Effects of glass content and oxidation on the spectra of SNC-like basalts: Applications to Mars remote sensing
|
|
|
- Primrose Thompson
- 5 years ago
- Views:
Transcription
1 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 107, NO. E5, 5030, /2001JE001518, 2002 Effects of glass content and oxidation on the spectra of SNC-like basalts: Applications to Mars remote sensing Michelle E. Minitti, 1 John F. Mustard, and Malcolm J. Rutherford Department of Geological Sciences, Brown University, Providence, Rhode Island, USA Received 16 May 2001; revised 10 October 2001; accepted 26 October 2001; published 18 May [1] We have investigated the effects of crystallization and oxidation on the visible and nearinfrared (VISNIR) and mid-infrared (mid-ir) spectra of materials with a Shergotty-Nakhla- Chassigny (SNC) basaltic composition in order to investigate potential links between Martian VISNIR spectra, Martian mid-ir spectra, and SNC meteorite data. SNC basalt samples of varying crystallinities were synthesized and subsequently oxidized in air at 700 C for 1 7 days, and reflectance spectra of both fine (75 mm) and coarse ( mm) basalt samples were obtained before and after oxidation. The effects of crystallization on the spectral properties are readily observed in the unoxidized products. In the VISNIR, increasing crystallization is accompanied by an increasing influence of pyroxene absorptions and decreasing influence of glass absorptions. In the mid-ir, changes in spectral properties with crystallization are due to the variations in relative amounts of glass, pyroxene, and plagioclase. Oxidation alters the glass and pyroxenes within the samples, leading to changes in VISNIR and mid-ir spectral shapes, although distinct signatures of oxidation products are not always observed. Both spectral and electron microscopy data indicate that hematite is the dominant oxidation product in the pigeonite and glass of the samples. In pigeonite, hematite is present in nanophase form and leads to the diminishment of the diagnostic 1- and 2-mm pyroxene absorptions in the VISNIR as well as pyroxene absorptions expressed between 800 and 1100 cm 1 in the mid-ir. In glass, hematite forms a near-surface layer that is detected with varying efficiencies in the VISNIR and mid-ir depending on the crystallinity and particle size of the analyzed sample. Oxidized SNC basalt samples of various crystallinities and fine grain sizes provide the best analogs to Mars remote-sensing data in the VISNIR and mid-ir. In the VISNIR, partially crystalline, oxidized SNC basalts can reproduce Martian dark region characteristics. In the mid-ir, similarities exist between the spectra of a partially crystalline, oxidized SNC basalt and the basalt lithology detected by the Thermal Emission Spectrometer (TES). The andesite lithology detected by TES is best matched by the spectrum of a fully crystalline, oxidized SNC basalt. These results imply that plagioclase-rich lithologies are not required by TES data and that observed variations in spectral character of dark regions across the Martian surface can be explained by SNC basalts influenced by oxidation coupled with variations in degree of crystallization. INDEX TERMS: 6225 Planetology: Solar System Objects: Mars; 5464 Planetology: Solid Surface Planets: Remote sensing; 5410 Planetology: Solid Surface Planets: Composition; 5470 Planetology: Solid Surface Planets: Surface materials and properties; KEYWORDS: Mars, hematite, Martian meteorites, basalts, remote sensing, glass, oxidation 1. Introduction [2] Remote-sensing studies, originating with Earth-based telescopic observations [e.g., Adams and McCord, 1969] and eventually including spacecraft [e.g., Sagan et al., 1973] and lander [e.g., Mutch et al., 1977] observations, have provided insight into the composition and distribution of Mars surface materials. Visible and near-infrared (VISNIR) remote-sensing data, sensitive to iron and hydroxyl mineralogy, first quantitatively established the spectral differences between the bright and dark regions on Mars [Singer and McCord, 1979] (Figure 1). Spectra of the bright regions are dominated by the signature of ferric iron, primarily in the form of nanophase hematite [Morris et al., 1989]. The spectra 1 Now at Department of Geological Sciences, Arizona State University, Tempe, Arizona, USA. Copyright 2002 by the American Geophysical Union /02/2001JE001518$09.00 of the dark regions, in addition to exhibiting the effects of ferric iron as seen in the bright regions, have a weak absorption at 1 mm and negative spectral slope beyond 1 mm [Singer and McCord, 1979]. The 1-mm feature is attributed to the presence of pyroxene, which in turn suggests that the observed dark regions consist of basalt [Adams and McCord, 1969]. The interpretation of the dark regions as pyroxene-bearing basalts is supported by the deconvolution of VISNIR data from the ISM on the Phobos II mission [Mustard and Sunshine, 1995]. Deconvolution results indicate that the observed dark regions consist of basalts containing subequal amounts of low- and high-calcium pyroxene, which agrees well with the pyroxene mineralogy of the basaltic shergottite group of the Shergotty-Nakhla-Chassigny (SNC), or Martian, meteorites. The most widely accepted cause of the negative spectral slope is the presence of a thin ferric coating intimately associated with a basaltic substrate whose absorption and scattering efficiencies change with wavelength [Singer and McCord, 1979; Singer, 1980; Mustard et al., 1993]. In addition to contributions from ferric coatings, Erard et al. [1991] demonstrated that contributions to the negative spectral slope arise from atmospheric aerosol 6-1
2 6-2 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA Figure 1. Representative spectra of Mars bright and dark regions in the VISNIR from Singer and McCord [1979]. backscattering and that the influence of the aerosols varies between observed dark regions. [3] The mid-ir spectra from the Thermal Emission Spectrometer (TES) on the Mars Global Surveyor mission, which are sensitive to silicate lattice mode vibrations, offer a different perspective of Martian surface materials. Syntheses of the TES global observations between 6 and 50 mm indicate that two spectral types characterize Mars dark regions (Figure 2). The first spectral type, based on comparison to terrestrial rock spectra, is classified as basaltic [Christensen et al., 2000a]. Deconvolution of the spectrum into component minerals results in a mineralogy of vol% plagioclase, vol% high-calcium pyroxene, and potentially vol% olivine and sheet silicates. The second spectral type, when compared to terrestrial rock spectra, best matches glassy andesites to basaltic andesites [Bandfield et al., 2000]. Spectral deconvolution of the andesite spectrum results in 45 vol% obsidian glass, 40 vol% plagioclase, and 10 vol% pyroxene. Dark regions in the northern hemisphere of Mars are dominated by the andesitic lithology, while dark regions in the southern hemisphere are dominated by the basaltic lithology. [4] Important differences exist between the VISNIR and mid-ir spectral data of Mars. The presence of glass is argued for in both wavelength ranges, but the nature of the proposed glass differs between the mid-ir (volcanic [Bandfield et al., 2000]) and VISNIR data (impact [Schultz and Mustard, 1998]). The signature of oxidation is a dominant component of the VISNIR data but is not apparent in the TES data. According to Christensen et al. [2000a], however, coatings that lead to the VISNIR oxidation signature are consistent with the TES data. Finally, the spectral deconvolution of VISNIR ISM spectra by Mustard and Sunshine [1995] indicates the presence of both low- and high-calcium pyroxene, while the deconvolution of mid-ir TES spectra indicates the presence of only high-calcium pyroxene. [5] While a good agreement in pyroxene mineralogy exists between VISNIR and basaltic SNC meteorite data [e.g., Mustard and Sunshine, 1995], the derived TES and observed basaltic SNC mineralogies are different. All calculated modes for the basaltic TES spectra have plagioclase/pyroxene ratios (vol%) between 1.3 and 2.8 [Christensen et al., 2000a], and the terrestrial rock with the best fit to the Martian basaltic andesite data has a plagioclase/ pyroxene ratio (vol%) of 3.5 [Bandfield et al., 2000]. In contrast, the range of plagioclase/pyroxene ratios (vol%) for basaltic shergottites is [Lodders, 1998], which correlates with the low Al 2 O 3 contents of calculated SNC parent melts [e.g., Hale et al., 1999]. The dissimilar TES and SNC plagioclase/pyroxene ratios are in keeping with lack of similarity between both TES spectral types and the SNC meteorite mid-ir spectra [Hamilton et al., 1997]. [6] The differences between the mid-ir, VISNIR, and SNC data raise the question of whether or not these data sets can be brought into agreement. Surface characteristics derived from VISNIR and TES data suggest two possible sources of the differences: oxidation and the presence of glass. Oxidation was shown to significantly affect the spectral signatures of pyroxenes in the VISNIR by Straub et al. [1991]. If the mid-ir spectral properties of pyroxenes were also changed by oxidation, how would such changes affect the contribution of pyroxenes to the bulk dark region spectra? In turn, could alteration of the pyroxene signal affect the plagioclase/ pyroxene ratio detected by TES? While glass is not a significant component of the SNC meteorites, it is feasible that in some volcanic environments eruptive products with SNC-like composition could contain varying amounts of glass. Is it possible that a wholly or partially glassy SNC composition sample could produce spectral features observed in the VISNIR and mid-ir? Additionally, if oxidation is invoked as a means of linking the mid-ir, VISNIR, and SNC data, the potential effect of oxidized glass must also be considered. Motivated by these questions, the goals of this study are to determine the effects of oxidation and degree of crystallization on the spectra of SNC-like basalts in order to establish if an SNC composition can yield spectra consistent with VISNIR and TES data. 2. Experimental Procedures 2.1. Basalt Crystallization [7] The basalt starting material synthesized for the experiments of this study was the A* composition, a calculated trapped melt Figure 2. Representative spectra of the two spectral lithologies detected by TES: basalt and andesite (J. L. Bandfield, personal communication, 2000).
3 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA 6-3 Table 1. Starting Material Compositions Oxide A* 3, wt.% A* 4, wt.% SiO Al 2 O FeO MgO CaO Na 2 O K 2 O P 2 O TiO MnO Total composition for Chassigny established by Johnson et al. [1991]. The composition of A* is essentially identical to early melt compositions established for other SNC meteorites [Minitti and Rutherford, 2000]; it is low in Al 2 O 3 (8.0 wt%) and high in total FeO (19 wt%) relative to terrestrial basaltic melt compositions. Two separate batches of A* were mixed and used as starting materials (Table 1). All experiments were conducted at the QFM oxygen buffer and at P = 1 atm. One-atmosphere, and therefore dry, experiments were conducted because basaltic SNC-like lavas or ash that might be analyzed by remote sensing would have likely completely degassed during emplacement at the Martian surface. Crystallization temperatures were varied between 1300 C and 980 C in order to achieve a wide range of crystallinities in the experimental products. For those experiments conducted at or above 1050 C, the tubing material (Ag 70 Pd 30 or Pt) was first saturated at the final experimental temperature using A* powder in order to prevent Fe loss in the actual experiment run. Experimental conditions are listed in Table 2. [8] In preparation for spectral analysis, the experiment products were ground in air using a mortar and pestle to one of three particle size ranges. Different particle size ranges were used because particle size can strongly affect both reflectance and emission spectra. A portion of all products was ground to a fine particle size of either <45 mm or <75 mm, while the remaining products were ground to a coarse particle size of mm. Morphology of the coarse particle size fragments created during grinding depended on the crystallinity of the samples. The glassiest samples broke into pieces that typically had some smooth, conchoidally fractured surfaces. The more crystalline samples broke into pieces with all rough surfaces. A similar distinction was observed in the fine particle size samples. Fine and coarse particle size products with similar crystallinities were created in several cases by conducting two separate experiments at the same temperature (Table 2). Final sample sizes after grinding ranged between 350 and 400 mg Basalt Oxidation [9] After synthesis, grinding, and initial chemical analysis (see section 3), the experimental products were subjected to a series of oxidation steps. Each sample was oxidized for 1, 3, and 7 days, and after each oxidation session the basalt was analyzed spectrally and several fragments were used to create a thin section. All oxidations were conducted in air at 700 C. Oxidation under these conditions does not likely represent the actual oxidation process on Mars; however, it is expected to produce oxidation products relevant to those found on Mars. For example, the oxidation experiments of Straub et al. [1991] were run in air from 7 hours to 28 days between 400 and 1000 C and yielded nanophase hematite, a likely oxidation product on Mars [Morris et al., 1989]. A sample before and after oxidation is pictured in Figure Analytical Procedures 3.1. Bulk Chemistry and Mineralogy [10] Prior to the oxidation experiments the residual glass (in experiments 1030 C) and phenocrysts from each experiment product were analyzed using the Brown University Cameca Camebax electron microprobe. We used a 10-nA defocused beam for the glasses and a 15-nA focused beam for the phenocrysts. The accelerating voltage during all analyses was 15 kv, and the sodium loss program of Nielsen and Sigurdsson [1974] was utilized during glass analyses. The electron microprobe data were used in the PETMIX mass balance program of Wright and Doherty [1970] in order to establish mode data for the experimental products. PETMIX revealed small degrees of Fe loss in some experimental products, but losses never exceeded 1.8 wt% Fe. In those experiments that did lose Fe, the modal amounts of the other phases present were normalized to account for the Fe loss. The modes from each experiment are listed in Table Reflectance Spectra [11] Spectral data were collected in the Keck/NASA RELAB facility at Brown University. At VISNIR wavelengths between 0.32 and 2.55 mm, reflectance spectra were obtained with a 10-nm sampling interval by the bidirectional spectrometer with a 30 incidence angle and a 0 emergence angle [Pieters, 1983; Mustard and Pieters, 1989]. For wavelengths between 1.8 and 26 mm, biconical reflectance spectra were obtained at 4-cm 1 intervals with the FTIR spectrometer with a 30 incidence angle and a 30 emergence angle. The biconcial reflectance data were translated into emissivity data via Kirchoff s law (e =1 r) for comparison to TES data. While use of Kirchoff s law is strictly viable only for directional hemispherical data [Salisbury et al., 1992], Mustard and Hays [1997] showed that biconical measurements are directly comparable to directional hemispherical and thermal emission measurements for fine particle sizes. Anomalous scattering may be present in coarse particle size samples when the complex index of refraction varies strongly with wavelength [Mustard and Hays, 1997]. [12] VISNIR data of the unoxidized, fine particle size samples were analyzed with the Modified Gaussian Model (MGM) of Table 2. Experimental Conditions a Sample Name Starting Material Temperature, C Duration, hours Tube Saturation Particle Size b MST-25 A* y coarse + fine MST-1 A* y fine MST-16 A* y coarse MST-26 A* y coarse MST-28 A* y fine MST-14 A* y fine MST-12 A* n fine MST-19 A* n coarse MST-13 A* n coarse + fine MST-17 A* n coarse a All experiments were conducted at 1 atm and fo2 = QFM. b Coarse + fine: single experiment product was analyzed at both coarse and fine ranges.
4 6-4 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA 3.3. Electron Microscopy [13] Transmission electron microscopy (TEM) analyses were conducted on fragments of an unoxidized sample (MST-19) and another sample (MST-16) oxidized for seven days using the Phillips 420 instrument at Brown University. Both samples were ion thinned in preparation for analysis; analyses were conducted with a 120-kV beam current. Images of the unoxidized and oxidized samples were obtained in bright-field mode, qualitative chemical analysis of the glass oxidation products was attempted with electron dispersive spectroscopy (EDS) using a 100-nm diameter beam, and diffraction patterns were obtained from the oxidation products using selected area diffraction in order to help constrain their composition. Scanning electron microscopy (SEM) analyses were carried out on oxidized coarse and fine particle size samples of 15 and 65% crystallinities using the JEOL JXA-8900 Superprobe at the Geophysical Laboratory of the Carnegie Institution of Washington. Surfaces of some of the coarse particle size samples were abraded with 400-grit emery paper before analysis. Backscattered electron images were obtained of the abraded and unabraded surfaces, and qualitative compositional analyses of materials at the surfaces of the particles were made using EDS. Imaging and compositional analyses were made at an accelerating voltage of 15 kv with a beam current of 1 na. Figure 3. Photomicrographs of unoxidized and oxidized glassbearing samples. Pictures are 2 mm across. (a) Sample MST-12 prior to oxidation. Phases present include pyroxene (px), plagioclase (pl), magnetite (m), and glass (gl) with 52 wt% SiO 2 and 19 wt% FeO. (b) MST-12 after oxidation for 1 day. Phases are still present, but the glassy areas (gl) have been transformed from orange to black in color. Sunshine et al. [1990] in order to establish the relative contributions of pyroxene and glass to the spectra. The MGM improves upon previous spectral deconvolution techniques by treating absorption bands as functions of bond length. To deconvolve an unknown spectrum, the model begins with prescribed absorption bands (position, width, strength) and iteratively adjusts the band parameters to obtain a fit to the unknown spectrum within the uncertainty associated with the unknown spectrum measurement. 4. Results 4.1. Macroscopic Properties [14] Intriguing macroscopic changes in the nature of the fine and coarse particle size samples were observed after the oxidation process. Prior to oxidation the samples were dark gray to black in color, and glassy samples had a vitreous luster. After oxidation the fine particle size samples had a brick red color, while the coarse particle size samples had a metallic sheen that was particularly well-developed on the smooth faces of the glassy, coarse particle fragments. When the surfaces of the coarse oxidized particles were abraded, the sheen on the surfaces was removed. Oxidation also caused both the fine and coarse particle size samples to become magnetic, despite containing no phenocrysts of magnetite SEM [15] Observations made at the macroscopic scale are confirmed and clarified at the microscopic scale by SEM imaging and EDS analyses. Unabraded surfaces of glassy, coarse particle size samples exhibit a relatively smooth, undulating texture and appear to be speckled with submicron particles (Figure 4a). The compositions of the unabraded surfaces are enriched in FeO and CaO and depleted in SiO 2 relative to the abraded surfaces, which are basaltic in composition. The surfaces of the glassy, fine particle samples are also smooth at the resolution of the SEM images (Figure 4b) and are covered with submicron phases visible at higher magnifications. These unabraded surfaces are enriched in FeO and CaO and Table 3. Mode Data for Basalt Synthesis Experiments Sample Name Experiment Temperature, C Percent Experimental Products, vol% Crystallization Glass Pig Aug Plag Mag Coarse Particle Size Products MST MST MST MST MST Fine Particle Size Products MST MST MST MST
5 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA 6-5 spherules (Figure 5a). The lack of similar features in the unoxidized glass confirms that the particles seen in the oxidized glass are a product of the oxidation process (Figure 6a). However, fine-scale structure is present in the unoxidized glass, perhaps a result of spinodal decomposition during quenching (R. A. Yund, personal communication, 2000). Images of pigeonite in the oxidized sample do not reveal the presence of a separate phase within the pigeonite or along the oxidized glass-pigeonite boundary (Figure 6b). [18] While attempts to chemically characterize the oxidation products in the oxidized glass with EDS met with limited success owing to the small size of the phases, selected area diffraction proved effective in determination of chemistry and microstructure Figure 4. (a) Backscattered electron images of the oxidized, 15% crystallized, coarse particle sample (MST-16). The boundary separates the abraded (dark) and unabraded (bright) portions of the surface. (b) Backscattered electron image of the oxidized, 15% crystallized, fine particle sample (MST-1). All surfaces are unabraded. Figures 4a and 4b reveal the relatively smooth nature of the individual faces of the glass-rich particles. (c) Backscattered electron image of the oxidized, 65% crystallized, coarse particle size sample (MST-19). Individual mineral grains, including plagioclase (pl), pyroxene (px) and Fe oxide (ox), are apparent at the surface. The smooth, medium-contrast surfaces represent the glass (gl) and have Ca- and Fe-rich compositions compared to the unoxidized glass. (d) Backscattered electron image of the oxidized, 65% crystallized, fine particle size sample (MST-12). Individual mineral grains are visible as in Figure 4c. depleted in SiO 2 relative to the abraded surface of the coarse particles, but the degree of FeO and CaO enrichment and SiO 2 depletion varies between the coarse and fine particle samples. [16] At the same magnification as in Figure 4a, the surfaces of the more crystalline samples (Figures 4c and 4d) are significantly rougher than the surface of the glassy sample, and individual mineral grains can be seen via their compositional contrast. Stoichiometric compositional analyses were obtained from the individual mineral grains (plagioclase, pyroxenes, Fe oxide), suggesting that the same material that covers the glassy sample surfaces does not cover the mineral grains. Outcrops of oxidized glass at the sample surfaces are FeO- and CaO-enriched and SiO 2 - depleted relative to the basaltic composition obtained from the abraded surface of the glassy, coarse particle sample, although the degrees of enrichment and depletion vary between each glassy area TEM [17] The TEM analyses provide even smaller-scale insight into the effects of oxidation. As is evident in Figure 3, the glassy portions of the basalts undergo significant change during the oxidation process. The bright-field images of an oxidized sample reveal the presence of very fine (25-nm) oxidation products throughout the glass (Figure 5). Some images suggest that oxidation products are arranged in larger-scale (2-mm) sectors or Figure 5. TEM bright-field image of oxidized (7 days) glassy areas in a 15% crystallized sample (MST-16). (a) Demonstration of the pervasive distribution of fine oxidation products throughout the glass and the organization of the particles into spherulite-like collections. Image is 4 mm across. (b) Higher magnification image reveals the small size of the individual particles (25 nm). Image is 1 mm across.
6 6-6 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA Figure 6. (a) TEM bright-field image of a pyroxene (pyx)-glass (gl) border in an unoxidized sample (MST-19). The glass displays fine-scale structure on the order of tens of nanometers, possibly owing to spinodal decomposition in the glass. Image is 2 mm across. (b) TEM bright-field image of pigeonite (pig) and glass (gl) within an oxidized sample (MST-16). The glass contains fine-scale oxidation products up to the boundary with the pyroxene, but no similar oxidation products are observed within the pyroxenes. Image is 2.2 mm across. of the glass oxidation products. Diffraction patterns, akin to powder diffraction, were obtained from a large (3.5-mm-diameter) glassy area containing a large number of oxidation products (e.g., Figure 5a) and also from crystallite clumps (e.g., Figure 5b) in the oxidized sample. The planar spacings derived from the oxidation products in the large glassy area diffraction pattern, when compared to the planar spacings of various candidate minerals, are best matched by those of the clinopyroxenes (Table 4). Diffraction patterns obtained from crystallite clumps have the appearance of single crystal diffraction patterns, suggesting that multiple individual particles in the clumps are subaligned with one another Spectroscopy Crystallization effects. [19] The spectra obtained of the unoxidized, variably crystallized samples at VISNIR and mid- IR wavelengths offer the first examination of crystallization effects on spectra of a SNC basaltic composition. In the fine particle VISNIR spectra with 15% crystallinity, the expression of the well-known 1- and 2-mm pyroxene crystal field absorptions is apparent (Figure 7a). With increasing degree of crystallinity, the pyroxene absorptions deepen, especially the band at 2.2 mm. The pure glass sample exhibits weak absorptions at 1.1 and 1.9 mm, and the expression of these absorptions decreases with increasing crystallinity. The spectra of the basalts crystallized >35% are still influenced by the glass bands, as evidenced by the inflections near 1.2 and 1.9 mm and the distortion of the 2.2-mm absorption relative to pure pyroxene spectra. [20] The coarse particle mid-ir data also reveal changes in spectral structure caused by changing degree of crystallization within the SNC basaltic samples (Figure 7d). The pure glass spectrum is featureless with one broad, asymmetrical feature near 1050 cm 1 and one narrower, symmetric feature near 500 cm 1. At as little as 15% crystallization an inflection develops near 1150 cm 1, individual features appear between 800 and 1100 cm 1, and the band between 400 and 600 cm 1 flattens and shifts slightly to higher wavenumbers. With increasing crystallization the inflection grows, the individual absorptions become more distinct, and the small wavenumber feature continues to broaden and shift to higher wavenumbers. Even with the development of features with increasing crystallization, the character of the pure glass spectrum influences the spectra of the samples crystallized up to 35%. [21] The complementary spectra to those in Figures 7a and 7d, the coarse particle VISNIR and the fine particle mid-ir data, are pictured in Figures 7b and 7c, respectively. The VISNIR spectra of the unoxidized, coarse particle samples are darker at all wavelengths and have a flatter continuum slope than the fine particle spectra (Figure 7b). This behavior is expected on the basis of the decreased amount of volume scattering in the coarse particle samples [Gaffey et al., 1993]. Despite the differences the coarse particle VISNIR spectra exhibit absorptions at the same wavelengths and display a loss of glass band expression with increasing crystallization as in the fine particle size data. In the mid-ir spectra the fine particle data exhibit a steep drop-off in emissivity at wavenumbers >1300 cm 1 and a reduction of spectral contrast between 600 and 1000 cm 1 (Figure 7c). Both of these effects can be attributed to volume scattering [Salisbury, 1993], which does not affect the general similarity in behavior of the coarse particle spectra relative to the fine particle size mid-ir spectra Oxidation effects. [22] Significant changes in both the VISNIR and mid-ir are apparent in a comparison of the unoxidized spectra and the spectra of the samples oxidized for just 1 day (Figure 8). In the fine particle VISNIR data the oxidized samples display a nearly complete loss of the 2.2-mm feature, a considerable decrease in spectral contrast of the 1-mm feature, and development of a flat continuum slope and a very strong absorption at wavelengths shorter than 0.7 mm. There is a trend of increasing reflectance between 0.7 and 2.5 mm with increasing duration of oxidation for each crystallization suite which is strongest in the least crystalline sample (Figure 8). There are few changes, however, in mineral absorption features with continued oxidation indicating that the duration of oxidation is less significant to the nature of the oxidized spectra than the oxidation mechanisms that are initiated by the first oxidation step. [23] The changes in mid-ir spectral characteristics caused by oxidation in the fine particle samples are crystallinity-dependent, with the greatest difference between the unoxidized and oxidized samples occurring in the glassiest sample (Figure 8d). In the glassiest sample spectrum the initial oxidation step leads to development of weak absorptions at 1100 cm 1 and between 400 and 500 cm 1, and longer oxidation times progressively strengthen these features. The feature at 1100 cm 1 correlates with a similar feature present in the spectra of unoxidized, crystalline samples (Figure 7c). In samples of greater crystallinity (Figures 8e and 8f), increasing degrees of oxidation mute the absorption features between 800 and 1100 cm 1 and lead to development of a weak feature between 400 and 500 cm 1 like that seen in the glassiest spectra (Figures 8e and 8f ). Overall, the development of features in the glassiest sample and the loss of features in the more crystalline samples ultimately lead the oxidized spectra of all crystallinities to converge toward a uniform spectral shape (Figure 9).
7 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA 6-7 Table 4. Interplanar Spacing Data for Oxidation Products and Selected Minerals a Measured, Å Fassaite, b Å Pigeonite, Å Augite, Å Bronzite, c Å Magnetite, Å Maghemite, Å Hematite, Å d d d d d d d d d d d d d d d d d d d d d a Interplanar spacings for all minerals except fassaite were obtained from Joint Committee on Selected Powder Diffraction Standards [1974]; all reported spacings (except for those of fassaite and bronzite) come from planes with reflection intensities > 0.10 I max. b Calculated d spacings based on cell parameters reported by Hazen and Finger [1977]. c All reported interplanar spacings in bronzite originate from planes with I > 0.30 I max. d Strongest intensities. Figure 7. VISNIR and mid-ir spectra of unoxidized experimental samples. Various colors indicate degree of crystallization from 0% (pure glass) to 80%. (a) Fine particle size (<45 and <75 mm) VISNIR data. (b) Coarse particle size ( mm) VISNIR data. (c) Fine particle size mid-ir data. Spectra colors follow legend in Figure 7a. (d) Coarse particle size mid-ir data. Spectra colors follow legend in Figure 7b. See color version of this figure at back of this issue.
8 6-8 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA Figure 8. Comparison of unoxidized and oxidized spectra of the fine particle size samples (<45 and <75 mm) in the VISNIR and mid-ir: VISNIR spectra for the (a) 15, (b) 55, and (c) 65% crystallized samples and mid-ir spectra for the (d) 15, (e) 55, and (f ) 65% crystallized samples. The legend in Figure 8a applies throughout Figure 8. The degree of crystallization is listed next to the figure label. [24] Spectra of the coarse particle size samples in the VISNIR and mid-ir offer different insights into the effects of oxidation (Figure 10). The same fundamental oxidation-induced changes that appear in fine particle VISNIR data also appear in the coarse particle VISNIR data (Figure 10a), with the addition of an absorption feature at 0.47 mm. The depth of this absorption decreases with increasing crystallinity (Figure 10a). In the mid-ir the spectra of the most glass-rich samples again undergo the largest oxidation-induced change, including development of three strong absorptions at 1250, 520, and 425 cm 1 and multiple weaker absorptions in the restrahlen band between 800 and 1100 cm 1 (Figure 10b). In the spectra of the more crystalline, coarse particle samples, individual absorptions are again muted by oxidation, but unlike the fine particle data (Figure 9), oxidation does not produce ultimate homogenization of coarse particle spectra. The absorptions present between 400 and 600 cm 1 in the 15% crystallized sample are only weakly expressed in the more crystallized samples. 5. Discussion 5.1. Nature of Glass Oxidation Products [25] The nature of the internal (clinopyroxene) and surficial (Ca- and Fe-rich coating ) oxidation products in the glasses of this study can be interpreted via the results of basaltic glass oxidation experiments [Cook et al., 1990; Cooper et al., 1996; Cook and Cooper, 2000]. In experiments in which oxidation temperatures were kept sufficiently low to avoid conventional surface devitrification (pyroxene nucleation [e.g., Cooper et al., 1996]), the three studies found that glasses exposed to oxidizing conditions responded in a manner dependent on the composition of the glass. Cook et al. [1990] found that Fe 2+ -bearing MgO-Al 2 O 3 - SiO 2 oxidized glass developed a surficial coating of MgO- (Mg,Fe) 3 O 4 and internal nanometer-sized MgFe 2 O 4 phases. Cooper et al. [1996] oxidized nepheline normative basaltic glass and found discrete CaO, MgO, and minor nepheline phases on the surface of the glass but no oxidation products internal to the glass. Cook and Cooper [2000] determined that oxidation of MgO- Al 2 O 3 -SiO 2 glass like that of Cook et al. [1990] but with roughly four times less FeO yielded Fe-enriched zones at the surface without formation of internal phases. In all these studies, glass oxidation is initiated by reaction of atmospheric oxygen with Fe 2+ in the glass to form Fe 3+. The oxidation front migrates into the sample by positive charge transfer from the newly formed Fe 3+ to a neighboring Fe 2+ within the sample. The transfer of positive charge into the sample is balanced by migration of positive divalent cations (Fe 2+,Ca 2+,Mg 2+ ) toward the sample surface. The movement of the divalent cations toward the surface of the oxidizing sample leads to the formation of the observed surficial oxidation products; surface phase precipitation occurs when the concentration of divalent cations becomes sufficiently high. The internal phases form when oxidation-produced Fe 3+, a network modifying cation, reaches a concentration that cannot be accommodated in the glass structure, thus leading to precipitation of Fe 3+ -bearing phases [e.g., Cook et al., 1990]. In glasses with sufficient Na 2 O, coupling of Na + and Fe 3+ creates a network-forming unit, thus allowing Fe 3+ to remain in the glass structure [e.g., Cooper et al., 1996]. [26] The oxidation mechanisms described in the above studies explain the oxidation of the SNC-based glasses of this study, controlled by the concentrations of Ca, Fe, Mg, and Na in the glasses. The elevated Ca and Fe surface concentrations in the SNCbased samples of this study are interpreted to result from outward migration of Ca 2+ and Fe 2+ driven by oxidizing surface conditions. The smooth-textured surface layer and the individual phases imaged on the oxidized surfaces (Figures 4a and 4b) are likely the manifestation of a Ca- and/or Fe-rich phase. The predominant involvement of Ca and Fe relative to Mg in the oxidation-induced cation migration suggests that the transport coefficients, controlled by concentration and diffusion coefficient, of Fe and Ca exceed that of Mg (R. F. Cooper, personal communication, 2000). This suggestion is supported by preoxidation analyses of the glasses indicating greater concentrations Fe and Ca in the glass relative to Mg at all crystallinities. Additionally, because the Fe concentrations of the glasses in this study are high relative to the basalts from the previous studies, Fe migration appears particularly important in Figure 9. Comparison of all the fine particle size mid-ir spectra oxidized for 7 days. Oxidation leads to homogenization of previously distinctive spectral shapes.
9 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA 6-9 the spectra of basaltic materials in the VISNIR [Singer and McCord, 1979] and the mid-ir [Johnson, 1999]. Figure 10. Coarse particle size samples oxidized for 7 days: (a) VISNIR spectra and (b) mid-ir spectra. The legend in Figure 10a also applies to Figure 10b. the oxidation of SNC glasses. Internal to the glasses, pyroxene formation is interpreted as the result of subsolidus devitrification but might also be due to nucleation of ferric-bearing pyroxene caused by a lack of sufficient Na + to charge-balance Fe 3+ within the glass. The pyroxenes within the glass, in addition to the phenocryst phases, are expected to complicate the glass oxidation process by altering the population of divalent cation phases available for migration; such an effect might explain why Fe and Ca levels vary at the measured oxidized glass surfaces. [27] The results of Cooper et al. [1996] can also be used to estimate the thickness of the Ca- and Fe-enriched layer at the surface of the glasses. On the basis of Rutherford backscattering spectroscopy measurements, Cooper et al. [1996] found that oxidation lasting 4 days at 600 C led to an enriched surface layer 0.4 mm thick. Assuming that the enriched layer thickness is enhanced by the greater oxidation temperature (700 C) and times (up to 7 days) of this study, a reasonable estimate for the enriched layer thickness in the oxidized samples is mm. [28] No matter what the specific oxidation mechanism, the signatures of the surficial Ca-bearing and Fe-bearing phases and the internal pyroxenes must be considered when interpreting the spectra of the experimental oxidized samples. Likewise, the potential influence of analogous oxidation products on the spectra of Martian surface samples must be taken into account. The effect of surficial oxidation products on Martian surface samples is especially important owing to the reported impact of coatings on 5.2. Spectroscopy Crystallization effects. [29] Changes due to crystallinity in the unoxidized spectra of the experimental SNC basalts can be interpreted through the characteristics of the individual phases in the basalts. VISNIR spectra of multiple Ferich basaltic glasses ( wt% FeO) created at a wide range of oxygen fugacities were studied by Bell et al. [1976], and each glass had absorptions near 1.0 and 1.8 mm, likely owing to crystal field and charge transfer transitions within the glasses. The positions of the VISNIR absorption bands in the experimental SNC composition glass fully coincide with the results of Bell et al. [1976], implying that the same mechanism is at work. The relationship between the observed VISNIR absorptions in the crystalline samples and those expected from pyroxenes within the samples can be assessed using the results of Cloutis and Gaffey [1991]. The sample crystallized 15% contains only one pyroxene, pigeonite. When absorption positions for the appropriate pigeonite composition are predicted using the data of Cloutis and Gaffey [1991], the predicted positions coincide well with those observed, despite the influence of the glass absorption bands on the pyroxene bands. Pyroxene absorption band positions become more difficult to interpret in the samples with 35% crystallization because of contributions from both pigeonite and augite. In reflectance spectra of Shergotty, which contains both pigeonite and augite, McFadden [1987] found that augite controlled the positions of the two major absorptions at 0.98 and 2.10 mm, despite a lower abundance of augite. Assuming that the spectra of the experimental SNC-based basalts are also dominated by augite absorptions, the observed bands at 1.0 and 2.26 mm (Figure 7a) both fall at wavelengths consistent with predicted augite absorption band positions from the Cloutis and Gaffey [1991] data. [30] Quantitative evaluation of the positions of absorptions from the SNC-based samples utilizing the Modified Gaussian Model [Sunshine et al., 1990] establishes the relative contributions of glass and pyroxene to the VISNIR spectra. Glass bands were initially assigned to the three absorption band positions observed in the 15% crystallized sample (0.92, 1.3, and 1.82 mm). The spectrum of the pure glass sample was not utilized because its steep continuum slope made it difficult to incorporate into the deconvolution. Pyroxene absorption band positions were initially assigned to the positions observed in the more crystalline samples (1.0 and 2.26 mm). With increasing crystallization, changes in the ratios of pyroxene band strength to glass band strength quantitatively demonstrate that the deepening of the 2.26 mm feature is due to the growing influence of the pyroxene bands (Figure 11). Cloutis et al. [1990] also observed strengthening of VISNIR pyroxene bands with increasing crystallinity in physical mixtures of pyroxenes, oxides, and basaltic glass. [31] The mid-ir data can also be interpreted in terms of the spectral characteristics of the individual SNC-based basalt components. The broad mid-ir absorptions present in the glass sample spectra are expected because the variety of bond lengths, strengths, and angles in glasses leads to absorptions at many wavelengths [Simon and McMahon, 1953]. Distinct features that appear in the more crystalline spectra are attributable to mineral absorptions and can be interpreted using data from the SNC meteorites. In the mid- IR spectrum of Zagami, a basaltic shergottite with subequal amounts of pigeonite and augite and subordinate amounts of plagioclase, Hamilton et al. [1997] observed features near 1100, 1010, 960, and 880 cm 1. The features at intermediate wavenumbers in the experimental SNC sample spectra fall at nearly these exact absorption positions (Figure 7d). This comparison suggests that the combined contribution of pigeonite, augite, and plagioclase leads to the observed absorption features in the experimental SNC spectra. The experimental SNC spectra should
10 6-10 MINITTI ET AL.: EFFECTS OF GLASS AND OXIDATION ON SNC BASALTIC SPECTRA Figure 11. Pyroxene/glass absorption band ratios plotted against degree of crystallization. The 1- and 2-mm pyroxene absorption bands are ratioed to the glass absorption band at 1.3 mm in the 15% crystallized sample. have a greater contribution from plagioclase relative to the actual SNC spectra because plagioclase absorption features weaken significantly at the shock level experienced by Zagami [Johnson et al., 2001]. The contribution from pyroxene absorptions to the experimental and actual SNC spectra should be similar because pyroxene absorption strengths are not significantly affected by shock [Johnson et al., 2001]. Thus, because of the relative effects of shock on plagioclase and pyroxene absorption strengths and the similarity of intermediate wavenumber absorption positions in the experimental and actual SNC spectra, we conclude that the major contributors to the intermediate wavenumber features are the pyroxenes. [32] The broad, flat, and relatively featureless absorption between 400 and 700 cm 1 in the experimental mid-ir spectra (Figure 7d) can be explained by the mineralogical and textural character of the samples. The gradual broadening and shallowing of the feature between 400 and 700 cm 1 with increasing crystallization can be accounted for with the increasing spectral influence of plagioclase. In pure mineral spectra at <700 cm 1, plagioclase absorptions are broader and less deep than pyroxene absorption features. Thus, as the proportion of plagioclase in the modes of the samples increases with increasing degree of crystallization (Table 3), the width and depth of the 400- to 700-cm 1 feature evolves toward a more plagioclase-like character. The featureless nature of the absorption resembles the character of fine-grained, volcanic rock spectra at low wavenumbers [Feely and Christensen, 1999; Hamilton and Christensen, 2000]. The Deccan basalt used as proxy for the Martian basaltic lithology by Christensen et al. [2000a], for example, is fine-grained and exhibits a similar featureless absorption between 400 and 700 cm 1. Thus the lack of features at small wavenumbers in the experimental sample spectra is in keeping with their fine grain size (15 mm) Oxidation effects. [33] The response of pyroxenerelated features to oxidation in the VISNIR is illustrated by the results of the pigeonite and augite oxidation experiments of Straub et al. [1991]. In pigeonite, Straub et al. found that nanophase hematite forms discrete phases which cause degradation of spectral contrast in the 1-mm feature and eventual elimination of the 2-mm feature. These observations are consistent with the behavior of the 1- and 2.26-mm features in the oxidized sample spectra; however, no nanophase hematite was detected in pigeonite using TEM (the signature of nanophase hematite is not expected to be detectable in the SEM-EDS analysis owing to the qualitative nature of the technique). Straub et al. found that the nanophase hematite created during pigeonite oxidation formed only at the exposed surfaces of the grains, leaving pristine pigeonite within the grains. Thus the lack of nanophase hematite within pigeonite and on the pigeonite surfaces in contact with the glass is consistent with the oxidation mechanism described by Straub et al. [1991]. In augite, Straub et al. found that oxidation leads to the incorporation of Fe 3+ into the pyroxene structure, which weakens the major augite absorptions. An increase in Fe 3+ within the experimental augites was not directly measured, but the behavior of the 1- and 2.26-mm absorptions is consistent with the development of Fe 3+ in augite. Overall, behavior of the 1- and 2.26-mm features in the VISNIR can be partially if not wholly attributed to the degradation of the pigeonite and augite absorptions caused by oxidation of the pyroxenes exposed at the particle surfaces. [34] Degradation of the pyroxene signal via oxidation is also suggested in the experimental mid-ir data. The features in the unoxidized spectra attributed to pigeonite and augite become more muted with increasing degrees of oxidation (Figures 8e and 8f), suggesting that oxidation lessens the contribution of pyroxene to the bulk rock spectra. The degradation of the pyroxene features is also consistent with the addition of a spectrally neutral material such as nanophase ferric oxide. [35] The other significant change between the unoxidized and oxidized spectra in the VISNIR is the development of the absorption at <0.7 mm in both the fine and coarse particle spectra (Figures 8a 8c and 10a). The observed feature is consistent with nanophase hematite [Morris et al., 1989], whose presence in the oxidized pigeonites of the experimental basalts is already suggested spectrally. However, coarser particle size hematite also exhibits a similar reflectance drop off and provides a better match to the minor absorption at 0.47 mm (Figures 10 and 12). The match between the hematite spectra and the experimental samples could be due to hematite produced through oxidation of magnetite that crystallized in the samples during the basalt syntheses. However, the feature is present in the spectra of samples that did not originally crystallize magnetite, suggesting that the hematite signal is not from oxidized phenocrystic magnetite. The presence of hematite is also consistent with the red color of the oxidized fine particle samples and the magnetic properties of the oxidized samples. [36] The hematite signal in the VISNIR, as well as the red coloration and magnetic character of the oxidized samples, can be explained if the Fe-enriched material at the surface of the oxidized samples is hematite. A hematite layer up to 1 mm thick (estimated thickness of the Fe-enriched layer) combined with the high n and k of hematite at <0.7 mm makes such a hematite layer optically thick in the VISNIR. Additionally, particle sizes of both the fine and coarse samples are sufficiently large relative to the VISNIR wavelengths of incident radiation that geometric optics governs particle-light interactions [Mustard and Hays, 1997]. In the VISNIR, two factors affect the strength of the hematite absorption: sample glass content and particle size. A stronger hematite signal is expected from more glass-rich samples because hematite is present at the surface of the oxidized glass. A weaker hematite signal is expected from finer particle size samples because of the increased number of scattering surfaces present. In addition, the multiple scattering surfaces within the fine particle size samples result in greater absorption and a more nanophase hematite-like spectrum (Figures 9a 9c) [Morris et al., 1989]. Indeed, the hematite absorption at 0.47 mm is strongest in a glassy, coarse sample (Figure 10a) and weakest in a more crystalline, fine sample (Figure 8c). The glassy, fine particle sample has a weaker hematite signal than the more crystalline, coarse particle sample, despite having a greater exposure of glass (Figures 8a and 10a). These latter two samples illustrate that the effects of glass content and particle size compete to establish the strength of hematite signal. The observed relative strengths of the hematite signal in these samples suggest that the scattering effect overrides the glass content effect.
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 106, NO. 7, PAGES 14,733 14,746, JULY 25, 2001
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 106, NO. 7, PAGES 14,733 14,746, JULY 25, 2001 Analysis of terrestrial and Martian volcanic compositions using thermal emission spectroscopy 2. Application to Martian
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 106, NO. 7, PAGES 14,733 14,746, JULY 25, 2001 Analysis of terrestrial and Martian volcanic compositions using thermal emission spectroscopy 2. Application to Martian
Discrimination of glass and phyllosilicate minerals in thermal infrared data
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2005je002474, 2005 Discrimination of glass and phyllosilicate minerals in thermal infrared data William C. Koeppen and Victoria E. Hamilton Hawaii
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 110,, doi:10.1029/2005je002474, 2005 Discrimination of glass and phyllosilicate minerals in thermal infrared data William C. Koeppen and Victoria E. Hamilton Hawaii
Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same
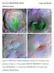 GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
GSA DATA REPOSITORY 2015222 Cannon and Mustard Additional examples Figure S1. CRISM maps of modeled mineralogy projected over CTX imagery (same parameters as Figure 1). A: Taytay Crater (CRISM ID: HRL00005B77).
Using the modified Gaussian model to extract quantitative data from lunar soils
 Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2006je002721, 2006 Using the modified Gaussian model to extract quantitative data from lunar soils Sarah K. Noble, 1,2
Click Here for Full Article JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 111,, doi:10.1029/2006je002721, 2006 Using the modified Gaussian model to extract quantitative data from lunar soils Sarah K. Noble, 1,2
UV-V-NIR Reflectance Spectroscopy
 UV-V-NIR Reflectance Spectroscopy Methods and Results A. Nathues Naturally-occurring inorganic substances with a definite and predictable chemical composition and physical properties Major groups: Silicates
UV-V-NIR Reflectance Spectroscopy Methods and Results A. Nathues Naturally-occurring inorganic substances with a definite and predictable chemical composition and physical properties Major groups: Silicates
Imagine the first rock and the cycles that it has been through.
 A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
A rock is a naturally formed, consolidated material usually composed of grains of one or more minerals The rock cycle shows how one type of rocky material gets transformed into another The Rock Cycle Representation
Pretty Green Mineral -- Pretty Dry Mars? Written by Linda M.V. Martel Hawai'i Institute of Geophysics and Planetology
 1 of 7 posted November 7, 2003 Pretty Green Mineral -- Pretty Dry Mars? Written by Linda M.V. Martel Hawai'i Institute of Geophysics and Planetology --- The discovery of olivine-bearing rocks on Mars underscores
1 of 7 posted November 7, 2003 Pretty Green Mineral -- Pretty Dry Mars? Written by Linda M.V. Martel Hawai'i Institute of Geophysics and Planetology --- The discovery of olivine-bearing rocks on Mars underscores
Assessing the limits of the Modified Gaussian Model for remote spectroscopic studies of pyroxenes on Mars
 Icarus 187 (2007) 442 456 www.elsevier.com/locate/icarus Assessing the limits of the Modified Gaussian Model for remote spectroscopic studies of pyroxenes on Mars Lisa C. Kanner, John F. Mustard, Aline
Icarus 187 (2007) 442 456 www.elsevier.com/locate/icarus Assessing the limits of the Modified Gaussian Model for remote spectroscopic studies of pyroxenes on Mars Lisa C. Kanner, John F. Mustard, Aline
Treatment of Data. Methods of determining analytical error -Counting statistics -Reproducibility of reference materials -Homogeneity of sample
 Treatment of Data Methods of determining analytical error -Counting statistics -Reproducibility of reference materials -Homogeneity of sample Detection Limits Assessment of analytical quality -Analytical
Treatment of Data Methods of determining analytical error -Counting statistics -Reproducibility of reference materials -Homogeneity of sample Detection Limits Assessment of analytical quality -Analytical
Geos 306, Mineralogy Final Exam, Dec 12, pts
 Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
Name: Geos 306, Mineralogy Final Exam, Dec 12, 2014 200 pts 1. (9 pts) What are the 4 most abundant elements found in the Earth and what are their atomic abundances? Create a reasonable hypothetical charge-balanced
Block: Igneous Rocks. From this list, select the terms which answer the following questions.
 Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
Geology 12 Name: Mix and Match: Igneous Rocks Refer to the following list. Block: porphyritic volatiles mafic glassy magma mixing concordant discontinuous reaction series igneous vesicular partial melting
EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4
 Name: EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4 Introduction In the previous lab, we learned about mineral characteristics, properties and identities as well as the three basic rock
Name: EPS 50 Lab 2: Igneous Rocks Grotzinger and Jordan, Chapter 4 Introduction In the previous lab, we learned about mineral characteristics, properties and identities as well as the three basic rock
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100
 Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Igneous Rock Classification, Processes and Identification Physical Geology GEOL 100 Ray Rector - Instructor Major Concepts 1) Igneous rocks form directly from the crystallization of a magma or lava 2)
Engineering Geology ECIV 2204
 Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
Engineering Geology ECIV 2204 Instructor : Dr. Jehad Hamad 2017-2016 Chapter (3) Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth Igneous Rocks Chapter 3: Rocks: Materials of the Solid Earth
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY
 WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
WORKING WITH ELECTRON MICROPROBE DATA FROM A HIGH PRESSURE EXPERIMENT CALCULATING MINERAL FORMULAS, UNIT CELL CONTENT, AND GEOTHERMOMETRY Brandon E. Schwab Department of Geology Humboldt State University
Essentials of Geology, 11e
 Essentials of Geology, 11e Igneous Rocks and Intrusive Activity Chapter 3 Instructor Jennifer Barson Spokane Falls Community College Geology 101 Stanley Hatfield Southwestern Illinois College Characteristics
Essentials of Geology, 11e Igneous Rocks and Intrusive Activity Chapter 3 Instructor Jennifer Barson Spokane Falls Community College Geology 101 Stanley Hatfield Southwestern Illinois College Characteristics
(Received 21 January 2015; revision accepted 23 October 2015)
 Meteoritics & Planetary Science 51, Nr 2, 207 234 (2016) doi: 10.1111/maps.12588 Complexities in pyroxene compositions derived from absorption band centers: Examples from Apollo samples, HED meteorites,
Meteoritics & Planetary Science 51, Nr 2, 207 234 (2016) doi: 10.1111/maps.12588 Complexities in pyroxene compositions derived from absorption band centers: Examples from Apollo samples, HED meteorites,
Rocks and Minerals. Tillery, Chapter 19. Solid Earth Materials
 Rocks and Minerals Tillery, Chapter 19 Science 330 Summer 2007 No other planet in the solar system has the unique combination of fluids of Earth. Earth has a surface that is mostly covered with liquid
Rocks and Minerals Tillery, Chapter 19 Science 330 Summer 2007 No other planet in the solar system has the unique combination of fluids of Earth. Earth has a surface that is mostly covered with liquid
Spectroscopy: The Study of Squiggly Lines. Reflectance spectroscopy: light absorbed at specific wavelengths corresponding to energy level transi8ons
 Spectroscopy: The Study of Squiggly Lines Reflectance spectroscopy: light absorbed at specific wavelengths corresponding to energy level transi8ons Interaction of Radiant Energy and Matter What causes
Spectroscopy: The Study of Squiggly Lines Reflectance spectroscopy: light absorbed at specific wavelengths corresponding to energy level transi8ons Interaction of Radiant Energy and Matter What causes
Lab 3 - Identification of Igneous Rocks
 Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
Lab 3 - Identification of Igneous Rocks Page - 1 Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly
(Received 18 October 2002; revision accepted 28 April 2003)
 Meteoritics & Planetary Science 38, Nr 6, 871 885 (2003) Abstract available online at http://meteoritics.org Searching for the source regions of martian meteorites using MGS TES: Integrating martian meteorites
Meteoritics & Planetary Science 38, Nr 6, 871 885 (2003) Abstract available online at http://meteoritics.org Searching for the source regions of martian meteorites using MGS TES: Integrating martian meteorites
The Nature of Igneous Rocks
 The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
The Nature of Igneous Rocks Form from Magma Hot, partially molten mixture of solid liquid and gas Mineral crystals form in the magma making a crystal slush Gases - H 2 O, CO 2, etc. - are dissolved in
Dept., Univ. of ela as are, 'i?ewark, DE Approximately 130 low specific gravity ((2.601, high silica
 HIGH (760%) SiO LUNAR GLASS%. ir.f. Glass, Geoloey Dept., Univ. of ela as are, 'i?ewark, DE. 1971 1 Approximately 130 low specific gravity ((2.601, high silica p602) glass particles recovered from a 4.88
HIGH (760%) SiO LUNAR GLASS%. ir.f. Glass, Geoloey Dept., Univ. of ela as are, 'i?ewark, DE. 1971 1 Approximately 130 low specific gravity ((2.601, high silica p602) glass particles recovered from a 4.88
Supporting Information
 Supporting Information Bindi et al. 10.1073/pnas.1111115109 Fig. S1. Electron microprobe X-ray elemental maps for the grain reported in Fig. 1B. Experimental details are given in Experimental Methods.
Supporting Information Bindi et al. 10.1073/pnas.1111115109 Fig. S1. Electron microprobe X-ray elemental maps for the grain reported in Fig. 1B. Experimental details are given in Experimental Methods.
Lab 4 - Identification of Igneous Rocks
 Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
Lab 4 - Identification of Igneous Rocks Page - Introduction A rock is a substance made up of one or more different minerals. Thus an essential part of rock identification is the ability to correctly recognize
A Rock is a solid aggregate of minerals.
 Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Quartz A Rock is a solid aggregate of minerals. Orthoclase Feldspar Plagioclase Feldspar Biotite Four different minerals are obvious in this piece of Granite. The average automobile contains: Minerals
Metcalf and Buck. GSA Data Repository
 GSA Data Repository 2015035 Metcalf and Buck Figure DR1. Secondary ionization mass-spectrometry U-Pb zircon geochronology plots for data collected on two samples of Wilson Ridge plutonic rocks. Data presented
GSA Data Repository 2015035 Metcalf and Buck Figure DR1. Secondary ionization mass-spectrometry U-Pb zircon geochronology plots for data collected on two samples of Wilson Ridge plutonic rocks. Data presented
23/9/2013 ENGINEERING GEOLOGY. Chapter 2: Rock classification:
 ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
ENGINEERING GEOLOGY Chapter 2: Rock classification: ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks
Spectroscopy of Loose and Cemented Sulfate-Bearing Soils: Implications for Duricrust on Mars
 Icarus 158, 42 55 (2002) doi:10.1006/icar.2002.6874 Spectroscopy of Loose and Cemented Sulfate-Bearing Soils: Implications for Duricrust on Mars Christopher D. Cooper and John F. Mustard Department of
Icarus 158, 42 55 (2002) doi:10.1006/icar.2002.6874 Spectroscopy of Loose and Cemented Sulfate-Bearing Soils: Implications for Duricrust on Mars Christopher D. Cooper and John F. Mustard Department of
Experimental petrology of the Mars Pathfinder rock composition: Constraints on the interpretation of Martian reflectance spectra
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 113,, doi:10.1029/2007je002983, 2008 Experimental petrology of the Mars Pathfinder rock composition: Constraints on the interpretation of Martian reflectance spectra
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 113,, doi:10.1029/2007je002983, 2008 Experimental petrology of the Mars Pathfinder rock composition: Constraints on the interpretation of Martian reflectance spectra
VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND
 VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND LEBN SCHUYLER Whitman College Sponsor: John Winter INTRODUCTION Iceland is exposed above sea level
VOLCANIC STRATIGRAPHY AND PETROLOGY OF THE NORTHERN SNAEFELLSNES RIFT, SOUTHERN LAXÁRDALSFJÖLL, ICELAND LEBN SCHUYLER Whitman College Sponsor: John Winter INTRODUCTION Iceland is exposed above sea level
Supplementary Figure 1 Panoramic view of four sites (CE-0005, CE-0006, CE-0007, and CE-0008) measured by APXS and VNIS. Images (a), (b), and (d) were
 Supplementary Figure 1 Panoramic view of four sites (CE-0005, CE-0006, CE-0007, and CE-0008) measured by APXS and VNIS. Images (a), (b), and (d) were acquired by the Panoramic Camera at the CE3-0007 site,
Supplementary Figure 1 Panoramic view of four sites (CE-0005, CE-0006, CE-0007, and CE-0008) measured by APXS and VNIS. Images (a), (b), and (d) were acquired by the Panoramic Camera at the CE3-0007 site,
The Tricky Business of Identifying Rocks on Mars
 1 of 8 posted May 22, 2002 The Tricky Business of Identifying Rocks on Mars --- A new analysis of thermal emission spectra suggests a new interpretation for the composition of the Martian surface. Written
1 of 8 posted May 22, 2002 The Tricky Business of Identifying Rocks on Mars --- A new analysis of thermal emission spectra suggests a new interpretation for the composition of the Martian surface. Written
Estimating Modal Abundances From the Spectra of
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 98, NO. E5, PAGES 9075-9087, MAY 25, 1993 Estimating Modal Abundances From the Spectra of Natural and Laboratory Pyroxene Mixtures Using the Modified Gaussian Model
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 98, NO. E5, PAGES 9075-9087, MAY 25, 1993 Estimating Modal Abundances From the Spectra of Natural and Laboratory Pyroxene Mixtures Using the Modified Gaussian Model
PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE
 PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE DANIEL HAWKINS Western Kentucky University Research Advisor: Andrew Wulff INTRODUCTION Round Point, in the
PETROGENESIS OF A SERIES OF MAFIC SHEETS WITHIN THE VINALHAVEN PLUTON, VINALHAVEN ISLAND, MAINE DANIEL HAWKINS Western Kentucky University Research Advisor: Andrew Wulff INTRODUCTION Round Point, in the
Near-infrared spectra of clinopyroxenes: Effects of calcium content and crystal structure
 Meteoritics & Planetary Science 46, Nr 3, 379 395 (2011) doi: 10.1111/j.1945-5100.2010.01158.x Near-infrared spectra of clinopyroxenes: Effects of calcium content and crystal structure Rachel L. KLIMA
Meteoritics & Planetary Science 46, Nr 3, 379 395 (2011) doi: 10.1111/j.1945-5100.2010.01158.x Near-infrared spectra of clinopyroxenes: Effects of calcium content and crystal structure Rachel L. KLIMA
Rocks. Types of Rocks
 Rocks Rocks are the most common material on Earth. They are naturally occurring aggregates of one or more minerals. 1 Igneous rocks, Types of Rocks Sedimentary rocks and Metamorphic rocks. 2 1 3 4 2 IGNEOUS
Rocks Rocks are the most common material on Earth. They are naturally occurring aggregates of one or more minerals. 1 Igneous rocks, Types of Rocks Sedimentary rocks and Metamorphic rocks. 2 1 3 4 2 IGNEOUS
Volatiles (H, C, N, F, S, Cl) in the lunar mantle, crust, and regolith: What questions remain and where to go next?
 Volatiles (H, C, N, F, S, Cl) in the lunar mantle, crust, and regolith: What questions remain and where to go next? Francis M. McCubbin & Charles K. Shearer Motivation behind this work 100, In Revision,
Volatiles (H, C, N, F, S, Cl) in the lunar mantle, crust, and regolith: What questions remain and where to go next? Francis M. McCubbin & Charles K. Shearer Motivation behind this work 100, In Revision,
Olivine-Pyroxene Distribution of S-type Asteroids Throughout the Main Belt
 Olivine-Pyroxene Distribution of S-type Asteroids Throughout the Main Belt Shaye Storm IfA REU 2007 and Massachusetts Institute of Technology Advisor: Schelte J. Bus Received ; accepted 2 ABSTRACT The
Olivine-Pyroxene Distribution of S-type Asteroids Throughout the Main Belt Shaye Storm IfA REU 2007 and Massachusetts Institute of Technology Advisor: Schelte J. Bus Received ; accepted 2 ABSTRACT The
9/4/2015. Feldspars White, pink, variable Clays White perfect Quartz Colourless, white, red, None
 ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
ENGINEERING GEOLOGY Chapter 1.0: Introduction to engineering geology Chapter 2.0: Rock classification Igneous rocks Sedimentary rocks Metamorphic rocks Chapter 3.0: Weathering & soils Chapter 4.0: Geological
How 2 nd half labs will work
 How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
How 2 nd half labs will work Continue to use your mineral identification skills Learn to describe, classify, interpret rock hand samples: Igneous sedimentary metamorphic volcanic plutonic (1 week) (1 wk)
MgO was dried at 1100 C overnight, then stoichiometrically combined with 95% enriched 57 Fe 2 O 3
 82 Appendix A A.1 Synthesis and Characterization A.1.1 (Mg 0.16 Fe 0.84 )O MgO was dried at 1100 C overnight, then stoichiometrically combined with 95% enriched 57 Fe 2 O 3 (Advanced Materials Techonologies
82 Appendix A A.1 Synthesis and Characterization A.1.1 (Mg 0.16 Fe 0.84 )O MgO was dried at 1100 C overnight, then stoichiometrically combined with 95% enriched 57 Fe 2 O 3 (Advanced Materials Techonologies
GSA DATA REPOSITORY
 GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
GSA DATA REPOSITORY 2013019 Supplemental information for The Solidus of Alkaline Carbonatite in the Deep Mantle Konstantin D. Litasov, Anton Shatskiy, Eiji Ohtani, and Gregory M. Yaxley EXPERIMENTAL METHODS
Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks
 321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
321 Lab 8 Instructor: L. Porritt - 1 - Igneous petrology EOSC 321 Laboratory 8: Intermediate and Felsic Volcanic Rocks. Pyroclastic Rocks Learning Goals. After this Lab, you should be able: Identify fine-grained
Minerals: Minerals: Building blocks of rocks. Atomic Structure of Matter. Building Blocks of Rocks Chapter 3 Outline
 Minerals: Building Blocks of Rocks Chapter 3 Outline Does not contain complete lecture notes. To be used to help organize lecture notes and home/test studies. Minerals: Building blocks of rocks Definition
Minerals: Building Blocks of Rocks Chapter 3 Outline Does not contain complete lecture notes. To be used to help organize lecture notes and home/test studies. Minerals: Building blocks of rocks Definition
Modeling Optical Properties of Martian Dust Using Mie Theory
 Modeling Optical Properties of Martian Dust Using Mie Theory Attila Elteto ATOC 5235: Remote Sensing of the Atmosphere and Oceans Spring, 2003 1. Introduction The Mie-Debye theory is a simple method for
Modeling Optical Properties of Martian Dust Using Mie Theory Attila Elteto ATOC 5235: Remote Sensing of the Atmosphere and Oceans Spring, 2003 1. Introduction The Mie-Debye theory is a simple method for
Written by G. Jeffrey Taylor Hawai i Institute of Geophysics and Planetology. A Plethera of Geochemical Data. May 8, 2009.
 1 of 8 May 8, 2009 --- Chemical analyses of rocks on the Martian surface indicate that the Martian crust was built of basalt lava flows not much different from those on Earth. Written by G. Jeffrey Taylor
1 of 8 May 8, 2009 --- Chemical analyses of rocks on the Martian surface indicate that the Martian crust was built of basalt lava flows not much different from those on Earth. Written by G. Jeffrey Taylor
Characterization of Montana Grow Product David Mogk January 16, 2014
 Characterization of Montana Grow Product David Mogk January 16, 2014 Samples of the raw material that is used in the production of Montana Grow products were characterized using X- ray Powder Diffraction
Characterization of Montana Grow Product David Mogk January 16, 2014 Samples of the raw material that is used in the production of Montana Grow products were characterized using X- ray Powder Diffraction
Mineralogy of Mars: Using our Experiences on Earth to Understand Processes on Mars. Liz Rampe (NASA-JSC) 8 July 2014
 Mineralogy of Mars: Using our Experiences on Earth to Understand Processes on Mars Liz Rampe (NASA-JSC) 8 July 2014 elizabeth.b.rampe@nasa.gov Topics of this Talk Introduction to mineralogy What are minerals
Mineralogy of Mars: Using our Experiences on Earth to Understand Processes on Mars Liz Rampe (NASA-JSC) 8 July 2014 elizabeth.b.rampe@nasa.gov Topics of this Talk Introduction to mineralogy What are minerals
The Surprising Lunar Maria
 1 of 5 posted June 23, 2000 The Surprising Lunar Maria Written by G. Jeffrey Taylor Hawai'i Institute of Geophysics and Planetology The lunar maria, the dark, smooth areas on the Moon, formed when lava
1 of 5 posted June 23, 2000 The Surprising Lunar Maria Written by G. Jeffrey Taylor Hawai'i Institute of Geophysics and Planetology The lunar maria, the dark, smooth areas on the Moon, formed when lava
Supplementary Materials for
 advances.sciencemag.org/cgi/content/full/4/5/eaar4378/dc1 Supplementary Materials for Discovery of moganite in a lunar meteorite as a trace of H2O ice in the Moon s regolith Masahiro Kayama, Naotaka Tomioka,
advances.sciencemag.org/cgi/content/full/4/5/eaar4378/dc1 Supplementary Materials for Discovery of moganite in a lunar meteorite as a trace of H2O ice in the Moon s regolith Masahiro Kayama, Naotaka Tomioka,
A Global View of Martian Surface Compositions from MGS-TES
 R ESEARCH A RTICLES A Global View of Martian Surface Compositions from MGS-TES Joshua L. Bandfield,* Victoria E. Hamilton, Philip R. Christensen Thermal Emission Spectrometer (TES) data from the Mars Global
R ESEARCH A RTICLES A Global View of Martian Surface Compositions from MGS-TES Joshua L. Bandfield,* Victoria E. Hamilton, Philip R. Christensen Thermal Emission Spectrometer (TES) data from the Mars Global
A4 CAI COMPOSITION: DIVALENT ELEMENT PARTITIONING AND CATHODOLUMINESCENCE. A4.1 Anorthite and Melt compositions for Synthetic CAI Composition 224SAM
 147 A4 CAI COMPOSITION: DIVALENT ELEMENT PARTITIONING AND CATHODOLUMINESCENCE A4.1 Anorthite and Melt compositions for Synthetic CAI Composition 224SAM Anorthite-melt partitioning of divalent elements
147 A4 CAI COMPOSITION: DIVALENT ELEMENT PARTITIONING AND CATHODOLUMINESCENCE A4.1 Anorthite and Melt compositions for Synthetic CAI Composition 224SAM Anorthite-melt partitioning of divalent elements
Matter and Minerals Earth: Chapter Pearson Education, Inc.
 Matter and Minerals Earth: Chapter 3 Minerals: Building Blocks of Rocks By definition a mineral is: Naturally occurring An inorganic solid Ordered internal molecular structure Definite chemical composition
Matter and Minerals Earth: Chapter 3 Minerals: Building Blocks of Rocks By definition a mineral is: Naturally occurring An inorganic solid Ordered internal molecular structure Definite chemical composition
Chapter 4 Rocks & Igneous Rocks
 Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Chapter 4 Rocks & Igneous Rocks Rock Definition A naturally occurring consolidated mixture of one or more minerals e.g, marble, granite, sandstone, limestone Rock Definition Must naturally occur in nature,
Engineering Geology. Igneous rocks. Hussien Al - deeky
 Igneous rocks Hussien Al - deeky 1 The Geology Definition of Rocks In Geology Rock is defined as the solid material forming the outer rocky shell or crust of the earth. There are three major groups of
Igneous rocks Hussien Al - deeky 1 The Geology Definition of Rocks In Geology Rock is defined as the solid material forming the outer rocky shell or crust of the earth. There are three major groups of
Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN, E. E. BEYER AND H. K. BRUECKNER
 Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Pressure- and stress-induced fabric transition in olivine from peridotites in the Western Gneiss Region (Norway): implications for mantle seismic anisotropy Q. WANG, Q-K. XIA, S. Y. O REILLY, W. L. GRIFFIN,
Objectives of this Lab. Introduction. The Petrographic Microscope
 Geological Sciences 101 Lab #9 Introduction to Petrology Objectives of this Lab 1. Understand how the minerals and textures of rocks reflect the processes by which they were formed. 2. Understand how rocks
Geological Sciences 101 Lab #9 Introduction to Petrology Objectives of this Lab 1. Understand how the minerals and textures of rocks reflect the processes by which they were formed. 2. Understand how rocks
Lecture 3 Rocks and the Rock Cycle Dr. Shwan Omar
 Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Rocks A naturally occurring aggregate of one or more minerals (e.g., granite), or a body of non-crystalline material (e.g., obsidian glass), or of solid organic material (e.g., coal). Rock Cycle A sequence
Student Name: College: Grade:
 Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
Student Name: College: Grade: Physical Geology Laboratory IGNEOUS MINERALS AND ROCKS IDENTIFICATION - INTRODUCTION & PURPOSE: In this lab you will learn to identify igneous rocks in hand samples from their
LAB 3: SPECTROSCOPY. GEOL104: Exploring the Planets
 LAB 3: SPECTROSCOPY OBJECTIVES: I. Review the basics of spectroscopy, including how to identify different materials on the basis of spectra. II. Develop an understanding of general spectroscopic features
LAB 3: SPECTROSCOPY OBJECTIVES: I. Review the basics of spectroscopy, including how to identify different materials on the basis of spectra. II. Develop an understanding of general spectroscopic features
Chapter 4 8/27/2013. Igneous Rocks. and Intrusive Igneous Activity. Introduction. The Properties and Behavior of Magma and Lava
 Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
Introduction Chapter 4 Igneous rocks form by the cooling of magma (or lava). Large parts of the continents and all the oceanic crust are composed of. and Intrusive Igneous Activity The Properties and Behavior
OCEAN/ESS 410. Lab 8. Igneous rocks
 Lab 8. Igneous rocks Today s exercise is an introduction to rock identification and the crustal structure of the seafloor. All rocks are composed of two or more minerals, and can be classified based on
Lab 8. Igneous rocks Today s exercise is an introduction to rock identification and the crustal structure of the seafloor. All rocks are composed of two or more minerals, and can be classified based on
Earth Science 11: Earth Materials: Rock Cycle
 Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Name: Date: Earth Science 11: Earth Materials: Rock Cycle Chapter 2, pages 44 to 46 2.1: Rock Cycle What is a Rock? A solid mass of mineral or mineral-like matter that occurs naturally as part of our planet
Effects of Very Fine Particle Size on Reflectance Spectra of Smectite and Palagonitic Soil
 Icarus 142, 557 570 (1999) Article ID icar.1999.6221, available online at http://www.idealibrary.com on Effects of Very Fine Particle Size on Reflectance Spectra of Smectite and Palagonitic Soil Christopher
Icarus 142, 557 570 (1999) Article ID icar.1999.6221, available online at http://www.idealibrary.com on Effects of Very Fine Particle Size on Reflectance Spectra of Smectite and Palagonitic Soil Christopher
Chapter 3: Igneous Rocks 3.2 IGNEOUS ROCK ORIGIN
 Chapter 3: Igneous Rocks Adapted by Lyndsay R. Hauber & Michael B. Cuggy (2018) University of Saskatchewan from Deline B, Harris R & Tefend K. (2015) "Laboratory Manual for Introductory Geology". First
Chapter 3: Igneous Rocks Adapted by Lyndsay R. Hauber & Michael B. Cuggy (2018) University of Saskatchewan from Deline B, Harris R & Tefend K. (2015) "Laboratory Manual for Introductory Geology". First
EPMA IMAGES. Figure 9. Energy-dispersive spectra of spot mineral analyses in sample 89GGR-33A for locations 1-5 in Figure 8.
 EPMA IMAGES The attached images and mineral data can be used to supplement an instrument-based lab, or serve as the basis for lab that can be completed without an instrument. Please provide credit for
EPMA IMAGES The attached images and mineral data can be used to supplement an instrument-based lab, or serve as the basis for lab that can be completed without an instrument. Please provide credit for
Quartz. ! Naturally occurring - formed by nature. ! Solid - not liquid or gas. Liquid water is not a mineral
 GEOL 110 - Minerals, Igneous Rocks Minerals Diamond Azurite Quartz Why Study Minerals?! Rocks = aggregates of minerals! Importance to Society?! Importance to Geology? 5 part definition, must satisfy all
GEOL 110 - Minerals, Igneous Rocks Minerals Diamond Azurite Quartz Why Study Minerals?! Rocks = aggregates of minerals! Importance to Society?! Importance to Geology? 5 part definition, must satisfy all
305 ATOMS, ELEMENTS, AND MINERALS
 DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
305 ATOMS, ELEMENTS, AND MINERALS
 DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT
 EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT Sources: University of Washington, Texas A&M University, University of Southern Alabama What is an igneous rock (a
EESC 4701: Igneous and Metamorphic Petrology IGNEOUS ROCK CLASSIFICATION LAB 2 HANDOUT Sources: University of Washington, Texas A&M University, University of Southern Alabama What is an igneous rock (a
305 ATOMS, ELEMENTS, AND MINERALS
 DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
Nitrogen speciation in upper mantle fluids and the origin of Earth s nitrogen-rich atmosphere
 Supporting Online Material for Nitrogen speciation in upper mantle fluids and the origin of Earth s nitrogen-rich atmosphere Sami Mikhail & Dimitri Sverjensky S1. Supplementary discussion S1.1 The selection
Supporting Online Material for Nitrogen speciation in upper mantle fluids and the origin of Earth s nitrogen-rich atmosphere Sami Mikhail & Dimitri Sverjensky S1. Supplementary discussion S1.1 The selection
Icarus 213 (2011) Contents lists available at ScienceDirect. Icarus. journal homepage:
 Icarus 213 (2011) 404 422 Contents lists available at ScienceDirect Icarus journal homepage: www.elsevier.com/locate/icarus A new systematic approach using the Modified Gaussian Model: Insight for the
Icarus 213 (2011) 404 422 Contents lists available at ScienceDirect Icarus journal homepage: www.elsevier.com/locate/icarus A new systematic approach using the Modified Gaussian Model: Insight for the
Nonlinear spectral mixing: Quantitative analysis of laboratory mineral mixtures
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 109,, doi:10.1029/2003je002179, 2004 Nonlinear spectral mixing: Quantitative analysis of laboratory mineral mixtures F. Poulet and S. Erard Institut d Astrophysique
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 109,, doi:10.1029/2003je002179, 2004 Nonlinear spectral mixing: Quantitative analysis of laboratory mineral mixtures F. Poulet and S. Erard Institut d Astrophysique
305 ATOMS, ELEMENTS, AND MINERALS
 DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
DATE DUE: Name: Instructor: Ms. Terry J. Boroughs Geology 305 ATOMS, ELEMENTS, AND MINERALS Instructions: Read each question carefully before selecting the BEST answer. Use GEOLOGIC VOCABULARY where APPLICABLE!
Statistical Analysis of the Links among Lunar Mare Soil Mineralogy, Chemistry, and Reflectance Spectra
 Icarus 155, 285 298 (2002) doi:10.1006/icar.2001.6749, available online at http://www.idealibrary.com on Statistical Analysis of the Links among Lunar Mare Soil Mineralogy, Chemistry, and Reflectance Spectra
Icarus 155, 285 298 (2002) doi:10.1006/icar.2001.6749, available online at http://www.idealibrary.com on Statistical Analysis of the Links among Lunar Mare Soil Mineralogy, Chemistry, and Reflectance Spectra
Classification of Igneous Rocks
 Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
Classification of Igneous Rocks Textures: Glassy- no crystals formed Aphanitic- crystals too small to see by eye Phaneritic- can see the constituent minerals Fine grained- < 1 mm diameter Medium grained-
IV. Governador Valadares clinopyroxenite, 158 grams find
 IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
IV. Governador Valadares clinopyroxenite, 158 grams find Figure IV-1. Photograph of Governador Valadares (158 grams) from Dr. Fernanda Ferrucci via Dr. Giuseppe Cavarretta. Photo taken by L. Spinozzi.
ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION
 Copyright JCPDS - International Centre for Diffraction Data 2003, Advances in X-ray Analysis, Volume 46. 204 ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION Robin M. Gonzalez,
Copyright JCPDS - International Centre for Diffraction Data 2003, Advances in X-ray Analysis, Volume 46. 204 ANALYSIS OF GEOLOGIC MATERIALS USING RIETVELD QUANTIATIVE X-RAY DIFFRACTION Robin M. Gonzalez,
Astrochemistry (2) Interstellar extinction. Measurement of the reddening
 Measurement of the reddening The reddening of stellar colours casts light on the properties of interstellar dust Astrochemistry (2) Planets and Astrobiology (2016-2017) G. Vladilo The reddening is measured
Measurement of the reddening The reddening of stellar colours casts light on the properties of interstellar dust Astrochemistry (2) Planets and Astrobiology (2016-2017) G. Vladilo The reddening is measured
Lecture 6 - Igneous Rocks and Volcanoes
 Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Lecture 6 - Igneous Rocks and Volcanoes Learning objectives Understand and be able to predict where and why magma will be forming at different tectonic settings Understand the factors controlling magma
Matter and Minerals. Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more).
 1 2 Matter and Minerals Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more). continued... 3 4 5 6 7 8 9 10 11 12 13 14 Also crystalline,
1 2 Matter and Minerals Earth 9 th edition Chapter 3 Minerals: summary in haiku form "Mineral" defined: natural, inorganic, solid (and two more). continued... 3 4 5 6 7 8 9 10 11 12 13 14 Also crystalline,
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS
 PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
PLATE TECTONICS, VOLCANISM AND IGNEOUS ROCKS PLATE TECTONICS TO IGNEOUS ROCKS Internal Heat Seafloor Spreading/Plate Tectonics Volcanism Plate Boundary Intra-plate (hot spot) Divergent Convergent Igneous
Estimating the water content of hydrated minerals using reflectance spectroscopy II. Effects of particle size
 Icarus 189 (2007) 574 588 www.elsevier.com/locate/icarus Estimating the water content of hydrated minerals using reflectance spectroscopy II. Effects of particle size Ralph E. Milliken, John F. Mustard
Icarus 189 (2007) 574 588 www.elsevier.com/locate/icarus Estimating the water content of hydrated minerals using reflectance spectroscopy II. Effects of particle size Ralph E. Milliken, John F. Mustard
EFFECTS OF LASER SPACE WEATHERING ON DERIVED IRON OXIDE CONTENT IN SAN CARLOS OLIVINE, PYROXENE, AND ANORTHOSITE
 EFFECTS OF LASER SPACE WEATHERING ON DERIVED IRON OXIDE CONTENT IN SAN CARLOS OLIVINE, PYROXENE, AND ANORTHOSITE Logan K. Magad-Weiss Department of Geology and Geophysics University of Hawai`i at Mānoa
EFFECTS OF LASER SPACE WEATHERING ON DERIVED IRON OXIDE CONTENT IN SAN CARLOS OLIVINE, PYROXENE, AND ANORTHOSITE Logan K. Magad-Weiss Department of Geology and Geophysics University of Hawai`i at Mānoa
Rocks: Materials of the Solid Earth
 1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
1 Rocks: Materials of the Solid Earth Presentation modified from: Instructor Resource Center on CD-ROM, Foundations of Earth Science,, 4 th Edition, Lutgens/Tarbuck, Rock Cycle Igneous Rocks Today 2 Rock
12 Chemistry (Mg,Fe) 2 SiO 4 Olivine is forms what is called an isomorphous solid solution series that ranges between two end members: Forsterite Mg
 11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
11 Olivine Structure Olivine is a common green or brown rock forming minerals which consists of a solid-solution series between Forsterite (Fo) and Fayalite (Fa). It is an orthorhombic orthosilicate with
Practice Test Rocks and Minerals. Name. Page 1
 Name Practice Test Rocks and Minerals 1. Which rock would be the best source of the mineral garnet? A) basalt B) limestone C) schist D) slate 2. Which mineral is mined for its iron content? A) hematite
Name Practice Test Rocks and Minerals 1. Which rock would be the best source of the mineral garnet? A) basalt B) limestone C) schist D) slate 2. Which mineral is mined for its iron content? A) hematite
ESR APPLICATIONS TO METEORITE SAMPLES. Chihiro y AMANAKA, Shin TOYODA and Motoji IKEY A
 Proc. NIPR Symp. Antarct. Meteorites, 6, 417-422, 1993 ESR APPLICATIONS TO METEORITE SAMPLES Chihiro y AMANAKA, Shin TOYODA and Motoji IKEY A Osaka University, Department of Earth and Space Science, J
Proc. NIPR Symp. Antarct. Meteorites, 6, 417-422, 1993 ESR APPLICATIONS TO METEORITE SAMPLES Chihiro y AMANAKA, Shin TOYODA and Motoji IKEY A Osaka University, Department of Earth and Space Science, J
The Main Point. How do light and matter interact? Lecture #7: Radiation and Spectra II. How is light absorbed and emitted?
 Lecture #7: Radiation and Spectra II How is light absorbed and emitted? Models of Atomic Structure. Formation of Spectral Lines. Doppler Shift. Applications in Solar System Studies Detecting gaseous phases
Lecture #7: Radiation and Spectra II How is light absorbed and emitted? Models of Atomic Structure. Formation of Spectral Lines. Doppler Shift. Applications in Solar System Studies Detecting gaseous phases
Martian Meteorites 1
 Martian Meteorites 1 The SNCs How do we know the SNCs are from Mars? [Taylor & McLennan, 2009] 2 The SNCs Abundances and isotopic compositions of gases trapped in impact melt glasses match those measured
Martian Meteorites 1 The SNCs How do we know the SNCs are from Mars? [Taylor & McLennan, 2009] 2 The SNCs Abundances and isotopic compositions of gases trapped in impact melt glasses match those measured
Igneous Rock. Magma Chamber Large pool of magma in the lithosphere
 Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Igneous Rock Magma Molten rock under the surface Temperature = 600 o 1400 o C Magma Chamber Large pool of magma in the lithosphere Magma chamber - most all magma consists of silicon and oxygen (silicate)
Supplementary Materials Detail Petrographic Description
 Supplementary Materials Detail Petrographic Description Figure S1 shows the results of a thin section analysis of all samples from Ijen Crater. All samples had a porphyritic texture composed of plagioclase,
Supplementary Materials Detail Petrographic Description Figure S1 shows the results of a thin section analysis of all samples from Ijen Crater. All samples had a porphyritic texture composed of plagioclase,
Final chemical results from the Mars Pathfinder alpha proton X-ray spectrometer
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. E12, 8096, doi:10.1029/2002je002019, 2003 Final chemical results from the Mars Pathfinder alpha proton X-ray spectrometer C. Nicole Foley, 1,2,3,4 Thanasis
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. E12, 8096, doi:10.1029/2002je002019, 2003 Final chemical results from the Mars Pathfinder alpha proton X-ray spectrometer C. Nicole Foley, 1,2,3,4 Thanasis
An Adulterated Martian Meteorite Written by G. Jeffrey Taylor Hawai'i Institute of Geophysics and Planetology
 1 of 5 posted July 20, 1999 An Adulterated Martian Meteorite Written by G. Jeffrey Taylor Hawai'i Institute of Geophysics and Planetology Martian meteorite Elephant Moraine (EET) A79001 is composed of
1 of 5 posted July 20, 1999 An Adulterated Martian Meteorite Written by G. Jeffrey Taylor Hawai'i Institute of Geophysics and Planetology Martian meteorite Elephant Moraine (EET) A79001 is composed of
Textures of Igneous Rocks
 Page 1 of 6 EENS 212 Prof. Stephen A. Nelson Petrology Tulane University This document last updated on 12-Feb-2004 Introduction to Igneous Rocks An igneous rock is any crystalline or glassy rock that forms
Page 1 of 6 EENS 212 Prof. Stephen A. Nelson Petrology Tulane University This document last updated on 12-Feb-2004 Introduction to Igneous Rocks An igneous rock is any crystalline or glassy rock that forms
Icarus 210 (2010) Contents lists available at ScienceDirect. Icarus. journal homepage:
 Icarus 210 (2010) 8 13 Contents lists available at ScienceDirect Icarus journal homepage: www.elsevier.com/locate/icarus Deconvolution of lunar olivine reflectance spectra: Implications for remote compositional
Icarus 210 (2010) 8 13 Contents lists available at ScienceDirect Icarus journal homepage: www.elsevier.com/locate/icarus Deconvolution of lunar olivine reflectance spectra: Implications for remote compositional
Thermal infrared emission spectroscopy of titanium-enriched pyroxenes
 JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. E8, 5095, doi:10.1029/2003je002052, 2003 Thermal infrared emission spectroscopy of titanium-enriched pyroxenes V. E. Hamilton Hawai i Institute of Geophysics
JOURNAL OF GEOPHYSICAL RESEARCH, VOL. 108, NO. E8, 5095, doi:10.1029/2003je002052, 2003 Thermal infrared emission spectroscopy of titanium-enriched pyroxenes V. E. Hamilton Hawai i Institute of Geophysics
Field Trips. Field Trips
 Field Trips Saturday field trips have been scheduled October 9, October 23 and December 4 Last all day (9:00 AM to 4:00 PM) Bus transportation provided from campus Joint with GG101 laboratory, GG101 Section
Field Trips Saturday field trips have been scheduled October 9, October 23 and December 4 Last all day (9:00 AM to 4:00 PM) Bus transportation provided from campus Joint with GG101 laboratory, GG101 Section
