68 MACROMOLECULES. Z + ln Z (2.233) C = T ds dt. Nk B (ln G) 2 [ 1 12 N 1. Thus, the specific heat per bond diverges in the thermodynamic limit.
|
|
|
- Vincent Carter
- 5 years ago
- Views:
Transcription
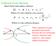
1 68 MACROMOLECULES Hence S = T ln Z + ln Z (2.232) ( T = x ln G ) Z + ln Z (2.233) x x = θ ln G x + ln(xn 1) ln(x 1). (2.234) S θ ln G (2.235) for Nɛ << 1. The entropy is proportional to the order parameter. For the specific heat we find C = T ds dt (2.236) in the neighbourhood of T c C k B (ln G) 2 d θ dɛ Nk B (ln G) 2 [ 1 12 N N 3 ɛ ] (2.237). (2.238) Thus, the specific heat per bond diverges in the thermodynamic limit. 2.5 Polyelectrolyte Polyelectrolytes are one of the least understood states of condensed matter, in contrast to neutral polymer solutions. Recall that a chemical compound composed of ions, in a solid, liquid or dissolved state is called a electrolyte. Such a system exhibits electrolytic conductivity and interionic interaction. The ions typically have charges of magnitude, equal to their valency z multiplied by the electronic charge e. A polymer having sufficient ionic substituents along the chain to be water-soluble is called a polyelectrolyte. Thus, polyelectrolytes are polymers with ionizable groups that can dissociate in solution, leaving ions of one sign bound to the chain
2 2.5 POLYELECTROLYTE 69 Figure 2.27: Electrolyte coil and anions in solution. They are soluble in water, in contrast to most neutral hydrocarbon polymers, which are only soluble in organic solvents. This a drastic effect on the polymer conformation. Whereas for the uncharged polymer we found a random coil we now will get more extended conformations as shown schematically in figure Also the the solution of the electrolyte and solvent molecules will be more viscous. A biological ion such as DNA, which is negatively charged due to phosphate groups will be surrounded by the positive anions K +, Na +, Ca +2 and Mg +2. What we are interested in are natural polyelectrolytes and chemically modified polyelectrolytes. Examples of natural polyelectrolytes are Nucleic acids (anionic) poly(l-lysine) (cationic) poly((l-glutamic acid) (anionic) The starting point of our development of a theory is the Debye-Hückel potential [43] (see figure 2.28). It form is given by
3 70 MACROMOLECULES The Deby-H ckel function compared to 1/r U_DH(r) ,5 1 1,5 2 2,5 3 r Deby-H ckel function 1/r Figure 2.28: The Debye-Hückel potential U DH (r) = k B T l B r exp( κr). (2.239) It is a short-range potential, where κ, the screening length, controls how fast the potential drops. κ is connected to the Bjerrum length by κ 2 = 4πl B I, (2.240) with I being the ionic strength of the solution (I = ionic species Z2 i c i, Z i being the valence and c i the concentration of species i). κ can vary considerably over at least two orders of magnitude The Poisson-Boltzmann equation We consider a collection of fixed charges surrounded by a gas of ions. The description of the equilibrium distribution of the electrolytic ions starts off with an approximate description. This is because for the full problem we would have to study the thermal motion of a strongly interacting many-body system with
4 2.5 POLYELECTROLYTE 71 long ranged Coulomb force. Hence the usual approach is to take the Poisson- Boltzmann equation as a starting point. Let c i (x) denote the number density of the ions. The charge is z i e. The density is given by the Boltzmann equation c i (x) = C exp( βφ(x)z i e) (2.241) where the internal energy is given by the product of the charge and the potential φ(x). The normalization constant c is to be determined from the condition that the concentration of each species c i eventually is that of the bulk concentration c 0 i (the bulk concentration of the i-th ionic component in the bulk electrolyte). Next the electrostatic potential is given by the Poisson equation d 2 φ(x) dx 2 = ρ(x)/ɛ. (2.242) The density can be related to the concentrations of the ion species by ρ(x) = i z i ec i (x) (2.243) Substituting equation (2.241) into (2.243) we obtain ρ(x) = i z i ec 0 i exp( βφ(x)z i e). (2.244) Finally, substituting the density into the Poisson equation, we arrive at the Boltzmann- Poisson equation. d 2 φ(x) dx 2 = 1 z i ec 0 i exp( βez i φ(x)) (2.245) ɛ i This equation can readily solved if we linearize by expanding the exponential term d 2 φ(x) dx 2 The solution to this equation is = 1 z i ec 0 i ɛ (1 βez iφ(x)). (2.246) i with d 2 φ(x) = l 2 dx 2 Dφ(x), (2.247)
5 72 MACROMOLECULES e2 β l D = zi 2 ɛ c0 i (2.248) being the Debye length l D. The Debye length is a measure for the region of a potential around a point charge, which causes screening of interactions. i Annealed polyelectrolytes The free energy of an annealed, fully stretched polyelectrolyte chain in a salty solution can be written as βf = + N/2 N/2 N/2 N/2 ds [f(s)(log f(s) 1) µf(s) (2.249) } ds f(s)f(s 1 b/λ ) D s s b e s s. (2.250) Here f(s) is the local charge distribution along the chain and λ D is the Debye screening length. The first term of eq is the entropy of a unidimensional ideal gas. The second term fixes the charge on the chain by the chemical potential µ and the third term represents the electrostatic interaction between the charges on the chain. Justification for eq can be obtained only if the average charge density < f > /b fulfills certain conditions. First we note, that < f > has to be small in order to make non-linear effects unimportant < f > l B b < f > u < 1 (2.251) Here, u = l B /b is a dimensionless parameter. Second < f > has to be large to make sure that a sufficiently large number of charges interact simultaneously < f > λ D b > 1, (2.252) i.e., the Manning parameter for the condensation of counterions on a partially charged rigid rod need to large enough. Minimizing eq the equilibrium charge density distribution on an annealed, fully stretched polyeletrolyte chain, up to first order in < f > l B /b is given by
6 2.5 POLYELECTROLYTE 73 Here f(s)/ < f > = 1+ < f > l B b + E 1 [( N 2 s ) b λ D E 1 (x) = x { E 1 [( N 2 + s ) b dt e t t ] 2 λ } D Nb λ D ] (2.253). (2.254) (2.255) Hence, we find a charge accumulation at the ends of the rod within a region of the order of the screening length. To generalize this to weakly charged flexible chains we use the most probable chain conformation r 0 (s) and the entropy of the freely jointed chain model βf [r 0 (s)] = N/2 N/2 ds [f(s)(log f(s) 1) (2.256) µf(s) + 3 ( ) 2 dr0 (s) (2.257) 2b 2 ds } N/2 + ds f(s)f(s ) e r 0(s) r 0 (s ) b/λ D. (2.258) r 0 (s) r 0 (s ) b N/2 This involves as a new length scale, the electrostatic blob size One blob thus contains ξ = b (uf) 1/3. (2.259) g = (ξ/b) 2 (2.260) monomers a chain consists of n b = N/g = N(uf 2 ) 2/3 (2.261) such electrostatic blobs. For a chain of finite length the blob size varies having the smallest (denoted by ξ 0 ) in the center and reaches ξ at the ends.
7 74 MACROMOLECULES With this, the result for the minimization turns out to be as before except for a rescaling of the Bjerrum and the Debye length l B = 3ξ 0 b l B (2.262) λ D = 3ξ 0 b λ D. (2.263) 2.6 Proteins Proteins (see figure 2.31 for an example ) are the machines and building blocks of living cells. They are polymers of the 20 naturally occuring amino acids listed in table 2.3. Recall from figure 2.2 that all amino acids have a COO and a NHHH part or a COOH carboxyl and NHH amino part. In addition, there is a side chain usually labeled R. The configuration of the side chain is called rotamer. This is due to the fact that the tetrahedral geometry stays the same and the main degree of freedom is rotation about the carbon bonds. In figure 2.29 is shown the amino acid Analine and its geometry. To form a protein, amino acids are bonded together in sequence and fold into a protein. Each protein has a unique three-dimensional structure. It was shown [46] that a protein in its natural environment folds into, i.e. vibrates around, a unique three dimensional structure, the native conformation, independent of the starting conformation. There are four levels of architecture in proteins Primary structure: The sequence of peptide-bonded amino acids (as in the example: RSDAEPHYLPQLRKDILEVICKYVQIDPEMVTVQLEQKDG- DISILELNVTLPEAEELK). This is determined by protein synthesis. Secondary structure: The regular, recurring arrangement in space of adjacent amino acid residues in a polypeptide chain. Two main types of secondary structures have been found in proteins, namely the α-helices and β-sheets. The α-helix-complex has already been studied in a previous section. In a β-sheet, two or more polypeptide chains run alongside of each other and are linked in a regular manner by hydrogen bonds between the
8 2.6 PROTEINS 75 Table 2.3: List of the 20 amino acids. The single letter code is used when comparing and aligning sequences of proteins amino acids 3-letter code single letter code Alanine Ala A Cysteine Cys C Aspartic AciD Asp D Glutamic Acid Glu E Phenylalanine Phe F Glycine Gly G Histidine His H Isoleucine Ile I Lysine Lys K Leucine Leu L Methionine Met M AsparagiNe Asn N Proline Pro P Glutamine Gln Q ARginine Arg R Serine Ser S Threonine Thr T Valine Val V Tryptophan Trp W TYrosine Tyr Y
9 76 MACROMOLECULES Figure 2.29: The amino acid Alanine. Note that the bond directions for carbon are the same as from the centroid of a tetrahedron to the vertices.
10 2.6 PROTEINS 77 Figure 2.30: β-sheet. The protein thioredoxin contains a five-stranded beta sheet comprised of three parallel strands and three antiparallel strands. The entire protein is shown as a cartoon with the beta strands (three parallel strands and three antiparallel strands) colored red and alpha helices colored yellow. main chain C=O and N-H groups. Hence, all hydrogen bonds in a β-sheet are between different segments of polypeptide. An example of one strand of a β-sheet is shown in figure A third type of secondary structure are loops. A loop is a section of the sequence that connects the other two kinds of secondary structures. Tertiary structure: The spatial arrangement among all amino acids in a polypeptide. The twisted shape is slightly flexible, and the chain folds upon itself. Quaternary structure: The spatial relationship of polypeptides or subunits. Several proteins interact and form complexes. From the point of view of polymer physics the protein is simply a polymer consisting of a long chain of amino acid residues, i.e. a polypeptides. An important protein which exists in both monomeric or globular (G-actin) and polymeric or filamentary (F-actin) forms is actin. The filaments can form a network of entangled and crosslinked filaments and is the basis for the cytoskeletal network.
11 78 MACROMOLECULES Figure 2.31: MinE protein showing α-helices and β-sheets Protein folding The long-standing question is: how do proteins fold? A protein folds due to the angles φ and ψ between the carbon atom of a residue and the neighboring atoms, i.e. N and CO, in the peptide bond -N-C-(CO)-. These angles can assume only a few values independently of each other. Denaturants such as urea added to the system caused proteins that are folded in the native conformation to loose tertiary structure and revert to a random coiled state. After removal of the denaturants, the protein folds back into the native conformation. The protein folding problem entails the mathematical prediction of (tertiary, 3- dimensional) protein structure given the (primary, linear) structure defined by the sequence of amino acids of the protein. With some exceptions, proteins fold spontaneously. What we want to have is a theoretical model that accurately predicts the folding and properties of the fold. The problem lies in the fact that a variety of globally different structures have very low energies, but within a few k B T of each other. Hence, we would need a very good energy function for possible predictions and the ensuing dynamics are glassy as we have seen before. What we would like to predict is for example the number of observable thermodynamic states
12 2.6 PROTEINS 79 the rate of folding the effect of specific mutations on the folding rate Folding is an interesting problem because it involves mathematical modeling and numerical analysis. It is a extremely challenging task which has not been satisfactorily solved to date. Here we can only give a very brief introduction into some current methods. Basically, we need to distinguish between continuous and discrete models. Within continuous space models, a crucial problem is of course the large number of degrees of freedom. The configuration space is an n dimensional space, where n = 3 number of atoms in molecule. For example, the bacteriorhodopsin has 3576 atoms and hence we have coordinates! This results in the Levinthal s paradox [47]: The 3-D structure of a protein is determined by the dihedral angles. These angles have a few preferred values that correspond to the local minima of torsion energy around each rotation bond. We only have to consider about 10 conformations per AA in a polypeptide chain. This means that we have to examine at least as many as 10 N conformations for a protein with N amino acids. Assuming that a protein can sample of the order of structures per second, would take this protein about seconds or years to examine all the possible conformations. This is longer than the age of the universe. Indeed, the problem of finding the minimum energy configuration is NP-complete under a variety of models. Consequently, it is still impossible to determine the minimum energy structure for larger proteins based on the knowledge of only their sequence. Since, for the foreseeable future it remains doubtful, that we find a satisfying solution for the molecular mechanics of the folding pathway, starting from the random coil conformation to the folded pattern that will emerge. The standard approach is to investigate models that are reduced in complexity. These can be discrete protein models on a lattice to reduce the conformational degrees of freedom or on the other end of the spectrum the reduction to paths in a random energy landscape model. We have already touched on the energy landscape models and will here focus on molecular modeling and lattice models.
13 80 MACROMOLECULES Numerical approaches Molecular Modeling To be able to predict the folded structure, we crucially depend on an energy function. The energy function of all the parameters are used to describe the protein structure. The task is then to find values of the parameters which minimize this function. Molecular mechanics describes the energy of a molecule in terms of a simple function which accounts for distortion from ideal bond distances and angles, as well as and for nonbonded van der Waals and Coulombic interactions. Thus, such force field methods ignore the electronic motions to calculate the energy of a system. To model macromolecular systems the CHARMM potential (Chemistry at HARvard Macromolecular Mechanics) [50, 51], AMBER and GROMOS (GROningen MOLecular Simulation System) force fields are often used. They are empirical force field parametrizations that consists in general of six terms: where V ({R}) = c i (l i l 0 ) 2 (2.264) bonds + c α (θ α θ 0 ) 2 (2.265) bond angles + c β (τ β τ 0 ) 2 (2.266) improper torsion angles + tri(ω) (2.267) dihedral angles Q i Q j + (2.268) ɛr ij charged pairs ( ) Ri + R j + c w Φ (2.269) r ij unbond pairs r ij = R i R j. (2.270)
14 2.6 PROTEINS 81 Here ɛ is the dielectric constant and Q i are the partial charges. The term tri refers to a linear combination of trigonometric functions and and multiples of ω. The term Φ refers to a Lennard-Jones potential. The parameters c etc. are usually fitted and derived from first principles. The approach taken by the Molecular Dynamics and the Langevin Dynamics method discussed in the next section is to solve the equations of motion resulting from a force field, such as the one above, numerically. Molecular Dynamics simulations The starting point for the Molecular Dynamics (MD) simulatuion [52, 53, 54, 55, 56, 57, 58] is thus a well-defined force field. Using Hamiltonian, Lagrangian or Newton s equations of motion these are approximated by suitable schemes Ψ such that they can be solved numerically. Let us be more general for the moment and use generalized coordinates p and q with the Hamiltonian instead of the usual coordinates used above H(q, p) = 1 2 pt M 1 p + V (q). (2.271) From this Hamiltonian we get the equations of motion d dt q = q H (2.272) d dt p = p H. (2.273) An important consideration for any numerical integration scheme is that we want to conserve as many quantities during a numerical evaluation as possible that are conserved due to symmetries etc. This leads us to the concept of symplectic methods. Symplectic methods preserve certain abstract invariants of Hamiltonian systems [59, 60, 61] and are stable for linear systems for sufficiently small values of the stepsize. We denote the trajectory that we want to generate by Γ Γ(t) = ( ) q p Then this trajectory obeys the equation of motion. (2.274)
15 82 MACROMOLECULES where I denotes the unit matrix. d Γ = J H(Γ) (2.275) dt ( ) 0 I J =, (2.276) I 0 We compute an observable A along the trajectories and hence average along the states we find along the path < A >= 1 n obs A(Γ ν (t)). (2.277) n obs ν=1 Here n obs is the number observations we took, i.e., how many interations we took in the numerical integration of the equations of motion. Let ρ 0 (Γ) denote the probability density at time t = 0: Γ(0) = ρ 0 (Γ) and let ρ(γ, t) denote the probability density for Γ(t). Then we have the Liouville theorem for the trajectories. ρ(γ, 0) = ρ 0 (2.278) ρ t + (ρj H) = 0 (2.279) This states that the y flow in phase space is that of an incompressible fluid. If ρ t = 0, then and with this ρ(γ) = H J ρ = 0 (2.280) e H(Γ)/k BT e H(Γ)/k BT dγ. (2.281) Now let Ψ be a numerical integrator, i.e. motion a discretization of the equations of then the phase space volume needs to be conserved Γ n+1 = Ψ(Γ n ), (2.282)
16 2.6 PROTEINS 83 The integrator is sympletic, if det Γ Ψ(Γ) = 1. (2.283) ( Γ ψ(γ) T J( Γ ψ(γ) = J (2.284) i.e. phase space volume and the energy is conserved. If, and only if, Ψ is symplectic, there exists a shadow Hamiltonian H t (Γ), such that for times n t e c/ t. H t (Γ n ) H t (Γ 0 ) = O(e c/ t ) (2.285) The most straightforward discretization of the equations of motion that involve differentials comes from the Taylor expansion. The idea is to base the algorithm on a discrete version of the differential operator. With suitable assumptions we can expand the variable r in a Taylor series r(t + ) = r(t) + n 1 i=1 where R n gives the error involved in the approximation. Using the forward t + and the backward difference t i i! r(i) (t) + R n. (2.286) r(t + ) = r(t) + v(t) + F (t) 2m 2 + d3 r 3 + R dt 3 4 3! (2.287) r(t ) = r(t) v(t) + F (t) 2m 2 d3 r 3 + R dt 3 4 3! (2.288) (2.289) If we add the two equations, we obtain r(t + ) = 2r(t) r(t ) + F (t) 2m 2 + R 4 (2.290) If we substract the two equations we obtain r(t + ) + r(t ) = 2v(t) + R 3 (2.291)
17 84 MACROMOLECULES Figure 2.32: Free energy landscape and hence an estimator for the velocity v(t) = r(t + ) + r(t ) 2 + R 2. (2.292) The estimator for the position and the velocity together comprise what is known as the Verlet algorithm [58]. The Verlet algorithm is a second-order method that is indeed symplectic. So, much hinges on the simulation step-size, since this determines the time-scales, that we can cover. As we have seen above the choice of stepsize is dominated by stability demands and not by accuracy demands. What we also need to stress is that we actually need to simulate rare events, namely the folding which becomes more and more glassy as time progresses. Langevin Dynamics simulations Another approach to understand how the primary structure gives rise to the tertiary structure is to model the protein chemically as realistic as possible and take the interaction of atoms with the environment into account only through their stochastic
18 2.6 PROTEINS 85 influence. The water molecules and all other possible solvent molecules are not explicitly taken into account. Thus we start off with a Langevin equation [48] Mẍ + ηẋ + V = ξ(t), (2.293) where M is the mass matrix, η the damping matrix and ξ a normalized white noise resulting from a Wiener process. Because of the independence of the coordinates, the Langevin equation (2.293) only depends on the covariance matrix of the noise ξ(t) = DẆ (t) (here D is the diffusion matrix and W a Wiener process) DD T =< ξξ T dt >. (2.294) The fluctuation-dissipation theorem relates the diffusion matrix to the damping matrix and the temperature DD T = 2k B T η. (2.295) Because of the lack of knowledge of the detailed damping a further reduction in complexity is usual taken by setting the damping matrix proportional to the mass matrix with a damping constant γ. We then get η = γm (2.296) D = (2k B T γm) 1/2. (2.297) Let E be the energy barrier which the molecule has to take to get to a new state. Then the activation energy can be related to the mean frequency of transition f by an Arrhenius law, i.e., the rate increases exponentially with the absolute temperature where h is the Plank constant. f = k BT h ( exp E ) k B T (2.298) We shall now assume that we are at low temperatures, where the motions of the involved atoms are small. The protein is further assumed in a local minimum x min
19 86 MACROMOLECULES and we are interested in the high frequency modes. The highest frequencies are of the order of sec (roughly the C-H bond vibrations). For simplicity we shall also assume that the eigenmodes are non-degenerate. Then we can expand the potential up to second order V (x) V (x min ) (x x min) T V (x xmin ) (2.299) where V = 2 V (x min ). Ignoring in the limit of low temperature and high frequency the damping and the random force term we obtain with the general solution Mẍ + V (x x min ) = 0 (2.300) x = x min + l u l exp(iω l t) (2.301) Here ω l are the frequencies and u l are the normal modes. Monte Carlo Method The dynamical Monte-Carlo-Method, to generate a sample from an ensemble can be specified as follows: Determine the matrix of transition probabilities from state to the other P = {p xy } = {p(x y)}, (2.302) that satisfies the following transition probabilities (i) x, y S n 0 : p (0) xy > 0 (ii) y S : x π xp xy = π y. A sufficient, but by no means necessary condition to satisfy (ii) is the condition of detailed balance for die for the distribution π, (ii ) x, y S : π x p xy = π y p yx.
Proteins: Characteristics and Properties of Amino Acids
 SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell
 Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
Translation. A ribosome, mrna, and trna.
 Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Dominant Paths in Protein Folding
 Dominant Paths in Protein Folding Henri Orland SPhT, CEA-Saclay France work in collaboration with P. Faccioli, F. Pederiva, M. Sega University of Trento Henri Orland Annecy meeting 2006 Outline Basic notions
Dominant Paths in Protein Folding Henri Orland SPhT, CEA-Saclay France work in collaboration with P. Faccioli, F. Pederiva, M. Sega University of Trento Henri Orland Annecy meeting 2006 Outline Basic notions
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
Section Week 3. Junaid Malek, M.D.
 Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Section Week 3 Junaid Malek, M.D. Biological Polymers DA 4 monomers (building blocks), limited structure (double-helix) RA 4 monomers, greater flexibility, multiple structures Proteins 20 Amino Acids,
Protein structure. Protein structure. Amino acid residue. Cell communication channel. Bioinformatics Methods
 Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Read more about Pauling and more scientists at: Profiles in Science, The National Library of Medicine, profiles.nlm.nih.gov
 2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
Amino Acids and Peptides
 Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
1. Amino Acids and Peptides Structures and Properties
 1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
Viewing and Analyzing Proteins, Ligands and their Complexes 2
 2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
From Amino Acids to Proteins - in 4 Easy Steps
 From Amino Acids to Proteins - in 4 Easy Steps Although protein structure appears to be overwhelmingly complex, you can provide your students with a basic understanding of how proteins fold by focusing
From Amino Acids to Proteins - in 4 Easy Steps Although protein structure appears to be overwhelmingly complex, you can provide your students with a basic understanding of how proteins fold by focusing
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013
 Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
Chemical Properties of Amino Acids
 hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
Major Types of Association of Proteins with Cell Membranes. From Alberts et al
 Major Types of Association of Proteins with Cell Membranes From Alberts et al Proteins Are Polymers of Amino Acids Peptide Bond Formation Amino Acid central carbon atom to which are attached amino group
Major Types of Association of Proteins with Cell Membranes From Alberts et al Proteins Are Polymers of Amino Acids Peptide Bond Formation Amino Acid central carbon atom to which are attached amino group
Exam I Answer Key: Summer 2006, Semester C
 1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
1. Which of the following tripeptides would migrate most rapidly towards the negative electrode if electrophoresis is carried out at ph 3.0? a. gly-gly-gly b. glu-glu-asp c. lys-glu-lys d. val-asn-lys
PROTEIN SECONDARY STRUCTURE PREDICTION: AN APPLICATION OF CHOU-FASMAN ALGORITHM IN A HYPOTHETICAL PROTEIN OF SARS VIRUS
 Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur. Lecture - 06 Protein Structure IV
 Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture - 06 Protein Structure IV We complete our discussion on Protein Structures today. And just to recap
Biochemistry Prof. S. DasGupta Department of Chemistry Indian Institute of Technology Kharagpur Lecture - 06 Protein Structure IV We complete our discussion on Protein Structures today. And just to recap
Exam III. Please read through each question carefully, and make sure you provide all of the requested information.
 09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
Molecular Modelling. part of Bioinformatik von RNA- und Proteinstrukturen. Sonja Prohaska. Leipzig, SS Computational EvoDevo University Leipzig
 part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 Protein Structure levels or organization Primary structure: sequence of amino acids (from
part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 Protein Structure levels or organization Primary structure: sequence of amino acids (from
UNIT TWELVE. a, I _,o "' I I I. I I.P. l'o. H-c-c. I ~o I ~ I / H HI oh H...- I II I II 'oh. HO\HO~ I "-oh
 UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
BCH 4053 Exam I Review Spring 2017
 BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
Packing of Secondary Structures
 7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
7.88 Lecture Notes - 4 7.24/7.88J/5.48J The Protein Folding and Human Disease Professor Gossard Retrieving, Viewing Protein Structures from the Protein Data Base Helix helix packing Packing of Secondary
Physiochemical Properties of Residues
 Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
Physiochemical Properties of Residues Various Sources C N Cα R Slide 1 Conformational Propensities Conformational Propensity is the frequency in which a residue adopts a given conformation (in a polypeptide)
2 Structure. 2.1 Coulomb interactions
 2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
2 Structure 2.1 Coulomb interactions While the information needed for reproduction of living systems is chiefly maintained in the sequence of macromolecules, any practical use of this information must
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
Biochemistry Quiz Review 1I. 1. Of the 20 standard amino acids, only is not optically active. The reason is that its side chain.
 Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Biochemistry Quiz Review 1I A general note: Short answer questions are just that, short. Writing a paragraph filled with every term you can remember from class won t improve your answer just answer clearly,
Dental Biochemistry Exam The total number of unique tripeptides that can be produced using all of the common 20 amino acids is
 Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Basic Principles of Protein Structures
 Basic Principles of Protein Structures Proteins Proteins: The Molecule of Life Proteins: Building Blocks Proteins: Secondary Structures Proteins: Tertiary and Quartenary Structure Proteins: Geometry Proteins
Basic Principles of Protein Structures Proteins Proteins: The Molecule of Life Proteins: Building Blocks Proteins: Secondary Structures Proteins: Tertiary and Quartenary Structure Proteins: Geometry Proteins
The Structure of Enzymes!
 The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes!
 The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
The Structure of Enzymes Levels of Protein Structure 0 order amino acid composition Primary Secondary Motifs Tertiary Domains Quaternary ther sequence repeating structural patterns defined by torsion angles
Problem Set 1
 2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
2006 7.012 Problem Set 1 Due before 5 PM on FRIDAY, September 15, 2006. Turn answers in to the box outside of 68-120. PLEASE WRITE YOUR ANSWERS ON THIS PRINTOUT. 1. For each of the following parts, pick
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Introduction to Comparative Protein Modeling. Chapter 4 Part I
 Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
Introduction to Comparative Protein Modeling Chapter 4 Part I 1 Information on Proteins Each modeling study depends on the quality of the known experimental data. Basis of the model Search in the literature
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Protein Secondary Structure Prediction
 part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
Protein Structure. Role of (bio)informatics in drug discovery. Bioinformatics
 Bioinformatics Protein Structure Principles & Architecture Marjolein Thunnissen Dep. of Biochemistry & Structural Biology Lund University September 2011 Homology, pattern and 3D structure searches need
Bioinformatics Protein Structure Principles & Architecture Marjolein Thunnissen Dep. of Biochemistry & Structural Biology Lund University September 2011 Homology, pattern and 3D structure searches need
Collision Cross Section: Ideal elastic hard sphere collision:
 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Enzyme Catalysis & Biotechnology
 L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
Peptides And Proteins
 Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
Kevin Burgess, May 3, 2017 1 Peptides And Proteins from chapter(s) in the recommended text A. Introduction B. omenclature And Conventions by amide bonds. on the left, right. 2 -terminal C-terminal triglycine
THE UNIVERSITY OF MANITOBA. PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR
 THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
THE UNIVERSITY OF MANITOBA 1 November 1, 2016 Mid-Term EXAMINATION PAPER NO: _1_ LOCATION: 173 Robert Schultz Theatre PAGE NO: 1 of 5 DEPARTMENT & COURSE NO: CHEM / MBIO 2770 TIME: 1 HOUR EXAMINATION:
Protein Structure Basics
 Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
Protein Structure Basics Presented by Alison Fraser, Christine Lee, Pradhuman Jhala, Corban Rivera Importance of Proteins Muscle structure depends on protein-protein interactions Transport across membranes
NH 2. Biochemistry I, Fall Term Sept 9, Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter
 Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Biochemistry I, Fall Term Sept 9, 2005 Lecture 5: Amino Acids & Peptides Assigned reading in Campbell: Chapter 3.1-3.4. Key Terms: ptical Activity, Chirality Peptide bond Condensation reaction ydrolysis
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
Protein Struktur (optional, flexible)
 Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
A Minimal Model for the Hydrophobic and Hydrogen Bonding Effects on Secondary and Tertiary Structure Formation in Proteins
 A Minimal Model for the Hydrophobic and Hydrogen Bonding Effects on Secondary and Tertiary Structure Formation in Proteins by Kyle Denison A thesis presented to the University of Waterloo in fulfillment
A Minimal Model for the Hydrophobic and Hydrogen Bonding Effects on Secondary and Tertiary Structure Formation in Proteins by Kyle Denison A thesis presented to the University of Waterloo in fulfillment
CHEMISTRY ATAR COURSE DATA BOOKLET
 CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
Dental Biochemistry EXAM I
 Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Model Mélange. Physical Models of Peptides and Proteins
 Model Mélange Physical Models of Peptides and Proteins In the Model Mélange activity, you will visit four different stations each featuring a variety of different physical models of peptides or proteins.
Model Mélange Physical Models of Peptides and Proteins In the Model Mélange activity, you will visit four different stations each featuring a variety of different physical models of peptides or proteins.
Rotamers in the CHARMM19 Force Field
 Appendix A Rotamers in the CHARMM19 Force Field The people may be made to follow a path of action, but they may not be made to understand it. Confucius (551 BC - 479 BC) ( ) V r 1 (j),r 2 (j),r 3 (j),...,r
Appendix A Rotamers in the CHARMM19 Force Field The people may be made to follow a path of action, but they may not be made to understand it. Confucius (551 BC - 479 BC) ( ) V r 1 (j),r 2 (j),r 3 (j),...,r
Lecture 10: Brownian Motion, Random Walk & Diffusion Side Chains of Amino Acids
 Lecture 10: Brownian Motion, Random Walk & Diffusion Side Chains of Amino Acids Lecturer: Prof. Brigita Urbanc (brigita@drexel.edu) PHYS 461 & 561, Fall 2009-2010 1 Stochastic Processes: Brownian Motion
Lecture 10: Brownian Motion, Random Walk & Diffusion Side Chains of Amino Acids Lecturer: Prof. Brigita Urbanc (brigita@drexel.edu) PHYS 461 & 561, Fall 2009-2010 1 Stochastic Processes: Brownian Motion
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability
 Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
Lecture 2 and 3: Review of forces (ctd.) and elementary statistical mechanics. Contributions to protein stability Part I. Review of forces Covalent bonds Non-covalent Interactions: Van der Waals Interactions
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
Lecture'18:'April'2,'2013
 CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
Resonance assignments in proteins. Christina Redfield
 Resonance assignments in proteins Christina Redfield 1. Introduction The assignment of resonances in the complex NMR spectrum of a protein is the first step in any study of protein structure, function
Resonance assignments in proteins Christina Redfield 1. Introduction The assignment of resonances in the complex NMR spectrum of a protein is the first step in any study of protein structure, function
Lecture 26: Polymers: DNA Packing and Protein folding 26.1 Problem Set 4 due today. Reading for Lectures 22 24: PKT Chapter 8 [ ].
![Lecture 26: Polymers: DNA Packing and Protein folding 26.1 Problem Set 4 due today. Reading for Lectures 22 24: PKT Chapter 8 [ ]. Lecture 26: Polymers: DNA Packing and Protein folding 26.1 Problem Set 4 due today. Reading for Lectures 22 24: PKT Chapter 8 [ ].](/thumbs/84/90810734.jpg) Lecture 26: Polymers: DA Packing and Protein folding 26.1 Problem Set 4 due today. eading for Lectures 22 24: PKT hapter 8 DA Packing for Eukaryotes: The packing problem for the larger eukaryotic genomes
Lecture 26: Polymers: DA Packing and Protein folding 26.1 Problem Set 4 due today. eading for Lectures 22 24: PKT hapter 8 DA Packing for Eukaryotes: The packing problem for the larger eukaryotic genomes
Secondary Structure. Bioch/BIMS 503 Lecture 2. Structure and Function of Proteins. Further Reading. Φ, Ψ angles alone determine protein structure
 Bioch/BIMS 503 Lecture 2 Structure and Function of Proteins August 28, 2008 Robert Nakamoto rkn3c@virginia.edu 2-0279 Secondary Structure Φ Ψ angles determine protein structure Φ Ψ angles are restricted
Bioch/BIMS 503 Lecture 2 Structure and Function of Proteins August 28, 2008 Robert Nakamoto rkn3c@virginia.edu 2-0279 Secondary Structure Φ Ψ angles determine protein structure Φ Ψ angles are restricted
Principles of Biochemistry
 Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
titin, has 35,213 amino acid residues (the human version of titin is smaller, with only 34,350 residues in the full length protein).
 Introduction to Protein Structure Proteins are large heteropolymers usually comprised of 50 2500 monomer units, although larger proteins are observed 8. The monomer units of proteins are amino acids. Proteins
Introduction to Protein Structure Proteins are large heteropolymers usually comprised of 50 2500 monomer units, although larger proteins are observed 8. The monomer units of proteins are amino acids. Proteins
Protein Struktur. Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep. wake up at slide 39
 Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
7.012 Problem Set 1. i) What are two main differences between prokaryotic cells and eukaryotic cells?
 ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
ame 7.01 Problem Set 1 Section Question 1 a) What are the four major types of biological molecules discussed in lecture? Give one important function of each type of biological molecule in the cell? b)
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
BIRKBECK COLLEGE (University of London)
 BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
BIRKBECK COLLEGE (University of London) SCHOOL OF BIOLOGICAL SCIENCES M.Sc. EXAMINATION FOR INTERNAL STUDENTS ON: Postgraduate Certificate in Principles of Protein Structure MSc Structural Molecular Biology
The Structure and Functions of Proteins
 Wright State University CORE Scholar Computer Science and Engineering Faculty Publications Computer Science and Engineering 2003 The Structure and Functions of Proteins Dan E. Krane Wright State University
Wright State University CORE Scholar Computer Science and Engineering Faculty Publications Computer Science and Engineering 2003 The Structure and Functions of Proteins Dan E. Krane Wright State University
Potentiometric Titration of an Amino Acid. Introduction
 NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
Dana Alsulaibi. Jaleel G.Sweis. Mamoon Ahram
 15 Dana Alsulaibi Jaleel G.Sweis Mamoon Ahram Revision of last lectures: Proteins have four levels of structures. Primary,secondary, tertiary and quaternary. Primary structure is the order of amino acids
15 Dana Alsulaibi Jaleel G.Sweis Mamoon Ahram Revision of last lectures: Proteins have four levels of structures. Primary,secondary, tertiary and quaternary. Primary structure is the order of amino acids
Lecture 14 - Cells. Astronomy Winter Lecture 14 Cells: The Building Blocks of Life
 Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
NAME. EXAM I I. / 36 September 25, 2000 Biochemistry I II. / 26 BICH421/621 III. / 38 TOTAL /100
 EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
BIS Office Hours
 BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
CHEM J-9 June 2014
 CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
CEM1611 2014-J-9 June 2014 Alanine (ala) and lysine (lys) are two amino acids with the structures given below as Fischer projections. The pk a values of the conjugate acid forms of the different functional
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS. Prokaryotes and Eukaryotes. DNA and RNA
 SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
BIOCHEMISTRY Unit 2 Part 4 ACTIVITY #6 (Chapter 5) PROTEINS
 BIOLOGY BIOCHEMISTRY Unit 2 Part 4 ACTIVITY #6 (Chapter 5) NAME NAME PERIOD PROTEINS GENERAL CHARACTERISTICS AND IMPORTANCES: Polymers of amino acids Each has unique 3-D shape Vary in sequence of amino
BIOLOGY BIOCHEMISTRY Unit 2 Part 4 ACTIVITY #6 (Chapter 5) NAME NAME PERIOD PROTEINS GENERAL CHARACTERISTICS AND IMPORTANCES: Polymers of amino acids Each has unique 3-D shape Vary in sequence of amino
A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of
 Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Biological Macromolecules
 Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
Introduction for Chem 493 Chemistry of Biological Macromolecules Dr. L. Luyt January 2008 Dr. L. Luyt Chem 493-2008 1 Biological macromolecules are the molecules of life allow for organization serve a
CHMI 2227 EL. Biochemistry I. Test January Prof : Eric R. Gauthier, Ph.D.
 CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
CHMI 2227 EL Biochemistry I Test 1 26 January 2007 Prof : Eric R. Gauthier, Ph.D. Guidelines: 1) Duration: 55 min 2) 14 questions, on 7 pages. For 70 marks (5 marks per question). Worth 15 % of the final
CHEM 3653 Exam # 1 (03/07/13)
 1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
Protein Structure. W. M. Grogan, Ph.D. OBJECTIVES
 Protein Structure W. M. Grogan, Ph.D. OBJECTIVES 1. Describe the structure and characteristic properties of typical proteins. 2. List and describe the four levels of structure found in proteins. 3. Relate
Protein Structure W. M. Grogan, Ph.D. OBJECTIVES 1. Describe the structure and characteristic properties of typical proteins. 2. List and describe the four levels of structure found in proteins. 3. Relate
Conformational Analysis
 Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
Conformational Analysis C01 3 C C 3 is the most stable by 0.9 kcal/mole C02 K eq = K 1-1 * K 2 = 0.45-1 * 0.048 = 0.11 C04 The intermediate in the reaction of 2 has an unfavorable syn-pentane interaction,
Protein structure (and biomolecular structure more generally) CS/CME/BioE/Biophys/BMI 279 Sept. 28 and Oct. 3, 2017 Ron Dror
 Protein structure (and biomolecular structure more generally) CS/CME/BioE/Biophys/BMI 279 Sept. 28 and Oct. 3, 2017 Ron Dror Please interrupt if you have questions, and especially if you re confused! Assignment
Protein structure (and biomolecular structure more generally) CS/CME/BioE/Biophys/BMI 279 Sept. 28 and Oct. 3, 2017 Ron Dror Please interrupt if you have questions, and especially if you re confused! Assignment
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation
 Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
Bchem 675 Lecture 9 Electrostatics-Lecture 2 Debye-Hückel: Continued Counter ion condensation Ion:ion interactions What is the free energy of ion:ion interactions ΔG i-i? Consider an ion in a solution
Computational Biology & Computational Medicine
 Computational Biology & Computational Medicine Homayoun Valafar Outline Why proteins? What are proteins? How do we compute them? How do we use computational approaches? Why Proteins? Molecular basis of
Computational Biology & Computational Medicine Homayoun Valafar Outline Why proteins? What are proteins? How do we compute them? How do we use computational approaches? Why Proteins? Molecular basis of
Supporting information to: Time-resolved observation of protein allosteric communication. Sebastian Buchenberg, Florian Sittel and Gerhard Stock 1
 Supporting information to: Time-resolved observation of protein allosteric communication Sebastian Buchenberg, Florian Sittel and Gerhard Stock Biomolecular Dynamics, Institute of Physics, Albert Ludwigs
Supporting information to: Time-resolved observation of protein allosteric communication Sebastian Buchenberg, Florian Sittel and Gerhard Stock Biomolecular Dynamics, Institute of Physics, Albert Ludwigs
Towards Understanding the Origin of Genetic Languages
 Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
Charged amino acids (side-chains)
 Proteins are composed of monomers called amino acids There are 20 different amino acids Amine Group Central ydrocarbon N C C R Group Carboxyl Group ALL amino acids have the exact same structure except
Proteins are composed of monomers called amino acids There are 20 different amino acids Amine Group Central ydrocarbon N C C R Group Carboxyl Group ALL amino acids have the exact same structure except
Programme Last week s quiz results + Summary Fold recognition Break Exercise: Modelling remote homologues
 Programme 8.00-8.20 Last week s quiz results + Summary 8.20-9.00 Fold recognition 9.00-9.15 Break 9.15-11.20 Exercise: Modelling remote homologues 11.20-11.40 Summary & discussion 11.40-12.00 Quiz 1 Feedback
Programme 8.00-8.20 Last week s quiz results + Summary 8.20-9.00 Fold recognition 9.00-9.15 Break 9.15-11.20 Exercise: Modelling remote homologues 11.20-11.40 Summary & discussion 11.40-12.00 Quiz 1 Feedback
Bioengineering 215. An Introduction to Molecular Dynamics for Biomolecules
 Bioengineering 215 An Introduction to Molecular Dynamics for Biomolecules David Parker May 18, 2007 ntroduction A principal tool to study biological molecules is molecular dynamics simulations (MD). MD
Bioengineering 215 An Introduction to Molecular Dynamics for Biomolecules David Parker May 18, 2007 ntroduction A principal tool to study biological molecules is molecular dynamics simulations (MD). MD
INTRODUCTION. Amino acids occurring in nature have the general structure shown below:
 Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015,
 Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Biochemistry,530:,, Introduc5on,to,Structural,Biology, Autumn,Quarter,2015, Course,Informa5on, BIOC%530% GraduateAlevel,discussion,of,the,structure,,func5on,,and,chemistry,of,proteins,and, nucleic,acids,,control,of,enzyma5c,reac5ons.,please,see,the,course,syllabus,and,
Molecular Mechanics. I. Quantum mechanical treatment of molecular systems
 Molecular Mechanics I. Quantum mechanical treatment of molecular systems The first principle approach for describing the properties of molecules, including proteins, involves quantum mechanics. For example,
Molecular Mechanics I. Quantum mechanical treatment of molecular systems The first principle approach for describing the properties of molecules, including proteins, involves quantum mechanics. For example,
Advanced Certificate in Principles in Protein Structure. You will be given a start time with your exam instructions
 BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
BIRKBECK COLLEGE (University of London) Advanced Certificate in Principles in Protein Structure MSc Structural Molecular Biology Date: Thursday, 1st September 2011 Time: 3 hours You will be given a start
CHAPTER - V MECHANISM OF OXIDATION OF AMINO ACIDS BY NBN
 37 CHAPTER - V MECHANISM OF OXIDATION OF AMINO ACIDS BY NBN Before proposing a probable mechanism for the oxidation of amino acids by NBN, the inetic results of the present investigation are summed up
37 CHAPTER - V MECHANISM OF OXIDATION OF AMINO ACIDS BY NBN Before proposing a probable mechanism for the oxidation of amino acids by NBN, the inetic results of the present investigation are summed up
Review of General & Organic Chemistry
 Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
Dihedral Angles. Homayoun Valafar. Department of Computer Science and Engineering, USC 02/03/10 CSCE 769
 Dihedral Angles Homayoun Valafar Department of Computer Science and Engineering, USC The precise definition of a dihedral or torsion angle can be found in spatial geometry Angle between to planes Dihedral
Dihedral Angles Homayoun Valafar Department of Computer Science and Engineering, USC The precise definition of a dihedral or torsion angle can be found in spatial geometry Angle between to planes Dihedral
