Balmer- and L-shell series of highly charged uranium in the experimental storage ring
|
|
|
- Easter Fleming
- 6 years ago
- Views:
Transcription
1 Balmer- and L-shell series of highly charged uranium in the experimental storage ring Summer student GSI Thomas Burschil Johann Wolfgang von Goethe-University, Frankfurt/Main 09/24/2007
2 Overview Physical basics Radiative emission Experiment Research at the ESR series Balmer series in hydrogen-like uranium L-shell series in helium- and lithium-like uranium
3 Overview Physical basics Radiative emission Experiment Research at the ESR series Balmer series in hydrogen-like uranium L-shell series in helium- and lithium-like uranium
4 Overview Physical basics Radiative emission Experiment Research at the ESR series Balmer series in hydrogen-like uranium L-shell series in helium- and lithium-like uranium
5 Radiative processes transition recombination electron capture radiative (REC) time reverse photo ionization non-radiative (NRC) three body interaction where momentum and energy is shared between the collision partner bremsstrahlung, etc.
6 Radiative processes transition recombination electron capture radiative (REC) time reverse photo ionization non-radiative (NRC) three body interaction where momentum and energy is shared between the collision partner bremsstrahlung, etc.
7 Radiative processes transition recombination electron capture radiative (REC) time reverse photo ionization non-radiative (NRC) three body interaction where momentum and energy is shared between the collision partner bremsstrahlung, etc.
8 Radiative processes transition recombination electron capture radiative (REC) time reverse photo ionization non-radiative (NRC) three body interaction where momentum and energy is shared between the collision partner bremsstrahlung, etc.
9 Radiative processes transition recombination electron capture radiative (REC) time reverse photo ionization non-radiative (NRC) three body interaction where momentum and energy is shared between the collision partner bremsstrahlung, etc.
10 Transition from energy level to energy level energy splitting so-called series finite state has the same main quantum number Lyman, Balmer, Paschen,... in hydrogen-like ions K-shell, L-shell, M-shell,... in other ions For example: ω = E f E i
11 Transition from energy level to energy level energy splitting so-called series finite state has the same main quantum number Lyman, Balmer, Paschen,... in hydrogen-like ions K-shell, L-shell, M-shell,... in other ions For example: ω = E f E i
12 Transition from energy level to energy level energy splitting so-called series finite state has the same main quantum number Lyman, Balmer, Paschen,... in hydrogen-like ions K-shell, L-shell, M-shell,... in other ions For example: ω = E f E i
13 Transition from energy level to energy level energy splitting so-called series finite state has the same main quantum number Lyman, Balmer, Paschen,... in hydrogen-like ions K-shell, L-shell, M-shell,... in other ions For example: ω = E f E i
14 Transition from energy level to energy level energy splitting so-called series finite state has the same main quantum number Lyman, Balmer, Paschen,... in hydrogen-like ions K-shell, L-shell, M-shell,... in other ions For example: ω = E f E i
15 REC and NRC ion captures electron from a target atom two possibilities: 1. non-radiative electron capture
16 REC and NRC ion captures electron from a target atom two possibilities: 1. non-radiative electron capture
17 REC and NRC ion captures electron from a target atom two possibilities: 1. radiative electron capture 2. non-radiative electron capture
18 REC and NRC ion captures electron from a target atom two possibilities: 1. radiative electron capture 2. non-radiative electron capture
19 REC and NRC cross section: σ REC Z T Z 5 P σ NRC Z 5 T Z 5 P
20 REC and NRC ion captures electron from a target atom two possibilities: 1. radiative electron capture 2. non-radiative electron capture
21 REC and NRC cross section: σ REC Z T Z 5 P v 5/2 σ NRC Z 5 T Z 5 P v 11
22 REC and NRC cross section: σ REC Z T Z 5 P v 5/2 σ NRC Z 5 T Z 5 P v 11
23 Research facility UNILAC SIS ESR electron cooler radiative recombination experiments gas-jet target electron capture experiments
24 Research facility UNILAC SIS ESR electron cooler radiative recombination experiments gas-jet target electron capture experiments
25 Research facility UNILAC SIS ESR electron cooler radiative recombination experiments gas-jet target electron capture experiments
26 Research facility UNILAC SIS ESR electron cooler radiative recombination experiments gas-jet target electron capture experiments
27 Observation places at the gas-jet target
28 Observation places at the gas-jet target
29 Recorded spectra
30 Recorded spectra
31 Recorded spectra
32 Recorded spectra
33 Calibration
34 Calibration system parameters U 92+ Ar observation angle: 35 beam energy: 88 MeV /u β = , γ = transfer factor between the laboratory and the emitter system: Doppler Shift = E labor sys = E emitter sys γ (1 β cos ϑ)
35 Calibration system parameters U 92+ Ar observation angle: 35 beam energy: 88 MeV /u β = , γ = transfer factor between the laboratory and the emitter system: Doppler Shift = Calibration E labor sys = E emitter sys γ (1 β cos ϑ) E emitter system E labor system channel 34.2 kev 46.9 kev 1784 ± kev 20.4 kev 733 ± 10
36 Calibration
37 Comparison Binding energies associated with transitions in a Balmer series peak E labor system E emitter system kev ± 0.44 kev kev ± 0.32 kev kev ± 0.43 kev kev ± 0.32 kev kev ± 0.60 kev kev ± 0.44 kev kev ± 0.43 kev kev ± 0.31 kev kev ± 0.49 kev kev ± 0.36 kev kev ± 0.43 kev kev ± 0.32 kev kev ± 0.62 kev kev ± 0.45 kev kev ± 0.60 kev kev ± 0.44 kev kev ± 0.47 kev kev ± 0.34 kev end kev ± 0.50 kev kev ± 0.36 kev
38 Comparison Binding energies associated with transitions in a Balmer series Example for transition energies
39 Comparison Binding energies associated with transitions in a Balmer series Example for transition energies
40 Comparison Binding energies associated with transitions in a Balmer series Example for transition energies
41 Comparison Binding energies associated with transitions in a Balmer series Example for transition energies
42 Comparison Binding energies associated with transitions in a Balmer series peak E emitter system E transition transition kev ± 0.32 kev kev 3s 1/2 2p 3/ kev ± 0.32 kev kev 3d 5/2 2p 3/ kev ± 0.44 kev kev 3s 1/2 2s 1/ kev ± 0.31 kev kev 3p 3/2 2s 1/ kev ± 0.36 kev kev 4d 5/2 2p 3/ kev ± 0.32 kev kev 5s 1/2 2p 3/ kev ± 0.45 kev kev 4p 3/2 2s 1/ kev ± 0.44 kev kev 5p 3/2 2s 1/ kev ± 0.34 kev kev 6s 1/2 2s 1/2 end kev ± 0.36 kev kev continuum
43 Results and Conclusion Spectra with associated transitions to the L-shell
44 Results and Conclusion Cascade
45 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton
46 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton
47 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton
48 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton
49 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton
50 L-shell series in helium- and lithium-like uranium experiment in August 2007 spectra with the first 4 L-shell lines data do not fit with the theoretical values more corrections: shielding correction every electron in the ion shields one proton E corr ( )
51 Spectrum of lithium-like uranium M- and L-shell series
52 Comparison Shielding correction Helium-like uranium in [kev ] peak E labor sys E emitter sys E shield corr. E theor
53 Comparison Shielding correction Lithium-like uranium in [kev ] peak E labor sys E emitter sys E shield corr. E theor
54 Conclusions E ( ) shielding correction is very rough approximation a more precise theory is needed
55 Conclusions E ( ) shielding correction is very rough approximation a more precise theory is needed
56 The end
57 The end Questions?
free electron plus He-like ion
 free electron plus He-like ion E e I p,n E 2 E 1 ΔE=E e +I p,n aber: ΔE=E 2 -E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012 2011-11-16 1 dielectronic recombination E 2 E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012
free electron plus He-like ion E e I p,n E 2 E 1 ΔE=E e +I p,n aber: ΔE=E 2 -E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012 2011-11-16 1 dielectronic recombination E 2 E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012
Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics
 Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics Andrey Surzhykov Technische Universität Braunschweig Physikalisch-Technische Bundesanstalt (PTB) Lecture 1
Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics Andrey Surzhykov Technische Universität Braunschweig Physikalisch-Technische Bundesanstalt (PTB) Lecture 1
Emitted Spectrum Summary of emission processes Emissivities for emission lines: - Collisionally excited lines - Recombination cascades Emissivities
 Emitted Spectrum Summary of emission processes Emissivities for emission lines: - Collisionally excited lines - Recombination cascades Emissivities for continuum processes - recombination - brehmsstrahlung
Emitted Spectrum Summary of emission processes Emissivities for emission lines: - Collisionally excited lines - Recombination cascades Emissivities for continuum processes - recombination - brehmsstrahlung
Physics 1C Lecture 29B
 Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Formation of excited states in high-z helium-like systems
 Hyperfine Interactions 127 (2000) 257 262 257 Formation of excited states in high-z helium-like systems S. Fritzsche a,th.stöhlker b,c, O. Brinzanescu c,d and B. Fricke a a Fachbereich Physik, Universität
Hyperfine Interactions 127 (2000) 257 262 257 Formation of excited states in high-z helium-like systems S. Fritzsche a,th.stöhlker b,c, O. Brinzanescu c,d and B. Fricke a a Fachbereich Physik, Universität
(b) The wavelength of the radiation that corresponds to this energy is 6
 Chapter 7 Problem Solutions 1. A beam of electrons enters a uniform 1.0-T magnetic field. (a) Find the energy difference between electrons whose spins are parallel and antiparallel to the field. (b) Find
Chapter 7 Problem Solutions 1. A beam of electrons enters a uniform 1.0-T magnetic field. (a) Find the energy difference between electrons whose spins are parallel and antiparallel to the field. (b) Find
Production of HCI with an electron beam ion trap
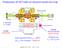 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
High Resolution Electron Spectrometry at the NESR. Ajay Kumar
 High Resolution Electron Spectrometry at the NESR Collaboration Ajay Kumar GSI, Darmstadt Stored Particles Atomic Physics Research Collaboration R. Mann G. Garcia X. Ma B. Sulik J. Ullrich L.C. Tribedi
High Resolution Electron Spectrometry at the NESR Collaboration Ajay Kumar GSI, Darmstadt Stored Particles Atomic Physics Research Collaboration R. Mann G. Garcia X. Ma B. Sulik J. Ullrich L.C. Tribedi
Auger electron and x-ray emission from high-z few-electron ions
 Auger electron and x-ray emission from high-z few-electron ions S. Fritzsche MPI für Kernphysik Heidelberg and GSI Darmstadt 4th August 2007 Main goal for studying high-z ions is the better understanding
Auger electron and x-ray emission from high-z few-electron ions S. Fritzsche MPI für Kernphysik Heidelberg and GSI Darmstadt 4th August 2007 Main goal for studying high-z ions is the better understanding
The GSI Anomaly. M. Lindner. Max-Planck-Institut für Kernphysik, Heidelberg. Sildes partially adopted from F. Bosch
 The GSI Anomaly M. Lindner Max-Planck-Institut für Kernphysik, Heidelberg Sildes partially adopted from F. Bosch What is the GSI Anomaly? Periodically modualted exponential β-decay law of highly charged,
The GSI Anomaly M. Lindner Max-Planck-Institut für Kernphysik, Heidelberg Sildes partially adopted from F. Bosch What is the GSI Anomaly? Periodically modualted exponential β-decay law of highly charged,
HL Chemistry. Tuesday August 25th Tuesday, August 25, 15
 HL Chemistry Tuesday August 25th 2015 Agenda Warm-Up NONE The Atom Project - Use your class time wisely HOMEWORK Questions? The Atom Project -Group Members? DUE August 31st 2015 Atomic Structure Questions
HL Chemistry Tuesday August 25th 2015 Agenda Warm-Up NONE The Atom Project - Use your class time wisely HOMEWORK Questions? The Atom Project -Group Members? DUE August 31st 2015 Atomic Structure Questions
X-ray spectroscopy. Crystal spectrometers. Reflection gratings. Transmission gratings. Solid state detectors
 X-ray spectroscopy Crystal spectrometers Reflection gratings Transmission gratings Solid state detectors Bragg s law How are X-rays reflected from a surface? At short wavelengths, mirrors do not work well.
X-ray spectroscopy Crystal spectrometers Reflection gratings Transmission gratings Solid state detectors Bragg s law How are X-rays reflected from a surface? At short wavelengths, mirrors do not work well.
Atomic Structure and Processes
 Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
Chapter 5 Atomic Structure and Processes 5.1 Elementary atomic structure Bohr Orbits correspond to principal quantum number n. Hydrogen atom energy levels where the Rydberg energy is R y = m e ( e E n
Theory of optically thin emission line spectroscopy
 Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
Theory of optically thin emission line spectroscopy 1 Important definitions In general the spectrum of a source consists of a continuum and several line components. Processes which give raise to the continuous
Lewis 2.1, 2.2 and 2.3
 Chapter 2(and 3) Cross-Sections TA Lewis 2.1, 2.2 and 2.3 Learning Objectives Understand different types of nuclear reactions Understand cross section behavior for different reactions Understand d resonance
Chapter 2(and 3) Cross-Sections TA Lewis 2.1, 2.2 and 2.3 Learning Objectives Understand different types of nuclear reactions Understand cross section behavior for different reactions Understand d resonance
HL Chemistry. Thursday August 20th Thursday, August 20, 15
 HL Chemistry Thursday August 20th 2015 Agenda Warm Up: Electron Configuration Practice Review Topic 2.2 Electron Configuration and begin Topic 12.1 Atomic Structure Warm Up You have 10 minutes ONLY! GO!
HL Chemistry Thursday August 20th 2015 Agenda Warm Up: Electron Configuration Practice Review Topic 2.2 Electron Configuration and begin Topic 12.1 Atomic Structure Warm Up You have 10 minutes ONLY! GO!
Feasibility Studies for the EXL Project at FAIR *
 * a,b,, S. Bagchi c, S. Diebold d, C. Dimopoulou a, P. Egelhof a, V. Eremin e, S. Ilieva a, N. Kalantar-Nayestanaki c, O. Kiselev a,f, T. Kröll f, Y.A. Litvinov a,g, M. Mutterer a, M.A. Najafi c, N. Petridis
* a,b,, S. Bagchi c, S. Diebold d, C. Dimopoulou a, P. Egelhof a, V. Eremin e, S. Ilieva a, N. Kalantar-Nayestanaki c, O. Kiselev a,f, T. Kröll f, Y.A. Litvinov a,g, M. Mutterer a, M.A. Najafi c, N. Petridis
HL Chemistry. Monday August 24th Monday, August 24, 15
 HL Chemistry Monday August 24th 2015 Agenda Warm Up: Ionization Energy of boron Review Topic 12.1 Atomic Structure Calculating Ionization Energy The Atom Project (?) Warm Up You have 5 minutes ONLY! GO!
HL Chemistry Monday August 24th 2015 Agenda Warm Up: Ionization Energy of boron Review Topic 12.1 Atomic Structure Calculating Ionization Energy The Atom Project (?) Warm Up You have 5 minutes ONLY! GO!
Physics 1C Lecture 29A. Finish off Ch. 28 Start Ch. 29
 Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
Time-modulation of electron-capture decay factor detected at GSI, Darmstadt
 Time-modulation of electron-capture decay factor detected at GSI, Darmstadt Byung Kyu Park Department of Physics University of California, Berkeley Physics 250 March 20, 2008 Byung Kyu Park (UC Berkeley)
Time-modulation of electron-capture decay factor detected at GSI, Darmstadt Byung Kyu Park Department of Physics University of California, Berkeley Physics 250 March 20, 2008 Byung Kyu Park (UC Berkeley)
High-energy collision processes involving intense laser fields
 High-energy collision processes involving intense laser fields Carsten Müller Max Planck Institute for Nuclear Physics, Theory Division (Christoph H. Keitel), Heidelberg, Germany EMMI Workshop: Particle
High-energy collision processes involving intense laser fields Carsten Müller Max Planck Institute for Nuclear Physics, Theory Division (Christoph H. Keitel), Heidelberg, Germany EMMI Workshop: Particle
Atomic Structure Ch , 9.6, 9.7
 Ch. 9.2-4, 9.6, 9.7 Magnetic moment of an orbiting electron: An electron orbiting a nucleus creates a current loop. A current loop behaves like a magnet with a magnetic moment µ:! µ =! µ B " L Bohr magneton:
Ch. 9.2-4, 9.6, 9.7 Magnetic moment of an orbiting electron: An electron orbiting a nucleus creates a current loop. A current loop behaves like a magnet with a magnetic moment µ:! µ =! µ B " L Bohr magneton:
Question: How are electrons arranged in an atom?
 Honors Chemistry: Coulomb s Law and periodic trends Question: How are electrons arranged in an atom? Coulomb s Law equation: 1. A) Define what each of the following variables in the equation represents.
Honors Chemistry: Coulomb s Law and periodic trends Question: How are electrons arranged in an atom? Coulomb s Law equation: 1. A) Define what each of the following variables in the equation represents.
Fundamentals of Spectroscopy for Optical Remote Sensing. Course Outline 2009
 Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
Fundamentals of Spectroscopy for Optical Remote Sensing Course Outline 2009 Part I. Fundamentals of Quantum Mechanics Chapter 1. Concepts of Quantum and Experimental Facts 1.1. Blackbody Radiation and
PHYS 202. Lecture 23 Professor Stephen Thornton April 25, 2005
 PHYS 202 Lecture 23 Professor Stephen Thornton April 25, 2005 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
PHYS 202 Lecture 23 Professor Stephen Thornton April 25, 2005 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
Chapter 4. Spectroscopy. Dr. Tariq Al-Abdullah
 Chapter 4 Spectroscopy Dr. Tariq Al-Abdullah Learning Goals: 4.1 Spectral Lines 4.2 Atoms and Radiation 4.3 Formation of the Spectral Lines 4.4 Molecules 4.5 Spectral Line Analysis 2 DR. T. AL-ABDULLAH
Chapter 4 Spectroscopy Dr. Tariq Al-Abdullah Learning Goals: 4.1 Spectral Lines 4.2 Atoms and Radiation 4.3 Formation of the Spectral Lines 4.4 Molecules 4.5 Spectral Line Analysis 2 DR. T. AL-ABDULLAH
Atomic double slit: Coherence transfer through excitation and (Auger) decay processes. S. Fritzsche, Kassel University Göteborg, 3rd June 2006
 Atomic double slit: Coherence transfer through excitation and (Auger) decay processes S. Fritzsche, Kassel University Göteborg, 3rd June 2006 Experiments with double slits (Feynman-Lectures 1962) Interference
Atomic double slit: Coherence transfer through excitation and (Auger) decay processes S. Fritzsche, Kassel University Göteborg, 3rd June 2006 Experiments with double slits (Feynman-Lectures 1962) Interference
Linear and circular accelerators
 Linear and circular accelerators Ion Accelerator Physics and Technology Oliver Boine-Frankenheim, Gesellschaft für Schwerionenforschung (GSI), Darmstadt Tel. 06159 712408, O.Boine-Frankenheim@gsi.de o
Linear and circular accelerators Ion Accelerator Physics and Technology Oliver Boine-Frankenheim, Gesellschaft für Schwerionenforschung (GSI), Darmstadt Tel. 06159 712408, O.Boine-Frankenheim@gsi.de o
5.111 Lecture Summary #5 Friday, September 12, 2014
 5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
5.111 Lecture Summary #5 Friday, September 12, 2014 Readings for today: Section 1.3 Atomic Spectra, Section 1.7 up to equation 9b Wavefunctions and Energy Levels, Section 1.8 The Principle Quantum Number.
The interaction of radiation with matter
 Basic Detection Techniques 2009-2010 http://www.astro.rug.nl/~peletier/detectiontechniques.html Detection of energetic particles and gamma rays The interaction of radiation with matter Peter Dendooven
Basic Detection Techniques 2009-2010 http://www.astro.rug.nl/~peletier/detectiontechniques.html Detection of energetic particles and gamma rays The interaction of radiation with matter Peter Dendooven
Atomic Physics with Stored and Cooled Ions
 Lecture #8 Atomic Physics with Stored and Cooled Ions Klaus Blaum Gesellschaft für Schwerionenforschung, GSI, Darmstadt and CERN, Physics Department, Geneva, Switzerland Summer School, Lanzhou, China,
Lecture #8 Atomic Physics with Stored and Cooled Ions Klaus Blaum Gesellschaft für Schwerionenforschung, GSI, Darmstadt and CERN, Physics Department, Geneva, Switzerland Summer School, Lanzhou, China,
PHYS 202. Lecture 23 Professor Stephen Thornton April 20, 2006
 PHYS 202 Lecture 23 Professor Stephen Thornton April 20, 2006 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
PHYS 202 Lecture 23 Professor Stephen Thornton April 20, 2006 Reading Quiz The noble gases (He, Ne, Ar, etc.) 1) are very reactive because they lack one electron of being in a closed shell. 2) are very
THE NATURE OF THE ATOM. alpha particle source
 chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
Astrophysical Radiation Processes
 PHY3145 Topics in Theoretical Physics Astrophysical Radiation Processes 3: Relativistic effects I Dr. J. Hatchell, Physics 407, J.Hatchell@exeter.ac.uk Course structure 1. Radiation basics. Radiative transfer.
PHY3145 Topics in Theoretical Physics Astrophysical Radiation Processes 3: Relativistic effects I Dr. J. Hatchell, Physics 407, J.Hatchell@exeter.ac.uk Course structure 1. Radiation basics. Radiative transfer.
The early periodic table based on atomic weight. (Section 5.1) Lets review: What is a hydrogen atom? 1 electron * nucleus H 1 proton
 PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
Chapter 22 Quantum Mechanics & Atomic Structure 22.1 Photon Theory of Light and The Photoelectric Effect Homework # 170
 22.1 Photon Theory of Light and The Photoelectric Effect Homework # 170 See Homework #95 in "Chapter 12-Electrostatics" for the table of "Useful nformation" on atomic particles. 01. What is the energy
22.1 Photon Theory of Light and The Photoelectric Effect Homework # 170 See Homework #95 in "Chapter 12-Electrostatics" for the table of "Useful nformation" on atomic particles. 01. What is the energy
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
Rb, which had been compressed to a density of 1013
 Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
Slide 1 / 57. Nuclear Physics & Nuclear Reactions Practice Problems
 Slide 1 / 57 Nuclear Physics & Nuclear Reactions Practice Problems Slide 2 / 57 Multiple Choice Slide 3 / 57 1 The atomic nucleus consists of: A B C D E Electrons Protons Protons and electrons Protons
Slide 1 / 57 Nuclear Physics & Nuclear Reactions Practice Problems Slide 2 / 57 Multiple Choice Slide 3 / 57 1 The atomic nucleus consists of: A B C D E Electrons Protons Protons and electrons Protons
Beam Cooling. Beam Cooling. M. Steck, GSI, Darmstadt CERN Accelerator School Chios, Greece September 18 30, Introduction. 1.
 Beam Cooling, GSI, Darmstadt CERN Accelerator School, September 18 30, 2011 Beam Cooling Introduction 1.Electron Cooling 2.Ionization Cooling 3.Laser Cooling 4.Stochastic Cooling Beam Cooling Beam cooling
Beam Cooling, GSI, Darmstadt CERN Accelerator School, September 18 30, 2011 Beam Cooling Introduction 1.Electron Cooling 2.Ionization Cooling 3.Laser Cooling 4.Stochastic Cooling Beam Cooling Beam cooling
ATHENA / AD-1. First production and detection of cold antihydrogen atoms. ATHENA Collaboration. Rolf Landua CERN
 ATHENA / AD-1 First production and detection of cold antihydrogen atoms ATHENA Collaboration Rolf Landua CERN 1 LONG TERM PHYSICS GOALS Antihydrogen = Hydrogen? CPT Gravity But... 2 FIRST GOAL PRODUCTION
ATHENA / AD-1 First production and detection of cold antihydrogen atoms ATHENA Collaboration Rolf Landua CERN 1 LONG TERM PHYSICS GOALS Antihydrogen = Hydrogen? CPT Gravity But... 2 FIRST GOAL PRODUCTION
Stellar Astrophysics: The Interaction of Light and Matter
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Gas 1: Molecular clouds
 Gas 1: Molecular clouds > 4000 known with masses ~ 10 3 to 10 5 M T ~ 10 to 25 K (cold!); number density n > 10 9 gas particles m 3 Emission bands in IR, mm, radio regions from molecules comprising H,
Gas 1: Molecular clouds > 4000 known with masses ~ 10 3 to 10 5 M T ~ 10 to 25 K (cold!); number density n > 10 9 gas particles m 3 Emission bands in IR, mm, radio regions from molecules comprising H,
CDO AP Chemistry Unit 5
 1. a. Calculate the wavelength of electromagnetic radiation that has a frequency of 5.56 MHz. b. Calculate the frequency of electromagnetic radiation that has a wavelength equal to 667 nm. 2. Electromagnetic
1. a. Calculate the wavelength of electromagnetic radiation that has a frequency of 5.56 MHz. b. Calculate the frequency of electromagnetic radiation that has a wavelength equal to 667 nm. 2. Electromagnetic
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
ATOMIC STRUCRURE
 ATOMIC STRUCRURE Long Answer Questions: 1. What are quantum numbers? Give their significance? Ans. The various orbitals in an atom qualitatively distinguished by their size, shape and orientation. The
ATOMIC STRUCRURE Long Answer Questions: 1. What are quantum numbers? Give their significance? Ans. The various orbitals in an atom qualitatively distinguished by their size, shape and orientation. The
Interstellar Astrophysics Summary notes: Part 2
 Interstellar Astrophysics Summary notes: Part 2 Dr. Paul M. Woods The main reference source for this section of the course is Chapter 5 in the Dyson and Williams (The Physics of the Interstellar Medium)
Interstellar Astrophysics Summary notes: Part 2 Dr. Paul M. Woods The main reference source for this section of the course is Chapter 5 in the Dyson and Williams (The Physics of the Interstellar Medium)
Nuclear Decays. Alpha Decay
 Nuclear Decays The first evidence of radioactivity was a photographic plate, wrapped in black paper and placed under a piece of uranium salt by Henri Becquerel on February 26, 1896. Like many events in
Nuclear Decays The first evidence of radioactivity was a photographic plate, wrapped in black paper and placed under a piece of uranium salt by Henri Becquerel on February 26, 1896. Like many events in
Experiments with hydrogen - discovery of the Lamb shift
 Experiments with hydrogen - discovery of the Lamb shift Haris Ðapo Relativistic heavy ion seminar, October 26, 2006 Outline 1 Pre-Lamb experiment The beginning (Bohr s formula) Fine structure (Dirac s
Experiments with hydrogen - discovery of the Lamb shift Haris Ðapo Relativistic heavy ion seminar, October 26, 2006 Outline 1 Pre-Lamb experiment The beginning (Bohr s formula) Fine structure (Dirac s
5-A / 9, WEA, Sat Nagar, Karol Bagh New Delhi Web:
 Keshaw Classes IIT/JEE Medical Classes 5-A 11028 / 9, WEA, Sat Nagar, Karol Bagh New Delhi-110005 Mob:9910915514,9953150192 Ph:011-45660510 E-mail : keshawclasses@gmail.com Web:www.keshawclasses.com MODERN
Keshaw Classes IIT/JEE Medical Classes 5-A 11028 / 9, WEA, Sat Nagar, Karol Bagh New Delhi-110005 Mob:9910915514,9953150192 Ph:011-45660510 E-mail : keshawclasses@gmail.com Web:www.keshawclasses.com MODERN
Photon transport mode in Serpent 2
 Photon transport mode in Serpent 2 Toni Kaltiaisenaho VTT Technical Research Centre of Finland, LTD Serpent User Group Meeting, Knoxville, TN October 13 16, 215 October 14, 215 1/21 Outline Photon physics
Photon transport mode in Serpent 2 Toni Kaltiaisenaho VTT Technical Research Centre of Finland, LTD Serpent User Group Meeting, Knoxville, TN October 13 16, 215 October 14, 215 1/21 Outline Photon physics
Beam Diagnostics and Instrumentation JUAS, Archamps Peter Forck Gesellschaft für Schwerionenforschnung (GSI)
 Beam Diagnostics and Instrumentation JUAS, Archamps Peter Forck Gesellschaft für Schwerionenforschnung (GSI), 2003, A dedicated proton accelerator for 1p-physics at the future GSI Demands facilities for
Beam Diagnostics and Instrumentation JUAS, Archamps Peter Forck Gesellschaft für Schwerionenforschnung (GSI), 2003, A dedicated proton accelerator for 1p-physics at the future GSI Demands facilities for
Beam-plasma atomic data needs for fusion devices
 Beam-plasma atomic data needs for fusion devices Contemporary areas of application on fusion machines: Beam stopping (H/D/T heating beams) Beam shinethrough in small machines and/or at low density. Power
Beam-plasma atomic data needs for fusion devices Contemporary areas of application on fusion machines: Beam stopping (H/D/T heating beams) Beam shinethrough in small machines and/or at low density. Power
APEX CARE INSTITUTE FOR PG - TRB, SLET AND NET IN PHYSICS
 Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
X-ray Energy Spectroscopy (XES).
 X-ray Energy Spectroscopy (XES). X-ray fluorescence as an analytical tool for element analysis is based on 3 fundamental parameters: A. Specificity: In determining an x-ray emission energy E certainty
X-ray Energy Spectroscopy (XES). X-ray fluorescence as an analytical tool for element analysis is based on 3 fundamental parameters: A. Specificity: In determining an x-ray emission energy E certainty
Bohr (1913) ( 3.4 ev) (L -shell) (n =2) Electron Binding Energy ev)
 Marian Smoluchowski-Emil Warburg-Prize Heavy-Ion Atom Collisions Atomic Physics under Extreme Conditions Andrzej Warczak One of the actual frontiers in physics is the study of matter exposed to extremely
Marian Smoluchowski-Emil Warburg-Prize Heavy-Ion Atom Collisions Atomic Physics under Extreme Conditions Andrzej Warczak One of the actual frontiers in physics is the study of matter exposed to extremely
Auger & X-ray Fluorescence
 At low energies or low temperature gas (plasma) the primary processes are photoionzation or excitation by particles (electron, atom, proton). Recombination takes place with emission of photons. In hot
At low energies or low temperature gas (plasma) the primary processes are photoionzation or excitation by particles (electron, atom, proton). Recombination takes place with emission of photons. In hot
Quantum Mechanics and Stellar Spectroscopy.
 Quantum Mechanics and Stellar Spectroscopy http://apod.nasa.gov/apod/ Recall the electric force. Like gravity it is a 1/r 2 force/ That is: F elec = Z 1 Z 2 e2 r 2 where Z 1 and Z 2 are the (integer) numbers
Quantum Mechanics and Stellar Spectroscopy http://apod.nasa.gov/apod/ Recall the electric force. Like gravity it is a 1/r 2 force/ That is: F elec = Z 1 Z 2 e2 r 2 where Z 1 and Z 2 are the (integer) numbers
 IONIZING RADIATION RADIOLOGICAL PHYSICS The science of ionizing radiation and its interaction with matter Special interest in the energy absorbed Radiation dosimetry quantitative determination of that
IONIZING RADIATION RADIOLOGICAL PHYSICS The science of ionizing radiation and its interaction with matter Special interest in the energy absorbed Radiation dosimetry quantitative determination of that
Interaction of Ionizing Radiation with Matter
 Type of radiation charged particles photonen neutronen Uncharged particles Charged particles electrons (β - ) He 2+ (α), H + (p) D + (d) Recoil nuclides Fission fragments Interaction of ionizing radiation
Type of radiation charged particles photonen neutronen Uncharged particles Charged particles electrons (β - ) He 2+ (α), H + (p) D + (d) Recoil nuclides Fission fragments Interaction of ionizing radiation
a) how many electrons do you see in the picture? How many protons? d) compare the energy from 3b with the energy in 2a and then in 2c.
 During Class Invention Question: How are electrons arranged in an atom? 1. Describe the nature of the interaction between protons and electrons in an atom? Consider using some or all of the following terms
During Class Invention Question: How are electrons arranged in an atom? 1. Describe the nature of the interaction between protons and electrons in an atom? Consider using some or all of the following terms
Report A+M/PSI Data Centre NRC Kurchatov Institute
 Report A+M/PSI Data Centre NRC Kurchatov Institute Yu.V.Martynenko 21st Meeting of the Atomic and Molecular Data Centers and ALADDIN Network Vienna, 07-09 September 2011 The main activities on A+M/PSI
Report A+M/PSI Data Centre NRC Kurchatov Institute Yu.V.Martynenko 21st Meeting of the Atomic and Molecular Data Centers and ALADDIN Network Vienna, 07-09 September 2011 The main activities on A+M/PSI
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
arxiv: v1 [physics.atom-ph] 21 Jun 2011
![arxiv: v1 [physics.atom-ph] 21 Jun 2011 arxiv: v1 [physics.atom-ph] 21 Jun 2011](/thumbs/80/80601513.jpg) arxiv:1106.4155v1 [physics.atom-ph] 21 Jun 2011 Differential energy measurement between He- and Li-like uranium intra-shell transitions M. Trassinelli 1, A. Kumar 2, H.F. Beyer 3, P. Indelicato 4, R. Märtin
arxiv:1106.4155v1 [physics.atom-ph] 21 Jun 2011 Differential energy measurement between He- and Li-like uranium intra-shell transitions M. Trassinelli 1, A. Kumar 2, H.F. Beyer 3, P. Indelicato 4, R. Märtin
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Optical Spectroscopy and Atomic Structure. PHYS 0219 Optical Spectroscopy and Atomic Structure 1
 Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Joint ICTP-IAEA Workshop on Fusion Plasma Modelling using Atomic and Molecular Data January 2012
 2327-4 Joint ICTP- Workshop on Fusion Plasma Modelling using Atomic and Molecular Data 23-27 January 2012 Atomic Processes Modeling in Plasmas Modeling Spectroscopic Observables from Plasmas Hyun-Kyung
2327-4 Joint ICTP- Workshop on Fusion Plasma Modelling using Atomic and Molecular Data 23-27 January 2012 Atomic Processes Modeling in Plasmas Modeling Spectroscopic Observables from Plasmas Hyun-Kyung
We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism".
 We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism". -Richard Feynman Quantum H Atom Review Radia Wave Function (1s):
We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism". -Richard Feynman Quantum H Atom Review Radia Wave Function (1s):
The FAIR Accelerator Facility
 The FAIR Accelerator Facility SIS300 existing GSI proton linac SIS18 UNILAC SIS100 HESR pbar target SuperFRS goals: higher intensity (low charge states) higher energy (high charge states) production of
The FAIR Accelerator Facility SIS300 existing GSI proton linac SIS18 UNILAC SIS100 HESR pbar target SuperFRS goals: higher intensity (low charge states) higher energy (high charge states) production of
Theory of electron cooling
 Theory of electron cooling Daria Astapovych 03/12/2014 HSC Meeting Outline Motivation and idea of the particle beam cooling Cooler Low energy, high energy beam Electron beam Kinetics of electron cooling
Theory of electron cooling Daria Astapovych 03/12/2014 HSC Meeting Outline Motivation and idea of the particle beam cooling Cooler Low energy, high energy beam Electron beam Kinetics of electron cooling
Spectroscopy. Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths.
 Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths. In contract, light emitted in low=pressure gas discharge contains only discrete individual wavelengths,
Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths. In contract, light emitted in low=pressure gas discharge contains only discrete individual wavelengths,
CHAPTER 2 RADIATION INTERACTIONS WITH MATTER HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION MR KAMARUL AMIN BIN ABDULLAH
 HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION CHAPTER 2 RADIATION INTERACTIONS WITH MATTER PREPARED BY: MR KAMARUL AMIN BIN ABDULLAH SCHOOL OF MEDICAL IMAGING FACULTY OF HEALTH SCIENCE Interactions
HDR 112 RADIATION BIOLOGY AND RADIATION PROTECTION CHAPTER 2 RADIATION INTERACTIONS WITH MATTER PREPARED BY: MR KAMARUL AMIN BIN ABDULLAH SCHOOL OF MEDICAL IMAGING FACULTY OF HEALTH SCIENCE Interactions
K.U.Leuven. : an Application of the MYRRHA Accelerator for Nuclear Physics. Piet Van Duppen IKS, KUL, Leuven (BE)
 ISOL@MYRRHA : an Application of the MYRRHA Accelerator for Nuclear Physics Piet Van Duppen IKS, KUL, Leuven (BE) MYRRHA ADS first step demo facility at power (50-100 MW) Flexible irradiation facility Need
ISOL@MYRRHA : an Application of the MYRRHA Accelerator for Nuclear Physics Piet Van Duppen IKS, KUL, Leuven (BE) MYRRHA ADS first step demo facility at power (50-100 MW) Flexible irradiation facility Need
THE OBSERVATION AND ANALYSIS OF STELLAR PHOTOSPHERES
 THE OBSERVATION AND ANALYSIS OF STELLAR PHOTOSPHERES DAVID F. GRAY University of Western Ontario, London, Ontario, Canada CAMBRIDGE UNIVERSITY PRESS Contents Preface to the first edition Preface to the
THE OBSERVATION AND ANALYSIS OF STELLAR PHOTOSPHERES DAVID F. GRAY University of Western Ontario, London, Ontario, Canada CAMBRIDGE UNIVERSITY PRESS Contents Preface to the first edition Preface to the
Dedicated Arrays: MEDEA GDR studies (E γ = MeV) Highly excited CN E*~ MeV, 4 T 8 MeV
 Dedicated Arrays: MEDEA GDR studies (E γ = 10-25 MeV) Highly excited CN E*~ 250-350 MeV, 4 T 8 MeV γ-ray spectrum intermediate energy region 10 MeV/A E beam 100 MeV/A - large variety of emitted particles
Dedicated Arrays: MEDEA GDR studies (E γ = 10-25 MeV) Highly excited CN E*~ 250-350 MeV, 4 T 8 MeV γ-ray spectrum intermediate energy region 10 MeV/A E beam 100 MeV/A - large variety of emitted particles
Lecture 5: the Hydrogen Atom
 Lecture 5: the Hydrogen Atom 1. Hydrogen atom: energy levels in the Bohr model 2. Emission lines: atoms releasing energy as electrons fall from level to level 3. Absorption lines: electrons being bumped
Lecture 5: the Hydrogen Atom 1. Hydrogen atom: energy levels in the Bohr model 2. Emission lines: atoms releasing energy as electrons fall from level to level 3. Absorption lines: electrons being bumped
Multiple Choice Questions
 Nuclear Physics & Nuclear Reactions Practice Problems PSI AP Physics B 1. The atomic nucleus consists of: (A) Electrons (B) Protons (C)Protons and electrons (D) Protons and neutrons (E) Neutrons and electrons
Nuclear Physics & Nuclear Reactions Practice Problems PSI AP Physics B 1. The atomic nucleus consists of: (A) Electrons (B) Protons (C)Protons and electrons (D) Protons and neutrons (E) Neutrons and electrons
Preliminary Quantum Questions
 Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Preliminary Quantum Questions Thomas Ouldridge October 01 1. Certain quantities that appear in the theory of hydrogen have wider application in atomic physics: the Bohr radius a 0, the Rydberg constant
Introduction. X-Ray Production and Quality. Fluorescence Yield. Fluorescence X-Rays. Initiating event. Initiating event 3/18/2011
 X-Ray Production and Quality Chapter 9 F.A. Attix, Introduction to Radiological Physics and Radiation Dosimetry Introduction Physics of x-ray generation Fluorescence x-rays Bremsstrahlung x-rays Beam quality
X-Ray Production and Quality Chapter 9 F.A. Attix, Introduction to Radiological Physics and Radiation Dosimetry Introduction Physics of x-ray generation Fluorescence x-rays Bremsstrahlung x-rays Beam quality
General Physics (PHY 2140)
 General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
Chapter Four (Interaction of Radiation with Matter)
 Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Four (Interaction of Radiation with Matter) Different types of radiation interact
Al-Mustansiriyah University College of Science Physics Department Fourth Grade Nuclear Physics Dr. Ali A. Ridha Chapter Four (Interaction of Radiation with Matter) Different types of radiation interact
Chapter Test B. Chapter: Arrangement of Electrons in Atoms. possible angular momentum quantum numbers? energy level? a. 4 b. 8 c. 16 d.
 Assessment Chapter Test B Chapter: Arrangement of Electrons in Atoms PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question
Assessment Chapter Test B Chapter: Arrangement of Electrons in Atoms PART I In the space provided, write the letter of the term or phrase that best completes each statement or best answers each question
FAIR AT GSI. P. Spiller, GSI, Darmstadt, Germany
 FAIR AT GSI P. Spiller, GSI, Darmstadt, Germany Abstract Based on the experience of the existing GSI facility and with the aim to apply new technical concepts in phase space cooling and fast ramping of
FAIR AT GSI P. Spiller, GSI, Darmstadt, Germany Abstract Based on the experience of the existing GSI facility and with the aim to apply new technical concepts in phase space cooling and fast ramping of
TARGET IIT JEE 2013 XIII MODERN PHYSICS C O N T E N T S. KEY CONCEPT...Page 2. EXERCISE I...Page 6. EXERCISE II...Page 7. EXERCISE III...
 PHYSICS TARGET IIT JEE 013 XIII MODERN PHYSICS C O N T E N T S KEY CONCEPT...Page EXERCISE I...Page 6 EXERCISE II...Page 7 EXERCISE III...Page 8 OBJECTIVE QUESTION BANK... Page 13 ANSWER KEY...Page KEY
PHYSICS TARGET IIT JEE 013 XIII MODERN PHYSICS C O N T E N T S KEY CONCEPT...Page EXERCISE I...Page 6 EXERCISE II...Page 7 EXERCISE III...Page 8 OBJECTIVE QUESTION BANK... Page 13 ANSWER KEY...Page KEY
An element Is a substance that cannot be split into simpler substance. It is composed of discrete particles called atoms.
 digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
digitalteachers.co.ug Atomic structure Inorganic chemistry deals with the physical and chemical properties of the elements of the the periodic table. An element Is a substance that cannot be split into
Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra
 Modern Atomic Physics: Experiment and Theory Preliminary plan of the lectures Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra Lecture 3 April 9 th, 014 1 15.04.015 Preliminary
Modern Atomic Physics: Experiment and Theory Preliminary plan of the lectures Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra Lecture 3 April 9 th, 014 1 15.04.015 Preliminary
LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS. Instructor: Kazumi Tolich
 LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS Instructor: Kazumi Tolich Lecture 23 2 29.1 Spectroscopy 29.2 Atoms The first nuclear physics experiment Using the nuclear model 29.3 Bohr s model of atomic quantization
LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS Instructor: Kazumi Tolich Lecture 23 2 29.1 Spectroscopy 29.2 Atoms The first nuclear physics experiment Using the nuclear model 29.3 Bohr s model of atomic quantization
The Electronic Structure of Atoms
 The Electronic Structure of Atoms Classical Hydrogen-like atoms: Atomic Scale: 10-10 m or 1 Å + - Proton mass : Electron mass 1836 : 1 Problems with classical interpretation: - Should not be stable (electron
The Electronic Structure of Atoms Classical Hydrogen-like atoms: Atomic Scale: 10-10 m or 1 Å + - Proton mass : Electron mass 1836 : 1 Problems with classical interpretation: - Should not be stable (electron
10 May 2018 (Thursday) 2:30-4:30 pm
 THE UNIVERSITY OF HONG KONG DEPARTMENT OF PHYSICS PHYS3652 Principles of Astronomy 10 May 2018 (Thursday) 2:30-4:30 pm A. Answer all FOUR questions (Marks for each question are listed) B. Define every
THE UNIVERSITY OF HONG KONG DEPARTMENT OF PHYSICS PHYS3652 Principles of Astronomy 10 May 2018 (Thursday) 2:30-4:30 pm A. Answer all FOUR questions (Marks for each question are listed) B. Define every
Neutron Interactions Part I. Rebecca M. Howell, Ph.D. Radiation Physics Y2.5321
 Neutron Interactions Part I Rebecca M. Howell, Ph.D. Radiation Physics rhowell@mdanderson.org Y2.5321 Why do we as Medical Physicists care about neutrons? Neutrons in Radiation Therapy Neutron Therapy
Neutron Interactions Part I Rebecca M. Howell, Ph.D. Radiation Physics rhowell@mdanderson.org Y2.5321 Why do we as Medical Physicists care about neutrons? Neutrons in Radiation Therapy Neutron Therapy
6. Stellar spectra. excitation and ionization, Saha s equation stellar spectral classification Balmer jump, H -
 6. Stellar spectra excitation and ionization, Saha s equation stellar spectral classification Balmer jump, H - 1 Occupation numbers: LTE case Absorption coefficient: κ ν = n i σ ν$ à calculation of occupation
6. Stellar spectra excitation and ionization, Saha s equation stellar spectral classification Balmer jump, H - 1 Occupation numbers: LTE case Absorption coefficient: κ ν = n i σ ν$ à calculation of occupation
Chapter 5 Light and Matter
 Chapter 5 Light and Matter Stars and galaxies are too far for us to send a spacecraft or to visit (in our lifetimes). All we can receive from them is light But there is much we can learn (composition,
Chapter 5 Light and Matter Stars and galaxies are too far for us to send a spacecraft or to visit (in our lifetimes). All we can receive from them is light But there is much we can learn (composition,
Nuclear Reactions A Z. Radioactivity, Spontaneous Decay: Nuclear Reaction, Induced Process: x + X Y + y + Q Q > 0. Exothermic Endothermic
 Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Different energy levels
 Different energy levels In the microscopic world energy is discrete www.cgrahamphysics.com Review Atomic electrons can only exist in certain discrete energy levels Light is made of photons When e s lose
Different energy levels In the microscopic world energy is discrete www.cgrahamphysics.com Review Atomic electrons can only exist in certain discrete energy levels Light is made of photons When e s lose
Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms. OR, Show me the Electrons!
 Lecture 2 362 January 16, 2019 Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms OR, Show me the Electrons! Color Red Yellow White Green Blue Violet Metal Flame
Lecture 2 362 January 16, 2019 Paradigm Shift: Development of Current Atomic Theory Spectroscopy and Energy Levels in Atoms OR, Show me the Electrons! Color Red Yellow White Green Blue Violet Metal Flame
Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling. Indian Institute of Technology Ropar
 Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling Specification of the ESR Particle detectors Re-injection to SIS Two 5 kv rf-cavities Fast Injection Schottky
Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling Specification of the ESR Particle detectors Re-injection to SIS Two 5 kv rf-cavities Fast Injection Schottky
Get Solution of These Packages & Learn by Video Tutorials on EXERCISE-1
 EXERCISE- SECTION (A) : PHOTON ON EMISSION FROM A SOURCE AND RADIATION A. A parallel beam of monochromatic light of wavelength 66 nm is incident on a totally reflecting plane mirror. The angle of incidence
EXERCISE- SECTION (A) : PHOTON ON EMISSION FROM A SOURCE AND RADIATION A. A parallel beam of monochromatic light of wavelength 66 nm is incident on a totally reflecting plane mirror. The angle of incidence
Worksheet 8 CHEM 121 Fall 2015
 Worksheet 8 CHEM 121 Fall 2015 Monday Name: Wednesday Name: Directions: As usual first 40 minutes without notes, with non-programmable calculator, with partner. Next 10 minutes with notes; last 25 minutes
Worksheet 8 CHEM 121 Fall 2015 Monday Name: Wednesday Name: Directions: As usual first 40 minutes without notes, with non-programmable calculator, with partner. Next 10 minutes with notes; last 25 minutes
Introduction to Ionizing Radiation
 Introduction to Ionizing Radiation Bob Curtis OSHA Salt Lake Technical Center Supplement to Lecture Outline V. 10.02 Basic Model of a Neutral Atom Electrons(-) orbiting nucleus of protons(+) and neutrons.
Introduction to Ionizing Radiation Bob Curtis OSHA Salt Lake Technical Center Supplement to Lecture Outline V. 10.02 Basic Model of a Neutral Atom Electrons(-) orbiting nucleus of protons(+) and neutrons.
