Potential, Kinetic Notes.notebook. April 16, 2012
|
|
|
- Joel Thompson
- 5 years ago
- Views:
Transcription
1 Kinetic Energy: Consider a baseball flying through the air. The ball is said to have "kinetic energy" by virtue of the fact that its in motion relative to the ground. You can see that it is has energy because it can do "work" on an object on the ground if it collides with it (either by pushing on it and/or damaging it during the collision). The formula for Kinetic energy, and for some of the other forms of energy described in this section will, is given in a later section of this primer. Apr 4 8:41 AM 1
2 Potential Energy: Consider a book sitting on a table. The book is said to have "potential energy" because if it is nudged off, gravity will accelerate the book, giving the book kinetic energy. Because the Earth's gravity is necessary to create this kinetic energy, and because this gravity depends on the Earth being present, we say that the "Earth book system" is what really possesses this potential energy, and that this energy is converted into kinetic energy as the book falls. 2
3 Chemical Energy: Consider the ability of your body to do work. The glucose (blood sugar) in your body is said to have "chemical energy" because the glucose releases energy when chemically reacted (combusted) with oxygen. Your muscles use this energy to generate mechanical force and also heat. Chemical energy is really a form of microscopic potential energy, which exists because of the electric and magnetic forces of attraction exerted between the different parts of each molecule the same attractive forces involved in thermal vibrations. These parts get rearranged in chemical reactions, releasing or adding to this potential energy. 3
4 Electrical Energy: Some material, particularly metals, have certain electrons that are only loosely attached to their atoms. They can easily be made to move from one atom to another if an electric field is applied to them. When those electrons move among the atoms of matter, a current of electricity is created. This is what happens in a piece of wire when an electric field, or voltage, is applied. The electrons pass from atom to atom, pushed by the electric field and by each other (they repel each other because like charges repel), thus creating the electrical current. The measure of how well something conducts electricity is called its conductivity, and the reciprocal of conductivity is called the resistance. 4
5 Nuclear Energy: The Sun, nuclear reactors, and the interior of the Earth, all have "nuclear reactions" as the source of their energy, that is, reactions that involve changes in the structure of the nuclei of atoms. In the Sun, hydrogen nuclei fuse (combine) together to make helium nuclei, in a process called fusion, which releases energy. In the interior of the Earth, Uranium nuclei split apart in a process called fission. If this didn't happen, the Earth's interior would have long gone cold! In both cases, fusion or fission, some of the matter making up the nuclei is actually converted into energy. How can this be? The answer is that matter itself is a form of energy! This concept involves one of the most famous formula's in physics, the formula, E=mc2. 5
Aim: What are the two types of Nuclear. Reactions? Do Now: 1. Get into your groups and compare your answers to your homework.
 Aim: What are the two types of Nuclear Reactions? Do Now: 1. Get into your groups and compare your answers to your homework. Nuclear Energy In nuclear reaction, mass is converted into energy; there is
Aim: What are the two types of Nuclear Reactions? Do Now: 1. Get into your groups and compare your answers to your homework. Nuclear Energy In nuclear reaction, mass is converted into energy; there is
Unit 1. Types of Energy and Energy Conservation
 Strand B. Energy Unit 1. Types of Energy and Energy Conservation Contents Page Energy Types 2 Gravitational Potential and Kinetic Energy 5 Conservation of Energy 8 B.1.1 Energy Types Energy is a physical
Strand B. Energy Unit 1. Types of Energy and Energy Conservation Contents Page Energy Types 2 Gravitational Potential and Kinetic Energy 5 Conservation of Energy 8 B.1.1 Energy Types Energy is a physical
FUSION NEUTRON DEUTERIUM HELIUM TRITIUM.
 FUSION AND FISSION THE SUN Nuclear Fusion Nuclear fusion is the process by which multiple nuclei join together to form a heavier nucleus. It is accompanied by the release or absorption of energy depending
FUSION AND FISSION THE SUN Nuclear Fusion Nuclear fusion is the process by which multiple nuclei join together to form a heavier nucleus. It is accompanied by the release or absorption of energy depending
Alta Chemistry CHAPTER 25. Nuclear Chemistry: Radiation, Radioactivity & its Applications
 CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
CHAPTER 25 Nuclear Chemistry: Radiation, Radioactivity & its Applications Nuclear Chemistry Nuclear Chemistry deals with changes in the nucleus The nucleus of an atom contains Protons Positively Charged
Low-mass Stellar Evolution
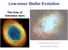 Low-mass Stellar Evolution The lives of low-mass stars And the lives of massive stars The Structure of the Sun Let s review: The Sun is held together by? The inward force is balanced by? Thinking about
Low-mass Stellar Evolution The lives of low-mass stars And the lives of massive stars The Structure of the Sun Let s review: The Sun is held together by? The inward force is balanced by? Thinking about
Energy Changes in Chemical and Nuclear Reactions
 Energy Changes in Chemical and Nuclear Reactions What happens when matter undergoes change? Clearly, new substances or states are produced, but energy changes also occur. If chemistry is the study of matter
Energy Changes in Chemical and Nuclear Reactions What happens when matter undergoes change? Clearly, new substances or states are produced, but energy changes also occur. If chemistry is the study of matter
Forces and Nuclear Processes
 Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
Forces and Nuclear Processes To understand how stars generate the enormous amounts of light they produce will require us to delve into a wee bit of physics. First we will examine the forces that act at
THE GASEOUS STATE OF MATTER
 THE GASEOUS STATE OF MATTER The gaseous state of matter is a form of matter in which the particles are in a high state of energy, which causes them to vibrate rapidly, experiencing a strong repulsion among
THE GASEOUS STATE OF MATTER The gaseous state of matter is a form of matter in which the particles are in a high state of energy, which causes them to vibrate rapidly, experiencing a strong repulsion among
Fundamental Forces of the Universe
 Fundamental Forces of the Universe There are four fundamental forces, or interactions in nature. Strong nuclear Electromagnetic Weak nuclear Gravitational Strongest Weakest Strong nuclear force Holds the
Fundamental Forces of the Universe There are four fundamental forces, or interactions in nature. Strong nuclear Electromagnetic Weak nuclear Gravitational Strongest Weakest Strong nuclear force Holds the
Energy Basics First Law of Thermodynamics and Energy Transformations Integrated Science 4 Honors Name: Per:
 Energy Basics First Law of Thermodynamics and Energy Transformations Integrated Science 4 Honors Name: Per: To understand how natural and human-built systems work, we must clearly define what matter and
Energy Basics First Law of Thermodynamics and Energy Transformations Integrated Science 4 Honors Name: Per: To understand how natural and human-built systems work, we must clearly define what matter and
Term 3 Week 2 Nuclear Fusion & Nuclear Fission
 Term 3 Week 2 Nuclear Fusion & Nuclear Fission Tuesday, November 04, 2014 Nuclear Fusion To understand nuclear fusion & fission Nuclear Fusion Why do stars shine? Stars release energy as a result of fusing
Term 3 Week 2 Nuclear Fusion & Nuclear Fission Tuesday, November 04, 2014 Nuclear Fusion To understand nuclear fusion & fission Nuclear Fusion Why do stars shine? Stars release energy as a result of fusing
turbine (a) (i) Which part of the power station provides thermal (heat) energy from a chain reaction?
 Nuclear fission and radiation 1 The diagram shows parts of a nuclear power station. control rods boiler steam generator electricity out turbine condenser nuclear reactor (a) (i) Which part of the power
Nuclear fission and radiation 1 The diagram shows parts of a nuclear power station. control rods boiler steam generator electricity out turbine condenser nuclear reactor (a) (i) Which part of the power
15.1 Energy and Its Forms. Energy and Work. How are energy and work related? Energy is the ability to do work. Work is a transfer of energy.
 Energy and Work How are energy and work related? Energy is the ability to do work. Work is a transfer of energy. Energy and Work Energy has different forms. A. The sun gives off energy in the form of heat
Energy and Work How are energy and work related? Energy is the ability to do work. Work is a transfer of energy. Energy and Work Energy has different forms. A. The sun gives off energy in the form of heat
Physical Science DCI Progression Chart
 DCI Progression Chart PS1: Matter and Its Interactions Grade Bands PS1.A Structure & Properties of Matter Grades K-2 Grades 3-5 Grades 6-8 Grades 9-12 Second Grade * Different kinds of matter exist and
DCI Progression Chart PS1: Matter and Its Interactions Grade Bands PS1.A Structure & Properties of Matter Grades K-2 Grades 3-5 Grades 6-8 Grades 9-12 Second Grade * Different kinds of matter exist and
7.6(B) distinguish between physical and chemical changes in matter in the digestive system; and
 7.6(B) distinguish between physical and chemical changes in matter in the digestive system; and 7.7(B) illustrate the transformation of energy within an organism such as the transfer from chemical energy
7.6(B) distinguish between physical and chemical changes in matter in the digestive system; and 7.7(B) illustrate the transformation of energy within an organism such as the transfer from chemical energy
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY. Instructor: Kazumi Tolich
 LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
LECTURE 25 NUCLEAR STRUCTURE AND STABILITY Instructor: Kazumi Tolich Lecture 25 2 30.1 Nuclear structure Isotopes Atomic mass 30.2 Nuclear stability Biding energy 30.3 Forces and energy in the nucleus
(b) The type of matter is irrelevant since the energy is directly proportional to mass only
 Exercise J.3.1. Answers 1. m = 800kg v = 70kmh -1 = 70 103 = 60 60 19.4ms-1 KK. EE = 1 2 mmvv2 = 800 19.42 2 = 150544JJ Using E= mc 2 and the kinetic energy of the car we obtain mm = EE cc 2 = 151235 (3
Exercise J.3.1. Answers 1. m = 800kg v = 70kmh -1 = 70 103 = 60 60 19.4ms-1 KK. EE = 1 2 mmvv2 = 800 19.42 2 = 150544JJ Using E= mc 2 and the kinetic energy of the car we obtain mm = EE cc 2 = 151235 (3
Chapter 14 Our Star Pearson Education, Inc.
 Chapter 14 Our Star Basic Types of Energy Kinetic (motion) Radiative (light) Potential (stored) Energy can change type, but cannot be created or destroyed. Thermal Energy: the collective kinetic energy
Chapter 14 Our Star Basic Types of Energy Kinetic (motion) Radiative (light) Potential (stored) Energy can change type, but cannot be created or destroyed. Thermal Energy: the collective kinetic energy
1. Four different processes are described in List A. The names of these processes are given in List B.
 Nuclear fission and nuclear fusion 1. Four different processes are described in List A. The names of these processes are given in List B. Draw a line to link each description in List A to its correct name
Nuclear fission and nuclear fusion 1. Four different processes are described in List A. The names of these processes are given in List B. Draw a line to link each description in List A to its correct name
Energy: Forms and Changes
 Energy: Forms and Changes Nature of Energy Energy is all around you! You can hear energy as sound. You can see energy as light. And you can feel it as wind. Nature of Energy You use energy when you: hit
Energy: Forms and Changes Nature of Energy Energy is all around you! You can hear energy as sound. You can see energy as light. And you can feel it as wind. Nature of Energy You use energy when you: hit
HW9 Concepts. Alex Alemi November 1, 2009
 HW9 Concepts Alex Alemi November 1, 2009 1 24.28 Capacitor Energy You are told to consider connecting a charged capacitor together with an uncharged one and told to compute (a) the original charge, (b)
HW9 Concepts Alex Alemi November 1, 2009 1 24.28 Capacitor Energy You are told to consider connecting a charged capacitor together with an uncharged one and told to compute (a) the original charge, (b)
Stellar Evolution. The lives of low-mass stars. And the lives of massive stars
 Stellar Evolution The lives of low-mass stars And the lives of massive stars The Structure of the Sun Let s review: The Sun is held together by what force? The inward force is balanced by what other force?
Stellar Evolution The lives of low-mass stars And the lives of massive stars The Structure of the Sun Let s review: The Sun is held together by what force? The inward force is balanced by what other force?
What is Energy? In science, energy is the ability to do work. Work is done when a force causes an object to move in the direction of the force.
 What is Energy? In science, energy is the ability to do work. Work is done when a force causes an object to move in the direction of the force. Energy Energy is the ability to do work. (reminder=what is
What is Energy? In science, energy is the ability to do work. Work is done when a force causes an object to move in the direction of the force. Energy Energy is the ability to do work. (reminder=what is
Conceptual Questions. Fig.8.51 EXERCISES. 8. Why can t electric field lines cross? 9. In which direction do charges always move in an electric field?
 EXERCISES Conceptual Questions 1. Explain why a neutral object can be attracted to a charged object. Why can this neutral object not be repelled by a charged object? 2. What is the function of an electroscope?
EXERCISES Conceptual Questions 1. Explain why a neutral object can be attracted to a charged object. Why can this neutral object not be repelled by a charged object? 2. What is the function of an electroscope?
A is called the mass number gives, roughly, the mass of the nucleus or atom in atomic mass units = amu = u
 5/5 A is called the mass number gives, roughly, the mass of the nucleus or atom in atomic mass units = amu = u The number of neutrons in the nucleus is given by the symbol N. Clearly, N = A Z. Isotope:
5/5 A is called the mass number gives, roughly, the mass of the nucleus or atom in atomic mass units = amu = u The number of neutrons in the nucleus is given by the symbol N. Clearly, N = A Z. Isotope:
Energy. Potential Kinetic
 Energy the ability to do work or cause change typically expressed in units of joules (J) can be transferred from one object to another two general types: Potential Kinetic Potential Energy (PE) stored
Energy the ability to do work or cause change typically expressed in units of joules (J) can be transferred from one object to another two general types: Potential Kinetic Potential Energy (PE) stored
Nuclear Physics and Nuclear Reactions
 Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
Slide 1 / 33 Nuclear Physics and Nuclear Reactions The Nucleus Slide 2 / 33 Proton: The charge on a proton is +1.6x10-19 C. The mass of a proton is 1.6726x10-27 kg. Neutron: The neutron is neutral. The
[2] State in what form the energy is released in such a reaction.... [1]
![[2] State in what form the energy is released in such a reaction.... [1] [2] State in what form the energy is released in such a reaction.... [1]](/thumbs/87/96058923.jpg) (a) The following nuclear reaction occurs when a slow-moving neutron is absorbed by an isotope of uranium-35. 0n + 35 9 U 4 56 Ba + 9 36Kr + 3 0 n Explain how this reaction is able to produce energy....
(a) The following nuclear reaction occurs when a slow-moving neutron is absorbed by an isotope of uranium-35. 0n + 35 9 U 4 56 Ba + 9 36Kr + 3 0 n Explain how this reaction is able to produce energy....
Figure 1. Page 1 of 6
 Page 1 of 6 Physical Science Unit 1 Test. Atomic & Nuclear Theory: Kinetic Molecular Theory: Gas Laws 1. According to Charles Law, if you have a balloon inside a car at noon during a hot summer day the
Page 1 of 6 Physical Science Unit 1 Test. Atomic & Nuclear Theory: Kinetic Molecular Theory: Gas Laws 1. According to Charles Law, if you have a balloon inside a car at noon during a hot summer day the
10/17/ A Closer Look at the Sun. Chapter 11: Our Star. Why does the Sun shine? Lecture Outline
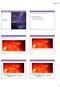 Lecture Outline 11.1 A Closer Look at the Sun Chapter 11: Our Star Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on FIRE? Chemical
Lecture Outline 11.1 A Closer Look at the Sun Chapter 11: Our Star Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on FIRE? Chemical
Name Class Date. Energy Energy
 CHAPTER 13 3 What Is Energy? SECTION Work and Energy KEY IDEAS As you read this section, keep these questions in mind: How are energy and work related? Why is potential energy called energy of position?
CHAPTER 13 3 What Is Energy? SECTION Work and Energy KEY IDEAS As you read this section, keep these questions in mind: How are energy and work related? Why is potential energy called energy of position?
ASTR 150. Last time: Mitigation Today: The Sun. Homework 4 due Monday night Night Observing starts next week. Music: Sonne Rammstein
 ASTR 150 Homework 4 due Monday night Night Observing starts next week Last time: Mitigation Today: The Sun Music: Sonne Rammstein 1 Imagine After being dropped into suspended animation in a Pizza accident
ASTR 150 Homework 4 due Monday night Night Observing starts next week Last time: Mitigation Today: The Sun Music: Sonne Rammstein 1 Imagine After being dropped into suspended animation in a Pizza accident
Stellar energy generation on the main sequence
 Stellar energy generation on the main sequence Once fusion reactions begin at the center of a cloud of gas, we call the object a star. For the bulk of its lifetime, a star fuses hydrogen into helium in
Stellar energy generation on the main sequence Once fusion reactions begin at the center of a cloud of gas, we call the object a star. For the bulk of its lifetime, a star fuses hydrogen into helium in
ELEMENTS OF PHYSICS ENERGY: WORK and POWER
 1 Pre-Test Directions: This will help you discover what you know about the subject of energy before you begin this lesson. Answer the following true or false. 1. To a physicist, energy is work or the capacity
1 Pre-Test Directions: This will help you discover what you know about the subject of energy before you begin this lesson. Answer the following true or false. 1. To a physicist, energy is work or the capacity
Today. Homework Due. Stars. Properties (Recap) Nuclear Reactions. proton-proton chain. CNO cycle. Stellar Lifetimes
 Today Stars Properties (Recap) Nuclear Reactions proton-proton chain CNO cycle Stellar Lifetimes Homework Due Stellar Properties Luminosity Surface Temperature Size Mass Composition Stellar Properties
Today Stars Properties (Recap) Nuclear Reactions proton-proton chain CNO cycle Stellar Lifetimes Homework Due Stellar Properties Luminosity Surface Temperature Size Mass Composition Stellar Properties
PhysicsAndMathsTutor.com 1
 PhysicsAndMathsTutor.com 1 1. Describe briefly one scattering experiment to investigate the size of the nucleus of the atom. Include a description of the properties of the incident radiation which makes
PhysicsAndMathsTutor.com 1 1. Describe briefly one scattering experiment to investigate the size of the nucleus of the atom. Include a description of the properties of the incident radiation which makes
Ch Radioactivity. Henry Becquerel, using U-238, discovered the radioactive nature of elements in 1896.
 Ch. 10 - Radioactivity Henry Becquerel, using U-238, discovered the radioactive nature of elements in 1896. Radioactivity the process in which an unstable atomic nucleus emits charged particles and energy
Ch. 10 - Radioactivity Henry Becquerel, using U-238, discovered the radioactive nature of elements in 1896. Radioactivity the process in which an unstable atomic nucleus emits charged particles and energy
The Story of Energy. Forms and Functions
 The Story of Energy Forms and Functions What are 5 things E helps us do? Batteries store energy! This car uses a lot of energy Even this sleeping puppy is using stored energy. We get our energy from FOOD!
The Story of Energy Forms and Functions What are 5 things E helps us do? Batteries store energy! This car uses a lot of energy Even this sleeping puppy is using stored energy. We get our energy from FOOD!
Jacob s Ladder Controlling Lightning
 Host: Fusion specialist: Jacob s Ladder Controlling Lightning PART 1 Jacob s ladder demonstration Video Teacher resources Phil Dooley European Fusion Development Agreement Peter de Vries European Fusion
Host: Fusion specialist: Jacob s Ladder Controlling Lightning PART 1 Jacob s ladder demonstration Video Teacher resources Phil Dooley European Fusion Development Agreement Peter de Vries European Fusion
The Twinkle in Mother Earth s Eye: Promising fusion power. What if you could have a miniature star powering your house, your computer, and your car?
 The Twinkle in Mother Earth s Eye: Promising fusion power What if you could have a miniature star powering your house, your computer, and your car? How cool would that be! Stars produce a lot of energy,
The Twinkle in Mother Earth s Eye: Promising fusion power What if you could have a miniature star powering your house, your computer, and your car? How cool would that be! Stars produce a lot of energy,
AST 301 Introduction to Astronomy
 AST 301 Introduction to Astronomy John Lacy RLM 16.332 471-1469 lacy@astro.as.utexas.edu Myoungwon Jeon RLM 16.216 471-0445 myjeon@astro.as.utexas.edu Bohua Li RLM 16.212 471-8443 bohuali@astro.as.utexas.edu
AST 301 Introduction to Astronomy John Lacy RLM 16.332 471-1469 lacy@astro.as.utexas.edu Myoungwon Jeon RLM 16.216 471-0445 myjeon@astro.as.utexas.edu Bohua Li RLM 16.212 471-8443 bohuali@astro.as.utexas.edu
Energy: Forms and Changes
 Energy: Forms and Changes The Energy Story Nature of Energy Energy is all around you! l You can hear energy as sound. l You can see energy as light. l And you can feel it as wind. Nature of Energy You
Energy: Forms and Changes The Energy Story Nature of Energy Energy is all around you! l You can hear energy as sound. l You can see energy as light. l And you can feel it as wind. Nature of Energy You
3 Types of Heat Transfer
 3 Types of Heat Transfer The movement of heat from a warmer object to a cooler object. Heat Transfer- 1. Conduction Heat transfer by direct contact of molecules. In other words, when one molecule runs
3 Types of Heat Transfer The movement of heat from a warmer object to a cooler object. Heat Transfer- 1. Conduction Heat transfer by direct contact of molecules. In other words, when one molecule runs
Birth and Death of Stars. Birth of Stars. Gas and Dust Clouds. Astronomy 110 Class 11
 Birth and Death of Stars Astronomy 110 Class 11 Birth of Stars Start in cloud of gas and dust Contraction and Fragmentation Gravitational collapse and heating Protostar and disk Main Sequence Star Gas
Birth and Death of Stars Astronomy 110 Class 11 Birth of Stars Start in cloud of gas and dust Contraction and Fragmentation Gravitational collapse and heating Protostar and disk Main Sequence Star Gas
Conceptual Physics. Luis A. Anchordoqui. Department of Physics and Astronomy Lehman College, City University of New York. Lesson II September 5, 2017
 Conceptual Physics Luis A. Anchordoqui Department of Physics and Astronomy Lehman College, City University of New York Lesson II September 5, 2017 https://arxiv.org/abs/1711.07445 L. A. Anchordoqui (CUNY)
Conceptual Physics Luis A. Anchordoqui Department of Physics and Astronomy Lehman College, City University of New York Lesson II September 5, 2017 https://arxiv.org/abs/1711.07445 L. A. Anchordoqui (CUNY)
Radioactivity pp Topic 9: Nuclear Physics Ch. 10. Radioactivity. Radioactivity
 Topic 9: Nuclear Physics Ch. 10 pp.244-249 results from radioactive decay, which is the process in which unstable atomic nuclei transform and emit radiation. has existed longer than the human race. Unstable
Topic 9: Nuclear Physics Ch. 10 pp.244-249 results from radioactive decay, which is the process in which unstable atomic nuclei transform and emit radiation. has existed longer than the human race. Unstable
s Traditionally, we use the calorie as a unit of energy. The nutritional Calorie, Cal = 1000 cal. Kinetic Energy and Potential Energy
 AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
AP Chemistry: Thermochemistry Lecture Outline 5.1 The Nature of Energy Thermodynamics is the study of energy and its transformations. Thermochemistry is the study of the relationships between chemical
Friday, 05/06/16 6) HW QUIZ MONDAY Learning Target (NEW)
 Friday, 05/06/16 1) Warm-up: If you start with 100g of a radioactive substance, how much will be left after 3 half-lives? 2) Review HW & Nuclear Notes 3) Complete Modeling Energy Investigation 4) Complete:
Friday, 05/06/16 1) Warm-up: If you start with 100g of a radioactive substance, how much will be left after 3 half-lives? 2) Review HW & Nuclear Notes 3) Complete Modeling Energy Investigation 4) Complete:
Nicholas J. Giordano. Chapter 30. Nuclear Physics. Marilyn Akins, PhD Broome Community College
 Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 30 Nuclear Physics Marilyn Akins, PhD Broome Community College Atomic Nuclei Rutherford s discovery of the atomic nucleus caused scientists
Nicholas J. Giordano www.cengage.com/physics/giordano Chapter 30 Nuclear Physics Marilyn Akins, PhD Broome Community College Atomic Nuclei Rutherford s discovery of the atomic nucleus caused scientists
Nuclear Fusion 1 of 24 Boardworks Ltd 2011
 Nuclear Fusion 1 of 24 Boardworks Ltd 2011 2 of 24 Boardworks Ltd 2011 How do we get energy from atoms? 3 of 24 Boardworks Ltd 2011 Energy is produced from atoms in power stations using the process of
Nuclear Fusion 1 of 24 Boardworks Ltd 2011 2 of 24 Boardworks Ltd 2011 How do we get energy from atoms? 3 of 24 Boardworks Ltd 2011 Energy is produced from atoms in power stations using the process of
Chapter 14 Lecture. Chapter 14: Our Star Pearson Education, Inc.
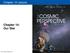 Chapter 14 Lecture Chapter 14: Our Star 14.1 A Closer Look at the Sun Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on FIRE?
Chapter 14 Lecture Chapter 14: Our Star 14.1 A Closer Look at the Sun Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on FIRE?
Energy: Forms and Changes
 Energy: Forms and Changes Nature of Energy Energy is all around you! You can hear energy as sound. You can see energy as light. And you can feel it as wind. Nature of Energy You use energy when you: hit
Energy: Forms and Changes Nature of Energy Energy is all around you! You can hear energy as sound. You can see energy as light. And you can feel it as wind. Nature of Energy You use energy when you: hit
10/18/ A Closer Look at the Sun. Chapter 11: Our Star. Why does the Sun shine? Lecture Outline
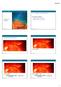 10/18/17 Lecture Outline 11.1 A Closer Look at the Sun Chapter 11: Our Star Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on
10/18/17 Lecture Outline 11.1 A Closer Look at the Sun Chapter 11: Our Star Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is it on FIRE? Is it on
The Sun Closest star to Earth - only star that we can see details on surface - easily studied Assumption: The Sun is a typical star
 The Sun Closest star to Earth - only star that we can see details on surface - easily studied Assumption: The Sun is a typical star Why is the Sun hot and bright? Surface Temperature of the Sun: T =
The Sun Closest star to Earth - only star that we can see details on surface - easily studied Assumption: The Sun is a typical star Why is the Sun hot and bright? Surface Temperature of the Sun: T =
Lecture 31 Chapter 22, Sections 3-5 Nuclear Reactions. Nuclear Decay Kinetics Fission Reactions Fusion Reactions
 Lecture Chapter, Sections -5 Nuclear Reactions Nuclear Decay Kinetics Fission Reactions Fusion Reactions Gamma Radiation Electromagnetic photons of very high energy Very penetrating can pass through the
Lecture Chapter, Sections -5 Nuclear Reactions Nuclear Decay Kinetics Fission Reactions Fusion Reactions Gamma Radiation Electromagnetic photons of very high energy Very penetrating can pass through the
Chapter 14 Our Star A Closer Look at the Sun. Why was the Sun s energy source a major mystery?
 Chapter 14 Our Star 14.1 A Closer Look at the Sun Our goals for learning Why was the Sun s energy source a major mystery? Why does the Sun shine? What is the Sun s structure? Why was the Sun s energy source
Chapter 14 Our Star 14.1 A Closer Look at the Sun Our goals for learning Why was the Sun s energy source a major mystery? Why does the Sun shine? What is the Sun s structure? Why was the Sun s energy source
 http://imgs.xkcd.com/comics/cell_phones.png Announcements: Thursday: Group A to Hayes 105 for hands-on experience Group B comes here to work on worksheet Both groups: bring your calculator! Next Tuesday
http://imgs.xkcd.com/comics/cell_phones.png Announcements: Thursday: Group A to Hayes 105 for hands-on experience Group B comes here to work on worksheet Both groups: bring your calculator! Next Tuesday
A Closer Look at the Sun
 Our Star A Closer Look at the Sun Our goals for learning Why was the Sun s energy source a major mystery? Why does the Sun shine? What is the Sun s structure? Why was the Sun s energy source a major mystery?
Our Star A Closer Look at the Sun Our goals for learning Why was the Sun s energy source a major mystery? Why does the Sun shine? What is the Sun s structure? Why was the Sun s energy source a major mystery?
Highlights of chapter 5, section 2
 Highlights of chapter 5, section 2 2 Conservation of Energy Changing Forms of Energy There are situations around us every day that involve energy changing from one form to another form. There are many
Highlights of chapter 5, section 2 2 Conservation of Energy Changing Forms of Energy There are situations around us every day that involve energy changing from one form to another form. There are many
1 A Solar System Is Born
 CHAPTER 16 1 A Solar System Is Born SECTION Our Solar System California Science Standards 8.2.g, 8.4.b, 8.4.c, 8.4.d BEFORE YOU READ After you read this section, you should be able to answer these questions:
CHAPTER 16 1 A Solar System Is Born SECTION Our Solar System California Science Standards 8.2.g, 8.4.b, 8.4.c, 8.4.d BEFORE YOU READ After you read this section, you should be able to answer these questions:
Card #1/28. Card #2/28. Science Revision P2. Science Revision P2. Science Revision P2. Card #4/28. Topic: F = ma. Topic: Resultant Forces
 Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Card #1/28 Card #2/28 Topic: Resultant Forces Topic: F = ma Topic: Distance-TIme Graphs Card #3/28 Card #4/28 Topic: Velocity-Time Graphs Card #2/28 Card #1/28 Card #4/28 Card #3/28 Card #5/28 Card #6/28
Physical Science Study Guide
 Name: Class: Date: Physical Science Study Guide Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The electrons in a water molecule are gathered nearest
Name: Class: Date: Physical Science Study Guide Multiple Choice Identify the choice that best completes the statement or answers the question. 1. The electrons in a water molecule are gathered nearest
What colour is the insulation around the wire connected to the live pin inside the plug? ... (1) (1)
 Q. Diagram shows a hairdryer. Diagram 2 shows how the heaters and fan of the hairdryer are connected to a 3-pin plug. The hairdryer does not have an earth wire. (a) What colour is the insulation around
Q. Diagram shows a hairdryer. Diagram 2 shows how the heaters and fan of the hairdryer are connected to a 3-pin plug. The hairdryer does not have an earth wire. (a) What colour is the insulation around
Chapter 8: E & M (Electricity & Magnetism or Electromagnetism)
 Chapter 8: E & M (Electricity & Magnetism or Electromagnetism) Electric charge & electric force Coulomb s Law Electrons & basic facts about atoms (mainly review) Charge conservation Electric current &
Chapter 8: E & M (Electricity & Magnetism or Electromagnetism) Electric charge & electric force Coulomb s Law Electrons & basic facts about atoms (mainly review) Charge conservation Electric current &
UNIT 2 MECHANICS CHAPTER 6 ENERGY
 UNIT 2 MECHANICS CHAPTER 6 ENERGY Chapter 6A The Nature of Energy Objectives: Discuss the importance of energy State what energy can do and the units in which energy is measured Define mechanical work
UNIT 2 MECHANICS CHAPTER 6 ENERGY Chapter 6A The Nature of Energy Objectives: Discuss the importance of energy State what energy can do and the units in which energy is measured Define mechanical work
Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes
 St Ninian s High School Chemistry Department National 5 Chemistry Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes Name Learning Outcomes After completing this topic you should be able to :
St Ninian s High School Chemistry Department National 5 Chemistry Unit 3: Chemistry in Society Nuclear Chemistry Summary Notes Name Learning Outcomes After completing this topic you should be able to :
Chapter 10 Section 4 Notes
 Chapter 10 Section 4 Notes This painting of an alchemist s laboratory was made around 1570. For centuries, these early scientists, known as alchemists, tried to use chemical reactions to make gold. The
Chapter 10 Section 4 Notes This painting of an alchemist s laboratory was made around 1570. For centuries, these early scientists, known as alchemists, tried to use chemical reactions to make gold. The
Today. Finish Ch. 6 on Momentum Start Ch. 7 on Energy
 Today Finish Ch. 6 on Momentum Start Ch. 7 on Energy Next three lectures (Sep 16, 20, 23) : Energy (Ch7) and Rotation (Ch.8) will be taught by Dr. Yonatan Abranyos, as I will be away at a research conference
Today Finish Ch. 6 on Momentum Start Ch. 7 on Energy Next three lectures (Sep 16, 20, 23) : Energy (Ch7) and Rotation (Ch.8) will be taught by Dr. Yonatan Abranyos, as I will be away at a research conference
Electrocution (large quantities of charge flow through the body to earth) Insulating mats or shoes
 P4 in 30 minutes P4a: Sparks Like charges repel and unlike charges attract. a positive charge due to lack of electrons a negative charge due to an excess of electrons. Atoms or molecules that have become
P4 in 30 minutes P4a: Sparks Like charges repel and unlike charges attract. a positive charge due to lack of electrons a negative charge due to an excess of electrons. Atoms or molecules that have become
Quantum Mechanics. Exam 3. Photon(or electron) interference? Photoelectric effect summary. Using Quantum Mechanics. Wavelengths of massive objects
 Exam 3 Hour Exam 3: Wednesday, November 29th In-class, Quantum Physics and Nuclear Physics Twenty multiple-choice questions Will cover:chapters 13, 14, 15 and 16 Lecture material You should bring 1 page
Exam 3 Hour Exam 3: Wednesday, November 29th In-class, Quantum Physics and Nuclear Physics Twenty multiple-choice questions Will cover:chapters 13, 14, 15 and 16 Lecture material You should bring 1 page
Mass number i. Example U (uranium 235) and U (uranium 238) atomic number e. Average atomic mass weighted of the isotopes of that element i.
 CP NT Ch. 4&25 I. Atomic Theory and Structure of the Atom a. Democritus all matter consists of very small, indivisible particles, which he named i. Atom smallest particle of an element that retains all
CP NT Ch. 4&25 I. Atomic Theory and Structure of the Atom a. Democritus all matter consists of very small, indivisible particles, which he named i. Atom smallest particle of an element that retains all
Name Class Date. What are the four fundamental forces in nature? How can forces affect the motion of an object? Why is friction sometime necessary?
 CHAPTER 11 SECTION Motion 3 Motion and Force KEY IDEAS As you read this section, keep these questions in mind: What are the four fundamental forces in nature? How can forces affect the motion of an object?
CHAPTER 11 SECTION Motion 3 Motion and Force KEY IDEAS As you read this section, keep these questions in mind: What are the four fundamental forces in nature? How can forces affect the motion of an object?
TYPES OF ENERGY and Energy
 TYPES OF ENERGY and Energy Conversions Mechanical, Electromagnetic, Electrical, Chemical, Nuclear, and Thermal What is Mechanical Energy? o Energy due to an object s motion (kinetic) or position (potential).
TYPES OF ENERGY and Energy Conversions Mechanical, Electromagnetic, Electrical, Chemical, Nuclear, and Thermal What is Mechanical Energy? o Energy due to an object s motion (kinetic) or position (potential).
LIGHT and SPECIAL RELATIVITY RELATIVISTIC MASS, MOMENTUM and ENERGY
 VISUAL PHYSICS ONLINE MODULE 7 NATURE OF LIGHT LIGHT and SPECIAL RELATIVITY RELATIVISTIC MASS, MOMENTUM and ENERGY Einstein s 1 st postulate states that the laws of physics are the same for all observers
VISUAL PHYSICS ONLINE MODULE 7 NATURE OF LIGHT LIGHT and SPECIAL RELATIVITY RELATIVISTIC MASS, MOMENTUM and ENERGY Einstein s 1 st postulate states that the laws of physics are the same for all observers
11/19/08. Gravitational equilibrium: The outward push of pressure balances the inward pull of gravity. Weight of upper layers compresses lower layers
 Gravitational equilibrium: The outward push of pressure balances the inward pull of gravity Weight of upper layers compresses lower layers Gravitational equilibrium: Energy provided by fusion maintains
Gravitational equilibrium: The outward push of pressure balances the inward pull of gravity Weight of upper layers compresses lower layers Gravitational equilibrium: Energy provided by fusion maintains
Today. Stars. Properties (Recap) Binaries. Stellar Lifetimes
 Today Stars Properties (Recap) Binaries Stellar Lifetimes 1 Exam Review Session This Tuesday, 6-8 PM, PHYS 1410 (the large lecture hall next to ours) Completely driven by your questions! The TAs will not
Today Stars Properties (Recap) Binaries Stellar Lifetimes 1 Exam Review Session This Tuesday, 6-8 PM, PHYS 1410 (the large lecture hall next to ours) Completely driven by your questions! The TAs will not
Chapter 14 Lecture. The Cosmic Perspective Seventh Edition. Our Star Pearson Education, Inc.
 Chapter 14 Lecture The Cosmic Perspective Seventh Edition Our Star 14.1 A Closer Look at the Sun Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is
Chapter 14 Lecture The Cosmic Perspective Seventh Edition Our Star 14.1 A Closer Look at the Sun Our goals for learning: Why does the Sun shine? What is the Sun's structure? Why does the Sun shine? Is
2 Energy from the Nucleus
 CHAPTER 4 2 Energy from the Nucleus SECTION Atomic Energy BEFORE YOU READ After you read this section, you should be able to answer these questions: What is nuclear fission? What is nuclear fusion? What
CHAPTER 4 2 Energy from the Nucleus SECTION Atomic Energy BEFORE YOU READ After you read this section, you should be able to answer these questions: What is nuclear fission? What is nuclear fusion? What
The SI unit for Energy is the joule, usually abbreviated J. One joule is equal to one kilogram meter squared per second squared:
 Chapter 2 Energy Energy is an extremely loaded term. It is used in everyday parlance to mean a number of different things, many of which bear at most a passing resemblance to the term as used in physical
Chapter 2 Energy Energy is an extremely loaded term. It is used in everyday parlance to mean a number of different things, many of which bear at most a passing resemblance to the term as used in physical
There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart?
 Question 32.1 The Nucleus There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart? a) Coulomb repulsive force doesn t act inside the nucleus b) gravity overpowers the Coulomb repulsive
Question 32.1 The Nucleus There are 82 protons in a lead nucleus. Why doesn t the lead nucleus burst apart? a) Coulomb repulsive force doesn t act inside the nucleus b) gravity overpowers the Coulomb repulsive
Nuclear Physics 2. D. atomic energy levels. (1) D. scattered back along the original direction. (1)
 Name: Date: Nuclear Physics 2. Which of the following gives the correct number of protons and number of neutrons in the nucleus of B? 5 Number of protons Number of neutrons A. 5 6 B. 5 C. 6 5 D. 5 2. The
Name: Date: Nuclear Physics 2. Which of the following gives the correct number of protons and number of neutrons in the nucleus of B? 5 Number of protons Number of neutrons A. 5 6 B. 5 C. 6 5 D. 5 2. The
Atomic Motion and Interactions
 Atomic Motion and Interactions 1. Handout: Unit Notes 2. Have You Seen an Atom Lately? 1. Lab: Oil Spreading on Water 2. Demo: Computer animation of spreading oil 3. Lab: Mixing Alcohol and Water 4. Demo:
Atomic Motion and Interactions 1. Handout: Unit Notes 2. Have You Seen an Atom Lately? 1. Lab: Oil Spreading on Water 2. Demo: Computer animation of spreading oil 3. Lab: Mixing Alcohol and Water 4. Demo:
What is it? What are the different types?
 What is it? What are the different types? Energy definition Energy is a challenging concept to understand and explain Energy is an abstract concept that has slightly different definitions depending on
What is it? What are the different types? Energy definition Energy is a challenging concept to understand and explain Energy is an abstract concept that has slightly different definitions depending on
Matter and Energy. Section 2.1 Chapter 2. Representations of Matter: Models and Symbols. Goal 1. Goal 2
 Section 2.1 Chapter 2 Matter and Energy Representations of Matter: Models and Symbols Goal 1 Goal 2 Identify and explain the difference among observations of matter at the macroscopic, microscopic, and
Section 2.1 Chapter 2 Matter and Energy Representations of Matter: Models and Symbols Goal 1 Goal 2 Identify and explain the difference among observations of matter at the macroscopic, microscopic, and
L-35 Modern Physics-3 Nuclear Physics 29:006 FINAL EXAM. Structure of the nucleus. The atom and the nucleus. Nuclear Terminology
 9:006 FINAL EXAM L-5 Modern Physics- Nuclear Physics The final exam is on Monday MAY 7:0 AM - 9:0 AM in W90 CB The FE is not cumulative, and will cover lectures through 6. (50 questions) The last regular
9:006 FINAL EXAM L-5 Modern Physics- Nuclear Physics The final exam is on Monday MAY 7:0 AM - 9:0 AM in W90 CB The FE is not cumulative, and will cover lectures through 6. (50 questions) The last regular
Nuclear Chemistry. The Nucleus. Isotopes. Slide 1 / 43. Slide 2 / 43. Slide 3 / 43
 Slide 1 / 43 Nuclear Chemistry The Nucleus Slide 2 / 43 Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons
Slide 1 / 43 Nuclear Chemistry The Nucleus Slide 2 / 43 Remember that the nucleus is comprised of the two nucleons, protons and neutrons. The number of protons is the atomic number. The number of protons
London Examinations IGCSE
 Centre No. Candidate No. Paper Reference(s) 4420/2H London Examinations IGCSE Physics Paper 2H Higher Tier Tuesday 9 November 2010 Morning Time: 2 hours Materials required for examination Nil Paper Reference
Centre No. Candidate No. Paper Reference(s) 4420/2H London Examinations IGCSE Physics Paper 2H Higher Tier Tuesday 9 November 2010 Morning Time: 2 hours Materials required for examination Nil Paper Reference
Nuclear Energy Learning Outcomes
 1 Nuclear Energy Learning Outcomes Describe the principles underlying fission and fusion. Interpret nuclear reactions. Discuss nuclear weapons. Describe the structure and operation of a nuclear reactor.
1 Nuclear Energy Learning Outcomes Describe the principles underlying fission and fusion. Interpret nuclear reactions. Discuss nuclear weapons. Describe the structure and operation of a nuclear reactor.
Nuclear Energy Learning Outcomes. Nuclear Fission. Chain Reaction
 by fastfission public domain by fastfission public domain 1 Nuclear Energy Learning Outcomes Describe the principles underlying fission and fusion. Interpret nuclear reactions. Discuss nuclear weapons.
by fastfission public domain by fastfission public domain 1 Nuclear Energy Learning Outcomes Describe the principles underlying fission and fusion. Interpret nuclear reactions. Discuss nuclear weapons.
Transfers and Transformations
 Chapter Introduction Lesson 1 Lesson 2 Lesson 3 Forms of Energy Energy Transfers and Transformations Particles in Motion Chapter Wrap-Up What is the difference between energy transfers and energy transformations?
Chapter Introduction Lesson 1 Lesson 2 Lesson 3 Forms of Energy Energy Transfers and Transformations Particles in Motion Chapter Wrap-Up What is the difference between energy transfers and energy transformations?
The Sun. the main show in the solar system. 99.8% of the mass % of the energy. Homework due next time - will count best 5 of 6
 The Sun the main show in the solar system 99.8% of the mass 99.9999...% of the energy 2007 Pearson Education Inc., publishing as Pearson Addison-Wesley Homework due next time - will count best 5 of 6 The
The Sun the main show in the solar system 99.8% of the mass 99.9999...% of the energy 2007 Pearson Education Inc., publishing as Pearson Addison-Wesley Homework due next time - will count best 5 of 6 The
Lecture Outline. 5.1 The Nature of Energy. Kinetic Energy and Potential Energy. 1 mv
 Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
Chapter 5. Thermochemistry Common Student Misconceptions Students confuse power and energy. Students confuse heat with temperature. Students fail to note that the first law of thermodynamics is the law
TEACHER BACKGROUND INFORMATION FORCE
 TEACHER BACKGROUND INFORMATION FORCE WHAT IS FORCE? Force is anything that can change the state of motion of a body. In simpler terms, force is a push or a pull. For example, wind pushing on a flag is
TEACHER BACKGROUND INFORMATION FORCE WHAT IS FORCE? Force is anything that can change the state of motion of a body. In simpler terms, force is a push or a pull. For example, wind pushing on a flag is
Physics (B): Physics in Context
 Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Physics
Centre Number Surname Candidate Number For Examinerʼs Use Other Names Candidate Signature Examinerʼs Initials General Certificate of Education Advanced Level Examination June 2012 Question 1 2 Mark Physics
1ST SEM MT CHAP 22 REVIEW
 1ST SEM MT CHAP 22 REVIEW Multiple Choice Identify the choice that best completes the statement or answers the question. (CAPITAL LETTERS ONLY PLEASE) 1. Mass defect is the difference between the mass
1ST SEM MT CHAP 22 REVIEW Multiple Choice Identify the choice that best completes the statement or answers the question. (CAPITAL LETTERS ONLY PLEASE) 1. Mass defect is the difference between the mass
Matter and Energy. Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law.
 Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
Fission & Fusion Matter and Energy Previous studies have taught us that matter and energy cannot be created nor destroyed We balance equations to obey this law. 2 H 2 O 2 H 2 + O 2 We now need to understand
Inner Transition Metals
 1 Inner Transition Metals Inner Transition Metals Inner Transition Metals The inner transition metals are found in the f-block, usually put at the bottom of the Periodic Table. These elements were sometimes
1 Inner Transition Metals Inner Transition Metals Inner Transition Metals The inner transition metals are found in the f-block, usually put at the bottom of the Periodic Table. These elements were sometimes
Missing words: mass hydrogen burning electrostatic repulsion. gravitationally hydrogen temperature protostar
 Instructions Fill in the blanks of each frame using the list of missing words given. Cut out each frame and arrange them on your page in order, then stick them down. The first two frames are already in
Instructions Fill in the blanks of each frame using the list of missing words given. Cut out each frame and arrange them on your page in order, then stick them down. The first two frames are already in
P4 Key facts sheet: Key Fact
 A: Sparks P4 Key facts sheet: Insulating materials can become charged when rubbed with another insulating material due to friction. This causes a transfer of electrons, resulting in both materials becoming
A: Sparks P4 Key facts sheet: Insulating materials can become charged when rubbed with another insulating material due to friction. This causes a transfer of electrons, resulting in both materials becoming
Science Unit Test Grade: 8 Unit 6: Gravity
 Science Unit Test Grade: 8 Unit 6: Gravity Name Date 1. Which of these is best used to measure weight in newtons? a. Spring scale b. Triple beam balance c. Double pan balance d. Simple lever 2. Which of
Science Unit Test Grade: 8 Unit 6: Gravity Name Date 1. Which of these is best used to measure weight in newtons? a. Spring scale b. Triple beam balance c. Double pan balance d. Simple lever 2. Which of
