Molecular modelling and simulation of electrolyte solutions, biomolecules, and wetting of component surfaces
|
|
|
- Alisha Harper
- 6 years ago
- Views:
Transcription
1 Molecular modelling and simulation of electrolyte solutions, biomolecules, and wetting of component surfaces M. Horsch, 1, S. Becker, 1 J. M. Castillo, 1 S. Deublein, 1 A. Fröscher, 1 S. Reiser, 1 S. Werth, 1 J. Vrabec, 2 and H. Hasse 1 Abstract Massively-parallel molecular dynamics simulation is applied to systems containing electrolytes, vapour-liquid interfaces, and biomolecules in contact with water-oil interfaces. Novel molecular models of alkali halide salts are presented and employed for the simulation of electrolytes in aqueous solution. The enzymatically catalysed hydroxylation of oleic acid is investigated by molecular dynamics simulation taking the internal degrees of freedom of the macromolecules into account. Thereby, Ewald summation methods are used to compute the long range electrostatic interactions. In systems with a phase boundary, the dispersive interaction, which is modelled by the Lennard-Jones potential here, has a more significant long range contribution than in homogeneous systems. This effect is accounted for by implementing the Janeček cutoff correction scheme. On this basis, the HPC infrastructure at the Steinbuch Centre for Computing was accessed and efficiently used, yielding new insights on the molecular systems under consideration. 1 Introduction Molecular simulation provides detailed information on processes on a level which is otherwise inaccessible, on the basis of physically realistic models of the intermolecular interactions. However, despite all efforts to keep these models as simple as possible, such simulations are extremely time consuming and belong to the most demanding applications of high performance computing. The scientific computing project Molecular simulation of static and dynamic properties of electrolyte systems, large molecules and wetting of component surfaces (MOCOS) aims at advancing the state of the art by developing the molecular 1 TU Kaiserslautern, Lehrstuhl für Thermodynamik, Erwin-Schrödinger-Str. 44, Kaiserslautern 2Universität Paderborn, Lehrstuhl für Thermodynamik und Energietechnik, Warburger Str. 100, Paderborn Corresponding author: Martin Horsch, phone: , martin.horsch@mv.uni-kl.de 1
2 2 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse simulation codes ms2 [1] as well as ls1 mardyn [2, 3], and by applying advanced simulation algorithms to complex molecular systems, employing the HPC resources at the Steinbuch Centre for Computing (SCC) in Karlsruhe. Various particular topics of relevance to the project are discussed in detail in dedicated publications to which the interested reader is referred here, concerning modelling and simulation of electrolyte solutions [4, 5], hydrogels [6, 7], and mixtures including hydrogen bonding fluids [8, 9, 10], as well as interfacial phenomena on small length scales [11, 12] and the role of long-range interactions in heterogeneous systems [12, 13]. The present article gives an overview, addressing the three main subjects of the MOCOS project by presenting recent scientific results which were facilitated by the computing resources at SCC: Section 2 discusses molecular model development for ions in aqueous solution and Section 3 reports on molecular simulation of large molecules, i.e. of enzymes and fatty acides. Interfacial phenomena such as the wetting behaviour of fluids in contact with a solid surface are discussed in Section 4 and a conclusion is given in Section 5. 2 Molecular modelling of aqueous electrolyte solutions 2.1 Force field development for alkali cations and halide anions Aqueous electrolyte solutions play an important role in many industrial applications and natural processes. The general investigation of electrolyte solutions is, hence, of prime interest. Their simulation on the molecular level is computationally expensive as the electrostatic long range interactions in the solution have to be taken into account by time consuming algorithms, such as Ewald summation [14]. The development of ion force fields in aqueous solutions is a challenging task because of the strong electrostatic interactions between the ions and the surrounding water molecules. Previously published parameterization strategies for the adjustment of the ion force fields of all alkali cations and halide anions yield multiple parameter sets for a single ion [15, 16] or are based on additional assumptions, e.g. the consideration of the aqueous alkali halide solution as a binary mixture of water and cations as well as anions, respectively [17]. The recent study of Deublein et al. [18], however, succeeded in obtaining one unique force field set for all alkali and halide ions in aqueous solution. Thereby, the ions are modelled as Lennard-Jones (LJ) spheres with a point charge (±1 e) in their centre of mass. Hence, the ion force fields have two adjustable parameters, namely the LJ size parameter σ and the LJ energy parameter ε. The σ parameter of the ions was adjusted to the reduced liquid solution density. The LJ energy parameter showed only a minor influence on the reduced density and was estimated to be ε = 100 K for all anions and cations [18]. In the present work, the influence of the LJ energy parameter on the self-diffusion coefficient of the alkali cations and the halide anions in aqueous solutions as well as
3 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 3 the position of the first maximum of the radial distribution function (RDF) of water around the ions was investigated systematically. Based on these results, a modified value is proposed for the LJ energy parameter. The new ε i parameter of the ion force fields is determined by a two step parametrization strategy. First, the LJ energy parameter of the ion force fields is adjusted to the self-diffusion coefficient of the ions in aqueous solution. Subsequently, the dependence of the position of the first maximum in the RDF of water around the ions on ε i is used to restrict the parameter range derived by considering the selfdiffusion coefficient. 2.2 Methods and simulation details In the present study, the self-diffusion coefficient of the ions in aqueous solutions is determined in equilibrium molecular dynamics by the Green-Kubo formalism. In this formalism, the self-diffusion coefficient is related to the time integral of the velocity autocorrelation function [19]. The radial distribution function g i O (r) of water around the ion i is defined by g i O (r)= ρ O(r) ρ O,bulk (1) where ρ O (r) is the local density of water as a function of the distance r from the ion i and ρ O,bulk is the density of water molecules in the bulk phase. In this case, the position of the water molecules is represented by the position of the oxygen atom. The radial distribution functions are evaluated by molecular dynamics (MD) simulation as well. The calculation of the self-diffusion coefficient by the Green-Kubo formalism and the evaluation of the RDF is time and memory consuming. The determination of D i and g i O (r) in electrolyte solutions is considerably more expensive due to additional time consuming algorithms, e.g. Ewald summation [14], required for permitting a truncation of the long range electrostatic interactions; however, it should be noted that a completely explicit evaluation of all pairwise interactions would be even much more expensive. In a first step, the density of the aqueous alkali halide solution was determined in an isobaric-isothermal (NpT) MD simulation at the desired temperature and pressure. The resulting density was used in a canonical (NVT) MD simulation at the same temperature, pressure and composition of the different alkali halide solutions. In this run, the self-diffusion coefficient of the ions as well as the radial distribution function, respectively, was determined. In case of the calculation of D i, the MD unit cell with periodic boundary conditions contained N = molecules, both for the NpT and the NVT simulation run. For the evaluation of the RDF, there were N = molecules in the simulation volume, i.e. 980 water molecules, 10 alkali cations and 10 halide anions. The radial
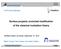
4 4 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse Fig. 1 Self-diffusion coefficient of alkali cations and halide anions in aqueous solutions (x S = mol/mol) at T = K and p=1 bar. Simulation results (full symbols) are compared to experimental data [20] (empty symbols). distribution function was sampled in the NVT simulation within a cutoff radius of 15 Å with 500 bins. 2.3 Self-diffusion coefficients and radial distribution functions The self-diffusion coefficient D i and the position of the first maximum r max,1 of the RDF g i O (r) of water around the ions was investigated for all alkali cations and halide anions in aqueous solution for ε = 200 K. These data were calculated at high dilution so that correlated motions and ion pairing between the cations and anions were avoided. Hence, D i and r max,1 are independent on the counter-ion in solution. The results for the self-diffusion coefficient D i are shown in Fig. 1. The overall agreement of the simulation results with the experimental data is excellent. The deviations are below 10 % for all ions, except for the sodium cation, where the deviation is about 20 %. These simulation results also follow the qualitative trends from experiment, i.e. D i increases with cation and anion size, respectively. This ion size dependence is directly linked to the electrostatic interaction between the ions and water. In aqueous solution, the cations and anions are surrounded by a shell of electrostatically bonded water molecules (hydration shell). The ions diffuse together with their hydration shell within the bulk water. For small ions, the hydration shell is firmly attached to the ion. Hence, the effective radius, that typically dominates ion motion, is larger for smaller ions than for larger ions, where the hydration shell is less pronounced. The results of r max,1 are shown in Fig. 2. In case of the alkali cations, the simulation results are within the range of the experimental data, except for Na + where the deviation from the experimental data is 5.3%. For the halide anions, only the simulation result for r max,1 of the RDF of water around the iodide anion is within the
5 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 5 range of the experimental data. The deviations of the simulation results for r max,1 from the experimental data are 12.1 % around F, 6.5 % around Cl, and 2.9 % around Br. Fig. 2 Position of the first maximum r max,1 of the RDF g i O (r) of water around the alkali cations and halide anions in aqueous solutions (x S = 0.01 mol/mol) at T = K and p=1 bar. Present simulation data (full symbols) are compared to the range of experimental r max,1 data [21, 22] (vertical lines). Comparing D i and r max,1 of the cesium cation and the fluoride anion, which have almost the same size, it can be seen that Cs + diffuses faster in aqueous solution and the water molecules of the hydration shell around F are closer to the ion. This can be attributed to the different orientations of the water molecules around the oppositely charged ions. The water molecules are able to build a stronger attached hydration shell around the fluoride ion which is closer to the ion. 2.4 Computational demands The molecular simulations in Section 2 were carried out with the MPI based molecular simulation program ms2, which was developed in our group, cf. Deublein et al. [1]. The total computing time for determining the self-diffusion coefficient of ions in aqueous solutions was 138 hours on 36 CPUs (48 hours for the NpT run and 90 hours for the NVT run). For these simulations a maximum virtual memory of 1.76 GB was used. For the evaluation of the radial distribution function of water around the ions a total computing time of 31 hours on 32 CPUs (10 hours for the NpT run and 21 hours for the NVT run) was required.
6 6 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse 3 Simulation of biomolecules with internal degrees of freedom 3.1 Catalysed hydroxylation of unsaturated fatty acids Producing polymers from regenerative feedstock is a highly interesting alternative to polymers made of naphtha. The first step for obtaining biopolymers is the synthesis and study of all possible basic materials that can be used as building blocks. Such materials can for instance be obtained by an enzymatically catalysed reaction of unsaturated fatty acids to dihydroxy-fatty acids. In nature, there are only mixtures saturated and unsaturated fatty acids rather than the pure compounds. Therefore, separation before or after the reaction is necessary to obtain pure products, e.g. by chromatography, using hydrotalcite as adsorbent and a mixture of water and isopropanol as solvent. Cytochrome P450 monooxygenase, an enzyme well-known for catalysing the hydroxylation of organic molecules, is a suitable catalyst for this process as well. The critical aspect of the catalytic reaction is the contact between the heme group, which contains the active centre of the enzyme, and the double bonds in the carbon chain of the fatty acid. The enzyme is denaturalized in the presence of organic molecules, and is only active in an aqueous phase. Fatty acids have a small dipole moment and are almost insoluble in water. So how does the contact between the active centre of the enzyme and the fatty acid take place? A series of molecular simulations was conducted to learn more about the distribution of molecules around the enzyme and the behaviour of the system at different conditions. For this purpose, the mixing behaviour of the systems fatty acid + water, fatty acid + water + isopropanol, and fatty acid + water + cytochrome P450 was investigated. In particular, it is relevant to know whether the fatty acid builds micelles in the water-isopropanol solvent, or how the enzyme catalyses the reaction despite the different phase behaviour of the solvent and the fatty acid. The fatty acid of interest in the present work is oleic acid. 3.2 Simulation details Molecular simulation of biological systems poses a challenge to scientific computing. The most important limitation in molecular simulation is the system size. As the number of molecules in the simulation box N increases, the computing time required for the simulation increases with O(N 2 ) if it is implemented in a naive way. The reason for this steep dependence is found in the computation of pair potentials, so that the distance and energy between interacting atoms needs to be computed at every simulation step. One option for decreasing the simulation time consists in following a coarsegraining approach [23]. By coarse-graining, a group of atoms is modelled as a single interaction centre, reducing the number of pair interactions that have to be cal-
7 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 7 culated. As we are interested in obtaining detailed atomistic information, we prefer to use a full atomistic model. The model we selected is OPLS [24], which has been successfully used to simulate a large variety of biological systems. The model for the heme group, inexistent in the original OPLS force field, was taken from a recent parameterization of this group compatible with the OPLS force field [25]. In these models, repulsion-dispersion interactions are treated with LJ potentials, while electrostatic interactions are taken into account considering point partial charges at the atomic positions. Internal molecular degrees of freedom are modelled via bond, bend, and torsion potentials. The cytochrome P450 enzyme contains more than atoms, and together with the fatty acid and the solvent molecules, we need to simulate up to atoms. Molecular simulation of such an enormous system would need months of simulation time unless we use advanced parallel simulation programs. For the present simulations of molecular biosystems, the GROMACS 4.6 MD program was used [26]. At the beginning of the simulation, the molecules were arranged in a randomized fashion within a periodic simulation box, and the energy was minimized. This procedure for generating a starting configuration attempts to mimic experimental conditions where initially the solutions are vigorously mixed. Then every molecule is assigned a random velocity (corresponding to the temperature of the system), and a short simulation of 2 ps is carried out for an initial equilibration. Subsequently, a long simulation (over a minimum simulation time of 20 ns) is run, where the properties of interest are calculated. The molecular positions are updated using a leapfrog algorithm to integrate Newton s equations of motion. We truncate the LJ potential at a cut-off radius of r c = 1.5 nm, and use the particle-mesh Ewald summation method [27] to calculate electro- Fig. 3 Cluster of oleic acid molecules in water-isopropanol solution. Dark blue, water; violet, isopropanol; light blue, oleic acid. The acid group of the oil molecules is painted in red.
8 8 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse static interactions. Temperature is controlled by velocity rescaling with a stochastic term, and pressure with a Berendsen barostat. More details about these simulation techniques can be found elsewhere [28]. Our simulations are performed under conditions for which reliable experimental data are available, i.e. at a temperature of 298 K and a pressure of 100 kpa. For the water-oleic acid system, we use a fatty acid mass fraction of 60 % and two different simulation boxes with approximately the same volume: A cubic box with V = (7.14nm) 3, and a orthorhombic box of the dimension nm 3. For the isopropanol-water-oleic acid system, we added 10 mg/ml of oleic acid to a mixture water/isopropanol with a mass fraction of 60 % (for isopropanol) in a cubic simulation box of (12 nm) 3. Finally, the oil/water concentration in the cytochrome P450 + water + oil system corresponded to a volume fraction of 30 % (for oleic acid) in a cubic box of (10 nm) 3, wherein a single cytochrome P450 enzyme was placed. Fig. 4 Cytochrome P450 in water-oleic acid solution. Cytochrome P450 is painted in orange; the heme group, in violet; the oil molecules, in light blue. The acid group of the oil molecules is painted in red, the double bond in green. For clarity, water has been removed in the picture. 3.3 Simulation results Molecular simulations of oil in the presence of water show a swift phase separation, as it is expected from experimental observation. The phase separation takes place
9 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 9 only after a few picoseconds. Independently of the shape of the simulation box, the oleic acid forms a bilayer, while other possible structures are not observed. Oil molecules form a ordered phase, where their acid groups point in the direction of the water phase, and their hydrocarbon tails are aligned. This is a consequence of the well known fact that the acid group is hydrophilic while the hydrocarbon tail is hydrophobic. When we add isopropanol to the water-oil mixture, the situation changes, as shown in Fig. 3. Water and isopropanol are miscible, and their behaviour is similar as in a pure water-isopropanol solution. Oil molecules are mostly surrounded by isopropanol, which can be easily inferred from the radial distribution functions. On the other hand, the double bonds in the oil clusters tend to be in contact with each other. Oil in the presence of water and cytochrome P450 behaves similarly as in wateroil solutions, cf. Fig. 4. After a few nanoseconds a clear phase separation takes place, where oil forms an ordered, separate phase. Cytochrome P450 stays solvated in water and in contact with the oil phase only at specific points. At the beginning of the simulation there are several oil molecules in the vicinity of cytochrome P450 at favourable contact sites. These molecules maintain their contact with the enzyme during the whole simulation in a position where an interaction with the active centre (which is situated within the heme group) is possible. 3.4 Computational demands The computational demands of our simulations were highly dependent on the type of simulation. For the energy minimization simulations, it was sufficient to use a single processor for several minutes. For equilibration, we used 16 CPUs running in parallel for a maximum of 3.5 hours. The most demanding simulations during production required 256 CPUs running for 180 hours and a virtual RAM of 2.8 GB. 4 Vapour-liquid interfaces and wetting of component surfaces 4.1 Planar vapour-liquid interfaces For simulating vapour-liquid coexistence on the molecular level, a long range correction is needed to obtain accurate results. Thereby, the dispersive contribution to the potential energy U i of a molecule i U i = u i j +Ui LRC (2) r i j <r c
10 10 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse Fig. 5 Strong scaling with N = CLJ particles and 512 slabs. The blue dots denote the run time of the whole program, while the red dots only denote the run time of the long range correction. The solid lines represent ideally linear scaling. is calculated as a sum of the explicitly computed part and the long range correction (LRC), where the latter consists of a summation over N s slabs, employing a periodic boundary condition. The LRC is only applied in the direction normal to the planar interface, corresponding to the y direction here Ui LRC N s = k u LRC i,k (y i,y k ), (3) given that the system is homogeneous in the other directions. The correction terms u LRC i,k (y i,y k ) for the slabs are calculated as an integral over the slab volume. The resulting term for the potential energy only depends on the distance between the slabs r, the density ρ and the thickness y of the slabs [29]. The slabwise interaction is computed in pairs, so that 2 1N2 s individual contributions have to be computed. Usually, the number of threads is much smaller than the number of slabs. For a scaling test, a system with N = two-centre Lennard-Jones (2CLJ) particles was simulated with different numbers of threads and 512 slabs. Fig. 5 shows the results for the strong scaling behaviour. For small numbers of threads, the program scales almost ideally, i.e. linearly. For larger numbers of threads, the communication between the threads and the decomposition of the particles becomes more time consuming. Eventually, the long range correction requires even more communication between the threads than the rest of the program, since the density profile has to be sent to every thread before the slab interaction begins. After the slab interaction has been calculated, the correction terms for the potential energy, the force and the virial have to be distributed to every thread. On the basis of the long range correction discussed above, vapour-liquid equilibria were considered by explicit MD simulation of the fluid phase coexistence (i.e. including an interface), so that the vapour-liquid surface tension γ could be computed following the virial route. For the (single centre) LJ fluid, a high precision
11 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 11 was obtained by simulating N = particles over roughly a million time steps, resulting in very small statistical uncertainties. From the resulting surface tension values, the regression term ( γ = T ) 1.23 ε T c σ 2 (4) was obtained, where the LJ critical temperature is given by Pérez Pellitero et al. [30] as T c = ε. 4.2 Curved vapour-liquid interfaces The influence of curvature on vapour-liquid interfacial properties was examined for the trunacted-shifted Lennard-Jones fluid (TSLJ), with a cutoff radius of r c = 2.5 σ, by simulating both curved (i.e. bubble and droplet) as well as planar interfaces by MD simulation of the canonical ensemble at a reduced temperature of T = 0.75 ε. For the system with planar symmetry, half of the simulation volume was filled with vapour and the other half with liquid, using a simulation box with a total elongation of 17 σ. The density profile from this simulation was compared with that of a bubble with an equimolar radius of R ρ = 12.6 σ as well as a droplet with R ρ = 12.4 σ, cf. Fig. 6. A system containing a bubble is subsaturated, whereas the fluid phases coexisting at the curved interface of a droplet are supersaturated with respect to the thermodynamic equilibrium condition for the bulk phases coexisting at saturation. In agreement with this qualitative statement from phenomenological thermodynamics, the vapour density (i.e. the minimal value from the density profile) was found to be smaller for the system containing a bubble (ρ = σ 3 ) and larger for the system containing a droplet ( σ 3 ), whereas the vapour density over the planar interface was σ 3. Analogous results were obtained for the chemical potential µ total = µ id + µ conf, which was determined as the sum of the ideal contribution µ id = T lnρ and the configurational contribution µ conf. The Widom test particle method [31] was implemented to compute a profile of µ conf, which in equilibrium is complementary to the logarithmic density profile, yielding an approximately constant profile for the total chemical potential, cf. Fig. 6. For the droplet, an average value of µ total = ( 3.31±0.04) ε was determined, in comparison to( 3.51±0.02) ε for the bubble and ( 3.37±0.02) ε for the planar slab. This shows that in the present case, curvature effects are more significant for a gas bubble than for a liquid droplet of the same size, suggesting that the surface tension of the bubble is larger than that of the droplet.
12 12 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse Fig. 6 Density profiles (top) and chemical potential profiles (bottom) for nanoscopic vapour-liquid interfaces of the TSLJ fluid from canonical ensemble MD simulation of systems containing planar liquid and vapour slabs, a liquid drop (surrounded by a supersaturated vapour), or a gas bubble (surrounded by a subsaturated liquid), respectively. The chemical potential profile includes information on both the configurational contribution (circles) and the total chemical potential (solid lines), as a function of the characteristic spatial coordinate of the system. The solid lines in the bottom left part of the two diagrams indicate the density of the vapour phase (top) as well as the average value of the total chemical potential (bottom). 4.3 Temperature dependence of contact angles The research on the wetting behaviour of surfaces is an active field in materials science. The goal is to reliably predict the wetting properties of component surfaces for design of components with new functional features. In this regard molecular simulations are particularly useful due to their high resolution. Moreover, molecular simulation permits the systematic investigation of the influence of particular parameters on the wetting behaviour. For the present study, the TSLJ potential was employed and the contact angle dependence on temperature for a non-wetting (θ > 90 ) and a partially wetting (θ < 90 ) scenario, cf. Fig. 7 was investigated by MD simulation. The solid wall was represented by a face centered cubic lattice with a lattice constant of a=1.55 σ and the (001) surface exposed to the fluid. For the unlike interaction between the solid and the fluid phase the size and energy parameters σ sf = σ ff and Fig. 7 Snapshots of simulation runs at different fluid-solid interaction parameters ζ of 0.35 (left) and 0.65 (right) at a temperature of 0.8 ε/k.
13 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 13 ε sf = ζε ff, (5) respectively, were applied. The simulations were carried out with the massively parallel MD program ls1 mardyn [32]. A total number of N = fluid particles was simulated in the NVT ensemble. In most cases, 64 processes were employed for parallel computation. The efficient simulation of heterogeneous systems, containing vapour-liquid interfaces and even a solid component, was facilitated here by a dynamic load balancing scheme based on k-dimensional trees [32]. At the beginning of the simulation, the fluid particles were arranged on regular lattice sites in form of a cuboid. The equilibration time of the three phase system was 2 ns, whereas the total simulation time was chosen to be 6 ns. Periodic boundary conditions were applied in all directions, leaving a channel with a distance of at least 35 σ between the wall and its periodic image, avoiding confinement effects at near-critical temperatures [33]. The contact angles were determined by the evaluation of density profiles averaged over a time span of 4 ns. Sessile drops were studied at two different interaction parameters, ζ = 0.35 and 0.65, corresponding to partially wetting and non-wetting conditions. The simulations were carried out in the temperature range between 0.7 and 1 ε, covering nearly the entire regime of stable vapour-liquid coexistence for the TSLJ fluid, as indicated by the triple point temperature T ε, cf. van Meel et al. [34], and the critical temperature T c ε, cf. Vrabec et al. [35]. It is well known that the respective wetting behaviour of the sytem, i.e. wetting or dewetting, is reinforced at higher temperatures, cf. Fig. 8. Close to the critical point of the fluid, the phenomenon of critical point wetting occurs [36], i.e. either the liquid or the vapour phase perfectly wets the solid surface cosθ(t T c ) 1. (6) Fig. 8 Simulation results and correlation for the dependence of the contact angle on the temperature for fluid-solid dispersive interaction strengths ζ of 0.35 (circles, dashed line) and 0.65 (triangles, dash-dotted line).
14 14 Horsch, Becker, Castillo, Deublein, Fröscher, Reiser, Werth, Vrabec, and Hasse 5 Conclusion Within the MOCOS project, a substantial progress was made regarding software development for massively-parallel molecular simulation. In particular, suitable algorithms for the long range contributions to the intermolecular interaction were implemented in ms2 (Ewald summation) and ls1 mardyn (Janeček correction for long range dispersive effects). In ls1 mardyn, a k-dimensional tree-based domain decomposition with dynamic load balancing was implemented to respond to an uneven distribution of the interaction sites over the simulation volume. Furthermore, GRO- MACS [26] was used to simulate large molecules, taking their internal degrees of freedom into account. In this way, systems with up to interaction sites were simulated for over 20 nanoseconds. New models were developed for alkali cations and halide anions in order to treat the dispersive interaction accurately in molecular simulations of electrolye solutions. These models were optimized by adjusting the LJ size parameter to the reduced density of aqueous solutions and the energy parameter to diffusion coefficients as well as pair correlation functions. On this basis, quantitatively reliable results were obtained regarding the structure of the hydration shells formed by the water molecules surrounding these ions. Important qualitative structural information on biomolecules at oil-water interfaces was deduced by analysing MD simulations of the cytochrome P450 enzyme, and the critical wetting behaviour as well as the properties of planar and curved vapour-liquid interfaces were characterized by massively-parallel MD simulation with the ls1 mardyn program. The scenarios disussed above show how molecular modelling can today be applied to practically relevant systems even if they exhibit highly complex structures and intermolecular interactions, including significant long range contributions. In the immediate future, molecular simulation as an application of high performance computing will therefore be able to play a crucial role not only by contributing to scientific progress, but also in its day-to-day use as a research and development tool in mechanical and process engineering as well as biotechnology. Acknowledgements The present work was carried out under the auspices of the Boltzmann-Zuse Society for Computational Molecular Engineering (BZS), and the molecular simulations were conducted on the XC4000 supercomputer at the Steinbuch Centre for Computing, Karlsruhe. The authors would like to thank Akshay Bedhotiya (Bombay) for his assistance regarding the simulation of gas bubbles and Sergey Lishchuk (Leicester), Martin Buchholz, Wolfgang Eckhardt, and Ekaterina Elts (München) and Gábor Rutkai (Paderborn) as well as Martin Bernreuther, Colin Glass, and Christoph Niethammer (Stuttgart) for contributing to the development of the ls1 mardyn and ms2 molecular simulation programs as well as Deutsche Forschungsgemeinschaft (DFG) for funding the Collaborative Research Centre (SFB) 926. Furthermore, they would like to acknowledge fruitful discussions with Cemal Engin, Michael Kopnarski, Birgit and Rolf Merz as well as Michael Schappals and Rajat Srivastava (Kaiserslautern), George Jackson and Erich Müller (London), Jonathan Walter (Ludwigshafen), Philippe Ungerer and Marianna Yiannourakou (Paris) as well as Nichola McCann (Visp).
15 Molecular simulation of electrolytes, biomolecules, and component surfaces (MOCOS) 15 References 1. S. Deublein, B. Eckl, J. Stoll, S.V. Lishchuk, G. Guevara Carrión, C.W. Glass, T. Merker, M. Bernreuther, H. Hasse, J. Vrabec, Comput. Phys. Comm. 182(11), 2350 (2011) 2. W. Eckhardt, A. Heinecke, R. Bader, M. Brehm, N. Hammer, H. Huber, H.G. Kleinhenz, J. Vrabec, H. Hasse, M. Horsch, M. Bernreuther, C.W. Glass, C. Niethammer, A. Bode, J. Bungartz, in Proc. ISC 2013 (Springer, Heidelberg, 2013), no in LNCS, pp M. Horsch, C. Niethammer, J. Vrabec, H. Hasse, Informat. Technol. 55(3), 97 (2013) 4. S. Deublein, S. Reiser, J. Vrabec, H. Hasse, J. Phys. Chem. B 116(18), 5448 (2012) 5. S. Deublein, S. Reiser, J. Vrabec, H. Hasse, submitted (2013) 6. J. Walter, V. Ermatchkov, J. Vrabec, H. Hasse, Fluid Phase Equilib. 296(2), 164 (2010) 7. J. Walter, J. Sehrt, J. Vrabec, H. Hasse, J. Phys. Chem. B 116(17), 5251 (2012) 8. Y.L. Huang, T. Merker, M. Heilig, H. Hasse, J. Vrabec, Ind. Eng. Chem. Res. 51(21), 7428 (2012) 9. S. Reiser, N. McCann, M. Horsch, H. Hasse, J. Supercrit. Fluids 68, 94 (2012) 10. S. Pařez, G. Guevara Carrión, H. Hasse, J. Vrabec, Phys. Chem. Chem. Phys. 15(11), 3985 (2013) 11. M. Horsch, H. Hasse, submitted (2013) 12. S. Werth, S.V. Lishchuk, M. Horsch, H. Hasse, Phys. A 392(10), 2359 (2013) 13. S. Werth, G. Rutkai, J. Vrabec, M. Horsch, H. Hasse, in preparation (2013) 14. P.P. Ewald, Ann. Phys. (Leipzig) 369(3), 253 (1921) 15. D. Horinek, S.I. Mamatkulov, R.R. Netz, J. Chem. Phys. 130, (2009) 16. M.M. Reif, P.H. Hünenberger, J. Chem. Phys. 134, (2011) 17. M.B. Gee, N.R. Cox, Y.F. Jiao, N. Bentenitis, S. Weerasinghe, J. Chem. Theory Computat. 7(5), 1369 (2011) 18. S. Deublein, J. Vrabec, H. Hasse, J. Chem. Phys. 136, (2012) 19. K. Gubbins, Statistical Mechanics, vol. 1 (Burlington House, London, 1972) 20. R. Mills, V. Lobo, Self-Diffusion in Electrolyte Solutions (Elsevier, New York, 1989) 21. H. Ohtaki, T. Radnai, Chem. Rev. 93(3), 1157 (1993) 22. J.L. Fulton, D.M. Pfund, S.L. Wallen, M. Newville, E.A. Stern, J. Ma, J. Chem. Phys. 105(6), 2161 (1996) 23. F. Müller Plathe, ChemPhysChem 3(9), 754 (2002) 24. W.L. Jørgensen, D.S. Maxwell, J. Tirado Rives, J. Am. Chem. Soc. 110(45), (1996) 25. V. Gogonea, J.M. Shy II., P.K. Biswas, J. Phys. Chem. B 110(45), (2006) 26. S. Pronk, P. Szilárd, R. Schulz, P. Larsson, P. Bjelkmar, R. Apostolov, M.R. Shirts, J.C. Smith, P.M. Kasson, D. van der Spoel, B. Hess, E. Lindahl, Bioinformatics 29(7), 845 (2013) 27. M.E. Tuckerman, G.J. Martyna, J. Phys. Chem. B 104(2), 159 (2000) 28. D. Frenkel, B. Smit, Understanding Molecular Simulation (Academic Press, San Diego, 2002) 29. J. Janeček, J. Phys. Chem. B 110(12), 6264 (2006) 30. J. Pérez Pellitero, P. Ungerer, G. Orkoulas, A.D. Mackie, J. Chem. Phys. 125, (2006) 31. B. Widom, J. Chem. Phys. 86(6), 869 (1982) 32. M. Buchholz, H.J. Bungartz, J. Vrabec, J. Computational Sci. 2(2), 124 (2011) 33. A. Oleinikova, I. Brovchenko, A. Geiger, Eur. Phys. J. B 52(4), 507 (2006) 34. J.A. van Meel, A.J. Page, R.P. Sear, D. Frenkel, J. Chem. Phys. 129, (2008) 35. J. Vrabec, G.K. Kedia, G. Fuchs, H. Hasse, Molec. Phys. 104(9), 1509 (2006) 36. J.W. Cahn, J. Chem. Phys. 66(8), 3667 (1977)
Scalable, performant, and resilient large-scale applications of molecular process engineering
 Scalable, performant, and resilient large-scale applications of molecular process engineering M. Horsch,1 P. Gralka,2 C. Niethammer,3 N. Tchipev,4 J. Vrabec,5 H. Hasse1 1 University of Kaiserslautern,
Scalable, performant, and resilient large-scale applications of molecular process engineering M. Horsch,1 P. Gralka,2 C. Niethammer,3 N. Tchipev,4 J. Vrabec,5 H. Hasse1 1 University of Kaiserslautern,
Molecular modeling of hydrogen bonding fluids: vapor-liquid coexistence and interfacial properties
 12 th HLRS Results and Review Workshop Molecular modeling of hydrogen bonding fluids: vapor-liquid coexistence and interfacial properties High Performance Computing Center Stuttgart (HLRS), October 8,
12 th HLRS Results and Review Workshop Molecular modeling of hydrogen bonding fluids: vapor-liquid coexistence and interfacial properties High Performance Computing Center Stuttgart (HLRS), October 8,
Surface tension of the two center Lennard-Jones plus quadrupole model fluid
 Surface tension of the two center Lennard-Jones plus quadrupole model fluid Stephan Werth, Martin Horsch 1, Hans Hasse Laboratory of Engineering Thermodynamics, Department of Mechanical and Process Engineering,
Surface tension of the two center Lennard-Jones plus quadrupole model fluid Stephan Werth, Martin Horsch 1, Hans Hasse Laboratory of Engineering Thermodynamics, Department of Mechanical and Process Engineering,
Molecular Modeling and Simulation of Phase Equilibria for Chemical Engineering
 InPROMT 2012, Berlin, 16. November 2012 DFG Transregio CRC 63 Molecular Modeling and Simulation of Phase Equilibria for Chemical Engineering Hans Hasse 1, Martin Horsch 1, Jadran Vrabec 2 1 Laboratory
InPROMT 2012, Berlin, 16. November 2012 DFG Transregio CRC 63 Molecular Modeling and Simulation of Phase Equilibria for Chemical Engineering Hans Hasse 1, Martin Horsch 1, Jadran Vrabec 2 1 Laboratory
arxiv: v1 [physics.comp-ph] 25 Jul 2015
![arxiv: v1 [physics.comp-ph] 25 Jul 2015 arxiv: v1 [physics.comp-ph] 25 Jul 2015](/thumbs/84/89137729.jpg) To appear in Molecular Physics Vol. 11, No. 17, 014, 1 13 arxiv:07.07130v1 [physics.comp-ph] 5 Jul 0 Long range correction for multi-site Lennard-Jones models and planar interfaces Stephan Werth 1, Gabor
To appear in Molecular Physics Vol. 11, No. 17, 014, 1 13 arxiv:07.07130v1 [physics.comp-ph] 5 Jul 0 Long range correction for multi-site Lennard-Jones models and planar interfaces Stephan Werth 1, Gabor
Large-scale MD simulation of heterogeneous systems with ls1 mardyn
 Large-scale MD simulation of heterogeneous systems with ls1 mardyn M. T. Horsch, R. Srivastava, S. J. Werth, C. Niethammer, C. W. Glass, W. Eckhardt, A. Heinecke, N. Tchipev, H.-J. Bungartz, S. Eckelsbach,
Large-scale MD simulation of heterogeneous systems with ls1 mardyn M. T. Horsch, R. Srivastava, S. J. Werth, C. Niethammer, C. W. Glass, W. Eckhardt, A. Heinecke, N. Tchipev, H.-J. Bungartz, S. Eckelsbach,
Dependence of the surface tension on curvature
 Dependence of the surface tension on curvature rigorously determined from the density profiles of nanodroplets Athens, st September M. T. Horsch,,, S. Eckelsbach, H. Hasse, G. Jackson, E. A. Müller, G.
Dependence of the surface tension on curvature rigorously determined from the density profiles of nanodroplets Athens, st September M. T. Horsch,,, S. Eckelsbach, H. Hasse, G. Jackson, E. A. Müller, G.
Molecular modelling and simulation of hydrogen bonding fluids
 Molecular modelling and simulation of hydrogen bonding fluids Martin Horsch, Alptekin Celik and Hans Hasse Lehrstuhl für Thermodynamik (LTD) Technische Universität Kaiserslautern Rostock, 27 th March 13
Molecular modelling and simulation of hydrogen bonding fluids Martin Horsch, Alptekin Celik and Hans Hasse Lehrstuhl für Thermodynamik (LTD) Technische Universität Kaiserslautern Rostock, 27 th March 13
Introduction to molecular dynamics
 1 Introduction to molecular dynamics Yves Lansac Université François Rabelais, Tours, France Visiting MSE, GIST for the summer Molecular Simulation 2 Molecular simulation is a computational experiment.
1 Introduction to molecular dynamics Yves Lansac Université François Rabelais, Tours, France Visiting MSE, GIST for the summer Molecular Simulation 2 Molecular simulation is a computational experiment.
Atomistic molecular simulations for engineering applications: methods, tools and results. Jadran Vrabec
 Atomistic molecular simulations for engineering applications: methods, tools and results Jadran Vrabec Motivation Simulation methods vary in their level of detail The more detail, the more predictive power
Atomistic molecular simulations for engineering applications: methods, tools and results Jadran Vrabec Motivation Simulation methods vary in their level of detail The more detail, the more predictive power
MD simulation of methane in nanochannels
 MD simulation of methane in nanochannels COCIM, Arica, Chile M. Horsch, M. Heitzig, and J. Vrabec University of Stuttgart November 6, 2008 Scope and structure Molecular model for graphite and the fluid-wall
MD simulation of methane in nanochannels COCIM, Arica, Chile M. Horsch, M. Heitzig, and J. Vrabec University of Stuttgart November 6, 2008 Scope and structure Molecular model for graphite and the fluid-wall
Density of methanolic alkali halide salt solutions by. experiment and molecular simulation
 Density of methanolic alkali halide salt solutions by experiment and molecular simulation Steffen Reiser, Martin Horsch, and Hans Hasse Laboratory of Engineering Thermodynamics, University of Kaiserslautern,
Density of methanolic alkali halide salt solutions by experiment and molecular simulation Steffen Reiser, Martin Horsch, and Hans Hasse Laboratory of Engineering Thermodynamics, University of Kaiserslautern,
Surface analysis algorithms in the mardyn program and the ls1 project
 Surface analysis algorithms in the mardyn program and the ls1 project Stuttgart, 15 th December 1 M. T. Horsch Surface tension The virial route Bakker-Buff equation: γ R 2 out in dz z Normal pressure decays
Surface analysis algorithms in the mardyn program and the ls1 project Stuttgart, 15 th December 1 M. T. Horsch Surface tension The virial route Bakker-Buff equation: γ R 2 out in dz z Normal pressure decays
Static and Dynamic Properties of Curved Vapour-Liquid Interfaces by Massively Parallel Molecular Dynamics Simulation
 Static and Dynamic Properties of Curved Vapour-Liquid Interfaces by Massively Parallel Molecular Dynamics Simulation M.T. Horsch, S.K. Miroshnichenko, J. Vrabec, C.W. Glass, C. Niethammer, M.F. Bernreuther,
Static and Dynamic Properties of Curved Vapour-Liquid Interfaces by Massively Parallel Molecular Dynamics Simulation M.T. Horsch, S.K. Miroshnichenko, J. Vrabec, C.W. Glass, C. Niethammer, M.F. Bernreuther,
Molecular Dynamic Simulation Study of the Volume Transition of PNIPAAm Hydrogels
 Molecular Dynamic Simulation Study of the Volume Transition of PNIPAAm Hydrogels Jonathan Walter 1, Jadran Vrabec 2, Hans Hasse 1 1 Laboratory of Engineering, University of Kaiserslautern, Germany 2 and
Molecular Dynamic Simulation Study of the Volume Transition of PNIPAAm Hydrogels Jonathan Walter 1, Jadran Vrabec 2, Hans Hasse 1 1 Laboratory of Engineering, University of Kaiserslautern, Germany 2 and
Supplementary material to Simultaneous description of bulk and interfacial properties of fluids by the Mie potential
 To appear in Molecular Physics Vol. 00, No. 00, Month 2016, 1 11 Supplementary material to Simultaneous description of bul and interfacial properties of fluids by the Mie potential Stephan Werth 1, Katrin
To appear in Molecular Physics Vol. 00, No. 00, Month 2016, 1 11 Supplementary material to Simultaneous description of bul and interfacial properties of fluids by the Mie potential Stephan Werth 1, Katrin
Sunyia Hussain 06/15/2012 ChE210D final project. Hydration Dynamics at a Hydrophobic Surface. Abstract:
 Hydration Dynamics at a Hydrophobic Surface Sunyia Hussain 6/15/212 ChE21D final project Abstract: Water is the universal solvent of life, crucial to the function of all biomolecules. Proteins, membranes,
Hydration Dynamics at a Hydrophobic Surface Sunyia Hussain 6/15/212 ChE21D final project Abstract: Water is the universal solvent of life, crucial to the function of all biomolecules. Proteins, membranes,
What is Classical Molecular Dynamics?
 What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential functions Newton s equations of motion are integrated
What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential functions Newton s equations of motion are integrated
On the calculation of solvation free energy from Kirkwood- Buff integrals: A large scale molecular dynamics study
 On the calculation of solvation free energy from Kirkwood- Buff integrals: A large scale molecular dynamics study Wynand Dednam and André E. Botha Department of Physics, University of South Africa, P.O.
On the calculation of solvation free energy from Kirkwood- Buff integrals: A large scale molecular dynamics study Wynand Dednam and André E. Botha Department of Physics, University of South Africa, P.O.
Long-range correction for dipolar fluids at planar interfaces
 To appear in Molecular Physics Vol. 00, No. 00, Month 2015, 1 16 Long-range correction for dipolar fluids at planar interfaces Stephan Werth, Martin Horsch and Hans Hasse Laboratory of Engineering Thermodynamics,
To appear in Molecular Physics Vol. 00, No. 00, Month 2015, 1 16 Long-range correction for dipolar fluids at planar interfaces Stephan Werth, Martin Horsch and Hans Hasse Laboratory of Engineering Thermodynamics,
Density of ethanolic alkali halide salt solutions by experiment and molecular simulation
 Density of ethanolic alkali halide salt solutions by experiment and molecular simulation Steffen Reiser, Martin Horsch, Hans Hasse Laboratory of Engineering Thermodynamics, University of Kaiserslautern
Density of ethanolic alkali halide salt solutions by experiment and molecular simulation Steffen Reiser, Martin Horsch, Hans Hasse Laboratory of Engineering Thermodynamics, University of Kaiserslautern
Water structure near single and multi-layer nanoscopic hydrophobic plates of varying separation and interaction potentials
 Bull. Mater. Sci., Vol. 31, No. 3, June 2008, pp. 525 532. Indian Academy of Sciences. Water structure near single and multi-layer nanoscopic hydrophobic plates of varying separation and interaction potentials
Bull. Mater. Sci., Vol. 31, No. 3, June 2008, pp. 525 532. Indian Academy of Sciences. Water structure near single and multi-layer nanoscopic hydrophobic plates of varying separation and interaction potentials
6 Hydrophobic interactions
 The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
The Physics and Chemistry of Water 6 Hydrophobic interactions A non-polar molecule in water disrupts the H- bond structure by forcing some water molecules to give up their hydrogen bonds. As a result,
 A Nobel Prize for Molecular Dynamics and QM/MM What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential
A Nobel Prize for Molecular Dynamics and QM/MM What is Classical Molecular Dynamics? Simulation of explicit particles (atoms, ions,... ) Particles interact via relatively simple analytical potential
Surface property corrected modification of the classical nucleation theory
 CCP5 Annual Meeting Surface property corrected modification of the classical nucleation theory Sheffield Hallam University, September 15, 2010 Martin Horsch, Hans Hasse, and Jadran Vrabec The critical
CCP5 Annual Meeting Surface property corrected modification of the classical nucleation theory Sheffield Hallam University, September 15, 2010 Martin Horsch, Hans Hasse, and Jadran Vrabec The critical
Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract
 Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract In this work, a simplified Lennard-Jones (LJ) sphere model is used to simulate the aggregation,
Structuring of hydrophobic and hydrophilic polymers at interfaces Stephen Donaldson ChE 210D Final Project Abstract In this work, a simplified Lennard-Jones (LJ) sphere model is used to simulate the aggregation,
We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions.
 CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
CHEM 2060 Lecture 10: Electrostatics L10-1 Electrostatics of Atoms & Molecules We have considered how Coulombic attractions and repulsions help to organize electrons in atoms and ions. We now look at Coulombic
CARBON 2004 Providence, Rhode Island. Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores
 CARBON Providence, Rhode Island Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores D. D. Do* and H. D. Do, University of Queensland, St. Lucia, Qld 7, Australia
CARBON Providence, Rhode Island Adsorption of Flexible n-butane and n-hexane on Graphitized Thermal Carbon Black and in Slit Pores D. D. Do* and H. D. Do, University of Queensland, St. Lucia, Qld 7, Australia
Supplemntary Infomation: The nanostructure of. a lithium glyme solvate ionic liquid at electrified. interfaces
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 207 Supplemntary Infomation: The nanostructure of a lithium glyme solvate ionic liquid
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 207 Supplemntary Infomation: The nanostructure of a lithium glyme solvate ionic liquid
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers
 Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
Supplementary Information for: Controlling Cellular Uptake of Nanoparticles with ph-sensitive Polymers Hong-ming Ding 1 & Yu-qiang Ma 1,2, 1 National Laboratory of Solid State Microstructures and Department
 Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Molecular Dynamics Simulation of a Nanoconfined Water Film
 Molecular Dynamics Simulation of a Nanoconfined Water Film Kyle Lindquist, Shu-Han Chao May 7, 2013 1 Introduction The behavior of water confined in nano-scale environment is of interest in many applications.
Molecular Dynamics Simulation of a Nanoconfined Water Film Kyle Lindquist, Shu-Han Chao May 7, 2013 1 Introduction The behavior of water confined in nano-scale environment is of interest in many applications.
Molecular models for cyclic alkanes and ethyl acetate as well as surface tension data from molecular simulation
 Molecular models for cyclic alkanes and ethyl acetate as well as surface tension data from molecular simulation Stefan Eckelsbach 1, Tatjana Janzen 1, Andreas Köster 1, Svetlana Miroshnichenko 1, Yonny
Molecular models for cyclic alkanes and ethyl acetate as well as surface tension data from molecular simulation Stefan Eckelsbach 1, Tatjana Janzen 1, Andreas Köster 1, Svetlana Miroshnichenko 1, Yonny
Hydrogels in charged solvents
 Hydrogels in charged solvents Peter Košovan, Christian Holm, Tobias Richter 27. May 2013 Institute for Computational Physics Pfaffenwaldring 27 D-70569 Stuttgart Germany Tobias Richter (ICP, Stuttgart)
Hydrogels in charged solvents Peter Košovan, Christian Holm, Tobias Richter 27. May 2013 Institute for Computational Physics Pfaffenwaldring 27 D-70569 Stuttgart Germany Tobias Richter (ICP, Stuttgart)
Molecular Dynamics Simulations. Dr. Noelia Faginas Lago Dipartimento di Chimica,Biologia e Biotecnologie Università di Perugia
 Molecular Dynamics Simulations Dr. Noelia Faginas Lago Dipartimento di Chimica,Biologia e Biotecnologie Università di Perugia 1 An Introduction to Molecular Dynamics Simulations Macroscopic properties
Molecular Dynamics Simulations Dr. Noelia Faginas Lago Dipartimento di Chimica,Biologia e Biotecnologie Università di Perugia 1 An Introduction to Molecular Dynamics Simulations Macroscopic properties
Mutual diffusion in the ternary mixture of water + methanol + ethanol: Experiments and Molecular Simulation
 - 1 - Mutual diffusion in the ternary mixture of water + methanol + ethanol: Experiments and Molecular Simulation Tatjana Janzen, Gabriela Guevara-Carrión, Jadran Vrabec University of Paderborn, Germany
- 1 - Mutual diffusion in the ternary mixture of water + methanol + ethanol: Experiments and Molecular Simulation Tatjana Janzen, Gabriela Guevara-Carrión, Jadran Vrabec University of Paderborn, Germany
arxiv: v3 [cond-mat.mes-hall] 12 Mar 2010
![arxiv: v3 [cond-mat.mes-hall] 12 Mar 2010 arxiv: v3 [cond-mat.mes-hall] 12 Mar 2010](/thumbs/96/128210233.jpg) Contact angle dependence on the fluid-wall arxiv:1001.2681v3 [cond-mat.mes-hall] 12 Mar 2010 dispersive energy Martin Horsch, Martina Heitzig,, Calin Dan, Jens Harting,, Hans Hasse, and Jadran Vrabec,
Contact angle dependence on the fluid-wall arxiv:1001.2681v3 [cond-mat.mes-hall] 12 Mar 2010 dispersive energy Martin Horsch, Martina Heitzig,, Calin Dan, Jens Harting,, Hans Hasse, and Jadran Vrabec,
Module 5: "Adsoption" Lecture 25: The Lecture Contains: Definition. Applications. How does Adsorption occur? Physisorption Chemisorption.
 The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
The Lecture Contains: Definition Applications How does Adsorption occur? Physisorption Chemisorption Energetics Adsorption Isotherms Different Adsorption Isotherms Langmuir Adsorption Isotherm file:///e
Phase Equilibria of binary mixtures by Molecular Simulation and PR-EOS: Methane + Xenon and Xenon + Ethane
 International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol.5, No.6, pp 2975-2979, Oct-Dec 2013 Phase Equilibria of binary mixtures by Molecular Simulation and PR-EOS: Methane +
International Journal of ChemTech Research CODEN( USA): IJCRGG ISSN : 0974-4290 Vol.5, No.6, pp 2975-2979, Oct-Dec 2013 Phase Equilibria of binary mixtures by Molecular Simulation and PR-EOS: Methane +
Supporting Information
 Projection of atomistic simulation data for the dynamics of entangled polymers onto the tube theory: Calculation of the segment survival probability function and comparison with modern tube models Pavlos
Projection of atomistic simulation data for the dynamics of entangled polymers onto the tube theory: Calculation of the segment survival probability function and comparison with modern tube models Pavlos
Specific Ion Solvtion in Ethylene Carbonate and Propylene Carbonate
 Specific Ion Solvtion in Ethylene Carbonate and Propylene Carbonate A. Arslanargin, A. Powers, S. Rick, T. Pollard, T. Beck Univ Cincinnati Chemistry Support: NSF, OSC TSRC 2016 November 2, 2016 A. Arslanargin,
Specific Ion Solvtion in Ethylene Carbonate and Propylene Carbonate A. Arslanargin, A. Powers, S. Rick, T. Pollard, T. Beck Univ Cincinnati Chemistry Support: NSF, OSC TSRC 2016 November 2, 2016 A. Arslanargin,
Mutual diffusion of binary liquid mixtures containing methanol, ethanol, acetone, benzene, cyclohexane, toluene and carbon tetrachloride
 - 1 - PLMMP 2016, Kyiv Mutual diffusion of binary liquid mixtures containing methanol, ethanol, acetone, benzene, cyclohexane, toluene and carbon tetrachloride Jadran Vrabec Tatjana Janzen, Gabriela Guevara-Carrión,
- 1 - PLMMP 2016, Kyiv Mutual diffusion of binary liquid mixtures containing methanol, ethanol, acetone, benzene, cyclohexane, toluene and carbon tetrachloride Jadran Vrabec Tatjana Janzen, Gabriela Guevara-Carrión,
EQUATION OF STATE DEVELOPMENT
 EQUATION OF STATE DEVELOPMENT I. Nieuwoudt* & M du Rand Institute for Thermal Separation Technology, Department of Chemical Engineering, University of Stellenbosch, Private bag X1, Matieland, 760, South
EQUATION OF STATE DEVELOPMENT I. Nieuwoudt* & M du Rand Institute for Thermal Separation Technology, Department of Chemical Engineering, University of Stellenbosch, Private bag X1, Matieland, 760, South
Activities in Aqueous Solutions of the Alkali Halide. Salts from Molecular Simulation
 Activities in Aqueous Solutions of the Alkali Halide Salts from Molecular Simulation Maximilian Kohns, Michael Schappals, Martin Horsch, and Hans Hasse Laboratory of Engineering Thermodynamics, University
Activities in Aqueous Solutions of the Alkali Halide Salts from Molecular Simulation Maximilian Kohns, Michael Schappals, Martin Horsch, and Hans Hasse Laboratory of Engineering Thermodynamics, University
Supporting Information
 Supporting Information Structure and Dynamics of Uranyl(VI) and Plutonyl(VI) Cations in Ionic Liquid/Water Mixtures via Molecular Dynamics Simulations Katie A. Maerzke, George S. Goff, Wolfgang H. Runde,
Supporting Information Structure and Dynamics of Uranyl(VI) and Plutonyl(VI) Cations in Ionic Liquid/Water Mixtures via Molecular Dynamics Simulations Katie A. Maerzke, George S. Goff, Wolfgang H. Runde,
Water models in classical simulations
 Water models in classical simulations Maria Fyta Institut für Computerphysik, Universität Stuttgart Stuttgart, Germany Water transparent, odorless, tasteless and ubiquitous really simple: two H atoms attached
Water models in classical simulations Maria Fyta Institut für Computerphysik, Universität Stuttgart Stuttgart, Germany Water transparent, odorless, tasteless and ubiquitous really simple: two H atoms attached
CE 530 Molecular Simulation
 1 CE 530 Molecular Simulation Lecture 1 David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Time/s Multi-Scale Modeling Based on SDSC Blue Horizon (SP3) 1.728 Tflops
1 CE 530 Molecular Simulation Lecture 1 David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Time/s Multi-Scale Modeling Based on SDSC Blue Horizon (SP3) 1.728 Tflops
MOLECULAR DYNAMIC SIMULATION OF WATER VAPOR INTERACTION WITH VARIOUS TYPES OF PORES USING HYBRID COMPUTING STRUCTURES
 MOLECULAR DYNAMIC SIMULATION OF WATER VAPOR INTERACTION WITH VARIOUS TYPES OF PORES USING HYBRID COMPUTING STRUCTURES V.V. Korenkov 1,3, a, E.G. Nikonov 1, b, M. Popovičová 2, с 1 Joint Institute for Nuclear
MOLECULAR DYNAMIC SIMULATION OF WATER VAPOR INTERACTION WITH VARIOUS TYPES OF PORES USING HYBRID COMPUTING STRUCTURES V.V. Korenkov 1,3, a, E.G. Nikonov 1, b, M. Popovičová 2, с 1 Joint Institute for Nuclear
The micro-properties of [hmpy+] [Tf 2 N-] Ionic liquid: a simulation. study. 1. Introduction
![The micro-properties of [hmpy+] [Tf 2 N-] Ionic liquid: a simulation. study. 1. Introduction The micro-properties of [hmpy+] [Tf 2 N-] Ionic liquid: a simulation. study. 1. Introduction](/thumbs/72/66941683.jpg) ISBN 978-1-84626-081-0 Proceedings of the 2010 International Conference on Application of Mathematics and Physics Volume 1: Advances on Space Weather, Meteorology and Applied Physics Nanjing, P. R. China,
ISBN 978-1-84626-081-0 Proceedings of the 2010 International Conference on Application of Mathematics and Physics Volume 1: Advances on Space Weather, Meteorology and Applied Physics Nanjing, P. R. China,
INTERMOLECULAR AND SURFACE FORCES
 INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
INTERMOLECULAR AND SURFACE FORCES SECOND EDITION JACOB N. ISRAELACHVILI Department of Chemical & Nuclear Engineering and Materials Department University of California, Santa Barbara California, USA ACADEMIC
Unit 10: Part 1: Polarity and Intermolecular Forces
 Unit 10: Part 1: Polarity and Intermolecular Forces Name: Block: Intermolecular Forces of Attraction and Phase Changes Intramolecular Bonding: attractive forces that occur between atoms WITHIN a molecule;
Unit 10: Part 1: Polarity and Intermolecular Forces Name: Block: Intermolecular Forces of Attraction and Phase Changes Intramolecular Bonding: attractive forces that occur between atoms WITHIN a molecule;
Lower critical solution temperature (LCST) phase. behaviour of an ionic liquid and its control by. supramolecular host-guest interactions
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Lower critical solution temperature (LCST) phase behaviour of an ionic liquid and its control by
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Lower critical solution temperature (LCST) phase behaviour of an ionic liquid and its control by
Supporting information for: Anomalous Stability of Two-Dimensional Ice. Confined in Hydrophobic Nanopore
 Supporting information for: Anomalous Stability of Two-Dimensional Ice Confined in Hydrophobic Nanopore Boxiao Cao, Enshi Xu, and Tianshu Li Department of Civil and Environmental Engineering, George Washington
Supporting information for: Anomalous Stability of Two-Dimensional Ice Confined in Hydrophobic Nanopore Boxiao Cao, Enshi Xu, and Tianshu Li Department of Civil and Environmental Engineering, George Washington
Hyeyoung Shin a, Tod A. Pascal ab, William A. Goddard III abc*, and Hyungjun Kim a* Korea
 The Scaled Effective Solvent Method for Predicting the Equilibrium Ensemble of Structures with Analysis of Thermodynamic Properties of Amorphous Polyethylene Glycol-Water Mixtures Hyeyoung Shin a, Tod
The Scaled Effective Solvent Method for Predicting the Equilibrium Ensemble of Structures with Analysis of Thermodynamic Properties of Amorphous Polyethylene Glycol-Water Mixtures Hyeyoung Shin a, Tod
Molecular dynamics simulation of nanofluidics and nanomachining
 Molecular dynamics simulation of nanofluidics and nanomachining M. T. Horsch,1, 4 S. Stephan,1 S. Becker,1 M. Heier,1 M. P. Lautenschläger,1 F. Diewald,2 R. Müller,2 H. M. Urbassek,3 and H. Hasse1 1 Engineering
Molecular dynamics simulation of nanofluidics and nanomachining M. T. Horsch,1, 4 S. Stephan,1 S. Becker,1 M. Heier,1 M. P. Lautenschläger,1 F. Diewald,2 R. Müller,2 H. M. Urbassek,3 and H. Hasse1 1 Engineering
Section 6.2A Intermolecular Attractions
 Section 6.2A Intermolecular Attractions As we know, molecules are held together by covalent bonds, but there are also attractive forces BETWEEN individual molecules (rather than within). These are called
Section 6.2A Intermolecular Attractions As we know, molecules are held together by covalent bonds, but there are also attractive forces BETWEEN individual molecules (rather than within). These are called
UB association bias algorithm applied to the simulation of hydrogen fluoride
 Fluid Phase Equilibria 194 197 (2002) 249 256 UB association bias algorithm applied to the simulation of hydrogen fluoride Scott Wierzchowski, David A. Kofke Department of Chemical Engineering, University
Fluid Phase Equilibria 194 197 (2002) 249 256 UB association bias algorithm applied to the simulation of hydrogen fluoride Scott Wierzchowski, David A. Kofke Department of Chemical Engineering, University
VOCABULARY. Set #2. Set #1
 VOCABULARY Set #1 1. Absolute zero 2. Accepted value 3. Accuracy 4. Celsius scale 5. Conversion factor 6. Density 7. Dimensional analysis 8. Experimental value 9. Gram 10. International system of units
VOCABULARY Set #1 1. Absolute zero 2. Accepted value 3. Accuracy 4. Celsius scale 5. Conversion factor 6. Density 7. Dimensional analysis 8. Experimental value 9. Gram 10. International system of units
Molecular dynamics simulation of the liquid vapor interface: The Lennard-Jones fluid
 Molecular dynamics simulation of the liquid vapor interface: The Lennard-Jones fluid Matthias Mecke and Jochen Winkelmann a) Institut für Physikalische Chemie, Universität Halle-Wittenberg, Geusaer Str.,
Molecular dynamics simulation of the liquid vapor interface: The Lennard-Jones fluid Matthias Mecke and Jochen Winkelmann a) Institut für Physikalische Chemie, Universität Halle-Wittenberg, Geusaer Str.,
Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids
 Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids Cameron F. Abrams Department of Chemical and Biological Engineering Drexel University Philadelphia, PA USA 9 June
Systematic Coarse-Graining and Concurrent Multiresolution Simulation of Molecular Liquids Cameron F. Abrams Department of Chemical and Biological Engineering Drexel University Philadelphia, PA USA 9 June
Multiscale Materials Modeling
 Multiscale Materials Modeling Lecture 02 Capabilities of Classical Molecular Simulation These notes created by David Keffer, University of Tennessee, Knoxville, 2009. Outline Capabilities of Classical
Multiscale Materials Modeling Lecture 02 Capabilities of Classical Molecular Simulation These notes created by David Keffer, University of Tennessee, Knoxville, 2009. Outline Capabilities of Classical
Molecular simulation of adsorption from dilute solutions
 Vol. 52 No. 3/2005 685 689 on-line at: www.actabp.pl Molecular simulation of adsorption from dilute solutions Werner Billes Rupert Tscheliessnig and Johann Fischer Institut für Verfahrens- und Energietechnik
Vol. 52 No. 3/2005 685 689 on-line at: www.actabp.pl Molecular simulation of adsorption from dilute solutions Werner Billes Rupert Tscheliessnig and Johann Fischer Institut für Verfahrens- und Energietechnik
CE 530 Molecular Simulation
 CE 530 Molecular Simulation Lecture 20 Phase Equilibria David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Thermodynamic Phase Equilibria Certain thermodynamic states
CE 530 Molecular Simulation Lecture 20 Phase Equilibria David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Thermodynamic Phase Equilibria Certain thermodynamic states
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE
 MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
MOLECULAR DYNAMICS SIMULATION OF HETEROGENEOUS NUCLEATION OF LIQUID DROPLET ON SOLID SURFACE Tatsuto Kimura* and Shigeo Maruyama** *Department of Mechanical Engineering, The University of Tokyo 7-- Hongo,
hydrated Nafion-117 for fuel cell application
 A molecular dynamics simulation study of oxygen within hydrated Nafion-117 for fuel cell application Jeffrey P. Fuller, Giuseppe F. Brunello, Seung Soon Jang School of Materials Science and Engineering
A molecular dynamics simulation study of oxygen within hydrated Nafion-117 for fuel cell application Jeffrey P. Fuller, Giuseppe F. Brunello, Seung Soon Jang School of Materials Science and Engineering
Computer simulation methods (2) Dr. Vania Calandrini
 Computer simulation methods (2) Dr. Vania Calandrini in the previous lecture: time average versus ensemble average MC versus MD simulations equipartition theorem (=> computing T) virial theorem (=> computing
Computer simulation methods (2) Dr. Vania Calandrini in the previous lecture: time average versus ensemble average MC versus MD simulations equipartition theorem (=> computing T) virial theorem (=> computing
Information Technology
 Information Technology Computational Molecular Engineering als aufstrebende Technologie in der Verfahrenstechnik Computational Molecular Engineering as an emerging technology in process engineering Martin
Information Technology Computational Molecular Engineering als aufstrebende Technologie in der Verfahrenstechnik Computational Molecular Engineering as an emerging technology in process engineering Martin
Supporting Information for: Physics Behind the Water Transport through. Nanoporous Graphene and Boron Nitride
 Supporting Information for: Physics Behind the Water Transport through Nanoporous Graphene and Boron Nitride Ludovic Garnier, Anthony Szymczyk, Patrice Malfreyt, and Aziz Ghoufi, Institut de Physique de
Supporting Information for: Physics Behind the Water Transport through Nanoporous Graphene and Boron Nitride Ludovic Garnier, Anthony Szymczyk, Patrice Malfreyt, and Aziz Ghoufi, Institut de Physique de
LAMMPS Performance Benchmark on VSC-1 and VSC-2
 LAMMPS Performance Benchmark on VSC-1 and VSC-2 Daniel Tunega and Roland Šolc Institute of Soil Research, University of Natural Resources and Life Sciences VSC meeting, Neusiedl am See, February 27-28,
LAMMPS Performance Benchmark on VSC-1 and VSC-2 Daniel Tunega and Roland Šolc Institute of Soil Research, University of Natural Resources and Life Sciences VSC meeting, Neusiedl am See, February 27-28,
Scientific Computing II
 Scientific Computing II Molecular Dynamics Simulation Michael Bader SCCS Summer Term 2015 Molecular Dynamics Simulation, Summer Term 2015 1 Continuum Mechanics for Fluid Mechanics? Molecular Dynamics the
Scientific Computing II Molecular Dynamics Simulation Michael Bader SCCS Summer Term 2015 Molecular Dynamics Simulation, Summer Term 2015 1 Continuum Mechanics for Fluid Mechanics? Molecular Dynamics the
Density dependence of dielectric permittivity of water and estimation of the electric field for the breakdown inception
 Journal of Physics: Conference Series PAPER OPEN ACCESS Density dependence of dielectric permittivity of water and estimation of the electric field for the breakdown inception To cite this article: D I
Journal of Physics: Conference Series PAPER OPEN ACCESS Density dependence of dielectric permittivity of water and estimation of the electric field for the breakdown inception To cite this article: D I
The Molecular Dynamics Method
 The Molecular Dynamics Method Thermal motion of a lipid bilayer Water permeation through channels Selective sugar transport Potential Energy (hyper)surface What is Force? Energy U(x) F = d dx U(x) Conformation
The Molecular Dynamics Method Thermal motion of a lipid bilayer Water permeation through channels Selective sugar transport Potential Energy (hyper)surface What is Force? Energy U(x) F = d dx U(x) Conformation
Cartoon courtesy of NearingZero.net. Chemical Bonding and Molecular Structure
 Cartoon courtesy of NearingZero.net Chemical Bonding and Molecular Structure Big Ideas in Unit 6 How do atoms form chemical bonds? How does the type of a chemical bond influence a compounds physical and
Cartoon courtesy of NearingZero.net Chemical Bonding and Molecular Structure Big Ideas in Unit 6 How do atoms form chemical bonds? How does the type of a chemical bond influence a compounds physical and
Supplementary Information. Surface Microstructure Engenders Unusual Hydrophobicity in. Phyllosilicates
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supplementary Information Surface Microstructure Engenders Unusual Hydrophobicity in Phyllosilicates
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supplementary Information Surface Microstructure Engenders Unusual Hydrophobicity in Phyllosilicates
A MOLECULAR DYNAMICS STUDY OF POLYMER/GRAPHENE NANOCOMPOSITES
 A MOLECULAR DYNAMICS STUDY OF POLYMER/GRAPHENE NANOCOMPOSITES Anastassia N. Rissanou b,c*, Vagelis Harmandaris a,b,c* a Department of Applied Mathematics, University of Crete, GR-79, Heraklion, Crete,
A MOLECULAR DYNAMICS STUDY OF POLYMER/GRAPHENE NANOCOMPOSITES Anastassia N. Rissanou b,c*, Vagelis Harmandaris a,b,c* a Department of Applied Mathematics, University of Crete, GR-79, Heraklion, Crete,
Electrons and Molecular Forces
 Electrons and Molecular Forces Chemistry 30 Ms. Hayduk Electron Configuration Atomic Structure Atomic Number Number of protons in the nucleus Defines the element Used to organize the periodic table 1 Bohr
Electrons and Molecular Forces Chemistry 30 Ms. Hayduk Electron Configuration Atomic Structure Atomic Number Number of protons in the nucleus Defines the element Used to organize the periodic table 1 Bohr
 Title Theory of solutions in the energy r of the molecular flexibility Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2003), 9702 Issue Date 2003-11-08 URL http://hdl.handle.net/2433/50354
Title Theory of solutions in the energy r of the molecular flexibility Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2003), 9702 Issue Date 2003-11-08 URL http://hdl.handle.net/2433/50354
Supporting information. The role of halide ions in the anisotropic growth. of gold nanoparticles: a microscopic, atomistic.
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 26 Supporting information The role of halide ions in the anisotropic growth of gold
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 26 Supporting information The role of halide ions in the anisotropic growth of gold
Multiscale simulations of complex fluid rheology
 Multiscale simulations of complex fluid rheology Michael P. Howard, Athanassios Z. Panagiotopoulos Department of Chemical and Biological Engineering, Princeton University Arash Nikoubashman Institute of
Multiscale simulations of complex fluid rheology Michael P. Howard, Athanassios Z. Panagiotopoulos Department of Chemical and Biological Engineering, Princeton University Arash Nikoubashman Institute of
Molecular Driving Forces
 Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
Molecular Driving Forces Statistical Thermodynamics in Chemistry and Biology SUBGfittingen 7 At 216 513 073 / / Ken A. Dill Sarina Bromberg With the assistance of Dirk Stigter on the Electrostatics chapters
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
1 Introduction. Stefan Eckelsbach 1, Thorsten Windmann 1, Ekaterina Elts 1, and Jadran Vrabec 1
 Simulation of liquid-liquid equilibria with molecular models optimized to vapor-liquid equilibria and model development for Hydrazine and two of its derivatives Stefan Eckelsbach 1, Thorsten Windmann 1,
Simulation of liquid-liquid equilibria with molecular models optimized to vapor-liquid equilibria and model development for Hydrazine and two of its derivatives Stefan Eckelsbach 1, Thorsten Windmann 1,
MARTINI simulation details
 S1 Appendix MARTINI simulation details MARTINI simulation initialization and equilibration In this section, we describe the initialization of simulations from Main Text section Residue-based coarsegrained
S1 Appendix MARTINI simulation details MARTINI simulation initialization and equilibration In this section, we describe the initialization of simulations from Main Text section Residue-based coarsegrained
Molecular Dynamics Simulation of Methanol-Water Mixture
 Molecular Dynamics Simulation of Methanol-Water Mixture Palazzo Mancini, Mara Cantoni University of Urbino Carlo Bo Abstract In this study some properties of the methanol-water mixture such as diffusivity,
Molecular Dynamics Simulation of Methanol-Water Mixture Palazzo Mancini, Mara Cantoni University of Urbino Carlo Bo Abstract In this study some properties of the methanol-water mixture such as diffusivity,
CE 530 Molecular Simulation
 1 CE 530 Molecular Simulation Lecture 14 Molecular Models David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Review Monte Carlo ensemble averaging, no dynamics easy
1 CE 530 Molecular Simulation Lecture 14 Molecular Models David A. Kofke Department of Chemical Engineering SUNY Buffalo kofke@eng.buffalo.edu 2 Review Monte Carlo ensemble averaging, no dynamics easy
Ionic Liquids simulations : obtention of structural and transport properties from molecular dynamics. C. J. F. Solano, D. Beljonne, R.
 Ionic Liquids simulations : obtention of structural and transport properties from molecular dynamics C. J. F. Solano, D. Beljonne, R. Lazzaroni Ionic Liquids simulations : obtention of structural and transport
Ionic Liquids simulations : obtention of structural and transport properties from molecular dynamics C. J. F. Solano, D. Beljonne, R. Lazzaroni Ionic Liquids simulations : obtention of structural and transport
Unit Cell-Level Thickness Control of Single-Crystalline Zinc Oxide Nanosheets Enabled by Electrical Double Layer Confinement
 Unit Cell-Level Thickness Control of Single-Crystalline Zinc Oxide Nanosheets Enabled by Electrical Double Layer Confinement Xin Yin, Yeqi Shi, Yanbing Wei, Yongho Joo, Padma Gopalan, Izabela Szlufarska,
Unit Cell-Level Thickness Control of Single-Crystalline Zinc Oxide Nanosheets Enabled by Electrical Double Layer Confinement Xin Yin, Yeqi Shi, Yanbing Wei, Yongho Joo, Padma Gopalan, Izabela Szlufarska,
Theory of Interfacial Tension of Partially Miscible Liquids
 Theory of Interfacial Tension of Partially Miscible Liquids M.-E. BOUDH-HIR and G.A. MANSOORI * University of Illinois at Chicago (M/C 063) Chicago, Illinois USA 60607-7052 Abstract The aim of this work
Theory of Interfacial Tension of Partially Miscible Liquids M.-E. BOUDH-HIR and G.A. MANSOORI * University of Illinois at Chicago (M/C 063) Chicago, Illinois USA 60607-7052 Abstract The aim of this work
Nucleation rate (m -3 s -1 ) Radius of water nano droplet (Å) 1e+00 1e-64 1e-128 1e-192 1e-256
 Supplementary Figures Nucleation rate (m -3 s -1 ) 1e+00 1e-64 1e-128 1e-192 1e-256 Calculated R in bulk water Calculated R in droplet Modified CNT 20 30 40 50 60 70 Radius of water nano droplet (Å) Supplementary
Supplementary Figures Nucleation rate (m -3 s -1 ) 1e+00 1e-64 1e-128 1e-192 1e-256 Calculated R in bulk water Calculated R in droplet Modified CNT 20 30 40 50 60 70 Radius of water nano droplet (Å) Supplementary
Engineering Molecular Models: Efficient Parameterization Procedure and Cyclohexanol as Case Study
 Engineering Molecular Models: Efficient Parameterization Procedure and Cyclohexanol as Case Study Thorsten Merker, Jadran Vrabec, and Hans Hasse Laboratory of Engineering Thermodynamics, University of
Engineering Molecular Models: Efficient Parameterization Procedure and Cyclohexanol as Case Study Thorsten Merker, Jadran Vrabec, and Hans Hasse Laboratory of Engineering Thermodynamics, University of
The spontaneous curvature of the water-hydrophobe interface
 THE JOURNAL OF CHEMICAL PHYSICS 137, 13512 (212) The spontaneous curvature of the water-hydrophobe interface Felix Sedlmeier and Roland R. Netz a) Physik Department, Technische Universität München, 85748
THE JOURNAL OF CHEMICAL PHYSICS 137, 13512 (212) The spontaneous curvature of the water-hydrophobe interface Felix Sedlmeier and Roland R. Netz a) Physik Department, Technische Universität München, 85748
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai
 Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Surface chemistry. Liquid-gas, solid-gas and solid-liquid surfaces. Levente Novák István Bányai Surfaces and Interfaces Defining of interfacial region Types of interfaces: surface vs interface Surface
Dynamic force matching: Construction of dynamic coarse-grained models with realistic short time dynamics and accurate long time dynamics
 for resubmission Dynamic force matching: Construction of dynamic coarse-grained models with realistic short time dynamics and accurate long time dynamics Aram Davtyan, 1 Gregory A. Voth, 1 2, a) and Hans
for resubmission Dynamic force matching: Construction of dynamic coarse-grained models with realistic short time dynamics and accurate long time dynamics Aram Davtyan, 1 Gregory A. Voth, 1 2, a) and Hans
Effect of surfactant structure on interfacial properties
 EUROPHYSICS LETTERS 15 September 2003 Europhys. Lett., 63 (6), pp. 902 907 (2003) Effect of surfactant structure on interfacial properties L. Rekvig 1 ( ), M. Kranenburg 2, B. Hafskjold 1 and B. Smit 2
EUROPHYSICS LETTERS 15 September 2003 Europhys. Lett., 63 (6), pp. 902 907 (2003) Effect of surfactant structure on interfacial properties L. Rekvig 1 ( ), M. Kranenburg 2, B. Hafskjold 1 and B. Smit 2
Chapter 4. Glutamic Acid in Solution - Correlations
 Chapter 4 Glutamic Acid in Solution - Correlations 4. Introduction Glutamic acid crystallises from aqueous solution, therefore the study of these molecules in an aqueous environment is necessary to understand
Chapter 4 Glutamic Acid in Solution - Correlations 4. Introduction Glutamic acid crystallises from aqueous solution, therefore the study of these molecules in an aqueous environment is necessary to understand
MOLECULAR INTERACTIONS NOTES
 - 1 - MOLECULAR INTERACTIONS NOTES Summary of Fundamental Molecular Interactions Q1Q Ion-Ion U ( r) = 4 r πεε o µ Q cosθ U ( r) = 4πεε o r µ 1µ U ( r) 3 r µ 1 µ U ( r) 6 r µ 1 α U ( r) 6 r Ion-Dipole Dipole-Dipole
- 1 - MOLECULAR INTERACTIONS NOTES Summary of Fundamental Molecular Interactions Q1Q Ion-Ion U ( r) = 4 r πεε o µ Q cosθ U ( r) = 4πεε o r µ 1µ U ( r) 3 r µ 1 µ U ( r) 6 r µ 1 α U ( r) 6 r Ion-Dipole Dipole-Dipole
Research Statement. Shenggao Zhou. November 3, 2014
 Shenggao Zhou November 3, My research focuses on: () Scientific computing and numerical analysis (numerical PDEs, numerical optimization, computational fluid dynamics, and level-set method for interface
Shenggao Zhou November 3, My research focuses on: () Scientific computing and numerical analysis (numerical PDEs, numerical optimization, computational fluid dynamics, and level-set method for interface
Molecular dynamics simulation of Aquaporin-1. 4 nm
 Molecular dynamics simulation of Aquaporin-1 4 nm Molecular Dynamics Simulations Schrödinger equation i~@ t (r, R) =H (r, R) Born-Oppenheimer approximation H e e(r; R) =E e (R) e(r; R) Nucleic motion described
Molecular dynamics simulation of Aquaporin-1 4 nm Molecular Dynamics Simulations Schrödinger equation i~@ t (r, R) =H (r, R) Born-Oppenheimer approximation H e e(r; R) =E e (R) e(r; R) Nucleic motion described
Solutions and Non-Covalent Binding Forces
 Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
Chapter 3 Solutions and Non-Covalent Binding Forces 3.1 Solvent and solution properties Molecules stick together using the following forces: dipole-dipole, dipole-induced dipole, hydrogen bond, van der
