THEORETICAL CALCULATION OF ELECTRON TRANSFER COEFFICIENT FOR PREDICTING THE FLOW OF ELECTRONS BY PM3, USING 20 AMINO ACIDS AND NICOTINE
|
|
|
- Maria Beasley
- 6 years ago
- Views:
Transcription
1 THEORETICAL CALCULATION OF ELECTRON TRANSFER COEFFICIENT FOR PREDICTING THE FLOW OF ELECTRONS BY PM3, USING 20 AMINO ACIDS AND NICOTINE Manuel Gonzalez Perez Doctor investigador SNI1, UPAEP, México Francisco Antonio Garcia Barrera Estudiante de Maestría, UPAEP, México Juan Francisco Mendez Diaz Estudiante de doctorado, Universitat Rovira i Virgili de Tarragona, España Maykel Gonzalez Torres Doctor investigador SNI1. CFATA-UNAM, México Juan Manuel Lopez Oglesby Doctor investigador SNIC, UPAEP, México Abstract In this work, an Electron Transfer Coefficient (ETC) it derived for various chemical species to help predict the flow of electrons in amino acidnicotine interactions. The quantum method "Semi-Empirical Parameterized Models Number 3" (SE-PM3) was set to calculate the variables HOMO- LUMO, the Gap Band (BG) and the Electrostatic Potential (EP) of 20 Amino Acids (AA) and Nicotine. In conclusion, the electrons need more energy to jump their BG when the ETC is large; thus, the electrons flow toward the lower value of the ETC. Tryptophan has an ETC = 19.1, Isoleucine has an ETC = , and Nicotine has an ETC = The ETC of nicotine is in the middle of the ETC of 20 different AA. Keywords: Electron Transfer Coefficient, Quantum Method PM3; HOMO- LUMO; Band Gap; Electrostatic Potential Introduction Electron transfer is an area widely studied in many fields of science. By examples: the chemistry of ceramics in which the consumption of ions is favored by decreasing the difference in Fermi energies (Fernández-Ibañez, 2000). The photochemical decarboxylation of carboxylic acids in organic chemistry (Yoshimi, 2010) and nanocomposites of fullerenes (Ito, 2012), 42
2 functionalized anthraquinone (Bezaine, 2005). The photo-induced electron transfers on a donor-acceptor system for converting solar energy into electrical power (Langa, 2012). Because of these fields of study, researchers have achieved the construction of optoelectronic devices (Kadish, 2010) and, it developed processes that are more efficient. In biochemistry, electron transfer generates reactions essential for life (Van der Ham, 2014). In the emerging field of the metalloprotein designs, researchers seek to prepare artificial proteins whose properties can mimic and perhaps improve many features found in natural metalloenzymes (Ogawa, 2006). Investigators have studied nicotine in its interactions with living matter extensively. An interesting study is the formation of carcinogens indoors, and its dangers from second and third-hand smoke (Sleimana, 2010). Quantum chemistry is a useful tool in these endeavors and has an important role in describing electron transfer. Relatively simple methods, such as SE-PM3 are available for calculating the nuclear reorganization of energy matrix elements and electronic coupling (Amini, 2003). There are different ways and methods to estimate the same parameter or variable. As an example, the variables and parameters of compounds used to treat Parkinson's disease, such as 4-acetylamino-2- (3, 5-dimethyl pyrazol-1-y l)- 6-pyridyl pyrimidine were calculated successfully (Morales-Bayuelo, 2014). Quantum methods allow the detection of various molecular structures before undertaking a synthetic extensive program (Greerling, 2002), (Abdallah, 2012). In this work, it proposed and derived using computational quantum chemistry methods an ETC to predict the flow of electrons in a given chemical system. As a test case, it calculated the coefficients for 20 AA and nicotine by the SE-PM3 method to organize the flow of electrons, predict chemical reactions, or transfer electrons from a molecule to molecule. Materials and methods It bought the molecular simulator Hyper Chem (HC). (Hyper Chem. Hypercube, MultiON for Windows. Serial # MultiON. Insurgentes Sur Tlacoquemecatl Col. del Valle, Delegación Benito Juárez, D. F., México CP ) It used HC SE-PM3 to draw the corresponding molecules. Then it selected, SE-PM3. It optimized the geometry with the Polak Ribiere method and conducted the calculation of the variables of HOMO-LUMO, BG, EP and other properties, resulting in a Tab-delimited table for BG and EP for AA. The specific parameters selected for each of the simulations were as follows: 43
3 SEMIEMPIRICAL PM3 Total Charge 0. Spin Multiplicity1. Spin Pairing RHF. State Lowest Converget Limit Interation Limit 50. Accelerate Convergence Yes. Polarizibility Not. Geometry Optimization: Algoritm Polak-Ribiere (Conjugate gradient). Termination condition RMS gradient of 0.1 kcal/amol. Algoritm Polak-Ribiere (Conjugate gradient), Termination condition or 255 maximun cycles. Algoritm Polak-Ribiere (Conjugate gradient) Termination condition or In vacuo. Screen refresh period 1 cyclest. PLOT OF ELECTROSTATIC POTENTIAL Molecular Property: Property Electrostatic Potential, Representation 3D. Mapped Isosurface. Isosufrace Grid: Grid Mesh Size Fine. Isosufrace Grid: Grid Layout Defoult. Contour Grid Starting Value Defoult. Contour Grid Increment Mapped Funtion Options Defoult. Transparency Level a criteria. Isosurface Rendering. Total charge density Rendering Wire mesh. FORMULATION BG was calculated by the formula BG = HOMO-LUMO (Odunola, 2007) and EP from the formula EP = E+ - E-. The ETC it calculated as enunciated in equation 1. HARDWARE Microsoft. Hardware ATA ST500DM002 IDB14SCSI Result and discussion Table 1 shows the BG values calculated in ev from lowest to highest in the fourth column. It observed that the Serine has the largest amount of BG. In contrast, Tryptophan has the lowest value of BG; thus, the valence electrons of tryptophan cross the smaller BG while the Serine electrons must pass a larger BG. Table 1 Values of BG of 20 AA in ev No. AA HOMO LUMO BG 1 Tryptophan Nicotine Methionine Tyrosine Cysteine Phenylalanine Histidine Arginine Threonine Aspartic acid Glycine Lysine Prolyne Leucine
4 15 Glutamic acid Glutamine Valine Asparagine Alanine Isoleucine Serine Given the same electrostatic potential for these listed AA, then the Tryptophan electrons need fewer times their electrostatic potential to cross the BP. By its nature, the EP varies in each AA, as shown in Table 2. Here, it was necessary to create an ETC to compare and quantify the flow direction of electron in chemical species. Table 2.Values of EP of 20 AA and Nicotine (ev) No. AA E+ E- EP 1 Leucine Isoleucine Valine Nicotine Tryptophan Glycine Phenylalanine Lysine Serine Alanine Cysteine Threonine Asparagine Methionine Glutamic acid Proline Glutamine Aspartic acid Arginine Tyrosine Histidine Table 2 shows EP values, lowest to highest in the fourth column. It observed that Histidine has the highest value of EP, whereas Leucine has the lowest value. It interpreted that the valence Histidine electrons have more energy available to cross its BG than Leucine electrons. With these two variables calculated for the chemical species in question, it is possible postulate a definition for the ETC: Equation 1 ETC = BG/EP 45
5 The ETC between molecules is equal to the division of the value of the BG (Table 1) by the value of EP (Table 2). The calculated ETC values it found in Table 3. Table 3 Values of ETC of 20 AA and Nicotine No. AA ETC 1 Tryptophan Tyrosine Histidine Methionine Arginine Cysteine Aspartic acid Glutamine Proline Threonine Nicotine Glutamic acid Asparagine Phenylalanine Alanine Lysine Glycine Serine Valine Leucine Isoleucine Interpretation of etc Higher values of ETC indicates that electrons have greater difficulty in passing from molecule to molecule of the same chemical species (which is similar to the electronic impedance), and lower values of ETC indicate easier electron transfer. The ETC is the multiple of its EP a chemical species needs to hop through its BG. In the particular case of ETC values of the studied AA, tryptophan needs 9.1 times its EP for its valence electrons to jump to a neighboring molecule of the same species. In the case of cysteine, whose ETC is , it would interpret this as meaning it requires times its PE to transfer its electrons to the same system. Nicotine With the same methodology, the ETC of nicotine it calculated as , placing it in the middle of all AA values. Given identical conditions for AA and nicotine, the electrons of nicotine have similar difficulties to jump its BG because nicotine is reactive or transfers its electrons with 20 AA. The cross bands of nicotine with two AA tryptophan and isoleucine it calculated, with the results shown in Table 4 for the nicotine-tryptophan interaction and Table 5 for the nicotine-isoleucine interaction. 46
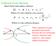
6 Where: NE+, NE-, TE+, TE-, IE+, IE- are the particular EPs of Nicotine, Tryptophan, and Isoleucine, respectively, and NL, NH, TL, TH, IL, IH, is HOMO-LUMO of Nicotine, Tryptophan, and Isoleucine, respectively. Table 4 Cross-Band of Nicotine vs Tryptophan Cross EP Cross BG ETC NE+ vs NE NL vs NH NE+ vs TE NL vs TH TE+ vs TE TL vs TH TE+ vs NE TL vs NH Table 5 Cross-Band or Nicotine vs Isoleucine Cross EP Cross BG ETC N+ vs N NL vs NH N+ vs I NL vs IH I+ vs I IL vs IH I+ vs N IL vs NH The most probable direction of the electrons at the first intersection is TH to TL (Fig. 1) due to the lower value of the ETC, while for the second crossing it is NH to IL (Fig 2). In the first case, there is a very low probability that nicotine reacts with tryptophan. The second case is very likely that nicotine reacts with isoleucine. Tryptophan TH TL ETC = 19.1 Nicotine NL NH ETC = Figure 1 Cross-band Tryptophan vs. Nicotine 47
7 Nicotine NH NL ETC = ETC = Isoleucine IH ETC = IL ETC = Figure 2 Cross-band Isoleucine vs. Nicotine Conclusion The electrons can jump a BG where its ETC has a lower value. If the ETC is lower in the same species, then the electrons jump the BG more quickly. Because of this, the substance is more likely to be stable and has a lower probability of reactivity. In the test cases, tryptophan has an ETC = 19.1, isoleucine has an ETC = , and nicotine has an ETC = The ETC for nicotine is in the center of the 20 AA ETCs. In the example: nicotine (40.873) vs. tryptophan (19.1) and nicotine vs. Isoleucine (54.573), nicotine would react with Isoleucine, but probably not with tryptophan. In general, when the internal ETC is highest (including cross-band ETCs) the probability of a chemical reaction is high. In other words, the valence electrons will move where the ETC has the smallest value. References: Abdallah H, Marvi J, Repic M, Sanghiran L, Habibah A. Chemical Reaction of Soybean Flavonoids with DNA: A Computational Study Using the Implicit Solvent Model. Int. J. Mol. Sci. 2012;(13): p Amini A, Harriman A. Computational methods for electron-transfer systems. Journal of Photochemistry and Photobiology C: Photochemistry Reviews. 2003; 4: p Bezaine A, Terme T, Vanelle P. An Electron Transfer Approach to the Preparation of Highly Functionalized Anthraquinones. Molecules. 2005;(10): p
8 Fernández-Ibáñez P, Malato S, De Las Nieves FJ. Oxide/Electrolyte interface: Electron transfer phenomena. Bol. Soc. Esp. Cerám. Vidrio. 2000; 39(4): p Greerling P, De Proft F. Chemical Reactivity as Described by Quantum Chemical Methods. Int. J. Mol. Sci (3): p Ito O, D Souza F. Recent Advances in Photoinduced Electron Transfer Processes of Fullerene-Based Molecular Assemblies and Nanocomposites. Molecules. 2012; 1(17): p Kadish KM, Smith KM, Guilard R. Self-Assembly and Photoinduced Electron Transfer. Handbook of Porphyrin Sciencie Singapore: World Scientific Publishing; Langa F, Nierengarten JF. Electron-Transfer Proces of Fullerenes Studied with Time-Resolved Spectroscopies. 2nd ed.: RCS; Morales-Bayuelo A, Valdiris V, Vivas-Reues R. Mathematical Analysis of a Series of 4-Acetylamino-2-(3,5-dimethylpyrazol-1-yl)-6-pyridylpyrimidines: A Simple Way to Relate Quantum Similarity to Local Chemical Reactivity Usingthe Gaussian Orbitals Localized Theory. Journal of Theoretical Chemistry. 2014; 2014: p Odunola OA, Semire B, Conformational Analysis (Semi Empirical PM3) and Electronic Properties of Functionalized Oligo(hexylpyrroles). E-Journal of Chemistry. 2007; 4(3): p Ogawa MY, Fan J, Fedorova A, Hong J, Kharenko OA, Kornilova AY, et al. Electron-Transfer Functionality of Synthetic Coiled-Coil Metalloproteins. J. Braz. Chem. Soc. 2006; 17(8): p Sleimana M, Gundela AL, Pankowb JF, Jacob P, Singera BC, Destaillatsa H. Formation of cancinogens indoors by surface-mediated reaction of nicotine with nitrous acid, leanding to potential thirdhand smoke hazards. 2010; 107(15): p Van der Ham, C. J., Koper, M. T., & Hetterscheid, D. G. Challenges in reduction of dinitrogen by proton and electron transfer. Chemical Society reviews. (2014). Yoshimi Y, Hayashi S, Nishikawa K, Haga Y, Maeda K, Morita T, et al. Influence of Solvent, Electron Acceptors and Arenes on Photochemical Decarboxylation of Free Carboxylic Acids via Single Electron Transfer (SET). Molecules 2010; 1(15): p
Applied quantum chemistry. Analysis of the rules of Markovnikov and anti-markovnikov
 Applied quantum chemistry. Analysis of the rules of Markovnikov and anti-markovnikov An explanation of the "controversy" in the history of these rules, using quantum chemistry methodology Dr. Manuel González-Pérez.
Applied quantum chemistry. Analysis of the rules of Markovnikov and anti-markovnikov An explanation of the "controversy" in the history of these rules, using quantum chemistry methodology Dr. Manuel González-Pérez.
Proteins: Characteristics and Properties of Amino Acids
 SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
SBI4U:Biochemistry Macromolecules Eachaminoacidhasatleastoneamineandoneacidfunctionalgroupasthe nameimplies.thedifferentpropertiesresultfromvariationsinthestructuresof differentrgroups.thergroupisoftenreferredtoastheaminoacidsidechain.
PROTEIN STRUCTURE AMINO ACIDS H R. Zwitterion (dipolar ion) CO 2 H. PEPTIDES Formal reactions showing formation of peptide bond by dehydration:
 PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
PTEI STUTUE ydrolysis of proteins with aqueous acid or base yields a mixture of free amino acids. Each type of protein yields a characteristic mixture of the ~ 20 amino acids. AMI AIDS Zwitterion (dipolar
Protein Secondary Structure Prediction
 part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
part of Bioinformatik von RNA- und Proteinstrukturen Computational EvoDevo University Leipzig Leipzig, SS 2011 the goal is the prediction of the secondary structure conformation which is local each amino
Exam III. Please read through each question carefully, and make sure you provide all of the requested information.
 09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
09-107 onors Chemistry ame Exam III Please read through each question carefully, and make sure you provide all of the requested information. 1. A series of octahedral metal compounds are made from 1 mol
Range of Certified Values in Reference Materials. Range of Expanded Uncertainties as Disseminated. NMI Service
 Calibration and Capabilities Amount of substance,, Russian Federation (Ural Scientific and Research Institiute Metrology, Rosstandart) (D.I. Mendeleyev Institute Metrology, Rosstandart) The uncertainty
Calibration and Capabilities Amount of substance,, Russian Federation (Ural Scientific and Research Institiute Metrology, Rosstandart) (D.I. Mendeleyev Institute Metrology, Rosstandart) The uncertainty
INTRODUCTION. Amino acids occurring in nature have the general structure shown below:
 Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Biochemistry I Laboratory Amino Acid Thin Layer Chromatography INTRODUCTION The primary importance of amino acids in cell structure and metabolism lies in the fact that they serve as building blocks for
Lecture 14 - Cells. Astronomy Winter Lecture 14 Cells: The Building Blocks of Life
 Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Lecture 14 Cells: The Building Blocks of Life Astronomy 141 Winter 2012 This lecture describes Cells, the basic structural units of all life on Earth. Basic components of cells: carbohydrates, lipids,
Periodic Table. 8/3/2006 MEDC 501 Fall
 Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Periodic Table 8/3/2006 MEDC 501 Fall 2006 1 rbitals Shapes of rbitals s - orbital p -orbital 8/3/2006 MEDC 501 Fall 2006 2 Ionic Bond - acl Electronic Structure 11 a :: 1s 2 2s 2 2p x2 2p y2 2p z2 3s
Sequence comparison: Score matrices. Genome 559: Introduction to Statistical and Computational Genomics Prof. James H. Thomas
 Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas Informal inductive proof of best alignment path onsider the last step in the best
Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas Informal inductive proof of best alignment path onsider the last step in the best
Sequence comparison: Score matrices
 Sequence comparison: Score matrices http://facultywashingtonedu/jht/gs559_2013/ Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best
Sequence comparison: Score matrices http://facultywashingtonedu/jht/gs559_2013/ Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best
Protein structure. Protein structure. Amino acid residue. Cell communication channel. Bioinformatics Methods
 Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Cell communication channel Bioinformatics Methods Iosif Vaisman Email: ivaisman@gmu.edu SEQUENCE STRUCTURE DNA Sequence Protein Sequence Protein Structure Protein structure ATGAAATTTGGAAACTTCCTTCTCACTTATCAGCCACCT...
Properties of amino acids in proteins
 Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Properties of amino acids in proteins one of the primary roles of DNA (but not the only one!) is to code for proteins A typical bacterium builds thousands types of proteins, all from ~20 amino acids repeated
Sequence comparison: Score matrices. Genome 559: Introduction to Statistical and Computational Genomics Prof. James H. Thomas
 Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best alignment path onsider the last step in
Sequence comparison: Score matrices Genome 559: Introduction to Statistical and omputational Genomics Prof James H Thomas FYI - informal inductive proof of best alignment path onsider the last step in
Viewing and Analyzing Proteins, Ligands and their Complexes 2
 2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
2 Viewing and Analyzing Proteins, Ligands and their Complexes 2 Overview Viewing the accessible surface Analyzing the properties of proteins containing thousands of atoms is best accomplished by representing
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell
 Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Using Higher Calculus to Study Biologically Important Molecules Julie C. Mitchell Mathematics and Biochemistry University of Wisconsin - Madison 0 There Are Many Kinds Of Proteins The word protein comes
Amino Acids and Peptides
 Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Amino Acids Amino Acids and Peptides Amino acid a compound that contains both an amino group and a carboxyl group α-amino acid an amino acid in which the amino group is on the carbon adjacent to the carboxyl
Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin
 Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin PART 1 : Metabolic flux analysis and minimal bioreaction modelling PART 2 : Dynamic metabolic flux analysis of underdetermined networks 2
Hypergraphs, Metabolic Networks, Bioreaction Systems. G. Bastin PART 1 : Metabolic flux analysis and minimal bioreaction modelling PART 2 : Dynamic metabolic flux analysis of underdetermined networks 2
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration
 A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
A rapid and highly selective colorimetric method for direct detection of tryptophan in proteins via DMSO acceleration Yanyan Huang, Shaoxiang Xiong, Guoquan Liu, Rui Zhao Beijing National Laboratory for
CHAPTER 29 HW: AMINO ACIDS + PROTEINS
 CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
CAPTER 29 W: AMI ACIDS + PRTEIS For all problems, consult the table of 20 Amino Acids provided in lecture if an amino acid structure is needed; these will be given on exams. Use natural amino acids (L)
Proteome Informatics. Brian C. Searle Creative Commons Attribution
 Proteome Informatics Brian C. Searle searleb@uw.edu Creative Commons Attribution Section structure Class 1 Class 2 Homework 1 Mass spectrometry and de novo sequencing Database searching and E-value estimation
Proteome Informatics Brian C. Searle searleb@uw.edu Creative Commons Attribution Section structure Class 1 Class 2 Homework 1 Mass spectrometry and de novo sequencing Database searching and E-value estimation
Lecture 15: Realities of Genome Assembly Protein Sequencing
 Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
Lecture 15: Realities of Genome Assembly Protein Sequencing Study Chapter 8.10-8.15 1 Euler s Theorems A graph is balanced if for every vertex the number of incoming edges equals to the number of outgoing
BENG 183 Trey Ideker. Protein Sequencing
 BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
BENG 183 Trey Ideker Protein Sequencing The following slides borrowed from Hong Li s Biochemistry Course: www.sb.fsu.edu/~hongli/4053notes Introduction to Proteins Proteins are of vital importance to biological
C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... methods use different attributes related to mis sense mutations such as
 C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... 2.1Introduction smentionedinchapter1,severalmethodsareavailabletoclassifyhuman missensemutationsintoeitherbenignorpathogeniccategoriesandthese
C h a p t e r 2 A n a l y s i s o f s o m e S e q u e n c e... 2.1Introduction smentionedinchapter1,severalmethodsareavailabletoclassifyhuman missensemutationsintoeitherbenignorpathogeniccategoriesandthese
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I Dr. Glen B Legge
 EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
EXAM 1 Fall 2009 BCHS3304, SECTION # 21734, GENERAL BIOCHEMISTRY I 2009 Dr. Glen B Legge This is a Scantron exam. All answers should be transferred to the Scantron sheet using a #2 pencil. Write and bubble
How did they form? Exploring Meteorite Mysteries
 Exploring Meteorite Mysteries Objectives Students will: recognize that carbonaceous chondrite meteorites contain amino acids, the first step towards living plants and animals. conduct experiments that
Exploring Meteorite Mysteries Objectives Students will: recognize that carbonaceous chondrite meteorites contain amino acids, the first step towards living plants and animals. conduct experiments that
Structures in equilibrium at point A: Structures in equilibrium at point B: (ii) Structure at the isoelectric point:
 ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
ame 21 F10-Final Exam Page 2 I. (42 points) (1) (16 points) The titration curve for L-lysine is shown below. Provide (i) the main structures in equilibrium at each of points A and B indicated below and
Discussion Section (Day, Time):
 Chemistry 27 Spring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Chemistry 27 Spring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Discussion Section (Day, Time): TF:
 ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
ame: Chemistry 27 Professor Gavin MacBeath arvard University Spring 2004 Final Exam Thursday, May 28, 2004 2:15 PM - 5:15 PM Discussion Section (Day, Time): Directions: TF: 1. Do not write in red ink.
PROTEIN SECONDARY STRUCTURE PREDICTION: AN APPLICATION OF CHOU-FASMAN ALGORITHM IN A HYPOTHETICAL PROTEIN OF SARS VIRUS
 Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Int. J. LifeSc. Bt & Pharm. Res. 2012 Kaladhar, 2012 Research Paper ISSN 2250-3137 www.ijlbpr.com Vol.1, Issue. 1, January 2012 2012 IJLBPR. All Rights Reserved PROTEIN SECONDARY STRUCTURE PREDICTION:
Enzyme Catalysis & Biotechnology
 L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
L28-1 Enzyme Catalysis & Biotechnology Bovine Pancreatic RNase A Biochemistry, Life, and all that L28-2 A brief word about biochemistry traditionally, chemical engineers used organic and inorganic chemistry
Potentiometric Titration of an Amino Acid. Introduction
 NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
NAME: Course: DATE Sign-Off: Performed: Potentiometric Titration of an Amino Acid Introduction In previous course-work, you explored the potentiometric titration of a weak acid (HOAc). In this experiment,
Collision Cross Section: Ideal elastic hard sphere collision:
 Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Collision Cross Section: Ideal elastic hard sphere collision: ( r r 1 ) Where is the collision cross-section r 1 r ) ( 1 Where is the collision distance r 1 r These equations negate potential interactions
Chemistry Chapter 22
 hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
hemistry 2100 hapter 22 Proteins Proteins serve many functions, including the following. 1. Structure: ollagen and keratin are the chief constituents of skin, bone, hair, and nails. 2. atalysts: Virtually
Translation. A ribosome, mrna, and trna.
 Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Translation The basic processes of translation are conserved among prokaryotes and eukaryotes. Prokaryotic Translation A ribosome, mrna, and trna. In the initiation of translation in prokaryotes, the Shine-Dalgarno
Module No. 31: Peptide Synthesis: Definition, Methodology & applications
 PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
PAPER 9: TECHNIQUES USED IN MOLECULAR BIOPHYSICS I Module No. 31: Peptide Synthesis: Definition, Methodology & applications Objectives: 1. Introduction 2. Synthesis of peptide 2.1. N-terminal protected
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 03
 01) Which of the following statements is not true regarding the active site of an enzyme? a. An active site is normally on the surface of an enzyme. b. An active site is normally hydrophobic in nature.
01) Which of the following statements is not true regarding the active site of an enzyme? a. An active site is normally on the surface of an enzyme. b. An active site is normally hydrophobic in nature.
1. Amino Acids and Peptides Structures and Properties
 1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
1. Amino Acids and Peptides Structures and Properties Chemical nature of amino acids The!-amino acids in peptides and proteins (excluding proline) consist of a carboxylic acid ( COOH) and an amino ( NH
Studies Leading to the Development of a Highly Selective. Colorimetric and Fluorescent Chemosensor for Lysine
 Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Supporting Information for Studies Leading to the Development of a Highly Selective Colorimetric and Fluorescent Chemosensor for Lysine Ying Zhou, a Jiyeon Won, c Jin Yong Lee, c * and Juyoung Yoon a,
Protein Identification Using Tandem Mass Spectrometry. Nathan Edwards Informatics Research Applied Biosystems
 Protein Identification Using Tandem Mass Spectrometry Nathan Edwards Informatics Research Applied Biosystems Outline Proteomics context Tandem mass spectrometry Peptide fragmentation Peptide identification
Protein Identification Using Tandem Mass Spectrometry Nathan Edwards Informatics Research Applied Biosystems Outline Proteomics context Tandem mass spectrometry Peptide fragmentation Peptide identification
Chemical Properties of Amino Acids
 hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
hemical Properties of Amino Acids Protein Function Make up about 15% of the cell and have many functions in the cell 1. atalysis: enzymes 2. Structure: muscle proteins 3. Movement: myosin, actin 4. Defense:
Discussion Section (Day, Time):
 Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Chemistry 27 Spring 2005 Exam 1 Chemistry 27 Professor Gavin MacBeath arvard University Spring 2005 our Exam 1 Friday, February 25, 2005 11:07 AM 12:00 PM Discussion Section (Day, Time): TF: Directions:
Properties of Amino Acids
 Biochemistry Department Date:19/9/ 2017 Properties of Amino Acids Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology 1 Intended Learning Outcomes (ILOs) By the end of this lecture,
Biochemistry Department Date:19/9/ 2017 Properties of Amino Acids Prof.Dr./ FAYDA Elazazy Professor of Biochemistry and Molecular Biology 1 Intended Learning Outcomes (ILOs) By the end of this lecture,
Protein Structure Bioinformatics Introduction
 1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
1 Swiss Institute of Bioinformatics Protein Structure Bioinformatics Introduction Basel, 27. September 2004 Torsten Schwede Biozentrum - Universität Basel Swiss Institute of Bioinformatics Klingelbergstr
Read more about Pauling and more scientists at: Profiles in Science, The National Library of Medicine, profiles.nlm.nih.gov
 2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
2018 Biochemistry 110 California Institute of Technology Lecture 2: Principles of Protein Structure Linus Pauling (1901-1994) began his studies at Caltech in 1922 and was directed by Arthur Amos oyes to
Solutions In each case, the chirality center has the R configuration
 CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
CAPTER 25 669 Solutions 25.1. In each case, the chirality center has the R configuration. C C 2 2 C 3 C(C 3 ) 2 D-Alanine D-Valine 25.2. 2 2 S 2 d) 2 25.3. Pro,, Trp, Tyr, and is, Trp, Tyr, and is Arg,
UNIT TWELVE. a, I _,o "' I I I. I I.P. l'o. H-c-c. I ~o I ~ I / H HI oh H...- I II I II 'oh. HO\HO~ I "-oh
 UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
UNT TWELVE PROTENS : PEPTDE BONDNG AND POLYPEPTDES 12 CONCEPTS Many proteins are important in biological structure-for example, the keratin of hair, collagen of skin and leather, and fibroin of silk. Other
CHEMISTRY ATAR COURSE DATA BOOKLET
 CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
CHEMISTRY ATAR COURSE DATA BOOKLET 2018 2018/2457 Chemistry ATAR Course Data Booklet 2018 Table of contents Periodic table of the elements...3 Formulae...4 Units...4 Constants...4 Solubility rules for
The Select Command and Boolean Operators
 The Select Command and Boolean Operators Part of the Jmol Training Guide from the MSOE Center for BioMolecular Modeling Interactive version available at http://cbm.msoe.edu/teachingresources/jmol/jmoltraining/boolean.html
The Select Command and Boolean Operators Part of the Jmol Training Guide from the MSOE Center for BioMolecular Modeling Interactive version available at http://cbm.msoe.edu/teachingresources/jmol/jmoltraining/boolean.html
Part 4 The Select Command and Boolean Operators
 Part 4 The Select Command and Boolean Operators http://cbm.msoe.edu/newwebsite/learntomodel Introduction By default, every command you enter into the Console affects the entire molecular structure. However,
Part 4 The Select Command and Boolean Operators http://cbm.msoe.edu/newwebsite/learntomodel Introduction By default, every command you enter into the Console affects the entire molecular structure. However,
The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II)
 1046 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 Myung-Jae Lee and Ui-Rak Kim The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II) Myung-Jae Lee and Ui-Rak Kim
1046 Bull. Korean Chem. Soc. 2004, Vol. 25, No. 7 Myung-Jae Lee and Ui-Rak Kim The Calculation of Physical Properties of Amino Acids Using Molecular Modeling Techniques (II) Myung-Jae Lee and Ui-Rak Kim
Lecture'18:'April'2,'2013
 CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
CM'224' 'rganic'chemistry'ii Spring'2013,'Des'Plaines' 'Prof.'Chad'Landrie 2 3 N cysteine (Cys) S oxidation S S 3 N cystine N 3 Lecture'18:'April'2,'2013 Disaccharides+&+Polysaccharides Amino+acids++(26.1926.3)
Discussion Section (Day, Time):
 Chemistry 27 pring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University pring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion ection (Day, Time): TF: Directions: 1.
Chemistry 27 pring 2005 Exam 3 Chemistry 27 Professor Gavin MacBeath arvard University pring 2005 our Exam 3 Friday April 29 th, 2005 11:07 AM 12:00 PM Discussion ection (Day, Time): TF: Directions: 1.
BCH 4053 Exam I Review Spring 2017
 BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
BCH 4053 SI - Spring 2017 Reed BCH 4053 Exam I Review Spring 2017 Chapter 1 1. Calculate G for the reaction A + A P + Q. Assume the following equilibrium concentrations: [A] = 20mM, [Q] = [P] = 40fM. Assume
Principles of Biochemistry
 Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
Principles of Biochemistry Fourth Edition Donald Voet Judith G. Voet Charlotte W. Pratt Chapter 4 Amino Acids: The Building Blocks of proteins (Page 76-90) Chapter Contents 1- Amino acids Structure: 2-
All Proteins Have a Basic Molecular Formula
 All Proteins Have a Basic Molecular Formula Homa Torabizadeh Abstract This study proposes a basic molecular formula for all proteins. A total of 10,739 proteins belonging to 9 different protein groups
All Proteins Have a Basic Molecular Formula Homa Torabizadeh Abstract This study proposes a basic molecular formula for all proteins. A total of 10,739 proteins belonging to 9 different protein groups
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS. Prokaryotes and Eukaryotes. DNA and RNA
 SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
SEQUENCE ALIGNMENT BACKGROUND: BIOINFORMATICS 1 Prokaryotes and Eukaryotes 2 DNA and RNA 3 4 Double helix structure Codons Codons are triplets of bases from the RNA sequence. Each triplet defines an amino-acid.
Evidence from Evolution Activity 75 Points. Fossils Use your textbook and the diagrams on the next page to answer the following questions.
 Name(s): Biology Evidence from Evolution Activity 75 Points Fossils Use your textbook and the diagrams on the next page to answer the following questions. 1. What are fossils? How are most fossils formed?
Name(s): Biology Evidence from Evolution Activity 75 Points Fossils Use your textbook and the diagrams on the next page to answer the following questions. 1. What are fossils? How are most fossils formed?
QUESTION 1 Which two functional groups react to form the peptide link found in proteins?
 QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
QUESTION 1 Which two functional groups react to form the peptide link found in proteins? NH and NH and NH2 and and NH2 A 2 B 2 C D OH COOH OH COOH QUESTION 2 The elements present in proteins are A B C
Proteomics. November 13, 2007
 Proteomics November 13, 2007 Acknowledgement Slides presented here have been borrowed from presentations by : Dr. Mark A. Knepper (LKEM, NHLBI, NIH) Dr. Nathan Edwards (Center for Bioinformatics and Computational
Proteomics November 13, 2007 Acknowledgement Slides presented here have been borrowed from presentations by : Dr. Mark A. Knepper (LKEM, NHLBI, NIH) Dr. Nathan Edwards (Center for Bioinformatics and Computational
Dental Biochemistry EXAM I
 Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Dental Biochemistry EXAM I August 29, 2005 In the reaction below: CH 3 -CH 2 OH -~ ethanol CH 3 -CHO acetaldehyde A. acetoacetate is being produced B. ethanol is being oxidized to acetaldehyde C. acetaldehyde
Chapter 2. Methodology
 Chapter 2 Methodology Chapter 2 Methodology NORMA AUREA RANGEL-VÁZQUEZ 1 FRANCISCO RODRÍGUEZ FÉLIX 2 1 División de Estudios de Posgrado e Investigación del Instituto Tecnológico de Aguascalientes, Ave.
Chapter 2 Methodology Chapter 2 Methodology NORMA AUREA RANGEL-VÁZQUEZ 1 FRANCISCO RODRÍGUEZ FÉLIX 2 1 División de Estudios de Posgrado e Investigación del Instituto Tecnológico de Aguascalientes, Ave.
Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene
![Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene Molecular Selective Binding of Basic Amino Acids by a Water-soluble Pillar[5]arene](/thumbs/77/75727507.jpg) Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
Electronic supplementary information Molecular Selective Binding of Basic Amino Acids y a Water-solule Pillar[5]arene Chunju Li,* a, Junwei Ma, a Liu Zhao, a Yanyan Zhang, c Yihua Yu, c Xiaoyan Shu, a
12/6/12. Dr. Sanjeeva Srivastava IIT Bombay. Primary Structure. Secondary Structure. Tertiary Structure. Quaternary Structure.
 Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Dr. anjeeva rivastava Primary tructure econdary tructure Tertiary tructure Quaternary tructure Amino acid residues α Helix Polypeptide chain Assembled subunits 2 1 Amino acid sequence determines 3-D structure
Practice Midterm Exam 200 points total 75 minutes Multiple Choice (3 pts each 30 pts total) Mark your answers in the space to the left:
 MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
MITES ame Practice Midterm Exam 200 points total 75 minutes Multiple hoice (3 pts each 30 pts total) Mark your answers in the space to the left: 1. Amphipathic molecules have regions that are: a) polar
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor
 LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
LS1a Fall 2014 Problem Set #2 Due Monday 10/6 at 6 pm in the drop boxes on the Science Center 2 nd Floor Note: Adequate space is given for each answer. Questions that require a brief explanation should
Dental Biochemistry Exam The total number of unique tripeptides that can be produced using all of the common 20 amino acids is
 Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
Exam Questions for Dental Biochemistry Monday August 27, 2007 E.J. Miller 1. The compound shown below is CH 3 -CH 2 OH A. acetoacetate B. acetic acid C. acetaldehyde D. produced by reduction of acetaldehyde
BIS Office Hours
 BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
BIS103-001 001 ffice ours TUE (2-3 pm) Rebecca Shipman WED (9:30-10:30 am) TUE (12-1 pm) Stephen Abreu TUR (12-1 pm) FRI (9-11 am) Steffen Abel Lecture 2 Topics Finish discussion of thermodynamics (ΔG,
Patrick: An Introduction to Medicinal Chemistry 5e Chapter 04
 01) Which of the following statements is not true about receptors? a. Most receptors are proteins situated inside the cell. b. Receptors contain a hollow or cleft on their surface which is known as a binding
01) Which of the following statements is not true about receptors? a. Most receptors are proteins situated inside the cell. b. Receptors contain a hollow or cleft on their surface which is known as a binding
Edward Susko Department of Mathematics and Statistics, Dalhousie University. Introduction. Installation
 1 dist est: Estimation of Rates-Across-Sites Distributions in Phylogenetic Subsititution Models Version 1.0 Edward Susko Department of Mathematics and Statistics, Dalhousie University Introduction The
1 dist est: Estimation of Rates-Across-Sites Distributions in Phylogenetic Subsititution Models Version 1.0 Edward Susko Department of Mathematics and Statistics, Dalhousie University Introduction The
Protein Struktur (optional, flexible)
 Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
Protein Struktur (optional, flexible) 22/10/2009 [ 1 ] Andrew Torda, Wintersemester 2009 / 2010, AST nur für Informatiker, Mathematiker,.. 26 kt, 3 ov 2009 Proteins - who cares? 22/10/2009 [ 2 ] Most important
1. Wings 5.. Jumping legs 2. 6 Legs 6. Crushing mouthparts 3. Segmented Body 7. Legs 4. Double set of wings 8. Curly antennae
 Biology Cladogram practice Name Per Date What is a cladogram? It is a diagram that depicts evolutionary relationships among groups. It is based on PHYLOGENY, which is the study of evolutionary relationships.
Biology Cladogram practice Name Per Date What is a cladogram? It is a diagram that depicts evolutionary relationships among groups. It is based on PHYLOGENY, which is the study of evolutionary relationships.
5) An amino acid that doesn't exist in proteins: - Tyrosine - Tryptophan - Cysteine - ornithine* 6) How many tripeptides can be formed from using one
 Biochemistry First 1) An amino acid that does not have L & D: - proline - serine - Glycine* - valine 2) An amino acid that exists in beta-bends and turns: - Gly* - Pro - Arg - Asp 3) An amino acid that
Biochemistry First 1) An amino acid that does not have L & D: - proline - serine - Glycine* - valine 2) An amino acid that exists in beta-bends and turns: - Gly* - Pro - Arg - Asp 3) An amino acid that
Towards Understanding the Origin of Genetic Languages
 Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
Towards Understanding the Origin of Genetic Languages Why do living organisms use 4 nucleotide bases and 20 amino acids? Apoorva Patel Centre for High Energy Physics and Supercomputer Education and Research
ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity
 ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity Absorption/Acid-Base Case (p. 42) Question #1: Drug Cetirizine Clemastin e Functional groups
ANSWERS TO CASE STUDIES Chapter 2: Drug Design and Relationship of Functional Groups to Pharmacologic Activity Absorption/Acid-Base Case (p. 42) Question #1: Drug Cetirizine Clemastin e Functional groups
Biochemistry by Mary K. Campbell & Shawn O. Farrell 8th. Ed. 2016
 3 Biochemistry by Mary K. Campbell & Shawn. Farrell 8th. Ed. 2016 3-1 3 Amino Acids & Peptides 3-2 3 Learning bjectives 1. What are amino acids, and what is their threedimensional structure? 2. What are
3 Biochemistry by Mary K. Campbell & Shawn. Farrell 8th. Ed. 2016 3-1 3 Amino Acids & Peptides 3-2 3 Learning bjectives 1. What are amino acids, and what is their threedimensional structure? 2. What are
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013
 Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
Protein Structure Marianne Øksnes Dalheim, PhD candidate Biopolymers, TBT4135, Autumn 2013 The presentation is based on the presentation by Professor Alexander Dikiy, which is given in the course compedium:
Content : Properties of amino acids.. Separation and Analysis of Amino Acids
 قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
قسم الكيمياء الحيوية.دولت على سالمه د. استاذ الكيمياء الحيوية ٢٠١٥-٢٠١٤ المحاضرة الثانية 1 Content : Properties of amino acids.. Separation and Analysis of Amino Acids 2 3 Physical Properties of Amino
A Plausible Model Correlates Prebiotic Peptide Synthesis with. Primordial Genetic Code
 Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Electronic Supplementary Material (ESI) for ChemComm. This journal is The Royal Society of Chemistry 2018 A Plausible Model Correlates Prebiotic Peptide Synthesis with Primordial Genetic Code Jianxi Ying,
Systematic approaches to study cancer cell metabolism
 Systematic approaches to study cancer cell metabolism Kivanc Birsoy Laboratory of Metabolic Regulation and Genetics The Rockefeller University, NY Cellular metabolism is complex ~3,000 metabolic genes
Systematic approaches to study cancer cell metabolism Kivanc Birsoy Laboratory of Metabolic Regulation and Genetics The Rockefeller University, NY Cellular metabolism is complex ~3,000 metabolic genes
A Theoretical Inference of Protein Schemes from Amino Acid Sequences
 A Theoretical Inference of Protein Schemes from Amino Acid Sequences Angel Villahoz-Baleta angel_villahozbaleta@student.uml.edu ABSTRACT Proteins are based on tri-dimensional dispositions generated from
A Theoretical Inference of Protein Schemes from Amino Acid Sequences Angel Villahoz-Baleta angel_villahozbaleta@student.uml.edu ABSTRACT Proteins are based on tri-dimensional dispositions generated from
MULTIPLE CHOICE 2 points each
 Name: Date: Score: / 110 Chapter 1/ TEST 1 OPEN BOOK KEY Organic Chemistry MULTIPLE CHOICE 2 points each 1. An atom of which element would have an electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 1? a.
Name: Date: Score: / 110 Chapter 1/ TEST 1 OPEN BOOK KEY Organic Chemistry MULTIPLE CHOICE 2 points each 1. An atom of which element would have an electron configuration of 1s 2 2s 2 2p 6 3s 2 3p 1? a.
GENETIC CODE AS A HARMONIC SYSTEM: TWO SUPPLEMENTS. Miloje M. Rakočević
 GENETIC CODE AS A HARMONIC SYSTEM: TWO SUPPLEMENTS Miloje M. Rakočević Department of Chemistry, Faculty of Science, University of Niš, Ćirila i Metodija 2, Serbia (E-mail: m.m.r@eunet.yu) Abstract. The
GENETIC CODE AS A HARMONIC SYSTEM: TWO SUPPLEMENTS Miloje M. Rakočević Department of Chemistry, Faculty of Science, University of Niš, Ćirila i Metodija 2, Serbia (E-mail: m.m.r@eunet.yu) Abstract. The
Jack Smith. Center for Environmental, Geotechnical and Applied Science. Marshall University
 Jack Smith Center for Environmental, Geotechnical and Applied Science Marshall University -- Division of Science and Research WV Higher Education Policy Commission WVU HPC Summer Institute June 20, 2014
Jack Smith Center for Environmental, Geotechnical and Applied Science Marshall University -- Division of Science and Research WV Higher Education Policy Commission WVU HPC Summer Institute June 20, 2014
1014NSC Fundamentals of Biochemistry Semester Summary
 1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
1014NSC Fundamentals of Biochemistry Semester Summary Griffith University, Nathan Campus Semester 1, 2014 Topics include: - Water & ph - Protein Diversity - Nucleic Acids - DNA Replication - Transcription
National Nutrient Database for Standard Reference Release 28 slightly revised May, 2016
 National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 005, Cheese, cottage, lowfat, 2% milkfat Report Date: February 23, 208 02:7 EST values and weights are for
National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 005, Cheese, cottage, lowfat, 2% milkfat Report Date: February 23, 208 02:7 EST values and weights are for
Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids
 Anal. Chem. 2001, 73, 3028-3034 Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids Luther W. Beegle and Isik Kanik* Jet Propulsion
Anal. Chem. 2001, 73, 3028-3034 Electrospray Ionization High-Resolution Ion Mobility Spectrometry for the Detection of Organic Compounds, 1. Amino Acids Luther W. Beegle and Isik Kanik* Jet Propulsion
Contents. 1. Building a molecular model 2. Additional Procedures and Options: 3. Selecting a calculation method
 Getting started with HyperChem. Program description. In this project you will create simple organic molecules and learn how to manipulate them. However, before starting you should create your home directory
Getting started with HyperChem. Program description. In this project you will create simple organic molecules and learn how to manipulate them. However, before starting you should create your home directory
NAME. EXAM I I. / 36 September 25, 2000 Biochemistry I II. / 26 BICH421/621 III. / 38 TOTAL /100
 EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
EXAM I I. / 6 September 25, 2000 Biochemistry I II. / 26 BIH421/621 III. / 8 TOTAL /100 I. MULTIPLE HOIE (6 points) hoose the BEST answer to the question by circling the appropriate letter. 1. An amino
Course syllabus for Chemistry 109C Organic Chemistry
 Course syllabus for Chemistry 109C Organic Chemistry Class meets: Tue, Thu 5:00 6:15 PM Chem 1179 Spring 2005 Instructor: Prof. Kalju Kahn, Office: PSB-N 1511, E-mail: kalju@chem.ucsb.edu Phone: 893-6157
Course syllabus for Chemistry 109C Organic Chemistry Class meets: Tue, Thu 5:00 6:15 PM Chem 1179 Spring 2005 Instructor: Prof. Kalju Kahn, Office: PSB-N 1511, E-mail: kalju@chem.ucsb.edu Phone: 893-6157
Novel 2D Graphic Representation of Protein Sequence and Its Application
 Journal of Fiber Bioengineering and Informatics 7:1 (2014) 23 33 doi:10.3993/jfbi03201403 Novel 2D Graphic Representation of Protein Sequence and Its Application Yongbin Zhao, Xiaohong Li, Zhaohui Qi College
Journal of Fiber Bioengineering and Informatics 7:1 (2014) 23 33 doi:10.3993/jfbi03201403 Novel 2D Graphic Representation of Protein Sequence and Its Application Yongbin Zhao, Xiaohong Li, Zhaohui Qi College
7.05 Spring 2004 February 27, Recitation #2
 Recitation #2 Contact Information TA: Victor Sai Recitation: Friday, 3-4pm, 2-132 E-mail: sai@mit.edu ffice ours: Friday, 4-5pm, 2-132 Unit 1 Schedule Recitation/Exam Date Lectures covered Recitation #2
Recitation #2 Contact Information TA: Victor Sai Recitation: Friday, 3-4pm, 2-132 E-mail: sai@mit.edu ffice ours: Friday, 4-5pm, 2-132 Unit 1 Schedule Recitation/Exam Date Lectures covered Recitation #2
Review of General & Organic Chemistry
 Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
Review of General & Organic Chemistry Diameter of a nucleus is only about 10-15 m. Diameter of an atom is only about 10-10 m. Fig 3.1 The structure of an atom Periodic Table, shown below, is a representation
1 large 50g. 1 extra large 56g
 National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 023, Egg, whole, raw, fresh Report Date: February 08, 208 06:8 EST values and weights are for edible portion.
National base for Standard Reference Release 28 slightly revised May, 206 Full Report (All s) 023, Egg, whole, raw, fresh Report Date: February 08, 208 06:8 EST values and weights are for edible portion.
Protein Struktur. Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep. wake up at slide 39
 Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
Protein Struktur Biologen und Chemiker dürfen mit Handys spielen (leise) go home, go to sleep wake up at slide 39 Andrew Torda, Wintersemester 2016/ 2017 Andrew Torda 17.10.2016 [ 1 ] Proteins - who cares?
NSCI Basic Properties of Life and The Biochemistry of Life on Earth
 NSCI 314 LIFE IN THE COSMOS 4 Basic Properties of Life and The Biochemistry of Life on Earth Dr. Karen Kolehmainen Department of Physics CSUSB http://physics.csusb.edu/~karen/ WHAT IS LIFE? HARD TO DEFINE,
NSCI 314 LIFE IN THE COSMOS 4 Basic Properties of Life and The Biochemistry of Life on Earth Dr. Karen Kolehmainen Department of Physics CSUSB http://physics.csusb.edu/~karen/ WHAT IS LIFE? HARD TO DEFINE,
Journal of Chemical and Pharmaceutical Research
 Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(5):41-50 Molar extinction coefficients of some amino acids
Available online www.jocpr.com Journal of Chemical and Pharmaceutical Research ISSN No: 0975-7384 CODEN(USA): JCPRC5 J. Chem. Pharm. Res., 2011, 3(5):41-50 Molar extinction coefficients of some amino acids
Using an Artificial Regulatory Network to Investigate Neural Computation
 Using an Artificial Regulatory Network to Investigate Neural Computation W. Garrett Mitchener College of Charleston January 6, 25 W. Garrett Mitchener (C of C) UM January 6, 25 / 4 Evolution and Computing
Using an Artificial Regulatory Network to Investigate Neural Computation W. Garrett Mitchener College of Charleston January 6, 25 W. Garrett Mitchener (C of C) UM January 6, 25 / 4 Evolution and Computing
GENERAL BIOLOGY LABORATORY EXERCISE Amino Acid Sequence Analysis of Cytochrome C in Bacteria and Eukarya Using Bioinformatics
 GENERAL BIOLOGY LABORATORY EXERCISE Amino Acid Sequence Analysis of Cytochrome C in Bacteria and Eukarya Using Bioinformatics INTRODUCTION: All life forms undergo metabolic processes to obtain energy.
GENERAL BIOLOGY LABORATORY EXERCISE Amino Acid Sequence Analysis of Cytochrome C in Bacteria and Eukarya Using Bioinformatics INTRODUCTION: All life forms undergo metabolic processes to obtain energy.
CHEM 3653 Exam # 1 (03/07/13)
 1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
1. Using phylogeny all living organisms can be divided into the following domains: A. Bacteria, Eukarya, and Vertebrate B. Archaea and Eukarya C. Bacteria, Eukarya, and Archaea D. Eukarya and Bacteria
