Partial Periodic Table
|
|
|
- Allen Johns
- 5 years ago
- Views:
Transcription
1 CEM 33, Fall 00 Professor Walba Third our Exam November 8, 00 Scores: ) 0 ) 0 3) 0 4) 0 5) 0 00 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, have neither given nor received unauthorized assistance. Name (printed): KEY Signature: Recitation TA Name: Recitation day and time: This is a closed-book exam. The use of notes, calculators, scratch paper, or cell phones will not be allowed during the exam. You may use models brought in a clear ziplock bag. Please put all you answers on the test. Use the backs of the pages for scratch. PLEASE read the questions very carefully! Partial Periodic Table A 8A e A 3A 4A 5A 6A 7A Li Be B C N F Ne Na Mg 3 Al 4 Si 5 P 6 S 7 8 Ar Page of 9
2 ) (0 pts) A. Name the major mechanistic pathway (S N, S N, E, or E) for each of the following reactions. For each reaction the solvent is give under the arrow. C 3 + C 3 C 3 3 C C 3 S N C 3 E C 3 3 C S N each d) C 3 + C 3 E B. For each of the following pairs of nucleophiles, indicate by number the stronger nucleophile in the ethanol solvent (polar, proti, and in DMF solvent (polar, aproti. Each box should have a or a written inside. C 3 C (Et) Solvent N Solvent (DMF) F C 3 C S C 3 C each C 3 C CF 3 C d) C 3 C C 3 C Page of 9 6
3 ) -continued- C. Give the product of each of the following reactions. reaction N + C 3 solvent N reaction P + C 3 solvent P each D. Which of the reactions in part C is faster in methanol solvent? E. Which of the reactions in part C is faster in DMF solvent? Page 3 of 9 4
4 ) (0 pts) Give the single major organic product of each of the following reactions. f appropriate, carefully show stereochemistry using wedges and dashes. f a racemate is formed, show only one enantiomer of the product, and label it rac. Assume chiral starting materials are enantiomerically pure unless they are labeled rac. 4 each C 3 C 3 S 4 - C, Zn-Cu ether rac d) C 3 C 3 e) 3 C 3 C 0 Page 4 of 9
5 3 (0 pts) Propose reagents for accomplishing each of the following reactions. Make your reactions efficient (i.e. the target product should be the major product). More than one step reaction sequences may be required. Be careful to indicate the separate steps in the sequence. 4 each Na S C 3 S Na, acetone Na NaCN CN d) Mg, ether C 3 e) 0 Page 5 of 9
6 4) (0 pts) A) For each of the following pairs of isomers, circle the more stable one. f the isomers have the same stability, label them same. 4 each Page 6 of 9
7 4) -continued- B) Dissolving -bromo--methylbutane () in methanol solvent gives two alkene products, - methyl--butene (), and -methyl--butene (3) as shown below. For this question ignore other possible products. C 3 (solvent) pts 3 Circle the major alkene product of this reaction. Carefully complete the energy diagram below for this reaction. Assume both alkene products and 3 are more stable than the starting bromide, and that the reaction is irreversible. Be careful to indicate the correct relative stability of the two product alkenes and 3. gnore what happens to the. E 6 pts 3 Reaction Coordinate 8 Page 7 of 9
8 5) (0 pts) Propose arrow-pushing mechanisms for each of the following transformations. Show all intermediates in your mechanisms, but do not show transition states. Be sure structures are complete, including all loan pairs. 5 each C 3, C C C 0 Page 8 of 9
9 5) - continued C 3 C 3 5 each C 3 C 3 C 3 d) S 4, + 0 Page 9 of 9
Partial Periodic Table
 CEM 3311, Fall 2011 Professor Walba Third our Exam November 17, 2011 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither
CEM 3311, Fall 2011 Professor Walba Third our Exam November 17, 2011 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither
Partial Periodic Table
 Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Second Hour Exam October 18, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Second Hour Exam October 18, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
Partial Periodic Table
 CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
CHEM 33-00, Fall 03 Professor Walba Third Hour Exam November 9, 03 scores: ) ) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither given nor received
Partial Periodic Table
 Easily Legible Printed Name: CHEM 3451, Spring 2018 Professor Walba Second Hour Exam March 13, 2018 scores: 1) 2) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student,
Easily Legible Printed Name: CHEM 3451, Spring 2018 Professor Walba Second Hour Exam March 13, 2018 scores: 1) 2) 3) 4) 5) CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student,
Partial Periodic Table
 Easily Legible Printed Name: CEM 3351 (100), all 2015 Professor Walba Third our Exam November 17, 2015 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado
Easily Legible Printed Name: CEM 3351 (100), all 2015 Professor Walba Third our Exam November 17, 2015 scores: 1) 20 2) 20 3) 20 4) 20 5) 20 100 CU onor Code Pledge: n my honor, as a University of Colorado
Partial Periodic Table
 Printed ame: CEM 3371, Spring 2015 Professor Walba First our Exam March 10, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither given nor received unauthorized
Printed ame: CEM 3371, Spring 2015 Professor Walba First our Exam March 10, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither given nor received unauthorized
Partial Periodic Table
 Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba econd our Exam ctober 20, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither given
Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba econd our Exam ctober 20, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder tudent, I have neither given
Partial Periodic Table
 CEM 33-00, Fall 203 Professor Walba First our Exam September 24, 203 scores: ) 20 2) 20 3) 20 4) 20 5) 20 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
CEM 33-00, Fall 203 Professor Walba First our Exam September 24, 203 scores: ) 20 2) 20 3) 20 4) 20 5) 20 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Partial Periodic Table
 Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CEM 3351 (100), Fall 2015 Professor Walba First our Exam September 22, 2015 CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student, I have neither
Partial Periodic Table
 Easily Legible Printed Name: CEM 3311 (300), Fall 2014 Professor Walba First our Exam September 23, 2014 scores: 1) 2) 3) 4) 5) CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student,
Easily Legible Printed Name: CEM 3311 (300), Fall 2014 Professor Walba First our Exam September 23, 2014 scores: 1) 2) 3) 4) 5) CU onor Code Pledge: n my honor, as a University of Colorado at Boulder Student,
Partial Periodic Table
 Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Third Hour Exam November 15, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
Easily Legible Printed Name: CHEM 3351 (100), Fall 2016 Professor Walba Third Hour Exam November 15, 2016 CU Honor Code Pledge: On my honor, as a University of Colorado at Boulder Student, I have neither
CHEM Exam 2 October 25, 2007
 CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
CEM 3311-200 Exam 2 October 25, 2007 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on
First 2-Hour Exam. By printing your name below, you pledge that
 Chem 3331 Fall 2004 Exam # 1 1 Name First 2-our Exam By printing your name below, you pledge that "n my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized
Chem 3331 Fall 2004 Exam # 1 1 Name First 2-our Exam By printing your name below, you pledge that "n my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized
NOTE: Exams will be returned by your TA in recitation!
 CEM 3311-200 Exam 2 Fall 2008 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on this work."
CEM 3311-200 Exam 2 Fall 2008 By printing my name below, I pledge that "On my honor, as a University of Colorado at Boulder student, I have neither given nor received unauthorized assistance on this work."
Partial Periodic Table
 EMITY 3331, pring 2005 Professor Walba Final Exam, May 3 U onor ode Pledge: n my honor, as a University of olorado at oulder tudent, I have neither given nor received unauthorized assistance. scores: 1)
EMITY 3331, pring 2005 Professor Walba Final Exam, May 3 U onor ode Pledge: n my honor, as a University of olorado at oulder tudent, I have neither given nor received unauthorized assistance. scores: 1)
Partial Periodic Table 1A
 CEMISTRY 3311, all 2001 Professor Walba irst our Exam September 27, 2001 scores: 1) 20 2) 20 3) 15 This is a closed-book "open model" exam. You may use models, but no notes or books. Please put all your
CEMISTRY 3311, all 2001 Professor Walba irst our Exam September 27, 2001 scores: 1) 20 2) 20 3) 15 This is a closed-book "open model" exam. You may use models, but no notes or books. Please put all your
CHEM 3311 Exam 2 ANSWER KEY
 CEM 3311 Exam 2 ANSWER KEY March 11, 2014 Time: 2 ours Please sign the onor Pledge h in this sheet with your scantron. I pledge that n my honor, as a University of Colorado-Boulder student, I have neither
CEM 3311 Exam 2 ANSWER KEY March 11, 2014 Time: 2 ours Please sign the onor Pledge h in this sheet with your scantron. I pledge that n my honor, as a University of Colorado-Boulder student, I have neither
Exam 3 November 17, 2005 Professor Rebecca Hoenigman
 EM 3311-200, Fall 2005 Exam 3 November 17, 2005 Professor Rebecca oenigman Average Score = 54.8 igh Score = 98 Low Score = 8 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits
EM 3311-200, Fall 2005 Exam 3 November 17, 2005 Professor Rebecca oenigman Average Score = 54.8 igh Score = 98 Low Score = 8 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits
FALL 2018 CEM 251: Organic Chemistry I Sections September 25, 2018
 FALL 2018 CEM 251: Organic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only
FALL 2018 CEM 251: Organic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only
!!!! Organic Chemistry CHM 224. Exam I Questions
 ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
ld Exam I Questions - CHM 224 rganic Chemistry CHM 224 Exam I Questions This set of questions is a compilation of old exams, and does not represent the typical length of an exam - there are more examples,
FALL 2018 CEM 251: Organic Chemistry I Sections September 25, 2018
 ALL 2018 CEM 251: rganic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only what
ALL 2018 CEM 251: rganic Chemistry Sections 25-36 TUE-TU 8:00 9:20 AM September 25, 2018 Name: Section: PD: TA: This is a closed book and note examination. f boxes are provided for your answers, only what
Final Exam Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3311-100 Spring 2007 Final Exam Professor R. oenigman Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation number, day,
I pledge to uphold the CU onor Code: CEM 3311-100 Spring 2007 Final Exam Professor R. oenigman Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation number, day,
Chemistry 51 Exam #3. Name KEY November 20, 2001
 Chemistry 51 Exam #3 Name KEY November 20, 2001 This exam has nine (9) questions. Please check before beginning to make sure no questions are missing. All scratch work must be done on the attached blank
Chemistry 51 Exam #3 Name KEY November 20, 2001 This exam has nine (9) questions. Please check before beginning to make sure no questions are missing. All scratch work must be done on the attached blank
Final Exam Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3311-200 Fall 2006 Final Exam Professor. oenigman Average Score = 145 igh Score = 235 Low Score = 27 Signature Name (printed) Last four digits of your student ID
I pledge to uphold the CU onor Code: CEM 3311-200 Fall 2006 Final Exam Professor. oenigman Average Score = 145 igh Score = 235 Low Score = 27 Signature Name (printed) Last four digits of your student ID
CHEM Exam 2 ANSWER KEY
 CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
CEM 3311-200 Exam 2 ANSWER KEY ctober 23, 2012 Time: 2 ours Please copy and sign the onor Pledge on the scantron sheet in the space below the double lines. I pledge that n my honor, as a University of
EXAMINATION 2 Chemistry 3A
 1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
1 EXAMINATIN 2 Chemistry 3A Name: KEY Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
Chemistry Fall Semester 2007; Midterm 3 Exam
 1 Chemistry 2521 Fall Semester 2007; Midterm 3 Exam December 7, Friday, 1:00 to 1:50 pm This exam has 7 problems (100 pts) on 9 pages. Make sure your copy is complete and correct. Printed Name (Last, First)
1 Chemistry 2521 Fall Semester 2007; Midterm 3 Exam December 7, Friday, 1:00 to 1:50 pm This exam has 7 problems (100 pts) on 9 pages. Make sure your copy is complete and correct. Printed Name (Last, First)
Chemistry 143 Exam #2 November 15, Name: Student Number: Section Number: TA: INSTRUCTIONS:
 Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
Chemistry 143 Exam #2 November 15, 2017 Name: Student Number: Section Number: TA: INSTRUCTINS: This exam consists of 32 questions on 9 pages. Please make certain that your exam is complete. Write your
CHE 321 Summer 2012 Exam 2 Form Select the correct number of chirality centers in heroin, the illicit drug shown below.
 Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
Multiple Choice Questions: 50 points 1. Select the correct number of chirality centers in heroin, the illicit drug shown below. A 4 B 5 C 6 D 7 E 8 F more than 8 2. Choose the correct IUPAC name for the
CEM 351 2nd EXAM/Version A Friday, October 17, :50 2:40 p.m. Room 138, Chemistry
 Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
Name (print) Signature Student # Section Number CEM 351 2nd EXAM/Version A Friday, October 17, 2003 1:50 2:40 p.m. Room 138, Chemistry Ann Swerkey Grade? 1.(20 2.(20. 3.(20 4.(20 5.(20 6.(20 TOTAL 100
Chem 112A: Final Exam
 Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 15th, 2010 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
CHEM Fall Exam 2 Professor R. Hoenigman. Signature. Name (printed) Last four digits of your student ID number.
 Average = 68 I pledge to uphold the CU on Code: CEM 3311-200 Fall 2006 Exam 2 Profess R. oenigman igh = 98 Low = 18 Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation
Average = 68 I pledge to uphold the CU on Code: CEM 3311-200 Fall 2006 Exam 2 Profess R. oenigman igh = 98 Low = 18 Signature Name (printed) Last four digits of your student ID number Recitation TA Recitation
CHEM Exam 1
 CEM 3311-100 Exam 1 February 12, 2013 Time: 2 ours By printing my name and signing on this cover page, I pledge that On my honor, as a University of Colorado-Boulder student, I have neither given nor received
CEM 3311-100 Exam 1 February 12, 2013 Time: 2 ours By printing my name and signing on this cover page, I pledge that On my honor, as a University of Colorado-Boulder student, I have neither given nor received
CHEM 3311 (Richardson) Third Exam Nov. 27, 2018
 CHEM 3311 (Richardson) Third Exam Nov. 27, 2018 Your Name: Question Score Out of 1 15 Student ID: 2 20 3 20 Recitation (check one) O 10:00 Mon (Shafer Soars) 4 10 O 11:00 Mon (Matthew Farmer) O 1:00 Mon
CHEM 3311 (Richardson) Third Exam Nov. 27, 2018 Your Name: Question Score Out of 1 15 Student ID: 2 20 3 20 Recitation (check one) O 10:00 Mon (Shafer Soars) 4 10 O 11:00 Mon (Matthew Farmer) O 1:00 Mon
Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron sheet.
 Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Test 3 Chemistry 2321 April 25, 2003 McMurry, Chapters 9-11 103 Total Points Name Please Print Part I. Multiple choice. (4 points each.) Choose the one best answer and mark your answer on the ScanTron
Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat #
 Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Test 4 Name Chemistry 2321 Last name, First name (please print) April 22, 2008 McMurry, Chapters 10 and 11 Row # Seat # Part 1. Multiple Choice. Choose the one best answer, and clearly mark that answer
Please read and sign the Honor Code statement below:
 CHEM 3311 Exam #1 Name Dr. Minger June 7, 2010 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
CHEM 3311 Exam #1 Name Dr. Minger June 7, 2010 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor received
PLEASE DO NOT OPEN THIS EXAM UNTIL YOU ARE INSTRUCTED TO DO SO.
 CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
CEMISTRY 0 :5 AM Section EXAM 2 Nov 2009 Name: Note: Your exam should consist of 5 pages including the cover page and grade tabulation sheet. Skim the entire exam, and solve the easiest problems first.
Midterm #2 Chem 3A - Fall 2013 Nov. 12, :00 8:45 pm. Name SID
 Midterm #2 Chem 3A - Fall 2013 Nov. 12, 2013 7:00 8:45 pm Name SID Including the title page, there should be 6 total questions spread over 8 pages (printed on both sides). Please provide all answers in
Midterm #2 Chem 3A - Fall 2013 Nov. 12, 2013 7:00 8:45 pm Name SID Including the title page, there should be 6 total questions spread over 8 pages (printed on both sides). Please provide all answers in
FALL 2018 CEM 251: Organic Chemistry I Sections Oct 23 rd, 2018
 FALL 2018 CEM 251: Organic Chemistry I Sections 25-36 TUE-TU 8:00 9:20 AM Oct 23 rd, 2018 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only
FALL 2018 CEM 251: Organic Chemistry I Sections 25-36 TUE-TU 8:00 9:20 AM Oct 23 rd, 2018 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only
UCF - ORGANIC CHEMISTRY 1 - PROF. DAOUDI UCF PROF. DAOUDI EXAM 3 REVIEW.
 UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
UCF PROF. DAOUDI EXAM 3 REVIEW www.clutchprep.com 1 PRACTICE: Identify the most stable and the least stable alkene PRACTICE: Create the full arrow pushing mechanism which shows all intermediates and all
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
Organic Chemistry CHM 224
 rganic Chemistry CHM 224 Exam I Review Questions This set of questions is a compilation of old exams and additional questions, and does not represent the typical length of an exam - this is WAY longer!
rganic Chemistry CHM 224 Exam I Review Questions This set of questions is a compilation of old exams and additional questions, and does not represent the typical length of an exam - this is WAY longer!
CHEMISTRY MIDTERM # 3 March 31, The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck!
 Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
Name: CEMISTRY 1-01 MIDTERM # March 1, 2009 The total number of points in this midterm is 100. The total exam time is 120 min (2 h). Good luck! 1. (11 pts) Mark as true (T) false (F) the following statements.
I pledge on my honor that I have neither given nor received unauthorized aid on this examination
 Chemistry 220b, Section 1 Exam 2 (100 pts) Thursday, ebruary 26, 2015 Chapters 13, 15-19 ame Write and sign the VU onor Pledge: I pledge on my honor that I have neither given nor received unauthorized
Chemistry 220b, Section 1 Exam 2 (100 pts) Thursday, ebruary 26, 2015 Chapters 13, 15-19 ame Write and sign the VU onor Pledge: I pledge on my honor that I have neither given nor received unauthorized
Chem 30A Winter Mon March 21st
 Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
Last Name First Name MI Student ID Number: Total Score / 200 Chem 30A Winter 2005 FINAL (180 Min) Mon March 21st Course Grade ***DO NOT OPEN THIS EXAM UNTIL INSTRUCTED TO DO SO*** ONLY ANSWERS WRITTEN
Final Exam December 13, 2005 Professor Rebecca Hoenigman
 CEM 3311-200, all 2005 inal Exam December 13, 2005 Professor Rebecca oenigman Average core = 158 igh core = 276 Low core = 20 I pledge to uphold the CU onor Code: ignature Name (printed) Last four digits
CEM 3311-200, all 2005 inal Exam December 13, 2005 Professor Rebecca oenigman Average core = 158 igh core = 276 Low core = 20 I pledge to uphold the CU onor Code: ignature Name (printed) Last four digits
CEM 251, Fall 2011 Sections Tuesday Thursday 6:00 7:30 pm Midterm #2 October 27, 2011
 CEM 251, Fall 2011 Sections 25 36 Tuesday Thursday 6:00 7:30 pm ctober 27, 2011 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only what is
CEM 251, Fall 2011 Sections 25 36 Tuesday Thursday 6:00 7:30 pm ctober 27, 2011 Name: Section: PID: TA: This is a closed book and note examination. If boxes are provided for your answers, only what is
Chem 2061 Final Exam. Fall Andy Aspaas, Instructor. Thursday, December 15, Instructions: Please print: Last name: First name:
 Please print: Last name: First name: hem 2061 Final Exam Fall 2005 Andy Aspaas, Instructor Thursday, December 15, 2005 Instructions: You may start as soon as you arrive. The exam was designed to be finished
Please print: Last name: First name: hem 2061 Final Exam Fall 2005 Andy Aspaas, Instructor Thursday, December 15, 2005 Instructions: You may start as soon as you arrive. The exam was designed to be finished
Score: NAME (Print): Chemistry 320N Dr. Brent Iverson 3rd Homework February 3, 2017 SIGNATURE:
 omework Problem Set 3 Iverson 30N Due Friday February 10 NAME (Print): SIGNATURE: hemistry 30N Dr. ent Iverson 3rd omework February 3, 017 Please print the first three letters of your last name in the
omework Problem Set 3 Iverson 30N Due Friday February 10 NAME (Print): SIGNATURE: hemistry 30N Dr. ent Iverson 3rd omework February 3, 017 Please print the first three letters of your last name in the
Note: You must have your answers written in pen if you want a regrade!!!!
 NAME (Print): SIGNATURE: hemistry 310M/318M Dr. Brent Iverson 2nd Midterm November 1, 2007 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit
NAME (Print): SIGNATURE: hemistry 310M/318M Dr. Brent Iverson 2nd Midterm November 1, 2007 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit
Please note: We routinely xerox a number of exams following initial grading to guard against receiving altered answers during the regrading process.
 NAME (Print): SIGNATURE: Chemistry 320N Dr. Brent Iverson 1st Midterm Feb. 18, 2016 Please print the first three letters of your LAST name in the three boxes Please Note: This test may be a bit long, but
NAME (Print): SIGNATURE: Chemistry 320N Dr. Brent Iverson 1st Midterm Feb. 18, 2016 Please print the first three letters of your LAST name in the three boxes Please Note: This test may be a bit long, but
UCSC, Binder (First and Last) CHEM 8A, Organic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60)
 UCSC, Binder Name (First and Last) CEM 8A, rganic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60) 2 (65) 3 (60) 4 (70) 5 (75) 6 (70) Total (400) % In each of the following problems, use your knowledge
UCSC, Binder Name (First and Last) CEM 8A, rganic Chemistry I Summer 18 FINAL EXAM (400 points) 1 (60) 2 (65) 3 (60) 4 (70) 5 (75) 6 (70) Total (400) % In each of the following problems, use your knowledge
NAME (Print): Chemistry 610A/618A Dr. Brent Iverson 2nd Exam Oct. 29, 2003 SIGNATURE:
 NAME (Print): SIGNATURE: hemistry 610A/618A Dr. ent Iverson 2nd Exam ct. 29, 2003 Please Note: This test may be a bit long, but there is a reason. I would like to give you a lot of little questions, so
NAME (Print): SIGNATURE: hemistry 610A/618A Dr. ent Iverson 2nd Exam ct. 29, 2003 Please Note: This test may be a bit long, but there is a reason. I would like to give you a lot of little questions, so
Chem 3719 Example Exams. Chemistry 3719 Practice Exams
 Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Chem 3719 Example Exams Chemistry 3719 Practice Exams Fall 2018 Chemistry 3719, Fall 2017 Exam 1 Student Name: Y Number: This exam is worth 100 points out of a total of 700 points for Chemistry 3719/3719L.
Exam 1 February 16, 2006 Professor Rebecca Hoenigman
 EM 3311 Spring 2006 Exam 1 February 16, 2006 Professor Rebecca oenigman Average Score = 55 igh Score = 96 Low Score = 13 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits of
EM 3311 Spring 2006 Exam 1 February 16, 2006 Professor Rebecca oenigman Average Score = 55 igh Score = 96 Low Score = 13 I pledge to uphold the U onor ode: Signature Name (printed) Last four digits of
Final Exam CHM1321-B. Date: April 19 Length: 3 hrs Last Name: Professor Sandro Gambarotta Name:
 Faculté des sciences Faculty of Science Département de chimie et science biomoléculaire Department of Chemistry and Biomolecular Sciences Pavillon d Iorio Hall 10 Marie-Curie Ottawa ON Canada K1N 6N5 '
Faculté des sciences Faculty of Science Département de chimie et science biomoléculaire Department of Chemistry and Biomolecular Sciences Pavillon d Iorio Hall 10 Marie-Curie Ottawa ON Canada K1N 6N5 '
Chem 14D Spring 2010 Garg Midterm 1 Review / Practice Problems
 Some Topics to Study (very broad): Chem 14D Spring 2010 Garg Midterm 1 Review / Practice Problems Reactivity stuff Classify atoms as electrophilic or nucleophilic Definitions of nucleophiles/electrophiles
Some Topics to Study (very broad): Chem 14D Spring 2010 Garg Midterm 1 Review / Practice Problems Reactivity stuff Classify atoms as electrophilic or nucleophilic Definitions of nucleophiles/electrophiles
Chemistry 3A. Midterm 2. Dr. Steven Pedersen November 9, Student name: ANSWERS Student signature:
 r. Steven Pedersen ovember 9, 2015 Chemistry 3A Midterm 2 Student name: ASWERS Student signature: Problem 1 Problem 2 Problem 3 Problem 4 Problem 5 Problem 6 Problem 7 (18 pts) (30 pts) (32 pts) (18 pts)
r. Steven Pedersen ovember 9, 2015 Chemistry 3A Midterm 2 Student name: ASWERS Student signature: Problem 1 Problem 2 Problem 3 Problem 4 Problem 5 Problem 6 Problem 7 (18 pts) (30 pts) (32 pts) (18 pts)
= ( 3 ) + 2 ( 3 ) = ( ) = = ( ) = 20 H H
 Columbia University 92CORG12.DOC CEM S3443D Summer 1992 Professor Grace B. Borowitz Exam No. 2 June 17, 1992 Name: Grade: Please use a non-red pen. Answer questions in the provided space. If you write
Columbia University 92CORG12.DOC CEM S3443D Summer 1992 Professor Grace B. Borowitz Exam No. 2 June 17, 1992 Name: Grade: Please use a non-red pen. Answer questions in the provided space. If you write
PRACTICE PROBLEMS UNIT 8
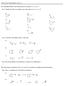 PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
PRACTICE PROBLEMS UNIT 8 8A. Identify halides and carbocations as being 1 o, 2 o, or 3 o 8A.1 assify the following halides and carbocations as 1 o, 2 o, or 3 o. 8A.2 Circle the most stable cation in each
EXAMINATION 2 Chemistry 3A
 000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
000402 1 EXAMINATION 2 Chemistry 3A Name: Print first name before second! Use capital letters! SID #: Peter Vollhardt April 11, 2017 GSI (if you are taking Chem 3AL): Please provide the following information
Note: You must have your answers written in pen if you want a regrade!!!!
 NAME (Print): SIGNATURE: hemistry 310M/318M Dr. ent Iverson 2nd Midterm November 1, 2007 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
NAME (Print): SIGNATURE: hemistry 310M/318M Dr. ent Iverson 2nd Midterm November 1, 2007 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long,
Chemistry 233 Exam 3. The Periodic Table
 Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
Name: Last First MI Chemistry 233 Exam 3 Fall 2017 Dr. J. sbourn Instructions: The first 8 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in the Scantron
diene. If stereoisomeric mixtures form, indicate by drawing one and writing "+ enantiomer" and/or "+ diastereomer" in the box.
 I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
I. (9 points) Page A. Consider how varying the conditions of a reaction can vastly influence the rate of a reaction as well as the product(s) formed. First draw the product(s) that form in the reference
Homework problems Chapters 6 and Give the curved-arrow formalism for the following reaction: CH 3 OH + H 2 C CH +
 omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
omework problems hapters 6 and 7 1. Give the curved-arrow formalism for the following reaction: : 3 - : 2 : 3 2-3 3 2. In each of the following sets, arrange the compounds in order of decreasing pka and
CH 253, Fall Question Points Possible Points Received
 CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
CH 253, Fall 2000 Exam #3 - M. Schwartz November 17, 2000 Name (Print Clearly) Question Points Possible Points Received 1 20 2 15 3 8 4 5 5 4 6 6 7 14 8 8 9 5 10 21 Subtotal 106 Extra Credit 2 Total 108
CHEM 203. Midterm Exam 2 November 13, 2014 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 2 ovember 13, 2014 Your name: ASWERS This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15 3.
CEM 203 Midterm Exam 2 ovember 13, 2014 Your name: ASWERS This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15 3.
Homework Problem Set 7 Iverson CH320M/328M Due Friday, Nov. 2
 NAME (Print): SIGNATURE: hemistry 320M/328M Dr. Brent Iverson 7th omework October 29, 2018 Please print the first three letters of your last name in the three boxes 1 Score: 2 1. For the following reactions,
NAME (Print): SIGNATURE: hemistry 320M/328M Dr. Brent Iverson 7th omework October 29, 2018 Please print the first three letters of your last name in the three boxes 1 Score: 2 1. For the following reactions,
1. (6 points) Provide IUPAC accepted names for the following compounds. 2. (6 points) Provide a structure for the following compounds.
 Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
Chem52 omework Set 1 This homework set is similar to a Chem 51 final exam. Please provide answers in the spaces provided or, preferably, on attached sheets. Point values are listed only to give you the
CHEM 8A Summer Student ID # Organic Chemistry EXAM 2 (300 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry EXAM 2 (300 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the questions
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry EXAM 2 (300 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the questions
Exam 4. Name CHEM 210. PBr 3. HBr. 1) NaH 2) Br H 2 SO 4. 1) NaOCH 3 2) dil. H 3 O + O CH 3 OH, H 2 SO 4. 1) LDA, -78 o C.
 Exam 4 Name CEM 210 1. (48 pts) Complete the equations for the following reactions. Show all organic products if two or more products form, indicate the major product. Indicate proper stereochemistry where
Exam 4 Name CEM 210 1. (48 pts) Complete the equations for the following reactions. Show all organic products if two or more products form, indicate the major product. Indicate proper stereochemistry where
Supplemental Exam CHM1321-B. Professor Sandro Gambarotta. Date:_6-July-2017 Length: 3 hrs Last Name:
 1 Supplemental Exam CHM1321-B Professor Sandro Gambarotta Date:_6-July-2017 Length: 3 hrs Last Name: First Name: Student # Seat # - Instructions: - Calculator permitted (Faculty approved or any other non-programmable
1 Supplemental Exam CHM1321-B Professor Sandro Gambarotta Date:_6-July-2017 Length: 3 hrs Last Name: First Name: Student # Seat # - Instructions: - Calculator permitted (Faculty approved or any other non-programmable
Organic Chemistry I. Summer Final Exam
 Print Name: rganic hemistry Summer 2014 Final Exam SPEAL NSTRUTNS: Please make sure to read each questions VERY carefully!! Some questions will look similar to questions on previous tests, but contain
Print Name: rganic hemistry Summer 2014 Final Exam SPEAL NSTRUTNS: Please make sure to read each questions VERY carefully!! Some questions will look similar to questions on previous tests, but contain
CHEM 3311 Exam 1 ANSWER KEY
 CEM 3311 Exam 1 ASWER KEY ebruary 11, 2014 Time: 2 ours By printing signing my name below (-2 if this information is not filled in), I pledge that On my honor, as a University of Colorado-Boulder student,
CEM 3311 Exam 1 ASWER KEY ebruary 11, 2014 Time: 2 ours By printing signing my name below (-2 if this information is not filled in), I pledge that On my honor, as a University of Colorado-Boulder student,
Please read and sign the Honor Code statement below:
 CHEM 3331 Exam #2 Name Dr. Minger July 27, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor
CHEM 3331 Exam #2 Name Dr. Minger July 27, 2015 Please read and sign the Honor Code statement below: I pledge that on my honor, as a University of Colorado at Boulder student, I have neither given nor
CHEM 203. Final Exam December 15, 2010 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
CEM 203 Final Exam December 15, 2010 Your name: ANSWERS This a closed-notes, closed-book exam You may use your set of molecular models This test contains 15 pages Time: 2h 30 min 1. / 16 2. / 15 3. / 24
Chemistry Exam 2. The Periodic Table
 Name: Last First MI Chemistry 234-002 Exam 2 Spring 2017 Dr. J. sbourn Instructions: The first 18 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in
Name: Last First MI Chemistry 234-002 Exam 2 Spring 2017 Dr. J. sbourn Instructions: The first 18 questions of this exam should be answered on the provided Scantron. You must use a pencil for filling in
You must have your answers written in PERMANENT ink if you want a regrade!!!! This means no test written in pencil or ERASABLE INK will be regraded.
 NAME (Print): SIGNATURE: Chemistry 310M/318M Dr. ent Iverson 3rd Midterm November 18, 2010 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit
NAME (Print): SIGNATURE: Chemistry 310M/318M Dr. ent Iverson 3rd Midterm November 18, 2010 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit
Dr. Steven Pedersen November 9, Chemistry 3A. Midterm 2
 Dr. Steven Pedersen November 9, 2016 Student name: ANSWER KEY Chemistry 3A Midterm 2 Student ID: (Also include your SID in the top left corner of each page) Student signature: Problem 1 Problem 2 Problem
Dr. Steven Pedersen November 9, 2016 Student name: ANSWER KEY Chemistry 3A Midterm 2 Student ID: (Also include your SID in the top left corner of each page) Student signature: Problem 1 Problem 2 Problem
Problem Point Value Points
 Math 70 TUFTS UNIVERSITY October 12, 2015 Linear Algebra Department of Mathematics Sections 1 and 2 Exam I Instructions: No notes or books are allowed. All calculators, cell phones, or other electronic
Math 70 TUFTS UNIVERSITY October 12, 2015 Linear Algebra Department of Mathematics Sections 1 and 2 Exam I Instructions: No notes or books are allowed. All calculators, cell phones, or other electronic
CHEM 231 (Davis) Organic Chemistry FINAL EXAM May 15, YOUR NAME (Last, First, M.I.) DISCUSSION SECTION #53 (5 Points)
 CHEM 231 (Davis) rganic Chemistry FINAL EXAM May 15, 2006 YUR NAME (Last, First, M.I.) DISCUSSIN SECTIN #53 (5 Points) Initial of last name Instructions Please fill in your name in the space above and
CHEM 231 (Davis) rganic Chemistry FINAL EXAM May 15, 2006 YUR NAME (Last, First, M.I.) DISCUSSIN SECTIN #53 (5 Points) Initial of last name Instructions Please fill in your name in the space above and
CHEM 8A Summer Student ID # Organic Chemistry FINAL EXAM (400 points)
 CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
CEM 8A Summer 2017 UCSC, Binder Name Student ID # rganic Chemistry FINAL EXAM (400 points) In each of the following problems, you will use your knowledge of organic chemistry conventions to answer the
CHE 321 Summer 2010 Exam 2 Form Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III
 CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
CE 321 Summer 2010 Exam 2 orm 2 Multiple Choice Questions: 60 points 1. Choose the structure(s) that represent cis-1-sec-butyl-4-methylcyclohexane. I II III (A) I only (B) II only (C) I and II only II
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2017 Dr. Rainer Glaser
 Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2017 Dr. Rainer Glaser Examination #3 Nucleophilic Substitutions & Eliminations, Alcohols, and Ethers Thursday, November 2, 2017, 8:25-9:15
Chemistry 2030 Introduction to Organic Chemistry Fall Semester 2017 Dr. Rainer Glaser Examination #3 Nucleophilic Substitutions & Eliminations, Alcohols, and Ethers Thursday, November 2, 2017, 8:25-9:15
Chem 232. Problem Set 9. Question 1. D. J. Wardrop
 Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Chem 232 D. J. Wardrop wardropd@uic.edu Problem Set 9 Question 1. a. Rank in order of increasing number of chirality centers (1 = fewest chirality centers; 5 = most chirality). 3 C C 3 b. Rank in order
Exam 3 Professor R. Hoenigman
 I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
I pledge to uphold the CU onor Code: CEM 3331-100 Spring 2008 Exam 3 Professor R. oenigman igh = 102 Low = 15 Average = 68 Signature ame (printed) Last four digits of your student ID number Recitation
Chem 341 Organic Chemistry I Final Exam December 12, 2007 N A M E K E Y
 Chem 341 rganic Chemistry I Final Exam December 12, 2007 N A M E K E Y Top 10 signs you ve had Chem 341: 10. You can read the ingredients list on the back of your shampoo bottle and understand what s in
Chem 341 rganic Chemistry I Final Exam December 12, 2007 N A M E K E Y Top 10 signs you ve had Chem 341: 10. You can read the ingredients list on the back of your shampoo bottle and understand what s in
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
ORGANIC CHEMISTRY I CHEM 221 /2 01 Final Examination December 20, 2005 1400-1700 Dr. Cerrie ROGERS x x molecular model kits (without instructions) programmable calculators must be reset Chem 221 --- ORGANIC
CHEM 203. Midterm Exam 2 November 12, 2013 ANSWERS. This a closed-notes, closed-book exam. You may use your set of molecular models
 CEM 203 Midterm Exam 2 November 12, 2013 ANSWERS Your name: This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15
CEM 203 Midterm Exam 2 November 12, 2013 ANSWERS Your name: This a closed-notes, closed-book exam You may use your set of molecular models This document contains 7 pages Time: 1h 30 min 1. / 10 2. / 15
Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm
 - 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
- 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
Chapter 9:Nucleophiles & Substitution Reactions
 1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
1. a. Place the following nucleophiles in order of strength (1= strongest; 3 = weakest). i. ii. b. Place the following in order of leaving group ability (1= best; 7 = worst). (A pka table may help you!)
You must have your answers written in PERMANENT ink if you want a regrade!!!! This means no test written in pencil or ERASABLE INK will be regraded.
 NAME (Print): SIGNATURE: Chemistry 320N Dr. Brent Iverson 1st Midterm Feb. 13, 2014 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
NAME (Print): SIGNATURE: Chemistry 320N Dr. Brent Iverson 1st Midterm Feb. 13, 2014 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
I. (32 points) Name. Page 1. (ii) its diastereomer its structural isomer its enantiomer. The product you have drawn will. H 3 CO Cl form with H 3 CO
 ame. ( points) Page Each of the reactions yields a PAR of main organic products, but you should only draw one. Then answer the other questions about the starting and the products formed. Check boxes are
ame. ( points) Page Each of the reactions yields a PAR of main organic products, but you should only draw one. Then answer the other questions about the starting and the products formed. Check boxes are
This is a long exam. Most students will not finish each problem. Carefully select your problems to work on.
 Chemistry 51 Exam 1, Spring 2008 =============================================== This is a closed book exam. The exam lasts 75 minutes. All answers must appear on the answer sheet. Only the answer sheet
Chemistry 51 Exam 1, Spring 2008 =============================================== This is a closed book exam. The exam lasts 75 minutes. All answers must appear on the answer sheet. Only the answer sheet
H C C C C H C H C C H OH. Cl H CH 3. Br CH 3. H Br H OH H 3 C. CHEMISTRY MIDTERM # 3 June 17, Name...
 Name... CEMISTRY 313-61 MIDTERM # 3 June 17, 2005 The total number of points in this midterm is 100. The total exam time is 90 min (1.5 h). Good luck! 1. (10 pts) Mark as true (T) false () the following
Name... CEMISTRY 313-61 MIDTERM # 3 June 17, 2005 The total number of points in this midterm is 100. The total exam time is 90 min (1.5 h). Good luck! 1. (10 pts) Mark as true (T) false () the following
CHE 230 Organic Chemistry Exam 4, May 4, 2000
 CE 230 rganic Chemistry Exam 4, May 4, 2000 Name Student ID No. Before you begin this exam : First: You are allowed to have a calculator and a simple model set at your seat. Please put away all other materials.
CE 230 rganic Chemistry Exam 4, May 4, 2000 Name Student ID No. Before you begin this exam : First: You are allowed to have a calculator and a simple model set at your seat. Please put away all other materials.
- NH2 or NH 3 H 2 O or CH 3 COO - BF 3 or F - I Br. or Br
 CEMSTRY 313-02 MDTERM # 3 answer key November 09, 2004 Statistics: Average: 71 pts (71%); ighest: 90 pts (90%); Lowest: 49 pts (49%) Number of students perfming at above average: 22 (51%) 1. (8 pts) Mark
CEMSTRY 313-02 MDTERM # 3 answer key November 09, 2004 Statistics: Average: 71 pts (71%); ighest: 90 pts (90%); Lowest: 49 pts (49%) Number of students perfming at above average: 22 (51%) 1. (8 pts) Mark
Organic Chemistry 1 CHM 2210 Exam 3 (November 9, 2001)
 Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Organic Chemistry 1 CM 2210 Exam 3 (November 9, 2001) Name (print): Signature: Student ID Number: There are 14 multiple choice problems (5 points each) on this exam, however, you will only be graded out
Note: You must have your answers written in pen if you want a regrade!!!!
 NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 1st Midterm Feb. 21, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
NAME (Print): SIGNATURE: Chemistry 310N Dr. Brent Iverson 1st Midterm Feb. 21, 2008 Please print the first three letters of your last name in the three boxes Please Note: This test may be a bit long, but
