University of Groningen
|
|
|
- Suzan Doyle
- 5 years ago
- Views:
Transcription
1 University of Groningen Synthesis of α-hydroxy-ω-amino poly(ethylene oxide) and its use in reaction injection moulding (RIM) Loontjens, Ton J.A.; Scholtens, Boudewijn J.R.; Belt, Wil J.W.; Frisch, Kurt C.; Wong, Shaiowen Published in: Polymer Bulletin DI: /BF IMPRTANT NTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below. Document Version Publisher's PDF, also known as Version of record Publication date: 1993 Link to publication in University of Groningen/UMCG research database Citation for published version (APA): Loontjens, T. J. A., Scholtens, B. J. R., Belt, W. J. W., Frisch, K. C., & Wong, S. (1993). Synthesis of α- hydroxy-ω-amino poly(ethylene oxide) and its use in reaction injection moulding (RIM). Polymer Bulletin, 30(5), DI: /BF Copyright ther than for strictly personal use, it is not permitted to download or to forward/distribute the text or part of it without the consent of the author(s) and/or copyright holder(s), unless the work is under an open content license (like Creative Commons). Take-down policy If you believe that this document breaches copyright please contact us providing details, and we will remove access to the work immediately and investigate your claim. Downloaded from the University of Groningen/UMCG research database (Pure): For technical reasons the number of authors shown on this cover page is limited to 10 maximum. Download date:
2 Polymer Bulletin 30, (1993) Polymer Bulletin 9 Springer-Verlag 1993 Synthesis of a-hydroxy-o~-amino poly(ethylene oxide) and its use in reaction injection moulding (RIM) Ton J. A. Loontjens *,I, Boudewijn J, R. Scholtens 1, Wil J. W. Belt 1, Kurt C, Frisch 2, and Shaio-wen Wong 2 1DSM Research, P.. Box 18, 6160 MD Geleen, The Netherlands 2polymer Technologies, Inc., University of Detroit Mercy, 4001 W. McNichols Road, Detroit, MI , USA SD~ARY Computer simulations show that oligomers with two different terminal groups with dissimilar reactivities for isoeyanates give a delayed viseosity rise in polyurethanes. This is a desired behmviour for RIM processes. Therefore, an a-hydroxy-m-amino poly(ethylene oxide) (HAPE) has been prepared. The synthesis was carried out by the ethoxylation of 2-hydroxyethyl phthalimide as a blocked amine. Hydrazinolysis appears to be the best way to obtain the deblocked oligomer. The product properties were eompared withanoligomeric diamine ether (Jeffamine D2000). The gel time of HAPE (~ : 500) and JAD2000 (M~ = 2000) was the same (2 see.). The product with HAPE had a higher modulus, a comparable impact and tensile strength and a lower elongation at break. INTRDUCTIN Reaction injection moulding (RIM) is an important processing technique, in particular for polyurethanes. The cycle time of thermosets produced with this technique is about the same as for injection moulded thermoplasts. The starting materials of RIM are low viscous liquids which are transformed into a three dimensional network in about two minutes. The gelation has to take place in a relatively short time interval. This imposes severe requirements on the chemistry of such a system. In structural RIM (SRIM) these requirements are even more severe, because the mould is partly filled with glass fibers. The initial viscosity must, therefore, be low and stay low as long as wetting out is not complete. However, subsequently the polymerization rate should be very high. Thus, one needs a reactive system in which the viscosity rise due to gelation is postponed until a high degree of conversion is reached. Yet, an acceptable level of cure is required in the end product. At the moment, RIM polyurethanes are composed by reacting an isocyanate with a polymeric polyol and a chain extender. The latter is often a low molecular hindered aromatic diamine [1]. In order to obtain faster systems, polyureas are often employed. However, these systems are so fast that they are difficult to process [2]. Polymeric diamines (e.g. Jeffamines) are processable if the molecular mass is not too low. However, in order to obtain products with high matrix moduli (SRIM), low molecular mass prepolymers are preferred. We report here the synthesis of a-hydroxy-m-amino terminated polyethers (HAPE) and their application with isoeyanates in polyurethane thermoset~ in order to establish whether these products can fill the gap between the *Corresponding author
3 490 polyurethanes and the polyureas. The amine group of the HAPE reacts very fast with an isocyanate but will still give low molecular mass products since mainly di- and trimers are formed initially. Until a conversion of about 50 % of the isocyanate groups, it is expected that the molecular mass remains relatively low. In the second stage of conversion, the polymerization continues by reactions between the hydroxy terminated reaction products and the remaining isocyanates. This results in gelation and network formation, but at a higher conversion than when a diamine is used instead of HAPE. EXPERIMENTAL Phthalimide blocked ~-hydroxy-m-amino poly(ethylene oxide) 2-Hydroxyethyl phthalimide (HEP) was prepared according to Billman [5]; g of HEP and 3.63 g NaCH 3 were heated to 140 ~ The methanol was removed by repeated vacuum distillation. The ethoxylation was performed at 170 ~ during 2 hours under a pressure of about 3 bar. In that period about I0 monomeric units are polymerized. Higher molecular masses can be obtained by extending the reaction time. ~-Hydroxy-m-amino polv(ethylene oxide) I00 g of the phthalimide blocked ~-hydroxy-~-amino poly(ethylene oxide) was dissolved in 200 ml ethanol. 9.2 g of a hydrazine hydrate (55%, equimolar) was added and the mixture was refluxed for 30 minutes. Next, 15 g concentrated HCI was added. Immediately a yellow precipitate appeared. The precipitate was removed by filtration at 0 ~ The filtrate was neutralized with NaH and the NaCI formed was filtered off. The oligomer was isolated by evaporation. Reaction injection moulding A smale scale apparatus (RCM-800 machine, Hi-Tech Engineering) was used to carry out the RIM experiments. A mixture of the oligomer and the diethyltoluene diamine (DETDA) and the catalyst was one stream, while the diisocyanate formed the other one. The oligomer tank was kept at ~ under 3-5 bar pressure of dry nitrogen. The diisocyanate was held at room temperature. The impingement mixing head pressure was about I00 bar. The flow rate was 40 g/s at ~ and the shot time 0.7 s. After the injection, the sample slab was post-cured in the closed mould for I hour at 120 ~ RESULTS AND DISCUSSIN The formation of networks through mixtures of difunctional resins with diand trifunctional crosslinkers has been modeled with computer simulations by one of us [3]. This model was originally developed for reactions between molecules containing the same reactive group within each molecule. However, it is possible to introduce substitution effects for some of the functional groups. A negative substitution effects means, that the second group reacts slower after the first one has reacted. The larger the negative substitution effect the larger the difference in reactivities. The actual difference in reactivity between an aliphatic amine and an alcohol with an isocyanate is more than a factor of thousand [2,4]. We used this model to estimate the increase of the molecular mass as function of the isoeyanate conversion. The reactivity ratio between an amine and an
4 491 alcohol with an isocyanate was chosen to be I000. A larger difference has no more influence on the results. The functionality of the HAPE was set at two. This implies that biuret formation was neglected. The functionality of the isocyanate was taken to be 2.3, which is a typical value for commercial systems. The molecular mass of the HAPE and the isocyanate was set at 1500 and 300, respectively. In Fig. I two curves are shown, calculated with the aforementioned network model [3]. The R curve is calculated on the basis of equal reactivities, typical for either diamines or diols with a molecular mass of 1500 as well. The S curve is based on a model HAPE, i.e. an oligomer with a negative substitution effect of In the S curve, the molecular mass of the product reaches the sum of the isocyanate and the HAPE at 50 % conversion of the isocyanate and 100 % of the amine. This is, on the average, what one expects with such a large difference in reactivities. After this point, the polymerization continues with the urethane formation. Because the average functionality is larger than two, the product will finally be crosslinked. The postponed gelation as function of the conversion is obvious from these calculations. With the model the ultimate network structures were verified to be equal. This theoretical result urged us to look for a synthetic route to produce these oligomers. IF..2 T $ 0 E IEI E ~E m / * Ka-.01 le converslon NC Fig.l. Calculated mass average molecular mass of the polyurethane as function of the conversion of the diisocyanate. There are several routes to synthesize HAPE. ne way is to transfer a polymeric diol or diamine into a HAPE. For instance, one could react a diol with acrylonitrile and hydrogenate the resulting product. Another route is to react a diamine with a caprolactone. However, this results always in a mixture of products which is difficult to purify. We have chosen to start with ethanolamine in which the amine group was blocked. Ethanolamine reacted readily and quantitatively with phthalic anhydride to form an imide, see scheme I [5]. Next, the polyether was prepared by the ethoxylation of the imide with ethylene oxide (E). The initiator was obtained by treating the imide with sodium methoxylate. The methanol formed was
5 492 removed by evaporation. E was dosed to the molten initiator at about 150 ~ The ethoxylation proceeded in 2 hours at 3 bar pressure. The product had a dark brown colour. The degree of ethoxylation depends on the amount of E added. Two products were prepared, one with on average of I0 additional E's and one with 21 additional E's. Scheme 1. Reaction scheme of the synthesis of ~-hydroxy-m-amino poly(ethylene oxide) ~ + H~N~H 0 N --~ H NaMe IL I" ~" NH~ ~ N H + ~ o ~ ~. The imide protecting group is not completely insensitive to the alkaline conditions used during the ethoxylation. According to the ~H-NMR spectrum (Fig. 2) about 10 % of the phthalimide has been opened during the ethoxylation. The peaks between 7.6 and 7.7 ppm are assigned to the ring opened product. This can give by-products because the formed carboxylate anion can also be ethoxylated. ~P. Fig. 2. IH-NMR of the phthalimide blocked =-hydroxy-e-amino poly(ethylene oxide) (M n = 500).
6 493 The deblocking reaction was performed with the product as obtained. Several deblocking reactions were at our disposal. The alkaline hydrolysis was rejected because this might degrade the poly(ethylene oxide) [6]. Aminolysis with methylamine in water gave a solid which was difficult to handle and to purify [7]. The hydrazinolysis appears to be the best choice [8]. The reaction is rather fast (0.5 hour) in ethanol as solvent. The hydrazine phthalimide was removed by precipitating the HCI salt. After filtration the HAPE was neutralized and isolated by evaporation of the solvent in a 85~ yield. This product was tested in a RIM recipe and compared with Jeffamine D2000, an s-m-amine terminal poly(propylene oxide) (PF) with a molecular mass of about The results are given in Table I. Table I. Polyurethane-urea RIM systems based on JAD2000 and HAPE 500. Reagent JAD2000 RAPE 500 Amine Eq. Wt. (g) I ligomer (g) DETDA i) (g) Dabco T-12 2) (g) Dabco 33 LV 3) (g) UL-28 4) (g) isonate 143L 5) (g) ligomer/detda/isonate 6) 1/5/6.3 1/1/2.1 Gel time (s) 2 2 Hardness shore D Tensile strength (MPa) Young's modulus (MPa) Elongation (%) Flex. strength (MPa) Flex. modulus (MPa) Izod (J/m) ) Diethyltoluenediamine, 2) dibutyltin dilaurate, 3) triethylenediamine in dipropylene glycol, 4) tin carboxylate, s) carbodiimide modified MDI and 6) molar ratio. The results show that the gel time for both systems was 2 seconds in spite of the higher concentration of reactive groups in HAPE. This gel time is in the desired range for RIM. Faster systems are difficult to process. The HAPE however, has only a molecular mass of 500 while the molecular mass of the JAD 2000 is This difference is clearly reflected in the modulus of the product, which is about twice as high for the HAPE product. The elongation at
7 494 break is lower, as expected. Fortunately, the impact and tensile strength are in the same range as with JAD2000. The difference between the flexural and the tensile strength for the HAPE system is not clear. In structural RIM applications, a great advantage is expected from the HAPE system, because of the lower starting viscosity and the resulting better wetting out of the glass fibers CNCLUSINS The idea that the gel point of a reactive system can be delayed by using an oligomer with reactive groups of very much different reactivities was verified theoretically and experimentally. According to the theory, the ultimate network structure is not affected by the difference in reactivities. An a-hydroxy-m-amino poly(ethylene oxide) (HAPE) was prepared by ethoxylation of 2-hydroxyethyl phthalimide. The blocking group was removed by hydrazinolysis. With this oligomer polyurethanes were prepared by reaction injection moulding (RIM). The results were compared with polyureas made with Jeffamines (oligomeric diamines). With HAPE (M n = 500 g/mol) the same gel time was obtained as with JAD2000 (M n = 2000 g/mol), although the concentration of reactive groups is much higher. As expected, the modulus was higher and the elongation at break lower. The tensile strength was about the same. Thus, the possibility to use HAPE with lower molecular masses than comparable diamines to produce polyurethanes with the same gel time, was experimentally confirmed. REFERENCES [I]. K.C.Frisch, Ruber Chem. and Teehn. 53, no i, , (1980). [2]. C.W.Macosko, Polym. Eng. and Sci., 28, nr I0, , (may 1988). [3]. B.J.R. Scholtens and G.P.J.M. Tiemersma Thoone, Computer Applications in Applied Polymer Science II: Automation, Modelling and Simulation, T. Provder, Ed., ACS Symposium Series 404, Am. Chem. Soc., Washington, DC, 1989, ch. 19, p. 213; G.P.J.M. Tiemersma-Thoone, B.J.R. Scholtens, K. Dusek and M. Gordon, J. Polym. Sci. Part B, Polym. Phys., 29, , (1991). [4]. K.C.Frisch, Advances in Urethane Sei. and Techn., vol 1, 1-30, (1971). [5]. J.H.Billman, J. Am. Chem. Soo., 65, , (1943). [6]. Methoden der rganisehen Chemie, Stiokstoffverbindugen II, 4e Auflage, Ed. Houben Well. [7]. S. Wolfe, Can. J. Chem., 48, no 22, [8]. H.R. Ing, J. Chem. Soo., 1926, Accepted March 2, 1993 C
A Glossary of Terms Used in the Adhesives, Coatings and Elastomers (ACE) Sector
 A Glossary of Terms Used in the Adhesives, Coatings and Elastomers (ACE) Sector Abrasion resistance The ability of the coating membrane to resist mechanical action such as foot traffic and particles, which
A Glossary of Terms Used in the Adhesives, Coatings and Elastomers (ACE) Sector Abrasion resistance The ability of the coating membrane to resist mechanical action such as foot traffic and particles, which
University of Groningen. Levulinic acid from lignocellulosic biomass Girisuta, Buana
 University of Groningen Levulinic acid from lignocellulosic biomass Girisuta, Buana IMPRTANT NTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Levulinic acid from lignocellulosic biomass Girisuta, Buana IMPRTANT NTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth
 University of Groningen Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen
 University of Groningen "Giant Surfactants" Created by the Fast and Efficient Functionalization of a DNA Tetrahedron with a Temperature-Responsive Polymer Wilks, Thomas R.; Bath, Jonathan; de Vries, Jan
University of Groningen "Giant Surfactants" Created by the Fast and Efficient Functionalization of a DNA Tetrahedron with a Temperature-Responsive Polymer Wilks, Thomas R.; Bath, Jonathan; de Vries, Jan
Citation for published version (APA): Sok, R. M. (1994). Permeation of small molecules across a polymer membrane: a computer simulation study s.n.
 University of Groningen Permeation of small molecules across a polymer membrane Sok, Robert Martin IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Permeation of small molecules across a polymer membrane Sok, Robert Martin IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
Precursor provides selectivity
 Precursor provides selectivity Novel route to isocyanate-free blocked isocyanates with functional groups. Ton Loontjens, Bart Plum, Tim Kidd, Boudewijn Scholtens, Polyurethane coatings, prepared from polyols
Precursor provides selectivity Novel route to isocyanate-free blocked isocyanates with functional groups. Ton Loontjens, Bart Plum, Tim Kidd, Boudewijn Scholtens, Polyurethane coatings, prepared from polyols
University of Groningen
 University of Groningen The Driving Mechanism Behind Attrition-Enhanced Deracemization Noorduin, Wim L.; Enckevort, Willem J.P. van; Meekes, Hugo; Kaptein, Bernard; Kellogg, Richard M.; Tully, John C.;
University of Groningen The Driving Mechanism Behind Attrition-Enhanced Deracemization Noorduin, Wim L.; Enckevort, Willem J.P. van; Meekes, Hugo; Kaptein, Bernard; Kellogg, Richard M.; Tully, John C.;
University of Groningen
 University of Groningen Dutch Resolution, A New Technology in Classical Resolution Broxterman, Quirinus B.; Echten, Erik van; Hulshof, Lumbertus A.; Kaptein, Bernard; Kellogg, Richard M.; Minnaard, Adriaan;
University of Groningen Dutch Resolution, A New Technology in Classical Resolution Broxterman, Quirinus B.; Echten, Erik van; Hulshof, Lumbertus A.; Kaptein, Bernard; Kellogg, Richard M.; Minnaard, Adriaan;
Synthesis of UV-Curable Hyperbranched Urethane Acrylates
 Synthesis of UV-Curable yperbranched Urethane Acrylates Ahmet ebioglu, James. Aerykssen, R. David Zopf, Igor V. Khudyakov, Bomar Specialties, Torrington, USA 1. Introduction yperbranched, highly branched,
Synthesis of UV-Curable yperbranched Urethane Acrylates Ahmet ebioglu, James. Aerykssen, R. David Zopf, Igor V. Khudyakov, Bomar Specialties, Torrington, USA 1. Introduction yperbranched, highly branched,
For more information about how to cite these materials visit
 Author(s): MELO 3D Project Team, 2012 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share-Alike 3.0 License: http://creativecommons.org/licenses/by-sa/2.0/
Author(s): MELO 3D Project Team, 2012 License: Unless otherwise noted, this material is made available under the terms of the Creative Commons Attribution Share-Alike 3.0 License: http://creativecommons.org/licenses/by-sa/2.0/
Curing Properties of Cycloaliphatic Epoxy Derivatives
 Curing Properties of Cycloaliphatic Epoxy Derivatives Hiroshi Sasaki Toagosei Co. Ltd. Nagoya, Japan Introduction UV-cationic-curing, based on the photo-generation of acid and consecutive cationic polymerization,
Curing Properties of Cycloaliphatic Epoxy Derivatives Hiroshi Sasaki Toagosei Co. Ltd. Nagoya, Japan Introduction UV-cationic-curing, based on the photo-generation of acid and consecutive cationic polymerization,
Supporting Information
 Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany A Distinctive Organocatalytic Approach to Complex Macromolecular Architectures Olivier Coulembier, Matthew 5. 5iesewetter, Andrew Mason, Philippe
Supporting Information Wiley-VCH 2007 69451 Weinheim, Germany A Distinctive Organocatalytic Approach to Complex Macromolecular Architectures Olivier Coulembier, Matthew 5. 5iesewetter, Andrew Mason, Philippe
University of Groningen. Laser Spectroscopy of Trapped Ra+ Ion Versolato, Oscar Oreste
 University of Groningen Laser Spectroscopy of Trapped Ra+ Ion Versolato, Oscar Oreste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Laser Spectroscopy of Trapped Ra+ Ion Versolato, Oscar Oreste IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Theoretical simulation of nonlinear spectroscopy in the liquid phase La Cour Jansen, Thomas
 University of Groningen Theoretical simulation of nonlinear spectroscopy in the liquid phase La Cour Jansen, Thomas IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF)
University of Groningen Theoretical simulation of nonlinear spectroscopy in the liquid phase La Cour Jansen, Thomas IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF)
University of Groningen
 University of Groningen Nonlinear optical properties of one-dimensional organic molecular aggregates in nanometer films Markov, R.V.; Plekhanov, A.I.; Shelkovnikov, V.V.; Knoester, Jasper Published in:
University of Groningen Nonlinear optical properties of one-dimensional organic molecular aggregates in nanometer films Markov, R.V.; Plekhanov, A.I.; Shelkovnikov, V.V.; Knoester, Jasper Published in:
Alcohols, Phenols and Ethers
 SUBJECTIVE PROBLEMS: Alcohols, Phenols and Ethers Q1. An organic liquid (A), containing C, H and O with boiling point: 78 o C, and possessing a rather pleasant odour, on heating with concentrated sulphuric
SUBJECTIVE PROBLEMS: Alcohols, Phenols and Ethers Q1. An organic liquid (A), containing C, H and O with boiling point: 78 o C, and possessing a rather pleasant odour, on heating with concentrated sulphuric
1.1 Basic Polymer Chemistry. 1.2 Polymer Nomenclature. 1.3 Polymer Synthesis. 1.4 Chain Growth Polymerization. Polymer =
 1.1 Basic Polymer hemistry Polymers are the largest class of soft materials: over 100 billion pounds of polymers made in US each year lassification systems 1.2 Polymer Nomenclature Polymer = Monomer =
1.1 Basic Polymer hemistry Polymers are the largest class of soft materials: over 100 billion pounds of polymers made in US each year lassification systems 1.2 Polymer Nomenclature Polymer = Monomer =
race to the Second Edition Preface to the First Edition 'kno\t'ledgements Contents
 Contents race to the Second Edition Preface to the First Edition 'kno\t'ledgements Chemistry and Basic Intermediates. Introduction. Basic Chemistry Basic Structure of a Polyurethane Elastomer. Synthesis
Contents race to the Second Edition Preface to the First Edition 'kno\t'ledgements Chemistry and Basic Intermediates. Introduction. Basic Chemistry Basic Structure of a Polyurethane Elastomer. Synthesis
SUPPLEMENTARY INFORMATION
 University of Groningen Direct observation of the spin-dependent Peltier effect Flipse, J.; Bakker, F. L.; Slachter, A.; Dejene, F. K.; van Wees, Bart Published in: Nature Nanotechnology DOI: 10.1038/NNANO.2012.2
University of Groningen Direct observation of the spin-dependent Peltier effect Flipse, J.; Bakker, F. L.; Slachter, A.; Dejene, F. K.; van Wees, Bart Published in: Nature Nanotechnology DOI: 10.1038/NNANO.2012.2
University of Groningen
 University of Groningen Identification of Early Intermediates of Caspase Activation Using Selective Inhibitors and Activity-Based Probes Berger, Alicia B.; Witte, Martin; Denault, Jean-Bernard; Sadaghiani,
University of Groningen Identification of Early Intermediates of Caspase Activation Using Selective Inhibitors and Activity-Based Probes Berger, Alicia B.; Witte, Martin; Denault, Jean-Bernard; Sadaghiani,
Electronic Supplementary Information for New Journal of Chemistry
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2015 Electronic Supplementary Information
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2015 Electronic Supplementary Information
Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n.
![Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n. Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n.](/thumbs/86/93829092.jpg) University of Groningen Fullerenes for organic electronics Kooistra, Floris Berend IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Fullerenes for organic electronics Kooistra, Floris Berend IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen
 University of Groningen Photosynthetic Quantum Yield Dynamics Hogewoning, Sander W.; Wientjes, Emilie; Douwstra, Peter; Trouwborst, Govert; van Ieperen, Wim; Croce, Roberta; Harbinson, Jeremy Published
University of Groningen Photosynthetic Quantum Yield Dynamics Hogewoning, Sander W.; Wientjes, Emilie; Douwstra, Peter; Trouwborst, Govert; van Ieperen, Wim; Croce, Roberta; Harbinson, Jeremy Published
University of Groningen. Enantioselective liquid-liquid extraction in microreactors Susanti, Susanti
 University of Groningen Enantioselective liquid-liquid extraction in microreactors Susanti, Susanti IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
University of Groningen Enantioselective liquid-liquid extraction in microreactors Susanti, Susanti IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite
Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth
 University of Groningen Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Carbon dioxide removal processes by alkanolamines in aqueous organic solvents Hamborg, Espen Steinseth IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
Study on Military Polyurethane Adhesive
 Research Paper Study on Military Polyurethane Adhesive Changming Zhang 1 *, Zhanggen Huang 1, Jun Ping Li 2 and Xiao Juan Lu 2 1 State Key Lab. of Coal Conversion, Institute of Coal Chemistry, Chinese
Research Paper Study on Military Polyurethane Adhesive Changming Zhang 1 *, Zhanggen Huang 1, Jun Ping Li 2 and Xiao Juan Lu 2 1 State Key Lab. of Coal Conversion, Institute of Coal Chemistry, Chinese
Thermoset Resins and Their Composites
 ACCE 22, September 13-14, 14, 22 Bio-based Thermoset Resins and Their Composites M. Misra,, L. T. Drzal, A. K. Mohanty,, L. Belchler, G. Mehta,, J-P. J Latere Dwan sisa Michigan State University 21 Engineering
ACCE 22, September 13-14, 14, 22 Bio-based Thermoset Resins and Their Composites M. Misra,, L. T. Drzal, A. K. Mohanty,, L. Belchler, G. Mehta,, J-P. J Latere Dwan sisa Michigan State University 21 Engineering
Supplementary Figure 1. Temperature profile of self-seeding method for polymer single crystal preparation in dilute solution.
 Supplementary Figure 1. Temperature profile of self-seeding method for polymer single crystal preparation in dilute solution. Supplementary Figure 2. 1 H nuclear magnetic resonance (NMR) spectra (a) and
Supplementary Figure 1. Temperature profile of self-seeding method for polymer single crystal preparation in dilute solution. Supplementary Figure 2. 1 H nuclear magnetic resonance (NMR) spectra (a) and
Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia
 University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
University of Groningen Peptide folding in non-aqueous environments investigated with molecular dynamics simulations Soto Becerra, Patricia IMPORTANT NOTE: You are advised to consult the publisher's version
Citation for published version (APA): Nouri-Nigjeh, E. (2011). Electrochemistry in the mimicry of oxidative drug metabolism Groningen: s.n.
 University of Groningen Electrochemistry in the mimicry of oxidative drug metabolism Nouri-Nigjeh, Eslam IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen Electrochemistry in the mimicry of oxidative drug metabolism Nouri-Nigjeh, Eslam IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen. Morphological design of Discrete-Time Cellular Neural Networks Brugge, Mark Harm ter
 University of Groningen Morphological design of Discrete-Time Cellular Neural Networks Brugge, Mark Harm ter IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Morphological design of Discrete-Time Cellular Neural Networks Brugge, Mark Harm ter IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
New Curing System of Urea-Formaldehyde Resins with Polyhydrazides I.
 455 Note [ MokuzaiGakkaishi Vol. 35, No. 5, p. 455-459 (1989) (Original Article)] New Curing System of Urea-Formaldehyde Resins with Polyhydrazides I. Curing with dihydrazide compounds* 1 Bunichiro TOMITA*
455 Note [ MokuzaiGakkaishi Vol. 35, No. 5, p. 455-459 (1989) (Original Article)] New Curing System of Urea-Formaldehyde Resins with Polyhydrazides I. Curing with dihydrazide compounds* 1 Bunichiro TOMITA*
University of Groningen
 University of Groningen Selective Pd-Catalyzed xidative Coupling of Anilides with lefins through C-H Bond Activation at Room Temperature Boele, Maarten D.K.; Strijdonck, Gino P.F. van; de Vries, André;
University of Groningen Selective Pd-Catalyzed xidative Coupling of Anilides with lefins through C-H Bond Activation at Room Temperature Boele, Maarten D.K.; Strijdonck, Gino P.F. van; de Vries, André;
Published in: ELECTRONIC PROPERTIES OF NOVEL MATERIALS - PROGRESS IN MOLECULAR NANOSTRUCTURES
 University of Groningen Fullerenes and nanostructured plastic solar cells Knol, Joop; Hummelen, Jan Published in: ELECTRONIC PROPERTIES OF NOVEL MATERIALS - PROGRESS IN MOLECULAR NANOSTRUCTURES IMPORTANT
University of Groningen Fullerenes and nanostructured plastic solar cells Knol, Joop; Hummelen, Jan Published in: ELECTRONIC PROPERTIES OF NOVEL MATERIALS - PROGRESS IN MOLECULAR NANOSTRUCTURES IMPORTANT
Supporting Information. Competitive Interactions of π-π Junctions and their Role on Microphase Separation of Chiral Block Copolymers
 Supporting Information Competitive Interactions of π-π Junctions and their Role on Microphase Separation of Chiral Block Copolymers Tao Wen, Jing-Yu Lee, Ming-Chia Li, Jing-Cherng Tsai and Rong-Ming Ho
Supporting Information Competitive Interactions of π-π Junctions and their Role on Microphase Separation of Chiral Block Copolymers Tao Wen, Jing-Yu Lee, Ming-Chia Li, Jing-Cherng Tsai and Rong-Ming Ho
Poly(N-substituted urethane)s with different molecular weights of polyethylene glycol
 Iranian Polymer Journal 14 (9), 005, 815-81 Synthesis and Characterization of N-Polyethylene Glycol Monomethyl Ether Substituted Polyurethane S. Mohammad Seyed Mohaghegh 1, Mehdi Barikani *, and Ali Akbar
Iranian Polymer Journal 14 (9), 005, 815-81 Synthesis and Characterization of N-Polyethylene Glycol Monomethyl Ether Substituted Polyurethane S. Mohammad Seyed Mohaghegh 1, Mehdi Barikani *, and Ali Akbar
University of Groningen. Extraction and transport of ion beams from an ECR ion source Saminathan, Suresh
 University of Groningen Extraction and transport of ion beams from an ECR ion source Saminathan, Suresh IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
University of Groningen Extraction and transport of ion beams from an ECR ion source Saminathan, Suresh IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish
Polymer Systems and Film Formation Mechanisms in High Solids, Powder, and UV Cure Systems
 Polymer Systems and Film Formation Mechanisms in High Solids, Powder, and UV Cure Systems J. Baghdachi, Ph.D. Coatings Research Institute Eastern Michigan University (734) 487-3192 Freshpaint@aol.com jamil.baghdachi@emich.edu
Polymer Systems and Film Formation Mechanisms in High Solids, Powder, and UV Cure Systems J. Baghdachi, Ph.D. Coatings Research Institute Eastern Michigan University (734) 487-3192 Freshpaint@aol.com jamil.baghdachi@emich.edu
Citation for published version (APA): Ruíz Duarte, E. An invitation to algebraic number theory and class field theory
 University of Groningen An invitation to algebraic number theory and class field theory Ruíz Duarte, Eduardo IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen An invitation to algebraic number theory and class field theory Ruíz Duarte, Eduardo IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen. Rheokinetics Cioffi, Mario
 University of Groningen Rheokinetics Cioffi, Mario IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below.
University of Groningen Rheokinetics Cioffi, Mario IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check the document version below.
Chemistry 283g- Experiment 3
 EXPERIMENT 3: xidation of Alcohols: Solid-Supported xidation and Qualitative Tests Relevant sections in the text: Fox & Whitesell, 3 rd Ed. pg. 448-452. A portion of this experiment is based on a paper
EXPERIMENT 3: xidation of Alcohols: Solid-Supported xidation and Qualitative Tests Relevant sections in the text: Fox & Whitesell, 3 rd Ed. pg. 448-452. A portion of this experiment is based on a paper
MODIFICATION WITH A SULFONATE MONOMER
 Thesis - MOLECULAR STRUCTURES AND FUNCTIONAL MODIFICATIONS OF POLY(VINYL ALCOHOL) CHAPTER 8 BY TOHEI MORITANI MODIFICATION WITH A SULFONATE MONOMER A functional monomer containing sodium sulfonate group,
Thesis - MOLECULAR STRUCTURES AND FUNCTIONAL MODIFICATIONS OF POLY(VINYL ALCOHOL) CHAPTER 8 BY TOHEI MORITANI MODIFICATION WITH A SULFONATE MONOMER A functional monomer containing sodium sulfonate group,
University of Groningen. Hollow-atom probing of surfaces Limburg, Johannes
 University of Groningen Hollow-atom probing of surfaces Limburg, Johannes IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
University of Groningen Hollow-atom probing of surfaces Limburg, Johannes IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
Citation for published version (APA): Shen, C. (2006). Wave Propagation through Photonic Crystal Slabs: Imaging and Localization. [S.l.]: s.n.
![Citation for published version (APA): Shen, C. (2006). Wave Propagation through Photonic Crystal Slabs: Imaging and Localization. [S.l.]: s.n. Citation for published version (APA): Shen, C. (2006). Wave Propagation through Photonic Crystal Slabs: Imaging and Localization. [S.l.]: s.n.](/thumbs/95/124821987.jpg) University of Groningen Wave Propagation through Photonic Crystal Slabs Shen, Chuanjian IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it.
University of Groningen Wave Propagation through Photonic Crystal Slabs Shen, Chuanjian IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it.
CHAPTER 3. FABRICATION TECHNOLOGIES OF CdSe/ZnS / Au NANOPARTICLES AND NANODEVICES. 3.1 THE SYNTHESIS OF Citrate-Capped Au NANOPARTICLES
 CHAPTER 3 FABRICATION TECHNOLOGIES OF CdSe/ZnS / Au NANOPARTICLES AND NANODEVICES 3.1 THE SYNTHESIS OF Citrate-Capped Au NANOPARTICLES Au NPs with ~ 15 nm were prepared by citrate reduction of HAuCl 4
CHAPTER 3 FABRICATION TECHNOLOGIES OF CdSe/ZnS / Au NANOPARTICLES AND NANODEVICES 3.1 THE SYNTHESIS OF Citrate-Capped Au NANOPARTICLES Au NPs with ~ 15 nm were prepared by citrate reduction of HAuCl 4
Supporting Information
 Supporting Information for Macromol. Chem. Phys, DOI: 10.1002/macp.201700302 Phase Segregation in Supramolecular Polymers Based on Telechelics Synthesized via Multicomponent Reactions Ansgar Sehlinger,
Supporting Information for Macromol. Chem. Phys, DOI: 10.1002/macp.201700302 Phase Segregation in Supramolecular Polymers Based on Telechelics Synthesized via Multicomponent Reactions Ansgar Sehlinger,
Citation for published version (APA): Hoekstra, S. (2005). Atom Trap Trace Analysis of Calcium Isotopes s.n.
 University of Groningen Atom Trap Trace Analysis of Calcium Isotopes Hoekstra, Steven IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Atom Trap Trace Analysis of Calcium Isotopes Hoekstra, Steven IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Sensitized solar cells with colloidal PbS-CdS core-shell quantum dots Lai, Lai-Hung; Protesescu, Loredana; Kovalenko, Maksym V.
 University of Groningen Sensitized solar cells with colloidal PbS-CdS core-shell quantum dots Lai, Lai-Hung; Protesescu, Loredana; Kovalenko, Maksym V.; Loi, Maria Published in: Physical Chemistry Chemical
University of Groningen Sensitized solar cells with colloidal PbS-CdS core-shell quantum dots Lai, Lai-Hung; Protesescu, Loredana; Kovalenko, Maksym V.; Loi, Maria Published in: Physical Chemistry Chemical
Module: 7. Lecture: 36
 Module: 7 Lecture: 36 DIMETHYL FORMAMIDE INTRODUCTION Dimethylformamide is an organic compound and denotes as DMF. The name is derived from the fact that it is a derivative of formamide, the amide of formic
Module: 7 Lecture: 36 DIMETHYL FORMAMIDE INTRODUCTION Dimethylformamide is an organic compound and denotes as DMF. The name is derived from the fact that it is a derivative of formamide, the amide of formic
University of Groningen. Statistical inference via fiducial methods Salomé, Diemer
 University of Groningen Statistical inference via fiducial methods Salomé, Diemer IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Statistical inference via fiducial methods Salomé, Diemer IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
18.8 Oxidation. Oxidation by silver ion requires an alkaline medium
 18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
18.8 Oxidation Oxidation by silver ion requires an alkaline medium Test for detecting aldehydes Tollens reagent to prevent precipitation of the insoluble silver oxide, a complexing agent is added: ammonia
Supporting Information for
 Supporting Information for Metal-Catalyzed Transesterification for Healing and Assembling of Thermosets Mathieu Capelot, Damien Montarnal, François Tournilhac, Ludwik Leibler* I. Epoxy-acid and epoxy-anhydride
Supporting Information for Metal-Catalyzed Transesterification for Healing and Assembling of Thermosets Mathieu Capelot, Damien Montarnal, François Tournilhac, Ludwik Leibler* I. Epoxy-acid and epoxy-anhydride
University of Groningen. Photophysics of nanomaterials for opto-electronic applications Kahmann, Simon
 University of Groningen Photophysics of nanomaterials for opto-electronic applications Kahmann, Simon IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Photophysics of nanomaterials for opto-electronic applications Kahmann, Simon IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
Citation for published version (APA): Fathi, K. (2004). Dynamics and morphology in the inner regions of spiral galaxies Groningen: s.n.
 University of Groningen Dynamics and morphology in the inner regions of spiral galaxies Fathi, Kambiz IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
University of Groningen Dynamics and morphology in the inner regions of spiral galaxies Fathi, Kambiz IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to
An Efficient Total Synthesis and Absolute Configuration. Determination of Varitriol
 An Efficient Total Synthesis and Absolute Configuration Determination of Varitriol Ryan T. Clemens and Michael P. Jennings * Department of Chemistry, University of Alabama, 500 Campus Dr. Tuscaloosa, AL
An Efficient Total Synthesis and Absolute Configuration Determination of Varitriol Ryan T. Clemens and Michael P. Jennings * Department of Chemistry, University of Alabama, 500 Campus Dr. Tuscaloosa, AL
Dual photo- and redox- active molecular switches for smart surfaces Ivashenko, Oleksii
 University of Groningen Dual photo- and redox- active molecular switches for smart surfaces Ivashenko, Oleksii IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Dual photo- and redox- active molecular switches for smart surfaces Ivashenko, Oleksii IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
SYNTHESIS OF AN AZO DYE revisited (1 or 2 credits)
 SYNTHESIS OF AN AZO DYE revisited (1 or 2 credits) This lab you can revisit the fist experiment of this quarter and synthesize more azo dyes of your choice. The old procedure is given below followed by
SYNTHESIS OF AN AZO DYE revisited (1 or 2 credits) This lab you can revisit the fist experiment of this quarter and synthesize more azo dyes of your choice. The old procedure is given below followed by
F G C G H H C B B A A DD E E
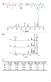 a D E F G H D E I F G H F F I DD E E G H H b c [1] 0 (M) [2a] 0 (M) [1] (M) [2a] (M) [3a] (M) K (M -1 ) a) 0.160 0.139 0.049 0.028 0.111 81 b) 0.147 0.180 0.025 0.058 0.122 84 c) 0.157 0.270 0.013 0.126
a D E F G H D E I F G H F F I DD E E G H H b c [1] 0 (M) [2a] 0 (M) [1] (M) [2a] (M) [3a] (M) K (M -1 ) a) 0.160 0.139 0.049 0.028 0.111 81 b) 0.147 0.180 0.025 0.058 0.122 84 c) 0.157 0.270 0.013 0.126
Electronic Supplementary Information
 Electronic Supplementary Information Effect of polymer chain conformation on field-effect transistor performance: synthesis and properties of two arylene imide based D-A copolymers Dugang Chen, a Yan Zhao,
Electronic Supplementary Information Effect of polymer chain conformation on field-effect transistor performance: synthesis and properties of two arylene imide based D-A copolymers Dugang Chen, a Yan Zhao,
Carbon dioxide removal by alkanolamines in aqueous organic solvents Hamborg, Espen S.; Derks, Peter W.J.; Elk, Edwin P. van; Versteeg, Geert F.
 University of Groningen Carbon dioxide removal by alkanolamines in aqueous organic solvents Hamborg, Espen S.; Derks, Peter W.J.; Elk, Edwin P. van; Versteeg, Geert F. Published in: Energy Procedia DOI:
University of Groningen Carbon dioxide removal by alkanolamines in aqueous organic solvents Hamborg, Espen S.; Derks, Peter W.J.; Elk, Edwin P. van; Versteeg, Geert F. Published in: Energy Procedia DOI:
Hyperbranched urethane-acrylates
 Hyperbranched urethane-acrylates Hyperbranched urethane-acrylates based on alkoxylated hydroxy acrylates combine high molecular weight, excellent reactivity and good coating properties, such as hardness,
Hyperbranched urethane-acrylates Hyperbranched urethane-acrylates based on alkoxylated hydroxy acrylates combine high molecular weight, excellent reactivity and good coating properties, such as hardness,
CHEM4. General Certificate of Education Advanced Level Examination January Unit 4 Kinetics, Equilibria and Organic Chemistry
 Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination January 2011 Question 1 2 Mark
Centre Number Surname Candidate Number For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination January 2011 Question 1 2 Mark
Department of Chemical Engineering, Massachusetts Institute of Technology, Cambridge, MA USA
 Supporting Information SELF-DECONTAMINATING FIBROUS MATERIALS REACTIVE TOWARD CHEMICAL THREATS Lev Bromberg, a Xiao Su, a Vladimir Martis, b Yunfei Zhang, a and T. Alan Hatton a* a Department of Chemical
Supporting Information SELF-DECONTAMINATING FIBROUS MATERIALS REACTIVE TOWARD CHEMICAL THREATS Lev Bromberg, a Xiao Su, a Vladimir Martis, b Yunfei Zhang, a and T. Alan Hatton a* a Department of Chemical
Methanolysis of Poly(ethylene terephthalate) in Supercritical Phase
 1 1 Vol.1 No.1 21 1 The Chinese Journal of Process Engineering Jan. 21 Methanolysis of Poly(ethylene terephthalate) in Supercritical Phase YANG Yong ( ) XIANG Hong-wei ( ) YANG Ji-li ( ), XU Yuan-yuan
1 1 Vol.1 No.1 21 1 The Chinese Journal of Process Engineering Jan. 21 Methanolysis of Poly(ethylene terephthalate) in Supercritical Phase YANG Yong ( ) XIANG Hong-wei ( ) YANG Ji-li ( ), XU Yuan-yuan
Supporting Information
 Supporting Information Thermoset Shape-Memory Polyurethane with Intrinsic Plasticity Enabled by Transcarbamoylation Ning Zheng, Zizheng Fang, Weike Zou, Qian Zhao,* and Tao Xie* anie_201602847_sm_miscellaneous_information.pdf
Supporting Information Thermoset Shape-Memory Polyurethane with Intrinsic Plasticity Enabled by Transcarbamoylation Ning Zheng, Zizheng Fang, Weike Zou, Qian Zhao,* and Tao Xie* anie_201602847_sm_miscellaneous_information.pdf
Experiment 15: Exploring the World of Polymers
 1 Experiment 15: Exploring the World of Polymers bjective: In this experiment, you will explore a class of chemical compounds known as polymers. You will synthesize and modify polymers, test their properties
1 Experiment 15: Exploring the World of Polymers bjective: In this experiment, you will explore a class of chemical compounds known as polymers. You will synthesize and modify polymers, test their properties
Substrate and Cation Binding Mechanism of Glutamate Transporter Homologs Jensen, Sonja
 University of Groningen Substrate and Cation Binding Mechanism of Glutamate Transporter Homologs Jensen, Sonja IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Substrate and Cation Binding Mechanism of Glutamate Transporter Homologs Jensen, Sonja IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
2K water-borne PU for furniture coatings
 2K water-borne PU for furniture coatings Water-borne 2K polyurethane systems are a new technology which is already successfully used in many applications also for wood coatings and more specifically furniture
2K water-borne PU for furniture coatings Water-borne 2K polyurethane systems are a new technology which is already successfully used in many applications also for wood coatings and more specifically furniture
Annals of West University of Timisoara
 Annals of West University of Timisoara Series Chemistry 13 (1) (2004) 27-32 SYNTHESIS IN NON-AQUEOUS MEDIUM OF SOME SYMMETRICAL DISAZO DIRECT DYES DERIVED FROM 4,4 -DIAMINOSTILBENE-2,2 -DISULPHONIC ACID
Annals of West University of Timisoara Series Chemistry 13 (1) (2004) 27-32 SYNTHESIS IN NON-AQUEOUS MEDIUM OF SOME SYMMETRICAL DISAZO DIRECT DYES DERIVED FROM 4,4 -DIAMINOSTILBENE-2,2 -DISULPHONIC ACID
University of Groningen. Event-based simulation of quantum phenomena Zhao, Shuang
 University of Groningen Event-based simulation of quantum phenomena Zhao, Shuang IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Event-based simulation of quantum phenomena Zhao, Shuang IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Supporting Information
 Supporting Information Syntheses and characterizations: Compound 1 was synthesized according to Scheme S-1. Scheme S-1 2 N N 5 i N 4 P Et Et iii N 6 ii P Et Et iv v, vi N N i) Fmoc-Su, DIPEA, Acetone;
Supporting Information Syntheses and characterizations: Compound 1 was synthesized according to Scheme S-1. Scheme S-1 2 N N 5 i N 4 P Et Et iii N 6 ii P Et Et iv v, vi N N i) Fmoc-Su, DIPEA, Acetone;
ELECTRONIC SUPPLEMENTARY INFORMATION
 Unprecedented Scissor Effect of Macromolecular Cross-linkers on the Glass Transition Temperature of Poly(Nvinylimidazole), Crystallinity Suppression of Poly(tetrahydrofuran) and Molecular Mobility by Solid
Unprecedented Scissor Effect of Macromolecular Cross-linkers on the Glass Transition Temperature of Poly(Nvinylimidazole), Crystallinity Suppression of Poly(tetrahydrofuran) and Molecular Mobility by Solid
Susterra Propanediol A Renewable Resource for Unsaturated Polyester Resins
 TECHNICAL BULLETIN Susterra Propanediol A Renewable Resource for Unsaturated Polyester Resins WILMINGTON, Del., July 9, 2008 Opportunities for Susterra Propanediol (1,3- propanediol) in unsaturated polyester
TECHNICAL BULLETIN Susterra Propanediol A Renewable Resource for Unsaturated Polyester Resins WILMINGTON, Del., July 9, 2008 Opportunities for Susterra Propanediol (1,3- propanediol) in unsaturated polyester
OCR (A) Chemistry A-level. Module 6: Organic Chemistry and Analysis
 OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
OCR (A) Chemistry A-level Module 6: Organic Chemistry and Analysis Organic Synthesis Notes by Adam Robertson DEFINITIONS Heterolytic fission: The breaking of a covalent bond when one of the bonded atoms
Chapter 5. Ionic Polymerization. Anionic.
 Chapter 5. Ionic Polymerization. Anionic. Anionic Polymerization Dr. Houston S. Brown Lecturer of Chemistry UH-Downtown brownhs@uhd.edu What you should know: What is anionic polymerization? What is MWD,
Chapter 5. Ionic Polymerization. Anionic. Anionic Polymerization Dr. Houston S. Brown Lecturer of Chemistry UH-Downtown brownhs@uhd.edu What you should know: What is anionic polymerization? What is MWD,
Decomposition Mechanism of Epoxy Resin in Nitric Acid for Recycling
 Decomposition Mechanism of Epoxy Resin in Nitric Acid for Recycling Weirong Dang I, Masatoshi Kubouchi I, Shurou Yamamoto I, Hideki Sembokuya I, Kazuyoshi Arai and Ken Tsuda Department of Chemical Engineering,
Decomposition Mechanism of Epoxy Resin in Nitric Acid for Recycling Weirong Dang I, Masatoshi Kubouchi I, Shurou Yamamoto I, Hideki Sembokuya I, Kazuyoshi Arai and Ken Tsuda Department of Chemical Engineering,
Supplementary Material for
 www.sciencemag.org/content/343/6173/873/suppl/dc1 Supplementary Material for Nonswellable Hydrogel Without Mechanical Hysteresis Hiroyuki Kamata, Yuki Akagi, Yuko Kayasuga-Kariya, Ung-il Chung, Takamasa
www.sciencemag.org/content/343/6173/873/suppl/dc1 Supplementary Material for Nonswellable Hydrogel Without Mechanical Hysteresis Hiroyuki Kamata, Yuki Akagi, Yuko Kayasuga-Kariya, Ung-il Chung, Takamasa
Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n.
![Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n. Citation for published version (APA): Kooistra, F. B. (2007). Fullerenes for organic electronics [Groningen]: s.n.](/thumbs/87/96826753.jpg) University of Groningen Fullerenes for organic electronics Kooistra, Floris Berend IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Fullerenes for organic electronics Kooistra, Floris Berend IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Working with Hazardous Chemicals
 A Publication of Reliable Methods for the Preparation of Organic Compounds Working with Hazardous Chemicals The procedures in Organic Syntheses are intended for use only by persons with proper training
A Publication of Reliable Methods for the Preparation of Organic Compounds Working with Hazardous Chemicals The procedures in Organic Syntheses are intended for use only by persons with proper training
Working with Hazardous Chemicals
 A Publication of Reliable Methods for the Preparation of Organic Compounds Working with Hazardous Chemicals The procedures in Organic Syntheses are intended for use only by persons with proper training
A Publication of Reliable Methods for the Preparation of Organic Compounds Working with Hazardous Chemicals The procedures in Organic Syntheses are intended for use only by persons with proper training
PROCESS FOR THE PREPARATION OF 5-CYANOPHTHALIDE AND INTERMEDIATES USEFUL THEREIN.
 ABSTRACT: PRCESS FR THE PREPARATIN F 5-CYANPHTHALIDE AND INTERMEDIATES USEFUL THEREIN. Process for the preparation of 5-cyanophthalide is disclosed which comprises reacting a pharmaceutically acceptable
ABSTRACT: PRCESS FR THE PREPARATIN F 5-CYANPHTHALIDE AND INTERMEDIATES USEFUL THEREIN. Process for the preparation of 5-cyanophthalide is disclosed which comprises reacting a pharmaceutically acceptable
University of Groningen. Statistical Auditing and the AOQL-method Talens, Erik
 University of Groningen Statistical Auditing and the AOQL-method Talens, Erik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
University of Groningen Statistical Auditing and the AOQL-method Talens, Erik IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please check
Honors Cup Synthetic Proposal
 onors Cup Synthetic Proposal Section: 270-V Group Members: Azhar Carim, Ian Cross, Albert Tang Title: Synthesis of indigo from -(2-bromoethyl)-2-nitrobenzamide Introduction: Indigo has been used as a dye
onors Cup Synthetic Proposal Section: 270-V Group Members: Azhar Carim, Ian Cross, Albert Tang Title: Synthesis of indigo from -(2-bromoethyl)-2-nitrobenzamide Introduction: Indigo has been used as a dye
CHEM4. General Certificate of Education Advanced Level Examination June Unit 4 Kinetics, Equilibria and Organic Chemistry
 Centre Number Surname Candidate Number For Examiner s Use ther Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Chemistry
Centre Number Surname Candidate Number For Examiner s Use ther Names Candidate Signature Examiner s Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Chemistry
Quiz 5 Introduction to Polymers
 100506 Quiz 5 Introduction to Polymers 1) Polyurethane in the video shown in class is formed from two liquids that are mixed. After mixing the solution foams and expands fairly rapidly forming a solid
100506 Quiz 5 Introduction to Polymers 1) Polyurethane in the video shown in class is formed from two liquids that are mixed. After mixing the solution foams and expands fairly rapidly forming a solid
Scheme 2: Formation of Di- Halide via Chloronium Intermediate
 Bromination of Alkenes CHM226 Background The carbon- carbon double bond, also known as an alkene, is a very important functional group in organic chemistry, and is often used as a precursor in the synthesis
Bromination of Alkenes CHM226 Background The carbon- carbon double bond, also known as an alkene, is a very important functional group in organic chemistry, and is often used as a precursor in the synthesis
Can a Hexapole magnet of an ECR Ion Source be too strong? Drentje, A. G.; Barzangy, F.; Kremers, Herman; Meyer, D.; Mulder, J.; Sijbring, J.
 University of Groningen Can a Hexapole magnet of an ECR Ion Source be too strong? Drentje, A. G.; Barzangy, F.; Kremers, Herman; Meyer, D.; Mulder, J.; Sijbring, J. Published in: Default journal IMPORTANT
University of Groningen Can a Hexapole magnet of an ECR Ion Source be too strong? Drentje, A. G.; Barzangy, F.; Kremers, Herman; Meyer, D.; Mulder, J.; Sijbring, J. Published in: Default journal IMPORTANT
Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji
 University of Groningen Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
University of Groningen Superfluid helium and cryogenic noble gases as stopping media for ion catchers Purushothaman, Sivaji IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's
Innovative. Technologies. Chemie des Klebens Chemistry of Adhesives. Dr. Jochen Stock, Laboratory Manager CRL Germany: Neuss, November 27 th, 2013
 Chemie des Klebens Chemistry of Adhesives Dr. Jochen Stock, Laboratory Manager CRL Germany: Neuss, November 27 th, 2013 Innovative Technologies 1 Overview Chemie des Klebens Chemistry of Adhesives Introduction
Chemie des Klebens Chemistry of Adhesives Dr. Jochen Stock, Laboratory Manager CRL Germany: Neuss, November 27 th, 2013 Innovative Technologies 1 Overview Chemie des Klebens Chemistry of Adhesives Introduction
Introduction to Polyurethane!
 Introduction to Polyurethane! Chemistry and Structure-Property Relationships 05/07/16 Monomers and Polymers Monomer (mono one; mer part): Small molecules Polymer Cons8tutes many monomers. Polymeriza8on
Introduction to Polyurethane! Chemistry and Structure-Property Relationships 05/07/16 Monomers and Polymers Monomer (mono one; mer part): Small molecules Polymer Cons8tutes many monomers. Polymeriza8on
4023 Synthesis of cyclopentanone-2-carboxylic acid ethyl ester from adipic acid diethyl ester
 NP 4023 Synthesis of cyclopentanone-2-carboxylic acid ethyl ester from adipic acid diethyl ester NaEt C 10 H 18 4 Na C 2 H 6 C 8 H 12 3 (202.2) (23.0) (46.1) (156.2) Classification Reaction types and substance
NP 4023 Synthesis of cyclopentanone-2-carboxylic acid ethyl ester from adipic acid diethyl ester NaEt C 10 H 18 4 Na C 2 H 6 C 8 H 12 3 (202.2) (23.0) (46.1) (156.2) Classification Reaction types and substance
CHEMISTRY (SALTERS) Chemistry of Materials. OXFORD CAMBRIDGE AND RSA EXAMINATIONS Advanced GCE
 XFRD CAMBRIDGE AND RSA EXAMINATINS Advanced GCE CHEMISTRY (SALTERS) Chemistry of Materials 2849 Monday 19 JUNE 2006 Afternoon 1 hour 30 minutes Candidates answer on the question paper. Additional materials:
XFRD CAMBRIDGE AND RSA EXAMINATINS Advanced GCE CHEMISTRY (SALTERS) Chemistry of Materials 2849 Monday 19 JUNE 2006 Afternoon 1 hour 30 minutes Candidates answer on the question paper. Additional materials:
Flexible Packaging Adhesives The Basics. Larry Jopko Rohm and Haas Company. Abstract
 Flexible Packaging Adhesives The Basics Larry Jopko Rohm and Haas Company Abstract Flexible packaging adhesives are predominately based on urethane and acrylic chemistry. Backbone options create unique
Flexible Packaging Adhesives The Basics Larry Jopko Rohm and Haas Company Abstract Flexible packaging adhesives are predominately based on urethane and acrylic chemistry. Backbone options create unique
Thermal and mechanical properties of several phthalonitrile resin system
 Thermal and mechanical properties of several phthalonitrile resin system Heng Zhou, Feng Liu, Yuehai Li, Tong Zhao* Institute of Chemistry, Chinese Academy of Sciences, Beijing, 100190, China Abstract:An
Thermal and mechanical properties of several phthalonitrile resin system Heng Zhou, Feng Liu, Yuehai Li, Tong Zhao* Institute of Chemistry, Chinese Academy of Sciences, Beijing, 100190, China Abstract:An
Supporting Information for
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
University of Groningen. Bifurcations in Hamiltonian systems Lunter, Gerard Anton
 University of Groningen Bifurcations in Hamiltonian systems Lunter, Gerard Anton IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
University of Groningen Bifurcations in Hamiltonian systems Lunter, Gerard Anton IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you wish to cite from it. Please
Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry
 Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry 30 Questions (5 pages); Time limit = 45 minutes Use of books or notes is not permitted. 1. When analyzed with a polarimeter, which of the
Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry 30 Questions (5 pages); Time limit = 45 minutes Use of books or notes is not permitted. 1. When analyzed with a polarimeter, which of the
Polymer Chemistry Prof. Dibakar Dhara Department of Chemistry Indian Institute of Technology, Kharagpur
 Polymer Chemistry Prof. Dibakar Dhara Department of Chemistry Indian Institute of Technology, Kharagpur Lecture - 10 Radical Chain Polymerization (Contd.) (Refer Slide Time: 00:28) Welcome back, and we
Polymer Chemistry Prof. Dibakar Dhara Department of Chemistry Indian Institute of Technology, Kharagpur Lecture - 10 Radical Chain Polymerization (Contd.) (Refer Slide Time: 00:28) Welcome back, and we
Polymer Bulletin 9 by Springer-Verlag 1980
 Polymer Bulletin 3, 391-398 (1980) Polymer Bulletin 9 by Springer-Verlag 1980 Homogeneous Epoxy-Acrylic Interpenetrating Polymer Networks: Preparation and Thermal Properties Alain Dubuisson, Dominique
Polymer Bulletin 3, 391-398 (1980) Polymer Bulletin 9 by Springer-Verlag 1980 Homogeneous Epoxy-Acrylic Interpenetrating Polymer Networks: Preparation and Thermal Properties Alain Dubuisson, Dominique
Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone
 Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone Chemical Concepts Carbonyl chemistry, base catalyzed aldol reaction, melting point, recrystallization Green
Experiment 2 Solvent-free Aldol Condensation between 3,4-dimethoxybenzaldehyde and 1-indanone Chemical Concepts Carbonyl chemistry, base catalyzed aldol reaction, melting point, recrystallization Green
