Mean-field theory of heteroepitaxial thin metal film morphologies
|
|
|
- Frederica Lewis
- 5 years ago
- Views:
Transcription
1 ELSVIER Surface Science 318 (1994) :~::~:~:~.::~:::4,:::~::: ::::;~:~~~.~,:,, :,~,::~,:~,~~,.,,.1.%x:.:.:.:(...:.:.~,...,... :.::.~.:~.::~ ii,..,.,:,:,.~?.:.::::>.x.:::$:.,:.: F ,Ci...:...A...,:. t,g>,;$$p ;:.(. surface ::.,.> l...:.:.h:...( ~...~IR:..~../,..._..,:(::.:.~::::::~.~~~.~.~:~:~:~~ : ~.~~.:~~:::::~.:::~~~:~::.::~,.~ :.:.:.:,/,..:.:... : Mean-field theory of heteroepitaxial thin metal film morphologies Tsun-Mei Chang, Emily A. Carter * Department of Chemistry and Biochemistry, Universiiy of California, Los Angeles, CA , USA Received 21 January 1994; accepted for publication 17 June 1994 Abstract We develop a mean-field statistical mechanical theory to examine the growth mechanisms and structures of fee (face-centered cubic) metal thin films grown on fc&o) metal substrates. Eleven hetero-interfaces with bulk cohesive energy differences as large as - 4 ev and lattice mismatches ranging from - 21% to 16% are examined in order to develop an understanding of how these intrinsic properties of the interface may determine film morphology. For small (up to - 5%) mismatched interfaces, pseudomorphic Stranski-Krastanov growth is predicted to be competitive with pseudomorphic layer-by-layer growth. When the lattice mismatch is 10% or greater, the growth mechanism is invariably incommensurate three-dimensional growth, where (111) cluster formation is energetically competitive with (100) cluster growth. The overlayer structures and growth modes correlate most closely with only one property: the lattice strain between the substrate and the adlayer. By contrast, the trends in the bulk cohesive energies do not allow even any qualitative prediction of the overlayer structures. However, surface energies can dictate minor changes in the growth modes of the films. Finally, no dependence of the film morphology on temperature is found over the temperature range K for the cases we examined, although the film morphologies can be markedly different from those predicted at lower temperatures. 1. Introduction The study of ultrathin metallic films has been a subject of great interest for many years [l-8]. Because of their unusual chemical and physical properties, these films may someday form the basis for new magnetic recording devices, new catalysts, and other technological applications. Due to advances in experimental techniques, it is now possible to have significant control over the growth conditions of epitaxial films. One hopes that the thickness, quality, and composition of the films will soon be tailored to specific applications. In the meantime, it is very * Corresponding author. Fax: eae@hem.ucla.edu. important to be able to characterize the structures and growth mechanisms of ultrathin films, in order to eventually manipulate film properties. There are many factors that may determine film structure: lattice mismatch, lattice symmetry, temperature, thickness or coverage, and the relative strength of the adatom-adatom, substrate atom-substrate atom, and adatom-substrate atom interactions. Depending on the relative importance of each of these factors, the structures of films may be significantly different from those of the related bulk materials. When the overlayer is ultrathin, and the lattice mismatch between the film and substrate is small, the overlayer often can adopt the structure of the substrate on which it is grown, regardless of its own bulk preferences. This is known as pseudomorphic growth. On the other hand, when no vectors in the 0039~6028/94/$ Elsevier Science B.V. AR rights reserved SSDI (94)
2 188 T.-M. Chang, EA. Carter/Siuface Science 318 (1994) reciprocal lattices of substrate and film are common to each other, this is known as incommensurate growth [6,9,10]. The structure of a film, if different from that of the bulk material, can have a pronounced impact on film properties. For example, the magnetic properties of Ni, Co, and Fe change dramatically when two-dimensional films of these metals are formed on other metal substrates [11,12]. The growth mechanism will determine the smoothness of the film surface, thus dictating the film quality and affecting its physical and chemical properties. It has been postulated that three distinct, major growth modes exist in the thermodynamic limit. One of them is known as the FM (Frank-van der Merwe) growth mode, which describes layer-bylayer growth [ll. In this model, the second adlayer will not form until the first adlayer is completed. This growth mechanism has been inferred, for example, by discontinuous changes in the slope of adlayer AES (Auger electron spectroscopy) signal versus coverage plots [13-161, or more definitively by forward scattering and scanning tunneling microscopy (STM) [14]. The VW (Volmer-Weber) growth mode model describes films that form via three-dimensional (3D) nucleation. This mechanism is thought to dominate when the adlayer metal AES signal simply shows a steady, continuous increase with coverage [13], or when direct observation, e.g. via STM, reveals 3D cluster formation. The SK (Stranski- Krastanov) growth mechanism model is a mixture of the above two. At the beginning, such a film is postulated to grow layer-by-layer for the first l-2 layers, followed by three-dimensional growth. In the past, much work has focused on understanding thermodynamic growth modes and equilibrium film structures. Such predictions provide an important thermodynamic baseline, in the same sense that thermochemistry provides a zeroth-order prediction of the tendency for a reaction to proceed. Determining the equilibrium film structures and growth modes sets the stage for understanding when kinetics are important, in the same manner that analysis of reaction kinetics is necessary in order to determine fully whether or not a reaction will actually occur. Furthermore, under certain experimental situations, equilibrium structures are expected [17]. For example, when surface diffusion is fast relative to the rate of deposition, equilibrium morphologies may be ob- served. This can be achieved at high substrate temperatures and low fluxes. Also, annealing epitaxial films often produces equilibrium structures [18]. For heteroepitaxy, it is also very important to develop a basic understanding of how static factors, such as strains, interactions, etc., affect the equilibrium film morphology. Bauer used thermodynamic arguments to propose the following criterion for determining heteroepitaxial growth modes [3]. Eschewing the morphology and length scale of the surface and its edges, he defined a quantity, A, which should predict the growth mechanism A=q+c~~-a,, (1) where oi is the film-substrate interfacial free energy per unit area, and or and us are the surface free energies per unit area of the film-vacuum and substrate-vacuum interfaces, respectively. For the case of A > 0, the growth mode should be dominated by the VW mechanism. For the case of A I 0, the growth mode should obey the FM mechanism if A I 0 for each additional layer, otherwise it should follow the SK mode. While this criterion makes excellent physical sense, in practice, it is very difficult to apply. In particular, it is unclear how or even if it is possible to extract ur from bulk specific free energies for application to ultrathin metallic films. Furthermore, there is no unique microscopic definition for the interfacial energy, ui. Bauer s criterion is closely related to the Young condition in wetting theory [ Many theoretical studies on growth modes have been based on the analog between these two phenomena, where FM, SK, and VW growth modes correspond to the cases of wetting, incomplete wetting, and non-wetting, respectively. Bruinsma and Zangwill [22] used a continuum model to study thin film morphology. By free energy minimization, they predicted that the FM mode is not thermodynamically stable for strained interfaces. Grabow and Gilmer subsequently reached a similar conclusion via molecular dynamics simulations [23]. This is a provocative result; however, it is not obvious how to apply their formula to real systems to test this idea further, since estimation of parameters in their formulation is not straightforward. More recently, Ratsch and Zangwill [24] used an analytic solution to the one-dimensional Frenkel-
3 T.-M. Chang, E.A. Carter/Surface Science 318 (1994) Kontorova model to minimize the free energies of diamond structures grown on a (100) substrate at 0 K. They found that only for very small mismatched interfaces (N 2%), is layer-by-layer growth the preferred thermal growth mode. The present work also deals with free energy minimization, but applied to real metal-on-metal systems within a microscopic framework. We shall see that the qualitative conclusions of these earlier studies are confirmed in the present work. Many studies have been devoted to understanding thin film growth using static mechanical energy calculations. The basic idea is to express the system s mechanical energy in terms of lattice parameters, and to try to minimize this energy with respect to these parameters analytically or numerically in order to find the lowest-energy structures, which are considered the equilibrium structures. Frank and van der Merwe developed a classical continuum theory 45 years ago [1,2], in which they assumed that the adatoms were connected to each other by harmonic springs, while experiencing a continuous, periodic potential due to the rigid substrate. The total energy of the system could then be described as a sum of only the adlayer-adlayer and adlayer-substrate interactions. From total mechanical energy minimization, they predicted that pseudomorphic growth can occur for up to 9% lattice mismatch between a substrate and an adlayer at one monolayer (1 ML) coverage. As we shall see, our work using a more sophisticated model leads in fact to a qualitatively similar conclusion. Subsequent work extended this idea using more realistic, atomistic potentials to describe the atomic interactions. For example, Dodson used a static calculation to study the structures of several uniformly strained fee metal overlayers on fee metal substrates. Using the embedded atom method (EAM) to calculate the particle interactions, he predicted the critical thickness (usually l-3 layers) up to which the thin films would maintain pseudomorphic growth [25]. He showed that the first overlayer can remain pseudomorphic for lattice mismatches up to 7-lo%, in good agreement with the earlier conclusions from the continuum models of Frank and van der Merwe, and, as shown below, consistent with our mean-field predictions as well. Although these static theories provided valuable insight into some aspects of film growth, they had some limitations. First, since temperature was not introduced in these formalisms, these theories only provided information at 0 K. Of course, the experiments typically are carried out above room temperature, therefore it would be useful to make predictions at finite temperatures. Furthermore, it has been shown theoretically and experimentally that changing the temperature can alter dramatically the film quality and morphology, suggesting that temperature can have a significant role to play [26,27]. Second, relaxation of the rigid lattice constraint on the substrate is critical for obtaining a realistic description of film growth, since the substrate will respond to the presence of the film. Third, many of the earlier studies used model interaction potentials not appropriate for metallic systems. Relaxation of the rigid lattice approximation and inclusion of temperature have been achieved via Monte Carlo (MC) and molecular dynamics (MD) simulations. Using MC and MD to simulate two-dimensional crystals with Lennard-Jones potentials, Dodson and Taylor found pseudomorphic growth up to 3-5 ML when the film-substrate lattice mismatch was less than 4% [28,29]. Using a lattice-based MC method with Lennard-Jones potentials, Faux et al. studied similar systems with lattice mismatches up to 10% They found that pseudomorphic layer-bylayer growth occurred for up to 2% lattice mismatch, while incommensurate three-dimensional growth was favored for more greatly mismatched interfaces. Performing a series of MD simulations for various degrees of misfit using Lennard-Jones and Stillinger-Weber potentials, Grabow and Gilmer [23] predicted that only when the substrate-adatom interaction is stronger than the adatom-adatom interaction and when there is no mismatch, is FM growth thermodynamically stable. For mismatched interfaces, the preference for VW or SK growth will depend on the relative strength of the substrateadatom and adatom-adatom interactions. (However, using nucleation theory, they predicted that the activation barrier for transformation from the FM mode to the VW or SK mode is very large. Thus, in many situations, FM persists as the metastable growth mode [23].) Bolding and Carter studied [26,31] fee metal overlayers deposited on a bee substrate, using MC simu-
4 190 T.-M. Chang, EA. Carter/Stirface Science 318 (1994) lations with the more realistic, pseudo-many-body EAM potentials. They examined the structures of thin films as a function of temperature and coverage. For the highly strained (- 18% in one direction) case of a Pd film on a model for Ta(ll0) or Nb(llO), they observed a direct pseudomorphic-incommensurate (P-I) structural transition for coverages above one bee monolayer at high temperatures, while the P-I transition was completely suppressed at low temperatures or submonolayer coverages. They also studied one case of an fee metal deposited on an fee substrate. In particular, they used MC simulations with EAM potentials to study growth of a Ni thin film on a Ag(100) surface [32]. Luedtke and Landman used MD simulations to study Au/Ni(lOO) and Ni/Au(lOO) interfaces [33]. Detailed comparisons of these latter studies to the present work is given later in the discussion section. Computer simulations have been shown to be very powerful for studying physical problems at the molecular level, by providing microscopic insight into physical phenomena. However, one is always restricted by computational limitations that induce restrictions on the size of the simulation. Making the connection between macroscopic phenomena and those observed via atomistic simulations is often still a tenuous undertaking. Therefore, it is useful to develop a theoretical approach that directly describes macroscopic quantities and is more computationally efficient than simulations, so that one may explore many systems to extract general trends. This is the goal of the present work. In this study, we develop a statistical mechanical theory to study the thermodynamically-controlled growth of strained epitaxial thin films. In Section 2, we present the theoretical formulation, which relies on a mean-field approximation. A similar approach has been applied fruitfully to the study of material failure under stress [34]. Section 3 discusses the details of our calculations of the free energies for model film-substrate interfaces under various growth conditions. By comparing the free energies of those configurations corresponding to different morphologies, the equilibrium structures of the interfaces and the growth mechanisms are determined. In Section 4, we apply this formulation to fee metal thin films on fcc(100) metal substrates using realistic pseudomany-body potentials to describe the interactions between the metal atoms. The effect of temperature, coverage, symmetry, and lattice mismatch will be investigated separately, and the results are compared to available experimental and simulation results in Section 5. We also discuss some limitations of this approach. In Section 6, we conclude. 2. Theoretical formulation Consider a system that consists of M substrate atoms and N adatoms. Then the configurational partition function, Z, of the system can be written as z= &/d&/d&... /dr,/dr,/dr,... X / dr, exp[-pu(r,,r,,...r,,r,,...r,)], (2) where Ri is the position of the substrate atom i, rj is the position of adatom j, and U is the potential energy, which involves a sum over the interactions of all the particles in the system. Here, p = l/k,t, k, is the Boltzmann constant, and T is the temperature of the system. If we restrict ourselves to heteroepitaxial systems of low to intermediate temperatures (i.e., far below the melting point), then it is reasonable to assume that each atom s position will fluctuate around its equilibrium lattice site. Because of the lattice mismatch-induced strain between the adatoms and substrate, the lattice parameters of the substrate and adlayer may prefer to be different. Here, we make the simplifying assumption that after adatoms are deposited on the substrate, the lattice constant of the substrate will remain at its bulk value. This is basically physically reasonable, since the substrate is effectively a three-dimensionally infinite bulk material. However, the situation is different for the overlayers. Due to the strain imposed by the substrate and the thinness of film, the lattice spacing of the adlayers will contract or expand in the directions parallel to the interface, with concomitant expansion or contraction perpendicular to the interface 132,351. In general, the deformation can be considered uniform, based on previous findings from MC simulations of the structures of lattice-mismatched semiconductor and metallic interfaces [32,35].
5 T.-M. Chang, EA. Carter/Surface Science 318 (1994) Region 1 Region ooooooooo ooooooooe ooooooooo Fig. 1. An example of overlayers growing in a layer-by-layer fashion. White and dark circles denote adatoms and substrate atoms, respectively. The presence of the interface between the film and the substrate breaks the translational symmetry in the direction perpendicular to the interface. The translational symmetry is also broken in the direction parallel to the interface unless the top-most adlayer completely covers the surface. We choose to consider the two most likely limiting cases, in which the adatoms either form a film layer-by-layer or form three-dimensional clusters on top of either the substrate or a complete adlayer. In a mean-field approximation, if we ignore edge effects, there are only two different environments that adatoms or substrate atoms experience in each layer. This can be more easily understood by examining some possible configurations. In Fig. 1, we consider the case in which the film grows in a layer-by-layer fashion. Suppose there are M, substrate layers with MXY atoms per layer, and N, complete adlayers with NXY atoms per layer. Suppose the last (N, + 11th adlayer partially covers the surface with a fractional surface area c. The system can then be divided into two subsystems, which are labelled as regions 1 and 2. The linear dimension of each subsystem is macroscopic in the lateral direction and is of microscopic thickness in the surface normal direction. This implies that the mean-field approximation can be only applied in the lateral direction. Therefore, in each subsystem, atoms are considered to experience an averaged environment within each layer, but experience different atomic interactions in different layers. Assuming that the system is free of defects such as vacancies, impurities, and dislocations, and with this mean-field approximation, the system s partition function can be reduced to a product of single particle partition functions. Now, the mean-field partition function, Z MF, of the system can be written as %4F(%~~%) (3) In the above, z~i(z) and z;i(z) are the partition functions of a single substrate atom, s, and adatom, a, in the (Y th and /3 th layer, respectively. The subscript l(2) denotes the two different environments. The factor of l/n!m! was dropped because now the particles are distinguishable. The single particle partition functions are defined as z ( Q,T;Q) = /, dr exp[ -PE,( R,aad;csb)], I The integration is performed over the Wigner-Seitz cell of the particle. Esca, is the sum of the interaction energies of the substrate atom (adatom) at position R(r) with the rest of the system at their equilibrium positions. usb and uad are the substrate and adlayer lattice parameters in the direction parallel to the interface, respectively. We allow the iuterlayer spacing to change as usb and uad are changed from their bulk values, as will be described in the next section. (The preferred bulk lattice constants of substrate and adatom are denoted as Use and a$, respectively.) From Eq. (31, the Helmholtz free energy of the system can be expressed as 4 adyt%l) = -k,t cm,,~ln zii + (1- c)m,.r?ln ziz [ a (I Nz+ 1 NZ 1 +cnxy c In zil + (1 -c)n,,cln z;z. (6) B B Note that A is a function of uad and T, and only parametrically depends on usb since we assume that (4)
6 192 T.-M. Chang, E.A. Carter/Surface Science 318 (1994) the lattice constant of the substrate does not change after adatoms are deposited. From Eq. (6), we evaluate A as a function of uad and T. The global minimum in the free energy then allows us to identify the equilibrium structure and preferred thermodynamic growth mode of the thin film. 3. Calculational details In general, the mean-field method can be applied to film-substrate interfaces with any lattice symmetry. However, to illustrate the use of this method, we limit our study to ultrathin fee metal films on fcc(100) metal substrates. It is well known that simple two-body (e.g., Lennard-Jones) potentials are not adequate for describing metallic bonding [36,37]. Therefore, we chose the Rafii-Tabar-Sutton-Chen parameterization of the Finnis-Sinclair two-body potential to represent the adatom-adatom, adatom-substrate, and substrate-substrate interactions, which accounts for many-body interactions through the environment dependence of the functional form [ These potential functions have been developed for bee [38], fee [39], and hcp [41] metals and metallic alloys [40]. For an atom i in a crystal, the Finnis-Sinclair potential has the following form where a is the experimental bulk lattice parameter, rij is the distance between atoms i and j, n and m are positive integers, E is a constant with dimensions of energy, and 6 is a positive dimensionless parameter. The first term of the potential represents the repulsive pair interaction between atoms, and the second term describes the effective many-body cohesion. The parameters E, b, m, n have been determined for all 10 fee metals by fitting to their bulk properties. These potentials have been shown to give reasonable predictions of bulk moduli, elastic constants, and enthalpies of mixing for alloys [39,40] and have been used to study fee metal interfaces, self-diffusion, and phonon spectral densities at surfaces [ In this study, the potential radial cutoff is at two lattice constants, which is typically 7-10 K in most cases. When we consider heteroepitaxial growth, unless the two materials have exactly the same lattice constants and symmetries, a mismatch will occur at the interface in a periodic fashion. The periodicity depends on the structural differences between the two materials. Near the interface, each adatom and substrate atom experiences different atomic environments, thus yielding different interaction energies. In order to take this effect into consideration to at least a first approximation, we evaluate the interactions in a configuration-averaged manner. Suppose that the films have uniformly contracted (or expanded) with some parallel lattice constant, which is different from the substrate lattice constant. For each adatom, we can find a nearest substrate atom. The local atomic arrangement of this particular adatom can be specified uniquely by the distance and orientation of the adatom with respect to its nearest substrate atom. Averaging over all possible configurations of the adatom-substrate combination gives the averaged orientation of the substrate with respect to the adatoms. The same procedure is also performed for the substrate atom. Now, instead of evaluating partition functions for each particle with different environments, we only need to calculate one averaged particle partition function per layer per region (see Fig. 1). This significantly reduces the computational effort. In evaluating the partition functions, the algorithm of extended Simpson s rule with an adjustable stepsize was used to perform the numerical integration [45]. The final results were considered converged when the difference between two sequential integrations, divided by the integral, was less than Up to this point, we have made the assumption that the deformation of the adlayers is uniform in the direction parallel to the interface. It is known from low-energy electron diffraction (LEED) intensity analyses that film expansion (contraction) in the lateral direction will cause contraction (expansion) in the film s interlayer spacing in order to reduce strain in the system [46]. As mentioned previously, MC simulations also suggested that the film naturally adjusts the interlayer spacing to compensate for the volume change induced by the parallel deformation of the overlayers [32,35,47]. We therefore adjust the
7 T.-M. Gang, EA. Carter/Surface Science 318 (1994) interlayer spacing accordingly. For example, a 10% expansion of the lattice spacing in the lateral direction of the film leads to a 17.4% contraction in the interlayer spacing. For simplicity of numerical computation, once the interlayer spacing was determined, the particles were not allowed to move in the perpendicular direction. Note that this constraint also implies that we do not consider alloy formation (via place exchange) at the interface. In order to determine the preferred morphology of the film, we evaluate free energies from Eq. (6) for several ideal configurations that are typical for different growth modes. We consider configurations in which adatoms form complete layers on top of the substrate, typical of layer-by-layer growth. We also consider configurations in which adatoms form several three-dimensional clusters n layers thick on top of a bare substrate. Based on kinetic considerations, we only allow n up to five, since it is probably unlikely that metal adatoms will form needle-like features on the substrate. Note that deviations in the free energy due to island boundaries are ignored in our model. Since we are interested in the macroscopic free energy and make the assumption that the three-dimensional clusters are of macroscopic size, we should be able to neglect safely the effect of the edge energies and therefore the actual shape and size of the clusters are irrelevant in this mean-field approximation. In particular, we only calculate the free energy of the interior atoms inside the three-dimensional clusters. In other words, we are actually modeling growth of large islands or complete layers where contributions to the total free energy from island or layer edges are small. At higher coverages (> 1 ML), we also consider configurations in which adatoms form three-dimensional clusters n layers thick on top of one complete adlayer. Note that in Eq. (61, MxY is set equal to 1, since we care only about the fraction c that defines the region. NxY is determined by the parallel lattice constant of the adlayers and the bulk substrate lattice constant, and is equal to M,,a~~/a$. We can then compare the free energies of different structures containing the same numbers of adatoms and substrate atoms. The structure with the lowest free energy is thermodynamically the most stable. The thermodynamically preferred growth mechanism can thus be inferred by comparing relative free energies. In this study, we are only interested in the initial stage of growth of metallic thin films. As the coverage increases, the growth mode can change. When the film is thick enough, the influence of the substrate vanishes, and it is known that for homoepitaxial systems, the thermodynamic growth mechanism must be the FM mode, except under kineticallylimited conditions [48]. However, this transition between growth modes is beyond the scope of this investigation, as we are only interested in how the substrate may influence the initial growth mode and subsequent ultrathin film structure. 4. Results and discussion We apply the mean-field approach presented above to study heteroepitaxy of fee metal films. Eleven cases are chosen to sample a wide range of substrate-film bulk lattice mismatches ( - 21% to 16%), where the lattice mismatch parameter is defined as (azja& - 1) X 100%. Thus, positive lattice mismatches occur when a:, is greater than a$. The effect of lattice mismatch, symmetry, temperature, and coverage will be considered separately in order to obtain an overall understanding of heteroepitaxial growth in the thermodynamic limit. In this study, the coverage is expressed in terms of monolayers (ML), where 1 ML is defined to be the coverage where there exists one adatom per surface substrate atom. In order to study the isolated effect of lattice mismatch, the thin films are first chosen to have the same fcc(100) symmetry as the substrate. We evaluate the free energies for different film morphologies as described previously. The parallel lattice parameter of the film is varied in the free energy calculation, with the perpendicular lattice parameter of the film adjusted to conserve volume, again as discussed in the previous section. Note that we assume the parallel lattice constant is independent of which adlayer of the film we consider; this is clearly an approximation, but for the ultrathin (l-3 ML) films with which we are concerned, it should be a minor perturbation. For thicker films, a linear interpolation between the substrate and film bulk lattice constants might be a better representation of actual thick films. This remains for future study. In Figs. 2-7, we show some typical curves for the
8 194 T.-M. Chang, EA. Carter/Surface Science 318 (1994) calculated film plus substrate free energy versus film parallel lattice parameter, a,,, for different growth modes. The value of the free energy, A, was calculated from Eq. (6) with MXY set to one. Thus, A is the total system free energy normalized by the number of surface substrate atoms and is in units of ev per surface substrate atom. However, since the substrate atoms that are more than two lattice constants (the potential cutoff) below the interface are not influenced by the presence of the overlayers, the free energies associated with these substrate atoms will only add a constant value to the system s free energy. Therefore, these constant contributions are not included in the calculations of A. The sharp dips in the free energy curves occur at the lattice constant of the underlying substrate metal, corresponding to pseudomorphic growth. This is favored because it minimizes the interfacial strain. The label FM in the figure captions indicates the free energy curves for the FM (layer-by-layer) growth mode. VW(n) represents the VW growth mode with adatoms forming three-dimensional n-layer clusters on top of a bare substrate. SK(n) indicates the SK mode where adatoms form a three-dimensional cluster IZ layers thick on top of one complete monolayer. Note in our calculations, we have included all the contributions to the free energy from each unique atom in different environments, with the exception of edge atoms, whose contribution in the limit of macroscopic island size can be neglected (as argued earlier). The temperature of the systems shown in Figs. 2-7 is 800 K. In the present work, to the extent the Finnis-Sinclair potentials are accurate to 0.01 ev/ atom, we can make conclusions about the preferred film morphology. Given that the size of errors in the potential is unknown, these predictions may be subject to change as more information about the quality of the potential becomes available from future comparisons with experiment. Note that although in some Fig. 2. Free energies (ev/m,, = ev/surface substrate atom) as a function of the parallel lattice spacing (a,, in & of the adlayer for Pt grown on Pd(100) at 800 K, for 0, = 0.9 ML (a) and 1.5 ML (b). In Fig. 2a, 0, f, 0, and X correspond to the free energies for FM, VW(2), VW(3), and VW(4) growth, respectively. In Fig. 2b, 0, +, and 0 correspond to FM, SK(2), and SK(3) growth, respectively. See Section 4 for definitions of FM, VW(n), and SK(n) growth modes.
9 T.-M. Chang, E.A. Carter/Surface Science 318 (1994) Table 1 Bulk lattice constants (in A), surface energies (ev/atom), and bulk cohesive energies (ev/atom) for the perfect (1001 surfaces of fee metals System Bulk lattice Surface Bulk cohesive constant energy b energy Ni (4.44) Pd (3.89) Pt (5.84) Ag (2.95) cu (3.49) Au (3.81) Pb (2.03) Taken from Ref. [60], p. 23. b Energy of formation of surface. See text for details. Bulk cohesive energies are calculated using Finnis-Sinclair potentials, and the experimental values are in parentheses [taken from Ref. [60], p Slight discrepancies exist because the Finnis-Sinclair potentials were fitted to earlier experimental data taken from Kittel(1971). figures, the free energy differences for different growth modes are sometimes less than the thermal energy, transformations between different film morphologies would require collective motion of all the adatoms, which is likely to have a large effective barrier [23]. Thermal fluctuations are unlikely to provide enough energy to overcome this barrier. Fig. 2 displays the free energy as a function of parallel lattice constant for a Pt film grown on a Pd(100) substrate. The small 0.77% lattice mismatch between Pd and Pt leads us to expect pseudomorphic growth, however, whether it will grow layer-by-layer (FM) or via three-dimensional islanding (SK or VW) is less clear. Given that the surface energy of Pt is slightly lower than Pd (see Table 0, FM growth is expected to be slightly favored. Figs. 2a and 2b show the free energy curves at coverages of 0.9 and 1.5 ML, respectively, where we indeed see that the Pt overlayers grow commensurately with the substrate kjd = 3.89 A, see Table 1). It is also clear from the Fig. 3. Free energies. (ev/m,,) as a function of the parallel lattice spacing (a,, in A) of the adlayer for Ag grown on Pt(100) at 800 K, for f& = 0.9 ML (a) and 1.5 ML (b). In Fig. 3a, 0, +, 0, X, and A correspond to the free energies for FM, VW(2), VW(31, VW(41, and VW(51 growth, respectively. In Fig. 3b, 0, +, 0, and X correspond to FM, SK(21, SK(3), and SK(4) growth, respectively.
10 196 T.-M. Chang, E.A. Carter/Surface Science 318 (1994) free energy curves that FM growth has slightly (0.01 ev/atom) lower free energies than SK and VW growth, indicating that layer-by-layer growth is preferred for Pt on Pd(100). We find that pseudomorphic FM growth persists as the slightly thermodynamically favored mechanism for coverages up to at least 3 ML. Figs. 3a and 3b depict the free energy curves for Ag deposited on Pt(100) at coverages of 0.9 and 1.5 ML. This interface is characterized by a 4.3% lattice mismatch. (Note that while clean Pt(100) is known to reconstruct [49], adsorption on Pt(100) lifts the reconstruction; therefore, since we are always examining the case where adsorption has occurred, we are free to consider the unreconstructed Pt(100) lattice.) At all coverages& Ag adopts the substrate lattice constant of 3.92 A, i.e., the film grows pseudomorphically. It is obvious from the free energy curves that at submonolayer coverages, Ag prefers to fill in the first layer instead of forming three-dimensional islands. However, as the coverage exceeds 1 ML, Fig. 3b shows that adatoms can form three-dimensional clusters on top of the first adlayer, since SK structures have slightly (0.02 ev/atom) lower free energies than FM structures. Thus, we predict the thermodynamically preferred growth mode for Ag on Pt(100) is the SK mechanism. Figs. 4a and 4b display free energy curves for Cu grown on Pd(100) at coverages of 0.9 and 1.5 ML, respectively. The lattice mismatch of this interface is %. For coverages up to 2 ML, we find that Cu adlayers grow pseudomorphically with the substrate. At submonolayer coverages, the global minimum in the free energy corresponds to FM growth, indicating that Cu adatoms prefer to form a complete layer instead of three-dimensional clusters. However, as with Ag on Pt(lOO), the situation changes as the coverage increases. Above 1 ML, SK growth has slightly (0.01 ev/atom) lower free energies than the FM growth, suggesting that pseudomorphic SK Fig. 4. Free energieso(ev/mx,) as a function of the parallel lattice spacing (a,, in A) of the adlayer for Cu grown on Pd(100) at 800 K, for Ocu =0.9ML(a)and1.5ML(b).Ina,O, +, 0, and X correspond to the free energies for FM, VW(2), VW(3), and VW(4) growth, respectively. In b, 0, +, and 0 correspond to FM, SK(2), and SK(3) growth, respectively.
11 T.-M. Chang, EA. Carter/Surface Science 318 (1994) growth may be the thermodynamically preferred growth mechanism for Cu grown on Pd(lOO), similar to Ag on Pt(100). Fig. 5 shows free energy curves for growth of Ag on Ni(lOO) at 0.9 ML coverage. The lattice mismatch in this case is extreme: 16.2%. We find that the favored growth mechanism and film morphology does not change with coverage, at least up to 2 ML of Ag. Although there is a local minimum in the fre,e energy at the pseudomorphic lattice constant (3.52 A for Ni(lOO)), the global minimum0 lies close to the bulk Ag lattice constant of 4.09 A. Thus the compressive strain from the large lattice mismatch is immediately relieved, producing an incommensurate overlayer. Interestingly, we predict the growth mode to change as the lattice constant of the film expands, with FM growth for the compressed layer local minimum and VW growth for the fully relaxed global minimum structure. Thus, left to its own devices, Ag immediately will form incommensurate three-dimensional clusters on top of Ni(lOO). Clearly, l l /I Ii,/I,i,P 10 I II 12 (,I,(.%) Fig. 6. Free energieso(ev/m,,) as a function of the parallel lattice spacing (a,, in A) of the adlayer for Ni grown on Ag(100) at 800 K, for ONi = 0.9 ML. 0, +,, X, and A correspond to the free energies for FM, VW(2), VW(3), VW(4), and VW(S) growth, respectively. -7 Fig. 5. Free energieso(ev/mx,) as a function of the parallel lattice spacing (a,, in A) of the adlayer for Ag grown on Ni(100) atsook,foro,,=0.9ml.o, +, 0, X, a,and+correspond to the free energies for FM, VW(2), VW(3), FM,,,, VW,,,(2), and V%,,,(3) growth, respectively. the extreme strain results in the preference for a roughened film, while our results suggest that an externally applied stress on these clusters might force them (depending on the barrier) to collapse ultimately into a smooth commensurate film! Fig. 6 displays free energy curves for the reverse case of Ni grown on Ag(100) at a coverage of 0.9 ML, where we again find these curves to be essentially independent of coverage up to 2 ML. The large negative ( %) lattice mismatch produces quite different behavior than the large positive mismatch case. In particular, when the film has a natural bulk lattice constant larger than the substrate s, incommensurate growth is expected whereas the reverse situation often produces pseudomorphic growth. This point is discussed in more detail below. Indeed, while a shallow local minimum exists near the bulk Ni lattice constant of 3.52 A, the global minimum lies at that of the Ag substrate ( m 4.09 A), leading to a pseudomorphic film. In this case, VW growth is
12 198 T.-M. Chnng, EA. Carter/Surface Science 318 (1994) Table 2 Summary of the thermodynamically preferred growth modes and structures of heteroepitaxial fee metal films, as a function of lattice mismatch and cohesive energy System a Lattice Ratio of Growth mode d P/I e mismatch b Emh Ag/Ni 16.2% 0.66 VW I Pd/Ni 10.5% 0.88 VW I Ag/Pd 5.14% 0.76 SK P Ag/Pt 4.34% 0.51 SK P Pt/Pd 0.77% 1.50 FM P Pd/Pt % 0.67 SK P Pd/Ag % 1.32 VW P Cu/Pd % 0.90 SK P Ni/Pt % 0.76 VW P Ni/Ag % 1.51 VW P Pt/Pb % 2.88 VW I a These results are generally independent of temperature over the range K. System lists the film metal first and the substrate metal second. b Lattice mismatch = (azd /azb - 1) X loo%, where uf and azb are the bulk lattice constants of the metal in the adlayer film and the metal in the substrate, respectively. Ecoh is the bulk cohesive energy of the metal. The ratio is for the film Emh to substrate Eco,,. d Thermodynamic growth mechanism: FM = layer by layer, VW = three-dimensional clustering, SK = mixture of FM and VW (see text). P/I classifies the metal film as growing pseudomorphically (P) or incommensurately (I) on top of the substrate metal. favored over layer-by-layer growth for all lattice constants of the film. Therefore, in contrast to Ag grown on Ni(lOO), no amount of external stress could force this film to grow layer by layer. Table 2 provides a summary for all the film-substrate interfaces we examined. It is clear from Table 2 that film-substrate lattice mismatch is a major factor determining the growth mode and structure of the film, and that relative bulk cohesive energies of the films do not correlate at all with growth mode or structure. Pseudomorphic growth is found for all mismatches in the range of 5.14% to -13.9%. Recall that positive lattice mismatches refer to cases where the film adatoms are larger than the substrate atoms. We see that it is easier to grow a commensurate overlayer for smaller adatoms. This can be understood by realizing that the repulsive part of pairwise atomic interactions generally is steeper than the attractive part, and it costs more energy to compress than to expand atoms by the same degree. Thus, it is less energetically favorable for larger adatoms to be compressed in order to grow pseudomorphically. On the other hand, the smaller adatoms can expand to maintain registry with the substrate in order to release the misfit energy near the interface at a significantly smaller cost in energy. We predict that all large mismatched systems, where the lattice mismatch is at least 10% in absolute value, prefer to grow as three-dimensional clusters (VW growth). For the small mismatched systems with lattice mismatches in the range of -0.77% to 5.14%, the FM or SK growth mode is primarily favored. This is physically reasonable, since these small mismatched interfaces may more closely resemble homoepitaxial systems and the thermodynamic growth mechanism for homoepitaxy must be the FM mode. Deviations from the homoepitaxial case may result in SK growth. SK or FM growth is expected especially for interfaces where the substrate-adatom interaction is stronger than the adatom-adatom interactions. However, as the mismatch increases, more strain energy will be built up in the film if the adatoms remain pseudomorphic with the substrate. Thus, the structures of the films eventually will become incommensurate. At the same time, the growth mode will change to the SK mode in order to minimize the interfacial misfit energies. In order to explain completely the results in Table 2, other factors also need to be taken into account. In Table 1, we list calculated values of the perfect (100) surface energies and the bulk cohesive energies of the fee metals we studied. The surface energies were calculated in the following manner. First, we calculated the total potential energy of a three-dimensionally infinite crystal via periodic boundary conditions in all three directions and where the atoms were placed in their experimentally determined bulk lattice sites. Next, we removed the periodic boundary condition in one direction, thereby creating two surfaces, and calculated the new total potential energy. The surface energy is just half the difference of the two potential energies. Note that this is an upper bound to the true surface energy, since we did not allow the surface to relax. We note that our upper bounds on the surface energies are all at most 10% larger than the values reported by Todd and Lynden-Bell, who calculated surface energies for relaxed surfaces [SO]. We are not concerned here
13 T.-M. Gang, EA. Carter/Surface Science 318 (1994) with reconstruction energetics, only with energy to form a perfect surface. This allows one to more naturally compare surface energies of different metals, since we expect that epitaxial addition of adatoms to form a film will lift any reconstruction anyway. Interestingly, Table 1 shows that the surface energy does not correlate well with the corresponding bulk cohesive energy, and since the surface energy is the critical value here, we concentrate on its impact on the film morphology. In order to distinguish the differences in the growth modes of the small mismatched interfaces, the surface energy itself has to be considered. We suggest the reason that Pd may grow on Pt(100) with the SK mode, while Pt on Pd(100) is predicted to follow the FM mode, is simply that Pd has a slightly higher surface energy than Pt, leading to a slightly larger tendency for Pd to cluster and a slightly larger tendency for Pt to wet surfaces. Since both Ag and Cu have much lower surface energies than Pt or Pd, Ag and Cu will have a strong tendency to wet (cover) a Pt or Pd surface. Interestingly, the difference in surface energies induces an SK growth mode for Ag or Cu grown on a Pd substrate, while the higher surface energy of Pd yields a VW growth mode for Pd grown on a Ag or Cu substrate. Besides lattice mismatch, lattice symmetry might be an important factor in determining the growth mode. For example, a pseudomorphic-incommensurate structural transition from a bcc010) to an fcc(ll1) overlayer has been observed in Monte Carlo simulations for fcc/bcc metal interfaces [31], in agreement with LEED data. For large (N 16%) mismatched fee/ fcc(100) interfaces, it has been observed experimentally that the adatoms may form fu$lll) islands on top of the substrate instead of fcc000) structures [51]. The stability of fcc(ll1) films on an fcc(100) substrate can be understood in the following way. For a large mismatched system, misfit dislocations will exist at the interface, since the preferred structures are incommensurate in order to relieve strain. If now the adlayers form more close-packed f&11) structures, the surface energy may be reduced. Therefore, in certain cases, the (111) orientation of the adlayem might be energetically more stable than fcc(100). Thus, we also calculated the free energies for two high symmetry orientations (i.e., rotations) of fcc(ll1) clusters on fcc(100) Fig. 7. Free energies0 (ev/m,,) as a function of the parallel lattice spacing (a,, in A) of the adlayer for F t grown on Pb(100) at 800 K, for f&, =0.9 ML. 0, +, 0, X, A, *, 0, and 0 correspond to the free energies for FM, VW(2), VW(3), VW(4), FM,,,, VWI11(2), VW,,,(3) and VW,,,(4) growth, respectively. substrates for the most likely cases where this may play a role: namely, the two largest mismatched systems, Ag/Ni(lOO) and Pt/Pb@O), where these are representative of both large positive and negative mismatched interfaces. In Figs. 5 and 7, we will only show results for the fcc(lll)/ fcc(100) orientation that has the lowest free energy. In Fig. 5, we display free energy curves for Ag on Ni(100) at & = 0.9 ML, comparing the FM and VW growth modes for both (100) and (111) film symmetries. The subscript 111 in the figure caption denotes the fcc(lll) symmetry. We see that the differences in free energies between (100) and (111) films is pronounced for the FM growth mode, but is negligible for the global minimum, namely for the tallest cluster formed via the VW growth mode. Thus once the film grows as incommensurate, three-dimensional clusters, it does not care if those clusters are (lllj- or (100)~like. In Fig. 7, we show the free energy curves of Pt on Pb(100) at 800 K for a coverage of 0.9 ML, although
14 200 T.-M. Chang, EA. Carter/Surface Science 318 (1994) the behavior is similar for coverages up to 1.5 ML. In this figure, again the results for both fcc(100) and fcc(ll1) film symmetries are presented. Interestingly, in this case the free energy curves for fcc(100) and fcc(ll1) symmetries behave quite differently. For fcc(ll1) films, FM growth is higher in energy than all VW clusters, with the energy decreasing continuously for increased height of the clusters. Furthermore, all VW(n) clusters are predicted to grow incommensurately, with th, preferred lattice constant close to that of Pt (3.92 A). Once again, we see that large mismatched interfaces favor incommensurate growth of thin three-dimensional clusters. Note also that Pt has a much higher surface energy than Pb (see Table 1). Thus, to simultaneously reduce the interfacial strain and the surface energy, Pt( 111) grows via three-dimensional clustering. By contrast, fcc(100) films grown via the FM mode have a commensurateoglobal minimum at the lattice constant of Pb (4.95 A), while again VW(n) clusters grow incommensurately and are lower in energy for all possible cluster sizes. Thus, the free energy can be reduced most effectively by forming three-dimensional Pt(100) clusters on top of Pb(100). The global minimum in the free energy for VW growth is now shifted to the Pt bulk lattice constant, leading again to incommensurate film structures. We find that both fcc(100) and fcc(ll1) clusters have very close global minimum free energies. Therefore, both large lattice-mismatched cases prefer incommensurate three-dimensional growth, with no strong preference for either (100) or (111) crystallites. In order to examine the effect of temperature on thin film growth, we also evaluated the free energies at room temperature, 300 K, of several of the cases shown in Table 2: Ag on Ni(lOO), Pd on Ni(lOO), Ag on Pt(lOO), Ni on Pt(lOO), and Pt on Pb(lOO). For all of these cases, the growth mode is not predicted to change over the temperature range we examined ( K). Thus, at least in this range and for fee/ fcc(100) interfaces, temperature does not seem important in determining the thermodynamic growth mechanism or the film structure. We observe, however, that relative free energy differences between different film morphologies are modified by temperature. For example, we found that the energy difference between pseudomorphic layer-by-layer growth and incommensurate three-dimensional growth for Pd on Ni(100) decreased with decreasing temperature, although incommensurate VW growth consistently had a lower free energy over the temperature range we studied. We expect that, while the interfaces examined here did not exhibit a temperature dependence over a 500 K range above room temperature, the structures of films grown at temperatures below room temperature will in fact be temperature-dependent due to a decreasing contribution of thermal fluctuations as the temperature is lowered. To verify this, we examined 0 K structures of the Pd/ Ni(lOO) interface via steepest descent energy minimization as a representative case. This simulation utilized six layers of substrate atoms with 242 atoms per layer (the positions of only the top two substrate layers were minimized). This cell size was chosen to minimize the strain induced by periodic boundary conditions [52]. We studied film structures for various morphologies at coverages of 0.8 and 1.5 ML. Using the steepest descent method, we found that layer-by-layer growth is energetically more stable compared to three-dimensional VW growth for Pd on Ni(100) at 0 K, in contrast to the mean-field results at higher temperatures ( K) where three-dimensional growth is the preferred thermal growth mode. We also performed mean-field free energy calculations for Pd grown on Ni(100) at 100 K. Again, we found that layer-by-layer growth is thermodynamically more favorable, contrary to higher-temperature mean-field results. Therefore, it is obvious that mean-field theory is able to predict film morphology as a function of temperature. In addition, both low-temperature studies show that the growth morphology indeed changes with temperature, providing evidence for the need to carry out calculations at finite temperatures. As mentioned previously, Monte Carlo simulations have also suggested that the equilibrium structures sometimes can change with temperature, with pseudomorphic films possible at low temperatures, and incommensurate films at higher temperatures [26]. Zinke-Allmang et al. used a macroscopic equilibrium model to also predict film morphology changes at non-zero temperatures We caution that temperature is bound to have other effects that cannot be probed by the mean-field approach. In particular, the mean-field approximation is only valid when the temperature is not too
* Present address: MSRC, MSIN K1-90, 906 Battelle Blvd., Richland, Structures and Growth Mechanisms for Heteroepitaxial fcc(ll1) Thin Metal Films?
 .I. Phys. Chem. 1995, 99, 7637-7648 7637 Structures and Growth Mechanisms for Heteroepitaxial fcc(ll1) Thin Metal Films? Tsun-Mei Chang' and Emily A. Carter* Department of Chemistry and Biochemistry, University
.I. Phys. Chem. 1995, 99, 7637-7648 7637 Structures and Growth Mechanisms for Heteroepitaxial fcc(ll1) Thin Metal Films? Tsun-Mei Chang' and Emily A. Carter* Department of Chemistry and Biochemistry, University
Reconstruction and intermixing in thin Ge layers on Si 001
 Reconstruction and intermixing in thin Ge layers on Si 001 L. Nurminen, 1 F. Tavazza, 2 D. P. Landau, 1,2 A. Kuronen, 1 and K. Kaski 1 1 Laboratory of Computational Engineering, Helsinki University of
Reconstruction and intermixing in thin Ge layers on Si 001 L. Nurminen, 1 F. Tavazza, 2 D. P. Landau, 1,2 A. Kuronen, 1 and K. Kaski 1 1 Laboratory of Computational Engineering, Helsinki University of
Lecture 30: Kinetics of Epitaxial Growth: Surface Diffusion and
 Lecture 30: Kinetics of Epitaxial Growth: Surface Diffusion and Nucleation Today s topics Understanding the basics of epitaxial techniques used for surface growth of crystalline structures (films, or layers).
Lecture 30: Kinetics of Epitaxial Growth: Surface Diffusion and Nucleation Today s topics Understanding the basics of epitaxial techniques used for surface growth of crystalline structures (films, or layers).
SEMICONDUCTOR GROWTH TECHNIQUES. Introduction to growth techniques (bulk, epitaxy) Basic concepts in epitaxy (MBE, MOCVD)
 SEMICONDUCTOR GROWTH TECHNIQUES Introduction to growth techniques (bulk, epitaxy) Basic concepts in epitaxy (MBE, MOCVD) Growth Processes Bulk techniques (massive semiconductors, wafers): Si, compounds
SEMICONDUCTOR GROWTH TECHNIQUES Introduction to growth techniques (bulk, epitaxy) Basic concepts in epitaxy (MBE, MOCVD) Growth Processes Bulk techniques (massive semiconductors, wafers): Si, compounds
Concepts in Surface Physics
 M.-C. Desjonqueres D. Spanjaard Concepts in Surface Physics Second Edition With 257 Figures Springer 1. Introduction................................. 1 2. Thermodynamical and Statistical Properties of
M.-C. Desjonqueres D. Spanjaard Concepts in Surface Physics Second Edition With 257 Figures Springer 1. Introduction................................. 1 2. Thermodynamical and Statistical Properties of
Available online at Physics Procedia 15 (2011) Stability and Rupture of Alloyed Atomic Terraces on Epitaxial Interfaces
 Available online at www.sciencedirect.com Physics Procedia 15 (2011) 64 70 Stability and Rupture of Alloyed Atomic Terraces on Epitaxial Interfaces Michail Michailov Rostislaw Kaischew Institute of Physical
Available online at www.sciencedirect.com Physics Procedia 15 (2011) 64 70 Stability and Rupture of Alloyed Atomic Terraces on Epitaxial Interfaces Michail Michailov Rostislaw Kaischew Institute of Physical
Bulk Structures of Crystals
 Bulk Structures of Crystals 7 crystal systems can be further subdivided into 32 crystal classes... see Simon Garrett, "Introduction to Surface Analysis CEM924": http://www.cem.msu.edu/~cem924sg/lecturenotes.html
Bulk Structures of Crystals 7 crystal systems can be further subdivided into 32 crystal classes... see Simon Garrett, "Introduction to Surface Analysis CEM924": http://www.cem.msu.edu/~cem924sg/lecturenotes.html
Potentials, periodicity
 Potentials, periodicity Lecture 2 1/23/18 1 Survey responses 2 Topic requests DFT (10), Molecular dynamics (7), Monte Carlo (5) Machine Learning (4), High-throughput, Databases (4) NEB, phonons, Non-equilibrium
Potentials, periodicity Lecture 2 1/23/18 1 Survey responses 2 Topic requests DFT (10), Molecular dynamics (7), Monte Carlo (5) Machine Learning (4), High-throughput, Databases (4) NEB, phonons, Non-equilibrium
Follow this and additional works at: Part of the Physics Commons Department: Physics
 Scholars' Mine Doctoral Dissertations Student Research & Creative Works Summer 2010 A study of the growth and structure of chromium and iron oxide films on Pd(001) using X-ray photoelectron diffraction
Scholars' Mine Doctoral Dissertations Student Research & Creative Works Summer 2010 A study of the growth and structure of chromium and iron oxide films on Pd(001) using X-ray photoelectron diffraction
Lecture 10 Thin Film Growth
 Lecture 10 Thin Film Growth 1/76 Announcements Homework: Homework Number 2 is returned today, please pick it up from me at the end of the class. Solutions are online. Homework 3 will be set Thursday (2
Lecture 10 Thin Film Growth 1/76 Announcements Homework: Homework Number 2 is returned today, please pick it up from me at the end of the class. Solutions are online. Homework 3 will be set Thursday (2
Morphological evolution of single-crystal ultrathin solid films
 Western Kentucky University From the SelectedWorks of Mikhail Khenner March 29, 2010 Morphological evolution of single-crystal ultrathin solid films Mikhail Khenner, Western Kentucky University Available
Western Kentucky University From the SelectedWorks of Mikhail Khenner March 29, 2010 Morphological evolution of single-crystal ultrathin solid films Mikhail Khenner, Western Kentucky University Available
QUASI-EQUILIBRIUM MONTE-CARLO: OFF-LATTICE KINETIC MONTE CARLO SIMULATION OF HETEROEPITAXY WITHOUT SADDLE POINTS
 QUASI-EQUILIBRIUM MONTE-CARLO: OFF-LATTICE KINETIC MONTE CARLO SIMULATION OF HETEROEPITAXY WITHOUT SADDLE POINTS Henry A. Boateng University of Michigan, Ann Arbor Joint work with Tim Schulze and Peter
QUASI-EQUILIBRIUM MONTE-CARLO: OFF-LATTICE KINETIC MONTE CARLO SIMULATION OF HETEROEPITAXY WITHOUT SADDLE POINTS Henry A. Boateng University of Michigan, Ann Arbor Joint work with Tim Schulze and Peter
Mal. Res. Soc. Symp. Proc. Vol Materials Research Society
 91 MOLECULAR-DYNAMICS SIMULATION OF THIN-FILM GROWTH MATTHIAS SCHNEIDER,* IVAN K. SCHULLER,* AND A. RAHMAN Materials Science Division, Argonne National Laboratory, Argonne, IL 60439 Supercomputer Institute,
91 MOLECULAR-DYNAMICS SIMULATION OF THIN-FILM GROWTH MATTHIAS SCHNEIDER,* IVAN K. SCHULLER,* AND A. RAHMAN Materials Science Division, Argonne National Laboratory, Argonne, IL 60439 Supercomputer Institute,
DIFFUSION AND GROWTH ON STRAINED SURFACES
 DIFFUSION AND GROWTH ON STRAINED SURFACES A.C. SCHINDLER AND D.E. WOLF Theoretische Physik, FB 10, Gerhard Mercator Universität Duisburg, 47048 Duisburg, Germany E-mail: A.Schindler@Uni-Duisburg.de Using
DIFFUSION AND GROWTH ON STRAINED SURFACES A.C. SCHINDLER AND D.E. WOLF Theoretische Physik, FB 10, Gerhard Mercator Universität Duisburg, 47048 Duisburg, Germany E-mail: A.Schindler@Uni-Duisburg.de Using
Kinetic Monte Carlo: from transition probabilities to transition rates
 Kinetic Monte Carlo: from transition probabilities to transition rates With MD we can only reproduce the dynamics of the system for 100 ns. Slow thermallyactivated processes, such as diffusion, cannot
Kinetic Monte Carlo: from transition probabilities to transition rates With MD we can only reproduce the dynamics of the system for 100 ns. Slow thermallyactivated processes, such as diffusion, cannot
Epitaxial Growth of Mn on Si(111)
 105 Chapter 7 Epitaxial Growth of Mn on Si(111) 7.1 Introduction There are a few reports and experiments concerning the adsoption of Mn on Si(111), where film growth with and without a Bi surfactant layer
105 Chapter 7 Epitaxial Growth of Mn on Si(111) 7.1 Introduction There are a few reports and experiments concerning the adsoption of Mn on Si(111), where film growth with and without a Bi surfactant layer
Sliding Friction in the Frenkel-Kontorova Model
 arxiv:cond-mat/9510058v1 12 Oct 1995 to appear in "The Physics of Sliding Friction", Ed. B.N.J. Persson (Kluwer Academic Publishers), 1995 Sliding Friction in the Frenkel-Kontorova Model E. Granato I.N.P.E.,
arxiv:cond-mat/9510058v1 12 Oct 1995 to appear in "The Physics of Sliding Friction", Ed. B.N.J. Persson (Kluwer Academic Publishers), 1995 Sliding Friction in the Frenkel-Kontorova Model E. Granato I.N.P.E.,
Monte Carlo simulation of thin-film growth on a surface with a triangular lattice
 Vacuum 52 (1999) 435 440 Monte Carlo simulation of thin-film growth on a surface with a triangular lattice Wei Helin*, Liu Zuli, Yao Kailun Department of Physics, Huazhong University of Science and Technology,
Vacuum 52 (1999) 435 440 Monte Carlo simulation of thin-film growth on a surface with a triangular lattice Wei Helin*, Liu Zuli, Yao Kailun Department of Physics, Huazhong University of Science and Technology,
2. Surface geometric and electronic structure: a primer
 2. Surface geometric and electronic structure: a primer 2.1 Surface crystallography 2.1.1. Crystal structures - A crystal structure is made up of two basic elements: lattice + basis Basis: Lattice: simplest
2. Surface geometric and electronic structure: a primer 2.1 Surface crystallography 2.1.1. Crystal structures - A crystal structure is made up of two basic elements: lattice + basis Basis: Lattice: simplest
Properties of Individual Nanoparticles
 TIGP Introduction technology (I) October 15, 2007 Properties of Individual Nanoparticles Clusters 1. Very small -- difficult to image individual nanoparticles. 2. New physical and/or chemical properties
TIGP Introduction technology (I) October 15, 2007 Properties of Individual Nanoparticles Clusters 1. Very small -- difficult to image individual nanoparticles. 2. New physical and/or chemical properties
Lecture 11 - Phonons II - Thermal Prop. Continued
 Phonons II - hermal Properties - Continued (Kittel Ch. 5) Low High Outline Anharmonicity Crucial for hermal expansion other changes with pressure temperature Gruneisen Constant hermal Heat ransport Phonon
Phonons II - hermal Properties - Continued (Kittel Ch. 5) Low High Outline Anharmonicity Crucial for hermal expansion other changes with pressure temperature Gruneisen Constant hermal Heat ransport Phonon
Kinetic Monte Carlo simulation of nucleation on patterned substrates
 PHYSICAL REVIEW B, VOLUME 63, 035407 Kinetic Monte Carlo simulation of nucleation on patterned substrates L. Nurminen, A. Kuronen, and K. Kaski Helsinki University of Technology, Laboratory of Computational
PHYSICAL REVIEW B, VOLUME 63, 035407 Kinetic Monte Carlo simulation of nucleation on patterned substrates L. Nurminen, A. Kuronen, and K. Kaski Helsinki University of Technology, Laboratory of Computational
Energy Spectroscopy. Ex.: Fe/MgO
 Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
Energy Spectroscopy Spectroscopy gives access to the electronic properties (and thus chemistry, magnetism,..) of the investigated system with thickness dependence Ex.: Fe/MgO Fe O Mg Control of the oxidation
Two simple lattice models of the equilibrium shape and the surface morphology of supported 3D crystallites
 Bull. Nov. Comp. Center, Comp. Science, 27 (2008), 63 69 c 2008 NCC Publisher Two simple lattice models of the equilibrium shape and the surface morphology of supported 3D crystallites Michael P. Krasilnikov
Bull. Nov. Comp. Center, Comp. Science, 27 (2008), 63 69 c 2008 NCC Publisher Two simple lattice models of the equilibrium shape and the surface morphology of supported 3D crystallites Michael P. Krasilnikov
EXCHANGE COUPLING IN MAGNETIC MULTILAYERS GROWN ON IRON WHISKERS (INVITED)
 EXCHANGE COUPLING IN MAGNETIC MULTILAYERS GROWN ON IRON WHISKERS (INVITED) J. Unguris, R. J. Celotta, D. A. Tulchinsky, and D. T. Pierce Electron Physics Group, National Institute of Standards and Technology,
EXCHANGE COUPLING IN MAGNETIC MULTILAYERS GROWN ON IRON WHISKERS (INVITED) J. Unguris, R. J. Celotta, D. A. Tulchinsky, and D. T. Pierce Electron Physics Group, National Institute of Standards and Technology,
THE CHEMICAL PHYSICS OF SOLID SURFACES
 THE CHEMICAL PHYSICS OF SOLID SURFACES EDITED BY D.A.KING B.Sc, Ph.D. (Rand), Sc.D. (East Anglia), F.R.S. 1920 Professor of Physical Chemistry, University of Cambridge AND D.P.WOODRUFF B.Sc. (Bristol),
THE CHEMICAL PHYSICS OF SOLID SURFACES EDITED BY D.A.KING B.Sc, Ph.D. (Rand), Sc.D. (East Anglia), F.R.S. 1920 Professor of Physical Chemistry, University of Cambridge AND D.P.WOODRUFF B.Sc. (Bristol),
MatSci 331 Homework 4 Molecular Dynamics and Monte Carlo: Stress, heat capacity, quantum nuclear effects, and simulated annealing
 MatSci 331 Homework 4 Molecular Dynamics and Monte Carlo: Stress, heat capacity, quantum nuclear effects, and simulated annealing Due Thursday Feb. 21 at 5pm in Durand 110. Evan Reed In this homework,
MatSci 331 Homework 4 Molecular Dynamics and Monte Carlo: Stress, heat capacity, quantum nuclear effects, and simulated annealing Due Thursday Feb. 21 at 5pm in Durand 110. Evan Reed In this homework,
Heterostructures and sub-bands
 Heterostructures and sub-bands (Read Datta 6.1, 6.2; Davies 4.1-4.5) Quantum Wells In a quantum well, electrons are confined in one of three dimensions to exist within a region of length L z. If the barriers
Heterostructures and sub-bands (Read Datta 6.1, 6.2; Davies 4.1-4.5) Quantum Wells In a quantum well, electrons are confined in one of three dimensions to exist within a region of length L z. If the barriers
From Atoms to Materials: Predictive Theory and Simulations
 From Atoms to Materials: Predictive Theory and Simulations Week 3 Lecture 4 Potentials for metals and semiconductors Ale Strachan strachan@purdue.edu School of Materials Engineering & Birck anotechnology
From Atoms to Materials: Predictive Theory and Simulations Week 3 Lecture 4 Potentials for metals and semiconductors Ale Strachan strachan@purdue.edu School of Materials Engineering & Birck anotechnology
Surface Structure and Morphology 2D Crystallography
 Surface Structure and Morphology 2D Crystallography Selvage (or selvedge (it. cimosa)): Region in the solid in the vicinity of the mathematical surface Surface = Substrate (3D periodicity) + Selvage (few
Surface Structure and Morphology 2D Crystallography Selvage (or selvedge (it. cimosa)): Region in the solid in the vicinity of the mathematical surface Surface = Substrate (3D periodicity) + Selvage (few
arxiv: v1 [cond-mat.mtrl-sci] 1 Jun 2017
![arxiv: v1 [cond-mat.mtrl-sci] 1 Jun 2017 arxiv: v1 [cond-mat.mtrl-sci] 1 Jun 2017](/thumbs/76/73160256.jpg) Stranski-Krastanov mechanism of growth and the effect of misfit sign on quantum dots nucleation arxiv:1706.00235v1 [cond-mat.mtrl-sci] 1 Jun 2017 J.E. Prieto 1 and I. Markov 2 1 Centro de Microanálisis
Stranski-Krastanov mechanism of growth and the effect of misfit sign on quantum dots nucleation arxiv:1706.00235v1 [cond-mat.mtrl-sci] 1 Jun 2017 J.E. Prieto 1 and I. Markov 2 1 Centro de Microanálisis
8 Summary and outlook
 91 8 Summary and outlook The main task of present work was to investigate the growth, the atomic and the electronic structures of Co oxide as well as Mn oxide films on Ag(001) by means of STM/STS at LT
91 8 Summary and outlook The main task of present work was to investigate the growth, the atomic and the electronic structures of Co oxide as well as Mn oxide films on Ag(001) by means of STM/STS at LT
Surface Physics Surface Diffusion. Assistant: Dr. Enrico Gnecco NCCR Nanoscale Science
 Surface Physics 008 8. Surface Diffusion Assistant: Dr. Enrico Gnecco NCCR Nanoscale Science Random-Walk Motion Thermal motion of an adatom on an ideal crystal surface: - Thermal excitation the adatom
Surface Physics 008 8. Surface Diffusion Assistant: Dr. Enrico Gnecco NCCR Nanoscale Science Random-Walk Motion Thermal motion of an adatom on an ideal crystal surface: - Thermal excitation the adatom
Solid Surfaces, Interfaces and Thin Films
 Hans Lüth Solid Surfaces, Interfaces and Thin Films Fifth Edition With 427 Figures.2e Springer Contents 1 Surface and Interface Physics: Its Definition and Importance... 1 Panel I: Ultrahigh Vacuum (UHV)
Hans Lüth Solid Surfaces, Interfaces and Thin Films Fifth Edition With 427 Figures.2e Springer Contents 1 Surface and Interface Physics: Its Definition and Importance... 1 Panel I: Ultrahigh Vacuum (UHV)
Document Version Publisher s PDF, also known as Version of Record (includes final page, issue and volume numbers)
 Parametrization of modified embedded-atom-method potentials for Rh, Pd, Ir, and Pt based on density functional theory calculations, with applications to surface properties Beurden, van, P.; Kramer, G.J.
Parametrization of modified embedded-atom-method potentials for Rh, Pd, Ir, and Pt based on density functional theory calculations, with applications to surface properties Beurden, van, P.; Kramer, G.J.
First Principles Investigation of Nickel-Graphene Interfaces
 Research Experience for Undergraduates Report First Principles Investigation of Nickel-Graphene Interfaces by Andrew J. Ross (Saint Anselm College) Advisers: L. Adamska Y. Lin I. I. Oleynik Physics Department
Research Experience for Undergraduates Report First Principles Investigation of Nickel-Graphene Interfaces by Andrew J. Ross (Saint Anselm College) Advisers: L. Adamska Y. Lin I. I. Oleynik Physics Department
Nucleation theory and the early stages of thin film growth
 Nucleation theory and the early stages of thin film growth C. Ratsch a) Department of Mathematics, University of California, Los Angeles, California 90095-1555 J. A. Venables Department of Physics and
Nucleation theory and the early stages of thin film growth C. Ratsch a) Department of Mathematics, University of California, Los Angeles, California 90095-1555 J. A. Venables Department of Physics and
Introductory Nanotechnology ~ Basic Condensed Matter Physics ~
 Introductory Nanotechnology ~ Basic Condensed Matter Physics ~ Atsufumi Hirohata Department of Electronics Go into Nano-Scale Lateral Size [m] 10-3 10-6 Micron-scale Sub-Micron-scale Nano-scale Human hair
Introductory Nanotechnology ~ Basic Condensed Matter Physics ~ Atsufumi Hirohata Department of Electronics Go into Nano-Scale Lateral Size [m] 10-3 10-6 Micron-scale Sub-Micron-scale Nano-scale Human hair
Monte Carlo simulations of alloy segregation in PtAg octahedral nanoparticles
 Monte Carlo simulations of alloy segregation in PtAg octahedral nanoparticles Louis C. Jones 6/8/12 Abstract Simulations were carried out to investigate phase segregation of insoluble alloy nanoparticles
Monte Carlo simulations of alloy segregation in PtAg octahedral nanoparticles Louis C. Jones 6/8/12 Abstract Simulations were carried out to investigate phase segregation of insoluble alloy nanoparticles
Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic Structure Analyses
 Materials 2010, 3, 3965-3993; doi:10.3390/ma3073965 Review OPEN ACCESS materials ISSN 1996-1944 www.mdpi.com/journal/materials Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic
Materials 2010, 3, 3965-3993; doi:10.3390/ma3073965 Review OPEN ACCESS materials ISSN 1996-1944 www.mdpi.com/journal/materials Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic
AN ALTERNATIVE MODEL OF THE RECONSTRUCTED (100) SURFACES OF GOLD, PLATINUM AND IRIDIUM
 Surface Science 97 (1980) 257-263 0 North-Holland Publishing Company AN ALTERNATIVE MODEL OF THE RECONSTRUCTED (100) SURFACES OF GOLD, PLATINUM AND IRIDIUM JP. BIBERIAN Centre de Recherches sur les M&wtismes
Surface Science 97 (1980) 257-263 0 North-Holland Publishing Company AN ALTERNATIVE MODEL OF THE RECONSTRUCTED (100) SURFACES OF GOLD, PLATINUM AND IRIDIUM JP. BIBERIAN Centre de Recherches sur les M&wtismes
Relaxation of a Strained Elastic Film on a Viscous Layer
 Mat. Res. Soc. Symp. Proc. Vol. 695 Materials Research Society Relaxation of a Strained Elastic Film on a Viscous Layer R. Huang 1, H. Yin, J. Liang 3, K. D. Hobart 4, J. C. Sturm, and Z. Suo 3 1 Department
Mat. Res. Soc. Symp. Proc. Vol. 695 Materials Research Society Relaxation of a Strained Elastic Film on a Viscous Layer R. Huang 1, H. Yin, J. Liang 3, K. D. Hobart 4, J. C. Sturm, and Z. Suo 3 1 Department
Quantum dot heterostructures: fabrication, properties, lasers Review
 SEMICONDUCTORS VOLUME 32, NUMBER 4 APRIL 1998 Quantum dot heterostructures: fabrication, properties, lasers Review N. N. Ledentsov, V. M. Ustinov, V. A. Shchukin. P. S. Kop ev, and Zh. I. Alferov A. F.
SEMICONDUCTORS VOLUME 32, NUMBER 4 APRIL 1998 Quantum dot heterostructures: fabrication, properties, lasers Review N. N. Ledentsov, V. M. Ustinov, V. A. Shchukin. P. S. Kop ev, and Zh. I. Alferov A. F.
Off-Lattice KMC Simulation of Quantum Dot Formation
 Off-Lattice KMC Simulation of Quantum Dot Formation T. P. Schulze University of Tennessee Peter Smereka & Henry Boateng University of Michigan Research supported by NSF-DMS-0854920 Heteroepitaxy and Quantum
Off-Lattice KMC Simulation of Quantum Dot Formation T. P. Schulze University of Tennessee Peter Smereka & Henry Boateng University of Michigan Research supported by NSF-DMS-0854920 Heteroepitaxy and Quantum
Reactive potentials and applications
 1.021, 3.021, 10.333, 22.00 Introduction to Modeling and Simulation Spring 2011 Part I Continuum and particle methods Reactive potentials and applications Lecture 8 Markus J. Buehler Laboratory for Atomistic
1.021, 3.021, 10.333, 22.00 Introduction to Modeling and Simulation Spring 2011 Part I Continuum and particle methods Reactive potentials and applications Lecture 8 Markus J. Buehler Laboratory for Atomistic
Kinetic Monte Carlo (KMC)
 Kinetic Monte Carlo (KMC) Molecular Dynamics (MD): high-frequency motion dictate the time-step (e.g., vibrations). Time step is short: pico-seconds. Direct Monte Carlo (MC): stochastic (non-deterministic)
Kinetic Monte Carlo (KMC) Molecular Dynamics (MD): high-frequency motion dictate the time-step (e.g., vibrations). Time step is short: pico-seconds. Direct Monte Carlo (MC): stochastic (non-deterministic)
G. L. KELLOGG. Sandia National Laboratories Albuquerque, NM USA. 1. Abstract
 ' C' 54NB 9s - q27 r, coup -C/606222-- AN ATOMC VEW OF CLUSTER DFFUSON ON METAL SURFACES G. L. KELLOGG Sandia National Laboratories Albuquerque, NM 8785-43 USA. Abstract Field ion microscope measurements
' C' 54NB 9s - q27 r, coup -C/606222-- AN ATOMC VEW OF CLUSTER DFFUSON ON METAL SURFACES G. L. KELLOGG Sandia National Laboratories Albuquerque, NM 8785-43 USA. Abstract Field ion microscope measurements
CO Adsorption on Co(0001)-Supported Pt Overlayers
 Int. J. Mol. Sci. 2001, 2, 246-250 International Journal of Molecular Sciences ISSN 1422-0067 2001 by MDPI www.mdpi.org/ijms/ CO Adsorption on Co(0001)-Supported Pt Overlayers P. Légaré 1 *, B. Madani
Int. J. Mol. Sci. 2001, 2, 246-250 International Journal of Molecular Sciences ISSN 1422-0067 2001 by MDPI www.mdpi.org/ijms/ CO Adsorption on Co(0001)-Supported Pt Overlayers P. Légaré 1 *, B. Madani
STM spectroscopy (STS)
 STM spectroscopy (STS) di dv 4 e ( E ev, r) ( E ) M S F T F Basic concepts of STS. With the feedback circuit open the variation of the tunneling current due to the application of a small oscillating voltage
STM spectroscopy (STS) di dv 4 e ( E ev, r) ( E ) M S F T F Basic concepts of STS. With the feedback circuit open the variation of the tunneling current due to the application of a small oscillating voltage
3.091 Introduction to Solid State Chemistry. Lecture Notes No. 6a BONDING AND SURFACES
 3.091 Introduction to Solid State Chemistry Lecture Notes No. 6a BONDING AND SURFACES 1. INTRODUCTION Surfaces have increasing importance in technology today. Surfaces become more important as the size
3.091 Introduction to Solid State Chemistry Lecture Notes No. 6a BONDING AND SURFACES 1. INTRODUCTION Surfaces have increasing importance in technology today. Surfaces become more important as the size
Elastic constants and the effect of strain on monovacancy concentration in fcc hard-sphere crystals
 PHYSICAL REVIEW B 70, 214113 (2004) Elastic constants and the effect of strain on monovacancy concentration in fcc hard-sphere crystals Sang Kyu Kwak and David A. Kofke Department of Chemical and Biological
PHYSICAL REVIEW B 70, 214113 (2004) Elastic constants and the effect of strain on monovacancy concentration in fcc hard-sphere crystals Sang Kyu Kwak and David A. Kofke Department of Chemical and Biological
Machine learning the Born-Oppenheimer potential energy surface: from molecules to materials. Gábor Csányi Engineering Laboratory
 Machine learning the Born-Oppenheimer potential energy surface: from molecules to materials Gábor Csányi Engineering Laboratory Interatomic potentials for molecular dynamics Transferability biomolecular
Machine learning the Born-Oppenheimer potential energy surface: from molecules to materials Gábor Csányi Engineering Laboratory Interatomic potentials for molecular dynamics Transferability biomolecular
Microscopical and Microanalytical Methods (NANO3)
 Microscopical and Microanalytical Methods (NANO3) 06.11.15 10:15-12:00 Introduction - SPM methods 13.11.15 10:15-12:00 STM 20.11.15 10:15-12:00 STS Erik Zupanič erik.zupanic@ijs.si stm.ijs.si 27.11.15
Microscopical and Microanalytical Methods (NANO3) 06.11.15 10:15-12:00 Introduction - SPM methods 13.11.15 10:15-12:00 STM 20.11.15 10:15-12:00 STS Erik Zupanič erik.zupanic@ijs.si stm.ijs.si 27.11.15
Phase diagrams for surface alloys
 Downloaded from orbit.dtu.dk on: Mar 06, 2019 Phase diagrams for surface alloys Christensen, Asbjørn; Ruban, Andrei; Stoltze, Per; Jacobsen, Karsten Wedel; Skriver, Hans Lomholt; Nørskov, Jens Kehlet;
Downloaded from orbit.dtu.dk on: Mar 06, 2019 Phase diagrams for surface alloys Christensen, Asbjørn; Ruban, Andrei; Stoltze, Per; Jacobsen, Karsten Wedel; Skriver, Hans Lomholt; Nørskov, Jens Kehlet;
Effect of Strain on Structure and Morphology of Ultrathin Ge Films on Si(001)
 Chem. Rev. 1997, 97, 1045 1061 1045 Effect of Strain on Structure and Morphology of Ultrathin Ge Films on Si(001) Feng Liu, Fang Wu, and M. G. Lagally* University of WisconsinsMadison, Madison, Wisconsin
Chem. Rev. 1997, 97, 1045 1061 1045 Effect of Strain on Structure and Morphology of Ultrathin Ge Films on Si(001) Feng Liu, Fang Wu, and M. G. Lagally* University of WisconsinsMadison, Madison, Wisconsin
5.8 Auger Electron Spectroscopy (AES)
 5.8 Auger Electron Spectroscopy (AES) 5.8.1 The Auger Process X-ray and high energy electron bombardment of atom can create core hole Core hole will eventually decay via either (i) photon emission (x-ray
5.8 Auger Electron Spectroscopy (AES) 5.8.1 The Auger Process X-ray and high energy electron bombardment of atom can create core hole Core hole will eventually decay via either (i) photon emission (x-ray
"Enhanced Layer Coverage of Thin Films by Oblique Angle Deposition"
 Mater. Res. Soc. Symp. Proc. Vol. 859E 2005 Materials Research Society JJ9.5.1 "Enhanced Layer Coverage of Thin Films by Oblique Angle Deposition" * karabt@rpi.edu Tansel Karabacak *, Gwo-Ching Wang, and
Mater. Res. Soc. Symp. Proc. Vol. 859E 2005 Materials Research Society JJ9.5.1 "Enhanced Layer Coverage of Thin Films by Oblique Angle Deposition" * karabt@rpi.edu Tansel Karabacak *, Gwo-Ching Wang, and
Formation and coarsening of Ag(110) bilayer islands on NiAl(110): STM analysis and atomistic lattice-gas modeling
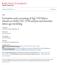 Chemistry Publications Chemistry 2010 Formation and coarsening of Ag(110) bilayer islands on NiAl(110): STM analysis and atomistic lattice-gas modeling Yong Han Iowa State University, y27h@iastate.edu
Chemistry Publications Chemistry 2010 Formation and coarsening of Ag(110) bilayer islands on NiAl(110): STM analysis and atomistic lattice-gas modeling Yong Han Iowa State University, y27h@iastate.edu
Polymer Solution Thermodynamics:
 Polymer Solution Thermodynamics: 3. Dilute Solutions with Volume Interactions Brownian particle Polymer coil Self-Avoiding Walk Models While the Gaussian coil model is useful for describing polymer solutions
Polymer Solution Thermodynamics: 3. Dilute Solutions with Volume Interactions Brownian particle Polymer coil Self-Avoiding Walk Models While the Gaussian coil model is useful for describing polymer solutions
Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic Structure Analyses
 Physics and Astronomy Publications Physics and Astronomy 7-2010 Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic Structure Analyses Yong Han Iowa State University Baris
Physics and Astronomy Publications Physics and Astronomy 7-2010 Nanoscale Quantum Islands on Metal Substrates: Microscopy Studies and Electronic Structure Analyses Yong Han Iowa State University Baris
Kinetic Monte Carlo Simulation of Two-dimensional Semiconductor Quantum Dots Growth
 Kinetic Monte Carlo Simulation of Two-dimensional Semiconductor Quantum Dots Growth by Ernie Pan Richard Zhu Melissa Sun Peter Chung Computer Modeling and Simulation Group The University of Akron Outline
Kinetic Monte Carlo Simulation of Two-dimensional Semiconductor Quantum Dots Growth by Ernie Pan Richard Zhu Melissa Sun Peter Chung Computer Modeling and Simulation Group The University of Akron Outline
Today we begin the first technical topic related directly to the course that is: Equilibrium Carrier Concentration.
 Solid State Devices Dr. S. Karmalkar Department of Electronics and Communication Engineering Indian Institute of Technology, Madras Lecture - 3 Equilibrium and Carrier Concentration Today we begin the
Solid State Devices Dr. S. Karmalkar Department of Electronics and Communication Engineering Indian Institute of Technology, Madras Lecture - 3 Equilibrium and Carrier Concentration Today we begin the
3.320 Lecture 23 (5/3/05)
 3.320 Lecture 23 (5/3/05) Faster, faster,faster Bigger, Bigger, Bigger Accelerated Molecular Dynamics Kinetic Monte Carlo Inhomogeneous Spatial Coarse Graining 5/3/05 3.320 Atomistic Modeling of Materials
3.320 Lecture 23 (5/3/05) Faster, faster,faster Bigger, Bigger, Bigger Accelerated Molecular Dynamics Kinetic Monte Carlo Inhomogeneous Spatial Coarse Graining 5/3/05 3.320 Atomistic Modeling of Materials
SUPPLEMENTARY INFORMATION
 In the format provided by the authors and unedited. Intrinsically patterned two-dimensional materials for selective adsorption of molecules and nanoclusters X. Lin 1,, J. C. Lu 1,, Y. Shao 1,, Y. Y. Zhang
In the format provided by the authors and unedited. Intrinsically patterned two-dimensional materials for selective adsorption of molecules and nanoclusters X. Lin 1,, J. C. Lu 1,, Y. Shao 1,, Y. Y. Zhang
Christian Ratsch, UCLA
 Strain Dependence of Microscopic Parameters and its Effects on Ordering during Epitaxial Growth Christian Ratsch, UCLA Institute for Pure and Applied Mathematics, and Department of Mathematics Collaborators:
Strain Dependence of Microscopic Parameters and its Effects on Ordering during Epitaxial Growth Christian Ratsch, UCLA Institute for Pure and Applied Mathematics, and Department of Mathematics Collaborators:
Energy Spectroscopy. Excitation by means of a probe
 Energy Spectroscopy Excitation by means of a probe Energy spectral analysis of the in coming particles -> XAS or Energy spectral analysis of the out coming particles Different probes are possible: Auger
Energy Spectroscopy Excitation by means of a probe Energy spectral analysis of the in coming particles -> XAS or Energy spectral analysis of the out coming particles Different probes are possible: Auger
Adsorption of Iodine on Pt(111) surface. Alexandre Tkachenko Marcelo Galván Nikola Batina
 Adsorption of Iodine on Pt(111) surface Alexandre Tkachenko Marcelo Galván Nikola Batina Outline Motivation Experimental results Geometry Ab initio study Conclusions Motivation Unusual structural richness
Adsorption of Iodine on Pt(111) surface Alexandre Tkachenko Marcelo Galván Nikola Batina Outline Motivation Experimental results Geometry Ab initio study Conclusions Motivation Unusual structural richness
ABSTRACT INTRODUCTION
 Mat. Res. Soc. Symp. Proc. Vol. 696 22 Materials Research Society Surface reconstruction and induced uniaxial magnetic fields on Ni films R. A. Lukaszew, B. McNaughton 1, V. Stoica 2 and R. Clarke 2 Department
Mat. Res. Soc. Symp. Proc. Vol. 696 22 Materials Research Society Surface reconstruction and induced uniaxial magnetic fields on Ni films R. A. Lukaszew, B. McNaughton 1, V. Stoica 2 and R. Clarke 2 Department
Rustam Z. Khaliullin University of Zürich
 Rustam Z. Khaliullin University of Zürich Molecular dynamics (MD) MD is a computational method for simulating time evolution of a collection of interacting atoms by numerically integrating Newton s equation
Rustam Z. Khaliullin University of Zürich Molecular dynamics (MD) MD is a computational method for simulating time evolution of a collection of interacting atoms by numerically integrating Newton s equation
Part III: Theoretical Surface Science Adsorption at Surfaces
 Technische Universität München Part III: Theoretical Surface Science Adsorption at Surfaces Karsten Reuter Lecture course: Solid State Theory Adsorption at surfaces (T,p) Phase II Phase I Corrosion Growth
Technische Universität München Part III: Theoretical Surface Science Adsorption at Surfaces Karsten Reuter Lecture course: Solid State Theory Adsorption at surfaces (T,p) Phase II Phase I Corrosion Growth
Semiconductor physics I. The Crystal Structure of Solids
 Lecture 3 Semiconductor physics I The Crystal Structure of Solids 1 Semiconductor materials Types of solids Space lattices Atomic Bonding Imperfection and doping in SOLIDS 2 Semiconductor Semiconductors
Lecture 3 Semiconductor physics I The Crystal Structure of Solids 1 Semiconductor materials Types of solids Space lattices Atomic Bonding Imperfection and doping in SOLIDS 2 Semiconductor Semiconductors
disordered, ordered and coherent with the substrate, and ordered but incoherent with the substrate.
 5. Nomenclature of overlayer structures Thus far, we have been discussing an ideal surface, which is in effect the structure of the topmost substrate layer. The surface (selvedge) layers of the solid however
5. Nomenclature of overlayer structures Thus far, we have been discussing an ideal surface, which is in effect the structure of the topmost substrate layer. The surface (selvedge) layers of the solid however
Finite-temperature magnetism of ultrathin lms and nanoclusters PhD Thesis Booklet. Levente Rózsa Supervisor: László Udvardi
 Finite-temperature magnetism of ultrathin lms and nanoclusters PhD Thesis Booklet Levente Rózsa Supervisor: László Udvardi BME 2016 Background of the research Magnetic materials continue to play an ever
Finite-temperature magnetism of ultrathin lms and nanoclusters PhD Thesis Booklet Levente Rózsa Supervisor: László Udvardi BME 2016 Background of the research Magnetic materials continue to play an ever
PHYSICAL REVIEW B 67, Two-dimensional alloy of immiscible metals: Single and binary monolayer films of Pb and Sn on Rh 111
 PHYSICAL REVIEW B 67, 195407 2003 Two-dimensional alloy of immiscible metals: Single and binary monolayer films of Pb and Sn on Rh 111 J. Yuhara, 1,2, * M. Schmid, 2 and P. Varga 2 1 Department of Crystalline
PHYSICAL REVIEW B 67, 195407 2003 Two-dimensional alloy of immiscible metals: Single and binary monolayer films of Pb and Sn on Rh 111 J. Yuhara, 1,2, * M. Schmid, 2 and P. Varga 2 1 Department of Crystalline
Surface Sensitivity & Surface Specificity
 Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Surface Sensitivity & Surface Specificity The problems of sensitivity and detection limits are common to all forms of spectroscopy. In its simplest form, the question of sensitivity boils down to whether
Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces
 Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces R. M. Feenstra Carnegie Mellon University Department of Physics Pittsburgh, PA 15213, USA. J. E. Northrup Palo Alto Research Center, Inc. 3333
Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces R. M. Feenstra Carnegie Mellon University Department of Physics Pittsburgh, PA 15213, USA. J. E. Northrup Palo Alto Research Center, Inc. 3333
An EAM potential for the dynamical simulation of Ni-Al alloys
 J. At. Mol. Sci. doi: 10.4208/jams.022310.031210a Vol. 1, No. 3, pp. 253-261 August 2010 An EAM potential for the dynamical simulation of Ni-Al alloys Jian-Hua Zhang, Shun-Qing Wu, Yu-Hua Wen, and Zi-Zhong
J. At. Mol. Sci. doi: 10.4208/jams.022310.031210a Vol. 1, No. 3, pp. 253-261 August 2010 An EAM potential for the dynamical simulation of Ni-Al alloys Jian-Hua Zhang, Shun-Qing Wu, Yu-Hua Wen, and Zi-Zhong
First-principles calculations of bulk and interfacial thermodynamic properties for fcc-based Al-Sc alloys
 PHYSICAL REVIEW B VOLUME 57, NUMBER 18 1 MAY 1998-II First-principles calculations of bulk and interfacial thermodynamic properties for fcc-based Al-Sc alloys Mark Asta, S. M. Foiles, and A. A. Quong*
PHYSICAL REVIEW B VOLUME 57, NUMBER 18 1 MAY 1998-II First-principles calculations of bulk and interfacial thermodynamic properties for fcc-based Al-Sc alloys Mark Asta, S. M. Foiles, and A. A. Quong*
LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb
 LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb O.D. DUBON, P.G. EVANS, J.F. CHERVINSKY, F. SPAEPEN, M.J. AZIZ, and J.A. GOLOVCHENKO Division of Engineering and Applied Sciences,
LOW-TEMPERATURE Si (111) HOMOEPITAXY AND DOPING MEDIATED BY A MONOLAYER OF Pb O.D. DUBON, P.G. EVANS, J.F. CHERVINSKY, F. SPAEPEN, M.J. AZIZ, and J.A. GOLOVCHENKO Division of Engineering and Applied Sciences,
Flux heterogeneity through incidence angle and particle energy in steering-enhanced growth
 Flux heterogeneity through incidence angle and particle energy in steering-enhanced growth Herbert Wormeester* and Bene Poelsema MESA Research Institute, University of Twente, P.O. Box 217, 7500 AE Enschede,
Flux heterogeneity through incidence angle and particle energy in steering-enhanced growth Herbert Wormeester* and Bene Poelsema MESA Research Institute, University of Twente, P.O. Box 217, 7500 AE Enschede,
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
DOI: 10.1038/NCHEM.2491 Experimental Realization of Two-dimensional Boron Sheets Baojie Feng 1, Jin Zhang 1, Qing Zhong 1, Wenbin Li 1, Shuai Li 1, Hui Li 1, Peng Cheng 1, Sheng Meng 1,2, Lan Chen 1 and
Lecture contents. Stress and strain Deformation potential. NNSE 618 Lecture #23
 1 Lecture contents Stress and strain Deformation potential Few concepts from linear elasticity theory : Stress and Strain 6 independent components 2 Stress = force/area ( 3x3 symmetric tensor! ) ij ji
1 Lecture contents Stress and strain Deformation potential Few concepts from linear elasticity theory : Stress and Strain 6 independent components 2 Stress = force/area ( 3x3 symmetric tensor! ) ij ji
Surface physics, Bravais lattice
 Surface physics, Bravais lattice 1. Structure of the solid surface characterized by the (Bravais) lattice + space + point group lattice describes also the symmetry of the solid material vector directions
Surface physics, Bravais lattice 1. Structure of the solid surface characterized by the (Bravais) lattice + space + point group lattice describes also the symmetry of the solid material vector directions
Southeast University, Nanjing, China 2 Department of Applied Physics, Aalto University,
 Supplementary Information to Solubility of Boron, Carbon and Nitrogen in Transition Metals: Getting Insight into Trends from First-Principles Calculations Xiaohui Hu, 1,2 Torbjörn Björkman 2,3, Harri Lipsanen
Supplementary Information to Solubility of Boron, Carbon and Nitrogen in Transition Metals: Getting Insight into Trends from First-Principles Calculations Xiaohui Hu, 1,2 Torbjörn Björkman 2,3, Harri Lipsanen
Monte Carlo Simulation of Pulsed Laser Deposition
 Monte Carlo Simulation of Pulsed Laser Deposition Pui-Man Lam @, S. J. Liu, and C.H. Woo Department of Mechanical Engineering, The Hong Kong Polytechnic University, Hong Kong ABSTRACT Using the Monte Carlo
Monte Carlo Simulation of Pulsed Laser Deposition Pui-Man Lam @, S. J. Liu, and C.H. Woo Department of Mechanical Engineering, The Hong Kong Polytechnic University, Hong Kong ABSTRACT Using the Monte Carlo
Plan for Lectures #4, 5, & 6. Theme Of Lectures: Nano-Fabrication
 Plan for Lectures #4, 5, & 6 Theme Of Lectures: Nano-Fabrication Quantum Wells, SLs, Epitaxial Quantum Dots Carbon Nanotubes, Semiconductor Nanowires Self-assembly and Self-organization Two Approaches
Plan for Lectures #4, 5, & 6 Theme Of Lectures: Nano-Fabrication Quantum Wells, SLs, Epitaxial Quantum Dots Carbon Nanotubes, Semiconductor Nanowires Self-assembly and Self-organization Two Approaches
Tensor-LEED Analysis of Oxide Surfaces
 Chapter 6 Tensor-LEED Analysis of xide Surfaces 6.1 The Ag(100) Surface 6.1.1 Introduction The use of LEED as a technique for surface structural determination began with the study of clean surfaces. Low
Chapter 6 Tensor-LEED Analysis of xide Surfaces 6.1 The Ag(100) Surface 6.1.1 Introduction The use of LEED as a technique for surface structural determination began with the study of clean surfaces. Low
Introduction to Solid State Physics or the study of physical properties of matter in a solid phase
 Introduction to Solid State Physics or the study of physical properties of matter in a solid phase Prof. Germar Hoffmann 1. Crystal Structures 2. Reciprocal Lattice 3. Crystal Binding and Elastic Constants
Introduction to Solid State Physics or the study of physical properties of matter in a solid phase Prof. Germar Hoffmann 1. Crystal Structures 2. Reciprocal Lattice 3. Crystal Binding and Elastic Constants
(1 2) (1 3) Excess free energy (Jm -2 ) Coverage (ML)
 0.06 0.04 (1 2) (1 3) Excess free energy (Jm -2 ) 0.02 0.0-0.02-0.04-0.06-0.08-0.1-0.12 0 1 2 3 4 5 6 Coverage (ML) Atomic exchange mean eld study of intermixing of Au on Ag(110) J. A. Nieminen Department
0.06 0.04 (1 2) (1 3) Excess free energy (Jm -2 ) 0.02 0.0-0.02-0.04-0.06-0.08-0.1-0.12 0 1 2 3 4 5 6 Coverage (ML) Atomic exchange mean eld study of intermixing of Au on Ag(110) J. A. Nieminen Department
In order to determine the energy level alignment of the interface between cobalt and
 SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
SUPPLEMENTARY INFORMATION Energy level alignment of the CuPc/Co interface In order to determine the energy level alignment of the interface between cobalt and CuPc, we have performed one-photon photoemission
arxiv:cond-mat/ v2 [cond-mat.mtrl-sci] 11 May 2000
![arxiv:cond-mat/ v2 [cond-mat.mtrl-sci] 11 May 2000 arxiv:cond-mat/ v2 [cond-mat.mtrl-sci] 11 May 2000](/thumbs/88/117731541.jpg) Coherent Stranski-Krastanov growth in 1+1 dimensions with anharmonic interactions: An equilibrium study arxiv:cond-mat/0001180v2 [cond-mat.mtrl-sci] 11 May 2000 Elka Korutcheva 1,, Antonio M. Turiel 2
Coherent Stranski-Krastanov growth in 1+1 dimensions with anharmonic interactions: An equilibrium study arxiv:cond-mat/0001180v2 [cond-mat.mtrl-sci] 11 May 2000 Elka Korutcheva 1,, Antonio M. Turiel 2
Scanning Tunneling Microscopy. how does STM work? the quantum mechanical picture example of images how can we understand what we see?
 Scanning Tunneling Microscopy how does STM work? the quantum mechanical picture example of images how can we understand what we see? Observation of adatom diffusion with a field ion microscope Scanning
Scanning Tunneling Microscopy how does STM work? the quantum mechanical picture example of images how can we understand what we see? Observation of adatom diffusion with a field ion microscope Scanning
Why Heteroepitaxy? Difficult to Model Multiple Species Dislocations and point defects important Species Flux important
 Why Heteroepitaxy? Difficult to Model Multiple Species Dislocations and point defects important Species Flux important The Silicon Age is Ending New materials and growth required Useful in design of Devices
Why Heteroepitaxy? Difficult to Model Multiple Species Dislocations and point defects important Species Flux important The Silicon Age is Ending New materials and growth required Useful in design of Devices
Module 16. Diffusion in solids II. Lecture 16. Diffusion in solids II
 Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
EFFECTS OF STOICHIOMETRY ON POINT DEFECTS AND IMPURITIES IN GALLIUM NITRIDE
 EFFECTS OF STOICHIOMETRY ON POINT DEFECTS AND IMPURITIES IN GALLIUM NITRIDE C. G. VAN DE WALLE AND J. E. NORTHRUP Palo Alto Research Center, 3333 Coyote Hill Road, Palo Alto, CA 930, USA E-mail: vandewalle@parc.com
EFFECTS OF STOICHIOMETRY ON POINT DEFECTS AND IMPURITIES IN GALLIUM NITRIDE C. G. VAN DE WALLE AND J. E. NORTHRUP Palo Alto Research Center, 3333 Coyote Hill Road, Palo Alto, CA 930, USA E-mail: vandewalle@parc.com
Sponsored by. Contract No. N K-0073: Modification P00006 DARPA Order 5674 NR
 OTIC FILE COP Study of Interfacial Chemistry between Metals and Their Effects on Electronic Systems q. o Sponsored by 00 Defense Advanced Research Projects Agency (DOD) and The Office of Naval Research
OTIC FILE COP Study of Interfacial Chemistry between Metals and Their Effects on Electronic Systems q. o Sponsored by 00 Defense Advanced Research Projects Agency (DOD) and The Office of Naval Research
Kinetic Monte Carlo simulation of semiconductor quantum dot growth
 Solid State Phenomena Online: 2007-03-15 ISSN: 1662-9779, Vols. 121-123, pp 1073-1076 doi:10.4028/www.scientific.net/ssp.121-123.1073 2007 Trans Tech Publications, Switzerland Kinetic Monte Carlo simulation
Solid State Phenomena Online: 2007-03-15 ISSN: 1662-9779, Vols. 121-123, pp 1073-1076 doi:10.4028/www.scientific.net/ssp.121-123.1073 2007 Trans Tech Publications, Switzerland Kinetic Monte Carlo simulation
Surface atoms/molecules of a material act as an interface to its surrounding environment;
 1 Chapter 1 Thesis Overview Surface atoms/molecules of a material act as an interface to its surrounding environment; their properties are often complicated by external adsorbates/species on the surface
1 Chapter 1 Thesis Overview Surface atoms/molecules of a material act as an interface to its surrounding environment; their properties are often complicated by external adsorbates/species on the surface
Supplementary information
 Supplementary information Supplementary Figure S1STM images of four GNBs and their corresponding STS spectra. a-d, STM images of four GNBs are shown in the left side. The experimental STS data with respective
Supplementary information Supplementary Figure S1STM images of four GNBs and their corresponding STS spectra. a-d, STM images of four GNBs are shown in the left side. The experimental STS data with respective
Review of Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces
 M RS Internet Journal Nitride Semiconductor Research Review of Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces R. M. Feenstra 1, J. E. Northrup 2 and Jörg Neugebauer 3 1 Department of Physics,
M RS Internet Journal Nitride Semiconductor Research Review of Structure of Bare and Adsorbate-Covered GaN(0001) Surfaces R. M. Feenstra 1, J. E. Northrup 2 and Jörg Neugebauer 3 1 Department of Physics,
