Characterization of highly basic molecules in terms of site-specific protonation parameters
|
|
|
- Brian Reed
- 5 years ago
- Views:
Transcription
1 Characterization of highly basic molecules in terms of site-specific protonation parameters Theses of doctoral (PhD) dissertation Gábor Orgován Semmelweis University Doctoral School of Pharmaceutical and Pharmacological Sciences Supervisor: Opponents: Head of examination committee: Members of examination committee: Prof. Dr. Béla Noszál, D.Sc. Dr. Ádám Demeter, Ph.D. Dr. Tamás Tábi, Ph.D. Prof. Dr. György Bagdy, D.Sc. Prof. Dr. Pál Perjési, C.Sc. Prof. Dr. Huba Kalász, D.Sc. Semmelweis University Department of Pharmaceutical Chemistry Budapest 2011
2 1. Introduction Most bio- and drug molecules contain two or more basic sites. The fate of ionizable drug molecules in the body (pharmacokinetic parameters) depends on external parameters (ph, temperature, relative permittivity, extramolecular environment), and also internal ones, such as molecular structure, lipophilicity, charge and charge distribution. Latters are influenced and can be characterized by various acid-base parameters, e.g. protonation constant (logk). Knowledge of these values are of high importance in every stage of drug development. In regulatory processes the protonation constants need to be determined with high precision. Binding of drug molecules to the target depends not only on their gross charge. Rather, it is highly affected by the protonation state of ionoizable groups. Site-specific basicities can be quantified by the microscopic protonation constants, which provide information on their submolecular basicity. Microconstants can be determined either by a ph-dependent, groupspecific spectroscopical method or by transferring basicities of structurally related analogues. A large number of biologically and pharmaceutically important compounds contain strongly basic functional groups (e,g. guanidino, amidine). The protonation of these groups takes place in strongly basic media, where the accurate ph-determination is a difficult task. In fact, most literature protonation constants on very basic molecules are highly erroneous, due to the distorted ph determination near the upper end of the ph-scale. We have therefore elaborated an NMR-based ph determination method that provide correct acidity data in highly basic solutions. This method allowed the determination of logk values for molecules that could not be accurately characterized before. 1
3 2. Objectives The aim of this work was the determination of macroscopic and microscopic protonation constants of molecules of high biological/pharmaceutical importance and strong basicity. Our studies have unfortunately showed that most literature logk values were incorrect, mainly due to the lack of an accurate ph-determination method. To quantify these equilibria a method for the accurate determination of extremely high ph values was needed. Our aim was to develop a 1 H NMR method, in which ph can be accurately calculated from chemical shifts of indicator molecules throughout the ph-scale (0 14). A subsequent aim was to accurately determine the microscopic protonation constants of the most basic natural amino acid, arginine, by 1 H NMR-pH titrations. Calculation of the microconstants could be achieved by including auxiliary compounds and deductive methods. The structure of the molecules are shown on Figure 1. We also investigated the protonation processes, in particular, the groupspecific basicities of the guanidino groups, of streptomycin, the classical aminoglycoside antibiotic drug (Figure 1). Introducing streptidine (Figure 1), the only available model compound, we were able to quantify the protonation processes of the major streptomycin microspecies. Furthermore, we have also aimed at developing a 15 N NMR method for the determination of group-specific basicities of individual amino groups. This method enabled us to determine the microconstants of linear biological polyamines and their nor-derivatives (Figure 1), a family of compounds that play a crucial rule in the regulation of the cell cycle. 2
4 Figure 1. Structure of the studied molecules 3
5 3. Methods All the measurements were carried out at 25 C. The ionic strength was adjusted to a constant value (1.00 M or 0.15 M) by an inert salt, KCl. The concentration of the molecules were: mm indicator molecules (dichloroacetic acid, acetone oxime, sarcosine, chloroacetic acid, formic acid, acetic acid, imidazole, TRIS, tert-butylamine, cytosine, methylguanidine, Figure 1); 2 mm biguanides (metformin, phenformin); 5 mm arginine and model compounds (arginine, citrulline, arginine amide, citrulline methyl ester), 5 mm streptidine, 20 mm streptomycin, 50 mm polyamines (spermidine, spermine, norspermidine, norspermine). NMR-pH titrations. Titrations were carried out on a Varian Unity 600 MHz spectrometer. The solution contained 5 % (v/v) D 2 O. Water resonance signal was suppressed by double spin echo or presaturation pulses. ph of the samples was measured by a Metrohm combined glass electrode and/or calculated from chemical shifts of indicator molecules. 1H NMR spectra were recorded in the titrations of the indicator molecules, arginine and model compounds and the biguanides. The chemical shifts of the overlapping proton signals of streptidine and streptomycin were determined by 13 C HSQC titrations. The hydrogen chemical shifts of polyamines were measured by 1 H and if needed, 13 C HSQC spectra, while 15 N HMBC measurements were carried out for the determination of nitrogen chemical shifts. Titration curves were evaluated by the OriginPro 8.0 software. Potentiometric titrations. ph-metric titrations were performed using a Metrohm combined glass electrode attached to a Metrohm 716 DMS Titrino autoburette, at constant ionic strength (1.00 M). For the determination of macroconstants acidified solution of 50 mm polyamine was titrated with KOH solution. The simultaneous evaluation of potentiometric and NMR-pH titrations were done by OriginPro 8.0 software. 4
6 4. Results, new scientific statements 1. A 1H NMR-based ph-determination method was developed. By this method the ph can be precisely and accurately determined in the entire (0 14) ph-range. The indicator parameters of the indicator molecules were determined at the two most often applied ionic strengths (0.15 M and 1.00 M). The results showed that ph can be determined by five indicator molecules (dichloroacetic acid, acetone oxime, sarcosine, acetic acid and imidazole) in the whole ph-range, at 1.00 M ionic strength. The useful ph-range was calculated by keeping the maximal error or ph-determination at 0.03 log units (Table 1). Table 1. The most important indicator parameters and the useful phrange of the selected indicator molecules. logk δ Ind δ HInd ph-range Dichloroacetic acid 1.04 (2) (1) (2) < 2.0 Acetone oxime 1.73 (1) (1) (1) Sarcosine (CH 2 ) (1) (1) (1) Sarcosine (CH 3 ) (1) (1) Acetic acid 4.57 (1) (1) (1) Imidazole (H2) (1) (1) (1) Imidazole (H4) (1) (1) Sarcosine (CH 2 ) (1) (1) (1) Sarcosine (CH 3 ) (1) (1) Acetone oxime (1) (1) (1) Acetone oxime (1) (1) Imidazole (H2) (2) (3) (1) > 13 This series of indicator molecules makes possible the accurate measurement of extremely high ph values and the subsequent determination of protonation constants of strongly basic molecules. 5
7 2. The protonation constants of two strongly basic biguanide-type oral antidiabetics, metformin and phenformin were determined. We have shown, that the logk 1 values found in the literature are significantly lower than our protonation constant. Table 2. Protonation constants of biguanides logk 1 logk 2 Metformin ± ± 0.02 Phenformin ± ± The microconstants of arginine, the most basic natural amino acid were determined by combining the accurate NMR-pH titrations and the deductive method. The molecule contains three basic sites: a guanidino (G), a primary amino (A) and a carboxylate (C) moiety (Figure 2). Inherent basicities of these groups are well-known to be different, the microconstants can therefore be calculated by deductive method, since the abundance of the minor species is lower by several orders of magnitude. We chose two isoelectronic model compounds, which contain a reduced number of ionizable groups: citrulline and arginine amide. The calculations were validated with an independent experiment: the adequate microconstant of arginine was compared to the macroconstant of citrulline methyl ester, a close derivative. The constants are in good agreement. The literature value of logk 1 falls as much as one log unit below than our constant, thus the isoelectric point is higher (11,41). The isoelectric point of citrulline is 5,5 log units lower (5,91), which is so far the largest difference between two isoelectronic compounds. The results showed that the major species predominate over the minor ones by 5 and 10 orders of magnitude. 6
8 Figure 2. Microscopic protonation scheme and constants of arginine 4. The protonation constants of streptomycin, the first-discoverd aminoglycoside-type antibiotic drug and its aglycone, streptidine were determined. The molecules contain three and two ionizable groups, respectively (two guanidino and a secondary amino; and two guanidino). The pair interactivity parameter between the two guanidino groups was determined for the first time. The value is significantly lower than the interactivity parameter between the amino groups of 1,3- diaminopropane-type compounds. This phenomenon can be interpreted as the charge on the protonated guanidino group is located near the remote nitrogen to a large extent, Due to the extensive delocalization, however, the charge cannot be assigned to a particular nitrogen atom. The site specific basicities of the streptomycin guanidino groups were calculated by transferring the interactivity parameter of streptidine. The guanidino group in position 1 is twice as basic as the sterically hindered G3 group (Figure 3). Streptomycin is so strong base that 25 % of the molecules are still protonated even in 1 M NaOH solution. 7
9 Figure 3. The major protonation pathways and related constants of streptomycin. 5. The macro- and microconstants of two linear triamines (spermidine and norspermidine, Figure 4) and two tetraamines (spermine and norspermine Figure 5). The macroconstants were determined by the simultaneous evaluation of potentiometric and 1 H NMR-pH titrations. In order to calculate the microconstants 15 N NMR-pH titrations were carried out. Literature values have been reported on the basis of 13 C NMR-pH titrations, but the reliability of these results is questionable, since the basic sites are very close, the chemical shifts of the carbons adjacent to the amino groups are therefore not at all selective indicators of the site-specific protonation. The microconstants of the minor species, however, cannot be determined accurately from the 15 N NMR-pH titration curves, as their contribution to the analytical signal is negligible. We have therefore used the deductive method: we transferred the interactivity parameter of a model compound with reduced number of ionizable groups. All the microconstants of the tetraamines cannot be calculated from the experimental data, by principle, two additional pieces of information are needed. We have, therefore, transferred the adequate pair interactivity parameters of spermidine and norspermidine, thus the microconstants could be calculated. 8
10 Figure 4. Protonation scheme and microconstants of spermidine and norspermidine Figure 5: Protonation scheme and microconstants of spermine and norspermine 9
11 Among the protonation isomers those are of higher abundance, where the positive charges are more separated. In case of diprotonated isomers those are AA and AB species, while for the triprotonated ones, the ABA species. 6. The standard error of the microconstants was calculated using the quadratic error propagation rule of Gauss. It was shown that one can get reliable microconstants by deductive methods, if the model compound corresponds to the minor species. By selectively monitoring the basic sites, the concentration of the microspecies have to be comparable to get the microconstants with reasonable standard deviation. 5. Summary A new 1 H NMR-based method was developed for the accurate determination of high ph values. This series of indicator molecules made possible the accurate determination of microscopic and macroscopic protonation constants of molecules of high basicity and biological/pharmaceutical importance. The protonation constants of two strongly basic biguanide-type oral antidiabetics, metformin and phenformin were determined. All the microconstants of the most basic natural amino acid, arginine were calculated by combination of NMR-pH and deductive method. Protonation constants of streptomycin, an aminoglycoside type antibiotic drug containing two guanidino groups, were determined. The pair interactivity parameter between two guanidino groups was also calculated. The microconstants of two triamines and two tetraamines were determined by 15 N NMR-pH titrations, by using as few deductive elements as possible. The standard errors of the microconstants were quantified by the error propagation rule of Gauss. 10
12 6. Publications Papers of the thesis work 1. Orgován G., Noszál B. (2011) Electrodeless, accurate ph determination in highly basic media using a new set of 1 H NMR ph indicators. J. Pharm. Biomed. Anal. 54 (5): Orgován G., Noszál B. (2011) The complete microspeciation of arginine and citrulline. J. Pharm. Biomed. Anal. 54 (5): Related papers 1. Orgován G., Tihanyi K., Noszál B. (2009) NMR analysis, protonation equilibria and decomposition kinetics of tolperisone. J. Pharm. Biomed. Anal. 50 (5): Orgován G., Noszál B. (2011) Transporterization: a tool for drug delivery. In: Tihanyi K., Vastag M. (eds) Solubility, Delivery and ADME problems of drugs and drug-candidates. Bentham Science Publishers, accepted for publication. 11
Characterization of various cyclodextrin complexes in terms of complex stability and enantioseparation. Tamás Sohajda
 Characterization of various cyclodextrin complexes in terms of complex stability and enantioseparation PhD Thesis Tamás Sohajda Semmelweis University School of PhD Studies, Pharmaceutical Sciences Supervisor:
Characterization of various cyclodextrin complexes in terms of complex stability and enantioseparation PhD Thesis Tamás Sohajda Semmelweis University School of PhD Studies, Pharmaceutical Sciences Supervisor:
Chapter 8. Acidity, Basicity and pk a
 Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Structure Determination: Nuclear Magnetic Resonance Spectroscopy
 Structure Determination: Nuclear Magnetic Resonance Spectroscopy Why This Chapter? NMR is the most valuable spectroscopic technique used for structure determination More advanced NMR techniques are used
Structure Determination: Nuclear Magnetic Resonance Spectroscopy Why This Chapter? NMR is the most valuable spectroscopic technique used for structure determination More advanced NMR techniques are used
Physico-chemical Profiling of the ACE-inhibitor Lisinopril: Acidbase
 ADMET & DMPK 1(2) (2013) 6-16; doi: 10.5599/admet.1.2.3 Open Access : ISSN : 1848-7718 Original scientific paper http://www.pub.iapchem.org/ojs/index.php/admet/index Physico-chemical Profiling of the ACE-inhibitor
ADMET & DMPK 1(2) (2013) 6-16; doi: 10.5599/admet.1.2.3 Open Access : ISSN : 1848-7718 Original scientific paper http://www.pub.iapchem.org/ojs/index.php/admet/index Physico-chemical Profiling of the ACE-inhibitor
OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry
 OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry Question No. 1 of 10 Question 1. Which statement concerning NMR spectroscopy is incorrect? Question #01 (A) Only nuclei
OAT Organic Chemistry - Problem Drill 19: NMR Spectroscopy and Mass Spectrometry Question No. 1 of 10 Question 1. Which statement concerning NMR spectroscopy is incorrect? Question #01 (A) Only nuclei
A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of
 Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
Questions with Answers- Amino Acids & Peptides A. Two of the common amino acids are analyzed. Amino acid X and amino acid Y both have an isoionic point in the range of 5.0-6.5 (Questions 1-4) 1. Which
4. NMR spectra. Interpreting NMR spectra. Low-resolution NMR spectra. There are two kinds: Low-resolution NMR spectra. High-resolution NMR spectra
 1 Interpreting NMR spectra There are two kinds: Low-resolution NMR spectra High-resolution NMR spectra In both cases the horizontal scale is labelled in terms of chemical shift, δ, and increases from right
1 Interpreting NMR spectra There are two kinds: Low-resolution NMR spectra High-resolution NMR spectra In both cases the horizontal scale is labelled in terms of chemical shift, δ, and increases from right
10/16/17 ACIDS AND BASES, DEFINED WATER IS AMPHOTERIC OUTLINE. 9.1 Properties of Acids and Bases. 9.2 ph. 9.3 Buffers
 ACIDS AND BASES, DEFINED A hydrogen atom contains a proton and an electron, thus a hydrogen ion (H + ) is a proton: Acids: Proton (H + ) transfer between molecules is the basis of acid/base chemistry Ø
ACIDS AND BASES, DEFINED A hydrogen atom contains a proton and an electron, thus a hydrogen ion (H + ) is a proton: Acids: Proton (H + ) transfer between molecules is the basis of acid/base chemistry Ø
2. Acids and Bases. Grossman, CHE Definitions.
 Grossman, CE 230 2. Acids and Bases. 2.1 Definitions. Brønsted acids are proton donors, and Brønsted bases are proton acceptors. Examples of Brønsted acids: Cl, Br, 2 S 4,, + 2, + 4, 3, C 3 C 2, C 2 CC
Grossman, CE 230 2. Acids and Bases. 2.1 Definitions. Brønsted acids are proton donors, and Brønsted bases are proton acceptors. Examples of Brønsted acids: Cl, Br, 2 S 4,, + 2, + 4, 3, C 3 C 2, C 2 CC
Module 13: Chemical Shift and Its Measurement
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy CHE_P12_M13_e-Text TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Shielding and deshielding
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy CHE_P12_M13_e-Text TABLE OF CONTENTS 1. Learning Outcomes 2. Introduction 3. Shielding and deshielding
Chapter 16. Acid-Base Equilibria
 Chapter 16. Acid-Base Equilibria 16.1 Acids and Bases: A Brief Review Acids taste sour and cause certain dyes to change color. Bases taste bitter and feel soapy. Arrhenius concept of acids and bases: An
Chapter 16. Acid-Base Equilibria 16.1 Acids and Bases: A Brief Review Acids taste sour and cause certain dyes to change color. Bases taste bitter and feel soapy. Arrhenius concept of acids and bases: An
UNIT 3 CHEMISTRY. Fundamental Principles in Chemistry
 UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
UNIT 3 CHEMISTRY NOTE: This list has been compiled based on the topics covered in the 2016 Master Class program. Once all of the 2017 Chemistry program materials have been finalised, this summary will
1) Which of the following represents the breaking of a noncovalent interaction? Topic: The Nature of Noncovalent Interactions
 Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Multiple Choice Questions 1) Which of the following represents the breaking of a noncovalent interaction? A) hydrolysis of an ester B) dissolving of salt crystals C) ionization of water D) decomposition
Amino Acids and Peptides
 Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
The resonance frequency of the H b protons is dependent upon the orientation of the H a protons with respect to the external magnetic field:
 Spin-Spin Splitting in Alkanes The signal arising from a proton or set of protons is split into (N+1) lines by the presence of N adjacent nuclei Example 1: Bromoethane The resonance frequency of the H
Spin-Spin Splitting in Alkanes The signal arising from a proton or set of protons is split into (N+1) lines by the presence of N adjacent nuclei Example 1: Bromoethane The resonance frequency of the H
CM Chemical Spectroscopy and Applications. Final Examination Solution Manual AY2013/2014
 NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
NANYANG TECHNOLOGICAL UNIVERSITY DIVISION OF CHEMISTRY AND BIOLOGICAL CHEMISTRY SCHOOL OF PHYSICAL & MATHEMATICAL SCIENCES CM 3011 - Chemical Spectroscopy and Applications Final Examination Solution Manual
Acids and Bases. Moore, T. (2016). Acids and Bases. Lecture presented at PHAR 422 Lecture in UIC College of Pharmacy, Chicago.
 Acids and Bases Moore, T. (2016). Acids and Bases. Lecture presented at PHAR 422 Lecture in UIC College of Pharmacy, Chicago. Drug dissolution can impact buffering capacity of the body Most enzymes require
Acids and Bases Moore, T. (2016). Acids and Bases. Lecture presented at PHAR 422 Lecture in UIC College of Pharmacy, Chicago. Drug dissolution can impact buffering capacity of the body Most enzymes require
Bio-elements. Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components.
 Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Bio-elements Living organisms requires only 27 of the 90 common chemical elements found in the crust of the earth, to be as its essential components. Most of the chemical components of living organisms
Nuclear magnetic resonance spectroscopy II. 13 C NMR. Reading: Pavia Chapter , 6.7, 6.11, 6.13
 Nuclear magnetic resonance spectroscopy II. 13 NMR Reading: Pavia hapter 6.1-6.5, 6.7, 6.11, 6.13 1. General - more/better/additional structural information for larger compounds -problems: a) isotopes
Nuclear magnetic resonance spectroscopy II. 13 NMR Reading: Pavia hapter 6.1-6.5, 6.7, 6.11, 6.13 1. General - more/better/additional structural information for larger compounds -problems: a) isotopes
Practice Final for Chem 104
 Practice Final for Chem 104 These questions cover the material that will be covered on the Final Exam. The distribution of the questions is about the same as the Final Exam. The forms of the questions
Practice Final for Chem 104 These questions cover the material that will be covered on the Final Exam. The distribution of the questions is about the same as the Final Exam. The forms of the questions
Ch 20 Carboxylic Acids and Nitriles
 Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
Ch 20 Carboxylic Acids and Nitriles Carboxylic Acids (RCO 2 H) are compounds with an OH attached to a carbonyl. Nitriles (RC N) are compounds a carbon-nitrogen triple bond. Naming Carboxylic Acids 1. Replace
Chapter 24. Amines. Based on McMurry s Organic Chemistry, 7 th edition
 Chapter 24. Amines Based on McMurry s Organic Chemistry, 7 th edition Amines Organic Nitrogen Compounds Organic derivatives of ammonia, NH 3, Nitrogen atom with a lone pair of electrons, making amines
Chapter 24. Amines Based on McMurry s Organic Chemistry, 7 th edition Amines Organic Nitrogen Compounds Organic derivatives of ammonia, NH 3, Nitrogen atom with a lone pair of electrons, making amines
Chemistry 12 April 1998 Provincial Examination
 Chemistry 12 April 1998 Provincial Examination ANSWER KEY / SCORING GUIDE CURRICULUM: Organizers 1. Reaction Kinetics 2. Dynamic Equilibrium 3. Solubility Equilibria 4. Acids, Bases, and Salts 5. Oxidation
Chemistry 12 April 1998 Provincial Examination ANSWER KEY / SCORING GUIDE CURRICULUM: Organizers 1. Reaction Kinetics 2. Dynamic Equilibrium 3. Solubility Equilibria 4. Acids, Bases, and Salts 5. Oxidation
Certificate Of Analysis
 Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
Certificate f Analysis Compound: -Hydroxy Propranolol Molecular Formula: C H N. (mw base =.) Structure: MW =. base H 8 0 H C H N -Hydroxy Propranolol "FR CHEMICAL USE NLY" Synonyms: (CA index name) -[(-Methylethyl)amino]--(-{-hydroxy}-naphthalenyloxy)--Propanol
AMINES. 3. Secondary When two hydrogen atoms are replaced by two alkyl or aryl groups.
 AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
AMINES Amine may be regarded as derivative of ammonia formed by replacement of one or more hydrogen atoms by corresponding number of alkyl or aryl group CLASSIFICATION 1. Ammonia 2. Primary amine 3. Secondary
Determining Chemical Structures with NMR Spectroscopy the ADEQUATEAD Experiment
 Determining Chemical Structures with NMR Spectroscopy the ADEQUATEAD Experiment Application Note Author Paul A. Keifer, Ph.D. NMR Applications Scientist Agilent Technologies, Inc. Santa Clara, CA USA Abstract
Determining Chemical Structures with NMR Spectroscopy the ADEQUATEAD Experiment Application Note Author Paul A. Keifer, Ph.D. NMR Applications Scientist Agilent Technologies, Inc. Santa Clara, CA USA Abstract
OXFORD H i g h e r E d u c a t i o n Oxford University Press, All rights reserved.
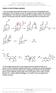 Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Patrick, An Introduction to dicinal hemistry 4e Answers to end-of-chapter questions 1) The mechanism below shows the release of one molecule of formaldehyde from methenamine. The mechanism can then be
Technical Note. Introduction
 Technical Note Characterization of Eleven 2,5-Dimethoxy-N-(2-methoxybenzyl)- phenethylamine (NBOMe) Derivatives and Differentiation from their 3- and 4- Methoxybenzyl Analogues - Part II Patrick A. Hays*,
Technical Note Characterization of Eleven 2,5-Dimethoxy-N-(2-methoxybenzyl)- phenethylamine (NBOMe) Derivatives and Differentiation from their 3- and 4- Methoxybenzyl Analogues - Part II Patrick A. Hays*,
NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule.
 Chapter 13: Nuclear magnetic resonance spectroscopy NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule. 13.2 The nature of
Chapter 13: Nuclear magnetic resonance spectroscopy NMRis the most valuable spectroscopic technique for organic chemists because it maps the carbon-hydrogen framework of a molecule. 13.2 The nature of
CHEM Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W
 CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
CHEM 2423. Chapter 13. Nuclear Magnetic Spectroscopy (Homework) W Short Answer 1. For a nucleus to exhibit the nuclear magnetic resonance phenomenon, it must be magnetic. Magnetic nuclei include: a. all
AP Chemistry Common Ion Effect; 16.6 ionization constants, will. Equilibria with Weak Acids and and the preparation of buffer
 Instructional Unit Acid-Base Equibria 16.1 Acid-Ionizaation Equilibria; Students will perform Students will distinguish Oral response, written 3.1.12C, 16.2 Polyprotic Acids; 16.3 Base- calculations involving
Instructional Unit Acid-Base Equibria 16.1 Acid-Ionizaation Equilibria; Students will perform Students will distinguish Oral response, written 3.1.12C, 16.2 Polyprotic Acids; 16.3 Base- calculations involving
AMINES. 4. From your knowledge of the effects involved, predict or explain experimental results. Important areas include:
 AMINES A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC or common name given the structure, and draw the structure given the name of amines and common nitrogen heterocycles (pyrrole, pyridine, purine, pyrimidine,
AMINES A STUDENT SHOULD BE ABLE TO: 1. Give the IUPAC or common name given the structure, and draw the structure given the name of amines and common nitrogen heterocycles (pyrrole, pyridine, purine, pyrimidine,
NECTA A-Level CHEMISTRY 2 May 2004
 NECTA A-Level CHEMISTRY 2 May 2004 [04/2] SECTION A 1. (a) Draw a sketch of a cell formed by combining Zn 2+ (aq)/zn(s) and Cu 2+ (aq)/cu(s) half-cells. Indicate on the sketch the following: The positive
NECTA A-Level CHEMISTRY 2 May 2004 [04/2] SECTION A 1. (a) Draw a sketch of a cell formed by combining Zn 2+ (aq)/zn(s) and Cu 2+ (aq)/cu(s) half-cells. Indicate on the sketch the following: The positive
New cases of prototropic tautomerism in substituted pyridines
 SIFT DESK Journal of Chemical Engineering And Bioanalytical Chemistry New cases of prototropic tautomerism in substituted pyridines Research Author: Talsi Valentine September 2017 Copy rights: This is
SIFT DESK Journal of Chemical Engineering And Bioanalytical Chemistry New cases of prototropic tautomerism in substituted pyridines Research Author: Talsi Valentine September 2017 Copy rights: This is
2.1 Representing Molecules. 2.1 Representing Molecules. 2.2 Bond-line Structures. Chapter 2 Molecular Representations
 2.1 Representing Molecules Which representations are adequate to represent only isopropanol and not its constitutional isomers? Chapter 2 Molecular Representations Copyright 2014 by John Wiley & Sons,
2.1 Representing Molecules Which representations are adequate to represent only isopropanol and not its constitutional isomers? Chapter 2 Molecular Representations Copyright 2014 by John Wiley & Sons,
Chapter 2 Polar Covalent Bonds; Acids and Bases SAMPLE. Chapter Outline
 Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
Chapter 2 Polar Covalent Bonds; Acids and Bases Chapter utline I. Polar covalent bonds (Sections 2.1 2.3). A. Electronegativity (Section 2.1). 1. Although some bonds are totally ionic and some are totally
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
(Refer Slide Time: 1:03)
 Principles and Applications of NMR spectroscopy Professor Hanudatta S. Atreya NMR Research Centre Indian Institute of Science Bangalore Module 1 Lecture No 05 Welcome back! In the last class we looked
Principles and Applications of NMR spectroscopy Professor Hanudatta S. Atreya NMR Research Centre Indian Institute of Science Bangalore Module 1 Lecture No 05 Welcome back! In the last class we looked
Write your name and date on the cover page Do not open exam until instructed to do so
 Write your name and date on the cover page Do not open exam until instructed to do so Name: Date: Exam III hem. 210 Do not open exam until told to do so. Get out your pencil, eraser, and scientific nongraphing
Write your name and date on the cover page Do not open exam until instructed to do so Name: Date: Exam III hem. 210 Do not open exam until told to do so. Get out your pencil, eraser, and scientific nongraphing
Chapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the H s and C s of a molecules
 hapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the s and s of a molecules Nuclei are positively charged and spin on an axis; they create a tiny magnetic field + + Not all
hapter 13: Nuclear Magnetic Resonance (NMR) Spectroscopy direct observation of the s and s of a molecules Nuclei are positively charged and spin on an axis; they create a tiny magnetic field + + Not all
PAPER No. 12: ORGANIC SPECTROSCOPY. Module 19: NMR Spectroscopy of N, P and F-atoms
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy CHE_P12_M19_e-Text TABLE OF CONTENTS 1. Learning Outcomes 2. 15 N NMR spectroscopy 3. 19 F NMR spectroscopy
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy CHE_P12_M19_e-Text TABLE OF CONTENTS 1. Learning Outcomes 2. 15 N NMR spectroscopy 3. 19 F NMR spectroscopy
Aromatic Hydrocarbons
 Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
Aromatic Hydrocarbons Aromatic hydrocarbons contain six-membered rings of carbon atoms with alternating single and double carbon-carbon bonds. The ring is sometimes shown with a circle in the center instead
ESI for. A rotaxane host system containing integrated triazole C H hydrogen bond donors for anion recognition. Nicholas G. White & Paul D.
 ESI for A rotaxane host system containing integrated triazole C H hydrogen bond donors for anion recognition Nicholas G. White & Paul D. Beer* Contents 1 Details of instrumentation 2 NMR Spectra of new
ESI for A rotaxane host system containing integrated triazole C H hydrogen bond donors for anion recognition Nicholas G. White & Paul D. Beer* Contents 1 Details of instrumentation 2 NMR Spectra of new
Calculate a rate given a species concentration change.
 Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Kinetics Define a rate for a given process. Change in concentration of a reagent with time. A rate is always positive, and is usually referred to with only magnitude (i.e. no sign) Reaction rates can be
Saba Al Fayoumi. Tamer Barakat. Dr. Mamoun Ahram + Dr. Diala Abu-Hassan
 1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
1 Saba Al Fayoumi Tamer Barakat Dr. Mamoun Ahram + Dr. Diala Abu-Hassan What is BIOCHEMISTRY??? Biochemistry = understanding life Chemical reactions are what makes an organism (An organism is simply atoms
Chapter 4: Amino Acids
 Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Chapter 4: Amino Acids All peptides and polypeptides are polymers of alpha-amino acids. lipid polysaccharide enzyme 1940s 1980s. Lipids membrane 1960s. Polysaccharide Are energy metabolites and many of
Solvation in the Cramer-Truhlar Groups
 Solvation in the Cramer-Truhlar Groups Developing solvation models that permit the accurate study of dynamical processes in aqueous and nonaqueous solution February 2007 Adam Chamberlin Natalie Elmasry
Solvation in the Cramer-Truhlar Groups Developing solvation models that permit the accurate study of dynamical processes in aqueous and nonaqueous solution February 2007 Adam Chamberlin Natalie Elmasry
Atmaram K. Mapari *Department of Chemistry, Ramnarain Ruia College, Matunga (E), Mumbai: , Maharashtra, India. Abstract
 International Journal of Applied Chemistry. ISS 0973-79 Volume 3, umber (07) pp. -8 Research India Publications http://www.ripublication.com Stability Constants of ixed Ligand Complexes of Transition etal(ii)
International Journal of Applied Chemistry. ISS 0973-79 Volume 3, umber (07) pp. -8 Research India Publications http://www.ripublication.com Stability Constants of ixed Ligand Complexes of Transition etal(ii)
Tuesday, January 13, NMR Spectroscopy
 NMR Spectroscopy NMR Phenomenon Nuclear Magnetic Resonance µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (μ). If these
NMR Spectroscopy NMR Phenomenon Nuclear Magnetic Resonance µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (μ). If these
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry
 UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
UNIT 4 REVISION CHECKLIST CHEM 4 AS Chemistry Topic 4.1 Kinetics a) Define the terms: rate of a reaction, rate constant, order of reaction and overall order of reaction b) Deduce the orders of reaction
Application of NMR spectroscopy in the quantification of Drug. and its Salt with chemical shift changes
 Chapter 2 22 Application of NMR spectroscopy in the quantification of Drug and its Salt with chemical shift changes 2.1 Introduction NMR spectroscopy is one of the most important and widespread analytical
Chapter 2 22 Application of NMR spectroscopy in the quantification of Drug and its Salt with chemical shift changes 2.1 Introduction NMR spectroscopy is one of the most important and widespread analytical
Spectroscopy. Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD)
 Spectroscopy Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD) A)From a structure: B)From a molecular formula, C c H h N n O o X x, Formula for saturated hydrocarbons: Subtract the
Spectroscopy Empirical Formula: Chemical Formula: Index of Hydrogen Deficiency (IHD) A)From a structure: B)From a molecular formula, C c H h N n O o X x, Formula for saturated hydrocarbons: Subtract the
Chapter 2 Polar Covalent Bonds; Acids and Bases. Chapter Outline
 rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
rganic Chemistry 9th Edition McMurry SLUTINS MANUAL Full clear download at: https://testbankreal.com/download/organic-chemistry-9th-edition-mcmurrysolutions-manual/ rganic Chemistry 9th Edition McMurry
The Use of NMR Spectroscopy
 Spektroskopi Molekul Organik (SMO): Nuclear Magnetic Resonance (NMR) Spectroscopy All is adopted from McMurry s Organic Chemistry The Use of NMR Spectroscopy Used to determine relative location of atoms
Spektroskopi Molekul Organik (SMO): Nuclear Magnetic Resonance (NMR) Spectroscopy All is adopted from McMurry s Organic Chemistry The Use of NMR Spectroscopy Used to determine relative location of atoms
Ionic Equilibria. In the Brönsted Lowry classification, acids and bases may be anions such as HSO 4
 Ionic Equilibria Brönsted Lowry Theory According to the Brönsted Lowry theory, an acid is a substance, charged or uncharged, that is capable of donating a proton, and a base is a substance, charged or
Ionic Equilibria Brönsted Lowry Theory According to the Brönsted Lowry theory, an acid is a substance, charged or uncharged, that is capable of donating a proton, and a base is a substance, charged or
Atmaram K. Mapari* Department of Chemistry, Ramnarain Ruia College, Matunga (E), Mumbai: , Maharashtra, India.
 International Journal of ChemTech Research CDE (USA): IJCRGG, ISS: 0974-490, ISS(nline):455-9555 Vol.0 o.5, pp 73-77, 07 Stability Constants of ixed Ligand Complexes of Transition etal(ii) ions with-[(e)--(-chloro-6-
International Journal of ChemTech Research CDE (USA): IJCRGG, ISS: 0974-490, ISS(nline):455-9555 Vol.0 o.5, pp 73-77, 07 Stability Constants of ixed Ligand Complexes of Transition etal(ii) ions with-[(e)--(-chloro-6-
Acid Base Equilibria
 Acid Base Equilibria Acid Ionization, also known as acid dissociation, is the process in where an acid reacts with water to produce a hydrogen ion and the conjugate base ion. HC 2 H 3 O 2(aq) H + (aq)
Acid Base Equilibria Acid Ionization, also known as acid dissociation, is the process in where an acid reacts with water to produce a hydrogen ion and the conjugate base ion. HC 2 H 3 O 2(aq) H + (aq)
Chapter 16: Acid Base Equilibria Chapter 16 Acid-Base Equilibria Learning Standards & Objectives;
 Chapter 16: Acid Base Equilibria Chapter 16 Acid-Base Equilibria Learning Standards & Objectives; Chapter 16 AP16-1,2-01 AP16-1,2-02 AP16-1,2-03 AP16-3,4-01 AP16-3,4-02 AP16-5-01 AP16-6,7-01 AP16-6,7-02
Chapter 16: Acid Base Equilibria Chapter 16 Acid-Base Equilibria Learning Standards & Objectives; Chapter 16 AP16-1,2-01 AP16-1,2-02 AP16-1,2-03 AP16-3,4-01 AP16-3,4-02 AP16-5-01 AP16-6,7-01 AP16-6,7-02
3.15 Nuclear Magnetic Resonance Spectroscopy, NMR
 3.15 Nuclear Magnetic Resonance Spectroscopy, NMR What is Nuclear Magnetic Resonance - NMR Developed by chemists and physicists together it works by the interaction of magnetic properties of certain nuclei
3.15 Nuclear Magnetic Resonance Spectroscopy, NMR What is Nuclear Magnetic Resonance - NMR Developed by chemists and physicists together it works by the interaction of magnetic properties of certain nuclei
Chapter 25. Organic and Biological Chemistry. Organic and
 Chapter 25 Calculate grade: (Add exam1 - exam 4 scores)x1.5 Add 7 best quizzes (each quiz is worth 29) Gives you the # points you have so far. Final (worth 200 points) Grades: 800-4 750-3.5 700-3 650-2.5
Chapter 25 Calculate grade: (Add exam1 - exam 4 scores)x1.5 Add 7 best quizzes (each quiz is worth 29) Gives you the # points you have so far. Final (worth 200 points) Grades: 800-4 750-3.5 700-3 650-2.5
a. Always start with the species added to water.
 Calculation of ph Proton Balance Equations According to the Brönsted Lowry theory, every proton donated by an acid must be accepted by a base. Thus, an equation accounting for the total proton transfers
Calculation of ph Proton Balance Equations According to the Brönsted Lowry theory, every proton donated by an acid must be accepted by a base. Thus, an equation accounting for the total proton transfers
Acceptable Answers Reject Mark. Same atomic number but different mass number only = (1)
 (a) Atoms (of an element) with the same number of protons () But with different number of neutrons () Same atomic number but different mass number only = () Element(s) with same number of protons but different
(a) Atoms (of an element) with the same number of protons () But with different number of neutrons () Same atomic number but different mass number only = () Element(s) with same number of protons but different
HWeb27 ( ; )
 HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
HWeb27 (9.1-9.2; 9.12-9.18) 28.1. Which of the following cannot be determined about a compound by mass spectrometry? [a]. boiling point [b]. molecular formula [c]. presence of heavy isotopes (e.g., 2 H,
ACID-BASE EXTRACTION
 ACID-BASE EXTRACTION An acid-base extraction is a type of liquid-liquid extraction. It typically involves different solubility levels in water and an organic solvent. The organic solvent may be any carbon-based
ACID-BASE EXTRACTION An acid-base extraction is a type of liquid-liquid extraction. It typically involves different solubility levels in water and an organic solvent. The organic solvent may be any carbon-based
World Journal of Pharmaceutical and Life Sciences WJPLS
 wjpls, 06, Vol., Issue 5, 7-4. Research Article ISSN 454-9 Santosh. WJPLS www.wjpls.org SJIF Impact Factor: 3.347 STABILITY CONSTANTS OF IXED LIGAND COPLEXES OF TRANSITION ETAL(II) IONS WIT-{(E)-[(4-BROO--
wjpls, 06, Vol., Issue 5, 7-4. Research Article ISSN 454-9 Santosh. WJPLS www.wjpls.org SJIF Impact Factor: 3.347 STABILITY CONSTANTS OF IXED LIGAND COPLEXES OF TRANSITION ETAL(II) IONS WIT-{(E)-[(4-BROO--
Protein Structure Determination using NMR Spectroscopy. Cesar Trinidad
 Protein Structure Determination using NMR Spectroscopy Cesar Trinidad Introduction Protein NMR Involves the analysis and calculation of data collected from multiple NMR techniques Utilizes Nuclear Magnetic
Protein Structure Determination using NMR Spectroscopy Cesar Trinidad Introduction Protein NMR Involves the analysis and calculation of data collected from multiple NMR techniques Utilizes Nuclear Magnetic
NMR NEWS June To find tutorials, links and more, visit our website
 Department of Chemistry NMR Facilities Director: Dr. Carlos A. Steren NMR NEWS June 2014 To find tutorials, links and more, visit our website www.chem.utk.edu/facilities/nmr Computers and software updates
Department of Chemistry NMR Facilities Director: Dr. Carlos A. Steren NMR NEWS June 2014 To find tutorials, links and more, visit our website www.chem.utk.edu/facilities/nmr Computers and software updates
Supplementary Data. Design, synthesis and evaluation of molecularly targeted hypoxia-activated prodrugs
 Supplementary Data Design, synthesis and evaluation of molecularly targeted hypoxia-activated prodrugs Liam J. O Connor 1,2, Cindy Cazares-Körner 1,2,, Jaideep Saha 1,, Charles. G. Evans 1,2 Michael R.
Supplementary Data Design, synthesis and evaluation of molecularly targeted hypoxia-activated prodrugs Liam J. O Connor 1,2, Cindy Cazares-Körner 1,2,, Jaideep Saha 1,, Charles. G. Evans 1,2 Michael R.
Name: Student Number: University of Manitoba - Department of Chemistry CHEM Introductory Organic Chemistry II - Term Test 1
 Name: Student Number: University of Manitoba - Department of Chemistry CEM 2220 - Introductory Organic Chemistry II - Term Test 1 Thursday, February 16, 2012; 7-9 PM This is a 2-hour test, marked out of
Name: Student Number: University of Manitoba - Department of Chemistry CEM 2220 - Introductory Organic Chemistry II - Term Test 1 Thursday, February 16, 2012; 7-9 PM This is a 2-hour test, marked out of
Topic 1: Quantitative chemistry
 covered by A-Level Chemistry products Topic 1: Quantitative chemistry 1.1 The mole concept and Avogadro s constant 1.1.1 Apply the mole concept to substances. Moles and Formulae 1.1.2 Determine the number
covered by A-Level Chemistry products Topic 1: Quantitative chemistry 1.1 The mole concept and Avogadro s constant 1.1.1 Apply the mole concept to substances. Moles and Formulae 1.1.2 Determine the number
CHEMISTRY (CHE) CHE 104 General Descriptive Chemistry II 3
 Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
Chemistry (CHE) 1 CHEMISTRY (CHE) CHE 101 Introductory Chemistry 3 Survey of fundamentals of measurement, molecular structure, reactivity, and organic chemistry; applications to textiles, environmental,
CHAPTER 2. Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules
 CHAPTER 2 Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules 2-1 Kinetics and Thermodynamics of Simple Chemical Processes Chemical thermodynamics: Is concerned with the extent that
CHAPTER 2 Structure and Reactivity: Acids and Bases, Polar and Nonpolar Molecules 2-1 Kinetics and Thermodynamics of Simple Chemical Processes Chemical thermodynamics: Is concerned with the extent that
τ 1 > 1/J - if this lifetime is significantly shortened, the coupling (splitting of the signal) will not be observed
 It is often advantageous to reverse or remove the splitting caused by spin-spin coupling This is called spin decoupling Spin decoupling (or just decoupling) can be used for several reasons - to simplify
It is often advantageous to reverse or remove the splitting caused by spin-spin coupling This is called spin decoupling Spin decoupling (or just decoupling) can be used for several reasons - to simplify
PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008
 PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008 1 Name SECTION B. Answer each question in this section by writing the letter corresponding to the best answer on the line provided (2 points each; 60
PHARMACEUTICAL CHEMISTRY EXAM #1 Februrary 21, 2008 1 Name SECTION B. Answer each question in this section by writing the letter corresponding to the best answer on the line provided (2 points each; 60
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature14137 1. Supplementary Methods 2. Supplementary Text 2.1 Physico-Chemical Properties and Structural Elucidation of Compound 1. 2.2 Physico-Chemical Properties and Structural Elucidation
doi:10.1038/nature14137 1. Supplementary Methods 2. Supplementary Text 2.1 Physico-Chemical Properties and Structural Elucidation of Compound 1. 2.2 Physico-Chemical Properties and Structural Elucidation
Switching to OCR A from Pearson (Edexcel)
 Switching to OCR A from Pearson (Edexcel) The content within the OCR Chemistry A specification covers the key concepts of chemistry and will be very familiar. We ve laid it out in a logical progression
Switching to OCR A from Pearson (Edexcel) The content within the OCR Chemistry A specification covers the key concepts of chemistry and will be very familiar. We ve laid it out in a logical progression
5.65 g = kg m = mm 174 ml = L. 711 kg = g 3.79 km = m L = μl g = mg 745 μm = cm 127 μl = ml 302 C = K 185 K = C 100 C = K
 WLHS / AP Bio / UNIT 1 Chemistry of Life Name AP Biology Summer Assignment Use Campbell CH 2-4 Biology is the study of life and living things. Before we can study and understand many biological principles,
WLHS / AP Bio / UNIT 1 Chemistry of Life Name AP Biology Summer Assignment Use Campbell CH 2-4 Biology is the study of life and living things. Before we can study and understand many biological principles,
Titration curve of amino acids. BCH 312 [Practical]
![Titration curve of amino acids. BCH 312 [Practical] Titration curve of amino acids. BCH 312 [Practical]](/thumbs/95/122777605.jpg) Titration curve of amino acids BCH 312 [Practical] Titration curve Titration Curves are produced by monitoring the ph of a given volume of a sample solution after successive addition of acid or alkali.
Titration curve of amino acids BCH 312 [Practical] Titration curve Titration Curves are produced by monitoring the ph of a given volume of a sample solution after successive addition of acid or alkali.
Chapter 13 Structure t Determination: Nuclear Magnetic Resonance Spectroscopy
 John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 13 Structure t Determination: ti Nuclear Magnetic Resonance Spectroscopy Revisions by Dr. Daniel Holmes MSU Paul D. Adams University of Arkansas
John E. McMurry www.cengage.com/chemistry/mcmurry Chapter 13 Structure t Determination: ti Nuclear Magnetic Resonance Spectroscopy Revisions by Dr. Daniel Holmes MSU Paul D. Adams University of Arkansas
Benchtop NMR Combined with GC/MS Confirms Identity of Forensic Case Sample
 APPLICATION NOTE Benchtop NMR Combined with GC/MS Confirms Identity of Forensic Case Sample No. AN52889 Authors: Dean Antic, Ph.D., Thermo Fisher Scientific, San Jose, CA, USA WanLi Wei, Senior Engineer,
APPLICATION NOTE Benchtop NMR Combined with GC/MS Confirms Identity of Forensic Case Sample No. AN52889 Authors: Dean Antic, Ph.D., Thermo Fisher Scientific, San Jose, CA, USA WanLi Wei, Senior Engineer,
Unit-8 Equilibrium. Rate of reaction: Consider the following chemical reactions:
 Unit-8 Equilibrium Rate of reaction: Consider the following chemical reactions: 1. The solution of sodium chloride and silver nitrate when mixed, there is an instantaneous formation of a precipitate of
Unit-8 Equilibrium Rate of reaction: Consider the following chemical reactions: 1. The solution of sodium chloride and silver nitrate when mixed, there is an instantaneous formation of a precipitate of
cis trans-amide isomerism of the 3,4-dehydroproline residue, the unpuckered proline
 Supporting Information for cis trans-amide isomerism of the 3,4-dehydroproline residue, the unpuckered proline Vladimir Kubyshkin*, Nediljko Budisa* Address: Institute of Chemistry, Technical University
Supporting Information for cis trans-amide isomerism of the 3,4-dehydroproline residue, the unpuckered proline Vladimir Kubyshkin*, Nediljko Budisa* Address: Institute of Chemistry, Technical University
Nuclear Spin States. NMR Phenomenon. NMR Instrumentation. NMR Active Nuclei. Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance NMR Phenomenon µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (!). E Nuclear Spin States aligned
Nuclear Magnetic Resonance NMR Phenomenon µ A spinning charged particle generates a magnetic field. A nucleus with a spin angular momentum will generate a magnetic moment (!). E Nuclear Spin States aligned
Unit 11 Instrumentation. Mass, Infrared and NMR Spectroscopy
 Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Unit 11 Instrumentation Mass, Infrared and NMR Spectroscopy Spectroscopic identification of organic compounds Qualitative analysis: presence but not quantity (i.e. PEDs) Quantitative analysis: quantity
Amino Acids, Polypeptides and Proteins. An -amino acid is a carboxylic acid which has an amino group attached to the carbon to the COOH.
 Amino Acids, Polypeptides and Proteins An -amino acid is a carboxylic acid which has an amino group attached to the carbon to the OO. Polypeptides and proteins are composed, either completely or partially,
Amino Acids, Polypeptides and Proteins An -amino acid is a carboxylic acid which has an amino group attached to the carbon to the OO. Polypeptides and proteins are composed, either completely or partially,
BIOC 460 General Chemistry Review: Chemical Equilibrium, Ionization of H 2 O, ph, pk a
 BIOC 460 General Chemistry Review: Chemical Equilibrium, Ionization of H 2 O, ph, pk a General Equilibrium: What are the UNITS of K eq? Example reactions: A --> B units of K eq? A --> B + C units of K
BIOC 460 General Chemistry Review: Chemical Equilibrium, Ionization of H 2 O, ph, pk a General Equilibrium: What are the UNITS of K eq? Example reactions: A --> B units of K eq? A --> B + C units of K
Course Syllabus. Department: Science & Technology. Date: April I. Course Prefix and Number: CHM 212. Course Name: Organic Chemistry II
 Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
Department: Science & Technology Date: April 2012 I. Course Prefix and Number: CHM 212 Course Name: Organic Chemistry II Course Syllabus Credit Hours and Contact Hours: 5 credit hours and 7 (3:3:1) contact
PAPER No.12 :Organic Spectroscopy MODULE No.29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I
 Subject Chemistry Paper No and Title Module No and Title Module Tag 12: rganic Spectroscopy 29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I CHE_P12_M29 TABLE F CNTENTS 1. Learning utcomes
Subject Chemistry Paper No and Title Module No and Title Module Tag 12: rganic Spectroscopy 29: Combined problem on UV, IR, 1 H NMR, 13 C NMR and Mass - Part I CHE_P12_M29 TABLE F CNTENTS 1. Learning utcomes
3 Use of Mass Spectra to Obtain Structural Information
 3 Use of Mass Spectra to Obtain Structural Information 1 Mass Spectrometry One of the most sensitive and versatile analytical tools More sensitive than other spectroscopic methods (e.g. IR spectroscopy)
3 Use of Mass Spectra to Obtain Structural Information 1 Mass Spectrometry One of the most sensitive and versatile analytical tools More sensitive than other spectroscopic methods (e.g. IR spectroscopy)
A selection of resources to start advanced Chemistry lessons for year olds
 A selection of resources to start advanced Chemistry lessons for 14-18 year olds Kristy Turner and Catherine Smith RSC School Teacher Fellows 2011-12 Foreword Kristy graduated from the University of Bradford
A selection of resources to start advanced Chemistry lessons for 14-18 year olds Kristy Turner and Catherine Smith RSC School Teacher Fellows 2011-12 Foreword Kristy graduated from the University of Bradford
Unit title: Chemistry for Applied Biologists
 Unit title: Chemistry for Applied Biologists Unit code: K/601/0292 QCF level: 5 Credit value: 15 Aim This unit covers bonding, thermodynamics, reaction rates, equilibrium, oxidation and reduction and organic
Unit title: Chemistry for Applied Biologists Unit code: K/601/0292 QCF level: 5 Credit value: 15 Aim This unit covers bonding, thermodynamics, reaction rates, equilibrium, oxidation and reduction and organic
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Supporting Information Kinetic Resolution of Constitutional Isomers Controlled by Selective Protection inside a Supramolecular Nanocapsule Simin Liu, Haiying Gan, Andrew T. Hermann,
SUPPLEMENTARY INFORMATION Supporting Information Kinetic Resolution of Constitutional Isomers Controlled by Selective Protection inside a Supramolecular Nanocapsule Simin Liu, Haiying Gan, Andrew T. Hermann,
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding.
 Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
Molecular Geometry: VSEPR model stand for valence-shell electron-pair repulsion and predicts the 3D shape of molecules that are formed in bonding. Sigma and Pi Bonds: All single bonds are sigma(σ), that
1. (8 pts) Circle the formula (only one) that best fits each of the following descriptions:
 1. (8 pts) Circle the formula (only one) that best fits each of the following descriptions: a. largest radius 2 b. stronger acid (first ionization) HN 3 H 3 P 4 H 2 S 4 c. largest radius N 3 2 F e. highest
1. (8 pts) Circle the formula (only one) that best fits each of the following descriptions: a. largest radius 2 b. stronger acid (first ionization) HN 3 H 3 P 4 H 2 S 4 c. largest radius N 3 2 F e. highest
Module 20: Applications of PMR in Structural Elucidation of Simple and Complex Compounds and 2-D NMR spectroscopy
 Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Module 20: Applications of PMR in Structural Elucidation of Simple and Complex Compounds and 2-D NMR spectroscopy
Subject Chemistry Paper No and Title Module No and Title Module Tag Paper 12: Organic Spectroscopy Module 20: Applications of PMR in Structural Elucidation of Simple and Complex Compounds and 2-D NMR spectroscopy
Chemistry 12 August 2008 Form A Provincial Examination Answer Key
 Cognitive Processes K = Knowledge U = Understanding H = Higher Mental Processes Topics 1. Reaction Kinetics 2. Dynamic Equilibrium 3. Solubility Equilibria 4. Acids, Bases, and Salts 5. Oxidation Reduction
Cognitive Processes K = Knowledge U = Understanding H = Higher Mental Processes Topics 1. Reaction Kinetics 2. Dynamic Equilibrium 3. Solubility Equilibria 4. Acids, Bases, and Salts 5. Oxidation Reduction
Synthetic and 1 H and 13 C NMR Spectral Studies on N-(Mono-substitutedphenyl)-acetamides H 4. NH-CO- CH 3 i. ; X = Cl, CH 3
 Synthetic and 1 H and 13 C NMR Spectral Studies on N-(Mono-substitutedphenyl)-acetamides and Substituted Acetamides, 2/3/4-YC 6 H 4 NH-CO- CH 3 i X i (Y=CH 3, F, Cl, Br, NO 2 ; X = Cl, CH 3 ; i =0,1,2,3)
Synthetic and 1 H and 13 C NMR Spectral Studies on N-(Mono-substitutedphenyl)-acetamides and Substituted Acetamides, 2/3/4-YC 6 H 4 NH-CO- CH 3 i X i (Y=CH 3, F, Cl, Br, NO 2 ; X = Cl, CH 3 ; i =0,1,2,3)
Find the ph and the degree of ionization for an 0.10 M solution of formic acid:
 148 Find the ph and the degree of ionization for an 0.10 M solution of formic acid: Value of Ka from Ebbing, page A-13 Define "x" as the change in hydronium ion concentration Assume "x" is small compared
148 Find the ph and the degree of ionization for an 0.10 M solution of formic acid: Value of Ka from Ebbing, page A-13 Define "x" as the change in hydronium ion concentration Assume "x" is small compared
tert-butyl alcohol n-butyl alcohol methyl propyl ether (c) Explain briefly why n-butyl alcohol has a much higher bp than methyl propyl ether.
 1. (15 points) Three 4 10 isomers are shown below, along with their boiling points. ( 3 ) 3 3 2 2 2 3 2 2 3 tert-butyl alcohol n-butyl alcohol methyl propyl ether bp: 82 118 39 (a) Based on their boiling
1. (15 points) Three 4 10 isomers are shown below, along with their boiling points. ( 3 ) 3 3 2 2 2 3 2 2 3 tert-butyl alcohol n-butyl alcohol methyl propyl ether bp: 82 118 39 (a) Based on their boiling
