X-ray spectroscopy. Crystal spectrometers. Reflection gratings. Transmission gratings. Solid state detectors
|
|
|
- Dortha Austin
- 5 years ago
- Views:
Transcription
1 X-ray spectroscopy Crystal spectrometers Reflection gratings Transmission gratings Solid state detectors
2 Bragg s law
3 How are X-rays reflected from a surface? At short wavelengths, mirrors do not work well. Reflectivity of all materials becomes nearly zero at energies beyond 30 ev for normal incidence. At grazing incidence, reflectivity is maintained up to much higher energies. Spectrometers have to use as few reflections as possible.
4 How are X-rays reflected from a surface? Reflection of x rays from a surface is caused by the electrons which give it a refractive index n<1 at x ray wavelengths (solids have n>1at optical wavelengths). X rays striking vacuum-metal interfaces are going from optically moredense to optically less-dense media. In these circumstances total internal reflection of the light can occur. If the incidence angle is great enough (or the grazing angle is small enough) the X-rays are reflected from the surface. Value of the critical angle depends on the electron density of the mirror material. This number is particularly large for gold, platinum, iridium Reflectivity of a gold mirror Incidence angle 90 o 2 o 0.5 o Photon energy (ev)
5 Multilayer optics for x-rays Reflectivity of X-rays from a solid surface can be enhanced by using multilayers with nanometer spacing. Change of refractive index at each interface causes a partial reflection Interference effects enhance constructively total reflection efficiency Reflectivity Wavelength (nm) Reflectivity of molybdenum/silicon multilayer mirror 40 layers 6.9 nm period Reflectivity is tailored to optimize a certain wavelength of interest
6 Multilayer optics for x-rays Curved mirror geometries are also possible X-ray analysis of chemical elements in microscopic samples
7 Crystal x-ray spectrometers Some types: Von Hamos (cylindrical) Dumond (crystal in transmission) Johann (cylindrical) High resolution spectra (up to R=20000) can be obtained.
8 The Rowland mounting a l 1 detector slit l 2 grating Geometry suggested by Rowland in Combines the focusing properties of a spherical mirror with the diffraction properties of a grating. If a source and the grating are placed on a circle with half the radius R of the grating, all wavelengths will be focussed on the same circle: The Rowland circle.
9 Von Hamos crystal x-ray spectrometer By bending crystals in sophisticated ways, focusing and imaging of x-ray sources can be achieved.
10 Transmission crystal x-ray spectrometer Dumond geometry: crystal works in transmission not affected by refractive index of crystal material application for hard x-rays, which are transmitted through crystal and partially reflected by many lattice layers
11 Johann x-ray spectrometer
12 The equivalence of mass and energy The direct test of Einstein s equation is based on the prediction that when a nucleus captures a neutron, the resulting isotope (mass number A+1) is somewhat lighter than the sum of the masses of the original nucleus (mass number A) and the free neutron (mass number 1). The energy equivalent to this mass difference is emitted as a spectra of gamma-rays. The mass difference in Einstein s equation using two silicon isotopes Si and two sulphur isotopes S has been measured with very high accuracy at the MIT, using a novel experimental technique.
13 The equivalence of mass and energy The mass differences betweeen the nuclei involved in the reaction are converted into photon energy. The masses are weighted in a Penning trap. The photon energy is measured with the crystal spectrometer: The quantity 1-Δmc 2 /E = (-1.4 +/- 4.4) 10-7 is obtained. Equivalence of mass and energy tested to a level of at least %.
14 Mass measurement at the MIT Penning trap The mass difference Δm is measured by simultaneous comparisons of the cyclotron frequencies (inversely proportional to the mass) of ions of the initial and final isotopes confined over a period of weeks in a Penning trap. (S. Rainville et al., Science 2004)
15 Wavelength measurement at ILL The energies of the gamma-rays emitted after neutron capture by both silicon and sulphur have been measured at the using the world s highest resolution gamma-ray interferometer GAMS4. Precision of better than 5 parts in
16 Wavelength measurement at ILL High angular resolution is obtained by using high precision optical interferometers. The crystals are mounted on interferometer arms which can be rotated in angular steps as small as arcsec (10-7 or rad).
17 Measurement of the 788 kev transition in 36 Cl.
18 Data for testing E=mc 2 a) Wavelength data and fitted curves. The energy of the emitted 5,421-keV γ- rays is measured from the angular separation of the second-order Bragg peaks resulting from diffraction from a silicon crystal. b) Measured mass ratio M[ 33 S]/M[ 32 SH] (which largely determines the mass change, m) as a function of the distance between the ions, s, in the Penning trap. The final mass ratio (solid line) is determined with fractional accuracy of 7 parts in (dashed lines). Simon Rainville et al., Nature 438 (2005) 1096
19 Flat crystal x-ray spectrometer
20 Absolute x-ray spectroscopy light beams laser crystal A novel methode removes the largest error sources found in crystal spectrometers x-ray source (EBIT) mirror fiducial 2 x-ray line fiducial 1 CCD Bruhns, Braun et al., Rev. Sci. Inst. 76, (2005)
21 Laboratory setup
22 Absolute measurements CCD 2 Γ = 180-2Θ 180-2Θ θ EBIT a/b θ crystal 180-2Θ ξ CCD 1 Bond method (W.L. Bond, Acta Cryst. 13, 814 (1960))
23 The Lyman-α spectrum of hydrogenic Ar
24 Hydrogenic standards? Current x-ray standards impaired by satellites Calibration line Fe Kα (1 ppm) Best HCI line elsewhere Ar 17+ Ly α1 (5 ppm) Counts Wavelength (pm) satellites
25 The Lyman-alpha spectrum of hydrogenic Ar ~2 ev
26 The Lyman-alpha spectrum of hydrogenic Ar
27 The He-like S 14+ spectrum Relative measurements Heidelberg EBIT Absolute measurements
28 Two-electron QED Contributions to ground state in He-like Ar ev 5 ev 1 ev + + h. o. experimental error bar: ± 0.01 ev E nuclear recoil 0.06 ev higher orders ev 0.1 ev
29 Lamb shift in Li-like Bi 80+ Measurement of the Bi 80+ 2s 1/2-2p 3/2 transition energy Hyperfine splitting of the F=4,5 levels
30 Lamb shift in Li-like Bi 80+ Hyperfine splitting of the F=4,5 levels
31 Grazing incidence spectrometer
32 Reflection grating spectrum
33
34 Seya-Namioka VUV spectrometer The Seya-Namioka mounting is used at wavelengths > 30 nm for which reflectivity at normal incidence is still sufficient Special case of the Rowland mounting with an acceptable spectral resolution. It is widely used in plasma diagnostics and at synchrotrons.
35 Transmission gratings for x-rays Height of grating structure has a strong effect on grating efficiency by imposing an additional diffrection condition The HETG gratings have a period of 0.2μm or 2000Å for the high-energy gratings, and 0.4μm or 4000Å, for the medium energy gratings. Gratings with grooves/mm (1000Å) are used already
36 A pinhole transmission grating spectrometer two spectra X-ray experiment with a two pinhole grating (a and b) spectrograph. PFS: plasma focus x-ray source C: capacitor for energy storage to feed discharge SH: shutter PH: pinhole to collimate x rays PG: pinhole gratings
37 Grazing incidence soft-x-ray spectrometer At wavelengths shorter than 30 nm, small grazing angles are needed. Detectors are mounted on the Rowland circle and moved under vacuum with bellows. Curved photographic plates and MCPs can also be used.
38 Grazing incidence imaging optics Kirkpatrik-Baez geometry uses two cylindrical x-ray mirrors at grazing incidence Each of the two cylindrical mirrors focuses in one coordinate.
39 Wolter telescope Grazing incidence mirror telescope for x rays in satellites Uses concentric conic surfaces with a single common point Paraboloid-hyperboloid combination with two internal reflections.
40 Wolter telescope and Rowland spectrometer After focusing by mirror, transmission or reflection gratings are used to add spectral dispersion when needed This spectrograph produces images in individual wavelengths
41 Chandra s Wolter telescope
42 Spectral imaging with XMM and Chandra Composite image Images of a supernova remnant at different soft x-ray wavelengths
43 Space based x-ray telescopes Coronal Diagnostic Spectrometer (CDS) on SOHO (ESA/NASA Solar and Heliospheric Observatory) Two compementary systems; the Normal Incidence Spectrometer (NIS) and the Grazing Incidence Spectrometer (GIS). Spectral range of operation
44 Diagnostics of fusion plasmas with VUV and x-rays The diagnostic of temperature, density, plasma composition, radiative losses material transport in fusion reactors in tokamaks requires the use of many spectrometers
45 The spectrum of hydrogen One needs three quantum numbers to define the state of a hydrogen (hydrogen-like) atom: n = 1, 2, 3 (principal) l = 0,, n-1 (orbital) m = -l,, +l (magnetic) The energy depends only on the principal quantum number n: E n 2 Z = 2n 2 ψ ( r) = ψ ( r, θ, ϕ) = R ( r) Y ( θ, ϕ) Energy (au) 0-1/18-1/8 3s (n=3, l=0) 2s (n=2, l=0).... nl 3p (n=3, l=1) 2p (n=2, l=1) lm free electron bound electron 3d (n=3, l=2) i.e. in a non-relativistic theory the (l,m) states are degenerate! -1/2 1s (n=1, l=0)
46 Dirac theory E nj = mc 2 1+ n j + 1/ 2 + Zα ( j + 1/ 2) 2 ( Zα) mc2 Positive continuum bound states e - n: principle quantum number j: total angular momentum α: fine structure constant- α = 1/ ) mc 2 : electron rest mass (511 kev) 0 Dirac-Theorie: (complete relativistic treatment of single electron systems) - mc 2 All levels with the same n and j are degenerate. Negative energy continuum e +
47 The Lamb shift Experiment: Lamb-Retherford 1947; Theoretical Explanation: Bethe 1947 Experiment proves that 2s 1/2 and 2p 1/2 are not degenerate Contradicts Dirac theory 2s 1/2 2E1 1s 1/2 2p 3/2 Lyα 1 (E1) Lyα 2 (E1) 2p 1/2 Lamb shift (LS) includes all deviations from the Dirac theory for a point like nucleus Consequence of the interaction of the bound electron with its own radiation field (self energy). Level scheme for hydrogen
48 The effects of QED on the binding energy increase with the strength of the electric field. s electrons are most affected since their. Lamb shift (three effects) QED Self-energy Self energy Bohr-trajectory Vacuum polarization Vacuum polarization Z Sizeof thenucleus
49 Laser spectroscopy of hydrogen relative accuracy laser spectroscopy frequency measurements year
50 Lamb shift: An effect of strong fields QED corrections to binding energy ΔE scale as: ΔE Z 4 /n s 2s Z: nuclear charge n: principal quantum number ψ Probability density in the region of highest field gradients is essential r (r) nucleus 2p 1/2 2p 3/ , radius [fm]
51 Average field strength of 1s electron <E> (V/cm) s changes by six orders of magnitude H-like U H atom Nuclear charge Z
52 Self energy (SE) Self-energy: Describes the emission and reabsorption of a virtual photon (Bethe 1947) The self energy decreases the binding energy. virtual photon electron in the outer field (Coulomb field of the nucleus) Mass renormalization m = me m m e m: measurable electron mass. It already includes the radiation correction. The self-energy decreases the binding energy. Classical theory: infinitely high mass corresponding to an infinitely high self energy. First calculations for H were made by Bethe based on an idea of Kramer: mass renormalization. The interaction with virtual photon modifies the electron mass. Measured electron mass m already includes this radiation correction. The self-energy correction for a bound state is equivalent to mass difference between the bound electron and a non-interacting free electron of the mass m e. m electron mass of the free electron without interaction with a field.
53 Vacuum Polarization (VP) Virtual creation and annihilation of an e + e pair VP acts attractive
54 Summary of Lamb shift contributions ΔΕ=α/π (αz) 4 F(αZ) m e c 2 Self energy Vacuum polarization s Lamb Shift, ΔE / Z 4 [mev] self energy (-) vacuum polarization Lamb- Shift nuclear size Individual corrections in detail 2p 1/2 1s 1 /2 Fine structure 2sLS 1sLS Hydrogen, H 10.2 ev 45.4 μev 4.4 μev 35.6 μev H-like uranium, U kev 4.56 kev 75.3 ev ev nuclear charge number, Z
55 Bound-State QED: 1s Lamb Shift at High-Z Sum of all corrections, leading to deviations from the Dirac theory for a point-like nucleus Self energy Vacuum polarization U 92+ SE VP NS ev ev ev Goal: ΔΕ=α/π (αz) 4 F(αZ) m e c 2 ±1 ev Low Z-Regime: αz << 1 F(αZ): series expansion in αz High Z-Regime: αz 1 F(αZ): series expansion in αz not appropriate
56 QED correction in second order α SE SE VP VP SE VP S(VP)E
57 GSI accelerator gacility UNILAC ESR 11.4 MeV/u U MeV/u U 92+ up to 1000 MeV/u U 92+ SIS
58 U 91+ X-Ray Spectroscopy at the ESR Storage Ring Storage rings for heavy ions particle detection U 92+ U MeV/u ESR (GSI, Darmstadt) 48º Ge(i) 90º 132º 48º GAS JET Elektronenkühlker particle detection U 91+ Ge(i) Operation parameters v/c = β 0.65 Revolution frequency f 10 6 s -1 Circumfence: 108 m Number of ions: 10 8 Production of characteristic x-rays by electron capture into bare ions (electron cooler or jet-target)
59 ESR Storage Ring
60 Storage rings: cooled ion beams electron collector electron gun electron collector electron gun high voltage platform high voltage platform magnetic field electron beam ion beam magnetic field electron beam ion beam
61 Electron cooling in storage rings I: ma U: kv Ions Electrons Ions Properties of the cold ions: Momentum spread Δp/p : Beam diameter 2 mm Ions interact 10 6 times per second with a collinear beam of cold electrons at nearly the same speed: cooling in the cold electron bath. The transversal components of the ion motion are efficiently cooled.
62 The effect of cooling before cooling after cooling ion intensity rel. ion velocity v/v 0 Ions interact 10 6 times per second with collinear beam of cold electrons Propertiesof thecoldionbeam Momentum spread Δp/p : Beamdiameter 2 mm
63 Merged-beams kinematics 17 ev Relative energy, E rel (ev) ev Electron energy, E e (ev) Provides precise access to low relative collision energies m e E cool = m E ion ion relative velocity: v rel = v e v ion relative energy: E rel = ( E e - E cool ) 2
64 The GSI Electron Cooler Electron Cooler 2.5 m interaction zone Voltage: 5 to 200 kv Current: 10 to 1000 ma
65 Dielectronic recombination: SR technique electron beam cathode voltage U cath ion beam recombination detector electron cooler merged-beams rate coefficient: α = σv U cath meas time cool
66 Experimental challenges Relativistic Doppler transformation E lab Eproj = γ (1 β cosθ lab E lab : Photon energy in the laboratory system E proj : Photon energy in the emitter system ) E lab /E proj MeV/u (β =0.69) 220 MeV/u (β =0.59) 68 MeV/u (β =0.36) 49 MeV/u (β =0.31) Doppler correction: Strong dependence on the velocity and the observation angle θ LAB observation anglel, θ lab [deg] 1 γ = ; β = 1 β 2 v c
67 X-Ray Spectroscopy at the ESR Storage Ring Injection Energy 400 MeV/u deceleration 10 MeV/u Excited states are produced by electron capture (gas jet target) / recombination (electron target)
68 0 o Spectroscopy at the Electron Cooler After capture of one electron by a U 92+, a photon is emitted and detected 600 H-like Uranium Lyα 1 Dipole Magnet Coincidence with the downcharged projectile (U 91+ ) reduces background counts Balmer L-RR j=1/2 j=3/2 Lyα 2 K-RR Energy [kev] Blue shift has its maximum β 0.29 E lab 1.43 E proj Δθ LAB not critical, almost no Doppler width Uncertainty caused by Δβ has its maximum
69 Ground state Lamb shift in H-like uranium 200 Lyα 1 Counts Lyα 2 50 Lyβ K-RR Photon energy (kev) 1s Lamb shift in U ±2.3±3.5 ev statistical 4.6 ev uncertainty in β
70 Ground state Lamb shift in H-like uranium 2s 1/2 M1 2p 3/2 Lyα 1 (E1) 2p 1/2 Lyα 2 (E1) counts Lyα 2 Lyα 1 1s Lamb shift 1s 1/2 50 2p 3/2 B. E. Lyβ photon energy [kev] K-RR Presently most accurate test of the bound-state QED for one-electron systems in the regime of strong fields carried out at the ESR.
71 Test of quantum electrodynamics 1s Lamb shift in H-like uranium counts Lyα 1 Lyα 2 K-RR Experiment: ev ± 4.6 ev Theory: ev 2s 1/2 M1 2p 3/2 Lyα 1 (E1) 2p 1/2 Lyα 2 (E1) photon energy [kev] 1s Lamb shift 1s 1/2 Research Highlights Nature 435, (16 June 2005) Lamb Shift [ev] U 91+ Gasjet Cooler Decelerated Ions: Jet Decelerated Ions: Cooler (our exp.) Year Theory A. Gumberidze PhD thesis 2003, PRL 94, (2005)
free electron plus He-like ion
 free electron plus He-like ion E e I p,n E 2 E 1 ΔE=E e +I p,n aber: ΔE=E 2 -E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012 2011-11-16 1 dielectronic recombination E 2 E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012
free electron plus He-like ion E e I p,n E 2 E 1 ΔE=E e +I p,n aber: ΔE=E 2 -E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012 2011-11-16 1 dielectronic recombination E 2 E 1 n n n n n n=1 n=2 n=3 AAMOP 2011-2012
Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra
 Modern Atomic Physics: Experiment and Theory Preliminary plan of the lectures Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra Lecture 3 April 9 th, 014 1 15.04.015 Preliminary
Modern Atomic Physics: Experiment and Theory Preliminary plan of the lectures Experiments Lamb shift, hyperfine structure, quasimolecules and MO spectra Lecture 3 April 9 th, 014 1 15.04.015 Preliminary
Balmer- and L-shell series of highly charged uranium in the experimental storage ring
 Balmer- and L-shell series of highly charged uranium in the experimental storage ring Summer student program @ GSI - 2007 Thomas Burschil Johann Wolfgang von Goethe-University, Frankfurt/Main 09/24/2007
Balmer- and L-shell series of highly charged uranium in the experimental storage ring Summer student program @ GSI - 2007 Thomas Burschil Johann Wolfgang von Goethe-University, Frankfurt/Main 09/24/2007
Experiments with hydrogen - discovery of the Lamb shift
 Experiments with hydrogen - discovery of the Lamb shift Haris Ðapo Relativistic heavy ion seminar, October 26, 2006 Outline 1 Pre-Lamb experiment The beginning (Bohr s formula) Fine structure (Dirac s
Experiments with hydrogen - discovery of the Lamb shift Haris Ðapo Relativistic heavy ion seminar, October 26, 2006 Outline 1 Pre-Lamb experiment The beginning (Bohr s formula) Fine structure (Dirac s
Production of HCI with an electron beam ion trap
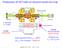 Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Production of HCI with an electron beam ion trap I=450 ma E= 5 kev axially: electrodes radially: electron beam space charge total trap potential U trap 200 V (U trap ion charge) 10000 ev 15000 A/cm 2 n
Lecture 4. Beyound the Dirac equation: QED and nuclear effects
 Lecture 4 Beyound the Dirac equation: QED and nuclear effects Plan of the lecture Reminder from the last lecture: Bound-state solutions of Dirac equation Higher-order corrections to Dirac energies: Radiative
Lecture 4 Beyound the Dirac equation: QED and nuclear effects Plan of the lecture Reminder from the last lecture: Bound-state solutions of Dirac equation Higher-order corrections to Dirac energies: Radiative
This work was performed under the auspices of the U.S. Department of Energy by Lawrence Livermore National Laboratory under contract
 This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
This work was performed under the auspices of the U.S. Department of Energy by under contract DE-AC52-7NA27344. Lawrence Livermore National Security, LLC The ITER tokamak Tungsten (W) is attractive as
Development of a detector setup to determine the 2s hyperfine transition of 209 Bi 80+ at the Experimental Storage Ring at GSI
 Denis Anielski 28.01.2011 1 Development of a detector setup to determine the 2s hyperfine transition of 209 Bi 80+ at the Experimental Storage Ring at GSI Denis Anielski Westfälische Wilhelms-Universität
Denis Anielski 28.01.2011 1 Development of a detector setup to determine the 2s hyperfine transition of 209 Bi 80+ at the Experimental Storage Ring at GSI Denis Anielski Westfälische Wilhelms-Universität
The World s Smallest Extreme Laboratories:
 The World s Smallest Extreme Laboratories: Probing QED with Highly Charged Ions. Joan Marler Clemson University Outline About HCIs About Ion trapping Precision spectroscopy with HCIs Highly Charged Ions
The World s Smallest Extreme Laboratories: Probing QED with Highly Charged Ions. Joan Marler Clemson University Outline About HCIs About Ion trapping Precision spectroscopy with HCIs Highly Charged Ions
Rb, which had been compressed to a density of 1013
 Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
Modern Physics Study Questions for the Spring 2018 Departmental Exam December 3, 2017 1. An electron is initially at rest in a uniform electric field E in the negative y direction and a uniform magnetic
X-Ray Photoelectron Spectroscopy (XPS)
 X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect was enunciated
X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect was enunciated
Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics
 Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics Andrey Surzhykov Technische Universität Braunschweig Physikalisch-Technische Bundesanstalt (PTB) Lecture 1
Physics of heavy multiply-charged ions: Studies on the borderile of atomic and nuclear physics Andrey Surzhykov Technische Universität Braunschweig Physikalisch-Technische Bundesanstalt (PTB) Lecture 1
PHYS 4 CONCEPT PACKET Complete
 PHYS 4 CONCEPT PACKET Complete Written by Jeremy Robinson, Head Instructor Find Out More +Private Instruction +Review Sessions WWW.GRADEPEAK.COM Need Help? Online Private Instruction Anytime, Anywhere
PHYS 4 CONCEPT PACKET Complete Written by Jeremy Robinson, Head Instructor Find Out More +Private Instruction +Review Sessions WWW.GRADEPEAK.COM Need Help? Online Private Instruction Anytime, Anywhere
X- & γ-ray Instrumentation
 X- & γ-ray Instrumentation Used nuclear physics detectors Proportional Counters Scintillators The Dark Ages Simple collimators HEAO A1 & A2: 2 x 8 degree field of view Confusion limit is about 200 sources
X- & γ-ray Instrumentation Used nuclear physics detectors Proportional Counters Scintillators The Dark Ages Simple collimators HEAO A1 & A2: 2 x 8 degree field of view Confusion limit is about 200 sources
Diffraction Gratings, Atomic Spectra. Prof. Shawhan (substituting for Prof. Hall) November 14, 2016
 Diffraction Gratings, Atomic Spectra Prof. Shawhan (substituting for Prof. Hall) November 14, 2016 1 Increase number of slits: 2 Visual Comparisons 3 4 8 2 Diffraction Grating Note: despite the name, this
Diffraction Gratings, Atomic Spectra Prof. Shawhan (substituting for Prof. Hall) November 14, 2016 1 Increase number of slits: 2 Visual Comparisons 3 4 8 2 Diffraction Grating Note: despite the name, this
Chapter Six: X-Rays. 6.1 Discovery of X-rays
 Chapter Six: X-Rays 6.1 Discovery of X-rays In late 1895, a German physicist, W. C. Roentgen was working with a cathode ray tube in his laboratory. He was working with tubes similar to our fluorescent
Chapter Six: X-Rays 6.1 Discovery of X-rays In late 1895, a German physicist, W. C. Roentgen was working with a cathode ray tube in his laboratory. He was working with tubes similar to our fluorescent
Relativistic corrections of energy terms
 Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
Lectures 2-3 Hydrogen atom. Relativistic corrections of energy terms: relativistic mass correction, Darwin term, and spin-orbit term. Fine structure. Lamb shift. Hyperfine structure. Energy levels of the
APEX CARE INSTITUTE FOR PG - TRB, SLET AND NET IN PHYSICS
 Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
Page 1 1. Within the nucleus, the charge distribution A) Is constant, but falls to zero sharply at the nuclear radius B) Increases linearly from the centre, but falls off exponentially at the surface C)
Nuclear Physics. (PHY-231) Dr C. M. Cormack. Nuclear Physics This Lecture
 Nuclear Physics (PHY-31) Dr C. M. Cormack 11 Nuclear Physics This Lecture This Lecture We will discuss an important effect in nuclear spectroscopy The Mössbauer Effect and its applications in technology
Nuclear Physics (PHY-31) Dr C. M. Cormack 11 Nuclear Physics This Lecture This Lecture We will discuss an important effect in nuclear spectroscopy The Mössbauer Effect and its applications in technology
Bannerman High School Physics Department. Making Accurate Statements. Higher Physics. Quanta and Waves
 Bannerman High School Physics Department Making Accurate Statements Higher Physics Quanta and Waves Mandatory Key Area: Particle Physics 1. Use your knowledge of physics to estimate the ratio of the smallest
Bannerman High School Physics Department Making Accurate Statements Higher Physics Quanta and Waves Mandatory Key Area: Particle Physics 1. Use your knowledge of physics to estimate the ratio of the smallest
THE NATURE OF THE ATOM. alpha particle source
 chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
chapter THE NATURE OF THE ATOM www.tutor-homework.com (for tutoring, homework help, or help with online classes) Section 30.1 Rutherford Scattering and the Nuclear Atom 1. Which model of atomic structure
High Resolution Electron Spectrometry at the NESR. Ajay Kumar
 High Resolution Electron Spectrometry at the NESR Collaboration Ajay Kumar GSI, Darmstadt Stored Particles Atomic Physics Research Collaboration R. Mann G. Garcia X. Ma B. Sulik J. Ullrich L.C. Tribedi
High Resolution Electron Spectrometry at the NESR Collaboration Ajay Kumar GSI, Darmstadt Stored Particles Atomic Physics Research Collaboration R. Mann G. Garcia X. Ma B. Sulik J. Ullrich L.C. Tribedi
Auger electron and x-ray emission from high-z few-electron ions
 Auger electron and x-ray emission from high-z few-electron ions S. Fritzsche MPI für Kernphysik Heidelberg and GSI Darmstadt 4th August 2007 Main goal for studying high-z ions is the better understanding
Auger electron and x-ray emission from high-z few-electron ions S. Fritzsche MPI für Kernphysik Heidelberg and GSI Darmstadt 4th August 2007 Main goal for studying high-z ions is the better understanding
SECTION A Quantum Physics and Atom Models
 AP Physics Multiple Choice Practice Modern Physics SECTION A Quantum Physics and Atom Models 1. Light of a single frequency falls on a photoelectric material but no electrons are emitted. Electrons may
AP Physics Multiple Choice Practice Modern Physics SECTION A Quantum Physics and Atom Models 1. Light of a single frequency falls on a photoelectric material but no electrons are emitted. Electrons may
Optical Spectroscopy and Atomic Structure. PHYS 0219 Optical Spectroscopy and Atomic Structure 1
 Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
Optical Spectroscopy and Atomic Structure PHYS 0219 Optical Spectroscopy and Atomic Structure 1 Optical Spectroscopy and Atomic Structure This experiment has four parts: 1. Spectroscope Setup - Your lab
is the minimum stopping potential for which the current between the plates reduces to zero.
 Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
Module 1 :Quantum Mechanics Chapter 2 : Introduction to Quantum ideas Introduction to Quantum ideas We will now consider some experiments and their implications, which introduce us to quantum ideas. The
U n 3 n Ba Kr (D) Br (C) Kr (B) Rb (E) 94 37
 1984 36. The critical angle for a transparent material in air is 30. The index of refraction of the material is most nearly (A) 0.33 (B) 0.50 (C) 1.0 (D) 1.5 (E) 2.0 37. An object is placed as shown in
1984 36. The critical angle for a transparent material in air is 30. The index of refraction of the material is most nearly (A) 0.33 (B) 0.50 (C) 1.0 (D) 1.5 (E) 2.0 37. An object is placed as shown in
10. Wavelength measurement using prism spectroscopy
 Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
Spk 0. Wavelength measurement using prism spectroscopy 0. Introduction The study of emitted spectra of electromagnetic waves by excited atoms makes for one of the most important methods to investigate
MANIPAL INSTITUTE OF TECHNOLOGY
 SCHEME OF EVAUATION MANIPA INSTITUTE OF TECHNOOGY MANIPA UNIVERSITY, MANIPA SECOND SEMESTER B.Tech. END-SEMESTER EXAMINATION - MAY SUBJECT: ENGINEERING PHYSICS (PHY/) Time: 3 Hrs. Max. Marks: 5 Note: Answer
SCHEME OF EVAUATION MANIPA INSTITUTE OF TECHNOOGY MANIPA UNIVERSITY, MANIPA SECOND SEMESTER B.Tech. END-SEMESTER EXAMINATION - MAY SUBJECT: ENGINEERING PHYSICS (PHY/) Time: 3 Hrs. Max. Marks: 5 Note: Answer
Physics 126 Practice Exam #4 Professor Siegel
 Physics 126 Practice Exam #4 Professor Siegel Name: Lab Day: 1. Light is usually thought of as wave-like in nature and electrons as particle-like. In which one of the following instances does light behave
Physics 126 Practice Exam #4 Professor Siegel Name: Lab Day: 1. Light is usually thought of as wave-like in nature and electrons as particle-like. In which one of the following instances does light behave
PC1144 Physics IV. Atomic Spectra
 PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
PC1144 Physics IV Atomic Spectra 1 Objectives Investigate how well the visible light wavelengths of hydrogen predicted by the Bohr theory agree with experimental values. Determine an experimental value
The Lamb shift in hydrogen and muonic hydrogen and the proton charge radius
 The Lamb shift in hydrogen and muonic hydrogen and the proton charge radius Savely Karshenboim Pulkovo Observatory (ГАО( РАН) ) (St. Petersburg) & Max-Planck Planck-Institut für Quantenoptik (Garching)
The Lamb shift in hydrogen and muonic hydrogen and the proton charge radius Savely Karshenboim Pulkovo Observatory (ГАО( РАН) ) (St. Petersburg) & Max-Planck Planck-Institut für Quantenoptik (Garching)
Chapter 28. Atomic Physics
 Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Chapter 28 Atomic Physics Quantum Numbers and Atomic Structure The characteristic wavelengths emitted by a hot gas can be understood using quantum numbers. No two electrons can have the same set of quantum
Chapter 37 Early Quantum Theory and Models of the Atom
 Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
Chapter 37 Early Quantum Theory and Models of the Atom Units of Chapter 37 37-7 Wave Nature of Matter 37-8 Electron Microscopes 37-9 Early Models of the Atom 37-10 Atomic Spectra: Key to the Structure
X-Ray Photoelectron Spectroscopy (XPS)
 X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
X-Ray Photoelectron Spectroscopy (XPS) Louis Scudiero http://www.wsu.edu/~scudiero; 5-2669 Fulmer 261A Electron Spectroscopy for Chemical Analysis (ESCA) The basic principle of the photoelectric effect
MIDSUMMER EXAMINATIONS 2001 PHYSICS, PHYSICS WITH ASTROPHYSICS PHYSICS WITH SPACE SCIENCE & TECHNOLOGY PHYSICS WITH MEDICAL PHYSICS
 No. of Pages: 6 No. of Questions: 10 MIDSUMMER EXAMINATIONS 2001 Subject PHYSICS, PHYSICS WITH ASTROPHYSICS PHYSICS WITH SPACE SCIENCE & TECHNOLOGY PHYSICS WITH MEDICAL PHYSICS Title of Paper MODULE PA266
No. of Pages: 6 No. of Questions: 10 MIDSUMMER EXAMINATIONS 2001 Subject PHYSICS, PHYSICS WITH ASTROPHYSICS PHYSICS WITH SPACE SCIENCE & TECHNOLOGY PHYSICS WITH MEDICAL PHYSICS Title of Paper MODULE PA266
Chap. 3. Elementary Quantum Physics
 Chap. 3. Elementary Quantum Physics 3.1 Photons - Light: e.m "waves" - interference, diffraction, refraction, reflection with y E y Velocity = c Direction of Propagation z B z Fig. 3.1: The classical view
Chap. 3. Elementary Quantum Physics 3.1 Photons - Light: e.m "waves" - interference, diffraction, refraction, reflection with y E y Velocity = c Direction of Propagation z B z Fig. 3.1: The classical view
MIDTERM 3 REVIEW SESSION. Dr. Flera Rizatdinova
 MIDTERM 3 REVIEW SESSION Dr. Flera Rizatdinova Summary of Chapter 23 Index of refraction: Angle of reflection equals angle of incidence Plane mirror: image is virtual, upright, and the same size as the
MIDTERM 3 REVIEW SESSION Dr. Flera Rizatdinova Summary of Chapter 23 Index of refraction: Angle of reflection equals angle of incidence Plane mirror: image is virtual, upright, and the same size as the
Chemistry (
 Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Question 2.1: (i) Calculate the number of electrons which will together weigh one gram. (ii) Calculate the mass and charge of one mole of electrons. Answer 2.1: (i) Mass of one electron = 9.10939 10 31
Nuclear Effects in Electron Capture into Highly Charged Heavy Ions
 Nuclear Effects in Electron Capture into Highly Charged Heavy Ions W. Scheid 1,A.Pálffy 2,Z.Harman 2, C. Kozhuharov 3, and C. Brandau 3 1 Institut für Theoretische Physik der Justus-Liebig-Universität
Nuclear Effects in Electron Capture into Highly Charged Heavy Ions W. Scheid 1,A.Pálffy 2,Z.Harman 2, C. Kozhuharov 3, and C. Brandau 3 1 Institut für Theoretische Physik der Justus-Liebig-Universität
arxiv: v1 [physics.atom-ph] 21 Jun 2011
![arxiv: v1 [physics.atom-ph] 21 Jun 2011 arxiv: v1 [physics.atom-ph] 21 Jun 2011](/thumbs/80/80601513.jpg) arxiv:1106.4155v1 [physics.atom-ph] 21 Jun 2011 Differential energy measurement between He- and Li-like uranium intra-shell transitions M. Trassinelli 1, A. Kumar 2, H.F. Beyer 3, P. Indelicato 4, R. Märtin
arxiv:1106.4155v1 [physics.atom-ph] 21 Jun 2011 Differential energy measurement between He- and Li-like uranium intra-shell transitions M. Trassinelli 1, A. Kumar 2, H.F. Beyer 3, P. Indelicato 4, R. Märtin
Physics 1C Lecture 29B
 Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Physics 1C Lecture 29B Emission Spectra! The easiest gas to analyze is hydrogen gas.! Four prominent visible lines were observed, as well as several ultraviolet lines.! In 1885, Johann Balmer, found a
Exercise 1 Atomic line spectra 1/9
 Exercise 1 Atomic line spectra 1/9 The energy-level scheme for the hypothetical one-electron element Juliettium is shown in the figure on the left. The potential energy is taken to be zero for an electron
Exercise 1 Atomic line spectra 1/9 The energy-level scheme for the hypothetical one-electron element Juliettium is shown in the figure on the left. The potential energy is taken to be zero for an electron
CHAPTER 12 TEST REVIEW
 IB PHYSICS Name: Period: Date: # Marks: 76 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 12 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
IB PHYSICS Name: Period: Date: # Marks: 76 Raw Score: IB Curve: DEVIL PHYSICS BADDEST CLASS ON CAMPUS CHAPTER 12 TEST REVIEW 1. An alpha particle is accelerated through a potential difference of 10 kv.
1. Nuclear Size. A typical atom radius is a few!10 "10 m (Angstroms). The nuclear radius is a few!10 "15 m (Fermi).
 1. Nuclear Size We have known since Rutherford s! " scattering work at Manchester in 1907, that almost all the mass of the atom is contained in a very small volume with high electric charge. Nucleus with
1. Nuclear Size We have known since Rutherford s! " scattering work at Manchester in 1907, that almost all the mass of the atom is contained in a very small volume with high electric charge. Nucleus with
The interaction of radiation with matter
 Basic Detection Techniques 2009-2010 http://www.astro.rug.nl/~peletier/detectiontechniques.html Detection of energetic particles and gamma rays The interaction of radiation with matter Peter Dendooven
Basic Detection Techniques 2009-2010 http://www.astro.rug.nl/~peletier/detectiontechniques.html Detection of energetic particles and gamma rays The interaction of radiation with matter Peter Dendooven
1 Photoelectric effect - Classical treatment. 2 Photoelectric effect - Quantum treatment
 1 OF 5 NOTE: This problem set is to be handed in to my mail slot (SMITH) located in the Clarendon Laboratory by 5:00 PM Tuesday, 10 May. 1 Photoelectric effect - Classical treatment A laser beam with an
1 OF 5 NOTE: This problem set is to be handed in to my mail slot (SMITH) located in the Clarendon Laboratory by 5:00 PM Tuesday, 10 May. 1 Photoelectric effect - Classical treatment A laser beam with an
= : K A
 Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
Atoms and Nuclei. State two limitations of JJ Thomson s model of atom. 2. Write the SI unit for activity of a radioactive substance. 3. What observations led JJ Thomson to conclusion that all atoms have
2. Determine the excess charge on the outer surface of the outer sphere (a distance c from the center of the system).
 Use the following to answer question 1. Two point charges, A and B, lie along a line separated by a distance L. The point x is the midpoint of their separation. 1. Which combination of charges will yield
Use the following to answer question 1. Two point charges, A and B, lie along a line separated by a distance L. The point x is the midpoint of their separation. 1. Which combination of charges will yield
Stellar Astrophysics: The Interaction of Light and Matter
 Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Stellar Astrophysics: The Interaction of Light and Matter The Photoelectric Effect Methods of electron emission Thermionic emission: Application of heat allows electrons to gain enough energy to escape
Solar UV Spectroscopy and Coronagraphy
 Solar UV Spectroscopy and Coronagraphy Werner Curdt Outline motivation the Sun s electromagnetic spectrum spectroscopic methods observational examples instrumental aspects optical design detectors others
Solar UV Spectroscopy and Coronagraphy Werner Curdt Outline motivation the Sun s electromagnetic spectrum spectroscopic methods observational examples instrumental aspects optical design detectors others
Photoelectron spectroscopy Instrumentation. Nanomaterials characterization 2
 Photoelectron spectroscopy Instrumentation Nanomaterials characterization 2 RNDr. Věra V Vodičkov ková,, PhD. Photoelectron Spectroscopy general scheme Impact of X-ray emitted from source to the sample
Photoelectron spectroscopy Instrumentation Nanomaterials characterization 2 RNDr. Věra V Vodičkov ková,, PhD. Photoelectron Spectroscopy general scheme Impact of X-ray emitted from source to the sample
Sliced multilayer gratings (SMG) as dispersive elements for the soft X-rays
 Sliced multilayer gratings (SMG) as dispersive elements for the soft X-rays E. A. Bugaev, V.A. Chirkov, R.M. Feshchenko*, V.P. Petukhov, A.V. Vinogradov, D.L. Voronov, V.A. Tokarev International Conference
Sliced multilayer gratings (SMG) as dispersive elements for the soft X-rays E. A. Bugaev, V.A. Chirkov, R.M. Feshchenko*, V.P. Petukhov, A.V. Vinogradov, D.L. Voronov, V.A. Tokarev International Conference
Highenergy Nuclear Optics of Polarized Particles
 Highenergy Nuclear Optics of Polarized Particles Vladimir G. Baryshevsky Research Institute for Nuclear Problems Belarusian State University 1> World Scientific NEW JERSEY LONDON SINGAPORE BEIJING SHANGHAI
Highenergy Nuclear Optics of Polarized Particles Vladimir G. Baryshevsky Research Institute for Nuclear Problems Belarusian State University 1> World Scientific NEW JERSEY LONDON SINGAPORE BEIJING SHANGHAI
Particles and Waves Particles Waves
 Particles and Waves Particles Discrete and occupy space Exist in only one location at a time Position and velocity can be determined with infinite accuracy Interact by collisions, scattering. Waves Extended,
Particles and Waves Particles Discrete and occupy space Exist in only one location at a time Position and velocity can be determined with infinite accuracy Interact by collisions, scattering. Waves Extended,
Any first year text, sections on atomic structure, spectral lines and spectrometers
 Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Physics 33 Experiment 5 Atomic Spectra References Any first year text, sections on atomic structure, spectral lines and spectrometers Any modern physics text, eg F.K. Richtmeyer, E.H. Kennard and J.N.
Atomic structure and dynamics
 Atomic structure and dynamics -- need and requirements for accurate atomic calculations Analysis and interpretation of optical and x-ray spectra (astro physics) Isotope shifts and hyperfine structures
Atomic structure and dynamics -- need and requirements for accurate atomic calculations Analysis and interpretation of optical and x-ray spectra (astro physics) Isotope shifts and hyperfine structures
Two-body weak decay of highly charged ions, a tool to study neutrino properties?
 Two-body weak decay of highly charged ions, a tool to study neutrino properties? The detector: ESR Experimental Storage Ring cooling: electron-, stochastic ion detection: Schottky-noise, particle detector
Two-body weak decay of highly charged ions, a tool to study neutrino properties? The detector: ESR Experimental Storage Ring cooling: electron-, stochastic ion detection: Schottky-noise, particle detector
LIGHT. Question. Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light.
 LIGHT Question Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light. What kind of information can we get from light? 1 Light
LIGHT Question Until very recently, the study of ALL astronomical objects, outside of the Solar System, has been with telescopes observing light. What kind of information can we get from light? 1 Light
Energy Level Energy Level Diagrams for Diagrams for Simple Hydrogen Model
 Quantum Mechanics and Atomic Physics Lecture 20: Real Hydrogen Atom /Identical particles http://www.physics.rutgers.edu/ugrad/361 physics edu/ugrad/361 Prof. Sean Oh Last time Hydrogen atom: electron in
Quantum Mechanics and Atomic Physics Lecture 20: Real Hydrogen Atom /Identical particles http://www.physics.rutgers.edu/ugrad/361 physics edu/ugrad/361 Prof. Sean Oh Last time Hydrogen atom: electron in
Particle nature of light & Quantization
 Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
Particle nature of light & Quantization A quantity is quantized if its possible values are limited to a discrete set. An example from classical physics is the allowed frequencies of standing waves on a
An Introduction to Diffraction and Scattering. School of Chemistry The University of Sydney
 An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
An Introduction to Diffraction and Scattering Brendan J. Kennedy School of Chemistry The University of Sydney 1) Strong forces 2) Weak forces Types of Forces 3) Electromagnetic forces 4) Gravity Types
High-precision studies in fundamental physics with slow neutrons. Oliver Zimmer Institut Laue Langevin
 High-precision studies in fundamental physics with slow neutrons Oliver Zimmer Institut Laue Langevin ILL, 20 September 2016 Topics The impossible particle and its properties Search for an electric dipole
High-precision studies in fundamental physics with slow neutrons Oliver Zimmer Institut Laue Langevin ILL, 20 September 2016 Topics The impossible particle and its properties Search for an electric dipole
PHYSICS 359E: EXPERIMENT 2.2 THE MOSSBAUER EFFECT: RESONANT ABSORPTION OF (-RAYS
 PHYSICS 359E: EXPERIMENT 2.2 THE MOSSBAUER EFFECT: RESONANT ABSORPTION OF (-RAYS INTRODUCTION: In classical physics resonant phenomena are expected whenever a system can undergo free oscillations. These
PHYSICS 359E: EXPERIMENT 2.2 THE MOSSBAUER EFFECT: RESONANT ABSORPTION OF (-RAYS INTRODUCTION: In classical physics resonant phenomena are expected whenever a system can undergo free oscillations. These
Particle Nature of Matter. Chapter 4
 Particle Nature of Matter Chapter 4 Modern physics When my grandfather was born, atoms were just an idea. That year, 1897, was marked by the discovery of the electron by J.J. Thomson. The nuclear model
Particle Nature of Matter Chapter 4 Modern physics When my grandfather was born, atoms were just an idea. That year, 1897, was marked by the discovery of the electron by J.J. Thomson. The nuclear model
Gamma-ray decay. Introduction to Nuclear Science. Simon Fraser University Spring NUCS 342 March 7, 2011
 Gamma-ray decay Introduction to Nuclear Science Simon Fraser University Spring 2011 NUCS 342 March 7, 2011 NUCS 342 (Lecture 18) March 7, 2011 1 / 31 Outline 1 Mössbauer spectroscopy NUCS 342 (Lecture
Gamma-ray decay Introduction to Nuclear Science Simon Fraser University Spring 2011 NUCS 342 March 7, 2011 NUCS 342 (Lecture 18) March 7, 2011 1 / 31 Outline 1 Mössbauer spectroscopy NUCS 342 (Lecture
Unified School District of De Pere Physics Benchmarks
 Content Standards: A. Students will understand that among the science disciplines, there are unifying themes: systems, order, organization, and interactions; evidence, models, and explanations; constancy,
Content Standards: A. Students will understand that among the science disciplines, there are unifying themes: systems, order, organization, and interactions; evidence, models, and explanations; constancy,
Introduction to the physics of highly charged ions. Lecture 7: Direct and resonant photoionization of HCI
 Introduction to the physics of highly charged ions Lecture 7: Direct and resonant photoionization of HCI Zoltán Harman harman@mpi-hd.mpg.de Universität Heidelberg, 02.12.2013 So far: radiative decay and
Introduction to the physics of highly charged ions Lecture 7: Direct and resonant photoionization of HCI Zoltán Harman harman@mpi-hd.mpg.de Universität Heidelberg, 02.12.2013 So far: radiative decay and
Chandra was launched aboard Space Shuttle Columbia on July 23, 1999!!!
 Chandra was launched aboard Space Shuttle Columbia on July 23, 1999!!! Crew Lost During Re-Entry Modern X-ray Telescopes and Detectors X-ray Telescopes X-ray Instruments Some early highlights Observations
Chandra was launched aboard Space Shuttle Columbia on July 23, 1999!!! Crew Lost During Re-Entry Modern X-ray Telescopes and Detectors X-ray Telescopes X-ray Instruments Some early highlights Observations
Chapter V: Interactions of neutrons with matter
 Chapter V: Interactions of neutrons with matter 1 Content of the chapter Introduction Interaction processes Interaction cross sections Moderation and neutrons path For more details see «Physique des Réacteurs
Chapter V: Interactions of neutrons with matter 1 Content of the chapter Introduction Interaction processes Interaction cross sections Moderation and neutrons path For more details see «Physique des Réacteurs
FACTS WHY? C. Alpha Decay Probability 1. Energetics: Q α positive for all A>140 nuclei
 C. Alpha Decay Probability 1. Energetics: Q α positive for all A>140 nuclei 2. Range of Measured Half-Lives (~10 44 ) 10 16 y > t 1/2 > 10 21 s 3. Why α? a. Proton & Neutron Emission: Q p, Q n are negative
C. Alpha Decay Probability 1. Energetics: Q α positive for all A>140 nuclei 2. Range of Measured Half-Lives (~10 44 ) 10 16 y > t 1/2 > 10 21 s 3. Why α? a. Proton & Neutron Emission: Q p, Q n are negative
Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002
 Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002 More Quantum Physics We know now how to detect light (or photons) One possibility to detect
Particle Detectors and Quantum Physics (2) Stefan Westerhoff Columbia University NYSPT Summer Institute 2002 More Quantum Physics We know now how to detect light (or photons) One possibility to detect
CHARGED PARTICLE INTERACTIONS
 CHARGED PARTICLE INTERACTIONS Background Charged Particles Heavy charged particles Charged particles with Mass > m e α, proton, deuteron, heavy ion (e.g., C +, Fe + ), fission fragment, muon, etc. α is
CHARGED PARTICLE INTERACTIONS Background Charged Particles Heavy charged particles Charged particles with Mass > m e α, proton, deuteron, heavy ion (e.g., C +, Fe + ), fission fragment, muon, etc. α is
Experiment 7: Spectrum of the Hydrogen Atom
 Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
Experiment 7: Spectrum of the Hydrogen Nate Saffold nas2173@columbia.edu Office Hour: Mondays, 5:30-6:30PM INTRO TO EXPERIMENTAL PHYS-LAB 1493/1494/2699 Introduction The physics behind: The spectrum of
2. X-ray Sources 2.1 Electron Impact X-ray Sources - Types of X-ray Source - Bremsstrahlung Emission - Characteristic Emission
 . X-ray Sources.1 Electron Impact X-ray Sources - Types of X-ray Source - Bremsstrahlung Emission - Characteristic Emission. Synchrotron Radiation Sources - Introduction - Characteristics of Bending Magnet
. X-ray Sources.1 Electron Impact X-ray Sources - Types of X-ray Source - Bremsstrahlung Emission - Characteristic Emission. Synchrotron Radiation Sources - Introduction - Characteristics of Bending Magnet
Problem Solving. radians. 180 radians Stars & Elementary Astrophysics: Introduction Press F1 for Help 41. f s. picture. equation.
 Problem Solving picture θ f = 10 m s =1 cm equation rearrange numbers with units θ factors to change units s θ = = f sinθ fθ = s / cm 10 m f 1 m 100 cm check dimensions 1 3 π 180 radians = 10 60 arcmin
Problem Solving picture θ f = 10 m s =1 cm equation rearrange numbers with units θ factors to change units s θ = = f sinθ fθ = s / cm 10 m f 1 m 100 cm check dimensions 1 3 π 180 radians = 10 60 arcmin
The Bohr Model of Hydrogen
 The Bohr Model of Hydrogen Suppose you wanted to identify and measure the energy high energy photons. One way to do this is to make a calorimeter. The CMS experiment s electromagnetic calorimeter is made
The Bohr Model of Hydrogen Suppose you wanted to identify and measure the energy high energy photons. One way to do this is to make a calorimeter. The CMS experiment s electromagnetic calorimeter is made
LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS. Instructor: Kazumi Tolich
 LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS Instructor: Kazumi Tolich Lecture 23 2 29.1 Spectroscopy 29.2 Atoms The first nuclear physics experiment Using the nuclear model 29.3 Bohr s model of atomic quantization
LECTURE 23 SPECTROSCOPY AND ATOMIC MODELS Instructor: Kazumi Tolich Lecture 23 2 29.1 Spectroscopy 29.2 Atoms The first nuclear physics experiment Using the nuclear model 29.3 Bohr s model of atomic quantization
NPRE 446: Interaction of Radiation with Matter Homework Assignments
 NPRE 446: Interaction of Radiation with Matter Homework Assignments Prof. Y Z Department of Nuclear, Plasma, and Radiological Engineering Department of Materials Science and Engineering Department of Electrical
NPRE 446: Interaction of Radiation with Matter Homework Assignments Prof. Y Z Department of Nuclear, Plasma, and Radiological Engineering Department of Materials Science and Engineering Department of Electrical
Ion traps. Trapping of charged particles in electromagnetic. Laser cooling, sympathetic cooling, optical clocks
 Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
Ion traps Trapping of charged particles in electromagnetic fields Dynamics of trapped ions Applications to nuclear physics and QED The Paul trap Laser cooling, sympathetic cooling, optical clocks Coulomb
UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM PHYSICAL SCIENCE TEST SERIES # 4. Atomic, Solid State & Nuclear + Particle
 UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM BOOKLET CODE PH PHYSICAL SCIENCE TEST SERIES # 4 Atomic, Solid State & Nuclear + Particle SUBJECT CODE 05 Timing: 3: H M.M: 200 Instructions 1.
UGC ACADEMY LEADING INSTITUE FOR CSIR-JRF/NET, GATE & JAM BOOKLET CODE PH PHYSICAL SCIENCE TEST SERIES # 4 Atomic, Solid State & Nuclear + Particle SUBJECT CODE 05 Timing: 3: H M.M: 200 Instructions 1.
Chapter II: Interactions of ions with matter
 Chapter II: Interactions of ions with matter 1 Trajectories of α particles of 5.5 MeV Source: SRIM www.srim.org 2 Incident proton on Al: Bohr model v=v 0 E p =0.025 MeV relativistic effect E p =938 MeV
Chapter II: Interactions of ions with matter 1 Trajectories of α particles of 5.5 MeV Source: SRIM www.srim.org 2 Incident proton on Al: Bohr model v=v 0 E p =0.025 MeV relativistic effect E p =938 MeV
UNIT VIII ATOMS AND NUCLEI
 UNIT VIII ATOMS AND NUCLEI Weightage Marks : 06 Alpha-particles scattering experiment, Rutherford s model of atom, Bohr Model, energy levels, Hydrogen spectrum. Composition and size of Nucleus, atomic
UNIT VIII ATOMS AND NUCLEI Weightage Marks : 06 Alpha-particles scattering experiment, Rutherford s model of atom, Bohr Model, energy levels, Hydrogen spectrum. Composition and size of Nucleus, atomic
Mossbauer Effect and Spectroscopy. Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln
 Mossbauer Effect and Spectroscopy Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln Emission E R γ-photon E transition hν = E transition - E R Photon does not carry
Mossbauer Effect and Spectroscopy Kishan Sinha Xu Group Department of Physics and Astronomy University of Nebraska-Lincoln Emission E R γ-photon E transition hν = E transition - E R Photon does not carry
Nuclear Reactions A Z. Radioactivity, Spontaneous Decay: Nuclear Reaction, Induced Process: x + X Y + y + Q Q > 0. Exothermic Endothermic
 Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Radioactivity, Spontaneous Decay: Nuclear Reactions A Z 4 P D+ He + Q A 4 Z 2 Q > 0 Nuclear Reaction, Induced Process: x + X Y + y + Q Q = ( m + m m m ) c 2 x X Y y Q > 0 Q < 0 Exothermic Endothermic 2
Some Topics in Optics
 Some Topics in Optics The HeNe LASER The index of refraction and dispersion Interference The Michelson Interferometer Diffraction Wavemeter Fabry-Pérot Etalon and Interferometer The Helium Neon LASER A
Some Topics in Optics The HeNe LASER The index of refraction and dispersion Interference The Michelson Interferometer Diffraction Wavemeter Fabry-Pérot Etalon and Interferometer The Helium Neon LASER A
The Final Exam (Exam 4) will be on FRIDAY MAY 11 From 3 5 PM in LR1 VAN
 1 --------------------------------------------------------------------------------------------------------------------- 29:006 SPRING 2012 PRACTICE EXAM 4 ---------------------------------------------------------------------------------------------------------------------
1 --------------------------------------------------------------------------------------------------------------------- 29:006 SPRING 2012 PRACTICE EXAM 4 ---------------------------------------------------------------------------------------------------------------------
Energy levels and atomic structures lectures chapter one
 Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Structure of Atom An atom is the smallest constituent unit of ordinary matter that has the properties of a element. Every solid, liquid, gas, and plasma is composed of neutral or ionized atoms. Atoms are
Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling. Indian Institute of Technology Ropar
 Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling Specification of the ESR Particle detectors Re-injection to SIS Two 5 kv rf-cavities Fast Injection Schottky
Experimental Storage Ring - ESR E max = 420 MeV/u, 10 Tm, electron-, stochastic- and laser cooling Specification of the ESR Particle detectors Re-injection to SIS Two 5 kv rf-cavities Fast Injection Schottky
Cauchois Johansson x-ray spectrograph for kev energy range
 REVIEW OF SCIENTIFIC INSTRUMENTS VOLUME 72, NUMBER 2 FEBRUARY 2001 Cauchois Johansson x-ray spectrograph for 1.5 400 kev energy range E. O. Baronova a) and M. M. Stepanenko RRC Kurchatov Institute, 123182,
REVIEW OF SCIENTIFIC INSTRUMENTS VOLUME 72, NUMBER 2 FEBRUARY 2001 Cauchois Johansson x-ray spectrograph for 1.5 400 kev energy range E. O. Baronova a) and M. M. Stepanenko RRC Kurchatov Institute, 123182,
High-energy collision processes involving intense laser fields
 High-energy collision processes involving intense laser fields Carsten Müller Max Planck Institute for Nuclear Physics, Theory Division (Christoph H. Keitel), Heidelberg, Germany EMMI Workshop: Particle
High-energy collision processes involving intense laser fields Carsten Müller Max Planck Institute for Nuclear Physics, Theory Division (Christoph H. Keitel), Heidelberg, Germany EMMI Workshop: Particle
Physics 23 Fall 1998 Lab 4 - The Hydrogen Spectrum
 Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
Physics 3 Fall 998 Lab 4 - The Hydrogen Spectrum Theory In the late 800's, it was known that when a gas is excited by means of an electric discharge and the light emitted is viewed through a diffraction
Sunday Monday Thursday. Friday
 Nuclear Structure III experiment Sunday Monday Thursday Low-lying excited states Collectivity and the single-particle degrees of freedom Collectivity studied in Coulomb excitation Direct reactions to study
Nuclear Structure III experiment Sunday Monday Thursday Low-lying excited states Collectivity and the single-particle degrees of freedom Collectivity studied in Coulomb excitation Direct reactions to study
The FAIR Accelerator Facility
 The FAIR Accelerator Facility SIS300 existing GSI proton linac SIS18 UNILAC SIS100 HESR pbar target SuperFRS goals: higher intensity (low charge states) higher energy (high charge states) production of
The FAIR Accelerator Facility SIS300 existing GSI proton linac SIS18 UNILAC SIS100 HESR pbar target SuperFRS goals: higher intensity (low charge states) higher energy (high charge states) production of
Interaction of particles with matter - 2. Silvia Masciocchi, GSI and University of Heidelberg SS2017, Heidelberg May 3, 2017
 Interaction of particles with matter - 2 Silvia Masciocchi, GSI and University of Heidelberg SS2017, Heidelberg May 3, 2017 Energy loss by ionization (by heavy particles) Interaction of electrons with
Interaction of particles with matter - 2 Silvia Masciocchi, GSI and University of Heidelberg SS2017, Heidelberg May 3, 2017 Energy loss by ionization (by heavy particles) Interaction of electrons with
Atomic Physics with Stored and Cooled Ions
 Lecture #8 Atomic Physics with Stored and Cooled Ions Klaus Blaum Gesellschaft für Schwerionenforschung, GSI, Darmstadt and CERN, Physics Department, Geneva, Switzerland Summer School, Lanzhou, China,
Lecture #8 Atomic Physics with Stored and Cooled Ions Klaus Blaum Gesellschaft für Schwerionenforschung, GSI, Darmstadt and CERN, Physics Department, Geneva, Switzerland Summer School, Lanzhou, China,
Exam 4. P202 Spring 2004 Instructor: Prof. Sinova
 Exam 4 P202 Spring 2004 Instructor: Prof. Sinova Name: Date: 4/22/04 Section: All work must be shown to get credit for the answer marked. You must show or state your reasoning. If the answer marked does
Exam 4 P202 Spring 2004 Instructor: Prof. Sinova Name: Date: 4/22/04 Section: All work must be shown to get credit for the answer marked. You must show or state your reasoning. If the answer marked does
Perspectives on High Resolution X-ray Spectroscopy
 Perspectives on High Resolution X-ray Spectroscopy Rewards and Challenges Claude R. Canizares MIT July 11-13, 2007 X-ray Spectroscopy Workshop Cambridge, MA Rewards of High Resolution X-ray Spectroscopy
Perspectives on High Resolution X-ray Spectroscopy Rewards and Challenges Claude R. Canizares MIT July 11-13, 2007 X-ray Spectroscopy Workshop Cambridge, MA Rewards of High Resolution X-ray Spectroscopy
The University of Hong Kong Department of Physics
 The University of Hong Kong Department of Physics Physics Laboratory PHYS3551 Introductory Solid State Physics Experiment No. 3551-2: Electron and Optical Diffraction Name: University No: This experiment
The University of Hong Kong Department of Physics Physics Laboratory PHYS3551 Introductory Solid State Physics Experiment No. 3551-2: Electron and Optical Diffraction Name: University No: This experiment
The Grating Spectrometer and Atomic Spectra
 PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
PHY 192 Grating Spectrometer Spring 2012 1 The Grating Spectrometer and Atomic Spectra Introduction In the previous experiment diffraction and interference were discussed and at the end a diffraction grating
