Orderly Arranged Fluorescence Dyes as a Highly Efficient Chemiluminescence Resonance Energy Transfer Probe for Peroxynitrite
|
|
|
- Jayson Barton
- 5 years ago
- Views:
Transcription
1 Supporting Information: Orderly Arranged Fluorescence Dyes as a Highly Efficient Chemiluminescence Resonance Energy Transfer Probe for Peroxynitrite Zhihua Wang, Xu Teng and Chao Lu* State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing , China. Fax/Tel.: E mail: luchao@mail.buct.edu.cn. This Supporting Information includes: (1) Figure S1 and Figure S2 schematic diagram of the CL setup; (2) Figure S3 powder XRD patterns of NO 3 LDHs, and SDS LDHs, TEM images of NO 3 LDHs and SDS LDHs, normalized fluorescence spectrum of sediments obtained by centrifuging calcein on the surface of SDS LDHs, and pictures of sediments obtained by centrifuging calcein on the surface of SDS LDHs under visible light and ultraviolet light at 254 nm; (3) Figure S4 structures of anionic surfactant SDS molecule and calcein fluorescence dye molecule; (4) Figure S5 the adsorption time of calcein on the surface of SDS LDHs; (5) Figure S6 fluorescence spectra of calcein@sds solution mixed with different concentrations of OH, and fluorescence spectra of the collapsed calcein@sds solution (a holding time of 60 min) mixed with different concentrations of OH; (6) Figure S7 CL intensity from the reaction of calcein@sds solution with different concentrations of ClO ; (7) Figure S8 normalized CL spectrum ONOO system mixed with calcein@sds solution, and normalized fluorescence spectrum of calcein solution; (8) Figure S9 normalized CL spectrum of ONOOH *, and absorption spectrum of calcein@sds solution; (9) Figure S10 CL intensity from the reaction of uranine@sds solution with different ROS. Inset: absorption spectrum of uranine@sds solution; (10) Figure S11 detection of ONOO from SIN 1; (11) Table S1 comparison of zeta potential values for SDS LDHs and calcein assembled SDS LDHs; (12) Table S2 optimum experimental conditions for the CL determination of 10 µm ONOO ; (11) Table S3 tolerance limit of various coexistent substances on the determination of 10 µm ONOO. S-1
2 Figure S1. Schematic diagram of the CL setup. a, 0.02 M H 2 O M HCl at 1.6 ml/min; b, 10 µm NaNO 2 at 1.6 ml/min; c, 0.1 M NaOH at 0.8 ml/min; d, calcein or calcein@sds solution; P, peristaltic pump; V, six-way injection valve; W, waste; PMT, photomultiplier tube. S-2
3 Figure S2. Schematic diagram of the CL setup. a, sample or spiked sample solution at 0.8 ml/min; b, solution; P, peristaltic pump; V, six-way injection valve; W, waste; PMT, photomultiplier tube. S-3
4 1 2 Figure S3. (A) Powder XRD patterns of (1) NO 3 LDHs, and (2) SDS LDHs. Inset: TEM images of NO 3 LDHs and SDS LDHs; (B) Normalized fluorescence spectrum of sediments obtained by centrifuging calcein on the surface of SDS LDHs. Inset: pictures of sediments obtained by centrifuging calcein on the surface of SDS LDHs under visible light (left) and ultraviolet light at 254 nm (right). S-4
5 Figure S4. Structures of anionic surfactant SDS molecule and calcein fluorescence dye molecule. S-5
6 Figure S5. Effect of the adsorption time of calcein on the surface of SDS LDHs on the CL intensity. The concentrations of calcein and SDS for each solution were 1.0 mm and 0.1 M, respectively. The concentration of ONOO was 10 µm. The colloidal solution was acidified with 1.0 M HCl. S-6
7 Figure S6. (A) Fluorescence spectra of solution mixed with different concentrations of OH. (a) 0 mm, (b) 1 mm, (c) 10 mm, (d) 100 mm; (B) fluorescence spectra of the collapsed calcein@sds solution (a holding time of 60 min) mixed with different concentrations of OH. (a) 0 mm, (b) 1 mm, (c) 10 mm, (d) 100 mm. S-7
8 Figure S7. CL intensity from the reaction of solution with different concentrations of ClO. The concentrations of calcein and SDS were 1.0 mm and 0.1 M, respectively. The colloidal solution was acidified with 1.0 M HCl. S-8
9 Figure S8. (A) Normalized CL spectrum ONOO system mixed with solution; (B) normalized fluorescence spectrum of calcein solution. The concentrations of calcein and SDS were 1.0 mm and 0.1 M, respectively. The concentration of ONOO was 0.01 M. The colloidal solution was acidified with 1.0 M HCl. S-9
10 Figure S9. (A) Normalized CL spectrum of ONOOH * ; (B) Absorption spectrum of calcein@sds solution. The concentrations of calcein and SDS were 1.0 mm and 0.1 M, respectively. The colloidal solution was acidified with 1.0 M HCl. ONOOH * was prepared by 0.07 M acidified H 2 O 2 and 0.1 M NaNO 2. S-10
11 Figure S10. CL intensity from the reaction of solution with different ROS. Inset: absorption spectrum of solution. The concentrations of 1 O 2, H 2 O 2, OH, O 2 and ClO were 1.0 mm. The concentration of ONOO was 0.5 mm. The concentrations of uranine and SDS were 1.0 mm and 0.1 M, respectively. The colloidal solution was acidified with 1.0 M HCl. S-11
12 Figure S11. Detection of ONOO from SIN 1. The concentration of SIN 1 was 5 mm. S-12
13 Table S1 Comparison of zeta potential values for SDS LDHs and calcein assembled SDS LDHs. SDS LDHs Sample calcein assembled SDS LDHs Zeta Potential (mv) mv mv S-13
14 Table S2 Optimum experimental conditions for the CL determination of 10 µm ONOO. Parameter Range studied Optimum Concentration of HCl (mm) Concentration of H 2 O 2 (mm) Concentration of NaOH (mm) Concentration of calcein (mm) Flow rate for HCl-H 2 O 2 or NaNO 2 (ml/min) Flow rate for NaOH (ml/min) S-14
15 Table S3 Tolerance limit of various coexistent substances on the determination of 10 µm ONOO. Tolerance (mm) Coexistent substances 10 Glucose (3 7 mm) a 5 K + (2 4 mm), Mg 2+ (1 3 mm), Ca 2+ (1 3 mm), Ac - (2 4 mm), bilirubin (14 22 µm) 1 S 2- - (30 60 µm), NO 3 (1 10 µm), SO 2-4 (1 4 µm), C 2 O 2-4 (1 4 µm), H 2 PO - 4 (10 50 µm) 0.5 Pb ( µm), HPO 4 (10 60 µm), I - (1 5 µm), Al 3+ (2 5 µm), Cr 3+ ( µm), glutamine (1 4 µm), homocysteine (3 6 µm), uric acid ( µm) 0.1 Fe 3+ (20 25 µm), Cu 2+ (2 4 µm), PO 3-4 (20 50 µm), ascorbic acid (50 80 µm), cysteine (7 50 µm) a Values in brackets means the common concentration ranges of the interfering compounds in mouse blood plasma. S-15
Natural montmorillonite nanosheet colloid-catalyzed hydrogen peroxide
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary Material (ESI) for RSC Advances Supplementary Information Natural montmorillonite
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supplementary Material (ESI) for RSC Advances Supplementary Information Natural montmorillonite
Supporting Information. Hexagonal Cobalt Oxyhydroxide-Carbon Dots Hybridized Surface: High Sensitive Fluorescence Turn-on Probe for Monitoring of
 Supporting Information Hexagonal Cobalt Oxyhydroxide-Carbon Dots Hybridized Surface: High Sensitive Fluorescence Turn-on Probe for Monitoring of Ascorbic Acid in Rat Brain Following Brain Ischemia Linbo
Supporting Information Hexagonal Cobalt Oxyhydroxide-Carbon Dots Hybridized Surface: High Sensitive Fluorescence Turn-on Probe for Monitoring of Ascorbic Acid in Rat Brain Following Brain Ischemia Linbo
Supporting Information
 Supporting Information Visual Determination of Cu 2+ through Copper-Catalysed in-situ Formation of Ag Nanoparticles Xun Yuan a and Yi Chen* a,b a Key Laboratory of Analytical Chemistry for Living Biosystems,
Supporting Information Visual Determination of Cu 2+ through Copper-Catalysed in-situ Formation of Ag Nanoparticles Xun Yuan a and Yi Chen* a,b a Key Laboratory of Analytical Chemistry for Living Biosystems,
mediated chemiluminescence for hydrogen peroxide and glucose
 Electronic Supplementary Information (ESI) for Chemical Communications Electronic Supplementary Information CoFe 2 O 4 magnetic nanoparticles as peroxidase mimic mediated chemiluminescence for hydrogen
Electronic Supplementary Information (ESI) for Chemical Communications Electronic Supplementary Information CoFe 2 O 4 magnetic nanoparticles as peroxidase mimic mediated chemiluminescence for hydrogen
Electrogenerated Upconverted Emission from Doped Organic Nanowires
 Electrogenerated Upconverted Emission from Doped Organic Nanowires Qing Li, Chuang Zhang, Jian Yao Zheng, Yong Sheng Zhao*, Jiannian Yao* Electronic Supplementary Information (ESI) 1 Experimental details
Electrogenerated Upconverted Emission from Doped Organic Nanowires Qing Li, Chuang Zhang, Jian Yao Zheng, Yong Sheng Zhao*, Jiannian Yao* Electronic Supplementary Information (ESI) 1 Experimental details
Supporting Information
 Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
Copyright WILEY-VCH Verlag GmbH & Co. KGaA, 69469 Weinheim, Germany, 2018. Supporting Information for Small, DOI: 10.1002/smll.201801523 Ultrasensitive Surface-Enhanced Raman Spectroscopy Detection Based
A Novel Detection Technique of Hydrazine Hydrate: Modality Change of Hydrogen-Bonding Induced Rapid and Ultrasensitive Colorimetric Assay
 A Novel Detection Technique of Hydrazine Hydrate: Modality Change of Hydrogen-Bonding Induced Rapid and Ultrasensitive Colorimetric Assay Zhenlu Zhao, a Kelong Ai, b Guo Zhang, b Ying Gao, a Xiuyun Yang,*
A Novel Detection Technique of Hydrazine Hydrate: Modality Change of Hydrogen-Bonding Induced Rapid and Ultrasensitive Colorimetric Assay Zhenlu Zhao, a Kelong Ai, b Guo Zhang, b Ying Gao, a Xiuyun Yang,*
Highly Sensitive Fluorescence Molecular Switch for the Ratio Monitoring of Trace Change of Mitochondrial Membrane Potential
 Highly Sensitive Fluorescence Molecular Switch for the Ratio Monitoring of Trace Change of Mitochondrial Membrane Potential Caixia Wang, Ge Wang, Xiang Li, Kui Wang, Jing Fan, Kai Jiang,, Yuming Guo, Hua
Highly Sensitive Fluorescence Molecular Switch for the Ratio Monitoring of Trace Change of Mitochondrial Membrane Potential Caixia Wang, Ge Wang, Xiang Li, Kui Wang, Jing Fan, Kai Jiang,, Yuming Guo, Hua
ISSN X CODEN (USA): PCHHAX. Determination of captopril with potassium permanganate chemiluminescence system*
 Available online at www.derpharmachemica.com ISSN 975-413X CODEN (USA): PCHHAX Der Pharma Chemica, 16, 8(1):289-293 (http://derpharmachemica.com/archive.html) Determination of captopril with potassium
Available online at www.derpharmachemica.com ISSN 975-413X CODEN (USA): PCHHAX Der Pharma Chemica, 16, 8(1):289-293 (http://derpharmachemica.com/archive.html) Determination of captopril with potassium
General Chemistry Experiment 3Lecture
 General Chemistry Experiment 3Lecture Part 1 Ionic Bonds Ionic Compounds Ions are positively and negatively charged atoms or groups of atoms that are each formed by the loss or gain of an electron.....
General Chemistry Experiment 3Lecture Part 1 Ionic Bonds Ionic Compounds Ions are positively and negatively charged atoms or groups of atoms that are each formed by the loss or gain of an electron.....
Supplementary Information for
 Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Visible-light Driven Plasmonic Photocatalyst Helical Chiral TiO 2 Nanofibers
 Visible-light Driven Plasmonic Photocatalyst Ag/AgCl @ Helical Chiral TiO 2 Nanofibers Dawei Wang, Yi Li*, Gianluca Li Puma, Chao Wang, Peifang Wang, Wenlong Zhang, and Qing Wang Fig. S1. The reactor of
Visible-light Driven Plasmonic Photocatalyst Ag/AgCl @ Helical Chiral TiO 2 Nanofibers Dawei Wang, Yi Li*, Gianluca Li Puma, Chao Wang, Peifang Wang, Wenlong Zhang, and Qing Wang Fig. S1. The reactor of
AP Chemistry Note Outline Chapter 4: Reactions and Reaction Stoichiometry:
 AP Chemistry Note Outline Chapter 4: Reactions and Reaction Stoichiometry: Water as a solvent Strong and Weak Electrolytes Solution Concentrations How to Make up a solution Types of Reactions Introduction
AP Chemistry Note Outline Chapter 4: Reactions and Reaction Stoichiometry: Water as a solvent Strong and Weak Electrolytes Solution Concentrations How to Make up a solution Types of Reactions Introduction
Supporting Information. Cells. Mian Wang, Yanglei Yuan, Hongmei Wang* and Zhaohai Qin*
 Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Fluorescent and Colorimetric Probe Containing Oxime-Ether for Pd 2+ in Pure
Electronic Supplementary Material (ESI) for Analyst. This journal is The Royal Society of Chemistry 2015 Supporting Information Fluorescent and Colorimetric Probe Containing Oxime-Ether for Pd 2+ in Pure
Fast and Slow Ligand Exchange at the Surface of Colloidal Gold Nanoparticles
 Fast and Slow Ligand Exchange at the Surface of Colloidal Gold Nanoparticles Rebecca Dinkel 1, Björn Braunschweig 1,2 * and Wolfgang Peukert 1,2 1 Institute of Particle Technology (LFG), Friedrich-Alexander
Fast and Slow Ligand Exchange at the Surface of Colloidal Gold Nanoparticles Rebecca Dinkel 1, Björn Braunschweig 1,2 * and Wolfgang Peukert 1,2 1 Institute of Particle Technology (LFG), Friedrich-Alexander
Supporting Information
 Supporting Information A Superlattice of Alternately Stacked Ni-Fe Hydroxide Nanosheets and Graphene for Efficient Splitting of Water Wei Ma,,, Renzhi Ma,, * Chengxiang Wang, Jianbo Liang, Xiaohe Liu,,
Supporting Information A Superlattice of Alternately Stacked Ni-Fe Hydroxide Nanosheets and Graphene for Efficient Splitting of Water Wei Ma,,, Renzhi Ma,, * Chengxiang Wang, Jianbo Liang, Xiaohe Liu,,
and their Maneuverable Application in Water Treatment
 Hierarchical Films of Layered Double Hydroxides by Using a Sol-Gel Process and their Maneuverable Application in Water Treatment Yufei Zhao, Shan He, Min Wei,* David G. Evans, Xue Duan State Key Laboratory
Hierarchical Films of Layered Double Hydroxides by Using a Sol-Gel Process and their Maneuverable Application in Water Treatment Yufei Zhao, Shan He, Min Wei,* David G. Evans, Xue Duan State Key Laboratory
graphene oxide nanocomposite with high performance for
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information One-pot in situ synthesis of CoFe 2 O 4 nanoparticles-reduced graphene
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2015 Supporting Information One-pot in situ synthesis of CoFe 2 O 4 nanoparticles-reduced graphene
A stable dual-functional system of visible-light-driven Ni(II) reduction to a nickel nanoparticle catalyst and robust in situ hydrogen production
 Electronic Supporting Information A stable dual-functional system of visible-light-driven Ni(II) reduction to a nickel nanoparticle catalyst and robust in situ hydrogen production Chuanjun Wang, a Shuang
Electronic Supporting Information A stable dual-functional system of visible-light-driven Ni(II) reduction to a nickel nanoparticle catalyst and robust in situ hydrogen production Chuanjun Wang, a Shuang
AP Chapter 4: Reactions in Aqueous Solution Name
 AP Chapter 4: Reactions in Aqueous Solution Name WarmUps (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. AP Chapter 4: Reactions in Aqueous Solution 2 WarmUps
AP Chapter 4: Reactions in Aqueous Solution Name WarmUps (Show your work for credit) Date 1. Date 2. Date 3. Date 4. Date 5. Date 6. Date 7. Date 8. AP Chapter 4: Reactions in Aqueous Solution 2 WarmUps
Practice Exam Topic 9: Oxidation & Reduction
 Name Practice Exam Topic 9: Oxidation & Reduction 1. What are the oxidation numbers of the elements in sulfuric acid, H 2 SO 4? Hydrogen Sulfur Oxygen A. +1 +6 2 B. +1 +4 2 C. +2 +1 +4 D. +2 +6 8 2. Consider
Name Practice Exam Topic 9: Oxidation & Reduction 1. What are the oxidation numbers of the elements in sulfuric acid, H 2 SO 4? Hydrogen Sulfur Oxygen A. +1 +6 2 B. +1 +4 2 C. +2 +1 +4 D. +2 +6 8 2. Consider
Supporting Information. for
 Supporting Information for Near-Infrared Fluorescent Turn-On Probe with a Remarkable Large Stokes Shift for Imaging Selenocysteine in Living Cells and Animals Weiyong Feng, Meixing Li, Yao Sun, and Guoqiang
Supporting Information for Near-Infrared Fluorescent Turn-On Probe with a Remarkable Large Stokes Shift for Imaging Selenocysteine in Living Cells and Animals Weiyong Feng, Meixing Li, Yao Sun, and Guoqiang
State Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical
 Electronic Supplementary Information Evidence for inhibition of HIF-1α Prolyl hydroxylase 3 activity by four biological active tetraazamacrocycles Jing Cao, Zhirong Geng, Xiaoyan Ma, Jinghan Wen, Yuxin
Electronic Supplementary Information Evidence for inhibition of HIF-1α Prolyl hydroxylase 3 activity by four biological active tetraazamacrocycles Jing Cao, Zhirong Geng, Xiaoyan Ma, Jinghan Wen, Yuxin
Homework #3 Chapter 4 Types of Chemical Reactions and Solution Stoichiometry
 Homework #3 Chapter 4 Types of Chemical Reactions and Solution Stoichiometry 13. Determine the concentrations of the solutions Solution A 4 particles 1.0 L Solution B 6 paticles 4.0 L Solution C 4 particles
Homework #3 Chapter 4 Types of Chemical Reactions and Solution Stoichiometry 13. Determine the concentrations of the solutions Solution A 4 particles 1.0 L Solution B 6 paticles 4.0 L Solution C 4 particles
ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. Sunday, August 18, 13
 ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. A solution is a homogenous mixture of 2 or more substances at the molecular level The solute(s) is(are)
ed. Brad Collins Aqueous Chemistry Chapter 5 Some images copyright The McGraw-Hill Companies, Inc. A solution is a homogenous mixture of 2 or more substances at the molecular level The solute(s) is(are)
Supporting Information. Synthesis and Upconversion Luminescence of BaY 2
 Supporting Information Synthesis and Upconversion Luminescence of BaY 2 F 8 :Yb 3+ /Er 3+ Nanobelts 5 Guofeng Wang, Qing Peng, and Yadong Li* Department of Chemistry and State Key Laboratory of New Ceramics
Supporting Information Synthesis and Upconversion Luminescence of BaY 2 F 8 :Yb 3+ /Er 3+ Nanobelts 5 Guofeng Wang, Qing Peng, and Yadong Li* Department of Chemistry and State Key Laboratory of New Ceramics
Graphene Sponge for Efficient and Repeatable Adsorption and Desorption of. Water Contaminations
 Supporting Information for Graphene Sponge for Efficient and Repeatable Adsorption and Desorption of Water Contaminations Jinping Zhao, Wencai Ren* and Hui-Ming Cheng Shenyang National Laboratory for Materials
Supporting Information for Graphene Sponge for Efficient and Repeatable Adsorption and Desorption of Water Contaminations Jinping Zhao, Wencai Ren* and Hui-Ming Cheng Shenyang National Laboratory for Materials
A reaction in which a solid forms is called a precipitation reaction. Solid = precipitate
 Chapter 7 Reactions in Aqueous Solutions 1 Section 7.1 Predicting Whether a Reaction Will Occur Four Driving Forces Favor Chemical Change 1. Formation of a solid 2. Formation of water 3. Transfer of electrons
Chapter 7 Reactions in Aqueous Solutions 1 Section 7.1 Predicting Whether a Reaction Will Occur Four Driving Forces Favor Chemical Change 1. Formation of a solid 2. Formation of water 3. Transfer of electrons
CONCENTRATION UNITS 0.800? concentration? What is the molar concentration of mercury? solution contain? 0.150? CHANGING CONCENTRATION UNITS
 CONCENTRATION UNITS 1. How many grams of CuSO 4 are required to make 650. ml of a 0.115 M solution? 2. How many grams of NaCl are required to prepare 250. ml of a 0.241 M solution? 3. How many grams of
CONCENTRATION UNITS 1. How many grams of CuSO 4 are required to make 650. ml of a 0.115 M solution? 2. How many grams of NaCl are required to prepare 250. ml of a 0.241 M solution? 3. How many grams of
Acetylene hydrochlorination over 13X zeolite. catalyst at high temperature
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Acetylene hydrochlorination over 13X zeolite catalyst at high temperature Zhijia Song, ab
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2016 Acetylene hydrochlorination over 13X zeolite catalyst at high temperature Zhijia Song, ab
Unit 5 Part 2: Redox Reactions and Electrochemistry
 Unit 5 Part 2: Redox Reactions and Electrochemistry Oxidation Numbers Oxidizing and Reducing Agents Balancing Redox Reactions Acidic solutions Basic solutions Galvanic Cells Nernst Equation This reaction
Unit 5 Part 2: Redox Reactions and Electrochemistry Oxidation Numbers Oxidizing and Reducing Agents Balancing Redox Reactions Acidic solutions Basic solutions Galvanic Cells Nernst Equation This reaction
Practice Final CH142, Spring 2012
 Practice Final CH142, Spring 2012 First here are a group of practice problems on Latimer Diagrams: 1. The Latimer diagram for nitrogen oxides in given below. Is NO stable with respect to disproportionation
Practice Final CH142, Spring 2012 First here are a group of practice problems on Latimer Diagrams: 1. The Latimer diagram for nitrogen oxides in given below. Is NO stable with respect to disproportionation
State Key Laboratory of Chemical Resource Engineering, Beijing University of Chemical Technology, Beijing , China
 Electronic Supplementary Material A Co-N/C hollow-sphere electrocatalyst derived from a metanilic CoAl layered double hydroxide for the oxygen reduction reaction, and its active sites in various ph media
Electronic Supplementary Material A Co-N/C hollow-sphere electrocatalyst derived from a metanilic CoAl layered double hydroxide for the oxygen reduction reaction, and its active sites in various ph media
2. In each of the following pairs of reactions, which would have the faster reaction rate?
 CHEMISTRY 12 REACTION RATES WORKSHEET 1. The following data were collected for the reaction: Zn (s) + 2HCl (aq) H 2(g) + ZnCl 2(aq) in which zinc metal was reacted with 0.200 M HCl (aq) : Time (seconds)
CHEMISTRY 12 REACTION RATES WORKSHEET 1. The following data were collected for the reaction: Zn (s) + 2HCl (aq) H 2(g) + ZnCl 2(aq) in which zinc metal was reacted with 0.200 M HCl (aq) : Time (seconds)
Novel dendrimer-like magnetic bio-sorbent based on modified orange peel. waste: adsorption-reduction behavior of arsenic
 Supplementary Information: Novel dendrimer-like magnetic bio-sorbent based on modified orange peel waste: adsorption-reduction behavior of arsenic Fanqing Meng1, Bowen Yang1, Baodong Wang 2, Shibo Duan1,
Supplementary Information: Novel dendrimer-like magnetic bio-sorbent based on modified orange peel waste: adsorption-reduction behavior of arsenic Fanqing Meng1, Bowen Yang1, Baodong Wang 2, Shibo Duan1,
M11/4/CHEMI/SPM/ENG/TZ2/XX CHEMISTRY STANDARD LEVEL PAPER 1. Monday 9 May 2011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES
 M11/4/CHEMI/SPM/ENG/TZ/XX 116116 CHEMISTRY STANDARD LEVEL PAPER 1 Monday 9 May 011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
M11/4/CHEMI/SPM/ENG/TZ/XX 116116 CHEMISTRY STANDARD LEVEL PAPER 1 Monday 9 May 011 (afternoon) 45 minutes INSTRUCTIONS TO CANDIDATES Do not open this examination paper until instructed to do so. Answer
ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation
 S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
Introduction to biochemical practicals. Vladimíra Kvasnicová
 Introduction to biochemical practicals Vladimíra Kvasnicová arrangement of practicals laboratory safety regulations laboratory equipment dealing with automatic pipette instructions: http://vyuka.lf3.cuni.cz/
Introduction to biochemical practicals Vladimíra Kvasnicová arrangement of practicals laboratory safety regulations laboratory equipment dealing with automatic pipette instructions: http://vyuka.lf3.cuni.cz/
Unit 8: Redox and Electrochemistry
 May 20, 2014 Unit 8: Redox and Electrochemistry http://www.firefly.org/firefly-pictures.html Oxidation Number numbers assigned to atoms that allow us to keep track of electrons. Rule #1: Oxidation number
May 20, 2014 Unit 8: Redox and Electrochemistry http://www.firefly.org/firefly-pictures.html Oxidation Number numbers assigned to atoms that allow us to keep track of electrons. Rule #1: Oxidation number
Reaction Kinetics Multiple Choice
 Reaction Kinetics Multiple Choice January 1999 1. Consider the reaction: Ca (s) + 2H 2 O (l) Ca(OH) 2 (aq) + H 2 (g) At a certain temperature, 2.50 g Ca reacts completely in 30.0 seconds. The rate of consumption
Reaction Kinetics Multiple Choice January 1999 1. Consider the reaction: Ca (s) + 2H 2 O (l) Ca(OH) 2 (aq) + H 2 (g) At a certain temperature, 2.50 g Ca reacts completely in 30.0 seconds. The rate of consumption
Specifically colorimetric recognition of calcium, strontium, barium. ions using 2-mercaptosuccinic acid-functionalized gold nanoparticles
 Electronic Supporting Information (ESI) for Specifically colorimetric recognition of calcium, strontium, barium ions using 2-mercaptosuccinic acid-functionalized gold nanoparticles and its use in reliable
Electronic Supporting Information (ESI) for Specifically colorimetric recognition of calcium, strontium, barium ions using 2-mercaptosuccinic acid-functionalized gold nanoparticles and its use in reliable
Cu-Creatinine- Metol system
 Quantification of Creatinine in Human Serum using Metol as a Chromogenic Probe Materials and methods 6.1. Reagents 6.1.1. N-methyl-p-aminophenol sulfate N-methyl-p-aminophenol sulfate also denoted as Metol
Quantification of Creatinine in Human Serum using Metol as a Chromogenic Probe Materials and methods 6.1. Reagents 6.1.1. N-methyl-p-aminophenol sulfate N-methyl-p-aminophenol sulfate also denoted as Metol
Study on the interaction behavior of catalase with. cephalosporins by chemiluminescence with flow injection
 tudy on the interaction behavior of catalase with cephalosporins by chemiluminescence with flow injection analysis Donghua Chen a, Zhuming Wang a,b, Yun Zhang a, Xunyu Xiong a and Zhenghua ong * a a Key
tudy on the interaction behavior of catalase with cephalosporins by chemiluminescence with flow injection analysis Donghua Chen a, Zhuming Wang a,b, Yun Zhang a, Xunyu Xiong a and Zhenghua ong * a a Key
Supplementary Information:
 Supplementary Information: One-Step and Rapid Synthesis of Clean and Monodisperse Dendritic Pt Nanoparticles and Their High Performance Toward Methanol Oxidation and p-nitrophenol Reduction Jun Wang, Xin-Bo
Supplementary Information: One-Step and Rapid Synthesis of Clean and Monodisperse Dendritic Pt Nanoparticles and Their High Performance Toward Methanol Oxidation and p-nitrophenol Reduction Jun Wang, Xin-Bo
Chapter 9. Chemical Names and Formulas
 Chapter 9 Chemical Names and Formulas 9.1 - Naming Ions Monatomic ions: Single atom with a positive or negative charge resulting from the loss or gain of one or more valence electrons. - Cations: Groups
Chapter 9 Chemical Names and Formulas 9.1 - Naming Ions Monatomic ions: Single atom with a positive or negative charge resulting from the loss or gain of one or more valence electrons. - Cations: Groups
Reactions in Aqueous Solution
 1 Reactions in Aqueous Solution Chapter 4 For test 3: Sections 3.7 and 4.1 to 4.5 Copyright The McGrawHill Companies, Inc. Permission required for reproduction or display. 2 A solution is a homogenous
1 Reactions in Aqueous Solution Chapter 4 For test 3: Sections 3.7 and 4.1 to 4.5 Copyright The McGrawHill Companies, Inc. Permission required for reproduction or display. 2 A solution is a homogenous
Electronic Supplementary Information (ESI) Tunable Phase and Visible-Light Photocatalytic Activity
 Electronic Supplementary Information (ESI) Metallic-Zinc Assistant Synthesis of Ti 3+ Self-Doped TiO 2 with Tunable Phase and Visible-Light Photocatalytic Activity Zhaoke Zheng, a Baibiao Huang,* a Xiaodong
Electronic Supplementary Information (ESI) Metallic-Zinc Assistant Synthesis of Ti 3+ Self-Doped TiO 2 with Tunable Phase and Visible-Light Photocatalytic Activity Zhaoke Zheng, a Baibiao Huang,* a Xiaodong
Chemistry 51 Chapter 8 TYPES OF SOLUTIONS. Some Examples of Solutions. Type Example Solute Solvent Gas in gas Air Oxygen (gas) Nitrogen (gas)
 TYPES OF SOLUTIONS A solution is a homogeneous mixture of two substances: a solute and a solvent. Solute: substance being dissolved; present in lesser amount. Solvent: substance doing the dissolving; present
TYPES OF SOLUTIONS A solution is a homogeneous mixture of two substances: a solute and a solvent. Solute: substance being dissolved; present in lesser amount. Solvent: substance doing the dissolving; present
DOUBLE DISPLACEMENT REACTIONS. Double your pleasure, double your fun
 DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
DOUBLE DISPLACEMENT REACTIONS Double your pleasure, double your fun Industrial processes produce unwanted by-products. Dissolved toxic metal ions-copper, mercury, and cadmium-are common leftovers in the
Electronic Supplementary Information. for. Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic. acid modified gold electrode
 Electronic Supplementary Information for Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic acid modified gold electrode Guangming Liu,* a Jingjing Li, b Li Wang b, Nana Zong b,
Electronic Supplementary Information for Discrimination of dopamine from ascorbic acid and uric acid on thioglycolic acid modified gold electrode Guangming Liu,* a Jingjing Li, b Li Wang b, Nana Zong b,
Supporting Information
 Supporting Information Robust Co-Catalytic Performance of Nanodiamonds Loaded on WO 3 for the Decomposition of Volatile Organic Compounds under Visible Light Hyoung il Kim, a Hee-na Kim, a Seunghyun Weon,
Supporting Information Robust Co-Catalytic Performance of Nanodiamonds Loaded on WO 3 for the Decomposition of Volatile Organic Compounds under Visible Light Hyoung il Kim, a Hee-na Kim, a Seunghyun Weon,
MOF-76: From Luminescent Probe to Highly Efficient U VI Sorption Material
 MOF-76: From Luminescent Probe to Highly Efficient U VI Sorption Material Weiting Yang, a Zhi-Qiang Bai, b Wei-Qun Shi*, b Li-Yong Yuan, b Tao Tian, a Zhi-Fang Chai*, c Hao Wang, a and Zhong-Ming Sun*
MOF-76: From Luminescent Probe to Highly Efficient U VI Sorption Material Weiting Yang, a Zhi-Qiang Bai, b Wei-Qun Shi*, b Li-Yong Yuan, b Tao Tian, a Zhi-Fang Chai*, c Hao Wang, a and Zhong-Ming Sun*
Supporting Information. Synthesis of Mg/ Al Layered Double Hydroxides for Adsorptive Removal of. Fluoride from Water: A Mechanistic and Kinetic Study
 Supporting Information Synthesis of Mg/ Al Layered Double Hydroxides for Adsorptive Removal of Fluoride from Water: A Mechanistic and Kinetic Study Gautam Kumar Sarma and Md. Harunar Rashid* Department
Supporting Information Synthesis of Mg/ Al Layered Double Hydroxides for Adsorptive Removal of Fluoride from Water: A Mechanistic and Kinetic Study Gautam Kumar Sarma and Md. Harunar Rashid* Department
Supporting Information
 Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Supporting Information A Low-Temperature Solid-Phase Method to Synthesize Highly Fluorescent Carbon Nitride Dots with Tunable Emission Juan Zhou, Yong Yang, and Chun-yang Zhang* Single-Molecule Detection
Chapter 9: Solutions
 9.1 Mixtures and Solutions Chapter 9: Solutions Heterogeneous mixtures are those in which the mixing is not uniform and have regions of different composition. Homogeneous mixtures are those in which the
9.1 Mixtures and Solutions Chapter 9: Solutions Heterogeneous mixtures are those in which the mixing is not uniform and have regions of different composition. Homogeneous mixtures are those in which the
Electronic Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information 3D ordered macroporous TiO 2 -supported
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supporting Information 3D ordered macroporous TiO 2 -supported
#35 balance the following chemical equations a) SiI 4(s) + 2Mg (s)! Si (s) + 2MgI 2(s) Si = 1 I = 4 Mg = 1 2. Si = 1 I = 2 4 Mg = 1 2 (1,2,1,2)
 #35 balance the following chemical equations a) SiI 4(s) 2Mg (s)! Si (s) 2MgI 2(s) Si = 1 I = 4 Mg = 1 2 Si = 1 I = 2 4 Mg = 1 2 SiI 4(s) 2Mg (s)! Si (s) 2MgI 2(s) (1,2,1,2) b) MnO 2(s) 2Mg (s)! Mn (s)
#35 balance the following chemical equations a) SiI 4(s) 2Mg (s)! Si (s) 2MgI 2(s) Si = 1 I = 4 Mg = 1 2 Si = 1 I = 2 4 Mg = 1 2 SiI 4(s) 2Mg (s)! Si (s) 2MgI 2(s) (1,2,1,2) b) MnO 2(s) 2Mg (s)! Mn (s)
Continuous Flow Analysis Methods David Davey University of South Australia. 21 February
 Continuous Flow Analysis Methods David Davey University of South Australia 21 February 2012 1 Flow Analysis Methods 1950s-60s Segmented Flow Methods Clinical Analysis Skeggs 1970s Flow Injection Analysis
Continuous Flow Analysis Methods David Davey University of South Australia 21 February 2012 1 Flow Analysis Methods 1950s-60s Segmented Flow Methods Clinical Analysis Skeggs 1970s Flow Injection Analysis
11.3 Reactions in Aqueous Essential Understanding Reactions that occur in aqueous solutions are double-replacement
 13. Is the following sentence true or false? Hydrocarbons, compounds of hydrogen and carbon, are often the reactants in combustion reactions. 14. Circle the letter of each compound that can be produced
13. Is the following sentence true or false? Hydrocarbons, compounds of hydrogen and carbon, are often the reactants in combustion reactions. 14. Circle the letter of each compound that can be produced
Synthesis of 2 ) Structures by Small Molecule-Assisted Nucleation for Plasmon-Enhanced Photocatalytic Activity
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Synthesis of Au@UiO-66(NH 2 ) Structures by Small Molecule-Assisted
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Synthesis of Au@UiO-66(NH 2 ) Structures by Small Molecule-Assisted
Name: Final Test. / n 2 Rydberg constant, R H. = n h ν Hydrogen energy, E n
 200 points Chemistry 121A Dr. Jay H. Baltisberger December 13, 1999 SHOW ALL CALCULATIONS & USE PROPER SIGNIFICANT FIGURES AND UNITS Avogadro s number, N A = 6.02x10 23 Planck s constant, h = 6.626x10
200 points Chemistry 121A Dr. Jay H. Baltisberger December 13, 1999 SHOW ALL CALCULATIONS & USE PROPER SIGNIFICANT FIGURES AND UNITS Avogadro s number, N A = 6.02x10 23 Planck s constant, h = 6.626x10
Novel fungus-titanate bio-nano composites as high performance. absorbents for the efficient removal of radioactive ions from.
 This journal is The Royal Society of Chemistry 0 Electronic Supplementary Information For Novel fungus-titanate bio-nano composites as high performance absorbents for the efficient removal of radioactive
This journal is The Royal Society of Chemistry 0 Electronic Supplementary Information For Novel fungus-titanate bio-nano composites as high performance absorbents for the efficient removal of radioactive
Lecture Presentation. Chapter 16. Aqueous Ionic Equilibrium. Sherril Soman Grand Valley State University Pearson Education, Inc.
 Lecture Presentation Chapter 16 Aqueous Ionic Equilibrium Sherril Soman Grand Valley State University The Danger of Antifreeze Each year, thousands of pets and wildlife species die from consuming antifreeze.
Lecture Presentation Chapter 16 Aqueous Ionic Equilibrium Sherril Soman Grand Valley State University The Danger of Antifreeze Each year, thousands of pets and wildlife species die from consuming antifreeze.
1. Forming a Precipitate 2. Solubility Product Constant (One Source of Ions)
 Chemistry 12 Solubility Equilibrium II Name: Date: Block: 1. Forming a Precipitate 2. Solubility Product Constant (One Source of Ions) Forming a Precipitate Example: A solution may contain the ions Ca
Chemistry 12 Solubility Equilibrium II Name: Date: Block: 1. Forming a Precipitate 2. Solubility Product Constant (One Source of Ions) Forming a Precipitate Example: A solution may contain the ions Ca
Redox and Electrochemistry
 Redox and Electrochemistry 1 Electrochemistry in Action! 2 Rules for Assigning Oxidation Numbers The oxidation number of any uncombined element is 0. The oxidation number of a monatomic ion equals the
Redox and Electrochemistry 1 Electrochemistry in Action! 2 Rules for Assigning Oxidation Numbers The oxidation number of any uncombined element is 0. The oxidation number of a monatomic ion equals the
Flow injection chemiluminescence determination of epinephrine using epinephrine-imprinted polymer as recognition material
 Analytica Chimica Acta 489 (2003) 183 189 Flow injection chemiluminescence determination of epinephrine using epinephrine-imprinted polymer as recognition material Jianxiu Du, Lihua Shen, Jiuru Lu Department
Analytica Chimica Acta 489 (2003) 183 189 Flow injection chemiluminescence determination of epinephrine using epinephrine-imprinted polymer as recognition material Jianxiu Du, Lihua Shen, Jiuru Lu Department
Electronic Supplementary Information
 Electronic Supplementary Information Formation of MS-Ag and MS (M=Pb, Cd, Zn) nanotubes via microwave-assisted cation exchange and their enhanced photocatalytic activities Yanrong Wang, a Wenlong Yang,
Electronic Supplementary Information Formation of MS-Ag and MS (M=Pb, Cd, Zn) nanotubes via microwave-assisted cation exchange and their enhanced photocatalytic activities Yanrong Wang, a Wenlong Yang,
Electrolytes do conduct electricity, in proportion to the concentrations of their ions in solution.
 Chapter 4 (Hill/Petrucci/McCreary/Perry Chemical Reactions in Aqueous Solutions This chapter deals with reactions that occur in aqueous solution these solutions all use water as the solvent. We will look
Chapter 4 (Hill/Petrucci/McCreary/Perry Chemical Reactions in Aqueous Solutions This chapter deals with reactions that occur in aqueous solution these solutions all use water as the solvent. We will look
Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly
 SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
SUPPORTING INFORMATION Highly Sensitive and Selective Colorimetric Visualization of Streptomycin in Raw Milk Using Au Nanoparticles Supramolecular Assembly Jiayu Sun, Jiechao Ge, Weimin Liu, Zhiyuan Fan,
Australian Institute of Innovative Materials, University of Wollongong, Australia.
 Photocatalytic Degradation of Methyl Orange by CeO 2 and Fe-doped CeO 2 Films under Visible Light Irradiation D. Channei a, B. Inceesungvorn a, N. Wetchakun b, S. Ukritnukun c, A. Nattestad d, J. Chen
Photocatalytic Degradation of Methyl Orange by CeO 2 and Fe-doped CeO 2 Films under Visible Light Irradiation D. Channei a, B. Inceesungvorn a, N. Wetchakun b, S. Ukritnukun c, A. Nattestad d, J. Chen
Amphiphilic diselenide-containing supramolecular polymers
 Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Amphiphilic diselenide-containing supramolecular polymers Xinxin Tan, Liulin Yang, Zehuan
Electronic Supplementary Material (ESI) for Polymer Chemistry. This journal is The Royal Society of Chemistry 2014 Amphiphilic diselenide-containing supramolecular polymers Xinxin Tan, Liulin Yang, Zehuan
Chapter 4. Aqueous Reactions and Solution Stoichiometry
 Sample Exercise 4.1 (p. 127) The diagram below represents an aqueous solution of one of the following compounds: MgCl 2, KCl, or K 2 SO 4. Which solution does it best represent? Practice Exercise 1 (4.1)
Sample Exercise 4.1 (p. 127) The diagram below represents an aqueous solution of one of the following compounds: MgCl 2, KCl, or K 2 SO 4. Which solution does it best represent? Practice Exercise 1 (4.1)
Reactions in Aqueous Solutions
 Chapter 4 Reactions in Aqueous Solutions Some typical kinds of chemical reactions: 1. Precipitation reactions: the formation of a salt of lower solubility causes the precipitation to occur. precipr 2.
Chapter 4 Reactions in Aqueous Solutions Some typical kinds of chemical reactions: 1. Precipitation reactions: the formation of a salt of lower solubility causes the precipitation to occur. precipr 2.
Supporting Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Adding refractory 5d transition metal W into PtCo
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2018 Supporting Information Adding refractory 5d transition metal W into PtCo
Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals.
 Chemistry 11 Notes on Chemical Reactions Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred:
Chemistry 11 Notes on Chemical Reactions Chemical Reaction Defn: Chemical Reaction: when starting chemical species form different chemicals. Evidence to indicate that a chemical reaction has occurred:
Supporting Information. Chemiluminescence of black phosphorus quantum dots
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Chemiluminescence of black phosphorus quantum dots induced by hypochlorite
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Supporting Information Chemiluminescence of black phosphorus quantum dots induced by hypochlorite
Chapter 9 Practice Worksheet: Reactions in Aqueous Solutions
 Chapter 9 Practice Worksheet: Reactions in Aqueous Solutions 1. The compound H 2 S is classified as a weak electrolyte. Describe/draw how it reacts when placed in water. Completely dissociates in water.
Chapter 9 Practice Worksheet: Reactions in Aqueous Solutions 1. The compound H 2 S is classified as a weak electrolyte. Describe/draw how it reacts when placed in water. Completely dissociates in water.
A Temperature Sensor Based on CdTe Quantum Dots/Layered Double. Hydroxide Ultrathin Films via Layer-by-Layer Assembly
 # Supplementary Material (ESI) for Chemical Communications # This journal is The Royal Society of Chemistry 2012 A Temperature Sensor Based on CdTe Quantum Dots/Layered Double Hydroxide Ultrathin Films
# Supplementary Material (ESI) for Chemical Communications # This journal is The Royal Society of Chemistry 2012 A Temperature Sensor Based on CdTe Quantum Dots/Layered Double Hydroxide Ultrathin Films
Acid / Base Properties of Salts
 Acid / Base Properties of Salts n Soluble ionic salts produce may produce neutral, acidic, or basic solutions depending on the acidbase properties of the individual ions. n Consider the salt sodium nitrate,
Acid / Base Properties of Salts n Soluble ionic salts produce may produce neutral, acidic, or basic solutions depending on the acidbase properties of the individual ions. n Consider the salt sodium nitrate,
Electrolytes vs Non electrolytes Electrolyte Non electrolyte A substance, that A substance that when dissolved in
 Definition Why Electrolytes vs Non electrolytes Electrolyte Non electrolyte A substance, that A substance that when dissolved in when dissolved in water DOES NOT water, conducts conduct electricity. electricity.
Definition Why Electrolytes vs Non electrolytes Electrolyte Non electrolyte A substance, that A substance that when dissolved in when dissolved in water DOES NOT water, conducts conduct electricity. electricity.
A solution is a homogeneous mixture of two or more substances.
 UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
UNIT (5) SOLUTIONS A solution is a homogeneous mixture of two or more substances. 5.1 Terminology Solute and Solvent A simple solution has two components, a solute, and a solvent. The substance in smaller
Solutions CHAPTER OUTLINE
 Chapter 8A Solutions 1 CHAPTER OUTLINE Type of Solutions Electrolytes & Nonelectrolytes Equivalents of Electrolytes Solubility & Saturation Soluble & Insoluble Salts Formation of a Solid Precipitation
Chapter 8A Solutions 1 CHAPTER OUTLINE Type of Solutions Electrolytes & Nonelectrolytes Equivalents of Electrolytes Solubility & Saturation Soluble & Insoluble Salts Formation of a Solid Precipitation
Reactions in Aqueous Solution
 Reading Assignments: Reactions in Aqueous Solution Chapter 4 Chapter 4 in R. Chang, Chemistry, 9 th Ed., McGraw-Hill, 2006. or previous editions. Or related topics in other textbooks. Consultation outside
Reading Assignments: Reactions in Aqueous Solution Chapter 4 Chapter 4 in R. Chang, Chemistry, 9 th Ed., McGraw-Hill, 2006. or previous editions. Or related topics in other textbooks. Consultation outside
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Experimental section Materials: Tannic acid (TA), silver nitrate
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information Experimental section Materials: Tannic acid (TA), silver nitrate
Supplementary Information for Scientific Reports. Synergistic Effect between Ultra-Small Nickel Hydroxide
 Supplementary Information for Scientific Reports Synergistic Effect between Ultra-Small Nickel Hydroxide Nanoparticles and Reduced Graphene Oxide Sheets for the Application in High-Performance Asymmetric
Supplementary Information for Scientific Reports Synergistic Effect between Ultra-Small Nickel Hydroxide Nanoparticles and Reduced Graphene Oxide Sheets for the Application in High-Performance Asymmetric
Supporting Information. Copyright Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, 2008
 Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2008 Highly Sensitive and Fast Responsive Fluorescence Turn-on Chemodosimeter for Cu 2+ and its Application in Live Cell
Supporting Information Copyright Wiley-VCH Verlag GmbH & Co. KGaA, 69451 Weinheim, 2008 Highly Sensitive and Fast Responsive Fluorescence Turn-on Chemodosimeter for Cu 2+ and its Application in Live Cell
A ratiometric luminescent sensing of Ag + ion via in situ formation of coordination polymers
 Electronic Supplementary Information (ESI) A ratiometric luminescent sensing of Ag + ion via in situ formation of coordination polymers Dong-Hua Li, a Jiang-Shan Shen, b Na Chen, a Yi-Bin Ruan a and Yun-Bao
Electronic Supplementary Information (ESI) A ratiometric luminescent sensing of Ag + ion via in situ formation of coordination polymers Dong-Hua Li, a Jiang-Shan Shen, b Na Chen, a Yi-Bin Ruan a and Yun-Bao
Preparation of sludge derived carbon with Fenton and NaClO activated and the application on the odor abatement
 Preparation of sludge derived carbon with Fenton and NaClO activated and the application on the odor abatement Lou Ziyang Dr. Associate Prof. 2015-07-02 Content Background SBC preparation Characterization
Preparation of sludge derived carbon with Fenton and NaClO activated and the application on the odor abatement Lou Ziyang Dr. Associate Prof. 2015-07-02 Content Background SBC preparation Characterization
CHAPTER 4. SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL
 93 CHAPTER 4 SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL 4.1 INTRODUCTION TiO 2 -derived nanotubes are expected to be applicable for several applications,
93 CHAPTER 4 SYNTHESIS, CHARACTERIZATION OF TiO 2 NANOTUBES AND THEIR APPLICATION IN DYE SENSITIZED SOLAR CELL 4.1 INTRODUCTION TiO 2 -derived nanotubes are expected to be applicable for several applications,
Supplementary Information
 Supplementary Information Visible Photocatalytic Water Splitting and Photocatalytic Two-Electron Oxygen Formation over Cu and Fe Doped g-c 3 N 4 Zhen Li a,b, Chao Kong a,b, Gongxuan Lu a* a State Key Laboratory
Supplementary Information Visible Photocatalytic Water Splitting and Photocatalytic Two-Electron Oxygen Formation over Cu and Fe Doped g-c 3 N 4 Zhen Li a,b, Chao Kong a,b, Gongxuan Lu a* a State Key Laboratory
Note Taking Guide: Episode electrochemistry the study of -related applications of - reactions
 Note Taking Guide: Episode 1401 Name electrochemistry the study of related applications of reactions oxidation numbers numbers assigned to the in a chemical that give the charge of the. In ionic compounds
Note Taking Guide: Episode 1401 Name electrochemistry the study of related applications of reactions oxidation numbers numbers assigned to the in a chemical that give the charge of the. In ionic compounds
Supporting Information
 Electronic Supplementary Material (ESI) for Analytical Methods. This journal is The Royal Society of Chemistry 2014 Supporting Information A rhodamine hydrazide-based fluorescent probe for sensitive and
Electronic Supplementary Material (ESI) for Analytical Methods. This journal is The Royal Society of Chemistry 2014 Supporting Information A rhodamine hydrazide-based fluorescent probe for sensitive and
Reactions in Aqueous Solutions
 Reactions in Aqueous Solutions Chapter 4 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. A solution is a homogenous mixture of 2 or more substances. The solute
Reactions in Aqueous Solutions Chapter 4 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. A solution is a homogenous mixture of 2 or more substances. The solute
CHAPTER - 3 ANALYTICAL PROFILE. 3.1 Estimation of Drug in Pharmaceutical Formulation Estimation of Drugs
 CHAPTER - 3 ANALYTICAL PROFILE 3.1 Estimation of Drug in Pharmaceutical Formulation 3.1.1 Estimation of Drugs ANALYTICAL PROFILE 84 3.1 ESTIMATION OF DRUG IN PHARMACEUTICAL FORMULATION. Agrawal A et al
CHAPTER - 3 ANALYTICAL PROFILE 3.1 Estimation of Drug in Pharmaceutical Formulation 3.1.1 Estimation of Drugs ANALYTICAL PROFILE 84 3.1 ESTIMATION OF DRUG IN PHARMACEUTICAL FORMULATION. Agrawal A et al
UNIT 10 Reduction/Oxidation Reactions & Electrochemistry NOTES
 Name Period CRHS Academic Chemistry UNIT 10 Reduction/Oxidation Reactions & Electrochemistry NOTES Quiz Date Lab Dates Exam Date Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry
Name Period CRHS Academic Chemistry UNIT 10 Reduction/Oxidation Reactions & Electrochemistry NOTES Quiz Date Lab Dates Exam Date Notes, Homework, Exam Reviews and Their KEYS located on CRHS Academic Chemistry
UNIT IV PPT #3 Ka and Kb KEY.notebook. November 23, WHAT IS Ka? UNIT IV. CALCULATIONS USING Ka. WHAT IS Ka? Nov 10 9:42 PM.
 WHAT IS Ka? Recall: Find ph of 0.100 M HCl. But What is ph of 0.10 M HF? UNIT IV Ka and Kb CALCULATIONS USING Ka I. [H 3O + ] (or ph) from Ka Ex. Find the [H 3O + ] in 0.10 M HF. WHAT IS Ka? Look at equilibrium
WHAT IS Ka? Recall: Find ph of 0.100 M HCl. But What is ph of 0.10 M HF? UNIT IV Ka and Kb CALCULATIONS USING Ka I. [H 3O + ] (or ph) from Ka Ex. Find the [H 3O + ] in 0.10 M HF. WHAT IS Ka? Look at equilibrium
Supplementary information
 Supplementary information Preparation of Few-Layer Bismuth Selenide by Liquid-Phase-Exfoliation and Its Optical Absorption Properties Liping Sun 1, Zhiqin Lin 1, Jian Peng 1, Jian Weng 1,3 *, Yizhong Huang
Supplementary information Preparation of Few-Layer Bismuth Selenide by Liquid-Phase-Exfoliation and Its Optical Absorption Properties Liping Sun 1, Zhiqin Lin 1, Jian Peng 1, Jian Weng 1,3 *, Yizhong Huang
Design and Fabrication of Hierarchically Porous Carbon with a Template-free Method
 Supplementary information Design and Fabrication of Hierarchically Porous Carbon with a Template-free Method Yutong Gong, Zhongzhe Wei, Jing Wang, Pengfei Zhang, Haoran Li and Yong Wang * Carbon Nano Materials
Supplementary information Design and Fabrication of Hierarchically Porous Carbon with a Template-free Method Yutong Gong, Zhongzhe Wei, Jing Wang, Pengfei Zhang, Haoran Li and Yong Wang * Carbon Nano Materials
Supporting Information
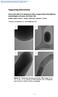 Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
Supporting Information Interaction effects of mesoporous silica nanoparticles with different morphologies on human red blood cells Madhura Joglekar, Robert A. Roggers, Yannan Zhao, and Brian G. Trewyn
5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES
 5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES LEARNING OUTCOMES a) Be able to write formulae of simple compounds b) Be able to write
5072 CHEMISTRY (NEW PAPERS WITH SPA) BASIC TECHNIQUES 5067 CHEMISTRY (NEW PAPERS WITH PRACTICAL EXAM) BASIC TECHNIQUES LEARNING OUTCOMES a) Be able to write formulae of simple compounds b) Be able to write
