Chapter 11 Outline: Ethers, Epoxides & Sulfides
|
|
|
- Domenic Atkins
- 5 years ago
- Views:
Transcription
1 Chapter 11 Outline: Ethers, Epoxides & Sulfides Review Nomenclature & Physical Properties on your own 1. Structure of Ethers & Epoxides 2. Preparation of Ethers & Thioethers 3. Reactions of Ethers 4. Preparation of Epoxides 5. Reactions of Epoxides 6. Practicing problems You should do all the problems in the textbook, but these are some suggested homework problems: , , 11.23, , 11.38, Brevetoxin B is a naturally occurring polyether exerts its neurotoxic effects by binding to sodium channels on nerve and muscle cell membranes, causing an excessive influx of sodium ions across the membranes, which leads to cell death. o It s synthesis was a 12-year effort by chemistry K. C. Nicolaou s group at Scripps Research Institute, (UC-San Diego J Am. Chem. Soc., 117, 1171 and 1173 (1995). o Structure that includes 11 trans-fused rings and 23 stereocenters. It was synthesized in 83 steps, overall yield of the synthesis was 0.043%; with average yield for each site was about 91%. Page 1 of 27
2 1. Structure & Physical Properties Which ones are strained? Crown Ethers: a special kind of cyclic ether 18-crown-6 12-crown-4 Page 2 of 27
3 What s the Utility of Crown Ethers? Page 3 of 27
4 2. Preparation Ethers & Thioethers a. Nucleophilic attack of RX with NaOR: in which direction does it lie? What mechanism does this undergo above? But how do you make a metal alkoxide like sodium ethoxide? Page 4 of 27
5 b. Williamson Ether Synthesis (WES) The WES involves a S N 2 reaction of a strong Nu and a suitable small RX. What is MTBE used for? MTBE = methyl tert-butl ether = One method of synthesis undergoes S N 2 (to afford an ether) and the other option will prefer E2 (to afford an alkene). Which synthetic route will provide S N 2? O Page 5 of 27
6 c. Acid-catalysed Dehydration of Alcohols Page 6 of 27
7 d. Acid-catalysed Addition of Alcohols to Alkenes Page 7 of 27
8 3. Reactions of Ethers -- how to make a good leaving group? a. Acid-catalyzed cleavage by concentrated HX (converting a ROR 2 RX) Determine the products for the following reactions: O excess HCl Page 8 of 27
9 Mechanism? Page 9 of 27
10 b. Silyl Ethers as Protecting Groups Sometimes one wants to transform one functional group in the presence of another, more acidic, functional group that they want to leave untouched. Who would be deprotonated first? Organic chemists use protecting groups for this purpose. Several reagents exist as protecting groups: Page 10 of 27
11 Silyl Ethers as Protecting Groups What is the driving force for the last step? Page 11 of 27
12 The precautionary tale of ethers: Low molecular weight ethers react with molecular oxygen to form peroxides which are explosive (no we will not be doing a demo of this!) If you see the precipitates at the bottom of the bottle, it should be removed by a bomb squad! Page 12 of 27
13 4. Preparation of Epoxides a. Synthesis of Epoxides Internal Nucleophilic Substitution in Halohydrins o Also thought of as an internal Williamson Ether Synthesis Mechanism? Page 13 of 27
14 b. Synthesis of Epoxides o Epoxidation occurs via syn (i.e. same side) addition of an O atom to either side of a planar double bond. o A cis alkene gives an epoxide with cis substituents. o A trans alkene gives an epoxide with trans substituents. Page 14 of 27
15 5. Reactions of Epoxides Due to ring strain, epoxides undergo a large number of ring opening reactions. There are only two reactions we will see: epoxides reacting in the presence of strong acid Since attack at either side occurs with equal probability, an equal amount of the two enantiomers is formed a racemic mixture. Optically inactive starting materials can give optically-active products. epoxides reacting in the presence of a strong Nu (absence of acid) What do you notice about the product stereochemistry? Anti or syn addition? Page 15 of 27
16 a. Acid-Catalyzed Ring Opening - In the presence of acid, an epoxide opens to form a glycol Reaction gives anti addition of nucleophile (i.e. trans products) Trans product is favored solely due to sterics (nucleophile attacks opposite side of where original epoxide points) Mechanism? Page 16 of 27
17 b. Acid-Catalyzed Ring Opening with HX- In the presence of HX, an epoxide opens and X - adds to MOST substituted carbon. o Reaction undergoes anti-addition to afford trans product o In the presence of acid, the Nu attacks at the more substituted C Mechanism? Page 17 of 27
18 c. Nucleophilic Ring Opening attacking an epoxide with a Nu Ethers are not usually susceptible to attack by strong nucleophiles alone (without first being protonated), but the ring strain in epoxides makes it undergo attack via S N 2 reactions more readily. O 1. NaBr 2. H 2 O HO Br o What overall two key features do you notice from above? o Mechanism? Page 18 of 27
19 6. Practicing Problems: O 1. NaBr 2. H 2 O HBr Mechanism? Mechanism? Page 19 of 27
20 A B C D E F G H I J K L M N O P Page 20 of 27
21 Determine the final product from the following steps: Page 21 of 27
22 Page 22 of 27
23 Page 23 of 27
24 Br 1. NaH 2. Cl 2 /H 2 O 3. NaH 4. HBr Page 24 of 27
25 Devise a synthesis for the following: Page 25 of 27
26 Page 26 of 27
27 Page 27 of 27
Chapter 11 - Alcohols and Ethers 1
 Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
Andrew Rosen Chapter 11 - Alcohols and Ethers 1 11.1 - Structure and Nomenclature - The common naming calls alcohols as alkyl alcohols (eg: methyl alcohol) - The common names of ethers have the groups
Ethers can be symmetrical or not:
 Chapter 14: Ethers, Epoxides, and Sulfides 175 Physical Properties Ethers can be symmetrical or not: linear or cyclic. Ethers are inert and make excellent solvents for organic reactions. Epoxides are very
Chapter 14: Ethers, Epoxides, and Sulfides 175 Physical Properties Ethers can be symmetrical or not: linear or cyclic. Ethers are inert and make excellent solvents for organic reactions. Epoxides are very
ORGANIC - CLUTCH CH ALCOHOLS, ETHERS, EPOXIDES AND THIOLS
 !! www.clutchprep.com CONCEPT: ALCOHOL NOMENCLATURE Glycols: Alcohols with two hydroxyls are called ; with three hydroxyls are called Always give most priority to the OH group. EXAMPLE: Provide the correct
!! www.clutchprep.com CONCEPT: ALCOHOL NOMENCLATURE Glycols: Alcohols with two hydroxyls are called ; with three hydroxyls are called Always give most priority to the OH group. EXAMPLE: Provide the correct
REACTION AND SYNTHESIS REVIEW
 REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
REACTION AND SYNTHESIS REVIEW A STUDENT SHOULD BE ABLE TO PREDICT PRODUCTS, IDENTIFY REACTANTS, GIVE REACTION CONDITIONS, PROPOSE SYNTHESES, AND PROPOSE MECHANISMS (AS LISTED BELOW). REVIEW THE MECHANISM
Ethers. Synthesis of Ethers. Chemical Properties of Ethers
 Page 1 of 6 like alcohols are organic derivatives of water, but lack the labile -OH group. As a result, ethers, except for epoxides, are usually not very reactive and are often used as solvents for organic
Page 1 of 6 like alcohols are organic derivatives of water, but lack the labile -OH group. As a result, ethers, except for epoxides, are usually not very reactive and are often used as solvents for organic
Alcohols, Ethers, & Epoxides
 Alcohols, Ethers, & Epoxides Alcohols Structure and Bonding Enols and Phenols Compounds having a hydroxy group on a sp 2 hybridized carbon enols and phenols undergo different reactions than alcohols. Chapter
Alcohols, Ethers, & Epoxides Alcohols Structure and Bonding Enols and Phenols Compounds having a hydroxy group on a sp 2 hybridized carbon enols and phenols undergo different reactions than alcohols. Chapter
Chapter 16. Ethers, Epoxides, and Sulfides. Class Notes. 2. Epoxides: cyclic ethers, 3-membered rings. 1. Symmetrical and unsymmetrical (mixed) ethers
 Chapter 16 Ethers, Epoxides, and Sulfides Chapter 16 suggested problems: 21, 25, 26, 31, 32, 33 Class Notes I. Nomenclature of ethers, epoxides, and sulfides A. General B. Ethers 1. Ethers: C-O-C 2. Epoxides:
Chapter 16 Ethers, Epoxides, and Sulfides Chapter 16 suggested problems: 21, 25, 26, 31, 32, 33 Class Notes I. Nomenclature of ethers, epoxides, and sulfides A. General B. Ethers 1. Ethers: C-O-C 2. Epoxides:
Chapter 8: Ethers and Epoxides. Diethyl ether in starting fluid
 Chapter 8: Ethers and Epoxides Diethyl ether in starting fluid 8.1 Nomenclature of Ethers Ethers are usually named by giving the name of each alkyl or aryl group, in alphabetical order, followed by the
Chapter 8: Ethers and Epoxides Diethyl ether in starting fluid 8.1 Nomenclature of Ethers Ethers are usually named by giving the name of each alkyl or aryl group, in alphabetical order, followed by the
Ch 18 Ethers and Epoxides
 Ch 18 Ethers and Epoxides Ethers (R-O-R ) are compounds with two organic groups attached to an sp 3 oxygen. Epoxides are cyclic ethers where the sp 3 O is a part of a 3-membered ring. Thiols (R-S-H ) and
Ch 18 Ethers and Epoxides Ethers (R-O-R ) are compounds with two organic groups attached to an sp 3 oxygen. Epoxides are cyclic ethers where the sp 3 O is a part of a 3-membered ring. Thiols (R-S-H ) and
2/26/18. Practice Questions. Practice Questions B F. How many steps are there in this reaction?
 Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Practice Questions Practice Questions D B F C E A G How many steps are there in this reaction? 1 Practice Questions D B F C E A G What is the highest-energy transitions state? Practice Questions D B F
Nuggets of Knowledge for Chapter 12 Alkenes (II) Chem reaction what is added to the C=C what kind of molecule results addition of HX HX only
 I. Addition Reactions of Alkenes Introduction Nuggets of Knowledge for Chapter 12 Alkenes (II) Chem 2310 An addition reaction always involves changing a double bond to a single bond and adding a new bond
I. Addition Reactions of Alkenes Introduction Nuggets of Knowledge for Chapter 12 Alkenes (II) Chem 2310 An addition reaction always involves changing a double bond to a single bond and adding a new bond
Learning Guide for Chapter 12 - Alkenes (II)
 Learning Guide for Chapter 12 - Alkenes (II) I. Addition reactions of alkenes Introduction to addition reactions Catalytic hydrogenation of alkenes Hydroxylation of alkenes Epoxidation of alkenes Cyclopropanation
Learning Guide for Chapter 12 - Alkenes (II) I. Addition reactions of alkenes Introduction to addition reactions Catalytic hydrogenation of alkenes Hydroxylation of alkenes Epoxidation of alkenes Cyclopropanation
(c) The intermediate carbocation resulting from 3-bromobut-1-ene is resonance stabilized.
 KT121 Sem II 09/10 SETIN B: 1.(a) Name of mechanism: E1 elimination. 7 marks (b) (i) + 3 3 3 (ii) + 3 (iii) l 8 marks (c) The intermediate carbocation resulting from 3-bromobut-1-ene is resonance stabilized.
KT121 Sem II 09/10 SETIN B: 1.(a) Name of mechanism: E1 elimination. 7 marks (b) (i) + 3 3 3 (ii) + 3 (iii) l 8 marks (c) The intermediate carbocation resulting from 3-bromobut-1-ene is resonance stabilized.
CHEM 263 Oct 25, stronger base stronger acid weaker acid weaker base
 CEM 263 ct 25, 2016 Reactions and Synthesis (Preparation) of R- Breaking the - Bond of R- with Metals R + Li 0 or Na 0 or K 0 metal R Li + 1/2 2 alkoxide Breaking the - Bond of R- by Acid-Base Reaction
CEM 263 ct 25, 2016 Reactions and Synthesis (Preparation) of R- Breaking the - Bond of R- with Metals R + Li 0 or Na 0 or K 0 metal R Li + 1/2 2 alkoxide Breaking the - Bond of R- by Acid-Base Reaction
Preparation of alkenes
 Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
Lecture 11 אלקנים הכנה ותגובות של אלקנים: הידרוגנציה, סיפוח הידרוהלוגנים )כלל מארקובניקוב(, סיפוח הלוגנים והסטראוכימיה של תוצרי הסיפוח, הידרובורציה, אפוקסידציה, אוזונוליזה. 1 Preparation of alkenes 1.
1. Radical Substitution on Alkanes. 2. Radical Substitution with Alkenes. 3. Electrophilic Addition
 1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
1. Radical Substitution on Alkanes Only Cl and Br are useful at the laboratory level. Alkane reactivity: tertiary > secondary > primary > methyl Numbers below products give their relative yield. Relative
Organic Chemistry. Alkenes
 Organic Chemistry by Nurlin Abu Samah, Dr. Md. Shaheen & Dr. Nadeem Akhtar Faculty of Industrial Sciences & Technology nurlin@ump.edu.my Chapter Description Aims The students should understand the fundamental
Organic Chemistry by Nurlin Abu Samah, Dr. Md. Shaheen & Dr. Nadeem Akhtar Faculty of Industrial Sciences & Technology nurlin@ump.edu.my Chapter Description Aims The students should understand the fundamental
water methanol dimethyl ether Ether can only act as a hydrogen bond acceptor H-bond acceptor O R
 Chapter 14: Ethers and Epoxides; Thiols and Sulfides 14.1 Introduction to Ethers An ether group is an oxygen atom that is bonded to two carbons. The ether carbons can be part of alkyl, aryl, or vinyl groups.
Chapter 14: Ethers and Epoxides; Thiols and Sulfides 14.1 Introduction to Ethers An ether group is an oxygen atom that is bonded to two carbons. The ether carbons can be part of alkyl, aryl, or vinyl groups.
Organic Chemistry Lecture 2 - Hydrocarbons, Alcohols, Substitutions
 ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
ALKANES Water-insoluble, low density C-C single bonds Higher MW -> higher BP, higher MP Branching -> lower BP, higher MP Forms cycloalkanes which can have ring strain Cyclohexane: chair vs. boat configuration
Alcohols, Ethers and Epoxides. Chapter Organic Chemistry, 8th Edition John McMurry
 Alcohols, Ethers and Epoxides Chapter 17-18 Organic Chemistry, 8th Edition John McMurry 1 Introduction Structure and Bonding Alcohols contain a hydroxy group (OH) bonded to an sp 3 hybridized carbon. 2
Alcohols, Ethers and Epoxides Chapter 17-18 Organic Chemistry, 8th Edition John McMurry 1 Introduction Structure and Bonding Alcohols contain a hydroxy group (OH) bonded to an sp 3 hybridized carbon. 2
Chapter 7. Alkenes: Reactions and Synthesis
 Chapter 7. Alkenes: Reactions and Synthesis 1 Synthesis of Alkenes: Elimination Reactions 1. Dehydrohalogenation of alkyl halides. loss of requires CH 2 CH 2 Cl Zaitsev s Rule: CH 2 C 2. Dehydration of
Chapter 7. Alkenes: Reactions and Synthesis 1 Synthesis of Alkenes: Elimination Reactions 1. Dehydrohalogenation of alkyl halides. loss of requires CH 2 CH 2 Cl Zaitsev s Rule: CH 2 C 2. Dehydration of
Organic Chemistry. Alkenes (2)
 For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
For updated version, please click on http://ocw.ump.edu.my Organic Chemistry Alkenes (2) by Dr. Seema Zareen & Dr. Izan Izwan Misnon Faculty Industrial Science & Technology seema@ump.edu.my & iezwan@ump.edu.my
CHEM 242 ETHERS, EPOXIDES, AND SULFIDES CHAP 17 ASSIGN
 CHEM 242 ETHERS, EPOXIDES, AND SULFIDES CHAP 17 ASSIGN 1. Which of the following is the correct name of the compound below? A) benzyl ethyl ether B) ethyl phenyl ether C) ethylbenzene ether D) ethoxy phenoxy
CHEM 242 ETHERS, EPOXIDES, AND SULFIDES CHAP 17 ASSIGN 1. Which of the following is the correct name of the compound below? A) benzyl ethyl ether B) ethyl phenyl ether C) ethylbenzene ether D) ethoxy phenoxy
Organic Chemistry I: Reactions and Overview
 Organic Chemistry I: Reactions and Overview Andrew Rosen Editor: Raghav Malik January 13, 2013 Contents I Library of Synthetic Reactions 3 II Organic Trends and Essentials 4 1 The Basics: Bonding and Molecular
Organic Chemistry I: Reactions and Overview Andrew Rosen Editor: Raghav Malik January 13, 2013 Contents I Library of Synthetic Reactions 3 II Organic Trends and Essentials 4 1 The Basics: Bonding and Molecular
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides"
 Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 6 Ionic Reactions-Nucleophilic Substitution and Elimination Reactions of Alkyl Halides" t Introduction" The polarity of a carbon-halogen bond leads to the carbon having a partial positive charge"
Chapter 8 Alkenes and Alkynes II: Addition Reactions
 Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8 Alkenes and Alkynes II: Addition Reactions Introduction: Additions to Alkenes Generally the reaction is exothermic because one π and one σ bond are converted to two σ bonds The π electrons of
Chapter 8 Alkenes and Alkynes II: Addition Reactions. Alkenes are electron rich. Additions to Alkenes
 Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
Additions to Alkenes Chapter 8 Alkenes and Alkynes II: Addition Reactions Generally the reaction is exothermic because one p and one s bond are converted to two s bonds Alkenes are electron rich The carbocation
CHEMISTRY 231 GENERAL ORGANIC CHEMISTRY I FALL 2014 List of Topics / Examination Schedule
 Page 1 of 5 CHEMISTRY 231 FALL 2014 List of Topics / Examination Schedule Unit Starts Topic of Study 20 Aug 2014 STRUCTURE AND BONDING Suggested Reading: Chapter 1 29 Aug 2014 ALKANES & CYCLOALKANES Suggested
Page 1 of 5 CHEMISTRY 231 FALL 2014 List of Topics / Examination Schedule Unit Starts Topic of Study 20 Aug 2014 STRUCTURE AND BONDING Suggested Reading: Chapter 1 29 Aug 2014 ALKANES & CYCLOALKANES Suggested
Alcohol Synthesis. Dr. Sapna Gupta
 Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
Alcohol Synthesis Dr. Sapna Gupta Synthesis of Alcohols Alcohols can be synthesized from several functional groups. Nucleophilic substitution of O - on alkyl halide ydration of alkenes water in acid solution
Organic Chemistry, Second Edition. Janice Gorzynski Smith University of Hawai i. Chapter 10 Alkenes
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Alkenes Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Alkenes Prepared by Rabi Ann Musah State University of New York at Albany Copyright The McGraw-Hill Companies,
Chapter 16: Ethers, Epoxides, and Sulfides
 Chapter 16: Ethers, Epoxides, and Sulfides 16.1: omenclature of Ethers, Epoxides, and Sulfides (Please read) 16.2: Structure and Bonding in Ethers and Epoxides The ether oxygen is sp 3 -hybridized and
Chapter 16: Ethers, Epoxides, and Sulfides 16.1: omenclature of Ethers, Epoxides, and Sulfides (Please read) 16.2: Structure and Bonding in Ethers and Epoxides The ether oxygen is sp 3 -hybridized and
Chapter 4. Reactions of alkenes. Addition reactions Carbocations Selectivity of reactions
 Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
Chapter 4 Reactions of alkenes Addition reactions Carbocations Selectivity of reactions Prob 47 p192. Give the reagents that would be required (including catalyst). Ch 4 #2 Electrophilic addition Ch 4
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Lecture Notes Chem 51C S. King. Chapter 20 Introduction to Carbonyl Chemistry; Organometallic Reagents; Oxidation & Reduction
 Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
Learning Guide for Chapter 11 - Alkenes I
 Learning Guide for Chapter 11 - Alkenes I I. Introduction to alkenes - p 1 bond structure, classifying alkenes, reactivity, physical properties, occurrences and uses, spectroscopy, stabilty II. Unsaturation
Learning Guide for Chapter 11 - Alkenes I I. Introduction to alkenes - p 1 bond structure, classifying alkenes, reactivity, physical properties, occurrences and uses, spectroscopy, stabilty II. Unsaturation
Chem 251 Fall Learning Objectives
 Learning Objectives Chapter 8 (last semester) 1. Write an electron-pushing mechanism for an SN2 reaction between an alkyl halide and a nucleophile. 2. Describe the rate law and relative rate of reaction
Learning Objectives Chapter 8 (last semester) 1. Write an electron-pushing mechanism for an SN2 reaction between an alkyl halide and a nucleophile. 2. Describe the rate law and relative rate of reaction
Aldehydes and Ketones : Aldol Reactions
 Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
Aldehydes and Ketones : Aldol Reactions The Acidity of the a Hydrogens of Carbonyl Compounds: Enolate Anions Hydrogens on carbons a to carbonyls are unusually acidic The resulting anion is stabilized by
But in organic terms: Oxidation: loss of H 2 ; addition of O or O 2 ; addition of X 2 (halogens).
 Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Reactions of Alcohols Alcohols are versatile organic compounds since they undergo a wide variety of transformations the majority of which are either oxidation or reduction type reactions. Normally: Oxidation
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Chapter 10 Outline: Alcohols
 Chapter 10 Outline: Alcohols Review Nomenclature, Structure & Physical Properties on your own 1. Acidity of Alcohols 2. Reactions of Alcohols 3. Reactions of Thiols You should do all the problems in the
Chapter 10 Outline: Alcohols Review Nomenclature, Structure & Physical Properties on your own 1. Acidity of Alcohols 2. Reactions of Alcohols 3. Reactions of Thiols You should do all the problems in the
Chapter 9. Nucleophilic Substitution and ß-Elimination
 Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Chapter 9 Nucleophilic Substitution and ß-Elimination Nucleophilic Substitution Nucleophile: From the Greek meaning nucleus loving. A molecule or ion that donates a pair of electrons to another atom or
Organic Reactions Susbstitution S N. Dr. Sapna Gupta
 Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
Organic Reactions Susbstitution S N 2 Dr. Sapna Gupta Kinetics of Nucleophilic Reaction Rate law is order of reaction 0 order is when rate of reaction is unaffected by change in concentration of the reactants
1) (100 pts) 5) (20 pts) 3) (35 pts) 4) (25pts. Total (200 pts) More Tutorial at
 Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Name: Perm number: Question 1) (100 pts) 2) (20 pts) 3) (35 pts) 4) (25pts 5) (20 pts) Total (200 pts) Your score 1. MULTIPLE CHOICE. Choose the one alternative that best completes the statement or answers
Module9. Nuclear Magnetic Resonance Spectroscopy Nuclear Magnetic Resonance (NMR) spectroscopy - Chemical shift - Integration of signal area
 1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
1 CHEMISTRY 263 HOME WORK Lecture Topics: Module7. Hydrogenation of Alkenes The Function of the Catalyst - Syn and anti- addition Hydrogenation of Alkynes - Syn- addition of hydrogen: Synthesis of cis-alkenes
Chapter 8 - Alkenes and Alkynes II Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles
 Andrew Rosen Chapter 8 - Alkenes and Alkynes II 8.1 - Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles 8.2 - Electrophilic Addition of Hydrogen Halides to Alkenes:
Andrew Rosen Chapter 8 - Alkenes and Alkynes II 8.1 - Addition Reactions of Alkenes - Electrons in the π bond of alkenes react with electrophiles 8.2 - Electrophilic Addition of Hydrogen Halides to Alkenes:
The Chemistry of Ethers, Epoxides, Glycols, and Sulfides
 The Chemistry of Ethers, Epoxides, Glycols, and Sulfides The chemistry of ethers is closely intertwined with the chemistry of alkyl halides, alcohols, and alkenes. Ethers, however, are considerably less
The Chemistry of Ethers, Epoxides, Glycols, and Sulfides The chemistry of ethers is closely intertwined with the chemistry of alkyl halides, alcohols, and alkenes. Ethers, however, are considerably less
DAMIETTA UNIVERSITY. Energy Diagram of One-Step Exothermic Reaction
 DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 5 Dr Ali El-Agamey 1 Energy Diagram of One-Step Exothermic Reaction The vertical axis in this graph represents the potential energy. The transition
DAMIETTA UNIVERSITY CHEM-103: BASIC ORGANIC CHEMISTRY LECTURE 5 Dr Ali El-Agamey 1 Energy Diagram of One-Step Exothermic Reaction The vertical axis in this graph represents the potential energy. The transition
Alcohols: Contain a hydroxy group( OH) bonded to an sp 2 or sp 3 hybridized
 Lecture Notes hem 51B S. King hapter 9 Alcohols, Ethers, and Epoxides I. Introduction Alcohols, ether, and epoxides are 3 functional groups that contain σ-bonds. Alcohols: ontain a hydroxy group( ) bonded
Lecture Notes hem 51B S. King hapter 9 Alcohols, Ethers, and Epoxides I. Introduction Alcohols, ether, and epoxides are 3 functional groups that contain σ-bonds. Alcohols: ontain a hydroxy group( ) bonded
2. Which functional groups and structural features are present in the following molecule (strychnine)?
 Chapter 1-2: 1. Which of the following species has a negative charge but NO lone pair of valance shell nonbonding electrons? [all atoms have complete valance shell of electrons, but lone pairs are not
Chapter 1-2: 1. Which of the following species has a negative charge but NO lone pair of valance shell nonbonding electrons? [all atoms have complete valance shell of electrons, but lone pairs are not
Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry
 Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry 1 Structure and Bonding 2 Structure and Bonding Rotation around the C=C bond is restricted 90 rotation The p orbitals are orthogonal
Alkenes (Olefins) Chapters 7 & 8 Organic Chemistry, 8 th Edition John McMurry 1 Structure and Bonding 2 Structure and Bonding Rotation around the C=C bond is restricted 90 rotation The p orbitals are orthogonal
Chapter 10 Lecture Outline
 Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
Organic Chemistry, Fifth Edition Janice Gorzynski Smith University of Hawai i Chapter 10 Lecture Outline Modified by Dr. Juliet Hahn Copyright 2017 McGraw-Hill Education. All rights reserved. No reproduction
Physical Properties. Alcohols can be: CH CH 2 OH CH 2 CH 3 C OH CH 3. Secondary alcohol. Primary alcohol. Tertiary alcohol
 Chapter 10: Structure and Synthesis of Alcohols 100 Physical Properties Alcohols can be: CH 3 CH 3 CH CH 2 OH * Primary alcohol CH 3 OH CH * CH 2 CH 3 Secondary alcohol CH 3 CH 3 * C OH CH 3 Tertiary alcohol
Chapter 10: Structure and Synthesis of Alcohols 100 Physical Properties Alcohols can be: CH 3 CH 3 CH CH 2 OH * Primary alcohol CH 3 OH CH * CH 2 CH 3 Secondary alcohol CH 3 CH 3 * C OH CH 3 Tertiary alcohol
Chemistry 210 Organic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar
 Chemistry 210 Organic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar Examination #3 Elimination Reactions Structure, Synthesis and Reactions of Alkenes and alkynes. Friday, July 23, 1999, 8:40 9:40
Chemistry 210 Organic Chemistry I Summer Semester 1999 Dr. Somnath Sarkar Examination #3 Elimination Reactions Structure, Synthesis and Reactions of Alkenes and alkynes. Friday, July 23, 1999, 8:40 9:40
LECTURE #14 Thurs., Oct.20, Midterm exam: Tues.Oct.25 during class Ch.1, , 7.10, 2, Sections
 CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
CHEM 221 section 01 LECTURE #14 Thurs., Oct.20, 2005 Midterm exam: Tues.Oct.25 during class Ch.1, 7.2-7.5, 7.10, 2, 3.1-3.5 ASSIGNED READINGS: TODAY S CLASS: NEXT LECTURE: Sections 4.7-4.10 finish Ch.4,
Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm
 - 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
- 1 - Chemistry 3351 Organic Chemistry/Final Exam Monday: Dec. 17 th from 1:30 pm 4:00pm Name: (please print, 1 pt) Page Possible Points Score 1 1 2 9 3 10 4 12 5 14 6 14 7 10 8 15 9 10 10 10 11 10 12
Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides"
 Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides The (E)-(Z) System for Designating Alkene Diastereomers The Cahn-Ingold-Prelog convention is used to assign
Chapter 7 Alkenes and Alkynes I: Properties and Synthesis Elimination Reactions of Alkyl Halides The (E)-(Z) System for Designating Alkene Diastereomers The Cahn-Ingold-Prelog convention is used to assign
Lecture Notes Chem 51B S. King I. Conjugation
 Lecture Notes Chem 51B S. King Chapter 16 Conjugation, Resonance, and Dienes I. Conjugation Conjugation occurs whenever p-orbitals can overlap on three or more adjacent atoms. Conjugated systems are more
Lecture Notes Chem 51B S. King Chapter 16 Conjugation, Resonance, and Dienes I. Conjugation Conjugation occurs whenever p-orbitals can overlap on three or more adjacent atoms. Conjugated systems are more
Chem Lecture 23 Page- 1 -
 Chem 230-001 Lecture 23 Page- 1 - ydroboration/ oxidation - acemic trans-2-methylcyclohexanol may be neatly synthesized from 1-methyl- 1-cyclohexene by using a reagent called borane, 3. 0.33 equivs. 3,
Chem 230-001 Lecture 23 Page- 1 - ydroboration/ oxidation - acemic trans-2-methylcyclohexanol may be neatly synthesized from 1-methyl- 1-cyclohexene by using a reagent called borane, 3. 0.33 equivs. 3,
Chapter 7 - Alkenes and Alkynes I
 Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
For example, 2-ethyloctan-3-ol which can also be called. 2-ethyl-3-octanol.
 Alcohols & Ethers Worksheet Part A Name: The focus of this chapter can be thought of as the organic version of water. When H 2 has one or both of its hydrogens replaced by an R group we get alcohols and
Alcohols & Ethers Worksheet Part A Name: The focus of this chapter can be thought of as the organic version of water. When H 2 has one or both of its hydrogens replaced by an R group we get alcohols and
CHEMISTRY 263 HOME WORK
 Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Lecture Topics: CHEMISTRY 263 HOME WORK Module7: Hydrogenation of Alkenes Hydrogenation - syn and anti- addition - hydrogenation of alkynes - synthesis of cis-alkenes -synthesis of trans-alkenes Text sections:
Chemistry 35 Exam 2 Answers - April 9, 2007
 Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Chemistry 35 Exam 2 Answers - April 9, 2007 Problem 1. (14 points) Provide the products for the reactions shown below. If stereochemistry is important, be sure to indicate stereochemistry clearly. If no
Ethers & Epoxides. Chapter 5. Dr. Seham ALTERARY. Chem 340-2nd semester
 Ethers & Epoxides Chapter 5 Dr. Seham ALTERARY Chapter s out line Ethers Definition; General formula; Classification and Types Nomenclature - Common Names. - IUPAC Naming. Physical Properties General methods
Ethers & Epoxides Chapter 5 Dr. Seham ALTERARY Chapter s out line Ethers Definition; General formula; Classification and Types Nomenclature - Common Names. - IUPAC Naming. Physical Properties General methods
Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy
 Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy Conjugated unsaturated systems have a p orbital on a carbon adjacent to a double bond The p orbital can come from another double (e.g.
Chapter 15: Conjugated Systems, Orbital Symmetry, and UV Spectroscopy Conjugated unsaturated systems have a p orbital on a carbon adjacent to a double bond The p orbital can come from another double (e.g.
Chem ORGANIC CHEMISTRY I
 ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
ORGANIC CHEMISTRY I CHEM 221 /4 02 Final Examination April 20, 2005 0900-1200 Dr. Cerrie ROGERS x x periodic table & pk a data table provided non-programmable calculators allowed molecular model kits allowed
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I. 5 Credit Hours. Prepared by: Richard A. Pierce
 JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
JEFFERSON COLLEGE COURSE SYLLABUS CHM200 ORGANIC CHEMISTRY I 5 Credit Hours Prepared by: Richard A. Pierce Revised Date: January 2008 by Ryan H. Groeneman Arts & Science Education Dr. Mindy Selsor, Dean
ELECTROPHILIC ADDITIONS OF ALKENES AS THE COUNTERPART OF ELIMINATIONS
 ELECTRPHILIC ADDITINS F ALKENES AS THE CUNTERPART F ELIMINATINS INTRDUCTIN - Chapter 8 is mostly about alkene reactions. That is, how one can transform alkenes into other functional groups. Most of these
ELECTRPHILIC ADDITINS F ALKENES AS THE CUNTERPART F ELIMINATINS INTRDUCTIN - Chapter 8 is mostly about alkene reactions. That is, how one can transform alkenes into other functional groups. Most of these
Double and Triple Bonds. The addition of an electrophile and a
 Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
Chapter 11 Additions to Carbon-Carbon Double and Triple Bonds The addition of an electrophile and a nucleophile to a C-C C double or triple bonds 11.1 The General Mechanism Pi electrons of the double bond
KOT 222 Organic Chemistry II
 KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
KOT 222 Organic Chemistry II Course Objectives: 1) To introduce the chemistry of alcohols and ethers. 2) To study the chemistry of functional groups. 3) To learn the chemistry of aromatic compounds and
11.5 PREPARATION AND OXIDATIVE CLEAVAGE OF GLYCOLS
 11.5 PEPAATIN AND XIDATIVE EAVAGE F GY 50 These reactions also provide methods for the formation of carbon carbon bonds. eactions that form carbon carbon bonds are especially important in organic chemistry
11.5 PEPAATIN AND XIDATIVE EAVAGE F GY 50 These reactions also provide methods for the formation of carbon carbon bonds. eactions that form carbon carbon bonds are especially important in organic chemistry
Chem 112A: Final Exam
 Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
Chem 112A: Final Exam December 14th, 2011 Please provide all answers in the spaces provided. You are not allowed to use a calculator for this exam, but you may use molecular model kits. nly cyclohexane
CHAPTER 9 HW SOLUTIONS: ALCOHOLS + ETHERS
 CAPTER 9 W SLUTNS: ALCLS + ETERS ALCL + ETER NMENCLATURE. Give the UPAC name for each compound. nclude cis/trans or R/S stereochemistry if necessary. Structure C Name -methyl--heptanol -t-butylcyclopentanol
CAPTER 9 W SLUTNS: ALCLS + ETERS ALCL + ETER NMENCLATURE. Give the UPAC name for each compound. nclude cis/trans or R/S stereochemistry if necessary. Structure C Name -methyl--heptanol -t-butylcyclopentanol
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Dr. Anand Gupta Mr Mahesh Kapil
 Dr. Anand Gupta Mr Mahesh Kapil 09356511518 09888711209 anandu71@yahoo.com mkapil_foru@yahoo.com Preparation of Haloalkanes From Alkanes Alkenes Alcohols Carboxylic Acids (Hundsdicker Reaction) Halide
Dr. Anand Gupta Mr Mahesh Kapil 09356511518 09888711209 anandu71@yahoo.com mkapil_foru@yahoo.com Preparation of Haloalkanes From Alkanes Alkenes Alcohols Carboxylic Acids (Hundsdicker Reaction) Halide
Chapter 8 Alkyl Halides and Elimination Reactions
 Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Organic Chemistry, Second Edition Janice Gorzynski Smith University of Hawai i Chapter 8 Alkyl Halides and Elimination Reactions Prepared by Rabi Ann Musah State University of New York at Albany Copyright
Part C- section 1 p-bonds as nucleophiles
 Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
Part C- section 1 p-bonds as nucleophiles Chemistry of Alkenes (Ch 8, 9, 10) - the double bond prevents free rotation - isomerism cis and trans - nomenclature E and Z (3 or 4 different substituents around
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams.
 2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
2311A and B Practice Problems to help Prepare for Final from Previous Marder Exams. Disclaimer.: Use only to help learn what you need to know and don t expect the final to be in the same form. 1 1. Short
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry
 Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry 30 Questions (5 pages); Time limit = 45 minutes Use of books or notes is not permitted. 1. When analyzed with a polarimeter, which of the
Hyperlearning MCAT Instructor Qualifying Exam Organic Chemistry 30 Questions (5 pages); Time limit = 45 minutes Use of books or notes is not permitted. 1. When analyzed with a polarimeter, which of the
REVIEW PROBLEM SET CH F. (b) Both cis and trans dibromides are obtained in the following reaction. Explain.
 REVIEW PRBLEM SET CH241-2017F [1] Provide brief answers to the following questions. (a) Write Newman projections for the chiral and meso forms of the following molecule looking down the bond connecting
REVIEW PRBLEM SET CH241-2017F [1] Provide brief answers to the following questions. (a) Write Newman projections for the chiral and meso forms of the following molecule looking down the bond connecting
ROADMAP FOR REACTIONS Chapter 6
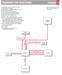 RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
RADMAP FR REACTINS Chapter 6 (A) (B) (C) (D) (E) (F) (G) (H) (I) (J) (K) (L) (M) (N) () Regiochemistry: Markovnikov addition to a bond Stereochemistry: anti-addition Regiochemistry: non-markovnikov addition
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution
 7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution Preview 7-4 7.1 Nucleophilic Substitution Reactions of Haloalkanes 7-5 Nucleophilic Substitution Mechanisms (7.1A) 7-5 The SN1
7: Reactions of Haloalkanes, Alcohols, and Amines. Nucleophilic Substitution Preview 7-4 7.1 Nucleophilic Substitution Reactions of Haloalkanes 7-5 Nucleophilic Substitution Mechanisms (7.1A) 7-5 The SN1
Organic Halogen Compounds
 8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
8 Organic alogen ompounds APTER SUMMARY 8.1 Introduction Although organic halogen compounds are rarely found in nature, they do have a variety of commercial applications including use as insecticides,
Ethers. Chapter 14: Ethers, Epoxides, & Sulfides. General Formula: Types: a) Symmetrical: Examples: b) Unsymmetrical: Examples: Physical Properties:
 Chamras Chemistry 106 Lecture Notes Examination 1 Materials Chapter 14: Ethers, Epoxides, & Sulfides Ethers General Formula: Types: a) Symmetrical: Examples: b) Unsymmetrical: Examples: Physical Properties:
Chamras Chemistry 106 Lecture Notes Examination 1 Materials Chapter 14: Ethers, Epoxides, & Sulfides Ethers General Formula: Types: a) Symmetrical: Examples: b) Unsymmetrical: Examples: Physical Properties:
1. CONCEPTS IN ORGANIC CHEMISTRY 2. SYNTHETIC ORGANIC CHEMISTRY 3. ISOMERISM II 4. HYDROCARBONS II 5. HALOALKANES. Vikasana - CET 2012
 CET OBJECTIVE QUESTION ON 1. CONCEPTS IN ORGANIC CHEMISTRY 2. SYNTHETIC ORGANIC CHEMISTRY 3. ISOMERISM II 4. HYDROCARBONS II 5. HALOALKANES 1.The inductive effect a. Implies the atoms ability to cause
CET OBJECTIVE QUESTION ON 1. CONCEPTS IN ORGANIC CHEMISTRY 2. SYNTHETIC ORGANIC CHEMISTRY 3. ISOMERISM II 4. HYDROCARBONS II 5. HALOALKANES 1.The inductive effect a. Implies the atoms ability to cause
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2
 C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
C h a p t e r S e v e n : Haloalkanes: Nucleophilc Substitution and Elimination Reactions S N 2 CHM 321: Summary of Important Concepts Concepts for Chapter 7: Substitution Reactions I. Nomenclature of
Chapter 10. Reactions of Alcohols, Amines, Ethers, and Epoxides
 Chapter 10. Reactions of Alcohols, Amines, Ethers, and Epoxides Learning objectives: 1. Provide both IUPAC and common (when applicable) names for alcohols and ethers. 2. Describe the physical properties
Chapter 10. Reactions of Alcohols, Amines, Ethers, and Epoxides Learning objectives: 1. Provide both IUPAC and common (when applicable) names for alcohols and ethers. 2. Describe the physical properties
CHM 292 Final Exam Answer Key
 CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
CHM 292 Final Exam Answer Key 1. Predict the product(s) of the following reactions (5 points each; 35 points total). May 7, 2013 Acid catalyzed elimination to form the most highly substituted alkene possible
Chapter 8: Alkene Structure and Preparation via Elimination Reactions
 Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Nature of the pi bond Chapter 8: Alkene Structure and Preparation via Elimination eactions [Sections: 8.1-8.13] C C 3 C C 3 bond length bond strength 2 C C 2 a C=C double bond is stronger than a C C single
Detailed Course Content
 Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
Detailed Course Content Chapter 1: Carbon Compounds and Chemical Bonds The Structural Theory of Organic Chemistry 4 Chemical Bonds: The Octet Rule 6 Lewis Structures 8 Formal Charge 11 Resonance 14 Quantum
How alkyl halides react
 Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
Chapter 10 1 How alkyl halides react δ+ δ- RCH 2 -X X= halogen X = higher EN C = lower EN This polar carbon-halogen bond causes alkyl halide to undergo S N and elimination reaction. 2 The mechanism of
ORGANIC - CLUTCH CH ALCOHOLS AND CARBONYL COMPOUNDS.
 !! www.clutchprep.com CONCEPT: INTRO TO REDOX Oxidation reactions involve an increase in the content of a molecule Reduction reactions involve an increase in the content of a molecule EXAMPLE: Label the
!! www.clutchprep.com CONCEPT: INTRO TO REDOX Oxidation reactions involve an increase in the content of a molecule Reduction reactions involve an increase in the content of a molecule EXAMPLE: Label the
Exam 1 (Monday, July 6, 2015)
 Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
Chem 231 Summer 2015 Assigned Homework Problems Last updated: Friday, July 24, 2015 Problems Assigned from Essential Organic Chemistry, 2 nd Edition, Paula Yurkanis Bruice, Prentice Hall, New York, NY,
CHEM 345 Problem Set 07 Key
 CHEM 345 Problem Set 07 Key 1.) Fill in the appropriate reaction arrow. The starting material is on the left, the product is on the right. Use. Simple Ring Size. 5 and 6 are favored. 3 is not. That s it.
CHEM 345 Problem Set 07 Key 1.) Fill in the appropriate reaction arrow. The starting material is on the left, the product is on the right. Use. Simple Ring Size. 5 and 6 are favored. 3 is not. That s it.
Suggested solutions for Chapter 19
 s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
s for Chapter 19 19 PRBLEM 1 Predict the orientation of addition to these alkenes. Simple examples of addition with regioselectivity. The first and last alkenes have different numbers of substituents at
REARRANGEMENTS NOTES Mechanistic Aspects of Rearrangements
 - 1 - REARRANGEMENTS NOTES Mechanistic Aspects of Rearrangements Nature of the Rearrangement It can vary from being truly stepwise to migration occurring in concert with initial ionisation. These two situations
- 1 - REARRANGEMENTS NOTES Mechanistic Aspects of Rearrangements Nature of the Rearrangement It can vary from being truly stepwise to migration occurring in concert with initial ionisation. These two situations
ORGANIC - CLUTCH CH ADDITION REACTIONS.
 !! www.clutchprep.com CONCEPT: GENERAL MECHANISM Addition reactions are ones in which 1 bond is broken and 2 new bonds are formed. They are the inverse of reactions EXAMPLE: Provide the mechanism for the
!! www.clutchprep.com CONCEPT: GENERAL MECHANISM Addition reactions are ones in which 1 bond is broken and 2 new bonds are formed. They are the inverse of reactions EXAMPLE: Provide the mechanism for the
Aldehydes and Ketones 2. Based on Organic Chemistry, J. G. Smith 3rde.
 Aldehydes and Ketones 2 Based on Organic Chemistry, J. G. Smith 3rde. The Wittig Reaction Wittig reaction, named for German chemist Georg Wittig, who was awarded the Nobel Prize in Chemistry in 1979 for
Aldehydes and Ketones 2 Based on Organic Chemistry, J. G. Smith 3rde. The Wittig Reaction Wittig reaction, named for German chemist Georg Wittig, who was awarded the Nobel Prize in Chemistry in 1979 for
Chapter 9 Aldehydes and Ketones Excluded Sections:
 Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
Chapter 9 Aldehydes and Ketones Excluded Sections: 9.14-9.19 Aldehydes and ketones are found in many fragrant odors of many fruits, fine perfumes, hormones etc. some examples are listed below. Aldehydes
Elimination Reactions. Chapter 6 1
 Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
Elimination Reactions Chapter 6 1 E1 Mechanism Step 1: halide ion leaves, forming a carbocation. Step 2: Base abstracts H + from adjacent carbon forming the double bond. Chapter 6 2 E1 Energy Diagram E1:
