Thermal energy 7 TH GRADE SCIENCE
|
|
|
- Jack Jefferson
- 6 years ago
- Views:
Transcription
1 Thermal energy 7 TH GRADE SCIENCE
2 Temperature There s more to temperature than the idea of hot and cold. Remember that all matter is made up of tiny particles that are constantly moving even in solid objects. Because they are moving, these particles have kinetic energy.
3 Temperature Temperature is a measurement of the average kinetic energy of the particles in matter. As something is heated, its particles will move faster. This means that their kinetic energy is higher and therefore their temperature will go up. Cold Hot
4 Temperature scales There are three scales for temperature. Fahrenheit Celsius Kelvin The difference between these are how much thermal energy each unit measures and where they start.
5 Daniel Gabriel Fahrenheit Temperature scales Fahrenheit has the smallest unit. 0 is the coldest the scientist who invented it could get salt water and 100 is about human body temperature.
6 Anders Celsius Temperature scales Celsius has larger units, almost twice the size of Fahrenheit. 0 is the freezing point of water and 100 the boiling point of water.
7 Temperature scales Kelvin has units the same size as Celsius. 0 Kelvin is the temperature known as absolute zero. William Thomson, Lord Kelvin
8 Absolute zero As the temperature of matter decreases the speed of its particles decrease. At some point the particles will stop moving. Absolute zero is the lowest possible temperature. Where particles stop moving and matter contains no thermal energy.
9
10 Thermal energy Thermal energy is the total energy of the particles in an object and depends on the temperature of the object.
11 Thermal energy constantly wants to even out with everything around it. Heat is the movement of thermal energy from a warmer object to a cooler object.
12 Specific heat Not every object heats up or cools down at the same rate. Different chemicals require different amounts of heat to change temperature. Specific heat is the amount of energy needed to raise the temperature of 1 kilogram of something 1 kelvin.
13 Specific heat Something with a high specific heat can absorb a large amount of thermal energy without changing its temperature very much.
14 Transferring heat Heat always flows from warmer objects to colder ones. As the temperature of one object increases the temperature of the other will decrease. Some materials transfer thermal energy better than others.
15 Conductors A conductor transfers thermal energy very well. Metals are good conductors. Good conductors feel cool when you touch them because they transfer heat away from your skin.
16 Insulators An insulator does NOT transfer thermal energy very well. Wood, wool, paper, and air are good insulators. Insulation prevents thermal energy from moving from one place to another very well.
17 States of matter A state is a form of matter. Matter can exist in three different states. Solid, liquid, or gas.
18 Solids An object s state of matter depends on how much thermal energy it has. Solids the particles of a solid cannot move out of their positions, they can only vibrate back and forth.
19 Liquids Liquids the particles in a liquid are close together, but they are not held in place like in solids so they can move around more. Therefore liquids don t have a shape.
20 Gasses Gasses the particles in a gas are moving so fast that they don t stay close together, they expand apart to fill the space they are in. They don t have a fixed shape or volume.
21 States of matter
22 Changes of state The change from one state of matter to another is called a change of state. A change of state happens when thermal energy is absorbed or released.
23
24 Changes of state Melting when a solid turns into a liquid. Freezing when a liquid turns into a solid. Evaporation/boiling When a liquid turns into a gas.
25 Changes of state Condensation when a gas turns into a liquid. Sublimation when a solid turns into a gas without first becoming a liquid.
26 Thermal expansion Thermal expansion is when matter spreads out and expands when it is heated.
27
28
29
30
31
32
33
Temperature and Heat. Chapter 10. Table of Contents. Chapter 10. Chapter 10. Bellringer. Objectives. Chapter 10. Chapter 10
 Heat and Heat Technology Table of Contents Temperature and Heat Section 3 Matter and Heat Bellringer Objectives The temperature of boiling water is 100 on the Celsius scale and 212 on the Fahrenheit scale.
Heat and Heat Technology Table of Contents Temperature and Heat Section 3 Matter and Heat Bellringer Objectives The temperature of boiling water is 100 on the Celsius scale and 212 on the Fahrenheit scale.
* Defining Temperature * Temperature is proportional to the kinetic energy of atoms and molecules. * Temperature * Internal energy
 * Defining Temperature * We associate temperature with how hot or cold an object feels. * Our sense of touch serves as a qualitative indicator of temperature. * Energy must be either added or removed from
* Defining Temperature * We associate temperature with how hot or cold an object feels. * Our sense of touch serves as a qualitative indicator of temperature. * Energy must be either added or removed from
Lecture 6. Temperature and Heat 27 September 2018
 Lecture 6. Temperature and Heat 27 September 2018 Wannapong Triampo, Ph.D. Korey Stringer 7-31-01 27 Yrs Old 6 3 335 lbs Eraste Autin 7-25-01 18 Yrs Old 6 2 250 lbs Preston Birdsong 8-13-00 18 Yrs Old
Lecture 6. Temperature and Heat 27 September 2018 Wannapong Triampo, Ph.D. Korey Stringer 7-31-01 27 Yrs Old 6 3 335 lbs Eraste Autin 7-25-01 18 Yrs Old 6 2 250 lbs Preston Birdsong 8-13-00 18 Yrs Old
The Kinetic Theory of Matter. Temperature. Temperature. Temperature. Temperature. Chapter 6 HEAT
 The Kinetic Theory of Matter Hewitt/Lyons/Suchocki/Yeh Conceptual Integrated Science Chapter 6 HEAT Kinetic Theory of Matter: Matter is made up of tiny particles (atoms or molecules) that are always in
The Kinetic Theory of Matter Hewitt/Lyons/Suchocki/Yeh Conceptual Integrated Science Chapter 6 HEAT Kinetic Theory of Matter: Matter is made up of tiny particles (atoms or molecules) that are always in
Thermal Energy and Heat Notes. Ch. 14
 Thermal Energy and Heat Notes Ch. 14 Temperature When scien
Thermal Energy and Heat Notes Ch. 14 Temperature When scien
Section 1: The Science of Energy¹
 SECTION1: THE SCIENCE OF ENERGY Section 1: The Science of Energy¹ What Is Energy? Energy is the ability to do work or the ability to make a change. Everything that happens in the world involves the exchange
SECTION1: THE SCIENCE OF ENERGY Section 1: The Science of Energy¹ What Is Energy? Energy is the ability to do work or the ability to make a change. Everything that happens in the world involves the exchange
Chapter 21: Temperature, Heat and Expansion
 Chapter 21: Temperature, Heat and Expansion All matter solid, liquid and gas is made of atoms or molecules, which are continually jiggling. As this jiggling is a movement, all these particles must have
Chapter 21: Temperature, Heat and Expansion All matter solid, liquid and gas is made of atoms or molecules, which are continually jiggling. As this jiggling is a movement, all these particles must have
Heat and Temperature
 Heat and Temperature Temperature What does temperature have to do with energy? What three temperature scales are commonly used? What makes things feel hot or cold? Intro: Discussion A person from Seattle
Heat and Temperature Temperature What does temperature have to do with energy? What three temperature scales are commonly used? What makes things feel hot or cold? Intro: Discussion A person from Seattle
Heat and temperature are different. Heat is a type of energy. Heat is the total energy of all the particles in an object.
 Heat & Temperature Heat vs. Temperature Heat and temperature are different. Heat is a type of energy. Heat is the total energy of all the particles in an object. Temperature is the average energy of each
Heat & Temperature Heat vs. Temperature Heat and temperature are different. Heat is a type of energy. Heat is the total energy of all the particles in an object. Temperature is the average energy of each
Chapter: Heat and States
 Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
Table of Contents Chapter: Heat and States of Matter Section 1: Temperature and Thermal Energy Section 2: States of Matter Section 3: Transferring Thermal Energy Section 4: Using Thermal Energy 1 Temperature
What does temperature have to do with energy? What three temperature scales are commonly used? What makes things feel hot or cold?
 Heat and Temperature Section 1: Temperature What does temperature have to do with energy? What three temperature scales are commonly used? What makes things feel hot or cold? 1 Intro: Discussion A person
Heat and Temperature Section 1: Temperature What does temperature have to do with energy? What three temperature scales are commonly used? What makes things feel hot or cold? 1 Intro: Discussion A person
Heating and Cooling Explained By The Particle Model. Notes: Part 2/4
 Heating and Cooling Explained By The Particle Model Notes: Part 2/4 Particles are the building blocks of all things. What are Particles? Some people call them molecules. Particles are NOT alive. How many
Heating and Cooling Explained By The Particle Model Notes: Part 2/4 Particles are the building blocks of all things. What are Particles? Some people call them molecules. Particles are NOT alive. How many
Kinetic Theory of Matter. Matter & Energy
 Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
Kinetic Theory of Matter Matter & Energy 1 Kinetic Theory of Matter All matter is made up of atoms and molecules that act as tiny particles. 2 Kinetic Theory of Matter These tiny particles are always in
Term Info Picture. Anything that has mass and takes up space; everything is made of matter.
 Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Characteristics, Changes, and States of Matter S8P1. Obtain, evaluate, and communicate information about the structure and properties of matter. B. Develop and use models to describe the movement of particles
Thermal Energy. Thermal Energy is the TRANSFER of kinetic energy between two objects that are at different temperatures.
 Thermal Energy Thermal Energy is the TRANSFER of kinetic energy between two objects that are at different temperatures. And remember: heat will always transfer from a warm object to a cold object. HEAT
Thermal Energy Thermal Energy is the TRANSFER of kinetic energy between two objects that are at different temperatures. And remember: heat will always transfer from a warm object to a cold object. HEAT
Science In Action 7 Heat and Temperature Section Quiz
 Section 2 Heat affects Matter in different ways 2.1 States of Matter and The Particle Model 1. Water has a distinct characteristic that sets it apart from other liquids on Earth. Water expands when it
Section 2 Heat affects Matter in different ways 2.1 States of Matter and The Particle Model 1. Water has a distinct characteristic that sets it apart from other liquids on Earth. Water expands when it
What Is Air Temperature?
 2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
2.2 Read What Is Air Temperature? In Learning Set 1, you used a thermometer to measure air temperature. But what exactly was the thermometer measuring? What is different about cold air and warm air that
2,000-gram mass of water compared to a 1,000-gram mass.
 11.2 Heat To change the temperature, you usually need to add or subtract energy. For example, when it s cold outside, you turn up the heat in your house or apartment and the temperature goes up. You know
11.2 Heat To change the temperature, you usually need to add or subtract energy. For example, when it s cold outside, you turn up the heat in your house or apartment and the temperature goes up. You know
8th Grade. Thermal Energy Study Guide.
 1 8th Grade Thermal Energy Study Guide 2015 10 09 www.njctl.org 2 Thermal Energy Study Guide www.njctl.org 3 Part 1 Define the following terms and/or concepts 4 1 Temperature 5 2 Kinetic Energy 6 3 Thermal
1 8th Grade Thermal Energy Study Guide 2015 10 09 www.njctl.org 2 Thermal Energy Study Guide www.njctl.org 3 Part 1 Define the following terms and/or concepts 4 1 Temperature 5 2 Kinetic Energy 6 3 Thermal
Slide 1 / 67. Slide 2 / 67. 8th Grade. Thermal Energy Study Guide Slide 3 / 67. Thermal Energy. Study Guide.
 Slide 1 / 67 Slide 2 / 67 8th Grade Thermal Energy Study Guide 2015-10-09 www.njctl.org Slide 3 / 67 Thermal Energy Study Guide www.njctl.org Slide 4 / 67 Part 1 Define the following terms and/or concepts
Slide 1 / 67 Slide 2 / 67 8th Grade Thermal Energy Study Guide 2015-10-09 www.njctl.org Slide 3 / 67 Thermal Energy Study Guide www.njctl.org Slide 4 / 67 Part 1 Define the following terms and/or concepts
Heat & Temperature. What are heat & temperature and how do they relate?
 Heat & Temperature What are heat & temperature and how do they relate? SPS7. Students will relate transformations and flow of energy within a system. a. Identify energy transformations within a system
Heat & Temperature What are heat & temperature and how do they relate? SPS7. Students will relate transformations and flow of energy within a system. a. Identify energy transformations within a system
Study Guide for Chapters 2, 3, and 10
 Study Guide for Chapters 2, 3, and 10 1. What is matter? Where can it be found? Anything that has mass and takes up space. 2. What units are used to measure volume? Liters and meters cubed 3. How would
Study Guide for Chapters 2, 3, and 10 1. What is matter? Where can it be found? Anything that has mass and takes up space. 2. What units are used to measure volume? Liters and meters cubed 3. How would
matter/index.html
 http://www.colorado.edu/physics/2000/index.pl http://www.harcourtschool.com/activity/states_of_ matter/index.html Thermal Energy Ch 6-1 Temperature and Heat Objectives Explain the kinetic theory of matter
http://www.colorado.edu/physics/2000/index.pl http://www.harcourtschool.com/activity/states_of_ matter/index.html Thermal Energy Ch 6-1 Temperature and Heat Objectives Explain the kinetic theory of matter
Thermal energy. Thermal energy is the internal energy of a substance. I.e. Thermal energy is the kinetic energy of atoms and molecules.
 Thermal energy Thermal energy is the internal energy of a substance. I.e. Thermal energy is the kinetic energy of atoms and molecules. Heat is the transfer of thermal energy between substances. Until the
Thermal energy Thermal energy is the internal energy of a substance. I.e. Thermal energy is the kinetic energy of atoms and molecules. Heat is the transfer of thermal energy between substances. Until the
Temperature and Its Measurement
 Temperature and Its Measurement When the physical properties are no longer changing, the objects are said to be in thermal equilibrium. Two or more objects in thermal equilibrium have the same temperature.
Temperature and Its Measurement When the physical properties are no longer changing, the objects are said to be in thermal equilibrium. Two or more objects in thermal equilibrium have the same temperature.
Chapter 3: Matter and Energy
 Chapter 3: Matter and Energy Convert between Fahrenheit, Celsius, and Kelvin temperature scales. Relate energy, temperature change, and heat capacity. The atoms and molecules that compose matter are in
Chapter 3: Matter and Energy Convert between Fahrenheit, Celsius, and Kelvin temperature scales. Relate energy, temperature change, and heat capacity. The atoms and molecules that compose matter are in
Solids, Liquids, and Gases. Chapter 14
 Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
Solids, Liquids, and Gases Chapter 14 Matter & Thermal Energy Matter can exist as a solid, a liquid, a gas or a plasma. The Molecular Kinetic Theory of Matter explains their differences and how they can
Properties of Matter. I like cheese
 Properties of Matter I like cheese Matter All the stuff in the universe that is not energy (and also, not Dark Matter or Dark Energy if you don t know what they are don t worry ). The stuff that makes
Properties of Matter I like cheese Matter All the stuff in the universe that is not energy (and also, not Dark Matter or Dark Energy if you don t know what they are don t worry ). The stuff that makes
Unit 4 - Energy & Heat SOL PS.6,7
 Unit 4 - Energy & Heat SOL PS.6,7 Bill Nye - Energy I. PS.6a - Potential & Kinetic Energy A. Energy is the ability to do work or cause change. B. Energy is either Potential or Kinetic. a. Potential Energy
Unit 4 - Energy & Heat SOL PS.6,7 Bill Nye - Energy I. PS.6a - Potential & Kinetic Energy A. Energy is the ability to do work or cause change. B. Energy is either Potential or Kinetic. a. Potential Energy
Topic 2: Heat Affects Matter in Different Ways
 Topic 2: Heat Affects Matter in Different Ways 1 2.1 States of Matter and the Particle Model of Matter A. States of 1. Matter is made up of tiny particles and exist in 3 states:, and. 2. Matter can change
Topic 2: Heat Affects Matter in Different Ways 1 2.1 States of Matter and the Particle Model of Matter A. States of 1. Matter is made up of tiny particles and exist in 3 states:, and. 2. Matter can change
CIE Physics IGCSE. Topic 2: Thermal Physics
 CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
CIE Physics IGCSE Topic 2: Thermal Physics Summary Notes Simple kinetic molecular model of matter Molecular model Solids Molecules close together in regular pattern Strong intermolecular forces of attraction
Temperature. Temperature Scales. Temperature (cont d) CHAPTER 14 Heat and Temperature
 Temperature CHAPTER 14 Heat and Temperature The temperature of a substance is proportional to the average kinetic energy of the substance s particles. As the average kinetic energy of the particles in
Temperature CHAPTER 14 Heat and Temperature The temperature of a substance is proportional to the average kinetic energy of the substance s particles. As the average kinetic energy of the particles in
Temperature Energy and Heat
 CHAPTER 3 Temperature Energy and Heat 3.1 Temperature What is temperature? Why is temperature important in chemistry? How is energy related to temperature? 2 3.1 Temperature Milk fat particles are being
CHAPTER 3 Temperature Energy and Heat 3.1 Temperature What is temperature? Why is temperature important in chemistry? How is energy related to temperature? 2 3.1 Temperature Milk fat particles are being
Before Statement After
 Thermal Energy Thermal Energy, Temperature, and Heat What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
Thermal Energy Thermal Energy, Temperature, and Heat What do you think? Read the two statements below and decide whether you agree or disagree with them. Place an A in the Before column if you agree with
kinetic molecular theory thermal energy.
 Thermal Physics 1 Thermal Energy The kinetic molecular theory is based on the assumption that matter is made up of tiny particles that are always in motion. In a hot object the particles are moving faster
Thermal Physics 1 Thermal Energy The kinetic molecular theory is based on the assumption that matter is made up of tiny particles that are always in motion. In a hot object the particles are moving faster
Matter & Energy. Objectives: properties and structures of the different states of matter.
 Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Matter & Energy Objectives: 1. Use the kinetic theory to describe the properties and structures of the different states of matter. 2. Describe energy transfers involved in changes of state. 3. Describe
Lecture 2: Zero law of thermodynamics
 Lecture 2: Zero law of thermodynamics 1. Thermometers and temperature scales 2. Thermal contact and thermal equilibrium 3. Zeroth law of thermodynamics 1. Thermometers and Temperature scales We often associate
Lecture 2: Zero law of thermodynamics 1. Thermometers and temperature scales 2. Thermal contact and thermal equilibrium 3. Zeroth law of thermodynamics 1. Thermometers and Temperature scales We often associate
Very Dynamic! Energy in the Earth s Atmosphere. How Does it Get Here? All Objects Radiate Energy!
 Energy in the Earth s Atmosphere Unit Essential Question: What are the different features of the atmosphere that characterize our weather. How does the atmosphere influence life and how does life influence
Energy in the Earth s Atmosphere Unit Essential Question: What are the different features of the atmosphere that characterize our weather. How does the atmosphere influence life and how does life influence
Matter, States of Matter, Gas Laws, Phase Changes, and Thermal Energy
 Matter, States of Matter, Gas Laws, Phase Changes, and Thermal Energy Double Jeopardy Jeopardy! Matter Tempera ture Phase Changes Heat Transfer Thermal Energy vs Heat 100 100 100 100 100 200 200 200 200
Matter, States of Matter, Gas Laws, Phase Changes, and Thermal Energy Double Jeopardy Jeopardy! Matter Tempera ture Phase Changes Heat Transfer Thermal Energy vs Heat 100 100 100 100 100 200 200 200 200
Chapter 1 Heating Processes
 Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
Chapter 1 Heating Processes Section 1.1 Heat and temperature Worked example: Try yourself 1.1.1 CALCULATING THE CHANGE IN INTERNAL ENERGY A student places a heating element and a paddle wheel apparatus
Notes for: How Can You Describe Matter, Mixtures, and Solutions? A. Matter is anything that has mass and takes up space
 Notes for: How Can You Describe Matter, Mixtures, and Solutions? I. Properties of Matter A. Matter is anything that has mass and takes up space 1. scientists describe and compare matter based on properties,
Notes for: How Can You Describe Matter, Mixtures, and Solutions? I. Properties of Matter A. Matter is anything that has mass and takes up space 1. scientists describe and compare matter based on properties,
All matter is made of moving particles
 All matter is made of moving particles I. Kinetic Theory of matter all particles in matter are constantly in motion a. Kinetic Energy is motion energy b. Therefore all particles in solids, liquids and
All matter is made of moving particles I. Kinetic Theory of matter all particles in matter are constantly in motion a. Kinetic Energy is motion energy b. Therefore all particles in solids, liquids and
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES
 THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
THE PARTICLE MODEL AND PROPERTIES OF THE GASES, LIQUIDS AND SOLIDS. STATES CHANGES The particle model of a gas A gas has no fixed shape or volume, but always spreads out to fill any container. There are
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Temperature, Thermal Energy, and Heat
 Temperature, Thermal Energy, and Heat Textbook pages 424 435 Section 10.1 Summary Before You Read We often use the terms heat and temperature interchangeably. Do you think they mean the same thing? Explain
Temperature, Thermal Energy, and Heat Textbook pages 424 435 Section 10.1 Summary Before You Read We often use the terms heat and temperature interchangeably. Do you think they mean the same thing? Explain
Preview. Heat Section 1. Section 1 Temperature and Thermal Equilibrium. Section 2 Defining Heat. Section 3 Changes in Temperature and Phase
 Heat Section 1 Preview Section 1 Temperature and Thermal Equilibrium Section 2 Defining Heat Section 3 Changes in Temperature and Phase Heat Section 1 TEKS The student is expected to: 6E describe how the
Heat Section 1 Preview Section 1 Temperature and Thermal Equilibrium Section 2 Defining Heat Section 3 Changes in Temperature and Phase Heat Section 1 TEKS The student is expected to: 6E describe how the
Matter: Properties and Change
 Matter: Properties and Change 6.P.2 Understand the structure, classifications and physical properties of matter. 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element are
Matter: Properties and Change 6.P.2 Understand the structure, classifications and physical properties of matter. 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element are
Recap: Bernoulli s Principle
 Recap: Bernoulli s Principle The sum of pressure plus kinetic energy per unit volume of a flowing fluid is constant. P + ½ρv 2 = constant pressure K.E. per unit volume (ρ = mass vol ) Result: Relates pressure
Recap: Bernoulli s Principle The sum of pressure plus kinetic energy per unit volume of a flowing fluid is constant. P + ½ρv 2 = constant pressure K.E. per unit volume (ρ = mass vol ) Result: Relates pressure
Heat & Temperature. Grade 7 Science - Unit 2 Pgs
 Heat & Temperature Grade 7 Science - Unit 2 Pgs 104-225 Temperature P Temperature is the measure of how much heat is in a substance. P Temperature is measured in degrees Celcius ( C) P It is difficult
Heat & Temperature Grade 7 Science - Unit 2 Pgs 104-225 Temperature P Temperature is the measure of how much heat is in a substance. P Temperature is measured in degrees Celcius ( C) P It is difficult
Atoms and molecules are in motion and have energy
 Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Atoms and molecules are in motion and have energy By now you know that substances are made of atoms and molecules. These atoms and molecules are always in motion and have attractions to each other. When
Conduction is the transfer of heat by the direct contact of particles of matter.
 Matter and Energy Chapter 9 energy flows from a material at a higher temperature to a material at a lower temperature. This process is called heat transfer. How is heat transferred from material to material,
Matter and Energy Chapter 9 energy flows from a material at a higher temperature to a material at a lower temperature. This process is called heat transfer. How is heat transferred from material to material,
OK, you ve probably seen the Bill Nye video and have learned that matter is everything and all stuff. That s right. Everything around you is matter.
 Matter & Chemistry OK, you ve probably seen the Bill Nye video and have learned that matter is everything and all stuff. That s right. Everything around you is matter. In fact, anything that has weight
Matter & Chemistry OK, you ve probably seen the Bill Nye video and have learned that matter is everything and all stuff. That s right. Everything around you is matter. In fact, anything that has weight
Properties of Matter. Heat. Summary
 Properties of Matter Heat Summary Heat is a form of energy that is measured in joules (J). The temperature of an object is a measure of the average kinetic energy of the particles in the object and is
Properties of Matter Heat Summary Heat is a form of energy that is measured in joules (J). The temperature of an object is a measure of the average kinetic energy of the particles in the object and is
Matter: Properties and Change
 Matter: Properties and Change Date: 6.P.2 Understand the structure, classifications and physical properties of matter. 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element
Matter: Properties and Change Date: 6.P.2 Understand the structure, classifications and physical properties of matter. 6.P.2.1 Recognize that all matter is made up of atoms and atoms of the same element
 Since ancient times, people have needed thermal energy (heat) to cook their food and to keep them warm. And since the beginning of time, uncontrolled heat has scorched and spoiled the taste of food and
Since ancient times, people have needed thermal energy (heat) to cook their food and to keep them warm. And since the beginning of time, uncontrolled heat has scorched and spoiled the taste of food and
Preview of Period 4: Transfer of Thermal Energy
 Preview of Period 4: Transfer of Thermal Energy 4.1 Temperature and Thermal Energy How is temperature measured? What temperature scales are used? 4.2 How is Thermal Energy Transferred? How do conduction,
Preview of Period 4: Transfer of Thermal Energy 4.1 Temperature and Thermal Energy How is temperature measured? What temperature scales are used? 4.2 How is Thermal Energy Transferred? How do conduction,
Study Guide Unit 3 Chapter 6 DRAFT
 Study Guide Unit 3 Chapter 6 DRAFT Unit 3 BIG IDEAS Energy can be transformed from one type into another. Energy transformation systems often involve thermal energy losses and are never 100 % efficient.
Study Guide Unit 3 Chapter 6 DRAFT Unit 3 BIG IDEAS Energy can be transformed from one type into another. Energy transformation systems often involve thermal energy losses and are never 100 % efficient.
Temperature, Heat, and Expansion
 Thermodynamics (Based on Chapters 21-24) Temperature, Heat, and Expansion (Ch 21) Warmth is the kinetic energy of atoms and molecules. Temperature (21.1) The measure of how hot and cold things are is temperature.
Thermodynamics (Based on Chapters 21-24) Temperature, Heat, and Expansion (Ch 21) Warmth is the kinetic energy of atoms and molecules. Temperature (21.1) The measure of how hot and cold things are is temperature.
Chapter 14 Heat and Temperature Notes
 Chapter 14 Heat and Temperature Notes Section 1: Temperature The degree of or of an object. Related to the of an object s atoms or molecules What makes something hot? o Particles that make up o They have
Chapter 14 Heat and Temperature Notes Section 1: Temperature The degree of or of an object. Related to the of an object s atoms or molecules What makes something hot? o Particles that make up o They have
What is a change of state? What happens during a change of state? What can happen when a substance loses or gains energy?
 CHAPTER 3 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
CHAPTER 3 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
Weather Unit. Welcome Back! Begin Semester 2 1/20/2010. Seating Changes Final Exam Averages S1 Routine Changes for S2 Review Expectations.
 Welcome Back! Begin Semester 2 Weather Unit I-2 to I-6 Seating Changes Final Exam Averages S1 Routine Changes for S2 Review Expectations Beginning of Class (ChemCatalyst) Chemistry Binders and note-taking
Welcome Back! Begin Semester 2 Weather Unit I-2 to I-6 Seating Changes Final Exam Averages S1 Routine Changes for S2 Review Expectations Beginning of Class (ChemCatalyst) Chemistry Binders and note-taking
How do we get information about heavenly bodies when they are so far??
 In Astronomy the most common unit to measure distances is---- Light Year: It is the distance traveled by light in one year. Speed of light ~ 300,000 kilometers/sec So in one hour lights travels = 300,000
In Astronomy the most common unit to measure distances is---- Light Year: It is the distance traveled by light in one year. Speed of light ~ 300,000 kilometers/sec So in one hour lights travels = 300,000
CHM Solids, Liquids, and Phase Changes (r15) Charles Taylor 1/9
 CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
A). Yes. B). No. Q15 Is it possible for a solid metal ball to float in mercury?
 Q15 Is it possible for a solid metal ball to float in mercury? A). Yes. B). No. The upward force is the weight of liquid displaced and the downward force is the weight of the ball. If the density of the
Q15 Is it possible for a solid metal ball to float in mercury? A). Yes. B). No. The upward force is the weight of liquid displaced and the downward force is the weight of the ball. If the density of the
Temperature and Heat 4.1. Temperature depends on particle movement Energy flows from warmer to cooler objects. 4.3
 Temperature and Heat NEW the BIG idea Heat is a flow of energy due to temperature differences. 4.1 Temperature depends on particle movement. 4.2 Energy flows from warmer to cooler objects. 4.3 The transfer
Temperature and Heat NEW the BIG idea Heat is a flow of energy due to temperature differences. 4.1 Temperature depends on particle movement. 4.2 Energy flows from warmer to cooler objects. 4.3 The transfer
Temperature, Thermal Energy and Heat
 Temperature You use the words hot and cold to describe temperature. Something is hot when its temperature is high. When you heat water on a stove, its temperature increases. How are temperature and heat
Temperature You use the words hot and cold to describe temperature. Something is hot when its temperature is high. When you heat water on a stove, its temperature increases. How are temperature and heat
Unit 11: Temperature and heat
 Unit 11: Temperature and heat 1. Thermal energy 2. Temperature 3. Heat and thermal equlibrium 4. Effects of heat 5. Transference of heat 6. Conductors and insulators Think and answer a. Is it the same
Unit 11: Temperature and heat 1. Thermal energy 2. Temperature 3. Heat and thermal equlibrium 4. Effects of heat 5. Transference of heat 6. Conductors and insulators Think and answer a. Is it the same
Name Class Date. How are temperature and energy related? What are the three common temperature scales? Why do objects feel hot or cold?
 CHAPTER 14 SECTION Heat and Temperature 1 Temperature KEY IDEAS As you read this section, keep these questions in mind: How are temperature and energy related? What are the three common temperature scales?
CHAPTER 14 SECTION Heat and Temperature 1 Temperature KEY IDEAS As you read this section, keep these questions in mind: How are temperature and energy related? What are the three common temperature scales?
Chapter 10 Test Form B
 Chapter 10 Test Form A 1. B 2. A 3. A 4. B 5. D 6. B 7. B 8. A 9. A 10. A 11. B 12. D 13. A 14. C 15. No, heat and cold do not flow between objects. Energy transferred between objects changes the temperature
Chapter 10 Test Form A 1. B 2. A 3. A 4. B 5. D 6. B 7. B 8. A 9. A 10. A 11. B 12. D 13. A 14. C 15. No, heat and cold do not flow between objects. Energy transferred between objects changes the temperature
Chapter 14 9/21/15. Solids, Liquids & Gasses. Essential Questions! Kinetic Theory! Gas State! Gas State!
 Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
Chapter 14 Solids, Liquids & Gasses Essential Questions What is the kinetic theory of matter? How do particles move in the different states of matter? How do particles behave at the boiling and melting
Section 16.3 Phase Changes
 Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
Section 16.3 Phase Changes Solid Liquid Gas 3 Phases of Matter Density of Matter How packed matter is (The amount of matter in a given space) Solid: Liquid: Gas: High Density Medium Density Low Density
WELCOME TO PERIOD 5: THERMAL ENERGY, THE MICROSCOPIC PICTURE. Homework #4 is due today at the beginning of class.
 WELCOME TO PERIOD 5: THERMAL ENERGY, THE MICROSCOPIC PICTURE Homework #4 is due today at the beginning of class. PHYSICS 1104 PERIOD 5 How are temperatures measured? How do atoms and molecules act at different
WELCOME TO PERIOD 5: THERMAL ENERGY, THE MICROSCOPIC PICTURE Homework #4 is due today at the beginning of class. PHYSICS 1104 PERIOD 5 How are temperatures measured? How do atoms and molecules act at different
Temp vs. Heat. Absolute Temperature Scales. Common Temperature Scales. Thermal Energy. Heat and Temperature are not the same!!
 Thermal Energy Heat and Temperature are not the same!! Cold is the absence of heat, not an energy Same concept as light/dark Cold can t come in, heat flows out Heat flows from High Temp Low Temp Temp vs.
Thermal Energy Heat and Temperature are not the same!! Cold is the absence of heat, not an energy Same concept as light/dark Cold can t come in, heat flows out Heat flows from High Temp Low Temp Temp vs.
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Bell Ringer. What are the formulas to obtain the force, acceleration, and mass? And corresponding units. F= ma M= f/a A= f/m
 Bell Ringer What are the formulas to obtain the force, acceleration, and mass? And corresponding units. F= ma M= f/a A= f/m F= N M= kg A= m/s^2 What did we learn about the acceleration rate and gravitational
Bell Ringer What are the formulas to obtain the force, acceleration, and mass? And corresponding units. F= ma M= f/a A= f/m F= N M= kg A= m/s^2 What did we learn about the acceleration rate and gravitational
SOLIDS, LIQUIDS, AND GASES
 CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
CHAPTER 2 SOLIDS, LIQUIDS, AND GASES SECTION 2 1 States of Matter (pages 56-60) This section explains how shape, volume, and the motion of particles are useful in describing solids, liquids, and gases.
Keep the Heat Test School Name. Team Number
 Keep the Heat Test 1-28-2012 School Name Team Number Circle the all of the correct answer to the below questions. One or more of the answers can be correct, if more than on one answer is correct, circle
Keep the Heat Test 1-28-2012 School Name Team Number Circle the all of the correct answer to the below questions. One or more of the answers can be correct, if more than on one answer is correct, circle
Physics 231 Lecture 29 Some slides relevant to Wed. Lecture. The notes for the lecture given by Dr. Nagy can be found at
 Physics 231 Lecture 29 Some slides relevant to Wed. Lecture. he notes for the lecture given by Dr. Nagy can be found at Main points : emperature and thermometers hermal expansion ΔL αδl; ΔA γa Δ; ΔV βvδ
Physics 231 Lecture 29 Some slides relevant to Wed. Lecture. he notes for the lecture given by Dr. Nagy can be found at Main points : emperature and thermometers hermal expansion ΔL αδl; ΔA γa Δ; ΔV βvδ
STATES OF MATTER. The Four States of Ma/er. Four States. Solid Liquid Gas Plasma
 STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
STATES OF MATTER The Four States of Ma/er Solid Liquid Gas Plasma Four States STATES OF MATTER Ø What makes a substance a par:cular state of ma
SI Measurements. (also known as metric system ) SI stands for System International or International System of Measurement
 SI Measurements (also known as metric system ) SI stands for System International or International System of Measurement SI Measurements (also known as metric system ) What about it? Common Language for
SI Measurements (also known as metric system ) SI stands for System International or International System of Measurement SI Measurements (also known as metric system ) What about it? Common Language for
Bernoulli s Principle. Application: Lift. Bernoulli s Principle. Main Points 3/13/15. Demo: Blowing on a sheet of paper
 Bernoulli s Principle Demo: Blowing on a sheet of paper Where the speed of a fluid increases, internal pressure in the fluid decreases. Due to continuous flow of a fluid: what goes in must come out! Fluid
Bernoulli s Principle Demo: Blowing on a sheet of paper Where the speed of a fluid increases, internal pressure in the fluid decreases. Due to continuous flow of a fluid: what goes in must come out! Fluid
Pg , Syllabus
 Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
Pg. 169 171, 173-175 Syllabus 5.7 5.14 www.cgrahamphysics.com What do you remember? End www.cgrahamphysics.com How do particles move? 3 of 30 Boardworks Ltd 2012 4 of 30 Boardworks Ltd 2012 States of matter
CERT Educational Series Heat Transfer
 Student Lab Sheet Answer Key CERT Educational Series Heat Transfer Name Date: Are HEAT and TEMPERATURE the same thing? YES NO Heat and Temperature are not the same thing. They have different units. Heat
Student Lab Sheet Answer Key CERT Educational Series Heat Transfer Name Date: Are HEAT and TEMPERATURE the same thing? YES NO Heat and Temperature are not the same thing. They have different units. Heat
Name Class Date. What is a change of state? What happens during a change of state? What can happen when a substance loses or gains energy?
 CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
CHAPTER 2 3 Changes of State SECTION States of Matter BEFORE YOU READ After you read this section, you should be able to answer these questions: What is a change of state? What happens during a change
the energy of motion!
 What are the molecules of matter doing all the time?! Heat and Temperature! Notes! All matter is composed of continually jiggling atoms or molecules! The jiggling is! If something is vibrating, what kind
What are the molecules of matter doing all the time?! Heat and Temperature! Notes! All matter is composed of continually jiggling atoms or molecules! The jiggling is! If something is vibrating, what kind
States of Matter. Liquid (water) H Gas (water vapor) Solid (ice)
 Liquid (water) FR EE ZI ME LT I SA E D C RA AP EV WATER MLECULE as (water vapor) Solid (ice) States of Matter You can skate on solid water. You can put solid water in your drink to make it cold. We call
Liquid (water) FR EE ZI ME LT I SA E D C RA AP EV WATER MLECULE as (water vapor) Solid (ice) States of Matter You can skate on solid water. You can put solid water in your drink to make it cold. We call
NATIONAL 5 PHYSICS THERMODYNAMICS
 NATIONAL 5 PHYSICS THERMODYNAMICS HEAT AND TEMPERATURE Heat and temperature are not the same thing! Heat Heat is a type of energy. Like all types of energy it is measured in joules (J). The heat energy
NATIONAL 5 PHYSICS THERMODYNAMICS HEAT AND TEMPERATURE Heat and temperature are not the same thing! Heat Heat is a type of energy. Like all types of energy it is measured in joules (J). The heat energy
Changing States of Matter By Cindy Grigg
 By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
By Cindy Grigg 1 On Earth, almost all matter exists in just three states. Matter is usually a solid, a liquid, or a gas. Plasma, the fourth state of matter, is rare on Earth. It sometimes can be found
States of matter. Book page , Syllabus /09/2016
 States of matter Book page 169 171, 173-175 Syllabus 5.7 5.14 05/09/2016 cgrahamphysics.com 2015 What is my state of matter? sand Decaffeinated coffee Glass Supercritical fluids Supercritical fluids Coldest
States of matter Book page 169 171, 173-175 Syllabus 5.7 5.14 05/09/2016 cgrahamphysics.com 2015 What is my state of matter? sand Decaffeinated coffee Glass Supercritical fluids Supercritical fluids Coldest
Checkpoint Identify matter and non-matter from the following air, temperature, table, thoughts, emotions, water, pen, prayer and phone.
 1 World of Matter What is matter? Definition Matter is something that occupies space and has mass. Checkpoint Identify matter and non-matter from the following air, temperature, table, thoughts, emotions,
1 World of Matter What is matter? Definition Matter is something that occupies space and has mass. Checkpoint Identify matter and non-matter from the following air, temperature, table, thoughts, emotions,
3. EFFECTS OF HEAT. Thus, heat can be defined as a form of energy that gives the sensation of hotness or coldness
 3. EFFECTS OF HEAT In the previous class you have learnt that heat is a form of energy. Heat can be obtained from various sources like the sun, fire, etc. When we read the weather forecast we observe that
3. EFFECTS OF HEAT In the previous class you have learnt that heat is a form of energy. Heat can be obtained from various sources like the sun, fire, etc. When we read the weather forecast we observe that
Matter and Its Properties. Unit 2
 Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
Matter and Its Properties Unit 2 Lesson 1: Physical & Chemical Properties & Changes Unit 2: Matter and Its Properties Section 1: Physical Properties & Change Lesson 1: Physical & Chemical Properties &
BUTTERFLY LAB METAMORPHOSIS & THE ENVIRONMENT. Handouts 6th & 7th Grade Science Unit EarthsBirthday.org
 METAMORPHOSIS & THE ENVIRONMENT Handouts 6th & 7th Grade Science Unit 1 800 698 4438 EarthsBirthday.org BUTTERFLY LAB CONTENTS Note: Answer Keys are in the Teacher Guide. Handout: Controlled Experiment
METAMORPHOSIS & THE ENVIRONMENT Handouts 6th & 7th Grade Science Unit 1 800 698 4438 EarthsBirthday.org BUTTERFLY LAB CONTENTS Note: Answer Keys are in the Teacher Guide. Handout: Controlled Experiment
0 o K is called absolute zero. Water Freezes: 273 o K Water Boils: 373 o K
 Part I Notes Temperature and Heat The terms at the right all mean the same thing. The heat energy of a substance is the sum of the kinetic and potential energies of all of the atoms and molecules in the
Part I Notes Temperature and Heat The terms at the right all mean the same thing. The heat energy of a substance is the sum of the kinetic and potential energies of all of the atoms and molecules in the
Chapter 14 Temperature and Heat
 Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 14 Temperature and Heat Thermodynamics Starting a different area of physics called thermodynamics Thermodynamics focuses on energy rather than
Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 14 Temperature and Heat Thermodynamics Starting a different area of physics called thermodynamics Thermodynamics focuses on energy rather than
Temperature and Heat. Chapter 17. PowerPoint Lectures for University Physics, Twelfth Edition Hugh D. Young and Roger A. Freedman
 Chapter 17 Temperature and Heat PowerPoint Lectures for University Physics, Twelfth Edition Hugh D. Young and Roger A. Freedman Lectures by James Pazun Goals for Chapter 17 To delineate the three different
Chapter 17 Temperature and Heat PowerPoint Lectures for University Physics, Twelfth Edition Hugh D. Young and Roger A. Freedman Lectures by James Pazun Goals for Chapter 17 To delineate the three different
Thermochemistry. The study of energy changes that occur during chemical reactions and changes in state.
 Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
Energy Thermochemistry The study of energy changes that occur during chemical reactions and changes in state. The Nature of Energy Energy - the ability to do work or produce heat Energy is stored in the
heat By cillian bryan and scott doyle
 heat By cillian bryan and scott doyle What is heat Heat energy is the result of the movement of tiny particles called atoms molecules or ions in solids, liquids and gases. Heat energy can be transferred
heat By cillian bryan and scott doyle What is heat Heat energy is the result of the movement of tiny particles called atoms molecules or ions in solids, liquids and gases. Heat energy can be transferred
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
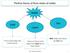 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Name Class Date. What are three kinds of energy transfer? What are conductors and insulators? What makes something a good conductor of heat?
 CHAPTER 14 SECTION Heat and Temperature 2 Energy Transfer KEY IDEAS As you read this section, keep these questions in mind: What are three kinds of energy transfer? What are conductors and insulators?
CHAPTER 14 SECTION Heat and Temperature 2 Energy Transfer KEY IDEAS As you read this section, keep these questions in mind: What are three kinds of energy transfer? What are conductors and insulators?
