Pd Supported on Carbon Nitride Boosts the Direct Hydrogen Peroxide Synthesis
|
|
|
- Deirdre Small
- 5 years ago
- Views:
Transcription
1 Supporting Information Pd Supported on Carbon Nitride Boosts the Direct Hydrogen Peroxide Synthesis Rosa Arrigo,* Manfred E. Schuster, Salvatore Abate, Gianfranco Giorgianni, Gabriele Centi, Siglinda Perathoner, Sabine Wrabetz, Verena Pfeifer, Markus Antonietti, δ Robert Schlögl,. Max-Planck-Institut für Chemische Energiekonversion, Stiftstrasse 34-36, Mülheim an der Ruhr, Germany Fritz-Haber Institut der Max-Planck Gesellschaft, Faradayweg 4-6, Berlin, Germany Università degli Studi di Messina, V.le F. Stagno D'Alcontres 31, Messina, Italy δ Max-Planck-Institut für Kolloid und Grenzflächenforschung, Am Mühlenberg 1 OT Golm, 14476, Potsdam, Germany *Correspondence to: rosa.arrigo@diamond.ac.uk Selectivity/CO differential heat of adsorption correlation Selectivity (S) was correlated to the CO microcalorimetric chemisorption results in order to identify the treshold value of differential heat of chemisorption for the selective path. The following formula was applied and solved analytically: S= mol of CO below the treshold value/ mol of total CO chemisorbed. The Pd1.8%873 after 20 minutes of reaction is characterized by a selectivity of 93%. Thus among the total 2 mol*g -1 of CO adsorbing, only 1.86 mol*g -1 of CO adsorb on the Pd atoms, which catalyze selectively the O2 hydrogenation. For the used Pd0.3%CxN after 180 minutes of reaction, the selectivity is 58%. In this case, among the 0.16 mol*g -1 of CO adsorbing, only 0.96 mol*g -1 of CO adsorb on the Pd atoms, which catalyze selectively the O2 hydrogenation. Those two catalysts actually give a pretty similar threshold value. This corresponds to a threshold value of 70 kj*mol -1 : all the sites probed by CO chemisorption above this value are unselective. On the other hand, for Pd1.8%473, if we consider the overall amount of CO adsorbed 3.4 mol*g -1, 2.1 mol*g -1 are responsible of the unselective pathway which gives a differential heat of adsorption as low as 40 kj*mol -1. For the sample Pd1.7%473 a better correlation was found when considering only the CO chemisorbed with a heat of adsorption above 40 kj*mol -1. In this case, 1.22 kj*mol -1 are considered unselective and 0.78 kj*mol -1 selective. This situation corresponds to a threshold differential heat of chemisorption of about 70 kj*mol -1, consistently with the other samples. If this is the case, chemisorption sites present on the Pd1.7%473 with a differential heat of chemisorption of 40 kj*mol -1 are not involved in the H2O2 synthesis from the elements. These chemisorption sites could be intuitively assigned to the Pd 2+ species highly abundant on this sample. Such a correlation could also explain the initial low activity observed for the PdAuNCNT473 catalysts discussed in reference 6. Those values could be used to model selective and unselective Pd sites and predict better catalysts for the H2O2 direct synthesis.
2 Scheme S 1: Reaction network of the direct H2O2 synthesis: direct formation of H2O2 and H2O, and decomposition of H2O2 to water. Table S1. Comparison of the performances in the direct H2O2 synthesis as reported in patents 1. Company Catalyst H2O2, % wt. Selectivity, % Reaction conditions Dupont Pd-Pt (Pt/Pd+Pt = 0.08) colloidal on alumina ENI 1% Pd-0.1%Pt on carbon BASF Pd on monolith HTI Degussa Pd(-Pt) on carbon black (140 m 2 /g)* 2.5% Pd-Au (95:5) on a- Al2O3 9,1 (276 g/g Pd.h) 5,1 (13.8 g/g Pd.h) bar, 5-8 C, 18% H2 in O2, aqueous acid solution (0.1N HCl) 100 bar, 8 C, (autoclave, after 600h), 3.6% H2, 11% O2 in inert 95:5 methanol:h2o solution (+ additives) 144 bar, 10% H2 in O2, methanol (+ additives) ~120 bar, ~35 C, (autoclave, after 600h), 3% H2 in air solvent and additives not indicated 50 bar, 25 C (trickle bed), 3% H2, 20% O2 methanol (+ additives) a) b) Figure S1. a) Deconvolved N1s XP spectra for NCNT473 (top) and NCNT873 (bottom) at KE 600eV. 12% and 8% are the N atomic abundance for NCNT473 and NCNT873, respectively. Fitting: [11] N1 (398.5 ev) is pyridine-like N; N2 (399.4 ev) is NH bond in amine and amide; N3 (400.1 ev) is pyrrole and lactam species; N4 (401.1 ev) is graphitic N. b) Deconvolved N1s XP spectra for CxN at KE 600eV in different sample spots. 16% and 8% are the N atomic abundance. The N5 component was attributed to N-O species.
3 Figure S 2: C1s spectra at 600eV KE, corresponding to a depth information of 2.5nm, normalized at the maximum peak intensity for comparison: CxN fresh (black line), after Pd immobilization (blue line), Pd0.3%CxN used (red line), NCNT functionalized with NH3 at 873K, NCN873 (green line) and HOPG (pale blue). The C1s spectrum of the NCN873 shows a peak with maximum at ev much broader than the asymmetric peak of HOPG at ev. Broadening to the higher binding energy side indicates C in sp 3 bonding or highly defective sp 2 carbon phase. C-N species contribute to this broadening. The CxN samples are characterized by a more symmetric main peak than the NCNT873, with maximum at 285 ev, indicating a comparatively reduced extension of the conjugated system. The component around 288 ev is attributed to parasitic surface C-O species. [11 ] Figure S 3: Survey spectra with 1100 ev excitation energy for Pd0.3%CxN fresh (red and blue line) and Pd0.3%CxN used (black).
4 Scheme S 2: Proposed formation pathways of the different N species in CxN.
5 a b c d e Figure S 4: Cumulative formation of H2O2 (red cycle), cumulative formation of H2O (black cycle) and H2O2 selectivity (green cycle) for Pd1.8%873 (a); Pd1.8%473 (b); Pd0.3%873SI (c) Pd0.3%873IWI (d) and Pd0.3%CxN (e). Changes in selectivity are triggered by the excessive exposure of the Pd surface to the reactive environment, O2 and H2, which is realized by the initial detachment of the PVA covering the nanoparticles surface facilitating the NPs overgrowth. This is the dominant process for most of the Pd NPs on NCNTs, due to the very low abundance of substitutional N species stabilizing small nanoparticles and, the more rigid carbon structure of the support. On CxN, the higher N abundance and the poorly graphitic surface realize a condition in which the surface of the Pd NPs is protected even at long reaction time resulting in the higher selectivity observed.
6 Figure S 5. HRTEM image and FFT (inset) of fresh Pd1.8%873. The arrows indicate nanoparticles with rough-like morphology. a b Figure S 6: SE (a) and BSE (b) images for Pd0.3%CxN fresh at 15 kv. Local EDX spectra corresponding to point 1 (blue spectrum), point 2 (red spectrum) and point 3 (black spectrum) are reported in the inset of Figure S6a. It is interesting to note that Pd is not detected where impurities of Al and Fe are present. a b c Figure S 7: Scanning electron micrograph at 1.5 kv of CxN (a); Pd0.3%CxN fresh (b) and Pd0.3%CxN used after 180 minutes of reaction (c).
7 a) b) Figure S 8: SE image of Pd0.3%CxN at 1.5 kv (a) and corresponding EDX spectrum (b). The EDX spectrum quantification in weight percentage is: 69% C, 29.4% N, 1% O, 0.4% Pd. Figure S 9 HAADF-STEM image and derived particle size distribution (insets) of Pd/NCNT: fresh Pd1.8%873 (a); 12 min (b) 180 minutes (c, d); fresh Pd1.7%473 (e); 12 minutes (f, g)..
8 Figure S 10. Fractional distribution of surface area of Pd particles derived from the particle size distribution obtained in STEM images (Insets Figure S9) Figure S 11: HAADF-STEM image fresh Pd0.3%CxN (a) and used after 180 minutes of reaction (b).
9 Figure S 12: Tilting series of HRTEM images for a NP of the Pd0.3%CxNused. Note the omnipresence of the carbonaceous fragments at both sides of the NPs. Figure S 13: Adsorption isotherm for: Pd1.8%873 used after 12 minutes of reaction (black square); Pd1.7%473 used after 12 minutes of reaction (red circle); Pd0.3%873IWI fresh (magenta rhombus); Pd0.3%CxN fresh (pale blue triangle); 1 st cycle of CO adsorption on Pd0.3%CxN used after 180 minutes of reaction (green rhombus) and 2 nd cycle of CO adsorption after desorption step (blue triangle). The best fit of the isotherms is the Langmuir isotherm model.
10 Figure S 14. XP Pd3d spectra of Pd1.8%873 (left panel) and Pd1.7%473 (right panel) as fresh and used catalyst. Fitting: [6, 13] bulk-like Pd 0 (335.1 ev); NP Pd 0 ( ev); Pd n+ at NP Pd 0 surface (336.9 ev); ionic Pd 2+ (338 ev). Figure S 15: N1s XP spectra for fresh (bottom) and after 12min reaction (top): Pd1.8%873 (A) and Pd1.7%473(B). Fitting as described in [11, 13]: N1 (red) Pyridine-like N; N2 (blue light) NH in amine and amide; N3 (magenta) in pyrrole-like and lactam N; N3 (green) quaternary N; N5 (dark blue) Pyridine oxide species. Table S2. N and Pd total absolute abundance and relative abundance of Pd species from XPS Catalyst N (at%) Pd (at%) Pd components (%) bulk-like Pd 0 (335.1 ev) NP Pd 0 ( ev) PdO x at Pd 0 surface (336.9 ev) ionic Pd 2+ (338 ev) Pd 1.7%473_fresh Pd 1.8%873_fresh Pd 1.7%473_12min Pd 1.8%873_12min Pd 1.8%873_180min
a b c Supplementary Figure S1
 a b c Supplementary Figure S1 AFM measurements of MoS 2 nanosheets prepared from the electrochemical Liintercalation and exfoliation. (a) AFM measurement of a typical MoS 2 nanosheet, deposited on Si/SiO
a b c Supplementary Figure S1 AFM measurements of MoS 2 nanosheets prepared from the electrochemical Liintercalation and exfoliation. (a) AFM measurement of a typical MoS 2 nanosheet, deposited on Si/SiO
Supporting Information
 Supporting Information Enhanced Stability of Immobilized Pt Nanoparticles on Carbon Nanotubes through Nitrogen Heteroatoms on Doped Carbon Supports Wen Shi, Kuang-Hsu Wu, Junyuan Xu ǁ, Qiang Zhang, Bingsen
Supporting Information Enhanced Stability of Immobilized Pt Nanoparticles on Carbon Nanotubes through Nitrogen Heteroatoms on Doped Carbon Supports Wen Shi, Kuang-Hsu Wu, Junyuan Xu ǁ, Qiang Zhang, Bingsen
The nature of the N-Pd interaction in nitrogen-doped carbon nanotube catalysts
 Supporting Information The nature of the N-Pd interaction in nitrogen-doped carbon nanotube catalysts Rosa Arrigo, * Manfred E. Schuster, Zailai Xie, Youngmi Yi, Gregor Wowsnick, Li L. Sun, Klaus E. Hermann,
Supporting Information The nature of the N-Pd interaction in nitrogen-doped carbon nanotube catalysts Rosa Arrigo, * Manfred E. Schuster, Zailai Xie, Youngmi Yi, Gregor Wowsnick, Li L. Sun, Klaus E. Hermann,
Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons
 Supporting Information Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons Mario Soorholtz, a Robin J. White,* b,c Tobias Zimmermann, a Maria-Magdalena Titirici, b Markus Antonietti, b Regina
Supporting Information Direct Methane Oxidation over Pt-modified Nitrogen-Doped Carbons Mario Soorholtz, a Robin J. White,* b,c Tobias Zimmermann, a Maria-Magdalena Titirici, b Markus Antonietti, b Regina
Depressing the hydrogenation and decomposition. nanoparticles on oxygen functionalized. carbon nanofibers. Supporting Information
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Depressing the hydrogenation and decomposition reaction in H 2 O 2 synthesis
Supplementary Information
 Supplementary Information Supplementary Figure 1. X-ray diffraction patterns of (a) pure LDH, (b) AuCl 4 ion-exchanged LDH and (c) the Au/LDH hybrid catalyst. The refined cell parameters for pure, ion-exchanged,
Supplementary Information Supplementary Figure 1. X-ray diffraction patterns of (a) pure LDH, (b) AuCl 4 ion-exchanged LDH and (c) the Au/LDH hybrid catalyst. The refined cell parameters for pure, ion-exchanged,
A Sustainable Synthesis of Nitrogen-Doped Carbon Aerogels
 A Sustainable Synthesis of Nitrogen-Doped Carbon Aerogels Supporting Information By Robin J. White, a, * Noriko Yoshizawa, b Markus Antonietti, a and Maria-Magdalena Titirici. a * e-mail: robin.white@mpikg.mpg.de
A Sustainable Synthesis of Nitrogen-Doped Carbon Aerogels Supporting Information By Robin J. White, a, * Noriko Yoshizawa, b Markus Antonietti, a and Maria-Magdalena Titirici. a * e-mail: robin.white@mpikg.mpg.de
NanoEngineering of Hybrid Carbon Nanotube Metal Composite Materials for Hydrogen Storage Anders Nilsson
 NanoEngineering of Hybrid Carbon Nanotube Metal Composite Materials for Hydrogen Storage Anders Nilsson Stanford Synchrotron Radiation Laboratory (SSRL) and Stockholm University Coworkers and Ackowledgement
NanoEngineering of Hybrid Carbon Nanotube Metal Composite Materials for Hydrogen Storage Anders Nilsson Stanford Synchrotron Radiation Laboratory (SSRL) and Stockholm University Coworkers and Ackowledgement
The influence of intercalated oxygen on the properties of graphene on polycrystalline Cu under various environmental conditions
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information The influence of intercalated oxygen on the
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2014 Electronic Supplementary Information The influence of intercalated oxygen on the
Supporting Information:
 Supporting Information: Supplementary Figure 1. (a) TEM and (b) HRTEM image of the Pd nanocube seeds. Inset of (b): the FFT pattern. (c) Magnified image of the square region highlighted in (b). Supplementary
Supporting Information: Supplementary Figure 1. (a) TEM and (b) HRTEM image of the Pd nanocube seeds. Inset of (b): the FFT pattern. (c) Magnified image of the square region highlighted in (b). Supplementary
Multifunktionale Elektrodenmaterialien von Hydrothermalem Kohlenstoff
 Multifunktionale Elektrodenmaterialien von Hydrothermalem Kohlenstoff AKK-Fachausschuss Neue Kohlenstoff-Formen Selb, 6. November 2014 Sylvia Reiche Max-Planck-Institut für Chemische Energiekonversion
Multifunktionale Elektrodenmaterialien von Hydrothermalem Kohlenstoff AKK-Fachausschuss Neue Kohlenstoff-Formen Selb, 6. November 2014 Sylvia Reiche Max-Planck-Institut für Chemische Energiekonversion
Supporting Information
 Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
Electronic Supplementary Material (ESI) for CrystEngComm. This journal is The Royal Society of Chemistry 217 Supporting Information Catalyst preparation A certain of aqueous NiCl 2 6H 2 O (2 mm), H 2 PtCl
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide Supporting online material Konstantin V. Emtsev 1, Aaron Bostwick 2, Karsten Horn
SUPPLEMENTARY INFORMATION Towards wafer-size graphene layers by atmospheric pressure graphitization of silicon carbide Supporting online material Konstantin V. Emtsev 1, Aaron Bostwick 2, Karsten Horn
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c,
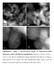 Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Supplementary Figure 1 HAADF-STEM images of 0.08%Pt/FeO x -R200 single-atom catalyst with different magnifications. Scale bar: a, 20 nm; b, 10 nm; c, 2 nm; d, 2 nm. The low magnification images demonstrate
Catalysis by supported metal clusters and model systems
 Catalysis by supported metal clusters and model systems Gianfranco Pacchioni Dipartimento di Scienza dei Materiali Università Milano-Bicocca Part I Catalysis by supported metal clusters and model systems
Catalysis by supported metal clusters and model systems Gianfranco Pacchioni Dipartimento di Scienza dei Materiali Università Milano-Bicocca Part I Catalysis by supported metal clusters and model systems
Shape-selective Synthesis and Facet-dependent Enhanced Electrocatalytic Activity and Durability of Monodisperse Sub-10 nm Pt-Pd Tetrahedrons and Cubes
 Supporting Information Shape-selective Synthesis and Facet-dependent Enhanced Electrocatalytic Activity and Durability of Monodisperse Sub-10 nm Pt-Pd Tetrahedrons and Cubes An-Xiang Yin, Xiao-Quan Min,
Supporting Information Shape-selective Synthesis and Facet-dependent Enhanced Electrocatalytic Activity and Durability of Monodisperse Sub-10 nm Pt-Pd Tetrahedrons and Cubes An-Xiang Yin, Xiao-Quan Min,
Special Properties of Au Nanoparticles
 Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Special Properties of Au Nanoparticles Maryam Ebrahimi Chem 7500/750 March 28 th, 2007 1 Outline Introduction The importance of unexpected electronic, geometric, and chemical properties of nanoparticles
Supporting Information
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Novel Carbon Nitride Composites with Improved Visible Light Absorption
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Novel Carbon Nitride Composites with Improved Visible Light Absorption
Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO
 Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
Supporing Information Very low temperature CO oxidation over colloidally deposited gold nanoparticles on Mg(OH) 2 and MgO Chun-Jiang Jia, Yong Liu, Hans Bongard, Ferdi Schüth* Max-Planck-Institut für Kohlenforschung,
BAE 820 Physical Principles of Environmental Systems
 BAE 820 Physical Principles of Environmental Systems Catalysis of environmental reactions Dr. Zifei Liu Catalysis and catalysts Catalysis is the increase in the rate of a chemical reaction due to the participation
BAE 820 Physical Principles of Environmental Systems Catalysis of environmental reactions Dr. Zifei Liu Catalysis and catalysts Catalysis is the increase in the rate of a chemical reaction due to the participation
Heterogeneous catalysis: the fundamentals Kinetics
 www.catalysiscourse.com Heterogeneous catalysis: the fundamentals Kinetics Prof dr J W (Hans) Niemantsverdriet Schuit Institute of Catalysis Catalysis is a cycle A B separation P catalyst P bonding catalyst
www.catalysiscourse.com Heterogeneous catalysis: the fundamentals Kinetics Prof dr J W (Hans) Niemantsverdriet Schuit Institute of Catalysis Catalysis is a cycle A B separation P catalyst P bonding catalyst
Acidic Water Monolayer on Ruthenium(0001)
 Acidic Water Monolayer on Ruthenium(0001) Youngsoon Kim, Eui-seong Moon, Sunghwan Shin, and Heon Kang Department of Chemistry, Seoul National University, 1 Gwanak-ro, Seoul 151-747, Republic of Korea.
Acidic Water Monolayer on Ruthenium(0001) Youngsoon Kim, Eui-seong Moon, Sunghwan Shin, and Heon Kang Department of Chemistry, Seoul National University, 1 Gwanak-ro, Seoul 151-747, Republic of Korea.
Fabrication of a One-dimensional Tube-in-tube Polypyrrole/Tin oxide Structure for Highly Sensitive DMMP Sensor Applications
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information (ESI) for Fabrication of a One-dimensional
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information (ESI) for Fabrication of a One-dimensional
Hydrogenation of Single Walled Carbon Nanotubes
 Hydrogenation of Single Walled Carbon Nanotubes Anders Nilsson Stanford Synchrotron Radiation Laboratory (SSRL) and Stockholm University Coworkers and Ackowledgement A. Nikitin 1), H. Ogasawara 1), D.
Hydrogenation of Single Walled Carbon Nanotubes Anders Nilsson Stanford Synchrotron Radiation Laboratory (SSRL) and Stockholm University Coworkers and Ackowledgement A. Nikitin 1), H. Ogasawara 1), D.
Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with. High Oxidation-Resistant Property as Efficient and Durable
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with High
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information Sub-10-nm Au-Pt-Pd Alloy Trimetallic Nanoparticles with High
Supporting Information
 Supporting Information High Performance Electrocatalyst: Pt-Cu Hollow Nanocrystals Xiaofei Yu, a Dingsheng, a Qing Peng a and Yadong Li* a a Department of Chemistry, Tsinghua University, Beijing, 100084
Supporting Information High Performance Electrocatalyst: Pt-Cu Hollow Nanocrystals Xiaofei Yu, a Dingsheng, a Qing Peng a and Yadong Li* a a Department of Chemistry, Tsinghua University, Beijing, 100084
Supplementary Information. ZIF-8 Immobilized Ni(0) Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane
 Supplementary Information ZIF-8 Immobilized Ni() Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane Pei-Zhou Li, a,b Kengo Aranishi, a and Qiang Xu* a,b
Supplementary Information ZIF-8 Immobilized Ni() Nanoparticles: Highly Effective Catalysts for Hydrogen Generation from Hydrolysis of Ammonia Borane Pei-Zhou Li, a,b Kengo Aranishi, a and Qiang Xu* a,b
Reactions at solid surfaces: From atoms to complexity. Gerhard Ertl Fritz Haber Institut der Max Planck-Gesellschaft Berlin, Germany
 Reactions at solid surfaces: From atoms to complexity Gerhard Ertl Fritz Haber Institut der Max Planck-Gesellschaft Berlin, Germany Jöns Jakob Berzelius 1779 1848 Wilhelm Ostwald 1853 1932 Nobel Prize
Reactions at solid surfaces: From atoms to complexity Gerhard Ertl Fritz Haber Institut der Max Planck-Gesellschaft Berlin, Germany Jöns Jakob Berzelius 1779 1848 Wilhelm Ostwald 1853 1932 Nobel Prize
1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO
 Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Supplementary Figures GO Supplementary Figure S1 1-amino-9-octadecene, HAuCl 4, hexane, ethanol 55 o C, 16h AuSSs on GO Schematic illustration of synthesis of Au square sheets on graphene oxide sheets.
Supporting information High density monodispersed cobalt nanoparticles filled into multi-walled carbon nanotubes
 Supporting information High density monodispersed cobalt nanoparticles filled into multi-walled carbon nanotubes Walid Baaziz a, Sylvie Begin-Colin a*, Benoit P. Pichon a, Ileana Florea a, Ovidiu Ersen
Supporting information High density monodispersed cobalt nanoparticles filled into multi-walled carbon nanotubes Walid Baaziz a, Sylvie Begin-Colin a*, Benoit P. Pichon a, Ileana Florea a, Ovidiu Ersen
Electronic Processes on Semiconductor Surfaces during Chemisorption
 Electronic Processes on Semiconductor Surfaces during Chemisorption T. Wolkenstein Translatedfrom Russian by E. M. Yankovskii Translation edited in part by Roy Morrison CONSULTANTS BUREAU NEW YORK AND
Electronic Processes on Semiconductor Surfaces during Chemisorption T. Wolkenstein Translatedfrom Russian by E. M. Yankovskii Translation edited in part by Roy Morrison CONSULTANTS BUREAU NEW YORK AND
Supporting Information
 Supporting Information Tandem Site and Size Controlled Pd Nanoparticles for the Directed Hydrogenation of Furfural Scott M. Rogers, 1,2 C. Richard. A. Catlow, 1,2,5 Carine E. Chan-Thaw, 3 Arunabhiram Chutia,
Supporting Information Tandem Site and Size Controlled Pd Nanoparticles for the Directed Hydrogenation of Furfural Scott M. Rogers, 1,2 C. Richard. A. Catlow, 1,2,5 Carine E. Chan-Thaw, 3 Arunabhiram Chutia,
Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward. High-efficiency C C Splitting of Ethanol in Conversion to CO 2
 Supporting Information Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward High-efficiency C C Splitting of Ethanol in Conversion to CO 2 Wei Zhu,, Jun Ke,, Si-Bo Wang, Jie Ren, Hong-Hui
Supporting Information Shaping Single-crystalline Trimetallic Pt Pd Rh Nanocrystals toward High-efficiency C C Splitting of Ethanol in Conversion to CO 2 Wei Zhu,, Jun Ke,, Si-Bo Wang, Jie Ren, Hong-Hui
Plasma driven ammonia decomposition on Fe-catalyst: eliminating surface nitrogen poisoning
 Supporting Information for Plasma driven ammonia decomposition on Fe-catalyst: eliminating surface nitrogen poisoning Contents: 1. Scheme of the DBD plasma-driven catalysis reactor, Scheme S1. 2. XRF analysis
Supporting Information for Plasma driven ammonia decomposition on Fe-catalyst: eliminating surface nitrogen poisoning Contents: 1. Scheme of the DBD plasma-driven catalysis reactor, Scheme S1. 2. XRF analysis
Supporting Information
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2018 Supporting Information The chemical identity, state and structure of catalytically active
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 2018 Supporting Information The chemical identity, state and structure of catalytically active
Surface structure vs. surface science; Atomic imaging of surface with STM; Nanoscale surface probing with STM; Exploration of surface catalysis.
 Lecture 7: STM Atomic Imaging of Metal and Semiconductor Surface Surface structure vs. surface science; Atomic imaging of surface with STM; Nanoscale surface probing with STM; Exploration of surface catalysis.
Lecture 7: STM Atomic Imaging of Metal and Semiconductor Surface Surface structure vs. surface science; Atomic imaging of surface with STM; Nanoscale surface probing with STM; Exploration of surface catalysis.
Supporting Information. Oxygen reduction and methanol oxidation behaviour of SiC based Pt. nanocatalysts for proton exchange membrane fuel cells
 Supporting Information Oxygen reduction and methanol oxidation behaviour of SiC based Pt nanocatalysts for proton exchange membrane fuel cells Rajnish Dhiman a *, Serban N. Stamatin a, Shuang M. Andersen
Supporting Information Oxygen reduction and methanol oxidation behaviour of SiC based Pt nanocatalysts for proton exchange membrane fuel cells Rajnish Dhiman a *, Serban N. Stamatin a, Shuang M. Andersen
Boosting the hydrogen evolution performance of ruthenium clusters. through synergistic coupling with cobalt phosphide
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Boosting the hydrogen evolution
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2018 Electronic Supplementary Information for Boosting the hydrogen evolution
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 218 Rel. intensity Rel. intensity Electronic Supplementary Information Under-cover stabilization
Electronic Supplementary Material (ESI) for Chemical Science. This journal is The Royal Society of Chemistry 218 Rel. intensity Rel. intensity Electronic Supplementary Information Under-cover stabilization
A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On
 Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Supplementary Information A Novel Electroless Method for the Deposition of Single-Crystalline Platinum Nanoparticle Films On an Organic Solid Matrix in the Presence of Gold Single Crystals Khaleda Banu,,,*
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using
 Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Supplementary Figure 1: Power dependence of hot-electrons reduction of 4-NTP to 4-ATP. a) SERS spectra of the hot-electron reduction reaction using 633 nm laser excitation at different powers and b) the
Direct-writing on monolayer GO with Pt-free AFM tips in the
 Supplementary Figure S1 Direct-writing on monolayer GO with Pt-free AFM tips in the presence of hydrogen. We replaced the Pt-coated tip with a gold-coated tip or an untreated fresh silicon tip, and kept
Supplementary Figure S1 Direct-writing on monolayer GO with Pt-free AFM tips in the presence of hydrogen. We replaced the Pt-coated tip with a gold-coated tip or an untreated fresh silicon tip, and kept
Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity
 Supporting Information Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity Neil M. Wilson, 1 Yung-Tin Pan, 1 Yu-Tsun Shao, 2 Jian-Min Zuo, 2
Supporting Information Direct Synthesis of H 2 O 2 on AgPt Octahedra: The Importance of Ag-Pt Coordination for High H 2 O 2 Selectivity Neil M. Wilson, 1 Yung-Tin Pan, 1 Yu-Tsun Shao, 2 Jian-Min Zuo, 2
Tunable nitrogen-doped carbon aerogels as sustainable electrocatalysts in the oxygen. reduction reaction Electronic Supplementary information (ESI)
 Tunable nitrogen-doped carbon aerogels as sustainable electrocatalysts in the oxygen reduction reaction Electronic Supplementary information (ESI) Stephanie-Angelika Wohlgemuth,* a Tim-Patrick Fellinger
Tunable nitrogen-doped carbon aerogels as sustainable electrocatalysts in the oxygen reduction reaction Electronic Supplementary information (ESI) Stephanie-Angelika Wohlgemuth,* a Tim-Patrick Fellinger
Nano-Enabled Catalysts for the Commercially Viable Production of H 2 O 2
 Nano-Enabled Catalysts for the Commercially Viable Production of H 2 O 2 Bing Zhou Headwaters Technology Innovation, LLC. Lawrenceville, NJ 08648 September 26, 2007 1 Headwaters Technology Development
Nano-Enabled Catalysts for the Commercially Viable Production of H 2 O 2 Bing Zhou Headwaters Technology Innovation, LLC. Lawrenceville, NJ 08648 September 26, 2007 1 Headwaters Technology Development
One-Pot Synthesis of Core-Shell-like Pt 3 Co Nanoparticle Electrocatalyst with Pt-enriched Surface for Oxygen Reduction Reaction in Fuel Cells
 Electronic Supplementary Information for One-Pot Synthesis of Core-Shell-like 3 Co Nanoparticle Electrocatalyst with -enriched Surface for Oxygen Reduction Reaction in Fuel Cells Ji-Hoon Jang b, Juyeong
Electronic Supplementary Information for One-Pot Synthesis of Core-Shell-like 3 Co Nanoparticle Electrocatalyst with -enriched Surface for Oxygen Reduction Reaction in Fuel Cells Ji-Hoon Jang b, Juyeong
Supplementary Information for
 Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Supplementary Information for Facile transformation of low cost thiourea into nitrogen-rich graphitic carbon nitride nanocatalyst with high visible light photocatalytic performance Fan Dong *a, Yanjuan
Supporting Information for. Size-Dependent Oxidation State and CO Oxidation Activity of Tin
 Supporting Information for Size-Dependent Oxidation State and CO Oxidation Activity of Tin Oxide Clusters Yusuke Inomata, Ken Albrecht,, Kimihisa Yamamoto *,, Laboratory for Chemistry and Life Science,
Supporting Information for Size-Dependent Oxidation State and CO Oxidation Activity of Tin Oxide Clusters Yusuke Inomata, Ken Albrecht,, Kimihisa Yamamoto *,, Laboratory for Chemistry and Life Science,
STUDY ON THE IMPROVEMENT OF THE REDUCTION CAPACITY OF ACTIVATED CARBON FIBER
 STUDY ON THE IMPROVEMENT OF THE REDUCTION CAPACITY OF ACTIVATED CARBON FIBER Chen Shuixia, Zeng Hanmin Materials Science Institute, Zhongshan University, Guangzhou 51275, China Key Laboratory for Polymeric
STUDY ON THE IMPROVEMENT OF THE REDUCTION CAPACITY OF ACTIVATED CARBON FIBER Chen Shuixia, Zeng Hanmin Materials Science Institute, Zhongshan University, Guangzhou 51275, China Key Laboratory for Polymeric
Supporting information
 Supporting information imetallic AuRh nanodendrites consisting of Au icosahedron cores and atomically ultrathin Rh nanoplate shells: synthesis and light-enhanced catalytic activity Yongqiang Kang 1,2,
Supporting information imetallic AuRh nanodendrites consisting of Au icosahedron cores and atomically ultrathin Rh nanoplate shells: synthesis and light-enhanced catalytic activity Yongqiang Kang 1,2,
Supporting information. Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability
 Supporting information Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability and Oxygen Reduction Reaction (ORR) Activity R. K. Singh, R. Rahul, M. Neergat 1 Department
Supporting information Stability Issues in Pd-based Catalysts: The Role of Surface Pt in Improving the Stability and Oxygen Reduction Reaction (ORR) Activity R. K. Singh, R. Rahul, M. Neergat 1 Department
Mesoporous N-Doped Carbons Prepared with Thermally Removable Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction
 Supporting Information Mesoporous N-Doped Carons Prepared with Thermally Removale Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction Wenhan Niu, a Ligui Li,* a Xiaojun Liu,
Supporting Information Mesoporous N-Doped Carons Prepared with Thermally Removale Nanoparticle Templates: an Efficient Electrocatalyst for Oxygen Reduction Reaction Wenhan Niu, a Ligui Li,* a Xiaojun Liu,
Catalyst Characterization Using Thermal Conductivity Detector *
 OpenStax-CNX module: m43579 1 Catalyst Characterization Using Thermal Conductivity Detector * Juan Velazquez Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons
OpenStax-CNX module: m43579 1 Catalyst Characterization Using Thermal Conductivity Detector * Juan Velazquez Andrew R. Barron This work is produced by OpenStax-CNX and licensed under the Creative Commons
Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 216 Cu 2 O/g-C 3 N 4 nanocomposites: An insight into the band structure tuning and catalytic efficiencies
Supporting Information
 Supporting Information Identification of the nearby hydroxyls role in promoting HCHO oxidation over a Pt catalyst Ying Huo #, Xuyu Wang #, Zebao Rui *, Xiaoqing Yang, Hongbing Ji * School of Chemical Engineering
Supporting Information Identification of the nearby hydroxyls role in promoting HCHO oxidation over a Pt catalyst Ying Huo #, Xuyu Wang #, Zebao Rui *, Xiaoqing Yang, Hongbing Ji * School of Chemical Engineering
Supporting Information. CdS/mesoporous ZnS core/shell particles for efficient and stable photocatalytic hydrogen evolution under visible light
 Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supporting Information CdS/mesoporous ZnS core/shell particles for efficient
Electronic Supplementary Material (ESI) for Energy & Environmental Science. This journal is The Royal Society of Chemistry 2014 Supporting Information CdS/mesoporous ZnS core/shell particles for efficient
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Using first principles to predict bimetallic catalysts for the ammonia decomposition reaction Danielle A. Hansgen, Dionisios G. Vlachos, Jingguang G. Chen SUPPLEMENTARY INFORMATION.
SUPPLEMENTARY INFORMATION Using first principles to predict bimetallic catalysts for the ammonia decomposition reaction Danielle A. Hansgen, Dionisios G. Vlachos, Jingguang G. Chen SUPPLEMENTARY INFORMATION.
Study on the Selective Hydrogenation of Nitroaromatics to N-aryl hydroxylamines using a Supported Pt nanoparticle Catalyst
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 204 Supporting Information Study on the Selective Hydrogenation of Nitroaromatics
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 204 Supporting Information Study on the Selective Hydrogenation of Nitroaromatics
Electronic Supplementary Information. Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered
 Electronic Supplementary Information Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered polyelectrolyte nanoreactors on a charged sphere Young-Min Chung,* a Yong-Tak
Electronic Supplementary Information Direct synthesis of H 2 O 2 catalyzed by Pd nanoparticles encapsulated in multi-layered polyelectrolyte nanoreactors on a charged sphere Young-Min Chung,* a Yong-Tak
Nitrogen and sulfur co-doped porous carbon derived from human hair as. highly efficient metal-free electrocatalyst for hydrogen evolution reaction
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Nitrogen and sulfur co-doped porous
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information Nitrogen and sulfur co-doped porous
Effect of Chloride Anions on the Synthesis and. Enhanced Catalytic Activity of Silver Nanocoral
 Supporting Information Effect of Chloride Anions on the Synthesis and Enhanced Catalytic Activity of Silver Nanocoral Electrodes for CO 2 Electroreduction Polyansky* Yu-Chi Hsieh, Sanjaya D. Senanayake,
Supporting Information Effect of Chloride Anions on the Synthesis and Enhanced Catalytic Activity of Silver Nanocoral Electrodes for CO 2 Electroreduction Polyansky* Yu-Chi Hsieh, Sanjaya D. Senanayake,
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 214 Electronic Supplementary Information Ultrathin and High-Ordered CoO Nanosheet
Molecular-Level Insight into Selective Catalytic Reduction of NO x with NH 3 to N 2
 Supporting Information Molecular-Level Insight into Selective Catalytic Reduction of NO x with to N 2 over Highly Efficient Bifunctional V a Catalyst at Low Temperature Ying Xin, Hao Li, Nana Zhang, Qian
Supporting Information Molecular-Level Insight into Selective Catalytic Reduction of NO x with to N 2 over Highly Efficient Bifunctional V a Catalyst at Low Temperature Ying Xin, Hao Li, Nana Zhang, Qian
Supporting Information. Solar-driven Reduction of Aqueous Protons Coupled to. Selective Alcohol Oxidation with a Carbon Nitride - Molecular
 Supporting Information Solar-driven Reduction of Aqueous Protons Coupled to Selective Alcohol Oxidation with a Carbon Nitride - Molecular Ni Catalyst System Hatice Kasap, 1 Christine A. Caputo, 1 Benjamin
Supporting Information Solar-driven Reduction of Aqueous Protons Coupled to Selective Alcohol Oxidation with a Carbon Nitride - Molecular Ni Catalyst System Hatice Kasap, 1 Christine A. Caputo, 1 Benjamin
Review of Nature Communications, NCOMMS In situ tracking of heterogeneous nanocatalytic processes. Reviewers' comments:
 Reviewers' comments: Reviewer #1 (Remarks to the Author): This manuscript describes measurement of surface enhanced Raman spectra (SERS) during CO oxidation catalysis on metal catalysts (E.g, Pt, FePt,
Reviewers' comments: Reviewer #1 (Remarks to the Author): This manuscript describes measurement of surface enhanced Raman spectra (SERS) during CO oxidation catalysis on metal catalysts (E.g, Pt, FePt,
Reviewers' Comments: Reviewer #1 (Remarks to the Author)
 Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript reports the synthesis of a series of Mo2C@NPC-rGO hybrid HER electrocatalysts by employing the precursor of PMo12 (H3PMo12O40)-PPy/rGO
Reviewers' Comments: Reviewer #1 (Remarks to the Author) The manuscript reports the synthesis of a series of Mo2C@NPC-rGO hybrid HER electrocatalysts by employing the precursor of PMo12 (H3PMo12O40)-PPy/rGO
Hydrogen adsorption by graphite intercalation compounds
 62 Chapter 4 Hydrogen adsorption by graphite intercalation compounds 4.1 Introduction Understanding the thermodynamics of H 2 adsorption in chemically modified carbons remains an important area of fundamental
62 Chapter 4 Hydrogen adsorption by graphite intercalation compounds 4.1 Introduction Understanding the thermodynamics of H 2 adsorption in chemically modified carbons remains an important area of fundamental
Role of Re and Ru in Re Ru/C Bimetallic Catalysts for the
 Role of Re and Ru in Re Ru/C Bimetallic Catalysts for the Aqueous Hydrogenation of Succinic Acid Xin Di a, Chuang Li a, Bingsen Zhang b, Ji Qi a, Wenzhen Li c, Dangsheng Su b, Changhai Liang a, * a Laboratory
Role of Re and Ru in Re Ru/C Bimetallic Catalysts for the Aqueous Hydrogenation of Succinic Acid Xin Di a, Chuang Li a, Bingsen Zhang b, Ji Qi a, Wenzhen Li c, Dangsheng Su b, Changhai Liang a, * a Laboratory
Supporting Information
 Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2019 Supporting Information Atomically dispersed Ni as the active site towards selective hydrogenation
Electronic Supplementary Material (ESI) for Green Chemistry. This journal is The Royal Society of Chemistry 2019 Supporting Information Atomically dispersed Ni as the active site towards selective hydrogenation
Electronic Supplementary Information
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Probing the Surface Reactivity of
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2017 Electronic Supplementary Information Probing the Surface Reactivity of
Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (b-f) bright-field images of the microstructure. Only two broad rings
 Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (bf) brightfield images of the microstructure. Only two broad rings were observed in the SAED pattern, as expected for amorphous
Supplementary Figure 1. TEM analysis of Co0.5 showing (a) a SAED pattern, and (bf) brightfield images of the microstructure. Only two broad rings were observed in the SAED pattern, as expected for amorphous
Metal free and Nonprecious Metal Materials for Energy relevant Electrocatalytic Processes. Shizhang Qiao ( 乔世璋 )
 Metal free and Nonprecious Metal Materials for Energy relevant Electrocatalytic Processes Shizhang Qiao ( 乔世璋 ) s.qiao@adelaide.edu.au The University of Adelaide, Australia 18 19 January 216, Perth 1.
Metal free and Nonprecious Metal Materials for Energy relevant Electrocatalytic Processes Shizhang Qiao ( 乔世璋 ) s.qiao@adelaide.edu.au The University of Adelaide, Australia 18 19 January 216, Perth 1.
Supporting Information
 Supporting Information Single-atom and Nano-clustered Pt Catalysts for Selective CO 2 Reduction Yuan Wang, a Hamidreza Arandiyan,* a,b Jason Scott,* a Kondo-Francois Aguey-Zinsou, c and Rose Amal* a Miss
Supporting Information Single-atom and Nano-clustered Pt Catalysts for Selective CO 2 Reduction Yuan Wang, a Hamidreza Arandiyan,* a,b Jason Scott,* a Kondo-Francois Aguey-Zinsou, c and Rose Amal* a Miss
SUPPORTING INFORMATION
 SUPPORTING INFORMATION Shape Control of Colloidal Cu 2-x S Polyhedral Nanocrystals by Tuning the Nucleation Rates Ward van der Stam, Sabine Gradmann, Thomas Altantzis, Xiaoxing Ke, Marc Baldus, Sara Bals
SUPPORTING INFORMATION Shape Control of Colloidal Cu 2-x S Polyhedral Nanocrystals by Tuning the Nucleation Rates Ward van der Stam, Sabine Gradmann, Thomas Altantzis, Xiaoxing Ke, Marc Baldus, Sara Bals
Supporting Information
 Supporting Information Selective CO 2 Reduction to CO in Water using Earth-Abundant Metal and Nitrogen-Doped Carbon Electrocatalysts Xin-Ming Hu, a,b Halvor Høen Hval, a,b Emil Tveden Bjerglund, a,b Kirstine
Supporting Information Selective CO 2 Reduction to CO in Water using Earth-Abundant Metal and Nitrogen-Doped Carbon Electrocatalysts Xin-Ming Hu, a,b Halvor Høen Hval, a,b Emil Tveden Bjerglund, a,b Kirstine
Electronic Supplementary Information (ESI)
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Multi-scale promoting effects
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2015 Electronic Supplementary Information (ESI) Multi-scale promoting effects
Topic 12 Transition Metals Revision Notes
 Topic 12 Transition Metals Revision Notes 1) Introduction Transition metals have 4 characteristic properties: they form complexes, they form coloured compounds, they have more than one oxidation state
Topic 12 Transition Metals Revision Notes 1) Introduction Transition metals have 4 characteristic properties: they form complexes, they form coloured compounds, they have more than one oxidation state
SUPPLEMENTARY INFORMATION
 Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 SUPPLEMENTARY INFORMATION From Melamine-Resorcinol-Formaldehyde to Nitrogen-Doped
Electronic Supplementary Material (ESI) for Journal of Materials Chemistry A. This journal is The Royal Society of Chemistry 2014 SUPPLEMENTARY INFORMATION From Melamine-Resorcinol-Formaldehyde to Nitrogen-Doped
Sol- Gel Synthesis Of Transi3on Metal Doped Silica Nanopar3cles
 Sol- Gel Synthesis Of Transi3on Metal Doped Silica Nanopar3cles Pat Downs Graduate Student Department of Material Science and Engineering Rensselaer Polytechnic Institute, Troy, NY, USA Advisors: Linda
Sol- Gel Synthesis Of Transi3on Metal Doped Silica Nanopar3cles Pat Downs Graduate Student Department of Material Science and Engineering Rensselaer Polytechnic Institute, Troy, NY, USA Advisors: Linda
Supporting information. Insight mechanism revealing the peroxidase mimetic catalytic activity of
 Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting information Insight mechanism revealing the peroxidase mimetic catalytic activity of
Electronic Supplementary Material (ESI) for Nanoscale. This journal is The Royal Society of Chemistry 2015 Supporting information Insight mechanism revealing the peroxidase mimetic catalytic activity of
Supplementary information. Derivatization and Interlaminar Debonding of Graphite-Iron Nanoparticles Hybrid
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Supplementary information Derivatization and Interlaminar Debonding of Graphite-Iron
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 2017 Supplementary information Derivatization and Interlaminar Debonding of Graphite-Iron
Supporting Information. Dissociative Water Adsorption by Al 3 O 4 + in the Gas Phase. Linnéstrasse 2, D Leipzig, Germany.
 Supporting Information Dissociative Water Adsorption by Al 3 O 4 + in the Gas Phase Matias R. Fagiani, 1,2, Xiaowei Song, 1,2 Sreekanta Debnath, 1,2 Sandy Gewinner, 2 Wieland Schöllkopf, 2 Knut R. Asmis,
Supporting Information Dissociative Water Adsorption by Al 3 O 4 + in the Gas Phase Matias R. Fagiani, 1,2, Xiaowei Song, 1,2 Sreekanta Debnath, 1,2 Sandy Gewinner, 2 Wieland Schöllkopf, 2 Knut R. Asmis,
Urchin-like Ni-P microstructures: A facile synthesis, properties. and application in the fast removal of heavy-metal ions
 SUPPORTING INFORMATION Urchin-like Ni-P microstructures: A facile synthesis, properties and application in the fast removal of heavy-metal ions Yonghong Ni *a, Kai Mi a, Chao Cheng a, Jun Xia a, Xiang
SUPPORTING INFORMATION Urchin-like Ni-P microstructures: A facile synthesis, properties and application in the fast removal of heavy-metal ions Yonghong Ni *a, Kai Mi a, Chao Cheng a, Jun Xia a, Xiang
4GLS. Workshop University College London. In situ XPS A Tool to bridge the Pressure Gap in Heterogeneous Catalysis
 Catalysis @ 4GLS Workshop 19.06.2006 University College London In situ XPS A Tool to bridge the Pressure Gap in Heterogeneous Catalysis Axel Knop-Gericke Fritz-Haber-Institut, Berlin, Department of Inorganic
Catalysis @ 4GLS Workshop 19.06.2006 University College London In situ XPS A Tool to bridge the Pressure Gap in Heterogeneous Catalysis Axel Knop-Gericke Fritz-Haber-Institut, Berlin, Department of Inorganic
Supplementary Figure 1 Morpholigical properties of TiO 2-x SCs. The statistical particle size distribution (a) of the defective {001}-TiO 2-x SCs and
 Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Supplementary Figure 1 Morpholigical properties of TiO 2-x s. The statistical particle size distribution (a) of the defective {1}-TiO 2-x s and their typical TEM images (b, c). Quantity Adsorbed (cm 3
Novel Nickel-Palladium Catalysts Encased in a Platinum Nanocage
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) Novel Nickel-Palladium Catalysts Encased in a Platinum
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Electronic Supplementary Information (ESI) Novel Nickel-Palladium Catalysts Encased in a Platinum
Supplementary Figure 1. XRD pattern for pristine graphite (PG), graphite oxide (GO) and
 Supplementary Figure 1. XRD pattern for pristine graphite (PG), graphite oxide (GO) and expanded graphites (EG-1hr and EG-5hr). The crystalline structures of PG, GO, EG-1hr, and EG-5hr were characterized
Supplementary Figure 1. XRD pattern for pristine graphite (PG), graphite oxide (GO) and expanded graphites (EG-1hr and EG-5hr). The crystalline structures of PG, GO, EG-1hr, and EG-5hr were characterized
Ab Initio Study of Hydrogen Storage on CNT
 Ab Initio Study of Hydrogen Storage on CNT Zhiyong Zhang, Henry Liu, and KJ Cho Stanford University Presented at the ICNT 2005, San Francisco Financial Support: GCEP (Global Climate and Energy Project)
Ab Initio Study of Hydrogen Storage on CNT Zhiyong Zhang, Henry Liu, and KJ Cho Stanford University Presented at the ICNT 2005, San Francisco Financial Support: GCEP (Global Climate and Energy Project)
ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation
 S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
S1 ph-depending Enhancement of Electron Transfer by {001} Facet-Dominating TiO 2 Nanoparticles for Photocatalytic H 2 Evolution under Visible Irradiation Masato M. Maitani a *, Zhan Conghong a,b, Dai Mochizuki
- Supporting Information -
 Protomers of Benzocaine: Solvent and Permittivity Dependence Stephan Warnke, Jongcheol Seo, Jasper Boschmans, Frank Sobott, James H. Scrivens, Christian Bleiholder,,? Michael T. Bowers, Sandy Gewinner,
Protomers of Benzocaine: Solvent and Permittivity Dependence Stephan Warnke, Jongcheol Seo, Jasper Boschmans, Frank Sobott, James H. Scrivens, Christian Bleiholder,,? Michael T. Bowers, Sandy Gewinner,
Photocatalytic degradation of dyes over graphene-gold nanocomposites under visible light irradiation
 Photocatalytic degradation of dyes over graphene-gold nanocomposites under visible light irradiation Zhigang Xiong, Li Li Zhang, Jizhen Ma, X. S. Zhao* Department of Chemical and Biomolecular Engineering,
Photocatalytic degradation of dyes over graphene-gold nanocomposites under visible light irradiation Zhigang Xiong, Li Li Zhang, Jizhen Ma, X. S. Zhao* Department of Chemical and Biomolecular Engineering,
Pd-P nanoalloys supported on porous carbon frame as efficient catalyst for benzyl alcohol oxidation
 Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2018 Supporting information Pd-P nanoalloys supported on porous carbon frame as
Electronic Supplementary Material (ESI) for Catalysis Science & Technology. This journal is The Royal Society of Chemistry 2018 Supporting information Pd-P nanoalloys supported on porous carbon frame as
Supporting Information. Modulating the photocatalytic redox preferences between
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Modulating the photocatalytic redox preferences between anatase TiO 2 {001}
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2016 Supporting Information Modulating the photocatalytic redox preferences between anatase TiO 2 {001}
Structural and Catalytic Investigation of Active-Site Isolation in Pd-Ga Intermetallic Compounds
 Structural and Catalytic Investigation of Active-Site Isolation in Pd-Ga Intermetallic Compounds, Rainer Giedigkeit, Kirill Kovnir, Rolf E. Jentoft, Marc Armbrüster, Yuri Grin, Robert Schlögl, Thorsten
Structural and Catalytic Investigation of Active-Site Isolation in Pd-Ga Intermetallic Compounds, Rainer Giedigkeit, Kirill Kovnir, Rolf E. Jentoft, Marc Armbrüster, Yuri Grin, Robert Schlögl, Thorsten
Rh 3d. Co 2p. Binding Energy (ev) Binding Energy (ev) (b) (a)
 Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Co 2p Co(0) 778.3 Rh 3d Rh (0) 307.2 810 800 790 780 770 Binding Energy (ev) (a) 320 315 310 305 Binding Energy (ev) (b) Supplementary Figure 1 Photoemission features of a catalyst precursor which was
Supporting Information. Graphene Oxide-Palladium Modified Ag-AgBr: A Novel Visible-Light- Responsive Photocatalyst for the Suzuki Coupling Reaction**
 Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Graphene Oxide-Palladium Modified Ag-AgBr: A Novel Visible-Light- Responsive
Electronic Supplementary Material (ESI) for RSC Advances. This journal is The Royal Society of Chemistry 2014 Supporting Information Graphene Oxide-Palladium Modified Ag-AgBr: A Novel Visible-Light- Responsive
Multiply twinned Pt Pd nanoicosahedrons as highly active electrocatalyst for methanol oxidation
 Supporting Information for Multiply twinned Pt Pd nanoicosahedrons as highly active electrocatalyst for methanol oxidation An-Xiang Yin, Xiao-Quan Min, Wei Zhu, Hao-Shuai Wu, Ya-Wen Zhang* and Chun-Hua
Supporting Information for Multiply twinned Pt Pd nanoicosahedrons as highly active electrocatalyst for methanol oxidation An-Xiang Yin, Xiao-Quan Min, Wei Zhu, Hao-Shuai Wu, Ya-Wen Zhang* and Chun-Hua
Thermodynamic calculations on the catalytic growth of carbon nanotubes
 Thermodynamic calculations on the catalytic growth of carbon nanotubes Christian Klinke, Jean-Marc Bonard and Klaus Kern Ecole Polytechnique Federale de Lausanne, CH-05 Lausanne, Switzerland Max-Planck-Institut
Thermodynamic calculations on the catalytic growth of carbon nanotubes Christian Klinke, Jean-Marc Bonard and Klaus Kern Ecole Polytechnique Federale de Lausanne, CH-05 Lausanne, Switzerland Max-Planck-Institut
Magnetite decorated graphite nanoplatelets as cost effective CO 2 adsorbent
 Supplementary Information Magnetite decorated graphite nanoplatelets as cost effective CO 2 adsorbent Ashish Kumar Mishra and Sundara Ramaprabhu * Alternative Energy and Nanotechnology Laboratory (AENL),
Supplementary Information Magnetite decorated graphite nanoplatelets as cost effective CO 2 adsorbent Ashish Kumar Mishra and Sundara Ramaprabhu * Alternative Energy and Nanotechnology Laboratory (AENL),
Catalysis Lectures W.H. Green 5.68J/10.652J Spring Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework
 Catalysis Lectures W.H. Green 5.68J/10.652J Spring 2003 Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework Major points: 1) Why reactions have barriers, and how catalysts
Catalysis Lectures W.H. Green 5.68J/10.652J Spring 2003 Handouts: Norskov et al., J. Catalysis Imbihl and Ertl, Chem. Rev. (partial) Homework Major points: 1) Why reactions have barriers, and how catalysts
