Magnetic Resonance Spectroscopy EPR and NMR
|
|
|
- Jessica Bishop
- 6 years ago
- Views:
Transcription
1 Magnetic Resonance Spectroscopy EPR and NMR A brief review of the relevant bits of quantum mechanics 1. Electrons have spin, - rotation of the charge about its axis generates a magnetic field at each electron. 2. Electrons in orbitals with two electrons are spin-paired, - one with m s +½ (, α), one with m s -½ (, β), - so that the spins and magnetic fields cancel ( ). 3. Most molecular bonds are formed by coalescence of atomic orbitals so as to satisfy the lower energy state arising from spin-pairing. Most molecules therefore have all orbitals occupied by magnetically silent electron pairs. 4. If an electron is added to or subtracted from such a molecule (by reduction or oxidation) an unpaired electron is introduced which is not spin coupled, and therefore acts as a magnet. Such molecules are said to be paramagnetic. 5. Some bonds have unpaired electrons, giving them an inherent paramagnetic property. 6. In EPR spectroscopy, this property is measured to provide specific information about the molecule and its environment. 7. Some nuclei also have magnetic properties, measured in NMR.
2 Electron Paramagnetic Resonance (EPR) spectroscopy. What can we use it for? Assay of concentration and concentration changes: redox state of paramagnetic species such as iron-sulfur clusters, semiquinones, flavin free-radicals, cytochromes, P450, photosynthetic reaction center components, amino acid radicals (Tyr, Trp), etc. Paramagnetic metals such as Mn 2+, vanadyl ions (Mg 2+ -analogue) at catalytic centers. Measurement of local polarity and local structural mobility using nitroxide spin probes. Measurement of distance through spin-coupling with remote paramagnetic centers. Measurement of structure through interaction with local nuclear spins. ENDOR and pulsed methods (ESEEM) provide both distances and angles to give details of structure in the reaction environment close to the paramagnetic center (NMR through EPR). Measurement of electron distribution on paramagnetic species. Measurement of interactions (H-bonding) with local solvent (H 2 O).
3 Nuclear Magnetic Resonance (NMR) spectroscopy 1. The atomic nucleus is made up of protons (+-ve charge) and neutrons (neutral). 2. Like electrons, protons and neutrons (or nucleons) are quantum mechanical entities, and their energetic properties can be described by operators, wave functions, and quantum numbers. 3. Both protons and neutrons have a spin ½, with spin quantum number, m I, which can have values of (up, +½, or α), or (down, -½, or β). 4. Total nuclear energy levels are lower if the constituent protons and neutrons are spin-paired, so that for most nuclei, the spins (and magnetic fields) cancel. Protons are spin-paired with protons, neutrons with neutrons. 5. Isotopes that have an odd number of either protons or neutrons have a net spin, according to the Table below: Number of protons Even Even Odd Odd Number of neutrons Even Odd Even Odd Nuclear spin, (I) 0 1 /2 or 3 /2 or 5 /2 1 /2 or 3 /2 or 5 /2 1 or 2 or 3
4 What can we use NMR spectroscopy for? Because of the wide range of nuclei that have spins, the applications of NMR are more widespread than those of EPR. Resolution can be enhanced by isotopic substitution for those nuclei that either are rare, or have no significant population, at natural abundance. Identification and characterization of reaction products in chemistry through characteristic chemical shifts in energy for particular nuclei. Proton exchange rates between -OH, -COOH, etc., and H 2 O Spin-spin coupling to identify bonding interactions. By extension of the above, - NMR can be used to obtain structures for macromolecules at atomic resolution in solution. Magnetic Resonance Imaging (MRI), - a major advance in medical imaging of internal tissues, - based on proton NMR.
5 Spin and magnetism Electrons and nuclei have magnetic properties only because they carry charges and move. The magnetic force generated by a moving charge is proportional to the current. In the case of a spinning entity, this translates into how fast it spins. Why do different isotopes have different nuclear magnetic properties? Since the nuclear charge is dependent on atomic number (the number of protons), and different isotopes have the same atomic number (otherwise they are different elements), they must all carry the same charge. Since there s no change in charge on adding neutrons, why is there a change in magnetic properties? If a nucleus has an even number of protons and an even number of neutrons, all the spins compensate, and the nucleus has no net spin. The nuclear magnetism arises from the spin introduced by an extra neutron, which gives a net angular momentum. The rate of spinning is dependent on angular momentum and therefore on mass. For any particular mass, a given increment in spin on adding a neutron will start the whole nucleus spinning at a rate dependent on the combined mass. The magnetic difference between isotopes therefore depends in a complicated way on the mass of the nucleus (the atomic weight), the number of charges it carries (the atomic number), and how many unpaired nucleons are present.
6 Some nuclear magnets of importance in biological studies As we shall see later, the gyromagnetic ratio (γ) determines the energy at which electromagnetic radiation will flip the spin of a nuclear magnet. A flip occurs when the energy matches (or is in resonance with) the energy of the transition. The energy needed (expressed in terms of frequency) depends on the applied magnetic field (the strength of the magnet), Tesla in this case.
7 NMR ~ T EPR 0.34 T RT=0.002 kcal/mol at 10 o K Energy of transition ν =E/h (E = hν) RT=0.593 kcal/mol at 298 o K ν -frequency Hz ~ ν - wavenumber cm -1 λ- wavelength nm ~ ν ν 1 = = λ c Energy (ν) Energy (ν) m I = ±½ 0 magnetic field strength (B 0 ) m s = ±½ 0 m I = -½ γ ν B 0 E = h = h 2π m I = +½ m s = -½ E = hν = m s = +½ magnetic field strength (B 0 ) gβ B 0
8 On the nature of electromagnetic waves, and the transitions that absorb or generate them Our knowledge of electromagnetic waves comes from their interactions with matter. We measure them by the transitions they generate in atoms, molecules, and other antennae, and they are generated by excitation of transitions that decay by emitting a photon. They are called electromagnetic, not because they are either electrical or magnetic entities, but because they generate or are generated by electrical transitions, which have a linked magnetic component. The energy of photons is expressed in terms of frequency, wavenumber or wavelength, all of which are related to energy through the Planck constant. All photons move at the speed of light (~1ft/ns), - this relates frequency to wavelength. The frequency is related to the timescale of the transition associated with absorption or generation of the photon. The transitions associated with EPR occur with timescales in the ns range; those for NMR with timescales in the µs range. These timescales are of importance in terms of saturation, and the use of pulsed techniques, as we will see later. Energy of transition ν =E/h (E = hν) ν -frequency Hz ν - wavenumber cm -1 λ- wavelength nm ~ ν ν 1 = = λ c
9 What is the difference between the electron and the nucleus as magnets? 1. The spin quantum numbers for the electron, proton and neutron, all have the same value of ½. This is an intrinsic angular momentum, - every electron, proton and neutron has this property. 2. Because of the relation between angular momentum and mass, a spin ½ body has a frequency of rotation determined by the mass. Because nuclear particles (nucleons) have masses 1,836 that of the electron, the nucleus spins much more slowly. 3. Because the magnetic field depends on the rate of rotation, electrons have magnetic fields ~2,000 that of protons, which have higher fields than more massive nuclei. Hence magnetic resonance occurs at much higher energies for EPR than NMR. 4. In addition to the angular momentum and magnetic properties arising from its spin, an electron in a molecular orbital has an orbital angular momentum, which also generates a magnetic field. The interaction between these two magnetic fields is called spin-orbit coupling. The antiparallel arrangement (bottom, right) has the lower energy, and is therefore favored. The electron magnetic field depends on the total angular momentum, J. In general, both electrons and nucleons spin-couple with other magnets in their environment.
10 Nuclear Magnetic Resonance m I = -½ In the absence of an external magnetic field, the spins of the nuclei are arranged more or less at random. When a magnetic field is applied, the spins align with the field, just like bar magnets. However, because the energy involved is so small, they can flip direction relatively easily. Energy (ν) m I = ±½ γ ν B 0 E = h = h 2π m I = +½ 0 magnetic field strength (B 0 ) The consequence is that the energy level of the nucleus splits in a magnetic field, as shown on the right. The population of spins in the m I +½ (α) state is (very slightly) higher than in the mi -½ (β) state, because the energy is lower.
11 When photons of the right energy are absorbed, the spin of the nucleus flips between the two states. If we look at the diagram, we can see that the energy gap between the two states is dependent on the strength of the applied magnetic field. As a consequence, photons are absorbed which have a particular energy at a particular field. This is what is meant by resonance. γ ν B 0 E = h = h 2π Here, γ is the gyromagnetic ratio for the nucleus, B 0 is the magnetic field strength, and h is Planck s constant. When we come to discuss pulsed NMR, we will need to refer to circular motion. Here the preferred frame of reference is that of the circular motion of precession. We define the precession or Larmor frequency: ω = 2πυ or by substitution in the above, ω = γb 0 0
12 Some consequences of the energy scale. The transitions leading to NMR absorption have energies in the radio frequency range, depending on nucleus (γ) and the strength of the magnetic field generated by the magnet. NMR machines are rated by the frequency at which the proton is in NMR resonance for the magnet they are built around, so we have 200 MHz, 500 MHz, 750 MHz and even 1 GHz NMR spectrometers. To achieve the higher fields, high electrical currents are needed, which can be achieved using superconducting coils, - these are generally called superconducting magnets (costing $M). Since ω = 2πν = γb 0, for a 200 MHz machine we need a magnet generating 4.7 Tesla; for a 500 MHz machine we need Tesla, etc. As we increase the energy gap (increase frequency), the small differences of energy for transitions of nuclear magnets in different environments are also increased, and our NMR spectrum will be better resolved. In addition, we also increase the population difference for the two spin states as we increase the energy ( E/RT is increased), as discussed in the next slide. These factors make a big difference in the amount of time needed to generate a data set, - for example in solution of the structure of a protein.
13 The energy levels of the two spin states of importance in EPR and NMR are relatively close. As we saw in previous slides, the E values for the transitions are much less than RT. As a consequence, the populations in the two states are almost equal (see next slide for details). This means that we have to worry about the phenomenon of stimulated emission. Photons are promiscuous, - they will take advantage of any transition that matches their energy. A down transition has the same energy gap as an up transition, so a photon can be adsorbed, and promote a transition, in either direction. The laws of energy conservation require that the energy absorbed must be given up, together with the energy of the transition. As a consequence, two photons are emitted, of equal energy to the one absorbed, to give one emitted net. If our two spins are in almost equal populations, all the spins will absorb one photon, and all the spins will emit one photon. We therefore do not see these changes in population.
14 As we have seen, the energy of electromagnetic waves is generally expressed in frequency, but energy scales are all related through Planck s constant and the speed of light, so we can express these energies in J/mol, ev, or any other energy units. For a particular temperature, any transition can be described in terms of an equilibrium between two states. For example, when we flip the energy level of a proton in a magnetic field, we have two states, or α, and or β, separated by E, calculated as above. Let us use N α and N β to represent the relative populations in the two states. We can represent the equilibration between these two states by N α N β. Then the ratio of these populations is given by the Boltzman distribution: N α / N β = e E / RT Remembering that the energies of the transitions associated with NMR (and those of EPR) are both very much less than RT, we can see that only a very small excess of states will be in the lower energy level. For the proton at 500 MHz, at ~25 o C, since E = hν, and Planck and Avagadro constants are J.s and mol -1 respectively, energies in the RF range (~500 MHz) are 0.2 J/mol. N α / N β = It is these one in ten thousand spins that are available to absorb a photon to provide an NMR signal.
15 Electron Paramagnetic Resonance The same general considerations apply to EPR. The photon energy for resonance depends on the applied field. The field aligns the spins, to give a splitting of the electron energy levels, with a population in the m s +½ state that is greater than that in the m s -½ state. Absorption of a photon at the resonance energy flips the spins. The net absorbance is due to transitions from m s +½ to m s -½ levels. No field External field applied Energy (ν) m s = ±½ 0 m s = -½ E = hν = m s = +½ magnetic field strength (B 0 ) gβ B 0 Absorption of microwaves changes the energy level so that the small fraction of spins in the lower energy state are flipped into the opposite orientation. Saturation occurs when flips in both directions occur with equal probability. The power needed to saturate depends on the relaxation time of the spin transition.
16 The general principles of EPR and NMR are essentially the same, but with the differences in energies already discussed. However, the nomenclature is different, mainly for historical reasons. The resonance condition for EPR is described by the equations: E E 0 = gβb m s = hν = E = 1 E 1 gβb 2 2 Here, g, the dimensionless Landé g-value, describes the resonance energy. Values around are found for simple free-radical systems. The term β is the Bohr magneton: + β = eh/(4π m e ) = x JT -1 in SI units, or β ν = β/h = e/(4π m e ) = x 10 9 Hz T -1 in frequency units, or β ev = β /(e/c) = x 10-5 ev T -1 in electron volt units, where (e/c) is the electron charge, x C. 0
17 Two different approaches to magnetic spectroscopy, CW and pulsed methods Continuous Wave (CW) The CW approach is that used in most EPR spectrometers. It was the method used in earlier NMR spectrometers, but has now been supplanted by Fourier Transform NMR (FT-NMR), which is a pulsed method that we will discuss later. The new generation of EPR spectrometers are also pulse-based instruments. In CW-EPR, the absorbance spectrum of the sample is measured over a range of energies appropriate for the magnetic field strength of the magnet used. However, for technical reasons, the spectrum is obtained by using a constant microwave frequency, and varying the magnetic field strength. The spectrum is usually presented as the first derivative of the absorption spectrum.
18 Comparison of an EPR spectrometer, and a UV-VIS spectrophotometer In this comparison, several points are noteworthy. 1. In a spectrophotometer, the spectrum is generated by varying the wavelength. As we just discussed, in a CW-EPR spectrometer, we use a fixed wavelength (frequency), but vary the magnetic field. This is achieved by the field scan and modulation coils associated with the magnet. 2. In the spectrophotometer shown, the light-beam is chopped by a rotating sector. This gives it a period function, and allows use of phase and frequency sensitive detection electronics, which improve the signal to noise ratio.
19 3. In the CW-EPR spectrometer, the signal is modulated at 100 khz by modulation of the magnetic field. This allows use of frequency and phase sensitive amplification to improve the S/N ratio. The intensity and modulation frequency are monitored to provide a reference channel. The output signal is an amplified difference. Because of the use of a modulated signal and frequency selective amplification, the detection electronics only respond to a change in absorbance in the sample. As a consequence, the spectrum is output as the first derivative of the absorbance spectrum.
20 Choice of EPR range Use of a fixed microwave frequency and a varying field in CW-EPR is determined by the difficulty of generating microwaves of sufficient intensity. The choice was originally dependent on the development of radar technology. Radar uses microwaves, with different frequencies for different purposes, and also needs a lot of power. Although alternative technologies are now available, the design of EPR spectrometers was based on the availability of klystron microwave sources from the radar applications, with the frequencies below. EPR spectroscopists talk about W- band, Q-band, S-band, etc., EPR spectrometers. Most work reported has been done at X-band, with microwave frequencies of ~9.5 GHz, and magnetic field strength ~3400 Gauss, or 0.34 Tesla.
21 electronics microwave bridge EPR tubes magnet computer for data analysis sample
Chem 325 NMR Intro. The Electromagnetic Spectrum. Physical properties, chemical properties, formulas Shedding real light on molecular structure:
 Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Physical properties, chemical properties, formulas Shedding real light on molecular structure: Wavelength Frequency ν Wavelength λ Frequency ν Velocity c = 2.998 10 8 m s -1 The Electromagnetic Spectrum
Magnetic Resonance Imaging (MRI)
 Magnetic Resonance Imaging Introduction The Components The Technology (MRI) Physics behind MR Most slides taken from http:// www.slideworld.org/ viewslides.aspx/magnetic- Resonance-Imaging- %28MRI%29-MR-Imaging-
Magnetic Resonance Imaging Introduction The Components The Technology (MRI) Physics behind MR Most slides taken from http:// www.slideworld.org/ viewslides.aspx/magnetic- Resonance-Imaging- %28MRI%29-MR-Imaging-
Nuclear magnetic resonance spectroscopy
 nuclear spin transitions O Nuclear magnetic resonance spectroscopy 1 H, 13 C, 2-dimensional which transitions? wavelength and intensity; ppm what happens if we change the environment of the nucleus? substituent
nuclear spin transitions O Nuclear magnetic resonance spectroscopy 1 H, 13 C, 2-dimensional which transitions? wavelength and intensity; ppm what happens if we change the environment of the nucleus? substituent
NMR Spectroscopy Laboratory Experiment Introduction. 2. Theory
 1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
1. Introduction 64-311 Laboratory Experiment 11 NMR Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy is a powerful and theoretically complex analytical tool. This experiment will introduce to
10.3 NMR Fundamentals
 10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
10.3 NMR Fundamentals nuclear spin calculations and examples NMR properties of selected nuclei the nuclear magnetic moment and precession around a magnetic field the spin quantum number and the NMR transition
Fundamental MRI Principles Module 2 N. Nuclear Magnetic Resonance. X-ray. MRI Hydrogen Protons. Page 1. Electrons
 Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Fundamental MRI Principles Module 2 N S 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons positively charged neutrons no significant charge electrons negatively charged Protons
Chapter 7. Nuclear Magnetic Resonance Spectroscopy
 Chapter 7 Nuclear Magnetic Resonance Spectroscopy I. Introduction 1924, W. Pauli proposed that certain atomic nuclei have spin and magnetic moment and exposure to magnetic field would lead to energy level
Chapter 7 Nuclear Magnetic Resonance Spectroscopy I. Introduction 1924, W. Pauli proposed that certain atomic nuclei have spin and magnetic moment and exposure to magnetic field would lead to energy level
DETECTION OF UNPAIRED ELECTRONS
 DETECTION OF UNPAIRED ELECTRONS There are experimental methods for the detection of unpaired electrons. One of the hallmarks of unpaired electrons in materials is interaction with a magnetic field. That
DETECTION OF UNPAIRED ELECTRONS There are experimental methods for the detection of unpaired electrons. One of the hallmarks of unpaired electrons in materials is interaction with a magnetic field. That
Biophysical Chemistry: NMR Spectroscopy
 Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Nuclear Magnetism Vrije Universiteit Brussel 21st October 2011 Outline 1 Overview and Context 2 3 Outline 1 Overview and Context 2 3 Context Proteins (and other biological macromolecules) Functional characterisation
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy
 Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Electron Spin Resonance, Basic principle of NMR, Application of NMR in the study of Biomolecules, NMR imaging and in vivo NMR spectromicroscopy Mitesh Shrestha Electron Spin Resonance Electron paramagnetic
Physical Background Of Nuclear Magnetic Resonance Spectroscopy
 Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
Physical Background Of Nuclear Magnetic Resonance Spectroscopy Michael McClellan Spring 2009 Department of Physics and Physical Oceanography University of North Carolina Wilmington What is Spectroscopy?
Fundamental MRI Principles Module Two
 Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
Fundamental MRI Principles Module Two 1 Nuclear Magnetic Resonance There are three main subatomic particles: protons neutrons electrons positively charged no significant charge negatively charged Protons
NUCLEAR MAGNETIC RESONANCE. The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei.
 14 Sep 11 NMR.1 NUCLEAR MAGNETIC RESONANCE The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei. Theory: In addition to its well-known properties of mass, charge,
14 Sep 11 NMR.1 NUCLEAR MAGNETIC RESONANCE The phenomenon of nuclear magnetic resonance will be used to study magnetic moments of nuclei. Theory: In addition to its well-known properties of mass, charge,
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r y even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
NMR Spectroscopy. for 1 st B.Tech INTRODUCTION Lecture -1 Indian Institute of Technology, Dhanbad
 NMR Spectroscopy for 1 st B.Tech Lecture -1 Indian Institute of Technology, Dhanbad by Dr. R P John & Dr. C. Halder INTRODUCTION Nucleus of any atom has protons and neutrons Both Proton and Neutron has
NMR Spectroscopy for 1 st B.Tech Lecture -1 Indian Institute of Technology, Dhanbad by Dr. R P John & Dr. C. Halder INTRODUCTION Nucleus of any atom has protons and neutrons Both Proton and Neutron has
Lecture 19: Building Atoms and Molecules
 Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r ψ even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 19: Building Atoms and Molecules +e r n = 3 n = 2 n = 1 +e +e r ψ even Lecture 19, p 1 Today Nuclear Magnetic Resonance Using RF photons to drive transitions between nuclear spin orientations in
Lecture 2 nmr Spectroscopy
 Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
Lecture 2 nmr Spectroscopy Pages 427 430 and Chapter 13 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure. Nuclear Magnetic Resonance (NMR)
 Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
Principles of Molecular Spectroscopy: Electromagnetic Radiation and Molecular structure Nuclear Magnetic Resonance (NMR) !E = h" Electromagnetic radiation is absorbed when the energy of photon corresponds
NMR, the vector model and the relaxation
 NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
NMR, the vector model and the relaxation Reading/Books: One and two dimensional NMR spectroscopy, VCH, Friebolin Spin Dynamics, Basics of NMR, Wiley, Levitt Molecular Quantum Mechanics, Oxford Univ. Press,
Chemistry 213 Practical Spectroscopy
 Chemistry 213 Practical Spectroscopy Dave Berg djberg@uvic.ca Elliott 314 A course in determining structure by spectroscopic methods Different types of spectroscopy afford different information about molecules
Chemistry 213 Practical Spectroscopy Dave Berg djberg@uvic.ca Elliott 314 A course in determining structure by spectroscopic methods Different types of spectroscopy afford different information about molecules
16.1 Introduction to NMR Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy. Spectroscopy 4/11/2013
 What is spectroscopy? NUCLEAR MAGNETIC RESONANCE (NMR) spectroscopy may be the most powerful method of gaining structural information about organic compounds. NMR involves an interaction between electromagnetic
What is spectroscopy? NUCLEAR MAGNETIC RESONANCE (NMR) spectroscopy may be the most powerful method of gaining structural information about organic compounds. NMR involves an interaction between electromagnetic
Magnetic Resonance Imaging
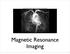 http://www.qldxray.com.au/filelibrary/mri_cardiovascular_system_ca_0005.jpg Magnetic Resonance Imaging 1 Overview 1. The magnetic properties of nuclei, and how they behave in strong magnetic fields. 2.
http://www.qldxray.com.au/filelibrary/mri_cardiovascular_system_ca_0005.jpg Magnetic Resonance Imaging 1 Overview 1. The magnetic properties of nuclei, and how they behave in strong magnetic fields. 2.
COPYRIGHTED MATERIAL. Production of Net Magnetization. Chapter 1
 Chapter 1 Production of Net Magnetization Magnetic resonance (MR) is a measurement technique used to examine atoms and molecules. It is based on the interaction between an applied magnetic field and a
Chapter 1 Production of Net Magnetization Magnetic resonance (MR) is a measurement technique used to examine atoms and molecules. It is based on the interaction between an applied magnetic field and a
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY
 ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
ELECTRON SPIN RESONANCE & MAGNETIC RESONANCE TOMOGRAPHY 1. AIM OF THE EXPERIMENT This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment
V27: RF Spectroscopy
 Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Martin-Luther-Universität Halle-Wittenberg FB Physik Advanced Lab Course V27: RF Spectroscopy ) Electron spin resonance (ESR) Investigate the resonance behaviour of two coupled LC circuits (an active rf
Nuclear Magnetic Resonance
 Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
Nuclear Magnetic Resonance PRINCIPLES OF NMR SPECTROSCOPY Contents Principles of nuclear magnetic resonance The nmr spectrometer Basic principles in nmr application NMR tools used to obtain information
The Basics of Magnetic Resonance Imaging
 The Basics of Magnetic Resonance Imaging Nathalie JUST, PhD nathalie.just@epfl.ch CIBM-AIT, EPFL Course 2013-2014-Chemistry 1 Course 2013-2014-Chemistry 2 MRI: Many different contrasts Proton density T1
The Basics of Magnetic Resonance Imaging Nathalie JUST, PhD nathalie.just@epfl.ch CIBM-AIT, EPFL Course 2013-2014-Chemistry 1 Course 2013-2014-Chemistry 2 MRI: Many different contrasts Proton density T1
Appendix II - 1. Figure 1: The splitting of the spin states of an unpaired electron
 Appendix II - 1 May 2017 Appendix II: Introduction to EPR Spectroscopy There are several general texts on this topic, and this appendix is only intended to give you a brief outline of the Electron Spin
Appendix II - 1 May 2017 Appendix II: Introduction to EPR Spectroscopy There are several general texts on this topic, and this appendix is only intended to give you a brief outline of the Electron Spin
Biochemistry 530 NMR Theory and Practice
 Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Biochemistry 530 NMR Theory and Practice Gabriele Varani Department of Biochemistry and Department of Chemistry University of Washington Lecturer: Gabriele Varani Biochemistry and Chemistry Room J479 and
Lecture 6: Physical Methods II. UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy
 Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Lecture 6: Physical Methods II UV Vis (electronic spectroscopy) Electron Spin Resonance Mossbauer Spectroscopy Physical Methods used in bioinorganic chemistry X ray crystallography X ray absorption (XAS)
Chancellor Phyllis Wise invites you to a birthday party!
 Chancellor Phyllis Wise invites you to a birthday party! 50 years ago, Illinois alumnus Nick Holonyak Jr. demonstrated the first visible light-emitting diode (LED) while working at GE. Holonyak returned
Chancellor Phyllis Wise invites you to a birthday party! 50 years ago, Illinois alumnus Nick Holonyak Jr. demonstrated the first visible light-emitting diode (LED) while working at GE. Holonyak returned
Química Orgânica I. Nuclear Magnetic Resonance Spectroscopy (I) Ciências Farmacêuticas Bioquímica Química AFB QO I 2007/08 1 AFB QO I 2007/08 2
 Química Orgânica I Ciências Farmacêuticas Bioquímica Química AFB QO I 2007/08 1 Nuclear Magnetic Resonance Spectroscopy (I) AFB QO I 2007/08 2 1 Adaptado de: Organic Chemistry, 6th Edition; L. G. Wade,
Química Orgânica I Ciências Farmacêuticas Bioquímica Química AFB QO I 2007/08 1 Nuclear Magnetic Resonance Spectroscopy (I) AFB QO I 2007/08 2 1 Adaptado de: Organic Chemistry, 6th Edition; L. G. Wade,
7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text , , 12.10)
 2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
2009, Department of Chemistry, The University of Western Ontario 7a.1 7a. Structure Elucidation: IR and 13 C-NMR Spectroscopies (text 11.1 11.5, 12.1 12.5, 12.10) A. Electromagnetic Radiation Energy is
Chapter 13 Spectroscopy
 hapter 13 Spectroscopy Infrared spectroscopy Ultraviolet-Visible spectroscopy Nuclear magnetic resonance spectroscopy Mass Spectrometry 13.1 Principles of Molecular Spectroscopy: Electromagnetic Radiation
hapter 13 Spectroscopy Infrared spectroscopy Ultraviolet-Visible spectroscopy Nuclear magnetic resonance spectroscopy Mass Spectrometry 13.1 Principles of Molecular Spectroscopy: Electromagnetic Radiation
ELECTRON PARAMAGNETIC RESONANCE
 ELECTRON PARAMAGNETIC RESONANCE = MAGNETIC RESONANCE TECHNIQUE FOR STUDYING PARAMAGNETIC SYSTEMS i.e. SYSTEMS WITH AT LEAST ONE UNPAIRED ELECTRON Examples of paramagnetic systems Transition-metal complexes
ELECTRON PARAMAGNETIC RESONANCE = MAGNETIC RESONANCE TECHNIQUE FOR STUDYING PARAMAGNETIC SYSTEMS i.e. SYSTEMS WITH AT LEAST ONE UNPAIRED ELECTRON Examples of paramagnetic systems Transition-metal complexes
Biomedical Imaging Magnetic Resonance Imaging
 Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
Biomedical Imaging Magnetic Resonance Imaging Charles A. DiMarzio & Eric Kercher EECE 4649 Northeastern University May 2018 Background and History Measurement of Nuclear Spins Widely used in physics/chemistry
10.4 Continuous Wave NMR Instrumentation
 10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
10.4 Continuous Wave NMR Instrumentation coherent detection bulk magnetization the rotating frame, and effective magnetic field generating a rotating frame, and precession in the laboratory frame spin-lattice
Lecture 02 Nuclear Magnetic Resonance Spectroscopy Principle and Application in Structure Elucidation
 Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
Application of Spectroscopic Methods in Molecular Structure Determination Prof. S. Sankararaman Department of Chemistry Indian Institution of Technology Madras Lecture 02 Nuclear Magnetic Resonance Spectroscopy
Atomic Quantum number summary. From last time. Na Optical spectrum. Another possibility: Stimulated emission. How do atomic transitions occur?
 From last time Hydrogen atom Multi-electron atoms This week s honors lecture: Prof. Brad Christian, Positron Emission Tomography Course evaluations next week Tues. Prof Montaruli Thurs. Prof. Rzchowski
From last time Hydrogen atom Multi-electron atoms This week s honors lecture: Prof. Brad Christian, Positron Emission Tomography Course evaluations next week Tues. Prof Montaruli Thurs. Prof. Rzchowski
Chemistry 431. Lecture 23
 Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
Chemistry 431 Lecture 23 Introduction The Larmor Frequency The Bloch Equations Measuring T 1 : Inversion Recovery Measuring T 2 : the Spin Echo NC State University NMR spectroscopy The Nuclear Magnetic
Ferdowsi University of Mashhad
 Spectroscopy in Inorganic Chemistry Nuclear Magnetic Resonance Spectroscopy spin deuterium 2 helium 3 The neutron has 2 quarks with a -e/3 charge and one quark with a +2e/3 charge resulting in a total
Spectroscopy in Inorganic Chemistry Nuclear Magnetic Resonance Spectroscopy spin deuterium 2 helium 3 The neutron has 2 quarks with a -e/3 charge and one quark with a +2e/3 charge resulting in a total
Electron spin resonance
 Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
Quick reference guide Introduction This is a model experiment for electron spin resonance, for clear demonstration of interaction between the magnetic moment of the electron spin with a superimposed direct
MRI Physics I: Spins, Excitation, Relaxation
 MRI Physics I: Spins, Excitation, Relaxation Douglas C. Noll Biomedical Engineering University of Michigan Michigan Functional MRI Laboratory Outline Introduction to Nuclear Magnetic Resonance Imaging
MRI Physics I: Spins, Excitation, Relaxation Douglas C. Noll Biomedical Engineering University of Michigan Michigan Functional MRI Laboratory Outline Introduction to Nuclear Magnetic Resonance Imaging
Chapter 13: Molecular Spectroscopy
 Chapter 13: Molecular Spectroscopy Electromagnetic Radiation E = hν h = Planck s Constant (6.63 x 10-34 J. s) ν = frequency (s -1 ) c = νλ λ = wavelength (nm) Energy is proportional to frequency Spectrum
Chapter 13: Molecular Spectroscopy Electromagnetic Radiation E = hν h = Planck s Constant (6.63 x 10-34 J. s) ν = frequency (s -1 ) c = νλ λ = wavelength (nm) Energy is proportional to frequency Spectrum
MOLECULAR SPECTROSCOPY AND PHOTOCHEMISTRY
 20 CHAPTER MOLECULAR SPECTROSCOPY AND PHOTOCHEMISTRY 20.1 Introduction to Molecular Spectroscopy 20.2 Experimental Methods in Molecular Spectroscopy 20.3 Rotational and Vibrational Spectroscopy 20.4 Nuclear
20 CHAPTER MOLECULAR SPECTROSCOPY AND PHOTOCHEMISTRY 20.1 Introduction to Molecular Spectroscopy 20.2 Experimental Methods in Molecular Spectroscopy 20.3 Rotational and Vibrational Spectroscopy 20.4 Nuclear
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction:
 Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Nuclear Magnetic Resonance (NMR) Spectroscopy Introduction: Nuclear magnetic resonance spectroscopy (NMR) is the most powerful tool available for organic structure determination. Like IR spectroscopy,
Topics. The concept of spin Precession of magnetic spin Relaxation Bloch Equation. Bioengineering 280A Principles of Biomedical Imaging
 Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
Bioengineering 280A Principles of Biomedical Imaging Fall Quarter 2006 MRI Lecture 1 Topics The concept of spin Precession of magnetic spin Relaxation Bloch Equation 1 Spin Intrinsic angular momentum of
Lecture 3 NMR Spectroscopy. January 26, 2016 Chemistry 328N
 Lecture 3 NMR Spectroscopy January 26, 2016 Please see me after class Enrollment issue Hugo Nicolas Eichner Nurbol Kaliyev E = hn Energy per photon Frequency Wave Length Electromagnetic Radiation Important
Lecture 3 NMR Spectroscopy January 26, 2016 Please see me after class Enrollment issue Hugo Nicolas Eichner Nurbol Kaliyev E = hn Energy per photon Frequency Wave Length Electromagnetic Radiation Important
PART 2 Electronic Structure and the Periodic Table. Reference: Chapter 7 8 in textbook
 PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Experiment to Discover Atom Structure -particle: He 2+ mass number = 4 Nucleus and Electron Model 2 Atomic Structure
PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Experiment to Discover Atom Structure -particle: He 2+ mass number = 4 Nucleus and Electron Model 2 Atomic Structure
With that first concept in mind, it is seen that a spinning nucleus creates a magnetic field, like a bar magnet
 NMR SPECTROSCOPY This section will discuss the basics of NMR (nuclear magnetic resonance) spectroscopy. Most of the section will discuss mainly 1H or proton spectroscopy but the most popular nuclei in
NMR SPECTROSCOPY This section will discuss the basics of NMR (nuclear magnetic resonance) spectroscopy. Most of the section will discuss mainly 1H or proton spectroscopy but the most popular nuclei in
4/4/11. Particles possess intrinsic angular momentum. Spin angular momentum is quantized (it can only take on discrete values)
 For the completely filled shells, subshell (4d 10 ) the orbital magnetic momentum is zero; for the 5s orbital M L is also zero. Hypothesis: the argent atom possesses no magnetic momentum >> they move in
For the completely filled shells, subshell (4d 10 ) the orbital magnetic momentum is zero; for the 5s orbital M L is also zero. Hypothesis: the argent atom possesses no magnetic momentum >> they move in
MRI Homework. i. (0.5 pt each) Consider the following arrangements of bar magnets in a strong magnetic field.
 MRI Homework 1. While x-rays are used to image bones, magnetic resonance imaging (MRI) is used to examine tissues within the body by detecting where hydrogen atoms (H atoms) are and their environment (e.g.
MRI Homework 1. While x-rays are used to image bones, magnetic resonance imaging (MRI) is used to examine tissues within the body by detecting where hydrogen atoms (H atoms) are and their environment (e.g.
Ala-Arg-Pro-Tyr-Asn-Phe-Cpa-Leu-NH 2
 Applied Spectroscop Ala-Arg-Pro-Tr-Asn-Phe-Cpa-Leu-NH 2 Cpa Ala Pro Guillermo Mona What is Spectroscop? Without going into latin or greek, spectroscop is the stud of the interactions between light and
Applied Spectroscop Ala-Arg-Pro-Tr-Asn-Phe-Cpa-Leu-NH 2 Cpa Ala Pro Guillermo Mona What is Spectroscop? Without going into latin or greek, spectroscop is the stud of the interactions between light and
Magnetic Resonance Spectroscopy ( )
 Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
Magnetic Resonance Spectroscopy In our discussion of spectroscopy, we have shown that absorption of E.M. radiation occurs on resonance: When the frequency of applied E.M. field matches the energy splitting
January 29, 2019 Chemistry 328N
 Lecture 3 NMR Spectroscopy January 29, 2019 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances and the correlation
Lecture 3 NMR Spectroscopy January 29, 2019 Molecular Spectroscopy Molecular spectroscopy: the study of the frequencies of electromagnetic radiation that are absorbed or emitted by substances and the correlation
Chapter 16 Nuclear Magnetic Resonance Spectroscopy
 hapter 16 Nuclear Magnetic Resonance Spectroscopy The Spinning Proton A spinning proton generates a magnetic field, resembling that of a small bar magnet. An odd number of protons in the nucleus creates
hapter 16 Nuclear Magnetic Resonance Spectroscopy The Spinning Proton A spinning proton generates a magnetic field, resembling that of a small bar magnet. An odd number of protons in the nucleus creates
10/4/2011. Tells you the number of protons
 Atomic Structure The arrangement of the subatomic particles within the atom determines the chemical properties of the elements How they interact with one another The types of ions and structures that they
Atomic Structure The arrangement of the subatomic particles within the atom determines the chemical properties of the elements How they interact with one another The types of ions and structures that they
NMR BMB 173 Lecture 16, February
 NMR The Structural Biology Continuum Today s lecture: NMR Lots of slides adapted from Levitt, Spin Dynamics; Creighton, Proteins; And Andy Rawlinson There are three types of particles in the universe Quarks
NMR The Structural Biology Continuum Today s lecture: NMR Lots of slides adapted from Levitt, Spin Dynamics; Creighton, Proteins; And Andy Rawlinson There are three types of particles in the universe Quarks
A Hands on Introduction to NMR Lecture #1 Nuclear Spin and Magnetic Resonance
 A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
A Hands on Introduction to NMR 22.920 Lecture #1 Nuclear Spin and Magnetic Resonance Introduction - The aim of this short course is to present a physical picture of the basic principles of Nuclear Magnetic
Measuring Spin-Lattice Relaxation Time
 WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
WJP, PHY381 (2009) Wabash Journal of Physics v4.0, p.1 Measuring Spin-Lattice Relaxation Time L.W. Lupinski, R. Paudel, and M.J. Madsen Department of Physics, Wabash College, Crawfordsville, IN 47933 (Dated:
Chapter 13 Nuclear Magnetic Resonance Spectroscopy
 Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 13 Nuclear Magnetic Resonance Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice Hall
Organic Chemistry, 6 th Edition L. G. Wade, Jr. Chapter 13 Nuclear Magnetic Resonance Spectroscopy Jo Blackburn Richland College, Dallas, TX Dallas County Community College District 2006, Prentice Hall
Nuclear Magnetic Resonance Imaging
 Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
Nuclear Magnetic Resonance Imaging Simon Lacoste-Julien Electromagnetic Theory Project 198-562B Department of Physics McGill University April 21 2003 Abstract This paper gives an elementary introduction
Nuclear Magnetic Resonance (NMR)
 Nuclear Magnetic Resonance (NMR) Nuclear Magnetic Resonance (NMR) The Nuclear Magnetic Resonance Spectroscopy (NMR) is one of the most important spectroscopic methods to explore the structure and dynamic
Nuclear Magnetic Resonance (NMR) Nuclear Magnetic Resonance (NMR) The Nuclear Magnetic Resonance Spectroscopy (NMR) is one of the most important spectroscopic methods to explore the structure and dynamic
NMR = Nuclear Magnetic Resonance
 NMR = Nuclear Magnetic Resonance NMR spectroscopy is the most powerful technique available to organic chemists for determining molecular structures. Looks at nuclei with odd mass numbers or odd number
NMR = Nuclear Magnetic Resonance NMR spectroscopy is the most powerful technique available to organic chemists for determining molecular structures. Looks at nuclei with odd mass numbers or odd number
An Introduction to Hyperfine Structure and Its G-factor
 An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
An Introduction to Hyperfine Structure and Its G-factor Xiqiao Wang East Tennessee State University April 25, 2012 1 1. Introduction In a book chapter entitled Model Calculations of Radiation Induced Damage
Last Updated:
 Last Updated: 2014 07 30 Generation of the EPR ignal MR and EPR are similar in a way that the amount of absorption energy required to for a transition between atomic or molecular states. pectroscopy generally
Last Updated: 2014 07 30 Generation of the EPR ignal MR and EPR are similar in a way that the amount of absorption energy required to for a transition between atomic or molecular states. pectroscopy generally
13/02/2017. Overview. Magnetism. Electron paramagnetic resonance (EPR) Electron Paramagnetic Resonance and Dynamic Nuclear Polarisation CH916
 Electron Paramagnetic Resonance and Dynamic Nuclear Polarisation CH916 Overview What it is Why it s useful Gavin W Morley, Department of Physics, University of Warwick Dynamic nuclear polarization Why
Electron Paramagnetic Resonance and Dynamic Nuclear Polarisation CH916 Overview What it is Why it s useful Gavin W Morley, Department of Physics, University of Warwick Dynamic nuclear polarization Why
Magnetic Resonance Imaging. Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics
 Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
Magnetic Resonance Imaging Pål Erik Goa Associate Professor in Medical Imaging Dept. of Physics pal.e.goa@ntnu.no 1 Why MRI? X-ray/CT: Great for bone structures and high spatial resolution Not so great
VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY
 1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
1 VIII. NUCLEAR MAGNETIC RESONANCE (NMR) SPECTROSCOPY Molecules are extremely small entities; thus, their direct detection and direct investigation is still almost impossible. For the detection and detailed
PART 2 Electronic Structure and the Periodic Table. Reference: Chapter 7 8 in textbook
 PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Early Atomic Models 2 Thomson s 1904 Model of the Atom Plumb Pudding Model He discovered the electron, a discovery
PART 2 Electronic Structure and the Periodic Table Reference: Chapter 7 8 in textbook 1 Early Atomic Models 2 Thomson s 1904 Model of the Atom Plumb Pudding Model He discovered the electron, a discovery
Saturation Absorption Spectroscopy of Rubidium Atom
 Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Saturation Absorption Spectroscopy of Rubidium Atom Jayash Panigrahi August 17, 2013 Abstract Saturated absorption spectroscopy has various application in laser cooling which have many relevant uses in
Introduction to NMR! Ravinder Reddy!
 Introduction to NMR! Ravinder Reddy! Brief History of NMR! First detection of NMR! MSNMR! FT NMR! 2D NMR! 2D-NMR and protein structure! Development of MRI! Outline! Concept of SPIN! Spin angular momentum!
Introduction to NMR! Ravinder Reddy! Brief History of NMR! First detection of NMR! MSNMR! FT NMR! 2D NMR! 2D-NMR and protein structure! Development of MRI! Outline! Concept of SPIN! Spin angular momentum!
Spectral Broadening Mechanisms
 Spectral Broadening Mechanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Spectral Broadening Mechanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Nuclear Spin and Stability. PHY 3101 D. Acosta
 Nuclear Spin and Stability PHY 3101 D. Acosta Nuclear Spin neutrons and protons have s = ½ (m s = ± ½) so they are fermions and obey the Pauli- Exclusion Principle The nuclear magneton is eh m µ e eh 1
Nuclear Spin and Stability PHY 3101 D. Acosta Nuclear Spin neutrons and protons have s = ½ (m s = ± ½) so they are fermions and obey the Pauli- Exclusion Principle The nuclear magneton is eh m µ e eh 1
Basic p rinciples COPYRIGHTED MATERIAL. Introduction. Atomic s tructure
 1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
1 Basic p rinciples Introduction 1 Atomic structure 1 Motion in the atom 2 MR active nuclei 2 The hydrogen nucleus 4 Alignment 4 Precession 8 The Larmor equation 9 Introduction The basic principles of
Geology 228/378 Applied & Environmental Geophysics Lecture 8. Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR)
 Geology 228/378 Applied & Environmental Geophysics Lecture 8 Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) 1. Time domain
Geology 228/378 Applied & Environmental Geophysics Lecture 8 Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) Induced Polarization (IP) and Nuclear Magnetic Resonance (NMR) 1. Time domain
Electronic Spectra of Complexes
 Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Electronic Spectra of Complexes Interpret electronic spectra of coordination compounds Correlate with bonding Orbital filling and electronic transitions Electron-electron repulsion Application of MO theory
Chemistry (
 Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Nuclear Magnetic Resonance (NMR)
 Nuclear Magnetic Resonance (NMR) E E increases with increasing magnetic field strength Boltzmann distribution at thermal equilibrium: N (m=-1/2) /N (m=+1/2) = e ( E/kT) with E = γ(h/2π)b o NMR Physical
Nuclear Magnetic Resonance (NMR) E E increases with increasing magnetic field strength Boltzmann distribution at thermal equilibrium: N (m=-1/2) /N (m=+1/2) = e ( E/kT) with E = γ(h/2π)b o NMR Physical
Introduction. Introduction. Introduction. Chem Experiment 4 NMR & Mass Spectroscopy and Biomolecular Structure. Fall, 2011
 hem 43 - Experiment 4 MR & Mass pectroscopy and Biomolecular tructure Fall, 2 What does MR measure? Introduction What information does MR provide us about the structures of biological macromolecules -
hem 43 - Experiment 4 MR & Mass pectroscopy and Biomolecular tructure Fall, 2 What does MR measure? Introduction What information does MR provide us about the structures of biological macromolecules -
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY
 ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
ATOMIC STRUCTURE, ELECTRONS, AND PERIODICITY All matter is made of atoms. There are a limited number of types of atoms; these are the elements. (EU 1.A) Development of Atomic Theory Atoms are so small
Introduction to MRI. Spin & Magnetic Moments. Relaxation (T1, T2) Spin Echoes. 2DFT Imaging. K-space & Spatial Resolution.
 Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
Introduction to MRI Spin & Magnetic Moments Relaxation (T1, T2) Spin Echoes 2DFT Imaging Selective excitation, phase & frequency encoding K-space & Spatial Resolution Contrast (T1, T2) Acknowledgement:
Overhauser Magnetometers For Measurement of the Earth s Magnetic Field
 Overhauser Magnetometers For Measurement of the Earth s Magnetic Field By: Dr. Ivan Hrvoic GEM Systems Inc. (Magnetic field Workshop on Magnetic Observatory Instrumentation Espoo, Finland. 1989) TABLE
Overhauser Magnetometers For Measurement of the Earth s Magnetic Field By: Dr. Ivan Hrvoic GEM Systems Inc. (Magnetic field Workshop on Magnetic Observatory Instrumentation Espoo, Finland. 1989) TABLE
NMR Spectroscopy. Guangjin Hou
 NMR Spectroscopy Guangjin Hou 22-04-2009 NMR History 1 H NMR spectra of water H NMR spectra of water (First NMR Spectra on Water, 1946) 1 H NMR spectra ethanol (First bservation of the Chemical Shift,
NMR Spectroscopy Guangjin Hou 22-04-2009 NMR History 1 H NMR spectra of water H NMR spectra of water (First NMR Spectra on Water, 1946) 1 H NMR spectra ethanol (First bservation of the Chemical Shift,
Principles of MRI. Vinyl Record. Last time: Today: Homework Due tonight! EE225E / BIO265. Transforms a temporal signal to a spatial signal
 What is this? ` Principles of MRI Lecture 05 EE225E / BIO265 Instructor: Miki Lustig UC Berkeley, EECS The first NMR spectrum of ethanol 1951. 1 2 Today Last time: Linear systems, Fourier Transforms, Sampling
What is this? ` Principles of MRI Lecture 05 EE225E / BIO265 Instructor: Miki Lustig UC Berkeley, EECS The first NMR spectrum of ethanol 1951. 1 2 Today Last time: Linear systems, Fourier Transforms, Sampling
Skoog Chapter 6 Introduction to Spectrometric Methods
 Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
Skoog Chapter 6 Introduction to Spectrometric Methods General Properties of Electromagnetic Radiation (EM) Wave Properties of EM Quantum Mechanical Properties of EM Quantitative Aspects of Spectrochemical
I. Multiple Choice Questions (Type-I)
 I. Multiple Choice Questions (Type-I) 1. Which of the following conclusions could not be derived from Rutherford s α -particle scattering experiement? (i) Most of the space in the atom is empty. (ii) The
I. Multiple Choice Questions (Type-I) 1. Which of the following conclusions could not be derived from Rutherford s α -particle scattering experiement? (i) Most of the space in the atom is empty. (ii) The
Spectral Broadening Mechanisms. Broadening mechanisms. Lineshape functions. Spectral lifetime broadening
 Spectral Broadening echanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
Spectral Broadening echanisms Lorentzian broadening (Homogeneous) Gaussian broadening (Inhomogeneous, Inertial) Doppler broadening (special case for gas phase) The Fourier Transform NC State University
The NMR Inverse Imaging Problem
 The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
The NMR Inverse Imaging Problem Nuclear Magnetic Resonance Protons and Neutrons have intrinsic angular momentum Atoms with an odd number of proton and/or odd number of neutrons have a net magnetic moment=>
Electron Paramagnetic Resonance. Ferro Magnetic Resonance. Proposed topics. -1- EPR fundamentals for an isotropic S=1/2 system,
 What is EPR? pg Electron Paramagnetic Resonance a.k.a. ESR a.k.a. FMR Electron Spin Resonance Ferro Magnetic Resonance Proposed topics pg 2-1- EPR fundamentals for an isotropic S=1/2 system, -2- anisotropy
What is EPR? pg Electron Paramagnetic Resonance a.k.a. ESR a.k.a. FMR Electron Spin Resonance Ferro Magnetic Resonance Proposed topics pg 2-1- EPR fundamentals for an isotropic S=1/2 system, -2- anisotropy
e 2m e c I, (7.1) = g e β B I(I +1), (7.2) = erg/gauss. (7.3)
 Chemistry 126 Molecular Spectra & Molecular Structure Week # 7 Electron Spin Resonance Spectroscopy, Supplement Like the hydrogen nucleus, an unpaired electron in a sample has a spin of I=1/2. The magnetic
Chemistry 126 Molecular Spectra & Molecular Structure Week # 7 Electron Spin Resonance Spectroscopy, Supplement Like the hydrogen nucleus, an unpaired electron in a sample has a spin of I=1/2. The magnetic
2. What is the wavelength, in nm, of light with an energy content of 550 kj/mol? a nm b nm c. 157 nm d. 217 nm e.
 1. What is the frequency associated with radiation of 4.59 x 10-8 cm wavelength? a. 6.54 x 10 17 s -1 b. 6.54 x 10 15 s -1 c. 1.53 x 10-8 s -1 d. 13.8 s -1 e. 2.18 x 10 7 s -1 1 2. What is the wavelength,
1. What is the frequency associated with radiation of 4.59 x 10-8 cm wavelength? a. 6.54 x 10 17 s -1 b. 6.54 x 10 15 s -1 c. 1.53 x 10-8 s -1 d. 13.8 s -1 e. 2.18 x 10 7 s -1 1 2. What is the wavelength,
Physics and Brain Imaging
 Physics and Brain Imaging Nuclear Magnetic Resonance (NMR) Magnetic Resonance Imaging (MRI) Functional MRI (fmri) Talk at Quarknet FSU Summer Workshop, July 24, 2017 Per Arne Rikvold Leonardo da Vinci
Physics and Brain Imaging Nuclear Magnetic Resonance (NMR) Magnetic Resonance Imaging (MRI) Functional MRI (fmri) Talk at Quarknet FSU Summer Workshop, July 24, 2017 Per Arne Rikvold Leonardo da Vinci
Physical fundamentals of magnetic resonance imaging
 Physical fundamentals of magnetic resonance imaging Stepan Sereda University of Bonn 1 / 26 Why? Figure 1 : Full body MRI scan (Source: [4]) 2 / 26 Overview Spin angular momentum Rotating frame and interaction
Physical fundamentals of magnetic resonance imaging Stepan Sereda University of Bonn 1 / 26 Why? Figure 1 : Full body MRI scan (Source: [4]) 2 / 26 Overview Spin angular momentum Rotating frame and interaction
Introduction of Key Concepts of Nuclear Magnetic Resonance
 I have not yet lost that sense of wonder, and delight, that this delicate motion should reside in all ordinary things around us, revealing itself only to those who looks for it. E. M. Purcell, Nobel Lecture.
I have not yet lost that sense of wonder, and delight, that this delicate motion should reside in all ordinary things around us, revealing itself only to those who looks for it. E. M. Purcell, Nobel Lecture.
Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift. Dr. Sapna Gupta
 Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift Dr. Sapna Gupta Introduction NMR is the most powerful tool available for organic structure determination.
Nuclear Magnetic Resonance H-NMR Part 1 Introduction to NMR, Instrumentation, Sample Prep, Chemical Shift Dr. Sapna Gupta Introduction NMR is the most powerful tool available for organic structure determination.
On Electron Paramagnetic Resonance in DPPH
 On Electron Paramagnetic Resonance in DPPH Shane Duane ID: 08764522 JS Theoretical Physics 5th Dec 2010 Abstract Electron Paramagnetic Resonance (EPR) was investigated in diphenyl pecryl hydrazyl (DPPH).
On Electron Paramagnetic Resonance in DPPH Shane Duane ID: 08764522 JS Theoretical Physics 5th Dec 2010 Abstract Electron Paramagnetic Resonance (EPR) was investigated in diphenyl pecryl hydrazyl (DPPH).
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis)
 Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Optical Spectroscopy 1 1. Absorption spectroscopy (UV/vis) 2 2. Circular dichroism (optical activity) CD / ORD 3 3. Fluorescence spectroscopy and energy transfer Electromagnetic Spectrum Electronic Molecular
Nuclear Magnetic Resonance Spectroscopy Thomas Wenzel Department of Chemistry Bates College, Lewiston ME
 Nuclear Magnetic Resonance Spectroscopy Thomas Wenzel Department of Chemistry Bates College, Lewiston ME 04240 twenzel@bates.edu The following textual material is designed to accompany a series of in-class
Nuclear Magnetic Resonance Spectroscopy Thomas Wenzel Department of Chemistry Bates College, Lewiston ME 04240 twenzel@bates.edu The following textual material is designed to accompany a series of in-class
Chemistry 121: Atomic and Molecular Chemistry Topic 3: Atomic Structure and Periodicity
 Text Chapter 2, 8 & 9 3.1 Nature of light, elementary spectroscopy. 3.2 The quantum theory and the Bohr atom. 3.3 Quantum mechanics; the orbital concept. 3.4 Electron configurations of atoms 3.5 The periodic
Text Chapter 2, 8 & 9 3.1 Nature of light, elementary spectroscopy. 3.2 The quantum theory and the Bohr atom. 3.3 Quantum mechanics; the orbital concept. 3.4 Electron configurations of atoms 3.5 The periodic
