Supporting Information
|
|
|
- Andra Powers
- 6 years ago
- Views:
Transcription
1 Supporting Information Supplemental Figure 1: Demonstration of polyelectrolyte complex formation ability. After mixing solutions of PA and PSS separately dissolved in the respective alcohol, increasing turbidity of the mixture and precipitation of the insoluble polyelectrolyte complex was observed.
2 Supplemental Figure 2. Alternating zeta-potential as a function of layer number indicates stepwise absorption of polymer layer that exhibit opposite charges (A). Increasing fluorescence intensity with every deposition of fluorescent labeled polymer (Polyallylamine-FITC) indicates subsequent increase of deposited polymer (B).
3 Supplemental Figure 3: IR spectra of polyelectrolyte complexes and their components
4 IR spectroscopic investigation: IR spectra of sulfonic acids often resemble those of sulfonic acid salts due to hydration to hydronium salts under normal working conditions. However, distinct features in IR spectra were observed allowing conclusion of polymer interactions. In summary the components of the spectra for the polyelectrolyte complex assembled in water resembles more those of PA hydrochloride and that of PSS sodium and components of the spectra for the polyelectrolyte complex assembled from 1-butanol resembles more those from PA and PSS. A detailed outline of spectral differences between PSS and PSS sodium PA and PA hydrochloride, PSS/PA complexes assembled from water or 1-butanol can be found in the following. It is remarkable that the spectra of the PSS/PA complex formed in butanol still resemble properties of free polystyrenesulfonic acid even though amine groups are roughly in 2.4 times molar excess. Comparing the spectra from PSS/PA complex assembled from water and Butanol (Spectra A) it can be seen that the spectra for aqueous LBL in the region of 1800 cm -1 to 1400 cm -1 is similar to that of polyallylamine hydrochloride and that of RP-LbL similar to that of polyallylamine. From Spectra B, it can bee seen that the peak at 1500 cm -1 becomes more defined upon PSS and PA complex formation in 1-butanol compared to the spectra of the free base polyallylamine, indicating amine-hydro salt formation. Differences between polyallylamine and polyallylamine hydrochloride can be observed at 1630 cm -1 and 1500 cm -1 which are bend and deformation vibration for free base primary amines and primary ammino salts, respectively (Spectra C).The bend vibration peak of ammonia salts is significantly more defined for the spectra of the polyelectrolyte complex assembled from water (Spectra C). The peak at 1200 cm -1 is sharp for polystyrenesulfonic acid sodium salt but a symmetric shoulder for polystyrenesulfonic acid with maxima at 1220 cm -1 and 1177 cm -1. Furthermore the C-S stretch peak at 620 cm -1 is more defined for the salt form compared to that of the free acid. Comparing the spectra of both polystyrenesulfonic sodium salt and PSS/PA polyelectrolyte complex from water and polystyrenesulfonic acid and PSS/PA polyelectrolyte complex from butanol in the
5 area from 1800 cm cm -1, it is clear in both cases that the peaks of the polyelectrolyte complex are N-H bend or deformation vibration and not found in polystyrenesulfonic acid or polystyrenesulfonic acid sodium salt (Spectra B and D). Comparing the spectra of polystyrenesulfonic acid and polystyrenesulfonic acid sodium salt, (Spectra E) only a few differences can be seen. When comparing the spectra of PSS/PA polyelectrolyte complex from butanol and PSS/PA polyelectrolyte complex from water, it becomes clear that the peak at 620 cm -1 becomes more defined in the spectra of PSS/PA polyelectrolyte complex from butanol compared to the spectra of the polystyrenesulfonic acid but less defined compared to the spectra of sodium polystyrenesulfonate or that of PSS/PA polyelectrolyte complex from water. When comparing the peak at 1200 cm -1 it can be seen that the peak has still a slight shoulder form for PSS/PA polyelectrolyte complex from butanol but not in the spectra od PSS/PA complex assembled from water. By taking into account the molar ratio of ammine groups to sulfonic acid groups that was used to precipitate the PSS/PA polyelectrolyte complex.
6 2Layer 4Layer 6Layer 8Layer 10 Layer Supplemental Figure 4: Collapsed polyelectrolyte microcapsules, demonstrating layer dependent increase in average thickness in their corresponding cross sections. Images of single capsules are zoom in areas of larger pictures which results in the limited resolution depicted.
7 Electronic Supplementary Material (ESI) for Soft Matter Supplemental Figure 5. Fluorescent micrograph of (PA-FITC/PSA)2PA-FITC microcapsules dispersed in cell culture medium after successfully performed phase transfer protocol. The irregular crystal shape can still be recognized after glucose core dissolution. A B Supplemental Figure 6. Fluorescent micrograph showing internalized microcapsules of (PA-FITC/PSA)2PA-FITC into IMR 90 fibroblast by quenching extracellular fluorescence with 0.004% w/v Trypan blue solution (A). Overlayed fluorescence and bright field micrographs reveal partial internalization depending on location and size of microcapsules (B).
Preparation and Characterization of Hydrogels
 Chapter 2 Preparation and Characterization of Hydrogels 2.1 Materials and Methods 2.1.1 Materials 2-Acrylamido-2-methylpropane sulfonic acid (AMPS) was obtained from Vinati Organic Ltd. Acrylamide (AM),
Chapter 2 Preparation and Characterization of Hydrogels 2.1 Materials and Methods 2.1.1 Materials 2-Acrylamido-2-methylpropane sulfonic acid (AMPS) was obtained from Vinati Organic Ltd. Acrylamide (AM),
K + -Recognition Capsules with Squirting Release Mechanisms
 Supplementary Material (ESI) for Chemical Communications K + -Recognition Capsules with Squirting Release Mechanisms Supplementary Material Zhuang Liu, Li Liu, Xiao-Jie Ju,* Rui Xie, Bao Zhang, and Liang-Yin
Supplementary Material (ESI) for Chemical Communications K + -Recognition Capsules with Squirting Release Mechanisms Supplementary Material Zhuang Liu, Li Liu, Xiao-Jie Ju,* Rui Xie, Bao Zhang, and Liang-Yin
One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts
 Electronic Supplementary Information One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts Ying Wang, Jia-Min Zheng, Kangnian
Electronic Supplementary Information One-pot Solvent-free Synthesis of Sodium Benzoate from the Oxidation of Benzyl Alcohol over Novel Efficient AuAg/TiO 2 Catalysts Ying Wang, Jia-Min Zheng, Kangnian
Dendritic Star Polymer of Polyacrylamide Based on β-cyclodextrin Trimer: A. Flocculant and Drug Vehicle
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2016 Electronic Supporting Information
Supporting Information
 Supporting Information Synthesis, Anion Exchange, and Delamination of Co-Al Layered Double Hydroxide. Assembly of the Exfoliated Nanosheet/Polyanion Composite Films and Magneto-optical Studies Zhaoping
Supporting Information Synthesis, Anion Exchange, and Delamination of Co-Al Layered Double Hydroxide. Assembly of the Exfoliated Nanosheet/Polyanion Composite Films and Magneto-optical Studies Zhaoping
Supporting Information for: Engineering the structure and properties of DNA-nanoparticle superstructures using polyvalent counterions
 Supporting Information for: Engineering the structure and properties of DNA-nanoparticle superstructures using polyvalent counterions Leo Y.T. Chou 1 ǂ, Fayi Song 1 ǂ, Warren C.W. Chan*ǂǁ ǂ Institute of
Supporting Information for: Engineering the structure and properties of DNA-nanoparticle superstructures using polyvalent counterions Leo Y.T. Chou 1 ǂ, Fayi Song 1 ǂ, Warren C.W. Chan*ǂǁ ǂ Institute of
BLAKEHURST HIGH SCHOOL
 BLAKEHURST HIGH SCHOOL YEAR 12 HALF YEARLY EXAM CHEMISTRY 2006 1½ HOURS NAME: Part A Total marks 10 Attempt Questions 1 10 Allow about 20 minutes for this part 1. The process of Steam Cracking can be written
BLAKEHURST HIGH SCHOOL YEAR 12 HALF YEARLY EXAM CHEMISTRY 2006 1½ HOURS NAME: Part A Total marks 10 Attempt Questions 1 10 Allow about 20 minutes for this part 1. The process of Steam Cracking can be written
Synthesis mechanism and gas-sensing application of. nanosheet-assembled tungsten oxide microspheres
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Synthesis mechanism and gas-sensing
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information for Synthesis mechanism and gas-sensing
Scanning Electron Microscopy (SEM) with Energy Dispersive Spectroscopy (EDS) Analysis The samples were also characterized by scanning electron
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Scanning Electron Microscopy (SEM) with Energy Dispersive Spectroscopy (EDS) Analysis The samples
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2016 Scanning Electron Microscopy (SEM) with Energy Dispersive Spectroscopy (EDS) Analysis The samples
ph = -log[h+], [H+] = 10-pH ph + poh = 14
![ph = -log[h+], [H+] = 10-pH ph + poh = 14 ph = -log[h+], [H+] = 10-pH ph + poh = 14](/thumbs/89/98654987.jpg) You may remove this page. ph = -log[h+], [H+] = 10-pH McVc = MdVd ph + poh = 14 NA = 6.02 x 1023 mol-1 JBA 2017 Chemistry Exam 3 Name: Score: /100 = /80 Multiple choice questions are worth two points each.
You may remove this page. ph = -log[h+], [H+] = 10-pH McVc = MdVd ph + poh = 14 NA = 6.02 x 1023 mol-1 JBA 2017 Chemistry Exam 3 Name: Score: /100 = /80 Multiple choice questions are worth two points each.
Conclusion and Future Work
 Chapter 7 7. Chapter 7 and Future Work Chapter 7 Abstract This chapter gives the details of correlations of the spectroscopic investigation results with those available from other studies and also summarizes
Chapter 7 7. Chapter 7 and Future Work Chapter 7 Abstract This chapter gives the details of correlations of the spectroscopic investigation results with those available from other studies and also summarizes
-organic Thin Film From an Aqueous Solution
 Fabrication and Characterization of -organic Thin Film From an Aqueous Solution Fabrication and Characterization of TiO 2 -organic Thin Film From an Aqueous Solution W.N. Mu 1 and S.Z. Shi 2 1 2 School
Fabrication and Characterization of -organic Thin Film From an Aqueous Solution Fabrication and Characterization of TiO 2 -organic Thin Film From an Aqueous Solution W.N. Mu 1 and S.Z. Shi 2 1 2 School
Solution. Types of Solutions. Concentration and Solution Stoichiometry
 Concentration and Solution Stoichiometry Solution homogenous mixture of 2 or more pure substances only one perceptible phase species do not react chemically Types of Solutions solid liquid gas Solutions
Concentration and Solution Stoichiometry Solution homogenous mixture of 2 or more pure substances only one perceptible phase species do not react chemically Types of Solutions solid liquid gas Solutions
Advanced Pharmaceutical Analysis
 Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
Lecture 2 Advanced Pharmaceutical Analysis IR spectroscopy Dr. Baraa Ramzi Infrared Spectroscopy It is a powerful tool for identifying pure organic and inorganic compounds. Every molecular compound has
The ph-responsive behaviour of aqueous solutions of poly(acrylic acid) is dependent on molar mass
 Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2016 The ph-responsive behaviour of aqueous solutions of poly(acrylic acid) is dependent on molar
Electronic Supplementary Material (ESI) for Soft Matter. This journal is The Royal Society of Chemistry 2016 The ph-responsive behaviour of aqueous solutions of poly(acrylic acid) is dependent on molar
Facile Assembly Enhanced Spontaneous Fluorescence Response of Ag + Ion Containing Polyelectrolyte Multilayer Films
 Supporting Information Facile Assembly Enhanced Spontaneous Fluorescence Response of Ag + Ion Containing Polyelectrolyte Multilayer Films Xiayun Huang and Nicole S. Zacharia*,, Department of Mechanical
Supporting Information Facile Assembly Enhanced Spontaneous Fluorescence Response of Ag + Ion Containing Polyelectrolyte Multilayer Films Xiayun Huang and Nicole S. Zacharia*,, Department of Mechanical
Label-Free Fluorimetric Detection of Histone Using Quaternized Carbon Dot-DNA Nanobiohybrid. Electronic Supplementary Information (ESI)
 Label-Free Fluorimetric Detection of Histone Using Quaternized Carbon Dot-DNA Nanobiohybrid Subhabrata Maiti, Krishnendu Das, and Prasanta Kumar Das* Department of Biological Chemistry, Indian Association
Label-Free Fluorimetric Detection of Histone Using Quaternized Carbon Dot-DNA Nanobiohybrid Subhabrata Maiti, Krishnendu Das, and Prasanta Kumar Das* Department of Biological Chemistry, Indian Association
In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes
 Supporting information for In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes Yuting Zhou, Jianmin Song, Lei Wang*, Xuting Xue, Xiaoman Liu, Hui Xie*, and Xin Huang* MIIT Key Laboratory
Supporting information for In Situ Gelation-Induced Death of Cancer Cells Based on Proteinosomes Yuting Zhou, Jianmin Song, Lei Wang*, Xuting Xue, Xiaoman Liu, Hui Xie*, and Xin Huang* MIIT Key Laboratory
TrioMol Isolation Reagent
 TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent
 TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
TrioMol Isolation Reagent Technical Manual No. 0242 Version 06142007 I Description... 1 II Key Features... 1 III Storage..... 1 IV General Protocol Using Triomol Isolation Reagent 1 V Troubleshooting.
Supporting Information for
 Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
Electronic Supplementary Material (ESI) for New Journal of Chemistry. This journal is The Royal Society of Chemistry and the Centre National de la Recherche Scientifique 2017 Supporting Information for
Supplementary Information. Omnidispersible Hedgehog Particles with Multilayer Coatings for. Multiplexed Biosensing
 Supplementary Information Omnidispersible Hedgehog Particles with Multilayer Coatings for Multiplexed Biosensing Douglas G. Montjoy 1, Joong Hwan Bahng 1,2, Aydin Eskafi 1, Harrison Hou 1, Nicholas A.
Supplementary Information Omnidispersible Hedgehog Particles with Multilayer Coatings for Multiplexed Biosensing Douglas G. Montjoy 1, Joong Hwan Bahng 1,2, Aydin Eskafi 1, Harrison Hou 1, Nicholas A.
1 Which of the following cannot be used to detect alcohol in a breathalyser test? Fractional distillation. Fuel cell. Infrared spectroscopy
 1 Which of the following cannot be used to detect alcohol in a breathalyser test? Fractional distillation Fuel cell Infrared spectroscopy Reduction of dichromate(vi) ions 2 Propanal, H 3 H 2 HO, and propanone,
1 Which of the following cannot be used to detect alcohol in a breathalyser test? Fractional distillation Fuel cell Infrared spectroscopy Reduction of dichromate(vi) ions 2 Propanal, H 3 H 2 HO, and propanone,
Identifying Unknowns
 Identifying Unknowns Testing for Identification Although non-characteristic properties can be shared by many different substances, they can still be useful in helping to identify an unknown Substances
Identifying Unknowns Testing for Identification Although non-characteristic properties can be shared by many different substances, they can still be useful in helping to identify an unknown Substances
Electronic Supplementary Information. Tubulation of Liposomes via the Interaction of Supramolecular Nanofibers
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Tubulation of Liposomes via the Interaction of Supramolecular
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Tubulation of Liposomes via the Interaction of Supramolecular
Di-Stimuli Responsive Diblock and Triblock Copolymer Particles
 Di-Stimuli Responsive Diblock and Triblock Copolymer Particles Nancy Weber, John Texter *, and Klaus Tauer Max Planck Institute for Colloids and Interfaces Department of Colloid Chemistry 14476 Golm, Germany
Di-Stimuli Responsive Diblock and Triblock Copolymer Particles Nancy Weber, John Texter *, and Klaus Tauer Max Planck Institute for Colloids and Interfaces Department of Colloid Chemistry 14476 Golm, Germany
Bottom Up Synthesis: Organic Colloids and Nano Particles Polymer latex dispersions
 Bottom Up Synthesis: Organic Colloids and Nano Particles Polymer latex dispersions Global production of synthetic polymers 2000: ca. 190 Mio. t ca. 200 Bio. sales Global production of synthetic polymers,
Bottom Up Synthesis: Organic Colloids and Nano Particles Polymer latex dispersions Global production of synthetic polymers 2000: ca. 190 Mio. t ca. 200 Bio. sales Global production of synthetic polymers,
SI0 UV-vis absorption spectra and emission response of 6MQz and 6MQc
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 SI0 UV-vis absorption spectra and emission response of 6MQz and 6MQc The absorption and emission
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2017 SI0 UV-vis absorption spectra and emission response of 6MQz and 6MQc The absorption and emission
CHAPTER 7: Solutions & Colloids 7.2 SOLUBILITY. Degrees of Solution. Page PHYSICAL STATES of SOLUTIONS SOLUTION
 CHAPTER 7: Solutions & Colloids Predict the relative solubility of materials on the basis of polarity Describe solution formation in terms of solutesolvent interactions Calculate solution concentrations
CHAPTER 7: Solutions & Colloids Predict the relative solubility of materials on the basis of polarity Describe solution formation in terms of solutesolvent interactions Calculate solution concentrations
Dynamic covalent assembly of stimuli responsive vesicle gels
 Supporting Information for Dynamic covalent assembly of stimuli responsive vesicle gels Christophe B. Minkenberg, Wouter E. Hendriksen, Feng Li, Eduardo Mendes, Rienk Eelkema, and Jan H. van Esch* Department
Supporting Information for Dynamic covalent assembly of stimuli responsive vesicle gels Christophe B. Minkenberg, Wouter E. Hendriksen, Feng Li, Eduardo Mendes, Rienk Eelkema, and Jan H. van Esch* Department
Synthesis of homochiral zeolitic imidazolate frameworks via solvent-assisted linker exchange for enantioselective sensing and separation
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Synthesis of homochiral zeolitic imidazolate frameworks via solvent-assisted
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Synthesis of homochiral zeolitic imidazolate frameworks via solvent-assisted
Electronic Supplementary Information. Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered
 Electronic Supplementary Information Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered polyelectrolyte nanoreactors on a charged sphere Young-Min Chung,* a Yong-Tak
Electronic Supplementary Information Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered polyelectrolyte nanoreactors on a charged sphere Young-Min Chung,* a Yong-Tak
Supplementary Information. Core-Shell Silver/Polymeric Nanoparticles-Based Combinatorial Therapy against Breast Cancer In-vitro
 Supplementary Information Core-Shell Silver/Polymeric Nanoparticles-Based Combinatorial Therapy against Breast Cancer In-vitro Nancy M. El-Baz 1,2, Laila Ziko 1,3, Rania Siam 1,3, Wael Mamdouh 1,2 * 1
Supplementary Information Core-Shell Silver/Polymeric Nanoparticles-Based Combinatorial Therapy against Breast Cancer In-vitro Nancy M. El-Baz 1,2, Laila Ziko 1,3, Rania Siam 1,3, Wael Mamdouh 1,2 * 1
Infrared Spectroscopy
 x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
x-rays ultraviolet (UV) visible Infrared (I) microwaves radiowaves near I middle I far I λ (cm) 8 x 10-5 2.5 x 10-4 2.5 x 10-3 2.5 x 10-2 µ 0.8 2.5 25 250 ν (cm -1 ) 13,000 4,000 400 40 ν (cm -1 1 ) =
Supporting Information
 Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Amphiphilic diselenide-containing supramolecular polymers
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Amphiphilic diselenide-containing supramolecular polymers Xinxin Tan, Liulin Yang, Zehuan
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Amphiphilic diselenide-containing supramolecular polymers Xinxin Tan, Liulin Yang, Zehuan
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water
 Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Chapter-2 (Page 22-37) Physical and Chemical Properties of Water Introduction About 70% of the mass of the human body is water. Water is central to biochemistry for the following reasons: 1- Biological
Bright line spectrum questions
 Base your answers to questions 1 and 2 on the information below and on your knowledge of chemistry. The bright-line spectra for four elements and a mixture of elements are shown in the diagram below. 1.
Base your answers to questions 1 and 2 on the information below and on your knowledge of chemistry. The bright-line spectra for four elements and a mixture of elements are shown in the diagram below. 1.
CH 221 Chapter Four Part II Concept Guide
 CH 221 Chapter Four Part II Concept Guide 1. Solubility Why are some compounds soluble and others insoluble? In solid potassium permanganate, KMnO 4, the potassium ions, which have a charge of +1, are
CH 221 Chapter Four Part II Concept Guide 1. Solubility Why are some compounds soluble and others insoluble? In solid potassium permanganate, KMnO 4, the potassium ions, which have a charge of +1, are
CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710)
 CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710) Identification of an Unknown by IR PRELAB (PreLab is due before entering the lab.) Every student has to prepare for each experiment by answering the Pre-Laboratory
CHEM 3760 Orgo I, F14 (Lab #11) (TECH 710) Identification of an Unknown by IR PRELAB (PreLab is due before entering the lab.) Every student has to prepare for each experiment by answering the Pre-Laboratory
Supporting Online Material. On-Chip Dielectrophoretic Co-Assembly of Live Cells and. Particles into Responsive Biomaterials
 Supporting Online Material On-Chip Dielectrophoretic Co-Assembly of Live Cells and Particles into esponsive Biomaterials Shalini Gupta, ossitza G. Alargova, Peter K. Kilpatrick and Orlin D. Velev* Description
Supporting Online Material On-Chip Dielectrophoretic Co-Assembly of Live Cells and Particles into esponsive Biomaterials Shalini Gupta, ossitza G. Alargova, Peter K. Kilpatrick and Orlin D. Velev* Description
Infrared Spectroscopic Study of the Interactions of Nylon-6 with Water
 Infrared Spectroscopic Study of the Interactions of Nylon-6 with Water REIKICHI IWAMOTO, 1 HIROSHI MURASE 2 1 Kansai Research Institute, Chudoji Minamimachi 134, Shimogyoku, Kyoto 600-8813, Japan 2 Toyobo
Infrared Spectroscopic Study of the Interactions of Nylon-6 with Water REIKICHI IWAMOTO, 1 HIROSHI MURASE 2 1 Kansai Research Institute, Chudoji Minamimachi 134, Shimogyoku, Kyoto 600-8813, Japan 2 Toyobo
Supporting Information. Light-induced Wide Range Color Switching of Liquid Crystal. Blue Phase doped with Hydrogen-bonded Chiral Azobenzene.
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Light-induced Wide Range Color Switching of Liquid Crystal Blue Phase
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Light-induced Wide Range Color Switching of Liquid Crystal Blue Phase
General Chemistry I. Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University. Module 6: Solutions
 General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 6: Solutions Definitions (solutions: solvent, solute, solubility, concentration; interactions between
General Chemistry I Dr. PHAN TẠI HUÂN Faculty of Food Science and Technology Nong Lam University Module 6: Solutions Definitions (solutions: solvent, solute, solubility, concentration; interactions between
Ultra-Sensitive ph Control of Supramolecular Polymers and Hydrogels: pk a Matching of Biomimetic Monomers
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2014 Supplementary Information for Ultra-Sensitive ph Control of Supramolecular Polymers and
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2014 Supplementary Information for Ultra-Sensitive ph Control of Supramolecular Polymers and
Lecture 1 Chapter 12 Sections 1-2. Solutions Solubility
 Lecture 1 Chapter 12 Sections 1-2 Solutions Solubility Chapt 12: Nature of Solutions Solution a homogeneous mixture of two or more substances, in which one is called the solvent and the other is the solute
Lecture 1 Chapter 12 Sections 1-2 Solutions Solubility Chapt 12: Nature of Solutions Solution a homogeneous mixture of two or more substances, in which one is called the solvent and the other is the solute
Calculate the total volume of hydrogen gas that was liberated at S.T.P conditions (Zn = 65.4, molar gas volume = 22.4 litres at S.T.
 MOLE CONCEPT 1. Zinc metal and hydrochloric acid react according to the following equation Zn (s) + 2HCl (aq) ZnCl (aq) + H 2(g) 1.96g of zinc were reacted with 100cm 3 of 0.2M Hydrochloric acid, (a) Determine
MOLE CONCEPT 1. Zinc metal and hydrochloric acid react according to the following equation Zn (s) + 2HCl (aq) ZnCl (aq) + H 2(g) 1.96g of zinc were reacted with 100cm 3 of 0.2M Hydrochloric acid, (a) Determine
EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY
 EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY Structure 7.1 Introduction Objectives 7.2 Principle 7.3 Requirements 7.4 Strategy for the Interpretation of IR Spectra 7.5 Practice Problems
EXPT. 7 CHARACTERISATION OF FUNCTIONAL GROUPS USING IR SPECTROSCOPY Structure 7.1 Introduction Objectives 7.2 Principle 7.3 Requirements 7.4 Strategy for the Interpretation of IR Spectra 7.5 Practice Problems
Mixtures. Chapters 12/13: Solutions and Colligative Properties. Types of Solutions. Suspensions. The Tyndall Effect: Colloid
 Mixtures Chapters 12/13: Solutions and Colligative Properties Solution - a homogeneous mixture of two or more substances in a single phase Soluble - capable of being dissolved Solutions - 2 Parts Solvent
Mixtures Chapters 12/13: Solutions and Colligative Properties Solution - a homogeneous mixture of two or more substances in a single phase Soluble - capable of being dissolved Solutions - 2 Parts Solvent
Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification
 Electronic Supporting Information: Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification Chunlong Kong, a Takuji Shintani b and Toshinori Tsuru*
Electronic Supporting Information: Pre-seeding -assisted synthesis of high performance polyamide-zeolite nanocomposie membrane for water purification Chunlong Kong, a Takuji Shintani b and Toshinori Tsuru*
Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
Switching shape of hollow layer-by-layer hydrogel microcontainers
 Electronic Supplementary Information Switching shape of hollow layer-by-layer hydrogel microcontainers Veronika Kozlovskaya, William Higgins, Jun Chen and Eugenia Kharlampieva* University of Alabama at
Electronic Supplementary Information Switching shape of hollow layer-by-layer hydrogel microcontainers Veronika Kozlovskaya, William Higgins, Jun Chen and Eugenia Kharlampieva* University of Alabama at
MARAKWET WEST DISTRICT 233/1 CHEMISTRY PAPER 1 JULY/AUGUST 2015 TIME: 2 HOURS
 MARAKWET WEST DISTRICT 233/1 CHEMISTRY PAPER 1 JULY/AUGUST 2015 TIME: 2 HOURS 1. State two reasons why we use the non- luminous flame for heating in the laboratory instead of using luminous flame. 2. The
MARAKWET WEST DISTRICT 233/1 CHEMISTRY PAPER 1 JULY/AUGUST 2015 TIME: 2 HOURS 1. State two reasons why we use the non- luminous flame for heating in the laboratory instead of using luminous flame. 2. The
Solutions. Solutions. How Does a Solution Form? Solutions. Energy Changes in Solution. How Does a Solution Form
 Ummm Solutions Solutions Solutions are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed uniformly throughout the solvent. Solutions The intermolecular forces
Ummm Solutions Solutions Solutions are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed uniformly throughout the solvent. Solutions The intermolecular forces
Solutions and Solubility. BHS Chemistry
 Solutions and Solubility BHS Chemistry MATTER Yes Can it be separated by physical means? No MIXTURES Pure SUBSTANCES Yes Is the composition uniform? Can it be decomposed by regular chemical means? No Yes
Solutions and Solubility BHS Chemistry MATTER Yes Can it be separated by physical means? No MIXTURES Pure SUBSTANCES Yes Is the composition uniform? Can it be decomposed by regular chemical means? No Yes
Supplementary Figure S1 Synthesis of tri(ethoxy)silane with boc-protected amino
 O O H 2 N NH 2 O OH NH 2 1 2 O LiAlH 4 NH 2 NH 2 2 3 NH 2 Boc 2 O 3 4 N H boc N H boc [Pt] HSi(OEt) 3 (EtO) 3 Si N H boc 4 5 Supplementary Figure S1 Synthesis of tri(ethoxy)silane with boc-protected amino
O O H 2 N NH 2 O OH NH 2 1 2 O LiAlH 4 NH 2 NH 2 2 3 NH 2 Boc 2 O 3 4 N H boc N H boc [Pt] HSi(OEt) 3 (EtO) 3 Si N H boc 4 5 Supplementary Figure S1 Synthesis of tri(ethoxy)silane with boc-protected amino
CHEM 3.2 (AS91388) 3 credits. Demonstrate understanding of spectroscopic data in chemistry
 CHEM 3.2 (AS91388) 3 credits Demonstrate understanding of spectroscopic data in chemistry Spectroscopic data is limited to mass, infrared (IR) and 13 C nuclear magnetic resonance (NMR) spectroscopy. Organic
CHEM 3.2 (AS91388) 3 credits Demonstrate understanding of spectroscopic data in chemistry Spectroscopic data is limited to mass, infrared (IR) and 13 C nuclear magnetic resonance (NMR) spectroscopy. Organic
Identification of Ions and Gases
 Identification of Ions and Gases Question Paper 7 Level IGCSE Subject Chemistry Exam Board CIE Topic Acids, Bases and Salts Sub-Topic Identification of Ions and Gases Paper Type Alternative to Practical
Identification of Ions and Gases Question Paper 7 Level IGCSE Subject Chemistry Exam Board CIE Topic Acids, Bases and Salts Sub-Topic Identification of Ions and Gases Paper Type Alternative to Practical
Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions)
 Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions) Metallic (electrostatic attraction between + metal ions and delocalised electrons) Group 1 ions 1+
Covalent (sharing of electron pairs) Ionic ( electrostatic attraction between oppositely charged ions) Metallic (electrostatic attraction between + metal ions and delocalised electrons) Group 1 ions 1+
1 of 43 Boardworks Ltd Chemistry 11. Solution Chemistry
 1 of 43 Boardworks Ltd 2009 Chemistry 11 Solution Chemistry Crash Course: Solutions 2 of 43 Boardworks Ltd 2009 https://www.youtube.com/watch?v=9h2f1bjr0p4&t=39s 3 of 43 Boardworks Ltd 2009 Solubility
1 of 43 Boardworks Ltd 2009 Chemistry 11 Solution Chemistry Crash Course: Solutions 2 of 43 Boardworks Ltd 2009 https://www.youtube.com/watch?v=9h2f1bjr0p4&t=39s 3 of 43 Boardworks Ltd 2009 Solubility
Solutions. LiCl (s) + H2O (l) LiCl (aq) 3/12/2013. Definitions. Aqueous Solution. Solutions. How Does a Solution Form? Solute Solvent solution
 Solutions Definitions A solution is a homogeneous mixture A solute is dissolved in a solvent. solute is the substance being dissolved solvent is the liquid in which the solute is dissolved an aqueous solution
Solutions Definitions A solution is a homogeneous mixture A solute is dissolved in a solvent. solute is the substance being dissolved solvent is the liquid in which the solute is dissolved an aqueous solution
Self-Assembly of Guanosine and Deoxy-Guanosine into Hydrogels: Monovalent Cation Guided Modulation of Gelation, Morphology and Self-Healing Properties
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2014 Supplementary Information Self-Assembly of Guanosine and Deoxy-Guanosine
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry B. This journal is The Royal Society of Chemistry 2014 Supplementary Information Self-Assembly of Guanosine and Deoxy-Guanosine
EXPERIMENT A4: PRECIPITATION REACTION AND THE LIMITING REAGENT. Learning Outcomes. Introduction
 1 EXPERIMENT A4: PRECIPITATION REACTION AND THE LIMITING REAGENT Learning Outcomes Upon completion of this lab, the student will be able to: 1) Demonstrate the formation of a precipitate in a chemical
1 EXPERIMENT A4: PRECIPITATION REACTION AND THE LIMITING REAGENT Learning Outcomes Upon completion of this lab, the student will be able to: 1) Demonstrate the formation of a precipitate in a chemical
Effects of methanol on crystallization of water in the deeply super cooled region
 Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
Effects of methanol on crystallization of water in the deeply super cooled region Ryutaro Souda Nanoscale Materials Center National Institute for Materials Science Japan PHYSICAL REVIEW B 75, 184116, 2007
Uniform properties throughout! SOLUTE(S) - component(s) of a solution present in small amounts.
 54 SOLUTIONS - a SOLUTION is a HOMOGENEOUS MIXTURE. Uniform properties throughout! - parts of a solution: SOLUTE(S) - component(s) of a solution present in small amounts. SOLVENT - the component of a solution
54 SOLUTIONS - a SOLUTION is a HOMOGENEOUS MIXTURE. Uniform properties throughout! - parts of a solution: SOLUTE(S) - component(s) of a solution present in small amounts. SOLVENT - the component of a solution
Supporting Information
 Supporting Information Janus Nanocomposite Hydrogels for Chirality-Dependent Cell Adhesion and Migration Andisheh Motealleh, a and Nermin Seda Kehr* a a Physikalisches Institut and CeNTech,Westfälische
Supporting Information Janus Nanocomposite Hydrogels for Chirality-Dependent Cell Adhesion and Migration Andisheh Motealleh, a and Nermin Seda Kehr* a a Physikalisches Institut and CeNTech,Westfälische
Fourier Transform IR Spectroscopy
 Fourier Transform IR Spectroscopy Absorption peaks in an infrared absorption spectrum arise from molecular vibrations Absorbed energy causes molecular motions which create a net change in the dipole moment.
Fourier Transform IR Spectroscopy Absorption peaks in an infrared absorption spectrum arise from molecular vibrations Absorbed energy causes molecular motions which create a net change in the dipole moment.
Chapter 4: Types of Chemical Reactions and Solution Stoichiometry
 Chapter 4: Types of Chemical Reactions and Solution Stoichiometry 4.1 Water, the Common Solvent 4.2 The Nature of Aqueous Solutions: Strong and Weak Electrolytes 4.3 The Composition of Solutions (MOLARITY!)
Chapter 4: Types of Chemical Reactions and Solution Stoichiometry 4.1 Water, the Common Solvent 4.2 The Nature of Aqueous Solutions: Strong and Weak Electrolytes 4.3 The Composition of Solutions (MOLARITY!)
CHAPTER IV HOFMANN REARRANGEMENT IN CROSSLINKED POLYMERIC MATRICES
 CHAPTER IV HOFMANN REARRANGEMENT IN CROSSLINKED POLYMERIC MATRICES The Hofmann degradation reaction has been used as a synthetic route for the preparation of amines 180-187 Tanaka and Senju reported the
CHAPTER IV HOFMANN REARRANGEMENT IN CROSSLINKED POLYMERIC MATRICES The Hofmann degradation reaction has been used as a synthetic route for the preparation of amines 180-187 Tanaka and Senju reported the
Application of IR Raman Spectroscopy
 Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Application of IR Raman Spectroscopy 3 IR regions Structure and Functional Group Absorption IR Reflection IR Photoacoustic IR IR Emission Micro 10-1 Mid-IR Mid-IR absorption Samples Placed in cell (salt)
Supplementary information
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the wner Societies 26 Supplementary information Structure and dynamics of water molecules confined in triglyceride
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the wner Societies 26 Supplementary information Structure and dynamics of water molecules confined in triglyceride
Electronic Supplementary Material
 Electronic Supplementary Material Synthesis and characterization of mesoporous Si MCM-41 materials and their application as solid acid catalysts in some esterification reactions by Tarun F Parangi (pp
Electronic Supplementary Material Synthesis and characterization of mesoporous Si MCM-41 materials and their application as solid acid catalysts in some esterification reactions by Tarun F Parangi (pp
A supramolecular approach for fabrication of photo- responsive block-controllable supramolecular polymers
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information A supramolecular approach for fabrication of photo- responsive
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Supporting Information A supramolecular approach for fabrication of photo- responsive
Change in physico-mechanical and thermal properties of polyamide / silica nanocomposite film
 International Journal of Engineering Research and Development e-issn: 2278-067X, p-issn: 2278-800X, www.ijerd.com Volume 7, Issue 6 (June 2013), PP. 01-05 Change in physico-mechanical and thermal properties
International Journal of Engineering Research and Development e-issn: 2278-067X, p-issn: 2278-800X, www.ijerd.com Volume 7, Issue 6 (June 2013), PP. 01-05 Change in physico-mechanical and thermal properties
3) Accounts for strands of DNA being held together into a double helix. 7) Accounts for the cohesive nature of water and its high surface tension
 AP Chemistry Test (Chapter 11) Multiple Choice (50%) Please use the following choices to answer questions 1-7. A) London dispersion forces B) Ion-ion attractions C) Dipole-dipole attractions D) Dipole-ion
AP Chemistry Test (Chapter 11) Multiple Choice (50%) Please use the following choices to answer questions 1-7. A) London dispersion forces B) Ion-ion attractions C) Dipole-dipole attractions D) Dipole-ion
The characterization of MnO nanostructures synthesized using the chemical bath deposition method
 The characterization of MnO nanostructures synthesized using the chemical bath deposition method LF Koao 1, F B Dejene 1* and HC Swart 2 1 Department of Physics, University of the Free State (Qwaqwa Campus),
The characterization of MnO nanostructures synthesized using the chemical bath deposition method LF Koao 1, F B Dejene 1* and HC Swart 2 1 Department of Physics, University of the Free State (Qwaqwa Campus),
Supporting Information
 Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Polystyrene Sulfonate Threaded in MIL-101Cr(III) as Stable and
Electronic Supplementary Material (ESI) for Dalton Transactions. This journal is The Royal Society of Chemistry 2016 Supporting Information Polystyrene Sulfonate Threaded in MIL-101Cr(III) as Stable and
Hour Examination # 4
 CHEM 346 Organic Chemistry I Fall 2014 Exam # 4 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I Fall 2014 Instructor: Paul Bracher Hour Examination # 4 Wednesday, December 3 rd, 2014 6:00 8:00
CHEM 346 Organic Chemistry I Fall 2014 Exam # 4 Solutions Key Page 1 of 12 CHEM 346 Organic Chemistry I Fall 2014 Instructor: Paul Bracher Hour Examination # 4 Wednesday, December 3 rd, 2014 6:00 8:00
World Journal of Pharmaceutical and Life Sciences WJPLS
 wjpls, 2016, Vol. 2, Issue 6, 542-549 Research Article ISSN 2454-2229 Prajapati et al. WJPLS www.wjpls.org SJIF Impact Factor: 3.347 SYNTHESIS, CHARACTERISATION AND APPLICATION PROPERTIES OF SOME NOVEL
wjpls, 2016, Vol. 2, Issue 6, 542-549 Research Article ISSN 2454-2229 Prajapati et al. WJPLS www.wjpls.org SJIF Impact Factor: 3.347 SYNTHESIS, CHARACTERISATION AND APPLICATION PROPERTIES OF SOME NOVEL
PAPER No. : 8 (PHYSICAL SPECTROSCOPY) MODULE NO. : 23 (NORMAL MODES AND IRREDUCIBLE REPRESENTATIONS FOR POLYATOMIC MOLECULES)
 Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
Subject Chemistry Paper No and Title Module No and Title Module Tag 8/ Physical Spectroscopy 23/ Normal modes and irreducible representations for polyatomic molecules CHE_P8_M23 TABLE OF CONTENTS 1. Learning
5070/04. UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY Paper 4 Alternative to Practical
 UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY Paper 4 Alternative to Practical 5070/04 May/June 2005 Candidates answer on the Question Paper.
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY Paper 4 Alternative to Practical 5070/04 May/June 2005 Candidates answer on the Question Paper.
Self-Assembly of Coated Colloidal Particles for Optical Applications
 Self-Assembly of Coated Colloidal Particles for Optical Applications Introduction Nearly two decades ago, theoretical predictions indicated the possibility of creating omnidirectional photonic-band-gap
Self-Assembly of Coated Colloidal Particles for Optical Applications Introduction Nearly two decades ago, theoretical predictions indicated the possibility of creating omnidirectional photonic-band-gap
X-Ray Fluorescent and Mid-Infrared Spectroscopic Measurement of Leaf Model
 X-Ray Fluorescent and Mid-Infrared Spectroscopic Measurement of Leaf Model Atsushi Hashimoto a, Toshito Niwa a, T. Yamamura a, Mizanur Rahman a, Kenichi Nakanishi a, Takaharu Kameoka b, Takao Kumon c,
X-Ray Fluorescent and Mid-Infrared Spectroscopic Measurement of Leaf Model Atsushi Hashimoto a, Toshito Niwa a, T. Yamamura a, Mizanur Rahman a, Kenichi Nakanishi a, Takaharu Kameoka b, Takao Kumon c,
Chapter 16. Solubility Equilibria 10/14/2010. Solubility Equilibria. Solubility Product (Constant), K sp. Solubility and the Solubility Product
 Solubility Equilibria These are associated with ionic solids dissolving in water to form aqueous solutions Chapter 16 Solubility Equilibria It is assumed that when an ionic compound dissolves in water,
Solubility Equilibria These are associated with ionic solids dissolving in water to form aqueous solutions Chapter 16 Solubility Equilibria It is assumed that when an ionic compound dissolves in water,
, are both macromolecules. They have similar physical properties but silicon(iv) oxide is acidic and zirconium(iv) oxide is amphoteric
 1 Silicon(IV) oxide, SiO 2, and zirconium(iv) oxide, ZrO 2, are both macromolecules. They have similar physical properties but silicon(iv) oxide is acidic and zirconium(iv) oxide is amphoteric. (a) Defi
1 Silicon(IV) oxide, SiO 2, and zirconium(iv) oxide, ZrO 2, are both macromolecules. They have similar physical properties but silicon(iv) oxide is acidic and zirconium(iv) oxide is amphoteric. (a) Defi
SOLUBILITY EQUILIBRIA (THE SOLUBILITY PRODUCT)
 SOLUBILITY EQUILIBRIA (THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Slightly soluble salts establish a dynamic equilibrium with the hydrated cations and
SOLUBILITY EQUILIBRIA (THE SOLUBILITY PRODUCT) Saturated solutions of salts are another type of chemical equilibria. Slightly soluble salts establish a dynamic equilibrium with the hydrated cations and
Heteroagglomeration of nanosilver with colloidal SiO2 and clay
 , 14, 1 8 Supplementary material Heteroagglomeration of nanosilver with colloidal SiO2 and clay Sébastien Maillette, A Caroline Peyrot, A Tapas Purkait, B Muhammad Iqbal, B Jonathan G. C. Veinot B and
, 14, 1 8 Supplementary material Heteroagglomeration of nanosilver with colloidal SiO2 and clay Sébastien Maillette, A Caroline Peyrot, A Tapas Purkait, B Muhammad Iqbal, B Jonathan G. C. Veinot B and
6.01 Solutions. The Chemistry of Matter in Water. Dr. Fred Omega Garces. Chemistry 111, Miramar College. 1 Solutions. January 10
 6.01 Solutions The Chemistry of Matter in Water Dr. Fred Omega Garces Chemistry 111, Miramar College 1 Solutions 6.01 Solutions ow water Dissolves Salts 2 Solutions Components of Solution omogeneous systems
6.01 Solutions The Chemistry of Matter in Water Dr. Fred Omega Garces Chemistry 111, Miramar College 1 Solutions 6.01 Solutions ow water Dissolves Salts 2 Solutions Components of Solution omogeneous systems
F G C G H H C B B A A DD E E
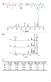 a D E F G H D E I F G H F F I DD E E G H H b c [1] 0 (M) [2a] 0 (M) [1] (M) [2a] (M) [3a] (M) K (M -1 ) a) 0.160 0.139 0.049 0.028 0.111 81 b) 0.147 0.180 0.025 0.058 0.122 84 c) 0.157 0.270 0.013 0.126
a D E F G H D E I F G H F F I DD E E G H H b c [1] 0 (M) [2a] 0 (M) [1] (M) [2a] (M) [3a] (M) K (M -1 ) a) 0.160 0.139 0.049 0.028 0.111 81 b) 0.147 0.180 0.025 0.058 0.122 84 c) 0.157 0.270 0.013 0.126
Chapter 16: Applications of Aqueous Equilibrium Part 3. Solubilities of Ionic Compounds and K sp
 Chapter 16: Applications of Aqueous Equilibrium Part 3 Solubilities of Ionic Compounds and K sp You ve already learned that not all ionic compounds are water soluble. You memorized the solubility rules
Chapter 16: Applications of Aqueous Equilibrium Part 3 Solubilities of Ionic Compounds and K sp You ve already learned that not all ionic compounds are water soluble. You memorized the solubility rules
EQUIVALENCE POINT. 8.8 millimoles is also the amount of acid left, and the added base gets converted to acetate ion!
 184 Another interesting point: The halfway point phenolphthalein color change buffer region EQUIVALENCE POINT What's special about it? It's the point where we have added half the required base to reach
184 Another interesting point: The halfway point phenolphthalein color change buffer region EQUIVALENCE POINT What's special about it? It's the point where we have added half the required base to reach
Fourier Transform Infrared Spectrophotometry Studies of Chromium Trioxide-Phthalic Acid Complexes
 DOI:10.7598/cst2016.1260 Chemical Science Transactions ISSN:2278-3458 2016, 5(3), 770-774 RESEARCH ARTICLE Fourier Transform Infrared Spectrophotometry Studies of Chromium Trioxide-Phthalic Acid Complexes
DOI:10.7598/cst2016.1260 Chemical Science Transactions ISSN:2278-3458 2016, 5(3), 770-774 RESEARCH ARTICLE Fourier Transform Infrared Spectrophotometry Studies of Chromium Trioxide-Phthalic Acid Complexes
Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage
 (Supporting Information) Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage Sanjay Kumar Singh, Xin-Bo Zhang, and Qiang Xu* National Institute of Advanced Industrial
(Supporting Information) Room Temperature Hydrogen Generation from Hydrous Hydrazine for Chemical Hydrogen Storage Sanjay Kumar Singh, Xin-Bo Zhang, and Qiang Xu* National Institute of Advanced Industrial
Synthesis of Dihydroquinoline Based Merocyanines as Naked Eye and Fluorogenic sensors for Hydrazine Hydrate in Aqueous Medium
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Synthesis of Dihydroquinoline Based Merocyanines as Naked Eye and Fluorogenic sensors for Hydrazine
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Synthesis of Dihydroquinoline Based Merocyanines as Naked Eye and Fluorogenic sensors for Hydrazine
Molecule smallest particle of a substance having its chemical properties Atoms connected via covalent bonds Examples:
 Ionic equations, calculations involving concentrations, stoichiometry MUDr. Jan Pláteník, PhD Molecule smallest particle of a substance having its chemical properties Atoms connected via covalent bonds
Ionic equations, calculations involving concentrations, stoichiometry MUDr. Jan Pláteník, PhD Molecule smallest particle of a substance having its chemical properties Atoms connected via covalent bonds
Infrared And Raman Spectra Of Inorganic And Coordination Compounds Applications In Coordination Or
 Infrared And Raman Spectra Of Inorganic And Coordination Compounds Applications In Coordination Or We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks
Infrared And Raman Spectra Of Inorganic And Coordination Compounds Applications In Coordination Or We have made it easy for you to find a PDF Ebooks without any digging. And by having access to our ebooks
C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy
 C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy Cl OH Cl An electron ionization mass spectrum of 2,5-dichlorophenol CHM 323: Summary of Important Concepts
C h a p t e r F o u r t e e n: Structure Determination: Mass Spectrometry and Infrared Spectroscopy Cl OH Cl An electron ionization mass spectrum of 2,5-dichlorophenol CHM 323: Summary of Important Concepts
Properties of Solutions (electrolytes) Chapter 4.1. Solution process (energy changes)
 Chapter 13 SOLUTIONS Properties of Solutions (electrolytes) Chapter 4.1 Solution process (energy changes) Solubility saturation supersaturation Factors that affect solubility intermolecular interactions
Chapter 13 SOLUTIONS Properties of Solutions (electrolytes) Chapter 4.1 Solution process (energy changes) Solubility saturation supersaturation Factors that affect solubility intermolecular interactions
Experiment 1: Extraction and Thin Layer Chromatography
 Experiment 1: Extraction and Thin Layer Chromatography Introduction: Chromatography is a useful tool in chemistry and can be very helpful in determining the composition of an unknown sample. In chromatography
Experiment 1: Extraction and Thin Layer Chromatography Introduction: Chromatography is a useful tool in chemistry and can be very helpful in determining the composition of an unknown sample. In chromatography
] [ SO 4 ] let sol y x x = x x be x = x 2 if sol y = 7.94 x 10 4 mol/l = 6.3 x 10 7
![] [ SO 4 ] let sol y x x = x x be x = x 2 if sol y = 7.94 x 10 4 mol/l = 6.3 x 10 7 ] [ SO 4 ] let sol y x x = x x be x = x 2 if sol y = 7.94 x 10 4 mol/l = 6.3 x 10 7](/thumbs/81/83813606.jpg) SCH 4U: UNIT 4 LESSONS Heterogeneous EQUILIBRIUM (Chap 17-pg 759) 1. Rationale: The principles of Chemical Equilibrium developed earlier (in Unit 3) also apply to weakly soluble salts. In this unit we
SCH 4U: UNIT 4 LESSONS Heterogeneous EQUILIBRIUM (Chap 17-pg 759) 1. Rationale: The principles of Chemical Equilibrium developed earlier (in Unit 3) also apply to weakly soluble salts. In this unit we
