Positive Regulation of Phytochrome B on Chlorophyll Biosynthesis and Chloroplast Development in Rice
|
|
|
- Erica Newman
- 6 years ago
- Views:
Transcription
1 Rice Science, 2013, 20(4): Copyright 2013, China National Rice Research Institute Published by Elsevier BV. All rights reserved DOI: /S (13)60133-X Positive Regulation of Phytochrome B on Chlorophyll Biosynthesis and Chloroplast Development in Rice ZHAO Jie 1, 2, ZHOU Jin-jun 1, WANG Ying-ying 1, GU Jian-wei 1, XIE Xian-zhi 1, 2 ( 1 Shandong Rice Research Institute, Jinan , China; 2 College of Life Sciences, Shandong Normal University, Jinan , China) Abstract: Phytochromes in rice are encoded by a gene family composed of three members, PHYA, PHYB, and PHYC. Through characterizing the phytochrome mutants and wild type (WT) in terms of photomorphogenesis, roles of individual phytochromes have been preliminarily explored in regulating rice de-etiolation, flowering time and fertility. However, little information has been reported about whether or how phytochromes affect chlorophyll biosynthesis and chloroplast development in rice. In this study, we compared the chlorophyll contents of wild type and the phya, phyb and phyaphyb mutants grown under either white light (WL) or red light (R). The results suggest that phyb perceives R to positively regulate chlorophyll biosynthesis, while the role of phya can be detected only in the phyb-deficient mutant. Analyses of the expression levels of genes involved in chlorophyll biosynthesis revealed that phytochromes affected the chlorophyll biosynthesis by regulating protochlorophyll oxidoreductase A (PORA) expression. The role of phyb in chloroplast development was also analyzed, and the results suggest that phyb perceives R to regulate chloroplast development by affecting the numbers of chloroplasts and grana, as well as the chloroplast membrane system. Key words: rice; phytochrome; chlorophyll biosynthesis; protochlorophyll oxidoreductase A; chloroplast development For higher plants, light is not only an energy source for photosynthesis, but also a key environmental factor that influences plant growth and development. Plants use an array of light receptors including phytochrome (phy), cryptochrome and phototropin, to sense external light conditions such as the duration, wavelength and intensity of incident light, and to make adjustments to adapt the changing environment (Suetsugu and Wada, 2007; Bae and Choi, 2008; Franklin and Quail, 2010). Among them, the phy family mainly perceives red light (R) and far-red light (FR), and participates in regulating many important processes in the plant life cycle (Franklin and Quail, 2010; Gu et al, 2011). Plant phytochromes are soluble chromoproteins existing in two photoconversible forms in the cells, inactive R-absorbing form (Pr) and active FR-absorbing (Pfr). Following conversion to the Pfr form, phytochromes translocate to the nucleus. In the nucleus, phytochromes interact with other proteins and trigger a wealth of gene expression to regulate the photomorphogenesis (Takano et al, 2005, 2009; Liu et al, 2006; Franklin and Quail, Received: 25 January 2013; Accepted: 28 March 2013 Corresponding author: XIE Xian-zhi (xzhxie2010@163.com) 2010; Chen and Chory, 2011). De-etiolation is one of the important photomorphogenic characteristics in plants. Dark-grown rice seedlings display a skotomorphogenic phenotype, which is characterized by the absence of chlorophyll. Light signals induce chlorophyll biosynthesis and plant greening. In Arabidopsis, phytochromes are involved in the regulation of chlorophyll biosynthesis as demonstrated by the observation that phyb mutant had lower chlorophyll content under white or red light compared to wild type (Reed et al, 1993). By comparing photomorphogenic characteristics between phyb and phyaphyb mutants in Arabidopsis, phya and cryptochromes were also shown to play roles in regulating the chlorophyll biosynthesis (Reed et al, 1994; Casal and Mazella, 1998; McCormac and Terry, 2002). Chlorophyll biosynthesis is a complicated process that involves many enzymes (Wu et al, 2008; Shi et al, 2009). Analysis of the complete genome of Arabidopsis thaliana showed that there were at least 27 genes encoding 15 enzymes involved in chlorophyll biosynthesis from glutamyl-trna to chlorophyll b (Wu et al, 2007). In recent years, extensive studies have revealed that phytochromes play important roles in regulating chlorophyll biosynthesis. McCormac et al (2001)
2 244 observed that phya, phyb, cry1 and cry2 regulated the expression of glutamyl-trna reductase gene (HEMA1), thus affecting chlorophyll biosynthesis. In Arabidopsis, phytochrome-interacting factor 1 (PIF1) directly or indirectly regulates the expression of the key genes controlling the chlorophyll biosynthetic pathway, including protochlorophyllide oxidoreductase (POR), ferrochelatase (FeChII) and heme oxygenase (HO3) (Moon et al, 2008). Except for chlorophyll biosynthesis, PIF1 and PIF3 were proposed to be negative regulators that integrate light and circadian control in the regulation of chloroplast development (Stephenson et al, 2009). Rice phytochrome gene family is composed of three members known as PHYA, PHYB and PHYC (Kay et al, 1989; Dehesh et al, 1991; Basu et al, 2000). It has been preliminary elucidated that individual phytochromes display both unique and overlapping roles in rice photomorphogenesis by characterization of all rice phytochrome mutants (Takano et al, 2005, 2009; Xie et al, 2007, 2011; Liu et al, 2012). Although leaf blade exhibits pale green in the phyb mutant, we still have no experimental evidence about whether and how phytochromes influence the content of chlorophyll in rice. Moreover, the effect of rice phytochromes on chloroplast development has not been reported. In this study, we examined chlorophyll content and analyzed the expression of genes involved in the chlorophyll biosynthetic pathway in the wild type (WT) and the phya, phyb and phyaphyb mutants. In addition, we also analyzed the influence of R on the development of chloroplast as well as the role of phyb in R signaling. It is verified that phyb-mediated R signal plays an important role in both chlorophyll biosynthesis and chloroplast membrane development. Rice materials MATERIALS AND METHODS The genetic background of phya, phyb and phyaphyb mutants and WT used in this study is Oryza sativa L., cv Nipponbare. The phya and phyb mutants are phya4 and phyb1, respectively (Takano et al, 2001, 2005). Methods Determination of chlorophyll content After being dehusked and surface-sterilized, seeds of WT and phytochrome mutants were grown in 0.4% agar and incubated overnight at 4 C. Then the seeds were transferred to white light (WL; 28 C, lx) Rice Science, Vol. 20, No. 4, 2013 or R [28 C, 15 mol/(m 2 s)] and grown for 7 d. The above-ground parts were used for measuring the chlorophyll content based on the methods described by Zhang (1985) with a slight modification. The fresh sample (0.2 g) was cut into pieces in a motor with a small amount of quartz sand and calcium carbonate powder, and then ground well in 10 ml 95% ethanol. After filtered into a brown volumetric flask, the extracts were brought to a total volume of 25 ml with 95% ethanol. Absorbance values were measured at the wavelengths of 665 nm and 649 nm, recorded as A 665 and A 649, respectively. The concentrations (mg/l) of chlorophyll a (C a ) and chlorophyll b (C b ) were calculated as follows: C a = A A 649 ; C b = A A 665. The content of chlorophyll (mg/g) = (C a + C b ) the volume of extracts / sample fresh weight. Gene expression analysis Seedlings of WT and phytochrome mutants were grown under R at 28 C for 7 d, the above-ground parts were harvested for gene expression analysis. The fluence intensity of R was 15 mol/(m 2 s). Total RNA was isolated from above-ground parts using the RNAiso reagent (TaKaRa, Dalian, China). First-strand cdnas were synthesized from DNase I- treated total RNA using PrimeScript RT Enzyme Mix I (TaKaRa), according to the manufacturer s instructions. Real-time PCR was performed on the ABI 7900HT Real-time PCR System (Applied Biosystems) using SYBR Premix Ex Taq (TaKaRa). Each reaction contained 10 L of 2 SYBR Premix Ex Taq TM, 2.0 L of cdna samples, and 0.2 L gene-specific primer pairs (Table 1) in a final volume of 20 L. As an internal control, the rice ubiquitin gene OsUBQ2 (accession number AK059011) was used to quantify the relative transcript level of each target gene. Three replicate biological experiments were performed. Transmission electron microscopy analysis of chloroplast ultrastructure Seeds of the WT and phytochrome mutants were surface-sterilized and incubated at 28 C for 3 d to induce seed germination. The most uniformly germinated seeds were sown in a 96-well plate with the bottom removed. The plate was floated in Yoshida culture solution in a growth chamber (13 h light of lx, 25 C/11 h dark, 23 C) to the 3-leaf stage of rice seedlings. Then, a set of seedlings was transferred to the continuous R [28 C, 15 mol/(m 2 s)], while the other set was transferred to the continuous WL (28 C,
3 ZHAO Jie, et al. Phytochrome B Positively Regulates Chlorophyll Biosynthesis and Chloroplast Development in Rice 245 Table 1. Real time RT-PCR primers used in the study. Gene name Accession number Gene product Primer pairs (5-3 ) GluTR AK Glutamyl trna reductase CCACACGCCATCTGTTTGAG TTCCCAAGCCTCCACTGTTT GSA AK Glutamate-1-semialdehyde aminotransferase TGATGGAGCCTGGAACCTAC GGTGAAGAAGAACCCGAACA CPO AK Coproporphyrinogen III oxidase GCATAAACCCAAAGGAATGG AGGCTACAGAGTGCGAGGAA PPOX-1 AK Protoporphyrinogen oxidase GCCACAAGCCATACCACAGTT CGAGGAACAATCCATCATAACC CS AK Chlorophyll synthetase CAGGTGGTCTTTCAGTTCCAA GATTGCTGCTTTCATCAGTGG PORA AK NADPH-protochlorophyllide oxidoreductase GTTCTTGGGCGTTCGTCTC CAAACAGAGCATCAGCACAGA OsUBQ2 AK Ubiquitin AACCAGCTGAGGCCCAAGA ACGATTGATTTAACCAGTCCATGA lx). When plants were cultured to the 4-leaf stage, the leaf section of 0.5 cm 0.5 cm were cut from the middle of the 4th leaf and quickly fixed in 2.5% glutaraldehyde and further fixed in 1% OsO 4, then the chloroplast ultrastructure was observed. RESULTS phyb-mediated red signal positively regulates chlorophyll biosynthesis Although several reports revealed that phytochromes affected chlorophyll biosynthesis in Arabidopsis, little information about that has been reported in rice. We compared chlorophyll contents of the WT, phya, phyb and phyaphyb mutants grown in either WL or R. The chlorophyll contents exhibited similar tendency in the materials grown under both light conditions. The chlorophyll content in the phya mutant was basically the same as that in WT, whereas the chlorophyll content was decreased in the phyb mutant compared to that in WT. The decrease was more obvious in the phyaphyb double mutants (Fig. 1). These results suggest that phyb plays an important role in chlorophyll biosynthesis. Although the deficiency of phya has little effect on the chlorophyll content, phya makes a big contribution to chlorophyll accumulation under the phyb-deficiency background. As shown in Fig. 1, the chlorophyll contents were obviously lower in all materials grown under R relative to those under WL, suggesting that lights with wavelengths other than R also regulate the metabolism of chlorophyll. The chlorophyll content in phyb mutant grown in R was significantly lower than that of the WT (Fig. 1), suggesting that phyb-mediated R signaling plays an important role in the chlorophyll biosynthesis. The chlorophyll content in the phyaphyb double mutant grown under R was almost under the measurable limit, which supports the conclusion by Takano et al (2009) that phytochromes are the sole photoreceptors for perceiving red/far-red light in rice. We further analyzed the expression of important genes involved in the chlorophyll biosynthetic pathway in the WT and phytochrome mutants grown under R (Table 1). The transcript levels of GSA and PPOX-1 were higher in the phya mutant, but lower in the phyb mutant, compared to those in the WT (Fig. 2). The transcript levels of CPO were lower in the phya and phyb mutants than in the WT. CS gene was similarly expressed in the WT, phya and phyb mutants. However, the transcript levels of GSA, CPO, PPOX-1 and CS were significantly decreased in the phyaphyb double mutant compared to those in the WT, phya and phyb mutants (Fig. 2). These results suggest that phya and phyb perceive R to positively regulate the expression of these genes in a highly redundant way. Fig. 1. Chlorophyll content in seedlings of wild type (WT), phya, phyb and phyaphyb mutants grown under continuous white light or red light. Seedlings of WT and phytochrome mutants were grown under continuous white light (7 900 lx) or continuous red light [15 mol/(m 2 s)] for 7 d. The above-ground parts were used to measure chlorophyll content. The error bars represent the standard error.
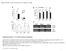
4 246 Rice Science, Vol. 20, No. 4, 2013 Effects of phyb-mediated light signals on chloroplast development Fig. 2. Expression levels of genes involved in chlorophyll biosynthesis in wild type (WT) and phytochrome mutants. Seedlings of WT and phytochrome mutants were grown under continuous red light [15 mol/(m 2 s)] for 7 d. The above-ground parts were used to analyze the gene expression. The error bars represent the standard error. The expression of PORA was slightly higher in the phya mutant, but lower in the phyb mutant relative to that in the WT (Fig. 2). By comparison, PORA mrna was greatly reduced in the phyaphyb double mutant (Fig. 2). These results suggest that phyb-mediated R signaling plays an important role in regulating PORA gene expression; phya also influences the PORA gene expression in the phyb-deficiency mutant. Strangely, the transcript levels of GluTR gene were higher in the phya and phyb mutants than in the WT, and were significantly decreased in the phyaphyb double mutant (Fig. 2). Taken together, the expression pattern of PORA gene under R is consistent with the chlorophyll content in the study, suggesting that phyb affects the chlorophyll biosynthesis probably via regulating PORA gene expression. Since phyb-mediated red signal regulates chlorophyll biosynthesis, it is interesting to analyze whether phyb-mediated red light signal also has a role in chloroplast development. To this end, we compared the development and ultrastructure of chloroplast in the WT and phyb mutant grown under either WL or R. The chloroplast numbers per mesophyll cell in the WT and phyb mutant grown under R were less than those under WL (Fig. 3-A). There were no significant differences in the chloroplast number per mesophyll cell in the WT and the phyb mutant grown under WL, whereas the chloroplast number per mesophyll cell in the phyb mutant was significantly reduced relative to that of the WT grown under R (Fig. 3-A). When plants were grown under WL, the number of grana per chloroplast was essentially the same in the WT as in the phyb mutant. By comparison, the number of grana per chloroplast in the phyb mutant was significantly less than that in the WT when grown under R (Fig. 3-B). These results suggest that phyb-mediated R signaling has an important role in chloroplast development and the formation of grana in rice. In addition, WT and phyb mutant have fewer number of chloroplasts and grana when grown under R than under WL (Fig. 3-A and -B). Thus, lights at wavelengths other than R also affect the development of chloroplasts and grana. Analysis of chloroplast ultrastructure revealed the obvious difference in the WT and the phyb mutant (Fig. 3-C). In plants grown under WL, the membrane Fig. 3. Comparison of chloroplast development in wild type (WT) and phyb mutant grown under either white light (WL) or red light (R). A, The number of chloroplasts per mesophyll cell in the leaves of the WT and phyb mutant under either continuous WL or continuous R. The means ± SE were obtained from, at least, 40 mesophyll cells plotted. B, The number of grana per chloroplast in the leaves of the WT and phyb mutant grown under either continuous WL or continuous R. The means ± SE were obtained from, at least, 30 chloroplast cells plotted. C, Ultrastructure of chloroplasts in the leaves of the WT and phyb mutant under either continuous WL or continuous R. * and ** denote significant differences at P 0.05 and P 0.01, respectively, according to t-test.
5 ZHAO Jie, et al. Phytochrome B Positively Regulates Chlorophyll Biosynthesis and Chloroplast Development in Rice 247 structure was relatively complete, and the granal stack obviously appeared (Fig. 3-C). Under R, however, the membrane structures in the chloroplast of the WT and the phyb mutant were not as complete as under WL (Fig. 3-C). Gaps appeared between grana lamellae in the wild-type chloroplasts, whereas membrane structures seemed seriously damaged in the phyb mutants grown under R, as indicated by less granal stack and obscure outer membrane of chloroplasts (Fig. 3-C). These results indicate that the phyb-mediated red signaling is necessary for the development of chloroplast membrane. DISCUSSION Our results showed that the chlorophyll content in the phyb mutant was less than half of that in the WT under R (Fig. 1), suggesting that phyb perceives R to positively regulate the chlorophyll biosynthesis in rice. The chlorophyll content in the phyaphyb double mutant grown under R was at the limit of measurement in the study (Fig. 1), indicating that phya is also involved in the chlorophyll biosynthesis. However, the chlorophyll content of the phya single mutant was the same as that of the WT (Fig. 1). From these results, we deduce that phya and phyb have highly redundant roles in the chlorophyll biosynthesis, and phya makes a big contribution to the chlorophyll biosynthesis under the phyb-deficient background. Such feature of phya functions has also been observed in diverse R- induced photomorphogenic responses in rice (Takano et al, 2005). Under continuous R, phyb mutants exhibited longer coleoptiles than the WT, while phya and WT had similar length of coleoptiles. However, the phyaphyb mutants grown under continuous R had coleoptiles as long as that of the etiolated seedlings (Takano et al, 2005). Under long-day conditions, the phya mutants flowered at the same time as the WT, and the flowering of the phyb was about 12 d earlier than the WT, whereas the phyaphyb double mutant flowered dramatically earlier than the phyb single mutant (Takano et al, 2005). The functional redundancy of phya and phyb was also observed in Arabidopsis, such as R-regulated inhibition of hypocotyl growth and expansion of cotyledons (Reed et al, 1994; Neff and Chory, 1998; Franklin and Quail, 2010). These results indicate that phya and phyb regulate R responses in a highly redundant way. In this study, phyb-mediated regulation of R on PORA gene expression was likely to be one of key factors controlling the chlorophyll content (Fig. 2). Similarly, phytochrome-mediated light signal induced the PORA gene expression in barley (Batsuschauer and Apel, 1984; Mösinger et al, 1985). Moreover, the activity of the PORA enzyme also requires the presence of light (Mapleston and Griffiths, 1980; Heyes and Hunter, 2005). In Arabidopsis, phya or phyb regulates chlorophyll biosynthetic pathway by interacting with PIF1 factor (Huq et al, 2004; Moon et al, 2008). To our knowledge, however, little information about light signal transduction pathway in rice has been reported, therefore, the mechanism by which phyb regulates the expression of PORA gene remains unclear. Under R, the phyb mutant had incomplete membrane and less granal stack (Fig. 3-C), which is similar to the phenotype of Arabidopsis pif1pif3 transferred from darkness to light (Stephenson et al, 2009). The chlorophyll biosynthesis happens in the thylakoid membrane (Rüdiger et al, 1980). Thus it is speculated that incomplete membrane structure in the chloroplasts of phyb mutant grown under R probably affects the chlorophyll biosynthesis. Moreover, it has been reported that transcript levels of Lhcb (light-harvesting chlorophyll a/b binding protein) gene in the phyb mutants were significantly lower than those in the WT seedlings grown under R (Takano et al, 2005), suggestive of developmental deficiency of chloroplasts in R-grown phyb mutants. In R-grown WT, the membrane structure of chloroplasts is well developed (Fig. 3-C), suggesting that phya (or phyc) perceives R to regulate chloroplast development. Under WL, there is not much difference in ultrastructures of chloroplasts in the WT and the phyb mutant, indicating the involvement of lights with wavelengths other than R in the chloroplast development. ACKNOWLEDGEMENTS This work was supported by the grants from the National Natural Science Foundations of China (Grant Nos and ), the National Major Science and Technology Project to Create New Crop Varieties Using Gene Transfer Technology, China (Grant No. 2009ZX B), and the Shandong Natural Science Funds for Distinguished Young Scholar, China (Grant No. JQ200911). REFERENCES Bae G, Choi G Decoding of light signals by plant phytochromes and their interacting proteins. Annu Rev Plant Biol, 59: Basu D, Dehesh K, Schneider-Poetsch H J, Harrington S E,
6 248 Rice Science, Vol. 20, No. 4, 2013 McCouch S R, Quail P H Rice PHYC gene: Structure, expression, map position and evolution. Plant Mol Biol, 44: Batsuschauer A, Apel K An inverse control by phytochrome of the expression of two nuclear genes in barley (Hordeum vulgare L.). Eur J Biochem, 143: Casal J J, Mazella M A Conditional synergism between cryptochrome 1 and phytochrome B is shown by the analysis of phya, phyb, and hy4 simple, double, and triple mutants in Arabidopsis. Plant Physiol, 118(1): Chen M, Chory J Phytochrome signaling mechanisms and the control of plant development. Trends Cell Biol, 21(11): Dehesh K, Tepperman J, Christensen A H, Quail P H phyb is evolutionarily conserved and constitutively expressed in rice seedling shoots. Mol Gen Genet, 225: Franklin K A, Quail P H Phytochrome functions in Arabidopsis development. J Exp Bot, 61: Gu J W, Liu J, Xue Y J, Zang X, Xie X Z Functions of phytochrome in rice growth and development. Rice Sci, 18(3): Heyes D J, Hunter C N Making light work of enzyme catalysis: Protochlorophyllide oxidoreductase. Trends Biochem Sci, 30: Huq E, Al-Sady B, Hudson M, Kim C, Apel K, Quail P H Phytochrome-interacting factor 1 is a critical bhlh regulator of chlorophyll biosynthesis. Science, 305: Kay S A, Keith B, Shinozaki K, Chua N H The sequence of the rice phytochrome gene. Nucl Acids Res, 17: Liu H W, Zhou R Y, Xu L Participation of phytochrome in sterility alteration of photoperiod-sensitive female sterile ramie. Acta Agron Sin, 32: (in Chinese with English abstract) Liu J, Zhang F, Zhou J J, Chen F, Wang B S, Xie X Z Phytochrome B control of total leaf area and stomatal density affects drought tolerance in rice. Plant Mol Biol, 78: Mapleston E R, Griffiths W T Light modulation of the activity of the protochlorophyllide oxidoreductase. Biochem J, 189: McCormac A C, Fischer A, Kumar A M, Söll D, Terry M J Regulation of HEMA1 expression by phytochrome and a plastid signal during de-etiolation in Arabidopsis thaliana. Plant J, 25: McCormac A C, Terry M J Loss of nuclear gene expression during the phytochrome A-mediated far-red block of greening response. Plant Physiol, 130: Moon J, Zhu L, Shen H, Huq E PIF1 directly and indirectly regulates chlorophyll biosynthesis to optimize the greening process in Arabidopsis. Proc Natl Acad Sci USA, 105: Mösinger E, Batschauer A, Schäfer E, Apel K Phytochrome control of in vitro transcription of specific genes in isolated nuclei from barley (Hordeum vulgare). Eur J Biochem, 147: Neff M M, Chory J Genetic interactions between phytochrome A, phytochrome B, and cryptochrome 1 during Arabidopsis development. Plant Physiol, 118(1): Reed J W, Nagpal P, Poole D S, Furuya M, Chory J Mutations in the gene for red/far-red light receptor phytochrome B alter cell elongation and physiological responses throughout Arabidopsis development. Plant Cell, 5: Reed J W, Nagatani K, Elich T D, Fagan M, Chory J Phytochrome A and phytochrome B have overlapping but distinct functions in Arabidopsis development. Plant Physiol, 104: Rüdiger W, Benz J, Guthoff C Detection and partial characterization of activity of chlorophyll synthetase in etioplast membranes. Eur J Biochem, 109: Shi D Y, Liu Z X, Jin W W Biosynthesis, catabolism and related signal regulations of plant chlorophyll. Hereditas, 31(7): (in Chinese with English abstract) Stephenson P G, Christian F, Matthew J T PIF3 is repressor of chloroplast development. Proc Natl Acad Sci USA, 106: Suetsugu N, Wada M Chloroplast photorelocation movement mediated by phototropin family proteins in green plants. Biol Chem, 388: Takano M, Kanegae H, Shinomura T, Miyao A, Hirohiko H, Furuya M Isolation and characterization of rice phytochrome A mutants. Plant Cell, 13: Takano M, Inagaki N, Xie X Z, Yuzurihara N, Hihara F, Ishizuka T, Yano M, Nishimura M, Miyao A, Hirochika H, Shinomura T Distinct and cooperative functions of phytochromes A, B and C in the control of deetiolation and flowering in rice. Plant Cell, 17: Takano M, Inagaki N, Xie X Z, Kiyota S, Baba-Kasai A, Tanabata T, Shinomura T Phytochromes are the sole photoreceptors for perceiving red/far-red light in rice. Proc Natl Acad Sci USA, 106: Wu Z M, Zhang X, He B, Diao L P, Sheng S L, Wang J L, Guo X P, Su N, Wang L F, Jiang L, Wang C M, Zhai H Q, Wan J M A chlorophyll-deficient rice mutant with impaired chlorophyllide esterification in chlorophyll biosynthesis. Plant Physiol, 145(1): Wu Z M, Zhang X, Wan J M Molecular regulation of chlorophyll biosynthesis. Plant Physiol Commun, 44(6): (in Chinese) Xie X Z, Shinomura T, Inagaki N, Kiyota S, Takano M Phytochrome-mediated inhibition of coleoptile growth in rice: Age-dependency and action spectra. Photochem Photobiol, 83: Xie X Z, Xue Y J, Zhou J J, Zhang B, Chang H, Takano M Phytochromes regulate SA and JA signaling pathways in rice and are required for developmentally controlled resistance to Magnaporthe grisea. Mol Plant, 4(4): Zhang Q D Methods to measure chlorophyll. Chin Bull Bot, 3: (in Chinese)
Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor in Rice, and Preliminary Analysis of Its Roles in Light Signal Transduction
 Rice Science, 2012, 19(4): 263 268 Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor
Rice Science, 2012, 19(4): 263 268 Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Expression Patterns of OsPIL11, a Phytochrome-Interacting Factor
Functions of Phytochrome in Rice Growth and Development
 Rice Science, 2011, 18(3): 231 237 Copyright 2011, China National Rice Research Institute Published by Elsevier BV. All rights reserved Functions of Phytochrome in Rice Growth and Development GU Jian-wei
Rice Science, 2011, 18(3): 231 237 Copyright 2011, China National Rice Research Institute Published by Elsevier BV. All rights reserved Functions of Phytochrome in Rice Growth and Development GU Jian-wei
Phytochromes are Involved in Elongation of Seminal Roots and Accumulation of Dry substances in Rice Seedlings
 Rice Science, 2013, 20(1): Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Phytochromes are Involved in Elongation of Seminal Roots and Accumulation
Rice Science, 2013, 20(1): Copyright 2012, China National Rice Research Institute Published by Elsevier BV. All rights reserved Phytochromes are Involved in Elongation of Seminal Roots and Accumulation
Phytochromes are Involved in Elongation of Seminal Roots and Accumulation of Dry Substances in Rice Seedlings
 Rice Science, 2013, 20(2): 88 94 Copyright 2013, China National Rice Research Institute Published by Elsevier BV. All rights reserved DOI: 10.1016/S1672-6308(13)60115-8 Phytochromes are Involved in Elongation
Rice Science, 2013, 20(2): 88 94 Copyright 2013, China National Rice Research Institute Published by Elsevier BV. All rights reserved DOI: 10.1016/S1672-6308(13)60115-8 Phytochromes are Involved in Elongation
Analysis of regulatory function of circadian clock. on photoreceptor gene expression
 Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Thesis of Ph.D. dissertation Analysis of regulatory function of circadian clock on photoreceptor gene expression Tóth Réka Supervisor: Dr. Ferenc Nagy Biological Research Center of the Hungarian Academy
Figure 18.1 Blue-light stimulated phototropism Blue light Inhibits seedling hypocotyl elongation
 Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Blue Light and Photomorphogenesis Q: Figure 18.3 Blue light responses - phototropsim of growing Corn Coleoptile 1. How do we know plants respond to blue light? 2. What are the functions of multiple BL
Electromagenetic spectrum
 Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
Light Controls of Plant Development 1 Electromagenetic spectrum 2 Light It is vital for photosynthesis and is also necessary to direct plant growth and development. It acts as a signal to initiate and
CBMG688R. ADVANCED PLANT DEVELOPMENT AND PHYSIOLOGY II G. Deitzer Spring 2006 LECTURE
 1 CBMG688R. ADVANCED PLANT DEVELOPMENT AND PHYSIOLOGY II G. Deitzer Spring 2006 LECTURE Photomorphogenesis and Light Signaling Photoregulation 1. Light Quantity 2. Light Quality 3. Light Duration 4. Light
1 CBMG688R. ADVANCED PLANT DEVELOPMENT AND PHYSIOLOGY II G. Deitzer Spring 2006 LECTURE Photomorphogenesis and Light Signaling Photoregulation 1. Light Quantity 2. Light Quality 3. Light Duration 4. Light
Plant Growth and Development
 Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
Plant Growth and Development Concept 26.1 Plants Develop in Response to the Environment Factors involved in regulating plant growth and development: 1. Environmental cues (e.g., day length) 2. Receptors
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E
 CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
CONTROL OF PLANT GROWTH AND DEVELOPMENT BI-2232 RIZKITA R E The development of a plant the series of progressive changes that take place throughout its life is regulated in complex ways. Factors take part
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering
 Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
Photoreceptor Regulation of Constans Protein in Photoperiodic Flowering by Valverde et. Al Published in Science 2004 Presented by Boyana Grigorova CBMG 688R Feb. 12, 2007 Circadian Rhythms: The Clock Within
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING. AnitaHajdu. Thesis of the Ph.D.
 THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
THE ROLE OF THE PHYTOCHROME B PHOTORECEPTOR IN THE REGULATION OF PHOTOPERIODIC FLOWERING AnitaHajdu Thesis of the Ph.D. dissertation Supervisor: Dr. LászlóKozma-Bognár - senior research associate Doctoral
Light Regulation of Flowering Time in Arabidopsis
 Chapter 38 Light Regulation of Flowering Time in Arabidopsis Xuhong Yu and Chentao Lin Introduction Plant development is dependent on not only endogenous conditions but also environmental factors. One
Chapter 38 Light Regulation of Flowering Time in Arabidopsis Xuhong Yu and Chentao Lin Introduction Plant development is dependent on not only endogenous conditions but also environmental factors. One
Light perception. phytochromes, cryptochromes, phototropins.
 Light perception phytochromes, cryptochromes, phototropins. all photoreceptors consist of proteins bound to light absorbing pigments i.e. chromophores. the spectral sensitivity of each photoreceptor depends
Light perception phytochromes, cryptochromes, phototropins. all photoreceptors consist of proteins bound to light absorbing pigments i.e. chromophores. the spectral sensitivity of each photoreceptor depends
Life Science Journal 2014;11(9) Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis
 Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Cryptochrome 2 negatively regulates ABA-dependent seed germination in Arabidopsis Sung-Il Kim 1, Sang Ik Song 3, Hak Soo Seo 1, 2, 4 * 1 Department of Plant Science and Research Institute of Agriculture
Synergistic and Antagonistic Action of Phytochrome (Phy) A and PhyB during Seedling De-Etiolation in Arabidopsis thaliana
 Int. J. Mol. Sci. 2015, 16, 12199-12212; doi:10.3390/ijms160612199 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Synergistic and Antagonistic
Int. J. Mol. Sci. 2015, 16, 12199-12212; doi:10.3390/ijms160612199 Article OPEN ACCESS International Journal of Molecular Sciences ISSN 1422-0067 www.mdpi.com/journal/ijms Synergistic and Antagonistic
Flower Development Pathways
 Developmental Leading to Flowering Flower Development s meristem Inflorescence meristem meristems organ identity genes Flower development s to Flowering Multiple pathways ensures flowering will take place
Developmental Leading to Flowering Flower Development s meristem Inflorescence meristem meristems organ identity genes Flower development s to Flowering Multiple pathways ensures flowering will take place
LECTURE 4: PHOTOTROPISM
 http://smtom.lecture.ub.ac.id/ Password: https://syukur16tom.wordpress.com/ LECTURE 4: PHOTOTROPISM LECTURE FLOW 1. 2. 3. 4. 5. INTRODUCTION DEFINITION INITIAL STUDY PHOTROPISM MECHANISM PHOTORECEPTORS
http://smtom.lecture.ub.ac.id/ Password: https://syukur16tom.wordpress.com/ LECTURE 4: PHOTOTROPISM LECTURE FLOW 1. 2. 3. 4. 5. INTRODUCTION DEFINITION INITIAL STUDY PHOTROPISM MECHANISM PHOTORECEPTORS
Light-Independent Phytochrome Signaling Mediated by Dominant GAF Domain Tyrosine Mutants of Arabidopsis Phytochromes in Transgenic Plants W OA
 The Plant Cell, Vol. 19: 2124 2139, July 2007, www.plantcell.org ª 2007 American Society of Plant Biologists Light-Independent Phytochrome Signaling Mediated by Dominant GAF Domain Tyrosine Mutants of
The Plant Cell, Vol. 19: 2124 2139, July 2007, www.plantcell.org ª 2007 American Society of Plant Biologists Light-Independent Phytochrome Signaling Mediated by Dominant GAF Domain Tyrosine Mutants of
Arabidopsis thaliana. Lucia Strader. Assistant Professor, Biology
 Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Arabidopsis thaliana Lucia Strader Assistant Professor, Biology Arabidopsis as a genetic model Easy to grow Small genome Short life cycle Self fertile Produces many progeny Easily transformed HIV E. coli
Nucleo-cytoplasmic partitioning of the plant photoreceptors phytochromes
 seminars in CELL & DEVELOPMENTAL BIOLOGY, Vol. 11, 2000: pp. 505 510 doi: 10.1006/scdb.2000.0202, available online at http://www.idealibrary.com on Nucleo-cytoplasmic partitioning of the plant photoreceptors
seminars in CELL & DEVELOPMENTAL BIOLOGY, Vol. 11, 2000: pp. 505 510 doi: 10.1006/scdb.2000.0202, available online at http://www.idealibrary.com on Nucleo-cytoplasmic partitioning of the plant photoreceptors
Phytochrome-mediated growth inhibition of seminal ro. The definitive version is available at Instructions for use
 Title Phytochrome-mediated growth inhibition of seminal ro Shimizu, Hisayo; Tanabata, Takanari; Xie, Xianzhi; I Author(s) Yamamoto, Kotaro T. CitationPhysiologia Plantarum, 137(3): 289-297 Issue Date 2009-11
Title Phytochrome-mediated growth inhibition of seminal ro Shimizu, Hisayo; Tanabata, Takanari; Xie, Xianzhi; I Author(s) Yamamoto, Kotaro T. CitationPhysiologia Plantarum, 137(3): 289-297 Issue Date 2009-11
Supplementary Materials for
 www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
www.sciencesignaling.org/cgi/content/full/9/452/ra106/dc1 Supplementary Materials for Stem-piped light activates phytochrome B to trigger light responses in Arabidopsis thaliana roots Hyo-Jun Lee, Jun-Ho
The role of the N-terminal NTE domain of PHYTOCHROMEs in red and far red light perception
 The role of the N-terminal NTE domain of PHYTOCHROMEs in red and far red light perception Theses of the Ph.D. dissertation János Bindics Supervisor: Dr. Ferenc Nagy Hungarian Academy of Sciences Biological
The role of the N-terminal NTE domain of PHYTOCHROMEs in red and far red light perception Theses of the Ph.D. dissertation János Bindics Supervisor: Dr. Ferenc Nagy Hungarian Academy of Sciences Biological
The signal transducing photoreceptors of plants
 Int. J. Dev. Biol. 49: 653-664 (2005) doi: 10.1387/ijdb.051989kf The signal transducing photoreceptors of plants KEARA A. FRANKLIN*, VICTORIA S. LARNER and GARRY C. WHITELAM Department of Biology, University
Int. J. Dev. Biol. 49: 653-664 (2005) doi: 10.1387/ijdb.051989kf The signal transducing photoreceptors of plants KEARA A. FRANKLIN*, VICTORIA S. LARNER and GARRY C. WHITELAM Department of Biology, University
Photoperiodic flowering in plants was the first photoperiodism
 Regulation of photoperiodic flowering by Arabidopsis photoreceptors Todd Mockler*, Hongyun Yang, XuHong Yu, Dhavan Parikh, Ying-chia Cheng, Sarah Dolan, and Chentao Lin Department of Molecular, Cell, and
Regulation of photoperiodic flowering by Arabidopsis photoreceptors Todd Mockler*, Hongyun Yang, XuHong Yu, Dhavan Parikh, Ying-chia Cheng, Sarah Dolan, and Chentao Lin Department of Molecular, Cell, and
Ph.D. thesis. Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana
 Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Ph.D. thesis Study of proline accumulation and transcriptional regulation of genes involved in this process in Arabidopsis thaliana Written by: Edit Ábrahám Temesváriné Supervisors: Dr. László Szabados
Cytokinin. Fig Cytokinin needed for growth of shoot apical meristem. F Cytokinin stimulates chloroplast development in the dark
 Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Cytokinin Abundant in young, dividing cells Shoot apical meristem Root apical meristem Synthesized in root tip, developing embryos, young leaves, fruits Transported passively via xylem into shoots from
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated
 Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Figure 1. Identification of UGT74E2 as an IBA glycosyltransferase. (A) Relative conversion rates of different plant hormones to their glucosylated form by recombinant UGT74E2. The naturally occurring auxin
Phytochrome E Influences Internode Elongation and Flowering Time in Arabidopsis
 The Plant Cell, Vol. 10, 1479 1487, September 1998, www.plantcell.org 1998 American Society of Plant Physiologists Phytochrome E Influences Internode Elongation and Flowering Time in Arabidopsis Paul F.
The Plant Cell, Vol. 10, 1479 1487, September 1998, www.plantcell.org 1998 American Society of Plant Physiologists Phytochrome E Influences Internode Elongation and Flowering Time in Arabidopsis Paul F.
OVEREXPRESSION OF RICE PHYTOCHROME A IN ARABIDOPSIS: DIVERSE ROLE IN MULTIPLE PHYSIOLOGICAL RESPONSES
 Pak. J. Bot., 43(6): 2835-2844, 2011. OVEREXPRESSION OF RICE PHYTOCHROME A IN ARABIDOPSIS: DIVERSE ROLE IN MULTIPLE PHYSIOLOGICAL RESPONSES CHUNFENG CHEN 1, 2, YOU CHEN 1, QING ZHANG 1, BENWEN CHEN 3,
Pak. J. Bot., 43(6): 2835-2844, 2011. OVEREXPRESSION OF RICE PHYTOCHROME A IN ARABIDOPSIS: DIVERSE ROLE IN MULTIPLE PHYSIOLOGICAL RESPONSES CHUNFENG CHEN 1, 2, YOU CHEN 1, QING ZHANG 1, BENWEN CHEN 3,
Summary. Introduction
 The Plant Journal (1998) 15(1), 69 77 Combinatorial interaction of light-responsive elements plays a critical role in determining the response characteristics of light-regulated promoters in Arabidopsis
The Plant Journal (1998) 15(1), 69 77 Combinatorial interaction of light-responsive elements plays a critical role in determining the response characteristics of light-regulated promoters in Arabidopsis
Phytochrome Signaling Mechanisms
 Phytochrome Signaling Mechanisms Authors: Jigang Li, Gang Li, Haiyang Wang, and Xing Wang Deng Source: The Arabidopsis Book, 2011(9) Published By: American Society of Plant Biologists URL: https://doi.org/10.1199/tab.0148
Phytochrome Signaling Mechanisms Authors: Jigang Li, Gang Li, Haiyang Wang, and Xing Wang Deng Source: The Arabidopsis Book, 2011(9) Published By: American Society of Plant Biologists URL: https://doi.org/10.1199/tab.0148
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach
 Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Characterisation of abiotic stress inducible plant promoters and bacterial genes for osmotolerance using transgenic approach ABSTRACT SUBMITTED TO JAMIA MILLIA ISLAMIA NEW DELHI IN PARTIAL FULFILMENT OF
Genetics: Published Articles Ahead of Print, published on March 6, 2009 as /genetics
 Genetics: Published Articles Ahead of Print, published on March 6, 2009 as 10.1534/genetics.108.099887 Blue light induces degradation of the negative regulator Phytochrome Interacting Factor 1 to promote
Genetics: Published Articles Ahead of Print, published on March 6, 2009 as 10.1534/genetics.108.099887 Blue light induces degradation of the negative regulator Phytochrome Interacting Factor 1 to promote
Phytochrome A is an irradiance-dependent red light sensor
 The Plant Journal (007) 50, 108 117 doi: 10.1111/j.165-1X.007.006.x Phytochrome A is an irradiance-dependent red light sensor Keara A. Franklin *, Trudie Allen and Garry C. Whitelam Department of Biology,
The Plant Journal (007) 50, 108 117 doi: 10.1111/j.165-1X.007.006.x Phytochrome A is an irradiance-dependent red light sensor Keara A. Franklin *, Trudie Allen and Garry C. Whitelam Department of Biology,
Cryptochromes Are Required for Phytochrome Signaling to the Circadian Clock but Not for Rhythmicity
 The Plant Cell, Vol. 12, 2499 2509, December 2000, www.plantcell.org 2000 American Society of Plant Physiologists Cryptochromes Are Required for Phytochrome Signaling to the Circadian Clock but Not for
The Plant Cell, Vol. 12, 2499 2509, December 2000, www.plantcell.org 2000 American Society of Plant Physiologists Cryptochromes Are Required for Phytochrome Signaling to the Circadian Clock but Not for
Involvement of Rice Cryptochromes in De-etiolation Responses and Flowering
 Plant Cell Physiol. 47(7): 915 925 (2006) doi:10.1093/pcp/pcj064, available online at www.pcp.oxfordjournals.org ß The Author 2006. Published by Oxford University Press on behalf of Japanese Society of
Plant Cell Physiol. 47(7): 915 925 (2006) doi:10.1093/pcp/pcj064, available online at www.pcp.oxfordjournals.org ß The Author 2006. Published by Oxford University Press on behalf of Japanese Society of
PLANTS modulate their growth and development in
 Copyright Ó 29 by the Genetics Society of America DOI: 1.1534/genetics.18.99887 Blue Light Induces Degradation of the Negative Regulator Phytochrome Interacting Factor 1 to Promote Photomorphogenic Development
Copyright Ó 29 by the Genetics Society of America DOI: 1.1534/genetics.18.99887 Blue Light Induces Degradation of the Negative Regulator Phytochrome Interacting Factor 1 to Promote Photomorphogenic Development
Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana
 Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana Jenna K. Bains, Selam Joseph, Shayan Shokoohi, Ian A. Villamin Abstract The presence of light and the presence of a mutation
Effect of light and the cer10 mutation on the growth rate of Arabidopsis thaliana Jenna K. Bains, Selam Joseph, Shayan Shokoohi, Ian A. Villamin Abstract The presence of light and the presence of a mutation
Seeing without eyes-how plants learn from light
 Seeing without eyes-how plants learn from light by STEPHEN DAY 1. INTRODUCTION Plants detect the intensity, direction, colour, and duration of light and use this information to regulate their growth and
Seeing without eyes-how plants learn from light by STEPHEN DAY 1. INTRODUCTION Plants detect the intensity, direction, colour, and duration of light and use this information to regulate their growth and
Photomorphogenesis. Edited by W. Shropshire, Jr. and H. Mohr
 Photomorphogenesis Edited by W. Shropshire, Jr. and H. Mohr Contributors K. Apel M. Black A.E. Canham J.A. De Greef M.J. Dring H. Egneus B. Frankland H. Fredericq L. Fukshansky M. Furuya V. Gaba A.W. Galston
Photomorphogenesis Edited by W. Shropshire, Jr. and H. Mohr Contributors K. Apel M. Black A.E. Canham J.A. De Greef M.J. Dring H. Egneus B. Frankland H. Fredericq L. Fukshansky M. Furuya V. Gaba A.W. Galston
23-. Shoot and root development depend on ratio of IAA/CK
 Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Balance of Hormones regulate growth and development Environmental factors regulate hormone levels light- e.g. phototropism gravity- e.g. gravitropism temperature Mode of action of each hormone 1. Signal
Light Control of Arabidopsis Development Entails Coordinated Regulation of Genome Expression and Cellular Pathways
 The Plant Cell, Vol. 13, 2589 2607, December 2001, www.plantcell.org 2001 American Society of Plant Biologists Light Control of Arabidopsis Development Entails Coordinated Regulation of Genome Expression
The Plant Cell, Vol. 13, 2589 2607, December 2001, www.plantcell.org 2001 American Society of Plant Biologists Light Control of Arabidopsis Development Entails Coordinated Regulation of Genome Expression
Supplemental Data. Chen and Thelen (2010). Plant Cell /tpc
 Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Supplemental Data. Chen and Thelen (2010). Plant Cell 10.1105/tpc.109.071837 1 C Total 5 kg 20 kg 100 kg Transmission Image 100 kg soluble pdtpi-gfp Plastid (PDH-alpha) Mito (PDH-alpha) GFP Image vector
Phytochromes and Shade-avoidance Responses in Plants
 Annals of Botany 96: 169 175, 2005 doi:10.1093/aob/mci165, available online at www.aob.oupjournals.org BOTANICAL BRIEFING Phytochromes and Shade-avoidance Responses in Plants KEARA A. FRANKLIN and GARRY
Annals of Botany 96: 169 175, 2005 doi:10.1093/aob/mci165, available online at www.aob.oupjournals.org BOTANICAL BRIEFING Phytochromes and Shade-avoidance Responses in Plants KEARA A. FRANKLIN and GARRY
Review Article Fluorescence and Photochemical Investigations of Phytochrome in Higher Plants
 Journal of Botany Volume, Article ID 587, 5 pages doi:.55//587 Review Article Fluorescence and Photochemical Investigations of Phytochrome in Higher Plants Vitaly A. Sineshchekov Physico-Chemical Biology,
Journal of Botany Volume, Article ID 587, 5 pages doi:.55//587 Review Article Fluorescence and Photochemical Investigations of Phytochrome in Higher Plants Vitaly A. Sineshchekov Physico-Chemical Biology,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL
 GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
GENETIC ANALYSES OF ROOT SYSTEM DEVELOPMENT IN THE TOMATO CROP MODEL Kelsey Hoth 1 Dr. Maria Ivanchenko 2 Bioresourse Research 1, Department of Botany and Plant Physiology 2, Oregon State University, Corvallis,
Light reaction. Dark reaction
 Photosynthesis Light reaction Dark reaction Electro-magnetic irradiance and sunlight CO 2 and O 2 fixation by Rubisco Oxygenic photosynthesis was established in Cyanobacteria Localisation of the
Photosynthesis Light reaction Dark reaction Electro-magnetic irradiance and sunlight CO 2 and O 2 fixation by Rubisco Oxygenic photosynthesis was established in Cyanobacteria Localisation of the
Major Plant Hormones 1.Auxins 2.Cytokinins 3.Gibberelins 4.Ethylene 5.Abscisic acid
 Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
Plant Hormones Lecture 9: Control Systems in Plants What is a Plant Hormone? Compound produced by one part of an organism that is translocated to other parts where it triggers a response in target cells
POTASSIUM IN PLANT GROWTH AND YIELD. by Ismail Cakmak Sabanci University Istanbul, Turkey
 POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
POTASSIUM IN PLANT GROWTH AND YIELD by Ismail Cakmak Sabanci University Istanbul, Turkey Low K High K High K Low K Low K High K Low K High K Control K Deficiency Cakmak et al., 1994, J. Experimental Bot.
Phytochrome A Regulates the Intracellular Distribution of Phototropin 1 Green Fluorescent Protein in Arabidopsis thaliana W
 The Plant Cell, Vol. 20: 2835 2847, October 2008, www.plantcell.org ã 2008 American Society of Plant Biologists Phytochrome A Regulates the Intracellular Distribution of Phototropin 1 Green Fluorescent
The Plant Cell, Vol. 20: 2835 2847, October 2008, www.plantcell.org ã 2008 American Society of Plant Biologists Phytochrome A Regulates the Intracellular Distribution of Phototropin 1 Green Fluorescent
15. PHOTOPERIODISM. 1. Short day plants
 15. PHOTOPERIODISM Photoperiodism is the phenomenon of physiological changes that occur in plants in response to relative length of day and night (i.e. photoperiod). The response of the plants to the photoperiod,
15. PHOTOPERIODISM Photoperiodism is the phenomenon of physiological changes that occur in plants in response to relative length of day and night (i.e. photoperiod). The response of the plants to the photoperiod,
Arabidopsis SHORT HYPOCOTYL UNDER BLUE1 Contains SPX and EXS Domains and Acts in Cryptochrome Signaling W
 The Plant Cell, Vol. 18, 921 934, April 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Arabidopsis SHORT HYPOCOTYL UNDER BLUE1 Contains SPX and EXS Domains and Acts in Cryptochrome
The Plant Cell, Vol. 18, 921 934, April 2006, www.plantcell.org ª 2006 American Society of Plant Biologists Arabidopsis SHORT HYPOCOTYL UNDER BLUE1 Contains SPX and EXS Domains and Acts in Cryptochrome
SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent Seed Germination Downstream of PIL5 W
 The Plant Cell, Vol. 20: 1260 1277, May 2008, www.plantcell.org ª 2008 American Society of Plant Biologists SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent
The Plant Cell, Vol. 20: 1260 1277, May 2008, www.plantcell.org ª 2008 American Society of Plant Biologists SOMNUS, a CCCH-Type Zinc Finger Protein in Arabidopsis, Negatively Regulates Light-Dependent
Hierarchical coupling of phytochromes and cryptochromes reconciles stability and light modulation of Arabidopsis development
 Development 128, 2291-2299 (21) Printed in Great Britain The Company of Biologists Limited 21 DEV361 2291 Hierarchical coupling of phytochromes and cryptochromes reconciles stability and light modulation
Development 128, 2291-2299 (21) Printed in Great Britain The Company of Biologists Limited 21 DEV361 2291 Hierarchical coupling of phytochromes and cryptochromes reconciles stability and light modulation
Light signals, phytochromes and cross-talk with other environmental cues
 Advance Access published December 12, 2003 Journal of Experimental Botany, Vol. 55, No. 395, Cross-talk in Plant Signal Transduction Special Issue, Page 1 of 6, January 2004 DOI: 10.1093/jxb/erh026 Light
Advance Access published December 12, 2003 Journal of Experimental Botany, Vol. 55, No. 395, Cross-talk in Plant Signal Transduction Special Issue, Page 1 of 6, January 2004 DOI: 10.1093/jxb/erh026 Light
Engineering light response pathways in crop plants for improved performance under high planting density
 Engineering light response pathways in crop plants for improved performance under high planting density Tom Brutnell Boyce Thompson Institute for Plant Research Cornell University, Ithaca NY 6000 years
Engineering light response pathways in crop plants for improved performance under high planting density Tom Brutnell Boyce Thompson Institute for Plant Research Cornell University, Ithaca NY 6000 years
Cryptochromes, Phytochromes, and COP1 Regulate Light-Controlled Stomatal Development in Arabidopsis W
 The Plant Cell, Vol. 21: 2624 2641, September 2009, www.plantcell.org ã 2009 American Society of Plant Biologists Cryptochromes, Phytochromes, and COP1 Regulate Light-Controlled Stomatal Development in
The Plant Cell, Vol. 21: 2624 2641, September 2009, www.plantcell.org ã 2009 American Society of Plant Biologists Cryptochromes, Phytochromes, and COP1 Regulate Light-Controlled Stomatal Development in
Marcelo J. Yanovsky and Steve A. Kay
 LIVING BY THE CALENDAR: HOW PLANTS KNOW WHEN TO FLOWER Marcelo J. Yanovsky and Steve A. Kay Reproductive processes in plants and animals are usually synchronized with favourable seasons of the year. It
LIVING BY THE CALENDAR: HOW PLANTS KNOW WHEN TO FLOWER Marcelo J. Yanovsky and Steve A. Kay Reproductive processes in plants and animals are usually synchronized with favourable seasons of the year. It
From seed germination to flowering, light controls plant development via the pigment phytochrome
 Proc. Natl. Acad. Sci. USA Vol. 93, pp. 12066-12071, October 1996 Symposium Paper This paper was presented at a symposium entitled "Frontiers in Plant Biology: How Plants Communicate" organized by Hans
Proc. Natl. Acad. Sci. USA Vol. 93, pp. 12066-12071, October 1996 Symposium Paper This paper was presented at a symposium entitled "Frontiers in Plant Biology: How Plants Communicate" organized by Hans
Changes in photoperiod or temperature alter the functional relationships between phytochromes and reveal roles for phyd and phye
 Edinburgh Research Explorer Changes in photoperiod or temperature alter the functional relationships between phytochromes and reveal roles for phyd and phye Citation for published version: Halliday, KJ
Edinburgh Research Explorer Changes in photoperiod or temperature alter the functional relationships between phytochromes and reveal roles for phyd and phye Citation for published version: Halliday, KJ
Supplemental Data. Perrella et al. (2013). Plant Cell /tpc
 Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Intensity Intensity Intensity Intensity Intensity Intensity 150 50 150 0 10 20 50 C 150 0 10 20 50 D 0 10 20 Distance (μm) 50 20 40 E 50 F 0 10 20 50 0 15 30 Distance (μm) Supplemental Figure 1: Co-localization
Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport
 Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Ph.D. thesis Arabidopsis PPR40 connects abiotic stress responses to mitochondrial electron transport Zsigmond Laura Supervisor: Dr. Szabados László Arabidopsis Molecular Genetic Group Institute of Plant
Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues
 Research Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues Daniel Kirchenbauer 1 *, Andras Viczian 2 *, Eva Adam 2, Zoltan
Research Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues Daniel Kirchenbauer 1 *, Andras Viczian 2 *, Eva Adam 2, Zoltan
The Circadian Clock Regulates the Photoperiodic Response of Hypocotyl Elongation through a Coincidence Mechanism in Arabidopsis thaliana
 The Circadian Clock Regulates the Photoperiodic Response of Hypocotyl Elongation through a Coincidence Mechanism in Arabidopsis thaliana Yusuke Niwa, Takafumi Yamashino * and Takeshi Mizuno Laboratory
The Circadian Clock Regulates the Photoperiodic Response of Hypocotyl Elongation through a Coincidence Mechanism in Arabidopsis thaliana Yusuke Niwa, Takafumi Yamashino * and Takeshi Mizuno Laboratory
Regulation of Phosphate Homeostasis by microrna in Plants
 Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
Regulation of Phosphate Homeostasis by microrna in Plants Tzyy-Jen Chiou 1 *, Kyaw Aung 1,2, Shu-I Lin 1,3, Chia-Chune Wu 1, Su-Fen Chiang 1, and Chun-Lin Su 1 Abstract Upon phosphate (Pi) starvation,
The shade avoidance syndrome: multiple responses mediated by multiple phytochromes
 Plant, Cell and Environment (1997) 20, 840 844 TECHNICAL REPORT (white this line if not required) The shade avoidance syndrome: multiple responses mediated by multiple phytochromes H. SMITH & G. C. WHITELAM
Plant, Cell and Environment (1997) 20, 840 844 TECHNICAL REPORT (white this line if not required) The shade avoidance syndrome: multiple responses mediated by multiple phytochromes H. SMITH & G. C. WHITELAM
SUMMARY Chlorophyll biosynthesis:
 Summary SUMMARY Plants are severely affected by various abiotic stresses due to their static nature. Abiotic stresses may be in the form of drought, salt, flooding, temperature, light or metal stress.
Summary SUMMARY Plants are severely affected by various abiotic stresses due to their static nature. Abiotic stresses may be in the form of drought, salt, flooding, temperature, light or metal stress.
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA
 NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
NATURAL VARIATION IN THE CYTOKININ METABOLIC NETWORK IN ARABIDOPSIS THALIANA PŘÍRODNÍ VARIACE METABOLISMU CYTOKININŮ U ARABIDOPSIS THALIANA Samsonová Z. 1, 2, 3, Kuklová A. 1, 2, Mazura P. 1, 2, Rotková
Véronique Bergougnoux 1 *, David Zalabák 2, Michaela Jandová 3, Ondřej Novák 1, Anika Wiese- Klinkenberg 4, Martin Fellner 1 * Abstract.
 Effect of Blue Light on Endogenous Isopentenyladenine and Endoreduplication during Photomorphogenesis and De-Etiolation of Tomato (Solanum lycopersicum L.) Seedlings Véronique Bergougnoux 1 *, David Zalabák
Effect of Blue Light on Endogenous Isopentenyladenine and Endoreduplication during Photomorphogenesis and De-Etiolation of Tomato (Solanum lycopersicum L.) Seedlings Véronique Bergougnoux 1 *, David Zalabák
Functional and signaling mechanism analysis of rice CRYPTOCHROME 1
 The Plant Journal (2006) 46, 971 983 doi: 10.1111/j.1365-313X.2006.02753.x Functional and signaling mechanism analysis of rice CRYPTOCHROME 1 Yan-Chun Zhang, Song-Fu Gong, Qing-Hua Li, Yi Sang and Hong-Quan
The Plant Journal (2006) 46, 971 983 doi: 10.1111/j.1365-313X.2006.02753.x Functional and signaling mechanism analysis of rice CRYPTOCHROME 1 Yan-Chun Zhang, Song-Fu Gong, Qing-Hua Li, Yi Sang and Hong-Quan
Citation for the original published paper (version of record): N.B. When citing this work, cite the original published paper.
 http://www.diva-portal.org This is the published version of a paper published in Scientific Reports. Citation for the original published paper (version of record): Dubreuil, C., Ji, Y., Strand, Å., Grönlund,
http://www.diva-portal.org This is the published version of a paper published in Scientific Reports. Citation for the original published paper (version of record): Dubreuil, C., Ji, Y., Strand, Å., Grönlund,
Blue light affects many aspects of plant growth and development.
 Plant blue-light receptors Chentao Lin Plants have several blue-light receptors, which regulate different aspects of growth and development. Recent studies have identified three such receptors: cryptochrome
Plant blue-light receptors Chentao Lin Plants have several blue-light receptors, which regulate different aspects of growth and development. Recent studies have identified three such receptors: cryptochrome
Significance. 1 To whom correspondence should be addressed.
 REDUCED CHLOROPLAST COVERAGE genes from Arabidopsis thaliana help to establish the size of the chloroplast compartment Robert M. Larkin a,b,c,1, Giovanni Stefano b, Michael E. Ruckle b,c,2, Andrea K. Stavoe
REDUCED CHLOROPLAST COVERAGE genes from Arabidopsis thaliana help to establish the size of the chloroplast compartment Robert M. Larkin a,b,c,1, Giovanni Stefano b, Michael E. Ruckle b,c,2, Andrea K. Stavoe
EMBO. Phytochrome-mediated photoperception and signal transduction in higher plants. reports. Eberhard Schäfer & Chris Bowler 1,+ Introduction
 EMBO reports Phytochrome-mediated photoperception and signal transduction in higher plants Eberhard Schäfer & Chris Bowler 1,+ Universitat Freiburg, Institut fur Biologie II/Botanik, Schanzlestrasse 1,
EMBO reports Phytochrome-mediated photoperception and signal transduction in higher plants Eberhard Schäfer & Chris Bowler 1,+ Universitat Freiburg, Institut fur Biologie II/Botanik, Schanzlestrasse 1,
Discovery of compounds that keep plants fresh ~ Controlling plant pore openings for drought tolerance and delay in leaf withering ~
 Discovery of compounds that keep plants fresh ~ Controlling plant pore openings for drought tolerance and delay in leaf withering ~ April 9, 2018 A team of scientists at Nagoya University has discovered
Discovery of compounds that keep plants fresh ~ Controlling plant pore openings for drought tolerance and delay in leaf withering ~ April 9, 2018 A team of scientists at Nagoya University has discovered
Report. Synergism of Red and Blue Light in the Control of Arabidopsis Gene Expression and Development
 Current Biology 19, 1216 1220, July 28, 2009 ª2009 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2009.05.062 Synergism of Red and Blue Light in the Control of Arabidopsis Gene Expression and Development
Current Biology 19, 1216 1220, July 28, 2009 ª2009 Elsevier Ltd All rights reserved DOI 10.1016/j.cub.2009.05.062 Synergism of Red and Blue Light in the Control of Arabidopsis Gene Expression and Development
Matthew Hudson Department of Crop Sciences, University of Illinois, Urbana, Illinois 61801
 Photoreceptor Biotechnology Matthew Hudson Department of Crop Sciences, University of Illinois, Urbana, Illinois 61801 I. Introduction and Background Plant photoreceptors influence or control almost all
Photoreceptor Biotechnology Matthew Hudson Department of Crop Sciences, University of Illinois, Urbana, Illinois 61801 I. Introduction and Background Plant photoreceptors influence or control almost all
Low-Fluence Red Light Increases the Transport and Biosynthesis of Auxin 1[C][W][OA]
![Low-Fluence Red Light Increases the Transport and Biosynthesis of Auxin 1[C][W][OA] Low-Fluence Red Light Increases the Transport and Biosynthesis of Auxin 1[C][W][OA]](/thumbs/72/66726940.jpg) Low-Fluence Red Light Increases the Transport and Biosynthesis of Auxin 1[C][W][OA] Xing Liu*, Jerry D. Cohen, and Gary Gardner Plant Biological Sciences Graduate Program, Department of Horticultural Science
Low-Fluence Red Light Increases the Transport and Biosynthesis of Auxin 1[C][W][OA] Xing Liu*, Jerry D. Cohen, and Gary Gardner Plant Biological Sciences Graduate Program, Department of Horticultural Science
Diurnal Dependence of Growth Responses to Shade in Arabidopsis: Role of Hormone, Clock, and Light Signaling
 RESEARCH ARTICLE Diurnal Dependence of Growth Responses to Shade in Arabidopsis: Role of Hormone, Clock, and Light Signaling Romina Sellaro, Manuel Pacín and Jorge J. Casal 1 IFEVA, Facultad de Agronomía,
RESEARCH ARTICLE Diurnal Dependence of Growth Responses to Shade in Arabidopsis: Role of Hormone, Clock, and Light Signaling Romina Sellaro, Manuel Pacín and Jorge J. Casal 1 IFEVA, Facultad de Agronomía,
Title. Author(s)Kurata, Tetsuya; Yamamoto, Kotaro T. CitationJournal of Plant Physiology, 151(3): Issue Date Doc URL.
 Title Light-stimulated root elongation in Arabidopsis thal Author(s)Kurata, Tetsuya; Yamamoto, Kotaro T. CitationJournal of Plant Physiology, 151(3): 346-351 Issue Date 1997 Doc URL http://hdl.handle.net/2115/44841
Title Light-stimulated root elongation in Arabidopsis thal Author(s)Kurata, Tetsuya; Yamamoto, Kotaro T. CitationJournal of Plant Physiology, 151(3): 346-351 Issue Date 1997 Doc URL http://hdl.handle.net/2115/44841
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids
 Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
Heterosis and inbreeding depression of epigenetic Arabidopsis hybrids Plant growth conditions The soil was a 1:1 v/v mixture of loamy soil and organic compost. Initial soil water content was determined
SYLLABUS, BIO 328D: DISCOVERY LABORATORY IN PLANT BIOLOGY
 SYLLABUS, BIO 328D: DISCOVERY LABORATORY IN PLANT BIOLOGY Instructor: Stan Roux; BIO 16; 471-4238; sroux@uts.cc.utexas.edu; Office hrs: TTh, 10-11:30 AM TA: Ashley Cannon; BIO 15; 471-1074; ashleycannon@utexas.edu;
SYLLABUS, BIO 328D: DISCOVERY LABORATORY IN PLANT BIOLOGY Instructor: Stan Roux; BIO 16; 471-4238; sroux@uts.cc.utexas.edu; Office hrs: TTh, 10-11:30 AM TA: Ashley Cannon; BIO 15; 471-1074; ashleycannon@utexas.edu;
** * * * Col-0 cau1 CAU1. Actin2 CAS. Actin2. Supplemental Figure 1. CAU1 affects calcium accumulation.
 Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Ca 2+ ug g -1 DW Supplemental Data. Fu et al. Plant Cell. (213). 1.115/tpc.113.113886 A 5 4 3 * Col- cau1 B 4 3 2 Col- cau1 ** * * ** C 2 1 25 2 15 1 5 Shoots Roots *
Ulrich Sperling, a Fabrice Franck, b Barbara van Cleve, a Geneviève Frick, a Klaus Apel, a and Gregory A. Armstrong a,1
 The Plant Cell, Vol. 10, 283 296, February 1998 1998 American Society of Plant Physiologists Etioplast Differentiation in Arabidopsis: Both PORA and PORB Restore the Prolamellar Body and Photoactive Protochlorophyllide
The Plant Cell, Vol. 10, 283 296, February 1998 1998 American Society of Plant Physiologists Etioplast Differentiation in Arabidopsis: Both PORA and PORB Restore the Prolamellar Body and Photoactive Protochlorophyllide
M P BHOJ (OPEN) UNIVERSITY, BHOPAL ASSIGNMENT QUESTION PAPER
 PAPER :I Cell and molecular Biology of plants Q.1 Describe various models of Plasma membrane with its significance? OR Discuss role of Plasmodesmata in movements of moleculer and macromolecules? Q.2 Give
PAPER :I Cell and molecular Biology of plants Q.1 Describe various models of Plasma membrane with its significance? OR Discuss role of Plasmodesmata in movements of moleculer and macromolecules? Q.2 Give
Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues
 Edinburgh Research Explorer Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues Citation for published version: Kirchenbauer,
Edinburgh Research Explorer Characterization of photomorphogenic responses and signaling cascades controlled by phytochrome-a expressed in different tissues Citation for published version: Kirchenbauer,
Time measurement and the control of flowering in plants
 Time measurement and the control of flowering in plants Alon Samach and George Coupland* Summary Many plants are adapted to flower at particular times of year, to ensure optimal pollination and seed maturation.
Time measurement and the control of flowering in plants Alon Samach and George Coupland* Summary Many plants are adapted to flower at particular times of year, to ensure optimal pollination and seed maturation.
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function
 Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Utilizing Illumina high-throughput sequencing technology to gain insights into small RNA biogenesis and function Brian D. Gregory Department of Biology Penn Genome Frontiers Institute University of Pennsylvania
Blue Light Receptors and Signal Transduction
 The Plant Cell, S207 S225, Supplement 2002, www.plantcell.org 2002 American Society of Plant Biologists Blue Light Receptors and Signal Transduction Chentao Lin 1 Department of Molecular, Cell and Developmental
The Plant Cell, S207 S225, Supplement 2002, www.plantcell.org 2002 American Society of Plant Biologists Blue Light Receptors and Signal Transduction Chentao Lin 1 Department of Molecular, Cell and Developmental
Antagonistic actions of Arabidopsis cryptochromes and phytochrome B in the regulation of floral induction
 Development 126, 73-82 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV214 73 Antagonistic actions of Arabidopsis cryptochromes and phytochrome B in the regulation of floral induction
Development 126, 73-82 (1999) Printed in Great Britain The Company of Biologists Limited 1999 DEV214 73 Antagonistic actions of Arabidopsis cryptochromes and phytochrome B in the regulation of floral induction
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and
 This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
This article appeared in a journal published by Elsevier. The attached copy is furnished to the author for internal non-commercial research and education use, including for instruction at the authors institution
Reproduction, Seeds and Propagation
 Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Reproduction, Seeds and Propagation Diploid (2n) somatic cell Two diploid (2n) somatic cells Telophase Anaphase Metaphase Prophase I One pair of homologous chromosomes (homologues) II Homologues condense
Red Light-Induced Phytochrome Relocation into the Nucleus in Adiantum capillus-veneris
 RESEARCH ARTICLE Red Light-Induced Phytochrome Relocation into the Nucleus in Adiantum capillus-veneris Hidenori Tsuboi a, Sachihiko Nakamura b, Eberhard Schäfer c and Masamitsu Wada a,b,c,d,1 a Kyushu
RESEARCH ARTICLE Red Light-Induced Phytochrome Relocation into the Nucleus in Adiantum capillus-veneris Hidenori Tsuboi a, Sachihiko Nakamura b, Eberhard Schäfer c and Masamitsu Wada a,b,c,d,1 a Kyushu
Distinct regulation of CAB and PHYB gene expression by similar circadian clocks
 The Plant Journal (2002) 32, 529 537 Distinct regulation of CAB and PHYB gene expression by similar circadian clocks Anthony Hall 1,László Kozma-Bognár 2, Ruth M. Bastow 1,y, Ferenc Nagy 2 and Andrew J.
The Plant Journal (2002) 32, 529 537 Distinct regulation of CAB and PHYB gene expression by similar circadian clocks Anthony Hall 1,László Kozma-Bognár 2, Ruth M. Bastow 1,y, Ferenc Nagy 2 and Andrew J.
Arabidopsis CONSTANS-LIKE3 Is a Positive Regulator of Red Light Signaling and Root Growth W
 The Plant Cell, Vol. 18, 70 84, January 2006, www.plantcell.org ª 2005 American Society of Plant Biologists Arabidopsis CONSTANS-LIKE3 Is a Positive Regulator of Red Light Signaling and Root Growth W Sourav
The Plant Cell, Vol. 18, 70 84, January 2006, www.plantcell.org ª 2005 American Society of Plant Biologists Arabidopsis CONSTANS-LIKE3 Is a Positive Regulator of Red Light Signaling and Root Growth W Sourav
1. (a) Why are the two kinds of self-incompatibiltiy (SI) mechanisms called sporophytic and gametophytic?
 Bio 328 -Spring 2005 NAME: Test #1 Please provide succinct answers in the space provided under each question. Unless otherwise noted in the margin the value of each question is 3 points. 1. (a) Why are
Bio 328 -Spring 2005 NAME: Test #1 Please provide succinct answers in the space provided under each question. Unless otherwise noted in the margin the value of each question is 3 points. 1. (a) Why are
LIGHT SIGNAL TRANSDUCTION IN HIGHER PLANTS
 Annu. Rev. Genet. 2004. 38:87 117 doi: 10.1146/annurev.genet.38.072902.092259 Copyright c 2004 by Annual Reviews. All rights reserved First published online as a Review in Advance on June 11, 2004 LIGHT
Annu. Rev. Genet. 2004. 38:87 117 doi: 10.1146/annurev.genet.38.072902.092259 Copyright c 2004 by Annual Reviews. All rights reserved First published online as a Review in Advance on June 11, 2004 LIGHT
Red-Light-Dependent Interaction of phyb with SPA1 Promotes COP1 SPA1 Dissociation and Photomorphogenic Development in Arabidopsis
 Research Article Red-Light-Dependent Interaction of phyb with SPA1 Promotes COP1 SPA1 Dissociation and Photomorphogenic Development in Arabidopsis Xue-Dan Lu 1, Chuan-Miao Zhou 2, Peng-Bo Xu 3, Qian Luo
Research Article Red-Light-Dependent Interaction of phyb with SPA1 Promotes COP1 SPA1 Dissociation and Photomorphogenic Development in Arabidopsis Xue-Dan Lu 1, Chuan-Miao Zhou 2, Peng-Bo Xu 3, Qian Luo
