0', where 0' is the angle between the direction of an applied field and the water F= E. (1) obtained. of the isolated molecule.
|
|
|
- Ethel King
- 5 years ago
- Views:
Transcription
1 THE DIELECTRIC CONSTANT OF LIQUID WATER AND VARIOUS FORMS OF ICE ACCORDING TO SIGNIFICANT STRUCTURE THEORY BY MARCUS E. HOBBS,* MU SHIK JHON,t AND HENRY EYRINGt DEPARTMENTS OF CHEMISTRY, DUKE UNIVERSITY, DURHAM, NORTH CAROLINA, AND UNIVERSITY OF UTAH, SALT LAKE CITY Communicated May 27, 1966 The electrostatic problem in treating hydrogen bonding and other dipolar liquids is to relate properly the molecular properties of the liquid molecules to the fundamental electrostatic equation (e - 1)/47r = P/E. Here E is the dielectric constant of the liquid, P the polarization of the dielectric, and E the field intensity. The solution of the, basic problem for homogeneous media with a sphere of dielectric constant e2 in a surrounding medium of dielectric constant E1 leads to the expression for the field,1 F, inside the sphere F= E. (1) 2E1+ e2 Onsager,2 3 and Kirkwood4 have called this the cavity field. In Onsager's excellent treatment of the problem of dipolar liquids, account was taken of long-range interactions between the dipole in the central cavity and the surrounding medium. In Kirkwood's approach the short-range interactions between nearest neighbors also were considered. In particular, on introduction of the internal dielectric constant, n2, and the assumption of spherical isotropically polarizable molecules, Onsager obtained e-n2) (2,E+ n2) _4 AV _2_212 (2) ( V \3kT/\ 3 / ( Here n is the refractive index appropriate to electronic and atomic polarizability, N is Avodagro's number, V is the molar volume of the liquid at temperature T, k is the Boltzmann constant, and M the value of the permanent dipole moment characteristic of the isolated molecule. In his derivation of equation (2), Onsager neglected short-range order. He assumed all angles between the molecular dipole and the applied field to be equally likely aside from the orientation energy in the local field. This postulate makes equation (2), as it stands, inapplicable to ice or water. This assumption cannot be even approximately true for a perfect single crystal of ice I. The dipole of a water molecule points from the oxygen atom to the midpoint between its two hydrogen atoms. There are six ways of selecting which two of the four neighboring hydrogen atoms in ice I are to belong to the central oxygen atom, and these six ways correspond to the water dipole pointing in the plus and minus direction of each of the three axes of an appropriately chosen rectangular coordinate system. Now if the water dipole were free to take up each of these six positions, the mean value of cos2 0', where 0' is the angle between the direction of an applied field and the water dipole, would be 1/3 as it is for free rotation and one would still arrive at equation (2). However, if ice had this sixfold degeneracy, there would be a corresponding entropy per mole of R In 6 persisting to low temperatures instead of the eu observed by Giauque,5 which, as Pauling6 points out, closely approximates an en- 31
2 32 CHEMISTRY: HOBBS, JHON, AND EYRING PROC. N. A. S. tropy of R In 6/4. Pauling's argument for 6/4 for the degeneracy proceeds as follows. Since neighboring oxygen atoms are equally likely to pre-empt the intermediate hydrogen atom, there is only a chance of (1/2)2 that a particular one of the six orientations of the water dipole be allowed. This gives the degeneracy 6/4 which is in close agreement with experiment. This degeneracy of 6/4 per water molecule allows some randomness of molecular orientation with respect to the direction of maximum polarization for a domain. The result is a decrease in the average component of the dipole moment,,u cos 0, in the direction of maximum polarization of the domain. Accordingly, the model discussed below differs from the Onsager model in one essential respect, namely, the method of obtaining the polarizability attributable to the permanent dipoles. In Onsager's model this polarizability, (T( + 2)2 is obtained by space averaging,u cos 0 over all possible values of the angle 0. This assumes that the relative population of dipole vectors is not constrained by shortrange directional interactions and leads to an incorrect value of this polarizability in cases where the dominant forces are directional short-range interactions. Kirkwood attempted to account for the short-range directional interactions by introduction of a correlation factor between a central molecule and nearest neighbors, but again it was assumed that the central dipole was free to orient in a local field, F. The model suggested by the present authors is that the Bernal and Fowler7 and Bjerrum8 proposals for the structure of water (see Fig. 1), and by implication many.~~~~~~~~~~..5 ~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~~... FIG. 1.-This model of the structure of ice was kindly supplied to us by Dr. Melvin E. Zandler. The small balls represent hydrogen atoms, while the big balls represent oxygen atoms. We see that the direction of every dipole points in the same direction-outward and down. Thus the projection of each dipole,u cos 0, along the direction of maximum polarization for such a domain, is itself.
3 VOL. 56, 1966 CHEMISTRY: HOBBS, JHON, AND EYRING 33 other hydrogen-bonding liquids and solids, be introduced immediately into the evaluation of the average dipole moment in the field direction attributable to permanent dipoles. This is done by assuming the following: (1) Solid or liquid water is made up of a mosaic of roughly bricklike domains with the dipoles in a particular domain having an average resultant moment /I cos 0 along the direction of maximum polarization for the domain while the direction of maximum polarization of neighboring domains is rotated through 1800 with respect to the first. For perfect tetrahedral bonding, cos 0 approaches its maximum value of 1, while for bent hydrogen bonds9 cos 0 is correspondingly smaller. (2) When an electric field is applied to the system, the directions of maximum polarization of domains orient in the field until all domains are either lined up with the field or against it. Further, these domains with their directions of maximum polarization oriented in the direction of the field increase in volume at the expense of domains appositively oriented until equilibrium is attained. The resulting mean dipole moment for solidlike molecules is then scos OF -ja cos0 F kt kt.u Cos Oe -M cos Oe _2 COS20F. cos OF = p cos0 F -coo F provided «1. (3) e kt kt k + e Here F is the local field. Thus for ice and the solidlike part of water the factor A2 JA~~~~~~~~~2 cos2 0 3kT in equation (2) must be replaced by ', while for the gaslike portion the factor -U-T is retained since such molecules oriented freely in the local field, F.10 (3) The interface between domains in the mosaic is made up of Bjerrum faults, vacancies, and related mismatch structures. According to the significant structure theory of liquids," the fraction of molecules having solidlike character is V8/V, where V8 is the molar volume of the solidlike structure in the liquid and V is the volume of the liquid at the temperature of observation. The remaining fraction of the molecules, (V - V8/V), is gaslike. Calculation of Dielectric Constants.-In the light of the foregoing discussion and the evidence from significant structure theory" that the fraction V,/V of the molecules of a liquid has solidlike properties and the remaining fraction (V-V-)IV has gaslike properties, we expect the dielectric constant of water to be given by (e - n2) (2E+ n2) = n4n (n + 2)2[V os A 0 + V VsT] (4) For ice, one has (E- n2) (2,E+n2) =rnm/2 cos20(n2 +2)2 (5) 3e V's kt ( 3) For convenience in actual calculations, equations (4) and (5) may be rearranged to give e 2 + _2_n+ 2 _2 1 Vs 1 E - + -y+ 67rN COS2O<(l )(77)- (i-3os + co2](6)
4 34 CHEMISTRY: HOBBS, JHON, AND EYRING PROC. N. A. S. and e=2 +S2 A2 )(n2+ 22( 1 E=2 2e 'knrn os 0'IV TI (7) The value of cos2 0 can be determined as a parameter by evaluation at a single temperature using experimental quantities for the other factors in equation (7). The small deviation of cos2 0 from unity reflects (1) any approximations in our model, (2) approximations in the value used for ;A and n, and (3) deviations from tetrahedral structures due to bending of hydrogen bonds, and finally (4) some decrease in polarization in a domain because of the degeneracy of 6/4 per molecule. As an application of equation (6), the calculated values of the dielectric constant of water are compared in Table 1 with observed values at several temperatures. In the calculation the one parameter, cos2 0, is taken as and,u as 1.84 debyes for both light and heavy water. The fact that for all forms of ice and water the values found for cos2 0 are nearly equal to unity is an extremely satisfying result. For comparison, Oster and Kirkwood4b obtained, using the Kirkwood correlation parameter, the data shown in Table 2. Judging from the data in Table 2 for the small temperature interval involved, the Kirkwood correlation parameter gives a poorer agreement with the temperature coefficient than that given by equation (6). Also, the Oster-Kirkwood model, which assumes free rotation about the hydrogen bond of the nearest neighbors of a central molecule, seems to be less appropriate for water than our model of water as a mosaic of domains. TABLE 1 COMPARISON OF CALCULATED AND OBSERVED VALUES OF THE DIELECTRIC CONSTANTS OF LIGHT AND HEAVY WATER H20 T, -K eobaa fcalc Vbc Vc n2d D20 T fobse eulc v Vc n2d a Values for 2730K and 3730K were taken from Malmborg, C. G., and A. A. Maryott, J. Ret. Nail. Bur. Std., 56, 1 (1956); and for 473 K and 573 K from Natl. Bur. Std. (U.S.), Circ. 514 (1951), p. 2. b Calculated from densities given in the American Institute of Physics Handbook (New York: McGraw-Hill Book Co., 1963), 2nd ed., pp c Values are taken from ref. 1lb. d Value at 273 K from footnote b above, pp. 6-90, and values at other temperatures were calculated from the molar refraction values of 3.66 cc using molar volumes given in the fourth column. e The values are taken from Collie, C. H., M. B. Hasted, and D. M. Ritson, Proc. Phys. Soc. (London), 60, 145 (1948). TABLE 2 DIELECTRIC CONSTANT OF WATER AS CALCULATED BY OSTER AND KIRKWOOD T, OK C-alo Cobs
5 VOL. 56, 1966 CHEMISTRY: HOBBS, JHON, AND EYRING 35 Ice I: Ice III: Ice V: Ice VI: TABLE 3 DIELECTRIC CONSTANT OF VARIous FORMS OF ICE T, OK Cobs fosla Constant used Density of Ice I = gm/cm' n2 = Density of Ice III = gm/cm n2= Density of IceV= gm/cm n2 = Density of Ice VI = gm/cm n2 = As an application of equation (7), the calculated values of the dielectric constant of various forms of ice are compared in Table 3 with observed values at several temperatures. Unfortunately, we have not sufficient experimental data available for a good comparison 223~~~~~~~~~~~~~~~~~~~~ except for ice I, 2210 which shows excellent agreement with experiment. In the calculations, the following values of cos2 0 are used for Ice I 0.90 for Ice III for Ice V for Ice VI Factors Affecting the Dipole Moment in Condensed Phases.-The factor (n2 ta2) corrects for the increase in dipole moment due to induction. The fact that for Ice VI the estimated value for cos2 0 is greater than 1 suggests that a slightly larger value of n2 Would probably be appropriate. Many solids containing polar molecules show little polarization arising from molecular dipoles in the solid state. This is to be expected if the dipoles are arranged in the solid so that their mean component along the direction of maximum polarization of a domain is small or zero. When the vapor pressure of a system gets high, the vacancies in the liquid tend to cluster like molecules in the vapor, and the gaslike part of the polarizability should 1 be multiplied by the factor An\2 - Xn HereXo aisnthe mole fraction of molen \A/ cules in clusters of n molecules having a resultant dipole moment An. Relaxation Times.-As Debye10 showed, the dielectric constant ea is a function of frequency and satisfies the equation E2 = (60 0) I +(,}2(8) 1+ Ca)2Tr2
6 36 CHEMISTRY: HOBBS, JHON, AND EYRING PROC. N. A. S. where according to absolute rate theory,'4 we write 1, kt _AGt - = -exp e RT. 2Tr h (9) Here eo and E. are the dielectric constants at low and high frequencies, and co is the circular frequency of the applied potential. The relaxation times for ice 13,250 given by Auty and Cole'5 are TH,0 = 5.30 X 10-6 e for ordinary ice and TDO 13,400 = 7.7 X 10-1 e RT for solid deuterium oxide. Using equation (9) we find kho -13,250-13,400 = 9.4 X 104 e for ordinary ice and kdo = 6.5 X 104 e for the specific rates of relaxation of light and heavy ice, respectively. ast A reasonable value for the frequency factor h e for a simple unimolecular rate constant where the entropy of activation, ASt, is small is If we divide 9.4 X 104 by 101', we obtained 9.4 X 10-9 for the fraction, f, of molecules which are situated so they can rotate from one domain into a neighboring domain better oriented with respect to the field. If, for each domain, only one molecule at a time is so situated that it can migrate to a neighboring domain, the linear dimension of the domain would be about a tenth of a micron. If more molecules can migrate at a time, the volume of the domain would be proportionately larger, and if the entropy of activation, ASt, is negative, the domain is correspondingly smaller. From this point of view the model seems reasonable. If the reaction coordinate were rotation about an axis normal to the axis of the dipole, the frequency factor would be larger for H20 than for D20 by a factor V2. The reported frequency factors are at least not inconsistent with this requirement. Manson, Cagle, and Eyring'6 have found 13,800 calories for the activation energy for the rate of growth of microcrystals of ice from supersaturated vapor up to a size where they can be seen. The saturated vapor was seeded with AgCl to nucleate the liquid drops. The rate-determining step is the growth of of one ice crystal at the expense of another. This correspondence in activation energies for crystal growth and passage between domains supports our model of the mechanism of dielectric relaxation in ice Ṁanson, Cagle, and Eyring'6 interpreted their results to show that a liquid domain of about 40 water molecules must freeze as a unit to form the bridge required in crystal growth. Jhon, Grosh, Ree, and Eyring"5 were able to explain the thermodynamic properties and viscosity of water on the assumption that it contained domains of about 46 molecules which changed as a unit from an "ice I-like" to an "ice III-like" state. These findings suggest the expected result that the domains which are hydrogen-bonded together should be much smaller in water than in ice, i.e., liquids lack long-range order. Because the domains of long-range order in water are much smaller than in ice and because of an abundance of fluidized vacancies in water, we expect that the relaxation time for water will have an f factor much inearer unity and an activation energy about 10,000 calories less than for ice. This is in accord with the value of
7 VOL. 56, 1966 CHEMISTRY: HOBBS, JHON, AND EYRING 37 r = 0.9 X reported by Collie, Hasted, and Ritson'7 for water. The relaxation time for D20 is about V2 times as long as for H20 in qualitative agreement with the findings in ice and with our expectations that the reaction coordinate will approximate a molecular rotation. Conclusions.-In this paper there are several points to be emphasized. (1) Short-range order in many solids and in hydrogen-bonded liquids prevents the reorientation of a molecular dipole by rotating locally in response to an electric field. However, only under extraordinary circumstances will there be long-range order extending through a macroscopic sample. Consequently, the result of an applied field will be to make those domains grow which are favorably oriented with respect to the field at the expense of the less favorably oriented until a steady state is achieved. (2) Because relaxation involves only molecules at the interface between domains, the relaxation time measures the size of the domains. The lack of sufficient degeneracy, as shown by the entropy, to allow local relaxation supports this point of view. (3) The lack of long-range order in liquids allows one to predict much shorter relaxation times for this state. (4) Significant structure theory by introducing the concept of solidlike and gaslike molecules leads to an explanation of the observed temperature coefficient of the dielectric constant in liquids. Debye's'9 calculation of the temperature dependence of the dielectric constant of water agrees well with experiment. It consists in assuming that each water molecule is restrained by a local potential field of about lokt. The difficulty with this explanation is that the relaxation time should be the reciprocal of a vibrational frequency which is more than an order of magnitude shorter than the experimental value. This forces us to turn to domains as the explanation of relaxation. The observed orientational entropy indicates there is too much local restriction to obtain polarizations of ice and water of the magnitude observed by rotation in situ. Domains with large electric moments would tend to line up parallel and antiparallel just as magnets juxtapose poles of opposite kinds. Our assumption that there are few domains with their direction of polarization oriented at angles intermediate between zero and 1800 is justified in this way. The presence of domains with intermediate orientation would lower the polarization and in the limit of complete randomness would lead to a factor of 1/3 multiplying 1s2/kT. This would require that the calculated Onsager field be low by just enough to compensate for this randomness. Investigators have frequently looked, unsuccessfully, for the domain structure in water which our considerations suggest may be present. The low-angle X-ray scattering in water does not differ appreciably from that found for liquid argon. A fairly broad distribution in domain volume around a mean value of about 46 molecules may explain this discrepancy. In subsequent publications, the dielectric constants of some of other well-known systems18 will be investigated using our proposed model in conjunction with the significant structure theory of liquids. One of the authors (M. S. Jhon) wishes to thank the National Science Foundation for support of this work. Dean Eyring wishes to express his appreciation for the opportunity of discussing some of the above problems with Professor Peter Debye. * Duke University. t University of Utah.
8 38 CHEMISTRY: HOBBS, JHON, AND EYRING PROC. N. A. S. 1Abraham, M., and R. Becker, The Classical Theory of Electricity and Magnetism (Hafner Publishing Company, Inc., 1949), 2nd ed, pp Onsager, L., J. Am. Chem. Soc., 58, 1486 (1936). 8 Onsager, L., and M. Dupuis, Rend. Scuola Intern. Fis. "Enrico Fermi," vol. 10, pp (a) Kirkwood, J. G., J. Chem. Phys., 7, 911 (1939); (b) Oster, G., and J. G. Kirkwood, J. Chem. Phys., 11, 175 (1943). 6 Giauque, W. F., and M. F. Ashley, Phys. Rev., 43, 81 (1933). 6 Pauling, L., J. Am. Chem. Soc., 57, 2680 (1935). 7Bernal, J. D., and R. H. Fowler, J. Chem. Phys., 1, 515 (1933). 8 Bjerrum, N., Science, 115, 385 (1952). 9 Pople, J. A., Proc. Roy. Soc. (London), A205, 163 (1951). 10 Debye, P., Polar Molecules (New York: Dover, 1945). 11 (a) Eyring, H., T. Ree, and N. Hirai, these PROCEEDINGS, 44, 683 (1958); (b) Jhon, M. S., J. Grosh, T. Ree, and H. Eyring, J. Chem. Phys., 44, 1465 (1966). 12Dorsay, N. E., Properties of Ordinary Water Substances (New York: Reinhold Publishing Corporation, 1940). 13Wilson, G. J., R. K. Chan, D. W. Davidson, and E. Whalley, private communication. 14 Eyring, H., D. Henderson, B. J. Stover, and E. M. Eyring, Statistical Mechanics and Dynamics (New York: John Wiley and Sons, 1964), p Auty, R. P., and R. H. Cole, J. Chem. Phys., 20, 1309 (1952). 16 Manson, J. E., F. W. Cagle, Jr., and H. Eyring, these PROCEEDINGS, 44, 156 (1958). 17 Collie, C. H., J. B. Hasted, and D. M. Ritson, Proc. Phys. Soc. (London), 60, 145 (1948). I8 Smyth, C. P., Dielectric Behavior and Structure (New York: McGraw-Hill Book Company, 1955). 19 Debye, P., Physik. Z., 36, 100, 193 (1935); Chem. Rev., 19, 171 (1936).
Chapter 2. Dielectric Theories
 Chapter Dielectric Theories . Dielectric Theories 1.1. Introduction Measurements of dielectric properties of materials is very important because it provide vital information regarding the material characteristics,
Chapter Dielectric Theories . Dielectric Theories 1.1. Introduction Measurements of dielectric properties of materials is very important because it provide vital information regarding the material characteristics,
INTERMOLECULAR FORCES: Polarity of Molecules. Seventh Course (General Chemistry) by Dr. Istadi
 INTERMOLECULAR FORCES: Polarity of Molecules Seventh Course (General Chemistry) by Dr. Istadi 1 Types of Intermolecular Forces The nature of the phases and their changes are due primarily to forces among
INTERMOLECULAR FORCES: Polarity of Molecules Seventh Course (General Chemistry) by Dr. Istadi 1 Types of Intermolecular Forces The nature of the phases and their changes are due primarily to forces among
The correlation between molecular motions and heat capacity in normal ice and water
 The correlation between molecular motions and heat capacity in normal ice and water Hai Bo Ke, Ping Wen *, Wei Hua Wang Institute of Physics, Chinese Academy of Sciences, Beijing 100190, P. R. China Abstract
The correlation between molecular motions and heat capacity in normal ice and water Hai Bo Ke, Ping Wen *, Wei Hua Wang Institute of Physics, Chinese Academy of Sciences, Beijing 100190, P. R. China Abstract
L(~<T) 3 N 2h2 1 - exp(-hv/kt)l (1) (2 rinkt) 3/eeV 3(8 7r2IkT) 1. 3(Sr2IkT) 1 1N/X
 THE SIGNIFICANT STRUCTURE THEORY APPLIED TO LIQUID OXYGEN BY KAK-CHOONG KIM,* WEI-CHEN Lu, TAIKYUE REE, AND HENRY EYRING DEPARTMENT OF CHEMISTRY, UNIVERSITY OF UTAH, SALT LAKE CITY Communicated January
THE SIGNIFICANT STRUCTURE THEORY APPLIED TO LIQUID OXYGEN BY KAK-CHOONG KIM,* WEI-CHEN Lu, TAIKYUE REE, AND HENRY EYRING DEPARTMENT OF CHEMISTRY, UNIVERSITY OF UTAH, SALT LAKE CITY Communicated January
The Direction of Spontaneous Change: Entropy and Free Energy
 The Direction of Spontaneous Change: Entropy and Free Energy Reading: from Petrucci, Harwood and Herring (8th edition): Required for Part 1: Sections 20-1 through 20-4. Recommended for Part 1: Sections
The Direction of Spontaneous Change: Entropy and Free Energy Reading: from Petrucci, Harwood and Herring (8th edition): Required for Part 1: Sections 20-1 through 20-4. Recommended for Part 1: Sections
3 - Ice and crystalline water
 3 - Ice and crystalline water All ice is not crystalline not all crystalline water is ice H 2 O solid phases (From Savage) Some general comments on the phase diagram All crystalline ice is tetrahedrically
3 - Ice and crystalline water All ice is not crystalline not all crystalline water is ice H 2 O solid phases (From Savage) Some general comments on the phase diagram All crystalline ice is tetrahedrically
Conduction Modeling in Mixed Alkali Borate Glasses
 International Journal of Pure & Applied Physics ISSN 0973-1776 Vol.1 No.2 (2005), pp. 191-197 Research India Publications http://www.ripub lication.com/ijpap.htm Conduction Modeling in Mixed Alkali Borate
International Journal of Pure & Applied Physics ISSN 0973-1776 Vol.1 No.2 (2005), pp. 191-197 Research India Publications http://www.ripub lication.com/ijpap.htm Conduction Modeling in Mixed Alkali Borate
Significant structure theory applied to liquid helium-3 (surface tension/ thermodynamic properties/ Fermi-Dirac integrals)
 Proc. Natl. Acad. Sci. USA Vol. 74, No., pp. 47394743, November 977 Chemistry Significant structure theory applied to liquid helium3 (surface tension/ thermodynamic properties/ FermiDirac integrals) YOUNGE
Proc. Natl. Acad. Sci. USA Vol. 74, No., pp. 47394743, November 977 Chemistry Significant structure theory applied to liquid helium3 (surface tension/ thermodynamic properties/ FermiDirac integrals) YOUNGE
Atomic and molecular interaction forces in biology
 Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
Atomic and molecular interaction forces in biology 1 Outline Types of interactions relevant to biology Van der Waals interactions H-bond interactions Some properties of water Hydrophobic effect 2 Types
Aqueous solutions. Solubility of different compounds in water
 Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Aqueous solutions Solubility of different compounds in water The dissolution of molecules into water (in any solvent actually) causes a volume change of the solution; the size of this volume change is
Physics 53. Thermal Physics 1. Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital.
 Physics 53 Thermal Physics 1 Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital. Arthur Koestler Overview In the following sections we will treat macroscopic systems
Physics 53 Thermal Physics 1 Statistics are like a bikini. What they reveal is suggestive; what they conceal is vital. Arthur Koestler Overview In the following sections we will treat macroscopic systems
Lennard-Jones as a model for argon and test of extended renormalization group calculations
 JOURNAL OF CHEMICAL PHYSICS VOLUME 111, NUMBER 2 22 NOVEMBER 1999 Lennard-Jones as a model for argon and test of extended renormalization group calculations John A. White Department of Physics, American
JOURNAL OF CHEMICAL PHYSICS VOLUME 111, NUMBER 2 22 NOVEMBER 1999 Lennard-Jones as a model for argon and test of extended renormalization group calculations John A. White Department of Physics, American
Chapter 10: Liquids and Solids
 Chapter 10: Liquids and Solids Chapter 10: Liquids and Solids *Liquids and solids show many similarities and are strikingly different from their gaseous state. 10.1 Intermolecular Forces Intermolecular
Chapter 10: Liquids and Solids Chapter 10: Liquids and Solids *Liquids and solids show many similarities and are strikingly different from their gaseous state. 10.1 Intermolecular Forces Intermolecular
Chem 112 Dr. Kevin Moore
 Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
Chem 112 Dr. Kevin Moore Gas Liquid Solid Polar Covalent Bond Partial Separation of Charge Electronegativity: H 2.1 Cl 3.0 H Cl δ + δ - Dipole Moment measure of the net polarity in a molecule Q Q magnitude
ME 262A - Physical Gas Dynamics 1996 Final Exam: Open Book Portion. h = 6.62 x J s Energy conversion factor: 1 calorie = 4.
 Name: ME 262A - Physical Gas Dynamics 1996 Final Exam: Open Book Portion Useful data and information: k = 1.38 x 10-23 J/K h = 6.62 x 10-34 J s Energy conversion factor: 1 calorie = 4.2 J 1. (40 points)
Name: ME 262A - Physical Gas Dynamics 1996 Final Exam: Open Book Portion Useful data and information: k = 1.38 x 10-23 J/K h = 6.62 x 10-34 J s Energy conversion factor: 1 calorie = 4.2 J 1. (40 points)
Intermolecular Forces and Liquids and Solids. Chapter 11. Copyright The McGraw Hill Companies, Inc. Permission required for
 Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw Hill Companies, Inc. Permission required for 1 A phase is a homogeneous part of the system in contact with other parts of the
Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw Hill Companies, Inc. Permission required for 1 A phase is a homogeneous part of the system in contact with other parts of the
B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry
 B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry ORGANIZING THEME/TOPIC UNIT 1: ATOMIC STRUCTURE Atomic Theory Electron configuration Periodic Trends Big Idea 1: The chemical
B L U E V A L L E Y D I S T R I C T C U R R I C U L U M Science AP Chemistry ORGANIZING THEME/TOPIC UNIT 1: ATOMIC STRUCTURE Atomic Theory Electron configuration Periodic Trends Big Idea 1: The chemical
CLASSICAL ELECTRICITY
 CLASSICAL ELECTRICITY AND MAGNETISM by WOLFGANG K. H. PANOFSKY Stanford University and MELBA PHILLIPS Washington University SECOND EDITION ADDISON-WESLEY PUBLISHING COMPANY Reading, Massachusetts Menlo
CLASSICAL ELECTRICITY AND MAGNETISM by WOLFGANG K. H. PANOFSKY Stanford University and MELBA PHILLIPS Washington University SECOND EDITION ADDISON-WESLEY PUBLISHING COMPANY Reading, Massachusetts Menlo
Force Field for Water Based on Neural Network
 Force Field for Water Based on Neural Network Hao Wang Department of Chemistry, Duke University, Durham, NC 27708, USA Weitao Yang* Department of Chemistry, Duke University, Durham, NC 27708, USA Department
Force Field for Water Based on Neural Network Hao Wang Department of Chemistry, Duke University, Durham, NC 27708, USA Weitao Yang* Department of Chemistry, Duke University, Durham, NC 27708, USA Department
Calorimetry. A calorimeter is a device in which this energy transfer takes place
 Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
Calorimetry One technique for measuring specific heat involves heating a material, adding it to a sample of water, and recording the final temperature This technique is known as calorimetry A calorimeter
Solid water floats on liquid water High surface tension Universal solvent High specific heat High heat of vaporization
 Solid water floats on liquid water High surface tension Universal solvent High specific heat High heat of vaporization Two Hydrogen One Oxygen Share electrons covalent bonds Bent molecule Covalent bonds
Solid water floats on liquid water High surface tension Universal solvent High specific heat High heat of vaporization Two Hydrogen One Oxygen Share electrons covalent bonds Bent molecule Covalent bonds
Intermolecular Forces and Liquids and Solids
 PowerPoint Lecture Presentation by J. David Robertson University of Missouri Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
PowerPoint Lecture Presentation by J. David Robertson University of Missouri Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction
Thermodynamics, Gibbs Method and Statistical Physics of Electron Gases
 Bahram M. Askerov Sophia R. Figarova Thermodynamics, Gibbs Method and Statistical Physics of Electron Gases With im Figures Springer Contents 1 Basic Concepts of Thermodynamics and Statistical Physics...
Bahram M. Askerov Sophia R. Figarova Thermodynamics, Gibbs Method and Statistical Physics of Electron Gases With im Figures Springer Contents 1 Basic Concepts of Thermodynamics and Statistical Physics...
(4) that, even though no chemical combination takes place between. (1) That the energy change represented above is a ftee energy change,4
 524 CHEMISTRY: T. J. WEBB ON THE FREE ENERGY OF HYDRATION OF IONS By T. J. WEBB* PHYSIKALISCHES INSTITUTZ DEPR EIDG. TsCHNISCHHN HOCHSCHULE, ZURICH Communicated July 1, 1926 PROC. N. A. S. 1. Calculations
524 CHEMISTRY: T. J. WEBB ON THE FREE ENERGY OF HYDRATION OF IONS By T. J. WEBB* PHYSIKALISCHES INSTITUTZ DEPR EIDG. TsCHNISCHHN HOCHSCHULE, ZURICH Communicated July 1, 1926 PROC. N. A. S. 1. Calculations
- intermolecular forces forces that exist between molecules
 Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
Chapter 11: Intermolecular Forces, Liquids, and Solids - intermolecular forces forces that exist between molecules 11.1 A Molecular Comparison of Liquids and Solids - gases - average kinetic energy of
Big Idea 1: Structure of Matter Learning Objective Check List
 Big Idea 1: Structure of Matter Learning Objective Check List Structure of Matter Mole Concept: Empirical Formula, Percent Composition, Stoichiometry Learning objective 1.1 The student can justify the
Big Idea 1: Structure of Matter Learning Objective Check List Structure of Matter Mole Concept: Empirical Formula, Percent Composition, Stoichiometry Learning objective 1.1 The student can justify the
Chapter 11. Intermolecular Forces and Liquids & Solids
 Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
Chapter 11 Intermolecular Forces and Liquids & Solids The Kinetic Molecular Theory of Liquids & Solids Gases vs. Liquids & Solids difference is distance between molecules Liquids Molecules close together;
Chemical Potential. Combining the First and Second Laws for a closed system, Considering (extensive properties)
 Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
Chemical Potential Combining the First and Second Laws for a closed system, Considering (extensive properties) du = TdS pdv Hence For an open system, that is, one that can gain or lose mass, U will also
POGIL 7 KEY Intermolecular Forces
 Honors Chem Block Name POGIL 7 KEY Intermolecular Forces In chemistry we talk a lot about properties of substances, since the object of chemistry is substances and their properties. After learning different
Honors Chem Block Name POGIL 7 KEY Intermolecular Forces In chemistry we talk a lot about properties of substances, since the object of chemistry is substances and their properties. After learning different
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline
 Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Chem 1075 Chapter 13 Liquids and Solids Lecture Outline Slide 2-3 Properties of Liquids Unlike gases, liquids respond dramatically to temperature and pressure changes. We can study the liquid state and
Intermolecular Forces and Liquids and Solids
 Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 A phase is a homogeneous part of the system in contact
Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 A phase is a homogeneous part of the system in contact
States of matter Part 1
 Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
Intermolecular Forces and Liquids and Solids
 Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. A phase is a homogeneous part of the system in contact
Intermolecular Forces and Liquids and Solids Chapter 11 Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. A phase is a homogeneous part of the system in contact
Chapter 11: Liquids, Solids, and Intermolecular Forces. Mrs. Brayfield
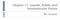 Chapter 11: Liquids, Solids, and Intermolecular Forces Mrs. Brayfield 11.1: Intermolecular Forces Intermolecular forces are attractive forces that exist between all molecules and atoms The state of matter
Chapter 11: Liquids, Solids, and Intermolecular Forces Mrs. Brayfield 11.1: Intermolecular Forces Intermolecular forces are attractive forces that exist between all molecules and atoms The state of matter
Ch 9 Liquids & Solids (IMF) Masterson & Hurley
 Ch 9 Liquids & Solids (IMF) Masterson & Hurley Intra- and Intermolecular AP Questions: 2005 Q. 7, 2005 (Form B) Q. 8, 2006 Q. 6, 2007 Q. 2 (d) and (c), Periodic Trends AP Questions: 2001 Q. 8, 2002 Q.
Ch 9 Liquids & Solids (IMF) Masterson & Hurley Intra- and Intermolecular AP Questions: 2005 Q. 7, 2005 (Form B) Q. 8, 2006 Q. 6, 2007 Q. 2 (d) and (c), Periodic Trends AP Questions: 2001 Q. 8, 2002 Q.
3.091 Introduction to Solid State Chemistry. Lecture Notes No. 6a BONDING AND SURFACES
 3.091 Introduction to Solid State Chemistry Lecture Notes No. 6a BONDING AND SURFACES 1. INTRODUCTION Surfaces have increasing importance in technology today. Surfaces become more important as the size
3.091 Introduction to Solid State Chemistry Lecture Notes No. 6a BONDING AND SURFACES 1. INTRODUCTION Surfaces have increasing importance in technology today. Surfaces become more important as the size
States of matter Part 1. Lecture 1. University of Kerbala. Hamid Alghurabi Assistant Lecturer in Pharmaceutics. Physical Pharmacy
 Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
Physical pharmacy I 1. States of matter (2 Lectures) 2. Thermodynamics (2 Lectures) 3. Solution of non-electrolyte 4. Solution of electrolyte 5. Ionic equilibria 6. Buffered and isotonic solution Physical
Thermodynamics and Equilibrium. Chemical thermodynamics is concerned with energy relationships in chemical reactions.
 1 of 7 Thermodynamics and Equilibrium Chemical thermodynamics is concerned with energy relationships in chemical reactions. In addition to enthalpy (H), we must consider the change in randomness or disorder
1 of 7 Thermodynamics and Equilibrium Chemical thermodynamics is concerned with energy relationships in chemical reactions. In addition to enthalpy (H), we must consider the change in randomness or disorder
Chapter 11. Freedom of Motion. Comparisons of the States of Matter. Liquids, Solids, and Intermolecular Forces
 Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
Liquids, Solids, and Intermolecular Forces Chapter 11 Comparisons of the States of Matter The solid and liquid states have a much higher density than the gas state The solid and liquid states have similar
Chapter 10 Liquids, Solids, and Intermolecular Forces
 Chapter 10 Liquids, Solids, and Intermolecular Forces The Three Phases of Matter (A Macroscopic Comparison) State of Matter Shape and volume Compressibility Ability to Flow Solid Retains its own shape
Chapter 10 Liquids, Solids, and Intermolecular Forces The Three Phases of Matter (A Macroscopic Comparison) State of Matter Shape and volume Compressibility Ability to Flow Solid Retains its own shape
Upon successful completion of this unit, the students should be able to:
 Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Unit 9. Liquids and Solids - ANSWERS Upon successful completion of this unit, the students should be able to: 9.1 List the various intermolecular attractions in liquids and solids (dipole-dipole, London
Chemistry: The Central Science
 Chemistry: The Central Science Fourteenth Edition Chapter 11 Liquids and Intermolecular Forces Intermolecular Forces The attractions between molecules are not nearly as strong as the intramolecular attractions
Chemistry: The Central Science Fourteenth Edition Chapter 11 Liquids and Intermolecular Forces Intermolecular Forces The attractions between molecules are not nearly as strong as the intramolecular attractions
Enduring Understandings & Essential Knowledge for AP Chemistry
 Enduring Understandings & Essential Knowledge for AP Chemistry Big Idea 1: The chemical elements are fundamental building materials of matter, and all matter can be understood in terms of arrangements
Enduring Understandings & Essential Knowledge for AP Chemistry Big Idea 1: The chemical elements are fundamental building materials of matter, and all matter can be understood in terms of arrangements
I. Intermolecular forces and changes in chemical state or phase
 General Chemistry II, in broad strokes I. Intermolecular forces and changes in chemical state or phase II. Solutions, solubility, and colligative properties III. Chemical Kinetics -- the study of rates
General Chemistry II, in broad strokes I. Intermolecular forces and changes in chemical state or phase II. Solutions, solubility, and colligative properties III. Chemical Kinetics -- the study of rates
Solutions. Solution Formation - Types of Solutions - Solubility and the Solution Process - Effects of Temperature and Pressure on Solubility
 Solutions Solutions Solution Formation - Types of Solutions - Solubility and the Solution Process - Effects of Temperature and Pressure on Solubility Colligative Properties - Ways of Expressing Concentration
Solutions Solutions Solution Formation - Types of Solutions - Solubility and the Solution Process - Effects of Temperature and Pressure on Solubility Colligative Properties - Ways of Expressing Concentration
n rr 'ih ifetl! * fn>p' ' -.A SEP <[ NOV 2 0 bba LIBRARY J2 2_ TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51
 CONVAIR S C I E N T I F I C R E S E A R C H L A B O R A T O R Y TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51 ifetl! * W. H. Gallaher JULY 1961 Distribution of this document is
CONVAIR S C I E N T I F I C R E S E A R C H L A B O R A T O R Y TWO PHASE FLOW ABOUT A STAGNATION POINT IN FILM BOILING RESEARC NOTE 51 ifetl! * W. H. Gallaher JULY 1961 Distribution of this document is
15.1 Water and Its Properties > Chapter 15 Water and Aqueous Systems Water and Its Properties Homogeneous Aqueous Systems
 Chapter 15 Water and Aqueous Systems 15.1 Water and Its Properties 15.2 Homogeneous Aqueous Systems 15.3 Heterogeneous Aqueous Systems 1 Copyright Pearson Education, Inc., or its affiliates. All Rights
Chapter 15 Water and Aqueous Systems 15.1 Water and Its Properties 15.2 Homogeneous Aqueous Systems 15.3 Heterogeneous Aqueous Systems 1 Copyright Pearson Education, Inc., or its affiliates. All Rights
Unit 7 Kinetics and Thermodynamics
 17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
17.1 The Flow of Energy Heat and Work Unit 7 Kinetics and Thermodynamics I. Energy Transformations A. Temperature 1. A measure of the average kinetic energy of the particles in a sample of matter B. Heat
cp final review part 2
 Name: Class: Date: cp final review part 2 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Standard conditions when working with gases are
Name: Class: Date: cp final review part 2 Multiple Choice Identify the letter of the choice that best completes the statement or answers the question. 1. Standard conditions when working with gases are
The change in free energy on transferring an ion from a medium of low dielectric constantε1 to one of high dielectric constant ε2:
 The Born Energy of an Ion The free energy density of an electric field E arising from a charge is ½(ε 0 ε E 2 ) per unit volume Integrating the energy density of an ion over all of space = Born energy:
The Born Energy of an Ion The free energy density of an electric field E arising from a charge is ½(ε 0 ε E 2 ) per unit volume Integrating the energy density of an ion over all of space = Born energy:
Intermolecular Forces and States of Matter AP Chemistry Lecture Outline
 Intermolecular Forces and States of Matter AP Chemistry Lecture Outline Name: Chemical properties are related only to chemical composition; physical properties are related to chemical composition AND the
Intermolecular Forces and States of Matter AP Chemistry Lecture Outline Name: Chemical properties are related only to chemical composition; physical properties are related to chemical composition AND the
Chapter 17. Free Energy and Thermodynamics. Chapter 17 Lecture Lecture Presentation. Sherril Soman Grand Valley State University
 Chapter 17 Lecture Lecture Presentation Chapter 17 Free Energy and Thermodynamics Sherril Soman Grand Valley State University First Law of Thermodynamics You can t win! The first law of thermodynamics
Chapter 17 Lecture Lecture Presentation Chapter 17 Free Energy and Thermodynamics Sherril Soman Grand Valley State University First Law of Thermodynamics You can t win! The first law of thermodynamics
Chapter 10: Liquids, Solids, and Phase Changes
 Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Chapter 10: Liquids, Solids, and Phase Changes In-chapter exercises: 10.1 10.6, 10.11; End-of-chapter Problems: 10.26, 10.31, 10.32, 10.33, 10.34, 10.35, 10.36, 10.39, 10.40, 10.42, 10.44, 10.45, 10.66,
Basic Chemistry 2014 Timberlake
 A Correlation of Basic Chemistry Timberlake Advanced Placement Chemistry Topics AP is a trademark registered and/or owned by the College Board, which was not involved in the production of, and does not
A Correlation of Basic Chemistry Timberlake Advanced Placement Chemistry Topics AP is a trademark registered and/or owned by the College Board, which was not involved in the production of, and does not
Thermal and Statistical Physics Department Exam Last updated November 4, L π
 Thermal and Statistical Physics Department Exam Last updated November 4, 013 1. a. Define the chemical potential µ. Show that two systems are in diffusive equilibrium if µ 1 =µ. You may start with F =
Thermal and Statistical Physics Department Exam Last updated November 4, 013 1. a. Define the chemical potential µ. Show that two systems are in diffusive equilibrium if µ 1 =µ. You may start with F =
M02M.1 Particle in a Cone
 Part I Mechanics M02M.1 Particle in a Cone M02M.1 Particle in a Cone A small particle of mass m is constrained to slide, without friction, on the inside of a circular cone whose vertex is at the origin
Part I Mechanics M02M.1 Particle in a Cone M02M.1 Particle in a Cone A small particle of mass m is constrained to slide, without friction, on the inside of a circular cone whose vertex is at the origin
CHEM Principles of Chemistry II Chapter 10 - Liquids and Solids
 CHEM 1212 - Principles of Chemistry II Chapter 10 - Liquids and Solids 10.1 Intermolecular Forces recall intramolecular (within the molecule) bonding whereby atoms can form stable units called molecules
CHEM 1212 - Principles of Chemistry II Chapter 10 - Liquids and Solids 10.1 Intermolecular Forces recall intramolecular (within the molecule) bonding whereby atoms can form stable units called molecules
Appendix 4. Appendix 4A Heat Capacity of Ideal Gases
 Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
Appendix 4 W-143 Appendix 4A Heat Capacity of Ideal Gases We can determine the heat capacity from the energy content of materials as a function of temperature. The simplest material to model is an ideal
Professor K. Intermolecular forces
 Professor K Intermolecular forces We've studied chemical bonds which are INTRAmolecular forces... We now explore the forces between molecules, or INTERmolecular forces which you might rightly assume to
Professor K Intermolecular forces We've studied chemical bonds which are INTRAmolecular forces... We now explore the forces between molecules, or INTERmolecular forces which you might rightly assume to
Phase Equilibria and Molecular Solutions Jan G. Korvink and Evgenii Rudnyi IMTEK Albert Ludwig University Freiburg, Germany
 Phase Equilibria and Molecular Solutions Jan G. Korvink and Evgenii Rudnyi IMTEK Albert Ludwig University Freiburg, Germany Preliminaries Learning Goals Phase Equilibria Phase diagrams and classical thermodynamics
Phase Equilibria and Molecular Solutions Jan G. Korvink and Evgenii Rudnyi IMTEK Albert Ludwig University Freiburg, Germany Preliminaries Learning Goals Phase Equilibria Phase diagrams and classical thermodynamics
Fundamentals. Statistical. and. thermal physics. McGRAW-HILL BOOK COMPANY. F. REIF Professor of Physics Universüy of California, Berkeley
 Fundamentals of and Statistical thermal physics F. REIF Professor of Physics Universüy of California, Berkeley McGRAW-HILL BOOK COMPANY Auckland Bogota Guatemala Hamburg Lisbon London Madrid Mexico New
Fundamentals of and Statistical thermal physics F. REIF Professor of Physics Universüy of California, Berkeley McGRAW-HILL BOOK COMPANY Auckland Bogota Guatemala Hamburg Lisbon London Madrid Mexico New
A Corresponding State Theory for the Viscosity of Liquids Bull. Korean Chem. Soc. 2008, Vol. 29, No Articles
 A Corresponding State Theory for the Viscosity of Liquids Bull. Korean Chem. Soc. 2008, Vol. 29, No. 1 33 Articles A Corresponding State Theory for the Viscosity of Liquids Wonsoo Kim * and Sukbae Lee
A Corresponding State Theory for the Viscosity of Liquids Bull. Korean Chem. Soc. 2008, Vol. 29, No. 1 33 Articles A Corresponding State Theory for the Viscosity of Liquids Wonsoo Kim * and Sukbae Lee
201. The Nature o f the Metallic Bond. III
 No. 8] 913 201. The Nature o f the Metallic Bond. III Atomic Interactions in Alloys. I By San-ichiro MIZUSHIMA, M.J.A., and Isao Ichishima Tokyo Research Institute, Yawata Iron and Steel Co. Ltd., Ida,
No. 8] 913 201. The Nature o f the Metallic Bond. III Atomic Interactions in Alloys. I By San-ichiro MIZUSHIMA, M.J.A., and Isao Ichishima Tokyo Research Institute, Yawata Iron and Steel Co. Ltd., Ida,
Dipole moments of glycerol, isopropyl alcohol, and isobutyl alcohol
 Dipole moments of glycerol, isopropyl alcohol, and isobutyl alcohol H. A. RIZK AND 1. M. ELANWAR Deprrr'fnzerit of Clzemistry, Frrcrrlty of Science, U~riversity of Crriro, Crriro, Egypt, U.A.R. Received
Dipole moments of glycerol, isopropyl alcohol, and isobutyl alcohol H. A. RIZK AND 1. M. ELANWAR Deprrr'fnzerit of Clzemistry, Frrcrrlty of Science, U~riversity of Crriro, Crriro, Egypt, U.A.R. Received
CH.7 Fugacities in Liquid Mixtures: Models and Theories of Solutions
 CH.7 Fugacities in Liquid Mixtures: Models and Theories of Solutions The aim of solution theory is to express the properties of liquid mixture in terms of intermolecular forces and liquid structure. The
CH.7 Fugacities in Liquid Mixtures: Models and Theories of Solutions The aim of solution theory is to express the properties of liquid mixture in terms of intermolecular forces and liquid structure. The
(a) Conventional molecular-orbital method, without configuration interaction. (b) Thomas-Fermi statistical method.
 372 PHYSICS: COULSON, MARCH AND ALTMANN PROC. N. A. S. T-ELECTRONS AND a-electrons* By C. A. COULSON,t N. H. MARCHi: AND S. ALTMANN WHEATSTONE PHYSICs DEPARTMENT, KING'S COLLEGE, LONDON, ENGLAND Communicated
372 PHYSICS: COULSON, MARCH AND ALTMANN PROC. N. A. S. T-ELECTRONS AND a-electrons* By C. A. COULSON,t N. H. MARCHi: AND S. ALTMANN WHEATSTONE PHYSICs DEPARTMENT, KING'S COLLEGE, LONDON, ENGLAND Communicated
 Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Title Super- and subcritical hydration of Thermodynamics of hydration Author(s) Matubayasi, N; Nakahara, M Citation JOURNAL OF CHEMICAL PHYSICS (2000), 8109 Issue Date 2000-05-08 URL http://hdl.handle.net/2433/50350
Modern Optics Prof. Partha Roy Chaudhuri Department of Physics Indian Institute of Technology, Kharagpur
 Modern Optics Prof. Partha Roy Chaudhuri Department of Physics Indian Institute of Technology, Kharagpur Lecture 08 Wave propagation in anisotropic media Now, we will discuss the propagation of electromagnetic
Modern Optics Prof. Partha Roy Chaudhuri Department of Physics Indian Institute of Technology, Kharagpur Lecture 08 Wave propagation in anisotropic media Now, we will discuss the propagation of electromagnetic
Chapter 13 Properties of Solutions
 Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 13 Properties of John D. Bookstaver St. Charles Community College St. Peters, MO 2006,
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 13 Properties of John D. Bookstaver St. Charles Community College St. Peters, MO 2006,
1.3 Molecular Level Presentation
 1.3.1 Introduction A molecule is the smallest chemical unit of a substance that is capable of stable, independent existence. Not all substances are composed of molecules. Some substances are composed of
1.3.1 Introduction A molecule is the smallest chemical unit of a substance that is capable of stable, independent existence. Not all substances are composed of molecules. Some substances are composed of
2. As gas P increases and/or T is lowered, intermolecular forces become significant, and deviations from ideal gas laws occur (van der Waal equation).
 A. Introduction. (Section 11.1) CHAPTER 11: STATES OF MATTER, LIQUIDS AND SOLIDS 1. Gases are easily treated mathematically because molecules behave independently. 2. As gas P increases and/or T is lowered,
A. Introduction. (Section 11.1) CHAPTER 11: STATES OF MATTER, LIQUIDS AND SOLIDS 1. Gases are easily treated mathematically because molecules behave independently. 2. As gas P increases and/or T is lowered,
At constant temperature, the first term is zero. Using Maxwell relation and for
 Issue 12 Number1 May A THEORITICAL STUDY OF INTERNAL PRESSURE AND FREE VOLUME FOR SINGLE MOLECULE OF A SAMPLE LIQUID D.Prakash and Dr. A.Mukunthan Department of Physics, Bharath UniversityChennai 7. Email:
Issue 12 Number1 May A THEORITICAL STUDY OF INTERNAL PRESSURE AND FREE VOLUME FOR SINGLE MOLECULE OF A SAMPLE LIQUID D.Prakash and Dr. A.Mukunthan Department of Physics, Bharath UniversityChennai 7. Email:
Chapter 11 Spontaneous Change and Equilibrium
 Chapter 11 Spontaneous Change and Equilibrium 11-1 Enthalpy and Spontaneous Change 11-2 Entropy 11-3 Absolute Entropies and Chemical Reactions 11-4 The Second Law of Thermodynamics 11-5 The Gibbs Function
Chapter 11 Spontaneous Change and Equilibrium 11-1 Enthalpy and Spontaneous Change 11-2 Entropy 11-3 Absolute Entropies and Chemical Reactions 11-4 The Second Law of Thermodynamics 11-5 The Gibbs Function
Instituut voor Theoretische Fysica, Rijksuniversiteit Utrecht, Utrecht, Tire Netherlands
 Physica 80A (1975) 398-404 North-Holland Publishing Co. SOME REMARKS ON THE THEORY OF THE DIELECTRIC CONSTANT OF NON-POLAR DENSE GASES B. R. A. NIJBOER Instituut voor Theoretische Fysica, Rijksuniversiteit
Physica 80A (1975) 398-404 North-Holland Publishing Co. SOME REMARKS ON THE THEORY OF THE DIELECTRIC CONSTANT OF NON-POLAR DENSE GASES B. R. A. NIJBOER Instituut voor Theoretische Fysica, Rijksuniversiteit
CHAPTER 1 THEORETICAL ASPECTS OF HEAT CAPACITY OF SOLIDS AND LIQUIDS
 CHAPTER 1 THEORETICAL ASPECTS OF HEAT CAPACITY OF SOLIDS AND LIQUIDS 1.1 CERTAIN THEORETICAL ASPECTS OF HEAT CAPACITY OF SOLIDS - BRIEF INTRODUCTION Heat capacity is considered as one of the most important
CHAPTER 1 THEORETICAL ASPECTS OF HEAT CAPACITY OF SOLIDS AND LIQUIDS 1.1 CERTAIN THEORETICAL ASPECTS OF HEAT CAPACITY OF SOLIDS - BRIEF INTRODUCTION Heat capacity is considered as one of the most important
Engineering Physics 1 Dr. B. K. Patra Department of Physics Indian Institute of Technology-Roorkee
 Engineering Physics 1 Dr. B. K. Patra Department of Physics Indian Institute of Technology-Roorkee Module-05 Lecture-02 Kinetic Theory of Gases - Part 02 (Refer Slide Time: 00:32) So, after doing the angular
Engineering Physics 1 Dr. B. K. Patra Department of Physics Indian Institute of Technology-Roorkee Module-05 Lecture-02 Kinetic Theory of Gases - Part 02 (Refer Slide Time: 00:32) So, after doing the angular
Chapter 10. Liquids and Solids
 Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Chapter 10 Liquids and Solids Chapter 10 Table of Contents 10.1 Intermolecular Forces 10.2 The Liquid State 10.3 An Introduction to Structures and Types of Solids 10.4 Structure and Bonding in Metals 10.5
Discipline Course-I. Semester-II. Paper No: Electricity and Magnetism. Lesson: Polarization and Dielectric Materials
 Discipline CourseI SemesterII Paper No: Electricity and Magnetism Lesson: Polarization and Dielectric Materials Lesson Developer: Dr. Amit Choudhary College/ Department: Physics Department, Deshbandhu
Discipline CourseI SemesterII Paper No: Electricity and Magnetism Lesson: Polarization and Dielectric Materials Lesson Developer: Dr. Amit Choudhary College/ Department: Physics Department, Deshbandhu
A critical remark on Planck s model of black body
 A critical remark on Planck s model of black body Andrea Carati Luigi Galgani 01/12/2003 Abstract We reexamine the model of matter radiation interaction considered by Planck in his studies on the black
A critical remark on Planck s model of black body Andrea Carati Luigi Galgani 01/12/2003 Abstract We reexamine the model of matter radiation interaction considered by Planck in his studies on the black
Chapter #16 Liquids and Solids
 Chapter #16 Liquids and Solids 16.1 Intermolecular Forces 16.2 The Liquid State 16.3 An Introduction to Structures and Types of Solids 16.4 Structure and Bonding of Metals 16.5 Carbon and Silicon: Network
Chapter #16 Liquids and Solids 16.1 Intermolecular Forces 16.2 The Liquid State 16.3 An Introduction to Structures and Types of Solids 16.4 Structure and Bonding of Metals 16.5 Carbon and Silicon: Network
Week 11/Th: Lecture Units 28 & 29
 Week 11/Th: Lecture Units 28 & 29 Unit 27: Real Gases Unit 28: Intermolecular forces -- types of forces between molecules -- examples Unit 29: Crystal Structure -- lattice types -- unit cells -- simple
Week 11/Th: Lecture Units 28 & 29 Unit 27: Real Gases Unit 28: Intermolecular forces -- types of forces between molecules -- examples Unit 29: Crystal Structure -- lattice types -- unit cells -- simple
Chapter 14. Liquids and Solids
 Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
Chapter 14 Liquids and Solids Section 14.1 Water and Its Phase Changes Reviewing What We Know Gases Low density Highly compressible Fill container Solids High density Slightly compressible Rigid (keeps
Let's look at the force on a current loop. In a uniform field it is zero: F = I I (dl B) =I I dl B =0 (4) since B is constant and comes outside the in
 Midterm: Mean 4.4/30, sigma = 5, high score - 25/30 Topic 3: Magnetostatic Fields in Matter Reading Assignment: Jackson Chapter 5.7-5. The treatment of magnetostatic fields in matter is quite parallel
Midterm: Mean 4.4/30, sigma = 5, high score - 25/30 Topic 3: Magnetostatic Fields in Matter Reading Assignment: Jackson Chapter 5.7-5. The treatment of magnetostatic fields in matter is quite parallel
Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras
 Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Lecture - 21 Quenching of Orbital Angular Momentum; Ferromagnetism In the last lecture, we saw
Condensed Matter Physics Prof. G. Rangarajan Department of Physics Indian Institute of Technology, Madras Lecture - 21 Quenching of Orbital Angular Momentum; Ferromagnetism In the last lecture, we saw
Condensed matter theory Lecture notes and problem sets 2012/2013
 Condensed matter theory Lecture notes and problem sets 2012/2013 Dmitri Ivanov Recommended books and lecture notes: [AM] N. W. Ashcroft and N. D. Mermin, Solid State Physics. [Mar] M. P. Marder, Condensed
Condensed matter theory Lecture notes and problem sets 2012/2013 Dmitri Ivanov Recommended books and lecture notes: [AM] N. W. Ashcroft and N. D. Mermin, Solid State Physics. [Mar] M. P. Marder, Condensed
 lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
lectures accompanying the book: Solid State Physics: An Introduction, by Philip ofmann (2nd edition 2015, ISBN-10: 3527412824, ISBN-13: 978-3527412822, Wiley-VC Berlin. www.philiphofmann.net 1 Bonds between
Final Exam for Physics 176. Professor Greenside Wednesday, April 29, 2009
 Print your name clearly: Signature: I agree to neither give nor receive aid during this exam Final Exam for Physics 76 Professor Greenside Wednesday, April 29, 2009 This exam is closed book and will last
Print your name clearly: Signature: I agree to neither give nor receive aid during this exam Final Exam for Physics 76 Professor Greenside Wednesday, April 29, 2009 This exam is closed book and will last
Dielectric polarization of 2-pyrrolidinone molecules in benzene solution - a quantum-chemical study
 Dielectric polarization of 2-pyrrolidinone molecules in benzene solution - a quantum-chemical study L. Gorb* ), J. Jadżyn $) and K. W. Wojciechowski #) Institute of Molecular Physics, Polish Academy of
Dielectric polarization of 2-pyrrolidinone molecules in benzene solution - a quantum-chemical study L. Gorb* ), J. Jadżyn $) and K. W. Wojciechowski #) Institute of Molecular Physics, Polish Academy of
CHAPTER 2 INTERATOMIC FORCES. atoms together in a solid?
 CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
CHAPTER 2 INTERATOMIC FORCES What kind of force holds the atoms together in a solid? Interatomic Binding All of the mechanisms which cause bonding between the atoms derive from electrostatic interaction
12A Entropy. Entropy change ( S) N Goalby chemrevise.org 1. System and Surroundings
 12A Entropy Entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result in products that
12A Entropy Entropy change ( S) A SPONTANEOUS PROCESS (e.g. diffusion) will proceed on its own without any external influence. A problem with H A reaction that is exothermic will result in products that
Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions
 Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions B.G. Potter, Jr., B.A. Tuttle, and V. Tikare Sandia National Laboratories Albuquerque, NM
Monte Carlo Simulation of Ferroelectric Domain Structure: Electrostatic and Elastic Strain Energy Contributions B.G. Potter, Jr., B.A. Tuttle, and V. Tikare Sandia National Laboratories Albuquerque, NM
Properties of Solutions
 Properties of Solutions The States of Matter The state a substance is in at a particular temperature and pressure depends on two antagonistic entities: The kinetic energy of the particles The strength
Properties of Solutions The States of Matter The state a substance is in at a particular temperature and pressure depends on two antagonistic entities: The kinetic energy of the particles The strength
INTERMEDIATE BONDING AND INTERMOLECULAR FORCES. Electronegativity
 INTERMEDIATE BNDING AND INTERMLECULAR FRCES Electronegativity is defined as follows Electronegativity Electronegativity is the ability of an atom within a covalent bond to attract the bonding pair of electrons.
INTERMEDIATE BNDING AND INTERMLECULAR FRCES Electronegativity is defined as follows Electronegativity Electronegativity is the ability of an atom within a covalent bond to attract the bonding pair of electrons.
Physical Pharmacy PHR 211. Lecture 1. Solubility and distribution phenomena.
 Physical Pharmacy PHR 211 Lecture 1 Solubility and distribution phenomena. Course coordinator Magda ELMassik, PhD Assoc. Prof. of Pharmaceutics 1 Objectives of the lecture After completion of thislecture,
Physical Pharmacy PHR 211 Lecture 1 Solubility and distribution phenomena. Course coordinator Magda ELMassik, PhD Assoc. Prof. of Pharmaceutics 1 Objectives of the lecture After completion of thislecture,
Solutions and Intermolecular Forces
 Solutions and Intermolecular Forces REVIEW Chemical Bonds Three basic types of bonds: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other
Solutions and Intermolecular Forces REVIEW Chemical Bonds Three basic types of bonds: Ionic Electrostatic attraction between ions Covalent Sharing of electrons Metallic Metal atoms bonded to several other
Surface interactions part 1: Van der Waals Forces
 CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
CHEM-E150 Interfacial Phenomena in Biobased Systems Surface interactions part 1: Van der Waals Forces Monika Österberg Spring 018 Content Colloidal stability van der Waals Forces Surface Forces and their
Chapter 13. Properties of Solutions. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Lecture Presentation Chapter 13 Properties of John D. Bookstaver St. Charles Community College Cottleville, MO are homogeneous mixtures of two or more pure substances. In a solution, the solute is dispersed
Thermodynamics II. Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
 Thermodynamics II Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Spontaneous Physical and Chemical Processes A waterfall runs downhill A lump of sugar dissolves
Thermodynamics II Copyright The McGraw-Hill Companies, Inc. Permission required for reproduction or display. 1 Spontaneous Physical and Chemical Processes A waterfall runs downhill A lump of sugar dissolves
Review of Chemical Equilibrium Introduction
 Review of Chemical Equilibrium Introduction Copyright c 2015 by Nob Hill Publishing, LLC This chapter is a review of the equilibrium state of a system that can undergo chemical reaction Operating reactors
Review of Chemical Equilibrium Introduction Copyright c 2015 by Nob Hill Publishing, LLC This chapter is a review of the equilibrium state of a system that can undergo chemical reaction Operating reactors
IMFA s. intermolecular forces of attraction Chez Chem, LLC All rights reserved.
 IMFA s intermolecular forces of attraction 2014 Chez Chem, LLC All rights reserved. **London Dispersion Forces Also know as Van der Waals forces A momentary non symmetrical electron distribution that can
IMFA s intermolecular forces of attraction 2014 Chez Chem, LLC All rights reserved. **London Dispersion Forces Also know as Van der Waals forces A momentary non symmetrical electron distribution that can
