|
|
|
- Emma Nicholson
- 5 years ago
- Views:
Transcription
1
2
3
4
5
6
7
8 Supplemental Figure and Movie Legends Figure S1. Time course experiments on the thermal stability of apo AT cpn-α by native PAGE (related to Figure 2B). The samples were heated, respectively, to (A) 45 C, (B) 50 C, (C) 55 C and (D) 60 C for 10 to 90 min before assayed by native PAGE. The lower bands represent monomers and the upper bands represent assemblies. Figure S2. ATP hydrolysis is not relevant to the thermal stabilities of AT thermosomes (related to Figure 3). (A, B) Thermal stability experiments on D400A mutants of cpn-α and cpn-β in absence (left) or presence (right) of ATP by native PAGE. (C) Thermal stability experiments on cpn-α (left) and cpn-β (right) with the existence of ADP. Figure S3. CryoEM reconstruction of AT cpn-α (related to Figure 4). (A) Raw cryoem micrograph of apo cpn-α (left) and profile of its power spectrum (right). The inset shows that the information limit is higher than 4.0 Å. (B) 2D classification of apo cpn-α. Endview particles with 8-fold symmetry are highlighted by the green rectangle and particles with 9-fold symmetry by the red rectangle. (C) Projection-matching diagrams for the reconstructions of apo cpn-α with 8- and 9-fold symmetries. (D) Structural comparison between 8-fold (magenta) and 9-fold (red) apo AT cpn-α. Superposition between the subunits from 8- and 9-fold cpn-α indicates their high similarity (left). Their packing modes within one ring are compared from top views (right) and the green arrow indicates the twisting angle between their subunits. Figure S4. The role of the lid domain in the thermal stability of AT cpn-α and cpn-β (related to Figure 4). (A) Thermal stability assays on the AT lidless cpn-α in the apo (left) and ATP-binding states (right). (B) Thermal stability assays on the AT lidless cpn-β in the apo (left) and ATP-binding states (right). Figure S5. Validation and reliability of the central density of AT cpn-β (related to Figure 5). (A) Volume rendering of cpn-β in the ATP-binding state (the cryoem map was filtered to 8.0 Å with a low-pass filter). (B) Asymmetric reconstruction of cpn-β in the ATP-binding state by using the low-pass filtered initial model with the central region
9 density masked out. Figure S6. The role of the N/C-termini for the thermal stability of AT thermosomes (related to Figure 6A-E). (A) Thermal stability assays of all the N/C-termini variants of AT cpn-α in the apo (left) and ATP-binding states (right). (B) Thermal stability assays of all the N/C-termini variants of AT cpn-β in the apo (left) and ATP-binding states (right). (C) The assembly of wild types and N/C-termini deletion mutants of AT cpn-α (left) and cpn-β (right) with (+) and without (-). Figure S7. Thermo stability assay of AT cpn-α and cpn-β in different ph values (related to Figure 6F). (A) Thermal stability assays on AT cpn-α in the apo (left) and ATP-binding states (right) in ph10.5, 9.0, 7.5, 6.0 and 4.5. (B) Thermal stability assays on AT cpn-β in the apo (left) and ATP-binding states (right) in ph10.5, 9.0, 7.5, 6.0 and 4.5. Movie S1. Volume rendering of AT cpn-β in the ATP-binding state. The cryoem map was filtered to 8.0 Å with a low-pass filter. Movie S2. Surface and volume rendering of AT cpn-β in the ATP-binding state from the asymmetric reconstruction.
10 Table S1. Statistics of the cryoem reconstructions and data entries Sample name AT apo cpn-α (8 fold) AT apo cpn-α (9 fold) AT apo cpn-β (9 fold) AT ATP-cpn-β (9 fold) MAG 1 Binn ing Pixel size Number of particles used Resolution (Å) at FSC 2 =0.5 Resolution (Å) at FSC 2 =0.143 EMDB 3 code (EMD-) PDB 4 code J1B J1C J1E J1F 1. MAG, nominal magnification. 2. FSC, Fourier shell correlation coefficient. 3. EMDB, electron microscopy data bank. 4. PDB, protein data bank.
11 Table S2. Summary of the N- and C-termini deletion and swapping variations of AT cpn-α and cpn-β. Sample name cpn-α-nδcδ cpn-α-nδcα cpn-α-nδcβ cpn-α-nαcδ cpn-α-nαcα cpn-α-nαcβ cpn-α-nβcδ cpn-α-nβcα cpn-α-nβcβ Explanation Main body of cpn-α, N- and C-termini deletion Main body of cpn-α, N-termini deletion, C-termini from cpn-α Main body of cpn-α, N-termini deletion, C-termini from cpn-β Main body of cpn-α, N-termini from cpn-α, C-termini deletion cpn-α itself Main body of cpn-α, N-termini from cpn-α, C-termini are from cpn-β Main body of cpn-α, N-termini from cpn-β, C-termini deletion Main body of cpn-α, N-termini from cpn-β, C-termini from cpn-α Main body of cpn-α, N- and C-termini both from cpn-β cpn-β-nδcδ cpn-β-nδcα cpn-β-nδcβ cpn-β-nαcδ cpn-β-nαcα cpn-β-nαcβ cpn-β-nβcδ cpn-β-nβcα cpn-β-nβcβ Main body of cpn-β, N- and C-termini deletion Main body of cpn-β, N-termini deletion, C-termini from cpn-α Main body of cpn-β, N-termini deletion, C-termini from cpn-β Main body of cpn-β, N-termini from cpn-α, C-termini deletion Main body of cpn-β, N- and C-termini both from cpn-α Main body of cpn-β, N-termini from cpn-α, C-termini are from cpn-β Main body of cpn-β, N-termini from cpn-β, C-termini deletion Main body of cpn-β, N-termini from cpn-β, C-termini from cpn-α cpn-β itself Δ: N- or C-terminus deletion Nα: N-terminus of cpn-α, from M1 to S18 Cα: C-terminus of cpn-α, from S533 to S563 Nβ: N-terminus of cpn-β, from M1 to Y27 Cβ: C-terminus of cpn-β, from G533 to D553
Purification, SDS-PAGE and cryo-em characterization of the MCM hexamer and Cdt1 MCM heptamer samples.
 Supplementary Figure 1 Purification, SDS-PAGE and cryo-em characterization of the MCM hexamer and Cdt1 MCM heptamer samples. (a-b) SDS-PAGE analysis of the hexamer and heptamer samples. The eluted hexamer
Supplementary Figure 1 Purification, SDS-PAGE and cryo-em characterization of the MCM hexamer and Cdt1 MCM heptamer samples. (a-b) SDS-PAGE analysis of the hexamer and heptamer samples. The eluted hexamer
SUPPLEMENTARY INFORMATION
 Supplementary materials Figure S1 Fusion protein of Sulfolobus solfataricus SRP54 and a signal peptide. a, Expression vector for the fusion protein. The signal peptide of yeast dipeptidyl aminopeptidase
Supplementary materials Figure S1 Fusion protein of Sulfolobus solfataricus SRP54 and a signal peptide. a, Expression vector for the fusion protein. The signal peptide of yeast dipeptidyl aminopeptidase
SUPPLEMENTARY INFORMATION
 SUPPLMTARY IFORMATIO a doi:10.108/nature10402 b 100 nm 100 nm c SAXS Model d ulers assigned to reference- Back-projected free class averages class averages Refinement against single particles Reconstructed
SUPPLMTARY IFORMATIO a doi:10.108/nature10402 b 100 nm 100 nm c SAXS Model d ulers assigned to reference- Back-projected free class averages class averages Refinement against single particles Reconstructed
Supplemental Data SUPPLEMENTAL FIGURES
 Supplemental Data CRYSTAL STRUCTURE OF THE MG.ADP-INHIBITED STATE OF THE YEAST F 1 C 10 ATP SYNTHASE Alain Dautant*, Jean Velours and Marie-France Giraud* From Université Bordeaux 2, CNRS; Institut de
Supplemental Data CRYSTAL STRUCTURE OF THE MG.ADP-INHIBITED STATE OF THE YEAST F 1 C 10 ATP SYNTHASE Alain Dautant*, Jean Velours and Marie-France Giraud* From Université Bordeaux 2, CNRS; Institut de
SUPPLEMENTARY INFORMATION
 www.nature.com/nature 1 Figure S1 Sequence alignment. a Structure based alignment of the plgic of E. chrysanthemi (ELIC), the acetylcholine binding protein from the snail Lymnea stagnalis (AchBP, PDB code
www.nature.com/nature 1 Figure S1 Sequence alignment. a Structure based alignment of the plgic of E. chrysanthemi (ELIC), the acetylcholine binding protein from the snail Lymnea stagnalis (AchBP, PDB code
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/10/eaat8797/dc1 Supplementary Materials for Single-molecule observation of nucleotide induced conformational changes in basal SecA-ATP hydrolysis Nagaraju Chada,
advances.sciencemag.org/cgi/content/full/4/10/eaat8797/dc1 Supplementary Materials for Single-molecule observation of nucleotide induced conformational changes in basal SecA-ATP hydrolysis Nagaraju Chada,
Supplementary Information: Long range allosteric regulation of the human 26S proteasome by 20S proteasome-targeting cancer drugs
 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Information: Long range allosteric regulation of the human 26S proteasome by 20S proteasome-targeting cancer drugs
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 Supplementary Information: Long range allosteric regulation of the human 26S proteasome by 20S proteasome-targeting cancer drugs
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. Purification of yeast CKM. (a) Silver-stained SDS-PAGE analysis of CKM purified through a TAP-tag engineered into the Cdk8 C-terminus. (b) Kinase activity
Supplementary Figures Supplementary Figure 1. Purification of yeast CKM. (a) Silver-stained SDS-PAGE analysis of CKM purified through a TAP-tag engineered into the Cdk8 C-terminus. (b) Kinase activity
SUPPLEMENTARY INFORMATION
 Supplementary Table 1: Amplitudes of three current levels. Level 0 (pa) Level 1 (pa) Level 2 (pa) TrkA- TrkH WT 200 K 0.01 ± 0.01 9.5 ± 0.01 18.7 ± 0.03 200 Na * 0.001 ± 0.01 3.9 ± 0.01 12.5 ± 0.03 200
Supplementary Table 1: Amplitudes of three current levels. Level 0 (pa) Level 1 (pa) Level 2 (pa) TrkA- TrkH WT 200 K 0.01 ± 0.01 9.5 ± 0.01 18.7 ± 0.03 200 Na * 0.001 ± 0.01 3.9 ± 0.01 12.5 ± 0.03 200
type GroEL-GroES complex. Crystals were grown in buffer D (100 mm HEPES, ph 7.5,
 Supplementary Material Supplementary Materials and Methods Structure Determination of SR1-GroES-ADP AlF x SR1-GroES-ADP AlF x was purified as described in Materials and Methods for the wild type GroEL-GroES
Supplementary Material Supplementary Materials and Methods Structure Determination of SR1-GroES-ADP AlF x SR1-GroES-ADP AlF x was purified as described in Materials and Methods for the wild type GroEL-GroES
SUPPLEMENTARY INFORMATION. doi: /nature07461
 Figure S1 Electrophysiology. a ph-activation of. Two-electrode voltage clamp recordings of Xenopus oocytes expressing in comparison to waterinjected oocytes. Currents were recorded at 40 mv. The ph of
Figure S1 Electrophysiology. a ph-activation of. Two-electrode voltage clamp recordings of Xenopus oocytes expressing in comparison to waterinjected oocytes. Currents were recorded at 40 mv. The ph of
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1737 Supplementary Table 1 variant Description FSEC - 2B12 a FSEC - 6A1 a K d (leucine) c Leucine uptake e K (wild-type like) K (Y18F) K (TS) K (TSY) K288A mutant, lipid facing side chain
doi:1.138/nature1737 Supplementary Table 1 variant Description FSEC - 2B12 a FSEC - 6A1 a K d (leucine) c Leucine uptake e K (wild-type like) K (Y18F) K (TS) K (TSY) K288A mutant, lipid facing side chain
PDBe TUTORIAL. PDBePISA (Protein Interfaces, Surfaces and Assemblies)
 PDBe TUTORIAL PDBePISA (Protein Interfaces, Surfaces and Assemblies) http://pdbe.org/pisa/ This tutorial introduces the PDBePISA (PISA for short) service, which is a webbased interactive tool offered by
PDBe TUTORIAL PDBePISA (Protein Interfaces, Surfaces and Assemblies) http://pdbe.org/pisa/ This tutorial introduces the PDBePISA (PISA for short) service, which is a webbased interactive tool offered by
SUPPLEMENTARY INFORMATION
 Dph2 SeMet (iron-free) # Dph2 (iron-free) Dph2-[4Fe-4S] Data collection Space group P2 1 2 1 2 1 P2 1 2 1 2 1 P2 1 2 1 2 1 Cell dimensions a, b, c (Å) 58.26, 82.08, 160.42 58.74, 81.87, 160.01 55.70, 80.53,
Dph2 SeMet (iron-free) # Dph2 (iron-free) Dph2-[4Fe-4S] Data collection Space group P2 1 2 1 2 1 P2 1 2 1 2 1 P2 1 2 1 2 1 Cell dimensions a, b, c (Å) 58.26, 82.08, 160.42 58.74, 81.87, 160.01 55.70, 80.53,
Supplemental Information. Molecular Basis of Spectral Diversity. in Near-Infrared Phytochrome-Based. Fluorescent Proteins
 Chemistry & Biology, Volume 22 Supplemental Information Molecular Basis of Spectral Diversity in Near-Infrared Phytochrome-Based Fluorescent Proteins Daria M. Shcherbakova, Mikhail Baloban, Sergei Pletnev,
Chemistry & Biology, Volume 22 Supplemental Information Molecular Basis of Spectral Diversity in Near-Infrared Phytochrome-Based Fluorescent Proteins Daria M. Shcherbakova, Mikhail Baloban, Sergei Pletnev,
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Structure of human carbamoyl phosphate synthetase: deciphering the on/off switch of human ureagenesis Sergio de Cima, Luis M. Polo, Carmen Díez-Fernández, Ana I. Martínez, Javier
SUPPLEMENTARY INFORMATION Structure of human carbamoyl phosphate synthetase: deciphering the on/off switch of human ureagenesis Sergio de Cima, Luis M. Polo, Carmen Díez-Fernández, Ana I. Martínez, Javier
Full wwpdb/emdatabank EM Map/Model Validation Report i
 Full wwpdb/emdatabank EM Map/Model Validation Report i Sep 25, 2018 07:01 PM EDT PDB ID : 6C0V EMDB ID: : EMD-7325 Title : Molecular structure of human P-glycoprotein in the ATP-bound, outwardfacing conformation
Full wwpdb/emdatabank EM Map/Model Validation Report i Sep 25, 2018 07:01 PM EDT PDB ID : 6C0V EMDB ID: : EMD-7325 Title : Molecular structure of human P-glycoprotein in the ATP-bound, outwardfacing conformation
ParM filament images were extracted and from the electron micrographs and
 Supplemental methods Outline of the EM reconstruction: ParM filament images were extracted and from the electron micrographs and straightened. The digitized images were corrected for the phase of the Contrast
Supplemental methods Outline of the EM reconstruction: ParM filament images were extracted and from the electron micrographs and straightened. The digitized images were corrected for the phase of the Contrast
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11054 Supplementary Fig. 1 Sequence alignment of Na v Rh with NaChBac, Na v Ab, and eukaryotic Na v and Ca v homologs. Secondary structural elements of Na v Rh are indicated above the
doi:10.1038/nature11054 Supplementary Fig. 1 Sequence alignment of Na v Rh with NaChBac, Na v Ab, and eukaryotic Na v and Ca v homologs. Secondary structural elements of Na v Rh are indicated above the
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11085 Supplementary Tables: Supplementary Table 1. Summary of crystallographic and structure refinement data Structure BRIL-NOP receptor Data collection Number of crystals 23 Space group
doi:10.1038/nature11085 Supplementary Tables: Supplementary Table 1. Summary of crystallographic and structure refinement data Structure BRIL-NOP receptor Data collection Number of crystals 23 Space group
Expanded View Figures
 The EMBO Journal Structure of a Dm peptide bound to the OT module Tobias Raisch et al Expanded View Figures A Hs Dm 262 297 685 8 HEAT HEAT MIF4G 9BD 1SHD 761 91 193 169 1152 1317 16 1376 1467 HEAT HEAT
The EMBO Journal Structure of a Dm peptide bound to the OT module Tobias Raisch et al Expanded View Figures A Hs Dm 262 297 685 8 HEAT HEAT MIF4G 9BD 1SHD 761 91 193 169 1152 1317 16 1376 1467 HEAT HEAT
Model building and validation for cryo-em maps at low resolution
 International Workshop of Advanced Image Processing of Cryo-Electron Microscopy 2015 Tsinghua University, Beijing, China June 3-7, 2015 Model building and validation for cryo-em maps at low resolution
International Workshop of Advanced Image Processing of Cryo-Electron Microscopy 2015 Tsinghua University, Beijing, China June 3-7, 2015 Model building and validation for cryo-em maps at low resolution
Cryo-EM data collection, refinement and validation statistics
 1 Table S1 Cryo-EM data collection, refinement and validation statistics Data collection and processing CPSF-160 WDR33 (EMDB-7114) (PDB 6BM0) CPSF-160 WDR33 (EMDB-7113) (PDB 6BLY) CPSF-160 WDR33 CPSF-30
1 Table S1 Cryo-EM data collection, refinement and validation statistics Data collection and processing CPSF-160 WDR33 (EMDB-7114) (PDB 6BM0) CPSF-160 WDR33 (EMDB-7113) (PDB 6BLY) CPSF-160 WDR33 CPSF-30
of the Guanine Nucleotide Exchange Factor FARP2
 Structure, Volume 21 Supplemental Information Structural Basis for Autoinhibition of the Guanine Nucleotide Exchange Factor FARP2 Xiaojing He, Yi-Chun Kuo, Tyler J. Rosche, and Xuewu Zhang Inventory of
Structure, Volume 21 Supplemental Information Structural Basis for Autoinhibition of the Guanine Nucleotide Exchange Factor FARP2 Xiaojing He, Yi-Chun Kuo, Tyler J. Rosche, and Xuewu Zhang Inventory of
SUPPLEMENTARY FIGURES. Structure of the cholera toxin secretion channel in its. closed state
 SUPPLEMENTARY FIGURES Structure of the cholera toxin secretion channel in its closed state Steve L. Reichow 1,3, Konstantin V. Korotkov 1,3, Wim G. J. Hol 1$ and Tamir Gonen 1,2$ 1, Department of Biochemistry
SUPPLEMENTARY FIGURES Structure of the cholera toxin secretion channel in its closed state Steve L. Reichow 1,3, Konstantin V. Korotkov 1,3, Wim G. J. Hol 1$ and Tamir Gonen 1,2$ 1, Department of Biochemistry
Supplemental Information. The Mitochondrial Fission Receptor MiD51. Requires ADP as a Cofactor
 Structure, Volume 22 Supplemental Information The Mitochondrial Fission Receptor MiD51 Requires ADP as a Cofactor Oliver C. Losón, Raymond Liu, Michael E. Rome, Shuxia Meng, Jens T. Kaiser, Shu-ou Shan,
Structure, Volume 22 Supplemental Information The Mitochondrial Fission Receptor MiD51 Requires ADP as a Cofactor Oliver C. Losón, Raymond Liu, Michael E. Rome, Shuxia Meng, Jens T. Kaiser, Shu-ou Shan,
Diphthamide biosynthesis requires a radical iron-sulfur enzyme. Pennsylvania State University, University Park, Pennsylvania 16802, USA
 Diphthamide biosynthesis requires a radical iron-sulfur enzyme Yang Zhang, 1,4 Xuling Zhu, 1,4 Andrew T. Torelli, 1 Michael Lee, 2 Boris Dzikovski, 1 Rachel Koralewski, 1 Eileen Wang, 1 Jack Freed, 1 Carsten
Diphthamide biosynthesis requires a radical iron-sulfur enzyme Yang Zhang, 1,4 Xuling Zhu, 1,4 Andrew T. Torelli, 1 Michael Lee, 2 Boris Dzikovski, 1 Rachel Koralewski, 1 Eileen Wang, 1 Jack Freed, 1 Carsten
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/6/e1700147/dc1 Supplementary Materials for Ribosome rearrangements at the onset of translational bypassing Xabier Agirrezabala, Ekaterina Samatova, Mariia Klimova,
advances.sciencemag.org/cgi/content/full/3/6/e1700147/dc1 Supplementary Materials for Ribosome rearrangements at the onset of translational bypassing Xabier Agirrezabala, Ekaterina Samatova, Mariia Klimova,
Sunhats for plants. How plants detect dangerous ultraviolet rays
 Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
Sunhats for plants How plants detect dangerous ultraviolet rays Anyone who has ever suffered sunburn will know about the effects of too much ultraviolet (UV) radiation, in particular UV-B (from 280-315
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11991 Supplementary Figure 1 - Refinement strategy for PIC intermediate assemblies by negative stain EM. The cryo-negative stain structure of free Pol II 1 (a) was used as initial reference
doi:10.1038/nature11991 Supplementary Figure 1 - Refinement strategy for PIC intermediate assemblies by negative stain EM. The cryo-negative stain structure of free Pol II 1 (a) was used as initial reference
Supporting Information
 Supporting Information Ottmann et al. 10.1073/pnas.0907587106 Fig. S1. Primary structure alignment of SBT3 with C5 peptidase from Streptococcus pyogenes. The Matchmaker tool in UCSF Chimera (http:// www.cgl.ucsf.edu/chimera)
Supporting Information Ottmann et al. 10.1073/pnas.0907587106 Fig. S1. Primary structure alignment of SBT3 with C5 peptidase from Streptococcus pyogenes. The Matchmaker tool in UCSF Chimera (http:// www.cgl.ucsf.edu/chimera)
THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN
 THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN HSP90 AND BOTH SCF E3 UBIQUITIN LIGASES AND KINETOCHORES Oliver Willhoft, Richard Kerr, Dipali Patel, Wenjuan Zhang, Caezar Al-Jassar, Tina
THE CRYSTAL STRUCTURE OF THE SGT1-SKP1 COMPLEX: THE LINK BETWEEN HSP90 AND BOTH SCF E3 UBIQUITIN LIGASES AND KINETOCHORES Oliver Willhoft, Richard Kerr, Dipali Patel, Wenjuan Zhang, Caezar Al-Jassar, Tina
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature12045 Supplementary Table 1 Data collection and refinement statistics. Native Pt-SAD X-ray source SSRF BL17U SPring-8 BL41XU Wavelength (Å) 0.97947 1.07171 Space group P2 1 2 1 2 1 P2
doi:10.1038/nature12045 Supplementary Table 1 Data collection and refinement statistics. Native Pt-SAD X-ray source SSRF BL17U SPring-8 BL41XU Wavelength (Å) 0.97947 1.07171 Space group P2 1 2 1 2 1 P2
Nature Structural & Molecular Biology: doi: /nsmb Supplementary Figure 1
 Supplementary Figure 1 Cryo-EM structure and model of the C. thermophilum 90S preribosome. a, Gold standard FSC curve showing the average resolution of the 90S preribosome masked and unmasked (left). FSC
Supplementary Figure 1 Cryo-EM structure and model of the C. thermophilum 90S preribosome. a, Gold standard FSC curve showing the average resolution of the 90S preribosome masked and unmasked (left). FSC
Supporting Protocol This protocol describes the construction and the force-field parameters of the non-standard residue for the Ag + -site using CNS
 Supporting Protocol This protocol describes the construction and the force-field parameters of the non-standard residue for the Ag + -site using CNS CNS input file generatemetal.inp: remarks file generate/generatemetal.inp
Supporting Protocol This protocol describes the construction and the force-field parameters of the non-standard residue for the Ag + -site using CNS CNS input file generatemetal.inp: remarks file generate/generatemetal.inp
Simulating the conformational transitions in Mm-cpn with Steered Molecular Dynamics. Technical report by G. Pintilie last updated March 2008
 Simulating the conformational transitions in Mm-cpn with Steered Molecular Dynamics Technical report by G. Pintilie last updated March 2008 G. Pintilie, Electrical Engineering and Computer Science, MIT
Simulating the conformational transitions in Mm-cpn with Steered Molecular Dynamics Technical report by G. Pintilie last updated March 2008 G. Pintilie, Electrical Engineering and Computer Science, MIT
R-factor-fit R-factor-non-fit
 Supplementary Figures and Legends R-factor-fit R-factor-non-fit R-factor-non-fit (overall) Supplementary figure 1 The areas where the R-factor-fit and the R-factor-non-fit were calculated. The edges of
Supplementary Figures and Legends R-factor-fit R-factor-non-fit R-factor-non-fit (overall) Supplementary figure 1 The areas where the R-factor-fit and the R-factor-non-fit were calculated. The edges of
Supplemental Information. Expanded Coverage of the 26S Proteasome. Conformational Landscape Reveals. Mechanisms of Peptidase Gating
 Cell Reports, Volume 24 Supplemental Information Expanded Coverage of the 26S Proteasome Conformational Landscape Reveals Mechanisms of Peptidase Gating Markus R. Eisele, Randi G. Reed, Till Rudack, Andreas
Cell Reports, Volume 24 Supplemental Information Expanded Coverage of the 26S Proteasome Conformational Landscape Reveals Mechanisms of Peptidase Gating Markus R. Eisele, Randi G. Reed, Till Rudack, Andreas
Structural basis for catalytically restrictive dynamics of a high-energy enzyme state
 Supplementary Material Structural basis for catalytically restrictive dynamics of a high-energy enzyme state Michael Kovermann, Jörgen Ådén, Christin Grundström, A. Elisabeth Sauer-Eriksson, Uwe H. Sauer
Supplementary Material Structural basis for catalytically restrictive dynamics of a high-energy enzyme state Michael Kovermann, Jörgen Ådén, Christin Grundström, A. Elisabeth Sauer-Eriksson, Uwe H. Sauer
Recognition of the amber UAG stop codon by release factor RF1
 Manuscript EMBO-2010-73984 Recognition of the amber UAG stop codon by release factor RF1 Andrei Korostelev, Jianyu Zhu, Haruichi Asahara and Harry F. Noller Corresponding author: Harry F. Noller, Univ.
Manuscript EMBO-2010-73984 Recognition of the amber UAG stop codon by release factor RF1 Andrei Korostelev, Jianyu Zhu, Haruichi Asahara and Harry F. Noller Corresponding author: Harry F. Noller, Univ.
SUPPLEMENTARY INFORMATION
 Supplementary Table 1: Data collection, phasing and refinement statistics ChbC/Ta 6 Br 12 Native ChbC Data collection Space group P4 3 2 1 2 P4 3 2 1 2 Cell dimensions a, c (Å) 132.75, 453.57 132.81, 452.95
Supplementary Table 1: Data collection, phasing and refinement statistics ChbC/Ta 6 Br 12 Native ChbC Data collection Space group P4 3 2 1 2 P4 3 2 1 2 Cell dimensions a, c (Å) 132.75, 453.57 132.81, 452.95
Spliceosome and Localization of Its Catalytic Core
 Molecular Cell, Volume 40 Supplemental Information 3D Cryo-EM Structure of an Active Step I Spliceosome and Localization of Its Catalytic Core Monika M. Golas, Bjoern Sander, Sergey Bessonov, Michael Grote,
Molecular Cell, Volume 40 Supplemental Information 3D Cryo-EM Structure of an Active Step I Spliceosome and Localization of Its Catalytic Core Monika M. Golas, Bjoern Sander, Sergey Bessonov, Michael Grote,
Structure and RNA-binding properties. of the Not1 Not2 Not5 module of the yeast Ccr4 Not complex
 Structure and RNA-binding properties of the Not1 Not2 Not5 module of the yeast Ccr4 Not complex Varun Bhaskar 1, Vladimir Roudko 2,3, Jerome Basquin 1, Kundan Sharma 4, Henning Urlaub 4, Bertrand Seraphin
Structure and RNA-binding properties of the Not1 Not2 Not5 module of the yeast Ccr4 Not complex Varun Bhaskar 1, Vladimir Roudko 2,3, Jerome Basquin 1, Kundan Sharma 4, Henning Urlaub 4, Bertrand Seraphin
Supporting Information
 Supporting Information Oxaliplatin binding to human copper chaperone Atox1 and protein dimerization Benny D. Belviso, 1 Angela Galliani, 2 Alessia Lasorsa, 2 Valentina Mirabelli, 1,3 Rocco Caliandro, 1
Supporting Information Oxaliplatin binding to human copper chaperone Atox1 and protein dimerization Benny D. Belviso, 1 Angela Galliani, 2 Alessia Lasorsa, 2 Valentina Mirabelli, 1,3 Rocco Caliandro, 1
Supplementary Figure 1. Aligned sequences of yeast IDH1 (top) and IDH2 (bottom) with isocitrate
 SUPPLEMENTARY FIGURE LEGENDS Supplementary Figure 1. Aligned sequences of yeast IDH1 (top) and IDH2 (bottom) with isocitrate dehydrogenase from Escherichia coli [ICD, pdb 1PB1, Mesecar, A. D., and Koshland,
SUPPLEMENTARY FIGURE LEGENDS Supplementary Figure 1. Aligned sequences of yeast IDH1 (top) and IDH2 (bottom) with isocitrate dehydrogenase from Escherichia coli [ICD, pdb 1PB1, Mesecar, A. D., and Koshland,
Image Assessment San Diego, November 2005
 Image Assessment San Diego, November 005 Pawel A. Penczek The University of Texas Houston Medical School Department of Biochemistry and Molecular Biology 6431 Fannin, MSB6.18, Houston, TX 77030, USA phone:
Image Assessment San Diego, November 005 Pawel A. Penczek The University of Texas Houston Medical School Department of Biochemistry and Molecular Biology 6431 Fannin, MSB6.18, Houston, TX 77030, USA phone:
Structure of the quaternary complex between SRP, SR, and translocon bound to the translating ribosome
 Structure of the quaternary complex between SRP, SR, and translocon bound to the translating ribosome Ahmad Jomaa 1, Yu-Hsien Hwang Fu 2, Daniel Boehringer 1, Marc Leibundgut 1, Shu-ou Shan 2, and Nenad
Structure of the quaternary complex between SRP, SR, and translocon bound to the translating ribosome Ahmad Jomaa 1, Yu-Hsien Hwang Fu 2, Daniel Boehringer 1, Marc Leibundgut 1, Shu-ou Shan 2, and Nenad
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/NCHEM.1290 Metal-directed, chemically tunable assembly of one-, two- and threedimensional crystalline protein arrays 1 2 1 1 1,3 Jeffrey D. Brodin 1, X. I. Ambroggio 2, Chunyan Tang 1, Kristin
DOI: 10.1038/NCHEM.1290 Metal-directed, chemically tunable assembly of one-, two- and threedimensional crystalline protein arrays 1 2 1 1 1,3 Jeffrey D. Brodin 1, X. I. Ambroggio 2, Chunyan Tang 1, Kristin
Molecular Mechanism for Conformational Dynamics of Ras GTP Elucidated from In-Situ Structural Transition in Crystal
 Molecular Mechanism for Conformational Dynamics of Ras GTP Elucidated from In-Situ Structural Transition in Crystal Shigeyuki Matsumoto, Nao Miyano, Seiki Baba, Jingling Liao, Takashi Kawamura, Chiemi
Molecular Mechanism for Conformational Dynamics of Ras GTP Elucidated from In-Situ Structural Transition in Crystal Shigeyuki Matsumoto, Nao Miyano, Seiki Baba, Jingling Liao, Takashi Kawamura, Chiemi
Nature Methods: doi: /nmeth Supplementary Figure 1. Atomic models in cryo-em maps.
 Supplementary Figure 1 Atomic models in cryo-em maps. (a) Two alpha (blue) and two beta (green) subunits of the T20S proteasome are shown as cartoon tubes fitted in a 3.2 Å potential map at isolevel 0.25
Supplementary Figure 1 Atomic models in cryo-em maps. (a) Two alpha (blue) and two beta (green) subunits of the T20S proteasome are shown as cartoon tubes fitted in a 3.2 Å potential map at isolevel 0.25
Computational Structural Biology and Molecular Simulation. Introduction to VMD Molecular Visualization and Analysis
 Computational Structural Biology and Molecular Simulation Introduction to VMD Molecular Visualization and Analysis Emad Tajkhorshid Department of Biochemistry, Beckman Institute, Center for Computational
Computational Structural Biology and Molecular Simulation Introduction to VMD Molecular Visualization and Analysis Emad Tajkhorshid Department of Biochemistry, Beckman Institute, Center for Computational
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11524 Supplementary discussion Functional analysis of the sugar porter family (SP) signature motifs. As seen in Fig. 5c, single point mutation of the conserved
SUPPLEMENTARY INFORMATION doi:10.1038/nature11524 Supplementary discussion Functional analysis of the sugar porter family (SP) signature motifs. As seen in Fig. 5c, single point mutation of the conserved
Supporting Information Converter domain mutations in myosin alter structural kinetics and motor function. Hershey, PA, MN 55455, USA
 Supporting Information Converter domain mutations in myosin alter structural kinetics and motor function Laura K. Gunther 1, John A. Rohde 2, Wanjian Tang 1, Shane D. Walton 1, William C. Unrath 1, Darshan
Supporting Information Converter domain mutations in myosin alter structural kinetics and motor function Laura K. Gunther 1, John A. Rohde 2, Wanjian Tang 1, Shane D. Walton 1, William C. Unrath 1, Darshan
Measuring quaternary structure similarity using global versus local measures.
 Supplementary Figure 1 Measuring quaternary structure similarity using global versus local measures. (a) Structural similarity of two protein complexes can be inferred from a global superposition, which
Supplementary Figure 1 Measuring quaternary structure similarity using global versus local measures. (a) Structural similarity of two protein complexes can be inferred from a global superposition, which
NGF - twenty years a-growing
 NGF - twenty years a-growing A molecule vital to brain growth It is twenty years since the structure of nerve growth factor (NGF) was determined [ref. 1]. This molecule is more than 'quite interesting'
NGF - twenty years a-growing A molecule vital to brain growth It is twenty years since the structure of nerve growth factor (NGF) was determined [ref. 1]. This molecule is more than 'quite interesting'
SUPPLEMENTARY INFORMATION
 Table of Contents Page Supplementary Table 1. Diffraction data collection statistics 2 Supplementary Table 2. Crystallographic refinement statistics 3 Supplementary Fig. 1. casic1mfc packing in the R3
Table of Contents Page Supplementary Table 1. Diffraction data collection statistics 2 Supplementary Table 2. Crystallographic refinement statistics 3 Supplementary Fig. 1. casic1mfc packing in the R3
RNA Polymerase I Contains a TFIIF-Related DNA-Binding Subcomplex
 Molecular Cell, Volume 39 Supplemental Information RNA Polymerase I Contains a TFIIFRelated DNABinding Subcomplex Sebastian R. Geiger, Kristina Lorenzen, Amelie Schreieck, Patrizia Hanecker, Dirk Kostrewa,
Molecular Cell, Volume 39 Supplemental Information RNA Polymerase I Contains a TFIIFRelated DNABinding Subcomplex Sebastian R. Geiger, Kristina Lorenzen, Amelie Schreieck, Patrizia Hanecker, Dirk Kostrewa,
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/5/243/ra68/dc1 Supplementary Materials for Superbinder SH2 Domains Act as Antagonists of Cell Signaling Tomonori Kaneko, Haiming Huang, Xuan Cao, Xing Li, Chengjun
www.sciencesignaling.org/cgi/content/full/5/243/ra68/dc1 Supplementary Materials for Superbinder SH2 Domains Act as Antagonists of Cell Signaling Tomonori Kaneko, Haiming Huang, Xuan Cao, Xing Li, Chengjun
Closing the Folding Chamber of the Eukaryotic Chaperonin Requires the Transition State of ATP Hydrolysis
 Cell, Vol. 113, 369 381, May 2, 2003, Copyright 2003 by Cell Press Closing the Folding Chamber of the Eukaryotic Chaperonin Requires the Transition State of ATP Hydrolysis Anne S. Meyer, 1,5 Joel R. Gillespie,
Cell, Vol. 113, 369 381, May 2, 2003, Copyright 2003 by Cell Press Closing the Folding Chamber of the Eukaryotic Chaperonin Requires the Transition State of ATP Hydrolysis Anne S. Meyer, 1,5 Joel R. Gillespie,
Nature Structural and Molecular Biology: doi: /nsmb Supplementary Figure 1. Definition and assessment of ciap1 constructs.
 Supplementary Figure 1 Definition and assessment of ciap1 constructs. (a) ciap1 constructs used in this study are shown as primary structure schematics with domains colored as in the main text. Mutations
Supplementary Figure 1 Definition and assessment of ciap1 constructs. (a) ciap1 constructs used in this study are shown as primary structure schematics with domains colored as in the main text. Mutations
SI Text S1 Solution Scattering Data Collection and Analysis. SI references
 SI Text S1 Solution Scattering Data Collection and Analysis. The X-ray photon energy was set to 8 kev. The PILATUS hybrid pixel array detector (RIGAKU) was positioned at a distance of 606 mm from the sample.
SI Text S1 Solution Scattering Data Collection and Analysis. The X-ray photon energy was set to 8 kev. The PILATUS hybrid pixel array detector (RIGAKU) was positioned at a distance of 606 mm from the sample.
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/310/5753/1513/dc1 Supporting Online Material for Structural Roles for Human Translation Factor eif3 in Initiation of Protein Synthesis Bunpote Siridechadilok, Christopher
www.sciencemag.org/cgi/content/full/310/5753/1513/dc1 Supporting Online Material for Structural Roles for Human Translation Factor eif3 in Initiation of Protein Synthesis Bunpote Siridechadilok, Christopher
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
DOI: 10.1038/ncb2215 Figure S1 Number of egfp-vps4a bursts versus cellular expression levels. The total number of egfp-vps4a bursts, counted at the end of each movie (frame 2000, after 1h 28 min) are plotted
Plasmid Relevant features Source. W18N_D20N and TrXE-W18N_D20N-anti
 Table S1. E. coli plasmids Plasmid Relevant features Source pdg680 T. reesei XynII AA 2-190 with C-terminal His 6 tag optimized for E. coli expression in pjexpress401 Wan et al. (in press) psbn44d psbn44h
Table S1. E. coli plasmids Plasmid Relevant features Source pdg680 T. reesei XynII AA 2-190 with C-terminal His 6 tag optimized for E. coli expression in pjexpress401 Wan et al. (in press) psbn44d psbn44h
Supporting Information for. Jesinghaus, Rachael Barry, Zemer Gitai, Justin Kollman and Enoch P. Baldwin
 Supporting Information for Inhibition of E. coli CTP synthetase by NADH and other nicotinamides, and their mutual interactions with CTP and GTP Chris Habrian, Adithi Chandrasekhara, Bita Shahrvini, Brian
Supporting Information for Inhibition of E. coli CTP synthetase by NADH and other nicotinamides, and their mutual interactions with CTP and GTP Chris Habrian, Adithi Chandrasekhara, Bita Shahrvini, Brian
Electron microscopy in molecular cell biology II
 Electron microscopy in molecular cell biology II Cryo-EM and image processing Werner Kühlbrandt Max Planck Institute of Biophysics Sample preparation for cryo-em Preparation laboratory Specimen preparation
Electron microscopy in molecular cell biology II Cryo-EM and image processing Werner Kühlbrandt Max Planck Institute of Biophysics Sample preparation for cryo-em Preparation laboratory Specimen preparation
Transmembrane Domains (TMDs) of ABC transporters
 Transmembrane Domains (TMDs) of ABC transporters Most ABC transporters contain heterodimeric TMDs (e.g. HisMQ, MalFG) TMDs show only limited sequence homology (high diversity) High degree of conservation
Transmembrane Domains (TMDs) of ABC transporters Most ABC transporters contain heterodimeric TMDs (e.g. HisMQ, MalFG) TMDs show only limited sequence homology (high diversity) High degree of conservation
Supplementary Information. Structural basis for precursor protein-directed ribosomal peptide macrocyclization
 Supplementary Information Structural basis for precursor protein-directed ribosomal peptide macrocyclization Kunhua Li 1,3, Heather L. Condurso 1,3, Gengnan Li 1, Yousong Ding 2 and Steven D. Bruner 1*
Supplementary Information Structural basis for precursor protein-directed ribosomal peptide macrocyclization Kunhua Li 1,3, Heather L. Condurso 1,3, Gengnan Li 1, Yousong Ding 2 and Steven D. Bruner 1*
Detailed description of overall and active site architecture of PPDC- 3dThDP, PPDC-2HE3dThDP, PPDC-3dThDP-PPA and PPDC- 3dThDP-POVA
 Online Supplemental Results Detailed description of overall and active site architecture of PPDC- 3dThDP, PPDC-2HE3dThDP, PPDC-3dThDP-PPA and PPDC- 3dThDP-POVA Structure solution and overall architecture
Online Supplemental Results Detailed description of overall and active site architecture of PPDC- 3dThDP, PPDC-2HE3dThDP, PPDC-3dThDP-PPA and PPDC- 3dThDP-POVA Structure solution and overall architecture
Protein structure analysis. Risto Laakso 10th January 2005
 Protein structure analysis Risto Laakso risto.laakso@hut.fi 10th January 2005 1 1 Summary Various methods of protein structure analysis were examined. Two proteins, 1HLB (Sea cucumber hemoglobin) and 1HLM
Protein structure analysis Risto Laakso risto.laakso@hut.fi 10th January 2005 1 1 Summary Various methods of protein structure analysis were examined. Two proteins, 1HLB (Sea cucumber hemoglobin) and 1HLM
Supplementary Figures
 Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Supplementary Figures Supplementary Figure 1. The asymmetric unit in para-iodio-phenylalanine crystal. The 50% probability ellipsoid representation was prepared using the Mercury Software. Colors are as
Nature Methods: doi: /nmeth Supplementary Figure 1
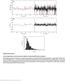 Supplementary Figure 1 Control experiments on the fluorescence stability of single Atto647N-biotin complexes. a, Exemplary signal trace (black line) in counts per second (cps) of a single Atto647N molecule
Supplementary Figure 1 Control experiments on the fluorescence stability of single Atto647N-biotin complexes. a, Exemplary signal trace (black line) in counts per second (cps) of a single Atto647N molecule
Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached
 Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached to HpUreI. Urea hydrolysis products 2NH 3 and 1CO 2
Supplementary Figure S1. Urea-mediated buffering mechanism of H. pylori. Gastric urea is funneled to a cytoplasmic urease that is presumably attached to HpUreI. Urea hydrolysis products 2NH 3 and 1CO 2
Dr. Yonca Yuzugullu PERG (Protein Engineering Research Group)
 Dr. Yonca Yuzugullu PERG (Protein Engineering Research Group) BSc, 1997 Ankara University, Turkey PhD, 2010 Middle East Technical University, Turkey Lecturer in Department of Biology Kocaeli University,
Dr. Yonca Yuzugullu PERG (Protein Engineering Research Group) BSc, 1997 Ankara University, Turkey PhD, 2010 Middle East Technical University, Turkey Lecturer in Department of Biology Kocaeli University,
Supplementary Figure 1 Crystal contacts in COP apo structure (PDB code 3S0R)
 Supplementary Figure 1 Crystal contacts in COP apo structure (PDB code 3S0R) Shown in cyan and green are two adjacent tetramers from the crystallographic lattice of COP, forming the only unique inter-tetramer
Supplementary Figure 1 Crystal contacts in COP apo structure (PDB code 3S0R) Shown in cyan and green are two adjacent tetramers from the crystallographic lattice of COP, forming the only unique inter-tetramer
Acta Crystallographica Section D
 Supporting information Acta Crystallographica Section D Volume 70 (2014) Supporting information for article: Structural characterization of the virulence factor Nuclease A from Streptococcus agalactiae
Supporting information Acta Crystallographica Section D Volume 70 (2014) Supporting information for article: Structural characterization of the virulence factor Nuclease A from Streptococcus agalactiae
Supplementary Figure 1. Biochemical and sequence alignment analyses the
 Supplementary Figure 1. Biochemical and sequence alignment analyses the interaction of OPTN and TBK1. (a) Analytical gel filtration chromatography analysis of the interaction between TBK1 CTD and OPTN(1-119).
Supplementary Figure 1. Biochemical and sequence alignment analyses the interaction of OPTN and TBK1. (a) Analytical gel filtration chromatography analysis of the interaction between TBK1 CTD and OPTN(1-119).
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/3/4/e1600663/dc1 Supplementary Materials for A dynamic hydrophobic core orchestrates allostery in protein kinases Jonggul Kim, Lalima G. Ahuja, Fa-An Chao, Youlin
advances.sciencemag.org/cgi/content/full/3/4/e1600663/dc1 Supplementary Materials for A dynamic hydrophobic core orchestrates allostery in protein kinases Jonggul Kim, Lalima G. Ahuja, Fa-An Chao, Youlin
Hands-on Course in Computational Structural Biology and Molecular Simulation BIOP590C/MCB590C. Course Details
 Hands-on Course in Computational Structural Biology and Molecular Simulation BIOP590C/MCB590C Emad Tajkhorshid Center for Computational Biology and Biophysics Email: emad@life.uiuc.edu or tajkhors@uiuc.edu
Hands-on Course in Computational Structural Biology and Molecular Simulation BIOP590C/MCB590C Emad Tajkhorshid Center for Computational Biology and Biophysics Email: emad@life.uiuc.edu or tajkhors@uiuc.edu
SUPPLEMENTARY INFORMATION
 Supplementary Results DNA binding property of the SRA domain was examined by an electrophoresis mobility shift assay (EMSA) using synthesized 12-bp oligonucleotide duplexes containing unmodified, hemi-methylated,
Supplementary Results DNA binding property of the SRA domain was examined by an electrophoresis mobility shift assay (EMSA) using synthesized 12-bp oligonucleotide duplexes containing unmodified, hemi-methylated,
Supplementary Materials belonging to
 Supplementary Materials belonging to Solution conformation of the 70 kda E.coli Hsp70 complexed with ADP and substrate. Eric B. Bertelsen 1, Lyra Chang 2, Jason E. Gestwicki 2 and Erik R. P. Zuiderweg
Supplementary Materials belonging to Solution conformation of the 70 kda E.coli Hsp70 complexed with ADP and substrate. Eric B. Bertelsen 1, Lyra Chang 2, Jason E. Gestwicki 2 and Erik R. P. Zuiderweg
Supplemental data for
 Supplemental data for A Real-Time Guanine Nucleotide Exchange Assay using NMR: Activation of RhoA by PDZ- RhoGEF. Geneviève M.C. Gasmi-Seabrook 1,3, Christopher B. Marshall 1,3, Melissa Cheung 1,3, Bryan
Supplemental data for A Real-Time Guanine Nucleotide Exchange Assay using NMR: Activation of RhoA by PDZ- RhoGEF. Geneviève M.C. Gasmi-Seabrook 1,3, Christopher B. Marshall 1,3, Melissa Cheung 1,3, Bryan
Cks1 CDK1 CDK1 CDK1 CKS1. are ice- lobe. conserved. conserved
 Cks1 d CKS1 Supplementary Figure 1 The -Cks1 crystal lattice. (a) Schematic of the - Cks1 crystal lattice. -Cks1 crystallizes in a lattice that contains c 4 copies of the t - Cks1 dimer in the crystallographic
Cks1 d CKS1 Supplementary Figure 1 The -Cks1 crystal lattice. (a) Schematic of the - Cks1 crystal lattice. -Cks1 crystallizes in a lattice that contains c 4 copies of the t - Cks1 dimer in the crystallographic
Protein Dynamics. The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron.
 Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
Protein Dynamics The space-filling structures of myoglobin and hemoglobin show that there are no pathways for O 2 to reach the heme iron. Below is myoglobin hydrated with 350 water molecules. Only a small
SUPPLEMENTARY INFORMATION
 Data collection Supplementary Table 1 Statistics of data collection, phasing and refinement Native Se-MAD Space group P2 1 2 1 2 1 P2 1 2 1 2 1 Cell dimensions a, b, c (Å) 50.4, 94.2, 115.4 49.8, 94.2,
Data collection Supplementary Table 1 Statistics of data collection, phasing and refinement Native Se-MAD Space group P2 1 2 1 2 1 P2 1 2 1 2 1 Cell dimensions a, b, c (Å) 50.4, 94.2, 115.4 49.8, 94.2,
The structure of vanadium nitrogenase reveals an unusual bridging ligand
 SUPPLEMENTARY INFORMATION The structure of vanadium nitrogenase reveals an unusual bridging ligand Daniel Sippel and Oliver Einsle Lehrstuhl Biochemie, Institut für Biochemie, Albert-Ludwigs-Universität
SUPPLEMENTARY INFORMATION The structure of vanadium nitrogenase reveals an unusual bridging ligand Daniel Sippel and Oliver Einsle Lehrstuhl Biochemie, Institut für Biochemie, Albert-Ludwigs-Universität
Supplementary Information. The protease GtgE from Salmonella exclusively targets. inactive Rab GTPases
 Supplementary Information The protease GtgE from Salmonella exclusively targets inactive Rab GTPases Table of Contents Supplementary Figures... 2 Supplementary Figure 1... 2 Supplementary Figure 2... 3
Supplementary Information The protease GtgE from Salmonella exclusively targets inactive Rab GTPases Table of Contents Supplementary Figures... 2 Supplementary Figure 1... 2 Supplementary Figure 2... 3
Supplementary figure 1. Comparison of unbound ogm-csf and ogm-csf as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine
 Supplementary figure 1. Comparison of unbound and as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine GM-CSF (slate) with bound GM-CSF in the GIF:GM-CSF complex (GIF: green,
Supplementary figure 1. Comparison of unbound and as captured in the GIF:GM-CSF complex. Alignment of two copies of unbound ovine GM-CSF (slate) with bound GM-CSF in the GIF:GM-CSF complex (GIF: green,
T H E J O U R N A L O F C E L L B I O L O G Y
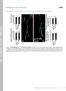 Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
Supplemental material Kasprowicz et al., http://www.jcb.org/cgi/content/full/jcb.201310090/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. NMJ morphology of shi 12-12B ; shi-4c not treated
SUPPLEMENTARY INFORMATION
 Fig. 1 Influences of crystal lattice contacts on Pol η structures. a. The dominant lattice contact between two hpol η molecules (silver and gold) in the type 1 crystals. b. A close-up view of the hydrophobic
Fig. 1 Influences of crystal lattice contacts on Pol η structures. a. The dominant lattice contact between two hpol η molecules (silver and gold) in the type 1 crystals. b. A close-up view of the hydrophobic
Structural and Molecular Basis for Coordination in a Viral. DNA Packaging Motor
 1 2 Structural and Molecular Basis for Coordination in a Viral DNA Packaging Motor 3 4 5 6 Huzhang Mao a*, Mitul Saha a*, Emilio Reyes-Aldrete a, Michael B. Sherman a, Michael Woodson a, Rockney Atz b,
1 2 Structural and Molecular Basis for Coordination in a Viral DNA Packaging Motor 3 4 5 6 Huzhang Mao a*, Mitul Saha a*, Emilio Reyes-Aldrete a, Michael B. Sherman a, Michael Woodson a, Rockney Atz b,
Supplementary Information. Overlap between folding and functional energy landscapes for. adenylate kinase conformational change
 Supplementary Information Overlap between folding and functional energy landscapes for adenylate kinase conformational change by Ulrika Olsson & Magnus Wolf-Watz Contents: 1. Supplementary Note 2. Supplementary
Supplementary Information Overlap between folding and functional energy landscapes for adenylate kinase conformational change by Ulrika Olsson & Magnus Wolf-Watz Contents: 1. Supplementary Note 2. Supplementary
Supporting Information
 Supporting Information Symmetry of Charge Partitioning in Collisional and UV Photon-induced Dissociation of Protein Assemblies Sem Tamara,,$, Andrey Dyachenko,,$, Kyle L. Fort,,$ Alexander Makarov,,# Richard
Supporting Information Symmetry of Charge Partitioning in Collisional and UV Photon-induced Dissociation of Protein Assemblies Sem Tamara,,$, Andrey Dyachenko,,$, Kyle L. Fort,,$ Alexander Makarov,,# Richard
Supplementary Figure 3 a. Structural comparison between the two determined structures for the IL 23:MA12 complex. The overall RMSD between the two
 Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Supplementary Figure 1. Biopanningg and clone enrichment of Alphabody binders against human IL 23. Positive clones in i phage ELISA with optical density (OD) 3 times higher than background are shown for
Nature Structural and Molecular Biology: doi: /nsmb.2938
 Supplementary Figure 1 Characterization of designed leucine-rich-repeat proteins. (a) Water-mediate hydrogen-bond network is frequently visible in the convex region of LRR crystal structures. Examples
Supplementary Figure 1 Characterization of designed leucine-rich-repeat proteins. (a) Water-mediate hydrogen-bond network is frequently visible in the convex region of LRR crystal structures. Examples
Nature Methods: doi: /nmeth Supplementary Figure 1. Overview of the density-guided rebuilding and refinement protocol.
 Supplementary Figure 1 Overview of the density-guided rebuilding and refinement protocol. (a) Flowchart of the protocol. The protocol uses 250 cycles of local backbone rebuilding, followed by five cycles
Supplementary Figure 1 Overview of the density-guided rebuilding and refinement protocol. (a) Flowchart of the protocol. The protocol uses 250 cycles of local backbone rebuilding, followed by five cycles
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature09450 Supplementary Table 1 Summary of kinetic parameters. Kinetic parameters were V = V / 1 K / ATP and obtained using the relationships max ( + m [ ]) V d s /( 1/ k [ ATP] + 1 k ) =,
doi:10.1038/nature09450 Supplementary Table 1 Summary of kinetic parameters. Kinetic parameters were V = V / 1 K / ATP and obtained using the relationships max ( + m [ ]) V d s /( 1/ k [ ATP] + 1 k ) =,
Phase problem: Determining an initial phase angle α hkl for each recorded reflection. 1 ρ(x,y,z) = F hkl cos 2π (hx+ky+ lz - α hkl ) V h k l
 Phase problem: Determining an initial phase angle α hkl for each recorded reflection 1 ρ(x,y,z) = F hkl cos 2π (hx+ky+ lz - α hkl ) V h k l Methods: Heavy atom methods (isomorphous replacement Hg, Pt)
Phase problem: Determining an initial phase angle α hkl for each recorded reflection 1 ρ(x,y,z) = F hkl cos 2π (hx+ky+ lz - α hkl ) V h k l Methods: Heavy atom methods (isomorphous replacement Hg, Pt)
Computational engineering of cellulase Cel9A-68 functional motions through mutations in its linker region. WT 1TF4 (crystal) -90 ERRAT PROVE VERIFY3D
 Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 218 Supplementary Material: Computational engineering of cellulase Cel9-68 functional
Electronic Supplementary Material (ESI) for Physical Chemistry Chemical Physics. This journal is the Owner Societies 218 Supplementary Material: Computational engineering of cellulase Cel9-68 functional
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature10955 Supplementary Figures Supplementary Figure 1. Electron-density maps and crystallographic dimer structures of the motor domain. (a f) Stereo views of the final electron-density maps
doi:10.1038/nature10955 Supplementary Figures Supplementary Figure 1. Electron-density maps and crystallographic dimer structures of the motor domain. (a f) Stereo views of the final electron-density maps
