Atomic spectroscopy (part I), 2017 Uwe Burghaus, Fargo, ND, USA
|
|
|
- Daniella Webb
- 5 years ago
- Views:
Transcription
1 Atomic spectroscopy (part I), 2017 Uwe Burghaus, Fargo, ND, USA
2 Last class: group theory Symmetry operations σ reflections C rotations S rotation-reflections I inversion E identity
3
4 Spectra of Atoms and Molecules, 3 rd Ed., Peter F. Bernath, Oxford University Press, chapter 5 Physics of Atoms and Molecules, B.H. Bransden, C.J. Joachain, Wiley, chapters 3-9 Foundations of Spectroscopy, S. Duckett, B. Gilbert, Oxford Chemistry Primers, Vol. 78 chapter 4 Molecular Spectroscopy, J.M. Brown, Oxford Chemistry Primers, Vol. 55 chapter 7
5 Jespersen, Hyslop, Chemistry the molecular nature of matter chapter 7.2; 7.3, page 338 (7 th Ed.) or R. Chang, Physical Chemistry for the Biosciences chapter 11.4, page 407- or P. Atkins / J. Paula, Physical chemistry for the life sciences chapter 9.7, page 338 (2 nd Ed.) or P. Atkins / J. Paula, Physical Chemistry chapter 11.1d, page 298 (7 th Ed.) or I.N. Levine, Physical chemistry chapter 18.3, page 602 (5 th Ed.) or T. Engel / P. Reid, Physical chemistry, chapter 12.7 (1 st Ed.) I PChem
6 Atomic spectroscopy Absorption spectroscopy Bohr model QM of H atom review Later in this class: Molecular spectroscopy, 2017 Uwe Burghaus, Fargo, ND, USA
7 Today Bohr Model
8 Niels Henrik David Bohr ( ) Danish physicist Nobel Prize in Physics in During 2 nd world war: UK USA Manhatten project in LosAlamos Also his son got a Nobel price. Similar case: Marie Curie & her daughter Who rejected a Nobel prize?
9 hydrogen (from the Greek ὑδρο- hydro meaning "water" and -γενής genes meaning "creator ): water is produced when hydrogen is burned Fe + H 2 O FeO + H 2 Robert Boyle apparently the 1 st who hade H 2 in 1671 Antoine-Laurent de Lavoisier named the element hydrogen in H 2 (g) + O 2 (g) 2 H 2 O(l)
10 Emission/Absorption spectra of atomic gases glowing gas sample Emission spectra of atoms bright line spectra PChem Quantum mechanics H He lamp Absorption spectra of atoms dark line spectra H Lyman series H Balmer series non-glowing gas sample H α H β H γ H οο
11 Emission/Absorption spectra of atomic gases PChem Quantum mechanics Experimental results Line spectra opposed to continuous blackbody radiation Different atoms different spectra Complex pattern of lines Series of lines Lines converge into continuum at one end
12 Emission (/Absorption) spectroscopy PChem Quantum mechanics light source slit lens diffraction gratings lens detector What is that, emission or absorption spectroscopy?
13 Light = electromagnetic wave PChem Quantum mechanics 1D figure Snapshot time = t 0 E(r,t) = E0cos(kr -ωt + φ) B(r,t) = B cos(kr -ωt + φ) electric field amplitude of electric E E k vector 0 field vector r coordinates ω angular t frequency φ phase lag B B 0 magnetic field vector 0 wave vector time amplitude of magnetic field vector Maxwell According to Maxwell's classical theory of light, electromagnetic waves are produced by accelerated electric charges. A charge oscillating at a frequency ν will emit radiation at that frequency.
14 Classical physics & spectroscopy PChem Quantum mechanics The spectroscopic results from Balmer, Lyman, etc. were inconsistent with classical physics. According to Maxwell's classical theory of light, electromagnetic waves are produced by accelerated electric charges. A charge oscillating at a frequency ν will emit radiation at that frequency. Bohr s postulates 1) Stationary states, E 1, E 2, E 3, Quantum mechanics Classical physics
15 Emission/Absorption spectra PChem Quantum mechanics Bohr s postulates 1) Stationary states, E 1, E 2, E 3, 2) No radiation on stationary states 3) E=E n -E n+1 = hν (transitions) 4) F(Coulomb) = F(Centrifugal) 5) Quantization of angular momentum l = n h/(2pi); n = 1,2,3, ν = 1/ λ = E E 4 2 e m0z 1 1 = ( ) π ε c m n n m Energy R (Rhydberg constant) m 0 : electron mass for speed = zero c: speed of light m, n: quantum # ε: dielectric constant Z: charge number λ: wavelength 1/λ: wave number e: electron charge h: Planck s const. h_bar = h/2π
16 Absorption / Emission of a photon Quantum mechanics E 2 E 2 photon E = hν E = hν photon E 1 E 1 absorption emission Bohr s postulates 1) Stationary states, E 1, E 2, E 3, 2) No radiation on stationary states 3) E=E n -E n+1 = hν (transitions).
17 Emission/Absorption spectra PChem Quantum mechanics ν = 1/ λ = E E 4 2 e m0z 1 1 = ( ) π ε c m n n m 0 Energy Energy 3 2 ν 1 n 2 R (Rhydberg constant) 0 1 m 0 : electron mass for speed = zero c: speed of light m, n: quantum # ε: dielectric constant Z: charge number λ: wavelength 1/λ: wave number e: electron charge h: Planck s const. h_bar = h/2π
18 Absorption spectra of atoms lamp non-glowing gas sample dark line spectra Classical limit n large (classical mechanics) ν = 1/ λ = E E 4 2 e m0z 1 1 = ( ) π ε c m n n m Electronic states of atoms n small: QM n large: classical mechanics Chang figure 11.12
19 Classical limit PChem Quantum mechanics QM classical mechanic n large h 0 Why do we know this? Spectroscopy, Bohr model Blackbody radiation Planck's equation Rayleigh s equation
20 Bohr s derivation PChem Quantum mechanics ν = 1/ λ = E E 4 2 e m0z 1 1 = ( ) π ε c m n n m Most books don t include the historic derivation, because it s simple but lengthy algebra, & it is not really correct centripetal force = coulomb force (4 th Bohr postulate) Quantization of angular momentum (5 th Bohr postulate) m 0 : electron mass for speed = zero c: speed of light m, n: quantum # ε: dielectric constant Z: charge number λ: wavelength 1/λ: wave number e: electron charge h: Planck s const. h_bar = h/2π k e Coulomb constant
21 Standing waves & stationary states in quantum mechanics PChem Quantum mechanics λ/2 2x λ/2 3x λ/2 Bohr s postulates 1) Stationary states, E 1, E 2, E 3, λ electron = h p Quantization of angular momentum (5 th Bohr postulate) wave fits in orbit wave does NOT fit in orbit
22 Important & well known numbers PChem Quantum mechanics How large is an atom? Bohr radius a 0 Ground state energy of H atom m 0 : electron mass for speed = zero c: speed of light m, n: quantum # ε: dielectric constant Z: charge number λ: wavelength 1/λ: wave number e: electron charge h: Planck s const. h_bar = h/2π k e Coulomb constant
23 More modern version of this looks like this lamp Absorption spectra of atoms dark line spectra diffraction grating light source mirror reference cell detector non-glowing gas sample 1.0 transparent mirror absorption (a. u.) Anthracene Absorption spectra of molecules mirror mirror sample cell detector wavelength (nm) Anthracene data from NDSU Pchem lab class 2006, U. Burghaus
24 Vibrations of small molecules - UVvis Absorption spectra of small molecules vibrations 1.0 electronic states absorption (a. u.) Anthracene wavelength (nm) Excitation spectra excitation wavelength scanned emission wavelength fixed Anthracene data from NDSU Pchem lab class 2006, U. Burghaus
25 Vibrations and rotations of small molecules - FTIR energy Electronic excitations vibrations rotations of molecules. PChem364 quantum mechanics excited state rotations distance j=3 j=2 j=1 j=0 v=3 v=2 electronic energy levels ground state v=0 v=1 distance electronic energy level vibrations
26 HCl/DCl IR data from NDSU Pchem lab class 2008 Standard example rot & vis spectra of HCl, DCl Vibration-rotation EXAMPLE HCl/DCl inensity (a.u.) 70 P branch R branch isotope splitting wave number in 1/cm inensity (a.u.) wave number in 1/cm v = 1 v = 0 j = 3 j = 2 j = 1 j = 0 j = 3 j = 2 j = 1 j = 0
27 31 Electronic structure of carbon nanotubes E 2 E 1 photon absorption E = hν metallic carbon nanotubes semiconducting carbon nanotubes energy conduction band energy conduction band E F E 11 E 11 E 22 E F valence band valence band Density of states Density of states
28 Absorption spectroscopy Energy 3 2 absorption (a. u.) 0.6 Anthracene wavelength (nm) energy conduction band vibrations electronic states E F 1 valence band Density of states H
29 So far this was a review of non chemistry major undergraduate knowledge
30 Atomic spectroscopy Absorption spectroscopy Bohr model QM of H atom review Later in this class: Molecular spectroscopy, 2017 Uwe Burghaus, Fargo, ND, USA
31 Survey of the quantum mechanics of the H atom Goal for the rest of this class Understand structure of the solution No mathematical details today Interaction potential coulomb potential Hamilton operator for H atom Definition of the problem Strategy spherical coordinates Idea of the solution Separation of variables Solution three equations quantum numbers
32 Spherical Coordinates PChem Quantum mechanics z (r,θ,φ) (x,y,z) z x φ Θ r y Θ r y (x,y,z) dv = dxdydz x (r,θ,φ) dv = r 2 dr sin(θ) dθ dφ
33 PChem Quantum mechanics H atom mathematical structure of the solution ( ) H x y z Ze r H E = + + = µ πε 0 ψ ψ ; Idea: use spherical coordinates, separate variables 3-separte differential equations ψ ψ πε φ ψ ψ ψ E r e r r r r r r m = Θ + Θ Θ Θ Θ )) ( sin 1 ) (sin sin 1 ) ( 1 [ 2 ) ( ) ( ) (,,,, φ θ ψ m m l l n m l n r R Φ Θ =
34 Schrödinger equation for the H atom in polar coordinates 2 Eq. (20.2) in Engel/Reid Compare this with 3D rigid rotor (chapter 18. 3) Eq. (18.7) in Engel/Reid Good news: angular part (20.2) = (18.7)
35 Engel s book page 436 Why are the solutions for the rigid rotor and the angular components of the H atom identical?
36 Separation of variables 3D rigid rotor Radial equation
37 Radial equation repulsive effective potential attractive
38 Spherical harmonics 3D rigid rotor
39
40 Radial functions
41 H atom wave functions PChem Quantum mechanics n l m ψ n l m
42 Real wave functions as linear combinations of complex wave functions
43 Quantum numbers for one electron system (H atom) PChem Quantum mechanics n = 1, 2, 3, l = 0, 1, 2, 3,, n-1 principal quantum number m l = 0, ±1, ± 2, ± 3,, ± l angular momentum quantum number magnetic quantum number
44 Paul Adrien Maurice Dirac ( ) was a British theoretical physicist. Dirac made fundamental contributions to the early development of both quantum mechanics and quantum electrodynamics. He held the Lucasian Chair of Mathematics at the University of Cambridge and spent the last fourteen years of his life at Florida State University. Among other discoveries, he formulated the Dirac equation, which describes the behaviour of fermions which led to the prediction of the existence of antimatter. Dirac shared the Nobel Prize in physics 1933 with Erwin Schrödinger, "for the discovery of new productive forms of atomic theory."
45 Stern-Gerlach experiment Ag: closed-shell + 5s electron No angular momentum Effect must be related to 5s electron l=0 for s-electrons It cannot be the angular momentum Electron spin
46 Electron spin vs. orbital angular momentum Orbital momentum L = l( l +1) l = 0,1,2, (n-1) S = s( s + 1) = Electron Spin 3 4 PChem Quantum mechanics s = 1/2 L z = m l m l = +l,,0, -l S z = m s m s = +1/2, -1/2 special case for l = 1
47 Degeneracy of states Two or more different quantum states are degenerate if they have the same energy. 0 Energy l = n n 3 l = m= n Allowed energies E = (degeneracy of states) 2 9 Number of states with identical energy n = 1 Rd 1
48 For a spherical problem use the Schrödinger Eq. in polar/spherical coordinates Use Coulomb potential V=-1/r Separation of variables nlm,. (,, ) = () Θ() Φ() ψ rφθ Rr θ φ Electron is confined to Coulomb potential, boundary condition, Quantum numbers n, l, m, s and rules for these E = -1/n 2, degeneracy of states Read this on a snowy weekend
49 Solving the Schrödinger equation. What do we get out of this? $ 1) wave functions probabilities, electron densities 2) e - confined to potential quantum numbers and rules how to use them 3) allowed energies interpretation of spectroscopic data
50 -) What is a Coulomb potential? -) Write the Hamiltonian for H atom in Cartesian coordinates. -) Outline the basic ideas of the solution of the H atoms in Q.M. -) Why are the solutions for the rigid rotor and the angular components of the H atom identical? -) In what functions does the wave function separate? -) What is a radial function? -) Why are linear combinations of the wave function used? -) What are the relevant quantum numbers? -) What are the energy eigenvalues? -) What is the degeneracy of states?
51 -) What is an emission spectrum? -) What is an absorption spectrum? -) Sketch qualitatively the emission pattern of a gas such as helium or hydrogen -) List Bohr s postulates -) What was the main accomplishment of Bohr s model? -) The Balmer series revers to good luck in a row when playing lottery? Yes/no. -) What is the Lyman series? -) What are stationary states in the framework of Bohr s model? -) Does an electron emit radiation in a stationary state? -) Only the energy is quantized but certainly not the angular momentum. Yes/no. -) The emission lines of Helium are evenly spaced in the frequency domain. Yes/no. -) Hydrogen and deuterium will result in the same emission spectra? Yes/no -) The Rydberg constant revers to the fact that the stock market always goes up over time, i.e. don t worry about your retirement as Rydberg told us already Yes/no -) Why is the reduced mass required? What is this?
52 Figure acknowledgement All images shown in this power point presentation were made by the author except the following with are excluded for the copyright of the author: xxx No part of this publication may be reproduced, stored in a retrieval system or transmitted in any form or by any means except as permitted by the United States Copyright Act, without prior written permission of the author. Trademarks and copyrights are property of their respective owners., 2016 Publisher and author: Uwe Burghaus, Fargo, ND, USA
Spectra of Atoms and Molecules, 3 rd Ed., Peter F. Bernath, Oxford University Press, chapter 5. Engel/Reid, chapter 18.3 / 18.4
 Last class Today Atomic spectroscopy (part I) Absorption spectroscopy Bohr model QM of H atom (review) Atomic spectroscopy (part II)-skipped Visualization of wave functions Atomic spectroscopy (part III)
Last class Today Atomic spectroscopy (part I) Absorption spectroscopy Bohr model QM of H atom (review) Atomic spectroscopy (part II)-skipped Visualization of wave functions Atomic spectroscopy (part III)
Vibrational spectroscopy., 2017 Uwe Burghaus, Fargo, ND, USA
 Vibrational spectroscopy, 017 Uwe Burghaus, Fargo, ND, USA CHEM761 Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states... microwave
Vibrational spectroscopy, 017 Uwe Burghaus, Fargo, ND, USA CHEM761 Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states... microwave
Vibrational spectroscopy., 2017 Uwe Burghaus, Fargo, ND, USA
 Vibrational spectroscopy, 017 Uwe Burghaus, Fargo, ND, USA CHEM761 Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states... by microwave
Vibrational spectroscopy, 017 Uwe Burghaus, Fargo, ND, USA CHEM761 Rotational spectroscopy is concerned with the measurement of the energies of transitions between quantized rotational states... by microwave
Physics 1C Lecture 29B
 Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Rotational spectroscopy., 2017 Uwe Burghaus, Fargo, ND, USA
 Rotational spectroscopy, 2017 Uwe Burghaus, Fargo, ND, USA Atomic spectroscopy (part I) Absorption spectroscopy Bohr model QM of H atom (review) Atomic spectroscopy (part II) Visualization of wave functions
Rotational spectroscopy, 2017 Uwe Burghaus, Fargo, ND, USA Atomic spectroscopy (part I) Absorption spectroscopy Bohr model QM of H atom (review) Atomic spectroscopy (part II) Visualization of wave functions
Stellar Astrophysics: The Interaction of Light and Matter
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Quantum Mechanics: The Hydrogen Atom
 Quantum Mechanics: The Hydrogen Atom 4th April 9 I. The Hydrogen Atom In this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen
Quantum Mechanics: The Hydrogen Atom 4th April 9 I. The Hydrogen Atom In this next section, we will tie together the elements of the last several sections to arrive at a complete description of the hydrogen
Atomic Structure and Atomic Spectra
 Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
Atomic Structure and Atomic Spectra Atomic Structure: Hydrogenic Atom Reading: Atkins, Ch. 10 (7 판 Ch. 13) The principles of quantum mechanics internal structure of atoms 1. Hydrogenic atom: one electron
A few Experimental methods for optical spectroscopy Classical methods Modern methods. Remember class #1 Generating fast LASER pulses
 A few Experimental methods for optical spectroscopy Classical methods Modern methods Shorter class Remember class #1 Generating fast LASER pulses, 2017 Uwe Burghaus, Fargo, ND, USA W. Demtröder, Laser
A few Experimental methods for optical spectroscopy Classical methods Modern methods Shorter class Remember class #1 Generating fast LASER pulses, 2017 Uwe Burghaus, Fargo, ND, USA W. Demtröder, Laser
The Photoelectric Effect
 The Photoelectric Effect Light can strike the surface of some metals causing an electron to be ejected No matter how brightly the light shines, electrons are ejected only if the light has sufficient energy
The Photoelectric Effect Light can strike the surface of some metals causing an electron to be ejected No matter how brightly the light shines, electrons are ejected only if the light has sufficient energy
Electronic structure of atoms
 Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
Chapter 1 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 1.1 The wave nature of light Much of our understanding
Chapter 6 Electronic structure of atoms
 Chapter 6 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 6.1 The wave nature of light Visible light is
Chapter 6 Electronic structure of atoms light photons spectra Heisenberg s uncertainty principle atomic orbitals electron configurations the periodic table 6.1 The wave nature of light Visible light is
Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths.
 Section7: The Bohr Atom Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths. Continuous Spectrum Everyone has seen the spectrum produced when white
Section7: The Bohr Atom Earlier we learned that hot, opaque objects produce continuous spectra of radiation of different wavelengths. Continuous Spectrum Everyone has seen the spectrum produced when white
5.111 Lecture Summary #6
 5.111 Lecture Summary #6 Readings for today: Section 1.9 (1.8 in 3 rd ed) Atomic Orbitals. Read for Lecture #7: Section 1.10 (1.9 in 3 rd ed) Electron Spin, Section 1.11 (1.10 in 3 rd ed) The Electronic
5.111 Lecture Summary #6 Readings for today: Section 1.9 (1.8 in 3 rd ed) Atomic Orbitals. Read for Lecture #7: Section 1.10 (1.9 in 3 rd ed) Electron Spin, Section 1.11 (1.10 in 3 rd ed) The Electronic
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY
 THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
THE UNIVERSITY OF QUEENSLAND DEPARTMENT OF PHYSICS PHYS2041 ATOMIC SPECTROSCOPY Warning: The mercury spectral lamps emit UV radiation. Do not stare into the lamp. Avoid exposure where possible. Introduction
The Hydrogen Atom. Dr. Sabry El-Taher 1. e 4. U U r
 The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Hydrogen Atom Atom is a 3D object, and the electron motion is three-dimensional. We ll start with the simplest case - The hydrogen atom. An electron and a proton (nucleus) are bound by the central-symmetric
The Photoelectric Effect
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours cannot be described by classical mechanics.
 A 10-MINUTE RATHER QUICK INTRODUCTION TO QUANTUM MECHANICS 1. What is quantum mechanics (as opposed to classical mechanics)? Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours
A 10-MINUTE RATHER QUICK INTRODUCTION TO QUANTUM MECHANICS 1. What is quantum mechanics (as opposed to classical mechanics)? Quantum mechanics (QM) deals with systems on atomic scale level, whose behaviours
Atomic Structure. Standing Waves x10 8 m/s. (or Hz or 1/s) λ Node
 Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Atomic Structure Topics: 7.1 Electromagnetic Radiation 7.2 Planck, Einstein, Energy, and Photons 7.3 Atomic Line Spectra and Niels Bohr 7.4 The Wave Properties of the Electron 7.5 Quantum Mechanical View
Introduction to Quantum Mechanics (Prelude to Nuclear Shell Model) Heisenberg Uncertainty Principle In the microscopic world,
 Introduction to Quantum Mechanics (Prelude to Nuclear Shell Model) Heisenberg Uncertainty Principle In the microscopic world, x p h π If you try to specify/measure the exact position of a particle you
Introduction to Quantum Mechanics (Prelude to Nuclear Shell Model) Heisenberg Uncertainty Principle In the microscopic world, x p h π If you try to specify/measure the exact position of a particle you
Ch 7 Quantum Theory of the Atom (light and atomic structure)
 Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Ch 7 Quantum Theory of the Atom (light and atomic structure) Electromagnetic Radiation - Electromagnetic radiation consists of oscillations in electric and magnetic fields. The oscillations can be described
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Electromagnetic Radiation All electromagnetic radiation travels at the same velocity: the speed of light (c), m/s.
 Chapter 6 Electronic Structure of Atoms Waves To understand the electronic structure of atoms, one must understand the nature of electromagnetic radiation. The distance between corresponding points on
Chapter 6 Electronic Structure of Atoms Waves To understand the electronic structure of atoms, one must understand the nature of electromagnetic radiation. The distance between corresponding points on
General Physics (PHY 2140)
 General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
General Physics (PHY 140) Lecture 33 Modern Physics Atomic Physics Atomic spectra Bohr s theory of hydrogen http://www.physics.wayne.edu/~apetrov/phy140/ Chapter 8 1 Lightning Review Last lecture: 1. Atomic
Chapter 6 - Electronic Structure of Atoms
 Chapter 6 - Electronic Structure of Atoms 6.1 The Wave Nature of Light To understand the electronic structure of atoms, one must understand the nature of electromagnetic radiation Visible light is an example
Chapter 6 - Electronic Structure of Atoms 6.1 The Wave Nature of Light To understand the electronic structure of atoms, one must understand the nature of electromagnetic radiation Visible light is an example
Electronic Structure of Atoms. Chapter 6
 Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
Electronic Structure of Atoms Chapter 6 Electronic Structure of Atoms 1. The Wave Nature of Light All waves have: a) characteristic wavelength, λ b) amplitude, A Electronic Structure of Atoms 1. The Wave
where n = (an integer) =
 5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
5.111 Lecture Summary #5 Readings for today: Section 1.3 (1.6 in 3 rd ed) Atomic Spectra, Section 1.7 up to equation 9b (1.5 up to eq. 8b in 3 rd ed) Wavefunctions and Energy Levels, Section 1.8 (1.7 in
Chapter 6: Electronic Structure of Atoms
 Chapter 6: Electronic Structure of Atoms Learning Outcomes: Calculate the wavelength of electromagnetic radiation given its frequency or its frequency given its wavelength. Order the common kinds of radiation
Chapter 6: Electronic Structure of Atoms Learning Outcomes: Calculate the wavelength of electromagnetic radiation given its frequency or its frequency given its wavelength. Order the common kinds of radiation
ONE AND MANY ELECTRON ATOMS Chapter 15
 See Week 8 lecture notes. This is exactly the same as the Hamiltonian for nonrigid rotation. In Week 8 lecture notes it was shown that this is the operator for Lˆ 2, the square of the angular momentum.
See Week 8 lecture notes. This is exactly the same as the Hamiltonian for nonrigid rotation. In Week 8 lecture notes it was shown that this is the operator for Lˆ 2, the square of the angular momentum.
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals
 Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
Chem 467 Supplement to Lecture 19 Hydrogen Atom, Atomic Orbitals Pre-Quantum Atomic Structure The existence of atoms and molecules had long been theorized, but never rigorously proven until the late 19
Chapter 37 Early Quantum Theory and Models of the Atom
 Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
298 Chapter 6 Electronic Structure and Periodic Properties of Elements
 98 Chapter 6 Electronic Structure and Periodic Properties of Elements 6. The Bohr Model By the end of this section, you will be able to: Describe the Bohr model of the hydrogen atom Use the Rydberg equation
98 Chapter 6 Electronic Structure and Periodic Properties of Elements 6. The Bohr Model By the end of this section, you will be able to: Describe the Bohr model of the hydrogen atom Use the Rydberg equation
UNIT : QUANTUM THEORY AND THE ATOM
 Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Name St.No. Date(YY/MM/DD) / / Section UNIT 102-10: QUANTUM THEORY AND THE ATOM OBJECTIVES Atomic Spectra for Hydrogen, Mercury and Neon. 1. To observe various atomic spectra with a diffraction grating
Spectroscopy. Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths.
 Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths. In contract, light emitted in low=pressure gas discharge contains only discrete individual wavelengths,
Hot self-luminous objects light the Sun or a light bulb emit a continuous spectrum of wavelengths. In contract, light emitted in low=pressure gas discharge contains only discrete individual wavelengths,
Modern Physics for Scientists and Engineers International Edition, 4th Edition
 Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
Modern Physics for Scientists and Engineers International Edition, 4th Edition http://optics.hanyang.ac.kr/~shsong Review: 1. THE BIRTH OF MODERN PHYSICS 2. SPECIAL THEORY OF RELATIVITY 3. THE EXPERIMENTAL
H atom solution. 1 Introduction 2. 2 Coordinate system 2. 3 Variable separation 4
 H atom solution Contents 1 Introduction 2 2 Coordinate system 2 3 Variable separation 4 4 Wavefunction solutions 6 4.1 Solution for Φ........................... 6 4.2 Solution for Θ...........................
H atom solution Contents 1 Introduction 2 2 Coordinate system 2 3 Variable separation 4 4 Wavefunction solutions 6 4.1 Solution for Φ........................... 6 4.2 Solution for Θ...........................
Experimental Determination of the Mass of a Neutron
 Experimental Determination of the Mass of a Neutron A. Andonian Department of Physics & Astronomy, Bates College, Lewiston, ME 04240, USA (Dated: April 13, 2016) The neutron is fundamental to our understanding
Experimental Determination of the Mass of a Neutron A. Andonian Department of Physics & Astronomy, Bates College, Lewiston, ME 04240, USA (Dated: April 13, 2016) The neutron is fundamental to our understanding
Lecture #21: Hydrogen Atom II
 561 Fall, 217 Lecture #21 Page 1 Lecture #21: Hydrogen Atom II Last time: TISE For H atom: final exactly solved problem Ĥ in spherical polar coordinates Separation: ψ nlml ( r,θ,φ) = R nl (r)y m l (θ,φ)
561 Fall, 217 Lecture #21 Page 1 Lecture #21: Hydrogen Atom II Last time: TISE For H atom: final exactly solved problem Ĥ in spherical polar coordinates Separation: ψ nlml ( r,θ,φ) = R nl (r)y m l (θ,φ)
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
E n = n h ν. The oscillators must absorb or emit energy in discrete multiples of the fundamental quantum of energy given by.
 Planck s s Radiation Law Planck made two modifications to the classical theory The oscillators (of electromagnetic origin) can only have certain discrete energies determined by E n = n h ν with n is an
Planck s s Radiation Law Planck made two modifications to the classical theory The oscillators (of electromagnetic origin) can only have certain discrete energies determined by E n = n h ν with n is an
Quantum Mechanics. Particle in a box All were partial answers, leading Schrödinger to wave mechanics
 Chemistry 4521 Time is flying by: only 15 lectures left!! Six quantum mechanics Four Spectroscopy Third Hour exam Three statistical mechanics Review Final Exam, Wednesday, May 4, 7:30 10 PM Quantum Mechanics
Chemistry 4521 Time is flying by: only 15 lectures left!! Six quantum mechanics Four Spectroscopy Third Hour exam Three statistical mechanics Review Final Exam, Wednesday, May 4, 7:30 10 PM Quantum Mechanics
Gilbert Kirss Foster. Chapter3. Atomic Structure. Explaining the Properties of Elements
 Gilbert Kirss Foster Chapter3 Atomic Structure Explaining the Properties of Elements Chapter Outline 3.1 Waves of Light 3.2 Atomic Spectra 3.3 Particles of Light: Quantum Theory 3.4 The Hydrogen Spectrum
Gilbert Kirss Foster Chapter3 Atomic Structure Explaining the Properties of Elements Chapter Outline 3.1 Waves of Light 3.2 Atomic Spectra 3.3 Particles of Light: Quantum Theory 3.4 The Hydrogen Spectrum
INTRODUCTION TO QUANTUM MECHANICS
 4 CHAPTER INTRODUCTION TO QUANTUM MECHANICS 4.1 Preliminaries: Wave Motion and Light 4.2 Evidence for Energy Quantization in Atoms 4.3 The Bohr Model: Predicting Discrete Energy Levels in Atoms 4.4 Evidence
4 CHAPTER INTRODUCTION TO QUANTUM MECHANICS 4.1 Preliminaries: Wave Motion and Light 4.2 Evidence for Energy Quantization in Atoms 4.3 The Bohr Model: Predicting Discrete Energy Levels in Atoms 4.4 Evidence
We now turn to our first quantum mechanical problems that represent real, as
 84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
84 Lectures 16-17 We now turn to our first quantum mechanical problems that represent real, as opposed to idealized, systems. These problems are the structures of atoms. We will begin first with hydrogen-like
Atoms. Radiation from atoms and molecules enables the most accurate time and length measurements: Atomic clocks
 Atoms Quantum physics explains the energy levels of atoms with enormous accuracy. This is possible, since these levels have long lifetime (uncertainty relation for E, t). Radiation from atoms and molecules
Atoms Quantum physics explains the energy levels of atoms with enormous accuracy. This is possible, since these levels have long lifetime (uncertainty relation for E, t). Radiation from atoms and molecules
Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum. All EM radiation travels at the speed of light, c = 3 x 10 8 m/s
 Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum V I B G Y O R All EM radiation travels at the speed of light, c = 3 x 10 8 m/s Electromagnetic radiation is a wave with a wavelength
Chapter 6: The Electronic Structure of the Atom Electromagnetic Spectrum V I B G Y O R All EM radiation travels at the speed of light, c = 3 x 10 8 m/s Electromagnetic radiation is a wave with a wavelength
Physics 280 Quantum Mechanics Lecture
 Spring 2015 1 1 Department of Physics Drexel University August 3, 2016 Objectives Review Early Quantum Mechanics Objectives Review Early Quantum Mechanics Schrödinger s Wave Equation Objectives Review
Spring 2015 1 1 Department of Physics Drexel University August 3, 2016 Objectives Review Early Quantum Mechanics Objectives Review Early Quantum Mechanics Schrödinger s Wave Equation Objectives Review
Terms to Know. 10.Angular quantum number 11.Magnetic quantum number 12.Spin quantum number
 Terms to Know 1. Photon 2. Atomic emission spectrum 3. Ground state 4. Atomic orbital 5. Aufbau principle 6. Pauli exclusion principle 7. Hunds rule 8. Electron configuration 9. Principle quantum number
Terms to Know 1. Photon 2. Atomic emission spectrum 3. Ground state 4. Atomic orbital 5. Aufbau principle 6. Pauli exclusion principle 7. Hunds rule 8. Electron configuration 9. Principle quantum number
Chapter 6. of Atoms. Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten
 Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall,
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall,
Chapter 6. of Atoms. Waves. Waves 1/15/2013
 Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall,
Chemistry, The Central Science, 10th edition Theodore L. Brown; H. Eugene LeMay, Jr.; and Bruce E. Bursten Chapter 6 John D. Bookstaver St. Charles Community College St. Peters, MO 2006, Prentice Hall,
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
Chapter 28 Atomic Physics Sir Joseph John Thomson J. J. Thomson 1856-1940 Discovered the electron Did extensive work with cathode ray deflections 1906 Nobel Prize for discovery of electron Early Models
10. Wavelength measurement using prism spectroscopy
 Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Optical Spectroscopy and Atomic Structure. PHYS 0219 Optical Spectroscopy and Atomic Structure 1
 Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall Duration: 2h 30m
 Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Final Exam Tuesday, May 8, 2012 Starting at 8:30 a.m., Hoyt Hall. ------------------- Duration: 2h 30m Chapter 39 Quantum Mechanics of Atoms Units of Chapter 39 39-1 Quantum-Mechanical View of Atoms 39-2
Quantum Theory of the Atom
 The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
The Wave Nature of Light Quantum Theory of the Atom Electromagnetic radiation carries energy = radiant energy some forms are visible light, x rays, and radio waves Wavelength ( λ) is the distance between
Early Quantum Theory and Models of the Atom
 Early Quantum Theory and Models of the Atom Electron Discharge tube (circa 1900 s) There is something ( cathode rays ) which is emitted by the cathode and causes glowing Unlike light, these rays are deflected
Early Quantum Theory and Models of the Atom Electron Discharge tube (circa 1900 s) There is something ( cathode rays ) which is emitted by the cathode and causes glowing Unlike light, these rays are deflected
General Chemistry. Contents. Chapter 9: Electrons in Atoms. Contents. 9-1 Electromagnetic Radiation. EM Radiation. Frequency, Wavelength and Velocity
 General Chemistry Principles and Modern Applications Petrucci Harwood Herring 8 th Edition Chapter 9: Electrons in Atoms Philip Dutton University of Windsor, Canada N9B 3P4 Contents 9-1 Electromagnetic
General Chemistry Principles and Modern Applications Petrucci Harwood Herring 8 th Edition Chapter 9: Electrons in Atoms Philip Dutton University of Windsor, Canada N9B 3P4 Contents 9-1 Electromagnetic
A Quantum Mechanical Model for the Vibration and Rotation of Molecules. Rigid Rotor
 A Quantum Mechanical Model for the Vibration and Rotation of Molecules Harmonic Oscillator Rigid Rotor Degrees of Freedom Translation: quantum mechanical model is particle in box or free particle. A molecule
A Quantum Mechanical Model for the Vibration and Rotation of Molecules Harmonic Oscillator Rigid Rotor Degrees of Freedom Translation: quantum mechanical model is particle in box or free particle. A molecule
CHEMISTRY Topic #1: Atomic Structure and Nuclear Chemistry Fall 2017 Dr. Susan Findlay See Exercises 3.1 to 3.3
 CHEMISTRY 1000 Topic #1: Atomic Structure and Nuclear Chemistry Fall 2017 Dr. Susan Findlay See Exercises 3.1 to 3.3 Light: Wave? Particle? Both! Modern models of the atom were derived by studying the
CHEMISTRY 1000 Topic #1: Atomic Structure and Nuclear Chemistry Fall 2017 Dr. Susan Findlay See Exercises 3.1 to 3.3 Light: Wave? Particle? Both! Modern models of the atom were derived by studying the
Electron Arrangement - Part 1
 Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
Brad Collins Electron Arrangement - Part 1 Chapter 8 Some images Copyright The McGraw-Hill Companies, Inc. Properties of Waves Wavelength (λ) is the distance between identical points on successive waves.
Chapter 6. Electronic Structure of Atoms. Lecture Presentation. John D. Bookstaver St. Charles Community College Cottleville, MO
 Lecture Presentation Chapter 6 John D. Bookstaver St. Charles Community College Cottleville, MO Waves To understand the electronic structure of atoms, one must understand the nature of electromagnetic
Lecture Presentation Chapter 6 John D. Bookstaver St. Charles Community College Cottleville, MO Waves To understand the electronic structure of atoms, one must understand the nature of electromagnetic
ATOMIC STRUCTURE. Kotz Ch 7 & Ch 22 (sect 4,5)
 ATOMIC STRUCTURE Kotz Ch 7 & Ch 22 (sect 4,5) properties of light spectroscopy quantum hypothesis hydrogen atom Heisenberg Uncertainty Principle orbitals ELECTROMAGNETIC RADIATION subatomic particles (electron,
ATOMIC STRUCTURE Kotz Ch 7 & Ch 22 (sect 4,5) properties of light spectroscopy quantum hypothesis hydrogen atom Heisenberg Uncertainty Principle orbitals ELECTROMAGNETIC RADIATION subatomic particles (electron,
64-311/5: Atomic and Molecular Spectra
 64-311-Questions.doc 64-311/5: Atomic and Molecular Spectra Dr T Reddish (Room 89-1 Essex Hall) SECTION 1: REVISION QUESTIONS FROM 64-310/14 ε ο = 8.854187817 x 10-1 Fm -1, h = 1.0545766 x 10-34 Js, e
64-311-Questions.doc 64-311/5: Atomic and Molecular Spectra Dr T Reddish (Room 89-1 Essex Hall) SECTION 1: REVISION QUESTIONS FROM 64-310/14 ε ο = 8.854187817 x 10-1 Fm -1, h = 1.0545766 x 10-34 Js, e
Physics 1C Lecture 29A. Finish off Ch. 28 Start Ch. 29
 Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
Physics 1C Lecture 29A Finish off Ch. 28 Start Ch. 29 Particle in a Box Let s consider a particle confined to a one-dimensional region in space. Following the quantum mechanics approach, we need to find
ATOMIC SPECTRA. To identify elements through their emission spectra. Apparatus: spectrometer, spectral tubes, power supply, incandescent lamp.
 ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
ATOMIC SPECTRA Objective: To measure the wavelengths of visible light emitted by atomic hydrogen and verify the measured wavelengths against those predicted by quantum theory. To identify elements through
We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism".
 We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism". -Richard Feynman Quantum H Atom Review Radia Wave Function (1s):
We now realize that the phenomena of chemical interactions, and, ultimately life itself, are to be understood in terms of electromagnetism". -Richard Feynman Quantum H Atom Review Radia Wave Function (1s):
Chapter 1. From Classical to Quantum Mechanics
 Chapter 1. From Classical to Quantum Mechanics Classical Mechanics (Newton): It describes the motion of a classical particle (discrete object). dp F ma, p = m = dt dx m dt F: force (N) a: acceleration
Chapter 1. From Classical to Quantum Mechanics Classical Mechanics (Newton): It describes the motion of a classical particle (discrete object). dp F ma, p = m = dt dx m dt F: force (N) a: acceleration
Particle nature of light & Quantization
 Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Chemistry 483 Lecture Topics Fall 2009
 Chemistry 483 Lecture Topics Fall 2009 Text PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon A. Background (M&S,Chapter 1) Blackbody Radiation Photoelectric effect DeBroglie Wavelength Atomic
Chemistry 483 Lecture Topics Fall 2009 Text PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon A. Background (M&S,Chapter 1) Blackbody Radiation Photoelectric effect DeBroglie Wavelength Atomic
Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures
 Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures Jeong Won Kang Department of Chemical Engineering Korea University Subjects Three Basic Types of Motions
Applied Statistical Mechanics Lecture Note - 3 Quantum Mechanics Applications and Atomic Structures Jeong Won Kang Department of Chemical Engineering Korea University Subjects Three Basic Types of Motions
The early periodic table based on atomic weight. (Section 5.1) Lets review: What is a hydrogen atom? 1 electron * nucleus H 1 proton
 PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
PERIODICITY AND ATOMIC STRUCTURE CHAPTER 5 How can we relate the structure of the atom to the way that it behaves chemically? The process of understanding began with a realization that many of the properties
Chapter 27 Early Quantum Theory and Models of the Atom Discovery and Properties of the electron
 Chapter 27 Early Quantum Theory and Models of the Atom 27-1 Discovery and Properties of the electron Measure charge to mass ratio e/m (J. J. Thomson, 1897) When apply magnetic field only, the rays are
Chapter 27 Early Quantum Theory and Models of the Atom 27-1 Discovery and Properties of the electron Measure charge to mass ratio e/m (J. J. Thomson, 1897) When apply magnetic field only, the rays are
ATOMIC STRUCRURE
 ATOMIC STRUCRURE Long Answer Questions: 1. What are quantum numbers? Give their significance? Ans. The various orbitals in an atom qualitatively distinguished by their size, shape and orientation. The
ATOMIC STRUCRURE Long Answer Questions: 1. What are quantum numbers? Give their significance? Ans. The various orbitals in an atom qualitatively distinguished by their size, shape and orientation. The
Sharif University of Technology Physics Department. Modern Physics Spring 2016 Prof. Akhavan
 Sharif University of Technology Physics Department Modern Physics Spring 2016 Prof. Akhavan Problems Set #5. Due on: 03 th of April / 15 th of Farvardin. 1 Blackbody Radiation. (Required text book is Modern
Sharif University of Technology Physics Department Modern Physics Spring 2016 Prof. Akhavan Problems Set #5. Due on: 03 th of April / 15 th of Farvardin. 1 Blackbody Radiation. (Required text book is Modern
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE Brooks/Cole - Thomson
 Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
Chapter 7 QUANTUM THEORY & ATOMIC STRUCTURE 1 7.1 The Nature of Light 2 Most subatomic particles behave as PARTICLES and obey the physics of waves. Light is a type of electromagnetic radiation Light consists
1.6. Quantum mechanical description of the hydrogen atom
 29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
29.6. Quantum mechanical description of the hydrogen atom.6.. Hamiltonian for the hydrogen atom Atomic units To avoid dealing with very small numbers, let us introduce the so called atomic units : Quantity
CHEM Course web page. Outline for first exam period
 CHEM 3 Course web page http://web.chemistry.gatech.edu/~barefield/3/chem3a.html Outline for first exam period Atomic structure and periodic properties Structures and bonding models for covalent compounds
CHEM 3 Course web page http://web.chemistry.gatech.edu/~barefield/3/chem3a.html Outline for first exam period Atomic structure and periodic properties Structures and bonding models for covalent compounds
Lecture 10. Central potential
 Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
Lecture 10 Central potential 89 90 LECTURE 10. CENTRAL POTENTIAL 10.1 Introduction We are now ready to study a generic class of three-dimensional physical systems. They are the systems that have a central
Georgia Institute of Technology CHEM 1310 revised 10/8/09 Spring The Development of Quantum Mechanics. ν (nu) = frequency (in s -1 or hertz)
 The Development of Quantum Mechanics Early physicists used the properties of electromagnetic radiation to develop fundamental ideas about the structure of the atom. A fundamental assumption for their work
The Development of Quantum Mechanics Early physicists used the properties of electromagnetic radiation to develop fundamental ideas about the structure of the atom. A fundamental assumption for their work
OpenStax-CNX module: m The Bohr Model. OpenStax College. Abstract
 OpenStax-CNX module: m51039 1 The Bohr Model OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By the end of this section, you will
OpenStax-CNX module: m51039 1 The Bohr Model OpenStax College This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 4.0 By the end of this section, you will
Chemistry 881 Lecture Topics Fall 2001
 Chemistry 881 Lecture Topics Fall 2001 Texts PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon MATHEMATICS for PHYSICAL CHEMISTRY, Mortimer i. Mathematics Review (M, Chapters 1,2,3 & 4; M&S,
Chemistry 881 Lecture Topics Fall 2001 Texts PHYSICAL CHEMISTRY A Molecular Approach McQuarrie and Simon MATHEMATICS for PHYSICAL CHEMISTRY, Mortimer i. Mathematics Review (M, Chapters 1,2,3 & 4; M&S,
Relativistic corrections of energy terms
 Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
Energy levels and atomic structures lectures chapter one
 Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of hydrogen atom.
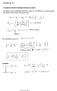 Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Lecture 20-21 Page 1 Lectures 20-21 Transitions between hydrogen stationary states The energy of the emitted light (photons) is given by the difference in energy between the initial and final states of
Chapter 39. Particles Behaving as Waves
 Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
Chapter 39 Particles Behaving as Waves 39.1 Electron Waves Light has a dual nature. Light exhibits both wave and particle characteristics. Louis de Broglie postulated in 1924 that if nature is symmetric,
( ( ; R H = 109,677 cm -1
 CHAPTER 9 Atomic Structure and Spectra I. The Hydrogenic Atoms (one electron species). H, He +1, Li 2+, A. Clues from Line Spectra. Reminder: fundamental equations of spectroscopy: ε Photon = hν relation
CHAPTER 9 Atomic Structure and Spectra I. The Hydrogenic Atoms (one electron species). H, He +1, Li 2+, A. Clues from Line Spectra. Reminder: fundamental equations of spectroscopy: ε Photon = hν relation
Quantum Mechanics & Atomic Structure (Chapter 11)
 Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
Quantum Mechanics & Atomic Structure (Chapter 11) Quantum mechanics: Microscopic theory of light & matter at molecular scale and smaller. Atoms and radiation (light) have both wave-like and particlelike
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
Chemistry 3502/4502 Final Exam Part I May 14, 2005 1. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle (e) The
12/04/2012. Models of the Atom. Quantum Physics versus Classical Physics The Thirty-Year War ( )
 Quantum Physics versus Classical Physics The Thirty-Year War (1900-1930) Interactions between Matter and Radiation Models of the Atom Bohr s Model of the Atom Planck s Blackbody Radiation Models of the
Quantum Physics versus Classical Physics The Thirty-Year War (1900-1930) Interactions between Matter and Radiation Models of the Atom Bohr s Model of the Atom Planck s Blackbody Radiation Models of the
Chapter 4. Table of Contents. Section 1 The Development of a New Atomic Model. Section 2 The Quantum Model of the Atom
 Arrangement of Electrons in Atoms Table of Contents Section 1 The Development of a New Atomic Model Section 2 The Quantum Model of the Atom Section 3 Electron Configurations Section 1 The Development of
Arrangement of Electrons in Atoms Table of Contents Section 1 The Development of a New Atomic Model Section 2 The Quantum Model of the Atom Section 3 Electron Configurations Section 1 The Development of
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model
 Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Today: general condition for threshold operation physics of atomic, vibrational, rotational gain media intro to the Lorentz model Laser operation Simplified energy conversion processes in a laser medium:
Indicate if the statement is True (T) or False (F) by circling the letter (1 pt each):
 Indicate if the statement is (T) or False (F) by circling the letter (1 pt each): False 1. In order to ensure that all observables are real valued, the eigenfunctions for an operator must also be real
Indicate if the statement is (T) or False (F) by circling the letter (1 pt each): False 1. In order to ensure that all observables are real valued, the eigenfunctions for an operator must also be real
Physics 228 Today: Ch 41: 1-3: 3D quantum mechanics, hydrogen atom
 Physics 228 Today: Ch 41: 1-3: 3D quantum mechanics, hydrogen atom Website: Sakai 01:750:228 or www.physics.rutgers.edu/ugrad/228 Happy April Fools Day Example / Worked Problems What is the ratio of the
Physics 228 Today: Ch 41: 1-3: 3D quantum mechanics, hydrogen atom Website: Sakai 01:750:228 or www.physics.rutgers.edu/ugrad/228 Happy April Fools Day Example / Worked Problems What is the ratio of the
Semiconductor Physics and Devices
 Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
Introduction to Quantum Mechanics In order to understand the current-voltage characteristics, we need some knowledge of electron behavior in semiconductor when the electron is subjected to various potential
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS
 QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
QUANTUM MECHANICS SECOND EDITION G. ARULDHAS Formerly, Professor and Head of Physics and Dean, Faculty of Science University of Kerala New Delhi-110001 2009 QUANTUM MECHANICS, 2nd Ed. G. Aruldhas 2009
Chemistry 3502/4502. Final Exam Part I. May 14, 2005
 Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Advocacy chit Chemistry 350/450 Final Exam Part I May 4, 005. For which of the below systems is = where H is the Hamiltonian operator and T is the kinetic-energy operator? (a) The free particle
Chapter 9: Electrons in Atoms
 General Chemistry Principles and Modern Applications Petrucci Harwood Herring 8 th Edition Chapter 9: Electrons in Atoms Philip Dutton University of Windsor, Canada N9B 3P4 Prentice-Hall 2002 Prentice-Hall
General Chemistry Principles and Modern Applications Petrucci Harwood Herring 8 th Edition Chapter 9: Electrons in Atoms Philip Dutton University of Windsor, Canada N9B 3P4 Prentice-Hall 2002 Prentice-Hall
Any first year text, sections on atomic structure, spectral lines and spectrometers
 Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Chapter 12: Phenomena
 Chapter 12: Phenomena K Fe Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected
Chapter 12: Phenomena K Fe Phenomena: Different wavelengths of electromagnetic radiation were directed onto two different metal sample (see picture). Scientists then recorded if any particles were ejected
Physics 401: Quantum Mechanics I Chapter 4
 Physics 401: Quantum Mechanics I Chapter 4 Are you here today? A. Yes B. No C. After than midterm? 3-D Schroedinger Equation The ground state energy of the particle in a 3D box is ( 1 2 +1 2 +1 2 ) π2
Physics 401: Quantum Mechanics I Chapter 4 Are you here today? A. Yes B. No C. After than midterm? 3-D Schroedinger Equation The ground state energy of the particle in a 3D box is ( 1 2 +1 2 +1 2 ) π2
