NiFe-Phosphate/Phosphide
Honeycomb-like Interconnected Network of Nickel Phosphide Hetero-nanoparticles
 Supporting Information Honeycomb-like Interconnected Network of Nickel Phosphide Hetero-nanoparticles with Superior Electrochemical Performance for Supercapacitors Shude Liu a, Kalimuthu Vijaya Sankar
Supporting Information Honeycomb-like Interconnected Network of Nickel Phosphide Hetero-nanoparticles with Superior Electrochemical Performance for Supercapacitors Shude Liu a, Kalimuthu Vijaya Sankar
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/6/eaap9360/dc1 Supplementary Materials for Oxygen-deficient triple perovskites as highly active and durable bifunctional electrocatalysts for oxygen electrode
advances.sciencemag.org/cgi/content/full/4/6/eaap9360/dc1 Supplementary Materials for Oxygen-deficient triple perovskites as highly active and durable bifunctional electrocatalysts for oxygen electrode
Supporting Information
 Supporting Information MoSe2 embedded CNT-Reduced Graphene Oxide (rgo) Composite Microsphere with Superior Sodium Ion Storage and Electrocatalytic Hydrogen Evolution Performances Gi Dae Park, Jung Hyun
Supporting Information MoSe2 embedded CNT-Reduced Graphene Oxide (rgo) Composite Microsphere with Superior Sodium Ion Storage and Electrocatalytic Hydrogen Evolution Performances Gi Dae Park, Jung Hyun
Supplementary Information. Unusual High Oxygen Reduction Performance in All-Carbon Electrocatalysts
 Supplementary Information Unusual High Oxygen Reduction Performance in All-Carbon Electrocatalysts Wei Wei 1, 4,, Ying Tao 1, 4,, Wei Lv 2,, Fang-Yuan Su 2, Lei Ke 2, Jia Li 2, Da-Wei Wang 3, *, Baohua
Supplementary Information Unusual High Oxygen Reduction Performance in All-Carbon Electrocatalysts Wei Wei 1, 4,, Ying Tao 1, 4,, Wei Lv 2,, Fang-Yuan Su 2, Lei Ke 2, Jia Li 2, Da-Wei Wang 3, *, Baohua
Reviewers' Comments: Reviewer #1 (Remarks to the Author)
 Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript reports the synthesis of a series of Mo2C@NPC-rGO hybrid HER electrocatalysts by employing the precursor of PMo12 (H3PMo12O40)-PPy/rGO
Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript reports the synthesis of a series of Mo2C@NPC-rGO hybrid HER electrocatalysts by employing the precursor of PMo12 (H3PMo12O40)-PPy/rGO
Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The. scale bars are 5 nm.
 Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The scale bars are 5 nm. S1 Supplementary Figure 2. TEM image of PtNi/Ni-B composite obtained under N 2 protection.
Supplementary Figure 1. HRTEM images of PtNi / Ni-B composite exposed to electron beam. The scale bars are 5 nm. S1 Supplementary Figure 2. TEM image of PtNi/Ni-B composite obtained under N 2 protection.
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Mesoporous cobalt-iron-organic frameworks: plasma-enhanced
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Mesoporous cobalt-iron-organic frameworks: plasma-enhanced
Operando Spectroscopic Analysis of an Amorphous Cobalt Sulfide Hydrogen Evolution Electrocatalyst
 Supporting information for: Operando Spectroscopic Analysis of an Amorphous Cobalt Sulfide Hydrogen Evolution Electrocatalyst Nikolay Kornienko 1, Joaquin Resasco 2, Nigel Becknell 1, Chang-Ming Jiang
Supporting information for: Operando Spectroscopic Analysis of an Amorphous Cobalt Sulfide Hydrogen Evolution Electrocatalyst Nikolay Kornienko 1, Joaquin Resasco 2, Nigel Becknell 1, Chang-Ming Jiang
Mesoporous N-Doped Carbons Prepared with Thermally Removable Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction
 Supporting Information Mesoporous N-Doped Carons Prepared with Thermally Removale Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction Wenhan Niu, a Ligui Li,* a Xiaojun Liu,
Supporting Information Mesoporous N-Doped Carons Prepared with Thermally Removale Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction Wenhan Niu, a Ligui Li,* a Xiaojun Liu,
Metal-Organic Framework Immobilized Cobalt Oxide Nanoparticles
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information Metal-Organic Framework Immobilized Cobalt Oxide
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Supporting information Metal-Organic Framework Immobilized Cobalt Oxide
Nanosheet-Constructed Porous BiOCl with Dominant {001} Facets for Superior Photosensitized Degradation
 Electronic Supplementary Information Nanosheet-Constructed Porous BiOCl with Dominant {001} Facets for Superior Photosensitized Degradation Dong-Hong Wang, ab Gui-Qi Gao, b Yue-Wei Zhang, a Li-Sha Zhou,
Electronic Supplementary Information Nanosheet-Constructed Porous BiOCl with Dominant {001} Facets for Superior Photosensitized Degradation Dong-Hong Wang, ab Gui-Qi Gao, b Yue-Wei Zhang, a Li-Sha Zhou,
Supporting Information. for Water Splitting. Guangxing Zhang, Jie Yang, Han Wang, Haibiao Chen, Jinlong Yang, and Feng Pan
 Supporting Information Co 3 O 4-δ Quantum Dots as a Highly Efficient Oxygen Evolution Reaction Catalyst for Water Splitting Guangxing Zhang, Jie Yang, Han Wang, Haibiao Chen, Jinlong Yang, and Feng Pan
Supporting Information Co 3 O 4-δ Quantum Dots as a Highly Efficient Oxygen Evolution Reaction Catalyst for Water Splitting Guangxing Zhang, Jie Yang, Han Wang, Haibiao Chen, Jinlong Yang, and Feng Pan
Rh 3d. Co 2p. Binding Energy (ev) Binding Energy (ev) (b) (a)
 Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
One-pot synthesis of bi-metallic PdRu tripods as an efficient catalyst for. electrocatalytic nitrogen reduction to ammonia
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information for One-pot synthesis of bi-metallic PdRu tripods
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information for One-pot synthesis of bi-metallic PdRu tripods
Revelation of the Excellent Intrinsic Activity. Evolution Reaction in Alkaline Medium
 Supporting Information Revelation of the Excellent Intrinsic Activity of MoS2 NiS MoO3 Nanowires for Hydrogen Evolution Reaction in Alkaline Medium Chuanqin Wang a,b, Bin Tian b, Mei Wu b, Jiahai Wang
Supporting Information Revelation of the Excellent Intrinsic Activity of MoS2 NiS MoO3 Nanowires for Hydrogen Evolution Reaction in Alkaline Medium Chuanqin Wang a,b, Bin Tian b, Mei Wu b, Jiahai Wang
Chemicals. Nickel foam (NF, thickness 1.6 mm, bulk density 0.45 g cm -3, porosity
 Supplementary Methods Chemicals. Nickel foam (NF, thickness 1.6 mm, bulk density 0.45 g cm -3, porosity 95%), stainless steel mesh (300 mesh, bulk density 0.25 g cm -3, wire diameter 0.04 mm, open area
Supplementary Methods Chemicals. Nickel foam (NF, thickness 1.6 mm, bulk density 0.45 g cm -3, porosity 95%), stainless steel mesh (300 mesh, bulk density 0.25 g cm -3, wire diameter 0.04 mm, open area
Pomegranate-Like N, P-Doped Nanospheres as Highly Active Electrocatalysts for Alkaline Hydrogen Evolution
 Supporting Information Pomegranate-Like N, P-Doped Mo2C@C Nanospheres as Highly Active Electrocatalysts for Alkaline Hydrogen Evolution Yu-Yun Chen,,,# Yun Zhang,,# Wen-Jie Jiang,, Xing Zhang,, Zhihui
Supporting Information Pomegranate-Like N, P-Doped Mo2C@C Nanospheres as Highly Active Electrocatalysts for Alkaline Hydrogen Evolution Yu-Yun Chen,,,# Yun Zhang,,# Wen-Jie Jiang,, Xing Zhang,, Zhihui
Trifunctional Ni-N/P-O-codoped graphene electrocatalyst enables
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Trifunctional Ni-N/P-O-codoped graphene electrocatalyst
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2019 Supporting Information Trifunctional Ni-N/P-O-codoped graphene electrocatalyst
B-site doping effects of NdBa 0.75 Ca 0.25 Co 2 O 5+δ double perovskite catalysts for oxygen evolution and reduction reactions
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 B-site doping effects of NdBa 0.75 Ca 0.25 Co 2 O 5+δ double perovskite
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 B-site doping effects of NdBa 0.75 Ca 0.25 Co 2 O 5+δ double perovskite
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Directly anchoring 2D NiCo metal-organic frameworks
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Directly anchoring 2D NiCo metal-organic frameworks
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Experimental Section Materials: Ti
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Experimental Section Materials: Ti
Boosting the hydrogen evolution performance of ruthenium clusters. through synergistic coupling with cobalt phosphide
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Boosting the hydrogen evolution
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Boosting the hydrogen evolution
One-Step Facile Synthesis of Cobalt Phosphides for Hydrogen Evolution Reaction Catalyst in Acidic and Alkaline Medium
 Supporting Information One-Step Facile Synthesis of Cobalt Phosphides for Hydrogen Evolution Reaction Catalyst in Acidic and Alkaline Medium Afriyanti Sumboja, a Tao An, a Hai Yang Goh, b Mechthild Lübke,
Supporting Information One-Step Facile Synthesis of Cobalt Phosphides for Hydrogen Evolution Reaction Catalyst in Acidic and Alkaline Medium Afriyanti Sumboja, a Tao An, a Hai Yang Goh, b Mechthild Lübke,
Supporting Information
 Supporting Information Bio-Inspired Engineering of Cobalt-Phosphonate Nanosheets for Robust Hydrogen Evolution Reaction Zhong-Sheng Cai, 1, Yi Shi, 2, Song-Song Bao, 1 Yang Shen, 1 Xing-Hua Xia,*,2 and
Supporting Information Bio-Inspired Engineering of Cobalt-Phosphonate Nanosheets for Robust Hydrogen Evolution Reaction Zhong-Sheng Cai, 1, Yi Shi, 2, Song-Song Bao, 1 Yang Shen, 1 Xing-Hua Xia,*,2 and
Phytic Acid-Assisted Formation of Hierarchical Porous CoP/C Nanoboxes for Enhanced Lithium Storage and Hydrogen Generation
 Phytic Acid-Assisted Formation of Hierarchical Porous CoP/C Nanoboxes for Enhanced Lithium Storage and Hydrogen Generation Xuxu Wang, ab Zhaolin Na, a Dongming Yin, a Chunli Wang, ab Yaoming Wu, a Gang
Phytic Acid-Assisted Formation of Hierarchical Porous CoP/C Nanoboxes for Enhanced Lithium Storage and Hydrogen Generation Xuxu Wang, ab Zhaolin Na, a Dongming Yin, a Chunli Wang, ab Yaoming Wu, a Gang
Electronic Supplementary Information Oxygen reduction reaction on neighboring Fe-N 4 and quaternary-n sites of pyrolized Fe/N/C catalyst
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information Oxygen reduction reaction on neighboring Fe-N
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information Oxygen reduction reaction on neighboring Fe-N
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Two-dimensional CoNi nanoparticles@s,n-doped
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Two-dimensional CoNi nanoparticles@s,n-doped
Supporting information
 a Supporting information Core-Shell Nanocomposites Based on Gold Nanoparticle@Zinc-Iron- Embedded Porous Carbons Derived from Metal Organic Frameworks as Efficient Dual Catalysts for Oxygen Reduction and
a Supporting information Core-Shell Nanocomposites Based on Gold Nanoparticle@Zinc-Iron- Embedded Porous Carbons Derived from Metal Organic Frameworks as Efficient Dual Catalysts for Oxygen Reduction and
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Jaemin Kim, Xi Yin, Kai-Chieh Tsao, Shaohua Fang and Hong Yang *
 Jaemin Kim, Xi Yin, Kai-Chieh Tsao, Shaohua Fang and Hong Yang * Department of Chemical and Biomolecular Engineering, University of Illinois at Urbana-Champaign, 114 Roger Adams Laboratory, MC-712, 600
Jaemin Kim, Xi Yin, Kai-Chieh Tsao, Shaohua Fang and Hong Yang * Department of Chemical and Biomolecular Engineering, University of Illinois at Urbana-Champaign, 114 Roger Adams Laboratory, MC-712, 600
Supplemental Information (SI): Cobalt-iron (oxy)hydroxide oxygen evolution electrocatalysts: The role of
 Supplemental Information (SI: Cobalt-iron (oxyhydroxide oxygen evolution electrocatalysts: The role of structure and composition on activity, stability, and mechanism Michaela S. Burke, Matthew G. Kast,
Supplemental Information (SI: Cobalt-iron (oxyhydroxide oxygen evolution electrocatalysts: The role of structure and composition on activity, stability, and mechanism Michaela S. Burke, Matthew G. Kast,
Supporting Information. Engineering the Composition and Crystallinity of Molybdenum Sulfide for High-performance Electrocatalytic Hydrogen Evolution
 Supporting Information Engineering the Composition and Crystallinity of Molybdenum Sulfide for High-performance Electrocatalytic Hydrogen Evolution Yanpeng Li 1,2 *, Yifei Yu 2, Robert A. Nielsen 3, William
Supporting Information Engineering the Composition and Crystallinity of Molybdenum Sulfide for High-performance Electrocatalytic Hydrogen Evolution Yanpeng Li 1,2 *, Yifei Yu 2, Robert A. Nielsen 3, William
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information Bismuth Nanodendrites as High Performance Electrocatalysts
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2016 Supporting Information Bismuth Nanodendrites as High Performance Electrocatalysts
Efficient Hydrogen Evolution. University of Central Florida, 4000 Central Florida Blvd. Orlando, Florida, 32816,
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2017 MoS 2 /TiO 2 Heterostructures as Nonmetal Plasmonic Photocatalysts for Highly
Supplementary Figure S1. AFM image and height profile of GO. (a) AFM image
 Supplementary Figure S1. AFM image and height profile of GO. (a) AFM image and (b) height profile of GO obtained by spin-coating on silicon wafer, showing a typical thickness of ~1 nm. 1 Supplementary
Supplementary Figure S1. AFM image and height profile of GO. (a) AFM image and (b) height profile of GO obtained by spin-coating on silicon wafer, showing a typical thickness of ~1 nm. 1 Supplementary
Electronic Supplementary Information:
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information: Platinum-Nickel Nanowire Catalysts
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information: Platinum-Nickel Nanowire Catalysts
Supplementary Figure S1: Particle size distributions of the Pt ML /Pd 9 Au 1 /C
 a 2 15 before cycle test mean particle size: 3.8 ± 1.2 nm b 2 15 after.6v - 1.V 1k cycle test mean particle size: 4.1 ± 1.5 nm Number 1 total number: 558 Number 1 total number: 554 5 5 1 2 3 4 5 6 7 8
a 2 15 before cycle test mean particle size: 3.8 ± 1.2 nm b 2 15 after.6v - 1.V 1k cycle test mean particle size: 4.1 ± 1.5 nm Number 1 total number: 558 Number 1 total number: 554 5 5 1 2 3 4 5 6 7 8
Supplementary Information
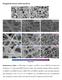 Supplementary Information Supplementary Figure 1. SEM images of samples. (a) PBC-0, and (b) PBSCF-0 powders after calcination; (c-e) electrospun PBSCF precursor fibers with controlled diameters before
Supplementary Information Supplementary Figure 1. SEM images of samples. (a) PBC-0, and (b) PBSCF-0 powders after calcination; (c-e) electrospun PBSCF precursor fibers with controlled diameters before
Supporting Information. Phenolic/resin assisted MOFs derived hierarchical Co/N-doping carbon
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry
Effect of Chloride Anions on the Synthesis and. Enhanced Catalytic Activity of Silver Nanocoral
 Supporting Information Effect of Chloride Anions on the Synthesis and Enhanced Catalytic Activity of Silver Nanocoral Electrodes for CO 2 Electroreduction Polyansky* Yu-Chi Hsieh, Sanjaya D. Senanayake,
Supporting Information Effect of Chloride Anions on the Synthesis and Enhanced Catalytic Activity of Silver Nanocoral Electrodes for CO 2 Electroreduction Polyansky* Yu-Chi Hsieh, Sanjaya D. Senanayake,
A General Approach to Ultrathin NiM (M = Fe, Co, Mn) Hydroxide Nanosheets as High-Performance Low-Cost. Electrocatalysts for Overall Water Splitting
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting information for A General Approach to Ultrathin NiM (M = Fe,
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Supporting information for A General Approach to Ultrathin NiM (M = Fe,
Tailorable and Wearable Textile Devices for Solar Energy Harvesting and Simultaneous Storage
 Supporting Information Tailorable and Wearable Textile Devices for Solar Energy Harvesting and Simultaneous Storage Zhisheng Chai,, Nannan Zhang,, Peng Sun, Yi Huang, Chuanxi Zhao, Hong Jin Fan, Xing Fan,*,
Supporting Information Tailorable and Wearable Textile Devices for Solar Energy Harvesting and Simultaneous Storage Zhisheng Chai,, Nannan Zhang,, Peng Sun, Yi Huang, Chuanxi Zhao, Hong Jin Fan, Xing Fan,*,
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Na3V2(PO4)2F3-SWCNT: A High Voltage Cathode for
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Na3V2(PO4)2F3-SWCNT: A High Voltage Cathode for
Facile synthesis of porous nitrogen-doped holey graphene as an efficient metal-free catalyst for the oxygen reduction reaction
 Electronic Supplementary Material Facile synthesis of porous nitrogen-doped holey graphene as an efficient metal-free catalyst for the oxygen reduction reaction Li Qin 1,2,5, Ruimin Ding 1,2, Huixiang
Electronic Supplementary Material Facile synthesis of porous nitrogen-doped holey graphene as an efficient metal-free catalyst for the oxygen reduction reaction Li Qin 1,2,5, Ruimin Ding 1,2, Huixiang
Co 3 O 4 Nanocrystals on Single-Walled Carbon Nanotubes as a Highly Efficient Oxygen-Evolving Catalyst
 Nano Res 95 Electronic Supplementary Material Co 3 O 4 Nanocrystals on Single-Walled Carbon Nanotubes as a Highly Efficient Oxygen-Evolving Catalyst Jian Wu 1, Yan Xue 1, Xin Yan 1, Wensheng Yan 2, Qingmei
Nano Res 95 Electronic Supplementary Material Co 3 O 4 Nanocrystals on Single-Walled Carbon Nanotubes as a Highly Efficient Oxygen-Evolving Catalyst Jian Wu 1, Yan Xue 1, Xin Yan 1, Wensheng Yan 2, Qingmei
Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT) sample. (a, b) TEM images of CNT; (c) EDS of CNT.
 1 Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT sample. (a, b TEM images of CNT; (c EDS of CNT. Cobalt is not detected in the original CNT sample (Note: The accidentally
1 Supplementary Figure 1 Morphology and composition of the original carbon nanotube (CNT sample. (a, b TEM images of CNT; (c EDS of CNT. Cobalt is not detected in the original CNT sample (Note: The accidentally
