Satish Chandra. Blackbody. Unit IV, BLACK BODY RADIATION. Radiation in a Hollow Enclosure. Pure Temperature Dependence
|
|
|
- Verity Gilmore
- 6 years ago
- Views:
Transcription
1 Lecture Notes Dated: Jan 04, 013 Blackbody The ability of a body to radiate is closely related to its ability to absorb radiation. This is to be expected, since a body at a constant temperature is in thermal equilibrium with its surroundings and must absorb energy from them at the same rate as it emits energy. It is convenient to consider as an ideal body one that absorbs all radiation incident upon it, regardless of frequency, and emit a radiation of universal character, but does not depend on the composition of the object. Such a body is called blackbody. A perfectly blackbody is one which absorb all the radiations of whatever frequency, incident on it. It neither reflects nor transmits any of the incident radiation and therefore, appears black whatever be the colour of radiation. Since all blackbodies behave identically, we can now disregard the precise nature of whatever is radiating. In practice, no substance possesses strictly the properties of a blackbody. Lamp black and the platinum black are the nearest approach to a blackbody. However, the bodies showing close approximation to a perfectly blackbody can be constructed. The blackbody due to Fery and the blackbody due to Wien are examples of a perfectly blackbody for experimental purpose. Radiation in a Hollow Enclosure In general, the nature of radiation and the amount of radiation per unit area per unit time, i.e., the quality and the quantity of radiation depends on the temperature as well as on the nature of the surface of radiating body. If however, we consider a nonconducting enclosure maintained at a constant temperature, i.e., nonconducting isothermal enclosure ; the quality and the quantity of radiation inside such an enclosure depend only on its temperature and are entirely independent of both the nature of the walls and the nature of the substances which may be inside it. Thus, we can say that, the energy density of the radiation in a uniform temperature enclosure depends on its temperature and is totally independent of the nature of the walls. Now, let us consider that the bodies A, B and C are placed inside an isothermal enclosure. Experience shows that each body will attain the temperature of the enclosure irrespective of its nature or original temperature. As the bodies are at the same temperature as that of the enclosure, each body must radiate energy at the same rate and exactly of same kind as its absorbs, each body must radiate energy at the same rate and exactly of same kind as it absorbs. In other words, we can say that, the bodies contained inside the enclosure have no effect on the quantity or quality of the radiant energy filled in the enclosure. Pure Temperature Dependence Let a blackbody be placed in an isothermal enclosure. The body will emit the full radiation of the enclosure after it is in thermal equilibrium with the enclosure. These radiations are independent of the nature of the substance. Clearly the radiation from an isothermal enclosure is identical with that from a blackbody at the same temperature. Therefore, the heat radiations in an isothermal enclosure are termed as blackbody radiation, and this radiation purely depends on its temperature. This characteristics of blackbody is termed as pure temperature dependence.
2 Blackbodies in Practice We have seen that if an enclosure be maintained at a constant temperature it becomes filled with radiation characteristic of a perfectly blackbody. A metallic cavity with a small hole is an example of a blackbody. Radiation entering the hole is eventually absorbed after successive reflections at the inner walls of the cavity. Some of the radiation re-emitted by the atoms in the walls of the cavity leaks out through the hole. This radiation (blackbody radiation) has the same character as that of any other perfect blackbody. A particular type of blackbody due to Fery (Figure 1) consists of a hollow copper sphere blackened inside with a very fine hole in the surface. When the radiations enter through the hole, they suffer a number of reflections at the inner walls of the sphere until it is completely absorbed. To avoid direct reflection from the surface opposite to the hole, a conical projection is made in front of the hole. Thus the hole acts as a blackbody absorber. When this sphere is placed in a bath such as water bath at a fixed temperature, the heat radiations comes out the hole. Thus only the hole not the walls of the sphere, acts as a black body radiator. Figure 1 Blackbody due to Fery. Figure Blackbody due to Wien. Another type of blackbody due to Wein consists of a hollow cylindrical metallic chamber C (Figure ) blackened inside and made of brass or platinum, depending upon the temperature at which it would be used. The cylinder is heated by an electric current passing through this platinum foil as indicated by thick dashes. The radiation then passes through a number of limiting diaphragms and emerges out of the hole O. The cylinder is surrounded by concentric porcelain tubes. The temperature of the blackbody is given by the thermo-element T. This is the type of blackbody now widely and commonly used for experimental purposes. Spectral Distribution of Blackbody Radiation The Lummer and Pringsheim in 1899 studied experimentally the distribution of energy among radiation of different frequency in the radiation spectrum of a blackbody. Experimental Arrangement to obtain Spectral Distribution A carbon tube heated electrically serves as the perfect blackbody S. A narrow beam of radiation from the blackbody is converted into a parallel beam by the use of a concave mirror M 1 as shown in figure 3. The parallel beam of radiation, then falls on flourspar prism P, which disperses the heat radiation into different frequencies in a similar manner as the glass prism does in the case of white light. The different frequencies in dispersed heat radiation are brought to focus at different points with the help of another concave mirror M. By slowly rotating the prism the different parts of the radiation spectrum in the form of a small band of frequencies can be made to fall on a linear bolometer B (a device to detect the thermal radiation), which measures the energy of the radiation in terms of the deflection in galvanometer G connected in the bolometer circuit. The spectral energy associated with the different frequencies is found to be different at a particular constant temperature.
3 Figure 3 Arrangement to study the blackbody radiation spectral distribution. The Spectrum of Blackbody Radiation The spectrum of blackbody radiation is shown in figure 4 for two temperatures, which is a plot of spectral energy density at each frequency, versus the frequency of the radiation. The following conclusions can be drawn from these curves: 1. At each constant temperature, the blackbody emits continuous heat radiation spectrum.. The energy associated with the radiation of a particular frequency increases with increase in temperature of the blackbody. 3. The energy distribution is not uniform, i.e., at a given temperature spectral energy density initially increases with frequency and after attaining a maxima, it decreases. 4. The frequency of maximum emission shifts towards greater frequency as the temperature of blackbody increases, i.e., ν max is proportional to absolute temperature,i.e., max T. This is in accordance to the Wien's displacement law. 5. The area enclosed between any curve and x-axis, which is total energy emitted per unit surface area per unit time at a temperature by a perfect blackbody is found to be proportional to the forth power of the absolute temperature, i.e., E T 4. This is the statement of Stefan-Boltzmann law of radiation. Figure 4 spectrum of blackbody radiation.
4 Explanation of Blackbody Spectrum Why does the blackbody spectrum have the shape shown in figure 4? Several efforts have been made to explain the results obtained in the spectrum of blackbody radiation, which is an outcome of convincing experiments. We shall discuss few important of them. Classical Explanation: Rayleigh Jeans Law To explain this spectrum, the classical calculation by Rayleigh and Jeans begins by considering a blackbody as a radiation filled cavity at the temperature T. Because the cavity walls are assumed to be perfect reflectors, the radiation must consist of standing electromagnetic waves, as in figure 5. In order for a node to occur at each wall, the path length from wall to wall, in any direction, must be an integral number of half wavelengths. Figure 5 If the cavity is a cube of length L on each side, this condition means that for standing waves in the x, y and z directions respectively, the possible wavelengths are such that n x = L =1,, 3,... number of half wavelengths in x direction n y = L =1,, 3,... number of half wavelengths in y direction (1) n z = L =1,, 3,.... number of half wavelengths in z direction For a standing wave in any arbitrary direction, it must be true that n x n y n z = L n x =1,,3,.... where n y =1,,3,... () n z =1,,3,... in order that the wave end in a node at its ends. To counts the number of standing waves g(λ)dλ within the cavity whose wavelengths lie between λ and λ + dλ, what we have to do is count the number of permissible sets of n x, n y, n z values that yield wavelengths in this interval. Let us imagine a n-space whose coordinate axes are n x, n y,and n z. Each point in the n-space corresponds
5 to a permissible set of n x, n y, n z values and thus corresponds to a standing wave also. If n is vector from origin to a particular point n x, n y, n z, its magnitude is n=n x n y n z The total number of wavelengths between λ and λ + dλ is the same as the number of points in n-space whose distances from the origin lie between n and n + dn. The volume of a spherical shell of radius n and thickness dn is (4π n dn), but we are only consider the octant (⅛ th )of this shell that includes positive values of n x, n y and n z. Also, for each standing wave counted in this way, there are two perpendicular directions of vibration. Hence, the number of independent standing waves in the cavity is g ndn=1/84 n dn=n dn (4) Now we consider the number of independent standing wave in the cavity as a function of their frequency ν instead of as a function of n. From Eqs. () and (3) we have n= L = L c and dn= L c d and so, the number of standing waves in the cavity whose frequency lies between ν and ν + dν, is g d = L L L3 d =8 d (5) c c c 3 As the cavity is a cube L long each side, the volume is therefore L 3. Then the number of independent standing waves G(ν)dν in the frequency interval between ν and ν + dν per unit volume in the cavity turned out to be G d = 1 L g d d 3 =8 (6) c 3 The formula is independent of the shape of the cavity. The higher the frequency ν, the shorter the wavelength and the greater the number of possible standing waves. The next step to find the average energy per standing wave. According to the classical theorem of equipartition of energy the average energy per degree of freedom of an entity that is part of the system of such entities in thermal equilibrium at the temperature T is ½kT. Each standing wave in a radiation filled cavity corresponds to two degrees of freedom, for a total of (½)kT, because each wave originates in an oscillator in the cavity wall. Such an oscillator has two degree of freedom, one that represents its kinetic energy and one that represents its potential energy. Thus, classical average energy per standing wave =kt (7) The energy u(ν)dν per unit volume in the cavity in frequency interval between ν to ν + dν, i.e., spectral energy density of blackbody is ud =G d = (3) 8 kt c 3 d (8) The result known as the Rayleigh-Jeans Law, for spectrum of blackbody radiation. This law agrees with experimental result only with lower frequencies (figure 6), but it fails miserably at higher frequencies (specially in ultraviolet region). The failure of Rayleigh-Jeans law in higher frequencies region is known as ultraviolet catastrophe.
Lecture 2 Blackbody radiation
 Lecture 2 Blackbody radiation Absorption and emission of radiation What is the blackbody spectrum? Properties of the blackbody spectrum Classical approach to the problem Plancks suggestion energy quantisation
Lecture 2 Blackbody radiation Absorption and emission of radiation What is the blackbody spectrum? Properties of the blackbody spectrum Classical approach to the problem Plancks suggestion energy quantisation
Notes on Black body spectrum
 Notes on Black body spectrum Stefano Atzeni October 9, 216 1 The black body Radiation incident on a body can be absorbed, reflected, transmitted. We call black body an ideal body that absorbs all incident
Notes on Black body spectrum Stefano Atzeni October 9, 216 1 The black body Radiation incident on a body can be absorbed, reflected, transmitted. We call black body an ideal body that absorbs all incident
The term "black body" was introduced by Gustav Kirchhoff in The light emitted by a black body is called black-body radiation.
 Black body (Redirected from Black-body radiation) As the temperature decreases, the peak of the black body radiation curve moves to lower intensities and longer wavelengths. The black-body radiation graph
Black body (Redirected from Black-body radiation) As the temperature decreases, the peak of the black body radiation curve moves to lower intensities and longer wavelengths. The black-body radiation graph
Module 5 : MODERN PHYSICS Lecture 23 : Particle and Waves
 Module 5 : MODERN PHYSICS Lecture 23 : Particle and Waves Objectives In this lecture you will learn the following Radiation (light) exhibits both wave and particle nature. Laws governing black body radiation,
Module 5 : MODERN PHYSICS Lecture 23 : Particle and Waves Objectives In this lecture you will learn the following Radiation (light) exhibits both wave and particle nature. Laws governing black body radiation,
Class 11: Thermal radiation
 Class : Thermal radiation By analyzing the results from a number of eperiments, Planck found the energy density of the radiation emitted by a black body in wavelength interval (, d + was well described
Class : Thermal radiation By analyzing the results from a number of eperiments, Planck found the energy density of the radiation emitted by a black body in wavelength interval (, d + was well described
Quantum Mechanics: Blackbody Radiation
 Blackbody Radiation Quantum Mechanics Origin of Quantum Mechanics Raleigh-Jeans law (derivation)-ultraviolet catastrophe, Wien s Distribution Law & Wein s Displacement law, Planck s radiation law (calculation
Blackbody Radiation Quantum Mechanics Origin of Quantum Mechanics Raleigh-Jeans law (derivation)-ultraviolet catastrophe, Wien s Distribution Law & Wein s Displacement law, Planck s radiation law (calculation
Quantum Physics Lecture 6
 Quantum Physics Lecture 6 Thermal Phenomena Specific Heats - Classical model, failure at low temperature - Einstein model Black Body radiation - Classical model, UV catastrophe - Planck model - Wien &
Quantum Physics Lecture 6 Thermal Phenomena Specific Heats - Classical model, failure at low temperature - Einstein model Black Body radiation - Classical model, UV catastrophe - Planck model - Wien &
Stellar Astrophysics: The Continuous Spectrum of Light
 Stellar Astrophysics: The Continuous Spectrum of Light Distance Measurement of Stars Distance Sun - Earth 1.496 x 10 11 m 1 AU 1.581 x 10-5 ly Light year 9.461 x 10 15 m 6.324 x 10 4 AU 1 ly Parsec (1
Stellar Astrophysics: The Continuous Spectrum of Light Distance Measurement of Stars Distance Sun - Earth 1.496 x 10 11 m 1 AU 1.581 x 10-5 ly Light year 9.461 x 10 15 m 6.324 x 10 4 AU 1 ly Parsec (1
Quantum Physics Lecture 5
 Quantum Physics Lecture 5 Thermal Phenomena - continued Black Body radiation - Classical model, UV catastrophe - Planck model, Wien & Stefan laws - Photoelectric effect revisited The hydrogen atom Planetary
Quantum Physics Lecture 5 Thermal Phenomena - continued Black Body radiation - Classical model, UV catastrophe - Planck model, Wien & Stefan laws - Photoelectric effect revisited The hydrogen atom Planetary
Modern Physics (Lec. 1)
 Modern Physics (Lec. 1) Physics Fundamental Science Concerned with the fundamental principles of the Universe Foundation of other physical sciences Has simplicity of fundamental concepts Divided into five
Modern Physics (Lec. 1) Physics Fundamental Science Concerned with the fundamental principles of the Universe Foundation of other physical sciences Has simplicity of fundamental concepts Divided into five
INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place.
 RADIATION INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place. Radiation: The energy emitted by matter in the form
RADIATION INTRODUCTION Radiation differs from conduction and convection in that it does not require the presence of a material medium to take place. Radiation: The energy emitted by matter in the form
Chapter 13. Phys 322 Lecture 34. Modern optics
 Chapter 13 Phys 3 Lecture 34 Modern optics Blackbodies and Lasers* Blackbodies Stimulated Emission Gain and Inversion The Laser Four-level System Threshold Some lasers Pump Fast decay Laser Fast decay
Chapter 13 Phys 3 Lecture 34 Modern optics Blackbodies and Lasers* Blackbodies Stimulated Emission Gain and Inversion The Laser Four-level System Threshold Some lasers Pump Fast decay Laser Fast decay
General Physics (PHY 2140)
 General Physics (PHY 2140) Lecture 27 Modern Physics Quantum Physics Blackbody radiation Plank s hypothesis http://www.physics.wayne.edu/~apetrov/phy2140/ Chapter 27 1 Quantum Physics 2 Introduction: Need
General Physics (PHY 2140) Lecture 27 Modern Physics Quantum Physics Blackbody radiation Plank s hypothesis http://www.physics.wayne.edu/~apetrov/phy2140/ Chapter 27 1 Quantum Physics 2 Introduction: Need
Physics 2D Lecture Slides Lecture 10. Jan.25, 2010
 Physics 2D Lecture Slides Lecture 10 Jan.25, 2010 Radiation from A Blackbody (a) Intensity of Radiation I =! R (#) d# " T 4 I =! T 4 (Area under curve) Stephan-Boltzmann Constant σ = 5.67 10-8 W / m 2
Physics 2D Lecture Slides Lecture 10 Jan.25, 2010 Radiation from A Blackbody (a) Intensity of Radiation I =! R (#) d# " T 4 I =! T 4 (Area under curve) Stephan-Boltzmann Constant σ = 5.67 10-8 W / m 2
Modern Physics, summer Modern physics. Historical introduction to quantum mechanics
 1 Modern physics 2 Gustav Kirchhoff (1824-1887) Surprisingly, the path to quantum mechanics begins with the work of German physicist Gustav Kirchhoff in 1859. Electron was discovered by J.J.Thomson in
1 Modern physics 2 Gustav Kirchhoff (1824-1887) Surprisingly, the path to quantum mechanics begins with the work of German physicist Gustav Kirchhoff in 1859. Electron was discovered by J.J.Thomson in
Sources of radiation
 Sources of radiation Most important type of radiation is blackbody radiation. This is radiation that is in thermal equilibrium with matter at some temperature T. Lab source of blackbody radiation: hot
Sources of radiation Most important type of radiation is blackbody radiation. This is radiation that is in thermal equilibrium with matter at some temperature T. Lab source of blackbody radiation: hot
Planck s Hypothesis of Blackbody
 Course : Bsc Applied Physical Science (Computer Science) Year Ist (Sem IInd) Paper title : Thermal Physics Paper No : 6 Lecture no. 20. Planck s Hypothesis of Blackbody Hello friends, in the last lecture
Course : Bsc Applied Physical Science (Computer Science) Year Ist (Sem IInd) Paper title : Thermal Physics Paper No : 6 Lecture no. 20. Planck s Hypothesis of Blackbody Hello friends, in the last lecture
Blackbody radiation. Main Laws. Brightness temperature. 1. Concepts of a blackbody and thermodynamical equilibrium.
 Lecture 4 lackbody radiation. Main Laws. rightness temperature. Objectives: 1. Concepts of a blackbody, thermodynamical equilibrium, and local thermodynamical equilibrium.. Main laws: lackbody emission:
Lecture 4 lackbody radiation. Main Laws. rightness temperature. Objectives: 1. Concepts of a blackbody, thermodynamical equilibrium, and local thermodynamical equilibrium.. Main laws: lackbody emission:
2. Fingerprints of Matter: Spectra
 2. Fingerprints of Matter: Spectra 2.1 Measuring spectra: prism and diffraction grating Light from the sun: white light, broad spectrum (wide distribution) of wave lengths. 19th century: light assumed
2. Fingerprints of Matter: Spectra 2.1 Measuring spectra: prism and diffraction grating Light from the sun: white light, broad spectrum (wide distribution) of wave lengths. 19th century: light assumed
5. Light-matter interactions: Blackbody radiation
 5. Light-matter interactions: Blackbody radiation REMINDER: no lecture on Monday Feb. 6th The electromagnetic spectrum Sources of light Boltzmann's Law Blackbody radiation The cosmic microwave background
5. Light-matter interactions: Blackbody radiation REMINDER: no lecture on Monday Feb. 6th The electromagnetic spectrum Sources of light Boltzmann's Law Blackbody radiation The cosmic microwave background
B. Sc. Physics (H.R.K) Chapter 49: Light and Quantum Physics LIGHT AND QUANTUM PHYSICS
 LIGHT AND QUANTUM PHYSICS 49.1 Thermal Radiations The radiations emitted by a body due to its temperature are called thermal radiations. All bodies not only emit the thermal radiations, but also absorb
LIGHT AND QUANTUM PHYSICS 49.1 Thermal Radiations The radiations emitted by a body due to its temperature are called thermal radiations. All bodies not only emit the thermal radiations, but also absorb
1. Historical perspective
 Atomic and Molecular Physics/Lecture notes presented by Dr. Fouad Attia Majeed/Third year students/college of Education (Ibn Hayyan)/Department of Physics/University of Babylon. 1. Historical perspective
Atomic and Molecular Physics/Lecture notes presented by Dr. Fouad Attia Majeed/Third year students/college of Education (Ibn Hayyan)/Department of Physics/University of Babylon. 1. Historical perspective
Chapter 27. Quantum Physics
 Chapter 27 Quantum Physics Need for Quantum Physics Problems remained from classical mechanics that relativity didn t explain Blackbody Radiation The electromagnetic radiation emitted by a heated object
Chapter 27 Quantum Physics Need for Quantum Physics Problems remained from classical mechanics that relativity didn t explain Blackbody Radiation The electromagnetic radiation emitted by a heated object
Chemistry 431. Lecture 1. Introduction Statistical Averaging Electromagnetic Spectrum Black body Radiation. NC State University
 Chemistry 431 Lecture 1 Introduction Statistical Averaging Electromagnetic Spectrum Black body Radiation NC State University Overview Quantum Mechanics Failure of classical physics Wave equation Rotational,
Chemistry 431 Lecture 1 Introduction Statistical Averaging Electromagnetic Spectrum Black body Radiation NC State University Overview Quantum Mechanics Failure of classical physics Wave equation Rotational,
Modern physics. Historical introduction to quantum mechanics
 2012-0-08 Modern physics dr hab. inż. Katarzyna ZAKRZEWSKA, prof. AGH KATEDRA ELEKTRONIKI, C-1, office 17, rd floor, phone 617 29 01, mobile phone 0 601 51 5 e-mail: zak@agh.edu.pl, Internet site http://home.agh.edu.pl/~zak
2012-0-08 Modern physics dr hab. inż. Katarzyna ZAKRZEWSKA, prof. AGH KATEDRA ELEKTRONIKI, C-1, office 17, rd floor, phone 617 29 01, mobile phone 0 601 51 5 e-mail: zak@agh.edu.pl, Internet site http://home.agh.edu.pl/~zak
Thermal Radiation By: Prof. K M Joshi
 Thermal Radiation By: Prof. K M Joshi Radiation originate due to emission of matter and its subsequent transports does not required any matter / medium. Que: Then what is the nature of this transport???
Thermal Radiation By: Prof. K M Joshi Radiation originate due to emission of matter and its subsequent transports does not required any matter / medium. Que: Then what is the nature of this transport???
I. INTRODUCTION AND HISTORICAL PERSPECTIVE
 I. INTRODUCTION AND HISTORICAL PERSPECTIVE A. Failures of Classical Physics At the end of the 19th century, physics was described via two main approaches. Matter was described by Newton s laws while radiation
I. INTRODUCTION AND HISTORICAL PERSPECTIVE A. Failures of Classical Physics At the end of the 19th century, physics was described via two main approaches. Matter was described by Newton s laws while radiation
Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240
 Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240 John D. Williams, Ph.D. Department of Electrical and Computer Engineering 406 Optics Building - UAHuntsville,
Lasers PH 645/ OSE 645/ EE 613 Summer 2010 Section 1: T/Th 2:45-4:45 PM Engineering Building 240 John D. Williams, Ph.D. Department of Electrical and Computer Engineering 406 Optics Building - UAHuntsville,
5. Light-matter interactions: Blackbody radiation
 5. Light-matter interactions: Blackbody radiation The electromagnetic spectrum Sources of light Boltzmann's Law Blackbody radiation why do hot things glow? The cosmic microwave background The electromagnetic
5. Light-matter interactions: Blackbody radiation The electromagnetic spectrum Sources of light Boltzmann's Law Blackbody radiation why do hot things glow? The cosmic microwave background The electromagnetic
Determination of Stefan-Boltzmann Constant.
 Determination of Stefan-Boltzmann Constant. An object at some non-zero temperature radiates electromagnetic energy. For the perfect black body, which absorbs all light that strikes it, it radiates energy
Determination of Stefan-Boltzmann Constant. An object at some non-zero temperature radiates electromagnetic energy. For the perfect black body, which absorbs all light that strikes it, it radiates energy
Chapter 1. Blackbody Radiation. Theory
 Chapter 1 Blackbody Radiation Experiment objectives: explore radiation from objects at certain temperatures, commonly known as blackbody radiation ; make measurements testing the Stefan-Boltzmann law in
Chapter 1 Blackbody Radiation Experiment objectives: explore radiation from objects at certain temperatures, commonly known as blackbody radiation ; make measurements testing the Stefan-Boltzmann law in
Chemistry 795T. Lecture 7. Electromagnetic Spectrum Black body Radiation. NC State University
 Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T. Black body Radiation. The wavelength and the frequency. The electromagnetic spectrum. Lecture 7
 Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Chemistry 795T Lecture 7 Electromagnetic Spectrum Black body Radiation NC State University Black body Radiation An ideal emitter of radiation is called a black body. Observation: that peak of the energy
Modern Physics. Unit 1: Classical Models and the Birth of Modern Physics Lecture 1.4: Blackbody Radiation and Photoelectric Effect
 Modern Physics Unit 1: Classical Models and the Birth of Modern Physics Lecture 1.4: Blackbody Radiation and Photoelectric Effect Ron Reifenberger Professor of Physics Purdue University 1 I. Blackbody
Modern Physics Unit 1: Classical Models and the Birth of Modern Physics Lecture 1.4: Blackbody Radiation and Photoelectric Effect Ron Reifenberger Professor of Physics Purdue University 1 I. Blackbody
Exam 2 Development of Quantum Mechanics
 PHYS40 (Spring 00) Riq Parra Exam # (Friday, April 1 th, 00) Exam Development of Quantum Mechanics Do NOT write your name on this exam. Write your class ID number on the top right hand corner of each problem
PHYS40 (Spring 00) Riq Parra Exam # (Friday, April 1 th, 00) Exam Development of Quantum Mechanics Do NOT write your name on this exam. Write your class ID number on the top right hand corner of each problem
Quantum Mechanics. Particle in a box All were partial answers, leading Schrödinger to wave mechanics
 Chemistry 4521 Time is flying by: only 15 lectures left!! Six quantum mechanics Four Spectroscopy Third Hour exam Three statistical mechanics Review Final Exam, Wednesday, May 4, 7:30 10 PM Quantum Mechanics
Chemistry 4521 Time is flying by: only 15 lectures left!! Six quantum mechanics Four Spectroscopy Third Hour exam Three statistical mechanics Review Final Exam, Wednesday, May 4, 7:30 10 PM Quantum Mechanics
CHAPTER 3 The Experimental Basis of Quantum Theory
 CHAPTER 3 The Experimental Basis of Quantum Theory 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Discovery of the X Ray and the Electron Determination of Electron Charge Line Spectra Quantization As far as I can
CHAPTER 3 The Experimental Basis of Quantum Theory 3.1 3.2 3.3 3.4 3.5 3.6 3.7 3.8 3.9 Discovery of the X Ray and the Electron Determination of Electron Charge Line Spectra Quantization As far as I can
Chapter 11 FUNDAMENTALS OF THERMAL RADIATION
 Chapter Chapter Fundamentals of Thermal Radiation FUNDAMENTALS OF THERMAL RADIATION Electromagnetic and Thermal Radiation -C Electromagnetic waves are caused by accelerated charges or changing electric
Chapter Chapter Fundamentals of Thermal Radiation FUNDAMENTALS OF THERMAL RADIATION Electromagnetic and Thermal Radiation -C Electromagnetic waves are caused by accelerated charges or changing electric
Planck s Hypothesis of Blackbody
 Course : Bsc Applied Physical Science (Computer Science) Year Ist (Sem IInd) Paper title : Thermal Physics Paper No : 6 Lecture no. 20. Planck s Hypothesis of Blackbody FAQs Q1. What were the shortcomings
Course : Bsc Applied Physical Science (Computer Science) Year Ist (Sem IInd) Paper title : Thermal Physics Paper No : 6 Lecture no. 20. Planck s Hypothesis of Blackbody FAQs Q1. What were the shortcomings
Blackbody Radiation. George M. Shalhoub
 Blackbody Radiation by George M. Shalhoub LA SALLE UNIVERSIY 900 West Olney Ave. Philadelphia, PA 94 shalhoub@lasalle.edu Copyright 996. All rights reserved. You are welcome to use this document in your
Blackbody Radiation by George M. Shalhoub LA SALLE UNIVERSIY 900 West Olney Ave. Philadelphia, PA 94 shalhoub@lasalle.edu Copyright 996. All rights reserved. You are welcome to use this document in your
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics Problem Solving 10: The Greenhouse Effect. Section Table and Group
 MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics 8.02 Problem Solving 10: The Greenhouse Effect Section Table and Group Names Hand in one copy per group at the end of the Friday Problem Solving
MASSACHUSETTS INSTITUTE OF TECHNOLOGY Department of Physics 8.02 Problem Solving 10: The Greenhouse Effect Section Table and Group Names Hand in one copy per group at the end of the Friday Problem Solving
Properties of Electromagnetic Radiation Chapter 5. What is light? What is a wave? Radiation carries information
 Concepts: Properties of Electromagnetic Radiation Chapter 5 Electromagnetic waves Types of spectra Temperature Blackbody radiation Dual nature of radiation Atomic structure Interaction of light and matter
Concepts: Properties of Electromagnetic Radiation Chapter 5 Electromagnetic waves Types of spectra Temperature Blackbody radiation Dual nature of radiation Atomic structure Interaction of light and matter
PH2200 Practice Final Exam Summer 2003
 INSTRUCTIONS 1. Write your name and student identification number on the answer sheet. 2. Please cover your answer sheet at all times. 3. This is a closed book exam. You may use the PH2200 formula sheet
INSTRUCTIONS 1. Write your name and student identification number on the answer sheet. 2. Please cover your answer sheet at all times. 3. This is a closed book exam. You may use the PH2200 formula sheet
TRANSMISSION OF HEAT
 TRANSMISSION OF HEAT Synopsis :. In general heat travels from one point to another whenever there is a difference of temperatures.. Heat flows from a body at higher temperature to a lower temperature..
TRANSMISSION OF HEAT Synopsis :. In general heat travels from one point to another whenever there is a difference of temperatures.. Heat flows from a body at higher temperature to a lower temperature..
Problems with Classical Physics. Blackbody Radiation Photoelectric Effect Compton Effect Bohr Model of Atom
 The Quantum Gang Problems with Classical Physics Blackbody Radiation Photoelectric Effect Compton Effect Bohr Model of Atom Why this shape? Why the drop? Blackbody Radiation A black body is an ideal system
The Quantum Gang Problems with Classical Physics Blackbody Radiation Photoelectric Effect Compton Effect Bohr Model of Atom Why this shape? Why the drop? Blackbody Radiation A black body is an ideal system
Black Body Radiation. Heated Bodies Radiate. How is Radiation Absorbed? Absorption and Emission. Observing the Black Body Spectrum.
 Black Body Radiation Michael Fowler University of Virginia Physics 252 Home Page Link to Previous Lecture Link to Next Lecture Heated Bodies Radiate We shall now turn to another puzzle confronting physicists
Black Body Radiation Michael Fowler University of Virginia Physics 252 Home Page Link to Previous Lecture Link to Next Lecture Heated Bodies Radiate We shall now turn to another puzzle confronting physicists
Phys 322 Lecture 34. Chapter 13. Modern optics. Note: 10 points will be given for attendance today and for the rest of the semester.
 Chapter 13 Phys 322 Lecture 34 Modern optics Note: 10 points will be given for attendance today and for the rest of the semester. Presentation schedule Name Topic Date Alip, Abylaikhan lasers Nov. 30th
Chapter 13 Phys 322 Lecture 34 Modern optics Note: 10 points will be given for attendance today and for the rest of the semester. Presentation schedule Name Topic Date Alip, Abylaikhan lasers Nov. 30th
Chapter 39. Particles Behaving as Waves
 Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Physics 2D Lecture Slides Lecture 10: Jan 26 th 2004
 Brian Wecht, the TA, is away this week. I will substitute for his office hours (in my office 3314 Mayer Hall, discussion and PS session. Pl. give all regrade requests to me this week Quiz 3 is This Friday
Brian Wecht, the TA, is away this week. I will substitute for his office hours (in my office 3314 Mayer Hall, discussion and PS session. Pl. give all regrade requests to me this week Quiz 3 is This Friday
ME 476 Solar Energy UNIT TWO THERMAL RADIATION
 ME 476 Solar Energy UNIT TWO THERMAL RADIATION Unit Outline 2 Electromagnetic radiation Thermal radiation Blackbody radiation Radiation emitted from a real surface Irradiance Kirchhoff s Law Diffuse and
ME 476 Solar Energy UNIT TWO THERMAL RADIATION Unit Outline 2 Electromagnetic radiation Thermal radiation Blackbody radiation Radiation emitted from a real surface Irradiance Kirchhoff s Law Diffuse and
STSF2223 Quantum Mechanics I
 STSF2223 Quantum Mechanics I What is quantum mechanics? Why study quantum mechanics? How does quantum mechanics get started? What is the relation between quantum physics with classical physics? Where is
STSF2223 Quantum Mechanics I What is quantum mechanics? Why study quantum mechanics? How does quantum mechanics get started? What is the relation between quantum physics with classical physics? Where is
Radiation Heat Transfer. Introduction. Blackbody Radiation. Definitions ,
 Radiation Heat Transfer Reading Problems 5-5-7 5-27, 5-33, 5-50, 5-57, 5-77, 5-79, 5-96, 5-07, 5-08 Introduction A narrower band inside the thermal radiation spectrum is denoted as the visible spectrum,
Radiation Heat Transfer Reading Problems 5-5-7 5-27, 5-33, 5-50, 5-57, 5-77, 5-79, 5-96, 5-07, 5-08 Introduction A narrower band inside the thermal radiation spectrum is denoted as the visible spectrum,
Chapter 7: Quantum Statistics
 Part II: Applications SDSMT, Physics 2014 Fall 1 Introduction Photons, E.M. Radiation 2 Blackbody Radiation The Ultraviolet Catastrophe 3 Thermal Quantities of Photon System Total Energy Entropy 4 Radiation
Part II: Applications SDSMT, Physics 2014 Fall 1 Introduction Photons, E.M. Radiation 2 Blackbody Radiation The Ultraviolet Catastrophe 3 Thermal Quantities of Photon System Total Energy Entropy 4 Radiation
Modern Physics Laboratory MP2 Blackbody Radiation
 Purpose MP2 Blackbody Radiation In this experiment, you will investigate the spectrum of the blackbody radiation and its dependence on the temperature of the body. Equipment and components Tungsten light
Purpose MP2 Blackbody Radiation In this experiment, you will investigate the spectrum of the blackbody radiation and its dependence on the temperature of the body. Equipment and components Tungsten light
Explain how Planck resolved the ultraviolet catastrophe in blackbody radiation. Calculate energy of quanta using Planck s equation.
 Objectives Explain how Planck resolved the ultraviolet catastrophe in blackbody radiation. Calculate energy of quanta using Planck s equation. Solve problems involving maximum kinetic energy, work function,
Objectives Explain how Planck resolved the ultraviolet catastrophe in blackbody radiation. Calculate energy of quanta using Planck s equation. Solve problems involving maximum kinetic energy, work function,
Thermal Radiation of Blackbodies Lab Partner 1 & Lab Partner 2 12 May 2011
 Thermal Radiation of Blackbodies Lab Partner 1 & Lab Partner 2 12 May 2011 We report on experiments investigating the thermal radiation from a blackbody. By finding the electromagnetic spectra emitted
Thermal Radiation of Blackbodies Lab Partner 1 & Lab Partner 2 12 May 2011 We report on experiments investigating the thermal radiation from a blackbody. By finding the electromagnetic spectra emitted
CHAPTER 3 The Experimental Basis of Quantum
 CHAPTER 3 The Experimental Basis of Quantum 3.1 Discovery of the X Ray and the Electron 3.2 Determination of Electron Charge 3.3 Line Spectra 3.4 Quantization 3.5 Blackbody Radiation 3.6 Photoelectric
CHAPTER 3 The Experimental Basis of Quantum 3.1 Discovery of the X Ray and the Electron 3.2 Determination of Electron Charge 3.3 Line Spectra 3.4 Quantization 3.5 Blackbody Radiation 3.6 Photoelectric
With certain caveats (described later) an object absorbs as effectively as it emits
 Figure 1: A blackbody defined by a cavity where emission and absorption are in equilibrium so as to maintain a constant temperature Blackbody radiation The basic principles of thermal emission are as follows:
Figure 1: A blackbody defined by a cavity where emission and absorption are in equilibrium so as to maintain a constant temperature Blackbody radiation The basic principles of thermal emission are as follows:
B.Tech. First Semester Examination Physics-1 (PHY-101F)
 B.Tech. First Semester Examination Physics-1 (PHY-101F) Note : Attempt FIVE questions in all taking least two questions from each Part. All questions carry equal marks Part-A Q. 1. (a) What are Newton's
B.Tech. First Semester Examination Physics-1 (PHY-101F) Note : Attempt FIVE questions in all taking least two questions from each Part. All questions carry equal marks Part-A Q. 1. (a) What are Newton's
Chapter 7: Quantum Statistics
 Part II: Applications SDSMT, Physics 2013 Fall 1 Introduction Photons, E.M. Radiation 2 Blackbody Radiation The Ultraviolet Catastrophe 3 Thermal Quantities of Photon System Total Energy Entropy 4 Radiation
Part II: Applications SDSMT, Physics 2013 Fall 1 Introduction Photons, E.M. Radiation 2 Blackbody Radiation The Ultraviolet Catastrophe 3 Thermal Quantities of Photon System Total Energy Entropy 4 Radiation
Experimental Basis for QM Ch3
 Experimental Basis for QM Ch3 This chapter describes the early evidence for quantization including Blackbody radiation Photoelectric effect Compton scattering X-rays and their spectra We ll see how early
Experimental Basis for QM Ch3 This chapter describes the early evidence for quantization including Blackbody radiation Photoelectric effect Compton scattering X-rays and their spectra We ll see how early
Princeton University, Physics 311/312 Blackbody Radiation, Page 1 BLACKBODY RADIATION
 Princeton University, Physics 311/312 Blackbody Radiation, Page 1 BLACKBODY RADIATION Introduction A black body is an object that absorbs all radiation falling on it. According to Kirchoff s Laws, such
Princeton University, Physics 311/312 Blackbody Radiation, Page 1 BLACKBODY RADIATION Introduction A black body is an object that absorbs all radiation falling on it. According to Kirchoff s Laws, such
Black Body Radiation
 Black Body Radiation Introduction: All objects are able to absorb electromagnetic waves, but those with temperature above absolute zero radiate as well. When incident electromagnetic radiation strikes
Black Body Radiation Introduction: All objects are able to absorb electromagnetic waves, but those with temperature above absolute zero radiate as well. When incident electromagnetic radiation strikes
Physics 1C. Lecture 27A
 Physics 1C Lecture 27A "Any other situation in quantum mechanics, it turns out, can always be explained by saying, You remember the experiment with the two holes? It s the same thing. " --Richard Feynman
Physics 1C Lecture 27A "Any other situation in quantum mechanics, it turns out, can always be explained by saying, You remember the experiment with the two holes? It s the same thing. " --Richard Feynman
Fundamentals of Blackbody Radiation
 Fundamentals of Blackbody Radiation Prof. Elias N. Glytsis Nov. 08, 206 School of Electrical & Computer Engineering National Technical Univerity of Athens This page was intentionally left blank... BLACKBODY
Fundamentals of Blackbody Radiation Prof. Elias N. Glytsis Nov. 08, 206 School of Electrical & Computer Engineering National Technical Univerity of Athens This page was intentionally left blank... BLACKBODY
Chapter 3 Energy Balance and Temperature. Astro 9601
 Chapter 3 Energy Balance and Temperature Astro 9601 1 Topics to be covered Energy Balance and Temperature (3.1) - All Conduction (3..1), Radiation (3.. and 3...1) Convection (3..3), Hydrostatic Equilibrium
Chapter 3 Energy Balance and Temperature Astro 9601 1 Topics to be covered Energy Balance and Temperature (3.1) - All Conduction (3..1), Radiation (3.. and 3...1) Convection (3..3), Hydrostatic Equilibrium
Physics 2D Lecture Slides Jan 27. Vivek Sharma UCSD Physics
 Physics 2D Lecture Slides Jan 27 Vivek Sharma UCSD Physics Ch 2 : Quantum Theory Of Light What is the nature of light? When it propagates? When it interacts with Matter? What is Nature of Matter? When
Physics 2D Lecture Slides Jan 27 Vivek Sharma UCSD Physics Ch 2 : Quantum Theory Of Light What is the nature of light? When it propagates? When it interacts with Matter? What is Nature of Matter? When
Blackbody Radiation. Rayleigh-Jeans law was an attempt to explain blackbody radiation based on classical ideas:
 Blackbody Radiation A Blackbody is an ideal system that absorbs all radiation incident on it. Emission of radiation by a blackbody is independent of the properties of its wall, but depends only on its
Blackbody Radiation A Blackbody is an ideal system that absorbs all radiation incident on it. Emission of radiation by a blackbody is independent of the properties of its wall, but depends only on its
Lecture 8. > Blackbody Radiation. > Photoelectric Effect
 Lecture 8 > Blackbody Radiation > Photoelectric Effect *Beiser, Mahajan & Choudhury, Concepts of Modern Physics 7/e French, Special Relativity *Nolan, Fundamentals of Modern Physics 1/e Serway, Moses &
Lecture 8 > Blackbody Radiation > Photoelectric Effect *Beiser, Mahajan & Choudhury, Concepts of Modern Physics 7/e French, Special Relativity *Nolan, Fundamentals of Modern Physics 1/e Serway, Moses &
Early Quantum Theory and Models of the Atom
 Early Quantum Theory and Models of the Atom Electron Discharge tube (circa 1900 s) There is something ( cathode rays ) which is emitted by the cathode and causes glowing Unlike light, these rays are deflected
Early Quantum Theory and Models of the Atom Electron Discharge tube (circa 1900 s) There is something ( cathode rays ) which is emitted by the cathode and causes glowing Unlike light, these rays are deflected
Mechanisms of heat transfer
 Lecture 4 Mechanisms of heat transfer Pre-reading: 17.7 Review Heat can be transferred from one object to another due to a temperature difference. The properties of many objects change with temperature:
Lecture 4 Mechanisms of heat transfer Pre-reading: 17.7 Review Heat can be transferred from one object to another due to a temperature difference. The properties of many objects change with temperature:
According to the law of Kirchhoff the ratio of the radiating power to. black-body. The human skin has its place among other radiators as regard
 THE RADIATION OF HEAT FROM THE HUMAN BODY III. THE HUMAN SKIN AS A BLACK-BODY RADIATOR BY JAMES D. HARDY (From The Russell Sage Institute of Pathology, in Affiliation nith the New York Hospital, New York
THE RADIATION OF HEAT FROM THE HUMAN BODY III. THE HUMAN SKIN AS A BLACK-BODY RADIATOR BY JAMES D. HARDY (From The Russell Sage Institute of Pathology, in Affiliation nith the New York Hospital, New York
Chapter 3 Energy Balance and Temperature. Topics to be covered
 Chapter 3 Energy Balance and Temperature Astro 9601 1 Topics to be covered Energy Balance and Temperature (3.1) - All Conduction (3..1), Radiation (3.. and31) 3...1) Convection (3..3), Hydrostatic Equilibrium
Chapter 3 Energy Balance and Temperature Astro 9601 1 Topics to be covered Energy Balance and Temperature (3.1) - All Conduction (3..1), Radiation (3.. and31) 3...1) Convection (3..3), Hydrostatic Equilibrium
Chapter One. The Old Quantum Theory. 1-1 Why Quantum Mechanics.
 Chapter One The Old Quantum Theory 1-1 Why Quantum Mechanics. The birth of quantum mechanics can be dated to 1925, when physicists such as Werner Heisenberg and Erwin Schrödinger invented mathematical
Chapter One The Old Quantum Theory 1-1 Why Quantum Mechanics. The birth of quantum mechanics can be dated to 1925, when physicists such as Werner Heisenberg and Erwin Schrödinger invented mathematical
ATMO/OPTI 656b Spring 2009
 Nomenclature and Definition of Radiation Quantities The various Radiation Quantities are defined in Table 2-1. Keeping them straight is difficult and the meanings may vary from textbook to textbook. I
Nomenclature and Definition of Radiation Quantities The various Radiation Quantities are defined in Table 2-1. Keeping them straight is difficult and the meanings may vary from textbook to textbook. I
Physics Lecture 6
 Physics 3313 - Lecture 6 Monday February 8, 2010 Dr. Andrew Brandt 1. HW1 Due today HW2 weds 2/10 2. Electron+X-rays 3. Black body radiation 4. Compton Effect 5. Pair Production 2/8/10 3313 Andrew Brandt
Physics 3313 - Lecture 6 Monday February 8, 2010 Dr. Andrew Brandt 1. HW1 Due today HW2 weds 2/10 2. Electron+X-rays 3. Black body radiation 4. Compton Effect 5. Pair Production 2/8/10 3313 Andrew Brandt
AT622 Section 3 Basic Laws
 AT6 Section 3 Basic Laws There are three stages in the life of a photon that interest us: first it is created, then it propagates through space, and finally it can be destroyed. The creation and destruction
AT6 Section 3 Basic Laws There are three stages in the life of a photon that interest us: first it is created, then it propagates through space, and finally it can be destroyed. The creation and destruction
Advanced Heat and Mass Transfer by Amir Faghri, Yuwen Zhang, and John R. Howell
 Advanced Heat and Mass Transfer by Amir Faghri, Yuwen Zhang, and John R. Howell 9.2 The Blackbody as the Ideal Radiator A material that absorbs 100 percent of the energy incident on it from all directions
Advanced Heat and Mass Transfer by Amir Faghri, Yuwen Zhang, and John R. Howell 9.2 The Blackbody as the Ideal Radiator A material that absorbs 100 percent of the energy incident on it from all directions
Physics 2D Lecture Slides Lecture 9: Jan 21st 2004
 Quiz 1 Blue Books will be returned to Student Affairs office (Ground Floor, Urey Hall Extension) this Wednesday by 10am (usually you can pick it up Tuesday 2pm) Regrade Requests must get to TA within 7
Quiz 1 Blue Books will be returned to Student Affairs office (Ground Floor, Urey Hall Extension) this Wednesday by 10am (usually you can pick it up Tuesday 2pm) Regrade Requests must get to TA within 7
Electromagnetic Radiation. Physical Principles of Remote Sensing
 Electromagnetic Radiation Physical Principles of Remote Sensing Outline for 4/3/2003 Properties of electromagnetic radiation The electromagnetic spectrum Spectral emissivity Radiant temperature vs. kinematic
Electromagnetic Radiation Physical Principles of Remote Sensing Outline for 4/3/2003 Properties of electromagnetic radiation The electromagnetic spectrum Spectral emissivity Radiant temperature vs. kinematic
(Total 1 mark) IB Questionbank Physics 1
 1. A transverse wave travels from left to right. The diagram below shows how, at a particular instant of time, the displacement of particles in the medium varies with position. Which arrow represents the
1. A transverse wave travels from left to right. The diagram below shows how, at a particular instant of time, the displacement of particles in the medium varies with position. Which arrow represents the
Chapter 27. Quantum Physics
 Chapter 27 Quantum Physics Need for Quantum Physics Problems remained from classical mechanics that relativity didn t explain Blackbody Radiation The electromagnetic radiation emitted by a heated object
Chapter 27 Quantum Physics Need for Quantum Physics Problems remained from classical mechanics that relativity didn t explain Blackbody Radiation The electromagnetic radiation emitted by a heated object
THERMODYNAMICS METHODS OF HEAT TRANSFER RADIATION
 VISUAL PHYSICS ONLINE THERMODYNAMICS METHODS OF HEAT TRANSFER RADIATION Radiation is the energy transferred by electromagnetic waves mainly infrared (IR), visible and ultraviolet (UV). All materials radiate
VISUAL PHYSICS ONLINE THERMODYNAMICS METHODS OF HEAT TRANSFER RADIATION Radiation is the energy transferred by electromagnetic waves mainly infrared (IR), visible and ultraviolet (UV). All materials radiate
The first quantitative conjecture based on experimental observation of hole radiation was:
 Black Body Radiation Heated Bodies Radiate We shall now turn to another puzzle confronting physicists at the turn of the century (1900): just how do heated bodies radiate? There was a general understanding
Black Body Radiation Heated Bodies Radiate We shall now turn to another puzzle confronting physicists at the turn of the century (1900): just how do heated bodies radiate? There was a general understanding
Fig. 8.1 illustrates the three measurements. air medium A. ray 1. air medium A. ray 2. air medium A. ray 3. Fig For Examiner s Use
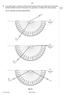 9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
Introduction to Modern Physics NE 131 Physics for Nanotechnology Engineering
 Introduction to Modern Physics NE 131 Physics for Nanotechnology Engineering Dr. Jamie Sanchez-Fortún Stoker Department of Physics, University of Waterloo Fall 2005 1 Introduction to Modern Physics 1.1
Introduction to Modern Physics NE 131 Physics for Nanotechnology Engineering Dr. Jamie Sanchez-Fortún Stoker Department of Physics, University of Waterloo Fall 2005 1 Introduction to Modern Physics 1.1
1. Radiative Transfer. 2. Spectrum of Radiation. 3. Definitions
 1. Radiative Transfer Virtually all the exchanges of energy between the earth-atmosphere system and the rest of the universe take place by radiative transfer. The earth and its atmosphere are constantly
1. Radiative Transfer Virtually all the exchanges of energy between the earth-atmosphere system and the rest of the universe take place by radiative transfer. The earth and its atmosphere are constantly
Semiconductor Physics and Devices
 Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
BLACKB ODY RADIATION LAWS. Stefan-Boltzmann. Blackbody E = 1. T is Temperature (Kelvin) R(T) = EaT. R(T) = at4. a = 5.67x10.
 LAWS a = 5.67x10 8 (m2 1() T is Temperature (Kelvin) R(T) = at4 Blackbody E = 1 R(T) = EaT 4 Stefan-Boltzmann BLACKB ODY RADIATION Wien Displacement AmaxT = b b = 2.898x10 3m K CONSIDER TWO OPAQUE QBJECTS
LAWS a = 5.67x10 8 (m2 1() T is Temperature (Kelvin) R(T) = at4 Blackbody E = 1 R(T) = EaT 4 Stefan-Boltzmann BLACKB ODY RADIATION Wien Displacement AmaxT = b b = 2.898x10 3m K CONSIDER TWO OPAQUE QBJECTS
Sensors - April A Certified-Emissivity Blackbody for Calibrating Infrared Thermometers
 Page 1 of 9 www.sensorsmag.com APRIL 2002 SENSOR TECHNOLOGY AND DESIGN A Certified-Emissivity Blackbody for Calibrating Infrared Thermometers As a practical matter, most commercial infrared thermometers
Page 1 of 9 www.sensorsmag.com APRIL 2002 SENSOR TECHNOLOGY AND DESIGN A Certified-Emissivity Blackbody for Calibrating Infrared Thermometers As a practical matter, most commercial infrared thermometers
Lecture 12. Measurements in Astronomy. Using Light. ASTR 111 Section 002. In astronomy, we need to make remote and indirect measurements
 Lecture 12 ASTR 111 Section 002 Measurements in Astronomy In astronomy, we need to make remote and indirect measurements Think of an example of a remote and indirect measurement from everyday life Using
Lecture 12 ASTR 111 Section 002 Measurements in Astronomy In astronomy, we need to make remote and indirect measurements Think of an example of a remote and indirect measurement from everyday life Using
Physics 2D Lecture Slides Lecture 11: Jan 27 th 2004
 Brian Wecht, the TA, is away this week. I will substitute for his office hours (in my office 3314 Mayer Hall, discussion and PS session. Pl. give all regrade requests to me this week (only) Quiz 3 is This
Brian Wecht, the TA, is away this week. I will substitute for his office hours (in my office 3314 Mayer Hall, discussion and PS session. Pl. give all regrade requests to me this week (only) Quiz 3 is This
Physics 576 Stellar Astrophysics Prof. James Buckley. Lecture 2 Radiation
 Physics 576 Stellar Astrophysics Prof. James Buckley Lecture 2 Radiation Reading/Homework Assignment Read chapter 1, sections 1.1, 1.2, 1.5 Homework will be assigned on Thursday. Radiation Radiation A
Physics 576 Stellar Astrophysics Prof. James Buckley Lecture 2 Radiation Reading/Homework Assignment Read chapter 1, sections 1.1, 1.2, 1.5 Homework will be assigned on Thursday. Radiation Radiation A
On the Law of Distribution of Energy in the Normal Spectrum
 On the Law of Distribution of Energy in the Normal Spectrum Max Planck Annalen der Physik vol.4, p.553 ff (1901) The recent spectral measurements made by O. Lummer and E. Pringsheim[1], and even more notable
On the Law of Distribution of Energy in the Normal Spectrum Max Planck Annalen der Physik vol.4, p.553 ff (1901) The recent spectral measurements made by O. Lummer and E. Pringsheim[1], and even more notable
1. How much heat was needed to raise the bullet to its final temperature?
 Name: Date: Use the following to answer question 1: A 0.0500-kg lead bullet of volume 5.00 10 6 m 3 at 20.0 C hits a block that is made of an ideal thermal insulator and comes to rest at its center. At
Name: Date: Use the following to answer question 1: A 0.0500-kg lead bullet of volume 5.00 10 6 m 3 at 20.0 C hits a block that is made of an ideal thermal insulator and comes to rest at its center. At
QM all started with - - The Spectrum of Blackbody Radiation
 QM all started with - - The Spectrum of Blackbody Radiation Thermal Radiation: Any object, not at zero temperature, emits electromagnetic called thermal. When we measure the intensity of a real object,
QM all started with - - The Spectrum of Blackbody Radiation Thermal Radiation: Any object, not at zero temperature, emits electromagnetic called thermal. When we measure the intensity of a real object,
LIGHT. Question. Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light.
 LIGHT Question Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light. What kind of information can we get from light? 1 Light
LIGHT Question Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light. What kind of information can we get from light? 1 Light
Elements of Statistical Mechanics
 Dr. Y. Aparna, Associate Prof., Dept. of Physics, JNTU College of ngineering, JNTU - H, lements of Statistical Mechanics Question: Discuss about principles of Maxwell-Boltzmann Statistics? Answer: Maxwell
Dr. Y. Aparna, Associate Prof., Dept. of Physics, JNTU College of ngineering, JNTU - H, lements of Statistical Mechanics Question: Discuss about principles of Maxwell-Boltzmann Statistics? Answer: Maxwell

 Infrared thermography In microwave radiometry hν
Infrared thermography In microwave radiometry hν