Introduction to Organometallic Chemistry. Stability of organometallic reagents
|
|
|
- Melina Harmon
- 6 years ago
- Views:
Transcription
1 Introduction to rganometallic hemistry. rganometallic chemistry is the chemistry of compounds that contain M- bonds, where M is either a main group metal or a transition metal.. ne common feature of all organometallic compounds is the metal is in a very low oxidation state.. Because of the low oxidation state on the metal, there are many d-electrons in the complex, so π-backbonding between the metal and ligands is very important. ne of the most common ligands in organometallic chemistry is carbonyl,.. rganometallic reagents are very important in synthesis because of their versatility. For example, metals will often activate the -H bond, which lowers the activation energy for many organic transformations.. rganometallic catalysts are used to make many important pharmaceuticals and are critical in the petrochemical industry. Stability of organometallic reagents. There is one problem with many organometallic reagents: because the metal is in a low oxidation state, if the complex is exposed to atmospheric oxygen the metal is readily oxidized.. Therefore, we carry out many organometallic reactions under an inert atmosphere of nitrogen or argon.. If a complex is exceptionally air sensitive, then we use a glove box:. If we do not need a glove box and can safely carry out the reaction in a hood, we usually use a Schlenck flask to protect the flask contents from atmospheric oxygen:
2 . We can even transfer liquids between two flasks under an inert atmosphere using cannula tubing:
3 Formalisms in rganometallic hemistry. There are four important things to know before being able to predict the reactivity of an organometallic complex: 1. the oxidation state of the metal, 2. the number of d-electrons on the metal, 3. the coordination number of the metal, and 4. the availability (or lack thereof) of vacant coordination sites on the metal.. It is important to realize that these formalisms are merely formal ways of looking at reality; they are not reality, they are not the truth, and they may not even be chemically reasonable. However, they allow us to predict trends among a very diverse set of chemical compounds.
4
5 General Trends for the Transition Metals Group 8 Metals 21 d 3 d 4 d 5 d 6 d 7 d 8 d 9 d 10 d 10 s 1 Sc Scandium 39 Y Yttrium 57 a anthanum 22 Ti Titanium 40 Zr Zirconium 72 Hf Hafnium 23 V Vanadium 41 Nb Niobium 73 Ta Tantalum 24 r hromium 42 Mo Molybdenum 74 W Tungsten 25 Mn Manganese 43 Tc Technetium 75 Re Rhenium 26 Fe Iron 44 Ru Ruthenum 76 s smium 27 o obalt 45 Rh Rhodium 77 Ir Iridium 28 Ni Nickel 46 Pd Palladium 78 Pt Platinum 29 u opper 47 Ag Silver 79 Au Early Transiton Metals ate Transition Metals Gold
6 Hapticity. Hapticity, η, eta is the number of atoms associated with the metal in an organometallic.. If only one atom is attached to the metal, we say the ligand is a monohapto or η 1 ligand.. yclopentadiene and the cyclopentadienyl anion, p, can bind to a metal through one to five carbon atoms: Bridging ligand µ x mu-x is the nomenclature used to indicate the presence of a bridging ligand between two or more metal centers. The x refers to the number of metal centers being bridged by the ligand. Usually most authors omit x = 2 and just use to indicate that the ligand is bridging the simplest case of two metals rdering There is no set method of naming or ordering the listing of metal and ligands in a metal/ligand complex that most authors follow. There are IUPA formalisms, but hardly anyone follows them. There are some qualitative rules that most authors seem to use in American hemical Society (AS) publications: 1) in formulas with p (cyclopentadienyl) ligands, the p usually comes first, followed by the metal center: p 2 Til 2 2) other anionic multi-electron donating ligands are also often listed in front of the metal. 3) in formulas with hydride ligands, the hydride is sometimes listed first. Rules # 1 & 2, however, take precedence over this rule: HRh()(PPh 3 ) 2 and p 2 TiH 2 4) bridging ligands are usually placed next to the metals in question, then followed by the other ligands (note that rules 1 & 2 take precedence): o 2 (µ-) 2 () 6, Rh 2 (µ-l) 2 () 4, p 2 Fe 2 (µ-) 2 () 2
7 ommon oordination Geometries 6-oordinate: ctahedral (90 & 180 angles) M M M 5-oordinate: Trigonal Bypyramidal or Square Pyramidial (90 & 120 ) (~100 & 90 ) axial apical M equitorial M basal 4-oordinate: Square Planar or Tetrahedral (90 & 180 ) (109 ) M M
8 Types of igand The arbonyl igand,. The carbonyl ligand is a σ-donor and very strong π-acceptor.. The homo has the appropriate symmetry for overlap with a metal UM:. Therefore, there is a σ -interaction between the HM and the Metal UM. σ -interaction. In addition, the metal HM can donate electron density into the UM since these orbitals have the appropriate symmetry for overlap:. Therefore, there is a π -interaction with the metal donating electron density into the π * UM of.. As electron density is donated from the metal into the antibonding π * UM of, there is a decrease in bond order when binds to a metal.. Therefore, the bond is weaker when bonded to a metal than in the gas phase. π -interaction. There are four different bonding modes.
9 The Hydride igand, H-. When coordinated to a metal, H is formally negative and is, therefore, called the hydride ligand.. Metal hydrides often behave as proton donors: Ho()4 + H2 > [o()4] - + H3 +. There are four different binding modes for hydrides:
10 Phosphines and Phosphites. Phosphines are ubiquitous in catalysis because their stereoelectronic properties are easily manipulated.. Phosphines are σ -donors and π -acceptors.. The more electronegative the substituents on P, the weaker the σ -donor ability and the stronger the π -acceptor ability.. For example, PF3 is a weak σ -donor but as strong a π -acceptor as.. onversely, P(tBu)3 is a strong σ -donor (because of the inductive electron release ability of the tbu groups) but a weak π -acceptor.. The π -accepting abilities of phosphines increase as: PF3 > P(Ph)3 > P(Me)3 > PMe3 > P(tBu)3. Steric effects in organometallic chemistry were first defined by Tolman in Bidentate phosphines are extremely important in catalysis.. Bidentate phosphines have two P-donor atoms per molecule.. Some examples of important bidentate phosphines, including BINAP, which was responsible for Nyori s and Knowles Noble Prize in 2001, are shown below:
11 π -Bonded rganic igands. In addition to being able to bind hydrogen, metals can also bind unsaturated organic molecules.. When Rh binds ethylene, for example, the = bond lengthens from Å for H2=H2 to Å in the Rh-( η2-h2=h2) complex.. This increase in = bond length upon coordination to a metal indicates that the metal donates electrons into the π* UM of the ethylene.. onsequently, the = bond weakens, which makes the = bond more susceptible to attack by an incoming nucleophile.. In other words, coordination of the olefin to a metal results in activation of the organic molecule.. There are two extremes to consider when an olefin binds to a metal: the one is pure 2 coordination, the other involves the formation of a metallocyclopropane: ther igands. Dinitrogen often forms an η 1-bond with a metal, analogous to : M-N 2.. Dihydrogen, on the other hand, forms an η 2-bond with a metal.
12 The 18 Electron Rule. The effective atomic number (EAN) rule states that most stable organometallic complexes have 18 electrons surrounding the metal. This is also known as the 18-electron rule.. Metals that contain 18 electrons around them are called coordinatively saturated.. Metals that contain less than 18 electrons around them are called coordinatively unsaturated and have vacant coordination sites.. To determine whether or not a metal is coordinatively saturated, we need to know how many electrons the ligand donates.. There are two ways to count electrons: ionic and covalent. Both give the same answer.. Ionic counting assumes the metal is in a formal oxidation state and the ligands are charged.. ovalent counting assumes the metal is in the zero oxidation state and the ligands are neutral.
13 The covalent counting method lasses of igands. We classify ligands by the number of electrons they donate to the metal: ne Electron Donors Two Electron Donors Three Electron Donors Alkyls, R Phosphines, PR3, PAr3 inear nitrosyl, N Aryls, Ar Phosphites, P(R)3 η 3-allyls, R2-R=R2 Bent nitrosyl, N arbonyl, η 3-p Hydride, H η 2-olefins, R2=R2 η 1-ligands Halides η 2-alkynes, RαR Amines, R3N-M Nitriles, RN-M Isonitriles, RN-M arbenes, M=R2. Notice that the hapticity of the ligand is generally equal to its electron count.. ertain ligands, like allyl and cyclopentadienyl, can bind in more than one manner:. When an η 3-allyl complex forms, the ligand bonds through all three carbon atoms and is considered a 3-electron donor. However, the allyl only occupies two coordination sites on the metal.. We are now in a position to consider any transition metal organometallic complex, assign the oxidation state at the metal, count the total number of electrons involved in bonding, and decide whether or not the complex is coordinatively saturated.
14 Example: Which of the following is coordinatively unsaturated: pfe()2(propyl) or Rh(PPh3)3l?
15 The ionic counting method To determine the electron count for a metal complex: 1) Determine the oxidation state of the transition metal center(s) and the metal centers resulting d-electron count. To do this one must: a) note any overall charge on the metal complex b) know the charges of the ligands bound to the metal center (ionic ligand method) c) know the number of electrons being donated to the metal center from each ligand (ionic ligand method) 2) Add up the electron counts for the metal center and ligands the charges and donor # for the common ligands: ationic 2e- donor: N (nitrosyl) Neutral 2e- donors: PR3 (phosphines), (carbonyl), R2=R2 (alkenes), R R (alkynes, can also donate 4 e-), N R (nitriles) Anionic 2e- donors: l (chloride), Br (bromide), I (iodide), H3 (methyl), R3 (alkyl), Ph (phenyl), H (hydride) The following can also donate 4 e- if needed, but initially count them as 2e- donors (unless they are acting as bridging ligands): R (alkoxide), SR (thiolate), NR2 (inorganic amide), PR2 (phosphide) Anionic 4e- donors: 3H5 (allyl), 2 (oxide), S2 (sulfide), NR2 (imide), R22 (alkylidene) and from the previous list: R (alkoxide), SR (thiolate), NR2 (inorganic amide), PR2 Anionic 6e- donors: p (cyclopentadienyl), 2 (oxide) Please note that we are using the Ionic Method of electron-counting. 95% of inorganic/organometallic chemists use the ionic method.
16 Example: R 3 P H 3 Re PR 3 1) There is no overall charge on the complex 2) There is one anionic ligand (H3, methyl group) 3) Since there is no overall charge on the complex (it is neutral), and since we have one anionic ligand present, the Re metal atom must have a +1 charge to compensate for the one negatively charged ligand. The +1 charge on the metal is also its oxidation state. So the Re is the in the +1 oxidation state. We denote this in two different ways: Re(+1), Re(I), or ReI. I prefer the Re(+1) nomenclature because it is clearer. Most chemistry journals, however, prefer the Roman numeral notation in parenthesis after the element. Now we can do our electron counting: Re(+1) d6 2 PR3 4e- 2 4e- H3 2e- H2=H2 2e- Total: 18e-
17 16-Electron Species Most stable organometallic compounds obey the 18-electron rule. However, stable complexes do exist with electron counts other than 18, as factors such as crystal field stabilisation energy and the nature of the bonding between the metal and the ligand affect the stability of the compound. The most widely encountered exceptions to the rule are 16-electron complexes of the transition metals on the right hand side of the d block, particularly Groups 9 and 10. These 16-electron, square-planar complexes commonly have d8 electron configurations, for example Rh(I), Ir(I), Ni(II), and Pd(II). Exceptions to the 18e Rule Group 8 Metals d 3 d 4 d 5 d 6 d 7 d 8 d 9 d 10 d 10 s Sc Ti V r Mn Fe o Ni u Scandium Titanium Vanadium hromium Manganese Iron obalt Nickel opper Y Zr Nb Mo Tc Ru Rh Pd Ag Yttrium Zirconium Niobium Molybdenum Technetium Ruthenum Rhodium Palladium Silver a Hf Ta W Re s Ir Pt Au anthanum Hafnium Tantalum Tungsten Rhenium smium Iridium Platinum Gold Early Transition Metals Middle Transition Metals ate Transition Metals 16e and sub-16e configurations are common 18e configurations are common 16e and sub-16e configurations are common oordination geometries higher than 6 oordination geometries of 6 are common oordination geometries of 5 or lower
18 Fundamental Reactions of Transition Metal omplex 1. igands Substitution 2. xidative Addition 3. Insertion 4. Transmetallation 5. Reductive Elimination 6. Dehydrometallation (Elimination of Hydrogen) 7. Nucleophilic Attack on igands oordinated to Transition Metals Figure 2.10 A hypothetical catalytic cycle with the precatalyst Mn_1 and four catalytic intermediates.
19 igands Substitution M n + xp M n-x P x + x The mechanism of this substitution will almost always depend on whether the parent Mn complex is coordinatively saturated or not! Substitutions reactions occur by a combination of ligand addition and ligand dissociation reactions. Mo - Mo +PMe 3 Mo PMe 3 18e- saturated complex 16e- unsaturated complex 18e- saturated complex igand Addition (association): this is when an incoming ligand coordinates to a metal center that has one or more empty orbitals available. Ph 3 P Ph 3 P Rh l + Ph 3 P Rh l PPh 3 This Rh(+1) complex is d 8 and only 14e-. Adding a ligand takes one to the more stable 16e- square-planar complex. igand Dissociation: this is when a ligand coordinated to a metal dissociates (falls off). The probability of a specific ligand dissociating depends on how strongly or weakly it is coordinated to the metal center and steric effects. igand Dissociation Ph 3 P Ph 3 P Rh l PPh 3 Ph 3 P Ph 3 P Rh l + PPh 3 The steric hindrence of the three bulky PPh 3 ligands favors dissociation of one to form the 14e- Rhl(PPh 3 ) 2 complex. The moderate electron-donating ability of the PPh 3 ligand (not a strongly coordinating ligand) makes this fairly facile.
20 Steric Factors Bulky (large) ligands occupy more space around a metal center and can block incoming ligands trying to access vacant coordination sites on a metal. Due to steric hindrance, however, they are also more often to dissociate to relieve the steric strain. onsider, for example, the following equilibrium: K D Ni(PR 3 ) 4 Ni(PR 3 ) 3 + PR 25º 3 igand: P(Et) 3 P(-p-tolyl) 3 P(-i-Pr) 3 P(-o-tolyl) 3 PPh 3 one angle: 109º 128º 130º 141º 145º K D : < x x x 10 2 > 1000 Solvent Effects l PPh 3 Pt Ph 3 P l -l l PPh 3 Pt Ph 3 P + solvent - solvent l PPh 3 Pt Ph 3 P solvent +Br l PPh 3 Pt Ph 3 P Br The 14e- three coordinate intermediate is actually almost immediately coordinated by a solvent molecule to produce the solvated 16e- complex shown to the far right. The solvent is usually weakly coordinated and readily dissociates to constantly produce the 14ereactive intermediate. Few organometallic chemists formally write solvated metal complexes down in their mechanisms, but they certainly are formed. The coordinating ability of the solvent, therefore, can often affect reactions. The presence of lone pairs and electron-rich donor atoms on the solvent usually makes it a better ligand. The polarity of the solvent can also have a definite impact on a reaction. Polar solvents
21 are usually quite good for reactions, such as that shown above, involving charged species. A non-polar hydrocarbon solvent (like toluene, for example) would probably inhibit the chloride dissociation mechanism. Instead, the dissociation of the neutral, less polar phosphine ligand would probably be favored. Some ommon oordinating Solvents: acetone, THF, DMS, water, alcohols, DME, DMF Trans Effect The trans effect concerns the electronic effect of one ligand on another ligand when they are trans to one another. The classical trans effect involves two σ-donating ligands trans to one another. The stronger σ-donor ligand preferentially weakens the bond of the weaker σ-donor ligand trans to it, making it easier to dissociate and do a ligand substitution reaction. l PEt 3 Pt Et 3 P -l Et 3 P Pt PEt 3 N N Et 3 P Pt PEt 3 Relative rate of substitution based on trans ligand : l = 1, Ph = 100, H 3 = 10 3, H = 10 4 There is a cis effect, but it is much weaker and basically ignored: The allyl anion has a similar facile ability to switch between η 3 and η 1 coordination modes that can promote ligand additions and/or substitutions. Mn Mn + η 3 η 1 η 1 η 3 usually not observed experimentally Mn can be stable and isolated - Mn
Reaction Mechanisms - Ligand Substitutions. ML n-x P x + xl
 Reaction chanisms - igand Substitutions igand Substitutions 1 A substitution reaction is one in which an existing ligand on a metal center is replaced by another ligand. Exactly how this occurs depends
Reaction chanisms - igand Substitutions igand Substitutions 1 A substitution reaction is one in which an existing ligand on a metal center is replaced by another ligand. Exactly how this occurs depends
Course 201N 1 st Semester Inorganic Chemistry Instructor: Jitendra K. Bera
 andout-9 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry xidative Addition, Reductive Elimination, Migratory Insertion, Elimination
andout-9 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry xidative Addition, Reductive Elimination, Migratory Insertion, Elimination
Chem 634. Introduction to Transition Metal Catalysis. Reading: Heg Ch 1 2 CS-B 7.1, , 11.3 Grossman Ch 6
 Chem 634 Introduction to Transition etal Catalysis eading: eg Ch 1 2 CS-B 7.1, 8.2 8.3, 11.3 Grossman Ch 6 Announcements Problem Set 1 due Thurs, 9/24 at beginning of class ffice our: Wed, 10:30-12, 220
Chem 634 Introduction to Transition etal Catalysis eading: eg Ch 1 2 CS-B 7.1, 8.2 8.3, 11.3 Grossman Ch 6 Announcements Problem Set 1 due Thurs, 9/24 at beginning of class ffice our: Wed, 10:30-12, 220
Reaction chemistry of complexes Three general forms: 1. Reactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b.
 eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
eaction chemistry of complexes Three general forms: 1. eactions involving the gain and loss of ligands a. Ligand Dissoc. and Assoc. (Bala) b. Oxidative Addition c. eductive Elimination d. Nucleophillic
Organometallic Chemistry and Homogeneous Catalysis
 Organometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N2 Kashiwa ampus, October 16, 2009 Properties I. Nature of II. Nature of L II.a. Ligands with -atom attached to II.b. Ligands
Organometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N2 Kashiwa ampus, October 16, 2009 Properties I. Nature of II. Nature of L II.a. Ligands with -atom attached to II.b. Ligands
Oxidative Addition/Reductive Elimination 1. Oxidative Addition
 Oxidative Addition Oxidative Addition/Reductive Elimination 1 An oxidative addition reaction is one in which (usually) a neutral ligand adds to a metal center and in doing so oxidizes the metal, typically
Oxidative Addition Oxidative Addition/Reductive Elimination 1 An oxidative addition reaction is one in which (usually) a neutral ligand adds to a metal center and in doing so oxidizes the metal, typically
π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles
 π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles M Transition metal alkene complexes The report in 1825 by William
π bonded ligands alkene complexes alkyne complexes allyl complexes diene complexes cyclopentadienyl complexes arene complexes metallacycles M Transition metal alkene complexes The report in 1825 by William
Organometallic Chemistry and Homogeneous Catalysis
 rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
rganometallic hemistry and omogeneous atalysis Dr. Alexey Zazybin Lecture N8 Kashiwa ampus, December 11, 2009 Types of reactions in the coordination sphere of T 3. Reductive elimination X-L n -Y L n +
O CH 3. Mn CH 3 OC C. 16eelimination
 igratory Insertion igratory Insertion/Elimination 1 A migratory insertion reaction is when a cisoidal anionic and neutral ligand on a metal complex couple together to generate a new coordinated anionic
igratory Insertion igratory Insertion/Elimination 1 A migratory insertion reaction is when a cisoidal anionic and neutral ligand on a metal complex couple together to generate a new coordinated anionic
Chapter 2 The Elementary Steps in TM Catalysis
 hapter 2 The Elementary Steps in TM atalysis + + ligand exchange A oxidative addition > n + A B n+2 reductive elimination < B n n+2 oxidative coupling + M' + M' transmetallation migratory insertion > (carbo-,
hapter 2 The Elementary Steps in TM atalysis + + ligand exchange A oxidative addition > n + A B n+2 reductive elimination < B n n+2 oxidative coupling + M' + M' transmetallation migratory insertion > (carbo-,
A Summary of Organometallic Chemistry
 A Summary of Organometallic Chemistry Counting valence electrons (v.e.) with the ionic model 1. Look at the total charge of the complex Ph 3 P Cl Rh Ph 3 P PPh 3 OC CO 2 Fe OC CO Co + charge:0 charge:
A Summary of Organometallic Chemistry Counting valence electrons (v.e.) with the ionic model 1. Look at the total charge of the complex Ph 3 P Cl Rh Ph 3 P PPh 3 OC CO 2 Fe OC CO Co + charge:0 charge:
Metal Hydrides, Alkyls, Aryls, and their Reactions
 Metal Hydrides, Alkyls, Aryls, and their Reactions A Primer on MO Theory σ-bonding in Organotransition Metal Complexes M-C Bond Energies in Organotransition Metal Complexes Thermodynamic Predictions
Metal Hydrides, Alkyls, Aryls, and their Reactions A Primer on MO Theory σ-bonding in Organotransition Metal Complexes M-C Bond Energies in Organotransition Metal Complexes Thermodynamic Predictions
Course 201N 1 st Semester Inorganic Chemistry Instructor: Jitendra K. Bera
 andout-8 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. Organometallic hemistry yclopentadienyl, Alkyl and Alkene yclopentadienyl p The cyclopentadienyl ligand
andout-8 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. Organometallic hemistry yclopentadienyl, Alkyl and Alkene yclopentadienyl p The cyclopentadienyl ligand
Insertion Reactions. 1) 1,1 insertion in which the metal and the X ligand end up bound to the same (1,1) atom
 Insertion Reactions xidative addition and substitution allow us to assemble 1e and 2e ligands on the metal, respectively. With insertion, and its reverse reaction, elimination, we can now combine and transform
Insertion Reactions xidative addition and substitution allow us to assemble 1e and 2e ligands on the metal, respectively. With insertion, and its reverse reaction, elimination, we can now combine and transform
Organometallic Chemistry and Homogeneous Catalysis
 Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N6 Kashiwa Campus, November 27, 2009 Group VIB: Cr, Mo, W -Oxidation states from -2 to +6 -While +2 and +3 for Cr are quite
Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N6 Kashiwa Campus, November 27, 2009 Group VIB: Cr, Mo, W -Oxidation states from -2 to +6 -While +2 and +3 for Cr are quite
PAPER No.11 : Inorganic Chemistry-II MODULE No.1 : Π-acceptor ligand, metal carbonyls, bonding modes of CO, classification of metal carbonyls
 Subject Paper No and Title Module No and Title Module Tag 11: INORGANIC CHEMISTRY-III (METAL π- COMPLEXES AND METAL CLUSTERS) 1: π-acidity, Metal carbonyls, their classification and general features CHE_P11_M1
Subject Paper No and Title Module No and Title Module Tag 11: INORGANIC CHEMISTRY-III (METAL π- COMPLEXES AND METAL CLUSTERS) 1: π-acidity, Metal carbonyls, their classification and general features CHE_P11_M1
Organometallic Chemistry and Homogeneous Catalysis
 Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N1 Kashiwa Campus, October 9, 2009 What compounds we can call organometallic compounds? Compounds containing direct metal-carbon
Organometallic Chemistry and Homogeneous Catalysis Dr. Alexey Zazybin Lecture N1 Kashiwa Campus, October 9, 2009 What compounds we can call organometallic compounds? Compounds containing direct metal-carbon
Ionic Compound Solubility. Ionic Compound Solubility. Nitrates (NO 3 - ) Chlorates (ClO 3 - ) Ionic Compound Solubility. Ionic Compound Solubility
 Nitrates (NO 3 - ) Chlorates (ClO 3 - ) Perchlorates (ClO 4 - ) Acetates (C 2 H 3 O 2 - ) Alkali Metal Compounds (Li +,Na +,K +,Rb +,Cs + ) Ammonium Compounds (NH 4 + ) Chlorides (Cl - ) Bromides (Br -
Nitrates (NO 3 - ) Chlorates (ClO 3 - ) Perchlorates (ClO 4 - ) Acetates (C 2 H 3 O 2 - ) Alkali Metal Compounds (Li +,Na +,K +,Rb +,Cs + ) Ammonium Compounds (NH 4 + ) Chlorides (Cl - ) Bromides (Br -
Repeated insertion. Multiple insertion leads to dimerization, oligomerization or polymerization. κ 1: mainly dimerization κ
 Repeated insertion ultiple insertion leads to dimerization, oligomerization or polymerization. k prop Et Key factor: k CT / k prop = κ κ 1: mainly dimerization κ 0.1-1.0: oligomerization (always mixtures)
Repeated insertion ultiple insertion leads to dimerization, oligomerization or polymerization. k prop Et Key factor: k CT / k prop = κ κ 1: mainly dimerization κ 0.1-1.0: oligomerization (always mixtures)
Inorganic Chemistry Year 3
 Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
Inorganic Chemistry Year 3 Transition Metal Catalysis Eighteen Electron Rule 1.Get the number of the group that the metal is in (this will be the number of d electrons) 2.Add to this the charge 1.Negative
Introduction to Organometallic Compounds. Metal. R MgX. Wilkinson s catalyst is used for hydrogenation of alkene and alkyne. Wilkinson's catalyst
 Introduction to rganometallic mpounds 1 Chapter 1 Introduction to rganometallic mpounds Introduction : Edward Frankland was father of organometallic chemistry for a complex to be organometallic compound,
Introduction to rganometallic mpounds 1 Chapter 1 Introduction to rganometallic mpounds Introduction : Edward Frankland was father of organometallic chemistry for a complex to be organometallic compound,
σ Bonded ligands: Transition Metal Alkyls and Hydrides
 σ Bonded ligands: Transition Metal Alkyls and Hydrides Simplest of organo-transitionmetal species Rare until and understanding of their stability in the 60 s and 70 s Metal alkyls can be considered as
σ Bonded ligands: Transition Metal Alkyls and Hydrides Simplest of organo-transitionmetal species Rare until and understanding of their stability in the 60 s and 70 s Metal alkyls can be considered as
Oxidative Addition and Reductive Elimination
 xidative Addition and Reductive Elimination red elim coord 2 ox add ins Peter.. Budzelaar xidative Addition Basic reaction: n + X Y n X Y The new -X and -Y bonds are formed using: the electron pair of
xidative Addition and Reductive Elimination red elim coord 2 ox add ins Peter.. Budzelaar xidative Addition Basic reaction: n + X Y n X Y The new -X and -Y bonds are formed using: the electron pair of
Organic Chemistry Laboratory Summer Lecture 6 Transition metal organometallic chemistry and catalysis July
 344 Organic Chemistry Laboratory Summer 2013 Lecture 6 Transition metal organometallic chemistry and catalysis July 30 2013 Summary of Grignard lecture Organometallic chemistry - the chemistry of compounds
344 Organic Chemistry Laboratory Summer 2013 Lecture 6 Transition metal organometallic chemistry and catalysis July 30 2013 Summary of Grignard lecture Organometallic chemistry - the chemistry of compounds
CHEM Core Chemistry 3. Reaction Mechanisms in Organometallic Chemistry
 E3012 - ore hemistry 3 eaction echanisms in Organometallic hemistry In an earlier section of this lecture course we considered the mechanisms of substitution reactions in organometallic species, and noted
E3012 - ore hemistry 3 eaction echanisms in Organometallic hemistry In an earlier section of this lecture course we considered the mechanisms of substitution reactions in organometallic species, and noted
Reductive Elimination
 Reductive Elimination Reductive elimination, the reverse of oxidative addition, is most often seen in higher oxidation states because the formal oxidation state of the metal is reduced by two units in
Reductive Elimination Reductive elimination, the reverse of oxidative addition, is most often seen in higher oxidation states because the formal oxidation state of the metal is reduced by two units in
Survey of Elements. 1s - very small atom All noble gases have very high first I.E. s which is in line with their chemical inertness
 Survey of Elements 354 Hydrogen 1s 1 Naturally, loss of one e - dominates 1s Helium 1s 2 - closed shell 1s - very small atom All noble gases have very high first I.E. s which is in line with their chemical
Survey of Elements 354 Hydrogen 1s 1 Naturally, loss of one e - dominates 1s Helium 1s 2 - closed shell 1s - very small atom All noble gases have very high first I.E. s which is in line with their chemical
Chapter 20: Aldehydes and Ketones
 hem A225 Notes Page 67 I. Introduction hapter 20: Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group (=) with no other heteroatoms attached. An aldehyde has at least one hydrogen attached;
hem A225 Notes Page 67 I. Introduction hapter 20: Aldehydes and Ketones Aldehydes and ketones contain a carbonyl group (=) with no other heteroatoms attached. An aldehyde has at least one hydrogen attached;
Chem Selected Aspects of Main Group Chemistry
 Selected Aspects of Main Group Chemistry For the rest of the course, we will look at some aspects of the chemistry of main group compounds. The basic principles that you have learned concerning atoms,
Selected Aspects of Main Group Chemistry For the rest of the course, we will look at some aspects of the chemistry of main group compounds. The basic principles that you have learned concerning atoms,
Bonding in Coordination Compounds. Crystal Field Theory. Bonding in Transition Metal Complexes
 Bonding in Transition Metal Complexes 1) Crystal Field Theory (ligand field theory) Crystal Field Theory Treat igands as negative charges (they repel the e- in the d orbitals deals only with d orbitals
Bonding in Transition Metal Complexes 1) Crystal Field Theory (ligand field theory) Crystal Field Theory Treat igands as negative charges (they repel the e- in the d orbitals deals only with d orbitals
75. A This is a Markovnikov addition reaction. In these reactions, the pielectrons in the alkene act as a nucleophile. The strongest electrophile will
 71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
71. B SN2 stands for substitution nucleophilic bimolecular. This means that there is a bimolecular rate-determining step. Therefore, the reaction will follow second-order kinetics based on the collision
11, Inorganic Chemistry III (Metal π-complexes and Metal Clusters) Module 31: Preparation and reactions of metal clusters
 1 Subject Paper No and Title Module No and Title Module Tag Chemistry 11, Inorganic Chemistry III (Metal π-complexes and Module 31: Preparation and reactions of metal clusters CHE_P11_M31 2 TABLE OF CONTENTS
1 Subject Paper No and Title Module No and Title Module Tag Chemistry 11, Inorganic Chemistry III (Metal π-complexes and Module 31: Preparation and reactions of metal clusters CHE_P11_M31 2 TABLE OF CONTENTS
Reductive Elimination
 Reductive Elimination Reductive elimination, the reverse of oxidative addition, is most often seen in higher oxidation states because the formal oxidation state of the metal is reduced by two units in
Reductive Elimination Reductive elimination, the reverse of oxidative addition, is most often seen in higher oxidation states because the formal oxidation state of the metal is reduced by two units in
Reducing Agents. Linda M. Sweeting 1998
 Reducing Agents Linda M. Sweeting 1998 Reduction is defined in chemistry as loss of oxygen, gain of hydrogen or gain of electrons; the gain of electrons enables you to calculate an oxidation state. Hydride
Reducing Agents Linda M. Sweeting 1998 Reduction is defined in chemistry as loss of oxygen, gain of hydrogen or gain of electrons; the gain of electrons enables you to calculate an oxidation state. Hydride
Chapter 25: The Chemistry of Life: Organic and Biological Chemistry
 Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Chemistry: The Central Science Chapter 25: The Chemistry of Life: Organic and Biological Chemistry The study of carbon compounds constitutes a separate branch of chemistry known as organic chemistry The
Module 10 : Reaction mechanism. Lecture 1 : Oxidative addition and Reductive elimination. Objectives. In this lecture you will learn the following
 Module 10 : Reaction mechanism Lecture 1 : Oxidative addition and Reductive elimination Objectives In this lecture you will learn the following The oxidative addition reactions. The reductive elimination
Module 10 : Reaction mechanism Lecture 1 : Oxidative addition and Reductive elimination Objectives In this lecture you will learn the following The oxidative addition reactions. The reductive elimination
Course 201N 1 st Semester Inorganic Chemistry Instructor: Jitendra K. Bera
 andout-10 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry omogeneous atalysis lefin ydrogenation; ydroformylation; Monsanto Acetic acid
andout-10 ourse 201N 1 st Semester 2006-2007 Inorganic hemistry Instructor: Jitendra K. Bera ontents 3. rganometallic hemistry omogeneous atalysis lefin ydrogenation; ydroformylation; Monsanto Acetic acid
Oxidative Addition oxidative addition reductive elimination
 Oxidative Addition We have seen how neutral ligands such as C 2 H 4 or CO can enter the coordination sphere of a metal by substitution. We now look at a general method for simultaneously introducing pairs
Oxidative Addition We have seen how neutral ligands such as C 2 H 4 or CO can enter the coordination sphere of a metal by substitution. We now look at a general method for simultaneously introducing pairs
PART 2. -BONDED ORGANOMETALLICS
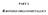 PART 2. -BODED ORGAOETALLIS V() 6 r() 6 o() 6 W() 6 Fe() 5 Ru() 5 Os() 5 i() 4 Figure 1. Binary, ononuclear etal arbonyls n n O Fe O O Fe O o O o n 2 () 10 Fe 2 () 9 o 2 () 8 Fe Fe Fe Ir() 3 Os Os Os o
PART 2. -BODED ORGAOETALLIS V() 6 r() 6 o() 6 W() 6 Fe() 5 Ru() 5 Os() 5 i() 4 Figure 1. Binary, ononuclear etal arbonyls n n O Fe O O Fe O o O o n 2 () 10 Fe 2 () 9 o 2 () 8 Fe Fe Fe Ir() 3 Os Os Os o
The 18 Electron Rule. References: Gray: chapter 5 OGN: chapter 18
 The 18 Electron Rule References: Gray: chapter 5 OGN: chapter 18 Element Groups Alkali metals nert or Noble gases Alkali earths alogens e Li Na Be Mg Transition metals B Al Si N P O S F l Ne Ar K Rb s
The 18 Electron Rule References: Gray: chapter 5 OGN: chapter 18 Element Groups Alkali metals nert or Noble gases Alkali earths alogens e Li Na Be Mg Transition metals B Al Si N P O S F l Ne Ar K Rb s
Organometallics Study Meeting
 rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
rganometallics Study eeting 04/21/2011.itsunuma 1. rystal field theory(ft) and ligand field theory(lft) FT: interaction between positively charged metal cation and negative charge on the non-bonding electrons
Basic Organic Chemistry
 Basic rganic hemistry ourse code: EM 12162 (Pre-requisites : EM 11122) hapter 06 hemistry of Aldehydes & Ketones Dr. Dinesh R. Pandithavidana ffice: B1 222/3 Phone: (+94)777-745-720 (Mobile) Email: dinesh@kln.ac.lk
Basic rganic hemistry ourse code: EM 12162 (Pre-requisites : EM 11122) hapter 06 hemistry of Aldehydes & Ketones Dr. Dinesh R. Pandithavidana ffice: B1 222/3 Phone: (+94)777-745-720 (Mobile) Email: dinesh@kln.ac.lk
Basic Organic Chemistry Course code : CHEM (Pre-requisites : CHEM 11122)
 Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
Basic Organic Chemistry Course code : CHEM 12162 (Pre-requisites : CHEM 11122) Chapter 01 Mechanistic Aspects of S N2,S N1, E 2 & E 1 Reactions Dr. Dinesh R. Pandithavidana Office: B1 222/3 Phone: (+94)777-745-720
PAPER No.11: Inorganic Chemistry-III MODULE No.29: Metal- metal bonds and their evidences
 1 Subject Chemistry Paper No and Title Module No and Title Module Tag 11: Inorganic Chemistry-III (Metal π-complexes and Metal Clusters) 29: Metal-metal bonds and their evidences CHE_P11_M29 2 TABLE OF
1 Subject Chemistry Paper No and Title Module No and Title Module Tag 11: Inorganic Chemistry-III (Metal π-complexes and Metal Clusters) 29: Metal-metal bonds and their evidences CHE_P11_M29 2 TABLE OF
N-Heterocyclic Carbenes (NHCs)
 N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
N-Heterocyclic Carbenes (NHCs) In contrast to Fischer and Schrock type carbenes NHCs are extremely stable, inert ligands when complexed to a metal centre. Similar to phosphine ligands they are electronically
Chem 263 Nov 3, 2016
 hem 263 Nov 3, 2016 Preparation of Aldehydes from Acid alides? + l l acid chloride aka acyl chloride aldehyde Needed: 2 Actual eagents: 2 /Pd Al This is lithium tri-t-butoxy aluminum hydride, a very sterically
hem 263 Nov 3, 2016 Preparation of Aldehydes from Acid alides? + l l acid chloride aka acyl chloride aldehyde Needed: 2 Actual eagents: 2 /Pd Al This is lithium tri-t-butoxy aluminum hydride, a very sterically
Recommended Reading: 20, , 13 (3rd edition); 19, , 13 (4th edition)
 Recommended Reading: 20, 21.2-21.4, 13 (3rd edition); 19, 20.2-20.4, 13 (4th edition) h 102 Problem Set 5 Due: Thursday, May 17 Before lass Problem 1 (2 points) Part A Hard and soft acids and bases (HSAB)
Recommended Reading: 20, 21.2-21.4, 13 (3rd edition); 19, 20.2-20.4, 13 (4th edition) h 102 Problem Set 5 Due: Thursday, May 17 Before lass Problem 1 (2 points) Part A Hard and soft acids and bases (HSAB)
Chapter 8. Acidity, Basicity and pk a
 Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
Chapter 8 Acidity, Basicity and pk a p182 In this reaction water is acting as a base, according to our definition above, by accepting a proton from HCl which in turn is acting as an acid by donating a
CHE1502. Tutorial letter 203/1/2016. General Chemistry 1B. Semester 1. Department of Chemistry
 E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
E1502/203/1/2016 Tutorial letter 203/1/2016 General hemistry 1B E1502 Semester 1 Department of hemistry This tutorial letter contains the answers to the questions in assignment 3. FIRST SEMESTER: KEY T
Problem 1 (4 points) D2h. C2v. Part A.
 Problem 1 (4 points) In 2004, a bimetallic Zr compound exhibiting side-on N2 binding was reported by Chirik and coworkers (Nature, 2004, 427, pp. 527-530). The crystal structure of this compound was obtained,
Problem 1 (4 points) In 2004, a bimetallic Zr compound exhibiting side-on N2 binding was reported by Chirik and coworkers (Nature, 2004, 427, pp. 527-530). The crystal structure of this compound was obtained,
EPIC LIGAND SURVEY: CARBON MONOXIDE
 EPIC LIGAND SURVEY: CARBON MONOXIDE As a young, growing field, organometallic chemistry may be taught in many ways. Some professors (e.g., Shaugnessy) spend a significant chunk of time discussing ligands,
EPIC LIGAND SURVEY: CARBON MONOXIDE As a young, growing field, organometallic chemistry may be taught in many ways. Some professors (e.g., Shaugnessy) spend a significant chunk of time discussing ligands,
Introduction to Organic Chemistry
 Introduction to rganic hemistry 59 Introduction to rganic hemistry andout 3 - chanism u u http://burton.chem.ox.ac.uk/teaching.html rganic hemistry J. layden,. Greeves, S. Warren Stereochemistry at a Glance
Introduction to rganic hemistry 59 Introduction to rganic hemistry andout 3 - chanism u u http://burton.chem.ox.ac.uk/teaching.html rganic hemistry J. layden,. Greeves, S. Warren Stereochemistry at a Glance
Name Date Class STUDY GUIDE FOR CONTENT MASTERY. covalent bond molecule sigma bond exothermic pi bond
 Covalent Bonding Section 9.1 The Covalent Bond In your textbook, read about the nature of covalent bonds. Use each of the terms below just once to complete the passage. covalent bond molecule sigma bond
Covalent Bonding Section 9.1 The Covalent Bond In your textbook, read about the nature of covalent bonds. Use each of the terms below just once to complete the passage. covalent bond molecule sigma bond
Hydrogen iodide is a strong acid and will drive the reverse reaction, meaning the forward reaction will not occur.
 EM 261 Oct 18, 2018 Photosynthesis and Related Reactions O 2 2 O 6 12 O 6 2 O N 3, S, Fe, u, o, other Natural Products D-Glucose R O R OR Ionic substitution S N 1 & R X X 2 hv Petroleum/ Alkanes R N 2
EM 261 Oct 18, 2018 Photosynthesis and Related Reactions O 2 2 O 6 12 O 6 2 O N 3, S, Fe, u, o, other Natural Products D-Glucose R O R OR Ionic substitution S N 1 & R X X 2 hv Petroleum/ Alkanes R N 2
Q.1 Predict what will happen when SiCl 4 is added to water.
 Transition etals F325 1 The aqueous chemistry of cations Hydrolysis when salts dissolve in water the ions are stabilised by polar water molecules hydrolysis can occur and the resulting solution can become
Transition etals F325 1 The aqueous chemistry of cations Hydrolysis when salts dissolve in water the ions are stabilised by polar water molecules hydrolysis can occur and the resulting solution can become
The elements 18.1 Occurrence and recovery 18.2 Physical properties Trends in chemical properties 18.3 Oxidation states across a series 18.
 Chapter 18 The d-block metals The elements 18.1 Occurrence and recovery 18.2 Physical properties Trends in chemical properties 18.3 Oxidation states across a series 18.4 Oxidation states down a group 18.5
Chapter 18 The d-block metals The elements 18.1 Occurrence and recovery 18.2 Physical properties Trends in chemical properties 18.3 Oxidation states across a series 18.4 Oxidation states down a group 18.5
Writing Chemical formula with polyatomic groups
 Writing Chemical formula with polyatomic groups 1. Use the Periodic table to determine the combining powers of single elements. Eg. Magnesium is in Group 2 and has a combining power of 2. 2. Use Table
Writing Chemical formula with polyatomic groups 1. Use the Periodic table to determine the combining powers of single elements. Eg. Magnesium is in Group 2 and has a combining power of 2. 2. Use Table
Chapter 11, Part 1: Polar substitution reactions involving alkyl halides
 hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
hapter 11, Part 1: Polar substitution reactions involving alkyl halides Overview: The nature of alkyl halides and other groups with electrophilic sp 3 hybridized leads them to react with nucleophiles and
Electron Configurations
 Section 3 Electron Configurations Key Terms electron configuration Pauli exclusion principle noble gas Aufbau principle Hund s rule noble-gas configuration Main Ideas Electrons fill in the lowest-energy
Section 3 Electron Configurations Key Terms electron configuration Pauli exclusion principle noble gas Aufbau principle Hund s rule noble-gas configuration Main Ideas Electrons fill in the lowest-energy
Introduction to Alkenes and Alkynes
 Introduction to Alkenes and Alkynes In an alkane, all covalent bonds between carbon were σ (σ bonds are defined as bonds where the electron density is symmetric about the internuclear axis) In an alkene,
Introduction to Alkenes and Alkynes In an alkane, all covalent bonds between carbon were σ (σ bonds are defined as bonds where the electron density is symmetric about the internuclear axis) In an alkene,
Q.1 Predict what will happen when SiCl 4 is added to water.
 Transition etals 2815 1 The aqueous chemistry of cations ydrolysis when salts dissolve in water the ions are stabilised by polar water molecules hydrolysis can occur and the resulting solution can become
Transition etals 2815 1 The aqueous chemistry of cations ydrolysis when salts dissolve in water the ions are stabilised by polar water molecules hydrolysis can occur and the resulting solution can become
Topic 9. Aldehydes & Ketones
 Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
Chemistry 2213a Fall 2012 Western University Topic 9. Aldehydes & Ketones A. Structure and Nomenclature The carbonyl group is present in aldehydes and ketones and is the most important group in bio-organic
14-1 Reactions Involving Gain or Loss of Ligands Reactions Involving Modification of Ligands
 Organometallic Reaction and Catalysis 14-1 Reactions Involving Gain or Loss of Ligands 14-2 Reactions Involving Modification of Ligands 14-3 Organometallic Catalysts 14-4 Heterogeneous Catalysts Inorganic
Organometallic Reaction and Catalysis 14-1 Reactions Involving Gain or Loss of Ligands 14-2 Reactions Involving Modification of Ligands 14-3 Organometallic Catalysts 14-4 Heterogeneous Catalysts Inorganic
Additions to the Carbonyl Groups
 Chapter 18 Additions to the Carbonyl Groups Nucleophilic substitution (S N 2andS N 1) reaction occurs at sp3 hybridized carbons with electronegative leaving groups Why? The carbon is electrophilic! Addition
Chapter 18 Additions to the Carbonyl Groups Nucleophilic substitution (S N 2andS N 1) reaction occurs at sp3 hybridized carbons with electronegative leaving groups Why? The carbon is electrophilic! Addition
The Organic Acids. Carboxylic Acids * *
 arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
arboxylic Acids The rganic Acids Some Notation: Acids and their conjugate bases pka ~ 15 - weak acid arboxylic Acid pka ~ 4 moderate acid - arboxylate Anion pka ~ -7 very strong acid l - l arboxylic acids
b.p.=100 C b.p.=65 C b.p.=-25 C µ=1.69 D µ=2.0 D µ=1.3 D
 Alcohols I eading: Wade chapter 10, sections 10-1- 10-12 Study Problems: 10-35, 10-37, 10-38, 10-39, 10-40, 10-42, 10-43 Key Concepts and Skills: Show how to convert alkenes, alkyl halides, and and carbonyl
Alcohols I eading: Wade chapter 10, sections 10-1- 10-12 Study Problems: 10-35, 10-37, 10-38, 10-39, 10-40, 10-42, 10-43 Key Concepts and Skills: Show how to convert alkenes, alkyl halides, and and carbonyl
All chemical bonding is based on the following relationships of electrostatics: 2. Each period on the periodic table
 UNIT VIII ATOMS AND THE PERIODIC TABLE 25 E. Chemical Bonding 1. An ELECTROSTATIC FORCE is All chemical bonding is based on the following relationships of electrostatics: The greater the distance between
UNIT VIII ATOMS AND THE PERIODIC TABLE 25 E. Chemical Bonding 1. An ELECTROSTATIC FORCE is All chemical bonding is based on the following relationships of electrostatics: The greater the distance between
Chapter 16. Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group. Physical Properties of Aldehydes and Ketones. Synthesis of Aldehydes
 Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
Nomenclature of Aldehydes and Ketones Chapter 16 Aldehydes and Ketones I. Aldehydes replace the -e of the parent alkane with -al The functional group needs no number Nucleophilic Addition to the Carbonyl
Chapter 20: Aldehydes and Ketones
 hapter 20: Aldehydes and Ketones [hapter 20 Sections: 20.1-20.7, 20.9-10.10, 20.13] 1. Nomenclature of Aldehydes and Ketones ketone ' aldehyde 2. eview of the Synthesis of Aldehydes and Ketones Br Br f
hapter 20: Aldehydes and Ketones [hapter 20 Sections: 20.1-20.7, 20.9-10.10, 20.13] 1. Nomenclature of Aldehydes and Ketones ketone ' aldehyde 2. eview of the Synthesis of Aldehydes and Ketones Br Br f
CHAPTER 2: Structure and Properties of Organic Molecules
 1 HAPTER 2: Structure and Properties of Organic Molecules Atomic Orbitals A. What are atomic orbitals? Atomic orbitals are defined by special mathematical functions called wavefunctions-- (x, y, z). Wavefunction,
1 HAPTER 2: Structure and Properties of Organic Molecules Atomic Orbitals A. What are atomic orbitals? Atomic orbitals are defined by special mathematical functions called wavefunctions-- (x, y, z). Wavefunction,
Atomic Structure. Atomic Structure. Atomic Structure. Atomic Structure. Electron Configuration. Electron Configuration
 Atomic Electron onfiguration Atomic Electron onfiguration z z z E 3rd SELL 3d 3p 3s y 2p x x y 2p y x y 2p z x 2nd SELL t SELL 2p x y z 2nd SELL 2p x y z y z x Atomic Ground State onfiguration Lowest energy
Atomic Electron onfiguration Atomic Electron onfiguration z z z E 3rd SELL 3d 3p 3s y 2p x x y 2p y x y 2p z x 2nd SELL t SELL 2p x y z 2nd SELL 2p x y z y z x Atomic Ground State onfiguration Lowest energy
Organic Chemistry Review: Topic 10 & Topic 20
 Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Organic Structure Alkanes C C σ bond Mechanism Substitution (Incoming atom or group will displace an existing atom or group in a molecule) Examples Occurs with exposure to ultraviolet light or sunlight,
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320
 Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Nuggets of Knowledge for Chapter 17 Dienes and Aromaticity Chem 2320 I. Isolated, cumulated, and conjugated dienes A diene is any compound with two or C=C's is a diene. Compounds containing more than two
Transition Metal Chemistry
 Transition Metal Chemistry 2 2011.12.2 Ⅰ Fundamental Organometallic Reactions Following four reactions are important formal reaction patterns in organotransition metal complexes, which would conveniently
Transition Metal Chemistry 2 2011.12.2 Ⅰ Fundamental Organometallic Reactions Following four reactions are important formal reaction patterns in organotransition metal complexes, which would conveniently
deactivation or decomposition is therefore quantified using the turnover number.
 A catalyst may be defined by two important criteria related to its stability and efficiency. Name both of these criteria and describe how they are defined with respect to stability or efficiency. A catalyst
A catalyst may be defined by two important criteria related to its stability and efficiency. Name both of these criteria and describe how they are defined with respect to stability or efficiency. A catalyst
Transition Metals and Coordination Chemistry
 Transition Metals and Coordination Chemistry Transition Metals Similarities within a given period and within a given group. Last electrons added are inner electrons (d s, f s). 20_431 Ce Th Pr Pa d U
Transition Metals and Coordination Chemistry Transition Metals Similarities within a given period and within a given group. Last electrons added are inner electrons (d s, f s). 20_431 Ce Th Pr Pa d U
Organometallic Rections 1: Reactions at the Metal
 E Organometallic Rections 1: Reactions at the Metal Three major classes of reactions: 1 Ligand Substitution associative (cf. S N 2) dissociative (cf. S N 1) interchange (not dealt with in this course)
E Organometallic Rections 1: Reactions at the Metal Three major classes of reactions: 1 Ligand Substitution associative (cf. S N 2) dissociative (cf. S N 1) interchange (not dealt with in this course)
Chapter 20: Aldehydes and Ketones
 Chapter 20: Aldehydes and Ketones [Chapter 20 Sections: 20.1-20.7, 20.9-10.10, 20.13] 1. Nomenclature of Aldehydes and Ketones ' ketone aldehyde f both aldehydes and ketones, the parent chain is the longest
Chapter 20: Aldehydes and Ketones [Chapter 20 Sections: 20.1-20.7, 20.9-10.10, 20.13] 1. Nomenclature of Aldehydes and Ketones ' ketone aldehyde f both aldehydes and ketones, the parent chain is the longest
Introduction & Definitions Catalytic Hydrogenations Dissolving Metal Reduction Reduction by Addition of H- and H+ Oxidation of Alcohols Oxidation of
 CEM 241- UNIT 4 xidation/reduction Reactions Redox chemistry 1 utline Introduction & Definitions Catalytic ydrogenations Dissolving Metal Reduction Reduction by Addition of - and + xidation of Alcohols
CEM 241- UNIT 4 xidation/reduction Reactions Redox chemistry 1 utline Introduction & Definitions Catalytic ydrogenations Dissolving Metal Reduction Reduction by Addition of - and + xidation of Alcohols
Chem 263 Notes March 2, 2006
 Chem 263 Notes March 2, 2006 Average for the midterm is 102.5 / 150 (approx. 68%). Preparation of Aldehydes and Ketones There are several methods to prepare aldehydes and ketones. We will only deal with
Chem 263 Notes March 2, 2006 Average for the midterm is 102.5 / 150 (approx. 68%). Preparation of Aldehydes and Ketones There are several methods to prepare aldehydes and ketones. We will only deal with
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions
 Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
Chapter 6: Organic Halogen Compounds; Substitution and Elimination Reactions Halogen compounds are important for several reasons. Simple alkyl and aryl halides, especially chlorides and bromides, are versatile
18-Electron Rule: Myth or Reality? An NBO Perspective
 18-Electron Rule: Myth or Reality? An NBO Perspective Eric CLOT UMR 5253 - CNRS, UM2, ENSCM, UM1 Interaction Experiment/Theory Experimental Chemists have Questions Where are the Nucleophilic and Electrophilic
18-Electron Rule: Myth or Reality? An NBO Perspective Eric CLOT UMR 5253 - CNRS, UM2, ENSCM, UM1 Interaction Experiment/Theory Experimental Chemists have Questions Where are the Nucleophilic and Electrophilic
Three Type Of Carbene Complexes
 Three Type f arbene omplexes arbene complexes have formal metal-to-carbon double bonds. Several types are known. The reactivity of the carbene and how it contributes to the overall electron counting is
Three Type f arbene omplexes arbene complexes have formal metal-to-carbon double bonds. Several types are known. The reactivity of the carbene and how it contributes to the overall electron counting is
Lecture Notes Chem 51C S. King. Chapter 20 Introduction to Carbonyl Chemistry; Organometallic Reagents; Oxidation & Reduction
 Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
Lecture Notes Chem 51C S. King Chapter 20 Introduction to Carbonyl Chemistry; rganometallic Reagents; xidation & Reduction I. The Reactivity of Carbonyl Compounds The carbonyl group is an extremely important
Learning Guide for Chapter 1 - Atoms and Molecules
 Learning Guide for hapter 1 - Atoms and Molecules I. Introduction to organic chemistry - p 1 II. Review of atomic structure - p 3 Elementary particles Periodic Table of Elements Electronegativity Atomic
Learning Guide for hapter 1 - Atoms and Molecules I. Introduction to organic chemistry - p 1 II. Review of atomic structure - p 3 Elementary particles Periodic Table of Elements Electronegativity Atomic
ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY )
 CHAPTER IV ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY ) n the end of the 1960s the leading specialist in homogeneous catalysis Jack Halpern wrote [1]: to develop
CHAPTER IV ACTIVATION OF C H BONDS BY LOW-VALENT METAL COMPLEXES ( THE ORGANOMETALLIC CHEMISTRY ) n the end of the 1960s the leading specialist in homogeneous catalysis Jack Halpern wrote [1]: to develop
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I. Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Ch.2 Polar Bonds and Their Consequences. 2.1 Polar Covalent Bonds and Electronegativity. polar covalent bonds: electron distribution is unsymmetrical
 2.1 Polar ovalent Bonds and Electronegativity polar covalent bonds: electron distribution is unsymmetrical Ionic haracter δ+ δ- + - X Y X Y X Y symmetrical covalent bond polar covalent bond ionic bond
2.1 Polar ovalent Bonds and Electronegativity polar covalent bonds: electron distribution is unsymmetrical Ionic haracter δ+ δ- + - X Y X Y X Y symmetrical covalent bond polar covalent bond ionic bond
Chapter 7 - Alkenes and Alkynes I
 Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
Andrew Rosen Chapter 7 - Alkenes and Alkynes I 7.1 - Introduction - The simplest member of the alkenes has the common name of ethylene while the simplest member of the alkyne family has the common name
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group
 Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Chapter 16 Aldehydes and Ketones I Nucleophilic Addition to the Carbonyl Group Nomenclature of Aldehydes and Ketones Aldehydes are named by replacing the -e of the corresponding parent alkane with -al
Lecture 11 Organic Chemistry 1
 EM 232 rganic hemistry I at hicago Lecture 11 rganic hemistry 1 Professor Duncan Wardrop February 16, 2010 1 Self Test Question What is the product(s) of the following reaction? 3 K( 3 ) 3 A 3 ( 3 ) 3
EM 232 rganic hemistry I at hicago Lecture 11 rganic hemistry 1 Professor Duncan Wardrop February 16, 2010 1 Self Test Question What is the product(s) of the following reaction? 3 K( 3 ) 3 A 3 ( 3 ) 3
Chapter 13: Alcohols and Phenols
 Chapter 13: Alcohols and Phenols [ Chapter 9 Sections: 9.10; Chapter 13 Sections: 13.1-13.3, 13.9-13.10] 1. Nomenclature of Alcohols simple alcohols C3 C3C2 Eddie Sachs 1927-1964 larger alcohols find the
Chapter 13: Alcohols and Phenols [ Chapter 9 Sections: 9.10; Chapter 13 Sections: 13.1-13.3, 13.9-13.10] 1. Nomenclature of Alcohols simple alcohols C3 C3C2 Eddie Sachs 1927-1964 larger alcohols find the
Chapter 7: Alcohols, Phenols and Thiols
 Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
Chapter 7: Alcohols, Phenols and Thiols 45 -Alcohols have the general formula R-OH and are characterized by the presence of a hydroxyl group, -OH. -Phenols have a hydroxyl group attached directly to an
CO 2 and CO activation
 2 and activation Most organic chemicals are currently made commercially from ethylene, a product of oil refining. Itispossiblethatinthenextseveraldecadeswemayhavetoshifttowardothercarbonsources for these
2 and activation Most organic chemicals are currently made commercially from ethylene, a product of oil refining. Itispossiblethatinthenextseveraldecadeswemayhavetoshifttowardothercarbonsources for these
Chemistry Higher level Paper 1
 M15/4/EMI/PM/ENG/TZ1/XX hemistry igher level Paper 1 Thursday 14 May 2015 (afternoon) 1 hour Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions.
M15/4/EMI/PM/ENG/TZ1/XX hemistry igher level Paper 1 Thursday 14 May 2015 (afternoon) 1 hour Instructions to candidates Do not open this examination paper until instructed to do so. Answer all the questions.
Chem 263 Nov 7, elimination reaction. There are many reagents that can be used for this reaction. Only three are given in this course:
 hem 263 Nov 7, 2013 Preparation of Ketones and Aldehydes from Alcohols xidation of Alcohols [] must have at least 1 E elimination reaction [] = oxidation; removal of electrons [] = reduction; addition
hem 263 Nov 7, 2013 Preparation of Ketones and Aldehydes from Alcohols xidation of Alcohols [] must have at least 1 E elimination reaction [] = oxidation; removal of electrons [] = reduction; addition
Organometallic Catalysis
 Organometallic Catalysis The catalysts we will study are termed homogeneous catalysts as they are dissolved in th e same solvent as the substrate. In contrast, heterogeneous catalysts, such as palladium
Organometallic Catalysis The catalysts we will study are termed homogeneous catalysts as they are dissolved in th e same solvent as the substrate. In contrast, heterogeneous catalysts, such as palladium
Chapter 8. Substitution reactions of Alkyl Halides
 Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
Chapter 8. Substitution reactions of Alkyl Halides There are two types of possible reaction in organic compounds in which sp 3 carbon is bonded to an electronegative atom or group (ex, halides) 1. Substitution
7. Haloalkanes (text )
 2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
2009, Department of hemistry, The University of Western Ontario 7.1 7. aloalkanes (text 7.1 7.10) A. Structure and Nomenclature Like hydrogen, the halogens have a valence of one. Thus, a halogen atom can
materials and their properties
 materials and their properties macroscopic properties phase state strength / stiffness electrical conductivity chemical properties color / transparence spectroscopical properties surface properties density
materials and their properties macroscopic properties phase state strength / stiffness electrical conductivity chemical properties color / transparence spectroscopical properties surface properties density
