Exam Prep Questions. (a) i. State the function of each of the following parts of the flower. Petal - Anther - (2 marks) Develops into a seed,
|
|
|
- Beatrix Fox
- 5 years ago
- Views:
Transcription
1 Exam Prep Questions 1. Fig. 1.1 shows a cross-section through a flower. (IGCSE 2011) (a) i. State the function of each of the following parts of the flower. Petal - Anther - (2 marks) ii. Name the part of the flower that, Develops into a seed, Develops into a fruit, (2 marks) (b) Flowers are involved in sexual reproduction. Complete Table 1.1 below to show whether each statement is true for asexual reproduction, sexual reproduction, both or neither. Use a tick ( ) for a correct statement and a cross ( ) for an incorrect statement. You must write either a tick or a cross in each space in the table. The first statement has been completed for you. Table 1.1 Statement Asexual Reproduction Sexual Reproduction Gametes are involved New individuals are produced A zygote is produced Offspring are always genetically identical
2 2. Fig 2.1 shows a pin in front of a plane mirror. (O Level 2012) A ray of light is incident on the mirror as shown. On Fig 2.1, Fig 2.1 (a) Draw the normal at the point where the ray is incident on the mirror, (1 mark) (b) Draw the reflected ray, (1 mark) (c) Mark the position of the image of the pin with an X. (1 mark) 3. Hydrocarbons are compounds which contain only the elements hydrogen and carbon. (a) The simplest hydrocarbon is methane (CH 4), which is an important fuel. (i) State 1 natural source of methane. (1 mark) (ii) Complete the following graphical formula of a methane molecule. (2 marks)
3 (iii) Carbon dioxide and carbon monoxide are compounds released into the atmosphere during combustion. Describe 1 environmental disadvantage of each compound. Carbon Dioxide: Carbon Monoxide: (2 marks)
4 4. When a light ray passes through a rectangular glass block, the ray is displaced from its original path onto a new path. This is shown in Fig 3.1. (IGCSE 2011/62) A student is doing an experiment to find out how the value of the displacement distance, d, varies with i, the angle of incidence. He places a glass block on a sheet of paper and draws the outline of the block on it. He Shines a narrow beam of light through the glass block He marks the path of the incident ray on the paper He measures i, the angle of incidence, and records the value in Table 2.1. He marks the new path of the light ray on the opposite side of the block. H removes the glass block from the paper and extends the line showing the original path of the light ray. He measures the displacement distance, d, shown in Fig. 3.1, to the nearest millimetre and records it in Table 2.1. He repeats the experiment, each time using a different angle of incidence, and records the results in Table 2.1.
5 Table 2.1 Experiment Number Angle of incidence, i/ Displacement d/mm (a) Fig 3.2 shows his diagram for experiment. (i) (ii) On fig 3.2, measure i, the angle of incidence, to the nearest degree. Record it in Table 3.1. (1 mark) On Fig. 3.2, measure the displacement distance, d, in millimetres to the nearest millimetre, and record it in Table 3.1. (1 mark)
6 (iii) On Fig. 3.2, measure the length, l, and the width, w, of the glass block in millimetres to the nearest millimetre. l = mm w = mm (2 marks) (b) (i) On the graph grid provided below, plot a graph of the displacement distance, d, (vertical axis) in millimetres against the angle of incidence, I, for each of the experiements in Table 2.1. Use values of i from 0 to 90, and values of d from 0-80 mm. Draw a smooth curve through the points and extend the line to point i = 90. (ii) Use your graph to find the value of the displacement distance, d 90, when the angle of incidence is 90. Show how you do this on the graph. d 90 = mm (2 marks)
7 5. (a) A student was studying cells under a microscope. She prepared cells from onion epidermis, placed them onto a slide and stained them using iodine solution. The student wanted to measure the average length of epidermis cells. She placed a ruler with a millimetre scale on the stage of the microscope to find the diameter of the field of view. She looked down the microscope and saw the field of view in Fig 4.1. (i) (ii) How wide is the field of view of the microscope? mm (1 mark) The student removed the ruler and placed her slide under the microscope. She counted 15 cells end to end across the field of view. Use your answer to (a)(i) to calculate the average length of 1 onion cell. Show your work and don t forget your units. (2 marks) (b) A group of cells is shown below in Fig 4.2.
8 (i) Make a large drawing of cell A in the space provided below. Label the nucleus and cell wall. (1 mark) (ii) Measure the length of cell A on your diagram at the longest part. See Fig 4.3 which shows the longest part. length of cell A on my diagram: mm (iii) using the value for the average cell length from (a)(ii) and the length of cell A on your diagram, calculate the magnification of your drawing. Show your work. Magnification = (3 marks)
9 6. When a reactive metal is added to a dilute acid, the metal reacts and dissolves and a gas is given off. (IGCSE, 2012/21) (i) Name one reactive metal that must not be added to a dilute acid.(1mark) Metal: Explain why this metal must not be added to the dilute acid below: (ii) Fig 5.1 shows how a student tested the gas given off when magnesium was added to dilute hydrochloric acid. Fig 5.1 State and explain what the student observed when he carried out this test. Observation: Explanation: (2 marks)
10 7. Some types of fireworks are made by filling cardboard tubes with firework mixture. Firework mixture is made from several solid substances, which have been powdered and mixed together. (IGCSE 2012/21) Fig 6.1 Shows a typical firework. When the paper fuse is lit, exothermic combustion reactions occur inside the firework. (a) (i) State TWO forms of energy that are released when the firework mixture reacts (b) Some firework mixtures contain aluminum which is oxidized when the firework is lit. Table 6.1 shows the numbers of protons and electrons in four particles, A, B, C and D, which are involved in the oxidation of aluminum.
11 (i) Atoms of the element aluminum have the proton number 13. State and explain which particle, B, or D, in Table 6.1 is an atom of aluminum. Particle: Explanation: (ii) State and explain which two particles in Table 6.1 could be found bonded together in aluminum oxide. Particles and Explanation: (c) Firework mixtures contain the compound potassium perchlorate, KClO 4 When potassium perchlorate is heated, a colourless gas is given off which re-lights a glowing splint. (i) State the name of the gas: 8. A student drew the following diagram to show the transmission of light through fibre optic cable (optical fibre). Assess the accuracy of the diagram.
12 9. The Alberta Energy website states Every two weeks Alberta produces enough coal to fill the Rogers Centre in Toronto. The Rogers Centre is a Baseball/Football stadium. i. Assuming the volume of the Rogers Centre is 1.6 million m 3. If all the coal produced was burned, and remember that coal is mostly carbon, in the space below estimate the mass (in tonnes) of carbon dioxide produced in a year. You can assume that a m 3 of coal has a mass of about 1 tonne (1000kg) (5 marks) (ii) What other product(s) are produced as the result of the burning of coal? (iii) What type of reaction is it when carbon monoxide is also produced when coal is burnt?
13 10. The diagram shows scientific principles that can be used in communication. What is the name of the scientific principle that Z represents? (a) Refraction (b) Binary transmission (c) Total Internal Reflection (d) Absorption 11. When a TV interviewer in Sydney Australia conducts a live interview with a person in New York, a satellite is used. The signals transmitted via the satellite travel at a speed of 3 x 10 8 m/s between the two cities. A person watching the interview observes a noticeable delay in the communication process. When a person in Sydney talks to a person in New York by phone, the signals pass through optical fibres between the two cities, traveling at a speed of 2 x 10 8 m/s, and there is no noticeable delay in the communication process. Why is the delay noticeable in the TV interview but not in the phone conversation? (a) The phone signals travel more efficiently through optical fibres than electromagnetic waves do through a vacuum. (b) Computers in a phone system make the process faster than satellite systems. (c) Satellites move so fast that the TV signals take time to catch up with them (d) The phone signal travels less distance than the TV signals 12. A heart medication has a coating which breaks down in acidic conditions. In which part of the digestive system will the coating break down? (a) mouth (b) stomach (c) small intestine (d) large intestine
14 13. Which of the following instructions always applies when working with household pesticides? (a) Do not use indoors (b) Wash hands after use (c) Store in a cool dark place (d) Keep away from naked flames 14. What feature of electromagnetic waves allow them to be used for communication? (a) They can be used in optical fibres (b) They can travel at very high speeds (c) They can be detected using a simple antenna (d) They can pass through the atmosphere without interference 15. Which statement makes a correct comparison between the use of an optical fibre and a copper wire for communication? (a) Both the optical fibre and copper wire transmit signals using electromagnetic waves (b) It is cheaper to send the same amount of data through a copper wire than through an optical fibre (c) More data can be transmitted per second through an optical fibre than through a copper wire (d) Electromagnetic waves are emitted from the optical fibre making the data less secure than data transmitted through the copper wire.
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education
 www.xtremepapers.com UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6608480670* CO-ORDINATED SCIENCES 0654/62 Paper 6 Alternative to Practical
www.xtremepapers.com UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education *6608480670* CO-ORDINATED SCIENCES 0654/62 Paper 6 Alternative to Practical
CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education PHYSICAL SCIENCE 0652/06
 www.xtremepapers.com Centre Number Candidate Number Name CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education PHYSICAL SCIENCE 0652/06 Paper 6 Alternative to Practical
www.xtremepapers.com Centre Number Candidate Number Name CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education PHYSICAL SCIENCE 0652/06 Paper 6 Alternative to Practical
Answer ALL questions.
 Answer ALL questions. 01. The three states of matter are solid, liquid and gas. The diagram shows how the particles are arranged in each of these states. (a) Use words from the box to show the changes
Answer ALL questions. 01. The three states of matter are solid, liquid and gas. The diagram shows how the particles are arranged in each of these states. (a) Use words from the box to show the changes
Paper Reference. Sample Assessment Material Time: 2 hours
 Centre No. Candidate No. Paper Reference(s) 4CH0/1C Edexcel IGCSE Chemistry Chemistry Paper 1 Sample Assessment Material Time: 2 hours Materials required for examination Nil Items included with question
Centre No. Candidate No. Paper Reference(s) 4CH0/1C Edexcel IGCSE Chemistry Chemistry Paper 1 Sample Assessment Material Time: 2 hours Materials required for examination Nil Items included with question
Q1. As the world population increases there is a greater demand for fertilisers.
 Q1. As the world population increases there is a greater demand for fertilisers. (a) Explain what fertilisers are used for............. (b) The amount of nitrogen in a fertiliser is important. How many
Q1. As the world population increases there is a greater demand for fertilisers. (a) Explain what fertilisers are used for............. (b) The amount of nitrogen in a fertiliser is important. How many
UNIT 2: CHEMICAL BONDING, APPLICATION OF CHEMICAL REACTIONS AND ORGANIC CHEMISTRY FOUNDATION TIER SAMPLE ASSESSMENT MATERIALS
 GCSE CHEMISTRY Sample Assessment Materials 71 Surname Other Names Centre Number Candidate Number GCSE CHEMISTRY UNIT 2: CHEMICAL BONDING, APPLICATION OF CHEMICAL REACTIONS AND ORGANIC CHEMISTRY FOUNDATION
GCSE CHEMISTRY Sample Assessment Materials 71 Surname Other Names Centre Number Candidate Number GCSE CHEMISTRY UNIT 2: CHEMICAL BONDING, APPLICATION OF CHEMICAL REACTIONS AND ORGANIC CHEMISTRY FOUNDATION
Q1. The diagram shows the apparatus for an experiment. Hydrated copper sulphate crystals were heated. They became anhydrous copper sulphate.
 Q1. The diagram shows the apparatus for an experiment. Hydrated copper sulphate crystals were heated. They became anhydrous copper sulphate. (a) Name a suitable piece of equipment to heat tube A.... (b)
Q1. The diagram shows the apparatus for an experiment. Hydrated copper sulphate crystals were heated. They became anhydrous copper sulphate. (a) Name a suitable piece of equipment to heat tube A.... (b)
Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2]
![Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2] Suggest one advantage and one disadvantage of using butane compared to charcoal as a source of heat [2]](/thumbs/78/78230619.jpg) High Demand Questions QUESTIONSHEET 1 A gas barbecue uses butane (C 4 H 10 ) as a fuel. The butane is stored as a liquid in a container. When the burner is turned on, the liquid turns to a gas and is lit
High Demand Questions QUESTIONSHEET 1 A gas barbecue uses butane (C 4 H 10 ) as a fuel. The butane is stored as a liquid in a container. When the burner is turned on, the liquid turns to a gas and is lit
Year 10 Revision. Atomic Structure C minutes. 75 marks. Page 1 of 28
 Year 0 Revision Atomic Structure C.-5 75 minutes 75 marks Page of 28 Q. A substance made of only one type of atom is called an element. The chemical symbols and positions of six elements in the periodic
Year 0 Revision Atomic Structure C.-5 75 minutes 75 marks Page of 28 Q. A substance made of only one type of atom is called an element. The chemical symbols and positions of six elements in the periodic
London Examinations IGCSE
 Centre No. Candidate No. Surname Signature Initial(s) Paper Reference(s) 4335/2H London Examinations IGCSE Chemistry Paper 2H Higher Tier Wednesday 16 May 2007 Morning Time: 2 hours Materials required
Centre No. Candidate No. Surname Signature Initial(s) Paper Reference(s) 4335/2H London Examinations IGCSE Chemistry Paper 2H Higher Tier Wednesday 16 May 2007 Morning Time: 2 hours Materials required
London Examinations IGCSE
 Centre No. Candidate No. Paper Reference(s) 4335/1F London Examinations IGCSE Chemistry Paper 1F Foundation Tier Wednesday 17 June 2009 Morning Time: 1 hour 30 minutes Materials required for examination
Centre No. Candidate No. Paper Reference(s) 4335/1F London Examinations IGCSE Chemistry Paper 1F Foundation Tier Wednesday 17 June 2009 Morning Time: 1 hour 30 minutes Materials required for examination
16+ ENTRANCE EXAMINATION
 ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2016 CHEMISTRY Time: 1 hour Candidates Name: Total marks available: 60 Questions Q1. This question is about the elements hydrogen and
ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2016 CHEMISTRY Time: 1 hour Candidates Name: Total marks available: 60 Questions Q1. This question is about the elements hydrogen and
CBSE Class X Science Board Paper 2011 (Set 1) Term II
 CBSE Class X Science Board Paper 2011 (Set 1) Term II Total time: 3 hrs Total marks: 80 General instructions: 1. The question paper comprises of two Sections, A and B. You are to attempt both the sections.
CBSE Class X Science Board Paper 2011 (Set 1) Term II Total time: 3 hrs Total marks: 80 General instructions: 1. The question paper comprises of two Sections, A and B. You are to attempt both the sections.
Heat energy change revision questions
 Name: Heat energy change revision questions Date: Time: Total marks available: 63 Total marks achieved: Q1. A student uses this apparatus to find the increase in temperature of water when methanol, CH
Name: Heat energy change revision questions Date: Time: Total marks available: 63 Total marks achieved: Q1. A student uses this apparatus to find the increase in temperature of water when methanol, CH
3 rd Year Revision. (from second year: Conservation of Mass Acids, Alkalis and Indicators)
 3 rd Year Revision (from second year: Conservation of Mass Acids, Alkalis and Indicators) To revise: Elements, compounds, mixtures. Periodic table names (eg groups, periods, transition metals, alkali metals,
3 rd Year Revision (from second year: Conservation of Mass Acids, Alkalis and Indicators) To revise: Elements, compounds, mixtures. Periodic table names (eg groups, periods, transition metals, alkali metals,
KENDRIYA VIDYALAYA SANGATHAN, HYDERABAD REGION
 KENDRIYA VIDYALAYA SANGATHAN, HYDERABAD REGION SAMPLE PAPER 01 FOR PERIODIC TEST II EXAM (2017-18) SUBJECT: SCIENCE (086) BLUE PRINT FOR HALF YEARLY EXAM: CLASS X Chapter Chemical Reactions and Equations
KENDRIYA VIDYALAYA SANGATHAN, HYDERABAD REGION SAMPLE PAPER 01 FOR PERIODIC TEST II EXAM (2017-18) SUBJECT: SCIENCE (086) BLUE PRINT FOR HALF YEARLY EXAM: CLASS X Chapter Chemical Reactions and Equations
7.1.2 Cell Functions. 104 minutes. 137 marks. Page 1 of 30
 7.1.2 Cell Functions 104 minutes 137 marks Page 1 of 30 ## Most pollen grains are transferred from one flower to another either by wind or by insects. Look at the drawings below which show pollen grains
7.1.2 Cell Functions 104 minutes 137 marks Page 1 of 30 ## Most pollen grains are transferred from one flower to another either by wind or by insects. Look at the drawings below which show pollen grains
London Examinations IGCSE
 Centre No. Candidate No. Surname Signature Initial(s) Paper Reference(s) 4437/5H London Examinations IGCSE Science (Double Award) Chemistry Paper 5H Higher Tier Monday 8 May 2006 Morning Time: 1 hour 30
Centre No. Candidate No. Surname Signature Initial(s) Paper Reference(s) 4437/5H London Examinations IGCSE Science (Double Award) Chemistry Paper 5H Higher Tier Monday 8 May 2006 Morning Time: 1 hour 30
GCSE COMBINED SCIENCE: SYNERGY
 GCSE COMBINED SCIENCE: SYNERGY Higher Tier Paper 4H H Specimen 2018 Time allowed: 1 hour 45 minutes Materials For this paper you must have: a ruler a calculator the periodic table (enclosed) the Physics
GCSE COMBINED SCIENCE: SYNERGY Higher Tier Paper 4H H Specimen 2018 Time allowed: 1 hour 45 minutes Materials For this paper you must have: a ruler a calculator the periodic table (enclosed) the Physics
London Examinations IGCSE
 Centre No. Candidate No. Surname Signature Initial(s) Paper Reference 4335/1F London Examinations IGCSE Chemistry Paper 1F Foundation Tier Monday 8 May 2006 Morning Time: 1 hour 30 minutes Materials required
Centre No. Candidate No. Surname Signature Initial(s) Paper Reference 4335/1F London Examinations IGCSE Chemistry Paper 1F Foundation Tier Monday 8 May 2006 Morning Time: 1 hour 30 minutes Materials required
The Periodic Table Question Paper 3
 The Periodic Table Question Paper 3 Level GCSE (9-1) Subject Chemistry Exam Board AQA Topic 4.1 Atomic structure and The Periodic Table Sub Topic The Periodic Table Difficulty level Bronze Level Booklet
The Periodic Table Question Paper 3 Level GCSE (9-1) Subject Chemistry Exam Board AQA Topic 4.1 Atomic structure and The Periodic Table Sub Topic The Periodic Table Difficulty level Bronze Level Booklet
Coimisiún na Scrúduithe Stáit State Examinations Commission
 2009. M 37 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION 2009 PHYSICS AND CHEMISTRY ORDINARY LEVEL MONDAY, 15 JUNE MORNING 9:30 TO 12:30 Six questions to be
2009. M 37 Coimisiún na Scrúduithe Stáit State Examinations Commission LEAVING CERTIFICATE EXAMINATION 2009 PHYSICS AND CHEMISTRY ORDINARY LEVEL MONDAY, 15 JUNE MORNING 9:30 TO 12:30 Six questions to be
Amounts of substances can be described in different ways. One mole of a substance is the relative formula mass in ...
 Q1.This question is about atoms and isotopes. (a) Atoms contain protons, neutrons and electrons. A lithium atom has the symbol Explain, in terms of sub-atomic particles, why the mass number of this lithium
Q1.This question is about atoms and isotopes. (a) Atoms contain protons, neutrons and electrons. A lithium atom has the symbol Explain, in terms of sub-atomic particles, why the mass number of this lithium
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY 5070/04
 www.xtremepapers.com Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY 5070/04 Paper 4 Alternative to Practical
www.xtremepapers.com Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS General Certificate of Education Ordinary Level CHEMISTRY 5070/04 Paper 4 Alternative to Practical
Specimen Paper. Further Additional Science Unit 2 Chemistry 3 [CODE] Time allowed 60 minutes
![Specimen Paper. Further Additional Science Unit 2 Chemistry 3 [CODE] Time allowed 60 minutes Specimen Paper. Further Additional Science Unit 2 Chemistry 3 [CODE] Time allowed 60 minutes](/thumbs/94/119883646.jpg) Centre Number Surname Candidate Number Specimen Paper For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Secondary Education Foundation Tier Question 1 Mark Further
Centre Number Surname Candidate Number Specimen Paper For Examiner s Use Other Names Candidate Signature Examiner s Initials General Certificate of Secondary Education Foundation Tier Question 1 Mark Further
Charles D Winters/Science Photo Library. How can you tell that a chemical reaction is taking place? ...
 Q1.The figure below shows magnesium burning in air. Charles D Winters/Science Photo Library (a) Look at the figure above. How can you tell that a chemical reaction is taking place? (b) Name the product
Q1.The figure below shows magnesium burning in air. Charles D Winters/Science Photo Library (a) Look at the figure above. How can you tell that a chemical reaction is taking place? (b) Name the product
1. A. Define the term rate of reaction. The measure of the amount of reactants being converted into products per unit amount of time
 Name answer key period IB topic 6 Kinetics 1. A. Define the term rate of reaction. The measure of the amount of reactants being converted into products per unit amount of time b. the reaction between C
Name answer key period IB topic 6 Kinetics 1. A. Define the term rate of reaction. The measure of the amount of reactants being converted into products per unit amount of time b. the reaction between C
Name and School: 2016 Academic Scholarship Preliminary Examination. Science. Time Allowed : One hour
 Name and School: 2016 Academic Scholarship Preliminary Examination Science Time Allowed : One hour Write your name on the question paper Write all your answers on the question paper Calculators are allowed
Name and School: 2016 Academic Scholarship Preliminary Examination Science Time Allowed : One hour Write your name on the question paper Write all your answers on the question paper Calculators are allowed
Scholarship Examination
 Write your name here Surname Other names Scholarship Examination Subject: Chemistry Time: 45 minutes You must have: Calculator Ruler Total Marks /45 Instructions Use black ink or ball-point pen. Fill in
Write your name here Surname Other names Scholarship Examination Subject: Chemistry Time: 45 minutes You must have: Calculator Ruler Total Marks /45 Instructions Use black ink or ball-point pen. Fill in
Section I: Synthesis reactions Synthesis reactions occur when two or more substances come together to form a single new substance.
 TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
TYPES OF CHEMICAL REACTIONS A Laboratory Investigation Purpose: Observe the five major types of reactions. Record observations for these reactions. Complete balanced equations for these reactions. Introduction:
Q1. Ammonia is used in the production of fertilisers. The flow diagram shows the main stages in the manufacture of ammonia.
 Q1. Ammonia is used in the production of fertilisers. The flow diagram shows the main stages in the manufacture of ammonia. Study the flow diagram and then answer the questions. (a) What is the purpose
Q1. Ammonia is used in the production of fertilisers. The flow diagram shows the main stages in the manufacture of ammonia. Study the flow diagram and then answer the questions. (a) What is the purpose
Page 2. Q1.This question is about mixtures and analysis. Which two substances are mixtures? Tick two boxes. Air. Carbon dioxide.
 Q1.This question is about mixtures and analysis. (a) Which two substances are mixtures? Tick two boxes. Air Carbon dioxide Graphite Sodium Chloride Steel (2) (b) Draw one line from each context to the
Q1.This question is about mixtures and analysis. (a) Which two substances are mixtures? Tick two boxes. Air Carbon dioxide Graphite Sodium Chloride Steel (2) (b) Draw one line from each context to the
The Atmosphere Exam questions
 The Atmosphere Exam questions 2012 - Higher What substance is formed when carbon is burned in oxygen? Give the effect of this substance on moist litmus paper. The table gives the % by volume of five gases/
The Atmosphere Exam questions 2012 - Higher What substance is formed when carbon is burned in oxygen? Give the effect of this substance on moist litmus paper. The table gives the % by volume of five gases/
C2 Revision Pack (Please keep this pack with you)
 Name: C2 Revision Pack (Please keep this pack with you) Follow all the steps below... 1) Practice all the maths and working scientifically questions PRACTICE ALL THESE QUESTIONS! Maths and Science Skills
Name: C2 Revision Pack (Please keep this pack with you) Follow all the steps below... 1) Practice all the maths and working scientifically questions PRACTICE ALL THESE QUESTIONS! Maths and Science Skills
IGCSE (9-1) Edexcel - Chemistry
 IGCSE (9-1) Edexcel - Chemistry Principles of Chemistry Chemical Formulae, Equations and Calculations NOTES 1.25: Write word equations and balanced chemical equations (including state symbols): For reactions
IGCSE (9-1) Edexcel - Chemistry Principles of Chemistry Chemical Formulae, Equations and Calculations NOTES 1.25: Write word equations and balanced chemical equations (including state symbols): For reactions
Describe how the inter-conversion of solids, liquids and gases are achieved and recall names used for these inter-conversions
 Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Understand the arrangements, movements and energy of the particle in each of the 3 states of matter : solid, liquid and gas Describe how the inter-conversion of solids, liquids and gases are achieved and
Topic: Atoms and the Periodic Table
 Topic: Atoms and the Periodic Table Atoms the basic building blocks of all substances 92 different naturally-occurring types of atom the smallest amount of any element we can have Fun Facts: One cubic
Topic: Atoms and the Periodic Table Atoms the basic building blocks of all substances 92 different naturally-occurring types of atom the smallest amount of any element we can have Fun Facts: One cubic
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *7007324309* CO-ORDINATED SCIENCES 0654/62 Paper 6 Alternative to Practical May/June 2017 1 hour
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *7007324309* CO-ORDINATED SCIENCES 0654/62 Paper 6 Alternative to Practical May/June 2017 1 hour
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *1137125136* PHYSICS 0625/33 Paper 3 Theory (Core) May/June 2016 1 hour 15 minutes Candidates answer
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *1137125136* PHYSICS 0625/33 Paper 3 Theory (Core) May/June 2016 1 hour 15 minutes Candidates answer
Science 1206 Chemistry Unit Sample Final Exam Key
 Science 1206 Chemistry Unit Sample Final Exam Key 1. A gas is proved to be carbon dioxide if: (a) a glowing splint bursts into flames in the gas. (b) a wet piece of red litmus goes blue in the gas. (c)
Science 1206 Chemistry Unit Sample Final Exam Key 1. A gas is proved to be carbon dioxide if: (a) a glowing splint bursts into flames in the gas. (b) a wet piece of red litmus goes blue in the gas. (c)
Structure and Bonding
 Structure and Bonding Foundation revision questions Name: Class: Date: Time: 66 minutes Marks: 65 marks Comments: Page of 25 The diagram represents a carbon atom. (a) Use words from the box to answer the
Structure and Bonding Foundation revision questions Name: Class: Date: Time: 66 minutes Marks: 65 marks Comments: Page of 25 The diagram represents a carbon atom. (a) Use words from the box to answer the
Year 10 Chemistry. Practice questions. Topics
 Year 10 Chemistry Practice questions Topics 1 Group 1 2 Group 7 3 Reactivity series 4 Air and Water 5 Rates of reaction 6 Electrolysis 7 Acids, Alkali and Salts Objective: Evaluate group 1 & 7 reactivity
Year 10 Chemistry Practice questions Topics 1 Group 1 2 Group 7 3 Reactivity series 4 Air and Water 5 Rates of reaction 6 Electrolysis 7 Acids, Alkali and Salts Objective: Evaluate group 1 & 7 reactivity
Page 2. Q1.Carbon dioxide is produced when copper carbonate is heated.
 Q1.Carbon dioxide is produced when copper carbonate is heated. A student investigated heating copper carbonate. The student used the apparatus to measure how long it took for carbon dioxide to be produced.
Q1.Carbon dioxide is produced when copper carbonate is heated. A student investigated heating copper carbonate. The student used the apparatus to measure how long it took for carbon dioxide to be produced.
Draw one line from each solution to the ph value of the solution. Solution ph value of the solution
 1 The ph scale is a measure of the acidity or alkalinity of a solution. (a) Draw one line from each solution to the ph value of the solution. Solution ph value of the solution 5 Acid 7 9 Neutral 11 13
1 The ph scale is a measure of the acidity or alkalinity of a solution. (a) Draw one line from each solution to the ph value of the solution. Solution ph value of the solution 5 Acid 7 9 Neutral 11 13
What do each of the hazard warning symbols below mean?
 Question 1 What do each of the hazard warning symbols below mean? Question 2 Draw a line to match the name of the separation technique to the type of mixture it is used to separate. filtration Used to
Question 1 What do each of the hazard warning symbols below mean? Question 2 Draw a line to match the name of the separation technique to the type of mixture it is used to separate. filtration Used to
Lower Sixth Chemistry. Sample Entrance Examination
 Lower Sixth Chemistry Sample Entrance Examination Time allowed: 60 minutes Name: Total : 60 Marks INSTRUCTIONS : Answer all questions Answers should be written in the spaces provided Dictionaries or reference
Lower Sixth Chemistry Sample Entrance Examination Time allowed: 60 minutes Name: Total : 60 Marks INSTRUCTIONS : Answer all questions Answers should be written in the spaces provided Dictionaries or reference
1. (a) The diagrams below show the arrangement of atoms or molecules in five different substances A, B, C, D and E.
 Level 7 Chemistry Questions 1. (a) The diagrams below show the arrangement of atoms or molecules in five different substances A, B, C, D and E. Each of the circles, and represents an atom of a different
Level 7 Chemistry Questions 1. (a) The diagrams below show the arrangement of atoms or molecules in five different substances A, B, C, D and E. Each of the circles, and represents an atom of a different
glossary 6 of. boxes with and units. question. the syllabus. to see if. nature. Day. Teacher. Pre readings. Topic. Mr Stocker.
 This revision booklet contains questions from the Coordinated examinations from 2007 2011. At the end of each question you will find a code corresponding to the year that the question came from. example
This revision booklet contains questions from the Coordinated examinations from 2007 2011. At the end of each question you will find a code corresponding to the year that the question came from. example
YEAR 8 - SCIENCE REVISION. Final Exam
 YEAR 8 - SCIENCE REVISION Final Exam 1. Give a comparative study of Copernicus model of solar system and Kepler s model of solar system. 2. Why do we follow Kepler s model today? 3. The figure below shows
YEAR 8 - SCIENCE REVISION Final Exam 1. Give a comparative study of Copernicus model of solar system and Kepler s model of solar system. 2. Why do we follow Kepler s model today? 3. The figure below shows
The names and formulae of three hydrocarbons in the same homologous series are:... (1) Which homologous series contains ethane, propane and butane?
 Q1. This question is about hydrocarbons. (a) The names and formulae of three hydrocarbons in the same homologous series are: Ethane C 2 H 6 Propane C 3 H 8 Butane C 4 H 10 The next member in the series
Q1. This question is about hydrocarbons. (a) The names and formulae of three hydrocarbons in the same homologous series are: Ethane C 2 H 6 Propane C 3 H 8 Butane C 4 H 10 The next member in the series
MARIYA INTERNATIONAL SCHOOL. Work sheet III. Term I. Level 8 Chemistry [MCQ] Name: CHEMICAL REACTIONS & SULFUR
![MARIYA INTERNATIONAL SCHOOL. Work sheet III. Term I. Level 8 Chemistry [MCQ] Name: CHEMICAL REACTIONS & SULFUR MARIYA INTERNATIONAL SCHOOL. Work sheet III. Term I. Level 8 Chemistry [MCQ] Name: CHEMICAL REACTIONS & SULFUR](/thumbs/95/123248955.jpg) MARIYA INTERNATIONAL SCHOOL Work sheet III Term I Level 8 Chemistry [MCQ] Name: CHEMICAL REACTIONS & SULFUR 1. A steel works and a chemical works are built near to a city. The limestone buildings in the
MARIYA INTERNATIONAL SCHOOL Work sheet III Term I Level 8 Chemistry [MCQ] Name: CHEMICAL REACTIONS & SULFUR 1. A steel works and a chemical works are built near to a city. The limestone buildings in the
Class X. Exercises solution
 Exercises solution Question 1: Which of the statements about the reaction below are incorrect? Lead is getting reduced. Carbon dioxide is getting oxidised. Carbon is getting oxidised. Lead oxide is getting
Exercises solution Question 1: Which of the statements about the reaction below are incorrect? Lead is getting reduced. Carbon dioxide is getting oxidised. Carbon is getting oxidised. Lead oxide is getting
Fig. 8.1 illustrates the three measurements. air medium A. ray 1. air medium A. ray 2. air medium A. ray 3. Fig For Examiner s Use
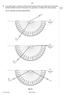 9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
9 9 9 14 8 In an optics lesson, a Physics student traces the paths of three s of light near the boundary between medium A and. The student uses a protractor to measure the various angles. Fig. 8.1 illustrates
SCIENCE. Year 10 Examination A 40 marks. Make sure that you have answered all the questions in paper 10B before you start this paper.
 NAME: SCIENCE TEACHER: (circle code) 10A SCIENCE Year 10 Examination 2013 10A 40 marks Make sure that you have answered all the questions in paper 10B before you start this paper. Time allowed for both
NAME: SCIENCE TEACHER: (circle code) 10A SCIENCE Year 10 Examination 2013 10A 40 marks Make sure that you have answered all the questions in paper 10B before you start this paper. Time allowed for both
The Fundamental Ideas in Chemistry
 The Fundamental Ideas in Chemistry Question Paper Level Subject Exam Board Unit Topic Difficulty Level Booklet GCSE Chemistry AQA C1 The Fundamental Ideas in Chemistry Silver Level Question Paper Time
The Fundamental Ideas in Chemistry Question Paper Level Subject Exam Board Unit Topic Difficulty Level Booklet GCSE Chemistry AQA C1 The Fundamental Ideas in Chemistry Silver Level Question Paper Time
Electrolysis. Specification points. Year 11 Electrolysis
 Electrolysis Specification points Year Electrolysis The process of electrolysis When an ionic compound is melted or dissolved in water, the ions are free to move about within the liquid or solution (the
Electrolysis Specification points Year Electrolysis The process of electrolysis When an ionic compound is melted or dissolved in water, the ions are free to move about within the liquid or solution (the
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 Cambridge International Examinations Cambridge International General Certificate of Secondary Education *6537478123* COMBINED SCIENCE 0653/62 Paper 6 Alternative to Practical May/June 2018 1 hour Candidates
Cambridge International Examinations Cambridge International General Certificate of Secondary Education *6537478123* COMBINED SCIENCE 0653/62 Paper 6 Alternative to Practical May/June 2018 1 hour Candidates
II, SUMMATIVE ASSESSMENT II, / SCIENCE. X / Class X. Time Allowed : 3 hours Maximum Marks : 90
 ... II, 06-7 SUMMATIVE ASSESSMENT II, 06-7 / SCIENCE X / Class X 90 Time Allowed : hours Maximum Marks : 90 4.. 4 6 0-0 6. 7 8 0-0 7. 9 4 70-70 8. 9. 4 6 K8CBTT General Instructions :. The question paper
... II, 06-7 SUMMATIVE ASSESSMENT II, 06-7 / SCIENCE X / Class X 90 Time Allowed : hours Maximum Marks : 90 4.. 4 6 0-0 6. 7 8 0-0 7. 9 4 70-70 8. 9. 4 6 K8CBTT General Instructions :. The question paper
ammonia carbon dioxide hydrogen nitrogen electrical heat solar sound (a) In air, the two most common gases are oxygen and...
 Chemistry C1 Foundation Questions Q1. Choose words from this list to complete the sentences, ammonia carbon dioxide hydrogen nitrogen electrical heat solar sound (a) In air, the two most common gases are
Chemistry C1 Foundation Questions Q1. Choose words from this list to complete the sentences, ammonia carbon dioxide hydrogen nitrogen electrical heat solar sound (a) In air, the two most common gases are
Edexcel Chemistry Checklist
 Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
Topic 1. Key concepts in chemistry Video: Developing the atomic model Describe how and why the atomic model has changed over time. Describe the difference between the plum-pudding model of the atom and
YEAR 10- Chemistry Term 1 plan
 YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
YEAR 10- Chemistry Term 1 plan 2016-2017 Week Topic Learning outcomes 1 1. The particulate nature of matter State the distinguishing properties of solids, liquids and gases. Describe the structure of solids,
Cambridge International Examinations Cambridge Ordinary Level
 Cambridge International Examinations Cambridge Ordinary Level *9280327505* CHEMISTRY 5070/42 Paper 4 Alternative to Practical May/June 2015 1 hour Candidates answer on the Question Paper. No Additional
Cambridge International Examinations Cambridge Ordinary Level *9280327505* CHEMISTRY 5070/42 Paper 4 Alternative to Practical May/June 2015 1 hour Candidates answer on the Question Paper. No Additional
London Examinations IGCSE
 Centre No. Candidate No. Paper Reference 4 3 3 5 2 H Paper Reference(s) 4335/2H London Examinations IGCSE Chemistry Paper 2H Higher Tier Wednesday 21 May 2008 Afternoon Time: 2 hours Surname Signature
Centre No. Candidate No. Paper Reference 4 3 3 5 2 H Paper Reference(s) 4335/2H London Examinations IGCSE Chemistry Paper 2H Higher Tier Wednesday 21 May 2008 Afternoon Time: 2 hours Surname Signature
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-ORDINATED SCIENCES 0654/22 Paper 2 (Core Theory), maximum
CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education MARK SCHEME for the May/June 2013 series 0654 CO-ORDINATED SCIENCES 0654/22 Paper 2 (Core Theory), maximum
London Examinations IGCSE
 Centre No. Candidate No. Paper Reference(s) 4437/5H London Examinations IGCSE Science (Double Award) Chemistry Paper 5H Higher Tier Tuesday 16 November 2010 Afternoon Time: 1 hour 30 minutes Materials
Centre No. Candidate No. Paper Reference(s) 4437/5H London Examinations IGCSE Science (Double Award) Chemistry Paper 5H Higher Tier Tuesday 16 November 2010 Afternoon Time: 1 hour 30 minutes Materials
Methane contains atoms of two elements, combined chemically. Methane is a mixture of two different elements.
 Q1.Methane (CH 4) is used as a fuel. (a) The displayed structure of methane is: Draw a ring around a part of the displayed structure that represents a covalent bond. (b) Why is methane a compound? Tick
Q1.Methane (CH 4) is used as a fuel. (a) The displayed structure of methane is: Draw a ring around a part of the displayed structure that represents a covalent bond. (b) Why is methane a compound? Tick
Chemistry. End of Course. Student Name and Date
 Chemistry HS End of Course Student Name and Date Baltimore City Public Schools Assessments End of Course High School Chemistry 1 Which of the following statements is 3 Which electron dot diagram NOT TRUE?
Chemistry HS End of Course Student Name and Date Baltimore City Public Schools Assessments End of Course High School Chemistry 1 Which of the following statements is 3 Which electron dot diagram NOT TRUE?
Website: Page 1. Page 14»Exercise» Page 15» Question 1:
 Page 14»Exercise» Question 1: Which of the statements about the reaction below are incorrect? (a) Lead is getting reduced. (b) Carbon dioxide is getting oxidised. (c) Carbon is getting oxidised. (d) Lead
Page 14»Exercise» Question 1: Which of the statements about the reaction below are incorrect? (a) Lead is getting reduced. (b) Carbon dioxide is getting oxidised. (c) Carbon is getting oxidised. (d) Lead
Cambridge International Examinations Cambridge International General Certificate of Secondary Education
 *7023149628* Cambridge International Examinations Cambridge International General Certificate of Secondary Education COMBINED SCIENCE 0653/61 Paper 6 Alternative to Practical May/June 2015 1 hour Candidates
*7023149628* Cambridge International Examinations Cambridge International General Certificate of Secondary Education COMBINED SCIENCE 0653/61 Paper 6 Alternative to Practical May/June 2015 1 hour Candidates
Cambridge International Examinations Cambridge Ordinary Level
 Cambridge International Examinations Cambridge Ordinary Level *7522989051* CHEMISTRY 5070/42 Paper 4 Alternative to Practical May/June 2018 1 hour Candidates answer on the Question Paper. No Additional
Cambridge International Examinations Cambridge Ordinary Level *7522989051* CHEMISTRY 5070/42 Paper 4 Alternative to Practical May/June 2018 1 hour Candidates answer on the Question Paper. No Additional
HKCEE Past Paper Questions: Part 9 Rate of Reactions Part A: Multiple Choices
 HKCEE Past Paper Questions: Part 9 Rate of Reactions Part A: Multiple Choices 1. HKCEE 1996 II Q11 In an experiment, 1.6 g of sulphur are burnt completely in air to form sulphur dioxide. What volume of
HKCEE Past Paper Questions: Part 9 Rate of Reactions Part A: Multiple Choices 1. HKCEE 1996 II Q11 In an experiment, 1.6 g of sulphur are burnt completely in air to form sulphur dioxide. What volume of
 Part I Short Answer Choose a letter to fill in the blanks. Use choices as many times as you wish. Only one choice is needed per blank. All are 3 points each. 1. First set. How can you tell these apart?
Part I Short Answer Choose a letter to fill in the blanks. Use choices as many times as you wish. Only one choice is needed per blank. All are 3 points each. 1. First set. How can you tell these apart?
0654 CO-ORDINATED SCIENCES
 CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
CAMBRIDGE INTERNATIONAL EXAMINATIONS Cambridge International General Certificate of Secondary Education MARK SCHEME for the May/June 2015 series 0654 CO-ORDINATED SCIENCES 0654/31 Paper 3 (Extended Theory),
The student might demonstrate the ability to achieve this standard by: Making a chart comparing the similar functions of plant and animal cells.
 1: Life, Cell Biology: All living organisms are composed of cells, from just one to many trillions, whose details are usually visible only through a microscope. s 1.a) Cells function similarly in all living
1: Life, Cell Biology: All living organisms are composed of cells, from just one to many trillions, whose details are usually visible only through a microscope. s 1.a) Cells function similarly in all living
Chemistry. Exam Choice. Student Number PRELIMINARY COURSE EXAMINATION. Total marks 75. General Instructions
 Student Number Exam Choice 2008 PRELIMINARY COURSE EXAMINATION Chemistry Total marks 75 General Instructions Reading time 5 minutes Working time 2 hours Write using black or blue pen Draw diagrams using
Student Number Exam Choice 2008 PRELIMINARY COURSE EXAMINATION Chemistry Total marks 75 General Instructions Reading time 5 minutes Working time 2 hours Write using black or blue pen Draw diagrams using
The Material World: Stoichiometry
 9. Stoichiometry A. Calculating Atomic Masses 3 Li 6.94 Atomic number (number of protons) Atomic mass (weighted average) a. The periodic table lists atomic masses for each element. b. The atomic mass is
9. Stoichiometry A. Calculating Atomic Masses 3 Li 6.94 Atomic number (number of protons) Atomic mass (weighted average) a. The periodic table lists atomic masses for each element. b. The atomic mass is
ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION. For entry in September Chemistry. Use of a calculator is permitted.
 ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2017 Chemistry Use of a calculator is permitted Time: 1 hour Candidate s Name: Total marks available: 60 St Edward's School 16+ Entrance
ST EDWARD S OXFORD 16+ ENTRANCE EXAMINATION For entry in September 2017 Chemistry Use of a calculator is permitted Time: 1 hour Candidate s Name: Total marks available: 60 St Edward's School 16+ Entrance
Kenya Certificate of Secondary Education (K.C.S.E.)
 Name: School:. Date:... 233/1 CHEMISTRY PAPER 1 JULY /AUGUST 2011 TIME: 2 HOURS Index No. Candidate s Sign.... Kenya Certificate of Secondary Education (K.C.S.E.) Chemistry Paper 1 INSTRUCTIONS TO THE
Name: School:. Date:... 233/1 CHEMISTRY PAPER 1 JULY /AUGUST 2011 TIME: 2 HOURS Index No. Candidate s Sign.... Kenya Certificate of Secondary Education (K.C.S.E.) Chemistry Paper 1 INSTRUCTIONS TO THE
OH, is an important feedstock for the chemical industry.
 1 Methanol, CH 3 OH, is an important feedstock for the chemical industry. In the manufacture of methanol, carbon dioxide and hydrogen are reacted together in the reversible reaction shown below. CO 2 (g)
1 Methanol, CH 3 OH, is an important feedstock for the chemical industry. In the manufacture of methanol, carbon dioxide and hydrogen are reacted together in the reversible reaction shown below. CO 2 (g)
SCIENCE HIGHER LEVEL
 J.37 PRE-JUNIOR CERTIFICATE EXAMINATION, 2014 SCIENCE HIGHER LEVEL TIME: 2 HOURS INSTRUCTIONS 1. Write your name, school s name and teacher s name in the boxes provided on this page. 2. Answer all questions.
J.37 PRE-JUNIOR CERTIFICATE EXAMINATION, 2014 SCIENCE HIGHER LEVEL TIME: 2 HOURS INSTRUCTIONS 1. Write your name, school s name and teacher s name in the boxes provided on this page. 2. Answer all questions.
Physical Science
 Physical Science 556 416 LaurenHill Academy Mid year Exam January 2005 Instructions: 1. Allowed materials: Non graphing calculator, and included periodic table (found at the end of the answer booklet feel
Physical Science 556 416 LaurenHill Academy Mid year Exam January 2005 Instructions: 1. Allowed materials: Non graphing calculator, and included periodic table (found at the end of the answer booklet feel
The City School PAF Chapter
 The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: INSTRUCTIONS: Write your name, index number, section, branch/campus
The City School PAF Chapter Comprehensive Worksheet May 2016 Science Class 7 Candidate Name: Index Number: Section: Branch/Campus: Date: INSTRUCTIONS: Write your name, index number, section, branch/campus
Foundation Tier 3 6 Test
 Foundation Tier 3 6 Test ame Class 1 Blood is pumped around the body by the heart. a Write the following in the order blood passes through them after leaving the heart. veins arteries capillaries First
Foundation Tier 3 6 Test ame Class 1 Blood is pumped around the body by the heart. a Write the following in the order blood passes through them after leaving the heart. veins arteries capillaries First
1 Exothermic and endothermic reactions
 1 Exothermic and endothermic reactions 2 2 26 3 45 Exothermic and endothermic reactions Question Paper 1 Level GCSE (9-1) Subject Chemistry Exam Board AQA Topic 4.5 Energy changes Sub Topic Exothermic
1 Exothermic and endothermic reactions 2 2 26 3 45 Exothermic and endothermic reactions Question Paper 1 Level GCSE (9-1) Subject Chemistry Exam Board AQA Topic 4.5 Energy changes Sub Topic Exothermic
Q1. (a) (i) Which acid should the student add to sodium hydroxide solution to make sodium sulphate? Formula:... (1)
 Q1. (a) (i) Which acid should the student add to sodium hydroxide solution to make sodium sulphate?... acid (ii) Use the table on the Data Sheet to help you to write the formula of sodium sulphate. Formula:...
Q1. (a) (i) Which acid should the student add to sodium hydroxide solution to make sodium sulphate?... acid (ii) Use the table on the Data Sheet to help you to write the formula of sodium sulphate. Formula:...
Chemical Reactions and Equations
 Chemical Reactions and Equations Question 1: Why should a magnesium ribbon be cleaned before burning in air? Magnesium is very reactive metal. When stored it reacts with oxygen to form a layer magnesium
Chemical Reactions and Equations Question 1: Why should a magnesium ribbon be cleaned before burning in air? Magnesium is very reactive metal. When stored it reacts with oxygen to form a layer magnesium
CBSE 10th Science 2013 Unsolved Paper All India
 Perfect solution to all problems Tips, Tricks, General Knowledge, Current Affairs, Latest Sample, Previous Year, Practice Papers with solutions. CBSE 10th Science 2013 Unsolved Paper All India Buy solution:
Perfect solution to all problems Tips, Tricks, General Knowledge, Current Affairs, Latest Sample, Previous Year, Practice Papers with solutions. CBSE 10th Science 2013 Unsolved Paper All India Buy solution:
1.4 Enthalpy. What is chemical energy?
 1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
1.4 Enthalpy What is chemical energy? Chemical energy is a form of potential energy which is stored in chemical bonds. Chemical bonds are the attractive forces that bind atoms together. As a reaction takes
"In Terms Of" 1. Explain, in terms of electron configuration, why arsenic and antimony are chemically similar.
 Name: Mrs. Vandergoot "In Terms Of" Regents Chemistry 1. Explain, in terms of electron configuration, why arsenic and antimony are chemically similar. 2. Base your answer to the following question on the
Name: Mrs. Vandergoot "In Terms Of" Regents Chemistry 1. Explain, in terms of electron configuration, why arsenic and antimony are chemically similar. 2. Base your answer to the following question on the
Allotropes (Diamond and Graphite) Revision Pack (C3)
 Allotropes: Allotropes are different forms of the same element in the same physical state; the atoms are bonded differently. Carbon has allotropes: - Diamond - Graphite - Buckminsterfullerene Diamond Properties
Allotropes: Allotropes are different forms of the same element in the same physical state; the atoms are bonded differently. Carbon has allotropes: - Diamond - Graphite - Buckminsterfullerene Diamond Properties
UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CHEMISTRY
 Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CHEMISTRY 06/06 Paper 6 Alternative to Practical Candidates
Centre Number Candidate Number Name UNIVERSITY OF CAMBRIDGE INTERNATIONAL EXAMINATIONS International General Certificate of Secondary Education CHEMISTRY 06/06 Paper 6 Alternative to Practical Candidates
PHYSICS (SPECIFICATION A) Unit 10 The Synoptic Unit
 Surname Centre Number Other Names Candidate Number Leave blank Candidate Signature General Certificate of Education June 2002 Advanced Level Examination PHYSICS (SPECIFICATION A) Unit 10 The Synoptic Unit
Surname Centre Number Other Names Candidate Number Leave blank Candidate Signature General Certificate of Education June 2002 Advanced Level Examination PHYSICS (SPECIFICATION A) Unit 10 The Synoptic Unit
Paper 1. Science test. First name. Last name. School KEY STAGE TIER
 Sc KEY STAGE 3 Science test Paper 1 TIER 5 7 2003 Please read this page, but do not open the booklet until your teacher tells you to start. Write your name and the name of your school in the spaces below.
Sc KEY STAGE 3 Science test Paper 1 TIER 5 7 2003 Please read this page, but do not open the booklet until your teacher tells you to start. Write your name and the name of your school in the spaces below.
Morning Time: 1 hour 15 minutes
 ADVANCED SUBSIDIARY GCE 2850/01 CHEMISTRY (SALTERS) Chemistry for Life WEDNESDAY 4 JUNE 2008 Morning Time: 1 hour 15 minutes *CUP/T46080* Candidates answer on the question paper Additional materials (enclosed):
ADVANCED SUBSIDIARY GCE 2850/01 CHEMISTRY (SALTERS) Chemistry for Life WEDNESDAY 4 JUNE 2008 Morning Time: 1 hour 15 minutes *CUP/T46080* Candidates answer on the question paper Additional materials (enclosed):
AQA Chemistry Checklist
 Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Topic 1. Atomic structure Video: Atoms, elements, compounds, mixtures Use the names and symbols of the first 20 elements in the periodic table, the elements in Groups 1 and 7, and other elements in this
Section 1: What is a Chemical Reaction
 Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
Section 1: What is a Chemical Reaction I can describe and give examples of physical and chemical changes. I can identify reactants and products. I can explain what happens to molecules in chemical reactions
2. Label animal and plant cells. 3. Differentiate between photosynthesis and cellular respiration
 SNC2D/2DN FINAL EXAM REVIEW ANSWERS UNIT 1 Cells, Tissues and Organs 1. Describe the structure and function of cell structures and organelles. Nucleus, cytoplasm, mitochondrion, chloroplast, Golgi apparatus
SNC2D/2DN FINAL EXAM REVIEW ANSWERS UNIT 1 Cells, Tissues and Organs 1. Describe the structure and function of cell structures and organelles. Nucleus, cytoplasm, mitochondrion, chloroplast, Golgi apparatus
2.3.1 Enthalpy Changes Exam Questions
 2.3.1 Enthalpy Changes Exam Questions 1. The standard enthalpy change of formation of hexane is 199 kj mol 1. Using the axes below, show the enthalpy profile diagram for the formation of hexane. On your
2.3.1 Enthalpy Changes Exam Questions 1. The standard enthalpy change of formation of hexane is 199 kj mol 1. Using the axes below, show the enthalpy profile diagram for the formation of hexane. On your
1 What is a chemical reaction?
 1 What is a chemical reaction? Substances on Earth are being continually changed due for example to extremes of hot and cold, to wind and weather, to acid rain and to solutions of different chemicals coming
1 What is a chemical reaction? Substances on Earth are being continually changed due for example to extremes of hot and cold, to wind and weather, to acid rain and to solutions of different chemicals coming
COMMON ENTRANCE STYLE EXAMINATION AT 13+ CHEMISTRY Practice Paper 2
 COMMON ENTRANCE STYLE EXAMINATION AT 13+ CHEMISTRY Practice Paper 2 1. Underline the option which best completes each of the following: (a) A substance which only contains one sort of atom is magnesium
COMMON ENTRANCE STYLE EXAMINATION AT 13+ CHEMISTRY Practice Paper 2 1. Underline the option which best completes each of the following: (a) A substance which only contains one sort of atom is magnesium
