Diffusion. Diffusion. Diffusion in Solid Materials
|
|
|
- Betty Johnson
- 6 years ago
- Views:
Transcription
1 Atoms movements in materials Diffusion Movement of atoms in solids, liquids and gases is very important Eamples: Hardening steel, chrome-plating, gas reactions, Si wafers.. etc. We will study: Atomic mechanisms of diffusion Mathematics of diffusion Factors affecting diffusion Eamples Steady state diffusion Nonsteady state diffusion Summary Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/1 Diffusion diffusion is the mass transport through atomic motion at high temperature can have self-diffusion or interdiffusion between two materials Self-diffusion occurs in pure elements Eample: Two chambers, each containing a different gas, separated by a removable barrier; when the barrier is pulled away, interdiffusion occurs O N driving force is the chemical or concentration gradient through materials or diffusion couple Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/ Diffusion in Solid Materials Recall Arhenius equation Heat temperature should be high enough to overcome energy barriers to atomic motion. N v N o ep Q kt where N v = equilibrium # of vacancies N o = total number of lattice sites k = Boltzman s constant Q = activation energy for vacancy formation Heat causes atoms to vibrate Vibration amplitude increases with temperature Melting occurs when vibrations are sufficient to rupture bonds Eample: lead (Pb) Eample: lead (Pb) Q slope = - k The process of substitutional diffusion requires the presence of vacancies (Vacancies give the atoms a place to move) Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/3 As T, number of vacancies, and energy, so diffusion is faster Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/4
2 Diffusion Mechanisms INTERSTITIAL DIFFUSION Atoms are constantly in motion and vibrating change of atomic position requires: vacant site energyto break atomic bonds Two types of diffusion mechanism: vacancy diffusion interstitial diffusion movement of vacancies in one direction is equivalent to atomic movement in the opposite direction Vacancy diffusion Interstitial diffusion Interstitial atom moves from one interstitial site to another (empty) Energy needed, again to squeeze past atoms. Eample:.. Usually much faster because many more empty interstitial sites and no vacancies are required Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/5 Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/6 Diffusion Diffusion is a.. process rate of diffusion is important diffusion flu (J) is defined as the mass, M, diffusing through unit area, A, per unit time, t M 1 dm J or J At A dt If flu does not change with time: oncentration gradient = Fick s 1 st law: Steady-State Diffusion d d d J D d Where D is the diffusion coefficient (diffusivity) or speed of diffusion (m /s). -ve because atoms diffuse down concentration gradient Eample of steady-state diffusion is gas diffusing through a metal plate (gas pressure constant). Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/7 Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/8
3 Eample The purification of H (gas) by diffusion through a Pd sheet was discussed in allister 5.3. ompute the number of kilograms of hydrogen that pass per hour through a 5 mm thick sheet of Pd having an area of 0.0 m at 500. Assume a diffusion coefficient of m /s, that the concentrations at the high and low pressure sides of the plate are.4 and 0.6 kg of H per m 3 of Pd, and that steady state conditions have been attained. hapter 5 allister 8e. (courtesy of Surface Division, Midland- Ross.) ase Hardening - Eample of interstitial diffusion is a case hardened gear. - Diffuse carbon atoms into the host iron atoms at the surface. Recall, flu is mass per unit time per unit area. Thus, multiplying J by area and time will give total mass. Result: The "ase" is - hard to deform: atoms "lock" planes from shearing. - hard to crack: atoms put the surface in compression. Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/9 Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/10 Steady State: the concentration profile doesn't change with time. J(left) Steady State Diffusion: Summary J(right) oncentration,, in the bo doesn t change w/time. Apply Fick's First Law: J D d d d d If J _left = J _right, then d left d right Result: the slope, d/d, must be constant (i.e., slope doesn't vary with position)! Steady State: J(left) = J(right) Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/11 Non-steady State Diffusion In most real situations diffusion is not.. Flu and concentration gradient vary with time The changes of the concentration profile is given in this case by a differential equation,. second law: D t If diffusion coefficient is independent of composition then: D t Solution of this equation is concentration profile as function of time, Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/1
4 Fick s Second Law t D Solution requires boundary conditions. A useful solution is for a semi-infinite solid when the surface concentration remains constant. What is semi-infinite solid? semi infinite bar: If non of the diffusing atoms reaches the bar end during the time over which the diffusion takes place. l >10(Dt), where l: bar length, D: diffusion coefficient and t: time Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/13 Fick s Second Law - Application Simple boundary condition is where the surface concentration is held constant, e.g. gas phase with constant partial pressure at the surface onditions are: before diffusion, solute atom have a homogeneous concentration of o is zero at the surface and increases with distance into the solid time is zero just before diffusion begins Mathematically, for t = 0, = o at 0 t > 0, = s at = 0 and = o at = s = constant surface concentration applying these boundary conditions gives: s 0 0 = 1 erf Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/14 Dt Application of Fick s Second Law Tabulation of Error Function Values 0 s 0 = 1 erf Dt s = Surface concentration which remains constant 0 = Initial concentration in solid = oncentration at distance into sample after time t. D = Diffusivity of solute in solvent, m s -1 t = Time, seconds erf = Gaussian error function, based on integration of the bell shaped curve z Dt Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/15 s 0 = 1 erf (z) z Dt 0 Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/16
5 Special case If it is desired to achieve a particular concentration of solute atoms in an alloy then: Dt Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/17 and 1 o s Dt o.. These equations facilitate the diffusion computation for this specific case (see eample 5.3). Factors Affecting Solid-State Diffusion Diffusing species and host material are important smaller atoms can squeeze in between host atoms more easily in lower packing density host material easier for atoms to migrate with fewer bonds to epand eg faster in more open lattice (B faster than F) (..) in lower melting point host material weaker bonds (easier to push apart) Temperature has a strong effect on diffusion rates: Qd D Doep RT Where: D o = temperature dependant pre-eponential constant Q d = activation energy for diffusion R = gas constant (8.31 J/mol K) T = absolute temperature Activation energy is the barrier to diffusion and controls the diffusion coefficient, D Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/18 Typical Diffusion Data Eperimental Determination of Activation Energy Qd D Doep RT Taking logs: Qd 1 logd logdo.3r T Typical values for preeponential (D o ) and activation energy (after Kittel, Solid State Physics 5 th ed.) Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/19 FIGURE 5.7 Plot of the logarithm of the diffusion coefficient versus the reciprocal of absolute temperature for several metals. plot log D vs. 1/T slope =.. intercept =.. Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/0
6 Eample An F Fe- alloy initially containing 0.35 wt. % is eposed to an oygenrich (and carbon-free) atmosphere at 1400 K (117 ). Under these conditions the carbon in the alloy diffuses toward the surface and reacts with the oygen in the atmosphere; that is, the carbon concentration at the surface is maintained essentially at 0 wt. %. (This process of carbon depletion is termed decarburization). At what position will the carbon concentration be 0.15 wt. % after a 10-hour treatment. The value of D at 1400 K is m /s. SUMMARY Diffusion FASTER for... Diffusion SLOWER for.... crystal structures structures.. melting T materials materials with bonding.. diffusing atoms density materials. melting T materials materials with.. bonding.. diffusing atoms density materials Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/1 Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/ Net topic: Mechanical properties of materials Dr. M. Medraj Mech. Eng. Dept. oncordia University Mech 1 lecture 9/3
Introduction To Materials Science FOR ENGINEERS, Ch. 5. Diffusion. MSE 201 Callister Chapter 5
 Diffusion MSE 201 Callister Chapter 5 1 Goals: Diffusion - how do atoms move through solids? Fundamental concepts and language Diffusion mechanisms Vacancy diffusion Interstitial diffusion Impurities Diffusion
Diffusion MSE 201 Callister Chapter 5 1 Goals: Diffusion - how do atoms move through solids? Fundamental concepts and language Diffusion mechanisms Vacancy diffusion Interstitial diffusion Impurities Diffusion
DIFFUSION IN SOLIDS. IE-114 Materials Science and General Chemistry Lecture-5
 DIFFUSION IN SOLIDS IE-114 Materials Science and General Chemistry Lecture-5 Diffusion The mechanism by which matter is transported through matter. It is related to internal atomic movement. Atomic movement;
DIFFUSION IN SOLIDS IE-114 Materials Science and General Chemistry Lecture-5 Diffusion The mechanism by which matter is transported through matter. It is related to internal atomic movement. Atomic movement;
Lecture 1: Atomic Diffusion
 Part IB Materials Science & Metallurgy H. K. D. H. Bhadeshia Course A, Metals and Alloys Lecture 1: Atomic Diffusion Mass transport in a gas or liquid generally involves the flow of fluid (e.g. convection
Part IB Materials Science & Metallurgy H. K. D. H. Bhadeshia Course A, Metals and Alloys Lecture 1: Atomic Diffusion Mass transport in a gas or liquid generally involves the flow of fluid (e.g. convection
Interfacial Defects. Grain Size Determination
 Interfacial Defects 4.27 For an FCC single crystal, would you expect the surface energy for a (00) plane to be greater or less than that for a () plane? Why? (Note: You may want to consult the solution
Interfacial Defects 4.27 For an FCC single crystal, would you expect the surface energy for a (00) plane to be greater or less than that for a () plane? Why? (Note: You may want to consult the solution
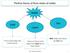
water adding dye partial mixing homogenization time
 iffusion iffusion is a process of mass transport that involves the movement of one atomic species into another. It occurs by ranom atomic jumps from one position to another an takes place in the gaseous,
iffusion iffusion is a process of mass transport that involves the movement of one atomic species into another. It occurs by ranom atomic jumps from one position to another an takes place in the gaseous,
HW #3. Concentration of nitrogen within the surface (at the location in question), C B = 2 kg/m3
 HW #3 Problem 5.7 a. To Find: The distance from the high-pressure side at which the concentration of nitrogen in steel equals 2.0 kg/m 3 under conditions of steady-state diffusion. b. Given: D = 6 10-11
HW #3 Problem 5.7 a. To Find: The distance from the high-pressure side at which the concentration of nitrogen in steel equals 2.0 kg/m 3 under conditions of steady-state diffusion. b. Given: D = 6 10-11
Module 16. Diffusion in solids II. Lecture 16. Diffusion in solids II
 Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Module 16 Diffusion in solids II Lecture 16 Diffusion in solids II 1 NPTEL Phase II : IIT Kharagpur : Prof. R. N. Ghosh, Dept of Metallurgical and Materials Engineering Keywords: Micro mechanisms of diffusion,
Steady-state diffusion is diffusion in which the concentration of the diffusing atoms at
 Chapter 7 What is steady state diffusion? Steady-state diffusion is diffusion in which the concentration of the diffusing atoms at any point, x, and hence the concentration gradient at x, in the solid,
Chapter 7 What is steady state diffusion? Steady-state diffusion is diffusion in which the concentration of the diffusing atoms at any point, x, and hence the concentration gradient at x, in the solid,
CHAPTER 4 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS
 4-1 CHAPTER 4 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS Vacancies and Self-Interstitials 4.1 In order to compute the fraction of atom sites that are vacant in copper at 1357 K, we must employ Equation
4-1 CHAPTER 4 IMPERFECTIONS IN SOLIDS PROBLEM SOLUTIONS Vacancies and Self-Interstitials 4.1 In order to compute the fraction of atom sites that are vacant in copper at 1357 K, we must employ Equation
Dopant Diffusion Sources
 Dopant Diffusion (1) Predeposition dopant gas dose control SiO Si SiO Doped Si region () Drive-in Turn off dopant gas or seal surface with oide profile control (junction depth; concentration) SiO SiO Si
Dopant Diffusion (1) Predeposition dopant gas dose control SiO Si SiO Doped Si region () Drive-in Turn off dopant gas or seal surface with oide profile control (junction depth; concentration) SiO SiO Si
Chapter 5: Diffusion (2)
 Chapter 5: Diffusin () ISSUES TO ADDRESS... Nn-steady state diffusin and Fick s nd Law Hw des diffusin depend n structure? Chapter 5-1 Class Eercise (1) Put a sugar cube inside a cup f pure water, rughly
Chapter 5: Diffusin () ISSUES TO ADDRESS... Nn-steady state diffusin and Fick s nd Law Hw des diffusin depend n structure? Chapter 5-1 Class Eercise (1) Put a sugar cube inside a cup f pure water, rughly
Solids (cont.) Describe the movement of particles in a solid and the forces between them.
 Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Solids A solid is matter that has a definite shape and a definite volume. The attractive forces between the particles in a solid are strong and pull them close together. Solids (cont.) Describe the movement
Most substances can be in three states: solid, liquid, and gas.
 States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
States of Matter Most substances can be in three states: solid, liquid, and gas. Solid Particles Have Fixed Positions The particles in a solid are very close together and have an orderly, fixed arrangement.
Diffusion. Diffusion = the spontaneous intermingling of the particles of two or more substances as a result of random thermal motion
 Diffusion Diffusion = the spontaneous intermingling of the particles of two or more substances as a result of random thermal motion Fick s First Law Γ ΔN AΔt Γ = flux ΔN = number of particles crossing
Diffusion Diffusion = the spontaneous intermingling of the particles of two or more substances as a result of random thermal motion Fick s First Law Γ ΔN AΔt Γ = flux ΔN = number of particles crossing
Chapter 14. Liquids and Solids
 Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
EMA5001 Lecture 2 Interstitial Diffusion & Fick s 1 st Law. Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University
 EMA500 Lecture Interstitial Diffusion & Fick s st Law Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University Substitutional Diffusion Different possibilities Exchange Mechanis
EMA500 Lecture Interstitial Diffusion & Fick s st Law Prof. Zhe Cheng Mechanical & Materials Engineering Florida International University Substitutional Diffusion Different possibilities Exchange Mechanis
Chemistry 101 Chapter 14 Liquids & Solids
 Chemistry 101 Chapter 14 Liquids & Solids States of matter: the physical state of matter depends on a balance between the kinetic energy of particles, which tends to keep them apart, and the attractive
Chemistry 101 Chapter 14 Liquids & Solids States of matter: the physical state of matter depends on a balance between the kinetic energy of particles, which tends to keep them apart, and the attractive
Driving force for diffusion and Fick s laws of diffusion
 Driving force for diffusion and Fick s laws of diffusion In the last lecture, it is eplained that diffusion of elements between different blocks is possible because of difference in chemical potential
Driving force for diffusion and Fick s laws of diffusion In the last lecture, it is eplained that diffusion of elements between different blocks is possible because of difference in chemical potential
Properties of Liquids and Solids
 Properties of Liquids and Solids World of Chemistry Chapter 14 14.1 Intermolecular Forces Most substances made of small molecules are gases at normal temperature and pressure. ex: oxygen gas, O 2 ; nitrogen
Properties of Liquids and Solids World of Chemistry Chapter 14 14.1 Intermolecular Forces Most substances made of small molecules are gases at normal temperature and pressure. ex: oxygen gas, O 2 ; nitrogen
Matter and Thermal Energy
 Section States of Matter Can you identify the states of matter present in the photo shown? Kinetic Theory The kinetic theory is an explanation of how particles in matter behave. Kinetic Theory The three
Section States of Matter Can you identify the states of matter present in the photo shown? Kinetic Theory The kinetic theory is an explanation of how particles in matter behave. Kinetic Theory The three
States of Matter 1 of 21 Boardworks Ltd 2016
 States of Matter 1 of 21 Boardworks Ltd 2016 States of Matter 2 of 21 Boardworks Ltd 2016 What are the three states of matter? 3 of 21 Boardworks Ltd 2016 At any given temperature, all substances exist
States of Matter 1 of 21 Boardworks Ltd 2016 States of Matter 2 of 21 Boardworks Ltd 2016 What are the three states of matter? 3 of 21 Boardworks Ltd 2016 At any given temperature, all substances exist
N = N A Pb A Pb. = ln N Q v kt. = kt ln v N
 5. Calculate the energy for vacancy formation in silver, given that the equilibrium number of vacancies at 800 C (1073 K) is 3.6 10 3 m 3. The atomic weight and density (at 800 C) for silver are, respectively,
5. Calculate the energy for vacancy formation in silver, given that the equilibrium number of vacancies at 800 C (1073 K) is 3.6 10 3 m 3. The atomic weight and density (at 800 C) for silver are, respectively,
Dopant Diffusion. (1) Predeposition dopant gas. (2) Drive-in Turn off dopant gas. dose control. Doped Si region
 Dopant Diffusion (1) Predeposition dopant gas dose control SiO Si SiO Doped Si region () Drive-in Turn off dopant gas or seal surface with oxide profile control (junction depth; concentration) SiO SiO
Dopant Diffusion (1) Predeposition dopant gas dose control SiO Si SiO Doped Si region () Drive-in Turn off dopant gas or seal surface with oxide profile control (junction depth; concentration) SiO SiO
Materials Engineering 272-C Fall 2001, Lecture 7 & 8 Fundamentals of Diffusion
 Materials Engineering 272-C Fall 2001, Lecture 7 & 8 Fundamentals f Diffusin Diffusin: Transprt in a slid, liquid, r gas driven by a cncentratin gradient (r, in the case f mass transprt, a chemical ptential
Materials Engineering 272-C Fall 2001, Lecture 7 & 8 Fundamentals f Diffusin Diffusin: Transprt in a slid, liquid, r gas driven by a cncentratin gradient (r, in the case f mass transprt, a chemical ptential
Appendix 4. Appendix 4A Heat Capacity of Ideal Gases
 Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
Outline. Tensile-Test Specimen and Machine. Stress-Strain Curve. Review of Mechanical Properties. Mechanical Behaviour
 Tensile-Test Specimen and Machine Review of Mechanical Properties Outline Tensile test True stress - true strain (flow curve) mechanical properties: - Resilience - Ductility - Toughness - Hardness A standard
Tensile-Test Specimen and Machine Review of Mechanical Properties Outline Tensile test True stress - true strain (flow curve) mechanical properties: - Resilience - Ductility - Toughness - Hardness A standard
CHEMISTRY Matter and Change. Chapter 12: States of Matter
 CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
CHEMISTRY Matter and Change Chapter 12: States of Matter CHAPTER 12 States of Matter Section 12.1 Section 12.2 Section 12.3 Section 12.4 Gases Forces of Attraction Liquids and Solids Phase Changes Click
Liquids and Solids Chapter 10
 Liquids and Solids Chapter 10 Nov 15 9:56 AM Types of Solids Crystalline solids: Solids with highly regular arrangement of their components Amorphous solids: Solids with considerable disorder in their
Liquids and Solids Chapter 10 Nov 15 9:56 AM Types of Solids Crystalline solids: Solids with highly regular arrangement of their components Amorphous solids: Solids with considerable disorder in their
Unit 4 Lesson 3 Mountain Building. Copyright Houghton Mifflin Harcourt Publishing Company
 Stressed Out How can tectonic plate motion cause deformation? The movement of tectonic plates causes stress on rock structures. Stress is the amount of force per unit area that is placed on an object.
Stressed Out How can tectonic plate motion cause deformation? The movement of tectonic plates causes stress on rock structures. Stress is the amount of force per unit area that is placed on an object.
Chapter 13 Lecture Lecture Presentation. Chapter 13. Chemical Kinetics. Sherril Soman Grand Valley State University Pearson Education, Inc.
 Chapter 13 Lecture Lecture Presentation Chapter 13 Chemical Kinetics Sherril Soman Grand Valley State University Ectotherms Lizards, and other cold-blooded creatures, are ectotherms animals whose body
Chapter 13 Lecture Lecture Presentation Chapter 13 Chemical Kinetics Sherril Soman Grand Valley State University Ectotherms Lizards, and other cold-blooded creatures, are ectotherms animals whose body
Chemical Kinetics and Equilibrium
 Chemical Kinetics and Equilibrium Part 1: Kinetics David A. Katz Department of Chemistry Pima Community College Tucson, AZ USA Chemical Kinetics The study of the rates of chemical reactions and how they
Chemical Kinetics and Equilibrium Part 1: Kinetics David A. Katz Department of Chemistry Pima Community College Tucson, AZ USA Chemical Kinetics The study of the rates of chemical reactions and how they
Chapter 11. Liquids, Solids, and Intermolecular Forces. Water, No Gravity. Lecture Presentation
 Lecture Presentation Chapter 11 Liquids, Solids, and Intermolecular Forces Water, No Gravity In the space station there are no spills. Rather, the water molecules stick together to form a floating, oscillating
Lecture Presentation Chapter 11 Liquids, Solids, and Intermolecular Forces Water, No Gravity In the space station there are no spills. Rather, the water molecules stick together to form a floating, oscillating
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline
 Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Stable Isotopes OUTLINE
 Stable Isotopes OUTLINE Reading: White Ch 9.1 to 9.7.1 (or digital p370-400) Exercise answer? What does the salt do? Today 1. 2 leftovers 2. Stable Isotopes for hydrologic and climate applications 1 CaCO
Stable Isotopes OUTLINE Reading: White Ch 9.1 to 9.7.1 (or digital p370-400) Exercise answer? What does the salt do? Today 1. 2 leftovers 2. Stable Isotopes for hydrologic and climate applications 1 CaCO
Section 4.5 Graphs of Logarithmic Functions
 6 Chapter 4 Section 4. Graphs of Logarithmic Functions Recall that the eponential function f ( ) would produce this table of values -3 - -1 0 1 3 f() 1/8 ¼ ½ 1 4 8 Since the arithmic function is an inverse
6 Chapter 4 Section 4. Graphs of Logarithmic Functions Recall that the eponential function f ( ) would produce this table of values -3 - -1 0 1 3 f() 1/8 ¼ ½ 1 4 8 Since the arithmic function is an inverse
LECTURE 1: Disordered solids: Structure properties
 LECTURE 1: Disordered solids: Structure properties What is an disordered solid: examples amorphous solids or glasses inorganic compounds (e.g. SiO 2 /silicates, B 2 O 3 /borates, GeO 2 /germanates, P 2
LECTURE 1: Disordered solids: Structure properties What is an disordered solid: examples amorphous solids or glasses inorganic compounds (e.g. SiO 2 /silicates, B 2 O 3 /borates, GeO 2 /germanates, P 2
compared to gases. They are incompressible. Their density doesn t change with temperature. These similarities are due
 Liquids and solids They are similar compared to gases. They are incompressible. Their density doesn t change with temperature. These similarities are due to the molecules being close together in solids
Liquids and solids They are similar compared to gases. They are incompressible. Their density doesn t change with temperature. These similarities are due to the molecules being close together in solids
Unit 4 Lesson 7 Mountain Building
 Indiana Standards 7.2.4 Explain how convection currents in the mantle cause lithospheric plates to move causing fast changes like earthquakes and volcanic eruptions, and slow changes like creation of mountains
Indiana Standards 7.2.4 Explain how convection currents in the mantle cause lithospheric plates to move causing fast changes like earthquakes and volcanic eruptions, and slow changes like creation of mountains
Properties of Error Function erf(z) And Complementary Error Function erfc(z)
 Properties of Error Function erf(z) And Complementary Error Function erfc(z) z erf (z) π e -y dy erfc (z) 1 - erf (z) erf () erf( ) 1 erf(- ) - 1 erf (z) d erf(z) dz π z for z
Properties of Error Function erf(z) And Complementary Error Function erfc(z) z erf (z) π e -y dy erfc (z) 1 - erf (z) erf () erf( ) 1 erf(- ) - 1 erf (z) d erf(z) dz π z for z
Matter and Change. Introductory Concepts for Physical Science or Chemistry
 Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
Matter and Change Introductory Concepts for Physical Science or Chemistry Properties of Matter Matter Anything that has mass and occupies space VOLUME WEIGHT RELIABILITY How does one describe the quantities
Chapter 19. Chemical Thermodynamics
 Chapter 19. Chemical Thermodynamics 19.1 Spontaneous Processes Chemical thermodynamics is concerned with energy relationships in chemical reactions. We consider enthalpy and we also consider entropy in
Chapter 19. Chemical Thermodynamics 19.1 Spontaneous Processes Chemical thermodynamics is concerned with energy relationships in chemical reactions. We consider enthalpy and we also consider entropy in
Macroscopic characterization with distribution functions. Physical Chemistry. Particles in a Box. Two equilibrium distributions
 Macroscopic characterization with distribution functions Physical Chemistry Lecture istributions and ransport Processes A macroscopic system contains a LARE number of particles, not all of which h the
Macroscopic characterization with distribution functions Physical Chemistry Lecture istributions and ransport Processes A macroscopic system contains a LARE number of particles, not all of which h the
Chapter 10. Lesson Starter. Why did you not smell the odor of the vapor immediately? Explain this event in terms of the motion of molecules.
 Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
Preview Lesson Starter Objectives The Kinetic-Molecular Theory of Gases The Kinetic-Molecular Theory and the Nature of Gases Deviations of Real Gases from Ideal Behavior Section 1 The Kinetic-Molecular
SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES
 30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
30 SOLIDS AND LIQUIDS - Here's a brief review of the atomic picture or gases, liquids, and solids GASES * Gas molecules are small compared to the space between them. * Gas molecules move in straight lines
The lower the energy of a substance, the interaction between its atoms and molecules.
 PHYSICAL STATES OF MATTER Kinetic Molecular Theory To understand the different states in which matter can exist, we need to understand something called the Kinetic Molecular Theory of Matter. Kinetic Molecular
PHYSICAL STATES OF MATTER Kinetic Molecular Theory To understand the different states in which matter can exist, we need to understand something called the Kinetic Molecular Theory of Matter. Kinetic Molecular
MSE 360 Exam 1 Spring Points Total
 MSE 360 Exam 1 Spring 011 105 Points otal Name(print) ID number No notes, books, or information stored in calculator memories may be used. he NCSU academic integrity policies apply to this exam. As such,
MSE 360 Exam 1 Spring 011 105 Points otal Name(print) ID number No notes, books, or information stored in calculator memories may be used. he NCSU academic integrity policies apply to this exam. As such,
CHAPTER 2 INTERATOMIC FORCES. atoms together in a solid?
 CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
collisions of electrons. In semiconductor, in certain temperature ranges the conductivity increases rapidly by increasing temperature
 1.9. Temperature Dependence of Semiconductor Conductivity Such dependence is one most important in semiconductor. In metals, Conductivity decreases by increasing temperature due to greater frequency of
1.9. Temperature Dependence of Semiconductor Conductivity Such dependence is one most important in semiconductor. In metals, Conductivity decreases by increasing temperature due to greater frequency of
Part I.
 Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
Part I bblee@unimp . Introduction to Mass Transfer and Diffusion 2. Molecular Diffusion in Gasses 3. Molecular Diffusion in Liquids Part I 4. Molecular Diffusion in Biological Solutions and Gels 5. Molecular
CHAPTER 13. States of Matter. Kinetic = motion. Polar vs. Nonpolar. Gases. Hon Chem 13.notebook
 CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
CHAPTER 13 States of Matter States that the tiny particles in all forms of matter are in constant motion. Kinetic = motion A gas is composed of particles, usually molecules or atoms, with negligible volume
Chapter 13. Properties of Solutions. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Chapter 10: Liquids and Solids
 Chapter 10: Liquids and Solids STATES OF MATTER: At any temperature above absolute zero, the atoms, molecules, or ions that make up a substance are moving. In the kinetic theory of gases, this motion is
Chapter 10: Liquids and Solids STATES OF MATTER: At any temperature above absolute zero, the atoms, molecules, or ions that make up a substance are moving. In the kinetic theory of gases, this motion is
Atoms, electrons and Solids
 Atoms, electrons and Solids Shell model of an atom negative electron orbiting a positive nucleus QM tells that to minimize total energy the electrons fill up shells. Each orbit in a shell has a specific
Atoms, electrons and Solids Shell model of an atom negative electron orbiting a positive nucleus QM tells that to minimize total energy the electrons fill up shells. Each orbit in a shell has a specific
They are similar to each other. Intermolecular forces
 s and solids They are similar to each other Different than gases. They are incompressible. Their density doesn t change much with temperature. These similarities are due to the molecules staying close
s and solids They are similar to each other Different than gases. They are incompressible. Their density doesn t change much with temperature. These similarities are due to the molecules staying close
EE 143 Microfabrication Technology Fall 2014
 EE 143 Microfabrication Technology Fall 2014 Prof. Clark T.-C. Nguyen Dept. of Electrical Engineering & Computer Sciences University of California at Berkeley Berkeley, CA 94720 EE 143: Microfabrication
EE 143 Microfabrication Technology Fall 2014 Prof. Clark T.-C. Nguyen Dept. of Electrical Engineering & Computer Sciences University of California at Berkeley Berkeley, CA 94720 EE 143: Microfabrication
Matter. Gas. Solid Liquid. Both shape and volume are not fixed. It has a fixed shape and a fixed volume.
 Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Matter Solid Liquid Gas It has a fixed shape and a fixed volume. It does not have a fixed shape, but it does have a fixed volume. Both shape and volume are not fixed. Felix Yung and John Polias 1 Matter
Week 3, Lectures 6-8, Jan 29 Feb 2, 2001
 Week 3, Lectures 6-8, Jan 29 Feb 2, 2001 EECS 105 Microelectronics Devices and Circuits, Spring 2001 Andrew R. Neureuther Topics: M: Charge density, electric field, and potential; W: Capacitance of pn
Week 3, Lectures 6-8, Jan 29 Feb 2, 2001 EECS 105 Microelectronics Devices and Circuits, Spring 2001 Andrew R. Neureuther Topics: M: Charge density, electric field, and potential; W: Capacitance of pn
Uniform properties throughout! SOLUTE(S) - component(s) of a solution present in small amounts.
 37 SOLUTIONS - a SOLUTION is a HOMOGENEOUS MIXTURE. Uniform properties throughout! - parts of a solution: SOLUTE(S) - component(s) of a solution present in small amounts. SOLVENT - the component of a solution
37 SOLUTIONS - a SOLUTION is a HOMOGENEOUS MIXTURE. Uniform properties throughout! - parts of a solution: SOLUTE(S) - component(s) of a solution present in small amounts. SOLVENT - the component of a solution
Matter, Atoms & Molecules
 Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Matter, Atoms & Molecules Matter is anything that has mass and takes up space. All matter is made of tiny particles called atoms, which are too small to see with the naked eye. Matter Matter is anything
Chapter 11. Freedom of Motion. Comparisons of the States of Matter. Liquids, Solids, and Intermolecular Forces
 Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
Liquids & Solids. For the condensed states the ave KE is less than the attraction between molecules so they are held together.
 Liquids & Solids Intermolecular Forces Matter exists in 3 states. The state of matter is influenced by the physical properties of a substance. For liquids & solids, the condensed states, many of the physical
Liquids & Solids Intermolecular Forces Matter exists in 3 states. The state of matter is influenced by the physical properties of a substance. For liquids & solids, the condensed states, many of the physical
CHEMICAL KINETICS (RATES OF REACTION)
 Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
Kinetics F322 1 CHEMICAL KINETICS (RATES OF REACTION) Introduction Chemical kinetics is concerned with the dynamics of chemical reactions such as the way reactions take place and the rate (speed) of the
Liquids & Solids. Mr. Hollister Holliday Legacy High School Regular & Honors Chemistry
 Liquids & Solids Mr. Hollister Holliday Legacy High School Regular & Honors Chemistry 1 Liquids 2 Properties of the States of Matter: Liquids High densities compared to gases. Fluid. The material exhibits
Liquids & Solids Mr. Hollister Holliday Legacy High School Regular & Honors Chemistry 1 Liquids 2 Properties of the States of Matter: Liquids High densities compared to gases. Fluid. The material exhibits
Diffusion in Solid State
 (Diffusion in Solid State): - Diffusion in Solid State Diffusion Mechanism It is known that at high temperature, the Amplitude of vibrations of atoms leads to expansion, while keeping the (Frequency of
(Diffusion in Solid State): - Diffusion in Solid State Diffusion Mechanism It is known that at high temperature, the Amplitude of vibrations of atoms leads to expansion, while keeping the (Frequency of
Chapter 12 Intermolecular Forces and Liquids
 Chapter 12 Intermolecular Forces and Liquids Jeffrey Mack California State University, Sacramento Why? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature
Chapter 12 Intermolecular Forces and Liquids Jeffrey Mack California State University, Sacramento Why? Why is water usually a liquid and not a gas? Why does liquid water boil at such a high temperature
P5 Heat and Particles Revision Kinetic Model of Matter: States of matter
 P5 Heat and Particles Revision Kinetic Model of Matter: States of matter State Size Shape Solid occupies a fixed volume has a fixed shape Liquid occupies a fixed volume takes the shape of its container
P5 Heat and Particles Revision Kinetic Model of Matter: States of matter State Size Shape Solid occupies a fixed volume has a fixed shape Liquid occupies a fixed volume takes the shape of its container
- As for the liquids, the properties of different solids often differ considerably. Compare a sample of candle wax to a sample of quartz.
 32 SOLIDS * Molecules are usually packed closer together in the solid phase than in the gas or liquid phases. * Molecules are not free to move around each other as in the liquid phase. Molecular/atomic
32 SOLIDS * Molecules are usually packed closer together in the solid phase than in the gas or liquid phases. * Molecules are not free to move around each other as in the liquid phase. Molecular/atomic
Unit 8 Kinetic Theory of Gases. Chapter 13-14
 Unit 8 Kinetic Theory of Gases Chapter 13-14 This tutorial is designed to help students understand scientific measurements. Objectives for this unit appear on the next slide. Each objective is linked to
Unit 8 Kinetic Theory of Gases Chapter 13-14 This tutorial is designed to help students understand scientific measurements. Objectives for this unit appear on the next slide. Each objective is linked to
Chemistry A: States of Matter Packet Name: Hour: Page 1. Chemistry A States of Matter Packet
 Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
Chemistry A: States of Matter Packet Name: Hour: Page 1 Chemistry A States of Matter Packet Chemistry A: States of Matter Packet Name: Hour: Page 2 Worksheet #1: States of Matter In this packet we will
3.091 Introduction to Solid State Chemistry. Lecture Notes No. 9a BONDING AND SOLUTIONS
 3.091 Introduction to Solid State Chemistry Lecture Notes No. 9a BONDING AND SOLUTIONS 1. INTRODUCTION Condensed phases, whether liquid or solid, may form solutions. Everyone is familiar with liquid solutions.
3.091 Introduction to Solid State Chemistry Lecture Notes No. 9a BONDING AND SOLUTIONS 1. INTRODUCTION Condensed phases, whether liquid or solid, may form solutions. Everyone is familiar with liquid solutions.
Materials at Equilibrium. G. Ceder Fall 2001 COURSE 3.20: THERMODYNAMICS OF MATERIALS. FINAL EXAM, Dec 18, 2001
 NAME: COURSE 3.20: THERMODYNAMICS OF MATERIALS FINAL EXAM, Dec 18, 2001 PROBLEM 1 (15 POINTS) PROBLEM 2 (15 POINTS) PROBLEM 3 (10 POINTS) PROBLEM 4 (20 POINTS) PROBLEM 5 (14 POINTS) PROBLEM 6 (14 POINTS)
NAME: COURSE 3.20: THERMODYNAMICS OF MATERIALS FINAL EXAM, Dec 18, 2001 PROBLEM 1 (15 POINTS) PROBLEM 2 (15 POINTS) PROBLEM 3 (10 POINTS) PROBLEM 4 (20 POINTS) PROBLEM 5 (14 POINTS) PROBLEM 6 (14 POINTS)
- A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged.
 POLARITY and shape: - A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged. POLARITY influences several easily observable properties. -
POLARITY and shape: - A polar molecule has an uneven distribution of electron density, making it have ends (poles) that are slightly charged. POLARITY influences several easily observable properties. -
Water SECTION The properties of water in all phases are determined by its structure.
 SECTION 10.5 Water Water commonly exists in all three physical states on Earth, where it is by far the most abundant liquid. It covers nearly three-quarters of Earth s surface. Water is an essential component
SECTION 10.5 Water Water commonly exists in all three physical states on Earth, where it is by far the most abundant liquid. It covers nearly three-quarters of Earth s surface. Water is an essential component
Ionic Bonds. H He: ... Li Be B C :N :O :F: :Ne:
 Ionic Bonds Valence electrons - the electrons in the highest occupied energy level - always electrons in the s and p orbitals - maximum of 8 valence electrons - elements in the same group have the same
Ionic Bonds Valence electrons - the electrons in the highest occupied energy level - always electrons in the s and p orbitals - maximum of 8 valence electrons - elements in the same group have the same
How do Elements Combine to Form Compounds?
 How do Elements Combine to Form Compounds? ACTIVITY What is it made of? Compounds account for the huge variety of matter on Earth All the compounds that exist on Earth are built from elements 118 elements
How do Elements Combine to Form Compounds? ACTIVITY What is it made of? Compounds account for the huge variety of matter on Earth All the compounds that exist on Earth are built from elements 118 elements
Phase Change DIagram
 States of Matter Phase Change DIagram Phase Change Temperature remains during a phase change. Water phase changes Phase Diagram What is a phase diagram? (phase diagram for water) Normal melting point:
States of Matter Phase Change DIagram Phase Change Temperature remains during a phase change. Water phase changes Phase Diagram What is a phase diagram? (phase diagram for water) Normal melting point:
Chapter 10. Liquids and Solids
 Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Chapter 7.1. States of Matter
 Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter 7.1 States of Matter In this chapter... we will learn about matter and different states of matter, many of which we are already familiar with! Learning about Kinetic Molecular Theory will help
Chapter-5 CHEMICAL EQUILIBRIUM
 hapter-5 HEMIAL EQUILIBRIUM On the basis of etent of reactions, chemical reactions can be classified in two categories reversible and irreversible reactions. Irreversible reactions goes for completion
hapter-5 HEMIAL EQUILIBRIUM On the basis of etent of reactions, chemical reactions can be classified in two categories reversible and irreversible reactions. Irreversible reactions goes for completion
Lecture Presentation. Chapter 11. Liquids and Intermolecular Forces. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 11 Liquids and Intermolecular Forces John D. Bookstaver St. Charles Community College Cottleville, MO Properties of Gases, Liquids, and Solids State Volume Shape of State Density
Lecture Presentation Chapter 11 Liquids and Intermolecular Forces John D. Bookstaver St. Charles Community College Cottleville, MO Properties of Gases, Liquids, and Solids State Volume Shape of State Density
CHM Solids, Liquids, and Phase Changes (r15) Charles Taylor 1/9
 CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
CHM 111 - Solids, Liquids, and Phase Changes (r15) - 2015 Charles Taylor 1/9 Introduction In CHM 110, we used kinetic theory to explain the behavior of gases. Now, we will discuss solids and liquids. While
States of Matter. We can explain the properties that we observe in the various states of matter with these postulates.
 States of Matter Kinetic Molecular Theory When discussing the properties of matter, it is not enough just to classify them. We must also create a model that helps to explain the properties that we see.
States of Matter Kinetic Molecular Theory When discussing the properties of matter, it is not enough just to classify them. We must also create a model that helps to explain the properties that we see.
Atoms and Ions Junior Science
 2018 Version Atoms and Ions Junior Science 1 http://msutoday.msu.edu Introduction Chemistry is the study of matter and energy and the interaction between them. The elements are the building blocks of all
2018 Version Atoms and Ions Junior Science 1 http://msutoday.msu.edu Introduction Chemistry is the study of matter and energy and the interaction between them. The elements are the building blocks of all
1 CHAPTER 4 THERMAL CONDUCTION
 1 CHAPTER 4 THERMAL CONDUCTION 4. The Error Function Before we start this chapter, let s just make sure that we are familiar with the error function erf a. We may need it during this chapter. 1 Here is
1 CHAPTER 4 THERMAL CONDUCTION 4. The Error Function Before we start this chapter, let s just make sure that we are familiar with the error function erf a. We may need it during this chapter. 1 Here is
Semiconductor Physics and Devices Chapter 3.
 Introduction to the Quantum Theory of Solids We applied quantum mechanics and Schrödinger s equation to determine the behavior of electrons in a potential. Important findings Semiconductor Physics and
Introduction to the Quantum Theory of Solids We applied quantum mechanics and Schrödinger s equation to determine the behavior of electrons in a potential. Important findings Semiconductor Physics and
Chapter 10: Liquids, Solids, and Phase Changes
 Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Chapter 17: Spontaneity, Entropy, and Free Energy
 Chapter 17: Spontaneity, Entropy, and Free Energy Review of Chemical Thermodynamics System: the matter of interest Surroundings: everything in the universe which is not part of the system Closed System:
Chapter 17: Spontaneity, Entropy, and Free Energy Review of Chemical Thermodynamics System: the matter of interest Surroundings: everything in the universe which is not part of the system Closed System:
Theoretical Models for Chemical Kinetics
 Theoretical Models for Chemical Kinetics Thus far we have calculated rate laws, rate constants, reaction orders, etc. based on observations of macroscopic properties, but what is happening at the molecular
Theoretical Models for Chemical Kinetics Thus far we have calculated rate laws, rate constants, reaction orders, etc. based on observations of macroscopic properties, but what is happening at the molecular
Electro - Principles I
 Electro - Principles I Page 10-1 Atomic Theory It is necessary to know what goes on at the atomic level of a semiconductor so the characteristics of the semiconductor can be understood. In many cases a
Electro - Principles I Page 10-1 Atomic Theory It is necessary to know what goes on at the atomic level of a semiconductor so the characteristics of the semiconductor can be understood. In many cases a
relatively narrow range of temperature and pressure.
 1) Of solids, liquids, and gases, the least common state of matter is the liquid state. a) Liquids can exist only within a relatively narrow range of temperature and pressure. 2) The kinetic-molecular
1) Of solids, liquids, and gases, the least common state of matter is the liquid state. a) Liquids can exist only within a relatively narrow range of temperature and pressure. 2) The kinetic-molecular
They are similar to each other
 They are similar to each other Different than gases. They are incompressible. Their density doesn t change much with temperature. These similarities are due to the molecules staying close together in solids
They are similar to each other Different than gases. They are incompressible. Their density doesn t change much with temperature. These similarities are due to the molecules staying close together in solids
Steady One-Dimensional Diffusion of One Species A through a Second Non-Transferring Species B. z y x. Liquid A
 Steady One-Dimensional Diffusion of One Species through a Second on-transferring Species B R. Shankar Subramanian Department of Chemical and Biomolecular Engineering Clarkson University Consider a simple
Steady One-Dimensional Diffusion of One Species through a Second on-transferring Species B R. Shankar Subramanian Department of Chemical and Biomolecular Engineering Clarkson University Consider a simple
SOLIDS, LIQUIDS AND GASES
 CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
CHEMIS TRY CONTENTS 17 SOLIDS, LIQUIDS AND GASES 17 Solids, Liquids and Gases 147 18 Solutions and Crystallisation 155 19 Separating Mixtures 162 20 Elements, Compounds and Mixtures 171 All materials exist
Liquids are collections of particles that are held together but they can flow (intermolecular forces)
 HW R&MN 4.5 P 1-5,10 Q 1,4,7 Ch. 4.6 P 1,2,4,6,7 Q 3,9 Chapter 4.6: Solids Bonding and Properties A solid is a collection of molecules, ions or atoms that are unable to flow (move around). Due to electromagnetic
HW R&MN 4.5 P 1-5,10 Q 1,4,7 Ch. 4.6 P 1,2,4,6,7 Q 3,9 Chapter 4.6: Solids Bonding and Properties A solid is a collection of molecules, ions or atoms that are unable to flow (move around). Due to electromagnetic
Unit 4: The Nature of Matter
 16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
16 16 Table of Contents Unit 4: The Nature of Matter Chapter 16: Solids, Liquids, and Gases 16.1: Kinetic Theory 16.2: Properties and Fluids 16.3: Behavior of Gases 16.1 Kinetic Theory Kinetic Theory kinetic
Chapter 11 SOLIDS, LIQUIDS AND GASES Pearson Education, Inc.
 Chapter 11 SOLIDS, LIQUIDS AND GASES States of Matter Because in the solid and liquid states particles are closer together, we refer to them as. The States of Matter The state of matter a substance is
Chapter 11 SOLIDS, LIQUIDS AND GASES States of Matter Because in the solid and liquid states particles are closer together, we refer to them as. The States of Matter The state of matter a substance is
Some notes on sigma and pi bonds:
 Some notes on sigma and pi bonds: SIGMA bonds are formed when orbitals overlap along the axis between two atoms. These bonds have good overlap between the bonding orbitals, meaning that they are strong.
Some notes on sigma and pi bonds: SIGMA bonds are formed when orbitals overlap along the axis between two atoms. These bonds have good overlap between the bonding orbitals, meaning that they are strong.
Lecture Presentation. Chapter 12. Solutions. Sherril Soman, Grand Valley State University Pearson Education, Inc.
 Lecture Presentation Chapter 12 Solutions Sherril Soman, Grand Valley State University Thirsty Seawater Drinking seawater can cause dehydration. Seawater Is a homogeneous mixture of salts with water Contains
Lecture Presentation Chapter 12 Solutions Sherril Soman, Grand Valley State University Thirsty Seawater Drinking seawater can cause dehydration. Seawater Is a homogeneous mixture of salts with water Contains
If a sodium atom gives an electron to a chlorine atom, both become more stable.
 3. STRUCTURE AND BONDING IONIC (ELECTROVALENT) BONDING Noble gases like neon or argon have eight electrons in their outer shells (or two in the case of helium). These noble gas structures are thought of
3. STRUCTURE AND BONDING IONIC (ELECTROVALENT) BONDING Noble gases like neon or argon have eight electrons in their outer shells (or two in the case of helium). These noble gas structures are thought of
Mat E 272 Lecture 25: Electrical properties of materials
 Mat E 272 Lecture 25: Electrical properties of materials December 6, 2001 Introduction: Calcium and copper are both metals; Ca has a valence of +2 (2 electrons per atom) while Cu has a valence of +1 (1
Mat E 272 Lecture 25: Electrical properties of materials December 6, 2001 Introduction: Calcium and copper are both metals; Ca has a valence of +2 (2 electrons per atom) while Cu has a valence of +1 (1
