The response of an electron in the image potential on the external steady fields
|
|
|
- Conrad Davis
- 6 years ago
- Views:
Transcription
1 1 The response of an electron in the image potential on the eternal steady fields 1,) P.A. Golovinski, M.A. Preobrahenskiy ) 1 ) Moscow Institute of Physics and Technology (State University), , Moscow, Russia ) Voroneh State University of Architecture and Civil ngineering, , Voroneh, Russia golovinski@bk.ru The eact wave functions, which describe the states of an electron, bound in the image potential, and the magnetic field, which is perpendicular to surface of a metal, are obtained. The correction terms to the energy of an electron in the first order of the perturbation theory with respect to the interaction with the eternal electric field are calculated. The possibility of eperimental measurement of a local electric field by the level shift is analyed. Low-dimensional systems (two-dimensional, one-dimensional and ero-dimensional) have a number of unusual properties that largely determine their role in the study and developing of nanoscale devices [1]. In particular, an electron near the metal surface interacts with induced on the surface polariation charge, attraction to which leads to appearance of localied states. Far from the surface in a vacuum this potential is well approimated by the classical long-range potential []. An electron near metal surface interacts with the field of its mirror image [3-5 interaction with which is asymptotically described by a one-dimensional Coulomb operator of an electron energy for the attraction to the metal surface [6] (the atomic system of units is used: e m 1). The model is corresponded to the "one-dimensional atom" with an effective charge Z 1/ 4, and the hydrogenlike electron states with energies v 1/ 3( a) are determined by the quantum-number and the quantum defect a, which weakly depends on v [3, 7]. This theoretical model [8-10] is confirmed by spectroscopic observations, such as two-photon resonance emission of electrons from the metal [11]. More sophisticated accounting of electron interaction with the surface plasmons is possible within the framework of multi-particle theory and is important for correct description of the width of the lower states with v 3. In this paper the response of an electron that is bound in the image potential on electric and magnetic fields is considered and the possibility of implementation this effect in spectroscopic measurement of electric field near the surface of metal is analyed. Nonrelativistic Hamiltonian of an electron in electromagnetic field can be epressed in terms of the operators of the momentum, the intrinsic magnetic moment, vector A and scalar potentials of electric field and magnetic field strength H [1] by equation:
2 1 A H ˆ pˆ μˆ H. (1) c Let s consider behavior of an electron in the image potential in homogeneous electric and magnetic fields, which are directed perpendicular to a metal surface. In this case, the scalar potential depends only on the coordinate and it is determined by eternal electric field strength by equation form A Hy, 0, Then, stationary Schrödinger equation has the form F. The vector potential can be taken in the pˆ pˆ pˆ pˆ Hy / c H y / c / F 1 4 H / c, y, 0 y It is assumed here that the operator.() μˆ H commutes with the Hamiltonian (1) and, therefore, the electron has a definite value of the spin projection. Hamiltonian of the q. () is additive with respect to the Cartesian coordinates. Hence, variables in the solution are entirely separated. Furthermore, because the Hamiltonian of the q. () does not contain the variable, it commutes with the component of the momentum operator pˆ. The corresponding component of the wave function is equal to the eigenfunction of operator pˆ normaliaed on δ-function it is described by a plane wave written as ep( ip ) /. With this in mind, wave function of electron can be, y, ep( ip ) y ( ) /. (3) Since the variables in the wave function (3) are divided, the total energy of the electron has the form: p y. (4) We obtain two independent equations for y and dependent components of the wave function by substituting the wave function (3) into q. (): Introducing new variable Y d / dy H y cp H / c p y H / cy 0 d / d F 1 / 4 0 y c p H and parameters, (5). (6) m p / y H / c; H / c (7) allows us to reduce the equation (5) to the Schrödinger equation for a harmonic oscillator [1] Component of the wave function in the form: d / dy m Y Y 0. (8) Y is epressed in terms of Hermite polynomials H m [1, 13]
3 3 1/ 4 n Y H / c m! 1/ ep HY / ch Y H / c. (9) Because equidistant discrete spectrum is m 1 ; m 1,,3 m,...,, (10) m and following q. (7) and q. (9), we obtain the energy component y that is linear on the magnetic field strength: m 1 H / c. (11) y p Thus, in the image potential field and magnetic field, which is perpendicular to the surface of metal, one has linear Zeeman. The value is determined by the q. (6) in the first-order of the perturbation theory with respect the interaction of an electron with an eternal electrostatic field. In the absence of an eternal electric field function () satisfies the equation Solution of the q. (1) is equal to 0 d / d 1 ( ) 0. (1) ) f /, where ( f coincides with the radial part of the wave function of s state of a hydrogen-like atom with charge Z 1/ 4 [1]. For negative values of energy 0 solutions of the q. (1), normalied to unity, are epressed in terms of confluent hypergeometric function 1,, / [13] 3 / ep / 1,, / 0 Quantum number is positive integer number. 1. (13) The dipole moment of an electron ( ) ( ) in the image potential is nonero because it has no mirror symmetry. Therefore, in contrast to the three-dimensional Coulomb potential, the level shift is defined by the epression F (14) in the first-order of the perturbation theory with respect to the interaction of an electron with an eternal electric field [14]. The calculation of the cross sections for single-photon transitions in the Coulomb potential between states with different energies i and f is usually based on the integral equations for the confluent hypergeometric functions [15], and the dipole moment of a state is obtained by a limit i f. For the static fields a direct algorithm for calculating the dipole moment of a discrete state, based on the reduction of epression for the matri element in q. (14) to the
4 4 superposition of the uler integrals [13], can be formulated. With taking into account the normaliation condition (13), it turns to the form min1, 3 3 s s... s 1 1!. (15) s0 s! s 1! s! The sum in the q. (15) for the states with quantum number contains two terms. Calculation leads to the relation 3 /, (16) that formally coincides with the average value of a distance r for the s-state in three-dimensional hydrogen atom. It is also true for the ground and first ecited state. Taking into account relation between quantum number and unperturbed energy 0, and q. (16), -component of the bound-state energy in an electrostatic field can be represented in the form 1/ 3F /. (17) The condition of validity for the first-order perturbation theory is determined by the relative smallness of the energy shift of the neighboring states compared with the distance between them, 4 which leads to the condition 1 / 3 1 takes a form F. For levels with 1 it simplifies and 5 F. Narrowing the range of applicability of the perturbation theory near the boundary of ioniation is eplained by the increased length of the electron cloud. Substituting y- component and the -component of the energy from q. (11) and q. (17) into q. (4) for the total energy of the electron, one can obtain equation m 1 H / c 1 3 F ( m, ). (18) Since the energy (18) does not depend on the component p of the momentum, the electron states, which are described by the wave functions (3), have an infinite degeneracy typical for the Landau levels. Dependences of energy shift (18) on the electric field strength for a number of highly ecited states of an electron in the image potential is shown in Fig. 1. These dependences demonstrate a linear rise of the energy distances between the levels with increasing the electric field strength.
5 5 Fig.1. The dependences of the energy shift of electron states, bound in the image potential, for quantum numbers (bottom to top) 0, 1,, 3, on the electric field strength. As it follows from q. (18), linear static Stark effect contributes to the energy of all states, which are bound by one-dimensional Coulomb interaction. In contrast, in three-dimensional Coulomb potential, the ground state is not shifted in a uniform electric field. This difference is determined by difference in the physical cause of the energy shift in three-dimensional potential shift, which is linear on the field, it is caused by interaction of degenerate levels, and in the onedimensional case it is caused by the mirror asymmetry of states. In accordance to the q. (18), the change in energy of transition between states with the equal quantum numbers m is determined by the electric field strength and quantum numbers i and f of levels, between which the radiation transition happens: / 3F. (0) i f The error in determining the electric field strength by spectroscopic measurements of the Rydberg states with =90 with high resolution in ultracold gas of cesium atoms is equal to 1 mv/cm [16]. The resolution is determined by a width of the line of the high-frequency resonance, which is equal to = 0 60 kh ( cm 1 ). Changing the energy of transition (in units cm-1) between the Rydberg states in the image potential, when quantum numbers differ by k, is equal to k k F (the electric field
6 6 strength is epressed in V/cm and it is assumed, that k )). The minimum electric field strength, measured by the spectroscopic methods in the image potential, limited by condition, is equal to F 510 / k. Thus, in the image potential field, the resolution k min of 1 mv/cm can be achieved at the transition between adjacent levels with quantum numbers ~ 50 and can be increased by increasing photon energy. In addition, the spectroscopic measurements in the image potential field are simplified (compared to the case of the Rydberg states of atoms) due to a more simple energetic structure of the bound states without quasidegenerate levels. In contrast to the electric field, the accuracy of the spectroscopic measurements of the magnetic field does not increase with increasing of the quantum numbers of the discrete states. However, the presence of the magnetic field facilitates the confinement of electrons, which allows localie the states in some space near the metal surface. This research has been supported by the RFFI through Grant and Ministry of ducation and Science of Russian Federation state order 014/
7 7 References [1] Nanoscale Science and Technology / d. by R.W. Kelsall, I.W. Hamly, M. Geoghegan. John Wiley & Sons, 006. [] Chulkov.V., Sarria I., Silkin V.M., Piyarke, chenique P.M. // Phys. Rev. Lett Vol. 80. P [3] chenique P.M., Uranga M.. // Surface Science Vol. 47. P. 15. [4] chenique P.M., Berndt R., Chulkov.V., Fauster T., Goldmann A., Höfer U. // Surface Science Reports Vol. 5. P. 19. [5] remeev S.V., Tsirkin S.S., Chulkov.V. // FTT Vol. 5. P (in Russian). [6] Landau L.D., Lifshits.M. lectrodynamics of continuous medium. Pergamon Press, [7] Chulkov.V., Silkin V.M., chenique P.M. // Surface Science Vol P [8] Höfer U., Shumay I. L., ReuCh., Thomann U., Wallauer W., Fauster Th. // Science Vol. 77. P [9] chenique, P. M., Pendry, J. B.// Prog. Surf. Sci Vol. 3. P [10] Dynamics at solid state surfaces and interfaces / d. by Bovenseipen U., Petek H., Wolf M. Wiley-VCH, 010. [11] Shumay I. L., Höfer U., Reuß Ch., Thomann U., Wallauer W., Fauster Th. // Phys. Rev. B Vol. 58, 0. P [1] Landau L.D., Lifshits.M. Quantum mechanics. Pergamon Press, [13] Bateman H., rdelyi Ar. Higher transcendental functions. Vol. 1. McGraw-Hill, [14] Sobelman I.I. Introduction to the theory of atomic spectra. Pergamon Press, 197. [15] Bethe H.A., Salpeter.. Quantum Mechanics of One- and Two-lectron Atoms. Dover Pub lications, 008. [16] Saβmannshausen H., Merkt F., Deigmayr J. // Phys. Rev. A Vol. 87. P
QUANTUM MECHANICS. Franz Schwabl. Translated by Ronald Kates. ff Springer
 Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
Franz Schwabl QUANTUM MECHANICS Translated by Ronald Kates Second Revised Edition With 122Figures, 16Tables, Numerous Worked Examples, and 126 Problems ff Springer Contents 1. Historical and Experimental
LECTURES ON QUANTUM MECHANICS
 LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
LECTURES ON QUANTUM MECHANICS GORDON BAYM Unitsersity of Illinois A II I' Advanced Bock Progrant A Member of the Perseus Books Group CONTENTS Preface v Chapter 1 Photon Polarization 1 Transformation of
Phase shift s influence of two strong pulsed laser waves on effective interaction of electrons
 Phase shift s influence of two strong pulsed laser waves on effective interaction of electrons S S Starodub, S P Roshchupkin and V V Dubov Institute of Applied Physics, National Academy of Sciences of
Phase shift s influence of two strong pulsed laser waves on effective interaction of electrons S S Starodub, S P Roshchupkin and V V Dubov Institute of Applied Physics, National Academy of Sciences of
Quantum Physics in the Nanoworld
 Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
Hans Lüth Quantum Physics in the Nanoworld Schrödinger's Cat and the Dwarfs 4) Springer Contents 1 Introduction 1 1.1 General and Historical Remarks 1 1.2 Importance for Science and Technology 3 1.3 Philosophical
ψ s a ˆn a s b ˆn b ψ Hint: Because the state is spherically symmetric the answer can depend only on the angle between the two directions.
 1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
1. Quantum Mechanics (Fall 2004) Two spin-half particles are in a state with total spin zero. Let ˆn a and ˆn b be unit vectors in two arbitrary directions. Calculate the expectation value of the product
Practical Quantum Mechanics
 Siegfried Flügge Practical Quantum Mechanics With 78 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Budapest Contents Volume I I. General Concepts 1. Law of probability
Siegfried Flügge Practical Quantum Mechanics With 78 Figures Springer-Verlag Berlin Heidelberg New York London Paris Tokyo Hong Kong Barcelona Budapest Contents Volume I I. General Concepts 1. Law of probability
P. W. Atkins and R. S. Friedman. Molecular Quantum Mechanics THIRD EDITION
 P. W. Atkins and R. S. Friedman Molecular Quantum Mechanics THIRD EDITION Oxford New York Tokyo OXFORD UNIVERSITY PRESS 1997 Introduction and orientation 1 Black-body radiation 1 Heat capacities 2 The
P. W. Atkins and R. S. Friedman Molecular Quantum Mechanics THIRD EDITION Oxford New York Tokyo OXFORD UNIVERSITY PRESS 1997 Introduction and orientation 1 Black-body radiation 1 Heat capacities 2 The
Relativistic corrections of energy terms
 Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
PHY4604 Introduction to Quantum Mechanics Fall 2004 Final Exam SOLUTIONS December 17, 2004, 7:30 a.m.- 9:30 a.m.
 PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
PHY464 Introduction to Quantum Mechanics Fall 4 Final Eam SOLUTIONS December 7, 4, 7:3 a.m.- 9:3 a.m. No other materials allowed. If you can t do one part of a problem, solve subsequent parts in terms
NERS 311 Current Old notes notes Chapter Chapter 1: Introduction to the course 1 - Chapter 1.1: About the course 2 - Chapter 1.
 NERS311/Fall 2014 Revision: August 27, 2014 Index to the Lecture notes Alex Bielajew, 2927 Cooley, bielajew@umich.edu NERS 311 Current Old notes notes Chapter 1 1 1 Chapter 1: Introduction to the course
NERS311/Fall 2014 Revision: August 27, 2014 Index to the Lecture notes Alex Bielajew, 2927 Cooley, bielajew@umich.edu NERS 311 Current Old notes notes Chapter 1 1 1 Chapter 1: Introduction to the course
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals
 Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
The 3 dimensional Schrödinger Equation
 Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Chapter 6 The 3 dimensional Schrödinger Equation 6.1 Angular Momentum To study how angular momentum is represented in quantum mechanics we start by reviewing the classical vector of orbital angular momentum
Quantum Physics II (8.05) Fall 2002 Outline
 Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Quantum Physics II (8.05) Fall 2002 Outline 1. General structure of quantum mechanics. 8.04 was based primarily on wave mechanics. We review that foundation with the intent to build a more formal basis
Basic quantum Hamiltonian s relativistic corrections. Abstract
 Basic quantum Hamiltonian s relativistic corrections Gintautas P. Kamuntavičius Physics Department, Vytautas Magnus University, Vileikos 8, Kaunas 44404, Lithuania (Dated: 2013.03.28) arxiv:1302.0491v2
Basic quantum Hamiltonian s relativistic corrections Gintautas P. Kamuntavičius Physics Department, Vytautas Magnus University, Vileikos 8, Kaunas 44404, Lithuania (Dated: 2013.03.28) arxiv:1302.0491v2
Quantum Mechanics: Fundamentals
 Kurt Gottfried Tung-Mow Yan Quantum Mechanics: Fundamentals Second Edition With 75 Figures Springer Preface vii Fundamental Concepts 1 1.1 Complementarity and Uncertainty 1 (a) Complementarity 2 (b) The
Kurt Gottfried Tung-Mow Yan Quantum Mechanics: Fundamentals Second Edition With 75 Figures Springer Preface vii Fundamental Concepts 1 1.1 Complementarity and Uncertainty 1 (a) Complementarity 2 (b) The
which implies that we can take solutions which are simultaneous eigen functions of
 Module 1 : Quantum Mechanics Chapter 6 : Quantum mechanics in 3-D Quantum mechanics in 3-D For most physical systems, the dynamics is in 3-D. The solutions to the general 3-d problem are quite complicated,
Module 1 : Quantum Mechanics Chapter 6 : Quantum mechanics in 3-D Quantum mechanics in 3-D For most physical systems, the dynamics is in 3-D. The solutions to the general 3-d problem are quite complicated,
3. Quantum Mechanics in 3D
 3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
3. Quantum Mechanics in 3D 3.1 Introduction Last time, we derived the time dependent Schrödinger equation, starting from three basic postulates: 1) The time evolution of a state can be expressed as a unitary
CONTENTS. vii. CHAPTER 2 Operators 15
 CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
CHAPTER 1 Why Quantum Mechanics? 1 1.1 Newtonian Mechanics and Classical Electromagnetism 1 (a) Newtonian Mechanics 1 (b) Electromagnetism 2 1.2 Black Body Radiation 3 1.3 The Heat Capacity of Solids and
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
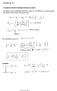 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Modern Physics for Scientists and Engineers International Edition, 4th Edition
 Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Qualifying Exam. Aug Part II. Please use blank paper for your work do not write on problems sheets!
 Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
Qualifying Exam Aug. 2015 Part II Please use blank paper for your work do not write on problems sheets! Solve only one problem from each of the four sections Mechanics, Quantum Mechanics, Statistical Physics
Phys 622 Problems Chapter 5
 1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
1 Phys 622 Problems Chapter 5 Problem 1 The correct basis set of perturbation theory Consider the relativistic correction to the electron-nucleus interaction H LS = α L S, also known as the spin-orbit
List of Comprehensive Exams Topics
 List of Comprehensive Exams Topics Mechanics 1. Basic Mechanics Newton s laws and conservation laws, the virial theorem 2. The Lagrangian and Hamiltonian Formalism The Lagrange formalism and the principle
List of Comprehensive Exams Topics Mechanics 1. Basic Mechanics Newton s laws and conservation laws, the virial theorem 2. The Lagrangian and Hamiltonian Formalism The Lagrange formalism and the principle
( ). Expanding the square and keeping in mind that
 One-electron atom in a Magnetic Field When the atom is in a magnetic field the magnetic moment of the electron due to its orbital motion and its spin interacts with the field and the Schrodinger Hamiltonian
One-electron atom in a Magnetic Field When the atom is in a magnetic field the magnetic moment of the electron due to its orbital motion and its spin interacts with the field and the Schrodinger Hamiltonian
a = ( a σ )( b σ ) = a b + iσ ( a b) mω 2! x + i 1 2! x i 1 2m!ω p, a = mω 2m!ω p Physics 624, Quantum II -- Final Exam
 Physics 624, Quantum II -- Final Exam Please show all your work on the separate sheets provided (and be sure to include your name). You are graded on your work on those pages, with partial credit where
Physics 624, Quantum II -- Final Exam Please show all your work on the separate sheets provided (and be sure to include your name). You are graded on your work on those pages, with partial credit where
PHYSICS OF SEMICONDUCTORS AND THEIR HETEROSTRUCTURES
 PHYSICS OF SEMICONDUCTORS AND THEIR HETEROSTRUCTURES Jasprit Singh University of Michigan McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London Madrid Mexico Milan Montreal
PHYSICS OF SEMICONDUCTORS AND THEIR HETEROSTRUCTURES Jasprit Singh University of Michigan McGraw-Hill, Inc. New York St. Louis San Francisco Auckland Bogota Caracas Lisbon London Madrid Mexico Milan Montreal
2.4. Quantum Mechanical description of hydrogen atom
 2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
2.4. Quantum Mechanical description of hydrogen atom Atomic units Quantity Atomic unit SI Conversion Ang. mom. h [J s] h = 1, 05459 10 34 Js Mass m e [kg] m e = 9, 1094 10 31 kg Charge e [C] e = 1, 6022
Announcements. Lecture 20 Chapter. 7 QM in 3-dims & Hydrogen Atom. The Radial Part of Schrodinger Equation for Hydrogen Atom
 Announcements! HW7 : Chap.7 18, 20, 23, 32, 37, 38, 45, 47, 53, 57, 60! Physics Colloquium: Development in Electron Nuclear Dynamics Theory on Thursday @ 3:40pm! Quiz 2 (average: 9), Quiz 3: 4/19 *** Course
Announcements! HW7 : Chap.7 18, 20, 23, 32, 37, 38, 45, 47, 53, 57, 60! Physics Colloquium: Development in Electron Nuclear Dynamics Theory on Thursday @ 3:40pm! Quiz 2 (average: 9), Quiz 3: 4/19 *** Course
Fundamentals of Spectroscopy for Optical Remote Sensing. Course Outline 2009
 Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
QUALIFYING EXAMINATION, Part 1. 2:00 PM 5:00 PM, Thursday September 3, 2009
 QUALIFYING EXAMINATION, Part 1 2:00 PM 5:00 PM, Thursday September 3, 2009 Attempt all parts of all four problems. Please begin your answer to each problem on a separate sheet, write your 3 digit code
QUALIFYING EXAMINATION, Part 1 2:00 PM 5:00 PM, Thursday September 3, 2009 Attempt all parts of all four problems. Please begin your answer to each problem on a separate sheet, write your 3 digit code
The general solution of Schrödinger equation in three dimensions (if V does not depend on time) are solutions of time-independent Schrödinger equation
 Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Lecture 17 Page 1 Lecture 17 L17.P1 Review Schrödinger equation The general solution of Schrödinger equation in three dimensions (if V does not depend on time) is where functions are solutions of time-independent
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
(n, l, m l ) 3/2/2016. Quantum Numbers (QN) Plots of Energy Level. Roadmap for Exploring Hydrogen Atom
 PHYS 34 Modern Physics Atom III: Angular Momentum and Spin Roadmap for Exploring Hydrogen Atom Today Contents: a) Orbital Angular Momentum and Magnetic Dipole Moment b) Electric Dipole Moment c) Stern
PHYS 34 Modern Physics Atom III: Angular Momentum and Spin Roadmap for Exploring Hydrogen Atom Today Contents: a) Orbital Angular Momentum and Magnetic Dipole Moment b) Electric Dipole Moment c) Stern
Chemistry 483 Lecture Topics Fall 2009
 Chemistry 483 Lecture Topics Fall 2009 Text PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon A. Background (M&S,Chapter 1) Blackbody Radiation Photoelectric effect DeBroglie Wavelength Atomic
Chemistry 483 Lecture Topics Fall 2009 Text PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon A. Background (M&S,Chapter 1) Blackbody Radiation Photoelectric effect DeBroglie Wavelength Atomic
DEPARTMENT OF PHYSICS UNIVERSITY OF PUNE PUNE SYLLABUS for the M.Phil. (Physics ) Course
 DEPARTMENT OF PHYSICS UNIVERSITY OF PUNE PUNE - 411007 SYLLABUS for the M.Phil. (Physics ) Course Each Student will be required to do 3 courses, out of which two are common courses. The third course syllabus
DEPARTMENT OF PHYSICS UNIVERSITY OF PUNE PUNE - 411007 SYLLABUS for the M.Phil. (Physics ) Course Each Student will be required to do 3 courses, out of which two are common courses. The third course syllabus
Lecture 4 Quantum mechanics in more than one-dimension
 Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Lecture 4 Quantum mechanics in more than one-dimension Background Previously, we have addressed quantum mechanics of 1d systems and explored bound and unbound (scattering) states. Although general concepts
Atomic Transitions and Selection Rules
 Atomic Transitions and Selection Rules The time-dependent Schrodinger equation for an electron in an atom with a time-independent potential energy is ~ 2 2m r2 (~r, t )+V(~r ) (~r, t )=i~ @ @t We found
Atomic Transitions and Selection Rules The time-dependent Schrodinger equation for an electron in an atom with a time-independent potential energy is ~ 2 2m r2 (~r, t )+V(~r ) (~r, t )=i~ @ @t We found
Atomic Physics 3 rd year B1
 Atomic Physics 3 rd year B1 P. Ewart Lecture notes Lecture slides Problem sets All available on Physics web site: http:www.physics.ox.ac.uk/users/ewart/index.htm Atomic Physics: Astrophysics Plasma Physics
Atomic Physics 3 rd year B1 P. Ewart Lecture notes Lecture slides Problem sets All available on Physics web site: http:www.physics.ox.ac.uk/users/ewart/index.htm Atomic Physics: Astrophysics Plasma Physics
Physics 606, Quantum Mechanics, Final Exam NAME ( ) ( ) + V ( x). ( ) and p( t) be the corresponding operators in ( ) and x( t) : ( ) / dt =...
 Physics 606, Quantum Mechanics, Final Exam NAME Please show all your work. (You are graded on your work, with partial credit where it is deserved.) All problems are, of course, nonrelativistic. 1. Consider
Physics 606, Quantum Mechanics, Final Exam NAME Please show all your work. (You are graded on your work, with partial credit where it is deserved.) All problems are, of course, nonrelativistic. 1. Consider
A. F. J. Levi 1 EE539: Engineering Quantum Mechanics. Fall 2017.
 A. F. J. Levi 1 Engineering Quantum Mechanics. Fall 2017. TTh 9.00 a.m. 10.50 a.m., VHE 210. Web site: http://alevi.usc.edu Web site: http://classes.usc.edu/term-20173/classes/ee EE539: Abstract and Prerequisites
A. F. J. Levi 1 Engineering Quantum Mechanics. Fall 2017. TTh 9.00 a.m. 10.50 a.m., VHE 210. Web site: http://alevi.usc.edu Web site: http://classes.usc.edu/term-20173/classes/ee EE539: Abstract and Prerequisites
Bound states of two particles confined to parallel two-dimensional layers and interacting via dipole-dipole or dipole-charge laws
 PHYSICAL REVIEW B VOLUME 55, NUMBER 8 15 FEBRUARY 1997-II Bound states of two particles confined to parallel two-dimensional layers and interacting via dipole-dipole or dipole-charge laws V. I. Yudson
PHYSICAL REVIEW B VOLUME 55, NUMBER 8 15 FEBRUARY 1997-II Bound states of two particles confined to parallel two-dimensional layers and interacting via dipole-dipole or dipole-charge laws V. I. Yudson
Free free scattering theory in circularly polarized laser field for polarized potential
 Free free scattering theory in circularly polarized laser field for polarized potential Kishori Yadav 1, Jeevan Jyoti Nakarmi 2 1 Patan Multiple Campus, 2 Central Department of Physics (T.U) ABSTRACT In
Free free scattering theory in circularly polarized laser field for polarized potential Kishori Yadav 1, Jeevan Jyoti Nakarmi 2 1 Patan Multiple Campus, 2 Central Department of Physics (T.U) ABSTRACT In
Solved radial equation: Last time For two simple cases: infinite and finite spherical wells Spherical analogs of 1D wells We introduced auxiliary func
 Quantum Mechanics and Atomic Physics Lecture 16: The Coulomb Potential http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Solved radial equation: Last time For two simple cases: infinite
Quantum Mechanics and Atomic Physics Lecture 16: The Coulomb Potential http://www.physics.rutgers.edu/ugrad/361 h / d/361 Prof. Sean Oh Solved radial equation: Last time For two simple cases: infinite
Joel Broida UCSD Fall 2009 Phys 130B QM II. Homework Set 4
 Joel Broida UCSD Fall 009 Phys 130B QM II Homework Set 4 1. Consider the particle-in-a-box problem but with a delta function potential H (x) = αδ(x l/) at the center (with α = const): H = αδ(x l/) 0 l/
Joel Broida UCSD Fall 009 Phys 130B QM II Homework Set 4 1. Consider the particle-in-a-box problem but with a delta function potential H (x) = αδ(x l/) at the center (with α = const): H = αδ(x l/) 0 l/
COPYRIGHTED MATERIAL. Index
 347 Index a AC fields 81 119 electric 81, 109 116 laser 81, 136 magnetic 112 microwave 107 109 AC field traps see Traps AC Stark effect 82, 84, 90, 96, 97 101, 104 109 Adiabatic approximation 3, 10, 32
347 Index a AC fields 81 119 electric 81, 109 116 laser 81, 136 magnetic 112 microwave 107 109 AC field traps see Traps AC Stark effect 82, 84, 90, 96, 97 101, 104 109 Adiabatic approximation 3, 10, 32
Ion traps. Trapping of charged particles in electromagnetic. Laser cooling, sympathetic cooling, optical clocks
 Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
A few principles of classical and quantum mechanics
 A few principles of classical and quantum mechanics The classical approach: In classical mechanics, we usually (but not exclusively) solve Newton s nd law of motion relating the acceleration a of the system
A few principles of classical and quantum mechanics The classical approach: In classical mechanics, we usually (but not exclusively) solve Newton s nd law of motion relating the acceleration a of the system
CLASSICAL ELECTRICITY
 CLASSICAL ELECTRICITY AND MAGNETISM by WOLFGANG K. H. PANOFSKY Stanford University and MELBA PHILLIPS Washington University SECOND EDITION ADDISON-WESLEY PUBLISHING COMPANY Reading, Massachusetts Menlo
CLASSICAL ELECTRICITY AND MAGNETISM by WOLFGANG K. H. PANOFSKY Stanford University and MELBA PHILLIPS Washington University SECOND EDITION ADDISON-WESLEY PUBLISHING COMPANY Reading, Massachusetts Menlo
Atomic Systems (PART I)
 Atomic Systems (PART I) Lecturer: Location: Recommended Text: Dr. D.J. Miller Room 535, Kelvin Building d.miller@physics.gla.ac.uk Joseph Black C407 (except 15/1/10 which is in Kelvin 312) Physics of Atoms
Atomic Systems (PART I) Lecturer: Location: Recommended Text: Dr. D.J. Miller Room 535, Kelvin Building d.miller@physics.gla.ac.uk Joseph Black C407 (except 15/1/10 which is in Kelvin 312) Physics of Atoms
2. The Schrödinger equation for one-particle problems. 5. Atoms and the periodic table of chemical elements
 1 Historical introduction The Schrödinger equation for one-particle problems 3 Mathematical tools for quantum chemistry 4 The postulates of quantum mechanics 5 Atoms and the periodic table of chemical
1 Historical introduction The Schrödinger equation for one-particle problems 3 Mathematical tools for quantum chemistry 4 The postulates of quantum mechanics 5 Atoms and the periodic table of chemical
Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid
 Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid Announcement This handout includes 9 problems. The first 5 are the problem set due. The last 4 cover material from the final few lectures
Quantum Physics II (8.05) Fall 2002 Assignment 12 and Study Aid Announcement This handout includes 9 problems. The first 5 are the problem set due. The last 4 cover material from the final few lectures
Lecture #13 1. Incorporating a vector potential into the Hamiltonian 2. Spin postulates 3. Description of spin states 4. Identical particles in
 Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
Lecture #3. Incorporating a vector potential into the Hamiltonian. Spin postulates 3. Description of spin states 4. Identical particles in classical and QM 5. Exchange degeneracy - the fundamental problem
2 Canonical quantization
 Phys540.nb 7 Canonical quantization.1. Lagrangian mechanics and canonical quantization Q: How do we quantize a general system?.1.1.lagrangian Lagrangian mechanics is a reformulation of classical mechanics.
Phys540.nb 7 Canonical quantization.1. Lagrangian mechanics and canonical quantization Q: How do we quantize a general system?.1.1.lagrangian Lagrangian mechanics is a reformulation of classical mechanics.
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall Duration: 2h 30m
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Atomic Structure and Processes
 Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
Physics 342 Lecture 27. Spin. Lecture 27. Physics 342 Quantum Mechanics I
 Physics 342 Lecture 27 Spin Lecture 27 Physics 342 Quantum Mechanics I Monday, April 5th, 2010 There is an intrinsic characteristic of point particles that has an analogue in but no direct derivation from
Physics 342 Lecture 27 Spin Lecture 27 Physics 342 Quantum Mechanics I Monday, April 5th, 2010 There is an intrinsic characteristic of point particles that has an analogue in but no direct derivation from
Introduction to Modern Quantum Optics
 Introduction to Modern Quantum Optics Jin-Sheng Peng Gao-Xiang Li Huazhong Normal University, China Vfe World Scientific» Singapore* * NewJerseyL Jersey* London* Hong Kong IX CONTENTS Preface PART I. Theory
Introduction to Modern Quantum Optics Jin-Sheng Peng Gao-Xiang Li Huazhong Normal University, China Vfe World Scientific» Singapore* * NewJerseyL Jersey* London* Hong Kong IX CONTENTS Preface PART I. Theory
3 Atomic Frequency Standards
 3 Atomic Frequency Standards 3-1 Basic Physics in the Atomic Frequency Standards Today, the second, unit of time, is defined by using the hyper fine transition of cesium atoms. In atomic clocks and atomic
3 Atomic Frequency Standards 3-1 Basic Physics in the Atomic Frequency Standards Today, the second, unit of time, is defined by using the hyper fine transition of cesium atoms. In atomic clocks and atomic
arxiv:cond-mat/ v1 9 Feb 1999
 Dressed States of a two component Bose-Einstein Condensate. P. B. Blakie and R. J. Ballagh Department of Physics, University of Otago, P. O. Bo 56, Dunedin, New Zealand. C. W. Gardiner Department of Physics,
Dressed States of a two component Bose-Einstein Condensate. P. B. Blakie and R. J. Ballagh Department of Physics, University of Otago, P. O. Bo 56, Dunedin, New Zealand. C. W. Gardiner Department of Physics,
Solutions Final exam 633
 Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
Solutions Final exam 633 S.J. van Enk (Dated: June 9, 2008) (1) [25 points] You have a source that produces pairs of spin-1/2 particles. With probability p they are in the singlet state, ( )/ 2, and with
14. Structure of Nuclei
 14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
14. Structure of Nuclei Particle and Nuclear Physics Dr. Tina Potter Dr. Tina Potter 14. Structure of Nuclei 1 In this section... Magic Numbers The Nuclear Shell Model Excited States Dr. Tina Potter 14.
University of Ljubljana Faculty of Mathematics and Physics. Photons in a double quantum resonator
 University of Ljubljana Faculty of Mathematics and Physics Photons in a double quantum resonator Author : Jernej Györköš 28010210 Mentor: Norma Mankoč Borštnik Date : 22.3.2006 Abstract The interaction
University of Ljubljana Faculty of Mathematics and Physics Photons in a double quantum resonator Author : Jernej Györköš 28010210 Mentor: Norma Mankoč Borštnik Date : 22.3.2006 Abstract The interaction
SCHRODINGER EQUATION. Chapter Schrodinger equation
 Chapter 2 SCHRODINGER EQUATION The first quantum mechanical theory, that gave the explanation of the discrete spectra of atomic emission, was based on the equation proposed by Schrodinger [2] in 1927.
Chapter 2 SCHRODINGER EQUATION The first quantum mechanical theory, that gave the explanation of the discrete spectra of atomic emission, was based on the equation proposed by Schrodinger [2] in 1927.
Preliminary Quantum Questions
 Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Modern Optical Spectroscopy
 Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
Modern Optical Spectroscopy With Exercises and Examples from Biophysics and Biochemistry von William W Parson 1. Auflage Springer-Verlag Berlin Heidelberg 2006 Verlag C.H. Beck im Internet: www.beck.de
University of Michigan Physics Department Graduate Qualifying Examination
 Name: University of Michigan Physics Department Graduate Qualifying Examination Part II: Modern Physics Saturday 17 May 2014 9:30 am 2:30 pm Exam Number: This is a closed book exam, but a number of useful
Name: University of Michigan Physics Department Graduate Qualifying Examination Part II: Modern Physics Saturday 17 May 2014 9:30 am 2:30 pm Exam Number: This is a closed book exam, but a number of useful
does not change the dynamics of the system, i.e. that it leaves the Schrödinger equation invariant,
 FYST5 Quantum Mechanics II 9..212 1. intermediate eam (1. välikoe): 4 problems, 4 hours 1. As you remember, the Hamilton operator for a charged particle interacting with an electromagentic field can be
FYST5 Quantum Mechanics II 9..212 1. intermediate eam (1. välikoe): 4 problems, 4 hours 1. As you remember, the Hamilton operator for a charged particle interacting with an electromagentic field can be
Physics 221A Fall 1996 Notes 19 The Stark Effect in Hydrogen and Alkali Atoms
 Physics 221A Fall 1996 Notes 19 The Stark Effect in Hydrogen and Alkali Atoms In these notes we will consider the Stark effect in hydrogen and alkali atoms as a physically interesting example of bound
Physics 221A Fall 1996 Notes 19 The Stark Effect in Hydrogen and Alkali Atoms In these notes we will consider the Stark effect in hydrogen and alkali atoms as a physically interesting example of bound
INTRODUCTION TO THE STRUCTURE OF MATTER
 INTRODUCTION TO THE STRUCTURE OF MATTER A Course in Modern Physics John J. Brehm and William J. Mullin University of Massachusetts Amherst, Massachusetts Fachberelch 5?@8hnlsdie Hochschule Darmstadt! HochschulstraSa
INTRODUCTION TO THE STRUCTURE OF MATTER A Course in Modern Physics John J. Brehm and William J. Mullin University of Massachusetts Amherst, Massachusetts Fachberelch 5?@8hnlsdie Hochschule Darmstadt! HochschulstraSa
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy. Often depends on position operator: Kinetic Energy 1-D case: 3-D case
 The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
The Postulates of Quantum Mechanics Common operators in QM: Potential Energy Often depends on position operator: Kinetic Energy 1-D case: 3-D case Time Total energy = Hamiltonian To find out about the
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
1 The postulates of quantum mechanics
 1 The postulates of quantum mechanics The postulates of quantum mechanics were derived after a long process of trial and error. These postulates provide a connection between the physical world and the
1 The postulates of quantum mechanics The postulates of quantum mechanics were derived after a long process of trial and error. These postulates provide a connection between the physical world and the
( ) in the interaction picture arises only
 Physics 606, Quantum Mechanics, Final Exam NAME 1 Atomic transitions due to time-dependent electric field Consider a hydrogen atom which is in its ground state for t < 0 For t > 0 it is subjected to a
Physics 606, Quantum Mechanics, Final Exam NAME 1 Atomic transitions due to time-dependent electric field Consider a hydrogen atom which is in its ground state for t < 0 For t > 0 it is subjected to a
arxiv: v2 [hep-th] 6 Jul 2009
![arxiv: v2 [hep-th] 6 Jul 2009 arxiv: v2 [hep-th] 6 Jul 2009](/thumbs/83/88697167.jpg) HIP-9-/TH Dirac Equation in Noncommutative Space for Hydrogen Atom arxiv:9.86v [hep-th] 6 Jul 9 T. C. Adorno, M. C. Baldiotti, M. Chaichian, D. M. Gitman and A. Tureanu Instituto de Física, Universidade
HIP-9-/TH Dirac Equation in Noncommutative Space for Hydrogen Atom arxiv:9.86v [hep-th] 6 Jul 9 T. C. Adorno, M. C. Baldiotti, M. Chaichian, D. M. Gitman and A. Tureanu Instituto de Física, Universidade
Electromagnetism II. Instructor: Andrei Sirenko Spring 2013 Thursdays 1 pm 4 pm. Spring 2013, NJIT 1
 Electromagnetism II Instructor: Andrei Sirenko sirenko@njit.edu Spring 013 Thursdays 1 pm 4 pm Spring 013, NJIT 1 PROBLEMS for CH. 6 http://web.njit.edu/~sirenko/phys433/phys433eandm013.htm Can obtain
Electromagnetism II Instructor: Andrei Sirenko sirenko@njit.edu Spring 013 Thursdays 1 pm 4 pm Spring 013, NJIT 1 PROBLEMS for CH. 6 http://web.njit.edu/~sirenko/phys433/phys433eandm013.htm Can obtain
2m 2 Ze2. , where δ. ) 2 l,n is the quantum defect (of order one but larger
 PHYS 402, Atomic and Molecular Physics Spring 2017, final exam, solutions 1. Hydrogenic atom energies: Consider a hydrogenic atom or ion with nuclear charge Z and the usual quantum states φ nlm. (a) (2
PHYS 402, Atomic and Molecular Physics Spring 2017, final exam, solutions 1. Hydrogenic atom energies: Consider a hydrogenic atom or ion with nuclear charge Z and the usual quantum states φ nlm. (a) (2
Time part of the equation can be separated by substituting independent equation
 Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
Lecture 9 Schrödinger Equation in 3D and Angular Momentum Operator In this section we will construct 3D Schrödinger equation and we give some simple examples. In this course we will consider problems where
1.6. Quantum mechanical description of the hydrogen atom
 29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
MOLECULAR SPECTROSCOPY
 MOLECULAR SPECTROSCOPY First Edition Jeanne L. McHale University of Idaho PRENTICE HALL, Upper Saddle River, New Jersey 07458 CONTENTS PREFACE xiii 1 INTRODUCTION AND REVIEW 1 1.1 Historical Perspective
MOLECULAR SPECTROSCOPY First Edition Jeanne L. McHale University of Idaho PRENTICE HALL, Upper Saddle River, New Jersey 07458 CONTENTS PREFACE xiii 1 INTRODUCTION AND REVIEW 1 1.1 Historical Perspective
Quantum Mechanics Solutions
 Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Quantum Mechanics Solutions (a (i f A and B are Hermitian, since (AB = B A = BA, operator AB is Hermitian if and only if A and B commute So, we know that [A,B] = 0, which means that the Hilbert space H
Lecture 10. Central potential
 Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
An Introduction to Hyperfine Structure and Its G-factor
 An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
Spectra of Atoms and Molecules. Peter F. Bernath
 Spectra of Atoms and Molecules Peter F. Bernath New York Oxford OXFORD UNIVERSITY PRESS 1995 Contents 1 Introduction 3 Waves, Particles, and Units 3 The Electromagnetic Spectrum 6 Interaction of Radiation
Spectra of Atoms and Molecules Peter F. Bernath New York Oxford OXFORD UNIVERSITY PRESS 1995 Contents 1 Introduction 3 Waves, Particles, and Units 3 The Electromagnetic Spectrum 6 Interaction of Radiation
FACULTY OF SCIENCES SYLLABUS FOR. B.Sc. (Non-Medical) PHYSICS PART-II. (Semester: III, IV) Session: , MATA GUJRI COLLEGE
 FACULTY OF SCIENCES SYLLABUS FOR B.Sc. (Non-Medical) PHYSICS PART-II (Semester: III, IV) Session: 2017 2018, 2018-2019 MATA GUJRI COLLEGE FATEHGARH SAHIB-140406, PUNJAB ----------------------------------------------------------
FACULTY OF SCIENCES SYLLABUS FOR B.Sc. (Non-Medical) PHYSICS PART-II (Semester: III, IV) Session: 2017 2018, 2018-2019 MATA GUJRI COLLEGE FATEHGARH SAHIB-140406, PUNJAB ----------------------------------------------------------
Nuclear magnetic resonance in condensed matter
 University of Ljubljana Faculty of mathematics and physics Physics department SEMINAR Nuclear magnetic resonance in condensed matter Author: Miha Bratkovič Mentor: prof. dr. Janez Dolinšek Ljubljana, October
University of Ljubljana Faculty of mathematics and physics Physics department SEMINAR Nuclear magnetic resonance in condensed matter Author: Miha Bratkovič Mentor: prof. dr. Janez Dolinšek Ljubljana, October
Helium fine structure theory for determination of α
 Journal of Physics: Conference Series Helium fine structure theory for determination of α To cite this article: Krzysztof Pachucki and Vladimir A Yerokhin 0 J. Phys.: Conf. Ser. 64 0007 View the article
Journal of Physics: Conference Series Helium fine structure theory for determination of α To cite this article: Krzysztof Pachucki and Vladimir A Yerokhin 0 J. Phys.: Conf. Ser. 64 0007 View the article
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS
 QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
We now turn to our first quantum mechanical problems that represent real, as
 84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
PHYSICS. Course Syllabus. Section 1: Mathematical Physics. Subject Code: PH. Course Structure. Electromagnetic Theory
 PHYSICS Subject Code: PH Course Structure Sections/Units Topics Section 1 Section 2 Section 3 Section 4 Section 5 Section 6 Section 7 Section 8 Mathematical Physics Classical Mechanics Electromagnetic
PHYSICS Subject Code: PH Course Structure Sections/Units Topics Section 1 Section 2 Section 3 Section 4 Section 5 Section 6 Section 7 Section 8 Mathematical Physics Classical Mechanics Electromagnetic
PHY 407 QUANTUM MECHANICS Fall 05 Problem set 1 Due Sep
 Problem set 1 Due Sep 15 2005 1. Let V be the set of all complex valued functions of a real variable θ, that are periodic with period 2π. That is u(θ + 2π) = u(θ), for all u V. (1) (i) Show that this V
Problem set 1 Due Sep 15 2005 1. Let V be the set of all complex valued functions of a real variable θ, that are periodic with period 2π. That is u(θ + 2π) = u(θ), for all u V. (1) (i) Show that this V
ATOMIC SPECTROSCOPY: Introduction to the Theory of Hyperfine Structure
 ATOMIC SPECTROSCOPY: Introduction to the Theory of Hyperfine Structure ATOMIC SPECTROSCOPY: Introduction to the Theory of Hyperfine Structure ANATOLI ANDREEV M.V. Lomonosov Moscow State University Moscow.
ATOMIC SPECTROSCOPY: Introduction to the Theory of Hyperfine Structure ATOMIC SPECTROSCOPY: Introduction to the Theory of Hyperfine Structure ANATOLI ANDREEV M.V. Lomonosov Moscow State University Moscow.
Common Exam Department of Physics University of Utah August 24, 2002
 Common Exam - 2002 Department of Physics University of Utah August 24, 2002 Examination booklets have been provided for recording your work and your solutions. Please note that there is a separate booklet
Common Exam - 2002 Department of Physics University of Utah August 24, 2002 Examination booklets have been provided for recording your work and your solutions. Please note that there is a separate booklet
Select/Special Topics in Atomic Physics Prof. P.C. Deshmukh Department Of Physics Indian Institute of Technology, Madras
 Select/Special Topics in Atomic Physics Prof. P.C. Deshmukh Department Of Physics Indian Institute of Technology, Madras Lecture - 37 Stark - Zeeman Spectroscopy Well, let us continue our discussion on
Select/Special Topics in Atomic Physics Prof. P.C. Deshmukh Department Of Physics Indian Institute of Technology, Madras Lecture - 37 Stark - Zeeman Spectroscopy Well, let us continue our discussion on
Quantum Field Theory. Kerson Huang. Second, Revised, and Enlarged Edition WILEY- VCH. From Operators to Path Integrals
 Kerson Huang Quantum Field Theory From Operators to Path Integrals Second, Revised, and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA I vh Contents Preface XIII 1 Introducing Quantum Fields
Kerson Huang Quantum Field Theory From Operators to Path Integrals Second, Revised, and Enlarged Edition WILEY- VCH WILEY-VCH Verlag GmbH & Co. KGaA I vh Contents Preface XIII 1 Introducing Quantum Fields
Super-reflection and Cloaking Based on Zero Index Metamaterial
 Super-reflection and Cloaking Based on Zero Index Metamaterial Jiaming Hao, Wei Yan, and Min Qiu Photonics and Microwave ngineering, Royal Institute of Technology (KTH), lectrum 9, 164 4, Kista, Sweden
Super-reflection and Cloaking Based on Zero Index Metamaterial Jiaming Hao, Wei Yan, and Min Qiu Photonics and Microwave ngineering, Royal Institute of Technology (KTH), lectrum 9, 164 4, Kista, Sweden
Magnetism of Atoms and Ions. Wulf Wulfhekel Physikalisches Institut, Karlsruhe Institute of Technology (KIT) Wolfgang Gaede Str. 1, D Karlsruhe
 Magnetism of Atoms and Ions Wulf Wulfhekel Physikalisches Institut, Karlsruhe Institute of Technology (KIT) Wolfgang Gaede Str. 1, D-76131 Karlsruhe 1 0. Overview Literature J.M.D. Coey, Magnetism and
Magnetism of Atoms and Ions Wulf Wulfhekel Physikalisches Institut, Karlsruhe Institute of Technology (KIT) Wolfgang Gaede Str. 1, D-76131 Karlsruhe 1 0. Overview Literature J.M.D. Coey, Magnetism and
MASSACHUSETTS INSTITUTE OF TECHNOLOGY 5.76 Modern Topics in Physical Chemistry Spring, Problem Set #2
 Reading Assignment: Bernath Chapter 5 MASSACHUSETTS INSTITUTE O TECHNOLOGY 5.76 Modern Topics in Physical Chemistry Spring 994 Problem Set # The following handouts also contain useful information: C &
Reading Assignment: Bernath Chapter 5 MASSACHUSETTS INSTITUTE O TECHNOLOGY 5.76 Modern Topics in Physical Chemistry Spring 994 Problem Set # The following handouts also contain useful information: C &
Motion of guiding center drift atoms in the electric and magnetic field of a Penning trap
 PHYSICS OF PLASMAS 1, 01101 (005) Motion of guiding center drift atoms in the electric and magnetic field of a Penning trap S. G. Kumin and T. M. O Neil Department of Physics, University of California
PHYSICS OF PLASMAS 1, 01101 (005) Motion of guiding center drift atoms in the electric and magnetic field of a Penning trap S. G. Kumin and T. M. O Neil Department of Physics, University of California
